Introduction
Charged particle therapy, including carbon ion beam
radiotherapy, is regarded as a promising treatment for various
types of human cancer (1). Although
clinical and pathological studies have shown that the mechanism by
which densely ionizing radiation induces apoptosis in cancer cells
involves redox regulation (2), a
study has revealed that cancer stem cells and minor fractions of
other cancer cells are able to survive radiation therapy (3). In contrast, charged particle therapy
is generally supposed to exert its antitumor effects directly on
cellular DNA, which is vulnerable to double-strand breaks (4). Because DNA double-strand breaks are
lethal to cells (5), this may be a
promising therapeutic mechanism for efficient eradication of cancer
cells.
A growing body of evidence has suggested that small
non-coding RNAs, known as microRNAs (miRNAs), are involved in the
regulation of tumor initiation and progression (4,6–9).
Several miRNAs have been found to be prognostic factors and
clinical targets for cancer treatment (9–13).
Zhang et al (9) demonstrated
that miR-205, which is downregulated in radioresistant breast
cancer cells, can be used as radiosensitizer in a preclinical
model. Wang et al (12)
found that miR-185 is downregulated in response to radiation and
that elevation of miR-185 sensitizes renal cell carcinoma cells to
X-ray irradiation. In this study, we focused on the expression of
miRNA in carbon ion beam-resistant mouse squamous carcinoma cell
lines (C30) to discover new radiosensitizers and prognostic
factors. We demonstrated that miR-374, a miRNA suppressed in C30,
can act as radiosensitizer when overexpressed in the human
pancreatic cancer cell lines PANC1 and MIA PaCa-2. This result
suggests that miR-374 is a potential prognostic factor for carbon
ion beam radiotherapy and may be a promising new
radiosensitizer.
Materials and methods
Cell lines
The NR-S1 (mouse squamous cell carcinoma; controls),
X60 (radioresistant to X-rays), and C30 (radioresistant to carbon
ions) cell lines were used in this study. The NR-S1 line was kindly
provided by Dr Koichi Ando of the Medicine and Biology Division,
Gunma University Heavy Ion Medical Center. The X60 and C30 cell
lines were kindly provided by Dr Katshutoshi Sato of the Advanced
Radiation Biology Research Program of the Research Center for
Charged Particle Therapy, National Institute of Radiological
Sciences (Chiba, Japan). The cells were cultured in Dulbecco's
modified Eagle's medium (DMEM; Sigma-Aldrich, Tokyo, Japan)
supplemented with 10% fetal bovine serum and
penicillin/streptomycin and maintained at 37°C in a 5%
CO2 incubator.
X-ray radioresistant cells
X60 cells were established by irradiating NR-S1
cells with 10 Gy of X-ray radiation once every 2 weeks. The cells
were irradiated with a total dose of 60 Gy of X-ray radiation and
cultured for 4 weeks after their final irradiation (14).
Carbon ion beam-resistant cells
C30 cells were established by irradiating NR-S1
cells with 5 Gy of carbon ion beam radiation once every 2 weeks.
The cells were irradiated with a total dose of 30 Gy of carbon ion
beam radiation and cultured for 4 weeks after their final
irradiation.
miRNA microarray analysis
Nucleotides isolated from cell lines were analyzed
by 3D-Gene microarrays (Toray Industries, Tokyo, Japan).
miRNA extraction and qRT-PCR
Total RNAs were extracted from the cultured cells
using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). Reverse
transcription for miR-374 was performed with the TaqMan MicroRNA
Reverse Transcription kit (Applied Biosystems, Carlsbad, CA, USA).
Quantitative reverse transcription PCR (qRT-PCR) was performed by
using the TaqMan Universal PCR Master Mix (Applied Biosystems).
Internal controls included snoRNA202, snoRNA234, and U6.
miRNA transfection
Transfection of 70–80% confluent cells was performed
using Lipofectamine RNAiMAX (Invitrogen) following the
manufacturer's instructions. The medium was replaced with new
culture medium 24 h after transfection.
