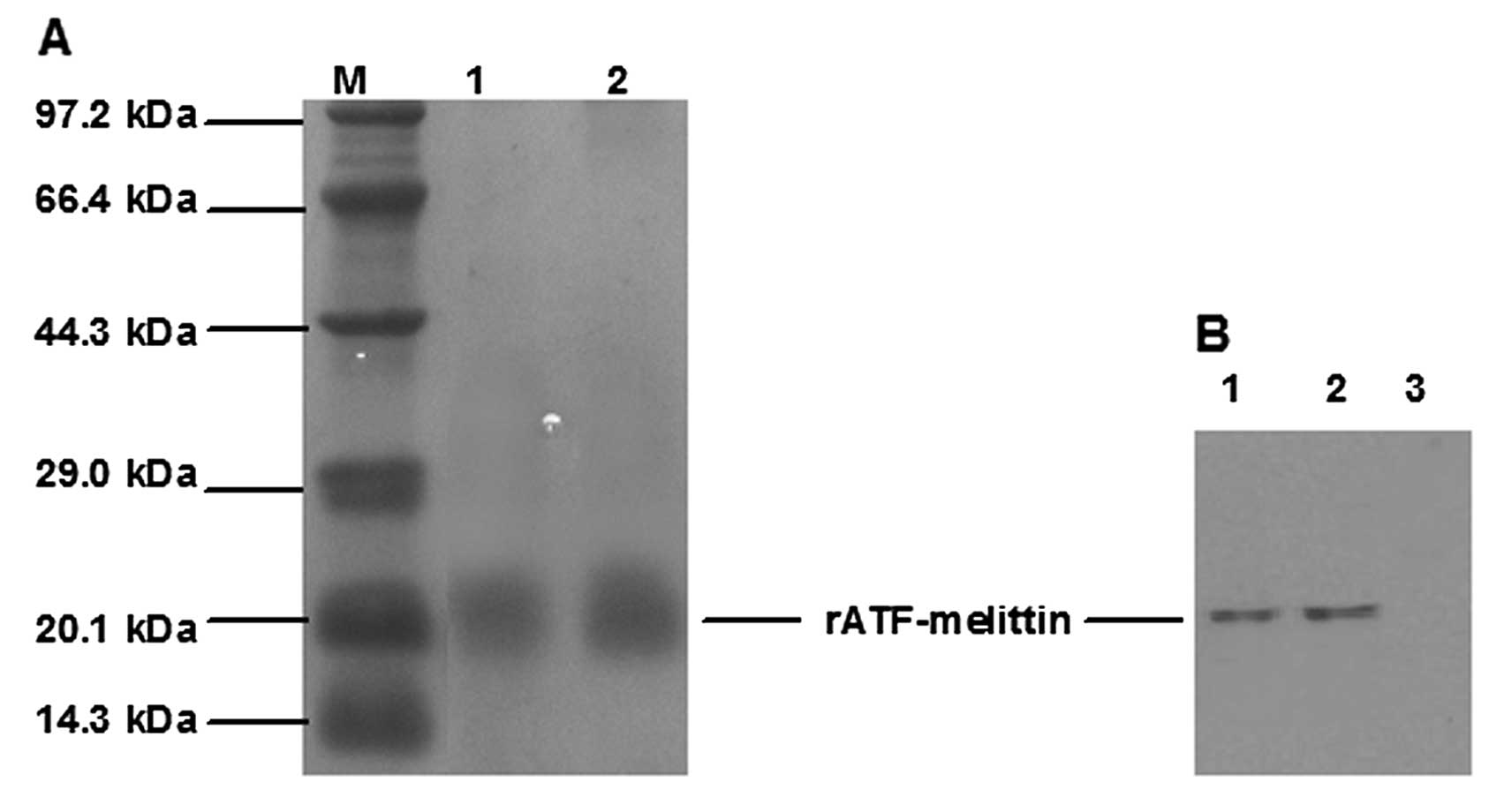
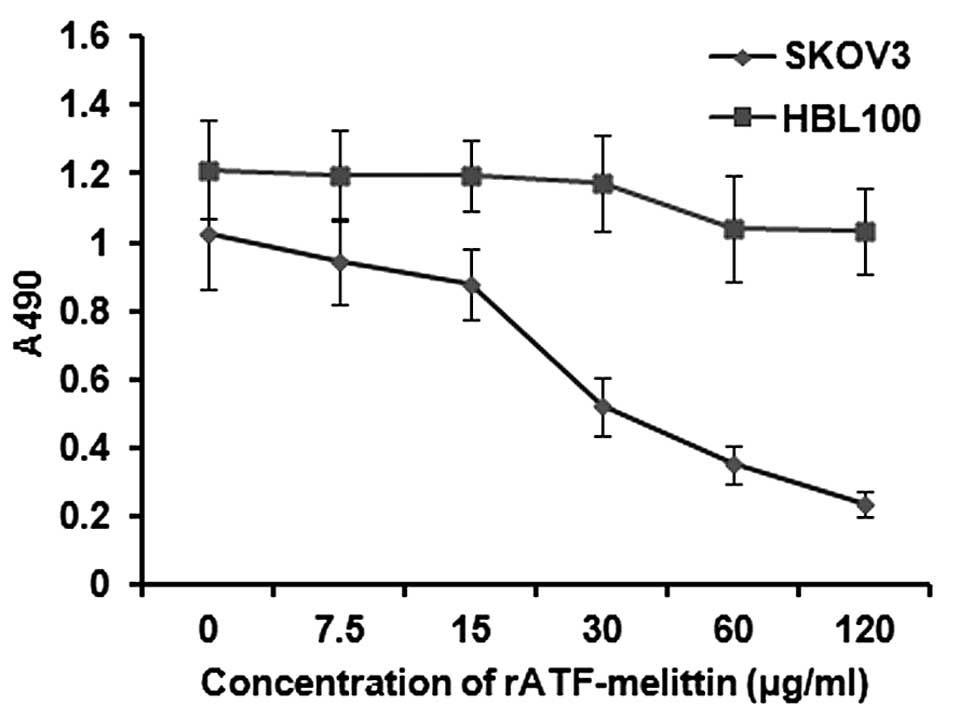
|
1
|
Raghuraman H and Chattopadhyay A:
Melittin: A membrane-active peptide with diverse functions. Biosci
Rep. 27:189–223. 2007. View Article : Google Scholar
|
|
2
|
Sommer A, Fries A, Cornelsen I, Speck N,
Koch-Nolte F, Gimpl G, Andrä J, Bhakdi S and Reiss K: Melittin
modulates keratinocyte function through P2 receptor-dependent ADAM
activation. J Biol Chem. 287:23678–23689. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Son DJ, Lee JW, Lee YH, Song HS, Lee CK
and Hong JT: Therapeutic application of anti-arthritis,
pain-releasing, and anti-cancer effects of bee venom and its
constituent compounds. Pharmacol Ther. 115:246–270. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang C, Chen T, Zhang N, Yang M, Li B, Lü
X, Cao X and Ling C: Melittin, a major component of bee venom,
sensitizes human hepatocellular carcinoma cells to tumor necrosis
factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis
by activating CaMKII-TAK1-JNK/p38 and inhibiting IkappaBalpha
kinase-NFkappaB. J Biol Chem. 284:3804–3813. 2009. View Article : Google Scholar
|
|
5
|
Jo M, Park MH, Kollipara PS, An BJ, Song
HS, Han SB, Kim JH, Song MJ and Hong JT: Anti-cancer effect of bee
venom toxin and melittin in ovarian cancer cells through induction
of death receptors and inhibition of JAK2/STAT3 pathway. Toxicol
Appl Pharmacol. 258:72–81. 2012. View Article : Google Scholar
|
|
6
|
Liu S, Yu M, He Y, Xiao L, Wang F, Song C,
