Introduction
Metastasis is a major obstacle in cancer therapy;
despite constant standardization of chemotherapy or surgical
procedures (1,2), some patients die due to distant
metastasis of tumor cells. The purpose of this experiment was to
explore a new method to inhibit the invasion of tumor cells more
efficiently.
Tumor metastasis is a multistage process during
which malignant cells spread from the primary tumor to distant
organs (3). With the participation
of multiple proteins and cytokines, tumor cells infiltrate the
circulatory system and avoid immune system attacks, eventually
reaching target organs for implantation and growth (4). The SIBLING (small integrin-binding
ligand N-linked glycoprotein) family of proteins regulates
malignant tumor behaviors such as malignant cell proliferation,
detachment, invasion, and metastasis by combining with integrin
protein (5). SIBLINGs are
overexpressed in many cancers (2,6–8). The
level of SIBLING protein in serum is often used to predict the
prognosis of many cancer patients, especially in patients with
prostate and breast cancer (9–11). Two
proteins in this family, namely, OPN (osteopontin) and BSP (bone
sialoprotein), have garnered the most attention, and their reported
levels of expression are closely correlated with tumor
aggressiveness. For invasion, BSP and OPN can activate specific
metalloproteinases (BSP activates MMP-2, OPN activates MMP-3) to
enhance the ability of cancer cells to hydrolyze the extracellular
matrix (ECM) (6,12). The binding of BSP and integrins
contributes to metastasis formation of breast cancer cells,
particularly bone metastasis (13).
Moreover, BSP-transfected breast cancer cells showed increased
primary tumor growth following injection into the mammary fat pad
of nude mice (14), and OPN
stimulated human prostate cancer (PCa) cell proliferation when
transferred to a mouse xenograft model system (15). These effects mainly occurred through
BSP and OPN activation of the epidermal growth factor receptor
(EGFR) and integrin-mediated intracellular Ca2+
signaling (16). Therefore, it is
important to identify a method that can inhibit the expression of
BSP and OPN in tumor cells to prevent tumor cell metastasis.
Studies have suggested that interleukin-8 (IL-8) and
its cognate receptors, namely, C-X-C chemokine receptor-1 (CXCR1)
and CX-C chemokine receptor-2 (CXCR2), mediate the initiation and
development of various types of cancers, including breast cancer,
PCa, lung cancer, colorectal carcinoma and melanoma (17–21).
IL-8 also integrates with multiple intracellular signaling pathways
to produce coordinated effects. In terms of invasion, IL-8 promotes
prostate and breast cancer expression of matrix metalloproteinases
(MMPs) (especially MMP-2 and MMP-9) to enhance their cell
aggressiveness (22,23). The ectopic expression of IL-8
stimulated by IL-1β and TNF-α can enhance the metastatic potential
of breast cancer since a high level of IL-8 can promote
angiogenesis and attract neutrophils to release enzymes involved in
tissue remodeling and tumor establishment (24). Increased IL-8 secretion by PCa cells
is similarly associated with the malignant biological behaviors of
cancer cells. IL-8/CXCR2 promotes castration-resistant growth and
proliferation of AIPC cells (androgen-independent PCa cells) by
activating cyclin D1 expression in a PI3K/Akt/mTOR and MAPK
pathway-dependent manner (25,26).
SB22502 is a specific CXCR2 receptor antagonist, and
studies have shown that SB225002 induces apoptosis in ovarian
cancer cells and cell death and cell cycle arrest in acute
lymphoblastic leukemia cells (27,28).
However, few studies have described the inhibition of cancer cell
invasion or metastasis by SB225002. This study shows for the first
time that SB225002-treated human PCa DU-145, LNCAP and PC-3 cells
exhibited reduced invasion ability. At the same time, we detected
the expression of BSP, OPN, MMP-2, MMP-9 and αvβ3 after treatment
with SB225002 and different signaling pathway inhibitors to further
clarify the underlying molecular mechanism of SB225002 function in
PCa cells.
Materials and methods
Cells and culture
Human androgen-independent prostate cancer DU-145
cells were obtained from Biotechnology Company (Shenyang, China)
and were cultured in MEM medium (Corning Inc., Corning, NY, USA),
supplemented with 10% FBS and 1% penicillin/streptomycin and
cultured in 5% CO2 at 37°C. Androgen-independent
prostate cancer PC-3 cells and androgen-dependent prostate cancer
LNCAP cells were provided by the Brain and Spinal Injury Laboratory
of Liaoning Province (Liaoning, China). PC-3 cells were cultured in
F-12, and LNCAP cells were cultured in RPMI-1640; the other culture
conditions were the same as those of DU-145 cells.
Reagents and treatment
LY294002 (Akt inhibitor), U0126 (ERK1/2 inhibitor),
SB203580 (p38 MAPK inhibitor), SP600125 (JNK1/2 inhibitor) and
SB225002 (CXCR2 receptor antagonist) were purchased from Selleck
Chemicals (Houston, TX, USA). The primary antibodies for PI3K (cat.
no. sc-71891), AKT (cat. no. sc-5270)/p-AKT (cat. no. sc-271966)
and mTOR (cat. no. sc-293089PE)/ p-mTOR (cat. no. sc-293133) were
purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Antibodies for MMP-2 (WL1579), MMP-9 (WL01580), and OPN (WL02848)
were obtained from Wanleibio (Shenyang, China). BSP (BA2329) was
purchased from Boster Biological Technology (Wuhan, China). αvβ3
(bs-1310R) and p-PI3K (bs-6417R) were purchased from Bioss
Biological Technology (Beijing, China). MTT and DAPI were provided
by Solarbio (Beijing, China), and Transwell Matrigel was purchased
from Corning Inc. The anti-human BSP Elisa kit was purchased from
AMEKO (Shanghai Lianshuo Biological Technology Co., Ltd., Shanghai,
China).
Cell viability analysis
Three PCa cell lines were seeded into 96-well plates
(at a cell density of 2×103 cells/well) cultured in
normal growth medium and treated with different concentrations of
SB225002 (0, 1, 3, 5, 10 and 15 µM) for 72 h. After incubation, MTT
solution (0.5 mg/ml) was added to each well, and the plates were
incubated in a humidified incubator at 37°C for 4 h. At the end of
the incubation period, the medium was removed, formazan was
dissolved in DMSO, and the optical densities were determined at 490
nm using a microplate reader. The cell growth inhibition rate was
calculated using the following equation: Cell growth inhibition
rate = 1-(absorbance of experimental value/absorbance of
control).
Migration and invasion assays
In vitro invasion was determined in 24-well
Transwell inserts with 8-µm pore-size filters. The basement
membrane Matrigel was diluted to 200 µg/ml with serum-free
RPMI-1640 medium, and the filters were coated with 100 µl of
basement membrane Matrigel, air-dried and hydrated with 100 µl of
serum-free RPMI-1640 per well for 30 min prior to cell addition.
