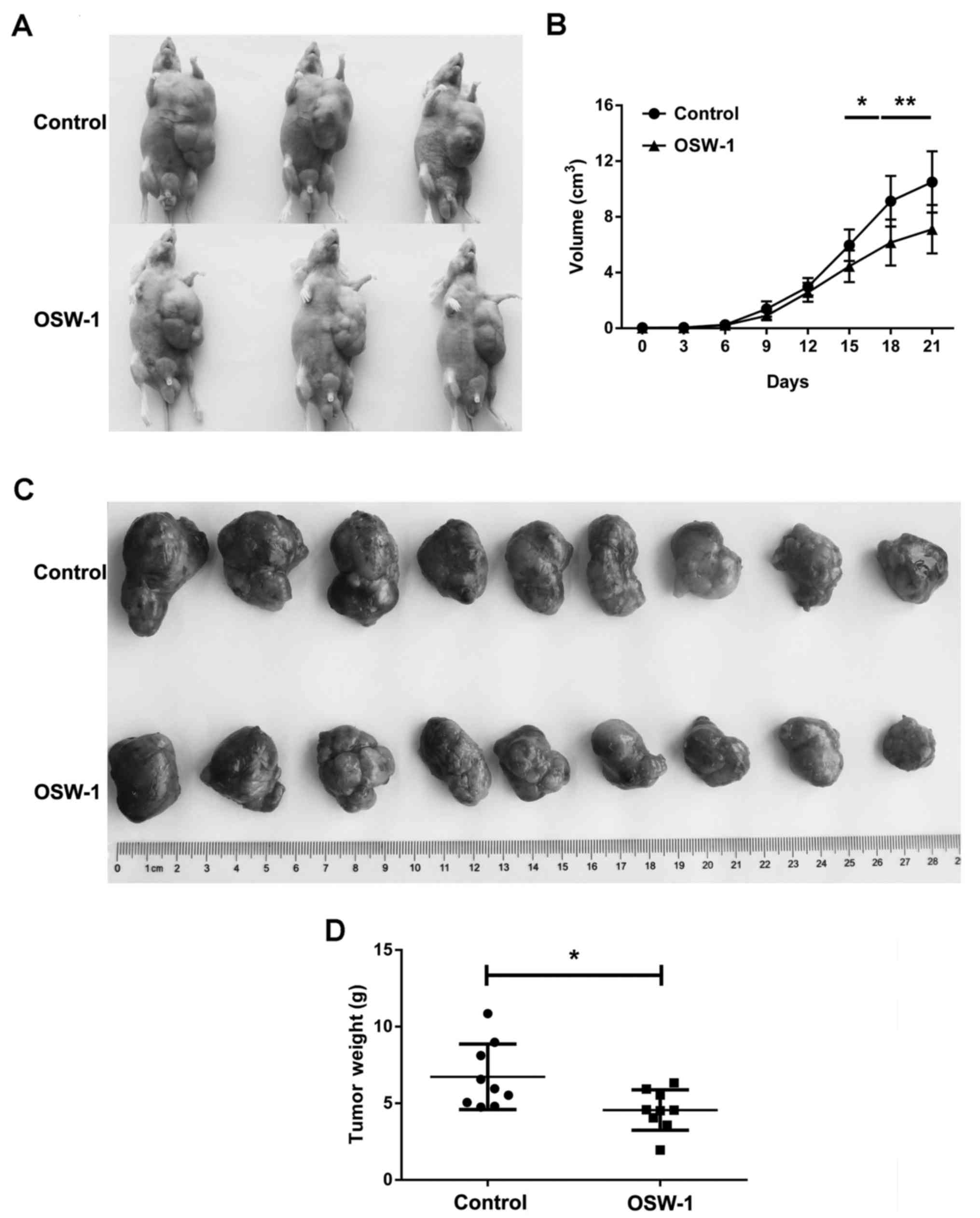
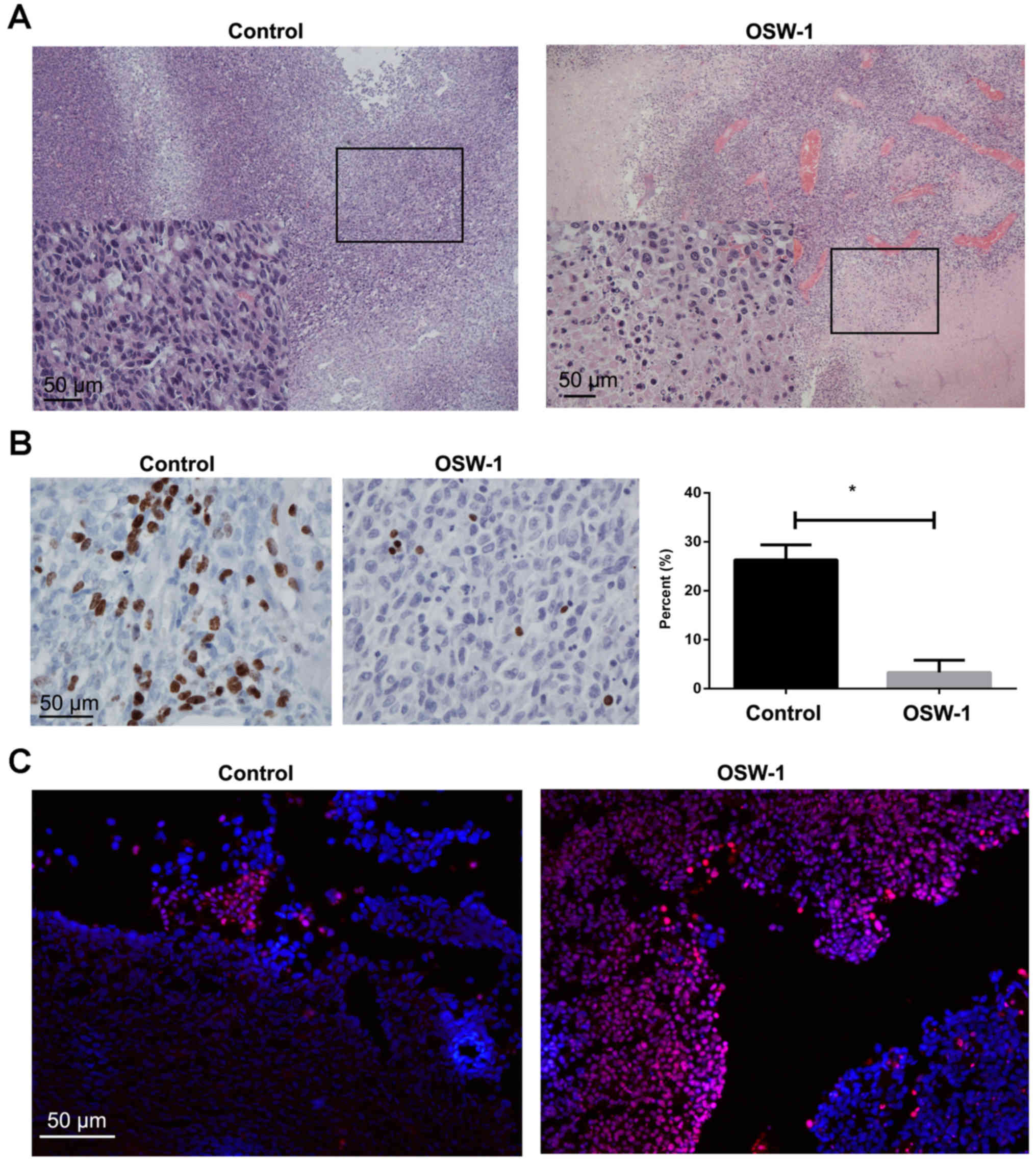
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wani MC, Taylor HL, Wall ME, Coggon P and
McPhail AT: Plant antitumor agents. VI. The isolation and structure
of taxol, a novel antileukemic and antitumor agent from Taxus
brevifolia. J Am Chem Soc. 93:2325–2327. 1971. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kubo S, Mimaki Y, Terao M, Sashida Y,
Nikaido T and Ohmoto T: Acylated cholestane glycosides from the
bulbs of Ornithogalum saundersiae. Phytochemistry. 31:3969–3973.
1992. View Article : Google Scholar
|
|
4
|
Tang Y, Li N, Duan JA and Tao W:
Structure, bioactivity, and chemical synthesis of OSW-1 and other
steroidal glycosides in the genus Ornithogalum. Chem Rev.
113:5480–5514. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhou Y, Garcia-Prieto C, Carney DA, Xu RH,
Pelicano H, Kang Y, Yu W, Lou C, Kondo S, Liu J, et al: OSW-1: A
natural compound with potent anticancer activity and a novel
mechanism of action. J Natl Cancer Inst. 97:1781–1785. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jin J, Jin X, Qian C, Ruan Y and Jiang H:
Signaling network of OSW-1-induced apoptosis and necroptosis in
hepatocellular carcinoma. Mol Med Rep. 7:1646–1650. 2013.PubMed/NCBI
|
|
7
|
Deng S, Yu B, Lou Y and Hui Y: First Total
Synthesis of an Exceptionally Potent Antitumor Saponin, OSW-1. J
Org Chem. 64:202–208. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ma X, Yu B, Hui Y, Xiao D and Ding J:
Synthesis of glycosides bearing the disaccharide of OSW-1 or its
1->4-linked analogue and their antitumor activities. Carbohydr
Res. 329:495–505. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yu W and Jin Z: A new strategy for the
stereoselective introduction of steroid side chain via alpha-alkoxy
vinyl cuprates: Total synthesis of a highly potent antitumor
natural product OSW-1. J Am Chem Soc. 123:3369–3370. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ma X, Yu B, Hui Y, Miao Z and Ding J:
Synthesis of steroidal glycosides bearing the disaccharide moiety
of OSW-1 and their antitumor activities. Carbohydr Res.
