Introduction
Lung cancer is the most common and fatal cancer
worldwide (1). In China, lung
cancer has been the leading cause of cancer-related deaths since
the 1990s (2). Recent trend
analyses indicate that the incidence of lung cancer has increased
over the last 20 years, and it is predicted that the disease burden
may continue to increase if no effective action is taken to control
lung cancer (3). In addition, the
lungs are the second most frequent site of metastasis, and
metastases to the lung complicate the course of as many as 40% of
other malignancies (4). For the
treatment of lung cancer, surgical resection, consisting of a
lobectomy and complete lymph node dissection, has long been
considered the standard treatment for resectable tumors and offers
the best curative chance for long-term survival (5). However, it is estimated that only a
third of patients with non-small cell lung cancer (NSCLC) are
suitable for curative resection (6). Therefore, non-invasive local
treatments that makes it possible to avoid resection of the
functioning parenchyma or prolonged general anaesthesia represent a
key point in the management of patients who are not eligible for
surgery (7).
As a minimally invasive approach for the local
treatment of lung cancer, radiofrequency thermal ablation (RFA) has
recently received much attention. RFA is a new image-guided
percutaneous technique that utilizes hyperthermic energy (8). Studies indicate that lung tumors are
ideal targets for RFA since the surrounding air in the adjacent
normal lung parenchyma provides an insulating effect, concentrating
the RF energy within the tumor tissue (9). The major problem with RFA treatment is
that thermal destruction may occur before achieving complete tumor
destruction (10). According to a
previous study, lung RFA is associated with increased rates of
local recurrence in tumors with large volumes since it is difficult
to reach sufficiently high temperatures further away from the heat
source, particularly in the treatment of larger tumors (11). Moreover, the rich blood supply of
the tumor is another cause for local recurrence, since an abundant
blood supply may help to dissipate the thermal energy and weaken
the ablation effect (12). A
greater number of clinical cases concerned with the over-growth of
residual tumors after RF ablation have been reported (11,13).
Ke et al confirmed that low RFA temperatures at target sites
could facilitate the rapid progression of residual hepatic VX2
carcinomas (14). Cumulative
evidence has demonstrated that residual tumors present after RFA
may exhibit an aggressive phenotype with an unfavorable prognosis,
eventually leading to the deterioration of the patient overall
condition.
However, the specific molecular mechanisms by which
overproliferation of residual lung tumor cells occurs following RFA
are still unclear. Hypoxia-inducible factor-1α (HIF-1α), a key
transcriptional regulator, plays a central role in the adaptation
of tumor cells to hypoxia by activating the transcription of genes
that regulate several biological processes including angiogenesis,
cell proliferation and migration (15). In our pervious study, we found that
HIF-1α can regulate the expression of multiple cytokines, such as
vascular endothelial growth factor-A (VEGF-A) (16), while promoting the proliferation and
angiogenesis potential of small cell lung cancers (SCLCs) (17). Heat-shock proteins (HSPs) are known
to serve as protein chaperones that assist in protein folding,
assembly, degradation and translocation. HSP70 is a member of the
HSP family, and it is constitutively expressed at low levels in
most tissues (18). The expression
of HSP70 is also significantly upregulated under thermal
stimulation (19). Previous studies
indicate that HSP70 interferes with the signaling pathways and
cellular responses to hypoxic stress; HIF-1α stability is
influenced by HSP70, which forms a long-lasting complex with HIF-1α
to increase the lifespan of HIF-1α (20). As far as the regulatory mechanism,
Yeh et al showed that PI3K/Akt contributes to promoting
HIF-1α expression by upregulating the expression of HSP70 (21). In the present study, we hypothesized
that insufficient RFA promoted the proliferation and angiogenesis
potential of residual lung cancer cells, which plays an important
role in the rapid proliferation of residual tumor cells after RFA.
Then, we investigated whether local hyperthermia could change the
microenvironment of ablated tumor tissues and the biological
characteristics of residual tumor cells. We found that these cells
exhibited rapid proliferation and upregulated angiogenesis
potential through a HSP70/HIF-1α-dependent mechanism.
Materials and methods
Materials
The PI3K/Akt inhibitor wortmannin (22), the HSP70 inhibitor VER-155008
(23)
5′-O-(4-cyanobenzyl)-8-[(3,4-dichlorobenzyl)amino]adenosine, the
HIF-1α inhibitor YC-1 (24) and
3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetra-zolium bromide (MTT)
were purchased from sigma-Aldrich (St. Louis, MO, USA). TRIzol
reagent was purchased from Invitrogen Corp. (Carlsbad, CA, USA).
RIPA lysis buffer was purchased from Beyotime Institute of
Biotechnology, China. Anti-HIF-1α used at a 1:500 dilution,
anti-HSP70 used at a 1:1,000 dilution, and anti-Akt used at a
1:1,000 dilution were purchased from Cell Signaling Technology
(Beverly, MA, USA). Anti-CD34 used at a 1:50 dilution was purchased
from Wuhan Boster Biological Engineering Technology Ltd., Co.
(Wuhan, China).
Cell lines and cell culture
According to our previous study (16,17),
the human NSCLC NCI-H1650 cell line was maintained in RPMI-1640
medium (sigma-Aldrich) supplemented with 10% fetal bovine serum
(FBS), 100 U/ml penicillin and 100 µg/ml kanamycin at 37°C
in a humidified atmosphere containing 5% CO2 and 20%
O2. The medium was routinely changed 2–3 days after
seeding. Cells were detached with trypsin/EDTA (Gibco-BRL, Paisley,
UK) and resuspended in a 1:1 solution of serum-free RPMI-1640
medium to a final concentration of ~5×105 cells/10
µl.
Heat treatment of lung cancer cells and
establishment of the sublines
In all of the experiments, the cells were exposed to
hyperthermic stress during the exponential phase in cell culture
plates. The NCI-H1650 cells were seeded into cell culture plates.
After 24, 48 or 72 h of incubation they were exposed to heat
stress. The plates were sealed with parafilm and submerged in a
water bath at the desired temperature for 10 min. Eight
temperatures ranging from 42–70°C were selected: 42, 46, 50, 54,
58, 62, 66 and 70°C. These temperatures match the ablation
temperatures applied during the clinical treatment of lung cancer.
After the hyperthermic treatment, fresh culture medium was added to
each well, and the surviving cells were maintained at 37°C in an
atmosphere containing 5% CO2 and 20% O2.
These cells separately cultured under 8 desired temperature points
were utilized as 8 sublines. One subline was generated from each
heat treatment.
MTT assay for lung cancer cell
viability
According to the study of Kong et al
(25), cells were cultured at a
concentration of 1×104 cells/well in 48-well plates. MTT
solution was added to each well at a final concentration of 0.5
mg/ml and incubated for 4 h. At the end of the incubation, formazan
crystals resulting from MTT reduction were dissolved by addition of
150 ml dimethyl sulfoxide (DMSO)/well. The optical density (OD) was
read at 570 nm, and the average values were determined from
replicate wells.
Real-time PCR analysis of HSP70 and
HIF-1α
Real-time PCR was performed using a SYBR ExScript
RT-PCR kit according to the manufacturer's protocol (Takara
Biotechnology Co., Ltd., Dalian, China) and the iCycler Real-Time
PCR Detection System (Bio-Rad Laboratories, Hercules, CA, USA). All
the RNA samples were extracted and run in duplicate on 96-well
optical PCR plates. The thermal cycling conditions were as follows:
1 cycle of 90.0°C for 10 min, 40 cycles of 95.0°C for 5 sec, 60.0°C
for 30 sec and 81 cycles of 55.0°C for 10 min (with an increase set
point temperature after cycle 2 of 0.5°C). β-actin was used as an
internal control. PCR primer sequences were as follows: HSP70
sense, 5′-CTGACAAGAAGAAGGTGCTGG-3′ and antisense,
5′-AGCAGCCATCAAGAGTCTGTC-3′, HIF-1α sense,
5′-CATCAGCTATTTGCGTGTGAGGA-3′ and antisense,
5′-AGCAATTCATCTGTGCTTTCATGTC-3′.
