Introduction
Acute myeloid leukemia (AML) is characterized by the
clonal proliferation of myeloid precursors. Leukemic blasts or
immature forms accumulate in the bone marrow, peripheral blood and
occasionally in other tissues, resulting in variable reductions in
the production of normal blood cells. Although some clinical
factors such as age, performance status or a history of prior
chemotherapy have been associated with the outcomes of patients
with AML, the most important factor in predicting the risk of
relapse has been chromosomal abnormalities detected at diagnosis
(1–4). Proto-oncogene mutations are an
important consideration in the risk stratification of patients with
AML (5). The mutational diagnosis
of NMP1, PML-RARA, CBFB-MYH11, FLT3 and
RAS has been applied to clinical practice and has
consequently impacted diagnoses, risk assessments and also guidance
related to available therapies. Changes in the expression of
specific genes (e.g., ABCB1, BAALC, ERG,
MN1, JAK2 and WT1) appear to influence the
prognoses of molecular subsets of patients with AML (3).
MicroRNAs (miRs) have been shown to negatively
regulate gene expression by binding to the mRNAs of protein-coding
genes, thereby degrading or blocking their translation. MicroRNAs
are also known to play an important role in various physiological
and pathological processes, such as apoptosis, cell proliferation
and differentiation, which indicates their functionality in
carcinogenesis as tumor-suppressor genes or proto-oncogenes
(6). MicroRNA expression profiling
has been used to distinguish between myeloid and lymphoid leukemia,
as well as distinct cytogenetic subtypes of AML based on the
upregulation or downregulation of specific miRNAs. MicroRNA
expression signatures have also been correlated with recurrent
molecular aberrations in AML (5).
miR-126-3p is known to play an important role in various
physiological processes. A previous study reported that miR-126-3p
(accession no. MIMAT0000445) inhibited cell apoptosis and increased
cell viability in AML cells (7).
Mature miR-126-3p is generated from the stem-loop sequence miR-126
(accession no. MI0000471). Although miR-126 also generates mature
miR-126-5p (accession no. MIMAT0000444), its function is less
clear.
Bioinformatics, microRNA.org,
predicted that miR-126-5p targets Klotho. The klotho gene
functions as an aging suppressor gene that extends the life-span.
Recent studies have revealed that Klotho plays an important role in
cancer tumorigenesis (8). The
Klotho protein can regulate multiple growth factor signaling
pathways, including IGF-1 and Wnt, as well as the activity of
multiple ion channels (9,10). The overexpression of Klotho was
previously shown to inhibit cancer cell proliferation and may act
as a potential therapeutic strategy in cancers (8).
It currently remains unclear whether miR-126-5p is
involved in the survival of patients with AML. The inhibitory
effects of miR-126-5p on the expression of Klotho mRNA have
not yet been elucidated in detail. We herein investigated the
survival benefits of miR-126-5p in patients with AML and the
inhibitory effects of a possible target mRNA of miR-126-5p and
Klotho.
Materials and methods
Reagents
The TaqMan gene expression assays, siPORT
NeoFX Transfection Agent and TRIzol reagent were obtained
from Life Technologies (Carlsbad, CA, USA). The QuantiTect primer
assay, miScript primer assay, miScript reverse transcription kit,
synthetic microRNA mimic, QIAamp RNA Blood Mini kit, AllStars
negative control siRNA and miScript SYBR-Green PCR kit were
purchased from Qiagen (Valencia, CA, USA). Real-time PCR Master Mix
Thunderbird SYBR qPCR Mix and reverse transcriptase, ReverTra Ace
were purchased from Toyobo Co., Ltd., (Osaka, Japan). Protease
inhibitor cocktail tablets (Complete, EDTA-free) were purchased
from Roche Diagnostics GmbH (Buckinghamshire, UK). RPMI-1640 medium
was purchased from Sigma-Aldrich (St. Louis, MO, USA). The human
acute myelogenous leukemia cell line, KG-1 (JCRB9051), and the
human myelogenous leukemia cell line, K562 (JCRB0019), were
purchased from the Japanese Collection of Research Bioresources
Cell Bank (Osaka, Japan). This cell line was tested and
authenticated by the JCRB Cell Bank. The human proximal tubular
cell line, HK-2, was purchased from the American Type Culture
Collection. Mouse monoclonal antibodies specific for anti-β-actin
(AC-74), Akt1 (5c10), anti-phosphorylated Akt1 (104A282),
anti-caspase-3 (NB500-210SS) and anti-JAK2 (1C1) were purchased
from Sigma-Aldrich, Enzo Life Sciences (Farmingdale, NY, USA),
Abcam (Cambridge, UK) and GeneTex (San Antonio, TX, USA),
respectively. Rabbit polyclonal antibodies specific for
phosphorylated-JAK2, STAT3, phosphorylated-STAT3, STAT5,
phosphorylated-STAT5 and Klotho were purchased from GeneTex. The
synthetic Klotho siRNA (sense, GGAUUGACCUUGAAUUUAATT and
antisense: UUAAAUUCAAGGUCAAUCCTT), PCR primers for Klotho
(sense, GCTCTCAAAGCCCACATACTG and antisense, GCAGCATAACGATAGAGGCC)
and PCR primers for ACTB actin, β (β-actin sense,
TGACGTGGACATCCG CAAAG and antisense, CTGGAAGGTGGACAGCGAGG)
(11,12) were obtained from Fasmac Co., Ltd.
