Introduction
Malignant effusion cell culture of patients with
different cancers have been used to assess the response to
chemotherapeutic agents, to provide in vitro
characterization of neoplastic cell lines, to investigate tumoral
heterogeneity and to understand the role of epithelial-mesenchymal
transition (EMT) and cancer stem cells (CSCs) in metastasis and in
high therapeutic failure rates (1–8).
Cytology of effusions (pleural, peritoneal and
pericardial fluid) and peritoneal lavage is a routine procedure in
pathological anatomy laboratories for the diagnosis, staging,
determining the primary site of metastatic cancer and follow-up of
patients with high incidence and mortality cancers such as
carcinoma of breast, lung, ovary and stomach (9–11). The
cell types usually identified by cytology of benign effusion are
mesothelial cells, macrophages and leukocytes. Besides these cells,
neoplastic cells may also be observed in malignant effusions
(9–11). Effusions with high cellularity and
characteristic morphological aspects enable to define the nature of
the malignant cells (9–11). However, it should be noted that
there are instances when the distinction between metastatic cancer
cells and reactive or neoplastic mesothelium can be challenging
(12–17). In these cases, immunocytochemistry
may elucidate the origin of the atypical cells (12–17).
Immunocytochemical panels typically include markers of mesothelial
and of malignant epithelial cells but these markers do not exhibit
high specificity and sensitivity and a broad panel of
immunocytochemical markers is used for differentiating these cells
(12–17).
Identification of cell types in culture of effusions
is even more difficult than in cytology because the growing cells
frequently undergo morphological and functional changes that in
turn may also result in different expression pattern of
immunocytochemical markers. In addition, there is the possibility
of growth of various cell types (mesothelial, macrophages and
epithelial malignant) simultaneously and failure to correctly
identify them can interfere with subsequent research results.
Therefore, in view of the potential use of culture of effusions and
the need for the correct identification of cell types grown in
culture, the aim of the present study was to identify by
immunocytochemistry cells in culture from malignant and
non-malignant effusions.
Materials and methods
Samples
Samples of effusions (pleural, peritoneal and
pericardial) and peritoneal lavage were obtained at the Department
of Pathology of Brasilia University Hospital, Brazil, between 2012
to 2015. The study protocol was approved by the Human Ethics Review
Committee of the Brasilia University.
Cytological and immunocytochemical
analyses
All samples were centrifuged and the sediment was
fixed in alcohol and stained by the Papanicolaou method. Cytology
was considered positive for malignancy, negative, suspicious or
unsatisfactory. Immunocytochemistry was performed in cytological
slides to evaluate malignancy: the samples were divided into
malignant (with positivity malignancy markers) and non-malignant
(lack of positivity for malignancy markers).
Cell culture
The initial volume of the samples ranged from 10 to
50 ml. The unfixed samples were centrifuged, the supernatant was
removed, and the cells were resuspended in 500 µl of medium and
then seeded onto a 4-well chamber slide, containing equal amount of
complete Dulbeccos modified Eagles medium (DMEM) supplemented with
20% bovine fetal serum (FBS) and antibiotics and antifungals 2%
(penicillin, streptomycin and amphotericin B, 200 U/ml, 200 mg/ml
and 2.5 µg/ml). Samples were seeded in duplicate and culture slides
were incubated at 37°C and 5% CO2 for a total of 7 days,
with medium change on the third day. On days 2 and 7, samples were
washed and fixed in alcohol. Immunocytochemistry was also performed
in culture slides for identification of cell types in growth.
Immunocytochemistry
For antigen retrieval, the slides were incubated for
45 min in a waterbath at 95–99°C with citrate buffer pH 6.0. For
blockade of endogenous tissue peroxide, the slides were immersed in
3% H2O2 solution at room temperature for 30
min. After washing with phosphate buffered saline (PBS), the slides
were incubated with primary antibody overnight at 4°C. The primary
antibodies used are shown in Table
I. After washing with PBS, the slides were incubated with a
secondary antibody for 30 min at room temperature and subsequently
with the streptavidin-biotin-peroxidase complex (LSAB+; DAKO A/S,
Glostrup, Denmark; K-690) for 30 min at room temperature. All
reactions were developed using a diaminobenzidine chromogen
solution (Dab substrate chromogen system-K3468; Dako). The
counterstaining was performed with Harris hematoxylin. The slides
were dehydrated, cleared and mounted. Positive and negative control
were used for each primary antibody according to the manufacturers
recommendations. Positive staining was defined as a strong brown
stain in more than 1% of cells in the cytoplasm (Pan-cytokeratin,
CD68, vimentin and melan), membrane (HBME and LCA), nucleus (WT1),
cytoplasm and membrane (MOC-31 and Claudin 4), cytoplasm and
nucleus (calretinin and IMP3). The expression of at least two of
the following markers was considered for identification of the
mesothelial cell: calretinin, HBME and WT1. The expression of at
least two of the following markers was considered for
identification of the malignant epithelial cells: MOC, IMP3 and
Claudin 4. The identification of macrophages was performed using
the marker CD68 and lack of expression of epithelial markers. LCA
and HMB-45 were used for identification of lymphoma and melanoma,
respectively.
 | Table I.Antibody used in
immunocytochemistry. |
Table I.