Western blot analysis
Cells were harvested and analyzed for the expression
of HIF-1α, HSP70, Akt and p-Akt. Briefly, proteins were extracted
by disrupting cells in RIPA lysis buffer, were separated on
polyacrylamide gels, and transferred to polyvinylidene difluoride
(PVDF) membranes. The membranes were then blocked at room
temperature for 1 h with 5% non-fat milk in Tris-buffered saline
containing Tween-20 (TBST). Then, the membranes were incubated with
anti-HIF-1α, anti-HSP70, anti-Akt, anti-phospho-Akt primary
antibodies at 37°C for 2 h. After, the membranes were incubated
with peroxidase-conjugated IgG at room temperature for 1 h. The
membranes were subsequently incubated with goat anti-rabbit
peroxidase-conjugated secondary antibodies, and immunoreactivity
was detected using an enhanced chemiluminescence kit and captured
on X-ray film. β-actin was used as an internal normalization
control.
Establishing xenografts in nude mice and
RFA treatment
Male congenital athymic BALB/c nude mice were
obtained from the Experimental Animal Center of the Shanghai Jiao
Tong University school of Medicine and maintained under
pathogen-free conditions in accordance with established
institutional guidance and approved protocols. All experiments were
carried out using 6- to 8-week-old mice weighing 16–22 g. In
vitro cultured NCI-H1650 cells (1×107) at a final
concentration of ~5×105 cells/10 µl were
suspended in phosphate-buffered saline (PBS) and were
subcutaneously injected into the flank area of mice. After tumors
reached 3–5 mm in diameter, mice were injected with 10% DMSO/PBS, 4
mg/kg twice weekly.
For RFA treatment, we applied the method of Xu et
al (26). RFA was performed
using a bipolar RFA device (Blade Opto-Electronic Technology
Development Co., Ltd., Beijing, China), which is a microRFA probe
with an active tip length of 10 mm. To simulate the clinical
settings of tumor recurrence following RFA, the 'incomplete RFA'
strategy using 180 sec of RFA at 1 W power was applied when the
size of the tumor reached nearly 3.3–3.6 cm3 on the 21st
day of tumor growth. This procedure resulted in incomplete ablation
of the tumors.
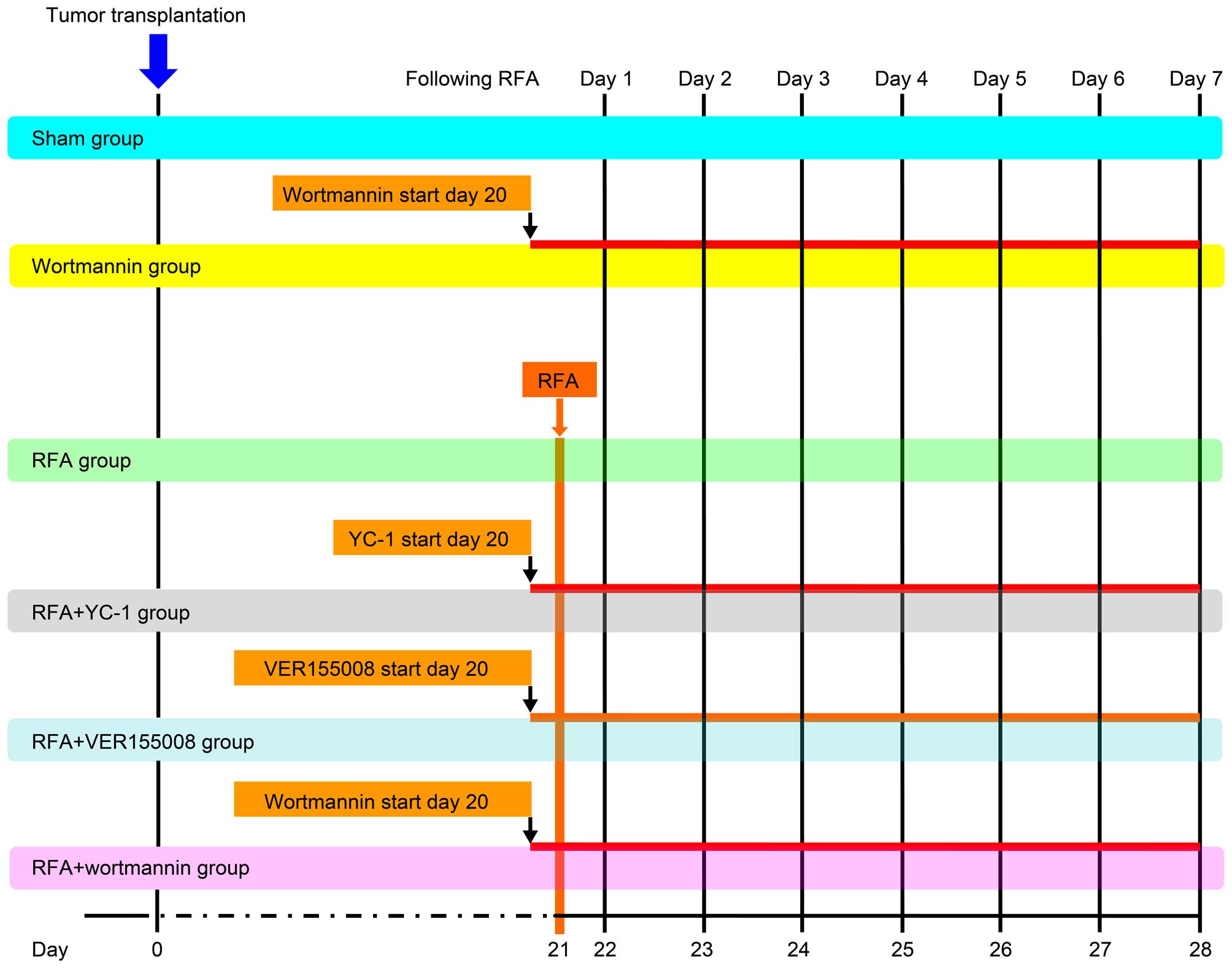
There were a total of 80 mice involved in the
experiment, which were divided into 6 groups: control (sham
puncture using an identical probe without applying energy), RFA,
VER-155008 + RFA (50 µg/day injection), YC-1 + RFA (600
µg/day injection), wortmannin (0.07 mg/day injection) and
wortmannin + RFA groups (0.07 mg/day injection). Each group
contained 10 mice (evenly divided into female and male genders). We
measured the tumor size daily with calipers from days 1–7 following
RFA treatment, and tumor volume was calculated according to the
formula: Volume = width2 × length × 0.5. All treatment
groups and schedules are shown in Fig.
1.
Immunohistochemistry detection for
CD34
Tissue sections of 4 µm were prepared and
endogenous peroxidases were inhibited with 0.3% hydrogen peroxide
in methanol for 30 min. Antigen retrieval was achieved using 0.05%
protease XIV at 37°C for 5 min. Sections were incubated with the
mouse anti-human CD34 primary antibody overnight at 4°C, then the
slides were incubated with rabbit anti-mouse secondary antibody at
room temperature for 45 min. The sections were subsequently
incubated with a streptavidin-biotin-peroxidase complex (Vectastain
ABC kit; Vector Laboratories, Burlingame, CA, USA) at room
temperature for 45 min. The reaction was visualized using chromogen
diaminobenzidine (DAB) for 10 sec. Finally, the slides were
counterstained with hematoxylin, and then mounted and examined on a
Nikon eclipse Ti microscope with a 40× objective. Microvessel
density (MVD) was defined as the number of positively stained
vessels per high-power field of view (HPF).
Statistical analysis
SPSS 13.0 software (SPSS, Inc., Chicago, IL, USA)
was applied to complete data processing. Independent-sample t-tests
were used to evaluate the differences in OD values. All data are
represented as mean ± SD for three independent experiments. Results
were considered statistically significant when the p-value was
<0.05.
