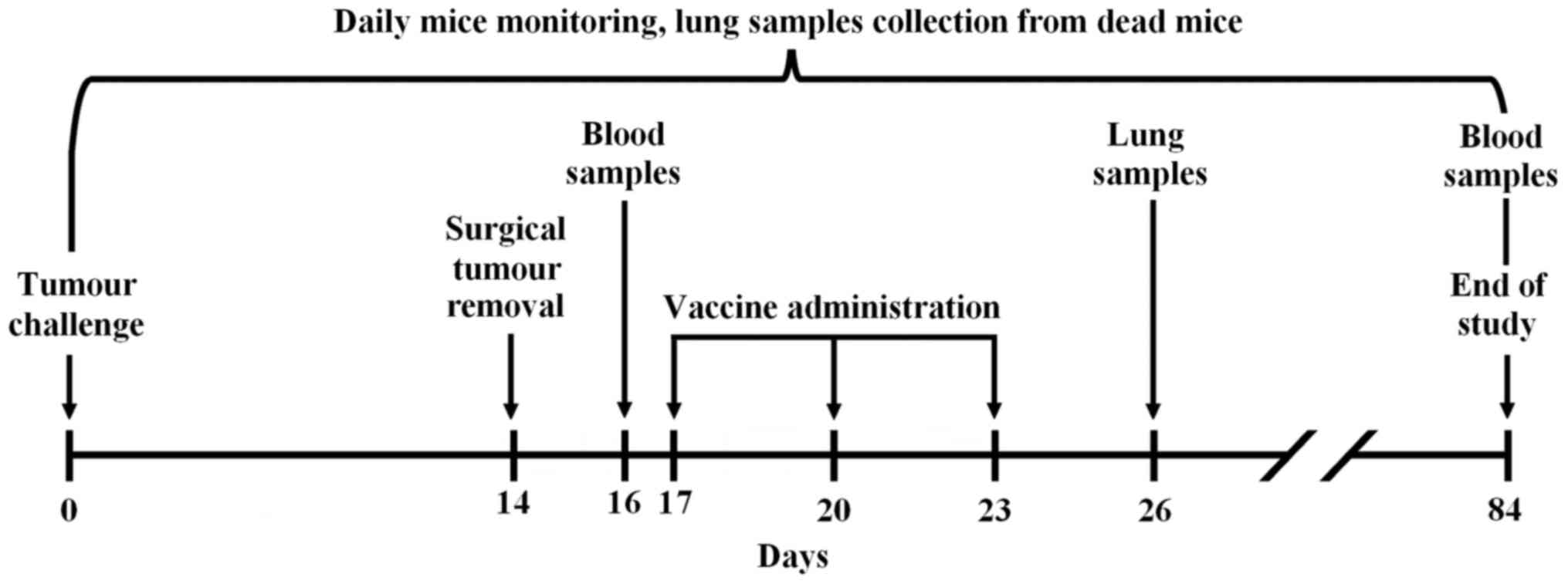
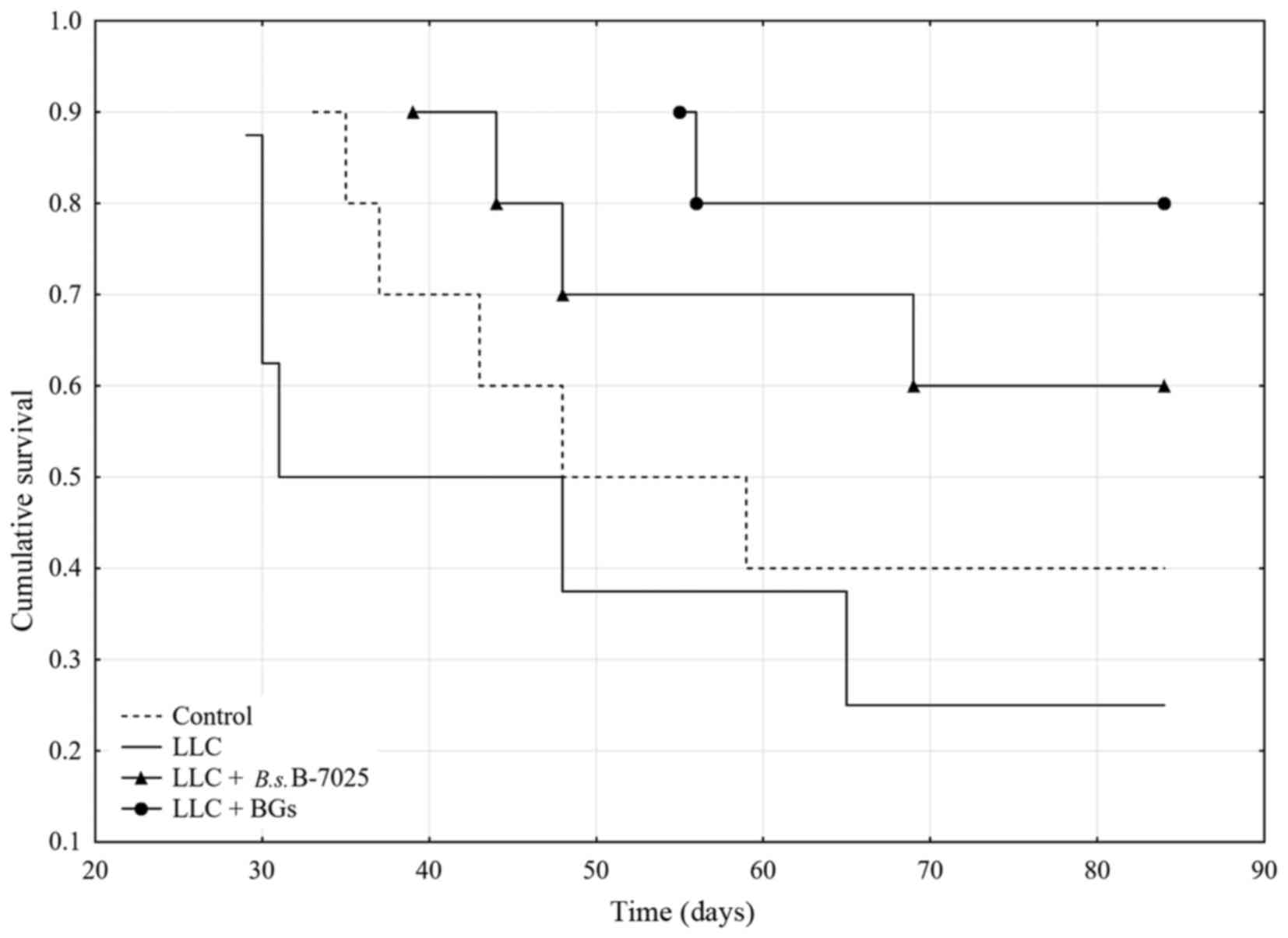
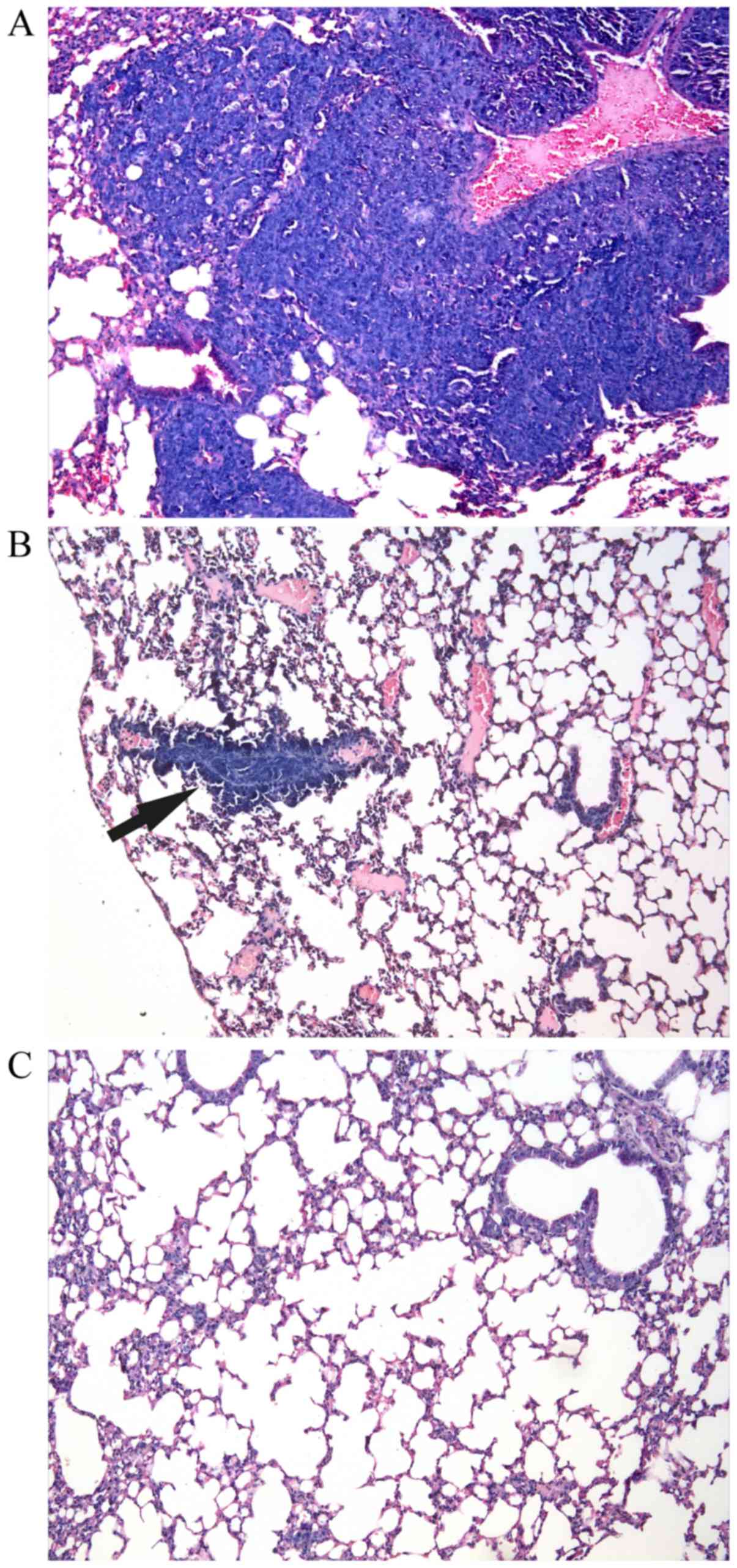
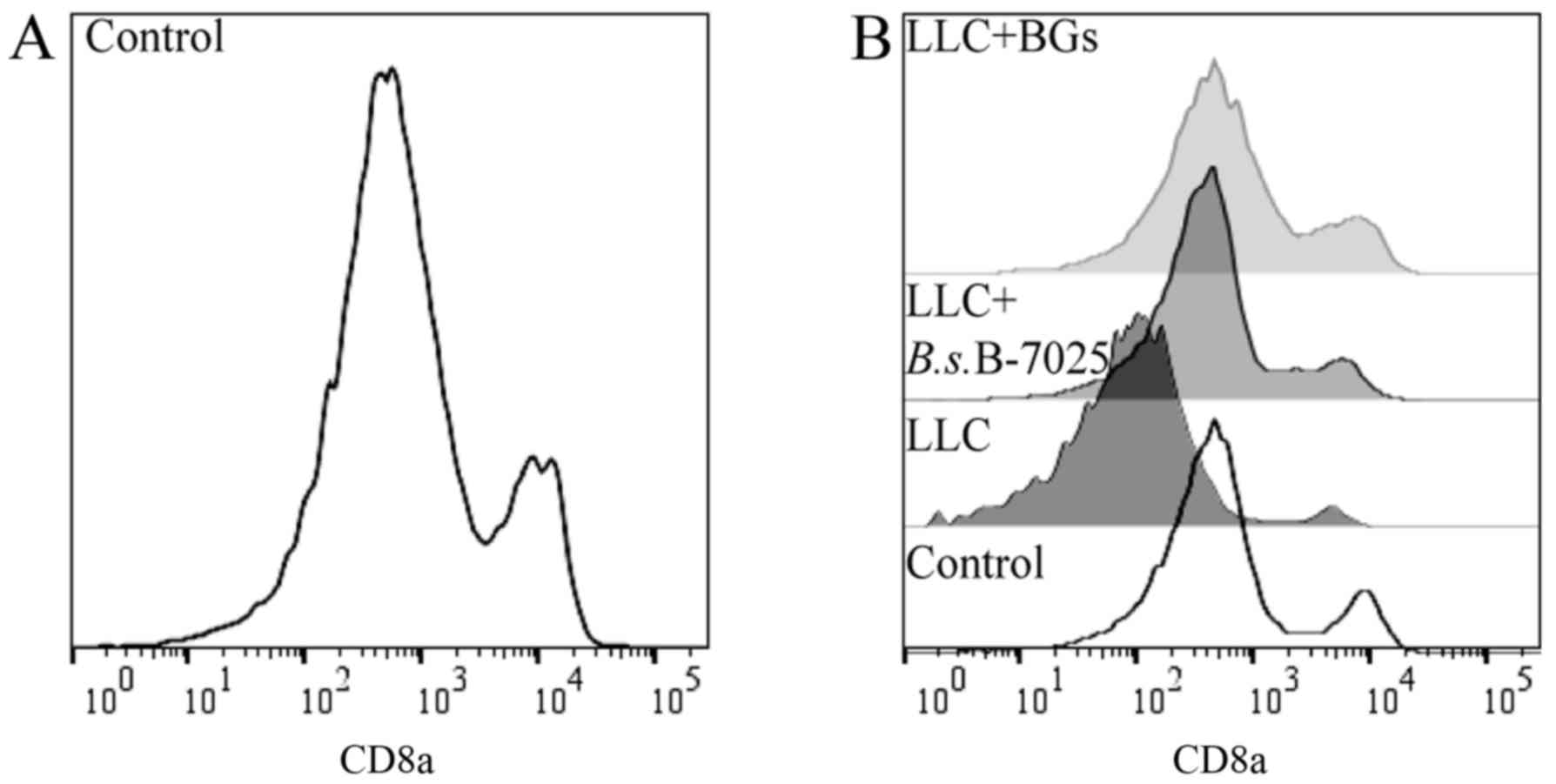
|
1
|
Vesely MD and Schreiber RD: Cancer
immunoediting: Antigens, mechanisms, and implications to cancer
immunotherapy. Ann NY Acad Sci. 1284:1–5. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zitvogel L, Tesniere A and Kroemer G:
Cancer despite immunosurveillance: Immunoselection and
immunosubversion. Nat Rev Immunol. 6:715–727. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bose A, Chakraborty T, Chakraborty K, Pal
