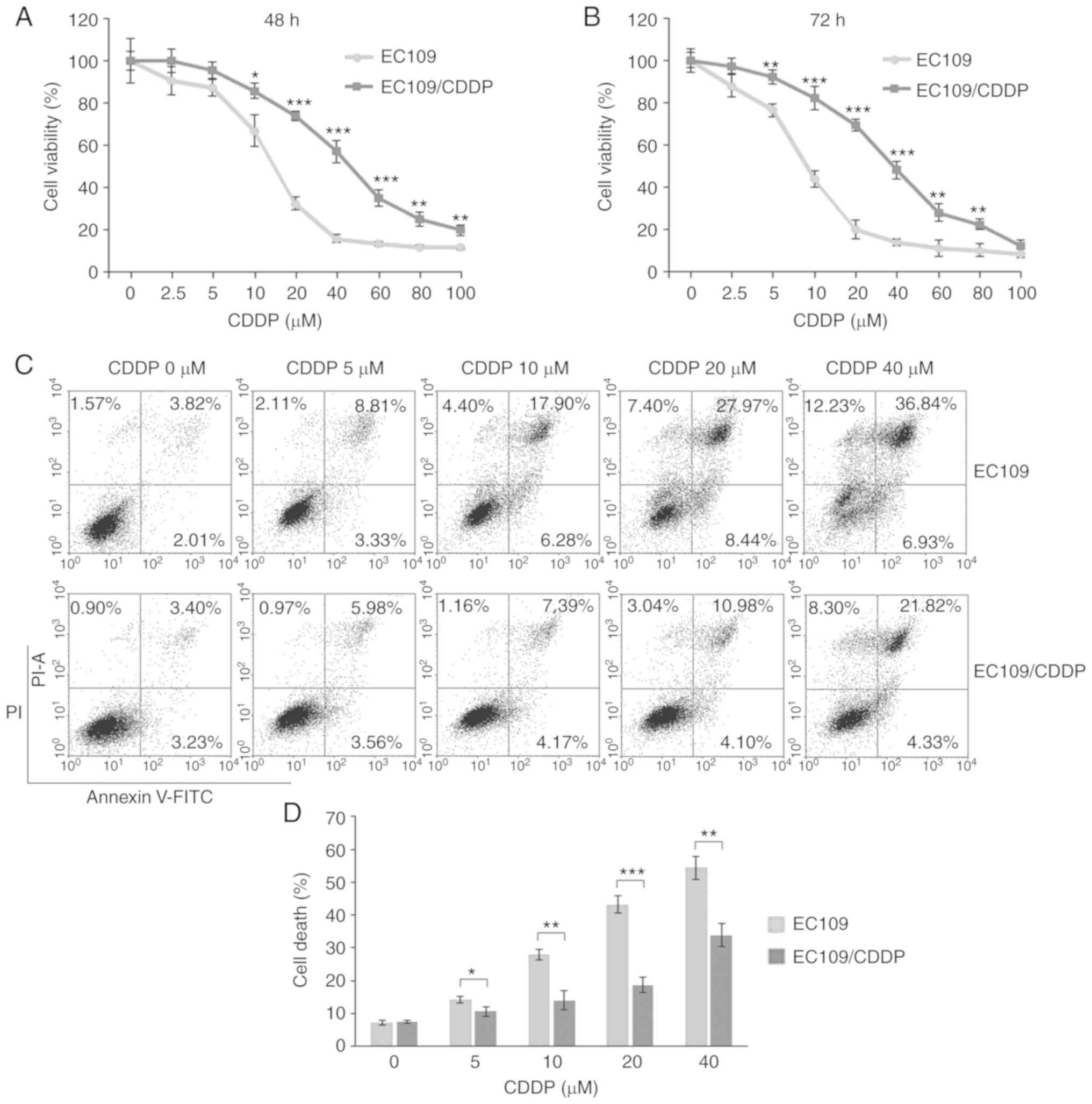