Results
In vitro heat treatment generates
NCI-H1650 cell sublines with increased viabilities and
proliferation activities
To detect the potential effect of hyperthermia on
the proliferative activity of lung cancer cells, first we monitored
cell viability at 24, 48 and 72 h after incubation at 42°C for 10
min. We found that the viability of the NCI-H1650 cells after
incubation at 42°C was significantly decreased compared to cells
that did not receive heat treatment; this may be due to cell
apoptosis and death. However, the cellular viability of the
parental cells not undergoing hyperthermia treatment and the cells
cultured at 42°C reached their highest viability at 72 h of
incubation (Fig. 2A). In addition,
the cell viability of NCI-H1650 cells after various heat treatments
was assessed in continuously passaged cells following exposed to 8
different heat conditions denoted in the methods. The cellular
viability of these 8 sublines was tested at 24, 48 and 72 h of
incubation. We found that these sublines exhibited significant
differences in cell viability compared to their parental cells,
particularly NCI-H1650 cells adapted to 54°C, which were
significantly more viable than their parental cells (Fig. 2B and C). We continued to monitor the
differences in proliferation activity between parental lung cancer
cells and these NCI-H1650 cell sublines. After this subline was
established, we cultured the cells for 7 days, drawing a
proliferation curve. From the proliferation curve, we found that
NCI-H1650 cells adapted to 54°C exhibited a significantly higher
proliferation rate compared to the parental cells (Fig. 2D). Therefore, we conclude that
thermal treatment can partially kill NCI-H1650 cells, while
generating sublines with increased viability and proliferation
activity. Therefore, we used the NCI-H1650 subline adapted to 54°C
for our subsequent research.
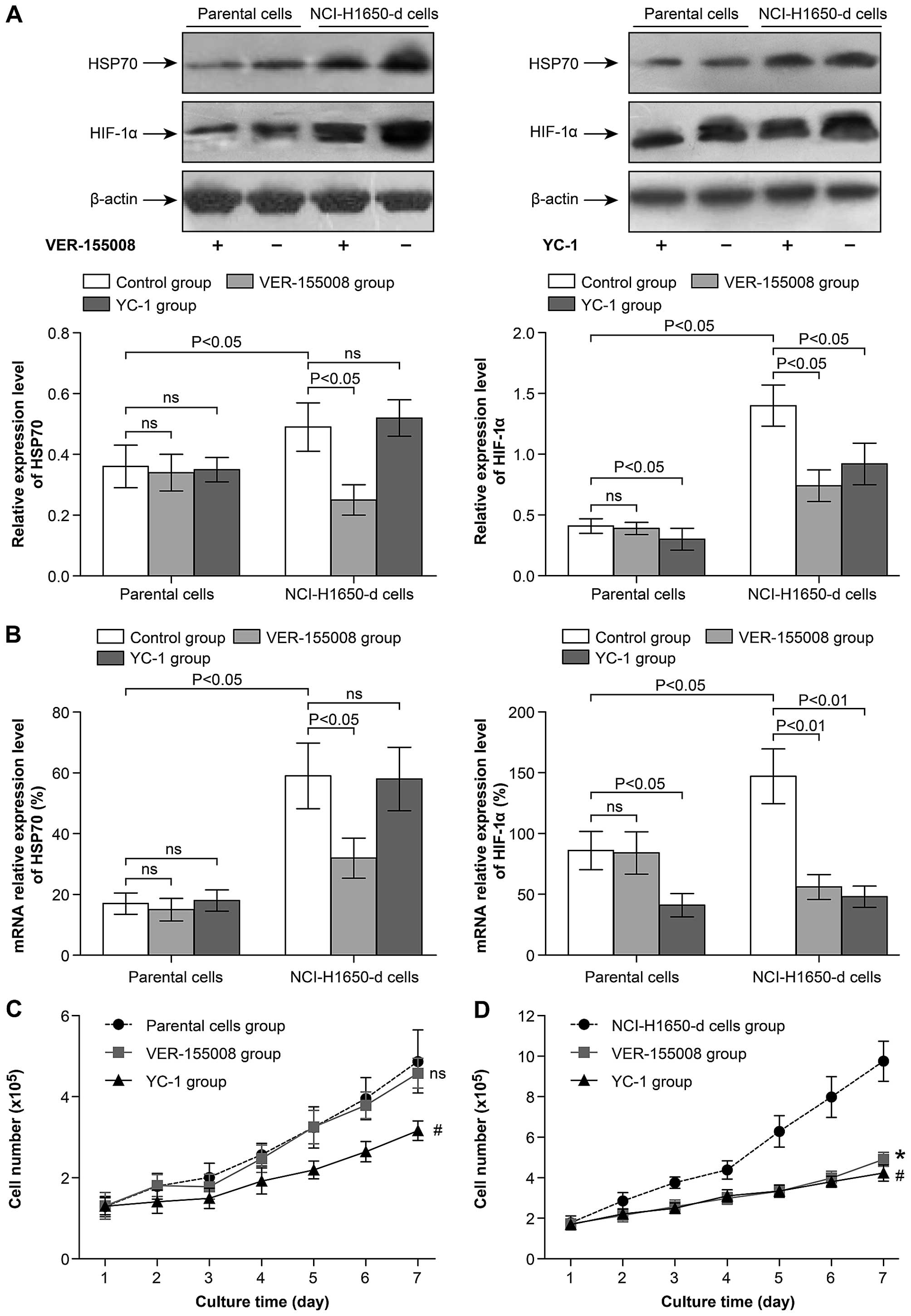
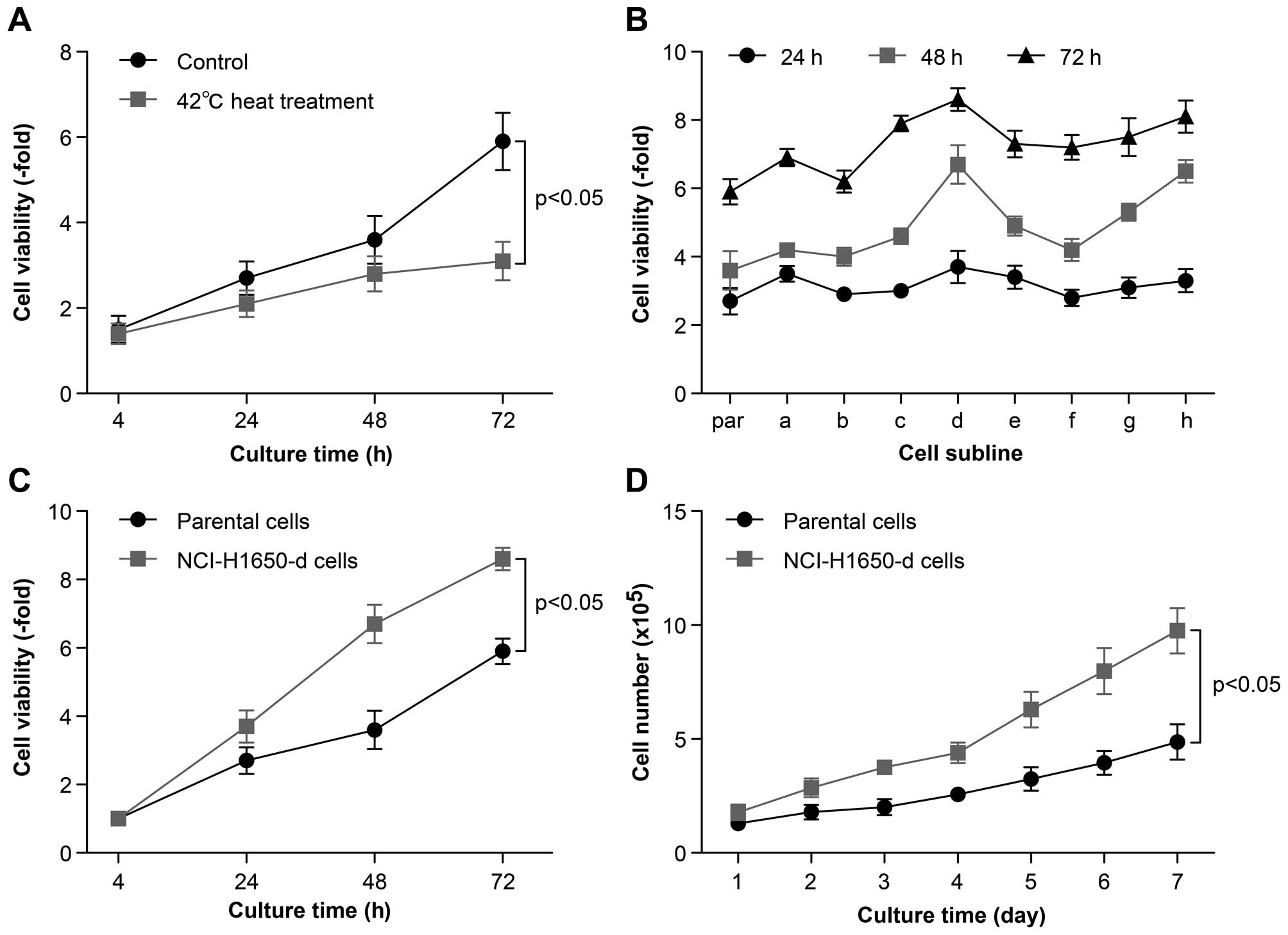 | Figure 2The viability of NCI-H1650 cells and
sublines derived from NCI-H1650 cells after hyperthermia. (A)
NCI-H1650 cells were cultured after 42°C heat treatment. The 24, 48
and 72 h cell viability of NCI-H1650 cells with or without 42°C
heat treatment were measured using MTT assay (p<0.05, control
group vs. 42°C heat treatment group). (B) Eight sublines were
established following 42, 46, 50, 54, 58, 62, 66 and 70°C heat
treatment for 15 min as described in Materials and methods. The 24,
48 and 72 h viability was evaluated by MTT assay after these 8
sublines had been established. par, parental NCI-H1650 cells; a–h,
sublines derived from the NCI-H1650 cells. (C) The 24, 48 and 72 h
