Introduction
Breast cancer is one of the most common cancers
worldwide and tends to metastasize to bone, resulting in osteolysis
and skeletal-related events (pain and fracture) in patients. The
mechanisms underlying this metastasis are complex and involve both
particular characteristics of the breast cancer cells and the bone
matrix (soil and seed concept). During metastasis, breast cancer
cells possess certain properties that enable them to grow in bone,
and the bone matrix provides a suitable microenvironment which
facilitates the growth of breast cancer cells. The bone
microenvironment facilitates dynamic bone resorption and bone
formation which maintains the balance of bone mass. Bone remodeling
consists of both resorption by osteoclasts (1–5) and
new bone formation by osteoblasts (3,5). The
two processes of remodeling are coupled temporally and spatially
(1,3,5,6) and
are controlled by an interplay between locally and systemically
derived signals, such as mechanical strain, growth factors,
hormones and other molecules (1,5,6).
Breast cancer cells disrupt this normal physiological process and
generate factors that directly or indirectly induce the formation
of osteoclasts in the bone microenvironment, which in turn releases
growth factors from the bone matrix (e.g., TGF-β) that stimulate
tumor growth and further osteolysis. This reciprocal interaction
between breast cancer cells and the bone microenvironment results
in a 'vicious cycle' that increases both bone destruction and the
tumor burden (7). The current
standard of care for breast cancer patients (i.e., anti-resorptive
therapy) with bone loss due to osteolytic bone metastases has some
positive effects, but is not curative with regard to tumor burden
(8,9).
Depression is a serious complication in women with
breast cancer which is often underestimated with a prevalence
varying between 10 and 25%. Depressed individuals display lower
bone mineral density (BMD) and higher bone resorption than
non-depressed subjects and the underlying mechanism is yet to be
elucidated. Research has shown that viscosity of platelet membrane
may be a bridge to psychiatric illness (10). Arachidonic acid increases with a
decrease in membrane viscosity (11–13),
which impairs the capacity of platelet and neuronal serotonin
receptors to capture serotonin 5-hydroxytryptamine (5-HT) (14). Serotonin does not cross the
blood-brain barrier which creates free serotonin in the circulation
and the excess of free serotonin (in depression) can induce
osteoporosis (15). The critical
step in 5-HT biosynthesis is catalyzed by the rate-limiting enzyme
Tph (Tph1 and Tph2). Tph1 is expressed mainly in the gut and pineal
gland (16), whereas Tph2 is
expressed only in neurons (17).
Since most circulating serotonin arises from synthesis in the
duodenum by specialized neuroendocrine enterochromaffin cells,
several studies have elucidated the effect of gut-derived serotonin
on the connection between the brain, bone and intestine (18–20).
Thus, in depression, a defect in the uptake of serotonin in
platelets and neurons increases circulating serotonin, which
influences bone mass and induces osteoporosis.
A recent study found that four 5-HT receptor (5-HTR)
subtypes (1A, 1B, 2B and 4) were differentially expressed in human
breast cancer (21). In human
breast cancer cells, 5-HT induced both parathyroid hormone-related
peptide (PTHrP) and the metastasis-associated transcription factor
Runx2/Cbfa1 (22). The mammary
epithelium possesses a local serotonin signaling system which
drives PTHrP expression during lactation and in breast cancer cells
which may be a passage of breast communication to bone through
serotonergic control of PTHrP (23). Evidence has shown that PTHrP plays
an important pathogenetic role in the establishment of osteolytic
bone lesions in breast cancer. Inhibition of PTHrP can reduce the
development of destructive bone lesions as well as the growth of
tumor cells in bone (24,25). This finding that serotonin regulates
the behavior of breast cancer cells and bone remodeling balance led
us to investigate the possible involvement of depression in the
control of breast cancer cell metastasis to the bone.
In the present study, we showed that depression
promotes the bone metastasis of breast cancer cells through
serotonin signaling. We confirmed that gut-derived serotonin
inhibits osteoblast differentiation and activates osteoclast
differentiation by stimulating the expression of RUNX2 in breast
cancer cells. Thus, activation of RUNX2 in breast cancer cells by
circulating serotonin appears to dissociate coupling between
osteoblasts and osteoclasts, suggesting that suppression of
gut-derived serotonin may decrease the rate of breast cancer bone
metastasis induced by depression.
Materials and methods
Intracardiac bone metastasis model
All experimental procedures were approved by the
Institution of Animal Care and Use Committee (IACUC) at Chongqing
Medical University. All the animals were handled in accordance to
the guidelines of IACUC animal usage standards. Female BALB/c nude
mice (4–6 weeks old) were purchased from the Laboratory Animal
Center (Chongqing Medical University) and quarantined within our
facility for 2 weeks prior to the experimentation. Nude mice were
group housed in plastic cages (n=5/cage) under standard laboratory
conditions with a 12-h dark/12-h light cycle, a constant
temperature of 20°C and humidity of 48%. Nude mice were housed in
sterile conditions and fed autoclaved standard chow.
MDA-MB-231 cells used in the present study were
gifted from the Key Laboratory for Biochemistry and Molecular
Pharmacology of Chongqing Medical University. MDA-MB-231 cells were
cultured in 10% fetal bovine serum (FBS) and Dulbecco's modified
Eagle's medium (DMEM) with 1% penicillin/streptomycin. Cells were
trypsinized at 70–90% confluency, rinsed and re-suspended in cold
PBS at 107 cells/ml. Athymic nude female mice aged 4–6
weeks were anesthetized and injected in the left cardiac ventricle
with 100 µl of the cell suspension (106
MDA-MB-231 cells). Bone metastasis was assessed weekly for 4 weeks
with Faxitron radiographic imaging (Faxitron MX-20 at 35 kV for 12
sec of exposure).
Chronic mild stress (CMS) and sucrose
consumption test
To establish the model of depression, the animals
were divided into two matched groups and placed in separate rooms.
One group was exposed to an initial 2 weeks of chronic mild
stressors and the other was left undisturbed. The stress protocol
consisted of one period of intermittent illumination, stroboscopic
light, grouping, food or water deprivation; two periods of soiled
cage and no stress; three periods of 45° box tilting. All the
stressors lasted from 10 to 14 h. During the 2 weeks of stress
stimulation, a sucrose consumption test was performed (three times
in the first week and two times in the last week). After the
initial 2 weeks of exposure to stress, the unchallenged and
stressed groups were subjected to the next experimental procedure
(intracardiac injection). Stress was continued for 4 weeks during
the entire period of treatment.
Radiographic analysis
Faxitron was used to quantify the bilateral
osteolytic lesions in the humeri, femora and tibiae at end
time-point. Presence of tumors within the bones was confirmed with
histology, and the lesion area was calculated as the average of
total osteolytic area per mouse. Data were double-blinded and
calculated by at least two independent researchers.
