Introduction
MicroRNAs are endogenous non-coding RNAs that can
regulate gene expression through translational repression and mRNA
degradation (1). Accumulating
evidence has shown their participation in the regulation of a wide
range of biological processes, including tumor formation (2), since almost 50% of identified human
miRNAs are found located at fragile sites on chromosomes that are
known to be associated with cancer (3).
Human prostate cancer (PCa) is one of the most
frequently diagnosed tumors, and the second significant cancer
killer in males in America (4).
Numerous studies have shown the involvement of miRNAs in prostate
carcinogenesis in recent years (5,6). There
are more than 50 miRNAs found deregulated in PCa (7,8),
including miR-34a, one of the tumor-suppressor miRNAs (9). As a miRNA that induces cell cycle
arrest and apoptosis (10), miR-34a
displayed CpG promoter methylation in ~80% of primary prostate
carcinomas (11) and remarkable
decrease in PCa cell lines (12).
Additionally, miR-34 family is involved in drug resistance in PCa
(12). However, the mechanism of
miR-34a in chemoresistance of PCa still needs further research.
Autophagy is a highly conserved, organized catabolic
process (13) and plays an
important role in cancer (14,15).
High-level in tumor cells following anticancer treatment,
therapeutic inhibitor targeting on the autophagic signaling pathway
represents a novel avenue to reduce the occurrence of
chemoresistance (16).
Autophagy-related gene 4B (ATG4B), a controller on autophagosome
maturation, is a particularly interesting target in this respect.
Recent studies have described that many microRNAs (miRNAs) are
involved in regulation of autophagic process by modulating
autophagy-related genes (ATGs). miR-101 and miR-376b negatively
regulates the expression of ATG4C and ATG4D (17,18).
miR-17 reduces ATG7 expression in glioblastoma cell lines (19). Notably, miR-34 represses autophagy
by reducing the expression of ATG9 in mammalian cells (20). However, the mechanism of miR-34a on
regulating autophagy and its role in drug resistance in PCa remains
largely unknown.
In the present study, we detected the methylation
condition of miR-34a in both PCa tissues and cell lines.
Additionally, the changes of apoptosis and drug sensitivity in
miR-34a methylated PCa cell lines were analyzed after transfection
of miR-34a or demethylation treatment using 5-azacytidine (5-Aza).
Furthermore, the target relation between miR-34a and ATG4B in PCa
was investigated. Finally, AMPK/mTOR pathway was examined for its
involvement in miR-34a-induced regulation process. Our research may
supply a novel choice for the future treatment on PCa
chemoresistance.
Materials and methods
Isolation of PCa and adjacent
tissues
Human PCa tissue and the corresponding adjacent
normal tissue were isolated during the operation, with the written
consent of the patients.
Cell culture
Human PCa cell lines, including PC-3, DU145 and
RWPE-1 were purchased from Auragene (Changsha, China). Cells were
cultured as monolayer in RPMI-1640 medium (HyClone, Logan, UT, USA)
supplemented with 10% fetal calf serum (Gibco, Gaithersburg, MD,
USA) and 2 mM glutamax.
For demethylation treatment, all PCa cells were
treated with 10 µmol/l 5-Azacytidine (Sigma-Aldrich, St.
Louis, MO, USA) for 48 h. Cells were then harvested for the
following investigations.
Transfection
PC-3 and Du145 cells were cultured as previously
described before transfection. Mimics NC (100 pmol) or pre-miR-34a
(Auragene) was diluted in 250 µl serum-free RPMI-1640
medium, which was then mixed with 10 µl of Lipofectamine
2000 (diluted in 250 µl serum-free medium). After incubation
at 37°C for 4 h, the medium was replaced by complete medium for
another 48 h culture. Cells were then collected for use.
MTT assay
For proliferation analysis of PCa cells, 5,000 cells
were plated in three 96-well plates. After every 24 h incubation at
37°C and 5% CO2, one plate was taken out and the cells
were treated with 20 µl MTT solution (5 mg/ml) for 4 h.
Subsequently, MTT solution was replaced by 150 µl of
dimethylsulfoxide (DMSO) to dissolve the tetrazolium crystals. The
OD values were read at a test wavelength of 570 nm with a
microplate reader within 10 min (Thermo Labsystems, Cheshire,
UK).
For the evaluation of the PCa cell sensitivity to
chemotherapy, after seeded on 96-well plates, the medium was
replaced by chemotherapeutic drug (Dox, 4.0 µmol/l; Topo, 5
mg/l). After 72 h, 20 µl MTT solution (5 mg/ml) was added to
each well. MTT solution was replaced by 150 µl of DMSO to
dissolve the tetrazolium crystals after 4 h incubation at room
temperature. The absorption was read at the wavelength of 570 nm
with a microplate reader within 10 min.
Apoptotic assay
Annexin V apoptosis detection kit (Life
Technologies, Grand Island, NY, USA) was used for apoptosis
detection. After cultured in 0.0625 µM Dox or 0.0195 mg/l
Topo for 48 h, cells were trypsinized, collected and washed twice
with cold phosphate-buffered saline (PBS). Binding buffer (500
µl) was used to resuspend cells. Annexin V-FITC (5
µl) and 5 µl propidium iodide were added to the
solution and mixed well. Following 15 min incubation at room
temperature in the dark, cells were analyzed using flow cytometric
analysis (BD Biosciences, San Jose, CA, USA).
Quantitative-polymerase chain reaction
(q-PCR) analysis of mRNA expression
Total RNA containing miRNA was extracted from PCa
and adjacent tissues, and PC-3 and DU145 cells with the use of
TRIzol reagent (Life Technologies, Shanghai, China) according to
the manufacturer's instructions, and cDNA was synthesized using
TaqMan MicroRNA Reverse Transcription kit (Applied Biosystems,
Foster City, CA, USA). q-PCR was performed with the use of
fluorescence quantitative PCR instrument (ABI 7500 thermocycler;
Life Technologies) and SYBR-Green Universal PCR Master Mix
(Bio-Rad, Hercules, CA, USA). The expression level of miR-34a was
normalized to RNU6B. Oligonucleotide sequences of the primer sets
used were as follows: miR-34a, HmiRQP0440; U6, HmiRQP9001; human
ATG4B (sense, 5′-ATGACTTCAATGATTGGTGCC-3′ and antisense,
5′-AGAAGAATCTGGACTTGGCAG-3′); human β-actin (sense,
5′-AGGGGCCGGACTCGTCATACT-3′ and antisense,
5′-GGCGGCACCACCATGTACCCT-3′). PCR was performed in a total volume
of 20 µl, which included 10 µl of 2X SYBR-Green qPCR
mix, 1 µl of each forward and reverse primer (10
µmol/l), 1 µl each cDNA sample and 7 µl
H2O. Amplifications were carried out in triplicate in
96-well microtiter plates. Thermal cycling conditions were as
follows: 95°C for 3 min, followed by 35 cycles of 95°C for 10 sec,
and 58°C for 30 sec, and finally by 95°C for 12 sec, 58°C for 50
sec.
