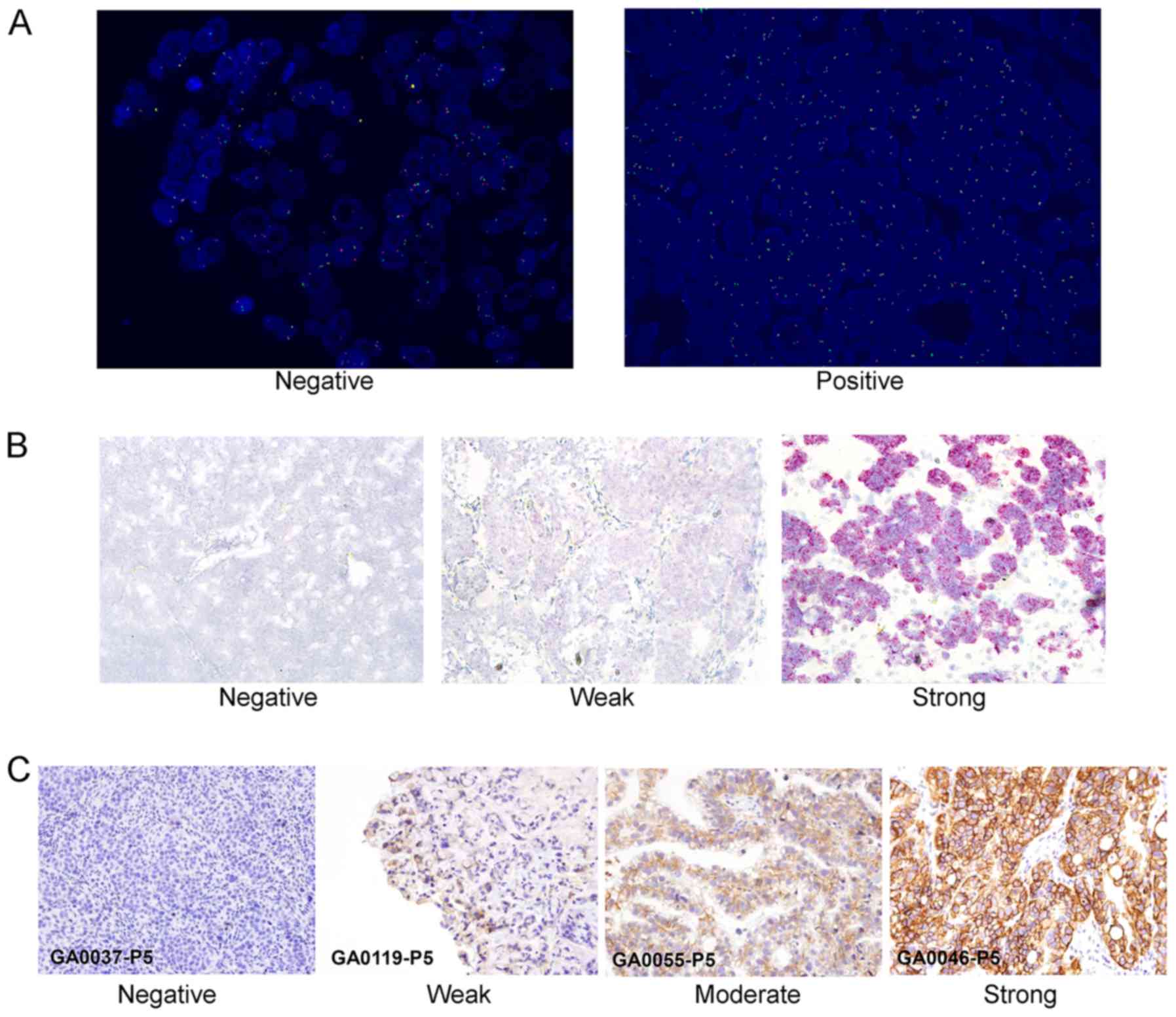
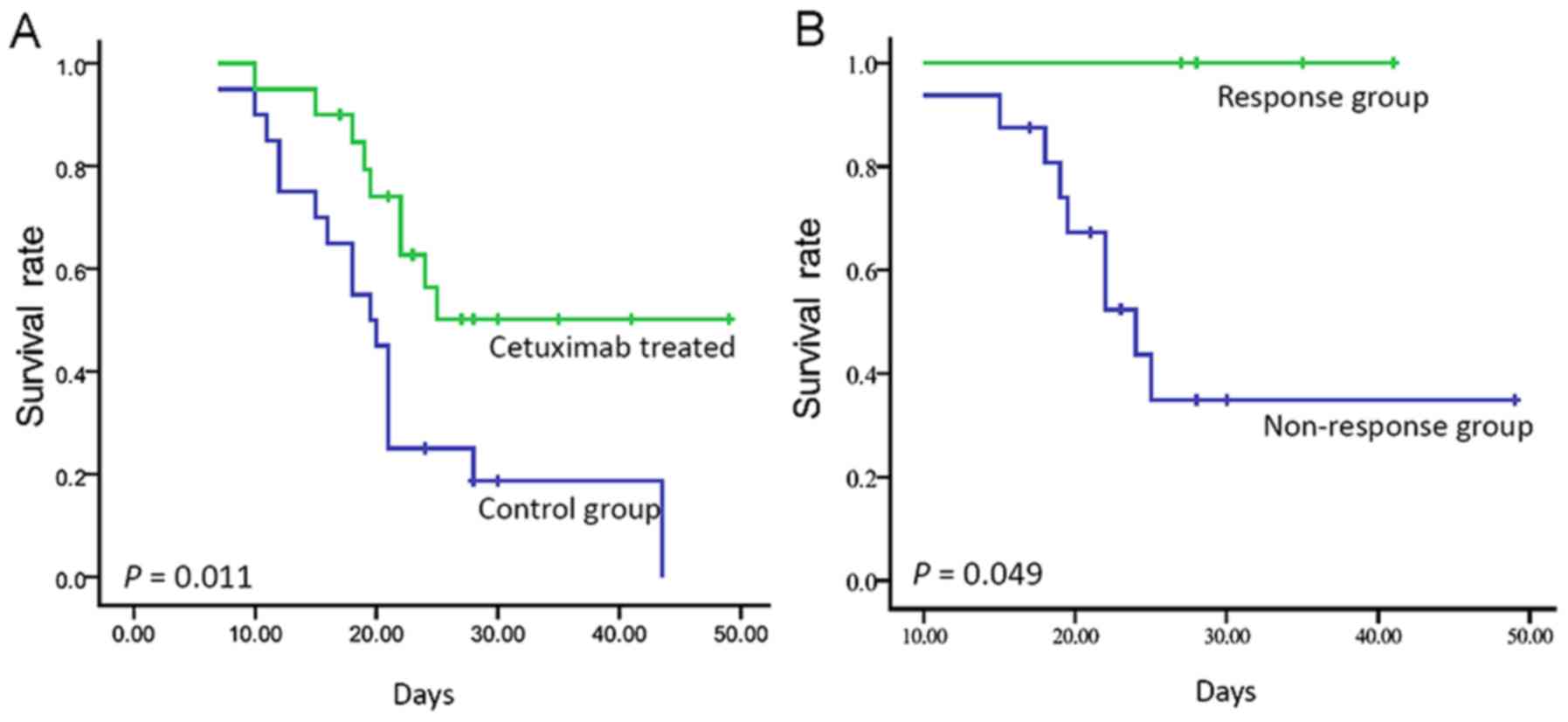
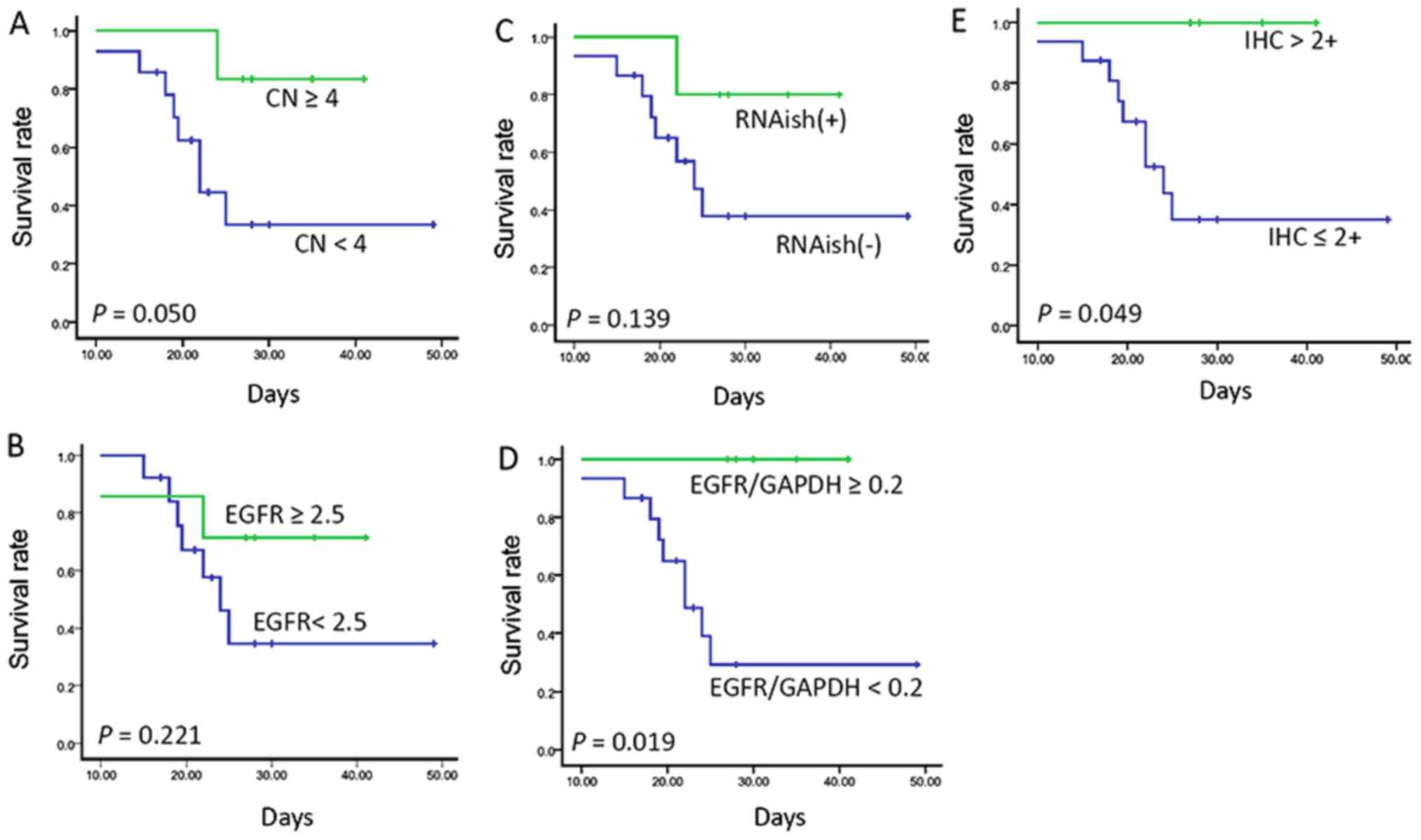
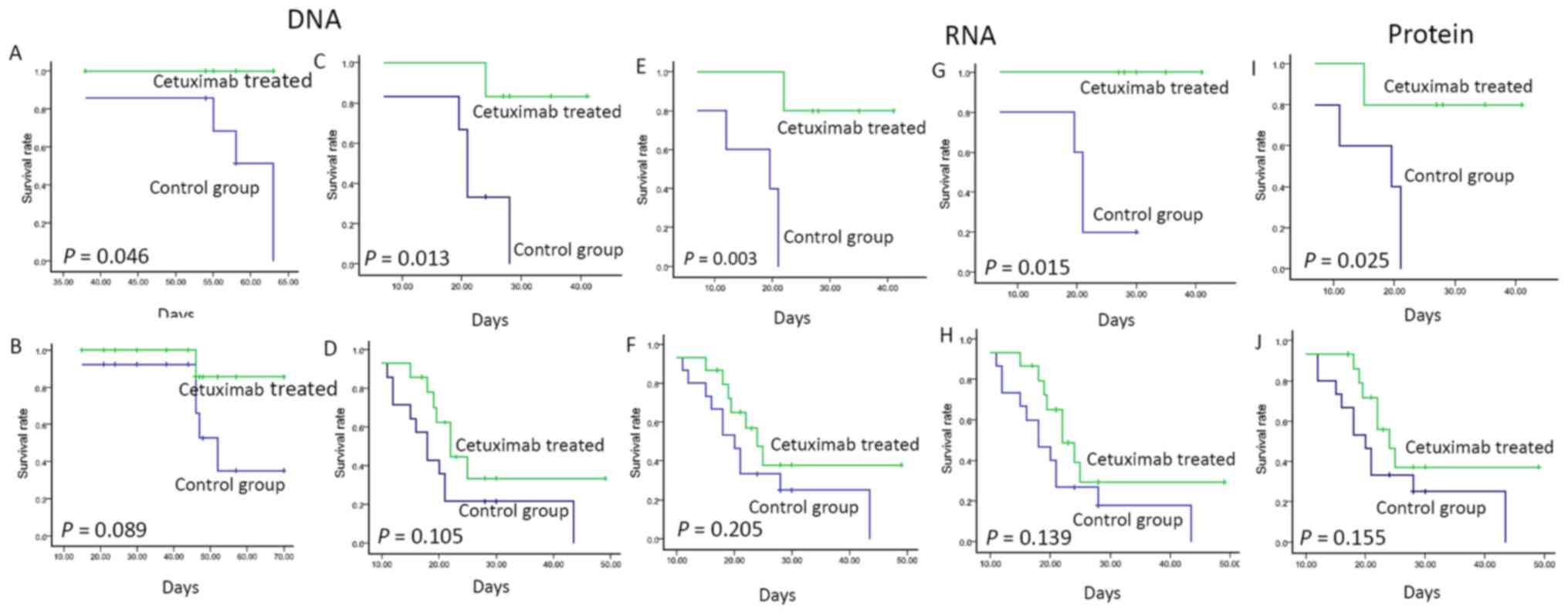
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gomez-Martin C, Plaza JC, Pazo-Cid R,
Salud A, Pons F, Fonseca P, Leon A, Alsina M, Visa L, Rivera F, et
al: Level of HER2 gene amplification predicts response and
overall survival in HER2-positive advanced gastric cancer treated
with trastuzumab. J Clin Oncol. 31:4445–4452. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Luo HY, Wei W, Shi YX, Chen XQ, Li YH,
Wang F, Qiu MZ, Li FH, Yan SL, Zeng MS, et al: Cetuximab enhances
the effect of oxaliplatin on hypoxic gastric cancer cell lines.