Microcomputed tomography (µCT)
analysis
Tibiae from each animal were dissected, cleaned,
fixed for 48 h in 10% formalin/PBS, transferred to 70% EtOH, and
then loaded into scanning tubes and imaged (vivaCT 40; Scanco
Medical). The scans were integrated into three-dimensional (3-D)
voxel images and a Gaussian filter (σ=0.8, support=1) was used to
reduce signal noise and a threshold of 300 was applied to all
analyzed scans. Scans were carried out at 12-mm resolution (E=70
kVp, I=112 µA). Four hundred transverse slices of the
proximal tibia were taken from the growth plate and extended
distally except from growth plate, and automated contouring using
voxel counting and sphere-filling distance transformation indices
was performed.
Histology
Bones were decalcified in 20% EDTA pH 7.4 at room
temperature for 3–4 days, and then dehydrated and embedded in
paraffin. Hematoxylin and eosin (H&E) staining was used to
quantify tumor burden and tumor number in 5-µm paraffin
sections. Tumor number and tumor burden were assessed in the long
bones presenting with tumors using the Bioquant system imaging
software (Bioquant Image Analysis Co., Nashville, TN, USA) as total
tumor area per combined area of the six long bones averaged from
three sections per bone.
Analysis of gene expression
Total RNA was isolated with TRIzol reagent
(Invitrogen Life Technologies, Carlsbad, CA, USA) and used to
generate cDNA templates by reverse transcription (RT) reaction with
hexamer and Superscript II RT. The first strand cDNA products were
further diluted 5- to 10-fold and used as templates for polymerase
chain reaction (PCR). All samples were normalized to the expression
level of GAPDH. The primers used in this study are shown in
Table I.
 | Table IGene-specific primers for human and
mouse. |
Table I
Gene-specific primers for human and
mouse.
| Genes | Primers |
|---|
| BSP (mouse) |
5′-AAGAGGAAGAATGAGAACGA-3′ |
|
5′-GCTTCTTCTCCGTTGTCTCC-3′ |
| Osteocalcin
(mouse) |
5′-CTGACCTCACAGATGCCAAG-3′ |
|
5′-GTAGCGCCGTGAGTCTGTTC-3′ |
| Collagen 1α1 |
5′-AGTTTCAGGTCTCTGCAGGT-3′ |
| (mouse) |
5′-AACTGGCAAGCAAGGAGACA-3′ |
| RANKL (mouse) |
5′-TGTACTTTCGAGCGCAGATG-3′ |
|
5′-CCACAATGTGTTGCAGTTCC-3′ |
| Cathepsin K |
5′-AAGTGGTAAGATGACGGGAC-3′ |
| (mouse) |
5′-TCTTCACAATGCCTCCGTTC-3′ |
| Calcitonin
receptor |
5′-CGGACTTTGACACAGCAGAA-3′ |
| (mouse) |
5′-GTCACCCTCTGGCAGCTAAG-3′ |
| GAPDH (mouse) |
5′-ACCCAGAAGACTGTGGATGG-3′ |
|
5′-CACATTGGGGGTAGGAACAC-3′ |
| PTHrP (human) |
5′-GGAAGCAACCAGCCCACCAG-3′ |
|
5′-ACCGCGTAGCTCAGCAGGAA-3′ |
| RUNX2 (human) |
5′-TTTGCACTGGGTCATGTGTT-3′ |
|
5′-TGGCTGCATTGAAAAGACTG-3′ |
| GAPDH (human) |
5′-CAACGAATTTGGCTACAGCA-3′ |
|
5′-AGGGGAGATTCAGTGTGGTG-3′ |
Western blotting
Western blotting was performed to examine the
protein expression levels in vitro. Protein concentration
was determined with a protein quantitative analysis kit, and 300 mg
of protein was run on a 10% gradient sodium dodecyl sulfate
polyacrylamide gel electrophoresis (SDS-PAGE) gel and blotted onto
PVDF membranes. The membranes were blocked with 5% BSA blocking
buffer at room temperature for 1 h, and then probed with the
primary antibody at room temperature for 1 h. After washing with
Tris-buffered saline with Tween-20 (TBST) three times, the PVDF
membranes were incubated with an appropriate secondary antibody
(antibodies diluted 1:5,000) at room temperature for 1 h, and then
the membranes were washed three times in TBST. Immune complexes
were detected using the chemiluminescent method, and immunoreactive
bands were quantified using an imaging system (Thermo Fisher
Scientific, Rockford, IL, USA).
ELISA
Mice were exposed to isoflurane for 30 sec, and
blood was collected by cardiac puncture to measure the serum
serotonin levels. Serum collection from all mice was conducted
between 9:00 a.m. and 12:00 p.m. Blood was centrifuged at maximum
speed at 4°C for 10 min. The serum supernatant was snap frozen and
stored at −80°C. Serum serotonin levels were quantified by ELISA
(serotonin kit; Sigma-Aldrich, St. Louis, MO, USA) according to the
manufacturer's instructions.
Co-culture studies
Boyden chambers (1-µm inserts; BD
Biosciences, Bedford, MA, USA) were used to test the influence of
MDA-MB-231 cells on osteoclasts. Bone marrow cells were collected
from the femur and tibiae of BALB/c female mice (4–6 weeks old).
Cells (5,000/well) were plated for 3 days and transferred to the
bottom of the chamber with RANKL (5 ng/ml). MDA-MB-231 cells
(1×105 cells/well) with or without serotonin (100
µm; Sigma-Aldrich) were plated in the upper layer for 7
days. Osteoclast formation was monitored by TRAP staining analysis.
For osteoblast co-culture studies, MC3T3 cells (5,000/well) were
harvested with MDA-MB-231 cells for 3 days. After the addition of
serotonin (100 µm) with 50 mg/ml ascorbate and 10 mM
β-glycerol phosphate (osteogenic media), the two cell lines were
cultured for 14 days. MC3T3 cells were fixed in 2% PFA and stained
for AP activity and Alizarin Red staining (ALZ)
(Sigma-Aldrich).
shRNA knockdown
MDA-MB-231 cells were transfected with shRNA
silencing vectors against human RUNX-2 or a scramble control as
previously described (47). After
transfection, RT-PCR and western blotting were performed to
evaluate RUNX-2-knockdown efficiency.
Statistical analysis
All data are presented as means ± SEM. Statistical
analyses were performed using one-way ANOVA for multiple
comparisons and the two-tailed Student's t-tests. For all analyses,
P<0.05 was considered to indicate a statistically significant
difference and each assay condition was performed in triplicate.
The results were repeated in at least three independent
experiments.
Results
Depression promotes breast cancer cell
metastasis to the bone and increases osteolytic lesions through
gut-derived serotonin
Several processes are involved in the metastasis of
breast cancer cells to bone which include cell release from the
primary tumor into the bloodstream, localization and proliferation
in the bone microenvironment. We used an established model of bone
metastasis in which MDA-MB-231 human breast cancer cells were
inoculated into nude mice by intracardiac injection to elucidate
the effect of depression on bone metastasis. CMS was chosen as a
model of depression in mice (26–28).
