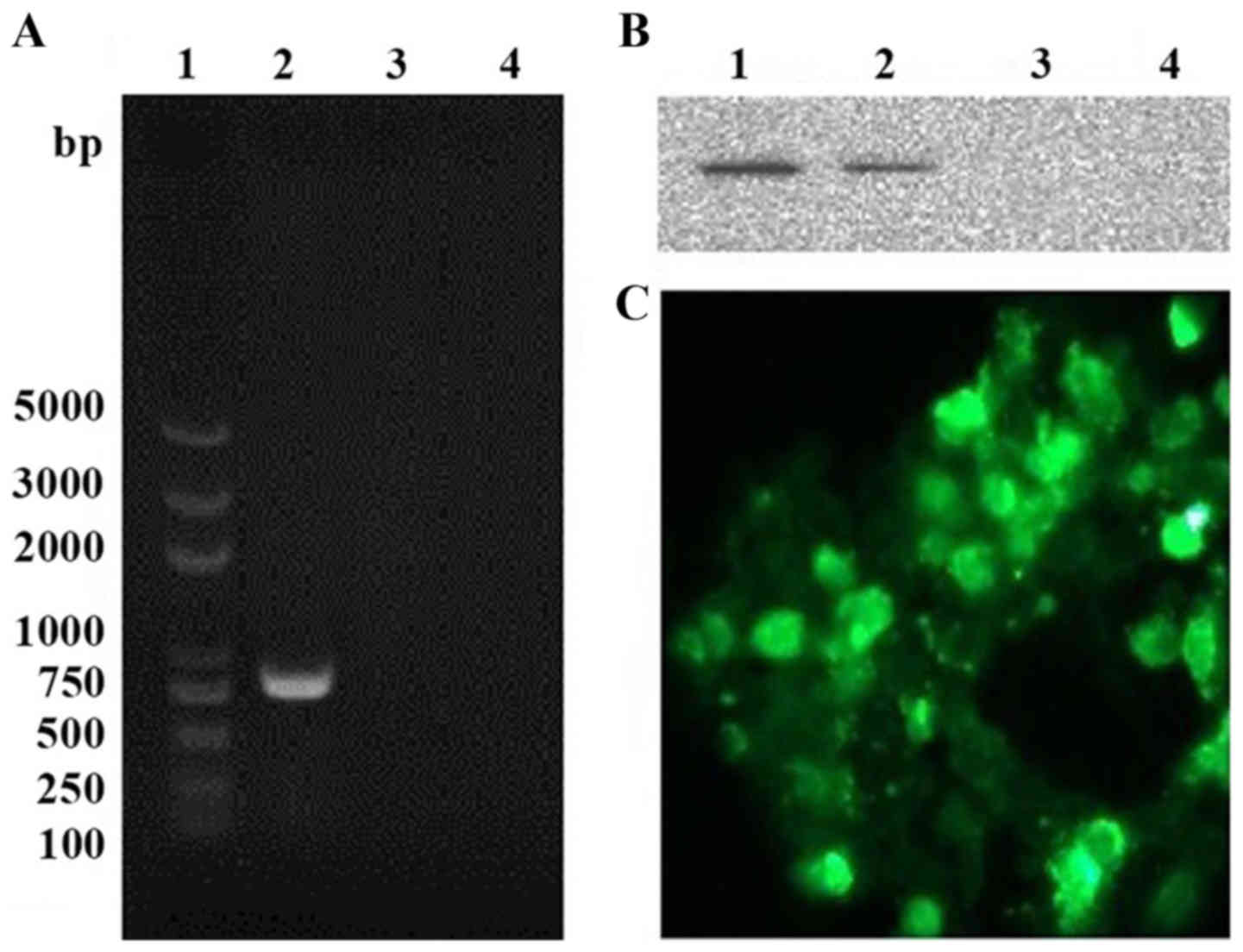
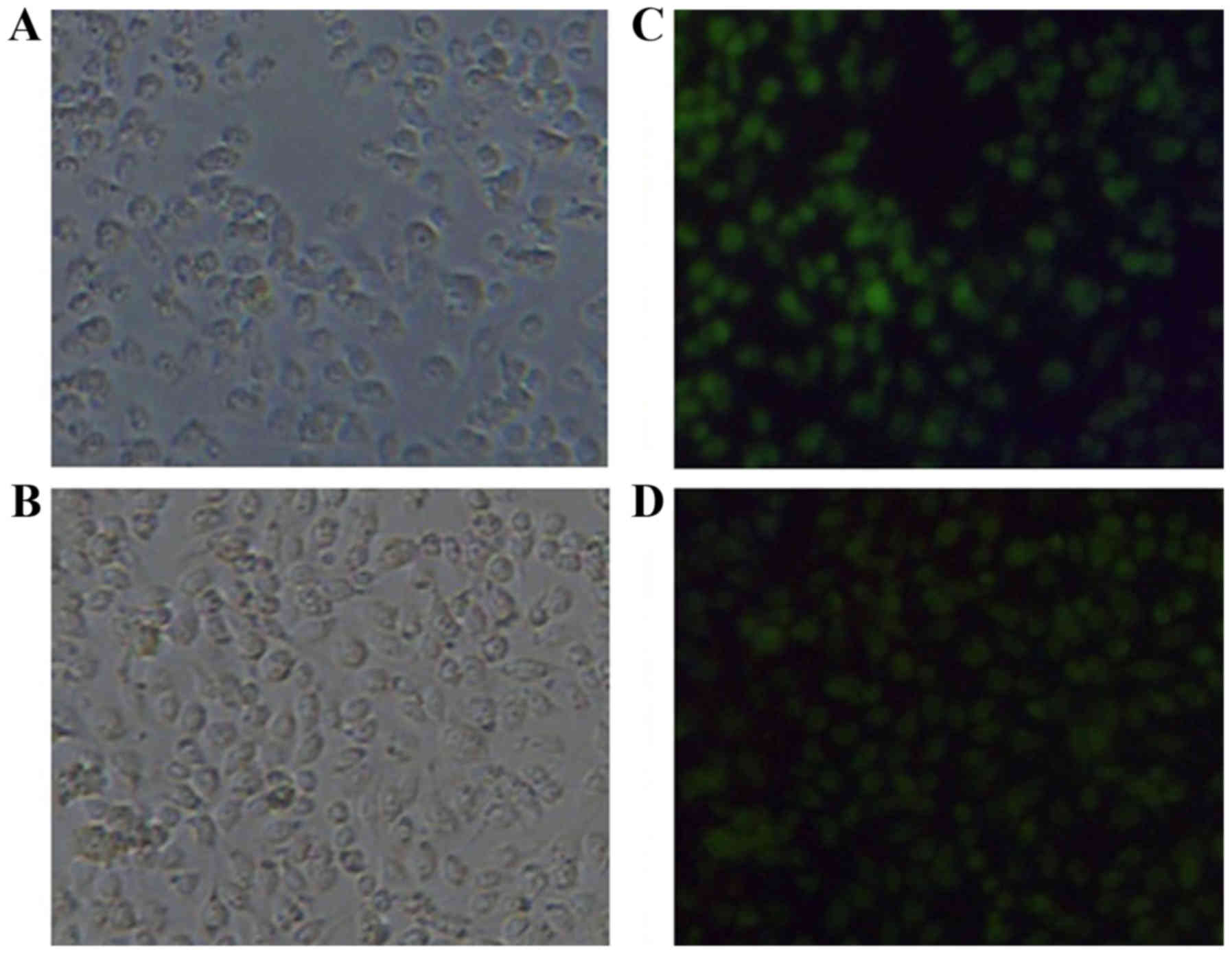
