Introduction
Nasopharyngeal carcinoma (NPC) is one of the most
common cancers in Southern China, Southeast Asia, the Arctic, and
the Middle East/North Africa (1).
Epstein-Barr virus infection, genetic factors, dietary and
environmental factors are risk factors for the development of NPC
(2). Due to the unique anatomical
location of the nasopharynx where many important nerves and vessels
are located, surgery is not the primary choice for NPC treatment.
Since NPC cells are sensitive to ionizing radiation (IR),
radiotherapy is now the primary therapy for NPC patients (3). However, some carcinoma cells are
resistant to IR. These radioresistant cells remain the major cause
of local recurrence and metastasis of NPC (4). Therefore, decreasing the
radioresistance of NPC cells could help improve NPC patient
prognosis.
SHP-1, also called PTPN6 (5), is an SH2 domain-containing protein
tyrosine phosphatase (PTP). It consists of 17 exons and 16 introns
and spans ~17 kb (6,7). SHP-1 is highly expressed in normal
hematopoietic cells (8) and is
weakly expressed in several hematological malignancies, including
Burkitt’s (9), natural killer cell
(10) and diffuse large cell
lymphomas, Hodgkin’s disease (11)
and chronic myeloid leukemia (12).
However, some studies have found that SHP-1 is highly expressed in
certain epithelial carcinoma cells, such as ovarian and breast cell
lines (13). Although many studies
have been conducted concerning SHP-1 in hematological tumors, the
function of SHP-1 in solid tumors, particularly in NPC, is mostly
unknown.
Our previous study found that SHP-1 is overexpressed
in NPC tissues and is associated with local recurrence after
radiotherapy (14). Knockdown of
SHP-1 by siRNA in the NPC cell line CNE-2 and in the non-small cell
lung cancer (NSCLC) cell line A549 resulted in increased
radiosensitivity (15,16). These results suggest that
overexpression of SHP-1 may be related to radioresistance.
In the present study, we aimed to further examine
whether increased SHP-1 contributes to radioresistance of NPC CNE-2
cells. We also investigated how it is related to DNA double-strand
break (DSB) repair, cell cycle arrest and cell apoptosis.
Materials and methods
Cell culture and irradiation
procedure
The NPC cell line CNE-2 was obtained from the Cell
Bank of the Sun Yat-Sen university (Guangzhou, China) and cultured
in RPMI-1640 medium (HyClone, Logan, UT, USA) supplemented with 12%
fetal bovine serum (Gibco, Grand Island, NY, USA) and 1%
penicillin/streptomycin (HyClone). The cells were maintained at
37°C in a humidified incubator with 5% CO2 and 95% room
air. Irradiation was performed at room temperature with single
doses of X-rays using a linear accelerator (Primus K; Siemens,
Munich, Bayern, Germany) with 6-Mv photons/100-cm focus-surface
distance and a dose rate of 2.0 Gy/min.
SHP-1 is upregulated by
lentiviral-mediated gene knock-in
Both the SHP-1 gene sequence and a negative oligo
sequence were inserted into pEZ-Lv201-green fluorescent protein
(GFP) lentiviral vectors (GeneCopoeia, Guangzhou, China). After
confirmation of the constructed plasmids by DNA sequencing, the
lentiviruses were then transfected into 293T cells. LP-H1802Lv201
is a lentivirus containing the SHP-1 gene and LP-NegLv201 is a
negative control containing a negative oligo sequence. Supernatants
containing the lentiviruses were harvested, purified and the titer
of lentiviruses was determined. Both lentiviral stocks were
transfected into the CNE-2 cells. Fifty microliters of lentivirus
stock (LP-H1802Lv201 and LP-NegLv201) was added to the CNE-2 cells.
Puromycin (Sigma-Aldrich, St. Louis, MO, USA) was used to screen
cells transfected with the lentivirus at a concentration of 2
μg/ml. RNA was extracted and RT-qPCR was used to detect
SHP-1 mRNA expression. Total protein was isolated and the
expression of SHP-1 was detected by western blotting. The
efficiency of infection was observed by fluorescence
microscopy.
Colony formation assay
Cells were seeded into 6-well culture plates and
irradiated the next day at distinct doses (0, 2, 4, 6 and 8 Gy).
The plates were incubated for 14 days, fixed with methanol, and
stained with Giemsa (both from Wuhan Google Biotechnology Ltd. Co.,
Wuhan, China), and colonies containing at least 50 cells were
counted as a clone. A multi-target single-hit model was used to
describe the survival fraction (SF). The equation SF = 1-(1 -
e−D/D0)N (where D is the radiation dose; e is
the bottom of the natural logarithm; D0 is the mean
death dose; and N is the extrapolated number) was used to fit the
cell survival curves.
Immunofluorescent assay (IFA)
Cells were irradiated with 2 Gy of X-rays, and
incubated for specified times after IR. The cells were harvested
and immunostained with anti-histone H2AX phosphorylation (γH2AX;
Abcam, Cambridge, UK) or anti-RAD51 (Millipore, Billerica, MA, USA)
antibodies. Then the cells were incubated with Dylight 549
goat-anti-rabbit IgG (Abbkine, Redlands, CA, USA). The nuclei were
visualized by staining with Hoechst 33258 (Wuhan Google
Biotechnology Ltd. Co.). Images were captured using an Olympus
laser scanning confocal microscope (Olympus Optical Co., Tokyo,
Honshu, Japan). For each treatment condition, fluorescently labeled
γH2AX foci or RAD51 were assessed by fluorescence microscopy in at
least 50 cells.
Cell cycle flow cytometry (FCM)
analysis
Cells were irradiated with 6 Gy of X-rays and
incubated for 24 h after IR. The cells were fixed overnight with
70% ethanol, and resuspended in PBS containing 1 mg/ml RNase and 50
μg/ml propidium iodide (both from Sigma-Aldrich). Cellular
DNA content was determined using a FACScan flow cytometer
(Becton-Dickinson, San Jose, CA, USA). Quantification of cells in
the g1, S, and g2/M phases was performed using CellQuest software
(BD Biosciences).
FCM analysis of apoptosis
Cells were exposed to 6 Gy of X-rays and incubated
for 24 h after IR. The cells were then collected and resuspended in
200 μl binding buffer and stained with 2 μl Annexin
V-APC and 4 μl of 7-AAD (all from Bestbio, Shanghai, China).
Analyses were performed using a FACScan flow cytometer
(Becton-Dickinson). Both APC-and 7-AAD-positive cells were
considered apoptotic cells.
