Introduction
Esophageal squamous cell carcinoma (ESCC), one of
the most prevalent and lethal cancers worldwide, results from the
uncontrolled proliferation of cells. Due to its poor prognosis, the
5-year survival rate is <15% (1–3).
Esophageal carcinogenesis is a multistep process that occurs due to
genetic alterations and disturbances of gene expression. In
addition, numerous genes have been identified to be essential
during carcinogenesis (4).
Moreover, in recent years, accumulating evidence has demonstrated
that small non-coding RNA microRNAs (miRNAs) and long non-coding
RNAs (lncRNAs), as transcript classes in the human genome, may play
an important role in tumorigenesis and tumor progression (5–7).
However, the underlying mechanisms of ESCC remain poorly
understood. Therefore, a better understanding of the detailed
mechanisms may be useful in identifying new therapeutic targets and
strategies for the treatment of ESCC.
miRNAs, a class of short, single-stranded,
non-coding RNA, bind to the complimentary recognition sequences in
the 3′-untranslated region (3′UTR) of target mRNA, and are
recognized as important post-transcriptional regulators of gene
expression (8). miRNA dysregulation
can therefore contribute to cell biological processes including
migration, invasion, apoptosis and metastasis by affecting relevant
transcripts (9–11). It has been reported that miR-34a
expression was significantly reduced in non-small cell lung cancer
compared with normal tissues (12).
miR-34a induces G1 arrest, apoptosis and senescence by repressing
the expression of Notch1 in RCC cell lines, and inhibit gastric
cancer tumorigenesis by targeting PDGFR and MET through the
PI3K/Akt pathway (13,14). miR-34a, which is located at
chromosome 1p36, is a well-known tumor-suppressor miRNA present in
various types of human cancer (15,16).
However, the detailed role of miR-34a in ESCC remains poorly
understood. In the present study, we found that upregulated miR-34a
increased apoptosis and decreased clonogenic formation, migration
and invasion by directly downregulating Yin Yang-1 (YY1) in TE-1
cells.
Materials and methods
Tissue samples
For real-time PCR analysis, 20 normal esophageal and
20 human ESCC tissues were used, as previously reported (17). These tissues were obtained
postoperatively between 2010 and 2012 from the Gastrointestinal
Center, Jiangyin People’s Hospital, Medical School of University of
Southeast of China (Jiangyin, China). All the patients provided
signed, informed consent for their tissues to be used for
scientific research. Ethics approval for the study was obtained
from the Jiangyin People’s Hospital.
RNA isolation and RT-PCR
Total miRNA was extracted from cultured cells and
esophageal tumor and normal esophageal tissues using TRIzol
(Invitrogen, Carlsbad, CA, USA). cDNAs were reverse transcribed to
cDNA using the RT-PCR kit (GenePharma, Shanghai, China).
Quantitative PCR was performed on a CFX96™ Real-Time PCR Detection
system (ABI 7500; Applied Biosystems, Foster City, CA, USA) using
SYBR® Premix Ex Taq™ II (GenePharma). The following PCR
conditions were used: denaturation at 95°C for 30 sec, followed by
40 cycles of annealing at 95°C for 10 sec and extension at 62°C for
30 sec. The data were normalized against the expression of the U6
snRNA. After amplification, melting curve analysis was performed to
ensure the specificity of the products.
Cell culture and transfection
The human ESCC TE-1 cell line was purchased from the
Type Culture Collection of the Chinese Academy of Sciences
(Shanghai, China). The cell lines were cultured in Dulbecco’s
modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine
serum (FBS) (both from Gibco, Grand Island, NY, USA) and incubated
at 37°C with 5% CO2. Cell transfections were performed
using Lipofectamine 2000 (Invitrogen-Life Technologies, Carlsbad,
CA, USA). Transfected miRNA mimics or inhibitors were purchased
from GenePharma. The miRNAs mimic or inhibitor sequences used were
: miR-34a mimic, sense, 5′-UGGCAGUGUCUUAGCUGGUUGU-3′ and antisense,
5′-AACCAGCUAAGACACUGCAAUU-3′; miR-34a NC, sense,
5′-UUCUCCGAACGUGUCAGGUTT-3′ and antisense,
5′-ACGUGACACGUUCGGAGAATT-3′; miR-34a inhibitor,
5′-ACAACCAGCUAAGACACUGCCA-3′; and miR-34a inhibitor NC,
5′-CAGUACUUUUGUGUAGUACAA-3′.
Measurement of apoptosis
Apoptosis was measured using propidium iodide
(PI)/Annexin V double-staining following the manufacturer’s
instructions (Keygen Biotech, Nanjing, China). Cells were harvested
24 h after transfection and apoptotic fractions were measured using
flow cytometry (Beckman Coulter, Brea, CA, USA). The Annexin
V+/PI− cells indicated early apoptotis and
Annexin V+/PI+ cells late apoptosis. The
percentage of the two types of cells was calculated.
Clonogenic formation assay
For the clonogenic formation assay, 1×103
cells were seeded in 6-well plates separately. After transfection,
the cells were incubated for ~10 days. The cells were then washed
with PBS, fixed and stained with Giemsa. The clone number (cells
population >50) was counted using a microscope.