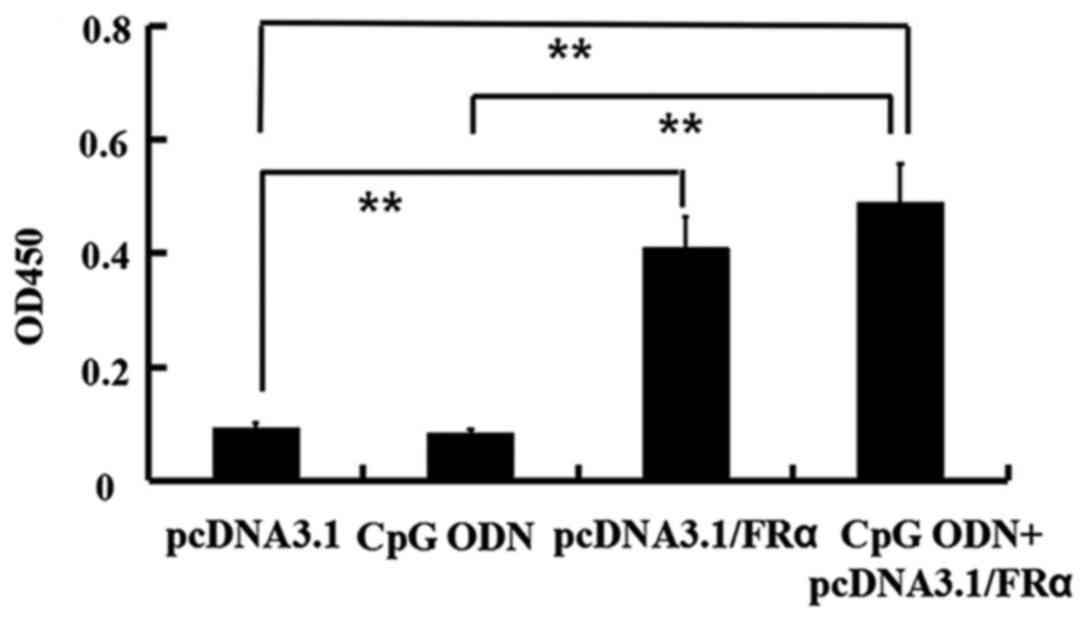
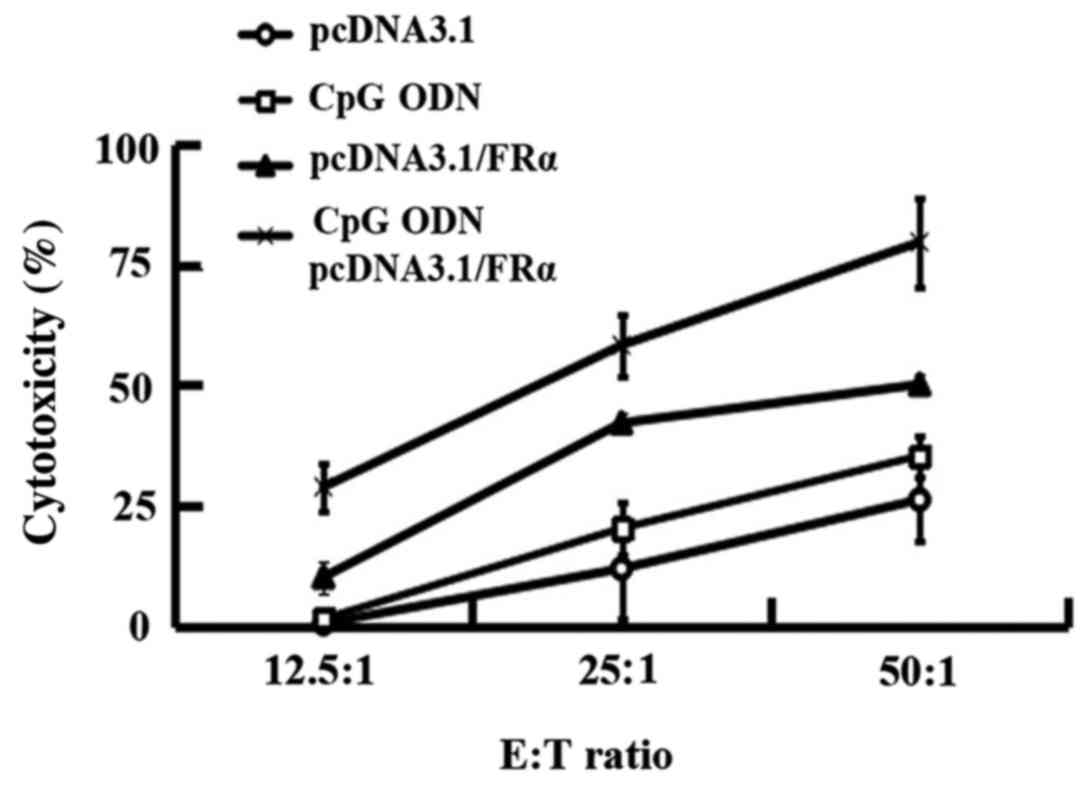
