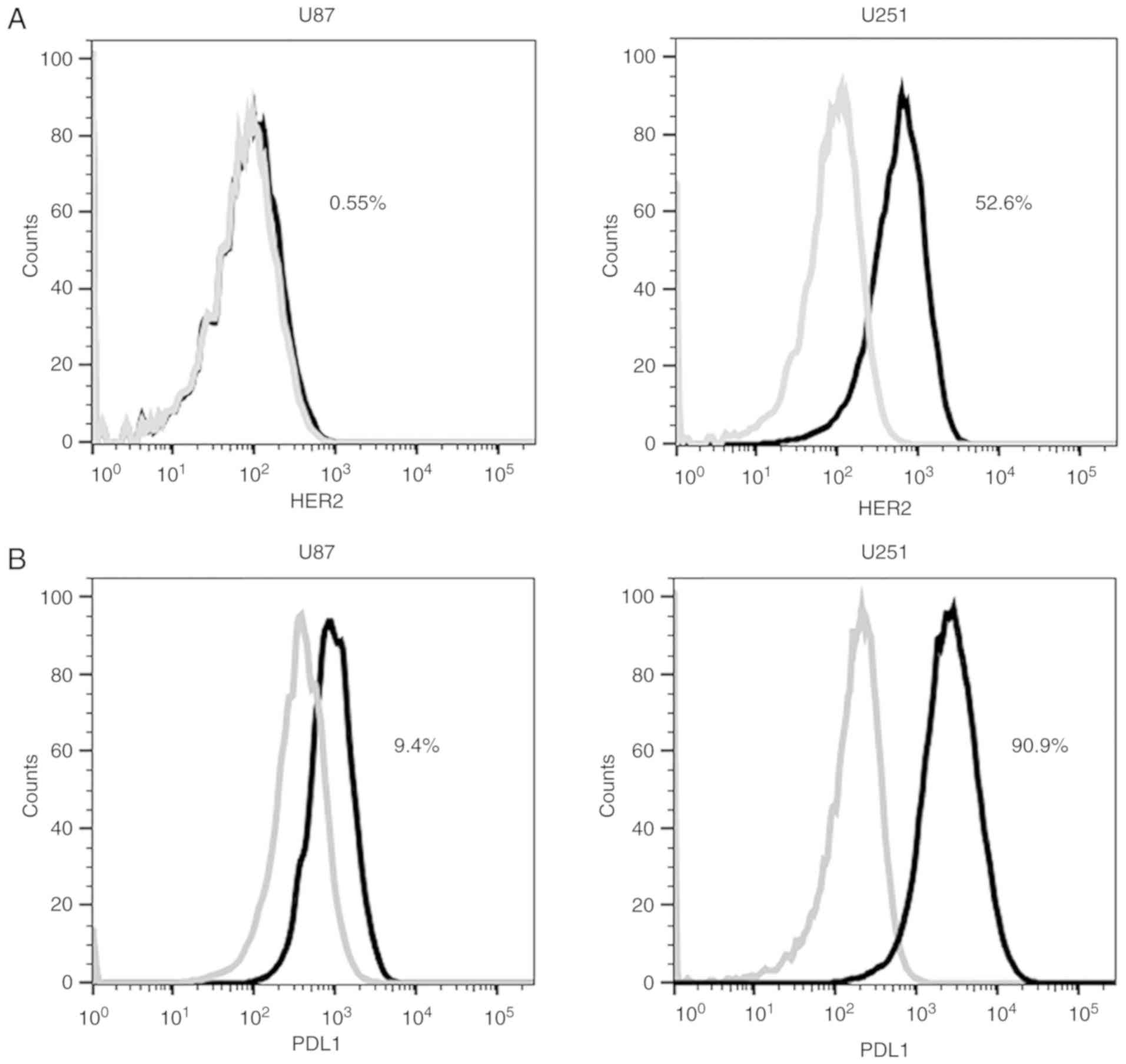
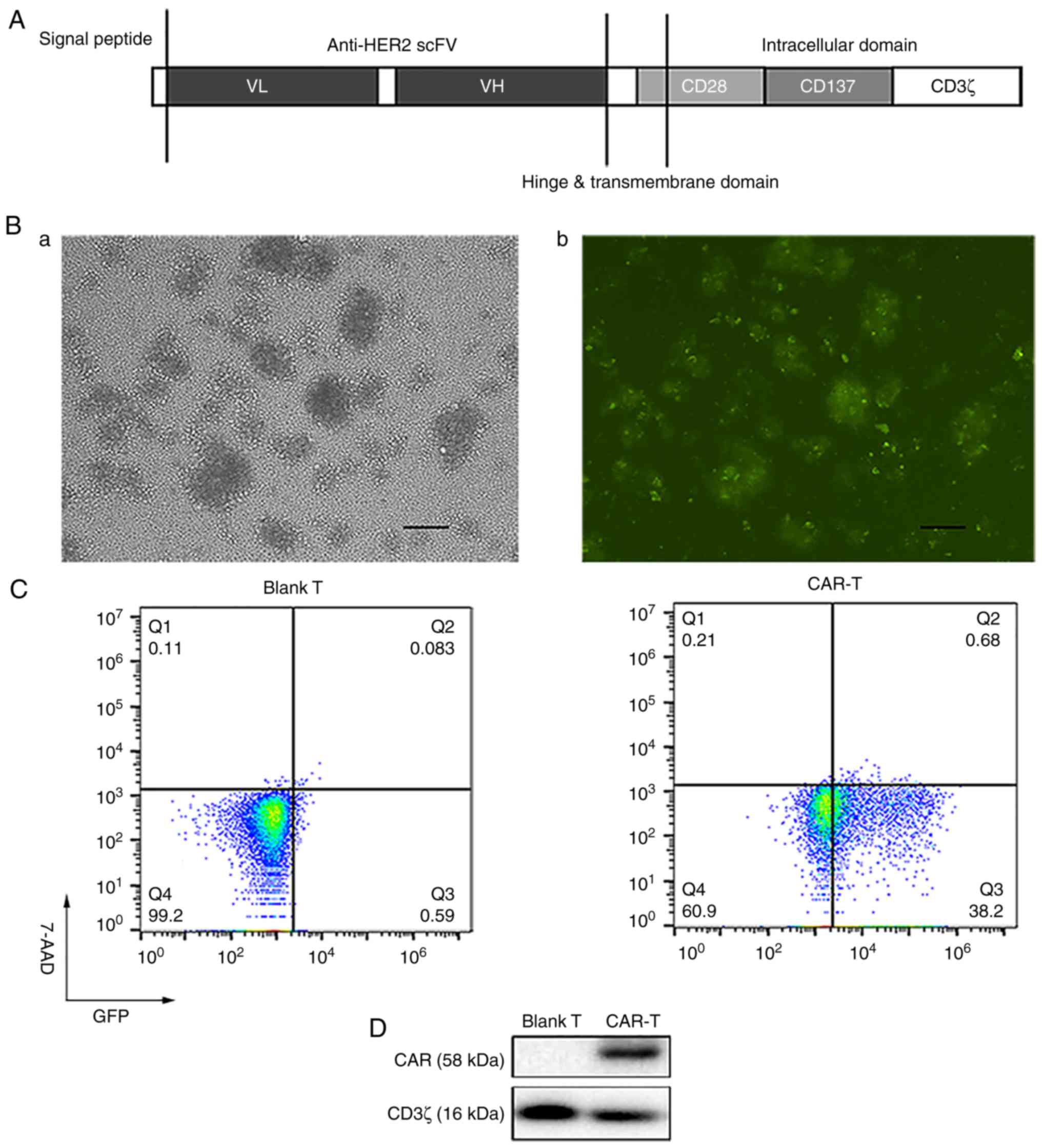
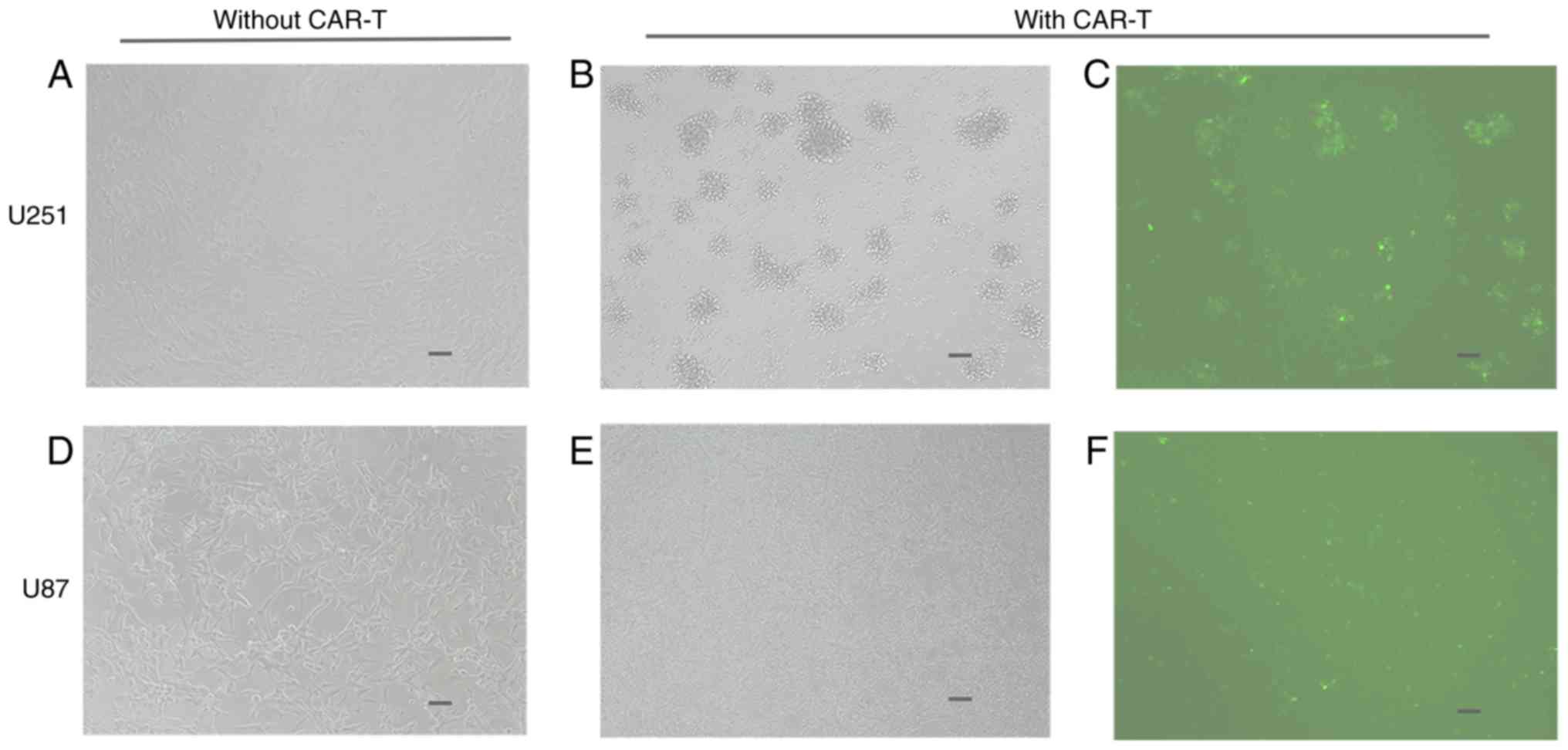
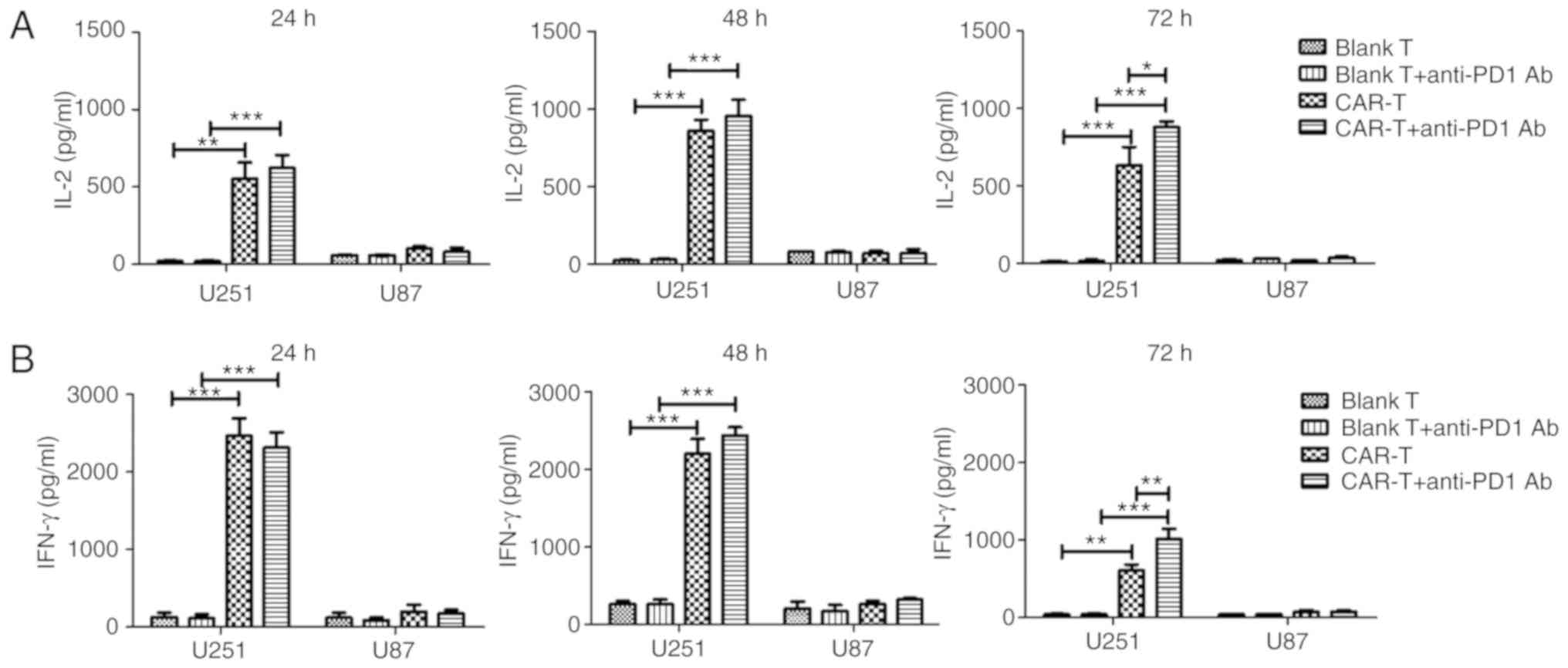
|
1
|
Reardon DA and Mitchell DA: The
development of dendritic cell vaccine-based immunotherapies for
glioblastoma. Semin Immunopathol. 39:225–239. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ohba S and Hirose Y: Current and Future
Drug Treatments for Glioblastomas. Curr Med Chem. 23:4309–4316.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Batash R, Asna N, Schaffer P, Francis N
and Schaffer M: Glioblastoma multiforme, diagnosis and treatment;
recent literature review. Curr Med Chem. 24:3002–3009. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hao L, Li T, Chang LJ and Chen X: Adoptive
immunotherapy for B-cell malignancies using CD19-targeted chimeric
antigen receptor T-cells: A systematic review of efficacy and
safety. Curr Med Chem. Aug 1–2017.(Epub ahead of print). doi:
10.2174/0929867324666170801101842. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jacoby E, Bielorai B, Avigdor A, Itzhaki
O, Hutt D, Nussboim V, Meir A, Kubi A, Levy M, Zikich D, et al:
Locally produced CD19 CAR T cells leading to clinical remissions in
medullary and extramedullary relapsed acute lymphoblastic leukemia.