Cells were added to the upper chamber inserts at a concentration of
5×104 cells in 0.2 ml of serum-free medium (at least 3
replicates for each sample). Three media (500 ml) containing 20%
FBS were added to the lower chamber. In the SB225002 group, 5 µM of
the drug was added. After incubation for 48 h at 37°C, cells in the
upper part of the Transwells were removed with a cotton swab, and
the chambers were washed with PBS. Cells that had migrated were
fixed with 4% paraformaldehyde and stained with 0.1% crystal violet
for 10 min (13). At the same time,
the experimental group and the control group without Matrigel were
set up to calculate the invasion rate of the cancer cells. For
migration, cells were plated in 6-well plates at a density of
5×105 cells per well until they reached 90% confluence.
A single wound was scraped with a pipette tip (200 µl was used) in
the center of the cell monolayer, and the wells were washed with
PBS to remove cell debris. After an additional 48 h of culture,
wound healing was visualized with an inverted microscope (Olympus
IX51; Olympus Corp., Tokyo, Japan).
Western blot analysis
Cells were harvested and lysed with lysis buffer
containing the protease inhibitor phenylmethylsulfonyl fluoride
(PMSF). Each group of protein samples was quantified using the BSA
Protein Quantification kit. Equal amounts of protein (20 µg/lane)
from the sample were electrophoresed on 10% SDS-PAGE gels and were
transferred to PVDF membranes. The membranes were blocked with 5%
skim milk in TBS containing 0.1% Tween-20 for 1 h at room
temperature. After washing with TBST three times, the membranes
were co-incubated with the primary antibody (1:500 for anti-BSP,
1:500 for anti-MMP2, 1:500 for GAPDH) overnight at 4°C in TBST.
After incubation with horseradish peroxidase (HRP) goat anti-rabbit
IgG (1:10,000) in TBST for 60 min, the proteins were visualized
using the ECL detection kit (Wanleibio). Imaging system used
ImageQuant™ LAS 4000 (GE Healthcare, Chicago, IL, USA).
Tumor xenografts
Fifteen BALB/c mice (as SIBLING proteins help tumor
cells escape immune system attack, nude mice were not selected)
weighing 18–22 g were obtained from the Brain and Spinal Injury
Laboratory of Liaoning (Liaoning, China) and reared at a
temperature 18–29°C; relative humidity 50–60%; ventilation 8–12
times/h; light 10–12 h/day, and were randomly divided into three
groups: untreated group, DU-145 implantation group and DU-145
implantation + SB225002 injection group. The number of cells
implanted was 1×106/mouse. All the procedures were
performed in accordance with the Regulations of Experimental Animal
Administration issued by the Ministry of Science and Technology of
China. The tumor volume (V) was measured at 1 week (W) and 2 W
after DU-145 implantation and SB225002 injection, and the
measurements were calculated as V = (length ×
width2)/2.
Immunohistochemistry
After xenograft implantation was completed at 1 W
and 2 W, the tumors were harvested, paraffin embedded,
deparaffinized and rehydrated through a gradient alcohol series
using standard protocols. Next, endogenous peroxidase was
inactivated with 3% hydrogen peroxide for 6 min, antigen unmasking
was performed by heat retrieval performed using citrate buffer (pH
6.1), and the slides were washed in PBS three times. Each group was
incubated with the respective primary antibody overnight at 4°C in
a humid chamber after blocking in goat serum at room temperature
for 15 min. Thereafter, anti-rabbit biotinylated secondary antibody
was added, followed by incubation at 37°C for 1 h, the addition of
HRP labeled streptavidin, and incubation at room temperature for 30
min with DAB color 3 min. Finally, hematoxylin was used to dye the
nuclei. The results were evaluated by two pathologists who
performed a double-blind reading as follows: Colorless, 0 points;
light yellow, 1 point; dark yellow, 2 points; brown, 3 points. The
percentage of positive cells was counted in each field. The
percentage of positive cells per 100 cells was counted under a
upright microscope (Olympus BX53; Olympus Corp.) (magnification,
×200): 0 indicates negative, 1 indicates the percentage of positive
cells <10%, 2 indicates the percentage of positive cells 10–50%,
3 indicates the percentage of positive cells >50–75%, and 4
indicates the percentage of positive cells >75%. The average
score for each group was calculated as the average color depth
multiplied by the percentage of the positive cell score. For the
final score, <1 indicates negative (−), 1–2 indicates weakly
positive (+), 3 −4 indicates positive (++), and >4 indicates
strongly positive (+++).
ELISA
Enzyme-linked immunosorbent assay was performed to
determine whether SB225002 reduced BSP and OPN secretion in
vivo. At the end of 7 days of SB225002 injection, mouse blood
was obtained through eye arteries. After centrifugation at 1,000
rpm for 10 min, the serum was separated from whole-blood samples,
and BSP and OPN were detected using the Human BSP/OPN ELISA
kit.
Immunofluorescence
DU-145 and PC-3 cells were seeded into 96-well
plates. When the cell density reached 60%, the medium was removed,
and the cells were fixed with 4% paraformaldehyde at room
temperature for 30 min. The cells were then permeabilized using
Triton X-100, and each well was treated with blocking buffer (1X
TBST, 3% goat serum) for 1 h at room temperature, followed by
overnight incubation at 4°C with primary antibodies (BSP, 1:300;
OPN, 1:300; αvβ3, 1:100) diluted in blocking buffer. The samples
were washed 3 times with 1X PBS and were incubated with secondary
antibodies for 1 h before mounting with Prolong Gold antifade
reagent (Solarbio, Beijing, China) with DAPI.
Statistical analysis
All the statistical analyses were evaluated using
SPSS 21.0 software (IBM Corp., Armonk, NY, USA). Data are presented
as the means ± SD (standard deviation). Statistical analysis was
performed using one-way ANOVA followed by the Bonferroni or Dunnett
(2-sided) test for comparisons. The level of significance was set
at P<0.05.