viability of parental NCI-H1650 and 54°C heat-adapted NCI-H1650
cells was evaluated by MTT assay (p<0.05, parental NCI-H1650
cells vs. NCI-H1650-d cells). (D) A growth curve of parental
NCI-H1650 and NCI-H1650-d cells was drawn. Data are the
representative results of three independent experiments (p<0.05
from day 2–7 parental NCI-H1650 cells vs. NCI-H1650-d cells). |
HSP70/HIF-1α are involved in regulating
the viability and proliferation activity of NCI-H1650 sublines
We further investigated the differences between
heat-adapted strains and parental lung cancer cells. Western
blotting demonstrated that the protein levels of HSP70 and HIF-1α
were upregulated in the NCI-H1650 cells adapted to 54°C compared to
the parental cells (Fig. 3A). At
the same time, the mRNA levels of HSP70 and HIF-1α were also
increased in the heat-adapted cells compared to the parental cells
(Fig. 3B). The HSP70 inhibitor
VER-155008 significantly decreased the protein and mRNA expression
of HSP70 and HIF-1α in the heat-adapted cells, but did not inhibit
the expression in the parental cells (Fig. 3A and B). Subsequently, we found that
treatment with VER-155008 or YC-1 significantly inhibited the
proliferation activity of the NCI-H1650 heat-adapted cells
(Fig. 3D). However YC-1, but not
VER-155008, inhibited the proliferation activity of the parental
NCI-H1650 cells (Fig. 3C). These
data indicate that the proliferation activity of the heat-adapted
NCI-H1650 cells was regulated by HSP70/HIF-1α, but the
proliferation activity of the parental cells was not linked to
HSP70.
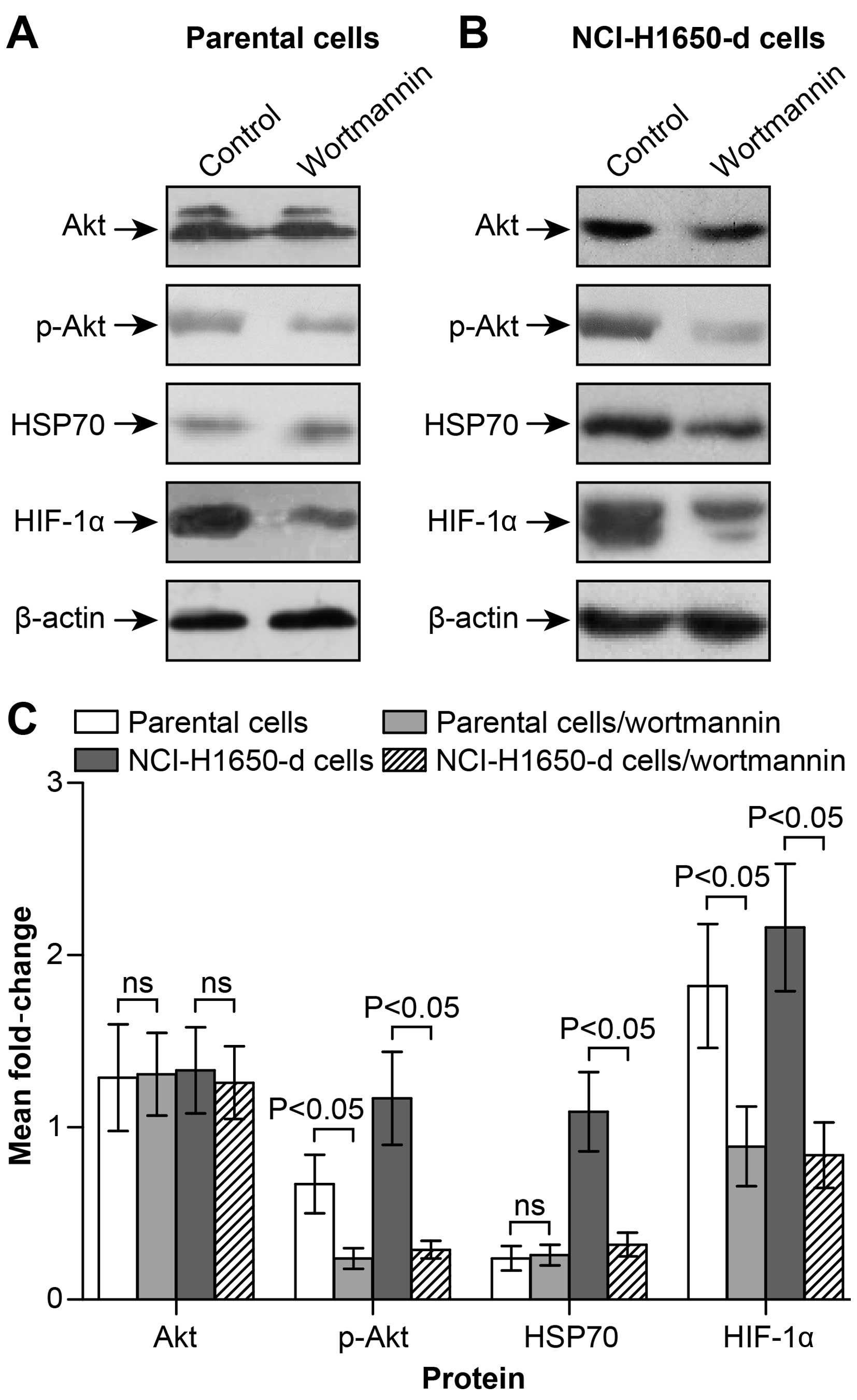
HSP70/HIF-1α expression is regulated by
the PI3K/Akt signaling pathway in heat-adapted NCI-H1650 cells
Western blot detection and semi-quantitative
analyses demonstrated that applying specific inhibitors of PI3K/Akt
(wortmannin) inhibited the expression of Akt phosphorylated active
form-p-Akt, but had little effect on the total expression of Akt
protein (Fig. 4A–C). Wortmannin
intervention also revealed the inhibition of HSP70 or HIF-1α
expression in the heat-adapted cells (Fig. 4B and C). However, for the parental
NCI-H1650 cell line, inhibiting PI3K/Akt signaling pathway
significantly inhibited HIF-1α protein expression (Fig. 4A and C). However, inhibiting
PI3K/Akt did not inhibit HSP70 expression at the protein level in
the parental cells (Fig. 4B). We
conclude that PI3K/Akt contributes to the reduction in HIF-1α
expression by inhibiting expression of HSP70 in heat-adapted cell
lines. However, for the parental lung cancer cells, PI3K/Akt
signaling directly regulated HIF-1α expression, and this biological
process was not associated with HSP70.