Am J Hematol. 93:1485–1492. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang D, Shi R, Wang Q and Li J:
Extramedullary relapse of acute lymphoblastic leukemia after
allogeneic hematopoietic stem cell transplantation treated by CAR
T-cell therapy: A case report. Onco Targets Ther. 11:6327–6332.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jena B, Dotti G and Cooper LJ: Redirecting
T-cell specificity by introducing a tumor-specific chimeric antigen
receptor. Blood. 116:1035–1044. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Potti A, Forseen SE, Koka VK, Pervez H,
Koch M, Fraiman G, Mehdi SA and Levitt R: Determination of
HER-2/neu overexpression and clinical predictors of survival in a
cohort of 347 patients with primary malignant brain tumors. Cancer
Invest. 22:537–544. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Koka V, Potti A, Forseen SE, Pervez H,
Fraiman GN, Koch M and Levitt R: Role of Her-2/neu overexpression
and clinical determinants of early mortality in glioblastoma
multiforme. Am J Clin Oncol. 26:332–335. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Seliger B, Rongcun Y, Atkins D, Hammers S,
Huber C, Storkel S and Kiessling R: HER-2/neu is expressed in human
renal cell carcinoma at heterogeneous levels independently of tumor
grading and staging and can be recognized by HLA-A2.1-restricted
cytotoxic T lymphocytes. Int J Cancer. 87:349–359. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang S, Saboorian MH, Frenkel E, Hynan L,
Gokaslan ST and Ashfaq R: Laboratory assessment of the status of
Her-2/neu protein and oncogene in breast cancer specimens:
Comparison of immunohistochemistry assay with fluorescence in situ
hybridisation assays. J Clin Pathol. 53:374–381. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li BT, Ross DS, Aisner DL, Chaft JE, Hsu
M, Kako SL, Kris MG, Varella-Garcia M and Arcila ME: HER2
amplification and HER2 mutation are distinct molecular targets in
lung cancers. J Thorac Oncol. 11:414–419. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Press MF, Cordon-Cardo C and Slamon DJ:
Expression of the HER-2/neu proto-oncogene in normal human adult
and fetal tissues. Oncogene. 5:953–962. 1990.PubMed/NCBI
|
|
14
|
Whilding LM and Maher J: ErbB-targeted CAR
T-cell immunotherapy of cancer. Immunotherapy. 7:229–241. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nowakowska P, Romanski A, Miller N,
Odendahl M, Bonig H, Zhang C, Seifried E, Wels WS and Tonn T:
Clinical grade manufacturing of genetically modified,
CAR-expressing NK-92 cells for the treatment of ErbB2-positive
malignancies. Cancer Immunol Immunother. 67:25–38. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu G, Ying H, Zeng G, Wheeler CJ, Black
KL and Yu JS: HER-2, gp100, and MAGE-1 are expressed in human
glioblastoma and recognized by cytotoxic T cells. Cancer Res.
64:4980–4986. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ahmed N, Salsman VS, Kew Y, Shaffer D,
Powell S, Zhang YJ, Grossman RG, Heslop HE and Gottschalk S:
HER2-specific T cells target primary glioblastoma stem cells and
induce regression of autologous experimental tumors. Clin Cancer
Res. 16:474–485. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Thomas CY, Chouinard M, Cox M, Parsons S,
Stallings-Mann M, Garcia R, Jove R and Wharen R: Spontaneous
activation and signaling by overexpressed epidermal growth factor
receptors in glioblastoma cells. Int J Cancer. 104:19–27. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Andersson U, Guo D, Malmer B, Bergenheim
AT, Brannstrom T, Hedman H and Henriksson R: Epidermal growth
factor receptor family (EGFR, ErbB2-4) in gliomas and meningiomas.
Acta Neuropathol. 108:135–142. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kuramitsu S, Yamamichi A, Ohka F, Motomura
K, Hara M and Natsume A: Adoptive immunotherapy for the treatment
of glioblastoma: Progress and possibilities. Immunotherapy.
8:1393–1404. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tlsty TD and Coussens LM: Tumor stroma and
regulation of cancer development. Annu Rev Pathol. 1:119–150. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Denko NC: Hypoxia, HIF1 and glucose
metabolism in the solid tumour. Nat Rev Cancer. 8:705–713. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zheng Y, Zha Y and Gajewski TF: Molecular
regulation of T-cell anergy. EMBO Rep. 9:50–55. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gajewski TF, Schreiber H and Fu YX: Innate
and adaptive immune cells in the tumor microenvironment. Nat
Immunol. 14:1014–1022. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Al-Zoughbi W, Huang J, Paramasivan GS,
Till H, Pichler M, Guertl-Lackner B and Hoefler G: Tumor
macroenvironment and metabolism. Semin Oncol. 41:281–295. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zou W and Chen L: Inhibitory B7-family
molecules in the tumour microenvironment. Nat Rev Immunol.