Western blotting
Whole-cell lysates were harvested and washed with
PBS, and then lysed in a buffer containing 150 mM NaCl, 1 mM PMSF,
NaVO4, aprotinin and leupeptin as protease inhibitors,
in 50 mM Tris-HCl pH 8.0, 0.2% SDS, 1% NP-40. Protein/sample (30
µg) was resolved on a SDS-PAGE gel with subsequent transfer
blotting. Membranes were incubated at 4°C with primary antibody
overnight [m-TOR, YT2915; p-m-TOR, YP0176; ATG4B, YT0394; (all from
Immunoway) Beclin-1, 2026-1; Epitomics; LC3B, ab51520; Abcam; Bax,
YT0456; Bcl2, YT0469 (both from Immunoway), caspase 3, ab2302;
caspase 9, ab2324 (both from Abcam)]. After washing, membranes were
incubated with the corresponding secondary antibody preparation for
1 h at room temperature, followed by chemiluminescence for
visualization. For control group, the membrane was stripped and
reprobed using an actin antibody after the probing of each membrane
with the primary antibody.
Dual-luciferase reporter gene assay
For the luciferase reporter experiments, a
3′-untranslated region (3′-UTR) segment of ATG4B gene (accession
no. NM_013325.4) was amplified by PCR from human genomic DNA using
primers that included an XhoI and a NotI tail on the
5′ and 3′ strands, respectively. PCR products were recombined with
psi-CHECK2. The PC-3 and DU145 cells were then transfected with the
firefly luciferase ATG4B-3′UTR-psi-CHECK2, combined with miR-34a
mimics. Twenty-four hours after transfection, cells were lysed with
a 1X passive lysis buffer and the activity of both Renilla
and firefly luciferase was assayed using the Dual-Luciferase
Reporter Assay system (Promega) according to the manufacturer's
instructions.
DNA methylation analysis using MSP
Genomic DNA from PCa tissues and cell lines was
isolated with Auragene Genomic DNA kit (Auragene Biotech, Changsha,
China). MSP was performed in a total volume of 20 µl using
1.5 units Platinum Taq Polymerase (Invitrogen) per reaction.
Oligonucleotide sequences used for the MSP were: miR-34a-MSP-F,
TGTTAGTTTTTTCGGGGAGTTTTCGG and miR-34a-MSP-R,
ACGCCAACTCCTCCCCCGTCCCGAAC; miR-34a-UMSP-F,
TAGTTTTTTTGGGGAGTTTTTGGTTT and miR-34a-UMSP-R,
TAACACCAACTCCTCCCCCATCCCAA. PCR was performed in a total volume of
15 µl, which included 1.5 µl of 10X LAmp buffer, 1.2
µl of dNTP mix, 0.5 µl of forward and reverse primer
(10 µM), 0.3 µl of Lamp Taq, 3 µl of 5X
C Solution I, 1 µl of template and 7 µl of
ddH2O. Amplifications were carried out in 96-well
microtiter plates. Thermal cycling conditions were: 94°C for 4 min,
followed by 35 cycles of 94°C for 30 sec, 58°C for 30 sec and 72°C
for 30 sec, and finally followed by 72°C for 5 min. Amplified
fragments were separated by electrophoresis on 8% polyacrylamide
gels and visualized by staining with ethidium bromide.
Statistical analysis
Data are expressed as mean ± standard deviation
(SD). Comparisons between groups were performed by one-way analysis
of variance (ANOVA) using SPSS 17.0 (SPSS, Inc., USA) and Prism 5.0
(GraphPad Software, USA). P<0.05 was considered to indicate a
statistically significant result.
Results
miR-34a is epigenetically downregulated
by DNA methylation in PCa tissue
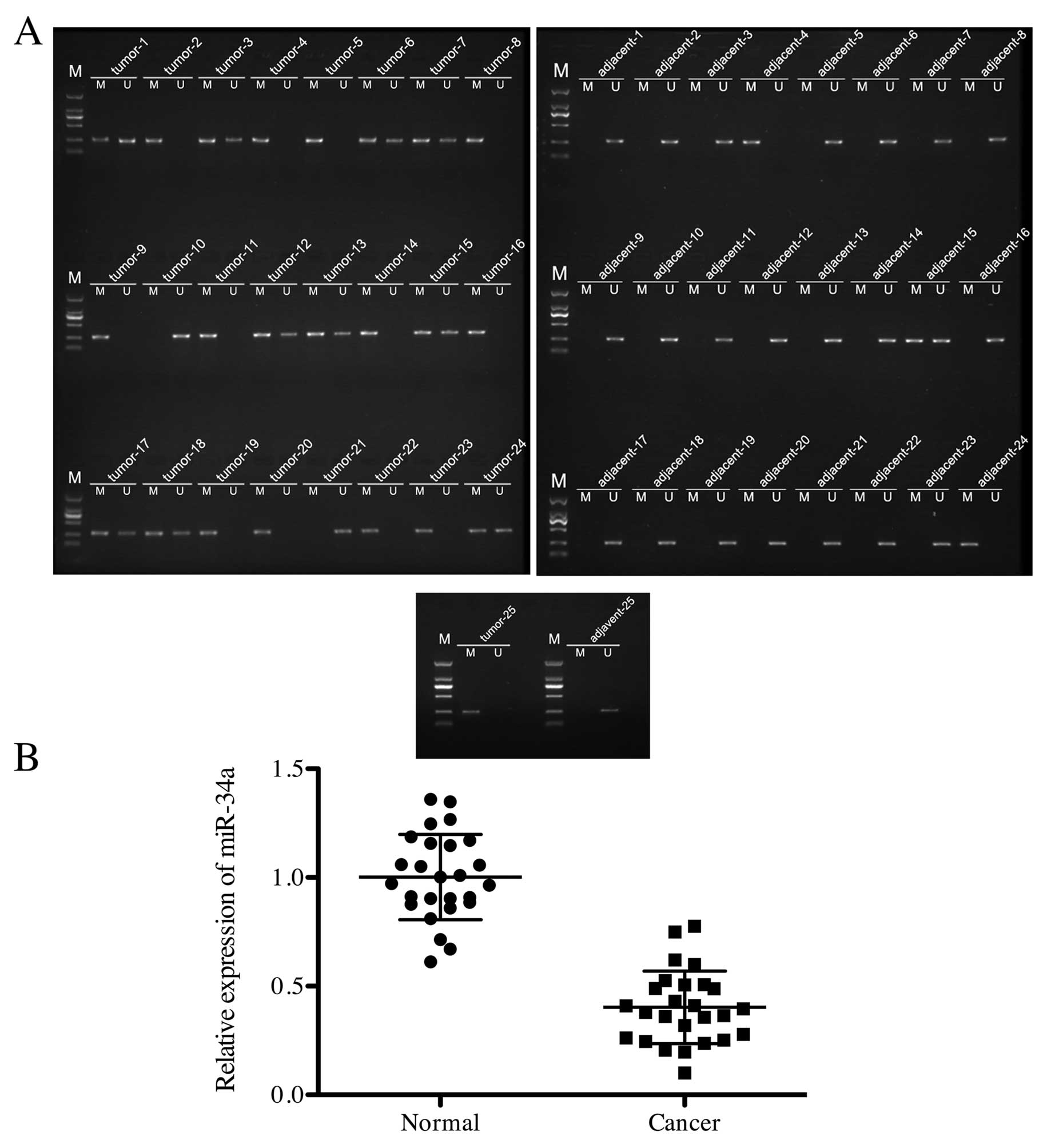
To determine the methylation condition of miR-34a in
PCa cells, 25 samples of PCa and adjacent tissues were collected
and measured in MSP, respectively. As shown in Fig. 1A, 23 in 25 (92%) PCa tissue samples
showed methylated miR-34a, while only 3 in 25 (12%) adjacent tissue
samples displayed positive methylation on miR-34a. Furthermore,
expression of miR-34a in PCa and cancer adjacent tissues was
quantified by q-PCR. The relative expression of miR-34a in adjacent
tissue was significantly higher than that in PCa tissue (Fig. 1B). This indicated that miR-34a was
hypermethylated in PCa tissue.