Sun S, Ling C and Xu Z: Melittin prevents liver cancer cell
metastasis through inhibition of the Rac1-dependent pathway.
Hepatology. 47:1964–1973. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Russell PJ, Hewish D, Carter T,
Sterling-Levis K, Ow K, Hattarki M, Doughty L, Guthrie R, Shapira
D, Molloy PL, et al: Cytotoxic properties of immunoconjugates
containing melittin-like peptide 101 against prostate cancer: In
vitro and in vivo studies. Cancer Immunol Immunother. 53:411–421.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Soman NR, Baldwin SL, Hu G, Marsh JN,
Lanza GM, Heuser JE, Arbeit JM, Wickline SA and Schlesinger PH:
Molecularly targeted nanocarriers deliver the cytolytic peptide
melittin specifically to tumor cells in mice, reducing tumor
growth. J Clin Invest. 119:2830–2842. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Moghadam BY, Hou WC, Corredor C,
Westerhoff P and Posner JD: Role of nanoparticle surface
functionality in the disruption of model cell membranes. Langmuir.
28:16318–16326. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Funayama JC, Pucca MB, Roncolato EC,
Bertolini TB, Campos LB and Barbosa JE: Production of human
antibody fragments binding to melittin and phospholipase A2 in
Africanised bee venom: Minimising venom toxicity. Basic Clin
Pharmacol Toxicol. 110:290–297. 2012. View Article : Google Scholar
|
|
11
|
Holle L, Song W, Holle E, Wei Y, Li J,
Wagner TE and Yu X: In vitro- and in vivo-targeted tumor lysis by
an MMP2 cleavable melittin-LAP fusion protein. Int J Oncol.