In the experimental protocol, nude mice were exposed to an initial
2 weeks of CMS and sucrose consumption was tested before and after
intracardiac injection. After 2 weeks of initial exposure to CMS,
the intake of sucrose solution was significantly diminished in the
stress group compared to the unchallenged animals, indicating a
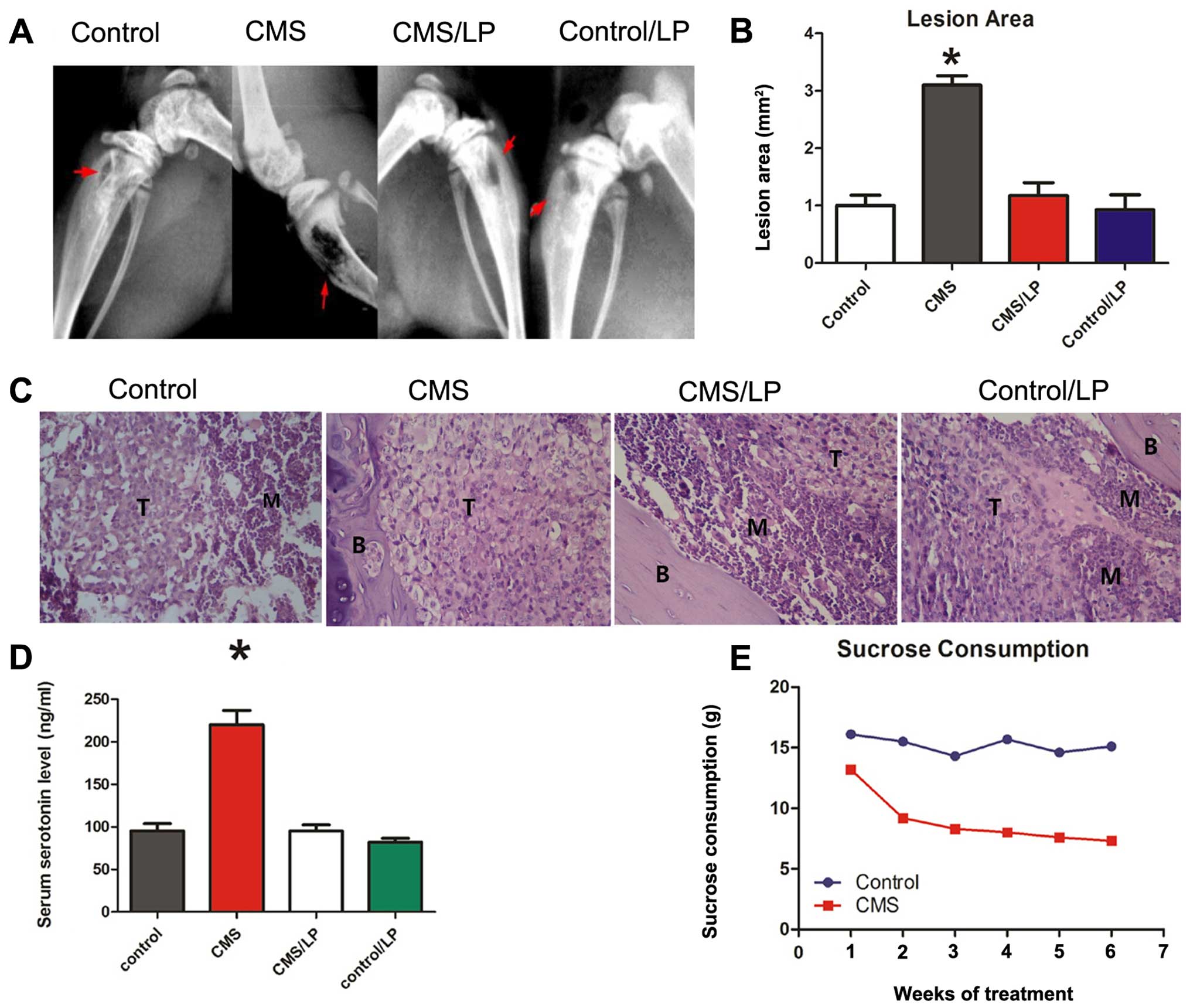
stress-induced decrease in sensitivity to reward (Fig. 1E). After inoculation of breast
cancer cells combined with 4 weeks of continuous exposure to CMS,
the area of osteolytic lesions increased significantly, measured by
Faxitron, compared to the no CMS group (Fig. 1A and B). CMS also significantly
increased the number of bone tumors, as measured by
histomorphometry (Fig. 1C).
LP533401 (Dalton Chemical Laboratories Inc., Toronto, ON, Canada),
a small-molecule inhibitor of Tph-1 which is the initial enzyme in
gut-derived serotonin (GDS) biosynthesis is currently being tested
for the treatment of irritable bowel syndrome with no overt
deleterious side effects (29).
Oral administration of LP533401 (250 mg/kg/day) for a continuous 6
weeks significantly decreased osteolytic lesions in the CMS group
compared to the PBS control (Fig.
1A–C). After 6 weeks of CMS treatment, the level of serum
serotonin was significantly increased, which was inhibited by
LP533401 (Fig. 1D).
These findings indicate that the distress induced by
CMS in mice increased the incidence of bone metastasis by breast
cancer cells, and that blockade of GDS by LP533401 perturbed the
bone metastasis, which implicates GDS in the process of breast
cancer cell metastasis to bone.