Antibody used in
immunocytochemistry.
| Antibody | Source | Clone | Dilution |
|---|
| Anti-epithelial
related antigen | Dako | MOC-31 | 1:100 |
| Anti-IMP3 | Dako | 69.1 | 1:500 |
| Anti-Claudin 4 | Abcam | AB15104 | 1:200 |
|
Anti-Calretinin | Dako | DAK-Calret 1 | 1:50 |
| Anti-human
mesothelial cell | Cell Marque | HBME-1 | 1:50 |
| Anti-Wilms tumor 1
(WT-1) | Dako | 6F-H2 | 1:300 |
| Anti-CD68 | BioCare
Medical | KP1 | 1:100 |
| Anti-vimentin | Dako | Clone V9 | 1:100 |
|
Anti-pan-cytokeratin (PANCK) | Dako | AE1/AE3 | 1:50 |
| Anti-LCA | Dako | 4KB5 | 1:50 |
| Anti-Melan-A | Dako | A103 | 1:25 |
Results
Samples
A total of 143 samples (pleural effusion n=76;
peritoneal effusion n=37; pericardial effusion n=4; and peritoneal
lavagen n=26) were analyzed. In 32.86% (47/143) of samples,
patients presented with current or previous histological diagnostic
of cancer (carcinoma n=45; lymphoma n=1; and melanoma n=1).
Cytology, culture and
immunocytochemistry
Cell growth was observed in 34.96% (50/143) of all
samples (pleural effusion n=31 and peritoneal effusion n=19). After
two days in culture, adherent cells formed a flat monolayer, and
appeared homogenous and polygonal in shape on the seventh day.
Because the cells were similar morphologically, immunocytochemistry
was performed to identify the different cellular types in
culture.
The identification of cell types in culture by
immunocytochemistry was conclusive only in 90% (45/50) of samples
that showed growth. In the remaining 5 samples, low cellularity
prevented the use of all necessary markers to complete
immunocytochemistry analysis. In 71.11% (32/45) of samples in which
immunocytochemistry was conclusive, patients presented with current
or previous histological diagnostic of cancer but only in 37.77%
(17/45), the samples were malignant (with positivity for carcinoma
markers). In 62.22% (28/45), the samples were non-malignant (lack
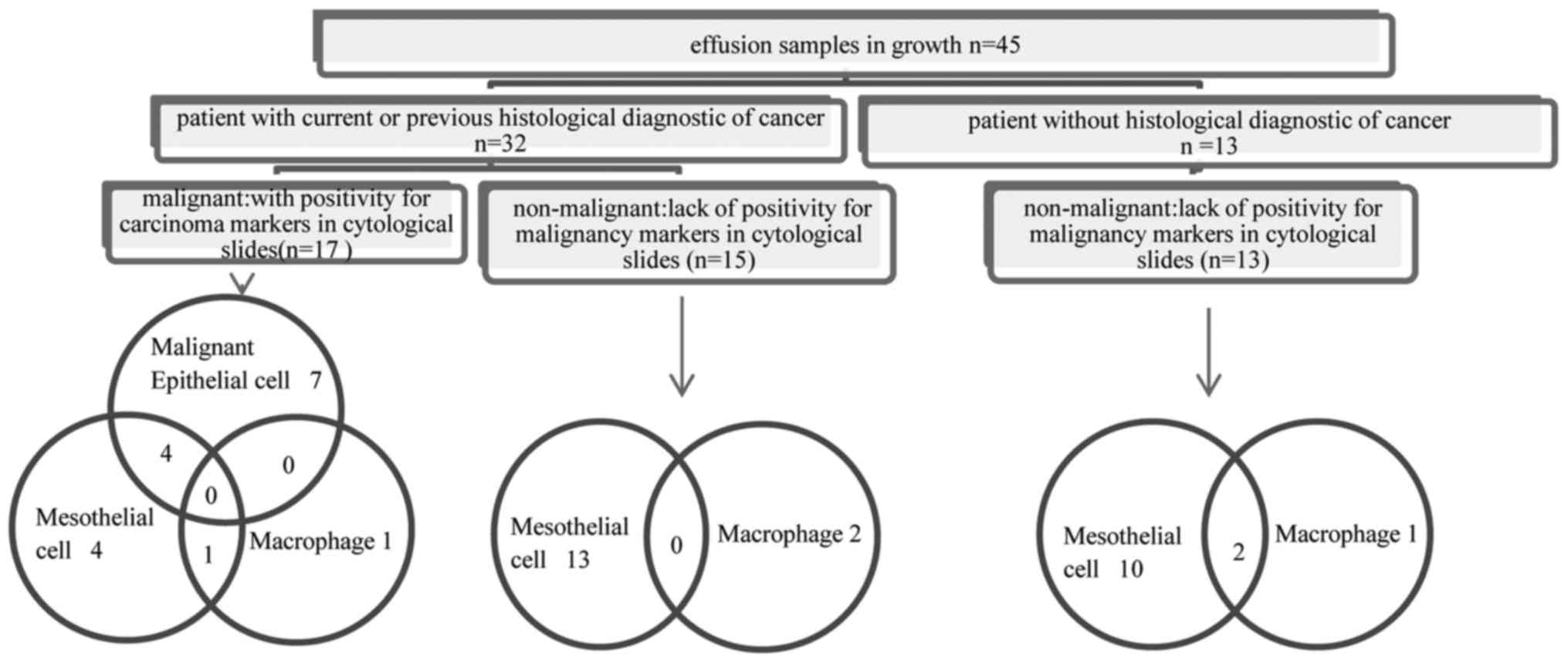
of positivity for malignancy markers) (Fig. 1).