334:159–164. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ma X, Yu B, Hui Y, Miao Z and Ding J:
Synthesis of OSW-1 analogues and a dimer and their antitumor
activities. Bioorg Med Chem Lett. 11:2153–2156. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Morzycki JW and Wojtkielewicz A: Synthesis
of a cholestane glycoside OSW-1 with potent cytostatic activity.
Carbohydr Res. 337:1269–1274. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yu W and Jin Z: Total synthesis of the
anticancer natural product OSW-1. J Am Chem Soc. 124:6576–6583.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Deng L, Wu H, Yu B, Jiang M and Wu J:
Synthesis of OSW-1 analogs with modified side chains and their
antitumor activities. Bioorg Med Chem Lett. 14:2781–2785. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Morzycki JW, Wojtkielewicz A and
Wołczyński S: Synthesis of analogues of a potent antitumor saponin
OSW-1. Bioorg Med Chem Lett. 14:3323–3326. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Peng W, Tang P, Hu X, Liu JO and Yu B:
Synthesis of the A, B-ring-truncated OSW saponin analogs and their
antitumor activities. Bioorg Med Chem Lett. 17:5506–5509. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tang P, Mamdani F, Hu X, Liu JO and Yu B:
Synthesis of OSW saponin analogs with modified sugar residues and
their antiproliferative activities. Bioorg Med Chem Lett.
17:1003–1007. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xue J, Liu P, Pan Y and Guo Z: A total
synthesis of OSW-1. J Org Chem. 73:157–161. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kang Y, Lou C, Ahmed KB, Huang P and Jin
Z: Synthesis of biotinylated OSW-1. Bioorg Med Chem Lett.
19:5166–5168. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zheng D, Guan Y, Chen X, Xu Y, Chen X and
Lei P: Synthesis of cholestane saponins as mimics of OSW-1 and
their cytotoxic activities. Bioorg Med Chem Lett. 21:3257–3260.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Guan Y, Zheng D, Zhou L, Wang H, Yan Z,
Wang N, Chang H, She P and Lei P: Synthesis of 5(6)-dihydro-OSW-1
analogs bearing three kinds of disaccharides linking at 15-hydroxy
and their antitumor activities. Bioorg Med Chem Lett. 21:2921–2924.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Maj J, Morzycki JW, Rárová L, Oklest'ková
J, Strnad M and Wojtkielewicz A: Synthesis and biological activity
of 22-deoxo-23-oxa analogues of saponin OSW-1. J Med Chem.
54:3298–3305. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sakurai K, Takeshita T, Hiraizumi M and
Yamada R: Synthesis of OSW-1 derivatives by site-selective
acylation and their biological evaluation. Org Lett. 16:6318–6321.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Guan YY, Song C and Lei PS: Synthesis of
three OSW-1 analogs with maltose side chains bearing different
protection groups. J Asian Nat Prod Res. 16:43–52. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zongxu Han: The preparation method of
compound Wannianqing capsules Chinese patent CN 1,352,986. June
12–2002
|
|
26
|
Zongxu Han: The preparation method of
anti-liver-cancer drug Chinese patent. CN 1,354,011. June
19–2002
|
|
27
|
Xinbo LV and Zongxu Han: The preparation
method of Ornithogalum saundersiae extract and its application
Chinese patent. CN 1,672,728. September 28–2005
|
|
28
|
Mimaki Y, Kuroda M, Kameyama A, Sashida Y,
Hirano T, Oka K, Maekawa R, Wada T, Sugita K and Beutler JA:
Cholestane glycosides with potent cytostatic activities on various
tumor cells from Ornithogalum saundersiae Bulbs. Bioorg Med Chem
Lett. 7:633–636. 1997. View Article : Google Scholar
|
|
29
|
Melstrom LG, Bentrem DJ, Salabat MR,
Kennedy TJ, Ding XZ, Strouch M, Rao SM, Witt RC, Ternent CA,
Talamonti MS, et al: Overexpression of 5-lipoxygenase in colon
polyps and cancer and the effect of 5-LOX inhibitors in vitro and
in a murine model. Clin Cancer Res. 14:6525–6530. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hewitt RE, McMarlin A, Kleiner D, Wersto
R, Martin P, Tsokos M, Stamp GW and Stetler-Stevenson WG:
Validation of a model of colon cancer progression. J Pathol.