Circulating serotonin increases
osteolytic lesions by inhibition of the proliferation and
differentiation of osteoblasts
A major mechanism of bone metastasis to the bone of
breast cancer cells is decoupled association of bone formation and
bone resorption, resulting in bone destruction. To test whether
depression has an effect on bone formation in the process of breast
cancer bone metastasis, we evaluated in vivo and in
vitro the influence of circulating serotonin on osteoblast
differentiation and proliferation. The level of circulating
serotonin increased significantly in the CMS treatment group
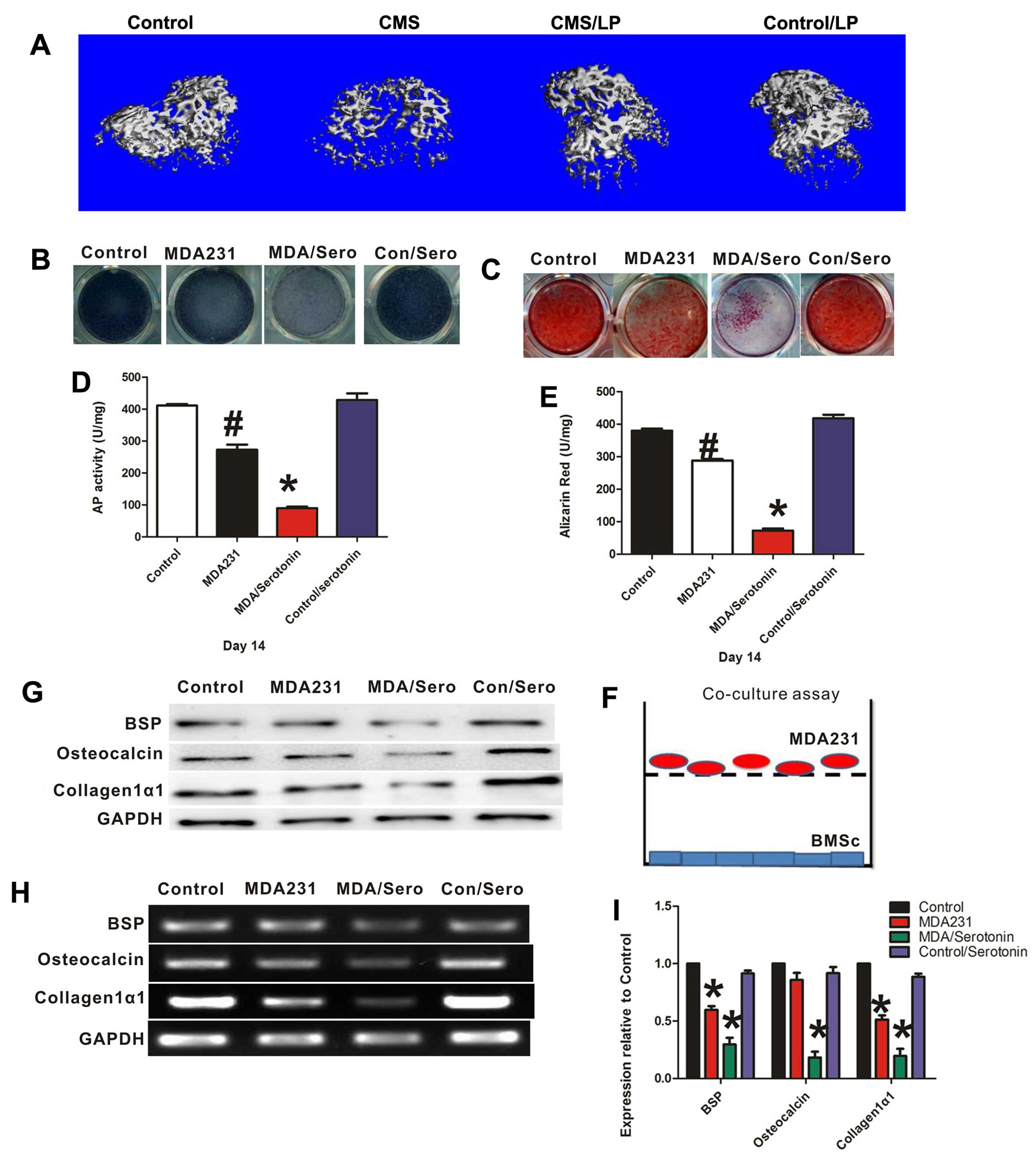
(Fig. 1D), whereas the bone volume
and bone formation rate decreased compared to the no CMS group
(Fig. 2A and Table II). These data showed that
osteoblast proliferation was suppressed in the CMS treatment group.
However, oral administration of LP53340 rescued the low bone
formation by inhibiting the biosynthesis of GDS. Thus, CMS
interferes with bone formation and facilitates the bone metastasis
of breast cancer cells through inhibition of osteoblast
proliferation.
 | Table IIµCT analysis of long bones
from the control, CMS, CMS/LP and control/LP mice. |
Table II
µCT analysis of long bones
from the control, CMS, CMS/LP and control/LP mice.
| Control | CMS | CMS/LP | Control/LP |
|---|
| (BV/TV) (%) | 5.78±1.6 | 2.95±1.2a | 4.92±1.8b | 5.34±1.5 |
| Conn.D | 26.82±8.9 | 18.3±6.4a | 25.43±7.8b | 26.78±8.4 |
| Tb.Nb | 2.01±0.6 | 1.53±0.3 | 1.87±0.5 | 2.12±0.5 |
| Tb.Th | 0.046±0.003 | 0.036±0.004 | 0.42±0.004 | 0.48±0.002 |
| Tb.Sp | 0.521±0.12 | 0.652±0.21 | 0.611±0.17 | 0.533±0.16 |
To demonstrate the role of serotonin and breast
cancer cells in osteoblast differentiation, Transwell assays were
performed. When MDA-MB-231 cells were plated in the Transwell
filter with MSCs in the bottom chamber, serotonin treatment (100
µM) significantly decreased the differentiation of
osteoblasts, as evidenced by the lower level of bone-specific
alkaline phosphatase (AP) (Fig. 2B and
D) activity and by decreased mineralization of extracellular
matrix, as measured by Alizarin Red staining (ALZ) (Fig. 2C and E). Furthermore,
osteoblast-associated gene and protein expression levels of BSP,
osteocalcin and collagen type Iα1, were significantly suppressed
following treatment with serotonin in the co-culture system
(Fig. 2G–I). However, serotonin
alone had no direct effect on osteoblast differentiation without
co-culture with breast cancer cells. These data showed that
serotonin indirectly inhibits osteoblast differentiation by acting
on breast cancer cells, which prevents bone formation.