(Atsugi, Japan). All other reagents were purchased from Wako Pure
Chemical Industries (Osaka, Japan).
Sampling of bone marrow
Bone marrow from 109 patients diagnosed with AML was
obtained from the North Japan Hematology Study Group. The
characteristics of the patients who received intensive therapy are
described in Table I. Chromosomal
aberrations were identified at participating centers using standard
procedures for bone marrow morphology, cytochemistry and flow
cytometry, along with cytogenetics using FISH and/or RT-PCR. An
informed written consent was obtained from all the patients that
participated in this study. The present study was approved by the
Ethics Committee of the Graduate School of Medicine, Hokkaido
University.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| miR-126-5p
| P-value |
|---|
| Low | High |
|---|
| Gender (M/F) | 29/25 | 31/24 | NS |
| Age, years |
| Mean | 58.2 | 57.3 | |
| Range | 20–78 | 17–79 | |
| Incipient
(initial/relapse) | 50/4 | 51/4 | NS |
| FAB classification,
n (%) | | | NS |
| M0 | 4 (7) | 2 (4) | |
| M1 | 14 (26) | 8 (15) | |
| M2 | 12 (22) | 21 (38) | |
| M3 | 10 (19) | 5 (9) | |
| M4 | 7 (13) | 10 (18) | |
| MDS | 5 (9) | 1 (2) | |
| Other | 2 (4) | 8 (14) | |
Cell culture and transfection assays
The human myeloid leukemia cell lines, KG-1 and
K562, were grown in RPMI-1640 medium. The human proximal tubular
cell line, HK-2, was grown in DMEM/Ham's F-12 medium. The medium
was supplemented with 10% fetal bovine serum, 2 mM glutamine, and
100 units/ml of penicillin at 37°C in a 5% CO2
humidified atmosphere. The synthetic miRNA inhibitor and precursor
were transfected using siPORT NeoFX transfection agent
according to the manufacturer's protocol. In 96-well plates, 3 pmol
of the inhibitor or precursor was transfected using 0.3 μl
of the siPORT NeoFX and the cells were harvested 72 h later
for the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium
bromide (MTT) assay. Cytarabine and idarubicin were added to the
wells and the cells were harvested at 120 and 48 h, respectively.
The cells were incubated with MTT solution, and incubation was
stopped with 20% sodium dodecyl sulfate solution. The 96-well
plates were shacken overnight in the dark and the absorbance was
then determined at 570 nm.
Real-time quantitative reverse
transcription-polymerase chain reaction (RT-PCR) analysis
The total RNA was isolated from the bone marrow
using the QIAamp RNA Blood Mini kit according to the manufacturer's
instructions. Single-stranded cDNA was synthesized by
reverse-transcriptase using ReverTra Ace and single-stranded cDNA
for the miRNA analysis was also synthesized by
reverse-transcriptase using the miScript reverse transcription kit
according to the manufacturer's instructions. Real-time PCR was
performed on the LightCycler 480 ΙΙ System (version 1.5; Roche
Diagnostics GmbH, Mannheim, Germany) using TaqMan gene expression
assays and Thunderbird qPCR Mix or the miScript SYBR-Green PCR kit
according to the manufacturer's instructions. The comparative
quantification cycle threshold (Cq) method was used to determine
the relative expression levels of the target genes. Cq values were
calculated by the 2nd derivative maximum method. Glucose-phosphate
isomerase (GPI) and RNU6B (U6) were analyzed as reference
genes for mRNA and miR, respectively (13,14).
The cycle number difference (ΔCq = reference genes - target genes)
was calculated for each replicate. Relative target gene expression
values were calculated using the mean of ΔCq from 3 replicates,
μ(ΔCq) =Σ(ΔCq)/3 and expressed as
2μ(ΔCq) (13).
Electrophoresis and western blot
analysis
Protein sampling and the western blot analysis were
performed as described previously (15). Whole cell homogenates (β-actin, 5
μg; other proteins, 20 μg) were separated using 10%
SDS polyacrylamide gels and proteins were then transferred to a
nitrocellulose membrane. The membranes were incubated with either
the AC-74 (10,000-fold dilution), 5C10 (1,000-fold dilution),
104A282 (1,000-fold dilution), 1C1 (500-fold dilution) or
NB500-210SS (2,000-fold dilution) monoclonal antibody and with the
rabbit polyclonal antibody (1,000-fold dilution) in the blocking
solution. The detection method has been described in detail
previously (15).
