Introduction
Normal and pathological tissue growth depends on
sufficient angiogenesis. Especially in cancer development,
neoangiogenesis plays a key role. During cancer progression two
different phases of cancer nutrition can be distinguished. At the
beginning of cancer development, nutrition is supplied by
diffusion. At a critical tumor volume of 1–2 mm3,
diffusion is no longer sufficient (1). Further tumor progression requires a
vascular network, which is induced by tumor cells via cytokines and
growth factors. This phenomenon is called the 'angiogenic switch'
(2). During tumor progression, the
tissue behaves like an inflamed wound (3). Hence, the migration of several cell
types, including human mesenchymal stem cells (hMSCs), is induced.
The latter is even equipped with a pro-angiogenic capability. In a
study conducted by Kinnaird et al, hMSCs released several
cytokines responsible for angiogenesis, e.g., vascular endothelial
growth factor (VEGF), placental growth factor (PIGF) and monocyte
chemoattractant protein-1 (MCP-1) (4). Further cytokines were detected in
hMSC-conditioned medium, especially interleukin 6 (IL-6) (5), which is a multifunctional cytokine
responsible for immune response as well as hematopoiesis (6,7).
hMSCs are self-renewing multipotent cells with the
ability to differentiate into various mesenchymal cell types, e.g.,
osteoblasts, chondrocytes and adipocytes (8). Recent data indicate that hMSCs are
pericytes whose pleiotropic nature allows them to sense and respond
to inflammatory processes in their microenvironment (9). The migratory behavior of systemically
administered hMSCs enables new therapeutic options, especially in
the field of drug delivery. Myers et al found that hMSCs
have been therapeutically applied in more than 200 active clinical
trials worldwide (9). However,
using hMSCs as a drug-carrier for cancer therapy is complicated,
since data concerning cancer and hMSC interaction are
contradictory. On the one hand, there are studies postulating tumor
progression and enhancement of tumor metastatic potential by hMSCs
via cell-cell contact as well as via secretion of cytokines and
growth factors (10–12). On the other hand, reports on the
anti- tumorigenic effects of hMSCs exist as well (13,14).
Different sources of stem cells with variable differentiation
status may contribute to the divergent results. The aim of the
present study was to investigate the cytokine patterns of the HNSCC
cell line FaDu, native hMSCs (hMSCs-nat), hMSCs differentiated into
adipocytes (hMSCs-adip) and osteocytes (hMSCs-ost). Furthermore,
the migration capabilities as well as the angiogenic effects of
hMSCs-nat, hMSCs-adip and hMSCs-ost were evaluated in a co-culture
with human umbilical vein endothelial cells (HUVECs).
Materials and methods
hMSC isolation and culture
hMSCs were isolated from five voluntary patients
undergoing surgery at the Department of Orthopedics,
Koenig-Ludwig-Haus (Wuerzburg). Informed consent was provided by
all patients. The study was approved by the Ethics Committee of the
Medical Faculty of the University of Wuerzburg (12/06). Ficoll
density gradient centrifugation (30 min, 1,300 rpm, density, 1,077
g/ml; Biochrom AG, Berlin, Germany) was performed as previously
described (15). After
centrifugation, cells in the interphase were collected. Several
washing steps with phosphate-buffered saline (PBS) (Roche
Diagnostics GmbH, Mannheim, Germany) containing 2% fetal calf serum
(FCS) (Linaris, Wertheim-Bettingen, Germany) followed. The cells
were resuspended in Dulbecco's modified Eagle's medium (DMEM)
containing 10% FCS, 1% penicillin and streptomycin (Sigma-Aldrich,
Schnelldorf, Germany). Flow cytometry (FACSCanto™; BD Bioscience,
Heidelberg, Germany) was performed to show the characteristic
surface markers of hMSCs. Furthermore, osteogenic and adipogenic
differentiation was carried out as described previously (16). Osteogenic medium consisted of
expansion medium (DMEM-EM) supplemented with 10−7 M
dexamethasone, 10−3 M β-glycerophosphate and 2–4 M
ascorbate-2-phosphate (all from Sigma-Aldrich). Adipogenic
differentiation medium was prepared by the addition of
10−7 M dexamethasone and 10−9 g/ml
recombinant human insulin (all from Sigma-Aldrich) to DMEM-EM.
Osteogenic differentiation was confirmed by Alizarin Red staining
and adipogenic differentiation by Oil Red O staining.
HUVEC isolation and culture
HUVECs were isolated from human umbilical veins of
three voluntary patients via 0.25% trypsin (Gibco Invitrogen,
Karlsruhe, Germany) digestion. First, the umbilical vein was washed
in D-Hank's solution. Next, a 10-min perfusion with a 0.25% trypsin
solution at 37°C followed. The enzyme reaction was blocked by
addition of FCS. After a centrifugation step at 1,000 rpm for 15
min, the cells were cultured in endothelial cell growth medium with
supplements (ECGM) (Provitro GmbH, Berlin, Germany) and 1%
penicillin/streptomycin.
HNSCC cell line FaDu
The head and neck squamous carcinoma (HNSCC) cell
line FaDu was used (17). The
cultivation of the FaDu cells was performed in DMEM-EM at 37°C with
5% CO2. Medium was replaced every other day.
Generation of multi-cellular
spheroids
HUVECs were labeled with the fluorochrome
1,1′-dioctadecyl-3,3,3′,3′-tetramethy-lindo-carbocyanine
perchlorate (DiI) (Gibco Invitrogen). The generation of
multi-cellular spheroids was performed as described previously
(16). After solidification of 0.1%
soft agar (Sigma-Aldrich) in a 96-multiwell plate, 0.2 ml DMEM-EM
containing 3×103 FaDu cells and 3×103 HUVECs
or 2×103 FaDu, 2×103 HUVECs and
2×103 hMSCs-nat were added.