Circulating serotonin increases
osteolytic lesions by promoting osteoclast differentiation
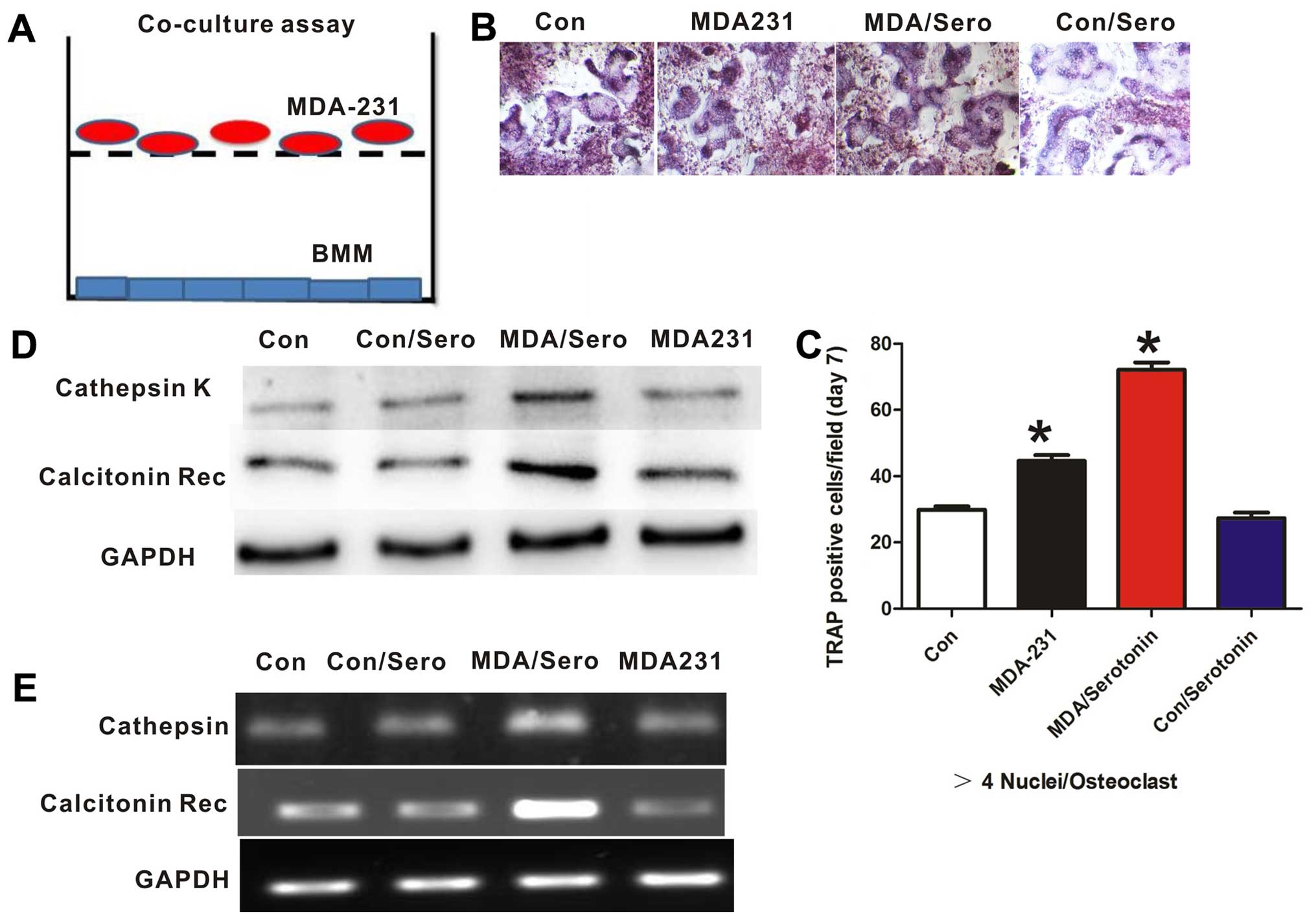
To test the effect of serotonin and MDA-MB-231 cells
on osteoclast differentiation, we incubated osteoclast progenitors
with breast cancer cells in a Transwell system. Osteoclast
differentiation was measured by staining cells for TRAP and
analyzing osteoclast-specific gene expression. In this co-culture
system, serotonin treatment significantly increased osteoclast
differentiation, as measured by TRAP activity (Fig. 3B and C). In addition, the expression
levels of osteoclast-related gene, cathepsin K, and calcitonin
receptor were significantly increased following serotonin treatment
(Fig. 3D and E). Thus, free
serotonin indirectly promotes osteoclast differentiation by acting
on breast cancer cells.
Gut-derived serotonin stimulates the
migration of breast cancer cells via RUNX2 signaling
In our previous studies, we found that RUNX2
expression in MDA-MB-231 cells was higher following serotonin
treatment (unpublished data). We hypothesized that serotonin might
promote bone metastasis by affecting RUNX2 expression in breast
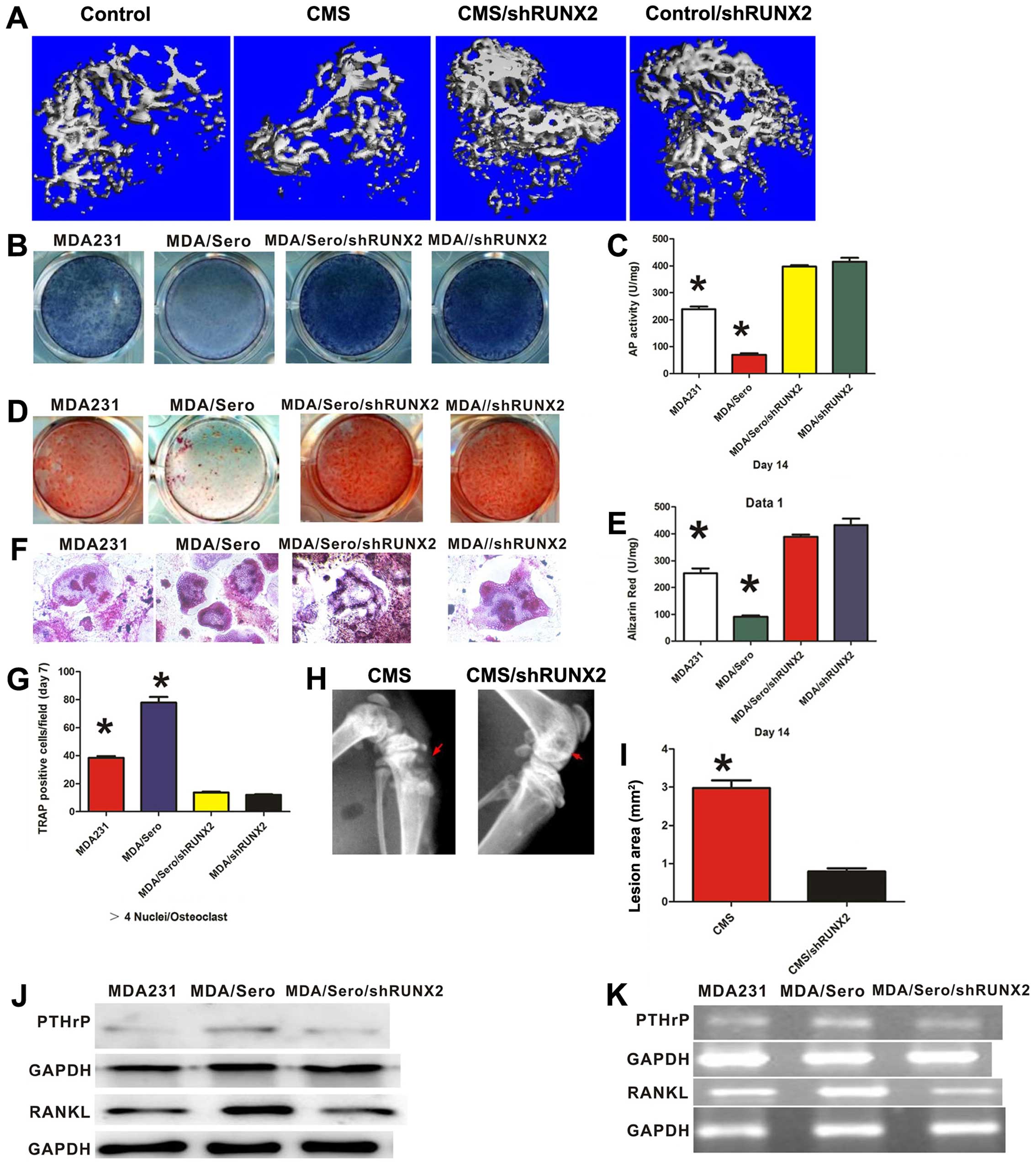
cancer cells. To address this question, we established an
adenovirus encoding a short hairpin (sh) interfering RNA for human
RUNX2. In the experiment, knockdown of RUNX2 by AD-shRUNX2 was
effective and specific (>90% reduction in RUNX2 expression by
western blotting and RT-PCR) compared to the scramble shRNA
control. RUNX2 deficiency reversed the suppression of osteo-blast
differentiation by serotonin co-culture with breast cancer cells,
as evidenced by AP and ALZ (Fig.