192:446–454. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Drewinko B, Romsdahl MM, Yang LY, Ahearn
MJ and Trujillo JM: Establishment of a human carcinoembryonic
antigen-producing colon adenocarcinoma cell line. Cancer Res.
36:467–475. 1976.PubMed/NCBI
|
|
32
|
Jing L, He MT, Chang Y, Mehta SL, He QP,
Zhang JZ and Li PA: Coenzyme Q10 protects astrocytes from
ROS-induced damage through inhibition of mitochondria-mediated cell
death pathway. Int J Biol Sci. 11:59–66. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Beauregard AP, Harquail J, Lassalle-Claux
G, Belbraouet M, Jean-Francois J, Touaibia M and Robichaud GA: CAPE
analogs induce growth arrest and apoptosis in breast cancer cells.
Molecules. 20:12576–12589. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Garcia-Prieto C, Ahmed KB Riaz, Chen Z,
Zhou Y, Hammoudi N, Kang Y, Lou C, Mei Y, Jin Z and Huang P:
Effective killing of leukemia cells by the natural product OSW-1
through disruption of cellular calcium homeostasis. J Biol Chem.
288:3240–3250. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhu J, Xiong L, Yu B and Wu J: Apoptosis
induced by a new member of saponin family is mediated through
caspase-8-dependent cleavage of Bcl-2. Mol Pharmacol. 68:1831–1838.
2005.PubMed/NCBI
|
|
36
|
Bianchi K, Rimessi A, Prandini A,
Szabadkai G and Rizzuto R: Calcium and mitochondria: Mechanisms and
functions of a troubled relationship. Biochim Biophys Acta.
1742:119–131. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zimmermann KC, Bonzon C and Green DR: The
machinery of programmed cell death. Pharmacol Ther. 92:57–70. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Oberst A, Bender C and Green DR: Living
with death: The evolution of the mitochondrial pathway of apoptosis
in animals. Cell Death Differ. 15:1139–1146. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Jin JC, Jin XL, Zhang X, Piao YS and Liu
SP: Effect of OSW-1 on microRNA expression profiles of hepatoma
cells and functions of novel microRNAs. Mol Med Rep. 7:1831–1837.
2013.PubMed/NCBI
|
|
40
|
Nagata S: Apoptosis by death factor. Cell.
88:355–365. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ellis RE, Yuan JY and Horvitz HR:
Mechanisms and functions of cell death. Annu Rev Cell Biol.
7:663–698. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Igney FH and Krammer PH: Death and
anti-death: Tumour resistance to apoptosis. Nat Rev Cancer.
2:277–288. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
43
|
Shen L, Li J, Xu L, Ma J, Li H, Xiao X,
Zhao J and Fang L: miR-497 induces apoptosis of breast cancer cells
by targeting Bcl-w. Exp Ther Med. 3:475–480. 2012.PubMed/NCBI
|
|
44
|
Kim SH and Choi KC: Anti-cancer effect and
underlying mechanism(s) of kaempferol, a phytoestrogen, on the
regulation of apoptosis in diverse cancer cell models. Toxicol Res.
29:229–234. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kasiri S, Ansari KI, Hussain I, Bhan A and
Mandal SS: Antisense oligonucleotide mediated knockdown of HOXC13
affects cell growth and induces apoptosis in tumor cells and over
expression of HOXC13 induces 3D-colony formation. RSC Advances.
3:3260–3269. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ponci V, Figueiredo CR, Massaoka MH, de
Farias CF, Matsuo AL, Sartorelli P and Lago JH: Neolignans from
Nectandra megapotamica (Lauraceae) display in vitro cytotoxic
activity and induce apoptosis in leukemia cells. Molecules.
20:12757–12768. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Green DR: Apoptotic pathways: Paper wraps
stone blunts scissors. Cell. 102:1–4. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Fulda S and Debatin KM: Extrinsic versus
intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene.
25:4798–4811. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kroemer G and Reed JC: Mitochondrial
control of cell death. Nat Med. 6:513–519. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Mattson MP and Chan SL: Calcium
orchestrates apoptosis. Nat Cell Biol. 5:1041–1043. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Zamzami N and Kroemer G: The mitochondrion
in apoptosis: How Pandora's box opens. Nat Rev Mol Cell Biol.
2:67–71. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Martinou JC and Green DR: Breaking the
mitochondrial barrier. Nat Rev Mol Cell Biol. 2:63–67. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kulikov AV, Shilov ES, Mufazalov IA,
Gogvadze V, Nedospasov SA and Zhivotovsky B: Cytochrome c: The
Achilles' heel in apoptosis. Cell Mol Life Sci. 69:1787–1797. 2012.
View Article : Google Scholar : PubMed/NCBI
|