Introduction
Esophageal carcinoma is the eighth most common
malignancy worldwide, with an estimated 455,800 new cases and
400,200 deaths annually (1), ~70%
of which occurred in China (2).
Approximately 60% of all patients have advanced disease at the time
of diagnosis, although advances in surgical techniques and
neoadjuvant and adjuvant chemoradiotherapy have led to increasingly
multimodal therapy for patients, the longevity remains
disappointingly low, with 5-year overall survival uniformly ~15%
(1,2). The two main histological types:
esophageal squamous cell carcinoma (ESCC) and adenocarcinoma (EAC),
exhibit many differences in etiology, epidemiology, genetic
landscape, clinical characteristics and prognosis (3,4), while
>90% of cases in China is ESCC (2). The application of targeted therapy is
mostly limited to EAC, few patients with advanced ESCC can be cured
with synthetic treatment modality (5). Therefore, it is important to discover
the molecular mechanism driving ESCC progression and develop novel
diagnostic and prognostic methods and potential therapeutic
targets.
RNA plays a central role in transmitting of genetic
information from DNA to protein. RNA helicases are a large family
of enzymes that perform diverse cellular functions in virtually all
steps of RNA metabolism, ranging from gene transcription to RNA
decay (6). DEAD-box helicases form
the largest family in RNA helicases and are ubiquitous from
bacteria to humans, play key roles in rearranging RNA-RNA and
RNA-protein interactions, participate in almost all aspects of
processes, including pre-mRNA splicing, ribosome biogenesis, RNA
transport, mRNA export, translation initiation and termination, and
organelle gene expression (7). In
addition, they also participate in an ATPase-independent manner in
basic processes such as transcriptional regulation (7). Several DEAD-box RNA helicases are
aberrantly expressed in tumor tissues and cell lines, and involved
in the processes that are key to cellular proliferation and cancer
progression and transformation, therefore, it is not surprising
that deregulation of expression or function of these proteins has
been implicated in cancer development or progression (8). These proteins are excellent potential
tumor-specific vulnerabilities that one might be able to capitalize
upon for the development of novel molecular-targeted therapies
(9,10).
DDX46 gene, also named as PRPF5 or Prp5, belongs to
DEAD-box RNA helicase family. In vitro biochemical assays
have shown that DDX46 is required for stable association of U2 in
the pre-spliceosome and have multiple functions in nuclear pre-mRNA
splicing before or during pre-spliceosome assembly (11,12).
In vivo zebrafish studies have shown that DDX46 is required
for the development of the digestive organs and brain through the
control of pre-mRNA splicing (13),
and multi-lineage differentiation of hematopoietic stem cells
through the regulation of specific gene expressions (14). Human DDX46 gene is located in
chromosome 5q31.1 (15). DDX46 has
not been reported involved in human cancers, except that Li and
colleagues (16) reported that
DDX46 expression was upregulated in colorectal carcinoma tissues
and critical for colorectal carcinoma cell proliferation. The
clinical significance and biologic role of DDX46 in ESCC, however,
have not been implicated previously. In this study, we found that
DDX46 was overexpressed in human ESCC tissues as compared with
adjacent normal tissues, and silencing of DDX46 expression
inhibited proliferation and induced apoptosis of human ESCC cells
in which DDX46 was amplified.
Materials and methods
Patients and tissue specimens
All tissue specimens were obtained from the 69 ESCC
patients who underwent esophagogastrostomy from January 2014 to
December 2014 at the Department of Thoracic Surgery, Lanzhou
University Second Hospital (Lanzhou, China). For histological
evaluation, hematoxylin-eosin stained sections from each specimen
were determined by pathologists. Preoperative clinical evaluation
was performed for all patients consisting of ultrasonography of the
neck, CT scan of the chest and abdomen, barium swallow examination,
upper gastrointestinal endoscopy with biopsy, Tc99m
whole body bone scan. No patient received chemo- or radiotherapy
prior to operation. All of the specimens had matched adjacent
normal tissues. The study protocol was approved by the medical
ethics committee of our hospital.
Immunohistochemistry
Immunohistochemistry (IHC) was performed using the
immunohistochemical SP kit (Maixin Biotech, Fuzhou, China)
according to the manufacturer's instructions. Briefly,
paraffin-embedded tissue sections were deparaffinized and
rehydrated. Endogenous peroxidase activity was blocked by
incubation in 3% hydrogen peroxide for 15 min at room temperature.
Antigen was retrieved by heating in a pressure cooker for 5 min in
sodium citrate buffer (pH 6.0). Non-specific antigens were blocked
by incubating in sheep serum for 30 min at room temperature. After
incubation with diluted rabbit anti-DDX46 (1:1,000 dilution,
Sigma-Aldrich, St. Louis, MO, USA) overnight at 4°C, the sections
were rinsed with PBS and secondary antibodies were applied at a
1:500 dilution in PBS for 30 min at room temperature. Then the
sections were incubated with streptavidin-horseradish peroxidase
(HRP) for 30 min at room temperature and rinsed with PBS and
incubated for 15 min with the chromogen diaminobenzidine (DAB) and
counterstained with hematoxylin. The images of sections were
captured under a transmission light microscope.
DDX46 expression was quantified using a visual
grading system based on the extent of staining (graded from 0 to 4:
0, none; 1, 1–25%; 2, 26–50%; 3, 51–75%; 4, >75%) and the
intensity of staining (graded from 0 to 3: 0, no staining; 1, weak
staining; 2, moderate staining; 3, strong staining). The index
value was calculated as a product of the extent of staining and
intensity of staining scores to evaluate the expression of DDX46.
DDX46 expression was classified into three grades: negative (index
values 0–3), weak positive (index values 4–8) and strong positive
(index values 9–12).
Cell culture and lentivirus
transfection
The human ESCC cell lines (TE-1, Eca-109, TE-11 and
KYSE-150) were purchased from the Cell Bank of the Chinese Academy
of Sciences (Shanghai, China), the human normal esophageal
epithelium cell line Het-1A was purchased from the Bioleaf Biotech
(Shanghai, China). All cell lines tested negative for any
mycoplasma contamination, and were maintained in a standard
humidified incubator at 37°C in 5% CO2 and used at
passage numbers 5–7 for the experiments.
Using specific short hairpin RNA (shRNA) mediated by
lentiviral vector (LV) to knockdown DDX46. Lentiviral constructs
expressing DDX46 shRNA (DDX46-shRNA-LV) and non-silencing negative
control LV (Control-LV) were designed and synthesized by Genechem
Chemtech (Shanghai, China) according to the sequence encoding human
DDX46 complementary DNA (NM_014829). The silencing shRNA sequence
was 5′-CATCCAAACCCAAGCTATT-3′, and non-silencing negative control
sequence was 5′-TTCTCCGAACGTGTCACGT-3′.
For lentivirus transfection, TE-1 and Eca-109 cells
were cultured in 6-well plates at a concentration of
2×105 cells per well on the day before shRNA
transfection. Then, DDX46-shRNA-LV was transfected into cells at a
multiplicity of infection (MOI) of 5 (TE-1) or 10 (Eca-109) using
polybrene (10 µg/ml) and enhanced infection solution
(Genechem Chemtech). At the same time, Control-LV was transfected
into cells using the same methods to control for the impact of the
viral vector. After incubation for 12 h, the medium was replaced
with fresh L-15 medium. After 72 h of incubation, cells were
observed under fluorescence microscopy (MicroPublisher 3.3RTV;
Olympus, Tokyo, Japan) to evaluate the efficiency of transfection.