HSP70/HIF-1α are involved in the
proliferation and angiogenesis of residual lung carcinomas
following incomplete RFA
At day 20 post-tumor implantation, no differences in
tumor size were observed, and the size of tumors in each group was
3.3–3.6 cm3. Tumors were then subjected to RFA treatment
(Fig. 5A). From days 1–7 following
RFA treatment, the tumor size was daily measured. According to the
slope of the growth curve, animals subjected to RFA treatment
showed a significantly increased growth rate compared to the
control group. These results indicate that RFA treatment may induce
residual tumor growth (Fig. 5B).
From day 1–7 following RFA treatment, there was a significant size
reduction in tumors in the VER-155008 + RFA and YC-1 + RFA groups
compared with the RFA only group (Fig.
5B). These data indicate that HSP70/HIF-1α is involved in
residual tumor growth.
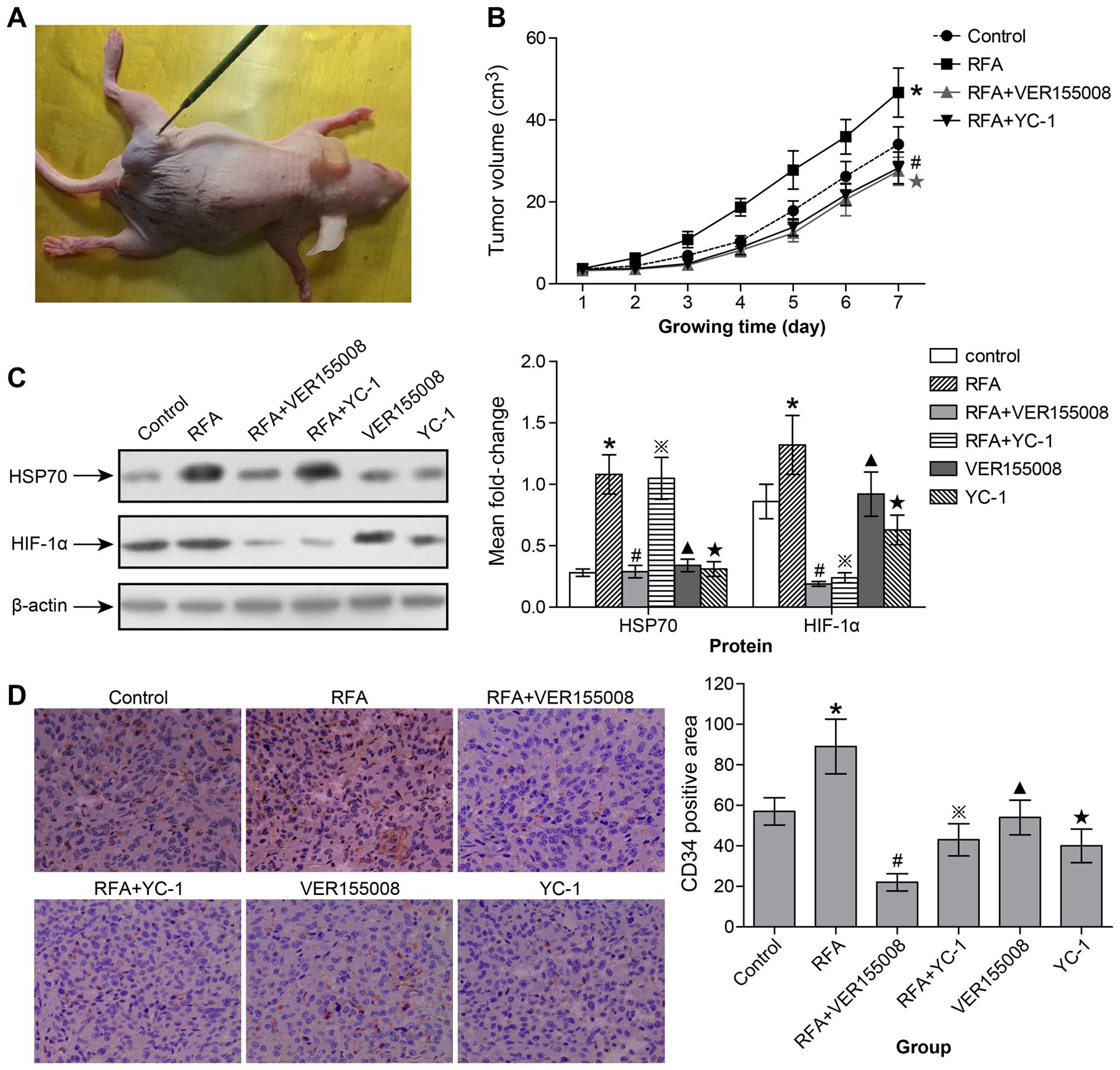 | Figure 5HSP70/HIF-1α are involved in
proliferation and angiogenesis of subcutaneously transplanted
tumors following incomplete radiofrequency ablation. (A) After the
subcutaneously transplanted tumor had formed, the RFA probe was
inserted into the tumor tissue. (B) Growth curve of subcutaneously
transplanted tumors in control, RFA, VER-155008 + RFA and YC-1 +
RFA group (*p<0.05 from day 3–7 control group vs. RFA
group; ★p<0.05 from day 3–7 RFA group vs. VER-155008
+ RFA group; #p<0.05 from day 3–7 RFA group vs. YC-1
+ RFA group). (C) In the control, RFA, VER-155008 + RFA, YC-1 +
RFA, VER-155008 alone and YC-1 alone groups, HSP70 and HIF-1α
expression in subcutaneously transplanted tumors was measured by
western blot and semi-quantitative analyses (*control
group vs. RFA group: HSP70, p<0.05; HIF-1α, p<0.05.
#RFA group vs. VER-155008 + RFA group: HSP70, p<0.05;
HIF-1α, p<0.01. ྿RFA group vs. YC-1 + RFA group:
HSP70, ns; HIF-1α, p<0.01. ▲Control group vs.
VER-155008 group: HSP70, ns; HIF-1α, ns. ★Control group
vs. YC-1 group: HSP70, ns; HIF-1α, p<0.05). (D) At day 7
following RFA treatment, the numbers of new microvessels marked
with CD34 expression in the subcutaneous tumors were detected by
immunohistochemistry analysis and quantified by performing counts
of 10 random fields at a magnification of ×400
(*p<0.05 control group vs. RFA group;
#p<0.05 RFA group vs. VER-155008 + RFA group;
྿p<0.05 RFA group vs. YC-1 + RFA group;
▲ns control group vs. VER-155008 group;
★p<0.05 control group vs. YC-1 group). ns, no
significance. |
At day 7 following RFA treatment, tumors were
resected for western blot analysis. We found that HSP70 and HIF-1α
were significantly upregulated in the RFA group when compared to
the control group (Fig. 5C). After
VER-155008 intervention, HSP70 and HIF-1α were both inhibited;
expression in the VER-155008 + RFA group was significantly
decreased when compared with the RFA group (Fig. 5C). However, after YC-1 intervention,
HIF-1α expression was inhibited, but HSP70 expression did not
significantly change (Fig. 5C).
Thus, we conclude that the HIF-1α expression may be regulated by
HSP70 in lung carcinomas following incomplete RFA. However, in the
VER-155008 group, without RFA treatment, HSP70 and HIF-1α
expression showed little change compared with the control group.
However, in the YC-1 group, HIF-1α expression was inhibited but
HSP70 expression did not significantly change compared with the
control group (Fig. 5C).