Results
SB225002 inhibits PCa cell
proliferation and invasion
The MTT results showed that, after 72 h of culture,
DU-145 and PC-3 proliferation was inhibited in a concentration- and
time-dependent manner upon treatment with SB225002. The growth
inhibition rate of the DU-145 and PC-3 cells reached 50% after
treatment with 5 µM SB225002 for 48 h, and it reached almost 100%
after treatment with 10 µM SB225002 for 72 h. For LNCAP cells, 15
µM SB225002 for 72 h showed an inhibition rate of 85% (Table I). To further explore the effect of
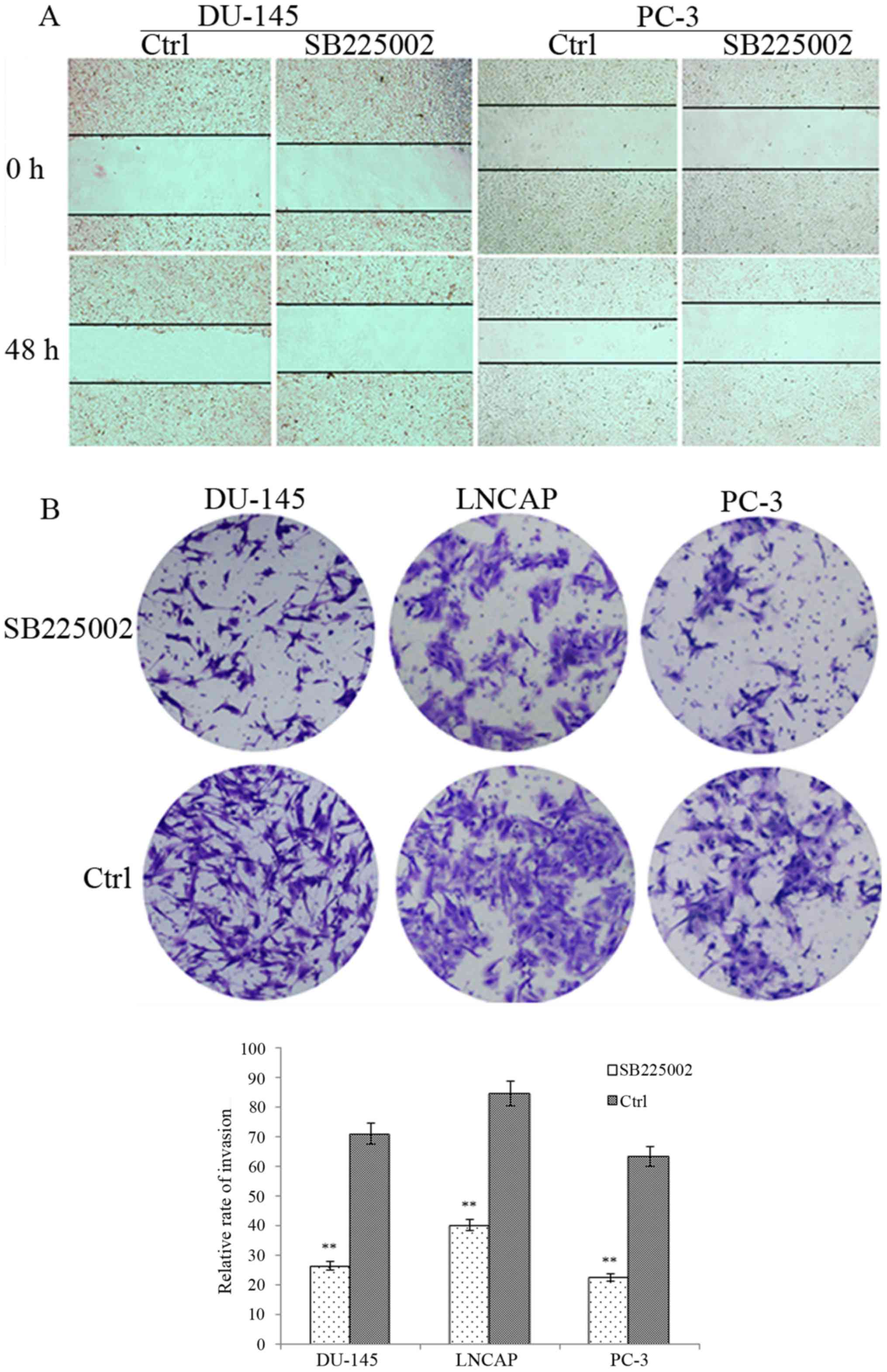
SB225002 on invasion ability, the Transwell assay was used to
validate the effect of SB225002 on the invasive ability of PCa
cells. After 48 h of treatment, the number of SB225002-treated
cancer cells from the three cell lines invading through the
Matrigel barrier was less than that of the control groups (Fig. 1B). After the cells were counted
under random high magnification, the DU-145 penetration rate of the
control group was 71.21% and that of the SB225002 group was 26.54%
(P=0.005). The LNCAP penetration rate of the control group was
84.67% for the control and 40.31% after SB225002 treatment
(P=0.005). Finally, the PC-3 penetration rate was 63.44% for the
control and 22.65% after SB225002 treatment (P=0.001). For the
migration assay (Fig. 1A), after 48
h of incubation, the wounds in the control groups of the DU-145 and
PC3 cells were significantly reduced by 25% for DU-145 and 30% for
PC-3; however, in the SB225002 group, the two cell lines migrated
poorly, showing a decrease of 10 and 5%, respectively, in wound
healing.
 | Table I.MTT assay was used to detect the
inhibitory effect of SB225002 on prostate cancer cell
proliferation. |
Table I.
MTT assay was used to detect the
inhibitory effect of SB225002 on prostate cancer cell
proliferation.
| A, DU-145 |
|---|
|
|---|
|
| OD (n=3, × ±
SD) | Inhibition rate of
cell growth (%) |
|---|
|
|
|
|
|---|
| SB225002
(µmol/l) | 24 h | 48 h | 72 h | 24 h | 48 h | 2 h |
|---|
| 0 | 0.211±0.023 | 0.274±0.197 | 0.281±0.026 |
0.00 |
0.00 |
0.00 |
| 5 | 0.109±0.017 | 0.137±0.013 | 0.110±0.028 | 48.34 | 50.00 | 60.85 |
| 10 | 0.059±0.015 | 0.042±0.010 | 0.003±0.001 | 72.22 | 84.85 | 98.91 |
| 15 | 0.026±0.012 | 0.021±0.009 | 0.002±0.001 | 87.81 | 92.34 | 99.01 |
|
| B, PC-3 |
|
|
| OD (n=3, × ±
SD) | Inhibition rate
of cell growth (%) |
|
|
|
|
| SB225002
(µmol/l) | 24 h | 48 h | 72 h | 24 h | 48 h | 2 h |
|
| 0 | 0.203±0.019 | 0.272±0.109 | 0.293±0.016 |
0.00 |
0.00 |
0.00 |
| 5 | 0.119±0.007 | 0.138±0.013 | 0.108±0.034 | 41.37 | 49.29 | 63.13 |
| 10 | 0.077±0.026 | 0.041±0.031 | 0.003±0.001 | 62.11 | 84.79 | 99.01 |
| 15 | 0.038±0.022 | 0.019±0.009 | 0.004±0.001 | 81.27 | 93.12 | 98.70 |
|
| C,
LNCAP |
|
|
| OD (n=3, × ±
SD) | Inhibition rate
of cell growth (%) |
|
|
|
|
| SB225002
(µmol/l) | 24 h | 48 h | 72 h | 24 h | 48 h | 2 h |
|
| 0 | 0.224±0.011 | 0.247±0.034 | 0.291±0.020 |
0.00 |
0.00 |
0.00 |
| 5 | 0.140±0.003 | 0.136±0.060 | 0.152±0.024 | 37.70 | 45.02 | 47.77 |
| 10 | 0.120±0.036 | 0.125±0.031 | 0.123±0.071 | 47.27 | 49.44 | 57.89 |
| 15 | 0.108±0.023 | 0.092±0.019 | 0.042±0.017 | 51.71 | 62.70 | 85.27 |
Co-expression of BSP/OPN and αvβ3 in
DU-145 and PC-3 cells
It was reported that BSP/OPN combine with integrin
proteins, especially αvβ3, on the cell surface to promote cell
migration and invasion. We used immunofluorescence to determine
whether BSP/OPN and αvβ3 were co-expressed in the PCa cells
(Fig. 2A and B). After labeling
with different colors of the fluorescent secondary antibody, BSP
and OPN were shown in the images as red fluorescence and αvβ3
showed green fluorescence. Additionally, nuclei stained with DAPI
exhibited blue fluorescence. Co-expression of BSP-αvβ3 and OPN-αvβ3
was evident in the DU-145 and PC-3 cells.