Methylation of miR-34a and expression of
ATG4B in PCa cell lines
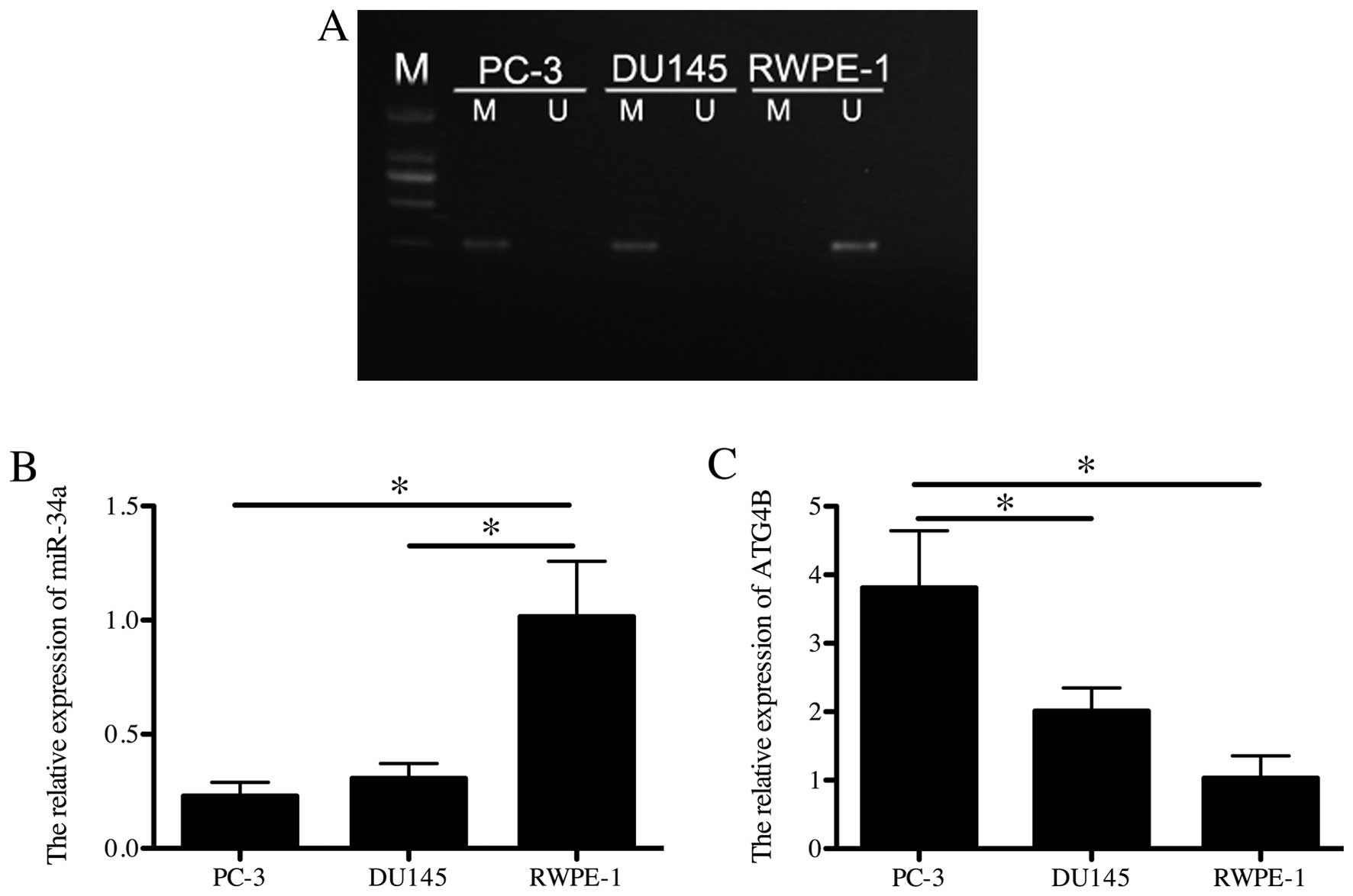
To analyze the methylation condition of miR-34a in
PCa cell lines in vitro, PC-3, DU145 and RWPE-1 were
detected in MSP. miR-34a showed methylated in PC-3 and DU145 while
RWPE-1 was unmethylated (Fig. 2A).
Additionally, the relative expression of miR-34a and mRNA of ATG4B
was respectively detected in all cell lines in q-PCR. Consistently,
PC-3 and DU145 showed significantly lower expression of miR-34a
than RWPE-1 (p<0.05; Fig. 2B),
while the mRNA level of ATG4B in RWPE-1 was lower than the other
two cell lines (p<0.05; Fig.
2C). The above demonstrated that PC-3 and DU145 showed miR-34a
methylation and kept low expression of miR-34a, while ATG4B was
highly expressed.