Western blot analysis
Total protein was extracted from the cells using
whole cell lysates (Beyotime, Nantong, China). The protein
concentrations of individual samples were assessed using a standard
bicinchoninic acid assay (Beyotime). For each sample, 30 μg
of protein was loaded on a 10% SDS-PAGE gel (Bio-Rad, Hercules, CA,
USA), transferred onto a polyvinylidene difluoride membrane and
blocked with 5% skimmed milk and 0.1% Tris-buffered saline Tween-20
(TBST) at room temperature for 1.5 h. The membranes were washed in
TBST three times and incubated overnight at 4°C with rabbit
anti-YY1, anti-MMP-2 and anti-MMP-9 antibody (1:1,000 dilution)
(all from Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) and
mouse anti-GAPDH and mouse anti-β-actin antibody (1:8,000 dilution)
(both from Beyotime). The membranes were then washed with TBST and
incubated with anti-rabbit IgG horseradish peroxidase-conjugated
secondary antibody (Cell Signaling Technology, Boston, MA, USA).
The protein expression was evaluated using chemiluminescence and
exposure to Kodak film (Kodak, Rochester, NY, USA).
YY1 overexpression plasmids and
shRNA
YY1 overexpression was obtained from OriGene
Technologies Inc. (Rockville, MD, USA). A shRNA negative vector and
two shRNA were constructed. Their targeting sequences are:
shRNA-NC, 5′-GTTCTCCGAACGTGTCACGT-3′; shRNA-1,
5′-GAACUCACCUCCUGAUUAU-3′; and shRNA-2,
5′-CAAAGAUGUUCAGGGAUAA-3′.
Luciferase reporter assay
The 3′UTR of YY1 was amplified using the primers:
forward, 5′-GCTCTAGAAAAGAAGAGA GAAGACCT-3′ and reverse,
5′-GCTCTAGACCATGTAACA GAAAGGGC-3′. The amplified fragmented was
inserted at the XbaI site of pGL3-promoter vector (Promega,
Madison, WI, USA). The construct was sequenced for confirmation.
For transfection, TE-1 cells were transfected by Lipofectamine 2000
(Invitrogen-Life Technologies). The luciferase reporter containing
a fragment of the YY1 3′UTR was co-transfected with pRL-TK
(Promega) and synthetic RNAs. Luciferase activity was measured
using the Dual-Luciferase Reporter Assay system (Promega). Promoter
activities were expressed as the ratio of firefly luciferase to
Renilla luciferase activity.
Cell viability assays
Cells were transfected with the indicated RNAs. The
transfected cells were seeded in 96-well plates at a density of
1×104 cells/well. MTT solution (20 μl of 5 mg/ml
MTT) was added to each well (for a total volume of 100 μl),
and the plates were incubated for 4 h at 37°C. Following removal of