The types of cells in culture identified by
immunocytochemistry were mesothelial cell, malignant epithelial
cell and macrophages.
In non-malignant samples, growth of mesothelial
cells, macrophages and of both cell types were identified in 82.14%
(23/28), 10.71% (3/28) and 7.14% (2/28), respectively. In malignant
samples, growth of malignant epithelial cells and of both malignant
epithelial and mesothelial cells was identified in 41.17% (7/17)
and 23.52% (4/17), respectively. In the remaining 35.29% (6/17) of
malignant samples, the only cells in growth were mesothelial and/or
macrophages instead of malignant epithelial cells (Fig. 1).
Presence of mesothelial cells in culture was
identified by positivity for at least two of the markers
calretinin, WT1 or HBME. The pattern of expression of the markers
in adherent mesothelial cells in culture was similar to that of
adjacent non-adherent mesothelial cells and to that of cells in
cytology; cytoplasm and nucleus for calretinin, membrane for HBME
and nucleus for WT1 (Figs. 2 and
3). Expression of non-specific
markers was also observed in the adherent mesothelial cells:
pan-cytokeratin and vimentin. Malignant epithelial cells in culture
were identified by positivity for at least two of the following
markers: MOC-31, Claudin 4 and IMP3. The pattern of expression of
the markers in adherent malignant epithelial cells in culture was
similar to that of adjacent non-adherent malignant epithelial cells
and to that of cells in cytology: cytoplasm and membrane for MOC-31
and Claudin 4, cytoplasm and nucleus for IMP3 (Fig. 3). Macrophages were identified by
positivity for CD68 and lack of expression of epithelial markers.
The staining pattern (cytoplasm) in adherent cells was similar to
that of non-adherent adjacent cells and to that of cells in
cytology.
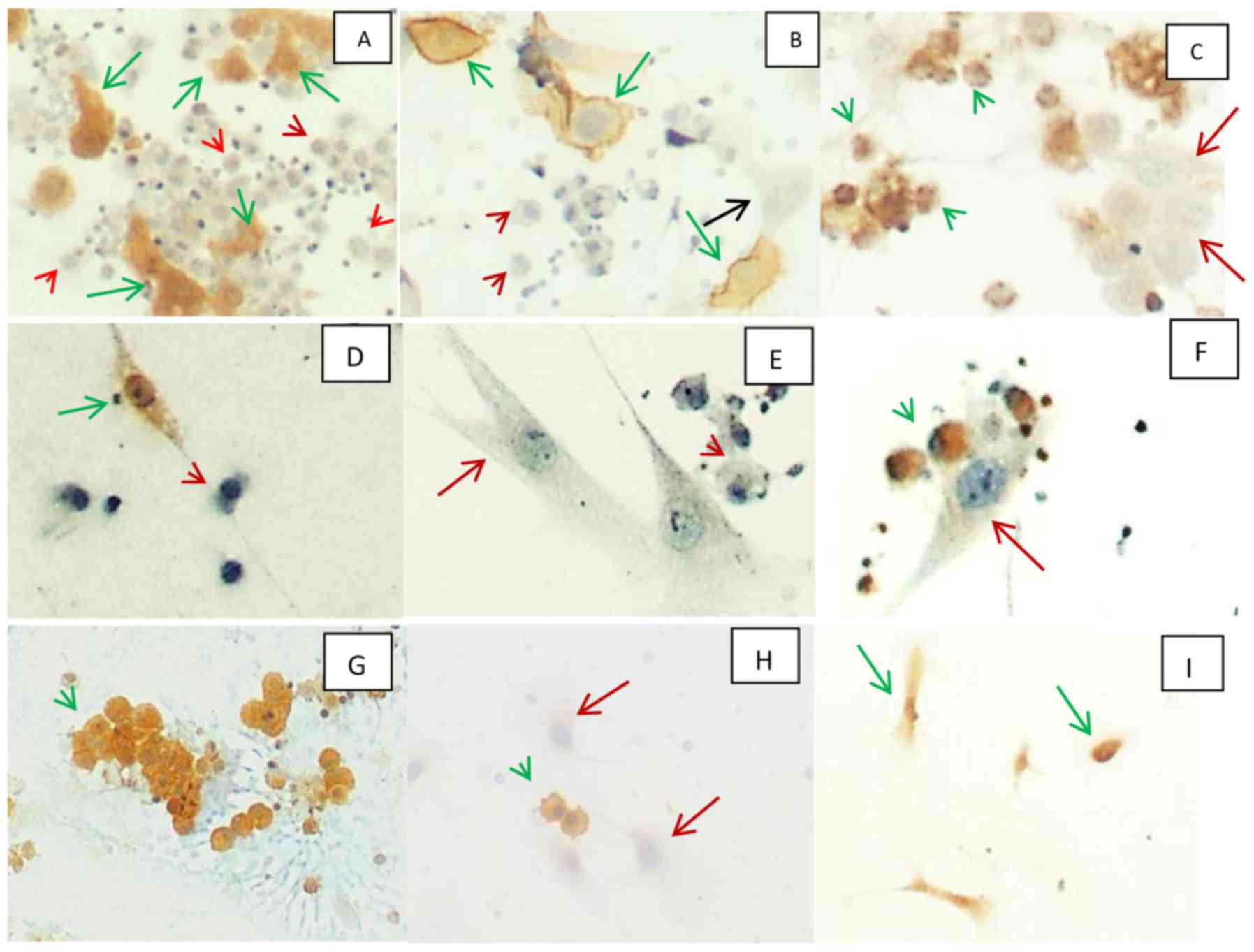 | Figure 2.(A-C), culture of mesothelial cells
from pleural effusion of patient with history of pneumonia. (A)
Immunocytochemistry for calretinin (magnification, ×100). Positive
in cytoplasm and nucleus of adherent mesothelial cell (green
arrow), negative in non-adherent macrophages (red arrowhead). (B)
Immunocytochemistry for HBME (magnification, ×100). Positive (green
arrow) and negative (black arrow) in membrane of adherent
mesothelial cells and negative in non-adherent macrophages (red
arrowhead). (C) Immunocytochemistry for CD68 (magnification, ×100).