Cytokine analysis of FaDu cells,
hMSCs-nat, hMSCs-adip and hMSCs-ost with the dot blot assay
The dot blot assay (RayBiotec Inc., Norcross, GA,
USA) is a semiquantitative method for investigating the cytokine
secretion of cells. FaDu cells, hMSCs-nat, hMSCs-ost and hMSCs-adip
were cultured in DMEM without supplements. Next, the supernatants
were collected and analyzed for the presence of cytokines. The
assay was performed according to the manufacturer's protocol. The
observation of labeled cytokines was achieved via enhanced
chemiluminescence using detection buffer and exposure to an X-ray
film. The cytokines are represented as dots with different
intensity and growth size.
hMSC migration towards FaDu cells
The migration assay was assessed in Transwells
(Corning Incorporated Costar, Wiesbaden, Germany), which were
coated with 50% Matrigel (Sigma-Aldrich). hMSCs-nat, hMSCs-adip or
hMSCs-ost were coated on the top surface of the membrane and
incubated with DMEM without FCS for 24 h at 37°C with 5%
CO2. A total of 5×104 FaDu cells was added to
the bottom of the well plate, followed by another incubation period
for 24 h. A cotton swab was used to remove the non-migratory cells
on the upper membrane. The migrated cells, which collected on the
lower surface of the membrane, were stained with 1% crystal violet
(Sigma-Aldrich) for 25 min. Another washing step with aqua
destillata followed. The cells were detached by incubation with 500
μl 10% acetic acid for 20 min. Extinction was measured at
570 nm using a multi-plate reader (Titertek Multiskan Plus MKII;
Labsystems, Helsinki, Finland). The migration of hMSCs towards
DMEM-FCS served as the control. The results of the control group
were defined as 100%.
Capillary tube formation assay
hMSCs-nat, hMSCs-adip and hMSCs-ost were incubated
on the bottom of μ-Slide Angiogenesis (Ibidi, Munich,
Germany) for 2 h. Next, 50% Matrigel (Sigma-Aldrich) was added to
the top of each cell system. After 30 min, 3×103
DiI-labeled HUVECs were coated on the Matrigel with ECGM. After 12
h, the tube formation was evaluated by inverted microscopy (Leica
DMI 4000B inverted microscope; Leica Microsystems, Wetzlar,
Germany). The tube length was measured using ImageJ software
(http://imagej.nih.gov/ij/).
Cultivation of hMSCs-nat on Matrigel
A total of 50% Matrigel was added to the bottom of
48-well plates for 30 min. After solidification, 104
hMSCs-nat were added and cultured with DMEM-EM. After 72 h, the
cells were observed by inverted microscopy.
Statistical analysis
All data were transferred to standard spreadsheets.
Differences between groups were examined for significance by the
Kruskal-Wallis test using GraphPad Prism 6.0 statistics software
(GraphPad Software, Inc., San Diego, CA, USA). P<0.05 was
considered to indicate a statistically significant difference.
Results
hMSC morphology and differentiation
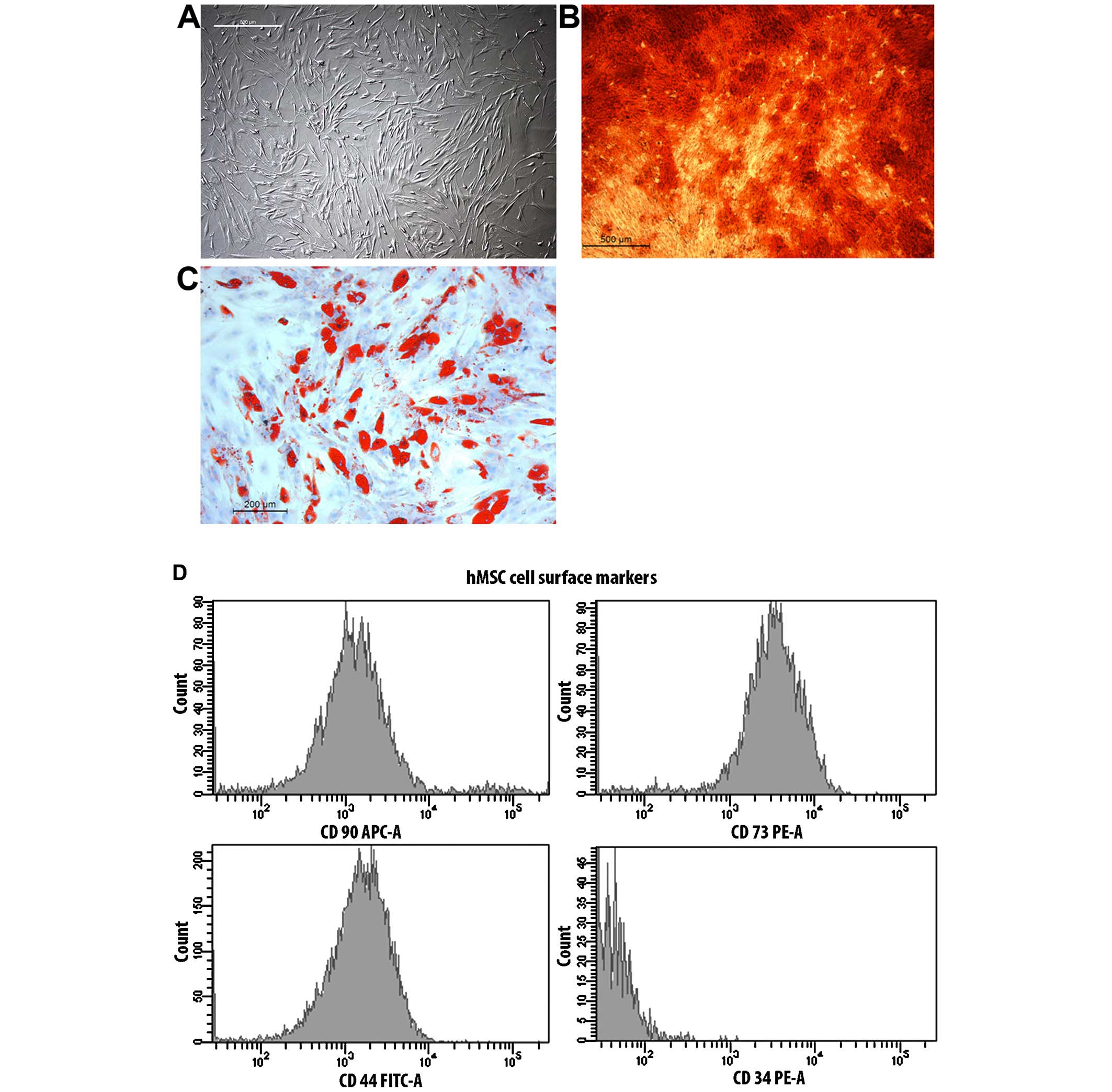
The hMSCs showed typical spindle-shaped structures.
Cells were positive for CD105, CD90 and CD44, and negative for
CD34. The differentiation into adipocytes was confirmed by Oil Red
O staining with typical intracellular lipid vacuoles. The
osteogenic differentiation was shown with Alizarin Red staining.
The extracellular calcium deposits were stained red (Fig. 1).