Oncol Rep. 23:1735–1745. 2010.PubMed/NCBI
|
|
4
|
Pinto C, Di Fabio F, Siena S, Cascinu S,
Llimpe Rojas FL, Ceccarelli C, Mutri V, Giannetta L, Giaquinta S,
Funaioli C, et al: Phase II study of cetuximab in combination with
FOLFIRI in patients with untreated advanced gastric or
gastroesophageal junction adenocarcinoma (FOLCETUX study). Ann
Oncol. 18:510–517. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Han SW, Oh DY, Im SA, Park SR, Lee KW,
Song HS, Lee NS, Lee KH, Choi IS, Lee MH, et al: Phase II study and
biomarker analysis of cetuximab combined with modified FOLFOX6 in
advanced gastric cancer. Br J Cancer. 100:298–304. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kim C, Lee JL, Ryu MH, Chang HM, Kim TW,
Lim HY, Kang HJ, Park YS, Ryoo BY and Kang YK: A prospective phase
II study of cetuximab in combination with XELOX (capecitabine and
oxaliplatin) in patients with metastatic and/or recurrent advanced
gastric cancer. Invest New Drugs. 29:366–373. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lordick F, Kang YK, Chung HC, Salman P, Oh
SC, Bodoky G, Kurteva G, Volovat C, Moiseyenko VM, Gorbunova V, et
al: Arbeitsgemeinschaft Internistische Onkologie and EXPAND
Investigators: Capecitabine and cisplatin with or without cetuximab
for patients with previously untreated advanced gastric cancer
(EXPAND): A randomised, open-label phase 3 trial. Lancet Oncol.
14:490–499. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Higaki E, Kuwata T, Nagatsuma AK, Nishida
Y, Kinoshita T, Aizawa M5, Nitta H, Nagino M and Ochiai A: Gene
copy number gain of EGFR is a poor prognostic biomarker in
gastric cancer: Evaluation of 855 patients with bright-field dual
in situ hybridization (DISH) method. Gastric Cancer. 19:63–73.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hotz B, Keilholz U, Fusi A, Buhr HJ and
Hotz HG: In vitro and in vivo antitumor activity of cetuximab in
human gastric cancer cell lines in relation to epidermal growth
factor receptor (EGFR) expression and mutational phenotype. Gastric
Cancer. 15:252–264. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhou C, Wu YL, Chen G, Feng J, Liu XQ,
Wang C, Zhang S, Wang J, Zhou S, Ren S, et al: Erlotinib versus
chemotherapy as first-line treatment for patients with advanced
EGFR mutation-positive non-small-cell lung cancer (OPTIMAL,
CTONG-0802): A multicentre, open-label, randomised, phase 3 study.
Lancet Oncol. 12:735–742. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Maemondo M, Inoue A, Kobayashi K, Sugawara
S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I,
et al: North-East Japan Study Group: Gefitinib or chemotherapy for
non-small-cell lung cancer with mutated EGFR. N Engl J Med.