Positive in cytoplasm of non-adherent macrophages (green
arrowhead), negative in adherent mesothelial cells (red arrow).
(D-F) Culture of mesothelial cells from pleural effusion of patient
with history of breast carcinoma and lack of positivity for
carcinoma markers in cytological slides. (D) Immunocytochemistry
for calretinin (magnification, ×200). Positive in cytoplasm and
nucleus of adherent mesothelial cells (green arrow), negative in
non-adherent macrophages (red arrowhead). (E) Immunocytochemistry
for MOC-31 (magnification, ×200). Negative in adherent mesothelial
cells (red arrow) and negative in non-adherent macrophages (red
arrowhead). (F) Immunocytochemistry for CD68 (magnification, ×200).
Positive in cytoplasm of non-adherent macrophages (green
arrowhead), negative in adherent mesothelial cells (red arrowhead).
(G-I) culture of mesothelial cells from pleural effusion of patient
with history of breast carcinoma and positivity for carcinoma
markers in cytological slides. (G and H) Immunocytochemistry for
MOC-31 (magnification, ×100). Positive in cytoplasm and membrane of
non-adherent epithelial malignant cells (green arrowhead) and
negative in adherent mesothelial cell (red arrow). (I)
immunocytochemistry for calretinin (magnification, ×100). Positive
in cytoplasm and nucleus of adherent mesothelial cells (green
arrow). |
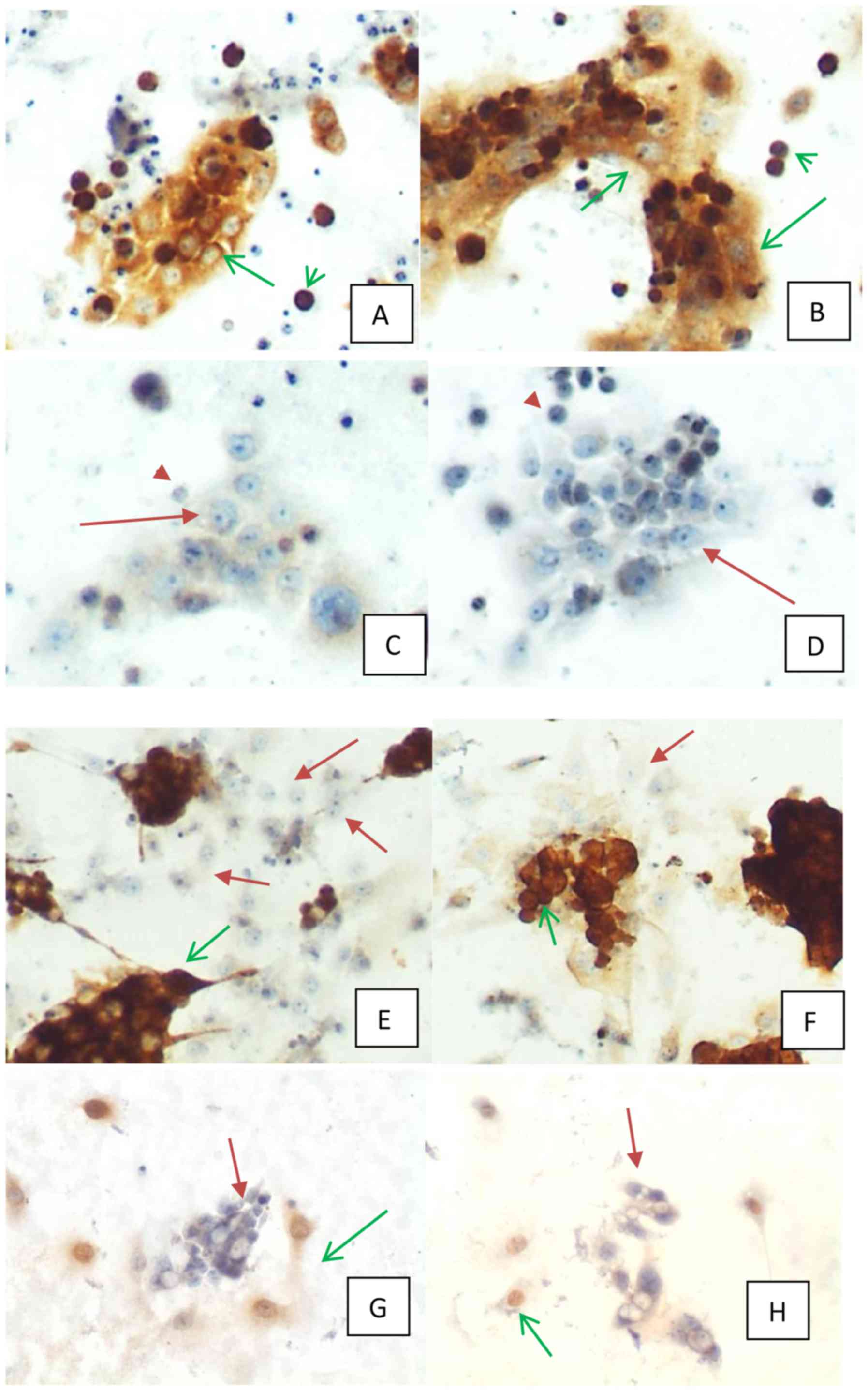 | Figure 3.(A-D) culture of malignant epithelial
cells from pleural effusion of a patient with history of esophagic
cancer and positivity for carcinoma markers in cytological slides.