At the indicated time-points, the cells were harvested for mRNA and
protein analysis as well as other assays.
Quantitative real-time PCR
The total RNA of cultured cells was isolated using
TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to the
manufacturer's instructions. The primers were designed by Beacon
Designer 2 software (Premier Biosoft, Palo Alto, CA, USA). DDX46
primers were as follows: forward, 5′-AAAATGGCGAGAAGAGCAACG-3′; and
reverse, 5′-CATCATCGTCCTCTAAACTCCAC-3′; glyceraldehyde-3-phosphate
dehydrogenase (GAPDH) was used as an internal control: forward,
5′-TGACTTCAACAGCGACACCCA-3′; and reverse,
5′-CACCCTGTTGCTGTAGCCAAA-3′). Quantitative real-time PCR (qRT-PCR)
was performed with the 7500 Real-Time PCR system (Applied
Biosystems, Foster City, CA, USA) using the SYBR Green Real-Time
PCR assay kit (Takara, Otsu, Japan). The relative expression levels
of mRNAs were calculated using the ΔCt (ΔCt = Ct sample − Ct
control) or 2−ΔΔCt (ΔΔCt = ΔCt sample − ΔCt control)
method.
Western blot analysis
The total protein was extracted from cultured cells
using total protein extraction kit as recommended by the
manufacturer (Sigma-Aldrich). The protein extracts (20 µg)
were separated on 10% sodiumdodecyl sulfate-polyacrylamide gels
(SDS-PAGE; Tanon Technology, Shanghai, China) and electrotransfered
onto polyvinylidene difluoride (PVDF) membranes (Millipore,
Billerica, MA, USA). The membranes were blocked in Tris-buffered
saline with Tween-20 (TBST) containing 5% powdered milk for 1 h at
room temperature, then incubated overnight at 4°C in blocking
solution with primary antibody as follows: anti-DDX46 (1:1,000
dilution, Sigma-Aldrich); anti-GAPDH (used as a loading control,
1:2,000 dilution, Santa Cruz Biotechnology, Santa Cruz, CA, USA).
After washing with TBST, the membranes were incubated with
HRP-conjugated secondary antibody (1:2,000 dilution, Beyotime,
Haimen, China) for 2 h at room temperature. The membranes were then
visualized using Pierce ECL Western Blotting Substrate (Thermo
Fisher Scientific, Rockford, IL, USA) according to the
manufacturer's instructions.
Cell growth assay
The cultured cells were seeded in 96-well plates at
a density of 2×103 cells per well. Dynamic growth of
green fluorescence protein (GFP)-labeled cells were monitored once
a day for 5 consecutive days using High Content Screening (HCS)
(Cellomics, ArrayScan VT1; Thermo Fisher Scientific). Cell growth
curves were drawn.
MTT cell viability assay
After the indicated treatments, the cells in the
logarithmic growth phase were seeded in 96-well plates in
triplicate at densities of 2×103 cells per well. Cell
proliferation was evaluated at 1, 2, 3, 4 and 5 days using the
methylthiazoltetrazolium (MTT) assay. In brief, at indicated
time-points, cells were incubated with 20 µl sterile MTT dye
(5 mg/ml, Sigma-Aldrich) for 4 h at 37°C, followed by removal of
the culture medium and addition of 100 µl dimethyl sulfoxide
(DMSO) to each well to stop the reaction. Then the 96-well plates
were shaken for 5 min, and the optical density (OD) values were
measured at 490 nm using a microplate reader (Bio-Rad, Hercules,
CA, USA).
Colony formation assay
Cells were plated in 6-well plates at 600 cells per
well and cultured for 15 days. After fixation with 4%
paraformaldehyde, the colonies were stained with Giemsa for 15 min.
Then the colonies were photographed and counted under a microscope.
Colonies consisting of ≥50 cells were counted as a clone.
Flow cytometry analysis
For detection of cell apoptotic activity, the cells
were cultured in 6-well plates. When the cells reached ~85%
confluence, they were collected and stained using the Annexin
V-Allophycocyanin (APC) single-staining Apoptosis Detection kit
(eBioscience, San Diego, CA, USA) according to the manufacturer's
instructions. For detection of the cell cycle, the cells were
cultured in 6-cm dishes. At ~85% confluence, cell cycle analysis
was performed after staining with propidium iodide (Sigma-Aldrich).
Both apoptosis and cell cycle distribution were quantified using a
Guava easy-Cyte HT flow cytometer (Millipore).
Stress and apoptosis signaling antibody
array
At ~85% confluence, cell extracts were prepared and
analyzed using the PathScan® Stress and Apoptosis
Signaling Antibody Array kit analysis (chemiluminescent readout,
Cell Signaling Technology, Danvers, MA, USA) according to the
manufacturer's instructions. Images were acquired by briefly
exposing the slide to standard chemiluminescent film.
Statistical analysis
All experiments were performed in triplicate. The
data are expressed as means ± standard deviation (SD) determined
from three independent experiments. Student's t-test or
Mann-Whitney U test was applied for statistical analysis using SPSS
software (version 16.0; SPSS Inc., Chicago, IL, USA). P-value
<0.05 was considered statistically significant.