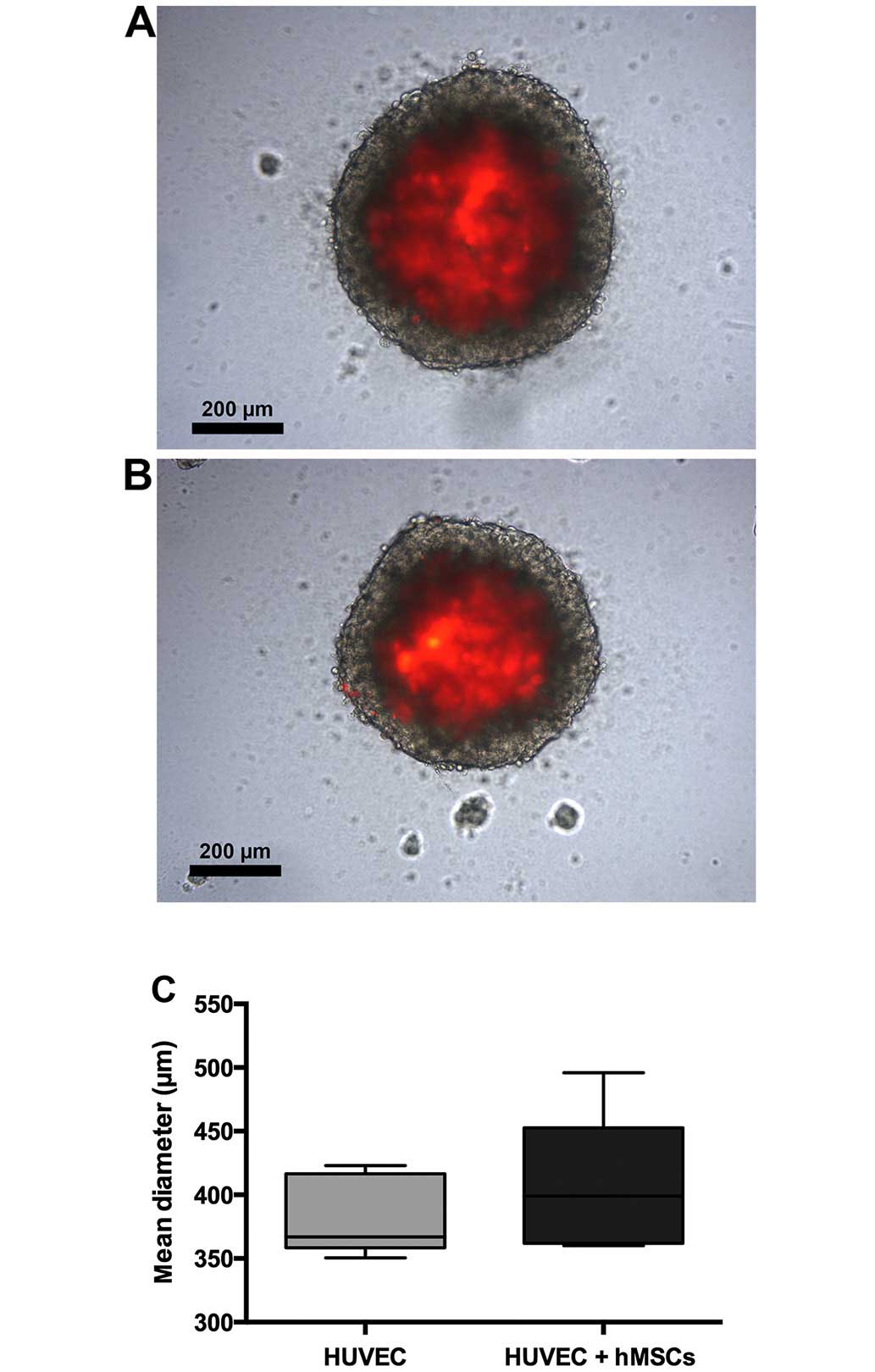
Morphology of the spheroids
There was no difference in spheroid growth from the
FaDu and HUVECs as compared to the spheroids from the FaDu cells,
hMSCs-nat and HUVECs. Spheroids had a well-shaped structure. The
mean diameter of the spheroids from the FaDu and the HUVECs was
383.4 μm. The mean diameter of the spheroids from the FaDu,
hMSCs-nat and HUVECs was 405.6 μm. The differences between
both groups were not statistically significant. Fluorescence
microscopy revealed that almost all HUVECs were located in the core
of the spheroids. There were only very few HUVECs observed in the
circumference (Fig. 2).
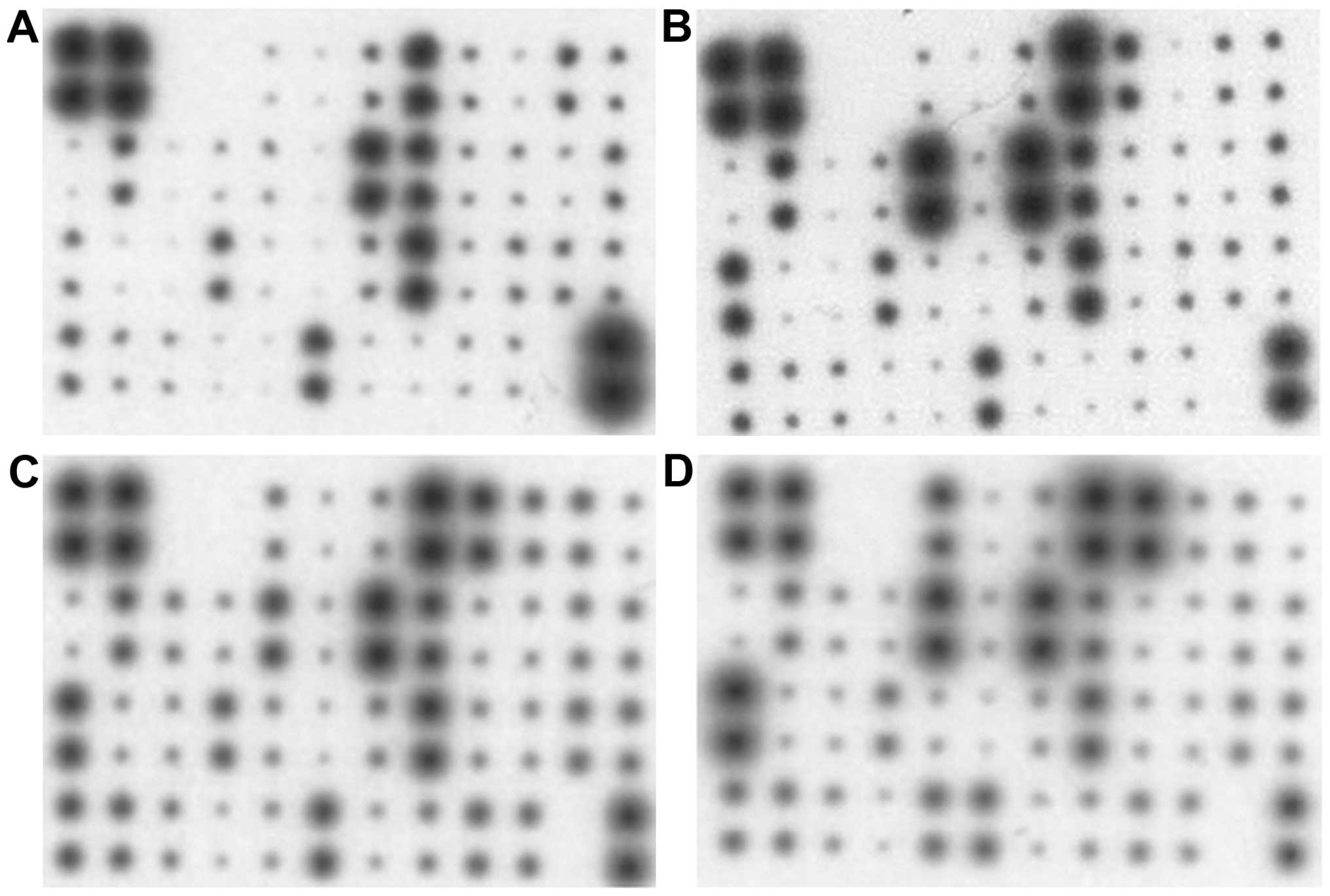
Cytokine analysis of hMSCs-nat,
hMSCs-adip, hMSCs-ost and FaDu cells
FaDu cells, hMSCs-nat, hMSCs-adip and hMSCs-ost
released different cytokines and growth factors responsible for
inflammation, angiogenesis and chemotaxis (Table I). Dots of the following cytokines
had a strong intensity in the FaDu cells: interleukin (IL)-3, IL-8,
IL-10, growth regulated oncogene (GRO)-α, RANTES, macrophage
colony-stimulating factor (MCSF), oncostatin M and tumor necrosis
factor (TNF)-α.
 | Table IArray map. |