S and Baral R: Dysregulation in immune functions is reflected in
tumor cell cytotoxicity by peripheral blood mononuclear cells from
head and neck squamous cell carcinoma patients. Cancer Immun.
8:102008.PubMed/NCBI
|
|
4
|
Noguchi A, Kaneko T, Naitoh K, Saito M,
Iwai K, Maekawa R, Kamigaki T and Goto S: Impaired and imbalanced
cellular immunological status assessed in advanced cancer patients
and restoration of the T cell immune status by adoptive T-cell
immunotherapy. Int Immunopharmacol. 18:90–97. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Galluzzi L, Vacchelli E, Bravo-San Pedro
JM, Buqué A, Senovilla L, Baracco EE, Bloy N, Castoldi F, Abastado
JP, Agostinis P, et al: Classification of current anticancer
immunotherapies. Oncotarget. 5:12472–12508. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Guo C, Manjili MH, Subjeck JR, Sarkar D,
Fisher PB and Wang XY: Therapeutic cancer vaccines: Past, present,
and future. Adv Cancer Res. 119:421–475. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Butterfield LH: Cancer vaccines. BMJ.
350:h9882015. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Palucka K and Banchereau J: Cancer
immunotherapy via dendritic cells. Nat Rev Cancer. 12:265–277.
2012. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schijns V, Tartour E, Michalek J,
Stathopoulos A, Dobrovolskienė NT and Strioga MM: Immune adjuvants
as critical guides directing immunity triggered by therapeutic
cancer vaccines. Cytotherapy. 16:427–439. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chiang CL, Coukos G and Kandalaft LE:
Whole tumor antigen caccines: Where Are We? Vaccines (Basel).