Statistical analysis
Comparisons between 2 groups were performed with the
Mann-Whitney U test or the Student's t-test. Comparisons between 3
groups were performed with the Tukey-Kramer test. Categorical
variables were analyzed with the two-tailed χ2 test or
Fisher's exact test (expected frequency <5). Survival was
plotted with the Kaplan-Meier curves, using the interval from the
date of the AML diagnosis to death or to the last contact.
Comparisons between each group were performed with the log-rank
test. Overall survival was evaluated using the Cox proportional
hazards model. All indicated P-values were two-sided: P<0.05,
P<0.01 and P<0.001.
Results
Relationships between the overall
survival and the expression levels of miR-126-5p and
miR-126-3p
The relationship between miR-126-5p expression level
and the overall survival duration in 109 patients who received
intensive therapy was evaluated. Patients were divided into two
groups: the high group expressed greater amounts of miR-126-5p than
the median value of miR-126-5p expression while the low group
expressed less. No significant differences were observed in the
patient characteristics or gene mutation statuses of the high and
low groups (Tables I and II). No significant differences were
observed in the expression levels of RNA-related abnormalities in
AML between the two groups (Table
III). Higher miR-126-5p expression levels (more than the median
value) correlated with a poorer probability of overall survival
(Fig. 1A). Higher miR-126-3p
expression levels also correlated with a poorer overall survival
(Fig. 1B). Cox proportional hazards
models also estimated a significant higher hazard ratio (HR) of
miR-126-5p and miR-126-3p in the high expression group (Fig. 1). In the Cox proportional hazard
regression model, the ‘miR-126-5p × miR-126-3p’ interaction was not
significant (p=0.73).
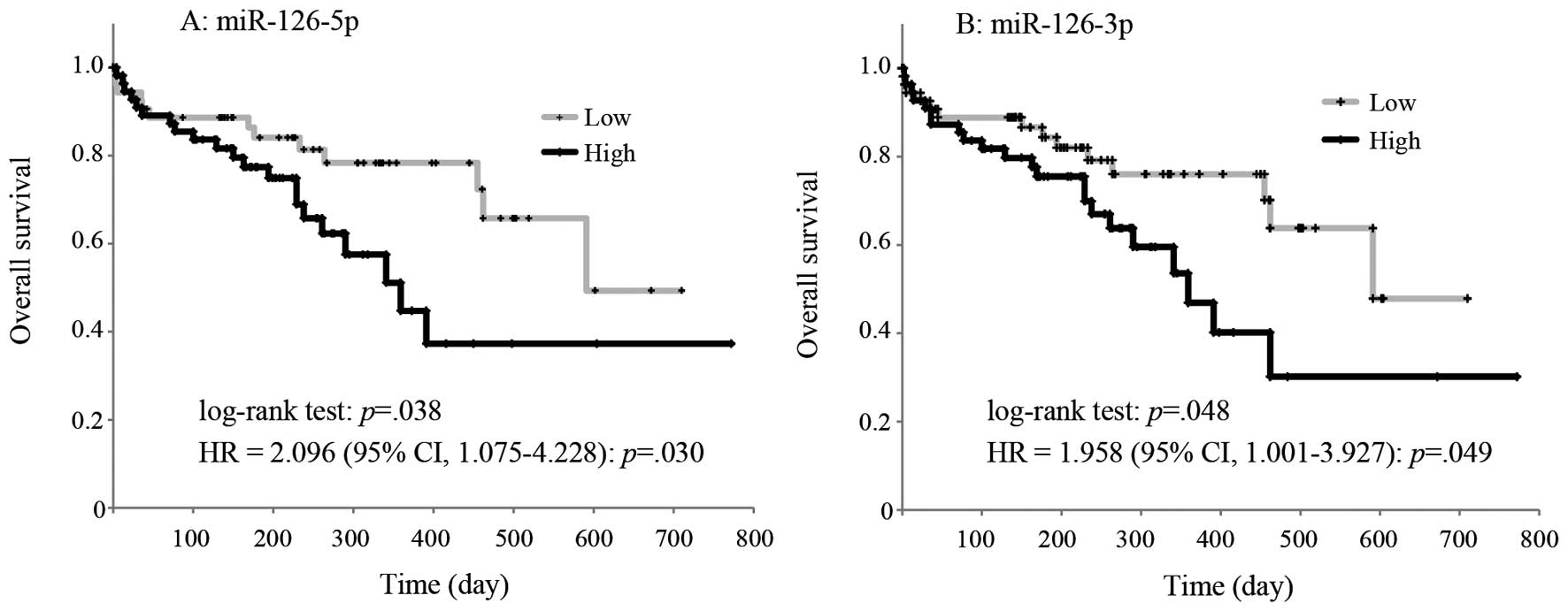 | Figure 1Kaplan-Meier overall survival curves
for patients with AML according to the low and high expression of
miR-126-5p and miR-126-3p. Kaplan-Meier plots showing estimates of
overall survival probabilities grouped based on the miR-126-5p
expression levels in a completely independent set of 109 AML
patients. (A) The black line curve represents the samples with high
(above median, 2−ΔΔCq value >1.1×10−4,
n=55) miR-126-5p expression levels, whereas the dotted line curve
corresponds to the samples with low (below median,
2−ΔΔCq value <1.1×10−4, n=54) miR-126-5p
expression levels. (B) The black line curve represents the samples
with high (above median, 2−ΔΔCq value
>2.0×10−4, n=54) miR-126-3p expression levels,
whereas the dotted line curve corresponds to the samples with low
(below median, 2−ΔΔCq value <2.0×10−4,
n=54) miR-126-3p expression levels. Comparisons between each group
were performed with the log-rank test. Cox proportional hazards