Table I
Array map.
| + | + | – | – | ENA-78 | GCSF | GM-CSF | GRO | GRO-α | I-309 | IL-1α | IL-1β |
| + | + | – | – | ENA-78 | GCSF | GM-CSF | GRO | GRO-α | I-309 | IL-1α | IL-1β |
| IL-2 | IL-3 | IL-4 | IL-5 | IL-6 | IL-7 | IL-8 | IL-10 | IL-12 | IL-13 | IL-15 | IFN-γ |
| IL-2 | IL-3 | IL-4 | IL-5 | IL-6 | IL-7 | IL-8 | IL-10 | IL-12 | IL-13 | IL-15 | IFN-γ |
| MCP-1 | MCP-2 | MCP-3 | MCSF | MCD | MIG | MIP-1d | RANTES | SCF | SDF-1TA | RC | TGF-β1 |
| MCP-1 | MCP-2 | MCP-3 | MCSF | MCD | MIG | MIP-1d | RANTES | SCF | SDF-1TA | RC | TGF-β1 |
| TNF-α | TNF-β | EGF | IGF-1 | Angogenin | Oncostatin M | Thrombopoietin | VEGF | PDGF-BB | Leptin | - | + |
| TNF-α | TNF-β | EGF | IGF-1 | Angogenin | Oncostatin M | Thrombopoietin | VEGF | PDGF-BB | Leptin | - | + |
Dots of the following cytokines had a strong
intensity in hMSCs-nat: IL-3, IL-6, IL-8, IL-10, GRO, GRO-α,
monocyte chemotactic protein (MCP)-1, MCSF, RANTES, TNF-α and
oncostatin M.
Dots of the following cytokines had a strong
intensity in the hMSCs-adip: IL-3, IL-6, IL-8, IL-10, GRO, GRO-α,
MCP-1, MCSF, RANTES, TNF-α, TNF-β, oncostatin M, platelet-derived
growth factor (PDGF)-BB and leptin.