Results
DDX46 is overexpressed in ESCC tissues
and cell lines
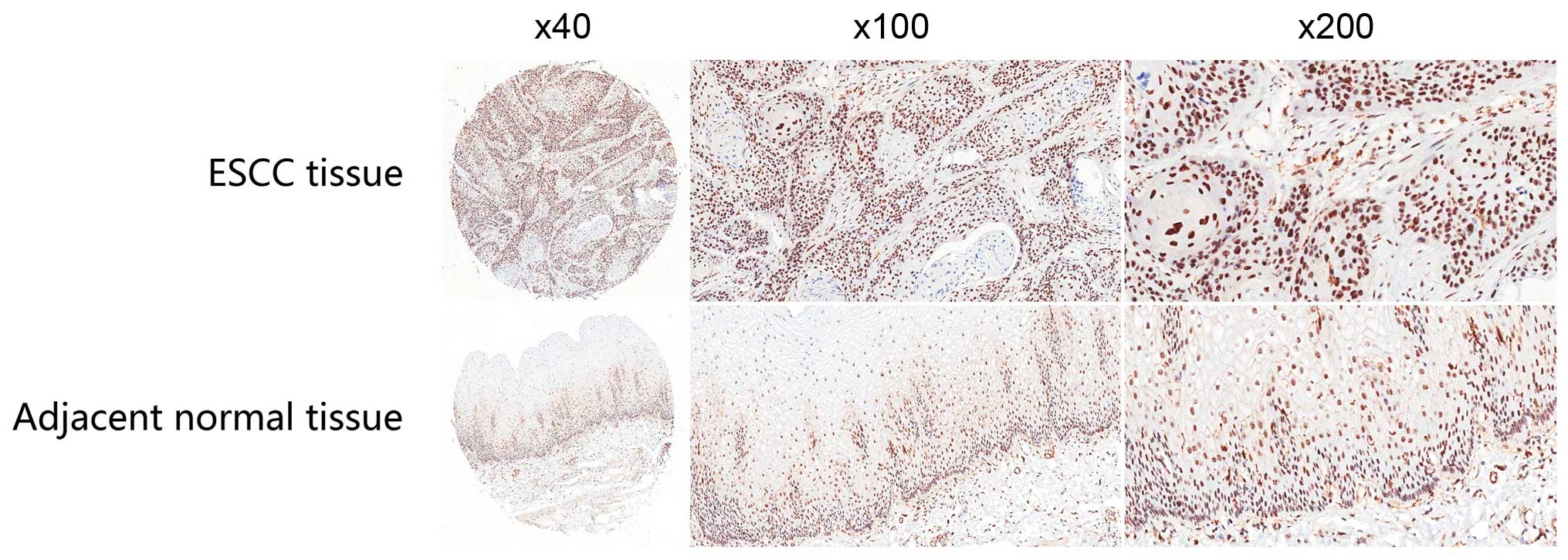
DDX46 protein expression was examined in 69 ESCC
tissues and adjacent normal tissues using IHC. It showed that DDX46
expression was predominantly located in the nucleus of ESCC cells,
and comparative analysis revealed that the expression levels of
DDX46 in ESCC tissues were higher than those in matched adjacent
non-tumor tissues (P<0.001; Table
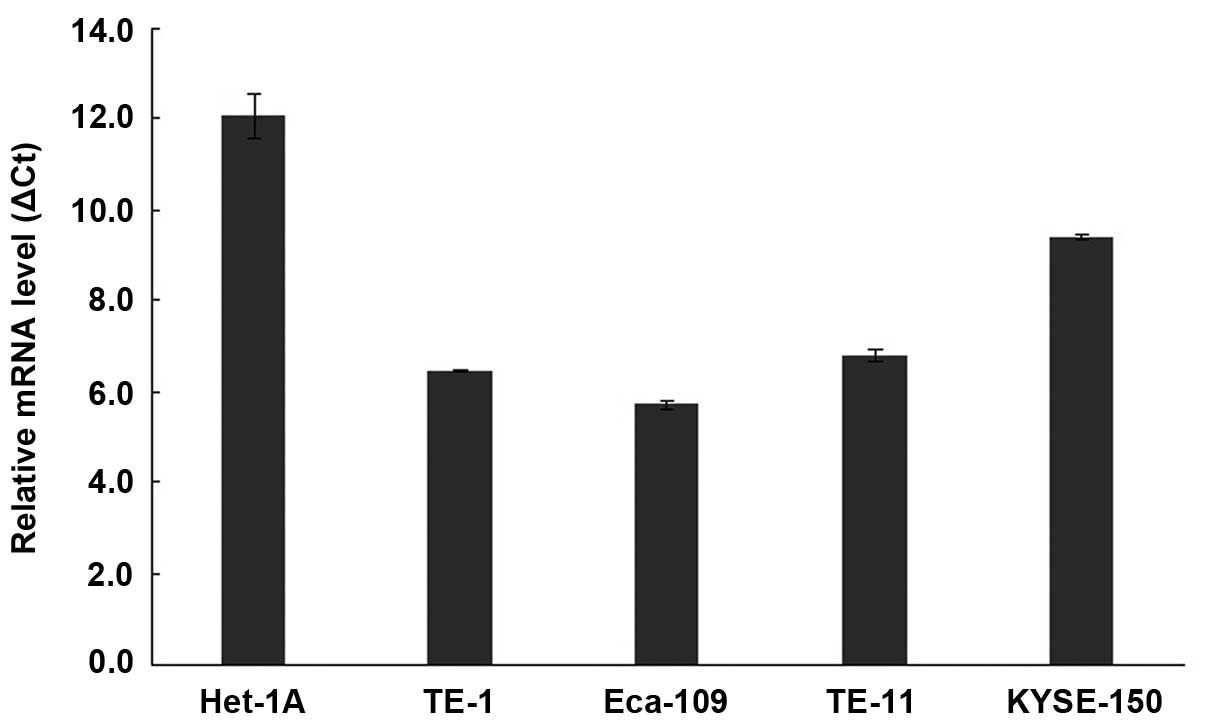
I and Fig. 1). qRT-PCR analysis
showed that DDX46 mRNA were significantly upregulated in all four
tested human ESCC cell lines (TE-1, Eca-109, TE-11 and KYSE-150),
compared with the human normal esophageal epithelium cell line
Het-1A (Fig. 2). These results
suggest that DDX46 expression was upregulated in ESCC.
 | Table IOverexpression of DDX46 detected in
ESCC tissues by IHC examination. |
Table I
Overexpression of DDX46 detected in
ESCC tissues by IHC examination.
| All cases | Negative | Weak positive | Strong
positive |
|---|
| ESCC tissues | 69 | 7 | 30 | 32 |
| Adjacent normal
tissues | 69 | 53 | 9 | 7 |