Upregulation of miR-34a promotes cell
apoptosis in PC-3 and DU145 cells
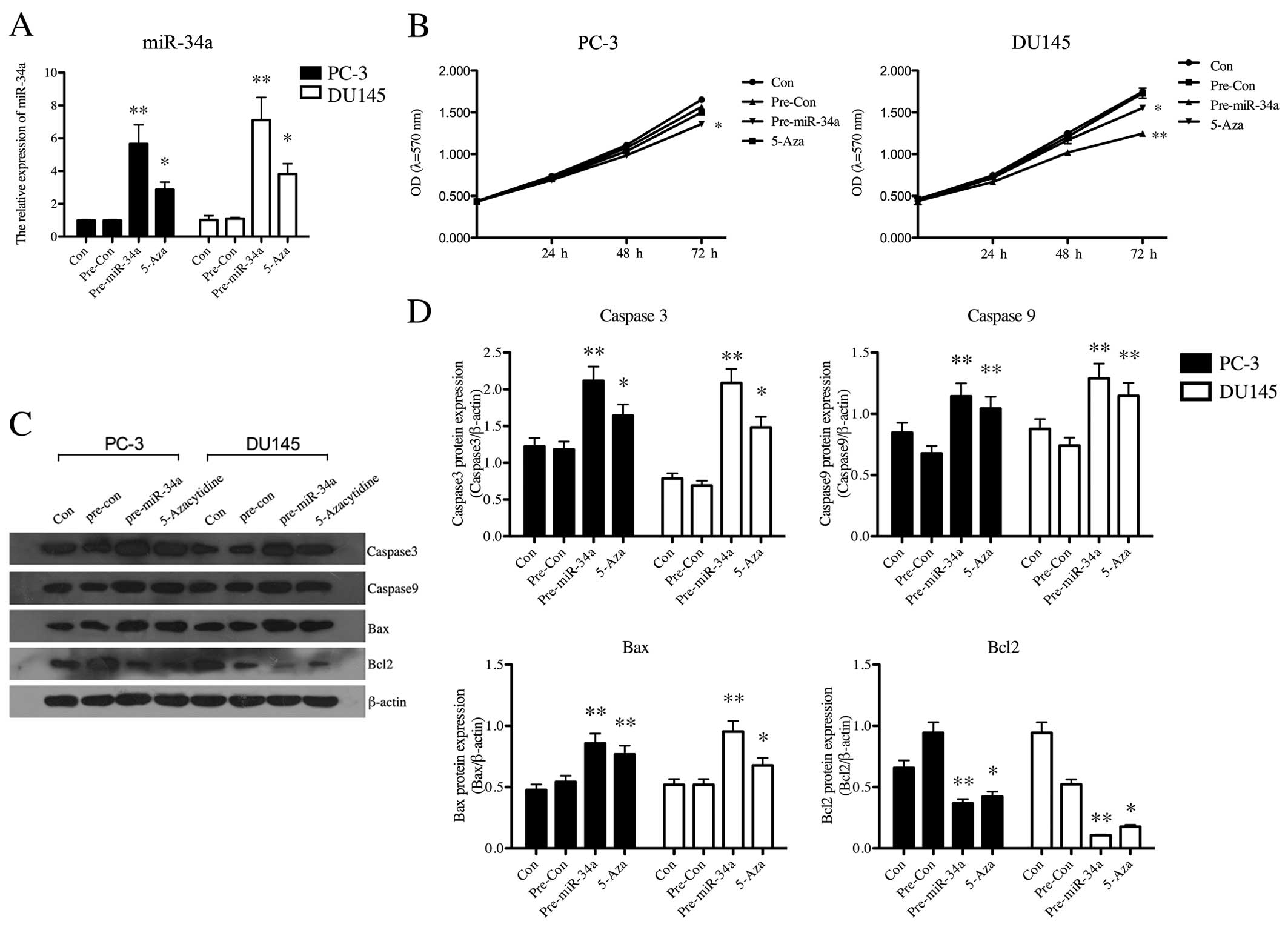
We performed gain-of-function studies using miR-34a
mimics, and miRNA transfection efficiency was determined by q-PCR.
After PC-3 and DU145 cells were transfected with miR-34a mimics,
the expression of miR-34a was significantly upregulated (p<0.01,
Fig. 3A). 5-Aza treatment also
increased the level of miR-34a (Fig.
3A). This indicated that the original low level of miR-34a in
PCa cells was related to methylation. Additionally, the effects of
upregulation of miR-34a either by transfection or by demethylation
treatment on cell proliferation in PC-3 and DU145 cells were
examined by MTT assay. Transfection of miR-34a mimics resulted in a
significant decrease in cell growth of PCa cell lines (PC-3,
p<0.05; DU145, p<0.01), and demethylation treatment also led
to a statistical decrease (p<0.05) in proliferation of DU145
cells (Fig. 3B). We further
investigated the effect of miR-34a on cell apoptosis using western
blotting (Fig. 3C). Apoptosis
related proteins, caspase 3 (p<0.01), caspase 9 (p<0.01) and
Bax (p<0.01), were all significantly upregulated after
transfection in both PC-3 and DU145 cells, and anti-apoptosis
protein Bcl-2 was statistically downregulated (p<0.01) (Fig. 3D). These data indicated that miR-34a
has a vital role in reducing the growth of PCa cells.