the culture medium, the remaining crystals were dissolved in DMSO,
and the absorbance at 490 nm was measured.
Invasion assays and wound-healing
assay
For the invasion assays, 5×104 cells were
plated in the top chamber containing a Matrigel-coated membrane
(24-well insert, 8-mm pore size; BD Biosciences). Twenty-four
serum-starved cells were plated in serum-free medium. The medium
supplemented with 10% serum was used as a chemoattractant in the
lower chamber. The cells were incubated for 48 h at 37°C in a
tissue culture incubator with 5% CO2. The non-invading
cells were removed from the upper sides of the Transwell membrane
filter inserts using cotton-tipped swabs. The invaded cells on the
lower sides of the inserts were stained with Giemsa, and the cells
were counted. Sterile ruler and skin marker tip were used to draw a
straight width line in a cell-covered plate. The cells were washed
with PBS, and cultured with serum-free medium. After 48 h, the
width of the line was observed using a microscope. Four random
views were taken and quantified under a microscope.
Statistical analysis
The SPSS 17.0 software was used for the statistical
analysis. The data are presented as the means ± SEM. Group
comparisons were evaluated by one-way ANOVA to determine
statistical significance. Differences were considered significant
when P<0.05.
Results
miR-34a is downregulated in esophageal
normal tissues compared with tumor tissues
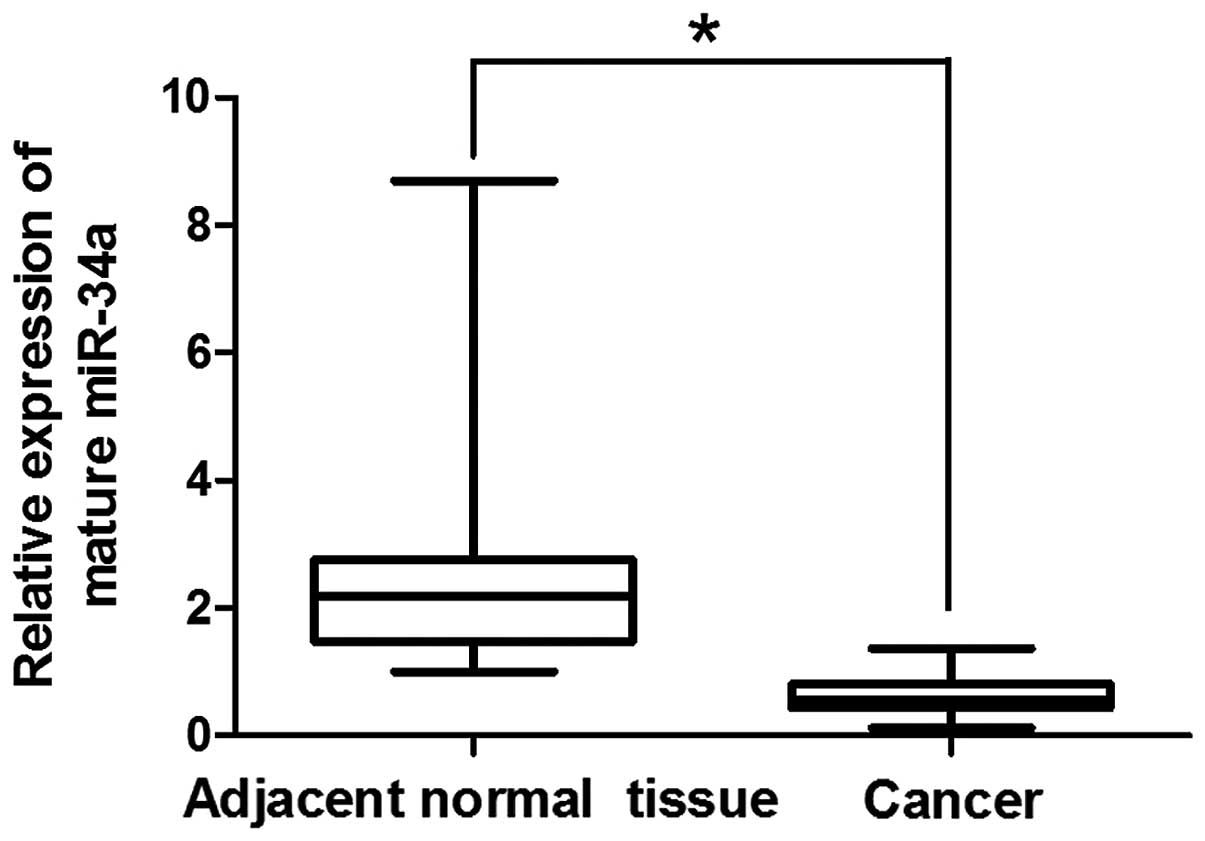
We first determined the expression of miR-34a in
esophageal cancer cells and matched adjacent normal esophageal
tissues. miR-34a expression in 20 human ESCC and 20 adjacent normal
esophageal tissues was determined by RT-qPCR analysis. Results
showed that the levels of miR-34a expression were significantly
decreased by 4.46-fold in the tumor tissues compared to the
adjacent normal mucosa tissues (Fig.
1). In these samples, miR-34a expression was decreased in 19
ESCC samples, suggesting that the down-regulation of miR-34a may be
a hallmark of ESCC.
miR-34a inhibits TE-1 cell proliferation,
colony formation and apoptosis, as well as migration and
invasion
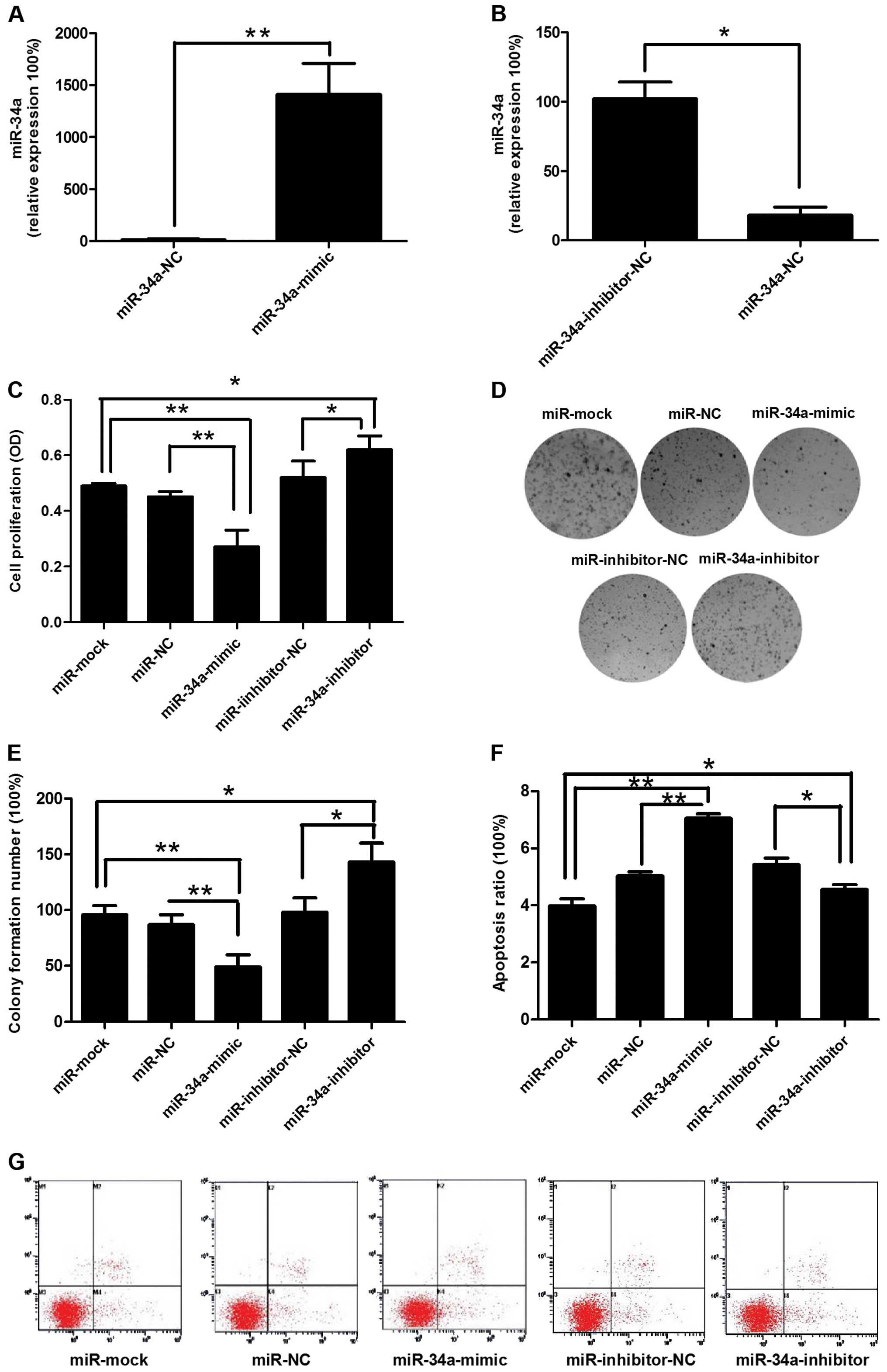
To investigate the effect of miR-34a on TE-1 cell
motilities, miRNA-NC, miR-34a mimics, miRNA inhibitor-NC, or
miR-34a inhibitor was transfected into TE-1 cells (Fig. 2A and B). miR-34a overexpression
reduced the proliferation rate and promoted apoptosis in cultured
TE-1 cells (Fig. 2C, G and F).