| P-value | <0.001 |
Lentivirus-mediated RNAi efficiently
decreases DDX46 expression
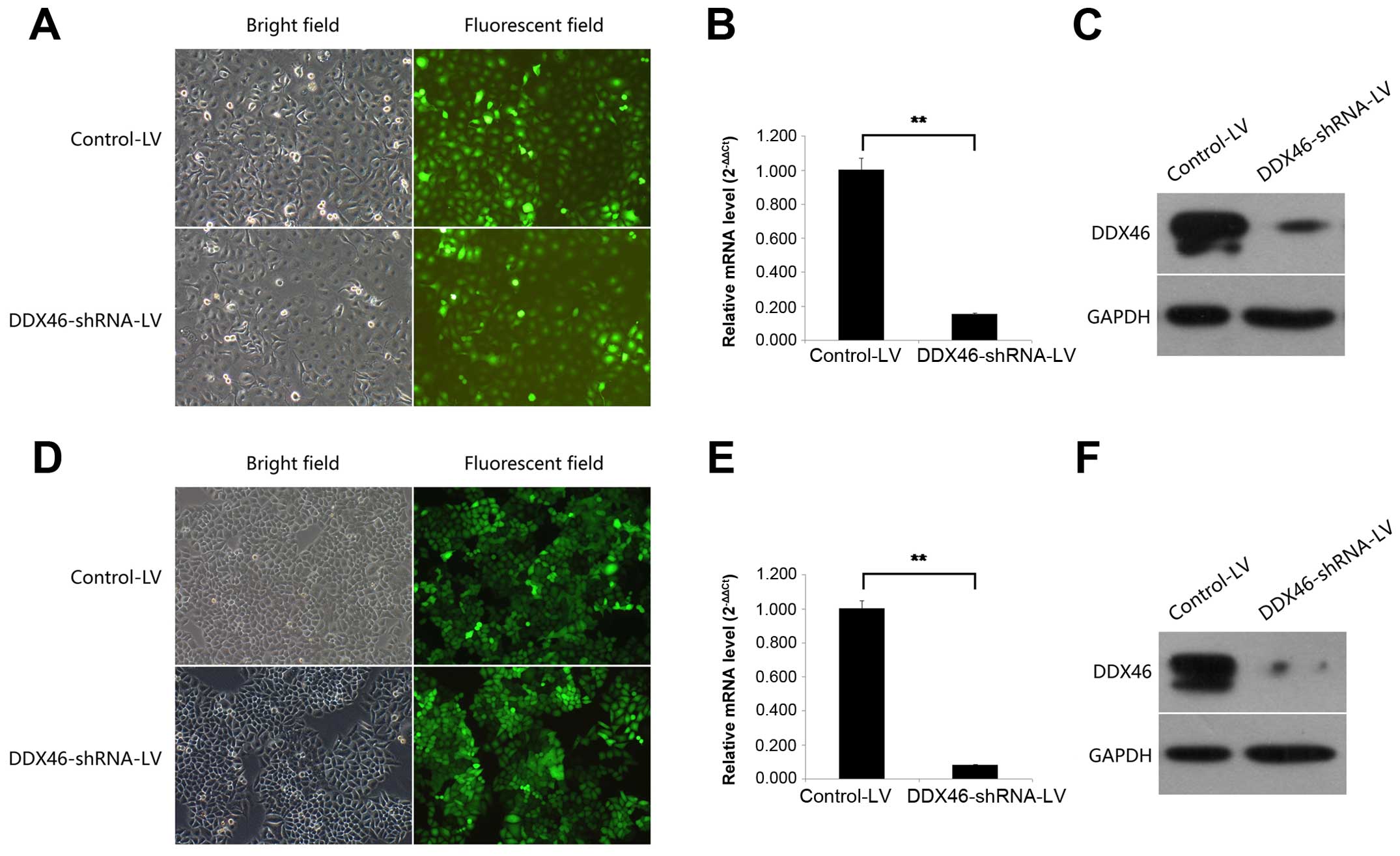
TE-1 and Eca-109 cells were chosen to silence the
endogenous DDX46 expression by DDX46-shRNA-LV. The transfection
efficiency of LV was confirmed by observing GFP-expressing cells
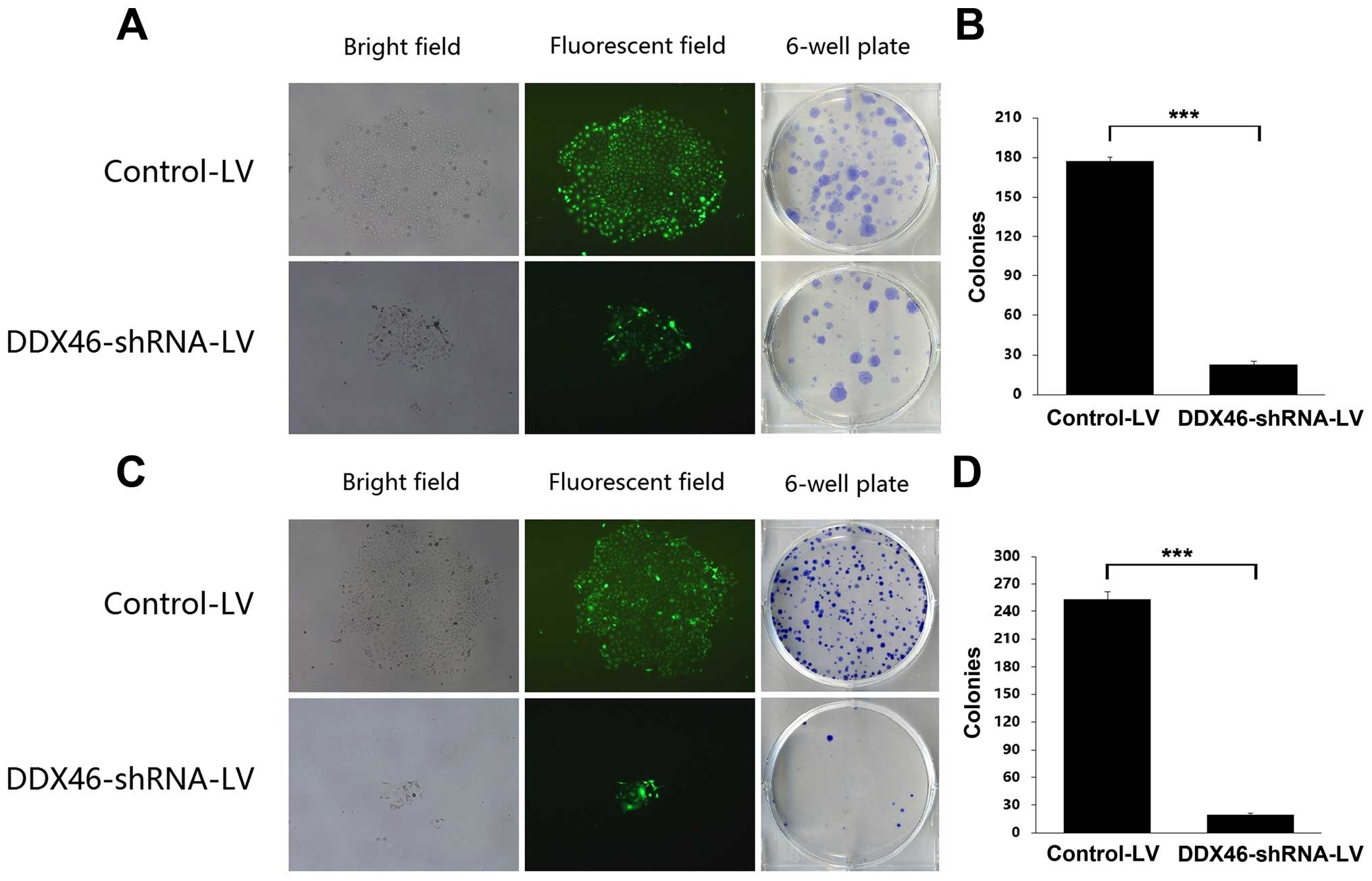
under a fluorescence microscope 72 h after incubation. As shown in
Fig. 3A and D, >90% of the TE-1
and Eca-109 cells were transfected. Then the silencing effect of LV
in TE-1 and Eca-109 cells was measured by qRT-PCR and western blot
analysis. Compared with the Control-LV group, DDX46 mRNA expression
level was reduced by 84.6% (Fig.
3B, P<0.01) and 91.8% (Fig.
3E, P<0.01) in TE-1 and Eca-109 cells, respectively, after
DDX46-shRNA-LV transfection. In parallel, DDX46 protein expression
level was also significantly decreased in the DDX46-shRNA-LV
transfected TE-1 and Eca-109 cells (Fig. 3C and F). These results indicated
that the lentivirus-mediated RNAi system was able to knock down the
endogenous expression of DDX46 in TE-1 and Eca-109 cells
specifically and effectively.