Dots of the following cytokines had a strong
intensity in the hMSCs-ost: IL-3, IL-6, IL-8, C-X-C motif chemokine
5, GRO, GRO-α, MCP-1, MCSF, RANTES, TNF-α, TNF-β, angiogenin,
oncostatin M, PDGF-BB and leptin (Fig.
3).
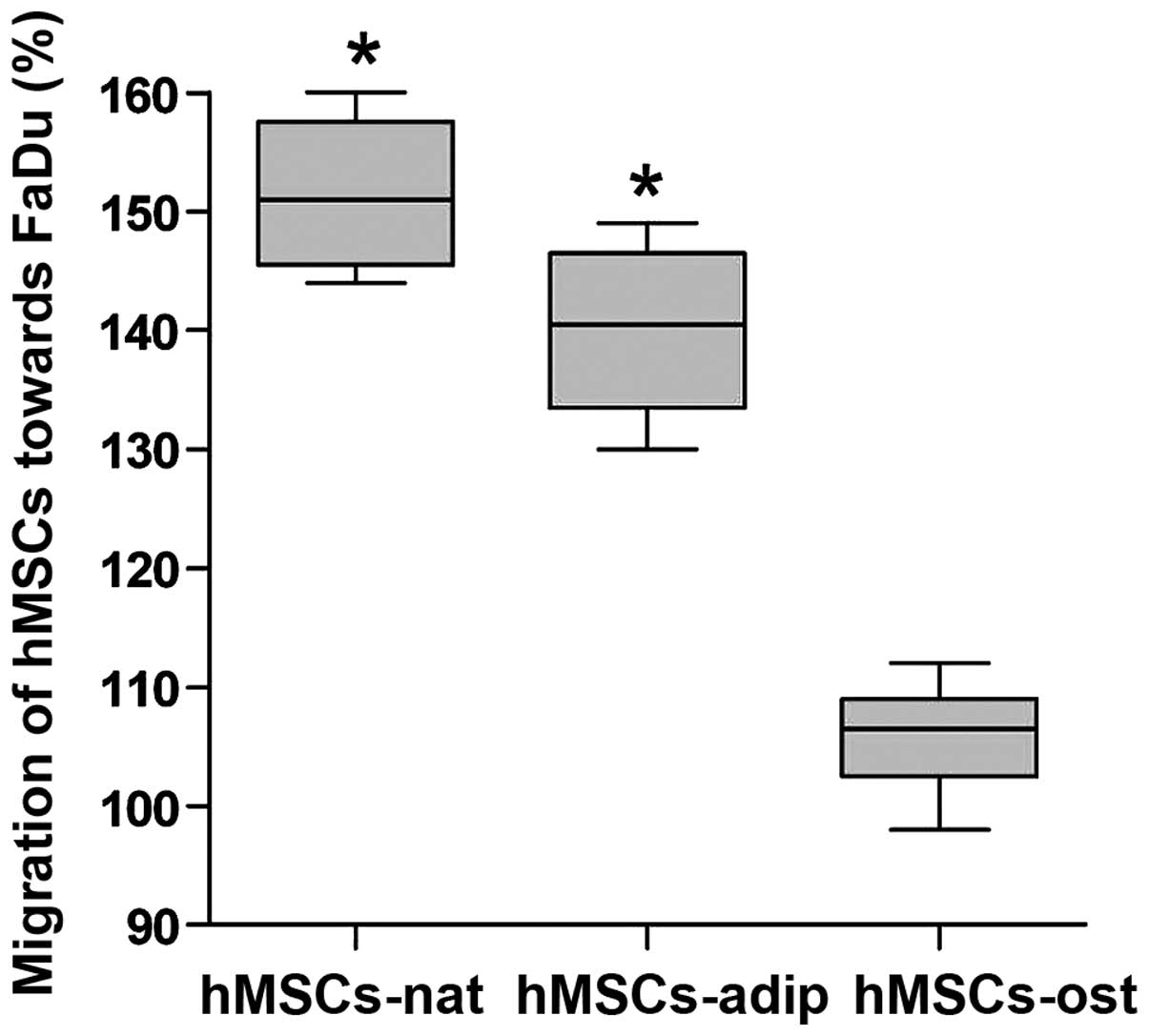
Migration of hMSCs-nat, hMSCs-adip and
hMSCs-ost towards FaDu cells
The migration assay was conducted in order to
evaluate the differences between hMSCs-nat, hMSCs-adip and
hMSCs-ost. The results of the control group (migration towards DMEM
+ 10% FCS) were defined as 100%. hMSCs-nat showed a significant
enhancement in migration by 52% towards the FaDu cells. The
differentiation of hMSCs into adipocytes (hMSCs-adip) did not
counteract the migration enhancement as noted in the hMSC-nat.
The differentiation of hMSCs into osteocytes
(hMSCs-ost) attenuated the migration capability significantly as
compared to the migration of hMSC-nat and hMSC-adip. However, there
were no significant differences between hMSCs-ost and the control
group (Fig. 4).
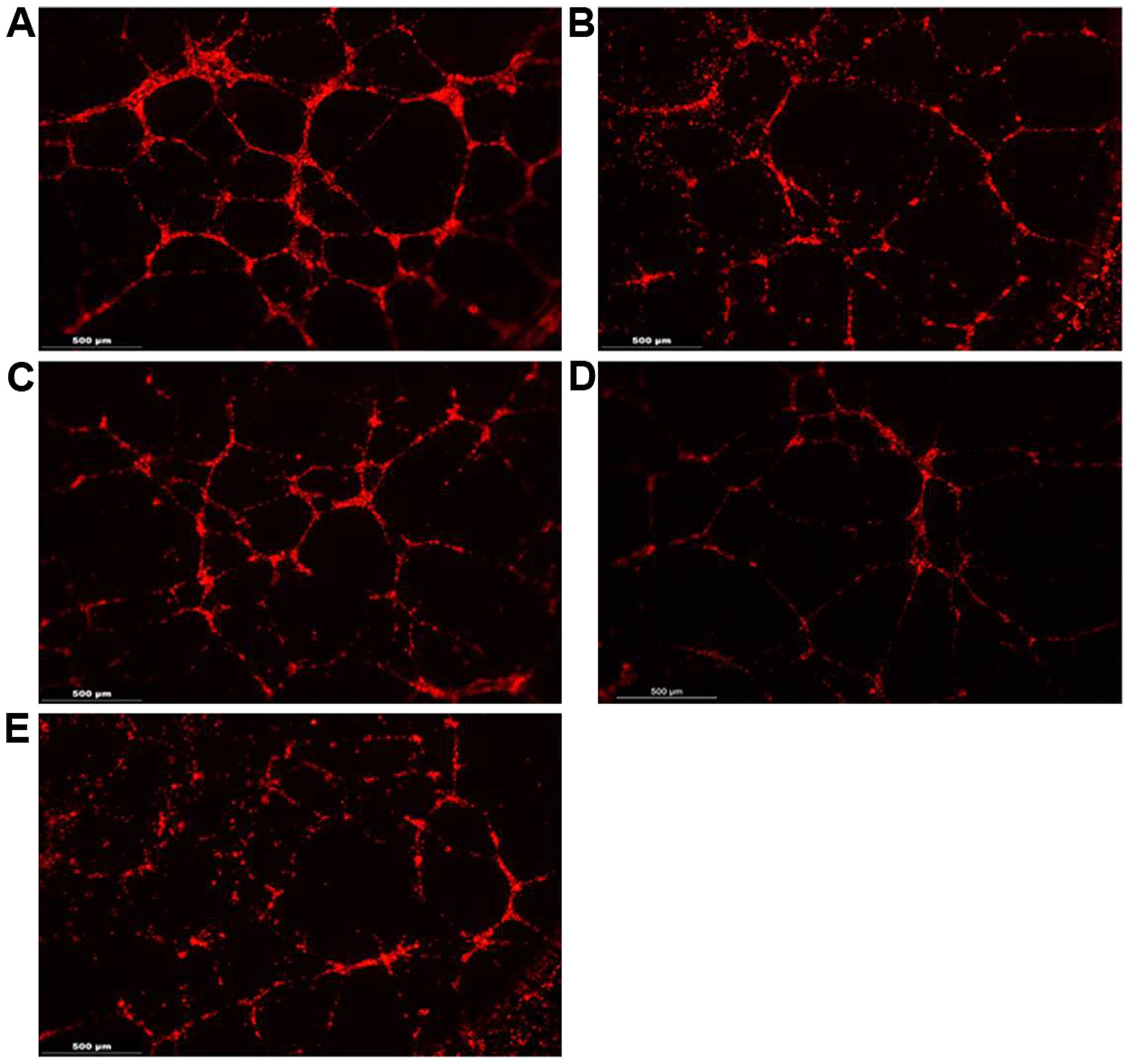
Capillary tube formation assay on
Matrigel
HUVECs formed tube-like structures on the Matrigel,
and the lengths of these tubes were measured with ImageJ software.