(A) Immunocytochemistry for MOC-31 (magnification, ×100). Positive
in membrane and cytoplasm of adherent (green arrow) and
non-adherent (green arrowhead) malignant epithelial cells. (B)
Immunocytochemistry for IMP3 (magnification, ×100). Positive in
cytoplasm of adherent (green arrow) and non-adherent malignant
epithelial cells (green arrowhead). (C) Immunocytochemistry for
calretinin (magnification, ×100). Negative in adherent (red arrow)
and non-adherent (red arrowhead) malignant epithelial cells. (D)
Immunocytochemistry for HBME (magnification, ×100). Negative in
adherent (red arrow) and non-adherent (red arrowhead) malignant
epithelial cells. (E-H) Culture of both malignant epithelial and
mesothelial cells from pleural effusion of patient with history of
gastric carcinoma and positivity for carcinoma markers in
cytological slides. (E) Immunocytochemistry for IMP3
(magnification, ×100). Positive in cytoplasm of adherent malignant
epithelial cells (green arrow), negative in adherent mesothelial
cells (red arrow). (F) Immunocytochemistry for MOC-31
(magnification, ×100). Positive in cytoplasm and membrane of
adherent malignant epithelial cells (green arrow), negative in
adherent mesothelial cells (red arrow). (G) Immunocytochemistry for
calretinin (magnification, ×100). Positive in cytoplasm and nucleus
of adherent mesothelial cells (green arrow), negative in adherent
malignant epithelial cells (red arrow). (H) Immunocytochemistry for
WT1 (magnification, ×100). Positive in nucleus of adherent
mesothelial cells (green arrow), negative in adherent malignant
epithelial cells (red arrow). |
Discussion
The aim of the present study was to identify, by a
panel of immunocytochemical markers, cell types in primary culture
from effusions. Although a large number of samples have been
subjected to culture in this study, growth was obtained in only 34%
of them. The low cellularity, mainly in peritoneal lavage, may
explain, at least partially, the low number of samples with growth.
To minimize the loss of cells, culture was performed directly on
chamber slides and the interval between sample collection and
culture of the samples was lesser than 48 h, a period when the
samples were kept at 4°C until culture. After culture and
cytological analyses, the remainder of the sample was kept at 4°C
for up to 2 weeks. Notably, in some of these samples, the presence
of viable and capable of growth mesothelial cells was noted after
such long-period storage (results not shown). Lack of adherence of
the cells was another limiting factor to obtain a higher number of
cultured samples. But cellularity and time of culture does not seem
to be a determinant of adherence and growth of malignant epithelial
cells in culture because in some samples in which many malignant
epithelial cells (isolated or grouped) were present, no cell growth
or only growth of mesothelial cells was identified, despite the
high cellularity of malignant epithelial cells and even after 7
days of culture. This is in accordance with recent results which
showed that cancer stem cells or tumor-initiating cells tend to
grow in three dimensional cultures (18).
Morphologically, adherent mesothelial and malignant
epithelial cells were indistinguishable in appearance and size,
whereas macrophages were proportionally smaller as compared with
those cells.
Using immunocytochemistry, it was possible to
identify cell types in 90% (45/50) of the samples in culture. The
expression pattern (nucleus, membrane and/or cytoplasm) of the
markers remained at 2 and 7 days of growth, but one limitation of
the present study was lack of analysis of markers expression for
longer periods of time.
The predominant cell type of effusions in culture
was mesothelial cells and they were present mainly in inflammatory
benign or malignant effusions. Mesothelial cells are specialized
epithelial cells that line the serous cavities (pleural,
pericardial and peritoneal) (19).
Under normal homeostasis, mesothelial cells exhibit limited cell
proliferation, with only 0.16–0.5% of cells within the mesothelium
undergoing mitosis at any one time (19,20).
The rate of mitosis increases to 30–60% following injury to the
mesothelium, and this is attributed in part to increased levels of
growth factors and cytokines (20–22).
Culture and isolation of mesothelial cells has been used in studies
on the role of mesothelial cells in the progression of cancer in
malignant effusion (23–25).