Colony formation assay
PANC-1 and MIA PaCa-2 underwent miRNA transfection
and irradiated by gamma-ray or carbon ion beam. Gamma-ray
irradiation was conducted by gamma cell (Best Theratronics Ltd.,
Ottawa, ON, Canada). Carbon ion beam irradiation was carried out at
the Heavy Ion Medical Accelerator in Chiba (HIMAC) at the National
Institute of Radiological Sciences, Japan. Irradiated cells were
seeded into 6-cm dishes and incubated for 12 days. Colonies were
then stained with crystal violet (0.5% w/v) and counted. The colony
consisted of at least 50 cells. Survival curves were drawn by using
Microsoft Excel software (Microsoft Corporation, Redmond, WA,
USA).
Sphere formation assay
PANC-1 and MIA PaCa-2 cell lines were transfected
with a negative control, miR-196 and miR-374. Cells (1,000, 500 and
100 per well) were seeded in a 96-well plate with serum-free medium
(DMEM/F12) supplemented with N2 supplement (Invitrogen), epidermal
growth factor (EGF; 20 ng/ml), and basic fibroblast growth factor
(bFGF; 20 ng/ml) (both from R&D Systems, Inc., Minneapolis, MN,
USA). After 14 days of incubation, spheres with diameters >100
μm were counted.
Statistical analysis
Each experiment was repeated three times or more.
Data are presented as the mean ± SD. Statistical significance was
determined with Student's t-tests using Microsoft Excel software
(Microsoft Corporation).
Results
miRNA expressions are differentially
changed in response to carbon ion beam radiation
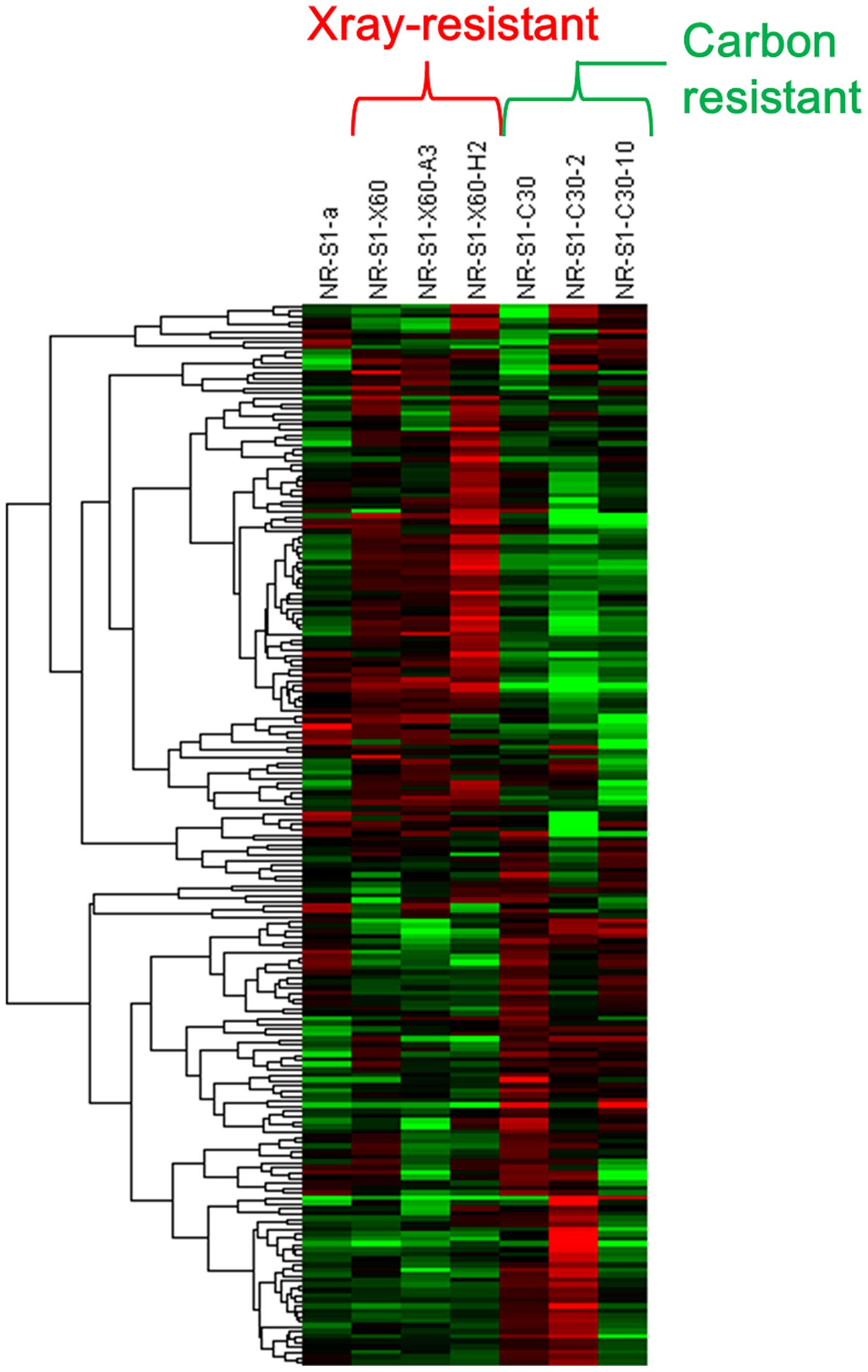
To identify miRNAs that may be involved in carbon
ion beam resistance, radioresistant cell lines were produced from a
mouse squamous cell carcinoma cell line, and then the miRNA
profiles of the resistant cells were obtained using miRNA
microarray analysis. The miRNA expression patterns of the
radioresistant cell lines produced by X-ray and carbon ion beam
irradiation were different (Fig.