4B–E). Furthermore, knockdown of RUNX2 significantly decreased
PTHrP expression in the breast cancer cells and RANKL expression in
osteoblast cells treated with serotonin (Fig. 4J and K). In the co-cultured system
of breast cancer cells and pre-osteoclasts isolated from mouse bone
marrow, knockdown of RUNX2 also profoundly decreased the
differentiation of osteoclasts, as measured by TRAP staining
(Fig. 4F and G). The implication of
these findings is that free serotonin disrupted the remodeling
process of the bone microenvironment through RUNX2 signaling. To
further elucidate the effect of RUNX2 on the bone metastasis of
breast cancer cells in depression, we inoculated MDA-MB-231 shRUNX2
and control cells (scrambles) via intracardiac injection in nude
mice exposed to CMS to mimic a depressed state. Knockdown of RUNX2
in the MDA-MB-231 cells inhibited bone formation as measured by
µCT (Fig. 4A and Table III) and eliminated the effect of
CMS on osteolytic lesions as measured by Faxitron (Fig. 4H and I). These results demonstrated
that RUNX2 signaling is involved in the regulatory function of
depression on breast cancer cell bone metastasis.
 | Table IIIµCT analysis of long bones
from control, CMS, CMS/shRUNX2 and control/shRUNX2 mice. |
Table III
µCT analysis of long bones
from control, CMS, CMS/shRUNX2 and control/shRUNX2 mice.
| Control | CMS | CMS/shRUNX2 |
Control/shRUNX2 |
|---|
| (BV/TV) (%) | 5.44±1.5 | 2.88±1.3a | 6.82±1.7b | 6.18±1.4 |
| Conn.D | 27.38±7.1 | 17.8±4.6a | 31.89±9.2b | 34.32±7.4 |
| Tb.Nb | 2.33±0.5 | 1.67±0.4 | 2.75±0.6 | 2.88±0.6 |
| Tb.Th | 0.043±0.002 | 0.032±0.001 | 0.54±0.002 | 0.58±0.003 |
| Tb.Sp | 0.547±0.21 | 0.678±0.18 | 0.436±0.12 | 0.421±0.17 |
Discussion
In the present study, we used both in vitro and
in vivo models to functionally test the effect of depression on
breast cancer-induced bone metastasis. We first established a model
of depression by CMS and a breast cancer bone metastatic model by
intracardiac injection. Our results demonstrated that CMS
significantly promoted osteolytic lesions of breast cancer by
inhibiting the proliferation and differentiation of osteoblasts and
by activating the differentiation of osteoclasts. Furthermore,
gut-derived serotonin signaling participated in the migratory
process of breast cancer cells in the state of depression. We
confirmed that circulating serotonin directly inhibits osteoblast
proliferation and indirectly blunts osteo-blast differentiation via
RUAN2 signaling in the breast cancer cells and indirectly
stimulates osteoclast differentiation by activating the
RUNX2-PTHrP-RANKL pathway in the interaction between tumor cells
and osteoblasts. Together, our data demonstrated that depression
produces a higher level of circulating serotonin, which is
essential for the bone metastasis of breast cancer cells, and RUNX2
signaling is the key mechanism in the process of colonization and
invasion to the bone microenvironment of tumor cells.
Recent prospective research with mixed types of
cancers indicates that depression at the time of diagnosis is
associated with an increase in mortality (30,31).
Depression is strongly associated with mortality in younger
patients with early stage breast cancer (32). Yet, little is known concerning the
effect of depression on bone metastasis of breast cancer. Here, we
found that depression facilitates the colonization of breast cancer
cells in the bone microenvironment and accelerates bone mass
destruction in a mouse model. To elucidate the relationship between
depression and bone loss, Yirmiya and Bab identified a total of 23
studies and found that depressed individuals had lower BMD and
higher bone resorption markers than non-depressed subjects
(33). Yadav and Ducy demonstrated
that circulating serotonin suppressed osteoblast proliferation by
binding to the Htr1b serotonin receptor in bone. Furthermore,
LP533401 inhibitor of the initial enzyme of GDS, reversed the
effect of serotonin (34). Similar
to these observations, our experiments found that the circulating
level of serotonin was significantly higher in depressed mice than
that in the control group (not depressed). Moreover, high free
serotonin is responsible for the inhibition of osteoblast
proliferation and this inhibition can be reversed by LP533401,
which is a selective Tph1 inhibitor, in vivo. More
interestingly, serotonin can also suppress osteoblast
differentiation when co-cultured with breast cancer cells. One
explanation for this is that serotonin has an indirectly regulatory
function on osteoblasts by acting on breast cancer. In contrast to
a study by Kode et al (35),
which showed no effect of serotonin on osteoclast differentiation,
our studies in vitro found that circulating serotonin had a
positive effect on osteoclast differentiation by acting on breast
cancer cells. These striking findings suggest that depression
decouples the remodeling process of the bone microenvironment by
elevating the level of free serotonin, which results in metastatic
bone loss by inhibiting bone formation and promoting bone
resorption. Thus, bone metastasis is generated by both increased
activation of osteoclasts and suppression of osteoblasts.
It has been suggested that cancer cells
preferentially metastasize to bone due to their ability to express
genes that are normally considered bone or bone-related (36). In this process, cancer cells are
equipped to home, adhere, survive and proliferate in the bone
microenvironment by osteomimetic factors including osteopontin
(OPN), osteocalcin, osteonectin, bone sialoprotein, RANKL and
PTHrP. Several of these molecules related to the recruitment and
differentiation of osteoclasts play prominent roles in the vicious
cycle, which can in turn contribute to tumor cell survival,
proliferation, adhesion and migration (37). Interestingly, many osteomimetic
factors are regulated by the same transcription factor, Runx2.
Runx2 is considered to be the major regulator of osteoblast
commitment and differentiation (38). RUNX2 is one of the primary
transcriptional regulators of new bone formation and skeletal
maintenance and ablation of this gene in mice can result in
chondrogenic and osteogenic maturation (39,40).
Recent studies showed that metastatic breast cancer cells express
the transcriptional factor RUNX2, which can inhibit osteoblast
differentiation. Moreover, expression of RUNX2 concurrently
enhanced osteoclast differentiation in a marrow stromal cell
culture (41–43). Consistent with these observations,
our findings showed that RUNX2 and RUNX2 target genes were
necessary for depression-induced bone metastasis of breast cancer.