8:467–477. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Keir ME, Butte MJ, Freeman GJ and Sharpe
AH: PD-1 and its ligands in tolerance and immunity. Annu Rev
Immunol. 26:677–704. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yuan W, Chen J, Cao Y, Yang L, Shen L,
Bian Q, Bin S, Li P, Cao J, Fang H, et al: Comparative analysis and
optimization of protocols for producing recombinant lentivirus
carrying the anti-Her2 chimeric antigen receptor gene. J Gene Med.
20:e30272018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang C, Hu W, Shen L, Dou R, Zhao S, Shan
D, Yu K, Huang R and Li H: Adoptive antitumor immunotherapy in
vitro and in vivo using genetically activated erbB2-specific T
cells. J Immunother. 37:351–359. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhong XS, Matsushita M, Plotkin J, Riviere
I and Sadelain M: Chimeric antigen receptors combining 4-1BB and
CD28 signaling domains augment PI3kinase/AKT/Bcl-XL activation and
CD8+ T cell-mediated tumor eradication. Mol Ther.
18:413–420. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Brentjens RJ, Santos E, Nikhamin Y, Yeh R,
Matsushita M, La Perle K, Quintas-Cardama A, Larson SM and Sadelain
M: Genetically targeted T cells eradicate systemic acute
lymphoblastic leukemia xenografts. Clin Cancer Res. 13:5426–5435.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Finney HM, Akbar AN and Lawson AD:
Activation of resting human primary T cells with chimeric
receptors: Costimulation from CD28, inducible costimulator, CD134,
and CD137 in series with signals from the TCR zeta chain. J
Immunol. 172:104–113. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Roger A, Finet A, Boru B, Beauchet A,
Mazeron JJ, Otzmeguine Y, Blom A, Longvert C, de Maleissye MF,
Funck-Brentano E and Saiag P: Efficacy of combined
hypo-fractionated radiotherapy and anti-PD-1 monotherapy in
difficult-to-treat advanced melanoma patients. Oncoimmunology.
7:e14421662018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Taquin H, Fontas E, Massol O, Chevallier
P, Balloti R, Beranger G, Lacour JP, Passeron T and Montaudie H:
Efficacy and safety data for checkpoint inhibitors in advanced
melanoma under real-life conditions: A monocentric study conducted
in Nice from 2010 to 2016. Ann Dermatol Venereol. 145:649–658.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang Y, Zhang X, Yang L, Xue J and Hu G:
Blockade of CCL2 enhances immunotherapeutic effect of anti-PD1 in
lung cancer. J Bone Oncol. 11:27–32. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zibetti Dal Molin G, Abrahao CM, Coleman
RL and Maluf FC: Response to pembrolizumab in a heavily treated
patient with metastatic ovarian carcinosarcoma. Gynecol Oncol Res
Pract. 5:62018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li S, Siriwon N, Zhang X, Yang S, Jin T,
He F, Kim YJ, Mac J, Lu Z, Wang S, et al: Enhanced cancer
immunotherapy by chimeric antigen receptor-modified t cells
engineered to secrete checkpoint inhibitors. Clin Cancer Res.
23:6982–6992. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Serganova I, Moroz E, Cohen I, Moroz M,
Mane M, Zurita J, Shenker L, Ponomarev V and Blasberg R:
Enhancement of PSMA-directed CAR adoptive immunotherapy by
PD-1/PD-L1 blockade. Mol Ther Oncolytics. 4:41–54. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liu X, Ranganathan R, Jiang S, Fang C, Sun
J, Kim S, Newick K, Lo A, June CH, Zhao Y and Moon EK: A chimeric
switch-receptor targeting PD1 augments the efficacy of
second-generation car t cells in advanced solid tumors. Cancer Res.
76:1578–1590. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Cherkassky L, Morello A, Villena-Vargas J,
Feng Y, Dimitrov DS, Jones DR, Sadelain M and Adusumilli PS: Human
CAR T cells with cell-intrinsic PD-1 checkpoint blockade resist
tumor-mediated inhibition. J Clin Invest. 126:3130–3144. 2016.
View Article : Google Scholar : PubMed/NCBI
|