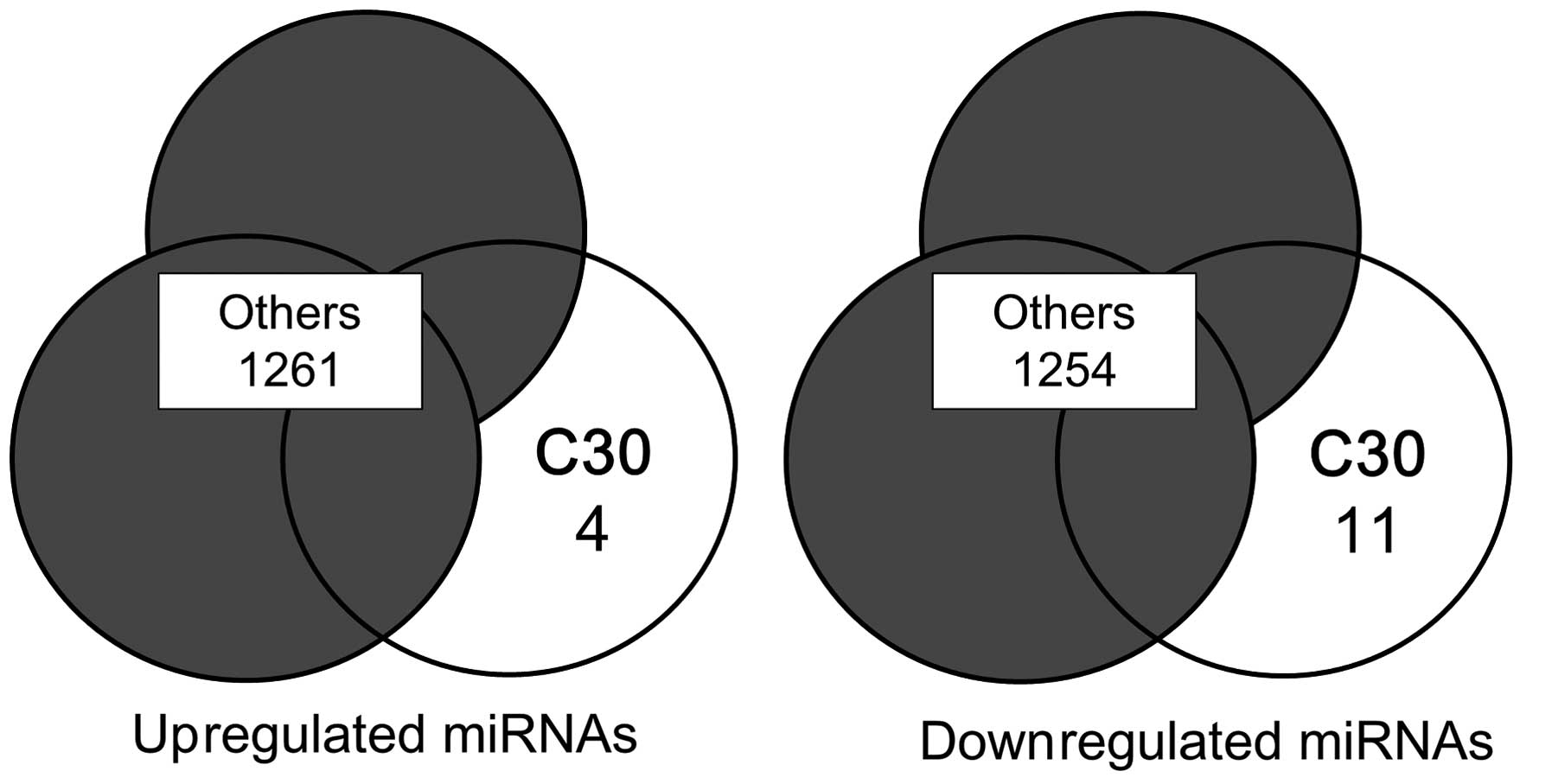
1). Among the 1,265 miRNAs analyzed, 4 were downregulated and
11 were upregulated (Fig. 2 and
Table I). To evaluate which of
these miRNAs were related to carbon ion beam resistance, miRNAs
were screened using the NCBI Gene database and miRBase: the miRNA
database. Each murine miRNA was searched to identify its function
and determine whether its sequence matched that of a human miRNA.