Colony formation of TE-1 cells was significantly decreased
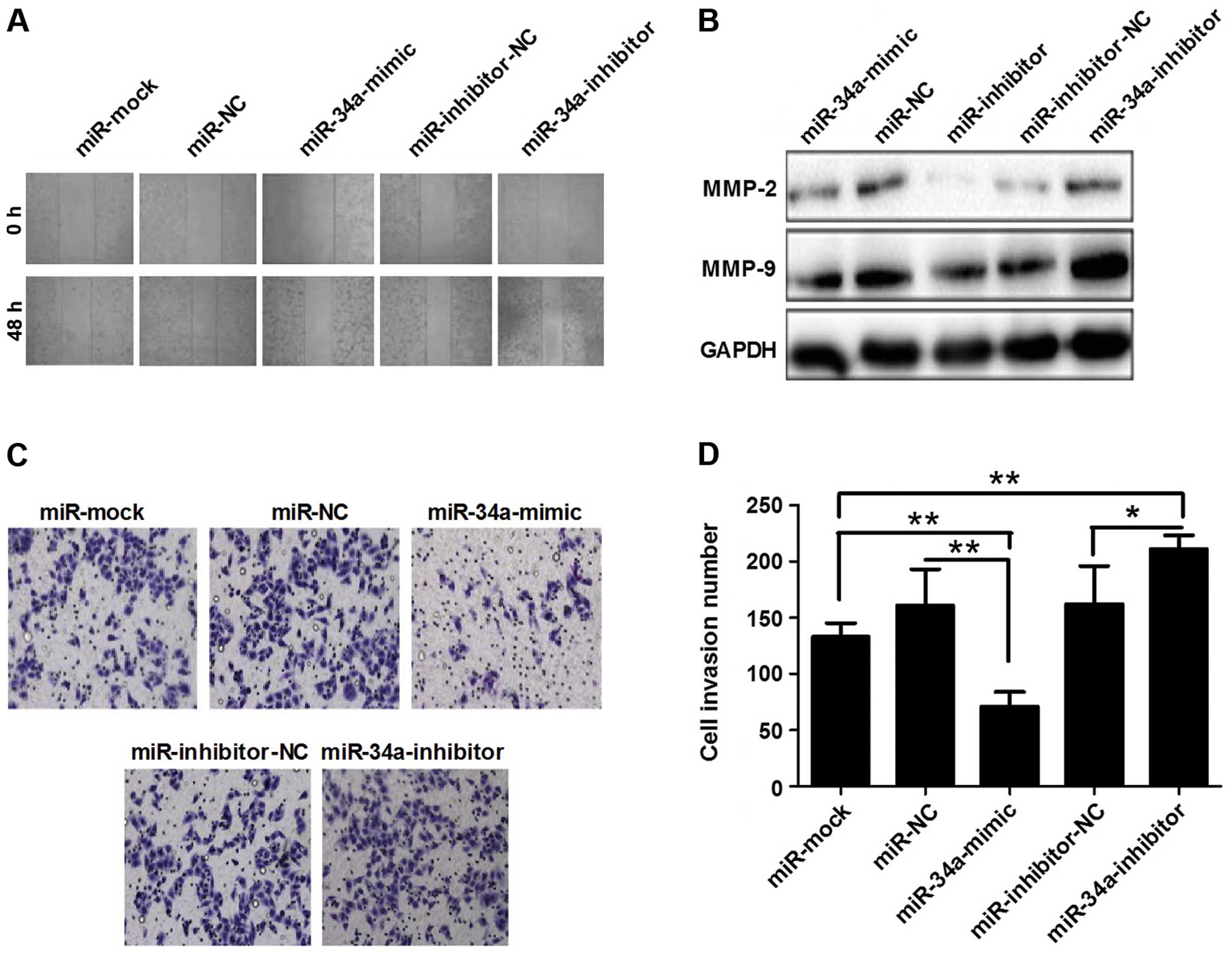
following the transfection of miR-34a mimics (Fig. 2D and E). Forty-eight hours after
transfection, the wound-healing was determined and the results
revealed that the width of line in the miR-34a mimics-transfected
group was wider than that of the miRNA-NC-transfected cells.