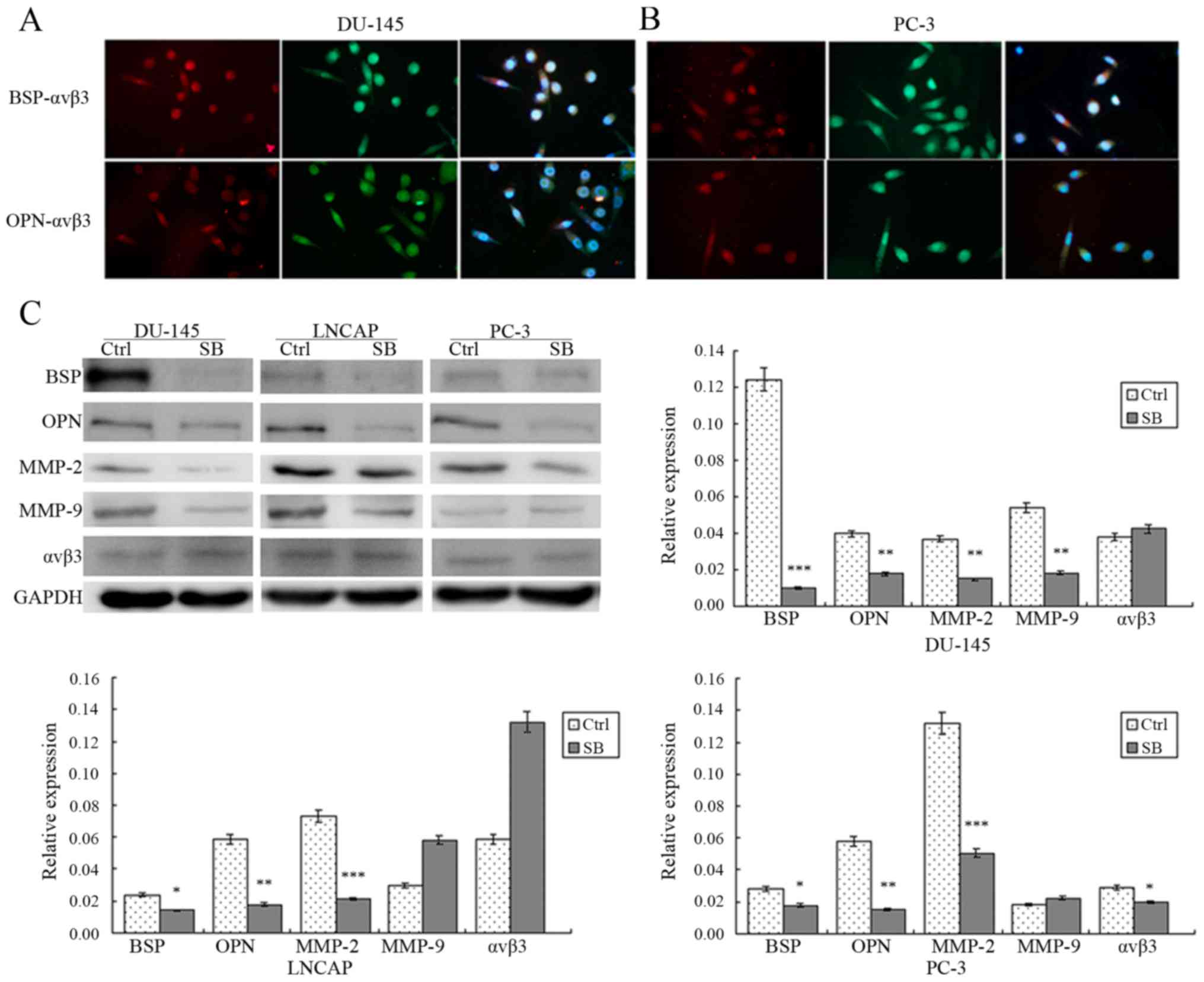 | Figure 2.(A and B) Immunofluorescence detected
that BSP/OPN and αvβ3 were co-expressed in DU-145 and PC-3 cells.
(C) Western blotting showed that SB225002 reduced expression of
BSP, OPN and MMP-2 in the PCa cells (P<0.05), but not that of
MMP-9 and αvβ3. Each histogram was compared with the
semi-quantitative result of western blot analysis; each experiment
was repeated at least 3 times, *P<0.05, **P<0.01,
***P<0.001. PCa, prostate cancer; BSP, bone sialoprotein; OPN,
osteopontin; MMP, metalloproteinase; αvβ3, integrin alpha v and
integrin beta 3; SB, SB22502 (a specific CXCR2 receptor
antagonist); Ctrl, control; LY, LY294002 (Akt inhibitor); U0, U0126
(ERK1/2 inhibitor); S, SB203580 (p38 MAPK inhibitor); SP, SP600125
(JNK1/2 inhibitor). (D-H) Effect of different signaling pathway
inhibitors on BSP, OPN, MMP-2, MMP-9 and αvβ3 expression. The
expression levels of BSP, OPN, MMP-2 and αvβ3 were decreased
significantly in the LY294002 group of the DU-145 and PC-3 cells;
however, levels of these five proteins were not obviously changed
in the LNCAP cells. Each histogram was compared with the
semi-quantitative result of western blot analysis; each experiment
was repeated at least 3 times, *P<0.05, **P<0.01,
***P<0.001. PCa, prostate cancer; BSP, bone sialoprotein; OPN,
osteopontin; MMP, metalloproteinase; αvβ3, integrin alpha v and
integrin beta 3; SB, SB22502 (a specific CXCR2 receptor
antagonist); Ctrl, control; LY, LY294002 (Akt inhibitor); U0, U0126
(ERK1/2 inhibitor); S, SB203580 (p38 MAPK inhibitor); SP, SP600125
(JNK1/2 inhibitor). (I) Expression of p-AKT and p-mTOR was
decreased obviously in the DU-145 and PC-3 cells after SB225002
treatment. Each histogram was compared with the semi-quantitative
result of western blot analysis; each experiment was repeated at
least 3 times, *P<0.05, **P<0.01, ***P<0.001. PCa,
prostate cancer; BSP, bone sialoprotein; OPN, osteopontin; MMP,
metalloproteinase; αvβ3, integrin alpha v and integrin beta 3; SB,
SB22502 (a specific CXCR2 receptor antagonist); Ctrl, control; LY,
LY294002 (Akt inhibitor); U0, U0126 (ERK1/2 inhibitor); S, SB203580
(p38 MAPK inhibitor); SP, SP600125 (JNK1/2 inhibitor). |
SB225002 inhibits the expression of
BSP, OPN, MMP2, MMP9 and αvβ3 in PCa cells
To explore the molecular mechanism of the
suppression of PCa invasion by SB225002, western blotting was
performed after treatment of the three PCa cell lines with SB225002
(5×10−6 M in 72 h). The results showed that the
expression levels of BSP, MMP-2 and OPN were reduced significantly
following SB225002 treatment compared with levels noted in the
control group: BSP was reduced by 9.04-fold in DU-145 cells, by
1.7-fold in LNCAP cells and by 5.4-fold in PC-3 cells; OPN was
reduced by 3.0-fold in DU-145 and PC-3 cells and by 2.8-fold in
LNCAP cells; MMP-2 was reduced by 3.2-fold in DU-145 cells, by
5.2-fold in PC-3 cells and by 4.5-fold in LNCAP cells, P<0.05
(Fig. 2C). Regarding MMP-9 and
αvβ3, the expression levels of both proteins were not reduced
significantly in the LNCAP cells (P=0.08), while MMP-9 expression
was decreased by 3.7-fold in the DU145 cells, and αvβ3 expression
was reduced by 1.3-fold in the PC-3 cells. These results indicate
that SB225002 may suppress PCa invasion through restraining the
expression of BSP, MMP-2 and OPN.