The miRNA mmu-miR-3068-3p was rejected because it does not exist in
humans. Based on the average log2 ratio values from the microarray
analysis, 3 upregulated miRNAs (mmu-miR-494-3p, mmu-miR-149-3p, and
mmu-miR-2861) and 2 downregulated miRNAs (mmu-miR-196a-5p and
mmu-miR-374c-5p) were selected for further study. To ensure that
the cellular expression of the selected miRNAs was consistent with
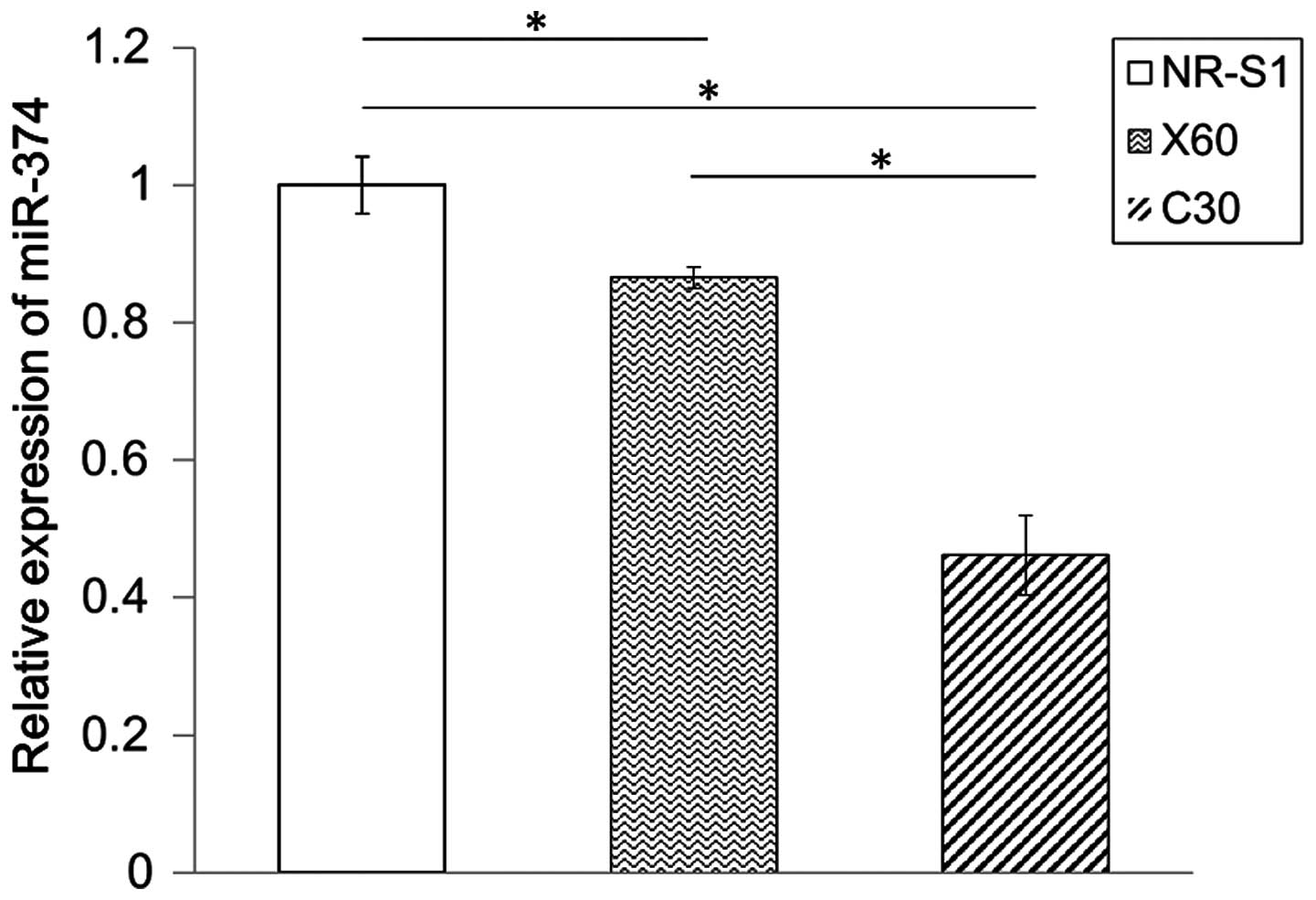
the microarray results, qRT-PCR was performed. Because the actual
expression of miR-532 was not consistent with the microarray
result, miR-532 was rejected (Fig.
3). Previous studies on the use of miRNAs, such as miR-205
(9) and miR-185 (12), as radiosensitizers focused on
downregulated miRNAs. Therefore, we first focused on downregulated
miRNAs, mmu-miR-196a-5p and mmu-miR-374c-5p.
 | Table IThe miRNAs specifically expressed in
C30 cell lines. |
Table I
The miRNAs specifically expressed in
C30 cell lines.
| Name | NR-S1 | X60 | X60-A3 | X60-H2 | C30 | C30-2 | C30-10 |
|---|
| mmu-miR-494-3p | −0.115 | −0.219 | −0.017 | −0.222 | 0.358 | 0.08 | 0.047 |
| mmu-miR-149-3p | −0.217 | −0.015 | −0.155 | −0.008 | 0.193 | 0.026 | 0.132 |