Western blotting
Cells were harvested and lysed after the different
treatments. Protein lysate concentrations were determined using the
BCA protein assay (Wuhan Google Biotechnology Ltd. Co.). Equal
amounts of protein were separated by 8–15% SDS-PAGE (according to
molecular weight; Wuhan Google Biotechnology Ltd. Co.) and
transferred to PVDF membranes (Millipore). The membranes were
blocked with 5% BSA (Wuhan Google Biotechnology Ltd. Co.) and then
probed with anti-SHP-1 (Epitomics, Burlingame, CA, USA), anti-RAD51
(Millipore), anti-phospho-p53 (Cell Signaling Technology, Danvers,
MA, USA), anti-γH2AX (Abcam), anti-phospho-ATM kinase and
anti-phospho-ATR protein (both from Cell Signaling Technology),
anti-phospho-CHK1 (Abcam), anti-phospho-CHK2 (Cell Signaling
Technology) or anti-GAPDH (Santa Cruz Biotechnology, Inc., Dallas,
TX, USA) antibodies. After washing by TBST (Wuhan Google
Biotechnology Ltd. Co.), the membranes were incubated with
goat-anti-rabbit or goat-anti-mouse IgG (Invitrogen, Carlsbad, CA,
USA) and visualized by a chemiluminescence detection system (UVP
OptiCam 600; UVP Inc., Upland, CA, USA) using a chemiluminescence
kit (Invitrogen). GAPDH protein levels were used as a control to
verify equal protein loading. Image J 1.43b software (NIH,
Bethesda, MD, USA) was used to scan the protein bands and to
measure the optical density values.
RT-qPCR
Total RNA was extracted with TRIzol (Invitrogen),
and reverse transcription was used to obtain cDNA, according to the
manufacturer’s instructions for the Takara RT-PCR kit (Takara,
Shiga, Japan). Then, qPCR was performed according to the
manufacturer’s instructions using SYBR-Green in a PCR amplifier,
ABI Prism 7000 (both from Applied Biosystems, Foster City, CA,
USA). The StepOne™ software v2.1 was used to analyze the data. The
primer sequences for SHP-1 were: forward, 5′-ACCATCATCCACCTCAAGT
ACC-3′ and reverse, 5′-CTGAGCACAGAAAGCACGAA-3′. β-actin was used as
an internal control, and the primer sequences were: forward,
5′-GATGAGATTGGCATGGC TTT-3′ and reverse,
5′-CACCTTCACCGTTCCAGTTT-3′.
Statistical analysis
Experimental data are expressed as the mean ± SD
from at least three or more independent experiments. Differences in
the measured variables of the experimental and control groups were
assessed using a t-test (SPSS 21.0 software). The criterion for
statistical significance was p<0.05.
Results
Overexpression of SHP-1 by
lentiviral-mediated gene knock-in in CNE-2 cells
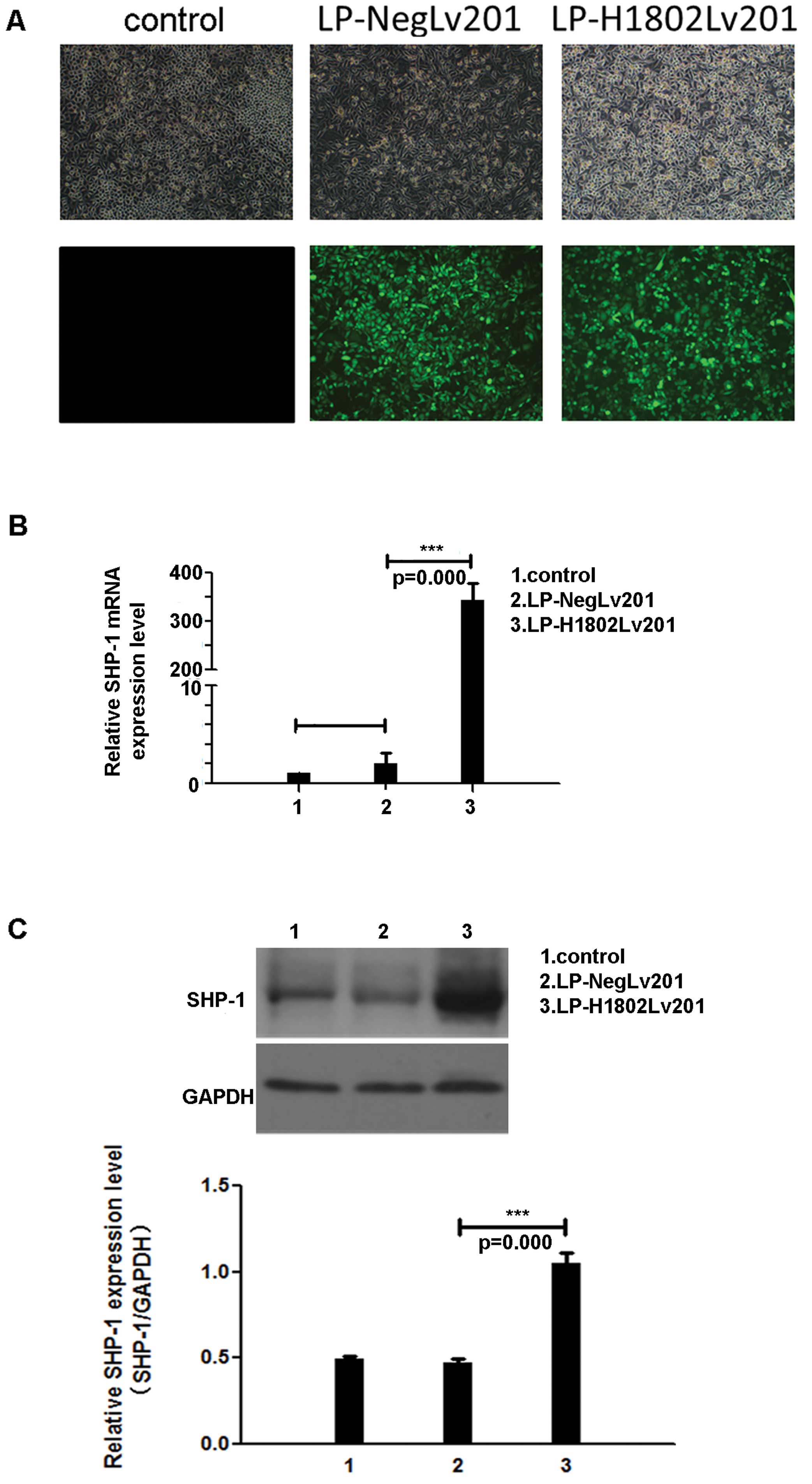
We transfected the NPC cell line CNE-2 with a
lentivirus containing the SHP-1 gene or a nonsense sequence
(referred to as LP-H1802Lv201 and LP-NegLv201 cells, respectively).
Fluorescence microscopy was used to observe GFP intensity and
transfection efficiency (Fig. 1A).
RT-PCR and western blot analyses were used to detect SHP-1
expression levels at the mRNA and protein levels, respectively.