Conversely, the miR-34a inhibitor-transfected group showed the
smallest width (Fig. 3A). The
results of the invasion assay showed that the invasion cell number
in the mimics group had fewer cells than that of the inhibitor
group (Fig. 3C and D). Western
blotting also demonstrated that miR-34a downregulated MMP-2 and -9
expression (Fig. 3B). These results
indicated that miR-34a inhibited migration, invasion and
proliferation in ESCC cells.
miR-34a directly targets YY1
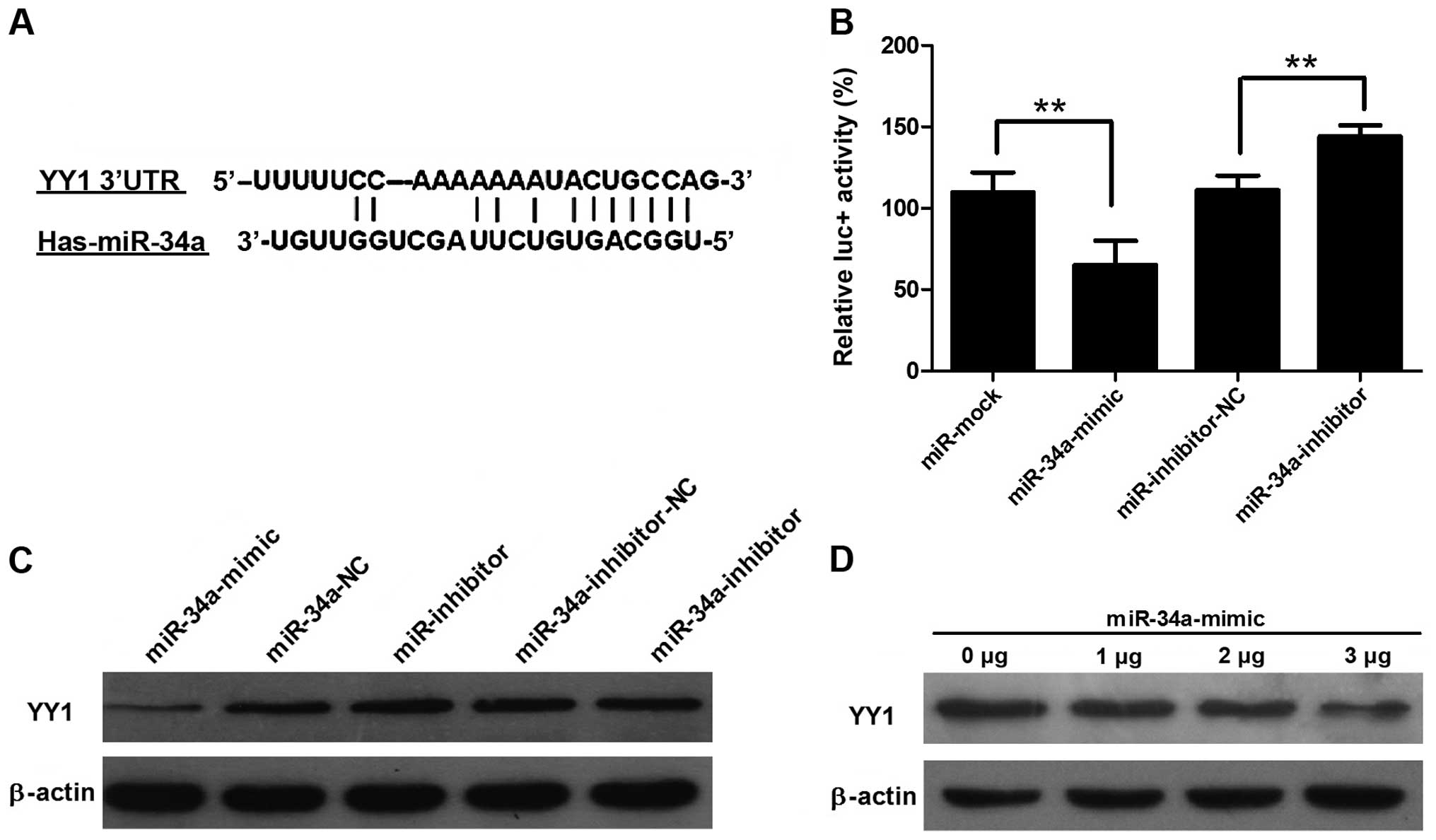
Bioinformatic tools predicted that human miR-34a may
target the 3′UTR of YY1 between position 710 and 726 bp (relative
to the YY1 stop codon, Fig. 4A). To
further confirm that miR-34a directly targets YY1, we performed
luciferase reporter assays to examine whether miR-34a regulates YY1
3′UTR. The 3′UTR of YY1 was cloned and inserted downstream of the
pGL3-promoter vector designated as pGL3-YY1 3′UTR. We found that
co-transfection of miR-34a with pGL3-YY1 3′UTR caused a significant
decrease in luciferase activity compared to the
miRNA-NC-co-transfected cells (Fig.
4B). Conversely, co-transfection of the miR-34a inhibitor
increased the luciferase activity, suggesting a suppressing role of
miR-34a. Western blotting demonstrated that YY1 expression was
significantly decreased in the miR-34a mimics-transfected group,
whereas miR-34a inhibitor transfection caused an increase in YY1
expression (Fig. 4C). The result
showed that YY1 expression was gradually reduced when the
concentration of transfected miR-34a was increased (Fig. 4D).