A major contribution of this study was to
demonstrate that in 65.62% of effusions of patient with current or
previous histological diagnosis of cancer and in 35.29% of
malignant effusion, the only cells found in culture were
mesothelial and/or macrophages instead of malignant epithelial
cells. Besides this, in 23.52% of malignant effusion, mesothelial
and malignant epithelial cells were growing simultaneously in
culture. When the objective is to obtain primary culture of
malignant epithelial cells from a malignant effusion, the ideal
sample should be without the presence of mesothelial cells and
macrophages which are cells generally present when there is
associated inflammation.
As the mesothelial cells were morphologically
indistinguishable from malignant epithelial cells and no single
marker is characterized by 100% specificity and sensitivity for
distinguishing these cells, a broad panel of markers should be used
for the growth of mesothelial cells and macrophages not to be
confused with growth of neoplastic cells in malignant effusions
culture.
To the best of our knowledge, we have for the first
time used a broad painel of immunocytochemical markers for
identification of cells in primary culture from malignant and
non-malignant effusions culture. The markers used here to identify
mesothelial cells in growth were calretinin, HBME and WT1, but
alternative markers could have be used such as thrombomodulin
(14). Calretinin is a high
sensitivity and specificity marker and widely used (14,16,26,27).
HBME was used, in the present study, for identification of
mesothelial cells but it has a low sensitivity when compared to
calretinin. Moreover, HBME can be expressed in certain types of
carcinoma, such as lung, ovarian, breast, colon and stomach
carcinoma (14,28). WT1 expression can be detected in
benign and malignant mesothelial cells. However, WT1 is a marker
that is expressed in most of primary ovarian carcinomas and has
been used to distinguish carcinoma of ovarian origin from carcinoma
of other primary sites. Thus, although the expression of HBME and
WT-1 could potentially cause some difficulty in the correct
interpretation of effusion specimens, concomitant usage of other
mesothelial cells and malignant epithelial cell markers could prove
helpful (15,29).
The markers used to identify malignant epithelial
cells were MOC-31, Claudin 4 and IMP3, but several other
adenocarcinomas markers have been used to distinguish malignant
epithelial cells from mesothelial cells such as B72.3, CEA and
Ber-EP4 (30–34). Besides, some primary site markers of
adenocarcinoma can also contribute to distinction between
mesothelial and epithelial malignant cells such as estrogen
receptor, progesterone receptor, GATA3, mammoglobin, GCPD15 for
breast; Napsin-A and TTF-1 for lung; CDX-2, villin, SATB2 for
gastrointestinal tract; PSA for prostate; PAX8, HBME and estrogen
receptor for ovary; CD10 and PAX 8 for kidney; hepatocyte-specific
antigen for liver. As no single marker presents 100% specificity
and sensitivity, positivity for a combination of markers have been
used for identification of primary site markers of adenocarcinomas
(35–45). For the diagnosis of squamous cell
carcinoma, the negativity for MOC-31, negativity for mesothelial
cell markers, and positivity for P63 may be useful (46).
In conclusion, the present study showed that in
culture of malignant effusions, mesothelial cells may be
simultaneously identified with malignant epithelial cell.
Mesothelial cells and macrophages may be the only cells identified
in malignant effusion culture. Therefore, a broad panel of cell
markers should be used for identification of cells in studies of
effusion primary culture. The ideal malignant effusion sample to
obtain culture of neoplastic cells should be that without the
presence of mesothelial cells and macrophages. These data will
prevent future errors in studies using cells isolated from effusion
cultures.
Acknowledgements
The present study was funded by the Fundação de
Apoio a Pesquisa do Distrito Federal (FAP-DF), Fundação de Ensino e
Pesquisa em Ciências da Saúde (FEPECS), FAHUB, CAPES and CNPQ.
References
|
1
|
Ruiz C, Kustermann S, Pietilae E, Vlajnic
T, Baschiera B, Arabi L, Lorber T, Oeggerli M, Savic S, Obermann E,
et al: Culture and drug profiling of patient derived malignant
pleural effusions for personalized cancer medicine. PLoS One.
11:e01608072016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Roscilli G, De Vitis C, Ferrara FF, Noto
A, Cherubini E, Ricci A, Mariotta S, Giarnieri E, Giovagnoli MR,
Torrisi MR, et al: Human lung adenocarcinoma cell cultures derived
from malignant pleural effusions as model system to predict
patients chemosensitivity. J Transl Med. 14:612016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bezdieniezhnykh N, Lykhova A, Semesiuk N,
Okhrimenko R and Kudryavets Y: Establishment and characterization
of new breast and ovarian cancer cell lines as a model for studying
cellular plasticity in vitro. Exp Oncol. 38:94–100. 2016.PubMed/NCBI
|
|
4
|
Ku JL, Park SC, Kim KH, Jeon YK, Kim SH,
Shin YK, Noh DY, Im SA, Bang YJ, Han W, et al: Establishment and
characterization of seven human breast cancer cell lines including
two triple-negative cell lines. Int J Oncol. 43:2073–2081. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Basak SK, Veena MS, Oh S, Huang G,
Srivatsan E, Huang M, Sharma S and Batra RK: The malignant pleural
effusion as a model to investigate intratumoral heterogeneity in
lung cancer. PLoS One. 4:e58842009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ricci A, De Vitis C, Noto A, Fattore L,
Mariotta S, Cherubini E, Roscilli G, Liguori G, Scognamiglio G,
Rocco G, et al: TrkB is responsible for EMT transition in malignant
pleural effusions derived cultures from adenocarcinoma of the lung.