3:344–372. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gerlinger M, Rowan AJ, Horswell S, Larkin
J, Endesfelder D, Gronroos E, Martinez P, Matthews N, Stewart A,
Tarpey P, et al: Intratumor heterogeneity and branched evolution
revealed by multiregion sequencing. N Engl J Med. 366:883–892.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Srivatsan S, Patel JM, Bozeman EN, Imasuen
IE, He S, Daniels D and Selvaraj P: Allogeneic tumor cell vaccines:
The promise and limitations in clinical trials. Hum Vaccin
Immunother. 10:52–63. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Schnurr M, Galambos P, Scholz C, Then F,
Dauer M, Endres S and Eigler A: Tumor cell lysate-pulsed human
dendritic cells induce a T-cell response against pancreatic
carcinoma cells: An in vitro model for the assessment of tumor
vaccines. Cancer Res. 61:6445–6450. 2001.PubMed/NCBI
|
|
14
|
Kuang DM, Zhao Q, Xu J, Yun JP, Wu C and
Zheng L: Tumor-educated tolerogenic dendritic cells induce
CD3epsilon down-regulation and apoptosis of T cells through
oxygen-dependent pathways. J Immunol. 181:3089–3098. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li H, Zhu H, Xu CJ and Yuan J: Cleavage of
BID by caspase 8 mediates the mitochondrial damage in the Fas
pathway of apoptosis. Cell. 94:491–501. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Schuster N and Krieglstein K: Mechanisms
of TGF-beta-mediated apoptosis. Cell Tissue Res. 307:1–14. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dong B, Dai G, Xu L, Zhang Y, Ling L, Sun
L and Lv J: Tumor cell lysate induces the immunosuppression and
apoptosis of mouse immunocytes. Mol Med Rep. 10:2827–2834.
2014.PubMed/NCBI
|
|
18
|
Strioga MM, Felzmann T, Powell DJ Jr,
Ostapenko V, Dobrovolskiene NT, Matuskova M, Michalek J and Schijns
VE: Therapeutic dendritic cell-based cancer vaccines: The state of
the art. Crit Rev Immunol. 33:489–547. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Beck B, Dörfel D, Lichtenegger FS, Geiger
C, Lindner L, Merk M, Schendel DJ and Subklewe M: Effects of TLR
agonists on maturation and function of 3-day dendritic cells from
AML patients in complete remission. J Transl Med. 9:1512011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kawahara M and Takaku H: Intradermal
immunization with combined baculovirus and tumor cell lysate
induces effective antitumor immunity in mice. Int J Oncol.
43:2023–2030. 2013.PubMed/NCBI
|
|
21
|
Temizoz B, Kuroda E and Ishii KJ: Vaccine
adjuvants as potential cancer immunotherapeutics. Int Immunol.
28:329–338. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kurooka M and Kaneda Y: Inactivated Sendai
virus particles eradicate tumors by inducing immune responses
through blocking regulatory T cells. Cancer Res. 67:227–236. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bungener L, Huckriede A, Wilschut J and
Daemen T: Delivery of protein antigens to the immune system by
fusion-active virosomes: A comparison with liposomes and ISCOMs.
Biosci Rep. 22:323–338. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu H, Tu Z, Feng F, Shi H, Chen K and Xu
X: Virosome, a hybrid vehicle for efficient and safe drug delivery
and its emerging application in cancer treatment. Acta Pharm.
65:105–116. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wiedermann U, Wiltschke C, Jasinska J,
Kundi M, Zurbriggen R, Garner-Spitzer E, Bartsch R, Steger G,
Pehamberger H, Scheiner O, et al: A virosomal formulated Her-2/neu
multi-peptide vaccine induces Her-2/neu-specific immune responses
in patients with metastatic breast cancer: A phase I study. Breast
Cancer Res Treat. 119:673–683. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Witte A, Wanner G, Sulzner M and Lubitz W:
Dynamics of PhiX174 protein E-mediated lysis of Escherichia coli.
Arch Microbiol. 157:381–388. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Henrich B, Lubitz W and Plapp R: Lysis of
Escherichia coli by induction of cloned phi X174 genes. Mol Gen
Genet. 185:493–497. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Montanaro J, Inic-Kanada A, Ladurner A,
Stein E, Belij S, Bintner N, Schlacher S, Schuerer N, Mayr UB,
Lubitz W, et al: Escherichia coli Nissle 1917 bacterial ghosts
retain crucial surface properties and express chlamydial antigen:
An imaging study of a delivery system for the ocular surface. Drug
Des Devel Ther. 9:3741–3754. 2015.PubMed/NCBI
|
|
29
|
Eko FO, Mayr UB, Attridge SR and Lubitz W:
Characterization and immunogenicity of Vibrio cholerae ghosts
expressing toxin-coregulated pili. J Biotechnol. 83:115–123. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kudela P, Koller VJ and Lubitz W:
Bacterial ghosts (BGs) - advanced antigen and drug delivery system.