Silencing DDX46 significantly inhibits
ESCC cell proliferation
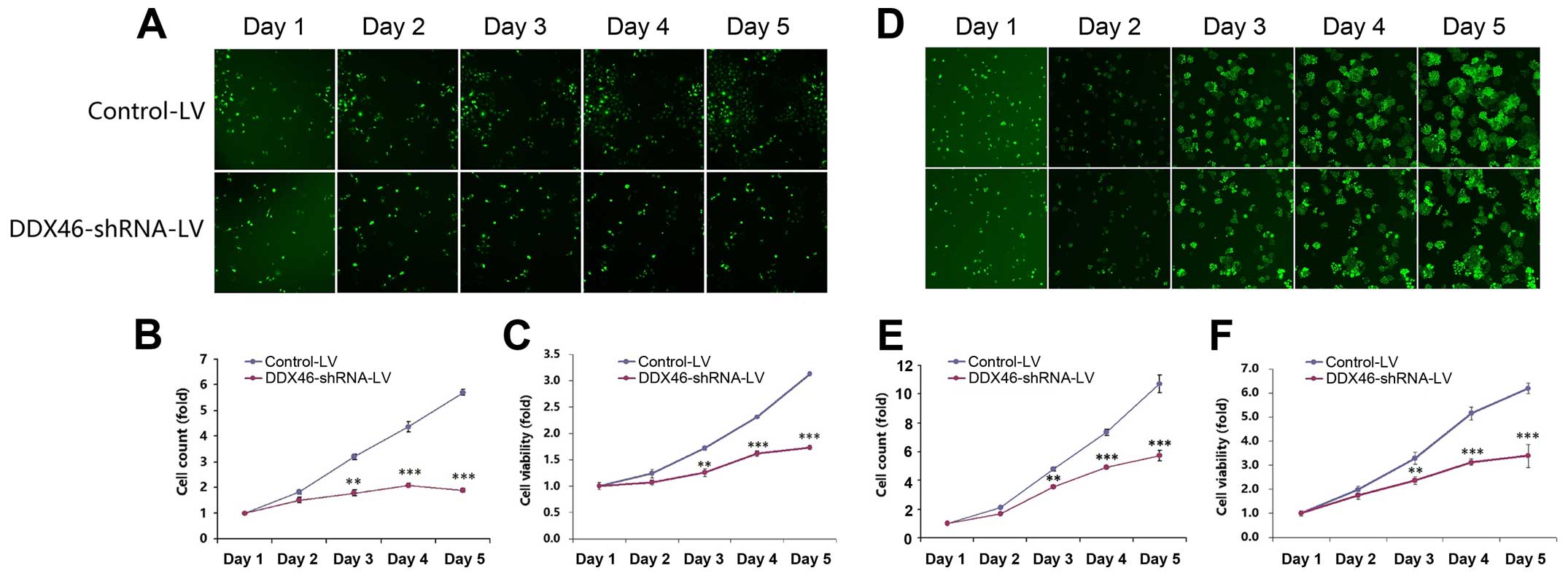
In order to determine the effects of DDX46 on cell
growth, cells with green fluorescence were counted once a day for 5
consecutive days using HCS (Fig. 4A and
D). The number of GFP-labeled cells in the DDX46-shRNA-LV group
was greatly decreased compared to the Control-LV group in both TE-1
(Fig. 4B) and Eca-109 (Fig. 4E) cells. Silencing DDX46 also
significantly inhibited cell viability that was analyzed by MTT
assays. As illustrated in Fig. 4C and
F, cell viability of DDX46-shRNA-LV transfected TE-1 and
Eca-109 cells were significantly reduced in a time-dependent
manner. Additionally, DDX46 knockdown significantly suppressed
colony formation capacity in ESCC cells. The number and size of
colonies in both DDX46-shRNA-LV transfected TE-1 and Eca-109 cells
were significantly reduced compared with the corresponding
Control-LV group (Fig. 5).
Collectively, these results suggested that overexpression of DDX46
played an essential role in proliferation of ESCC cells in
vitro.
Silencing DDX46 significantly induces
apoptosis and regulated cell cycle arrest in ESCC cells
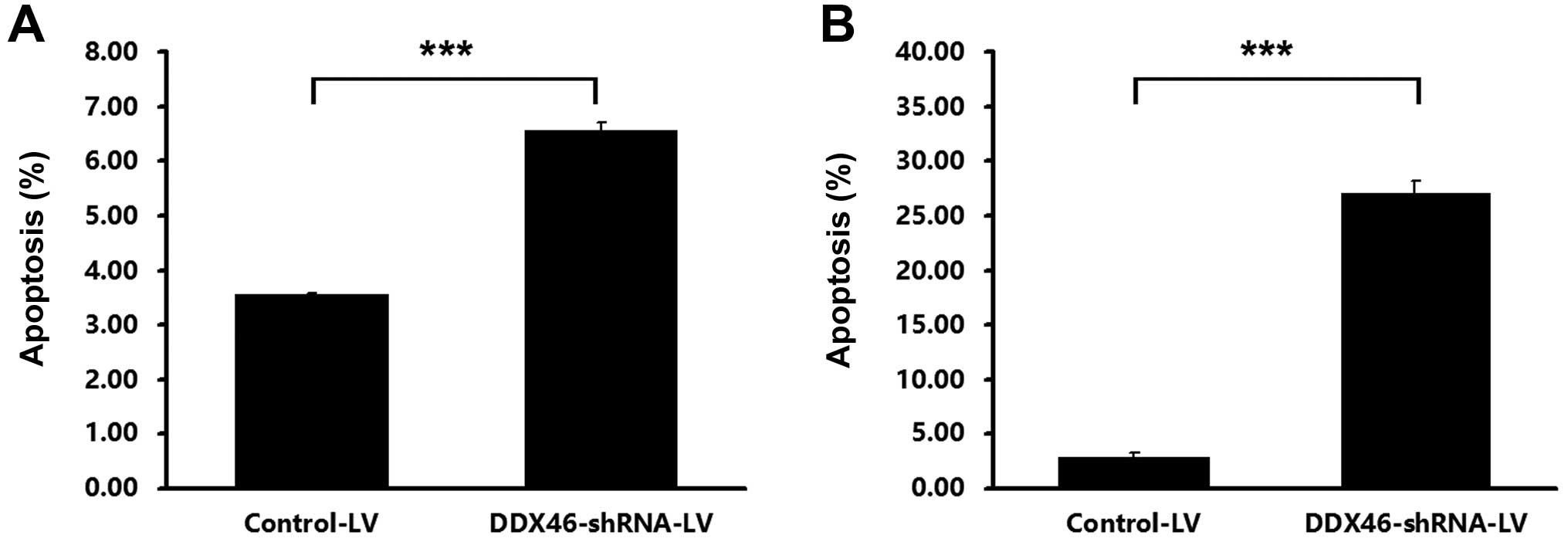
Transfection of DDX46-shRNA-LV resulted in a
significant increase in the percentage of apoptotic cells, compared
with the Control-LV, in TE-1 (Fig.