RT-qPCR showed that in the LP-H1802Lv201 cells, SHP-1 mRNA
expression levels were increased by 300-fold (Fig. 1B). Western blot analyses also
indicated an increased expression of SHP-1 protein in the
LP-H1802Lv201 cells (Fig. 1C).
Upregulation of SHP-1 results in enhanced
radioresistance
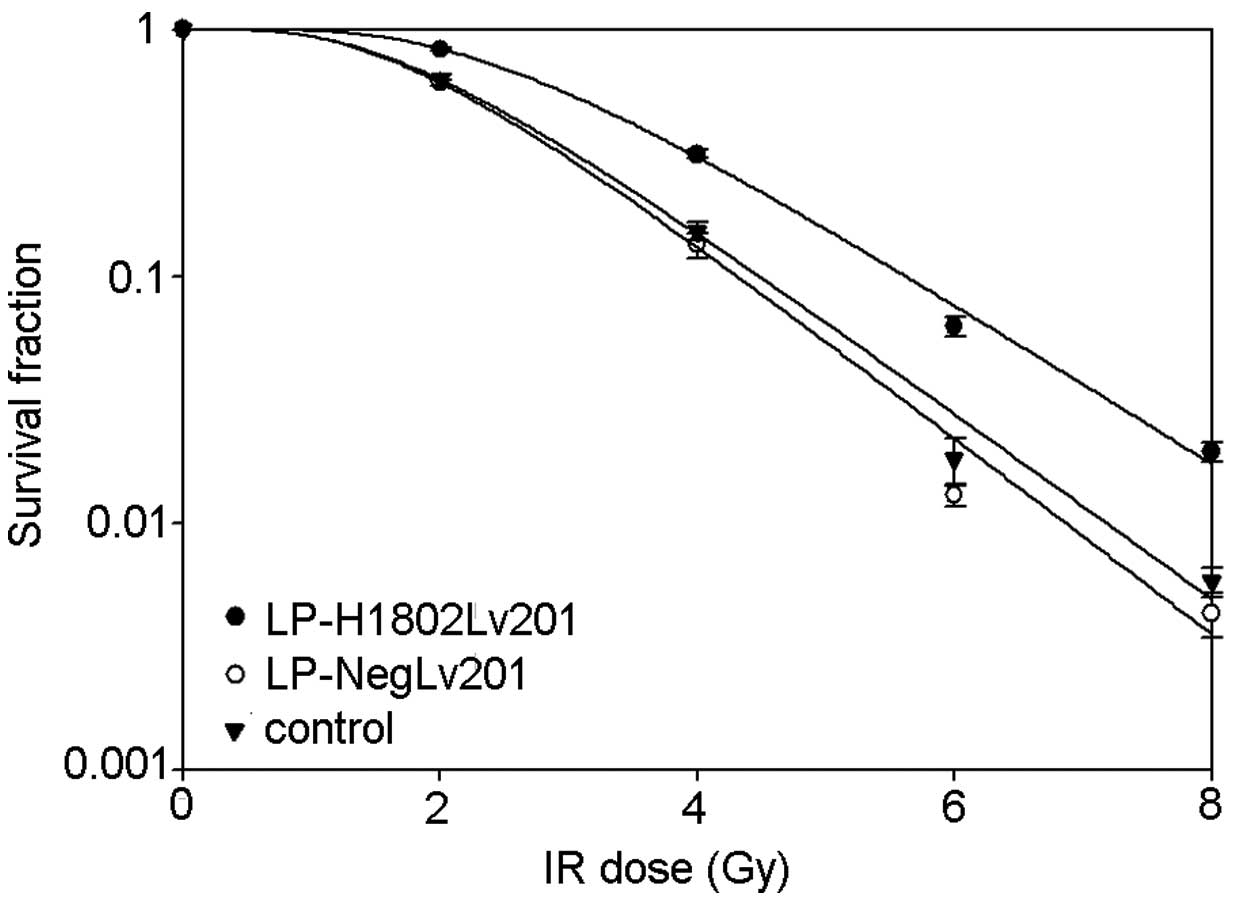
To determine the relationship between SHP-1 and
radio-resistance, colony formation assays were performed, and cell
survival curves were used to analyze the results. The shoulder area
under the survival curve was larger in the LP-H1802Lv201 cells
(Fig. 2). In contrast to the
control and LP-NegLv201 cells, LP-H1802Lv201 cells had increased
D0, Dq, N and SF2 (Table I) values, which represented a higher
radioresistance. The differences in these values were negligible
between the control and LP-NegLv201 cells. Therefore, we concluded
that upregulation of SHP-1 resulted in enhanced
radioresistance.
 | Table IParameters of the radiosensitivity in
the three cell lines. |
Table I
Parameters of the radiosensitivity in
the three cell lines.