We observed that RUNX2 knockdown impacted breast cancer cell growth
in bone and osteolytic lesions stimulated by depression. We also
observed that when co-cultured with breast cancer cells in
vitro, RUNX2 knockdown eliminated the inhibition of osteoblast
differentiation and activation of osteoclast differentiation under
a free serotonin condition. These observations showed that tumor
cells lacking intact Runx2 function, independent of growth, did not
activate local osteoclasts and suppress osteoblasts. Thus, our
results support the conclusion that Runx2 activity is required for
stimulation of bone metastasis by depression.
Previous studies suggest that production of PTHrP is
more common in metastatic breast cancer cells in bone than in the
primary tumor, and that PTHrP may be responsible for the local bone
destruction occurring in patients with breast cancer, even in the
absence of hypercalcemia or increased plasma PTHrP concentrations.
The role of PTHrP in bone metabolism is not fully understood, but
it is known to cause upregulation of RANKL and downregulation of
OPG, which can enhance osteoclast function leading to bone
degradation (44,45). PTHrP, which is secreted by breast
cancer cells, can induce osteoclastic bone resorption by
stimulating RANKL expression in osteoblasts (46).
Analogous to these observations, our study showed
that serotonin treatment increased the expression of PTHrP in
breast cancer cells and RANKL in osteoblasts which can be
suppressed by RUNX2 knockdown. The data presented here suggest the
possibility that PTHrP may be an effector of RUNX2 in bone
metastases, as overexpression of PTHrP in breast cancer cells
consistent with overexpressed RUNX2 resulted in accelerated bone
metastases. The effect of RUNX2 on tumor cells to stimulate PTHrP
may result in adverse effects when tumor cells are housed in bone.
Tumor cells in the bone microenvironment produce PTHrP, which
causes upregulation of RANKL in osteoblasts, thus enhancing
osteoclast function leading to bone degradation.
In summary, we used a mouse model of depression to
confirm that depression promotes bone metastasis of breast cancer
which is associated with decoupled interplay between osteoclasts
and osteoblasts. By knockdown of RUNX2, we found that the
RUNX2/PTHrP/RANKL pathway plays an important role in breast cancer
bone metastasis induced by depression. These findings are
significant for the elucidation of the mechanisms of breast cancer
bone metastasis and provide insight into the prevention and/or
treatment of the osteolysis caused by breast cancer.
Acknowledgments
The present study was supported by research grants
from the Natural Science Foundation of China (NSFC, 81272005 to
Z.-L.D.) and the Nature Science Foundation of Chongqing in China
(CSFCQ, 2013jjB10021 to Z.-L.D.).
Abbreviations:
|
CMS
|
chronic mild stress
|
|
RUNX-2
|
runt-related transcription factor
2
|
|
PTHrP
|
parathyroid hormone-related
protein
|
|
RANKL
|
receptor activator for nuclear
factor-κB ligand
|
|
GDS
|
gut-derived serotonin
|
|
µCT
|
microcomputed tomography
|
|
shRNA
|
short hairpin RNA
|
|
MSCs
|
mesenchymal stem cells
|
|
AP
|
bone-specific alkaline phosphatase
|
|
ALZ
|
Alizarin Red staining
|
|
TRAP
|
tartrate-resistant acid
phosphatase
|
References
|
1
|
Feng X and McDonald JM: Disorders of bone
remodeling. Annu Rev Pathol. 6:121–145. 2011. View Article : Google Scholar
|
|
2
|
Henriksen K, Bollerslev J, Everts V and
Karsdal MA: Osteoclast activity and subtypes as a function of
physiology and pathology - implications for future treatments of
osteoporosis. Endocr Rev. 32:31–63. 2011. View Article : Google Scholar
|
|
3
|
Khosla S, Westendorf JJ and Oursler MJ:
Building bone to reverse osteoporosis and repair fractures. J Clin
Invest. 118:421–428. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Novack DV and Teitelbaum SL: The
osteoclast: Friend or foe? Annu Rev Pathol. 3:457–484. 2008.
View Article : Google Scholar
|
|
5
|
Raggatt LJ and Partridge NC: Cellular and
molecular mechanisms of bone remodeling. J Biol Chem.
285:25103–25108. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zaidi M: Skeletal remodeling in health and
disease. Nat Med. 13:791–801. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Waning DL and Guise TA: Molecular
mechanisms of bone metastasis and associated muscle weakness. Clin
Cancer Res. 20:3071–3077. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lipton A: Implications of bone metastases
and the benefits of bone-targeted therapy. Semin Oncol. 37(Suppl
2): S15–S29. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tonyali O, Arslan C and Altundag K: The
role of zoledronic acid in the adjuvant treatment of breast cancer:
Current perspectives. Expert Opin Pharmacother. 11:2715–2725. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tonello L and Cocchi M: The cell membrane:
Is it a bridge from psychiatry to quantum consciousness?
Neuroquantology. 8:54–60. 2010. View Article : Google Scholar
|
|
11
|
Cocchi M and Tonello L: Bio molecular
considerations in major depression and ischemic cardiovascular
disease. Cent Nerv Syst Agents Med Chem. 10:97–107. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cocchi M, Tonello L, De Lucia A and Amato
P: Platelet and brain fatty acids: A model for the classifcation of
the animals? Part 1. Int J Anthropol. 24:69–76. 2009.
|
|
13
|
Cocchi M, Tonello L, De Lucia A and Amato
P: Platelet and brain fatty acids: A model for the classification
of the animals? Part 2. Platelet and brain fatty acid transfer:
Hypothesis on arachidonic acid and its relationship to major
depression. Int J Anthropol. 24:201–220. 2009.
|
|
14
|
Heron DS, Shinitzky M, Hershkowitz M and
Samuel D: Lipid fluidity markedly modulates the binding of
serotonin to mouse brain membranes. Proc Natl Acad Sci USA.
77:7463–7467. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bab IA and Yirmiya R: Depression and bone
mass. Ann NY Acad Sci. 1192:170–175. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Darmon MC, Guibert B, Leviel V, Ehret M,
Maitre M and Mallet J: Sequence of two mRNAs encoding active rat
tryptophan hydroxylase. J Neurochem. 51:312–316. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Walther DJ, Peter JU, Bashammakh S,
Hörtnagl H, Voits M, Fink H and Bader M: Synthesis of serotonin by
a second tryptophan hydroxylase isoform. Science. 299(76)2003.
View Article : Google Scholar
|
|
18
|
Yadav VK, Ryu JH, Suda N, Tanaka KF,
Gingrich JA, Schütz G, Glorieux FH, Chiang CY, Zajac JD, Insogna
KL, et al: Lrp5 controls bone formation by inhibiting serotonin
synthesis in the duodenum. Cell. 135:825–837. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Haney EM and Warden SJ: Skeletal effects
of serotonin (5-hydroxytryptamine) transporter inhibition: Evidence
from clinical studies. J Musculoskelet Neuronal Interact.
