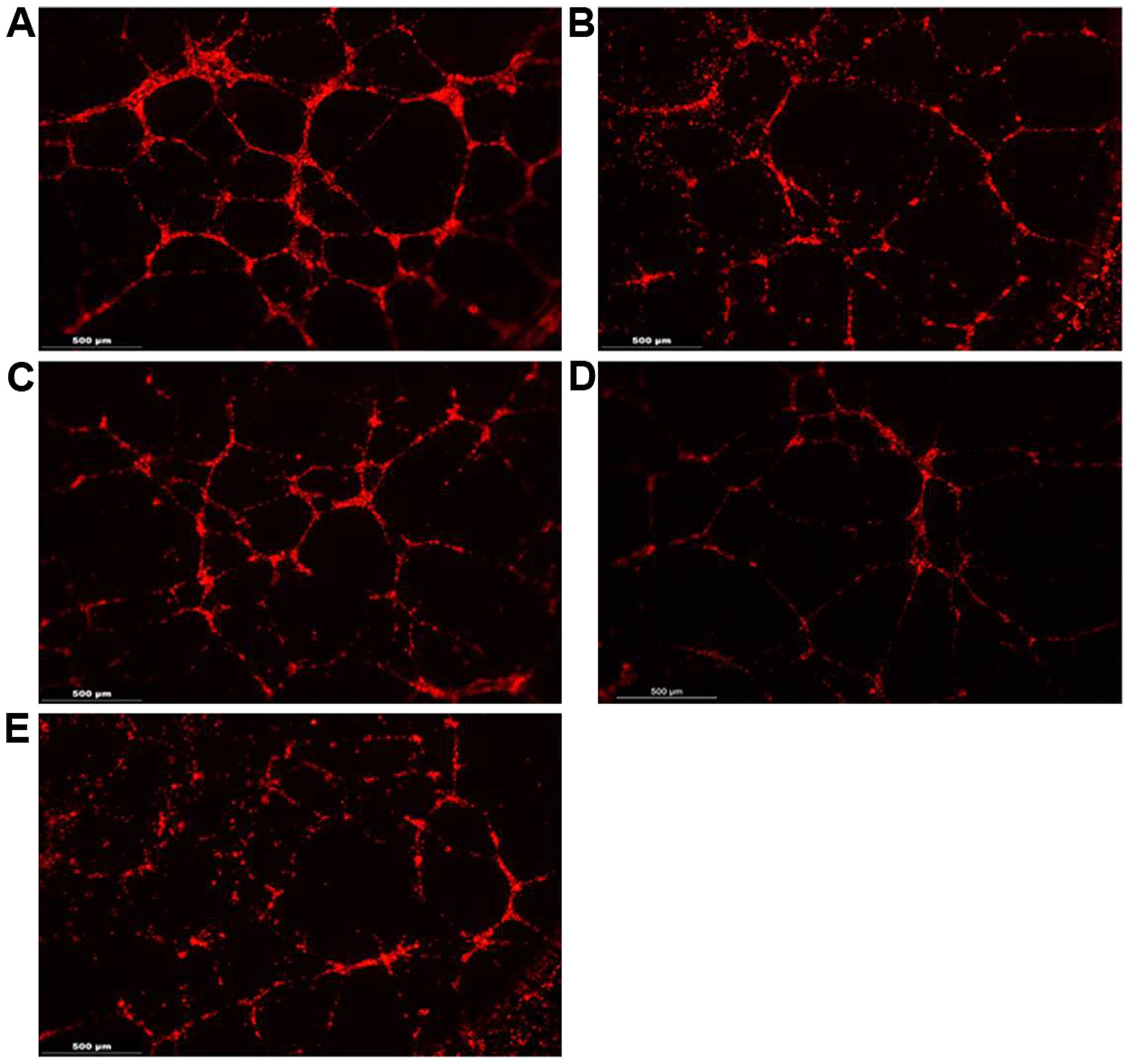
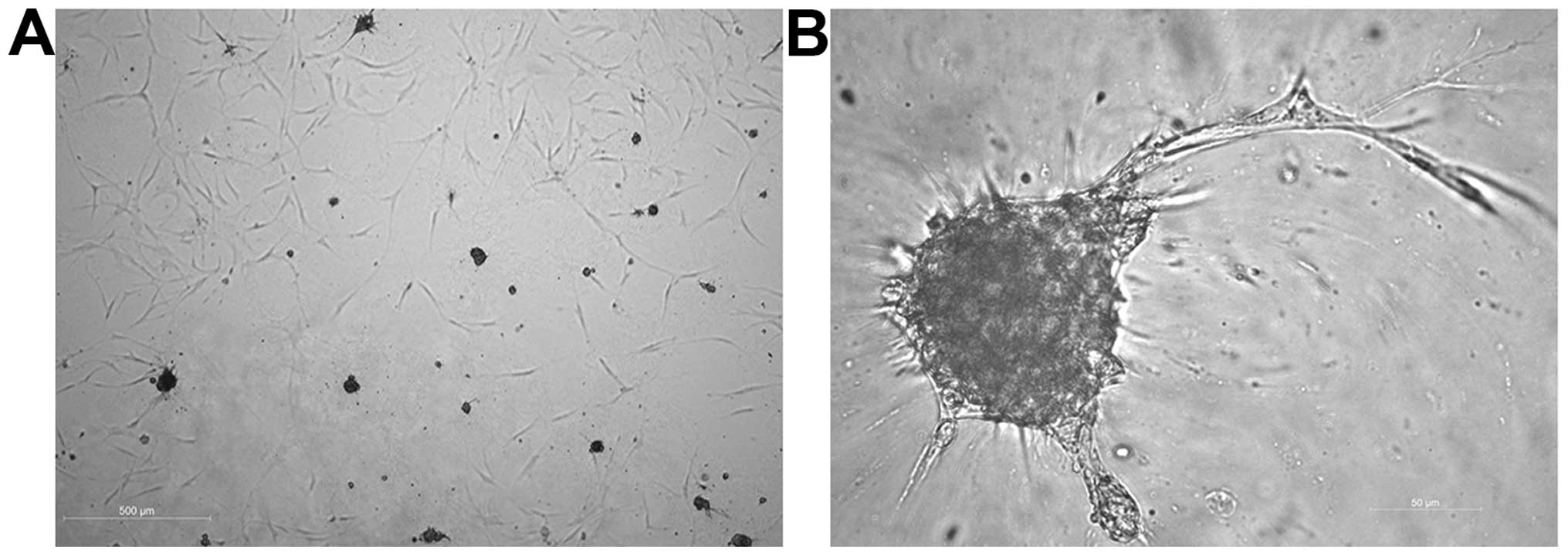
|
1
|
Folkman J: What is the evidence that
tumors are angiogenesis dependent? J Natl Cancer Inst. 82:4–6.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bergers G and Benjamin LE: Tumorigenesis
and the angiogenic switch. Nat Rev Cancer. 3:401–410. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dvorak HF: Tumors: Wounds that do not
heal. Similarities between tumor stroma generation and wound
healing. N Engl J Med. 315:1650–1659. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kinnaird T, Stabile E, Burnett MS, Shou M,
Lee CW, Barr S, Fuchs S and Epstein SE: Local delivery of
marrow-derived stromal cells augments collateral perfusion through
paracrine mechanisms. Circulation. 109:1543–1549. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hung SC, Pochampally RR, Chen SC, Hsu SC
and Prockop DJ: Angiogenic effects of human multipotent stromal
cell conditioned medium activate the PI3K-Akt pathway in hypoxic
endothelial cells to inhibit apoptosis, increase survival, and
stimulate angiogenesis. Stem Cells. 25:2363–2370. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kanazawa T, Nishino H, Hasegawa M, Ohta Y,
Iino Y, Ichimura K and Noda Y: Interleukin-6 directly influences
proliferation and invasion potential of head and neck cancer cells.
Eur Arch Otorhinolaryngol. 264:815–821. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kishimoto T: The biology of interleukin-6.
Blood. 74:1–10. 1989.PubMed/NCBI
|
|
8
|
Caplan AI: Mesenchymal stem cells. J
Orthop Res. 9:641–650. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Myers JT, Barkauskas DS and Huang AY:
Dynamic imaging of marrow-resident granulocytes interacting with
human mesenchymal stem cells upon systemic lipopolysaccharide
challenge. Stem Cells Int. 2013:6568392013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Karnoub AE, Dash AB, Vo AP, Sullivan A,
Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R and Weinberg
RA: Mesenchymal stem cells within tumour stroma promote breast
cancer metastasis. Nature. 449:557–563. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin WR, Brittan M and Alison MR: The role
of bone marrow-derived cells in fibrosis. Cells Tissues Organs.