362:2380–2388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mitsudomi T, Morita S, Yatabe Y, Negoro S,
Okamoto I, Tsurutani J, Seto T, Satouchi M, Tada H, Hirashima T, et
al: West Japan Oncology Group: Gefitinib versus cisplatin plus
docetaxel in patients with non-small-cell lung cancer harbouring
mutations of the epidermal growth factor receptor (WJTOG3405): An
open label, randomised phase 3 trial. Lancet Oncol. 11:121–128.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mazières J, Brugger W, Cappuzzo F, Middel
P, Frosch A, Bara I, Klingelschmitt G and Klughammer B: Evaluation
of EGFR protein expression by immunohistochemistry using H-score
and the magnification rule: Re-analysis of the SATURN study. Lung
Cancer. 82:231–237. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dahabreh IJ, Linardou H, Kosmidis P,
Bafaloukos D and Murray S: EGFR gene copy number as a
predictive biomarker for patients receiving tyrosine kinase
inhibitor treatment: A systematic review and meta-analysis in
non-small-cell lung cancer. Ann Oncol. 22:545–552. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Licitra L, Mesia R, Rivera F, Remenár E,
Hitt R, Erfán J, Rottey S, Kawecki A, Zabolotnyy D, Benasso M, et
al: Evaluation of EGFR gene copy number as a predictive
biomarker for the efficacy of cetuximab in combination with
chemotherapy in the first-line treatment of recurrent and/or
metastatic squamous cell carcinoma of the head and neck: EXTREME
study. Ann Oncol. 22:1078–1087. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang L, Yang J, Cai J, Song X, Deng J,
Huang X, Chen D, Yang M, Wery JP, Li S, et al: A subset of gastric
cancers with EGFR amplification and overexpression respond to
cetuximab therapy. Sci Rep. 3:29922013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Washington K: 7th edition of the AJCC
cancer staging manual: Stomach. Ann Surg Oncol. 17:3077–3079. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang M, Shan B, Li Q, Song X, Cai J, Deng
J, Zhang L, Du Z, Lu J, Chen T, et al: Overcoming erlotinib
resistance with tailored treatment regimen in patient-derived
xenografts from naïve Asian NSCLC patients. Int J Cancer.
132:E74–84. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Julien S, Merino-Trigo A, Lacroix L,
Pocard M, Goéré D, Mariani P, Landron S, Bigot L, Nemati F,
Dartigues P, et al: Characterization of a large panel of
patient-derived tumor xenografts representing the clinical
heterogeneity of human colorectal cancer. Clin Cancer Res.
18:5314–5328. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schleich N, Po C, Jacobs D, Ucakar B,
Gallez B, Danhier F and Préat V: Comparison of active, passive and
magnetic targeting to tumors of multifunctional
paclitaxel/SPIO-loaded nanoparticles for tumor imaging and therapy.
J Control Release. 194:82–91. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Waddell T, Chau I, Cunningham D, Gonzalez
D, Okines AF, Okines C, Wotherspoon A, Saffery C, Middleton G,
Wadsley J, et al: Epirubicin, oxaliplatin, and capecitabine with or
without panitumumab for patients with previously untreated advanced
oesophagogastric cancer (REAL3): A randomised, open-label phase 3
trial. Lancet Oncol. 14:481–489. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fox SB, Kumarasinghe MP, Armes JE, Bilous
M, Cummings MC, Farshid G, Fitzpatrick N, Francis GD, McCloud PI,
Raymond W, et al: Gastric HER2 Testing Study (GaTHER): An
evaluation of gastric/gastroesophageal junction cancer testing
accuracy in Australia. Am J Surg Pathol. 36:577–582. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tang D, Liu CY, Shen D, Fan S, Su X, Ye P,
Gavine PR and Yin X: Assessment and prognostic analysis of EGFR,
HER2, and HER3 protein expression in surgically resected gastric
adenocarcinomas. Onco Targets Ther. 8:7–14. 2014.PubMed/NCBI
|
|
24
|
Cappuzzo F, Hirsch FR, Rossi E, Bartolini
S, Ceresoli GL, Bemis L, Haney J, Witta S, Danenberg K, Domenichini
I, et al: Epidermal growth factor receptor gene and protein and
gefitinib sensitivity in non-small-cell lung cancer. J Natl Cancer
Inst. 97:643–655. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tsao MS, Sakurada A, Cutz JC, Zhu CQ,
Kamel-Reid S, Squire J, Lorimer I, Zhang T, Liu N, Daneshmand M, et
al: Erlotinib in lung cancer - molecular and clinical predictors of
outcome. N Engl J Med. 353:133–144. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Peled N, Yoshida K, Wynes MW and Hirsch
FR: Predictive and prognostic markers for epidermal growth factor
receptor inhibitor therapy in non-small cell lung cancer. Ther Adv
Med Oncol. 1:137–144. 2009. View Article : Google Scholar : PubMed/NCBI
|