In co-culture with hMSCs-nat, HUVECs developed significantly longer
tube-like structures as compared to the HUVECs alone or co-cultured
with the FaDu cells. The formation of tube-like structures was
counteracted by the co-cultivation with hMSCs-adip and hMSCs-ost.
The tube length was significantly attenuated after the
co-cultivation of the HUVECs with hMSCs-adip or hMSCs-ost. The
lengths as well as the ability to build tube-like structures were
negatively affected (Fig. 5).
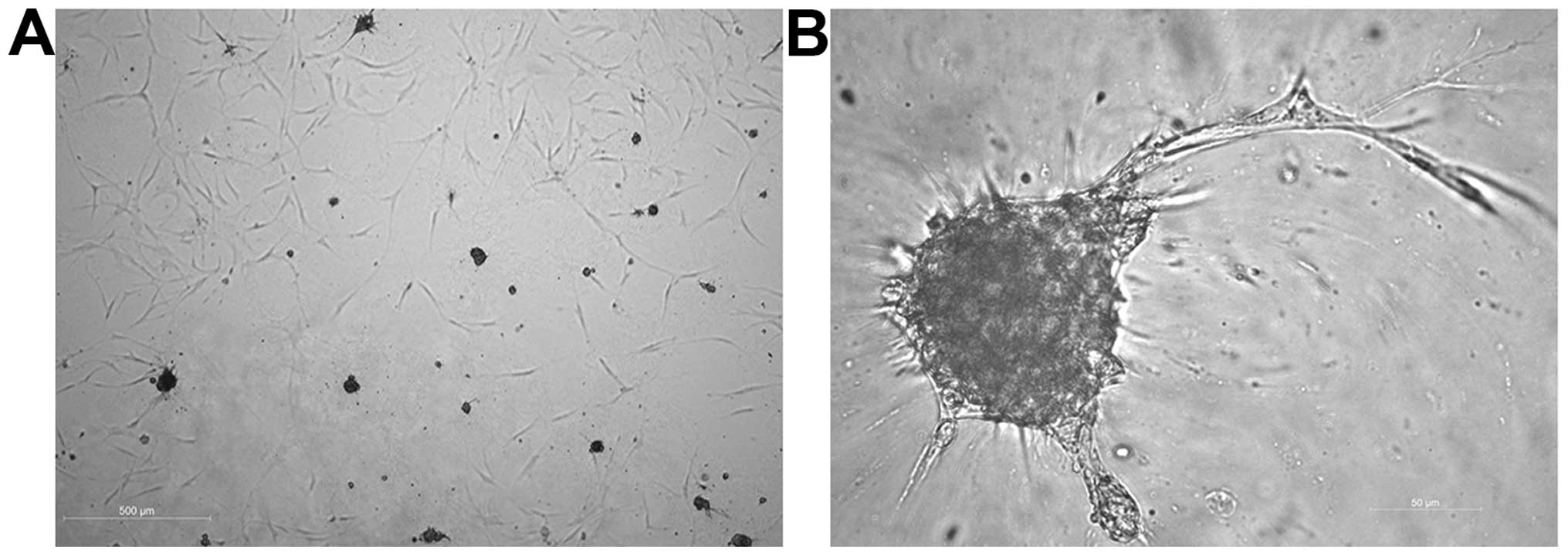
Cultivation of hMSCs-nat on Matrigel
After the cultivation of hMSCs-nat on Matrigel, some
of the cells took the shape of a spheroid. From these spheroids
tube-like structures sprouted out. The length of the tube-like
structures was not significantly higher as compared to the HUVECs
(Fig. 6).
Discussion
The induction of angiogenesis is one of the six
hallmarks of cancer as proposed by Hanahan and Weinberg. It differs
significantly in tumors compared to normal physiological processes.
In cases such as wound healing or female reproductive cycling the
angiogenesis process is turned on transiently, whereas in cancer
progression the so-called 'angiogenic switch' is activated
permanently (18). In addition to
nutrition and oxygen supply, neovascularization is a prerequisite
for cancer cells to circulate and induce metastasis (19). Furthermore, tumor vasculature may be
a reason for cancer therapy failure. The vessels can be leaky and
arranged chaotically (20). The
cancer vasculature system is abnormal with heterogeneous perfusion
areas, with the poorly perfused regions not being reached by
anticancer drugs (21). This may
lead to cancer resistance towards anticancer drugs. In the process
of angiogenesis, interactions between tumor cells with the
non-malignant tumor stroma are fundamental. Cytokines and growth
factors such as VEGF, fibroblast growth factors (FGF), angiopoietin
(Ang)-1 and Ang2, IL-6 and IL-8 are all part of the orchestration
(22). The dot blot assay revealed
the presence of these proteins in the present study. hMSCs as well
as FaDu cells were able to release cytokines and growth factors.
Cancer and stroma interact by direct cell-cell contact as well as
by soluble factors, which implies their symbiotic relationship. In
tissue engineering, especially in cardiology and the regeneration
of cardiomyocytes, hMSCs have already demonstrated their
pro-angiogenic features. These effects were mediated via secretion
of cytokines such as VEGF, angiopoietin, SDF-1 or integrin-linked
kinase (23–25). In ischemia studies, hMSCs showed
their pro-angiogenic effect, which was resolved via expression of
hypoxia inducible factor-1α (26,27).
The cultivation of hMSCs on Matrigel revealed capillary-like
formation. The potential of hMSCs to build capillary networks was
shown by several other groups (28–30).
The generation of this capillary network was induced in the
presence of VEGF and insulin-like growth factors. These results
demonstrate the direct involvement of hMSCs in angiogenesis by
forming capillary-like structures.
Cancer treatment must target several aspects of the
tumor including the tumor microenvironment. Here, anti-angiogenic
medications have recently gained popularity for the treatment of
several tumor entities (31–33).
However, targeted tumor therapy requires an appropriate vehicle,
and hMSCs show strong tumor tropism. Thus, hMSCs are often
considered to be an adequate candidate for tumor therapy (34–36).
Yet, the data in the literature concerning the applicability of
hMSCs are divergent. The majority of the studies show a
pro-tumorigenic effect of hMSCs. These effects are induced in
various different ways. Zhang et al demonstrated an
induction of autophagy and thereby an enhancement of cancer cell
survival (37). Jung et al
demonstrated an induction of epithelial-mesenchymal transition by
hMSCs, with a consecutive enhancement of the metastatic potential
of cancer cells (38). Cancer
growth was shown to be promoted by cytokine secretion from hMSCs
such as oncostatin M secretion (39). Cytokines appear to be primarily
responsible for cancer progression and metastasis.