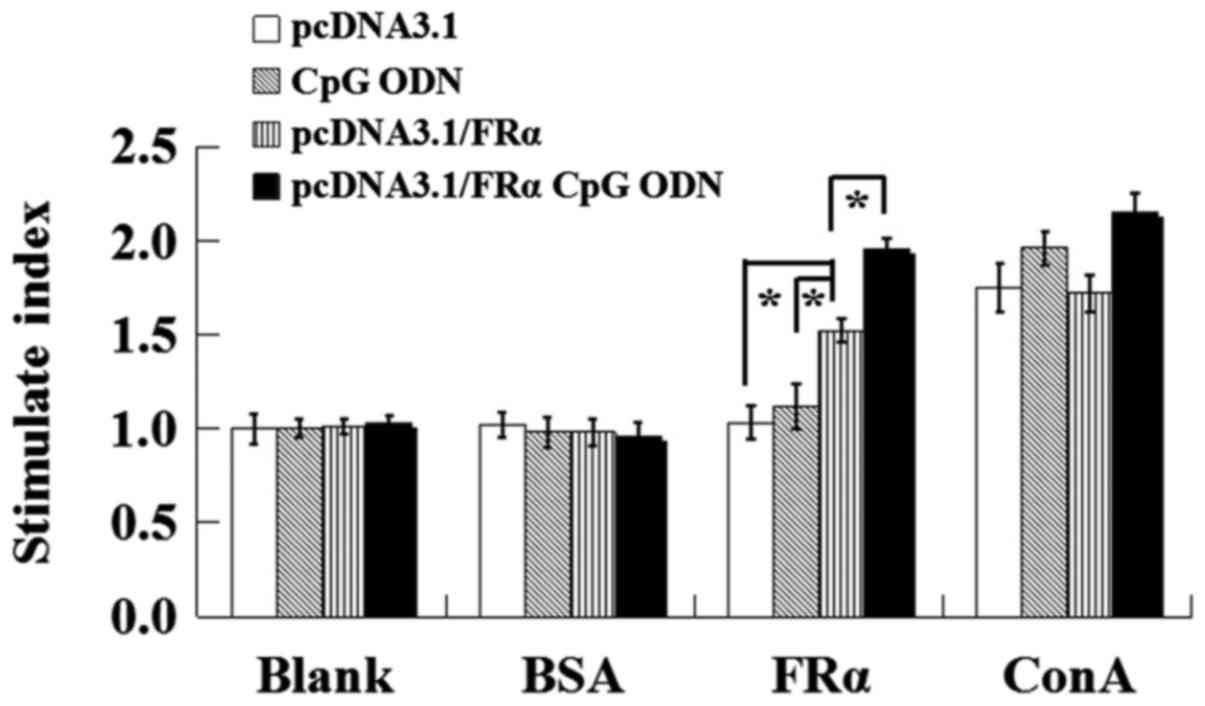
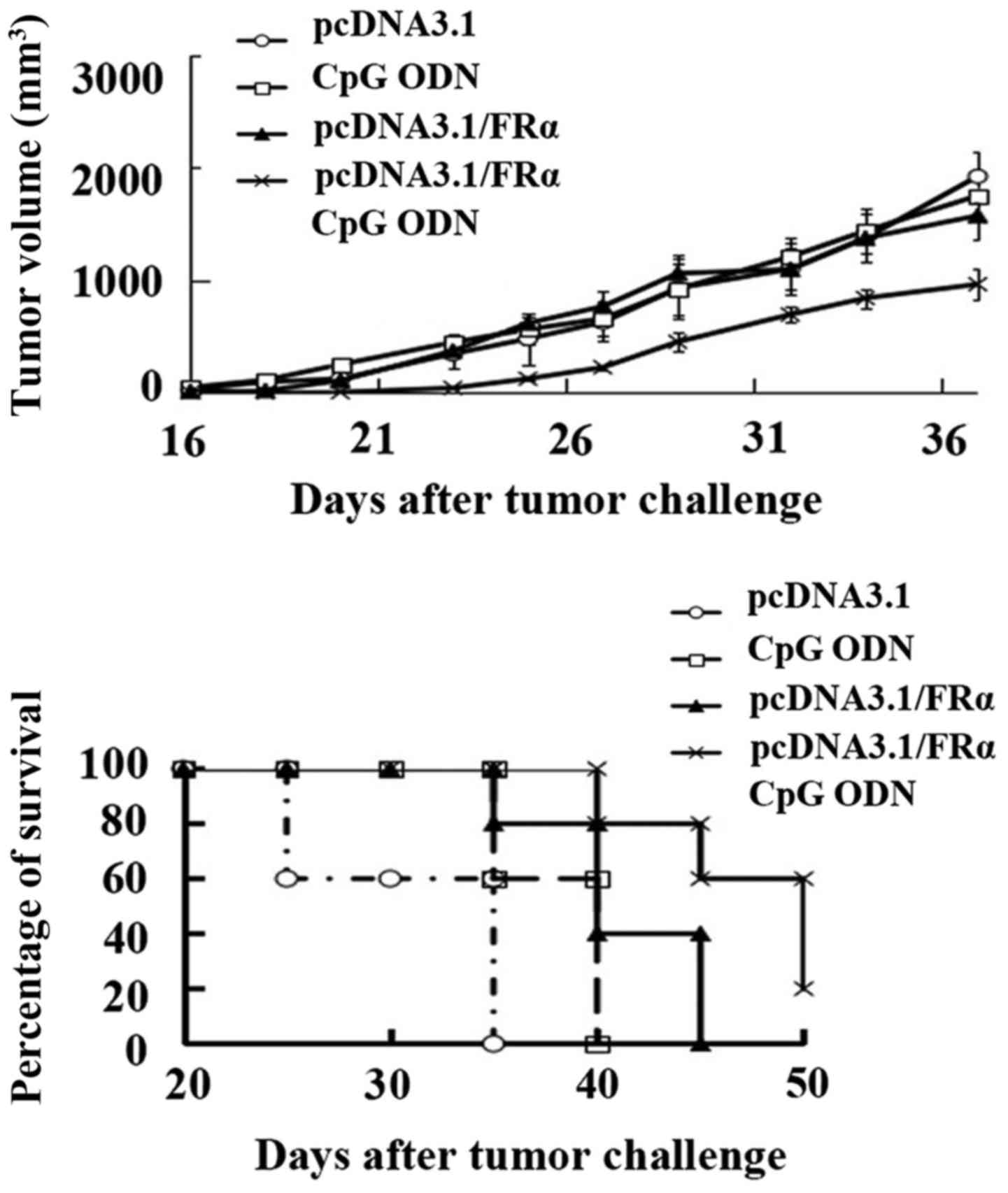
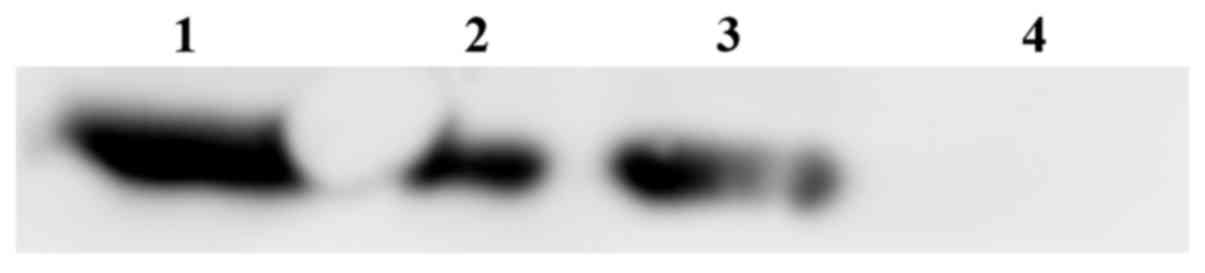
|
1
|
Delany I, Rappuoli R and De Gregorio E:
Vaccines for the 21st century. EMBO Mol Med. 6:708–720.