Upregulation of miR-34a enhances the
sensitivity of PC-3 and DU145 cells to Dox and Topo
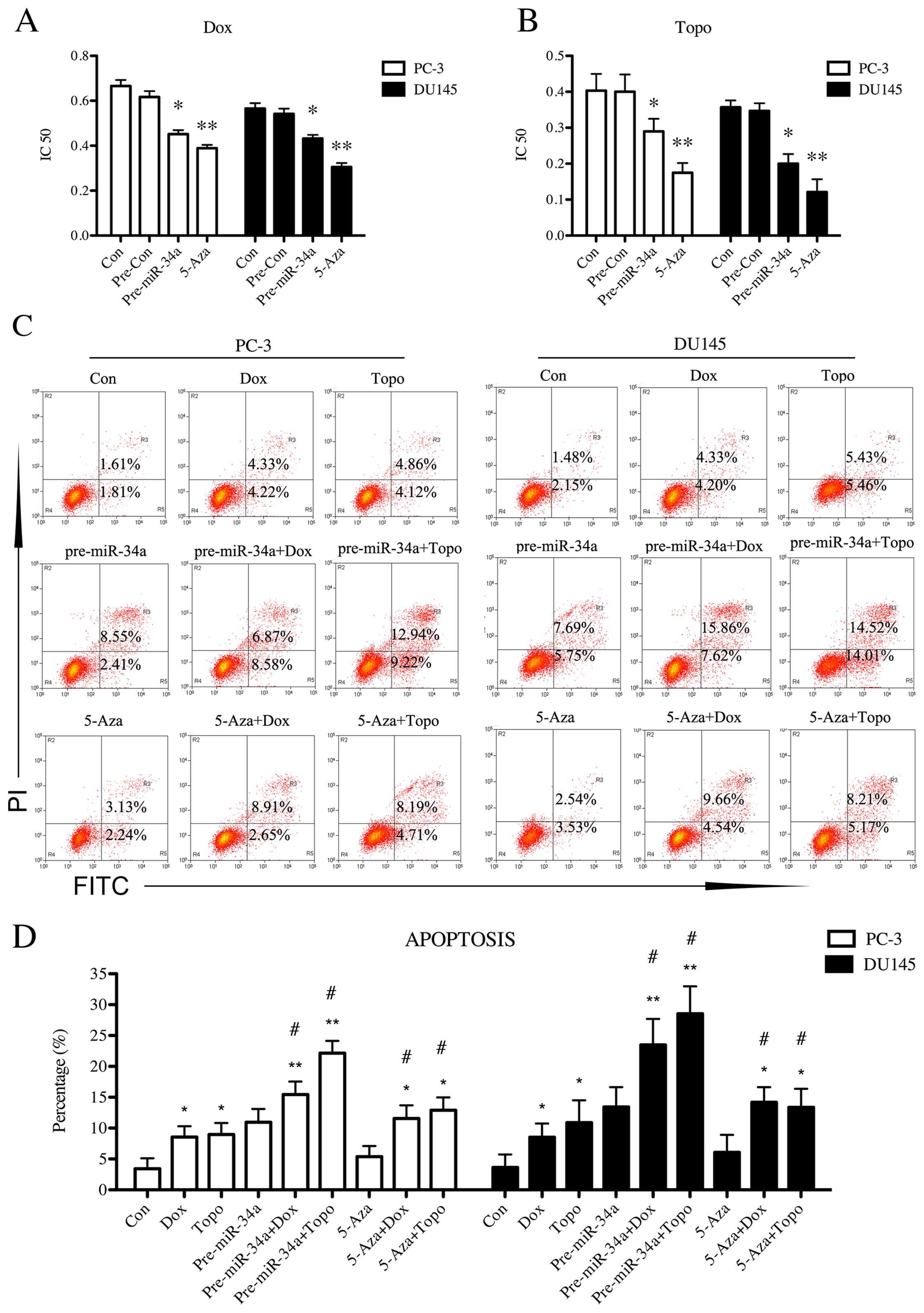
The effects of upregulation of miR-34a on cell
chemosensitivity in PC-3 and DU145 cells were examined by MTT
assay. Transfection with miR-34a mimics resulted in a significant
decrease on IC50 value when PCa cell lines were treated
with Dox (p<0.05, Fig. 4A) and
Topo (p<0.05, Fig. 4B),
respectively. 5-Aza treatment led to a statistical decrease
(p<0.01) on IC50 value of both cell lines when
treated with Dox and Topo (Fig. 4A and
B). This indicated that upregulation of miR-34a induced by
overexpression or demethylation enhanced the drug sensitivity in
PCa cells. We further investigated the effect of miR-34a
upregulation on cell apoptosis in Dox and Topo using flow cytometry
(Fig. 4C). Based on the data shown
in Fig. 4D, Dox (p<0.05) and
Topo (p<0.05) treatment led to an increased apoptosis percentage
in PCa cells compared with control. Upregulation of miR-34a in both
cell lines induced by mimics transfection (p<0.05) or 5-Aza
treatment (p<0.05) significantly increased the apoptosis
percentage of cells that were cultured in Dox and Topo, compared
with non-treated cells in Dox or Topo, respectively.
miR-34a-upregulated PC-3 and DU145 cells showed higher apoptosis
percentage in both Dox (p<0.01) and Topo (p<0.01) than the
control group. Collectively, these data demonstrated that
overexpression of miR-34a or demethylation on miR-34a in PC-3 and
DU145 cells improved cell chemosensitivity to Dox and Topo.
Upregulation of miR-34a impairs autophagy
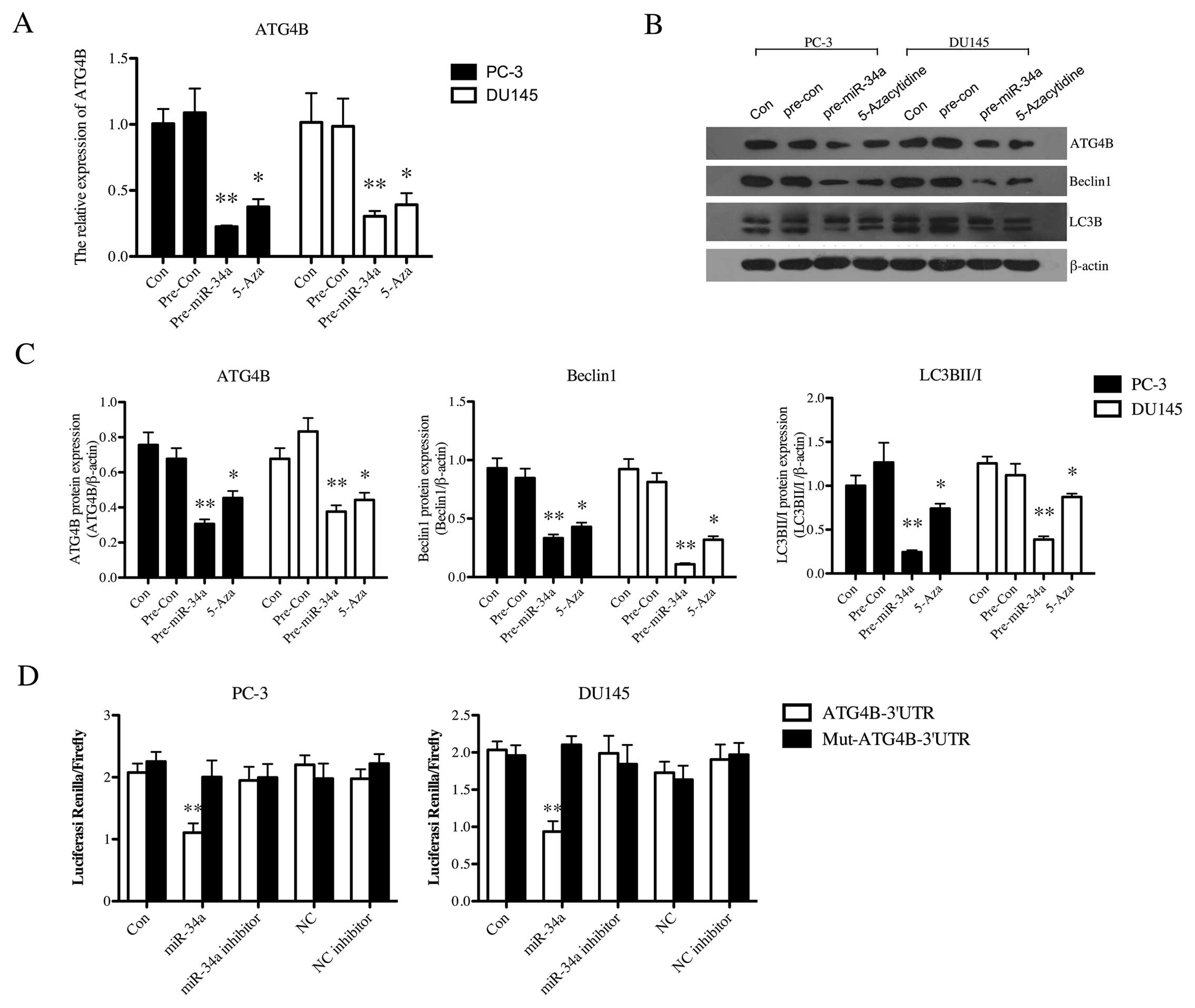
by directly down-regulating ATG4B in PC-3 and DU145 cells
For the analysis of the effects of miR-34a on
regulating autophagy-related proteins, the expression of AGT4B was
first examined in PCa cells. mRNA expression of ATG4B in PC-3
(p<0.01) and DU145 (p<0.01) was statistically reduced after
transfection of miR-34a mimics, which was similar to the effect of
5-Aza on PCa cells (PC-3, p<0.05; DU145, p<0.05) (Fig. 5A). The protein expression of ATG4B
and autophagy-related proteins, Beclin-1 and LC3B II/I, were then
detected in both cell lines (Fig.