models also estimated a significantly higher HR of miR-126-5p and
miR-126-3p in the high group: miR-126-5p: HR=2.096 (95% CI,
1.075–4.228), p=0.030 and miR-126-3p: HR=1.958 (95% CI,
1.001–3.927), p=0.049. In the Cox proportional hazard regression
model, the ‘miR-126-5p x miR-126-3p’ interaction was not
significant (p=0.73). CI, confidence interval; AML, acute myeloid
leukemia; HR, hazard ratio. |
 | Table IIGene mutation status. |
Table II
Gene mutation status.
| Gene | miR-126-5p
| P-value |
|---|
| Low, n (%) | High, n (%) |
|---|
| NPM1 | 13 (24) | 12 (22) | NS |
|
PML-RARA | 10 (19) | 5 (9) | NS |
| CEBPA | 13 (24) | 7 (13) | NS |
|
CBFb-MYH11 | 2 (4) | 6 (11) | NS |
|
FLT3-ITD | 6 (11) | 1 (2) | NS |
| FLT3
D835 | 1 (2) | 2 (4) | NS |
| N-RAS (codon
12/13) | 3 (6) | 3 (5) | NS |
| K-RAS (codon
12/13) | 1 (2) | 1 (2) | NS |
| Othera | 1 (2) | 7 (13) | |
 | Table IIIRelative expression levels of the
gene-related abnormalities in AML. |
Table III
Relative expression levels of the
gene-related abnormalities in AML.
| Gene | miR-126-5p
| P-value |
|---|
| Low | High |
|---|
| ABCB1 | 0.055±0.128 | 0.027±0.061 | NS |
| BAALC | 0.589±1.836 | 0.927±2.034 | NS |
| ERG | 0.251±0.346 | 0.184±0.190 | NS |
| MN1 | 0.137±0.493 | 0.117±0.215 | NS |
| JAK2 | 0.583±0.813 | 0.400±0.525 | NS |
| WT1 | 2,989±4,832 | 2,257±2,511 | NS |
Effects of miR-126-5p transfection on
drug sensitivity, Klotho expression and Akt phosphorylation
The miR-126-5p mimic was transfected into the human
cell lines, KG-1, K562 and HK-2. Transfection of the miR-126-5p
mimic into the KG-1 cells resulted in decreased sensitivity to
cytarabin, but not to idarubicin. Transfection of the miR-126-5p
inhibitor did not alter the drug sensitivity (Table IV). The expression level of
Klotho mRNA was decreased following transfection with the
miR-126-5p mimic. Transfection of the miR-126-5p mimic also
significantly decreased the expression levels of the Klotho protein
(relative expression level; control, 1.00±0.03; miR-126-5p mimic,
0.61±0.13, mean ± standard error; p=0.046, Student's t-test, n=3).
A typical western blot analysis is shown in Fig. 2A. The transfection of the miR-126-5p
mimic as well as the positive control siRNA downregulated the
expression of Klotho (Fig.
2A). The same actions were observed in the K562 and HK-2 cells
(Fig. 2B). Azuma et al
reported that Klotho mRNA and protein are abundantly
expressed in HK-2 cells (30);
thus, the transfection effect of miR-126-5p into HK-2 cells was
also evaluated. Transfections of the miR-126-5p mimic into KG-1 or
K562 cells as well as into the HK-2 cells downregulated the
expression of Klotho. The expression level of Klotho
mRNA was also decreased by the transfection with the miR-126-5p
mimic in the K562 or HK-2 cells. Expression levels of the Klotho
protein were decreased by the transfection with the miR-126-5p
mimic in the HK-2 cells (Fig. 2B).