188:178–188. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rhodes LV, Muir SE, Elliott S, Guillot LM,
Antoon JW, Penfornis P, Tilghman SL, Salvo VA, Fonseca JP, Lacey
MR, et al: Adult human mesenchymal stem cells enhance breast
tumori-genesis and promote hormone independence. Breast Cancer Res
Treat. 121:293–300. 2010. View Article : Google Scholar
|
|
13
|
Khakoo AY, Pati S, Anderson SA, Reid W,
Elshal MF, Rovira II, Nguyen AT, Malide D, Combs CA, Hall G, et al:
Human mesen-chymal stem cells exert potent antitumorigenic effects
in a model of Kaposi's sarcoma. J Exp Med. 203:1235–1247. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhu Y, Sun Z, Han Q, Liao L, Wang J, Bian
C, Li J, Yan X, Liu Y, Shao C, et al: Human mesenchymal stem cells
inhibit cancer cell proliferation by secreting DKK-1. Leukemia.
23:925–933. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Scherzed A, Hackenberg S, Froelich K, Rak
K, Technau A, Radeloff A, Nöth U, Koehler C, Hagen R and
Kleinsasser N: Effects of salinomycin on human bone marrow-derived
mesenchymal stem cells in vitro. Toxicol Lett. 218:207–214. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Scherzed A, Hackenberg S, Radeloff A,
Froelich K, Rak K, Hagen R and Kleinsasser N: Human mesenchymal
stem cells promote cancer motility and cytokine secretion in vitro.
Cells Tissues Organs. 198:327–337. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rangan SR: A new human cell line (FaDu)
from a hypopharyngeal carcinoma. Cancer. 29:117–121. 1972.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zetter BR: Angiogenesis and tumor
metastasis. Annu Rev Med. 49:407–424. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Grantab RH and Tannock IF: Penetration of
anticancer drugs through tumour tissue as a function of cellular
packing density and interstitial fluid pressure and its
modification by bortezomib. BMC Cancer. 12:2142012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jain RK: Normalizing tumor
microenvironment to treat cancer: Bench to bedside to biomarkers. J
Clin Oncol. 31:2205–2218. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gupta MK and Qin RY: Mechanism and its
regulation of tumor-induced angiogenesis. World J Gastroenterol.
9:1144–1155. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Copland IB: Mesenchymal stromal cells for
cardiovascular disease. J Cardiovasc Dis Res. 2:3–13. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang D, Fan GC, Zhou X, Zhao T, Pasha Z,
Xu M, Zhu Y, Ashraf M and Wang Y: Over-expression of CXCR4 on
mesenchymal stem cells augments myoangiogenesis in the infarcted
myocardium. J Mol Cell Cardiol. 44:281–292. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wen Z, Huang W, Feng Y, Cai W, Wang Y,
Wang X, Liang J, Wani M, Chen J, Zhu P, et al: MicroRNA-377
regulates mesenchymal stem cell-induced angiogenesis in ischemic
hearts by targeting VEGF. PLoS One. 9:e1046662014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hua J, He ZG, Qian DH, Lin SP, Gong J,
Meng HB, Yang TS, Sun W, Xu B, Zhou B, et al: Angiopoietin-1
gene-modified human mesenchymal stem cells promote angiogenesis and
reduce acute pancreatitis in rats. Int J Clin Exp Pathol.
7:3580–3595. 2014.PubMed/NCBI
|
|
27
|
Park IS, Chung PS and Ahn JC: Enhanced
angiogenic effect of adipose-derived stromal cell spheroid with
low-level light therapy in hind limb ischemia mice. Biomaterials.
35:9280–9289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jazayeri M, Allameh A, Soleimani M,
Jazayeri SH, Piryaei A and Kazemnejad S: Molecular and
ultrastructural characterization of endothelial cells
differentiated from human bone marrow mesenchymal stem cells. Cell
Biol Int. 32:1183–1192. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Aguirre A, Planell JA and Engel E:
Dynamics of bone marrow-derived endothelial progenitor
cell/mesenchymal stem cell interaction in co-culture and its
implications in angiogenesis. Biochem Biophys Res Commun.
400:284–291. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Malinowski M, Pietraszek K, Perreau C,
Boguslawski M, Decot V, Stoltz JF, Vallar L, Niewiarowska J,
Cierniewski C, Maquart FX, et al: Effect of lumican on the
migration of human mesenchymal stem cells and endothelial
progenitor cells: Involvement of matrix metalloproteinase-14. PLoS
One. 7:e507092012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
García-Alfonso P, Grande E, Polo E, Afonso
R, Reina JJ, Jorge M, Campos JM, Martínez V, Angeles C and Montagut
C: The role of antiangiogenic agents in the treatment of patients
with advanced colorectal cancer according to K-RAS status.