ERK, JNK, P38 and PI-3K signaling
pathways mediate the expression of BSP, OPN, MMP-2, MMP-9 and
αvβ3
To identify which signal transduction pathway(s)
regulate several of the abovementioned invasion-related protein
expression levels, we applied the inhibitors LY294002 (Akt
inhibitor), U0126 (ERK1/2 inhibitor), SB203580 (p38 MAPK
inhibitor), and SP600125 (JNK1/2 inhibitor) and detected the
expression of these proteins by western blotting. The three cancer
cell lines were treated with LY294002 (10−6 M), U0126
(10−6 M), SB203580 (10−6 M), and SP600125
(10−6 M) for 72 h. The findings revealed that BSP, OPN,
MMP-2 and αvβ3 protein levels were significantly lower in the
DU-145 and PC-3 cells after LY294004 treatment than after the other
inhibitor treatments (Fig. 2D-F and
H). This result is consistent with previous reports that PI3K
can regulate tumor cell invasion. Although the expression level of
MM-9 in the LY29402 treatment group was lower than that in the
control group (DU-145 and PC-3), the difference was not
statistically significant (Fig.
2G). Regarding LNCAP cells, although the BSP and MMP-9
expression levels were reduced, no statistically significant
difference was noted, and no obvious expression changes were found
in the remaining three proteins (OPN, MMP-2 and αvβ3) after
LY294002 treatment in LNCAP cells. Moreover, in the SP600125
treatment group, we found that the expression levels of BSP and
αvβ3 showed little reduction, especially in DU-145 and PC-3 cells,
suggesting that the JNK signaling pathway can also regulate tumor
cell invasion.
SB225002 inhibits the
phosphorylation/activation of AKT and mTOR
To identify the effect of SB225002 on the PI3K
signaling pathway, we treated the three PCa cell lines with
SB225002 (10−6 M for 72 h). The primary proteins of the
PI3K signaling pathway (PI3K/p-PI3K, AKT/p-AKT and mTOR/p-mTOR)
were detected by western blotting. The result shows that SB225002
did not significantly promote the phosphorylation of PI3K, and PI3K
expression was not significantly changed. When we further detected
the PI3K downstream protein AKT, only DU-145 cells demonstrated a
decrease in the expression of AKT, and AKT in LNCAP and PC-3 cells
was not changed; however, phosphorylated Akt in all three cancer
cell lines showed a significant downward trend. Finally, the
expression levels of mTOR and p-mTOR in the SB225002 group were
lower than those of the control group (Fig. 2I). The above results illustrate that
SB225002 has a certain blocking effect on the PI3K signaling
pathway, and this block effect may begin with the inhibition of
phosphorylation of AKT.
SB225002 suppresses PCa cell growth
and the secretion of BSP and OPN in
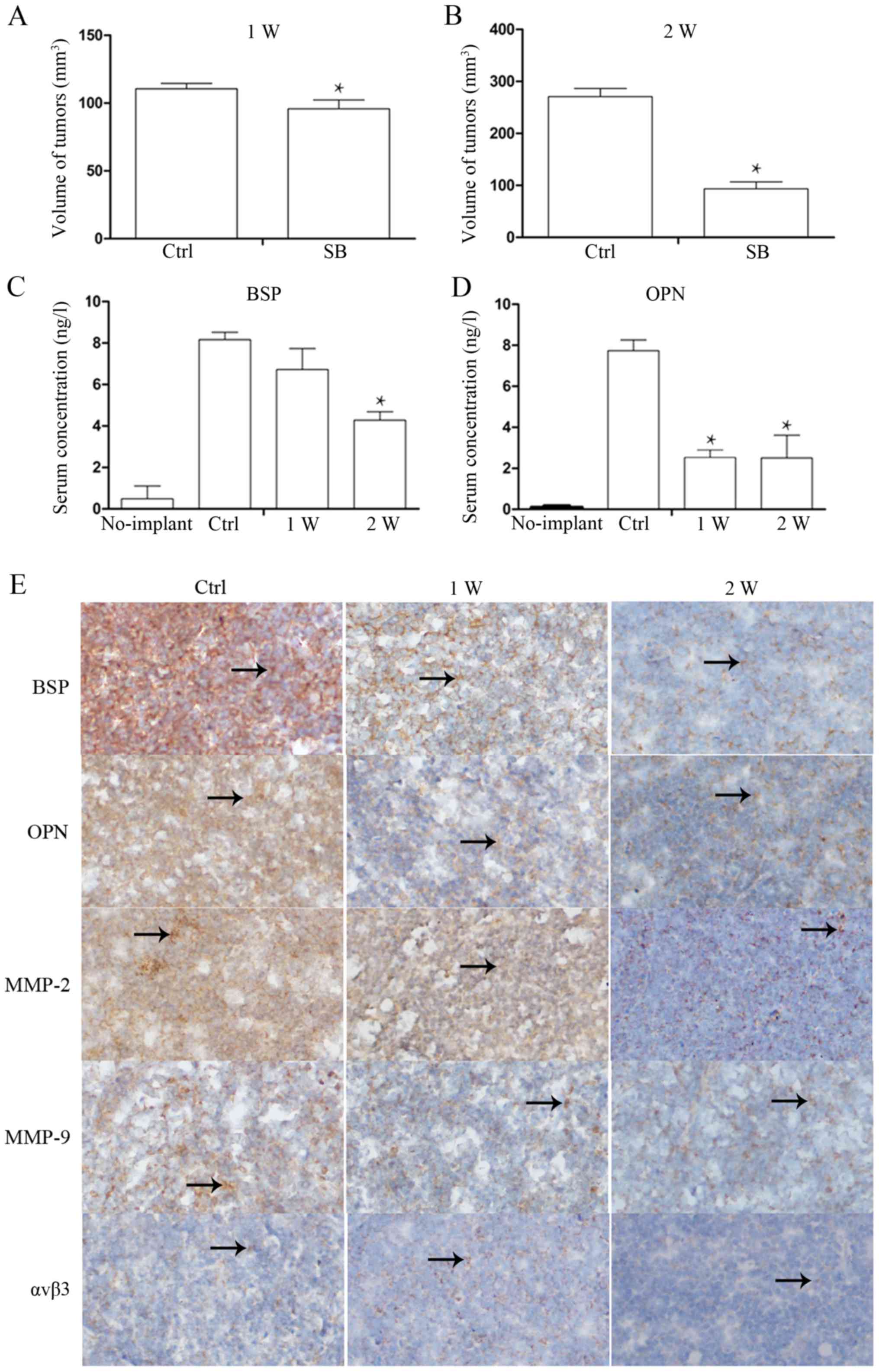
Toexamine whether SB225002 inhibits the secretion of
BSP and OPN from PCa cells in vivo, after the xenografts
were harvested at 1 week (W) and 2 W, whole blood from the treated
and control group mice was obtained from the eye artery, and
enzyme-linked immunosorbent assay was performed after centrifuging
the blood at 1,000 rpm for 10 min. The results revealed that the
serum BSP and OPN levels in the no implant mice were low BSP,
0.724±0.3 ng/l; OPN, 0.132±0.01 ng/l). However, after two weeks of
SB225002 administration, the serum levels of BSP (9.201±0.4 ng/l in
the control group and 4.821±0.6 ng/l in the SB225002 group; P=0.01)
and OPN (8.431±0.5 ng/l in the control group and 3.812±1.4 ng/l in
the SB225002 group; P=0.04) were decreased by 2-fold compared with
the levels in the control group (Fig.
3C and D), indicating that SB225002 can inhibit PCa cell
secretion of BSP and OPN in vivo. We also measured the
volume of the xenografts at 1 W and 2 W after SB225002 injection.
At the end of the first week, the tumor volume was 110.5709±3.98
mm3 in the control group and 95.8498±6.49 mm3
in the SB225002 group. At the end of the second week, the tumor
volume of the control group had reached 270.7950±15.59
mm3, and the tumor volume of the SB225002 group was
93.3554±13.34 mm3 (P=0.006) (Fig. 3A and B). The BSP, OPN and MMP-2
expression levels in xenografts were similarly determined by
immunohistochemistry. In xenografts, positively expressed proteins
were stained to yellow or brown yellow and appears in the shape of
dot or sheet. The arrows indicate protein positive expression