Transfection of the miR-126-5p mimic resulted in an elevation in
the phosphorylation of Akt in the KG-1 cells (Fig. 2C). Transfection of the miR-126-5p
mimic also significantly increased the expression of the
phosphorylated forms of Akt in the HK-2 cells (relative expression
level: control, 1.00±0.14; miR-126-5p mimic, 2.22±0.25, mean ±
standard error; p=0.005, Student's t-test, n=4; Fig. 2D). Transfection of the miR-126-5p
mimic or inhibitor did not affect the expression or phosphorylation
of JAK2, STAT3, STAT5 or Actin (Fig.
2C), or the mRNA expression of ABCB1, BAALC,
ERG, MN1, JAK2, WT1 or ACTB
(data not shown).
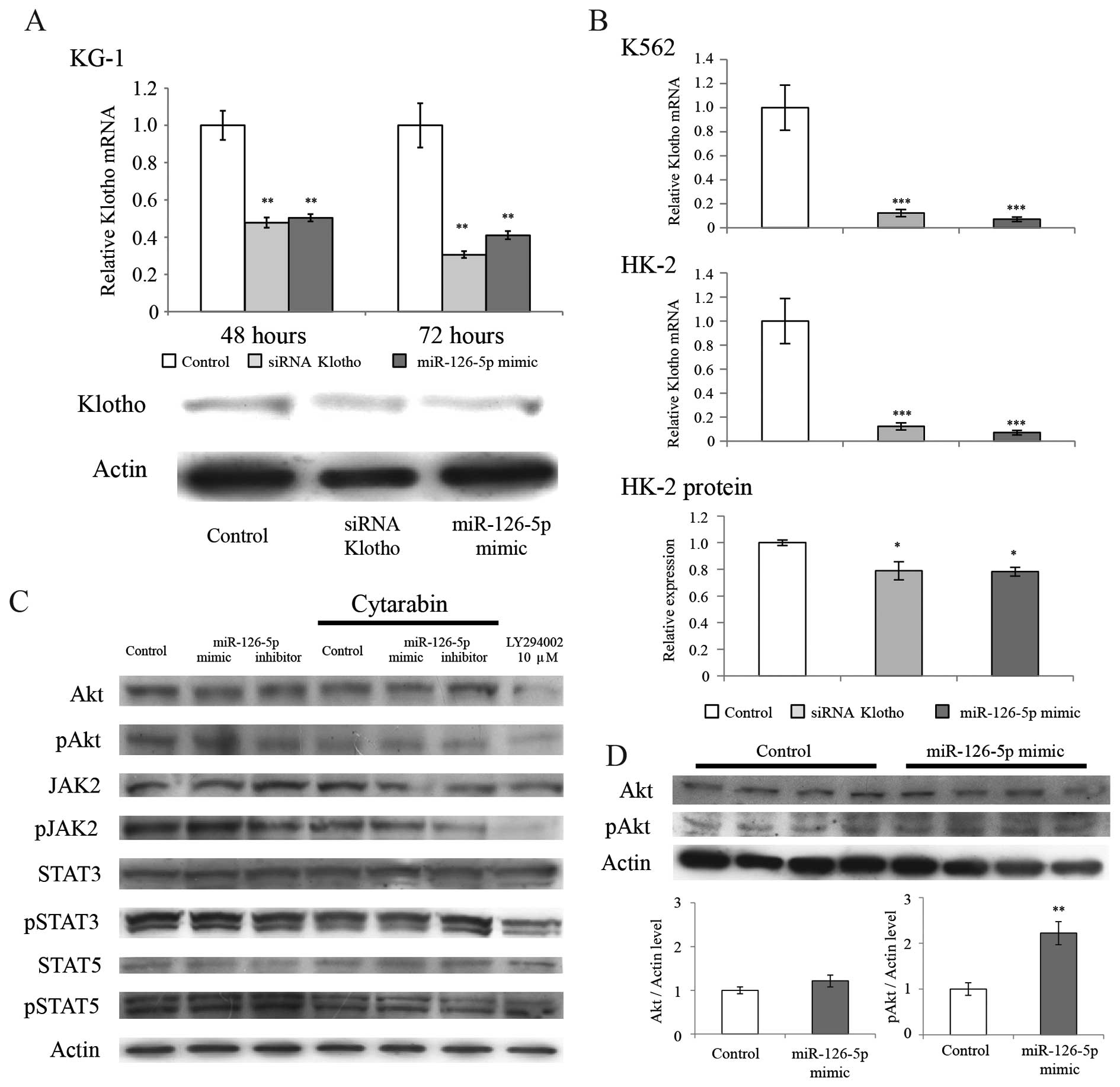 | Figure 2Effects of transfection with the
miR-126-5p mimic on the expression of Klotho, Akt, and JAK/STAT
kinases. (A) The relative level of the means and the standard error
of Klotho/Actin expression is indicated. Cells were
incubated for 48 or 72 h. Klotho mRNA expression levels were
significantly decreased following transfection of the miR-126-5p
mimic. A positive control, the siRNA of klotho, also
significantly decreased Klotho mRNA levels. Comparisons
between the 3 groups were performed with the Tukey-Kramer test,
**p<0.01 significantly different from the control
group. Transfection of the miR-126-5p mimic also significantly
decreased expression levels of the Klotho protein (relative
expression level: control, 1.00±0.03; miR-126-5p mimic, 0.61±0.13;
mean ± standard error, p=0.046, Student's t-test, n=3). This
transfection did not affect the expression level of actin. (B) The
miR-126-5p mimic or siRNA of Klotho was transfected into the
K562 or HK-2 cells. The relative level of the means and the
standard error of Klotho/Actin expression is indicated. The
cells were incubated for 72 h. Klotho mRNA expression levels
were also significantly decreased following transfection of the
miR-126-5p mimic. Comparisons between the 3 groups were performed
with the Tukey-Kramer test, *p<0.05,
***p<0.001, significantly different from the control
group. (C) Western blot analysis of Akt, the phosphorylated forms
of Akt at serine 473 (pAkt), JAK2, the phosphorylated forms of JAK2
at tyrosine 1007 (pJAK2), STAT3, the phosphorylated forms of STAT3
at tyrosine 705 (pSTAT3), STAT5, the phosphorylated forms of STAT5
at tyrosine 694 (pSTAT5) and actin. The miR-126-5p mimic or
inhibitor was transfected into KG-1 cells. The cells were incubated
for 24 h and then administered cytarabin at a dose of 60 nM for 120
h. The PI3K inhibitor, LY294002 was administered at a dose of 10
μM for 48 h as a positive control for the decline in the
phosphorylation of Akt. (D) Western blot analysis of Akt, the
phosphorylated forms of Akt at serine 473 (pAkt), and actin. The
miR-126-5p mimic was then transfected into KG-1 cells. Transfection
of the miR-126-5p mimic significantly increased the expression of
the phosphorylated forms of Akt (relative expression level:
control, 1.00±0.14; miR-126-5p mimic, 2.22±0.25; mean ± standard