In the minority of studies, cancer inhibition by
hMSCs was described. In a study conducted by Chen et al,
hMSCs derived from placenta were transfected with recombinant
adenoviruses expressing pigment epithelium-derived factor (PEDF),
and these hMSCs were able to inhibit melanoma cell growth. This
anticancer activity was discussed to be the result of PEDF
expression (40). Li et al
demonstrated inhibition of gastric cancer cells by hMSCs derived
from human foreskin (41).
The divergent results in the current literature may
be due to the use of hMSCs from different sources as well as the
donor's age. Nevertheless, hMSCs as a vehicle for cancer therapy
have been used in various studies (42–44),
particularly as an engineered vehicle administered via viral
transfection. In a study conducted by Martinez-Quintanilla et
al engineered hMSCs were able to co-express the pro-drug
converting enzyme as well as the herpes simplex virus thymidine
kinase (HSV-TK) and a potent and secretable variant of tumor
necrosis factor apoptosis-inducing ligand (S-TRAIL). Via these
capabilities caspase-mediated cancer cell death was inducted
(44). Other studies used
retroviral transfection of hMSCs with promising results (45,46).
However, such viral vectors may have risk factors, e.g.,
integration into the host chromosomes. Thus non-viral transfected
hMSCs or native hMSCs with antitumor properties may be the perfect
candidates for tumor-targeted therapy.
The present study revealed a pro-angiogenic effect
of hMSCs-nat. Interestingly, the differentiation of hMSCs
counteracted the pro-angiogenic effects as well as their migration
capability. The cause of this phenomenon needs to be investigated
to ascertain an alteration of the hMSC cytokine pattern during
differentiation. Counteracting the pro-angiogenic effects of hMSCs
may be suitable for the generation of a 'safe vehicle' for future
targeted tumor therapy.
Acknowledgments
The present study was supported by Rudolf Bartling
Stiftung, the Department of Orthopaedic Surgery, Koenig-Ludwig-Haus
(Professor Maximilian Rudert, Director) and Dr Marianne Schmidt,
Department of Otorhinolaryngology, Plastic, Aesthetic and
Reconstructive Head and Neck Surgery, Julius Maximilian University
of Wuerzburg, Germany.
References
|
1
|
Folkman J: What is the evidence that
tumors are angiogenesis dependent? J Natl Cancer Inst. 82:4–6.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bergers G and Benjamin LE: Tumorigenesis
and the angiogenic switch. Nat Rev Cancer. 3:401–410. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dvorak HF: Tumors: Wounds that do not
heal. Similarities between tumor stroma generation and wound
healing. N Engl J Med. 315:1650–1659. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kinnaird T, Stabile E, Burnett MS, Shou M,
Lee CW, Barr S, Fuchs S and Epstein SE: Local delivery of
marrow-derived stromal cells augments collateral perfusion through
paracrine mechanisms. Circulation. 109:1543–1549. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hung SC, Pochampally RR, Chen SC, Hsu SC
and Prockop DJ: Angiogenic effects of human multipotent stromal
cell conditioned medium activate the PI3K-Akt pathway in hypoxic
endothelial cells to inhibit apoptosis, increase survival, and
stimulate angiogenesis. Stem Cells. 25:2363–2370. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kanazawa T, Nishino H, Hasegawa M, Ohta Y,
Iino Y, Ichimura K and Noda Y: Interleukin-6 directly influences
proliferation and invasion potential of head and neck cancer cells.
Eur Arch Otorhinolaryngol. 264:815–821. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kishimoto T: The biology of interleukin-6.
Blood. 74:1–10. 1989.PubMed/NCBI
|
|
8
|
Caplan AI: Mesenchymal stem cells. J
Orthop Res. 9:641–650. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Myers JT, Barkauskas DS and Huang AY:
Dynamic imaging of marrow-resident granulocytes interacting with
human mesenchymal stem cells upon systemic lipopolysaccharide
challenge. Stem Cells Int. 2013:6568392013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Karnoub AE, Dash AB, Vo AP, Sullivan A,
Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R and Weinberg
RA: Mesenchymal stem cells within tumour stroma promote breast
cancer metastasis. Nature. 449:557–563. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin WR, Brittan M and Alison MR: The role
of bone marrow-derived cells in fibrosis. Cells Tissues Organs.
188:178–188. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rhodes LV, Muir SE, Elliott S, Guillot LM,
Antoon JW, Penfornis P, Tilghman SL, Salvo VA, Fonseca JP, Lacey
MR, et al: Adult human mesenchymal stem cells enhance breast
tumori-genesis and promote hormone independence. Breast Cancer Res
Treat. 121:293–300. 2010. View Article : Google Scholar
|
|
13
|
Khakoo AY, Pati S, Anderson SA, Reid W,
Elshal MF, Rovira II, Nguyen AT, Malide D, Combs CA, Hall G, et al:
Human mesen-chymal stem cells exert potent antitumorigenic effects
in a model of Kaposi's sarcoma. J Exp Med. 203:1235–1247. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhu Y, Sun Z, Han Q, Liao L, Wang J, Bian
C, Li J, Yan X, Liu Y, Shao C, et al: Human mesenchymal stem cells
inhibit cancer cell proliferation by secreting DKK-1. Leukemia.
23:925–933. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Scherzed A, Hackenberg S, Froelich K, Rak
K, Technau A, Radeloff A, Nöth U, Koehler C, Hagen R and
Kleinsasser N: Effects of salinomycin on human bone marrow-derived
mesenchymal stem cells in vitro. Toxicol Lett. 218:207–214. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Scherzed A, Hackenberg S, Radeloff A,
Froelich K, Rak K, Hagen R and Kleinsasser N: Human mesenchymal
stem cells promote cancer motility and cytokine secretion in vitro.
Cells Tissues Organs. 198:327–337. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rangan SR: A new human cell line (FaDu)
from a hypopharyngeal carcinoma. Cancer. 29:117–121. 1972.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zetter BR: Angiogenesis and tumor
metastasis. Annu Rev Med. 49:407–424. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Grantab RH and Tannock IF: Penetration of
anticancer drugs through tumour tissue as a function of cellular
packing density and interstitial fluid pressure and its
modification by bortezomib. BMC Cancer. 12:2142012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jain RK: Normalizing tumor
microenvironment to treat cancer: Bench to bedside to biomarkers. J
Clin Oncol. 31:2205–2218. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gupta MK and Qin RY: Mechanism and its
regulation of tumor-induced angiogenesis. World J Gastroenterol.