Angiogenesis. 17:805–821. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang Z, Wang M, Yang F, Nie W, Chen F, Xu
J and Guan X: Multitargeted antiangiogenic tyrosine kinase
inhibitors combined to chemotherapy in metastatic breast cancer: A
systematic review and meta-analysis. Eur J Clin Pharmacol.
70:531–538. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ohara T, Noma K, Urano S, Watanabe S,
Nishitani S, Tomono Y, Kimura F, Kagawa S, Shirakawa Y and Fujiwara
T: A novel synergistic effect of iron depletion on antiangiogenic
cancer therapy. Int J Cancer. 132:2705–2713. 2013. View Article : Google Scholar
|
|
34
|
Xin H, Kanehira M, Mizuguchi H, Hayakawa
T, Kikuchi T, Nukiwa T and Saijo Y: Targeted delivery of CX3CL1 to
multiple lung tumors by mesenchymal stem cells. Stem Cells.
25:1618–1626. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kolluri KK, Laurent GJ and Janes SM:
Mesenchymal stem cells as vectors for lung cancer therapy.
Respiration. 85:443–451. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kanehira M, Xin H, Hoshino K, Maemondo M,
Mizuguchi H, Hayakawa T, Matsumoto K, Nakamura T, Nukiwa T and
Saijo Y: Targeted delivery of NK4 to multiple lung tumors by bone
marrow-derived mesenchymal stem cells. Cancer Gene Ther.
14:894–903. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhang MH, Hu YD, Xu Y, Xiao Y, Luo Y, Song
ZC and Zhou J: Human mesenchymal stem cells enhance autophagy of
lung carcinoma cells against apoptosis during serum deprivation.
Int J Oncol. 42:1390–1398. 2013.PubMed/NCBI
|
|
38
|
Jung Y, Kim JK, Shiozawa Y, Wang J, Mishra
A, Joseph J, Berry JE, McGee S, Lee E, Sun H, et al: Recruitment of
mesen-chymal stem cells into prostate tumours promotes metastasis.
Nat Commun. 4:17952013. View Article : Google Scholar
|
|
39
|
Lee MJ, Heo SC, Shin SH, Kwon YW, Do EK,
Suh DS, Yoon MS and Kim JH: Oncostatin M promotes mesenchymal stem
cell-stimulated tumor growth through a paracrine mechanism
involving periostin and TGFBI. Int J Biochem Cell Biol.
45:1869–1877. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chen Q, Cheng P, Song N, Yin T, He H, Yang
L, Chen X and Wei Y: Antitumor activity of placenta-derived
mesenchymal stem cells producing pigment epithelium-derived factor
in a mouse melanoma model. Oncol Lett. 4:413–418. 2012.
|
|
41
|
Li Y, Zhao Y, Cheng Z, Zhan J, Sun X, Qian
H, Zhu W and Xu W: Mesenchymal stem cell-like cells from children
foreskin inhibit the growth of SGC-7901 gastric cancer cells. Exp
Mol Pathol. 94:430–437. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Katsuda T, Kosaka N, Takeshita F and
Ochiya T: The therapeutic potential of mesenchymal stem
cell-derived extracellular vesicles. Proteomics. 13:1637–1653.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mader EK, Butler G, Dowdy SC, Mariani A,
Knutson KL, Federspiel MJ, Russell SJ, Galanis E, Dietz AB and Peng
KW: Optimizing patient derived mesenchymal stem cells as virus
carriers for a phase I clinical trial in ovarian cancer. J Transl
Med. 11:202013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Martinez-Quintanilla J, Bhere D, Heidari
P, He D, Mahmood U and Shah K: Therapeutic efficacy and fate of
bimodal engineered stem cells in malignant brain tumors. Stem
Cells. 31:1706–1714. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Cavarretta IT, Altanerova V, Matuskova M,
Kucerova L, Culig Z and Altaner C: Adipose tissue-derived
mesenchymal stem cells expressing prodrug-converting enzyme inhibit
human prostate tumor growth. Mol Ther. 18:223–231. 2010. View Article : Google Scholar :
|
|
46
|
Kucerova L, Altanerova V, Matuskova M,
Tyciakova S and Altaner C: Adipose tissue-derived human mesenchymal
stem cells mediated prodrug cancer gene therapy. Cancer Res.
67:6304–6313. 2007. View Article : Google Scholar : PubMed/NCBI
|