error, **p<0.01, Student's t-test, n=4). This
transfection did not affect the expression levels of actin and
Akt. |
 | Table IVEffects of the mimic or inhibitor
miR-126-5p transfection on drug sensitivity in the KG-1 cells. |
Table IV
Effects of the mimic or inhibitor
miR-126-5p transfection on drug sensitivity in the KG-1 cells.
| NC | miR-126-5p
|
|---|
| Mimic | Inhibitor |
|---|
| Idarubicin | 38.0±1.6 | 43.8±2.4 | 46.0±1.6 |
| Cytarabin | 35.1±7.1 | 54.0±5.3a | 36.6±2.5 |
Discussion
The present study revealed two important issues:
Firstly, higher levels of miR-126-5p/3p resulted in a poorer
prognosis and secondly, miR-126-5p suppressed the expression of
Klotho mRNA. One hundred and nine eligible patients who
received intensive therapy were enrolled in the present study.
Patients were divided into high and low groups based on their
miR-126-5p expression levels. No significant differences were
observed in the patient characteristics or the gene mutation
statuses between the high and low groups. No significant
differences were observed in the expression levels of gene-related
abnormalities in AML between the two groups (Table III). These results suggest that
the expression levels of the gene-related abnormalities were not
related to prognosis in these groups.
The high expression group had a poorer prognosis
than that of the low group (Fig.
1). The functions of miR-126-3p have been reported previously
in an in vitro study; the miR-126-3p mimic was shown to
inhibit cell apoptosis, increase cell viability and enhance
proliferation and differentiation (7,16–19).
These findings suggest that the upregulation of miR-126-3p is a
predictor for poor prognosis. This was the first study to suggest
that higher expression of miR-126-3p results in a poorer prognosis
in AML patients. The present study showed that the high group had a
poorer prognosis and an interaction was not observed between the
hazard ratios of miR-126-5p and miR-126-3p (p=0.73). These results
suggest that the upregulation of miR-126-5p may enhance the
malignant potential of AML.
Recent studies have shown that miR-126-5p rescued
the proliferation of endothelial cells by suppressing Dlk1, and
decreased the proliferation of stromal cells (20–22).
The overexpression of miR-126-3p/5p was shown to significantly
induce apoptosis, and activate caspase-3 and caspase-7 by directly
regulating ADAM9 and MMP7 in melanoma (23). These findings suggested that the
upregulation of miR-126-5p resulted in opposing effects in
different cell types; miR-126-5p inhibited proliferation in stromal
cells, but activated proliferation in endothelial cells and
inhibited apoptosis in melanoma cells. In the present study, the
transfection of miR-126-5p into KG-1 cells induced drug resistance
to cytarabin (Table IV) and the
higher expression of miR-126-5p was associated with a poorer
prognosis. The results of the present study revealed that the
higher expression of miR-126-5p inhibited apoptosis induced by the
cytarabin treatment, leading to a poor prognosis.