| Parameters | CNE-2 | LP-NegLv201 | LP-H1802Lv201 |
|---|
| D0 | 1.153 | 1.092 | 1.329 |
| Dq | 1.768 | 1.774 | 2.255 |
| N | 5.078 | 5.401 | 7.170 |
| SF2 | 0.610 | 0.627 | 0.835 |
DNA DSB repair is enhanced in the
SHP-1-overexpressing cells
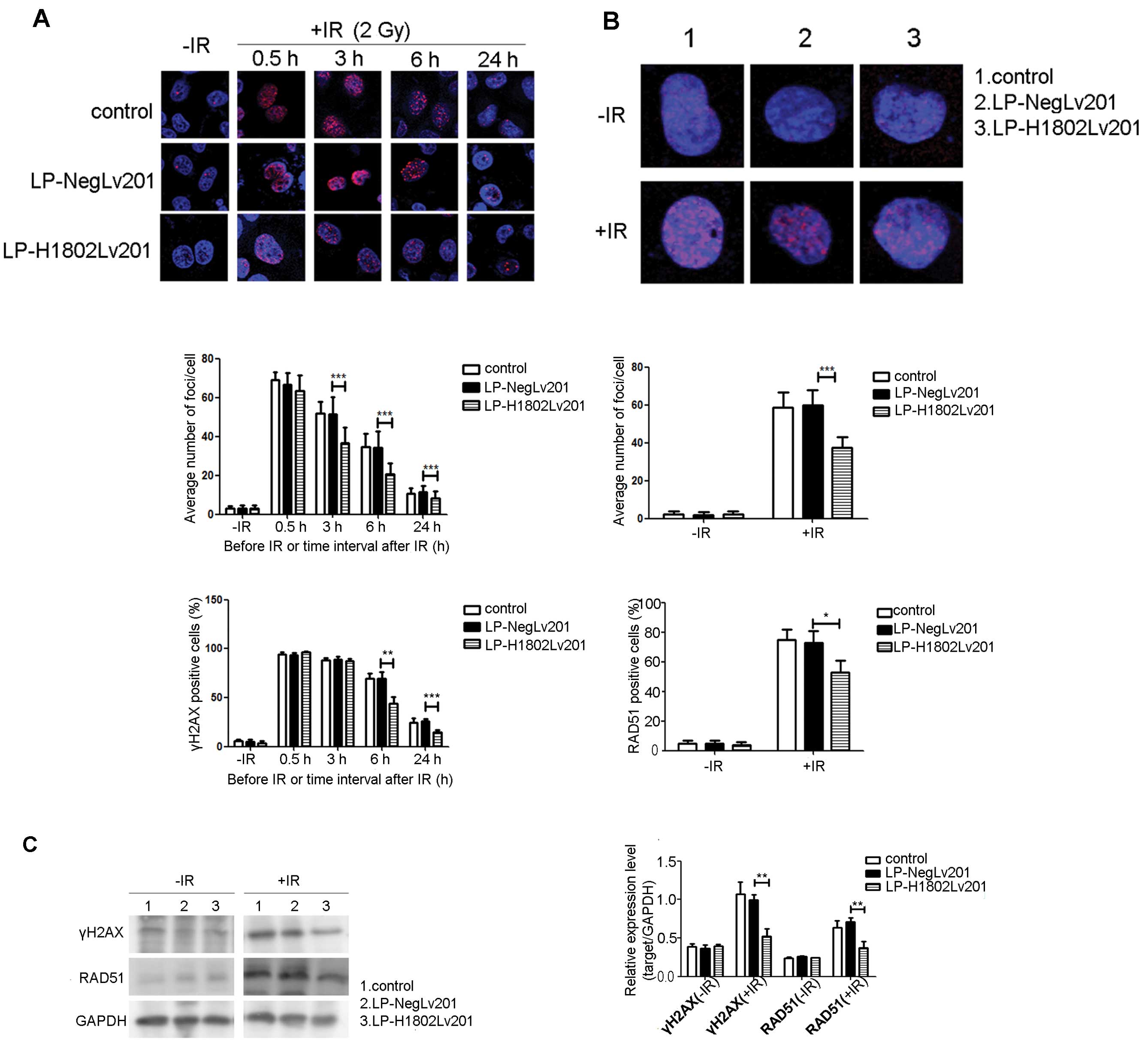
To determine DNA DSB repair, we used an anti-γH2AX
to immunofluorescently stain γH2AX foci at different time-points
after exposure to IR. As shown in Fig.
3A without exposure to IR, few γH2AX foci were observed in the
three cell groups. After irradiation with 2 Gy IR, γH2AX foci
rapidly increased. The numbers of γ-H2AX foci in the 3 groups were
almost equal at 0.5 h after IR. However, foci in the LP-H1802Lv201
cells disappeared more quickly. In contrast to the control and
LP-NegLv201 cells, γH2AX foci in the LP-H1802v201 cells were
significantly decreased at 3, 6 and 24 h after IR. Cells having
more than 10 γH2AX foci were scored as γH2AX-positive cells. We
found that the percentage of γH2AX-positive cells was significantly
decreased in the LP-H1802Lv201 cell group at 6 and 24 h after IR.
In contrast, at 0.5 and 3 h after IR, the percentages of
γH2AX-positive cells in the 3 groups did not differ significantly.
We also assessed RAD51 foci at 6 h after IR or under the condition
without IR. As shown in Fig. 3B,
without IR, RAD51 foci did not show a significant difference in the
3 groups. At 6 h after IR, the number of RAD51 foci in the
LP-H1802Lv201 cells was significantly less than that in the control
and LP-NegLv201 cells. Cell having more than 10 RAD51 foci were
scored as RAD51-positive cells. The percentage of RAD51-positive
cells was also lower in the LP-H1802Lv201 cell group. Western blot
analyses were also used to assess γH2AX and RAD51 expression at 24
h after IR or under the condition without IR (Fig. 3C). Without IR, expression levels of
γH2AX and RAD51 in the 3 cell groups were almost equal. At 24 h
after IR, expression levels of γH2AX and RAD51 were significantly
increased in contrast to the condition without IR. However,
expression levels of γH2AX and RAD51 were lower in the
LP-H1802Lv201 cells. We concluded that IR caused DNA DSBs equally
in the 3 cell groups. Yet, SHP-1-overexpressing cells showed an
enhanced DSB repair capacity.
SHP-1-overexpressing cells undergo
increased S phase arrest after IR
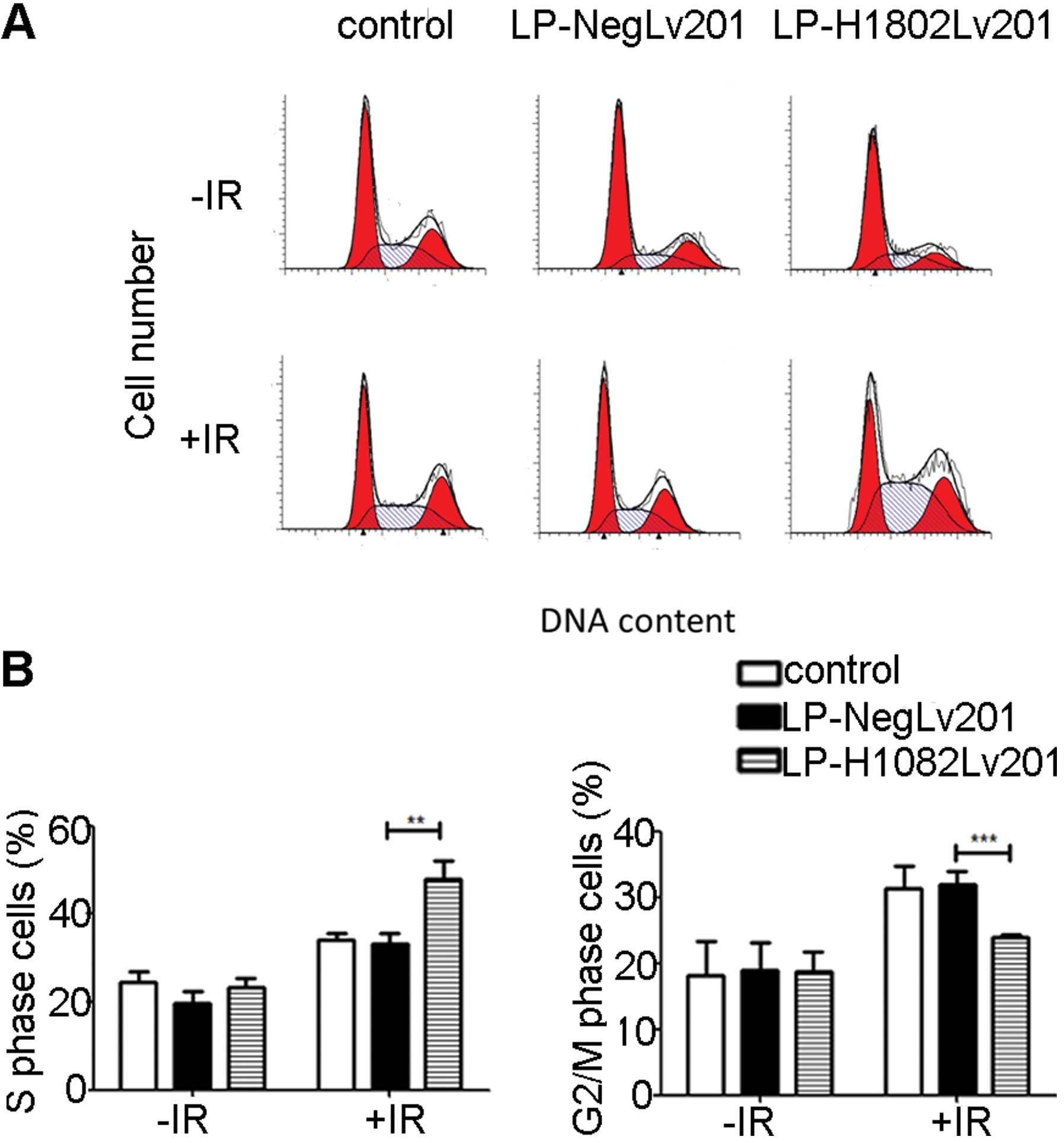
To evaluate how SHP-1 affects cell cycle
distribution, we used FCM to estimate the cell cycle changes.