| mmu-miR-2861 | −0.235 | −0.159 | −0.253 | −0.244 | 0.222 | 0.464 | 0.036 |
| mmu-miR-3068-3p | −0.083 | −0.52 | −0.784 | −0.132 | 0.196 | 0.556 | 0.322 |
| mmu-miR-18a-5p | 0.03 | 0.017 | 0.276 | 0.493 | −0.069 | −1.064 | −0.124 |
| mmu-miR-205-5p | 0.266 | 0.276 | 0.083 | 0.87 | −0.136 | −1.117 | −1.603 |
| mmu-miR-16-5p | 0.007 | 0.174 | 0.099 | 0.539 | −0.348 | −0.616 | −0.144 |
| mmu-miR-23a-3p | 0.062 | 0.127 | 0.168 | 0.529 | −0.402 | −0.395 | −0.357 |
| mmu-miR-677-3p | 0.117 | 0.039 | 0.319 | 0.596 | −0.283 | −0.682 | −0.547 |
| mmu-miR-7a-5p | 0.142 | 0.401 | 0.669 | 0.312 | −0.459 | −1.856 | −0.431 |
| mmu-miR-196a-5p | 0.31 | 0.554 | 0.521 | 0.792 | −1.085 | −1.956 | −1.334 |
| mmu-miR-374c-5p | 0.252 | 0.403 | 0.248 | 0.786 | −0.576 | −1.815 | −0.688 |
| mmu-miR-3968 | 0.012 | 0.226 | 0.147 | 0.299 | −0.105 | −0.569 | −0.182 |
| mmu-miR-6243 | 0.12 | 0.241 | 0.161 | 0.124 | −0.019 | −0.459 | −0.307 |
| mmu-miR-151-5p | 0.42 | 0.295 | 0.314 | 0.042 | −0.401 | −0.303 | −0.748 |
Overexpression of miR-374 radiosensitized
human pancreas cancer cell lines against carbon ion beam