Bioinformatics, microRNA.org
(http://www.microrna.org), is a comprehensive
resource of microRNA target predictions and expression profiles.
Target predictions are based on the development of a miRanda
algorithm, which incorporates current biological knowledge on
target rules and on the use of an up-to-date compendium of
mammalian microRNAs (24). Klotho
has been identified as an anti-aging gene, and also plays an
important role in cancer tumorigenesis, cell survival,
differentiation and metastasis (8).
Wang et al reported that the knockdown of Klotho inhibited
apoptosis, which significantly increased the expression of
phospho-Akt and increased resistance to cisplatin in A549/DDP cells
(11). A previous study reported
that Klotho exerted an inhibitory effect on the IGF-1 pathway in
both breast and pancreatic cancer cells (25–27).
The PI3K/Akt pathway is one of the important downstream pathways of
the IGF-1 pathway, and numerous studies have confirmed its role in
the apoptosis of cancer cells (28–30).
The present study demonstrated that miR-126-5p inhibited the
expression of Klotho and upregulated the phosphorylation of Akt
(Fig. 2). The present study and the
previous findings collectively suggest that miR-126-5p may induce
drug resistance by activating phospho-Akt, which targets
Klotho.
In conclusion, the results of the present study
demonstrated that the higher expression of miR-126-5p/3p in AML may
result in a poor prognosis. Furthermore, miR-126-5p induced drug
resistance to cytarabine by enhancing phosphorylation of Akt.
Acknowledgments
The authors would like to thank all the patients who
provided bone marrow samples, as well as patient information,
medical and nursing staff working at the North Japan Hematology
Study Group: The principal investigators of the study, in
alphabetical order by city: Asahikawa - Y. Kakinoki (Asahikawa City
Hospital); Hakodate - Y. Tsutsumi (Hakodate Municipal Hospital), T.
Kawamura (Nanae New Hospital); Kushiro - T. Miyagishima (Kushiro
Rosai Hospital); Obihiro - H. Kobayashi (Obihiro-Kosei Hospital);
Sapporo - A. Shigematsu, M. Nishio (Hokkaido University Hospital),
H. Iwasaki (Sapporo-Kosei General Hospital), K. Imai (Sapporo
Hokuyu Hospital), M. Kurosawa (National Hospital Organization,
Hokkaido Cancer Center), M. Morioka, A. Mori (Aiiku Hospital), S.
Yamamoto (Sapporo City General Hospital), Y. Haseyama (KKR Sapporo
Medical Center Tonan Hospital); Toyama - A. Wada (Toyama University
Hospital). The authors would like to thank Drs. Kumiko Kasashi and
Takehiro Yamada at the Department of Pharmacy, Hokkaido University
Hospital, for their helpful contributions. The present study was
supported by the Shimabara Science Promotion Foundation and the
Training Program for Oncology Professionals in Hokkaido by the
Ministry of Education, Culture, Sports Science and Technology,
Japan.
References
|
1
|
Mrózek K, Heerema NA and Bloomfield CD:
Cytogenetics in acute leukesmia. Blood Rev. 18:115–136. 2004.
View Article : Google Scholar
|
|
2
|
Burnett A, Wetzler M and Löwenberg B:
Therapeutic advances in acute myeloid leukemia. J Clin Oncol.
29:487–494. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Marcucci G, Haferlach T and Döhner H:
Molecular genetics of adult acute myeloid leukemia: Prognostic and
therapeutic Implications. J Clin Oncol. 29:475–486. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Byrd JC, Mrózek K, Dodge RK, et al:
Pretreatment cytogenetic abnormalities are predictive of induction
success, cumulative incidence of relapse, and overall survival in
adult patients with de novo acute myeloid leukemia: results from
Cancer and Leukemia Group B (CALGB 8461). Blood. 100:4325–4336.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Walker A and Marcucci G: Molecular
prognostic factors in cytogenetically normal acute myeloid
leukemia. Expert Rev Hematol. 5:547–558. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Esquela-Kerscher A and Slack FJ: Oncomirs
- microRNAs with a role in cancer. Nat Rev Cancer. 6:259–269. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li Z, Lu J, Sun M, et al: Distinct
microRNA expression profiles in acute myeloid leukemia with common
translocations. Proc Natl Acad Sci USA. 105:15535–15540. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhou X and Wang X: Klotho: a novel
biomarker for cancer. J Cancer Res Clin Oncol. Aug 3–2014.Epub
ahead of print. View Article : Google Scholar
|
|
9
|
Kuro-o M: Klotho as a regulator of
oxidative stress and senescence. Biol Chem. 389:233–241. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hu MC, Shiizaki K, Kuro-o M and Moe OW:
Fibroblast growth factor 23 and Klotho: physiology and
pathophysiology of an endocrine network of mineral metabolism. Annu
Rev Physiol. 75:503–533. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang Y, Chen L, Huang G, et al: Klotho
sensitizes human lung cancer cell line to cisplatin via PI3k/Akt
pathway. PLoS One. 8:e573912013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu F, Wu S, Ren H and Gu J: Klotho
suppresses RIG-I-mediated senescence-associated inflammation. Nat
Cell Biol. 13:254–262. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Baldus CD, Tanner SM, Ruppert AS, et al:
BAALC expression predicts clinical outcome of de novo acute myeloid
leukemia patients with normal cytogenetics: a Cancer and Leukemia
Group B Study. Blood. 102:1613–1618. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fukao T, Fukuda Y, Kiga K, et al: An
evolutionarily conserved mechanism for microRNA-223 expression
revealed by microRNA gene profiling. Cell. 129:617–631. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shibayama Y, Iwashita Y, Yoshikawa Y, et
al: Effect of 5-fluoro-uracil treatment on SN-38 absorption from
intestine in rats. Biol Pharm Bull. 34:1418–1425. 2011. View Article : Google Scholar
|
|
16
|
Zhang J, Du YY, Lin YF, Chen YT, Yang L,
Wang HJ and Ma D: The cell growth suppressor, miR-126, targets
IRS-1. Biochem Biophys Res Commun. 377:136–140. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ebrahimi F, Gopalan V, Smith RA and Lam
AK: miR-126 in human cancers: Clinical roles and current
perspectives. Exp Mol Pathol. 96:98–107. 2004. View Article : Google Scholar
|
|
18
|
Shen WF, Hu YL, Uttarwar L, Passegue E and
Largman C: MicroRNA-126 regulates HOXA9 by binding to the homeobox.
Mol Cell Biol. 28:4609–4619. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cammarata G, Augugliaro L, Salemi D, et
al: Differential expression of specific microRNA and their targets
in acute myeloid leukemia. Am J Hematol. 85:331–339.
2010.PubMed/NCBI
|
|
20
|
Schober A, Nazari-Jahantigh M, Wei Y, et
al: 2014. MicroRNA-126-5p promotes endothelial proliferation and
limits atherosclerosis by suppressing Dlk1. Nat Med. 20:368–376.
2014. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu Z, Yin H, Liu T, et al: MiR-126-5p
regulates osteoclast differentiation and bone resorption in giant
cell tumor through inhibition of MMP-13. Biochem Biophys Res
Commun. 443:944–949. 2014. View Article : Google Scholar
|
|
22
|
Zhou W, Yin H, Wang T, et al: MiR-126-5p
regulates osteolysis formation and stromal cell proliferation in
giant cell tumor through inhibition of PTHrP. Bone. 66:267–276.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Felli N, Felicetti F, Lustri AM, et al:
miR-126&126* restored expressions play a tumor suppressor role
by directly regulating ADAM9 and MMP7 in melanoma. PLoS One.
8:e568242013. View Article : Google Scholar
|
|
24
|
Betel D, Wilson M, Gabow A, Marks DS and
Sander C: 2008. The microRNA.org resource: targets and expression.
Nucleic Acids Res. 36:D149–D153. 2008. View Article : Google Scholar
|
|
25
|
Wolf I, Levanon-Cohen S, Bose S, et al:
Klotho: a tumor suppressor and a modulator of the IGF-1 and FGF
pathways in human breast cancer. Oncogene. 27:7094–7105. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Abramovitz L, Rubinek T, Ligumsky H, Bose
S, Barshack I, Avivi C, Kaufman B and Wolf I: KL1 internal repeat
mediates klotho tumor suppressor activities and inhibits bFGF and
IGF-I signaling in pancreatic cancer. Clin Cancer Res.
17:4254–4266. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen B, Wang X, Zhao W and Wu J: Klotho
inhibits growth and promotes apoptosis in human lung cancer cell
line A549. J Exp Clin Cancer Res. 29:992010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Poh TW and Pervaiz S: LY294002 and
LY303511 sensitize tumor cells to drug-induced apoptosis via
intracellular hydrogen peroxide production independent of the
phosphoinositide 3-kinase-Akt pathway. Cancer Res. 65:6264–6274.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hayakawa J, Ohmichi M, Kurachi H, et al:
Inhibition of BAD phosphorylation either at serine 112 via
extracellular signal-regulated protein kinase cascade or at serine
136 via Akt cascade sensitizes human ovarian cancer cells to
cisplatin. Cancer Res. 60:5988–5994. 2000.PubMed/NCBI
|
|
30
|
Shibata T, Kokubu A, Tsuta K and Hirohashi
S: Oncogenic mutation of PIK3CA in small cell lung carcinoma: A
potential therapeutic target pathway for chemotherapy-resistant
lung cancer. Cancer Lett. 283:203–211. 2009. View Article : Google Scholar : PubMed/NCBI
|
