6A) and Eca-109 (Fig. 6B)
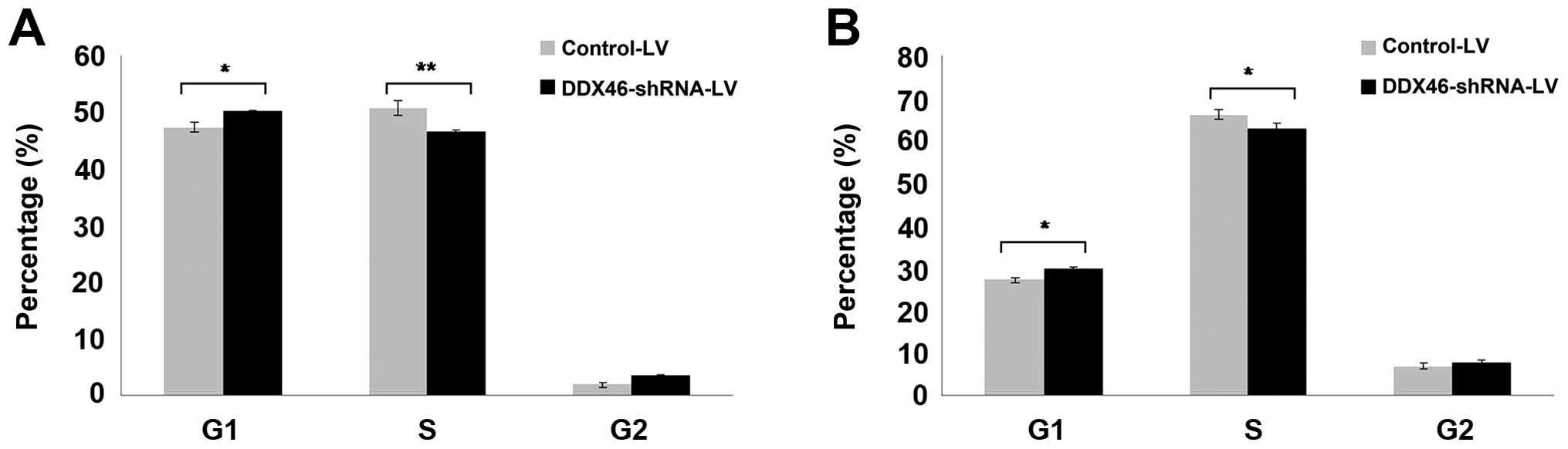
cells. Furthermore, flow cytometry assay indicated that silencing
of DDX46 expression markedly increased the percentage of G1-phase
cells and decreased the percentage of S-phase cells in TE-1
(Fig. 7A) and Eca-109 (Fig. 7B) cells, the cells were arrested in
the G1-phase after knockdown of DDX46. The above data indicated
that DDX46 was involved in the regulation of ESCC cell apoptosis
and cell cycle progression.
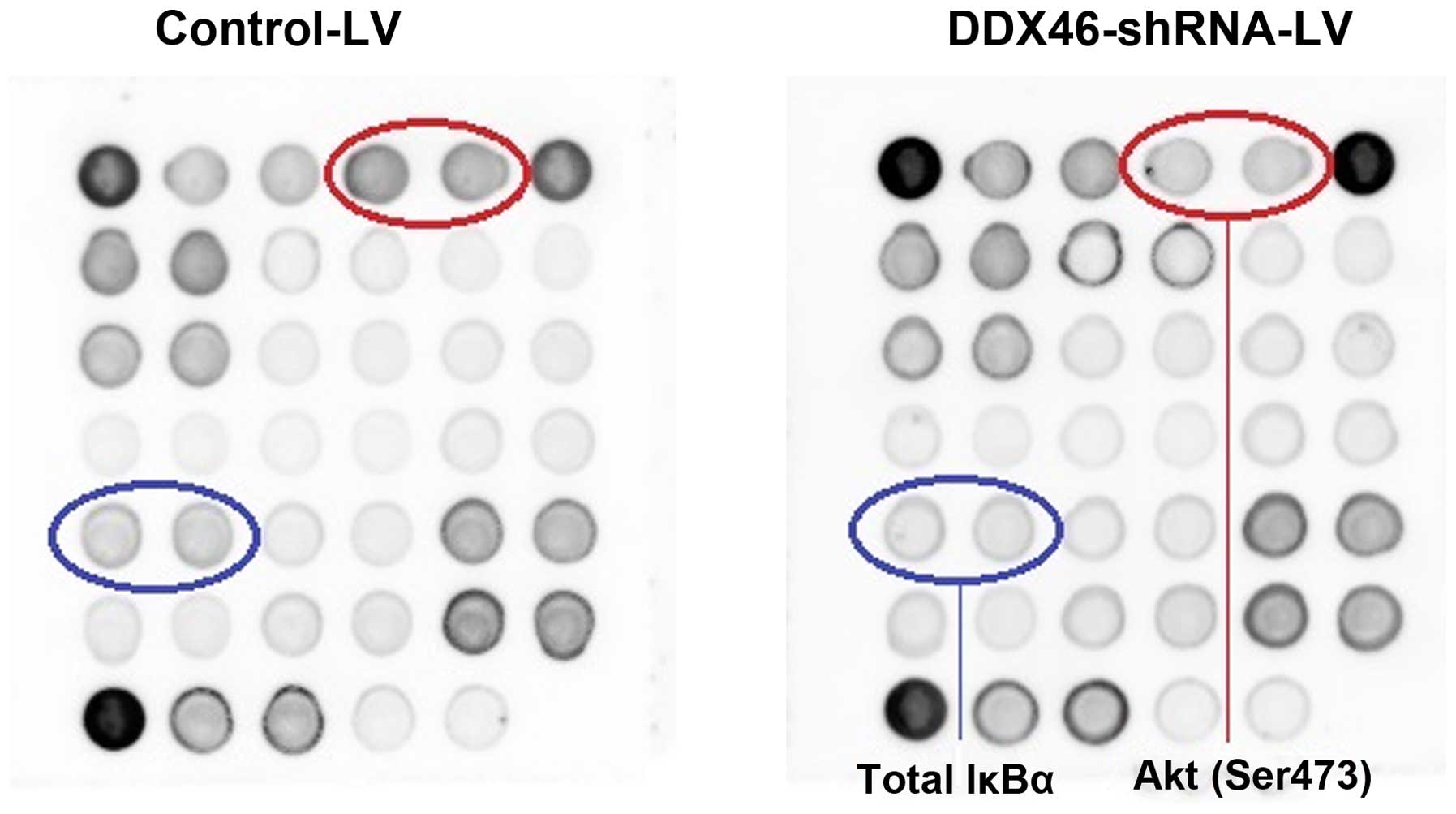
To further illuminate the molecular mechanisms by
which DDX46 regulated ESCC cell apoptosis, the stress and apoptosis
signaling antibody array kit was used to detect the changes of
signaling molecules in TE-1 cells with or without DDX46 knockdown.
As shown in Fig. 8, the
phosphorylation of Akt (Ser473) and IκBα (total) were downregulated
in DDX46-silenced cells, respectively. The data indicated that
DDX46 knockdown could significantly induce apoptosis of DDX46 cells
via blockade of Akt and IκBα activations.
Discussion
In this study, we found that DDX46 protein
expression in clinical specimens from ESCC tissues was upregulated
compared with matched adjacent non-tumor tissues; the expression of
DDX46 mRNA was significantly higher in all of the tested human ESCC
cell lines compared to the normal esophageal epithelium cell
Het-1A. In addition, silencing DDX46 inhibited proliferation and
induced apoptosis of ESCC cells. These data indicated that DDX46
overexpression played essential roles in the tumorigenicity of
human ESCC.