Without IR, the cell fractions in the S phase did not show a
significant difference in the 3 cell groups. At 24 h after IR, the
percentage of S phase cells was significantly increased and the
percentage of g2/M phase cell group was decreased in the
LP-H1802Lv201 cells compared with the control and LP-NegLv201 cells
(Fig. 4). The results suggest that
overexpression of SHP-1 led to increased IR-induced S phase arrest
and thus decreased g2/M phase cells.
Overexpression of SHP-1 causes an
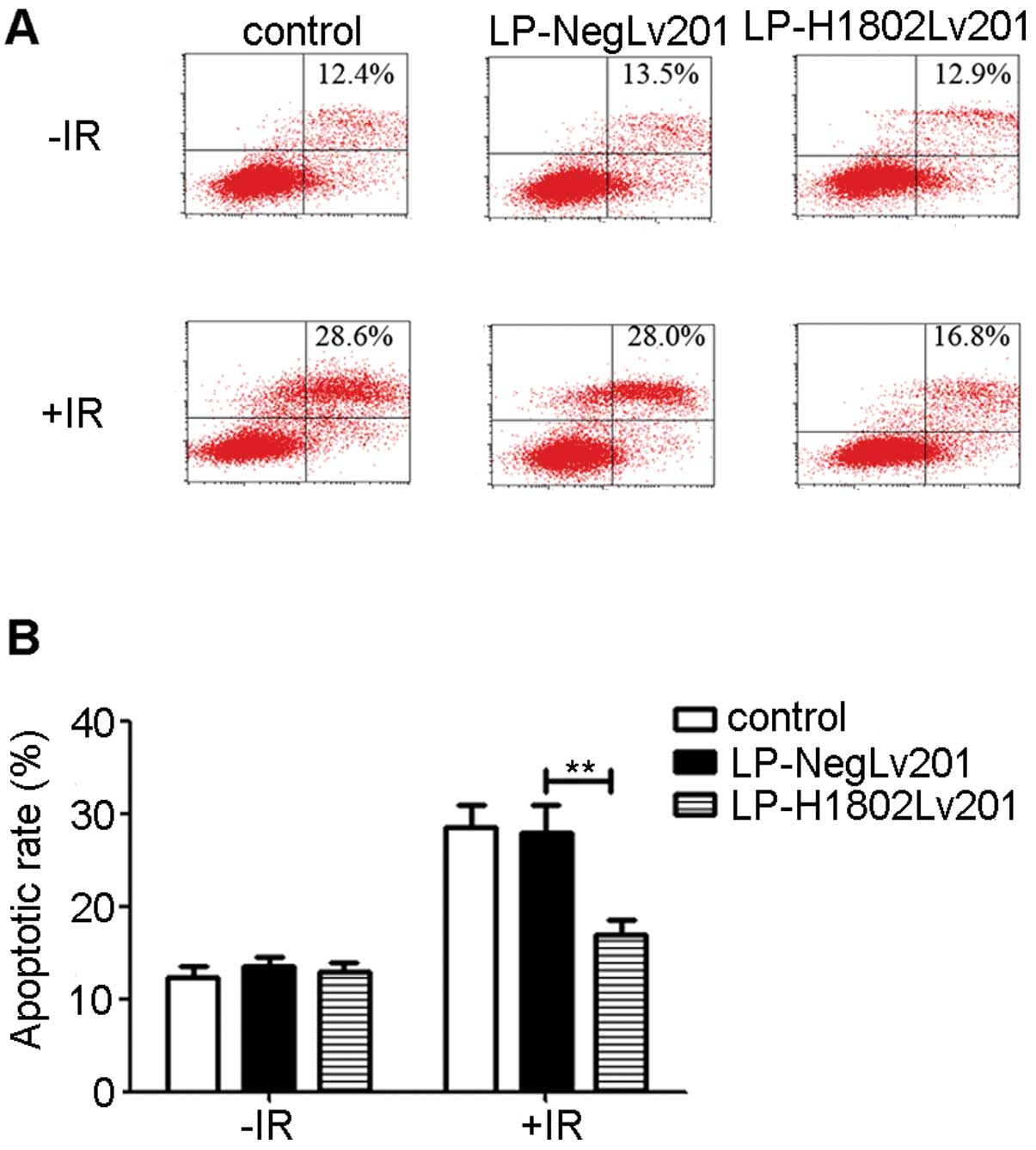
anti-apoptotic effect
Before IR, the apoptotic rate of the control,
LP-NegLv201 and LP-H1802Lv201 cells had no significant differences.
At 24 h after IR, the apoptotic rates of the 3 cell groups were
increased. However, the apoptotic rate of the LP-H1802Lv201 cells
was significantly lower than the rate in the control and
LP-NegLv201 cells (Fig. 5). These
data suggest that IR promoted apoptosis in the 3 cell groups.
However, LP-H1802Lv201 cells were more resistant to IR-induced
apoptosis. The results suggest that overexpression of SHP-1 had an
anti-apoptotic effect.