9:1144–1155. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Copland IB: Mesenchymal stromal cells for
cardiovascular disease. J Cardiovasc Dis Res. 2:3–13. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang D, Fan GC, Zhou X, Zhao T, Pasha Z,
Xu M, Zhu Y, Ashraf M and Wang Y: Over-expression of CXCR4 on
mesenchymal stem cells augments myoangiogenesis in the infarcted
myocardium. J Mol Cell Cardiol. 44:281–292. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wen Z, Huang W, Feng Y, Cai W, Wang Y,
Wang X, Liang J, Wani M, Chen J, Zhu P, et al: MicroRNA-377
regulates mesenchymal stem cell-induced angiogenesis in ischemic
hearts by targeting VEGF. PLoS One. 9:e1046662014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hua J, He ZG, Qian DH, Lin SP, Gong J,
Meng HB, Yang TS, Sun W, Xu B, Zhou B, et al: Angiopoietin-1
gene-modified human mesenchymal stem cells promote angiogenesis and
reduce acute pancreatitis in rats. Int J Clin Exp Pathol.
7:3580–3595. 2014.PubMed/NCBI
|
|
27
|
Park IS, Chung PS and Ahn JC: Enhanced
angiogenic effect of adipose-derived stromal cell spheroid with
low-level light therapy in hind limb ischemia mice. Biomaterials.
35:9280–9289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jazayeri M, Allameh A, Soleimani M,
Jazayeri SH, Piryaei A and Kazemnejad S: Molecular and
ultrastructural characterization of endothelial cells
differentiated from human bone marrow mesenchymal stem cells. Cell
Biol Int. 32:1183–1192. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Aguirre A, Planell JA and Engel E:
Dynamics of bone marrow-derived endothelial progenitor
cell/mesenchymal stem cell interaction in co-culture and its
implications in angiogenesis. Biochem Biophys Res Commun.
400:284–291. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Malinowski M, Pietraszek K, Perreau C,
Boguslawski M, Decot V, Stoltz JF, Vallar L, Niewiarowska J,
Cierniewski C, Maquart FX, et al: Effect of lumican on the
migration of human mesenchymal stem cells and endothelial
progenitor cells: Involvement of matrix metalloproteinase-14. PLoS
One. 7:e507092012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
García-Alfonso P, Grande E, Polo E, Afonso
R, Reina JJ, Jorge M, Campos JM, Martínez V, Angeles C and Montagut
C: The role of antiangiogenic agents in the treatment of patients
with advanced colorectal cancer according to K-RAS status.
Angiogenesis. 17:805–821. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang Z, Wang M, Yang F, Nie W, Chen F, Xu
J and Guan X: Multitargeted antiangiogenic tyrosine kinase
inhibitors combined to chemotherapy in metastatic breast cancer: A
systematic review and meta-analysis. Eur J Clin Pharmacol.
70:531–538. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ohara T, Noma K, Urano S, Watanabe S,
Nishitani S, Tomono Y, Kimura F, Kagawa S, Shirakawa Y and Fujiwara
T: A novel synergistic effect of iron depletion on antiangiogenic
cancer therapy. Int J Cancer. 132:2705–2713. 2013. View Article : Google Scholar
|
|
34
|
Xin H, Kanehira M, Mizuguchi H, Hayakawa
T, Kikuchi T, Nukiwa T and Saijo Y: Targeted delivery of CX3CL1 to
multiple lung tumors by mesenchymal stem cells. Stem Cells.
25:1618–1626. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kolluri KK, Laurent GJ and Janes SM:
Mesenchymal stem cells as vectors for lung cancer therapy.
Respiration. 85:443–451. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kanehira M, Xin H, Hoshino K, Maemondo M,
Mizuguchi H, Hayakawa T, Matsumoto K, Nakamura T, Nukiwa T and
Saijo Y: Targeted delivery of NK4 to multiple lung tumors by bone
marrow-derived mesenchymal stem cells. Cancer Gene Ther.
14:894–903. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhang MH, Hu YD, Xu Y, Xiao Y, Luo Y, Song
ZC and Zhou J: Human mesenchymal stem cells enhance autophagy of
lung carcinoma cells against apoptosis during serum deprivation.
Int J Oncol. 42:1390–1398. 2013.PubMed/NCBI
|
|
38
|
Jung Y, Kim JK, Shiozawa Y, Wang J, Mishra
A, Joseph J, Berry JE, McGee S, Lee E, Sun H, et al: Recruitment of
mesen-chymal stem cells into prostate tumours promotes metastasis.
Nat Commun. 4:17952013. View Article : Google Scholar
|
|
39
|
Lee MJ, Heo SC, Shin SH, Kwon YW, Do EK,
Suh DS, Yoon MS and Kim JH: Oncostatin M promotes mesenchymal stem
cell-stimulated tumor growth through a paracrine mechanism
involving periostin and TGFBI. Int J Biochem Cell Biol.
45:1869–1877. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chen Q, Cheng P, Song N, Yin T, He H, Yang
L, Chen X and Wei Y: Antitumor activity of placenta-derived
mesenchymal stem cells producing pigment epithelium-derived factor
in a mouse melanoma model. Oncol Lett. 4:413–418. 2012.
|
|
41
|
Li Y, Zhao Y, Cheng Z, Zhan J, Sun X, Qian
H, Zhu W and Xu W: Mesenchymal stem cell-like cells from children
foreskin inhibit the growth of SGC-7901 gastric cancer cells. Exp
Mol Pathol. 94:430–437. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Katsuda T, Kosaka N, Takeshita F and
Ochiya T: The therapeutic potential of mesenchymal stem
cell-derived extracellular vesicles. Proteomics. 13:1637–1653.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mader EK, Butler G, Dowdy SC, Mariani A,
Knutson KL, Federspiel MJ, Russell SJ, Galanis E, Dietz AB and Peng
KW: Optimizing patient derived mesenchymal stem cells as virus
carriers for a phase I clinical trial in ovarian cancer. J Transl
Med. 11:202013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Martinez-Quintanilla J, Bhere D, Heidari
P, He D, Mahmood U and Shah K: Therapeutic efficacy and fate of
bimodal engineered stem cells in malignant brain tumors. Stem
Cells. 31:1706–1714. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Cavarretta IT, Altanerova V, Matuskova M,
Kucerova L, Culig Z and Altaner C: Adipose tissue-derived
mesenchymal stem cells expressing prodrug-converting enzyme inhibit
human prostate tumor growth. Mol Ther. 18:223–231. 2010. View Article : Google Scholar :
|
|
46
|
Kucerova L, Altanerova V, Matuskova M,
Tyciakova S and Altaner C: Adipose tissue-derived human mesenchymal
stem cells mediated prodrug cancer gene therapy. Cancer Res.
67:6304–6313. 2007. View Article : Google Scholar : PubMed/NCBI
|