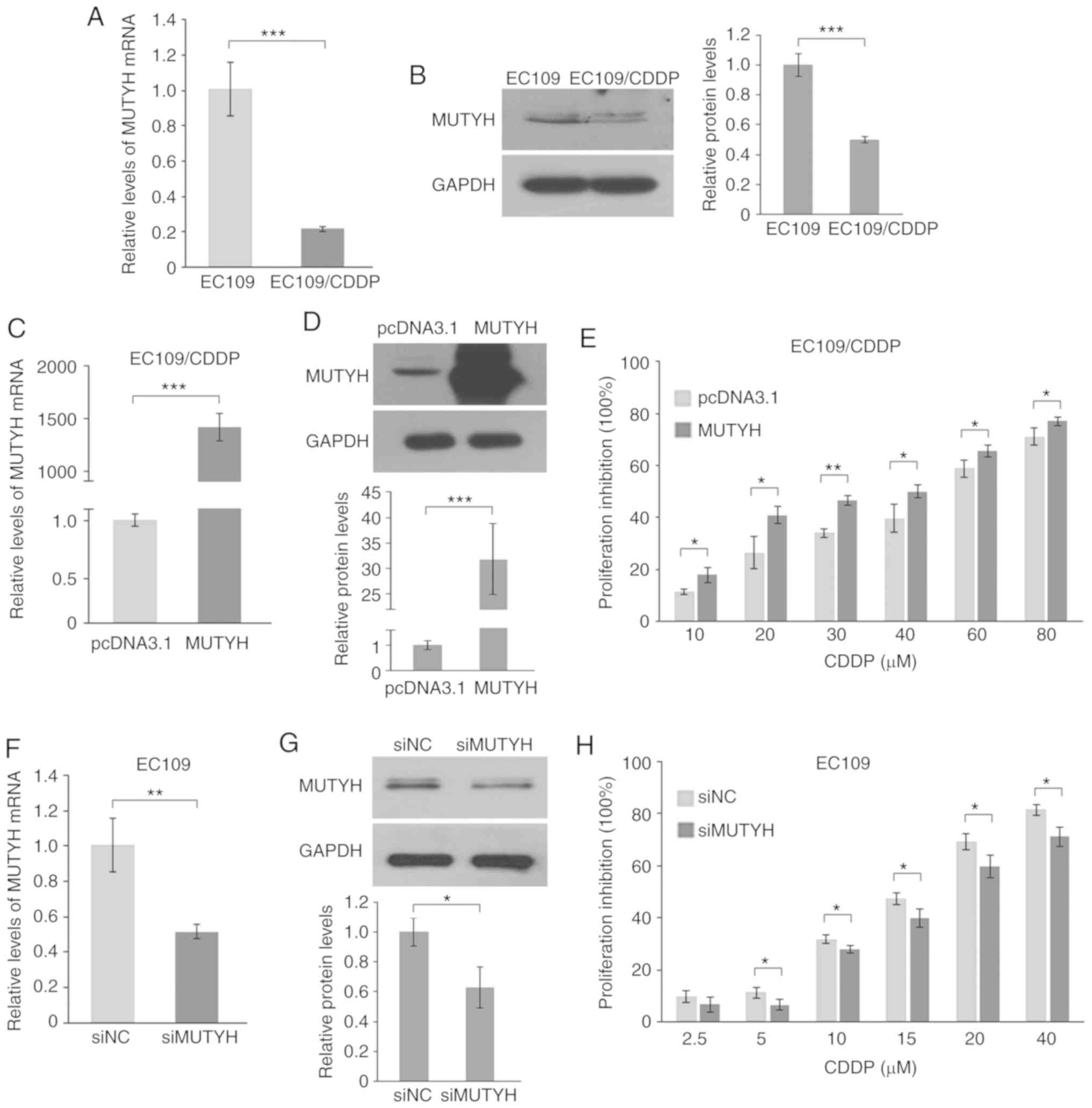