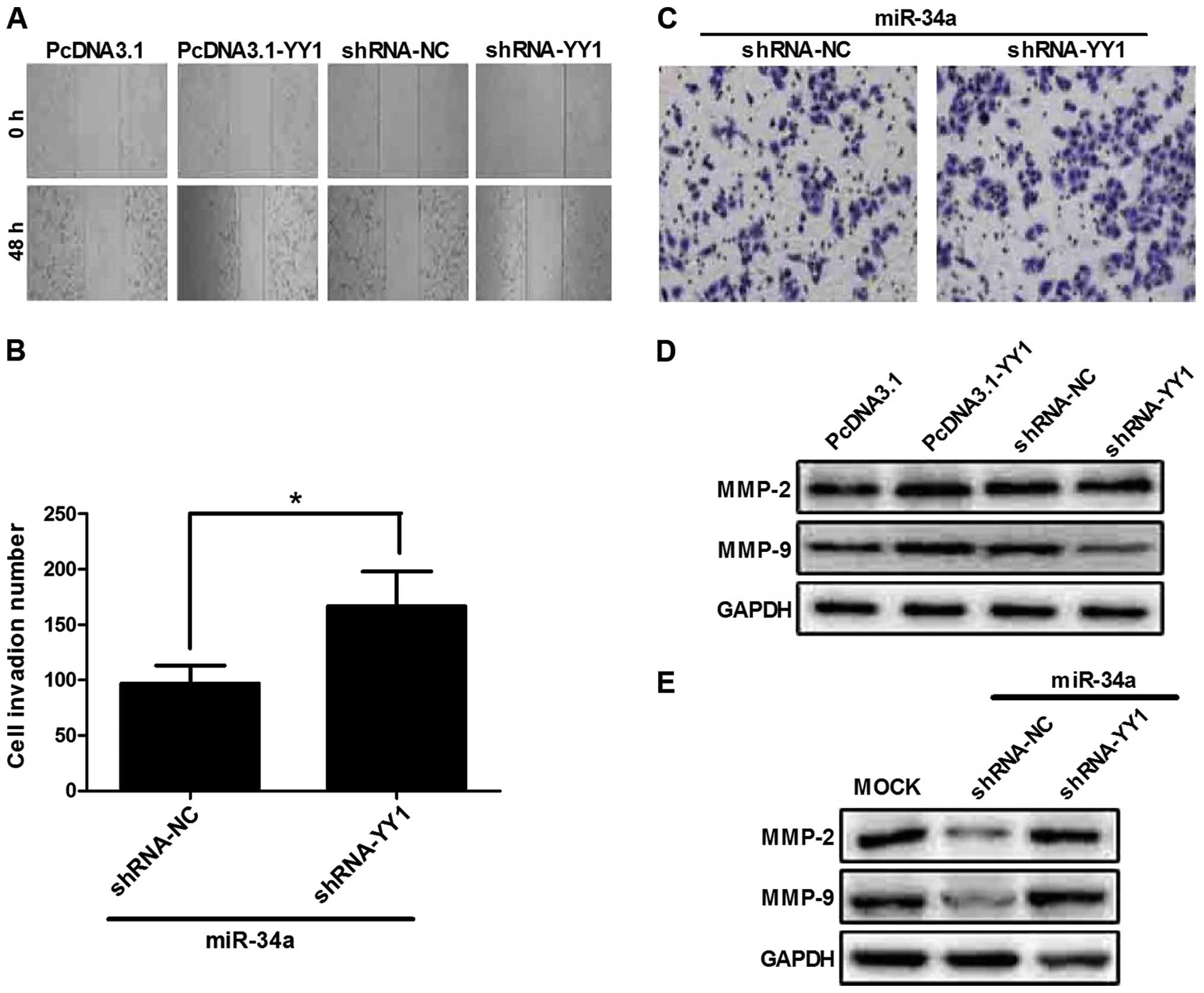
YY1 promotes TE-1 migation and
invasion
We investigated whether YY1 facilitates the
metastasis and invasion of ESCC cells. We previously reported that
YY1 promotes the invasion of TE-1 cells (18). To confirm this result, TE-1 cells
were transfected with pcDNA3.1-YY1 or pcDNA3.1 plus shRNA-NC or
shRNA-YY1. The wound-healing assay showed that YY1 overexpression
healed more rapidly than the remaining groups (Fig. 5A), which is consistent with the
results of our previous study.
The Transwell-based invasion assay was used to
identify the relationship between YY1 and miR-34a. The results
showed that co-transfected miR-34a and shRNA-YY1 significantly
increased the invasion ability of TE-1 cells (Fig. 5B and C). This result suggested that
shRNA targeting YY1 reversed the inhibitory effects of miR-34a on
the invasion ability of TE-1 cells. The results from western
blotting showed that MMP-2 and -9 expression was correlated with
the invasive status of cells. In the present study, MMP-9
expression in the shRNA-YY1-transfected group was significantly
decreased compared to that of pcDNA3.1-YY1. MMP-9 was markedly
increased in co-transfected miR-34a and shRNA-YY1, which is
consistent with the result of the invasion assay (Fig. 5D and E).
Discussion
miRNAs have demonstrated far-reaching effects on the
development of cellular biology and cancer (19,20).
miR-34a is reported to be significantly dysregulated in various
cancer types, such as colon, prostate and pancreatic cancer and
glioma (21–23). In the present study, the qPCR
examination revealed that miR-34a expression significantly
decreased in 13 of the 20 human esophageal tumor tissues compared
with the normal tissues, which suggested that dysregulation of
miR-34a may be involved in the development of human esophageal
cancer. Moreover, studies have reported that miR-34a can affect the
growth of proneural glioma and renal cell carcinoma cells (13,24).