(Fig. 3E). The expression levels of
BSP, OPN and MMP-9 were significantly decreased after SB225002
injection for one week, while MMP-2 expression level remained
strongly. But the MMP-2 expression was significantly reduced after
two weeks of injection. As for αVβ3, there was no significant
change before and after SB225002 injection. After 2 weeks, the
positive staining scores of the five proteins were calculated and
are shown in Table II.
 | Table II.Expression intensity assessments of
five proteins. |
Table II.
Expression intensity assessments of
five proteins.
|
| BSP | OPN | MMP-2 | MMP-9 | αvβ3 |
|---|
| Ctrl | +++ | +++ | +++ | ++ | + |
| 1 week | ++ | + | ++ | + | + |
| 2 weeks | + | + | + | + |
|
Discussion
Invasion and metastasis are two major obstacles to
the treatment of malignant tumors (1,2). Many
patients lose the chance of surgical treatment due to the transfer
of primary tumors to distant organs. For PCa, although most
patients respond initially to androgen removal, most patients
eventually develop castration resistance and have a high risk of
bone metastases (21). Thus, there
is an urgent need to explore effective strategies to prevent
distant tumor metastasis to improve the prognosis of patients.
At the molecular level, malignant cells must be able
to detach from their primary tissues, evade the host immune system,
cross the walls of the vasculature, penetrate through the
extracellular matrix in tissue, and finally take up residence and
survive in tissues quite different from their origins (3). Studies over recent years have
suggested that, besides MMP family proteins, small integrin binding
ligand N-linked glycoproteins (SIBLINGs) also regulate many of the
activities required for the distant metastasis of tumor cells,
especially for malignant bone metastases. Additionally, the serum
levels of BSP and OPN (two primary proteins of the SIBLING family
and the most frequently investigated) are often used to predict the
occurrence of bone metastases (2,7,9–11).
As mentioned previously, BSP and OPN contain an integrin-binding
RGD (Arg-Gly-Asp) sequence that can bind to integrins to enhance
the invasion and adhesion of tumor cells (29). However, OPN peptides must be cleaved
by MMP-9 first, followed by the enhancement of matrix degradation
by activating MMP-3 (38). In terms
of immune escape, after tumor cells enter the vasculature, these
two SIBLINGs can bind complement factor H and prevent tumor cells
from complement attack (30). In
addition, some studies have indicated that SIBLING proteins can
also be combined with the CD44 regulation of tumor cell
proliferation and apoptosis (3).
Because SIBLINGs are important factors in the regulation of tumor
metastasis, to decrease tumor cell expression, SIBLINGs may be an
efficiency strategy by which to overcome the distant metastasis of
PCa. Many studies have reported that phosphoinositide-3-kinase
(PI3K) is an important signaling pathway responsible for malignant
neoplasm growth and transformation processes (31). For invasion, the PI3K signaling
pathway mediates the expression of MMP-2 and MMP-9 (32,33).
Zhang et al demonstrated that the PI3K/Akt pathway inhibitor
LY294002 attenuated the migration, invasion, expression and
activity of MMP and expression of p-PI3K and p-Akt in U87 and U251
cells (34). However, few studies
have reported on the factors that regulate SIBLING expression. In
this study, a series of in vitro and in vivo
experiments confirmed that SB225002 could decrease PCa expression
of BSP and OPN through the PI3K pathway.
As our results showed, following treatment of three
prostate cell lines with different concentrations of SB225002,
concentration- and time-dependent growth inhibition was
demonstrated in DU145 and PC-3 cells but not in LNCAP cells. The
lack of an effect in LNCAP cells is likely due to LNCAP belonging
to the androgen-dependent cell group, and some reports have
demonstrated that IL-8 and its receptors are not expressed or
negligibly expressed in androgen-dependent PCa (18,35–37).
Additionally, the Transwell assay exhibited that SB225002 could
decrease the number of cancer cells that crossed the Matrigel
barrier, indicating that SB225002 can reduce the invasion of PCa
cells. Many studies have demonstrated SIBLING and integrin
expression in breast cancer, but few have been reported in PCa.