Vaccine. 28:5760–5767. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhu W, Yang G, Zhang Y, Yuan J and An L:
Generation of biotechnology-derived Flavobacterium columnare ghosts
by PhiX174 gene E-mediated inactivation and the potential as
vaccine candidates against infection in grass carp. J Biomed
Biotechnol. 2012:7607302012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cai K, Zhang Y, Yang B and Chen S:
Yersinia enterocolitica ghost with msbB mutation provides
protection and reduces proinflammatory cytokines in mice. Vaccine.
31:334–340. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Riedmann EM, Kyd JM, Cripps AW and Lubitz
W: Bacterial ghosts as adjuvant particles. Expert Rev Vaccines.
6:241–253. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Muhammad A, Champeimont J, Mayr UB, Lubitz
W and Kudela P: Bacterial ghosts as carriers of protein subunit and
DNA-encoded antigens for vaccine applications. Expert Rev Vaccines.
11:97–116. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Podgorskiĭ VS, Kovalenko EA, Getman EI,
Potebnia GP and Tanasienko OA: Lectin activity of antitumor
substances synthesized by Bacillus subtilis B-7025. Mikrobiol Z.
64:10–17. 2002.(In Russian). PubMed/NCBI
|
|
36
|
Tanasienko OA, Cheremshenko NL, Titova GP,
Potebnya MG, Gavrilenko MM, Nagorna SS and Kovalenko NK: Elevation
of the efficacy of antitumor vaccine prepared on the base of
lectines from B. subtilis B-7025 upon its combined application with
probiotics in vivo. Exp Oncol. 27:336–338. 2005.
|
|
37
|
Potebnia HP, Safronova LA, Cheremshenko
NL, Lisovenko HS, Sorokulova IB, Prykhodko VO, Trokhymenko NV,
Tanasiienko OA and Bombin AV: Influence of probiotic subalin on
efficiency of antitumor vaccine. Mikrobiol Zh. 68:51–58. 2006.(In
Ukrainian).
|
|
38
|
Potebnya GP, Lisovenko GS, Trokhimenko NV,
Cheremshenko NL, Didenko GV, Reder AS and Andronati SA: Elevation
of efficacy of cancer vaccine combined with interferon and inducer
of endogeneous interferon synthesis amixin. Exp Oncol. 30:319–323.
2008.PubMed/NCBI
|
|
39
|
Tanasienko OA, Rudyk MP, Pozur VV and
Potebnya GP: Influence of bacterial lectins on some reactions of
nonspecific immunity in sarcoma 37 transplanted mice. Exp Oncol.
32:254–257. 2010.PubMed/NCBI
|
|
40
|
Potebnya G, Cheremshenko N, Lisovenko G,
Voyekova I, Bazas' V, Todor I and Chekhun V: Antitumor efficacy of
autovaccines prepared from chemoresistant tumor cells with the use
of lectin OF B. subtilis B-7025. Exp Oncol. 29:277–280. 2007.
|
|
41
|
Potebnya GP, Kudryavets YY, Lisovenko GS,
Cheremshenko NL, Voeykova IM, Trokhimenko NV, Symchich TV and
Evstrateyva LM: Experimental study of the efficacy of combined use
of cancer vaccine and interferon. Exp Oncol. 29:102–105.
2007.PubMed/NCBI
|
|
42
|
Potebnya GP, Voeykova IM, Lisovenko GS,
Cheremshenko NL, Todor IM, Yudina OY, Kovtonyuk OV and Chekhun VF:
Antitumor and antimetastatic activities of vaccine prepared from
cisplatin-resistant lewis lung carcinoma. Exp Oncol. 31:226–230.