At day 7 following RFA treatment, the tumors were
resected and MVD (CD34-positive expression) was detected by
immunohistochemistry staining to analyze angiogenesis potential
(Fig. 5D). We found that RFA
treatment significantly stimulated the angiogenesis potential of
tumors compared to tumors not treated with RFA (Fig. 5D). However, with VER-155008 or YC-1
intervention to inhibit HSP70 and HIF-1α, respectively, the
angiogenesis potential of the tumor was also significantly
inhibited; MVD was attenuated in the VER-155008 + RFA and the YC-1
+ RFA groups compared with the RFA group. However, in the
VER-155008 group, MVD expression did not significantly change,
while it was significantly inhibited in the YC-1 group compared to
the control group (Fig. 5D).
Therefore, HIF-1α expression appeared to be regulated by HSP70,
while its expression inhibited the angiogenesis and proliferation
of residual lung carcinomas following incomplete RFA. However, in
tumors not treated with RFA, HSP70 did not appear to be involved in
this regulatory process.
Targeting the PI3K/Akt signaling pathway
inhibits tumor growth and HSP70/HIF-1α expression after RFA
treatment in nude mice
Considering the potential role of the PI3K/Akt
signaling pathway in HSP70/HIF-1α expression, we wanted to better
understand the involvement of Akt in the stability regulation of
HSP70/HIF-1α. To confirm that HSP70/HIF-1α downregulation occurs by
interfering with PI3K/Akt signaling and to exclude potential
side-effects of inhibitory drugs, we applied a specific inhibitor
of PI3K/Akt signaling, called wortmannin. Following RFA treatment,
the tumor size was again measured from day 1–7 and resected at day
7 for western blot analysis. We found that the combination of
wortmannin intervention with RFA treatment significantly decreased
HSP70 and HIF-1α expression (Fig.
6A). At the same time, a significant reduction in the size of
the tumor was observed in the wortmannin + RFA group compared with
the RFA group over the course of the 7 days of observation
(Fig. 6B). However, in the
wortmannin group we found that the HIF-1α expression level and
tumor volume were significantly decreased, while HSP70 expression
was largely unaffected compared to the control group (Fig. 6A and B). Thus, we suspected that
following incomplete RFA treatment, the PI3K/Akt signaling pathway
may play a central role in regulating HSP70/HIF-1α expression,
which affects the growth of the residual tumor. However, in tumors
not undergoing RFA treatment, PI3K/Akt signaling may directly
regulate HIF-1α expression, while heat-induced HSP70 is not
involved.
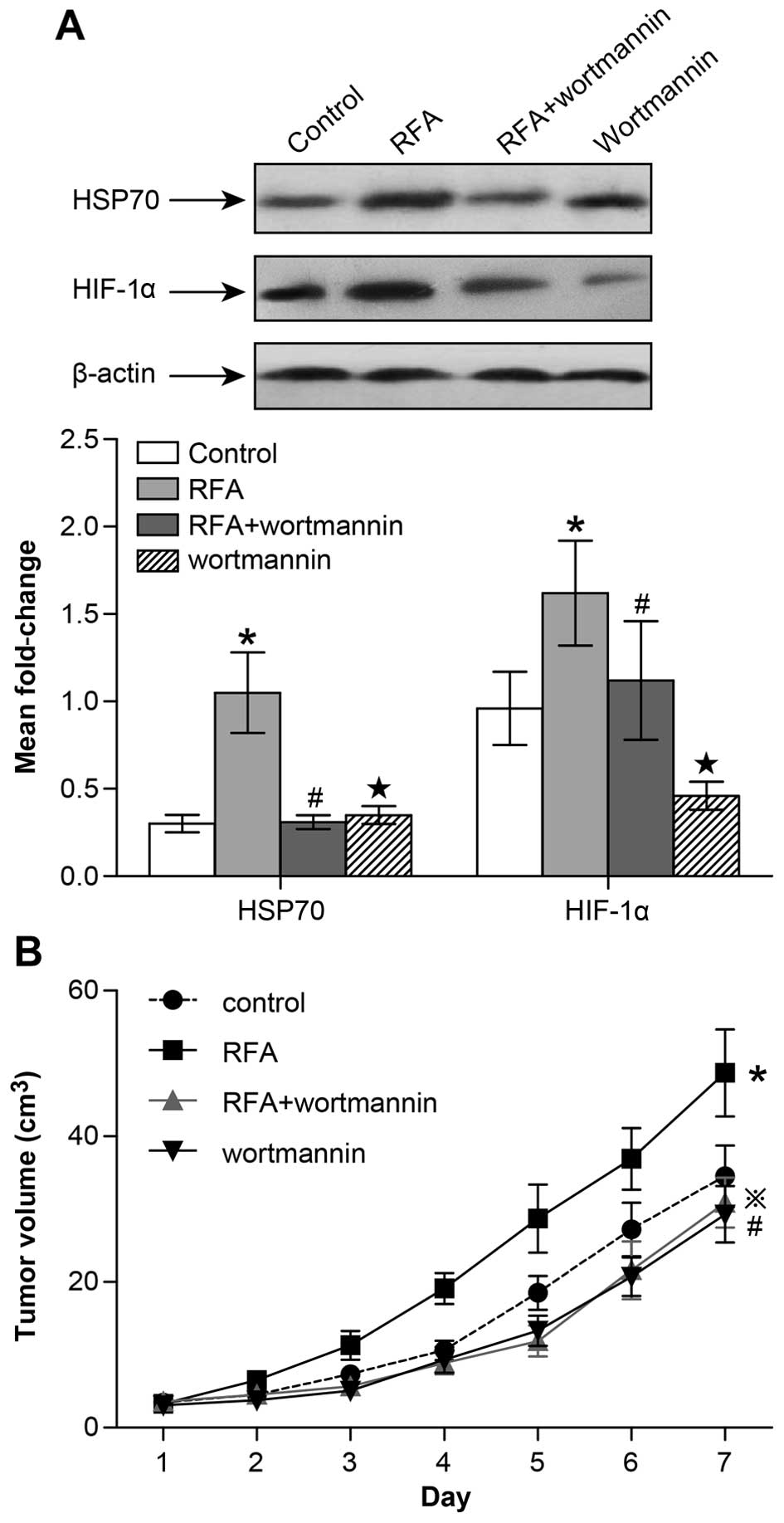 | Figure 6The PI3K/Akt signaling pathway
regulates HSP70/HIF-1α expression and residual tumor growth
following incomplete radiofrequency ablation. (A) In the control,
RFA, wortmannin + RFA and wortmannin groups, HSP70 and HIF-1α
expression in the subcutaneously transplanted tumors was measured
by western blot and semi-quantitative analyses (*control
group vs. RFA group: HSP70, p<0.05; HIF-1α, p<0.05;
#RFA group vs. wortmannin + RFA group: HSP70, p<0.05;
HIF-1α, p<0.05; ★control group vs. wortmannin group:
HSP70, ns; HIF-1α, p<0.05). (B) Growth curve of subcutaneously
transplanted tumors in the control, RFA, wortmannin + RFA and
wortmannin group (*p<0.05 from day 3–7 control group
vs. RFA group; ྿p<0.05 from day 3–7 RFA group vs.
wortmannin + RFA group; #p<0.05 from day 3–7 control
group vs. wortmannin group). |
Discussion
Radiofrequency ablation (RFA) is becoming an
increasingly accepted treatment for primary lung cancer in patients
who are not candidates for subsegmental resection or lobectomy
(27). Compared to radiotherapy and
chemotherapy, RFA is focused exclusively on the tumor area without
causing any damage to the normal surrounding tissue. Compared to
surgical treatment, it is superior in that it is minimally
invasive. For advanced lung cancers, including SCLCs, RFA can also
be maximally effective for palliative care (28). In its early stage, it is not clear
whether RFA may function best as a stand-alone therapy or in
combination with other modalities. Understanding and recognizing
the patterns of tumor recurrence is critical in evaluating the
efficacy of this new treatment and to determine optimal candidates
for this treatment (29).
Although most studies indicate that lung RFA is safe
and feasible, operation complications appear to be common in
clinical trails. The most common complication is pneumothorax or
hemothorax. However, with the progression of hemostasis techniques
and the improvements in surgical repair materials, the incidence of
these complications has been significantly reduced (30). Nevertheless, such as other
therapeutic methods, postoperative recurrence is still a
challenging problem to solve for the RFA treatment of lung cancers.