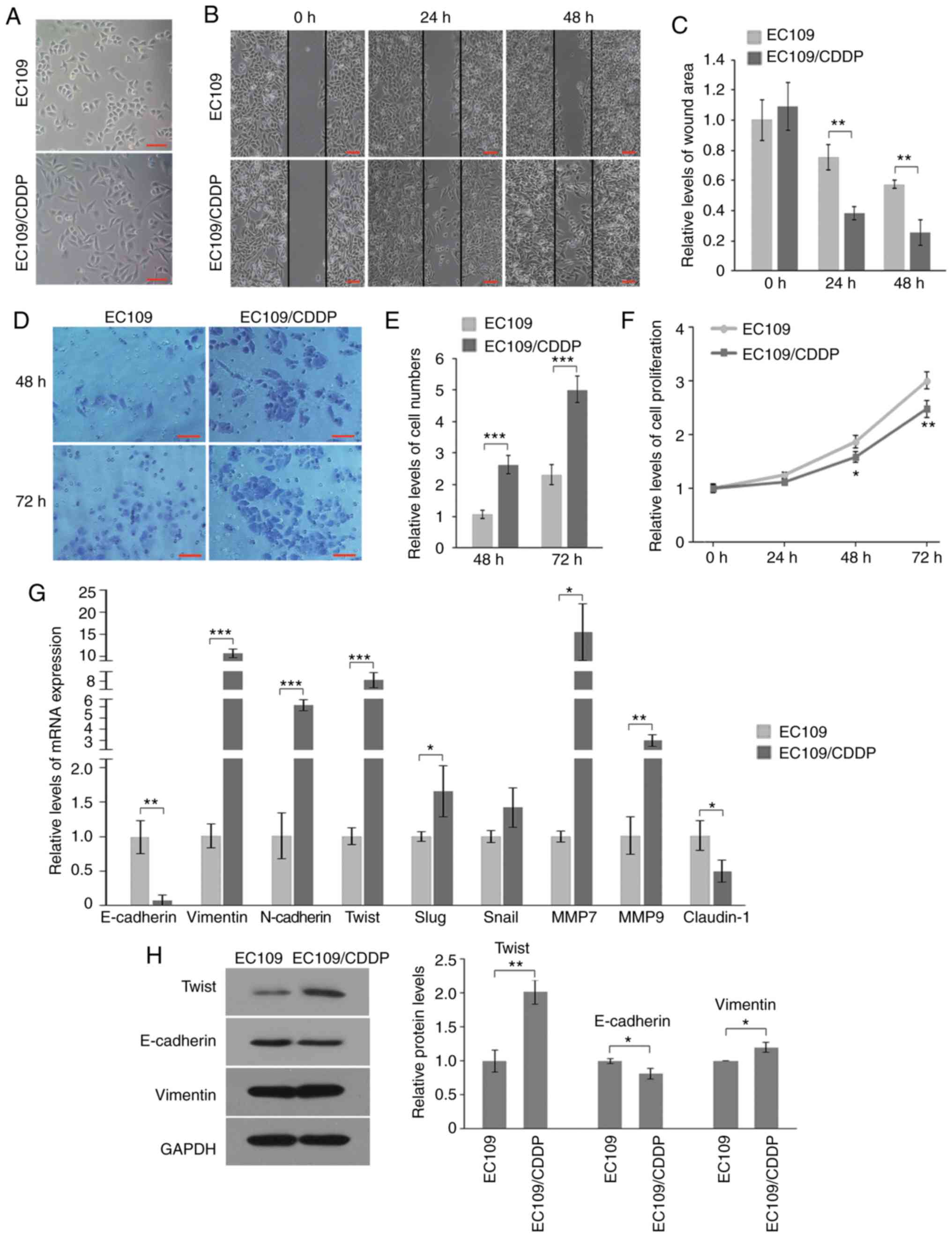