8:133–145. 2008.PubMed/NCBI
|
|
20
|
Rosen CJ: Bone remodeling, energy
metabolism, and the molecular clock. Cell Metab. 7:7–10. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kopparapu PK, Tinzl M, Anagnostaki L,
Persson JL and Dizeyi N: Expression and localization of serotonin
receptors in human breast cancer. Anticancer Res. 33:363–370.
2013.PubMed/NCBI
|
|
22
|
Hernandez LL, Gregerson KA and Horseman
ND: Mammary gland serotonin regulates parathyroid hormone-related
protein and other bone-related signals. Am J Physiol Endocrinol
Metab. 302:E1009–E1015. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Horseman ND and Hernandez LL: New concepts
of breast cell communication to bone. Trends Endocrinol Metab.
25:34–41. 2014. View Article : Google Scholar
|
|
24
|
Johnson RW, Nguyen MP, Padalecki SS,
Grubbs BG, Merkel AR, Oyajobi BO, Matrisian LM, Mundy GR and
Sterling JA: TGF-beta promotion of Gli2-induced expression of
parathyroid hormone-related protein, an important osteolytic factor
in bone metastasis, is independent of canonical Hedgehog signaling.
Cancer Res. 71:822–831. 2011. View Article : Google Scholar :
|
|
25
|
Guise TA, Yin JJ, Taylor SD, Kumagai Y,
Dallas M, Boyce BF, Yoneda T and Mundy GR: Evidence for a causal
role of parathyroid hormone-related protein in the pathogenesis of
human breast cancer-mediated osteolysis. J Clin Invest.
98:1544–1549. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu R, Dang W, Jianting M, Su C, Wang H,
Chen Y and Tan Q: Citalopram alleviates chronic stress induced
depression-like behaviors in rats by activating GSK3β signaling in
dorsal hippo-campus. Brain Res. 1467:10–17. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao Y, Wang Z, Dai J, Chen L, Huang Y and
Zhan Z: Beneficial effects of benzodiazepine diazepam on chronic
stress-induced impairment of hippocampal structural plasticity and
depression-like behavior in mice. Behav Brain Res. 228:339–350.
2012. View Article : Google Scholar
|
|
28
|
Conway CC, Hammen C, Brennan PA, Lind PA
and Najman JM: Interaction of chronic stress with serotonin
transporter and catechol-O-methyltransferase polymorphisms in
predicting youth depression. Depress Anxiety. 27:737–745.
2010.PubMed/NCBI
|
|
29
|
Liu Q, Yang Q, Sun W, Vogel P, Heydorn W,
Yu XQ, Hu Z, Yu W, Jonas B, Pineda R, et al: Discovery and
characterization of novel tryptophan hydroxylase inhibitors that
selectively inhibit serotonin synthesis in the gastrointestinal
tract. J Pharmacol Exp Ther. 325:47–55. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Satin JR, Linden W and Phillips MJ:
Depression as a predictor of disease progression and mortality in
cancer patients: A meta-analysis. Cancer. 115:5349–5361. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pinquart M and Duberstein PR: Depression
and cancer mortality: A meta-analysis. Psychol Med. 40:1797–1810.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Vodermaier A, Linden W, Rnic K, Young SN,
Ng A, Ditsch N and Olson R: Prospective associations of depression
with survival: A population-based cohort study in patients with
newly diagnosed breast cancer. Breast Cancer Res Treat.
143:373–384. 2014. View Article : Google Scholar :
|
|
33
|
Yirmiya R and Bab I: Major depression is a
risk factor for low bone mineral density: A meta-analysis. Biol
Psychiatry. 66:423–432. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yadav VK and Ducy P: Lrp5 and bone
formation: A serotonin-dependent pathway. Ann NY Acad Sci.
1192:103–109. 2010. View Article : Google Scholar
|
|
35
|
Kode A, Mosialou I, Silva BC, Rached MT,
Zhou B, Wang J, Townes TM, Hen R, DePinho RA, Guo XE, et al: FOXO1
orchestrates the bone-suppressing function of gut-derived
serotonin. J Clin Invest. 122:3490–3503. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rucci N and Teti A: Osteomimicry: How
tumor cells try to deceive the bone. Front Biosci (Schol Ed).
2:907–915. 2010. View
Article : Google Scholar
|
|
37
|
Standal T, Borset M and Sundan A: Role of
osteopontin in adhesion, migration, cell survival and bone
remodeling. Exp Oncol. 26:179–184. 2004.PubMed/NCBI
|
|
38
|
Marie PJ: Transcription factors
controlling osteoblastogenesis. Arch Biochem Biophys. 473:98–105.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Komori T, Yagi H, Nomura S, Yamaguchi A,
Sasaki K, Deguchi K, Shimizu Y, Bronson RT, Gao YH, Inada M, et al:
Targeted disruption of Cbfa1 results in a complete lack of bone
formation owing to maturational arrest of osteoblasts. Cell.
89:755–764. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Banerjee C, McCabe LR, Choi JY, Hiebert
SW, Stein JL, Stein GS and Lian JB: Runt homology domain proteins
in osteoblast differentiation: AML3/CBFA1 is a major component of a
bone-specific complex. J Cell Biochem. 66:1–8. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mendoza-Villanueva D, Zeef L and Shore P:
Metastatic breast cancer cells inhibit osteoblast differentiation
through the Runx2/CBFβ-dependent expression of the Wnt antagonist,
sclerostin. Breast Cancer Res. 13:R1062011. View Article : Google Scholar
|
|
42
|
Pratap J, Wixted JJ, Gaur T, Zaidi SK,
Dobson J, Gokul KD, Hussain S, van Wijnen AJ, Stein JL, Stein GS,
et al: Runx2 transcriptional activation of Indian Hedgehog and a
downstream bone metastatic pathway in breast cancer cells. Cancer
Res. 68:7795–7802. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Javed A, Barnes GL, Pratap J, Antkowiak T,
Gerstenfeld LC, van Wijnen AJ, Stein JL, Lian JB and Stein GS:
Impaired intranuclear trafficking of Runx2 (AML3/CBFA1)
transcription factors in breast cancer cells inhibits osteolysis in
vivo. Proc Natl Acad Sci USA. 102:1454–1459. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Karaplis AC and Goltzman D: PTH and PTHrP
effects on the skeleton. Rev Endocr Metab Disord. 1:331–341. 2000.
View Article : Google Scholar
|
|
45
|
Kozlow W and Guise TA: Breast cancer
metastasis to bone: Mechanisms of osteolysis and implications for
therapy. J Mammary Gland Biol Neoplasia. 10:169–180. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Mundy GR: Metastasis to bone: Causes,
consequences and therapeutic opportunities. Nat Rev Cancer.
2:584–593. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
47
|
Pratap J, Javed A, Languino LR, van Wijnen
AJ, Stein JL, Stein GS and Lian JB: The Runx2 osteogenic
transcription factor regulates matrix metalloproteinase 9 in bone
metastatic cancer cells and controls cell invasion. Mol Cell Biol.
25:8581–8591. 2005. View Article : Google Scholar : PubMed/NCBI
|