In the present study, we found that TE-1 cells grew slowly
following transfection with miR-34a mimics. Metastasis is a major
cause of death in esophageal cancer patients (25) and the MMP family has been considered
to be involved in cancer invasion and metastasis, especially MMP-2
and -9. Due to their ability to degrade type IV collagen, MMP-2 and
-9 have been correlated with the invasive stage of carcinomas
(26). It is reported that MMP-2
and -9 were involved in breast cancer initiation and growth in the
early stage of tumor genesis (27).
In the present study, the cell invasion number and expression of
MMP-2 and -9 were significantly decreased when transfected with
miR-34a mimics. This result suggested that miR-34a mediated the
metastasis of ESCC through MMP-2 and -9.
Moreover, using a bioinformatics approach and a
literature review, we found that miR-34a directly regulates
numerous target genes. Among them, transcription factor YY1 is
likely to be an important target gene of miR-34a. YY1 plays
important roles in cell proliferation and differentiation (28). Increased YY1 expression was observed
in various types of cancer, such as breast, ovarian, colon, bone,
liver, lung, bladder, cervical, prostate and esophageal cancer
(29–35). However, the specific function in
different cancer types has not been elucidated. In the present
study, luciferase activity identified that miR-34a can directly
target YY1, and western blotting showed that the expression of YY1
was inhibited by miR-34a. In addition, in the wound-healing and
invasion assays, the results suggest that YY1 is an important
factor in promoting cell metastasis.
In conclusion, we have demonstrated that the
miR-34a/YY1 axis contributes to esophageal cancer progression by
increasing apoptosis and inhibiting clonogenic formation, cell
proliferation and invasion. miR-34a negatively regulated YY1
expression in TE-1 cells. These results provide a therapeutic
target for esophageal cancer.
Acknowledgments
This study was supported by the National Natural
Science Foundation of China (nos. 81302382, 81472917, 81372433 and
81402626), the Key Programs of Natural Science Foundation of
Jiangsu, Educational Committee (no. 11KJA310001), the Priority
Academic Program Development of Jiangsu Higher Education
Institutions (PAPD) and the Natural Science Fund for Colleges and
Universities in Jiangsu Province (no. 12KJB330005).
References
|
1
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar
|
|
2
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lv Y, Zhang J and Qiao L: Quality of life
in patients with esophageal cancer receiving definitive
chemoradiotherapy or esophagectomy. Mol Clin Oncol. 2:870–874.
2014.PubMed/NCBI
|
|
4
|
Li SQ, Li F, Xiao Y, Wang CM, Tuo L, Hu J,
Yang XB, Wang JS, Shi WH, Li X, et al: Comparison of long non
coding RNAs, microRNAs and messenger RNAs involved in initiation
and progression of esophageal squamous cell carcinoma. Mol Med Rep.
10:652–662. 2014.PubMed/NCBI
|
|
5
|
Lu J, Xue L, Jin M and Lyu N: Expression
profiling of metastasis-related microRNAs in early esophageal
squamous cell carcinoma. Zhonghua Bing Li Xue Za Zhi. 43:313–317.
2014.In Chinese. PubMed/NCBI
|
|
6
|
Pan F, Yao J, Chen Y, Zhou C, Geng P, Mao
H and Fang X: A novel long non-coding RNA FOXCUT and mRNA FOXC1
pair promote progression and predict poor prognosis in esophageal
squamous cell carcinoma. Int J Clin Exp Pathol. 7:2838–2849.
2014.PubMed/NCBI
|
|
7
|
Ge H, Lu Y, Chen Y, Zheng X, Wang W and Yu
J: ERCC1 expression and tumor regression predict survival in
esophageal squamous cell carcinoma patients receiving combined
trimo-dality therapy. Pathol Res Pract. 210:656–661. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Caporali A and Emanueli C: MicroRNA
regulation in angio-genesis. Vascul Pharmacol. 55:79–86. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Banaudha KK and Verma M: The role of
microRNAs in the management of liver cancer. Methods Mol Biol.
863:241–251. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tavazoie SF, Alarcón C, Oskarsson T, Padua
D, Wang Q, Bos PD, Gerald WL and Massagué J: Endogenous human
microRNAs that suppress breast cancer metastasis. Nature.
451:147–152. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Valastyan S, Reinhardt F, Benaich N,
Calogrias D, Szász AM, Wang ZC, Brock JE, Richardson AL and
Weinberg RA: A pleiotropically acting microRNA, miR-31, inhibits
breast cancer metastasis. Cell. 137:1032–1046. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gallardo E, Navarro A, Viñolas N, Marrades
RM, Diaz T, Gel B, Quera A, Bandres E, Garcia-Foncillas J, Ramirez
J, et al: miR-34a as a prognostic marker of relapse in surgically
resected non-small-cell lung cancer. Carcinogenesis. 30:1903–1909.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang C, Mo R, Yin B, Zhou L, Liu Y and
Fan J: Tumor suppressor microRNA-34a inhibits cell proliferation by
targeting Notch1 in renal cell carcinoma. Oncol Lett. 7:1689–1694.
2014.PubMed/NCBI
|
|
14
|
Peng Y, Guo JJ, Liu YM and Wu XL:
MicroRNA-34a inhibits the growth, invasion and metastasis of
gastric cancer by targeting PDGFR and MET expression. Biosci Rep.