35:829–835. 2009.PubMed/NCBI
|
|
12
|
Botkjaer KA, Deryugina EI, Dupont DM,
Gårdsvoll H, Bekes EM, Thuesen CK, Chen Z, Ploug M, Quigley JP and
Andreasen PA: Targeting tumor cell invasion and dissemination in
vivo by an aptamer that inhibits urokinase-type plasminogen
activator through a novel multifunctional mechanism. Mol Cancer
Res. 10:1532–1543. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li J, Lin Y, Zhuang H and Hua ZC:
Expression, purification, and biological characterization of the
amino-terminal fragment of urokinase in Pichia pastoris. J
Microbiol Biotechnol. 23:1197–1205. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang L, Sajja HK, Cao Z, Qian W, Bender L,
Marcus AI, Lipowska M, Wood WC and Wang YA: uPAR-targeted optical
imaging contrasts as theranostic agents for tumor margin detection.
Theranostics. 4:106–118. 2013. View Article : Google Scholar
|
|
15
|
Ding Y, Zhang H, Zhong M, Zhou Z, Zhuang
Z, Yin H, Wang X and Zhu Z: Clinical significance of the uPA system
in gastric cancer with peritoneal metastasis. Eur J Med Res.
18:282013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Thummarati P, Wijitburaphat S, Prasopthum
A, Menakongka A, Sripa B, Tohtong R and Suthiphongchai T: High
level of urokinase plasminogen activator contributes to
cholangiocarcinoma invasion and metastasis. World J Gastroenterol.
18:244–250. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Foekens JA, Peters HA, Look MP, Portengen
H, Schmitt M, Kramer MD, Brünner N, Jänicke F, Meijer-van Gelder
ME, Henzen-Logmans SC, et al: The urokinase system of plasminogen
activation and prognosis in 2780 breast cancer patients. Cancer
Res. 60:636–643. 2000.PubMed/NCBI
|
|
18
|
Kim TD, Song KS, Li G, Choi H, Park HD,
Lim K, Hwang BD and Yoon WH: Activity and expression of
urokinase-type plasminogen activator and matrix metalloproteinases
in human colorectal cancer. BMC Cancer. 6:2112006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Laufs S, Schumacher J and Allgayer H:
Urokinase-receptor (u-PAR): an essential player in multiple games
of cancer: a review on its role in tumor progression, invasion,
metastasis, proliferation/dormancy, clinical outcome and minimal
residual disease. Cell Cycle. 5:1760–1771. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rockway TW, Nienaber V and Giranda VL:
Inhibitors of the protease domain of urokinase-type plasminogen
activator. Curr Pharm Des. 8:2541–2558. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Carriero MV and Stoppelli MP: The
urokinase-type plasminogen activator and the generation of
inhibitors of urokinase activity and signaling. Curr Pharm Des.
17:1944–1961. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li H, Soria C, Griscelli F, Opolon P,
Soria J, Yeh P, Legrand C, Vannier JP, Belin D, Perricaudet M, et
al: Amino-terminal fragment of urokinase inhibits tumor cell
invasion in vitro and in vivo: Respective contribution of the
urokinase plasminogen activator receptor-dependent or-independent
pathway. Hum Gene Ther. 16:1157–1167. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Longuespée R, Boyon C, Desmons A, Vinatier
D, Leblanc E, Farré I, Wisztorski M, Ly K, D'Anjou F, Day R, et al:
Ovarian cancer molecular pathology. Cancer Metastasis Rev.
31:713–732. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Su D, Katsaros D, Xu S, Xu H, Gao Y,
Biglia N, Feng J, Ying L, Zhang P, Benedetto C, et al:
ADP-ribosylation factor-like 4C (ARL4C), a novel ovarian cancer
metastasis suppressor, identified by integrated genomics. Am J
Transl Res. 7:242–256. 2015.PubMed/NCBI
|
|
25
|
Coosemans A, Baert T and Vergote I: A view
on dendritic cell immunotherapy in ovarian cancer: How far have we
come? Facts Views Vis Obgyn. 7:73–78. 2015.PubMed/NCBI
|
|
26
|
Sun D, Sun M, Zhu W, Wang Z, Li Y and Ma
J: The anti-cancer potency and mechanism of a novel tumor-activated
fused toxin, DLM. Toxins. 7:423–438. 2015. View Article : Google Scholar : PubMed/NCBI
|