2014.PubMed/NCBI
|
|
2
|
Vanneman M and Dranoff G: Combining
immunotherapy and targeted therapies in cancer treatment. Nat Rev
Cancer. 12:237–251. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Houghton AN: Cancer antigens: Immune
recognition of self and altered self. J Exp Med. 180:1–4. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wolff JA, Malone RW, Williams P, Chong W,
Acsadi G, Jani A and Felgner PL: Direct gene transfer into mouse
muscle in vivo. Science. 247:1465–1468. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fioretti D, Iurescia S, Fazio VM and
Rinaldi M: DNA vaccines: Developing new strategies against cancer.
J Biomed Biotechnol. 2010:1743782010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li L, Saade F and Petrovsky N: The future
of human DNA vaccines. J Biotechnol. 162:171–182. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Latz E, Schoenemeyer A, Visintin A,
Fitzgerald KA, Monks BG, Knetter CF, Lien E, Nilsen NJ, Espevik T
and Golenbock DT: TLR9 signals after translocating from the ER to
CpG DNA in the lysosome. Nat Immunol. 5:190–198. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Krieg AM: Therapeutic potential of
Toll-like receptor 9 activation. Nat Rev Drug Discov. 5:471–484.
2006. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Klinman DM, Currie D, Lee G, Grippe V and
Merkel T: Systemic but not mucosal immunity induced by AVA prevents
inhalational anthrax. Microbes Infect. 9:1478–1483. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ohno S, Okuyama R, Aruga A, Sugiyama H and
Yamamoto M: Phase I trial of Wilms' Tumor 1 (WT1) peptide vaccine
with GM-CSF or CpG in patients with solid malignancy. Anticancer
Res. 32:2263–2269. 2012.PubMed/NCBI
|
|
11
|
Mutwiri GK, Nichani AK, Babiuk S and
Babiuk LA: Strategies for enhancing the immunostimulatory effects
of CpG oligodeoxynucleotides. J Control Release. 97:1–17. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vollmer J and Krieg AM: Immunotherapeutic
applications of CpG oligodeoxynucleotide TLR9 agonists. Adv Drug
Deliv Rev. 61:195–204. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Halperin SA, van Nest G, Smith B, Abtahi
S, Whiley H and Eiden JJ: A phase I study of the safety and
immunogenicity of recombinant hepatitis B surface antigen
co-administered with an immunostimulatory phosphorothioate
oligonucleotide adjuvant. Vaccine. 21:2461–2467. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Elnakat H and Ratnam M: Role of folate
receptor genes in reproduction and related cancers. Front Biosci.
11:506–519. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Parker N, Turk MJ, Westrick E, Lewis JD,
Low PS and Leamon CP: Folate receptor expression in carcinomas and
normal tissues determined by a quantitative radioligand binding
assay. Anal Biochem. 338:284–293. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Basal E, Eghbali-Fatourechi GZ, Kalli KR,
Hartmann LC, Goodman KM, Goode EL, Kamen BA, Low PS and Knutson KL:
Functional folate receptor alpha is elevated in the blood of
ovarian cancer patients. PLoS One. 4:e62922009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kalli KR, Oberg AL, Keeney GL,
Christianson TJ, Low PS, Knutson KL and Hartmann LC: Folate
receptor alpha as a tumor target in epithelial ovarian cancer.
Gynecol Oncol. 108:619–626. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Toffoli G, Cernigoi C, Russo A, Gallo A,
Bagnoli M and Boiocchi M: Overexpression of folate binding protein
in ovarian cancers. Int J Cancer. 74:193–198. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Despierre E, Lambrechts S, Leunen K,
Berteloot P, Neven P, Amant F, O'Shannessy DJ, Somers EB and
Vergote I: Folate receptor alpha (FRA) expression remains unchanged
in epithelial ovarian and endometrial cancer after chemotherapy.