2009.PubMed/NCBI
|
|
43
|
Xu X, Huang Q, Mao Y, Cui Z, Li Y, Huang
Y, Rajput IR, Yu D and Li W: Immunomodulatory effects of Bacillus
subtilis (natto) B4 spores on murine macrophages. Microbiol
Immunol. 56:817–824. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lee SW, Park HJ, Park SH, Kim N and Hong
S: Immunomodulatory effect of poly-γ-glutamic acid derived from
Bacillus subtilis on natural killer dendritic cells. Biochem
Biophys Res Commun. 443:413–421. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Langemann T, Koller VJ, Muhammad A, Kudela
P, Mayr UB and Lubitz W: The Bacterial ghost platform system:
Production and applications. Bioeng Bugs. 1:326–336. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Didenko GV, Dvorschenko OS, Lisovenko GS,
Kovalenko NG, Potebnya GP, Kikot VV, Pozur VK and Golub AA: The
modification of cancer vaccine prepared on the base of metabolic
products of B. subtilis 7025 with the use of sorbents and
automacrophages. Exp Oncol. 25:116–118. 2003.
|
|
47
|
Didenko GV, Kuzmenko AP, Shpak EG,
Tawrovska IA, Nadirashvili NA, Blum IO and Potebnya GP:
Optimization of the methods of isolation, electrophoretic
characterization, and antitumor efficacy of cytotoxic metabolites
from the culture filtrate Bacillus subtilis B-7025. Dop. NAS
Ukraine. 7:185–190. 2012.
|
|
48
|
Lowry OH, Rosebrough NJ, Farr AL and
Randall RJ: Protein measurement with the Folin phenol reagent. J
Biol Chem. 193:265–275. 1951.PubMed/NCBI
|
|
49
|
Niu PG, Zhang YX, Shi DH, Liu Y, Chen YY
and Deng J: Cardamonin inhibits metastasis of Lewis lung carcinoma
cells by decreasing mTOR activity. PLoS One. 10:e01277782015.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Schneider CA, Rasband WS and Eliceiri KW:
NIH Image to ImageJ: 25 years of image analysis. Nat Methods.
9:671–675. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Schirrmacher V, Fournier P and Schlag P:
Autologous tumor cell vaccines for post-operative active-specific
immunotherapy of colorectal carcinoma: Long-term patient survival
and mechanism of function. Expert Rev Vaccines. 13:117–130. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Laufer I, Iorgulescu JB, Chapman T, Lis E,
Shi W, Zhang Z, Cox BW, Yamada Y and Bilsky MH: Local disease
control for spinal metastases following ‘separation surgery’ and
adjuvant hypofractionated or high-dose single-fraction stereotactic
radiosurgery: Outcome analysis in 186 patients. J Neurosurg Spine.
18:207–214. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Brandau S and Suttmann H: Thirty years of
BCG immunotherapy for non-muscle invasive bladder cancer: A success
story with room for improvement. Biomed Pharmacother. 61:299–305.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Rothman J and Paterson Y: Live-attenuated
Listeria-based immunotherapy. Expert Rev Vaccines. 12:493–504.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Jahan T, Hassan R, Alley E, Kindler H,
Antonia S, Whiting C, Coussens L, Murphy AL, Thomas A and
Brockstedt DG: 208O_PR: CRS-207 with chemotherapy (chemo) in
malignant pleural mesothelioma (MPM): results from a phase 1b
trial. J Thorac Oncol. 11:(4 Suppl). S1562016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Becht E, Giraldo NA, Dieu-Nosjean MC,
Sautès-Fridman C and Fridman WH: Cancer immune contexture and
immunotherapy. Curr Opin Immunol. 39:7–13. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Kawahara M and Takaku H: A tumor lysate is
an effective vaccine antigen for the stimulation of CD4(+) T-cell
function and subsequent induction of antitumor immunity mediated by
CD8(+) T cells. Cancer Biol Ther. 16:1616–1625. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Tjomsland V, Spångeus A, Sandström P,
Borch K, Messmer D and Larsson M: Semi mature blood dendritic cells
exist in patients with ductal pancreatic adenocarcinoma owing to
inflammatory factors released from the tumor. PLoS One.
5:e134412010. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Kúdela P, Schwarczová Z, Sedlák J and
Bizik J: Conditioned medium from HeLa cells enhances motility of
human monocyte-derived dendritic cells but abrogates their
maturation and endocytic activity. Neoplasma. 48:382–388.
2001.PubMed/NCBI
|