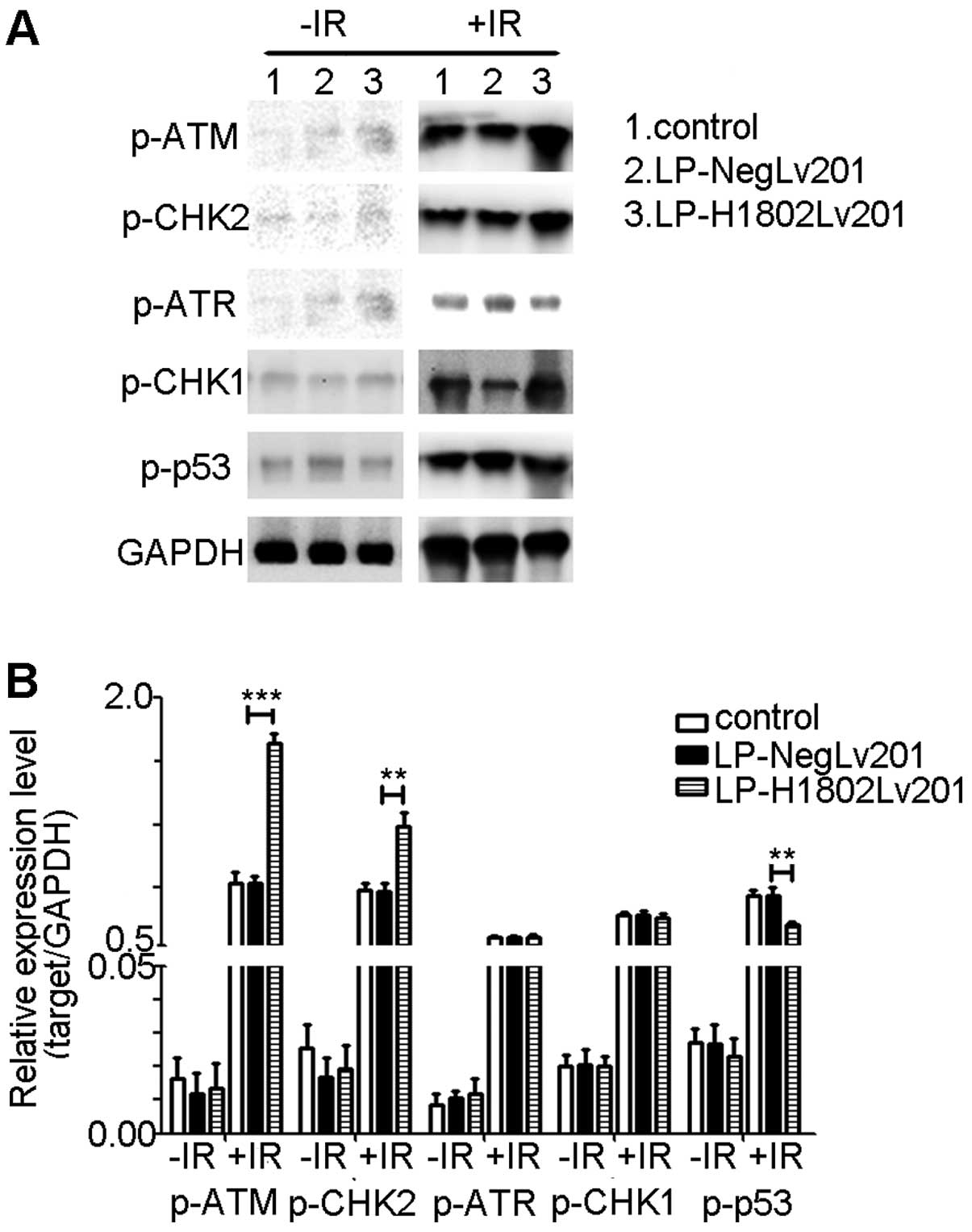
SHP-1-overexpressing cells show increased
activation of ATM and CHK2 and suppressed activation of p53 after
IR
To explore how the ATM/CHK1 and ATR/CHK1 pathways
were activated after IR, we determined the phosphorylation levels
of ATM (p-ATM), CHK2 (p-CHK2), ATR (p-ATR), CHK1 (p-CHK1) and p53
(p-p53). Results of the western blot analyses showed that the
phosphorylation levels of ATM, CHK2, ATR, CHK1 and p53 were
extremely low when cells did not receive IR. After radiation,
phosphorylation levels of these proteins were increased. Compared
with the control and LP-Neglv201 cells, LP-H1802lv201 cells had
relatively increased phosphor-ylation levels of ATM and CHK2, while
the phosphorylation of p53 was decreased. Phosphorylation of ATR
and CHK1 did not show a significantly difference (Fig. 6).
Discussion
In the past few decades, SHP-1 has been believed to
be a tumor-suppressor in many malignancies (10,17,18).
However, our previous research found that SHP-1 was overexpressed
in NPC tissues and was associated with local recurrence and
metastasis after radiotherapy (4).
Suppression of SHP-1 expression resulted in a higher
radiosensitivity (15,16). These results suggest that
overexpression of SHP-1 may be related to radioresistance and that
it may be a potential target to enhance NPC radiosensitivity.
In the present study, we investigated the effects of
SHP-1 on the radioresistance of NPC cells. We showed that
over-expression of SHP-1 enhanced DNA DSB repair, increased
IR-induced S phase arrest and decreased cell apoptosis, thus
resulting in radioresistance in CNE-2 cells. The result was
consistent with previous observations (15,16).
IR-induced cell death is a result of irreparable
DSBs (19). The repair response of
DSBs is one of the factors that influences radiosensitivity. DSBs
are mainly repaired by non-homologous end joining (NHEJ) and
homologous recombination. Repair of DSBs appeared within 30–60 min
after radiation. The majority of DSBs are repaired in 24 h. γH2AX
is a hallmark of DSB recognition and repair. Fewer γH2AX foci
represent a more rapid repair of DSBs and higher radio-resistance
(19–24). RAD51 is an important protein
involved in homologous recombination processes. Increased RAD51
expression is related to radioresistance of tumor cells (25). In the present study, we showed that
overexpression of SHP-1 in NPC cells decreased the expression of
γH2AX, which indicted enhanced repair of DSBs. Notably, RAD51
expression was decreased in the LP-H1802lv201 cells, which are
relatively radioresistant cells. One reason for this result may be
that we detected RAD51 expression only at one time-point after IR.
According to γH2AX expression, LP-H1802Lv201 cells had an enhanced
DSB repair capacity. Thus, the DSB repair peak of LP-H1802Lv201
cells should have appeared sooner than the control and LP-NegLv201
cells. When we detected the expression of RAD51, the DSB repair
peak may have transpired in the LP-H1802lv201 cells while this peak
was not yet achieved in the control and LP-Neglv201 cells.