Gynecol Oncol. 130:192–199. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ebel W, Routhier EL, Foley B, Jacob S,
McDonough JM, Patel RK, Turchin HA, Chao Q, Kline JB, Old LJ, et
al: Preclinical evaluation of MORAb-003, a humanized monoclonal
antibody antagonizing folate receptor-alpha. Cancer Immun. 7:6–13.
2007.PubMed/NCBI
|
|
21
|
Armstrong DK, White AJ, Weil SC, Phillips
M and Coleman RL: Farletuzumab (a monoclonal antibody against
folate receptor alpha) in relapsed platinum-sensitive ovarian
cancer. Gynecol Oncol. 129:452–458. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Walters CL, Arend RC, Armstrong DK,
Naumann RW and Alvarez RD: Folate and folate receptor alpha
antagonists mechanism of action in ovarian cancer. Gynecol Oncol.
131:493–498. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chu RS, Targoni OS, Krieg AM, Lehmann PV
and Harding CV: CpG oligodeoxynucleotides act as adjuvants that
switch on T helper 1 (Th1) immunity. J Exp Med. 186:1623–1631.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hailemichael Y and Overwijk WW: Cancer
vaccines: Trafficking of tumor-specific T cells to tumor after
therapeutic vaccination. Int J Biochem Cell Biol. 53:46–50. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lindblad EB: Aluminium compounds for use
in vaccines. Immunol Cell Biol. 82:497–505. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cooper CL, Davis HL, Angel JB, Morris ML,
Elfer SM, Seguin I, Krieg AM and Cameron DW: CPG 7909 adjuvant
improves hepatitis B virus vaccine seroprotection in
antiretroviral-treated HIV-infected adults. AIDS. 19:1473–1479.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Perales MA, Yuan J, Powel S, Gallardo HF,
Rasalan TS, Gonzalez C, Manukian G, Wang J, Zhang Y, Chapman PB, et
al: Phase I/II study of GM-CSF DNA as an adjuvant for a
multipeptide cancer vaccine in patients with advanced melanoma. Mol
Ther. 16:2022–2029. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Krug LM, Dao T, Brown AB, Maslak P, Travis
W, Bekele S, Korontsvit T, Zakhaleva V, Wolchok J, Yuan J, et al:
WT1 peptide vaccinations induce CD4 and CD8 T cell immune responses
in patients with mesothelioma and non-small cell lung cancer.
Cancer Immunol Immunother. 59:1467–1479. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Barve M, Bender J, Senzer N, Cunningham C,
Greco FA, McCune D, Steis R, Khong H, Richards D, Stephenson J, et
al: Induction of immune responses and clinical efficacy in a phase
II trial of IDM-2101, a 10-epitope cytotoxic T-lymphocyte vaccine,
in metastatic non-small-cell lung cancer. J Clin Oncol.
26:4418–4425. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Vansteenkiste J, Zielinski M, Linder A,
Dahabreh J, Gonzalez EE, Malinowski W, Lopez-Brea M, Vanakesa T,
Jassem J, Kalofonos H, et al: Adjuvant MAGE-A3 immunotherapy in
resected non-small-cell lung cancer: Phase II randomized study
results. J Clin Oncol. 31:2396–2403. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Baumgaertner P, Jandus C, Rivals JP, Derré
L, Lövgren T, Baitsch L, Guillaume P, Luescher IF, Berthod G,
Matter M, et al: Vaccination-induced functional competence of
circulating human tumor-specific CD8 T-cells. Int J Cancer.
130:2607–2617. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Speiser DE, Liénard D, Rufer N,
Rubio-Godoy V, Rimoldi D, Lejeune F, Krieg AM, Cerottini JC and
Romero P: Rapid and strong human CD8+ T cell responses
to vaccination with peptide, IFA, and CpG oligodeoxynucleotide
7909. J Clin Invest. 115:739–746. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Neglia F, Orengo AM, Cilli M, Meazza R,
Tomassetti A, Canevari S, Melani C, Colombo MP and Ferrini S: DNA
vaccination against the ovarian carcinoma-associated antigen folate
receptor alpha (FRalpha) induces cytotoxic T lymphocyte and
antibody responses in mice. Cancer Gene Ther. 6:349–357. 1999.
View Article : Google Scholar : PubMed/NCBI
|