irradiation
Carbon ion beam radiotherapy is a promising
treatment for pancreatic cell carcinoma, which is a
therapy-resistant cancer, with respectable treatment outcomes in a
clinical trial (15). We
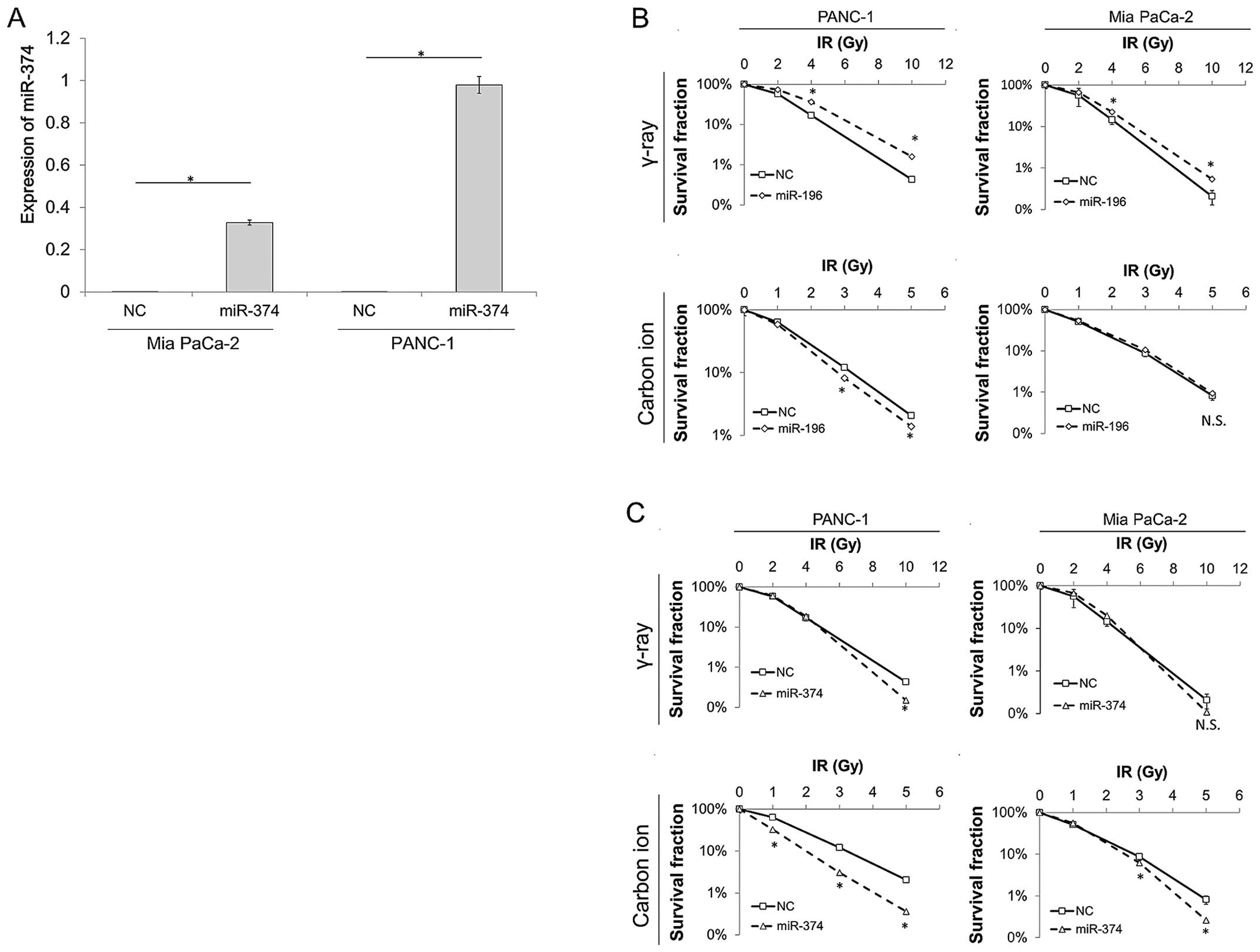
overexpressed miR-196 and miR-374 in human pancreatic cancer cell
lines PANC1 and MIA PaCa-2 using lipofectamine RNAiMAX. The actual
expression level was evaluated with qRT-PCR (Fig. 4A). A colony formation assay was
performed with gamma-ray and carbon ion beam irradiation to
investigate whether miRNA expression changes radiosensitivity. Cell
lines overexpressing miR-196 were more resistant to gamma-ray
irradiation than the control. PANC-1 cells overexpressing miR-196
were a little more sensitive to carbon ion beam irradiation than
the control; however, there was no change in carbon ion beam
sensitivity in the MIA PaCa-2 cell line (Fig. 4B). Overexpression of miR-374
increased the sensitivity of both PANC-1 and MIA PaCa-2 cells to
carbon ion beam irradiation. There was no change in gamma-ray
sensitivity (Fig. 4C). This result
suggests that miR-374 may be used as a new carbon ion
radiosensitizer.