Cells often respond to IR-induced DSBs by activating
cell cycle checkpoints, which play an importance role in
determining radiosensitivity. In general, S phase cells are the
most radioresistant, while G2/M phase cells are the most sensitive
to radiation (26). It has been
reported that abrogation of the G2/M checkpoint promotes IR-induced
cell death (27). The present data
showed that overexpression of SHP-1 increased the fraction of S
phase cells. At the same time, the cell fraction in the G2/M phase
was decreased, which may have resulted from S phase arrest. Thus,
we inferred that overexpression of SHP-1 contributed to the
radioresistance of NPC cells by increasing S phase arrest.
ATM kinase plays vital roles in IR-induced DNA
damage repair response (DDR). ATM is activated upon DNA damage, and
downstream effector kinases of ATM, including CHK2 and p53, are
also activated (28). Increased
activation of ATM/CHK2 enhances DNA damage repair, thus leading to
radioresistance. It has been reported that inhibition of ATM
activation increases apoptosis and enhances radiosensitivity. The
ATR/CHK1 pathway also has an influence on radio-resistance by
regulating homologous recombination repair. Overactivation of the
ATR/CHK1 pathway increased the radioresistance of tumor cells. When
DNA damage is not repaired, cells will be eliminated though
different mechanisms including p53-dependent apoptosis (28–32).
In the present study, we found that SHP-1-overexpressing cells had
an increased phosphorylation of the ATM/CHK2 pathway, while
phosphorylation of the ATR/CHK1 pathway did not show a difference
in the other two cell groups. These data suggest that
overexpression of SHP-1 enhanced DNA damage repair by activating
the ATM/CHK1 pathway, but not the ATR/CHK1 pathway, and enhanced
DNA damage repair resulted in decreased p53-dependent
apoptosis.
ATM also takes part in intra-S checkpoint response
to IR-induced DSBs through two separate pathways (33,34).
One pathway involves activation of CHK1, CHK2 and Cdc25A. CHK1 and
CHK2 are phosphorylated and activated by ATM, which leads to
phosphorylation and proteolysis of Cdc25A and then activates
intra-S checkpoint response (35).
The other pathway involves the cohesin subunits, Smc1 and Smc3.
Smc1 and Smc3 are phosphorylated by ATM and bind to Scc1 and SA1 or
SA2 to form cohesin, which plays important roles in homologous
recombination repair of DSBs (33,34,36–38).
In the present study, SHP-1-overexpressing cells showed an
increased S phase arrest accompanied by increased ATM and CHK2
activation after IR. However, activation of CHK1 was not increased
in the SHP-1-overexpressing cells. We inferred that IR-induced DSBs
activated the ATM/CHK2 pathway. Then Cdc25A was phosphorylated and
the intra-S checkpoint was activated, leading to S phase arrest.
However, whether the ATR/CHK1 pathway and ATM/Smc1/Smc3 are
involved in S phase arrest in SHP-1-overexpressing cells needs
further study.
In the present study, we demonstrated that
overexpression of SHP-1 was related to the acquired resistance to
IR in the NPC cell line CNE-2. Enhanced DSB repair, increased S
phase arrest and decreased apoptosis contributed to this acquired
radioresistance. Examining the SHP-1 expression level in tumor
tissues of NPC patients may help to predict prognosis.
Acknowledgments
The present study was supported by grants from the
Natural Sciences Foundation of China (no. 81301976) and the Wu
Jieping Medical Foundation.
Abbreviations:
|
NPC
|
nasopharyngeal carcinoma
|
|
IR
|
ionizing radiation
|
|
DSB
|
double-strand break
|
|
NSCLC
|
non-small cell lung cancer
|
|
GFP
|
green fluorescent protein
|
|
IFA
|
immunofluorescent assay
|
|
FCM
|
flow cytometry
|
|
DDR
|
DNA damage repair response
|
|
ATM
|
ataxia telangiectasia mutated
|
|
CHK2
|
checkpoint kinase 2
|
|
ATR
|
ataxia telangiectasia and Rad3-related
protein
|
|
CHK1
|
checkpoint kinase 1
|
|
NHEJ
|
non-homologous end joining
|
|
TKI
|
tyrosine kinase inhibitor
|
References
|
1
|
Chang ET and Adami HO: The enigmatic
epidemiology of nasopharyngeal carcinoma. Cancer Epidemiol
Biomarkers Prev. 15:1765–1777. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hung CM, Chang CC, Lin CW, Chen CC and Hsu
YC: GADD45γ induces g2/M arrest in human pharynx and nasopharyngeal
carcinoma cells by cucurbitacin E. Sci Rep. 4:64542014. View Article : Google Scholar
|
|
3
|
Lee AW, Sze WM, Au JS, Leung SF, Leung TW,
Chua DT, Zee BC, Law SC, Teo PM, Tung SY, et al: Treatment results
for nasopharyngeal carcinoma in the modern era: the Hong Kong
experience. Int J Radiat Oncol Biol Phys. 61:1107–1116. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Feng XP, Yi H, Li MY, Li XH, Yi B, Zhang
PF, Li C, Peng F, Tang CE, Li JL, et al: Identification of
biomarkers for predicting nasopharyngeal carcinoma response to
radiotherapy by proteomics. Cancer Res. 70:3450–3462. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lorenz U: SHP-1 and SHP-2 in T cells: two
phosphatases functioning at many levels. Immunol Rev. 228:342–359.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Banville D, Stocco R and Shen SH: Human
protein tyrosine phosphatase 1C (PTPN6) gene structure: alternate
promoter usage and exon skipping generate multiple transcripts.
Genomics. 27:165–173. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Evren S, Wan S, Ma XZ, Fahim S, Mody N,
Sakac D, Jin T and Branch DR: Characterization of SHP-1 protein
tyrosine phosphatase transcripts, protein isoforms and phosphatase
activity in epithelial cancer cells. Genomics. 102:491–499. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nakase K, Cheng J, Zhu Q and Marasco WA:
Mechanisms of SHP-1 P2 promoter regulation in hematopoietic cells
and its silencing in HTLv-1-transformed T cells. J Leukoc Biol.
85:165–174. 2009. View Article : Google Scholar :
|
|
9
|
Delibrias CC, Floettmann JE, Rowe M and
Fearon DT: Downregulated expression of SHP-1 in Burkitt lymphomas
and germinal center B lymphocytes. J Exp Med. 186:1575–1583. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Oka T, Yoshino T, Hayashi K, Ohara N,
Nakanishi T, Yamaai Y, Hiraki A, Sogawa CA, Kondo E, Teramoto N, et
al: Reduction of hematopoietic cell-specific tyrosine phosphatase
SHP-1 gene expression in natural killer cell lymphoma and various
types of lymphomas/leukemias: combination analysis with cDNA
expression array and tissue microarray. Am J Pathol. 159:1495–1505.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sato K, Horiuchi M, Yo R and Nakarai I: A
long survival case of small cell lung cancer synchronized with
renal cancer. Kyobu Geka. 44:251–253. 1991.In Japanese. PubMed/NCBI
|
|
12
|
Amin HM, Hoshino K, Yang H, Lin Q, Lai R
and Garcia-Manero G: Decreased expression level of SH2
domain-containing protein tyrosine phosphatase-1 (Shp1) is
associated with progression of chronic myeloid leukaemia. J Pathol.