Considering that SIBLINGs enhance invasion through combining with
integrin receptors, we evaluated the co-expression of BSP, OPN and
αvβ3 in DU-145 and PC-3 cells, and immunofluorescence analysis
indicated all three proteins were expressed in PCa. Simultaneously,
western blotting was performed to detect the influence of SB225002
on these invasion-related proteins, and SB225002 treatment was
found to decrease the expression of BSP, OPN and MMP-2 in the three
cell lines. However, MMP-9 expression was only reduced in DU-145
cells, and SB2250022 did not inhibit the expression of αvβ3. By
contrast, following treatment with SB225002 treatment, the αvβ3
expression levels showed an increasing trend in the three cell
lines. Next, we treated cells with different signaling pathway
inhibitors to detect which pathways control tumor cell invasion
primarily. After U0126, SP600125, SB230580 and LY294002 treatment,
the expression of the five proteins in the LY294002 group was
obviously inhibited, consistent with previous reports describing
that PI3K regulates the invasion of malignant neoplasms (32–34).
Next, we tested the signaling protein in the PI3K pathway in the
SB225002 and control groups to determine whether SB225002
suppresses PCa cell invasion through the PI3K pathway. Western
blotting showed that, in the SB225002 group, P-AKT expression was
decreased obviously, the expression levels of downstream protein
mTOR and P-mTOR were significantly reduced, and the expression
levels of PI3K and P-PI3K did not change, suggesting that the
function of SB225002 to restrain tumor cell invasion was achieved
by inhibiting the phosphorylation of AKT. Finally, we implanted
DU-145 cells into mice subcutaneously, through two weeks of
continuous intraperitoneal administration and confirmed that
SB225002 suppressed PCa cell expression and secretion of BSP and
OPN in vivo, in addition to MMP-2.
In conclusion, many studies have confirmed that
SB225002 is an IL-8 receptor antagonist (17,38,39).
SB225002 exhibits many antitumor effects by blocking the binding of
IL-8 to CXCR2 receptors. This experiment confirmed that SB225002
has an inhibitory effect on the expression of invasion-related
proteins. These findings may provide new ideas and methods to
prevent the distant metastasis of tumors in clinical practice.
Acknowledgements
We would like to thank Professor Huamao Jiang for
technical guidance in this experiment.
Funding
The present study was supported by the National
Natural Science Foundation of China (no. 81672265) and the
Distinguished Professor Fund of Liaoning Provincial Department of
Education [Liaojiaofa (2015) no. 153].
Availability of data and materials
The datasets analyzed during the current study are
available from the corresponding author on reasonable request.
Author's contributions
HJ and MX conceived and designed the study. MX, JL,
HW, BL and ZG performed the experiments. MX wrote the paper. HJ and
HW reviewed and edited the manuscript. All authors read and
approved the manuscript and agree to be accountable for all aspects
of the research in ensuring that the accuracy or integrity of any
part of the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
The animal experiment was approval by the JinZhou
University Laboratory Animal Ethics Review Committee (JinZhou,
China).
Patient consent for publication
Not applicable.
Competing interests
The authors state that they have no competing
interests.
References
|
1
|
Weigelt B, Peterse JL and van't Veer LJ:
Breast cancer metastasis: Markers and models. Nat Rev Cancer.
5:591–602. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rizzoli R, Body JJ, Brandi ML,
Cannate-Andia J, Chappard D, El Maghraoui A, Glüer CC, Kendler D,
Napoli N, Papaioannou A, et al: Cancer-associated bone disease.
Osteoporos Int. 24:2929–2953. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kruger TE, Miller AH, Godwin AK and Wang
J: Bone sialoprotein and osteopontin in bone metastasis of
osteotropic cancers. Crit Rev Oncol Hematol. 89:330–341. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen J, Rodriguez JA, Barnett B, Hashimoto
N, Tang J and Yoneda T: Bone sialoprotein promotes tumor cell
migration in both in vitro and in vivo models. Connect Tissue Res.
44 (Suppl 1):S279–S284. 2003. View Article : Google Scholar
|
|
5
|
Gordon JA, Sodek J, Hunter GK and Goldberg
HA: Bone sialoprotein stimulates focal adhesion-related signaling
pathways: Role in migration and survival of breast and prostate
cancer cells. J Cell Biochem. 107:1118–1128. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Anunobi CC, Koli K, Saxena G, Banjo AA and
Ogbureke KU: Expression of the SIBLINGs and their MMP partners in
human benign and malignant prostate neoplasms. Oncotarget.
7:48038–48049. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Righi L, Bollito E, Ceppi P, Mirabelli D,
Tavaglione V, Chiusa L, Porpiglia F, Brunelli M, Martignoni G,
Terrone C and Papotti M: Prognostic role of bone sialoprotein in
clear cell renal carcinoma. Anticancer Res. 33:2679–2687.
2013.PubMed/NCBI
|
|
8
|
Zhang L, Hou X, Lu S, Rao H, Hou J, Luo R,
Huang H, Zhao H, Jian H, Chen Z, et al: Predictive significance of
bone sialoprotein and osteopontin for bone metastases in resected
Chinese non-small-cell lung cancer patients: A large cohort
retrospective study. Lung Cancer. 67:114–119. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
D'Oronzo S, Brown J and Coleman R: The
value of biomarkers in bone metastasis. Eur J Cancer Care (Engl).
26:2017. View Article : Google Scholar
|
|
10
|
Wang Y, Zhang XF, Dai J, Zheng YC, Zhang
MG and He JJ: Predictive value of serum bone sialoprotein and
prostate-specific antigen doubling time in patients with bone
metastasis of prostate cancer. J Huazhong Univ Sci Technolog Med
Sci. 33:559–562. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wei RJ, Li TY, Yang XC, Jia N, Yang XL and
Song HB: Serum levels of PSA, ALP, ICTP, and BSP in prostate cancer
patients and the significance of ROC curve in the diagnosis of
prostate cancer bone metastases. Genet Mol Res. 15:gmr7707. 2016.
View Article : Google Scholar
|
|
12
|
Bellahcène A, Castronovo V, Ogbureke KU,
Fisher LW and Fedarko NS: Small integrin-binding ligand N-linked
glycoproteins (SIBLINGs): Multifunctional proteins in cancer. Nat
Rev Cancer. 8:212–226. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang J, Wang L, Xia B, Yang C, Lai H and
Chen X: BSP gene silencing inhibits migration, invasion, and bone
metastasis of MDA-MB-231BO human breast cancer cells. PLoS One.
8:e629362013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Waltregny D, Bellahcène A, de Leval X,
Florkin B, Weidle U and Castronovo V: Increased expression of bone
sialoprotein in bone metastases compared with visceral metastases
in human breast and prostate cancers. J Bone Miner Res. 15:834–843.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Khodavirdi AC, Song Z, Yang S, Zhong C,
Wang S, Wu H, Pritchard C, Nelson PS and Roy-Burman P: Increased
expression of osteopontin contributes to the progression of
prostate cancer. Cancer Res. 66:883–888. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lecrone V, Li W, Devoll RE, Logothetis C
and Farach-Carson MC: Calcium signals in prostate cancer cells:
Specific activation by bone-matrix proteins. Cell Calcium.