Discussion
The mechanisms of X-ray resistance have been studied
and the DNA repair processes that occur through the ATR-Chk1
(12,16) and ZEB1-Chk1 pathways (9,17) are
known to be involved. Carbon ion beam radiation is thought to be
more effective than X-ray radiation in inducing double-strand DNA
breaks; therefore, it should be more effective in treating
X-ray-resistant cancer cells. However, the mechanisms leading to
resistance to carbon ion beam radiation are not fully understood.
Several studies have shown that radiation-induced changes in miR
expression are often related to radioresistance. For example,
miR-205 and miR-185, which are downregulated after X-ray
irradiation, can radiosensitize cancer cells when overexpressed. We
investigated miRNAs that are downregulated in carbon ion
beam-irradiated cell lines, and evaluated whether overexpression of
these downregulated miRNAs can radiosensitize the cell toward
carbon ion beam radiation. In our study, miR-196 and miR-374 were
downregulated in C30 cells. Overexpression of miR-374 enhanced the
carbon ion beam radiosensitivity of human pancreatic cancer cell
lines PANC-1 and MIA PaCa-2. Overexpression of miR-196 decreased
the gamma-ray radiosensitivity of PANC-1 and MIA PaCa-2, and
slightly enhanced the carbon ion sensitivity of PANC-1 (Fig. 4B). On the contrary, overexpression
of miR-196 increased sphere formation in the MIA PaCa-2 cell line,
while miR-374c suppressed it. The sphere formation assay is often
used to evaluate cancer stem cell properties, and this result
suggests that miR-374 has a potential to suppress cancer stem
cells. Our findings suggest that miR-374 may be useful as a
radiosensitizer for carbon ion beam radiotherapy. While several
X-ray radiosensitizers have been studied to date, there are few
studies describing carbon ion beam radiosensitizers. Although
further investigations are required, including in vivo
clinical trials, miR-374 showed high potential to be used as a
carbon ion beam radiosensitizer or as a biomarker for carbon ion
beam sensitivity.
The limitation of this study is that the mechanism
through which miR-374 enhances carbon ion beam radiosensitivity is
unclear. We searched for target DNAs involved in the regulation of
carbon ion beam radiosensitivity using a target scan. None of the
DNAs known to be involved in the regulation of X-ray
radiosensitivity, such as ZEB19, ATR (12), were in the target DNA list of
miR-374. This suggests that carbon ion beam radiosensitivity is
regulated by a pathway different from that of X-ray
radiosensitivity.
In conclusion, we demonstrated the potential of
miR-374 to become the first carbon ion beam radiosensitizer. We
also showed that miR-374 can be used as a biomarker to determine
the optimal treatment for cancer. In the future, further detailed
studies of carbon ion beam radioresistance mechanisms and clinical
trials for the human evaluation of miR-374 are expected.
Acknowledgments
We thank all of our laboratory members for their
fruitful discussion and technical assistance. This study was
supported in part by a Grant-in-Aid for Scientific Research from
the Ministry of Education, Culture, Sports, Science, and Technology
(Japan); P-DIRECT; a Grant-in-Aid from the Ministry of Health,
Labor, and Welfare; a grant from the National Institute of
Biomedical Innovation; and a grant from the Osaka University Drug
Discovery Funds. Partial support was received from Taiho
Pharmaceutical Co., Ltd., Evidence Based Medical Research Center,
through institutional endowments. A part of study was performed as
a research project with heavy ions at NIRS-HIMAC (no. 15J183), and
supported by the Grants-in-Aid for Scientific Research in Japan
Society for the Promotion of Science (no. 15K15467).