Follow-up imaging data indicate that local treatment area,
intrapulmonary and nodal recurrences are commonly found during the
first 2 years after RFA, and that the most common area of
recurrence would be the local treatment area (27). Increasing tumor sizes and stages
have been significantly associated with the likelihood of
recurrence with RFA. Giraud et al proposed that microscopic
extensions of primary lung cancers should be encompassed in a
margin of 8 mm for adenocarcinomas and 6 mm for squamous cell
carcinomas. The aim of ablation should be to include an expected
ablation zone that includes the primary tumor plus at least an
additional 8–10 mm beyond the visible tumor margin in all
directions. Therefore, the target ablation diameter should be 1.6–2
cm greater than the tumor diameter (31). Various scholars suggest that it is
very difficult to achieve complete ablation due to heat sink
effects from adjacent vessels or the tumor angiogenesis network
(32). However, a new opinion was
brought forward that local recurrence following RFA may be the
result of the rapid growth of residual tumor cells, which has been
observed in numerous clinical centers (23).
Various studies demonstrate that residual tumor
cells are prone to proliferation, invasion and metastasis when the
local ablative temperature and thermal energy are not sufficiently
high. Additionally, angiogenesis potential induced by RFA-induced
hyperthermia also can play a role in the rapid growth of residual
tumor cells that have escaped ablation injury (14). This phenomenon is often referred to
as a 'malignant transformation', which is associated with RFA
therapies used to treat tumors (33). RFA is not only an important
therapeutic method, but the microenvironmental conditions present
with or induced by RFA can be replicated in in vitro cell
culture. The cell lines we used were already established and used
in previous studies, thus that further spontaneous transformation
under normal culture conditions would be unlikely. Therefore, we
concluded that the biological microenvironment associated with
tumor growth changed due to the exposure to heat stress.
Heat-adapted sublines became more proliferative, which became the
focus of our subsequent study to identify the molecular biological
mechanisms involved in tumor recurrence.
Previous studies indicate that RFA causes profound
hypoxia directly adjacent to the ablated area. The presence of
hypoxia has been reported in several solid tumors and is related to
tumor aggressiveness, proliferation and angiogenesis potential
(34). The increased levels of
hypoxia induced by RFA provided an additional growth-stimulating
microenvironment for the surviving tumor cells. HIF-1α is a
transcriptional factor that is induced by hypoxia and regulates
multiple biological processes including angiogenesis, cell
proliferation and migration (35).
In addition to hypoxia, HSPs are overexpressed at the border of the
ablated area. HSP70 is overexpressed under conditions of cellular
stress, such as heat activation (36). Kroeze et al proposed that
despite various conflicting results on the influence of HSP70 on
damaged cells there is strong evidence indicating that HSP70
overexpression plays an important role in increased cell survival
through the recovery of damaged cells, induction of cell
proliferation and inhibition of apoptosis (37). Increased expression of HSP70 in
surviving tumor cells at the border of thermally ablated tumors may
lead to the development of more aggressive tumor cells (38). Through pull-down assays and peptide
arrays, Zhou et al confirmed that HSP70 directly binds the
oxygen-dependent degradation domain (ODD) of HIF-1α, which
initiates PI3K/Akt-mediated degradation of HIF-1α under hypoxic
conditions. These data suggest that HSP70 constitutes an integral
player in HIF-1α accumulation and that PI3K/Akt contributes to
HIF-1α stabilization by provoking the expression of HSP70 (39). This agrees with our results that
hyperthermia-induced lung cancer cells exhibited enhanced
proliferation and angiogenesis potential through the
PI3K/Akt/HSP70/HIF-1α signaling pathway. In vivo the
peripheral region around the ablation zone highly expressed
HSP70/HIF-1α. When a specific inhibitor of PI3K/Akt was applied
in vitro and in vivo, the proliferation activity and
angiogenesis potential were both inhibited, regardless of cell
type.
RFA has been proposed as an acceptable alternative
treatment for lung cancer. Although the technique and condition of
ablation have steadily improved, including the application of
multipolar ablation and widening of the ablation margin, local
recurrence is still common. Multidisciplinary synthetic therapy,
which can combine RFA with other treatments, is also recently a
subject of increased interest. The present study demonstrated that
RFA-induced hyperthermia inherently changes the properties of
cells, creating different cancer sublines. These sublines exhibited
enhanced viability, proliferation and angiogenesis potential. The
PI3K/Akt/HSP70/HIF-1α signaling pathway played a central role
during this biological process. Therefore, RFA therapy supplemented
by molecular therapy targeting PI3K/Akt/HSP70/HIF-1α signaling may
decrease the incidence of local recurrence, improving patient
prognosis. The present study supplies additional theoretical basis
for the use of multidisciplinary synthetic therapies to treat lung
cancer.
Acknowledgments
The present study was funded by the National Nature
Science Foundation of China (no. 81302028). We would like to thank
the Research Center of the First Affiliated Hospital of Anhui
Medical University and the Laboratory Animal Center of Anhui
Medical University for providing technical assistance. The authors
would like to thank the Duoease Scientific Service Center for
excellent language editing service and suggestions for figure
revision.
Abbreviations:
|
RFA
|
radiofrequency ablation
|
|
NSCLC
|
non-small cell lung cancer
|
|
HIF-1α
|
hypoxia inducible factor-1α
|
|
HSP70
|
heat shock protein 70
|
|
MVD
|
microvessel density
|
References
|
1
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar
|
|
2
|
Chen WQ, Zhang SW, Zou XN and Zhao P: An
analysis of lung cancer mortality in China, 2004–2005. Zhonghua Yu
Fang Yi Xue Za Zhi. 44:378–382. 2010.In Chinese. PubMed/NCBI
|
|
3
|
Chen W, Zhang S and Zou X: Estimation and
projection of lung cancer incidence and mortality in China.
Zhongguo Fei Ai Za Zhi. 13:488–493. 2010.In Chinese. PubMed/NCBI
|
|
4
|
Mohammed TL, Chowdhry A, Reddy GP, Amorosa
JK, Brown K, Dyer DS, Ginsburg ME, Heitkamp DE, Jeudy J, Kirsch J,
et al: Expert Panel on Thoracic Imaging: ACR appropriateness
criteria® screening for pulmonary metastases. J Thorac
Imaging. 26:W1–W3. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Varela G and Thomas PA: Surgical
management of advanced non-small cell lung cancer. J Thorac Dis.
6(Suppl 2): S217–S223. 2014.PubMed/NCBI
|
|
6
|
Schreiner W, Semrau S, Fietkau R and Sirbu
H: Oligometastatic non-small cell lung cancer - surgical options
and therapy strategies. Zentralbl Chir. 139:335–341. 2014.In
German. PubMed/NCBI
|
|
7
|
Baisi A, De Simone M, Raveglia F and
Cioffi U: Thermal ablation in the treatment of lung cancer: Present
and future. Eur J Cardiothorac Surg. 43:683–686. 2013. View Article : Google Scholar
|
|
8
|
de Baere T, Farouil G and Deschamps F:
Lung cancer ablation: What is the evidence? Semin Intervent Radiol.
30:151–156. 2013. View Article : Google Scholar :
|
|
9
|
Goldberg SN, Gazelle GS, Compton CC and
McLoud TC: Radiofrequency tissue ablation in the rabbit lung:
efficacy and complications. Acad Radiol. 2:776–784. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ahmad A, Chen SL, Kavanagh MA, Allegra DP
and Bilchik AJ: Radiofrequency ablation of hepatic metastases from
colorectal cancer: Are newer generation probes better? Am Surg.
72:875–879. 2006.PubMed/NCBI
|
|
11
|
Shah DR, Green S, Elliot A, McGahan JP and
Khatri VP: Current oncologic applications of radiofrequency
ablation therapies. World J Gastrointest Oncol. 5:71–80. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lanuti M, Sharma A, Willers H, Digumarthy
SR, Mathisen DJ and Shepard JA: Radiofrequency ablation for stage I
non-small cell lung cancer: Management of locoregional recurrence.