5B). Both the PC-3 and DU145 cells after transfection displayed
significantly downregulated level of ATG4B protein (p<0.01),
Beclin-1 (p<0.01) and LC3B II/I (p<0.01) (Fig. 5C). 5-Aza treatment similarly led to
the decrease of ATG4B (p<0.05), Beclin-1 (p<0.05) and LC3B
II/I (p<0.05) (Fig. 5C).
Furthermore, dual-luciferase reporter gene assay was operated to
examine whether ATG4B is indeed functionally targeted by miR-34a.
Fig. 5D shows that miR-34a
inhibited the luciferase activity from the construct with the
ATG4B-3′-UTR segment both in PC-3 (p<0.01) and DU145 cells
(p<0.01). There was no change in the luciferase reporter
activity when the cells were cotransfected with the
miR-34a-inhibitor or negative controls. This demonstrated that
ATG4B was a direct target of miR-34a in PC-3 and DU145 cells.
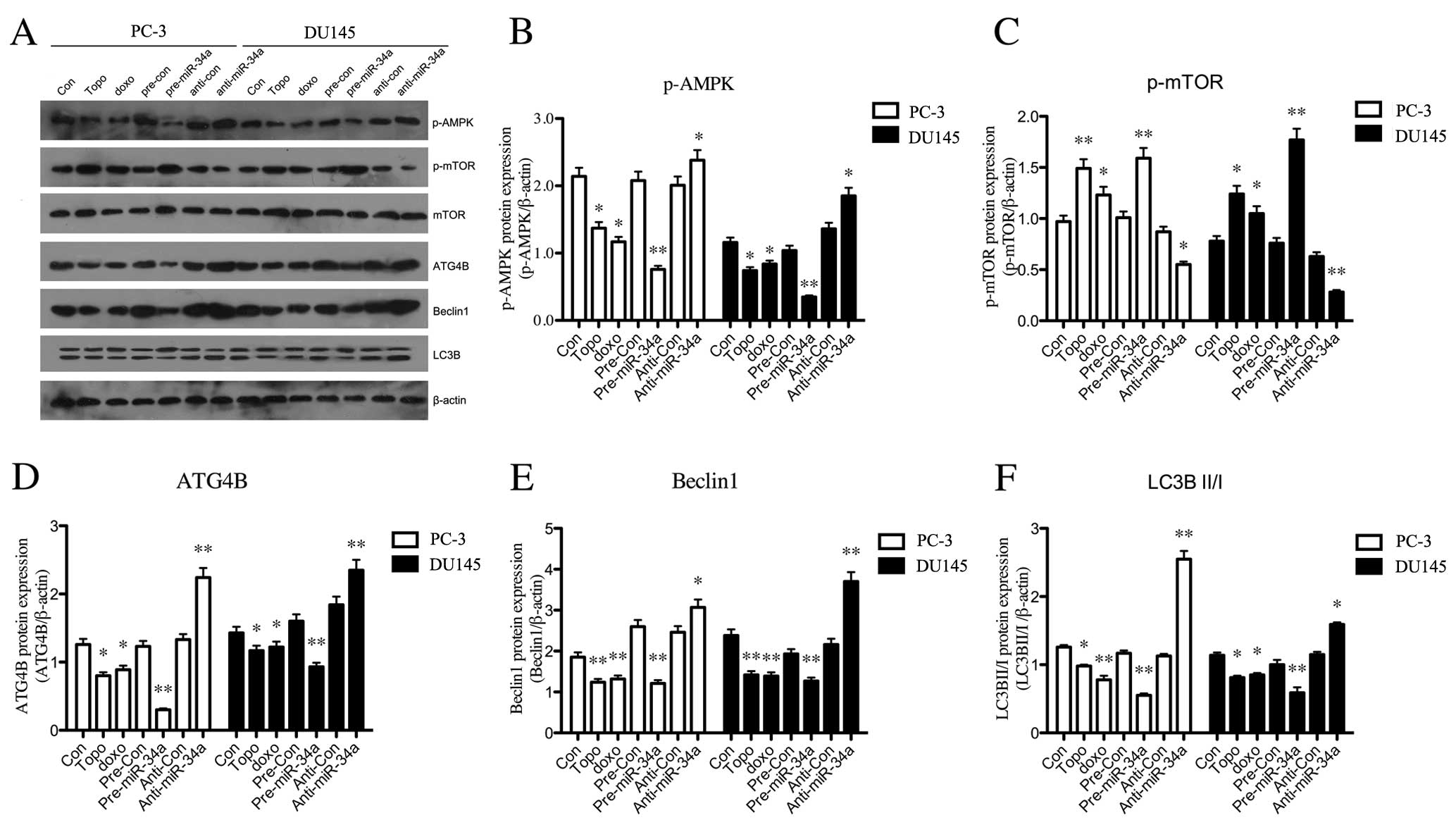
miR-34a regulates ATG4B-induced autophagy
through the AMPK/mTOR pathway
In order to investigate the pathway by which miR-34a
regulates autophagy in PCa cells, we performed western blotting for
the analysis of protein expression in the AMPK/mTOR pathway
(Fig. 6A). As shown in Fig. 6B, the level of phosphorylated AMPK
was significantly downregulated in chemotherapy-treated group.