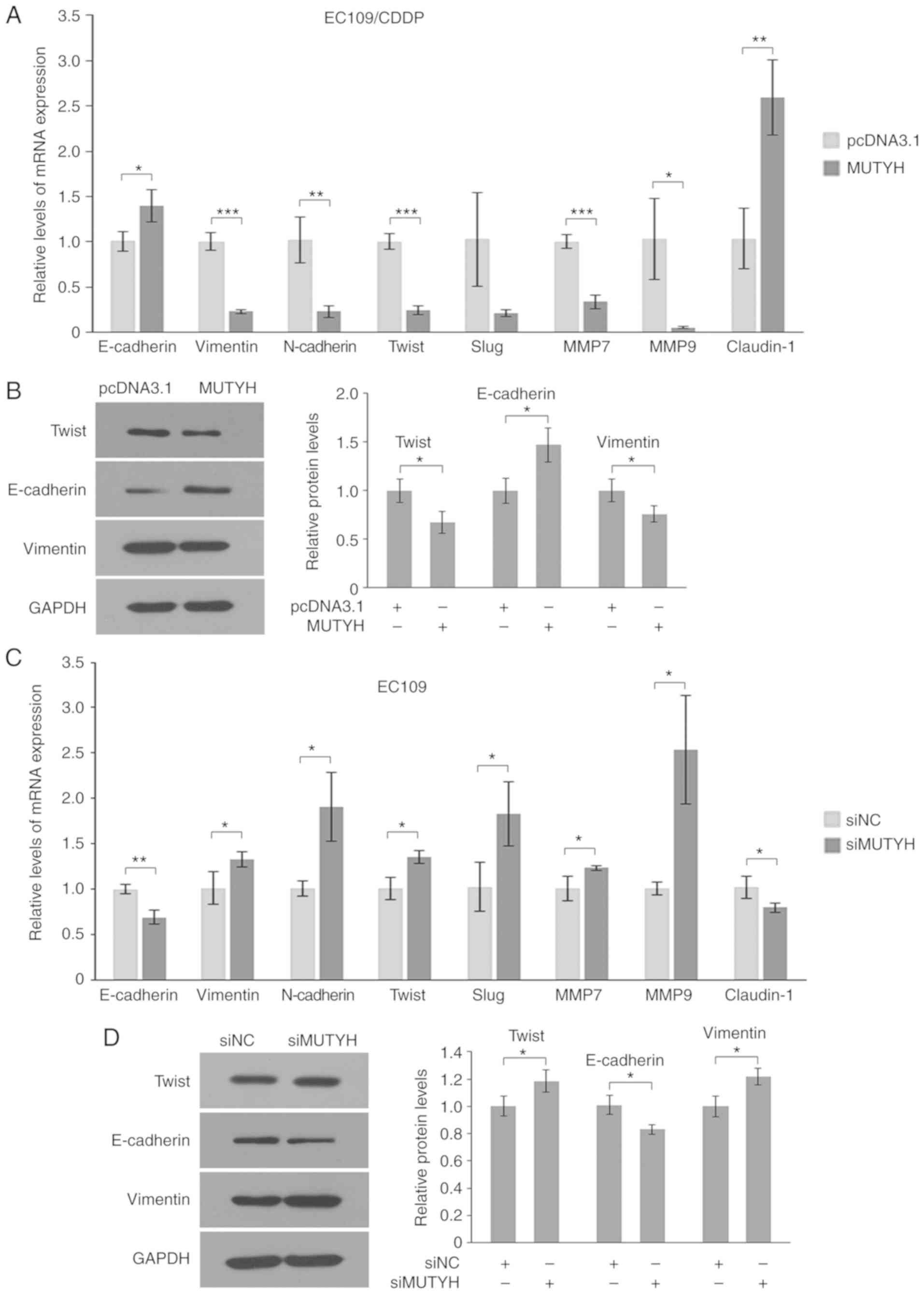