Cell Cycle. 12:1696–1703. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen SF, Lin YS, Jao SW, Chang YC, Liu CL,
Lin YJ and Nieh S: Pulmonary adenocarcinoma in malignant pleural
effusion enriches cancer stem cell properties during metastatic
cascade. PLoS One. 8:e546592013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yin T, Wang G, He S, Shen G, Su C, Zhang
Y, Wei X, Ye T, Li L, Yang S, et al: Malignant pleural effusion and
ascites induce epithelial-mesenchymal transition and cancer
stem-like cell properties via the vascular endothelial growth
factor (VEGF)/phosphatidylinositol 3-kinase (PI3K)/Akt/mechanistic
target of eapamycin (mTOR) pathway. J Biol Chem. 291:26750–26761.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Francis IM, Alath P, George SS, Jaragh M,
Al Jassar A and Kapila K: Metastatic breast carcinoma in pleural
fluid: Correlation of receptor and HER2 status with the primary
carcinoma - a pilot study. Diagn Cytopathol. 44:980–986. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kalogeraki A, Lazopoulos G, Papadakis GZ,
Tamiolakis D, Karvela-Kalogeraki I, Karvelas-Kalogerakis M,
Segredakis J, Papadakis M, Moustou E, Datseri G, et al: Cytology of
pericardial effusion due to malignancy. Rom J Intern Med.
54:179–183. 2016.PubMed/NCBI
|
|
11
|
Kalogeraki A, Tamiolakis D, Datseri G,
Lazopoulos G, Karvelas-Kalogerakis M, Segredakis J and Zoi I:
Pleural effusion cytology due to malignancy. A combined
cytomorphological-immunocytochemical study of 500 cases. Rev Port
Pneumol 2006. 22:290–291. 2016.PubMed/NCBI
|
|
12
|
Antonangelo L, Sales RK, Corá AP, Acencio
MM, Teixeira LR and Vargas FS: Pleural fluid tumour markers in
malignant pleural effusion with inconclusive cytologic results.
Curr Oncol. 22:e336–e341. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Westfall DE, Fan X and Marchevsky AM:
Evidence-based guidelines to optimize the selection of antibody
panels in cytopathology: Pleural effusions with malignant
epithelioid cells. Diagn Cytopathol. 38:9–14. 2010.PubMed/NCBI
|
|
14
|
Su XY, Li GD, Liu WP, Xie B and Jiang YH:
Cytological differential diagnosis among adenocarcinoma, epithelial
mesothelioma, and reactive mesothelial cells in serous effusions by
immunocytochemistry. Diagn Cytopathol. 39:900–908. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lyons-Boudreaux V, Mody DR, Zhai J and
Coffey D: Cytologic malignancy versus benignancy: How useful are
the ‘newer’ markers in body fluid cytology? Arch Pathol Lab Med.
132:23–28. 2008.PubMed/NCBI
|
|
16
|
Saleh HA, El-Fakharany M, Makki H, Kadhim
A and Masood S: Differentiating reactive mesothelial cells from
metastatic adenocarcinoma in serous effusions: The utility of
immunocytochemical panel in the differential diagnosis. Diagn
Cytopathol. 37:324–332. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim JH, Kim GE, Choi YD, Lee JS, Lee JH,
Nam JH and Choi C: Immunocytochemical panel for distinguishing
between adenocarcinomas and reactive mesothelial cells in effusion
cell blocks. Diagn Cytopathol. 37:258–261. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Qureshi-Baig K, Ullmann P, Rodriguez F,
Frasquilho S, Nazarov PV, Haan S and Letellier E: What do we learn
from spheroid culture systems? Insights from tumorspheres derived
from primary colon cancer tissue. PLoS One. 11:e01460522016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yung S and Chan TM: Mesothelial cells.
Perit Dial Int. 27 Suppl 2:S110–S115. 2007.PubMed/NCBI
|
|
20
|
Yung S, Li FK and Chan TM: Peritoneal
mesothelial cell culture and biology. Perit Dial Int. 26:162–173.
2006.PubMed/NCBI
|
|
21
|
Mutsaers SE, Whitaker D and Papadimitriou
JM: Stimulation of mesothelial cell proliferation by exudate
macrophages enhances serosal wound healing in a murine model. Am J
Pathol. 160:681–692. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou Q and Yu X: Isolation and propagation
of rat peritoneal mesothelial cells. Methods Mol Biol. 1397:25–34.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ranieri D, Raffa S, Parente A, Del Monte
Rossi S, Ziparo V and Torrisi MR: High adhesion of tumor cells to
mesothelial monolayers derived from peritoneal wash of disseminated
gastrointestinal cancers. PLoS One. 8:e576592013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tsukada T, Fushida S, Harada S, Yagi Y,
Kinoshita J, Oyama K, Tajima H, Fujita H, Ninomiya I, Fujimura T,
et al: The role of human peritoneal mesothelial cells in the
fibrosis and progression of gastric cancer. Int J Oncol.
41:476–482. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cabourne EJ, Roberts G, Goldin R, Ryder T,
Mobberly M and Ziprin P: Investigation of tumor-peritoneal
interactions in the pathogenesis of peritoneal metastases using a
novel ex vivo peritoneal model. J Surg Res. 164:e265–e272. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lv M, Leng JH, Hao YY, Sun Y, Cha N and Wu
GP: Expression and significance of MOC-31 and calretinin in pleural
fluid of patients with lung cancer. Diagn Cytopathol. 43:527–531.