Overexpression of miR-34a also led to a decrease on expression of
phosphorylated AMPK, while silence of miR-34a resulted in a
significant upregulation. Additionally, p-mTOR was significantly
reduced when miR-34a was overexpressed in PC-3 or DU145 cells, and
anti-miR-34a statistically upregulated the expression of p-mTOR
(Fig. 6C). Besides, the treatments
with Dox or Topo on cells resulted in an increase on p-mTOR
expression (Fig. 6C). Furthermore,
the expression of autophagy-related proteins were downregulated
after miR-34a overexpression, including ATG4B (Fig. 6D), Beclin-1 (Fig. 6E) and LC3B II/I (Fig. 6F). The anti-miR-34a transfection
reversed the low expression level of these proteins, with a
significant increase compared with control group (Fig. 6D–F). These data collectively
indicated that miR-34a induced the downregulation of
autophagy-related proteins, ATG4B, Beclin-1 and LC3B II/I, through
the AMPK/mTOR pathway.
Discussion
Accumulating evidences have shown the decrease of
miR-34a in prostate cancer (PCa) and its relation with drug
resistance (12). Autophagy also
plays an important role in chemoresistance of cancer (14–16).
However, whether the chemoresistance induced by miR-34a is related
to ATG4B in PCa, including their mechanism on chemoresistance,
still further research is needed. In the present study, we found
that upregulation of miR-34a induced by mimic transfection and
demethylation treatment using 5-azacytidine (5-Aza) resulted in the
enhanced apoptosis and chemosensitivity in PCa cells.
Overexpression of miR-34a reduced the expression of
autophagy-related proteins, ATG4B, Beclin-1 and LC3B II/I in PCa
cells by downregulating p-AMPK and upregulating p-mTOR in PCa
cells.
We detected the hypermethylation condition of
miR-34a in PCa tissues and cell lines such as PC-3 and DU145.
Previously, several lines of evidence demonstrated miR-34a promoter
methylation in many types of cancers (11). Moreover, Lodygin et al
demonstrated that the loss of miR-34a expression associated with
the methylation of promoter CpG island was found in the majority
(79.1%) of primary prostate carcinoma samples (11). Additionally, miR-34a expression was
markedly reduced in PCa cell lines (12). There data are in accordance with our
findings. Notably, miR-34a is even linked to PCa metastasis,
suggesting that it could be a potential biomarker for PCa (21).
In addition, we found miR-34a effective on PCa cell
apop-tosis. Previously, Bommer et al showed that Bcl-2 is
targeted by miR-34a (22). Welch
et al also found the re-expression of miR-34a induced
restoration of apoptosis through the activation of caspases 3 and 7
in neuroblastoma (23). Besides,
miR-34a synthetic mimics were proved to trigger growth inhibition
and apoptosis in multiple myeloma cells by down-regulating canonic
targets BCL-2, CDK6 and NOTCH1 in vitro and in vivo
(24). All these data were in
accordance with our findings. Additionally, Nalls et al
revealed that re-expression of miR-34a in pancreatic CSCs inhibited
cell growth, cell cycle progression, self-renewal, epithelial to
mesenchymal transition and invasion (25). Forced expression of miR-34a was able
to significantly suppress tumor growth in an orthotopic mouse model
of breast cancer (26). Using
chemically synthesized miR-34a and a lipid-based delivery vehicle
efficiently inhibited tumor growth without liver or kidney toxicity
and immune response in a mouse lung tumor model (27). Therefore, upregulation of miR-34a
could be an attractive new therapeutic strategy to improve
treatment outcome of cancer.
Furthermore, accumulating evidence demonstrated that
miR-34 family is involved in drug resistance in PCa. For example,
re-expression of miR-34a attenuated chemoresistance to anticancer
drug camptothecin via induction of apoptosis in PCa (12), also in agreement with our findings.
We found that upregulation of miR-34a by transfection of mimics
resulted in enhanced chemosensitivity in PCa cells, which proved
the role of miR-34a in chemoresistance. Besides, we also found
5-Aza treatment resulted in similar effect with transfection of
miR-34a mimics. This indicated the epigenetic regulation on miR-34a
in PCa was significantly responsible for the chemo-resistance in
PCa. Notably, recent studies have described that miRNAs are
involved in regulation of autophagic process by modulating
autophagy-related genes (ATGs). miR-101 and miR-376b negatively
regulates the expression of ATG4C and ATG4D (17,18).
miR-17 reduces ATG7 expression in glioblastoma cell lines (19). Furthermore, since the high-level
autophagy in tumor cells following anticancer treatment is
considered to be related with drug resistance (16), and Rothe et al demonstrated
that miR-34a regulated the expression of ATG4B in CML, and acted as
a factor on regulating drug resistance (28), we focused on the regulation role of
miR-34a on ATG4B in PCa. Notably, we found that ATG4B was the
direct target of miR-34a in PCa.
AMPK/mTOR is the essential regulator of cellular
autophagy (29). We have proved the
regulation of miR-34a on ATG4B, however, it is unclear whether the
regulation effect is induced by the AMPK/mTOR pathway. We evaluated
the expression of phosphorylated AMPK and mTOR after
over-expression or silencing of miR-34a in PCa cells. According to
its active role in autophagy (30,31),
p-AMPK was down-regulated in miR-34a overexpressed PCa cells,
followed by the upregulated p-mTOR, and in agreement with the
inhibitory function of mTOR in autophagy (32,33),
upregulated p-mTOR, induced by miR-34a overexpression finally led
to the decrease of autophagy-related proteins, including ATG4B,
Beclin-1 and LC3B II/I. We found miR-34a downregulated
autophagy-related proteins by the pathway of AMPK/mTOR.
In conclusion, we demonstrated that upregulation of
miR-34a by transfection or demethylation resulted in the enhanced
apoptosis and drug sensitivity in PCa cells. ATG4B, directly
regulated by miR-34a through AMPK/mTOR, was involved in this
process. Our research may supply a novel target for the future
treatment on PCa chemoresistance.
Abbreviations:
|
PCa
|
prostate cancer
|
|
miR-34a
|
microRNA-34a
|
|
Topo
|
Topotecan
|
|
Dox
|
doxorubicin
|
|
5-Aza
|
5-azacytidine
|
References
|
1
|
Filipowicz W, Bhattacharyya SN and
Sonenberg N: Mechanisms of post-transcriptional regulation by
microRNAs: Are the answers in sight? Nat Rev Genet. 9:102–114.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Calin GA, Sevignani C, Dumitru CD, Hyslop
T, Noch E, Yendamuri S, Shimizu M, Rattan S, Bullrich F, Negrini M,
et al: Human microRNA genes are frequently located at fragile sites
and genomic regions involved in cancers. Proc Natl Acad Sci USA.