212:402–410. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
López-Ruiz P, Rodriguez-Ubreva J, Cariaga
AE, Cortes MA and Colás B: SHP-1 in cell-cycle regulation.
Anticancer Agents Med Chem. 11:89–98. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Duró JC: Postmenopausal osteoporosis. Med
Clin (Barc). 85:506–509. 1985.In Spanish.
|
|
15
|
Peng G, Cao RB, Li YH, Zou ZW, Huang J and
Ding Q: Alterations of cell cycle control proteins SHP 1/2, p16,
CDK4 and cyclin D1 in radioresistant nasopharyngeal carcinoma
cells. Mol Med Rep. 10:1709–1716. 2014.PubMed/NCBI
|
|
16
|
Cao R, Ding Q, Li P, Xue J, Zou Z, Huang J
and Peng G: SHP1-mediated cell cycle redistribution inhibits
radiosensitivity of non-small cell lung cancer. Radiat Oncol.
8:1782013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Schibli RA: Lung biopsy in interstitial
pulmonary processes. Schweiz Med Wochenschr. 106:467–468. 1976.In
German. PubMed/NCBI
|
|
18
|
Tassidis H, Culig Z, Wingren AG and
Härkönen P: Role of the protein tyrosine phosphatase SHP-1 in
interleukin-6 regulation of prostate cancer cells. Prostate.
70:1491–1500. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jackson SP and Bartek J: The DNA-damage
response in human biology and disease. Nature. 461:1071–1078. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Reynolds P, Anderson JA, Harper JV, Hill
MA, Botchway SW, Parker AW and O’Neill P: The dynamics of Ku70/80
and DNA-PKcs at DSBs induced by ionizing radiation is dependent on
the complexity of damage. Nucleic Acids Res. 40:10821–10831. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jakob B, Splinter J, Conrad S, Voss KO,
Zink D, Durante M, Löbrich M and Taucher-Scholz G: DNA
double-strand breaks in heterochromatin elicit fast repair protein
recruitment, histone H2AX phosphorylation and relocation to
euchromatin. Nucleic Acids Res. 39:6489–6499. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lieber MR: The mechanism of double-strand
DNA break repair by the nonhomologous DNA end-joining pathway. Annu
Rev Biochem. 79:181–211. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Schmid TE, Dollinger G, Beisker W, Hable
V, Greubel C, Auer S, Mittag A, Tarnok A, Friedl AA, Molls M, et
al: Differences in the kinetics of gamma-H2AX fluorescence decay
after exposure to low and high LET radiation. Int J Radiat Biol.
86:682–691. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rao VA, Agama K, Holbeck S and Pommier Y:
Batracylin (NSC 320846), a dual inhibitor of DNA topoisomerases I
and II induces histone gamma-H2AX as a biomarker of DNA damage.
Cancer Res. 67:9971–9979. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Maacke H, Jost K, Opitz S, Miska S, Yuan
Y, Hasselbach L, Lüttges J, Kalthoff H and Stürzbecher HW: DNA
repair and recombination factor Rad51 is over-expressed in human
pancreatic adenocarcinoma. Oncogene. 19:2791–2795. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hall EJ and Giaccia A: Cell survival
curves. Radiobiology for the Radiologist. 7th edition. Lippincott
Williams and Wilkins; New York, NY: 2011
|
|
27
|
Feng Z, Xu S, Liu M, Zeng YX and Kang T:
Chk1 inhibitor Gö6976 enhances the sensitivity of nasopharyngeal
carcinoma cells to radiotherapy and chemotherapy in vitro and in
vivo. Cancer Lett. 297:190–197. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhou W, Sun M, Li GH, Wu YZ, Wang Y, Jin
F, Zhang YY, Yang L and Wang DL: Activation of the phosphorylation
of ATM contributes to radioresistance of glioma stem cells. Oncol
Rep. 30:1793–1801. 2013.PubMed/NCBI
|
|
29
|
Biddlestone-Thorpe L, Sajjad M, Rosenberg
E, Beckta JM, Valerie NC, Tokarz M, Adams BR, Wagner AF, Khalil A,
Gilfor D, et al: ATM kinase inhibition preferentially sensitizes
p53-mutant glioma to ionizing radiation. Clin Cancer Res.
19:3189–3200. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hu B, Wang H, Wang X, Lu HR, Huang C,
Powell SN, Huebner K and Wang Y: Fhit and CHK1 have opposing
effects on homologous recombination repair. Cancer Res.
65:8613–8616. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang H, Wang H, Powell SN, Iliakis G and
Wang Y: ATR affecting cell radiosensitivity is dependent on
homologous recombination repair but independent of nonhomologous
end joining. Cancer Res. 64:7139–7143. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang H, Hu B, Liu R and Wang Y: CHK1
affecting cell radio-sensitivity is independent of non-homologous
end joining. Cell Cycle. 4:300–303. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yazdi PT, Wang Y, Zhao S, Patel N, Lee EY
and Qin J: SMC1 is a downstream effector in the ATM/NBS1 branch of
the human S-phase checkpoint. Genes Dev. 16:571–582. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Falck J, Petrini JH, Williams BR, Lukas J
and Bartek J: The DNA damage-dependent intra-S phase checkpoint is
regulated by parallel pathways. Nat Genet. 30:290–294. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kaufmann WK: The human intra-S checkpoint
response to UVC-induced DNA damage. Carcinogenesis. 31:751–765.
2010. View Article : Google Scholar :
|
|
36
|
Kim ST, Xu B and Kastan MB: Involvement of
the cohesin protein, Smc1, in Atm-dependent and independent
responses to DNA damage. Genes Dev. 16:560–570. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Luo H, Li Y, Mu JJ, Zhang J, Tonaka T,
Hamamori Y, Jung SY, Wang Y and Qin J: Regulation of intra-S phase
checkpoint by ionizing radiation (IR)-dependent and IR-independent
phosphorylation of SMC3. J Biol Chem. 283:19176–19183. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sørensen CS, Syljuåsen RG, Falck J,
Schroeder T, Rönnstrand L, Khanna KK, Zhou BB, Bartek J and Lukas
J: Chk1 regulates the S phase checkpoint by coupling the
physiological turnover and ionizing radiation-induced accelerated
proteolysis of Cdc25A. Cancer Cell. 3:247–258. 2003. View Article : Google Scholar : PubMed/NCBI
|