DEAD-box RNA helicases was first described in 1989
(17), since then it has gradually
formed the largest helicases family (7). Members of this family share a
conserved core containing nine conserved motifs, including the
characteristic D-E-A-D (Asp-Glu-Ala-Asp, or DEAD in one-letter
code) motif, which gave the protein family its name; these motifs
confer the ATP hydrolysis and RNA unwinding activities that have
established them as RNA helicases (8). DEAD-box RNA helicases play key, and
often essential, roles in RNA metabolism. Overexpression of these
helicases can indicate increased mistakes during RNA and protein
production, possibly leading to carcinogenesis. A more recent study
of breast cancer gene expression and tissue microarrays, which
showed that DDX1 mRNA overexpression and elevated cytoplasmic DDX1
protein were associated with early recurrence and suggested that
DDX1 may be an independent prognostic marker for early recurrence
in breast cancer (18). DDX6 was
first suggested to be involved in tumor development through its
cloning as an RNA helicase gene from a human lymphoid cell line
with chromosomal breakpoint 11q23.3 (19). Subsequent reports demonstrated that
DDX6 protein was elevated in glioblastoma, rhabdosarcoma, lung
cancer (20), colorectal carcinoma
(21,22) and hepatitis C virus-related
hepatocellular carcinoma (23) as
compared with normal tissue. The first report of aberrant DDX5
expression in cancer came from a study which showed that DDX5 was
overexpressed and abnormally modified by polyubiquitylation in
colorectal carcinoma (24). This
was followed by several other reports showing DDX5, and in several
cases also DDX17, overexpression in a series of cancers including
colon (25,26), prostate (27), breast (28,29),
cutaneous squamous cell carcinoma (30) and glioma (31). DDX39 was overexpressed in lung
squamous cell carcinomas (32).
Recent proteomic study of gastrointestinal stromal tumors correlate
to elevated DDX39 levels with an increased risk of metastasis
following surgery (33). However,
the expression of DDX46 in human cancer tissues remains largely
unknown, only Li and colleagues (16) reported on colorectal carcinoma. For
the first time, our study assessed DDX46 expression in human ESCC
tissues by IHC. Our results showed that the cellular localization
of DDX46 was focused in the nucleus of cells, and DDX46 was more
strongly and highly stained in ESCC tissues than in matched normal
tissues. In vitro, silencing DDX46 inhibited cell
proliferation and colony formation by inducing cell apoptosis and
G1-phase cell cycle arrest. These results are similar to a previous
study (16), indicating a common
role of DDX46 in cancer cell proliferation.
Although numerous reports implied an involvement of
DEAD-box RNA helicases in cancer development, the precise mechanism
of its contribution to cancer was concealed at the time. Forced
overexpression of DDX6 in colorectal carcinoma cells stimulated Tcf
transcriptional activity and expression of Wnt pathway target genes
(34). DDX5 and DDX17 regulated the
β-catenin pathway by formed complexes with β-catenin and promoted
the ability of β-catenin to activate gene transcription (26). Some DEAD-box RNA helicases were
coactivators of tumor development transcription factors and
regulated the expression of downstream molecules. For instance,
DDX5 selectively activated p53-dependent p21 expression, thereby
influencing entry into cell cycle arrest and apoptosis after DNA
damage (35). Whether DDX46
regulates these signaling pathways or others needs further
exploration. Herein, we found that DDX46 may play an important role
in ESCC cells proliferation by inducing cell apoptosis and cell
cycle arrest in vitro. Silencing DDX46 significantly
impaired ESCC cell growth, blocked G0-G1 phase progression, and
increased the number of apoptotic cells in both TE-1 and Eca-109
cells. The stress and apoptosis signaling antibody array displayed
that DDX46 knockdown significantly reduced the phosphorylation of
Akt and IκBα. Akt plays a central role in tumorigenesis, its
function is dictated by phosphorylation status (36). Suppressed Akt activity could inhibit
proliferation and induce apoptosis (37). Activated Akt could stimulate the
phosphorylation and impact various downstream targets, including
IκBα (38), which is endogenous
inhibitor of NF-κB. NF-κB is a nuclear transcription factor that
regulates expression of a large number of genes that are critical
for the regulation of apoptosis and tumorigenesis. In its inactive
form, NF-κB is sequestered in the cytoplasm, bound by members of
the IκB family of inhibitor proteins, which include IκBα. The
various stimuli that activate NF-κB cause phosphorylation of IκBα,
which is followed by its ubiquitination and subsequent degradation.
This results in the exposure of the nuclear localization signals on
NF-κB subunits and the subsequent translocation of the molecule to
the nucleus. Therefore, cell apoptosis due to knockdown of DDX46
could be explained, at least in part, by inhibition of the activity
of NF-κB via the blockade of Akt and IκBα. However, the direct link
between DDX46 and the NF-κB signaling need to be investigated
further.
In conclusion, our observations demonstrated that
DDX46 was significantly upregulated in ESCC tissues and cell lines,
DDX46 knockdown led to decreased proliferation and increased
apoptosis in ESCC cells, which was mediated via decreased
endogenous IκBα phosphorylation as well as negative regulation of
NF-κB signaling. We hope that this study will provide the basis for
novel strategy in the development of effective ESCC diagnosis and
treatment. Further study is needed to clarify the specific
molecular mechanisms for the role of DDX46 in the development and
regulation of ESCC.
Acknowledgments
This study was supported by a grant (No.
1308RJZA123) from the Natural Science Foundation of Gansu Province,
China.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen W, Zheng R, Zeng H, Zhang S and He J:
Annual report on status of cancer in China, 2011. Chin J Cancer
Res. 27:2–12. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siewert JR and Ott K: Are squamous and
adenocarcinomas of the esophagus the same disease? Semin Radiat
Oncol. 17:38–44. 2007. View Article : Google Scholar
|
|
4
|
Gao YB, Chen ZL, Li JG, Hu XD, Shi XJ, Sun
ZM, Zhang F, Zhao ZR, Li ZT, Liu ZY, et al: Genetic landscape of
esophageal squamous cell carcinoma. Nat Genet. 46:1097–1102. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Moorcraft SY and Chau I: Investigational
therapies targeting the ErbB family in oesophagogastric cancer.
Expert Opin Investig Drugs. 23:1349–1363. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jankowsky E: RNA helicases at work:
Binding and rearranging. Trends Biochem Sci. 36:19–29. 2011.
View Article : Google Scholar :
|
|
7
|
Linder P and Fuller-Pace F: Happy
birthday: 25 years of DEAD-box proteins. Methods Mol Biol.
1259:17–33. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fuller-Pace FV: DEAD box RNA helicase
functions in cancer. RNA Biol. 10:121–132. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Heerma van Voss MR, Vesuna F, Trumpi K,
Brilliant J, Berlinicke C, de Leng W, Kranenburg O, Offerhaus GJ,
Bürger H, van der Wall E, et al: Identification of the DEAD box RNA
helicase DDX3 as a therapeutic target in colorectal cancer.