2015. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hyun TS, Barnes M and Tabatabai ZL: The
diagnostic utility of D2-40, calretinin, CK5/6, desmin and MOC-31
in the differentiation of mesothelioma from adenocarcinoma in
pleural effusion cytology. Acta Cytol. 56:527–532. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fetsch PA, Simsir A and Abati A:
Comparison of antibodies to HBME-1 and calretinin for the detection
of mesothelial cells in effusion cytology. Diagn Cytopathol.
25:158–161. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhao L, Guo M, Sneige N and Gong Y: Value
of PAX8 and WT1 immunostaining in confirming the ovarian origin of
metastatic carcinoma in serous effusion specimens. Am J Clin
Pathol. 137:304–309. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Oda T, Ogata S, Kawaguchi S, Minabe S,
Dokyu M, Takahashi H, Kumazawa F, Shimazaki H, Takano M, Hase K, et
al: Immunocytochemical utility of claudin-4 versus those of Ber-EP4
and MOC-31 in effusion cytology. Diagn Cytopathol. 44:499–504.
2016. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kundu UR and Krishnamurthy S: Use of the
monoclonal antibody MOC-31 as an immunomarker for detecting
metastatic adenocarcinoma in effusion cytology. Cancer Cytopathol.
119:272–278. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kim NI, Kim GE and Lee JS: Diagnostic
usefulness of claudin-3 and claudin-4 for immunocytochemical
differentiation between metastatic adenocarcinoma cells and
reactive mesothelial cells in effusion cell blocks. Acta Cytol.
60:232–239. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ikeda K, Tate G, Suzuki T, Kitamura T and
Mitsuya T: IMP3/L523S, a novel immunocytochemical marker that
distinguishes benign and malignant cells: The expression profiles
of IMP3/L523S in effusion cytology. Hum Pathol. 41:745–750. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hanley KZ, Facik MS, Bourne PA, Yang Q,
Spaulding BO, Bonfiglio TA and Xu H: Utility of anti-L523S antibody
in the diagnosis of benign and malignant serous effusions. Cancer.
114:49–56. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jing X, Li QK, Bedrossian U and Michael
CW: Morphologic and immunocytochemical performances of effusion
cell blocks prepared using 3 different methods. Am J Clin Pathol.
139:177–182. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ebata T, Okuma Y, Nakahara Y, Yomota M,
Takagi Y, Hosomi Y, Asami E, Omuro Y, Hishima T, Okamura T, et al:
Retrospective analysis of unknown primary cancers with malignant
pleural effusion at initial diagnosis. Thorac Cancer. 7:39–43.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lew M, Pang JC, Jing X, Fields KL and Roh
MH: Young investigator challenge: The utility of GATA3
immunohistochemistry in the evaluation of metastatic breast
carcinomas in malignant effusions. Cancer Cytopathol. 123:576–581.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Porcel JM, Palma R, Bielsa S, Esquerda A,
Gatius S, Matias-Guiu X and Salud A: TTF-1 and napsin A on cell
blocks and supernatants of pleural fluids for labeling malignant
effusions. Respirology. 20:831–833. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Moh M, Krings G, Ates D, Aysal A, Kim GE
and Rabban JT: SATB2 expression distinguishes ovarian metastases of
colorectal and appendiceal origin from primary ovarian Tumors of
mucinous or endometrioid type. Am J Surg Pathol. 40:419–432. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Seipel AH, Samaratunga H, Delahunt B,
Wiklund P, Clements M and Egevad L: Immunohistochemistry of ductal
adenocarcinoma of the prostate and adenocarcinomas of non-prostatic
origin: A comparative study. APMIS. 124:263–270. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
McKnight R, Cohen C and Siddiqui MT:
Utility of paired box gene 8 (PAX8) expression in fluid and
fine-needle aspiration cytology: an immunohistochemical study of
metastatic ovarian serous carcinoma. Cancer Cytopathol. 25:298–302.
2010. View Article : Google Scholar
|
|
42
|
Waters L, Crumley S, Truong L, Mody D and
Coffey D: PAX2 and PAX8: Useful markers for metastatic effusions.
Acta Cytol. 58:60–66. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Barr ML, Jilaveanu LB, Camp RL, Adeniran
AJ, Kluger HM and Shuch B: PAX-8 expression in renal tumours and
distant sites: A useful marker of primary and metastatic renal cell
carcinoma? J Clin Pathol. 68:12–17. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Chute DJ, Kong CS and Stelow EB:
Immunohistochemistry for the detection of renal cell carcinoma in
effusion cytology. Diagn Cytopathol. 39:118–123. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Karabork A, Kaygusuz G and Ekinci C: The
best immunohisto-chemical panel for differentiating hepatocellular
carcinoma from metastatic adenocarcinoma. Pathol Res Pract.
206:572–577. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Pu RT, Pang Y and Michael CW: Utility of
WT-1, p63, MOC31, mesothelin, and cytokeratin (K903 and CK5/6)
immunostains in differentiating adenocarcinoma, squamous cell
carcinoma, and malignant mesothelioma in effusions. Diagn
Cytopathol. 36:20–25. 2008. View
Article : Google Scholar : PubMed/NCBI
|