34:e001122014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Corcoran C, Rani S and O’Driscoll L:
miR-34a is an intracellular and exosomal predictive biomarker for
response to docetaxel with clinical relevance to prostate cancer
progression. Prostate. 74:1320–1334. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Daige CL, Wiggins JF, Priddy L,
Nelligan-Davis T, Zhao J and Brown D: Systemic delivery of a miR34a
mimic as a potential therapeutic for liver cancer. Mol Cancer Ther.
13:2352–2360. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhou J, Zhang S, Xie L, Liu P, Xie F, Wu
J, Cao J and Ding WQ: Overexpression of DNA polymerase iota (Polι)
in esophageal squamous cell carcinoma. Cancer Sci. 103:1574–1579.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Luo J, Jiang X, Cao L, Dai K, Zhang S, Ge
X, Zhou X and Lu X: Expression of YY1 correlates with progression
and metastasis in esophageal squamous cell carcinomas. Onco Targets
Ther. 7:1753–1759. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jones KB, Salah Z, Del Mare S, Galasso M,
Gaudio E, Nuovo GJ, Lovat F, LeBlanc K, Palatini J, Randall RL, et
al: miRNA signatures associate with pathogenesis and progression of
osteosarcoma. Cancer Res. 72:1865–1877. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Maire G, Martin JW, Yoshimoto M,
Chilton-MacNeill S, Zielenska M and Squire JA: Analysis of
miRNA-gene expression-genomic profiles reveals complex mechanisms
of microRNA deregulation in osteosarcoma. Cancer Genet.
204:138–146. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Siemens H, Neumann J, Jackstadt R,
Mansmann U, Horst D, Kirchner T and Hermeking H: Detection of
miR-34a promoter methylation in combination with elevated
expression of c-Met and β-catenin predicts distant metastasis of
colon cancer. Clin Cancer Res. 19:710–720. 2013. View Article : Google Scholar
|
|
22
|
Liu C, Kelnar K, Liu B, Chen X,
Calhoun-Davis T, Li H, Patrawala L, Yan H, Jeter C, Honorio S, et
al: The microRNA miR-34a inhibits prostate cancer stem cells and
metastasis by directly repressing CD44. Nat Med. 17:211–215. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guessous F, Zhang Y, Kofman A, Catania A,
Li Y, Schiff D, Purow B and Abounader R: microRNA-34a is tumor
suppressive in brain tumors and glioma stem cells. Cell Cycle.
9:1031–1036. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Silber J, Jacobsen A, Ozawa T, Harinath G,
Pedraza A, Sander C, Holland EC and Huse JT: miR-34a repression in
proneural malignant gliomas upregulates expression of its target
PDGFRA and promotes tumorigenesis. PLoS One. 7:e338442012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J and
Thun MJ: Cancer statistics, 2009. CA Cancer J Clin. 59:225–249.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Stetler-Stevenson WG: Type IV collagenases
in tumor invasion and metastasis. Cancer Metastasis Rev. 9:289–303.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Duffy MJ, Maguire TM, Hill A, McDermott E
and O’Higgins N: Metalloproteinases: role in breast carcinogenesis,
invasion and metastasis. Breast Cancer Res. 2:252–257. 2000.
View Article : Google Scholar
|
|
28
|
Leeman MF, Curran S and Murray GI: New
insights into the roles of matrix metalloproteinases in colorectal
cancer development and progression. J Pathol. 201:528–534. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jeong HM, Lee SH, Yum J, Yeo CY and Lee
KY: Smurf2 regulates the degradation of YY1. Biochim Biophys Acta.
1843:2005–2011. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Allouche A, Nolens G, Tancredi A,
Delacroix L, Mardaga J, Fridman V, Winkler R, Boniver J, Delvenne P
and Begon DY: The combined immunodetection of AP-2alpha and YY1
transcription factors is associated with ERBB2 gene overexpression
in primary breast tumors. Breast Cancer Res. 10:R92008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zaravinos A and Spandidos DA: Yin yang 1
expression in human tumors. Cell Cycle. 9:512–522. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kashyap V and Bonavida B: Role of YY1 in
the pathogenesis of prostate cancer and correlation with
bioinformatic data sets of gene expression. Genes Cancer. 5:71–83.
2014.PubMed/NCBI
|
|
33
|
Huerta-Yepez S, Liu H, Baritaki S, Del
Lourdes Cebrera-Muñoz M, Rivera-Pazos C, Maldonado-Valenzuela A,
Valencia-Hipolito A, Vega MI, Chen H, Berenson JR, et al:
Overexpression of Yin Yang 1 in bone marrow-derived human multiple
myeloma and its clinical significance. Int J Oncol. 45:1184–1192.
2014.PubMed/NCBI
|
|
34
|
Notarbartolo M, Giannitrapani L, Vivona N,
Poma P, Labbozzetta M, Florena AM, Porcasi R, Muggeo VM, Sandonato
L, Cervello M, et al: Frequent alteration of the Yin Yang 1/Raf-1
kinase inhibitory protein ratio in hepatocellular carcinoma. OMICS.
15:267–272. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Luo J, Zhou X, Ge X, Liu P, Cao J, Lu X,
Ling Y and Zhang S: Upregulation of Ying Yang 1 (YY1) suppresses
esophageal squamous cell carcinoma development through heme
oxygenase-1. Cancer Sci. 104:1544–1551. 2013. View Article : Google Scholar : PubMed/NCBI
|