101:2999–3004. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J and
Thun MJ: Cancer statistics, 2009. CA Cancer J Clin. 59:225–249.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shi XB, Xue L, Yang J, Ma AH, Zhao J, Xu
M, Tepper CG, Evans CP, Kung HJ and deVere White RW: An
androgen-regulated miRNA suppresses Bak1 expression and induces
androgen-independent growth of prostate cancer cells. Proc Natl
Acad Sci USA. 104:19983–19988. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lu Z, Liu M, Stribinskis V, Klinge CM,
Ramos KS, Colburn NH and Li Y: MicroRNA-21 promotes cell
transformation by targeting the programmed cell death 4 gene.
Oncogene. 27:4373–4379. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sun R, Fu X, Li Y, Xie Y and Mao Y: Global
gene expression analysis reveals reduced abundance of putative
microRNA targets in human prostate tumours. BMC Genomics.
10:932009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Volinia S, Calin GA, Liu CG, Ambs S,
Cimmino A, Petrocca F, Visone R, Iorio M, Roldo C, Ferracin M, et
al: A microRNA expression signature of human solid tumors defines
cancer gene targets. Proc Natl Acad Sci USA. 103:2257–2261. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pang Y, Young CY and Yuan H: MicroRNAs and
prostate cancer. Acta Biochim Biophys Sin. 42:363–369. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hermeking H: p53 enters the microRNA
world. Cancer Cell. 12:414–418. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lodygin D, Tarasov V, Epanchintsev A,
Berking C, Knyazeva T, Körner H, Knyazev P, Diebold J and Hermeking
H: Inactivation of miR-34a by aberrant CpG methylation in multiple
types of cancer. Cell Cycle. 7:2591–2600. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fujita Y, Kojima K, Hamada N, Ohhashi R,
Akao Y, Nozawa Y, Deguchi T and Ito M: Effects of miR-34a on cell
growth and chemoresistance in prostate cancer PC3 cells. Biochem
Biophys Res Commun. 377:114–119. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mizushima N: Autophagy: Process and
function. Genes Dev. 21:2861–2873. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Choi AM, Ryter SW and Levine B: Autophagy
in human health and disease. N Engl J Med. 368:651–662. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
White E: Deconvoluting the
context-dependent role for autophagy in cancer. Nat Rev Cancer.
12:401–410. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yang ZJ, Chee CE, Huang S and Sinicrope
FA: The role of autophagy in cancer: Therapeutic implications. Mol
Cancer Ther. 10:1533–1541. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Korkmaz G, le Sage C, Tekirdag KA, Agami R
and Gozuacik D: miR-376b controls starvation and mTOR
inhibition-related autophagy by targeting ATG4C and BECN1.
Autophagy. 8:165–176. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Frankel LB, Wen J, Lees M, Høyer-Hansen M,
Farkas T, Krogh A, Jäättelä M and Lund AH: microRNA-101 is a potent
inhibitor of autophagy. EMBO J. 30:4628–4641. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Comincini S, Allavena G, Palumbo S, Morini
M, Durando F, Angeletti F, Pirtoli L and Miracco C: microRNA-17
regulates the expression of ATG7 and modulates the autophagy
process, improving the sensitivity to temozolomide and low-dose
ionizing radiation treatments in human glioblastoma cells. Cancer
Biol Ther. 14:574–586. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang J, Chen D, He Y, Meléndez A, Feng Z,
Hong Q, Bai X, Li Q, Cai G, Wang J, et al: MiR-34 modulates
Caenorhabditis elegans lifespan via repressing the autophagy gene
atg9. Age. 35:11–22. 2013. View Article : Google Scholar :
|
|
21
|
Watahiki A and Wang Y, Morris J, Dennis K,
O'Dwyer HM, Gleave M, Gout PW and Wang Y: MicroRNAs associated with
metastatic prostate cancer. PLoS One. 6:e249502011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bommer GT, Gerin I, Feng Y, Kaczorowski
AJ, Kuick R, Love RE, Zhai Y, Giordano TJ, Qin ZS, Moore BB, et al:
p53-mediated activation of miRNA34 candidate tumor-suppressor
genes. Curr Biol. 17:1298–1307. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Welch C, Chen Y and Stallings RL:
MicroRNA-34a functions as a potential tumor suppressor by inducing
apoptosis in neuroblastoma cells. Oncogene. 26:5017–5022. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Di Martino MT, Leone E, Amodio N, Foresta
U, Lionetti M, Pitari MR, Cantafio ME, Gullà A, Conforti F, Morelli
E, et al: Synthetic miR-34a mimics as a novel therapeutic agent for
multiple myeloma: In vitro and in vivo evidence. Clin Cancer Res.
18:6260–6270. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nalls D, Tang SN, Rodova M, Srivastava RK
and Shankar S: Targeting epigenetic regulation of miR-34a for
treatment of pancreatic cancer by inhibition of pancreatic cancer
stem cells. PLoS One. 6:e240992011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li L, Xie X, Luo J, Liu M, Xi S, Guo J,
Kong Y, Wu M, Gao J, Xie Z, et al: Targeted expression of miR-34a
using the T-VISA system suppresses breast cancer cell growth and
invasion. Mol Ther. 20:2326–2334. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Trang P, Wiggins JF, Daige CL, Cho C,
Omotola M, Brown D, Weidhaas JB, Bader AG and Slack FJ: Systemic
delivery of tumor suppressor microRNA mimics using a neutral lipid
emulsion inhibits lung tumors in mice. Mol Ther. 19:1116–1122.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rothe K, Lin H, Lin KB, Leung A, Wang HM,
Malekesmaeili M, Brinkman RR, Forrest DL, Gorski SM and Jiang X:
The core autophagy protein ATG4B is a potential biomarker and
therapeutic target in CML stem/progenitor cells. Blood.
123:3622–3634. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Fan X, Wang J, Hou J, Lin C, Bensoussan A,
Chang D, Liu J and Wang B: Berberine alleviates ox-LDL induced
inflammatory factors by up-regulation of autophagy via AMPK/mTOR
signaling pathway. J Transl Med. 13:922015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Herrero-Martín G, Høyer-Hansen M,
García-García C, Fumarola C, Farkas T, López-Rivas A and Jäättelä
M: TAK1 activates AMPK-dependent cytoprotective autophagy in
TRAIL-treated epithelial cells. EMBO J. 28:677–685. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Matsui Y, Takagi H, Qu X, Abdellatif M,
Sakoda H, Asano T, Levine B and Sadoshima J: Distinct roles of
autophagy in the heart during ischemia and reperfusion: Roles of
AMP-activated protein kinase and Beclin 1 in mediating autophagy.
Circ Res. 100:914–922. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sudarsanam S and Johnson DE: Functional
consequences of mTOR inhibition. Curr Opin Drug Discov Devel.
13:31–40. 2010.PubMed/NCBI
|
|
33
|
Jung CH, Ro SH, Cao J, Otto NM and Kim DH:
mTOR regulation of autophagy. FEBS Lett. 584:1287–1295. 2010.
View Article : Google Scholar : PubMed/NCBI
|