27:35–42. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sueoka H, Hirano T, Uda Y, Iimuro Y,
Yamanaka J and Fujimoto J: Blockage of CXCR2 suppresses tumor
growth of intrahepatic cholangiocellular carcinoma. Surgery.
155:640–649. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Huang J, Yao JL, Zhang L, Bourne PA, Quinn
AM, di Sant'Agnese PA and Reeder JE: Differential expression of
interleukin-8 and its receptors in the neuroendocrine and
non-neuroendocrine compartments of prostate cancer. Am J Pathol.
166:1807–1815. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li X, Wang S, Zhu R, Li H, Han Q and Zhao
RC: Lung tumor exosomes induce a pro-inflammatory phenotype in
mesenchymal stem cells via NFκB-TLR signaling pathway. J Hematol
Oncol. 9:422016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Arenberg DA, Kunkel SL, Polverini PJ,
Glass M, Burdick MD and Strieter RM: Inhibition of interleukin-8
reduces tumorigenesis of human non-small cell lung cancer in SCID
mice. J Clin Invest. 97:2792–2802. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen K, Wu K, Jiao L, Wang L, Ju X, Wang
M, Di Sante G, Xu S, Wang Q, Li K, et al: The endogenous cell-fate
factor dachshund restrains prostate epithelial cell migration via
repression of cytokine secretion via a cxcl signaling module.
Cancer Res. 75:1992–2004. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Neveu B, Moreel X, Deschênes-Rompré MP,
Bergeron A, LaRue H, Ayari C, Fradet Y and Fradet V: IL-8 secretion
in primary cultures of prostate cells is associated with prostate
cancer aggressiveness. Res Rep Urol. 6:27–34. 2014.PubMed/NCBI
|
|
23
|
Ha NH, Park DG, Woo BH, Kim DJ, Choi JI,
Park BS, Kim YD, Lee JH and Park HR: Porphyromonas gingivalis
increases the invasiveness of oral cancer cells by upregulating
IL-8 and MMPs. Cytokine. 86:64–72. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
De Larco JE, Wuertz BR, Rosner KA,
Erickson SA, Gamache DE, Manivel JC and Furcht LT: A potential role
for interleukin-8 in the metastatic phenotype of breast carcinoma
cells. Am J Pathol. 158:639–646. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
MacManus CF, Pettigrew J, Seaton A, Wilson
C, Maxwell PJ, Berlingeri S, Purcell C, McGurk M, Johnston PG and
Waugh DJ: Interleukin-8 signaling promotes translational regulation
of cyclin D in androgen-independent prostate cancer cells. Mol
Cancer Res. 5:737–748. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Araki S, Omori Y, Lyn D, Singh RK,
Meinbach MD, Sandman Y, Lokeshwar VB and Lokeshwar BL:
Interleukin-8 is a molecular determinant of androgen independence
and progression in prostate cancer. Cancer Res. 67:6854–6862. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Du M, Qiu Q, Gruslin A, Gordon G, He M,
Chan CC, Li D and Tsang BK: SB225002 promotes mitotic catastrophe
in chemo-sensitive and -resistant ovarian cancer cells independent
of p53 status in vitro. PLoS One. 8:e545722013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
de Vasconcellos JF, Laranjeira AB, Leal
PC, Bhasin MK, Zenatti PP, Nunes RJ, Yunes RA, Nowill AE, Libermann
TA, Zerbini LF and Yunes JA: SB225002 induces cell death and cell
cycle arrest in acute lymphoblastic leukemia cells through the
activation of GLIPR1. PLoS One. 10:e01347832015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rapuano BE and MacDonald DE:
Structure-activity relationship of human bone sialoprotein
peptides. Eur J Oral Sci. 121:600–609. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fedarko NS, Fohr B, Robey PG, Young MF and
Fisher LW: Factor H binding to bone sialoprotein and osteopontin
enables tumor cell evasion of complement-mediated attack. J Biol
Chem. 275:16666–16672. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Okkenhaug K and Vanhaesebroeck B: PI3K in
lymphocyte development, differentiation and activation. Nat Rev
Immunol. 3:317–330. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ku MJ, Kim JH, Lee J, Cho JY, Chun T and
Lee SY: Maclurin suppresses migration and invasion of human
non-small-cell lung cancer cells via anti-oxidative activity and
inhibition of the Src/FAK-ERK-β-catenin pathway. Mol Cell Biochem.
402:243–252. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tseng CH, Tzeng CC, Chiu CC, Hsu CY, Chou
CK and Chen YL: Discovery of
2-[2-(5-nitrofuran-2-yl)vinyl)quinoline derivatives as a novel type
of antimetastatic agents. Bioorg Med Chem. 23:141–148. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang FY, Hu Y, Que ZY, Wang P, Liu YH,
Wang ZH and Xue YX: Shikonin inhibits the migration and invasion of
human glioblastoma cells by targeting phosphorylated β-catenin and
phosphorylated PI3K/Akt: A potential mechanism for the anti-glioma
efficacy of a traditional Chinese herbal medicine. Int J Mol Sci.
16:23823–23848. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Murphy C, Mcgurk M, Prttigrew J,
Santinelli A, Mazzucchelli R, Johnston PG, Montironi R and Waugh
DJ: Nonapical and cytoplasmic expression of interleukin-8, CXCR1,
and CXCR2 correlates with cell proliferation and microvessel
density in prostate cancer. Clin Cancer Res. 11:4117–4127. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Seaton A, Scullin P, Mxawell PJ, Wilson C,
Prttigrew J, Gallagher R, O'Sullivan JM, Johnston PG and Waugh DJ:
Interleukin-8 signaling promotes androgen-independent proliferation
of prostate cancer cells via induction of androgen receptor
expression and activation. Carcinogenesis. 29:1148–1156. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tanaka H, Kono E, Tran CP, Miyazaki H,
Yamashiro J, Shimomura T, Fazli L, Wada R, Huang J, Vessella RL, et
al: Monoclonal antibody targeting of N-cadherin inhibits prostate
cancer growth, metastasis and castration resistance. Nat Med.
16:1414–1420. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Takafuji V, Forgues ME, Unsworth E,
Goldsmith P and Wang X: An osteopontin fragment is essential for
tumor cell invasion in hepatocellular carcinoma. Oncogene.
26:6361–6371. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Manjavachi MN, Quintão NL, Campos MM,
Deschamps IK, Yunes RA, Nunes RJ, Leal PC and Calixto JB: The
effects of the selective and non-peptide CXCR2 receptor antagonist
SB225002 on acute and long-lasting models of nociception in mice.
Eur J Pain. 14:23–31. 2010. View Article : Google Scholar : PubMed/NCBI
|