Oncotarget. 6:28312–28326. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bol GM, Vesuna F, Xie M, Zeng J, Aziz K,
Gandhi N, Levine A, Irving A, Korz D, Tantravedi S, et al:
Targeting DDX3 with a small molecule inhibitor for lung cancer
therapy. EMBO Mol Med. 7:648–669. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xu YZ and Query CC: Competition between
the ATPase Prp5 and branch region-U2 snRNA pairing modulates the
fidelity of spliceosome assembly. Mol Cell. 28:838–849. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kosowski TR, Keys HR, Quan TK and Ruby SW:
DExD/H-box Prp5 protein is in the spliceosome during most of the
splicing cycle. RNA. 15:1345–1362. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hozumi S, Hirabayashi R, Yoshizawa A,
Ogata M, Ishitani T, Tsutsumi M, Kuroiwa A, Itoh M and Kikuchi Y:
DEAD-box protein Ddx46 is required for the development of the
digestive organs and brain in zebrafish. PLoS One. 7:e336752012.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hirabayashi R, Hozumi S, Higashijima S and
Kikuchi Y: Ddx46 is required for multi-lineage differentiation of
hematopoietic stem cells in zebrafish. Stem Cells Dev.
22:2532–2542. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Abdelhaleem M, Maltais L and Wain H: The
human DDX and DHX gene families of putative RNA helicases.
Genomics. 81:618–622. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li M, Ma Y, Huang P, Du A, Yang X, Zhang
S, Xing C, Liu F and Cao J: Lentiviral DDX46 knockdown inhibits
growth and induces apoptosis in human colorectal cancer cells.
Gene. 560:237–244. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Linder P, Lasko PF, Ashburner M, Leroy P,
Nielsen PJ, Nishi K, Schnier J and Slonimski PP: Birth of the
D-E-A-D box. Nature. 337:121–122. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Germain DR, Graham K, Glubrecht DD, Hugh
JC, Mackey JR and Godbout R: DEAD box 1: A novel and independent
prognostic marker for early recurrence in breast cancer. Breast
Cancer Res Treat. 127:53–63. 2011. View Article : Google Scholar
|
|
19
|
Lu D and Yunis JJ: Cloning, expression and
localization of an RNA helicase gene from a human lymphoid cell
line with chromosomal breakpoint 11q23.3. Nucleic Acids Res.
20:1967–1972. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Akao Y, Marukawa O, Morikawa H, Nakao K,
Kamei M, Hachiya T and Tsujimoto Y: The rck/p54 candidate
proto-oncogene product is a 54-kilodalton D-E-A-D box protein
differentially expressed in human and mouse tissues. Cancer Res.
55:3444–3449. 1995.PubMed/NCBI
|
|
21
|
Hashimoto K, Nakagawa Y, Morikawa H, Niki
M, Egashira Y, Hirata I, Katsu K and Akao Y: Co-overexpression of
DEAD box protein rck/p54 and c-myc protein in human colorectal
adenomas and the relevance of their expression in cultured cell
lines. Carcinogenesis. 22:1965–1970. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nakagawa Y, Morikawa H, Hirata I, Shiozaki
M, Matsumoto A, Maemura K, Nishikawa T, Niki M, Tanigawa N, Ikegami
M, et al: Overexpression of rck/p54, a DEAD box protein, in human
colorectal tumours. Br J Cancer. 80:914–917. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Miyaji K, Nakagawa Y, Matsumoto K, Yoshida
H, Morikawa H, Hongou Y, Arisaka Y, Kojima H, Inoue T, Hirata I, et
al: Overexpression of a DEAD box/RNA helicase protein, rck/p54, in
human hepatocytes from patients with hepatitis C virus-related
chronic hepatitis and its implication in hepatocellular
carcinogenesis. J Viral Hepat. 10:241–248. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Causevic M, Hislop RG, Kernohan NM, Carey
FA, Kay RA, Steele RJ and Fuller-Pace FV: Overexpression and
polyubiquitylation of the DEAD-box RNA helicase p68 in colorectal
tumours. Oncogene. 20:7734–7743. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yang L, Lin C and Liu ZR: Phosphorylations
of DEAD box p68 RNA helicase are associated with cancer development
and cell proliferation. Mol Cancer Res. 3:355–363. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shin S, Rossow KL, Grande JP and Janknecht
R: Involvement of RNA helicases p68 and p72 in colon cancer. Cancer
Res. 67:7572–7578. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Clark EL, Coulson A, Dalgliesh C, Rajan P,
Nicol SM, Fleming S, Heer R, Gaughan L, Leung HY, Elliott DJ, et
al: The RNA helicase p68 is a novel androgen receptor coactivator
involved in splicing and is overexpressed in prostate cancer.
Cancer Res. 68:7938–7946. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wortham NC, Ahamed E, Nicol SM, Thomas RS,
Periyasamy M, Jiang J, Ochocka AM, Shousha S, Huson L, Bray SE, et
al: The DEAD-box protein p72 regulates ERalpha-/oestrogen-dependent
transcription and cell growth, and is associated with improved
survival in ERalpha-positive breast cancer. Oncogene. 28:4053–4064.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mazurek A, Luo W, Krasnitz A, Hicks J,
Powers RS and Stillman B: DDX5 regulates DNA replication and is
required for cell proliferation in a subset of breast cancer cells.
Cancer Discov. 2:812–825. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang SJ, Zhang C, You Y and Shi CM:
Overexpression of RNA helicase p68 protein in cutaneous squamous
cell carcinoma. Clin Exp Dermatol. 37:882–888. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang R, Jiao Z, Li R, Yue H and Chen L:
p68 RNA helicase promotes glioma cell proliferation in vitro and in
vivo via direct regulation of NF-κB transcription factor p50.
Neuro-oncol. 14:1116–1124. 2012. View Article : Google Scholar
|
|
32
|
Sugiura T, Nagano Y and Noguchi Y: DDX39,
upregulated in lung squamous cell cancer, displays RNA helicase
activities and promotes cancer cell growth. Cancer Biol Ther.
6:957–964. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kikuta K, Kubota D, Saito T, Orita H,
Yoshida A, Tsuda H, Suehara Y, Katai H, Shimada Y, Toyama Y, et al:
Clinical proteomics identified ATP-dependent RNA helicase DDX39 as
a novel biomarker to predict poor prognosis of patients with
gastrointestinal stromal tumor. J Proteomics. 75:1089–1098. 2012.
View Article : Google Scholar
|
|
34
|
Lin F, Wang R, Shen JJ, Wang X, Gao P,
Dong K and Zhang HZ: Knockdown of RCK/p54 expression by RNAi
inhibits proliferation of human colorectal cancer cells in vitro
and in vivo. Cancer Biol Ther. 7:1669–1676. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Nicol SM, Bray SE, Black HD, Lorimore SA,
Wright EG, Lane DP, Meek DW, Coates PJ and Fuller-Pace FV: The RNA
helicase p68 (DDX5) is selectively required for the induction of
p53-dependent p21 expression and cell-cycle arrest after DNA
damage. Oncogene. 32:3461–3469. 2013. View Article : Google Scholar
|
|
36
|
Testa JR and Bellacosa A: AKT plays a
central role in tumorigenesis. Proc Natl Acad Sci USA.
98:10983–10985. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Mandal M, Kim S, Younes MN, Jasser SA,
El-Naggar AK, Mills GB and Myers JN: The Akt inhibitor KP372–1
suppresses Akt activity and cell proliferation and induces
apoptosis in thyroid cancer cells. Br J Cancer. 92:1899–1905. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Fresno Vara JA, Casado E, de Castro J,
Cejas P, Belda-Iniesta C and González-Barón M: PI3K/Akt signalling
pathway and cancer. Cancer Treat Rev. 30:193–204. 2004. View Article : Google Scholar : PubMed/NCBI
|