References
|
1
|
Kamada T, Tsujii H, Blakely EA, Debus J,
De Neve W, Durante M, Jäkel O, Mayer R, Orecchia R, Pötter R, et
al: Carbon ion radiotherapy in Japan: An assessment of 20 years of
clinical experience. Lancet Oncol. 16:e93–e100. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yasueda A, Urushima H and Ito T: Efficacy
and interaction of antioxidant supplements as adjuvant therapy in
cancer treatment: A Systematic Review. Integr Cancer Ther.
15:17–39. 2016. View Article : Google Scholar
|
|
3
|
Skvortsova I, Debbage P, Kumar V and
Skvortsov S: Radiation resistance: Cancer stem cells (CSCs) and
their enigmatic pro-survival signaling. Semin Cancer Biol.
35:39–44. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Baek SJ, Ishii H, Tamari K, Hayashi K,
Nishida N, Konno M, Kawamoto K, Koseki J, Fukusumi T, Hasegawa S,
et al: Cancer stem cells: The potential of carbon ion beam
radiation and new radiosensitizers (Review). Oncol Rep.
34:2233–2237. 2015.PubMed/NCBI
|
|
5
|
Kunkel TA: DNA replication fidelity. J
Biol Chem. 279:16895–16898. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Calin GA, Dumitru CD, Shimizu M, Bichi R,
Zupo S, Noch E, Aldler H, Rattan S, Keating M, Rai K, et al:
Frequent deletions and down-regulation of micro-RNA genes miR15 and
miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci
USA. 99:15524–15529. 2002. View Article : Google Scholar
|
|
7
|
He L, Thomson JM, Hemann MT,
Hernando-Monge E, Mu D, Goodson S, Powers S, Cordon-Cardo C, Lowe
SW, Hannon GJ, et al: A microRNA polycistron as a potential human
oncogene. Nature. 435:828–833. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ma L, Teruya-Feldstein J and Weinberg RA:
Tumour invasion and metastasis initiated by microRNA-10b in breast
cancer. Nature. 449:682–688. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang P, Wang L, Rodriguez-Aguayo C, Yuan
Y, Debeb BG, Chen D, Sun Y, You MJ, Liu Y, Dean DC, et al: miR-205
acts as a tumour radiosensitizer by targeting ZEB1 and Ubc13. Nat
Commun. 5:56712014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ma S, Tang KH, Chan YP, Lee TK, Kwan PS,
Castilho A, Ng I, Man K, Wong N, To KF, et al: miR-130b Promotes
CD133(+) liver tumor-initiating cell growth and self-renewal via
tumor protein 53-induced nuclear protein 1. Cell Stem Cell.
7:694–707. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu C, Kelnar K, Liu B, Chen X,
Calhoun-Davis T, Li H, Patrawala L, Yan H, Jeter C, Honorio S, et
al: The microRNA miR-34a inhibits prostate cancer stem cells and
metastasis by directly repressing CD44. Nat Med. 17:211–215. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang J, He J, Su F, Ding N, Hu W, Yao B,
Wang W and Zhou G: Repression of ATR pathway by miR-185 enhances
radiation-induced apoptosis and proliferation inhibition. Cell
Death Dis. 4:e6992013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huang F, Tang J, Zhuang X, Zhuang Y, Cheng
W, Chen W, Yao H and Zhang S: MiR-196a promotes pancreatic cancer
progression by targeting nuclear factor kappa-B-inhibitor alpha.
PLoS One. 9:e878972014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sato K, Imai T, Okayasu R and Shimokawa T:
Heterochromatin domain number correlates with X-ray and carbon-ion
radiation resistance in cancer cells. Radiat Res. 182:408–419.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tsujii H and Kamada T: A review of update
clinical results of carbon ion radiotherapy. Jpn J Clin Oncol.
42:670–685. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brezniceanu ML, Lau CJ, Godin N, Chénier
I, Duclos A, Ethier J, Filep JG, Ingelfinger JR, Zhang SL and Chan
JS: Reactive oxygen species promote caspase-12 expression and
tubular apoptosis in diabetic nephropathy. J Am Soc Nephrol.
21:943–954. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang P, Wei Y, Wang L, Debeb BG, Yuan Y,
Zhang J, Yuan J, Wang M, Chen D, Sun Y, et al: ATM-mediated
stabilization of ZEB1 promotes DNA damage response and
radioresistance through CHK1. Nat Cell Biol. 16:864–875. 2014.
View Article : Google Scholar : PubMed/NCBI
|