Ann Thorac Surg. 93:921–988. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zavaglia C, Corso R, Rampoldi A, Vinci M,
Belli LS, Vangeli M, Solcia M, Castoldi C, Prisco C, Vanzulli A, et
al: Is percutaneous radiofrequency thermal ablation of
hepatocellular carcinoma a safe procedure? Eur J Gastroenterol
Hepatol. 20:196–201. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ke S, Ding XM, Kong J, Gao J, Wang SH,
Cheng Y and Sun WB: Low temperature of radiofrequency ablation at
the target sites can facilitate rapid progression of residual
hepatic VX2 carcinoma. J Transl Med. 8:732010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Seeber LM, Horrée N, Vooijs MA, Heintz AP,
van der Wall E, Verheijen RH and van Diest PJ: The role of hypoxia
inducible factor-1alpha in gynecological cancer. Crit Rev Oncol
Hematol. 78:173–184. 2011. View Article : Google Scholar
|
|
16
|
Wan J, Ma J, Mei J and Shan G: The effects
of HIF-1alpha on gene expression profiles of NCI-H446 human small
cell lung cancer cells. J Exp Clin Cancer Res. 28:1502009.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wan J, Chai H, Yu Z, Ge W, Kang N, Xia W
and Che Y: HIF-1α effects on angiogenic potential in human small
cell lung carcinoma. J Exp Clin Cancer Res. 30:772011. View Article : Google Scholar
|
|
18
|
Son WY, Han CT, Hwang SH, Lee JH, Kim S
and Kim YC: Repression of hspA2 messenger RNA in human testes with
abnormal spermatogenesis. Fertil Steril. 73:1138–1144. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Huang WJ, Xia LM, Zhu F, Huang B, Zhou C,
Zhu HF, Wang B, Chen B, Lei P, Shen GX, et al: Transcriptional
upregulation of HSP70-2 by HIF-1 in cancer cells in response to
hypoxia. Int J Cancer. 124:298–305. 2009. View Article : Google Scholar
|
|
20
|
Tikhonova NS, Moskaleva OS, Margulis BA
and Guzhova IV: Molecular chaperone HSP70 protects neuroblastoma
SK-N-SH cells from hypoxic stress. Tsitologiia. 50:467–472. 2008.In
Russian.
|
|
21
|
Yeh CH, Hsu SP, Yang CC, Chien CT and Wang
NP: Hypoxic preconditioning reinforces HIF-alpha-dependent HSP70
signaling to reduce ischemic renal failure-induced renal tubular
apoptosis and autophagy. Life Sci. 86:115–123. 2010. View Article : Google Scholar
|
|
22
|
Li B, Wang Z, Zhong Y, Lan J, Li X and Lin
H: CCR9-CCL25 interaction suppresses apoptosis of lung cancer cells
by activating the pI3K/Akt pathway. Med oncol. 32:662015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wen W, Liu W, Shao Y and Chen L:
VER-155008, a small molecule inhibitor of HSP70 with potent
anti-cancer activity on lung cancer cell lines. Exp Biol Med.
239:638–645. 2014. View Article : Google Scholar
|
|
24
|
Na JI, Na JY, Choi WY, Lee MC, Park MS,
Choi KH, Lee JK, Kim KT, Park JT and Kim HS: The HIF-1 inhibitor
YC-1 decreases reactive astrocyte formation in a rodent ischemia
model. Am J Transl Res. 7:751–760. 2015.PubMed/NCBI
|
|
25
|
Kong J, Kong J, Pan B, Ke S, Dong S, Li X,
Zhou A, Zheng L and Sun WB: Insufficient radiofrequency ablation
promotes angiogenesis of residual hepatocellular carcinoma via
HIF-1α/VEGFA. PLoS One. 7:e372662012. View Article : Google Scholar
|
|
26
|
Xu M, Xie XH, Xie XY, Xu ZF, Liu GJ, Zheng
YL, Huang GL, Wang W, Zheng SG and Lü MD: Sorafenib suppresses the
rapid progress of hepatocellular carcinoma after insufficient
radiofrequency ablation therapy: An experiment in vivo. Acta
Radiol. 54:199–204. 2013. View Article : Google Scholar
|
|
27
|
Beland MD, Wasser EJ, Mayo-Smith WW and
Dupuy DE: Primary non-small cell lung cancer: Review of frequency,
location, and time of recurrence after radiofrequency ablation.
Radiology. 254:301–307. 2010. View Article : Google Scholar
|
|
28
|
Baisi A, Raveglia F, De Simone M and
Cioffi U: Palliative role of percutaneous radiofrequency ablation
for severe hemoptysis in an elderly patient with inoperable lung
cancer. J Thorac Cardiovasc Surg. 140:1196–1197. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pennathur A, Luketich JD, Abbas G, Chen M,
Fernando HC, Gooding WE, Schuchert MJ, Gilbert S, Christie NA and
Landreneau RJ: Radiofrequency ablation for the treatment of stage I
non-small cell lung cancer in high-risk patients. J Thorac
Cardiovasc Surg. 134:857–864. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kashima M, Yamakado K, Takaki H, Kodama H,
Yamada T, Uraki J and Nakatsuka A: Complications after 1000 lung
radio-frequency ablation sessions in 420 patients: A single
center's experiences. AJR Am J Roentgenol. 197:W576–W580. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Giraud P, Antoine M, Larrouy A, Milleron
B, Callard P, De Rycke Y, Carette MF, Rosenwald JC, Cosset JM,
Housset M, et al: Evaluation of microscopic tumor extension in
non-small-cell lung cancer for three-dimensional conformal
radiotherapy planning. Int J Radiat Oncol Biol Phys. 48:1015–1024.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Abbas G, Schuchert MJ, Pennathur A,
Gilbert S and Luketich JD: Ablative treatments for lung tumors:
Radiofrequency ablation, stereotactic radiosurgery, and microwave
ablation. Thorac Surg Clin. 17:261–271. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Obara K, Matsumoto N, Okamoto M, Kobayashi
M, Ikeda H, Takahashi H, Katakura Y, Matsunaga K, Ishii T, Okuse C,
et al: Insufficient radiofrequency ablation therapy may induce
further malignant transformation of hepatocellular carcinoma.
Hepatol Int. 2:116–123. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Seeber LM, Horrée N, van der Groep P, van
der Wall E, Verheijen RH and van Diest PJ: Necrosis related
HIF-1alpha expression predicts prognosis in patients with
endometrioid endometrial carcinoma. BMC Cancer. 10:3072010.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhao X, Gao S, Ren H, Sun W, Zhang H, Sun
J, Yang S and Hao J: Hypoxia-inducible factor-1 promotes pancreatic
ductal adenocarcinoma invasion and metastasis by activating
transcription of the actin-bundling protein fascin. Cancer Res.
74:2455–2464. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bhardwaj N, Dormer J, Ahmad F, Strickland
AD, Gravante G, Beckingham I, West K, Dennison AR and Lloyd DM:
Heat shock protein 70 expression following hepatic radiofrequency
ablation is affected by adjacent vasculature. J Surg Res.
173:249–257. 2012. View Article : Google Scholar
|
|
37
|
Kroeze SG, van Melick HH, Nijkamp MW,
Kruse FK, Kruijssen LW, van Diest PJ, Bosch JL and Jans JJ:
Incomplete thermal ablation stimulates proliferation of residual
renal carcinoma cells in a translational murine model. BJU Int.
110:E281–E286. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kim EK, Park JD, Shim SY, Kim HS, Kim BI,
Choi JH and Kim JE: Effect of chronic hypoxia on proliferation,
apoptosis, and HSP70 expression in mouse bronchiolar epithelial
cells. Physiol Res. 55:405–411. 2006.
|
|
39
|
Zhou J, Schmid T, Frank R and Brüne B:
PI3K/Akt is required for heat shock proteins to protect
hypoxia-inducible factor 1alpha from pVHL-independent degradation.
J Biol Chem. 279:13506–13513. 2004. View Article : Google Scholar : PubMed/NCBI
|