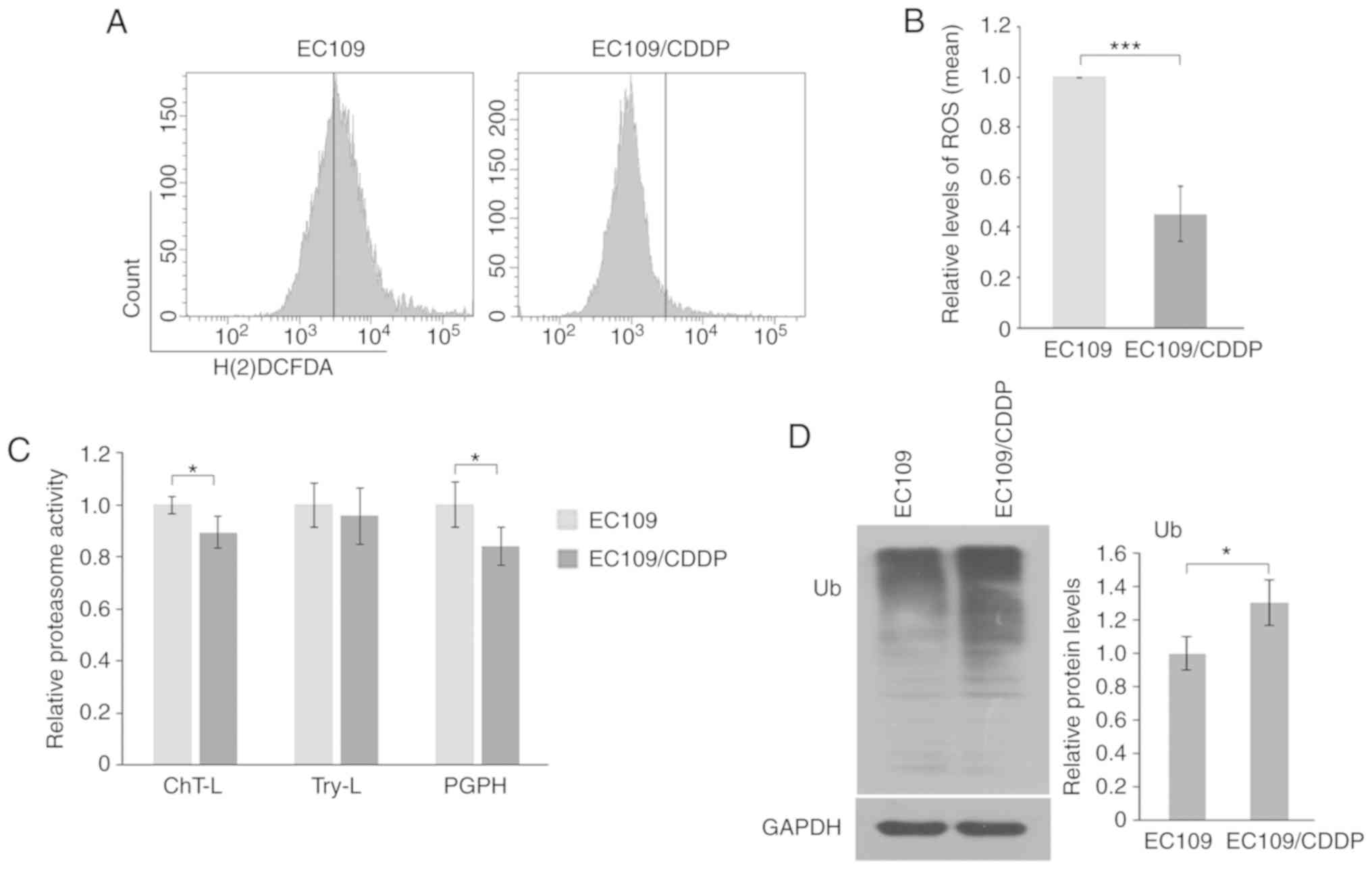
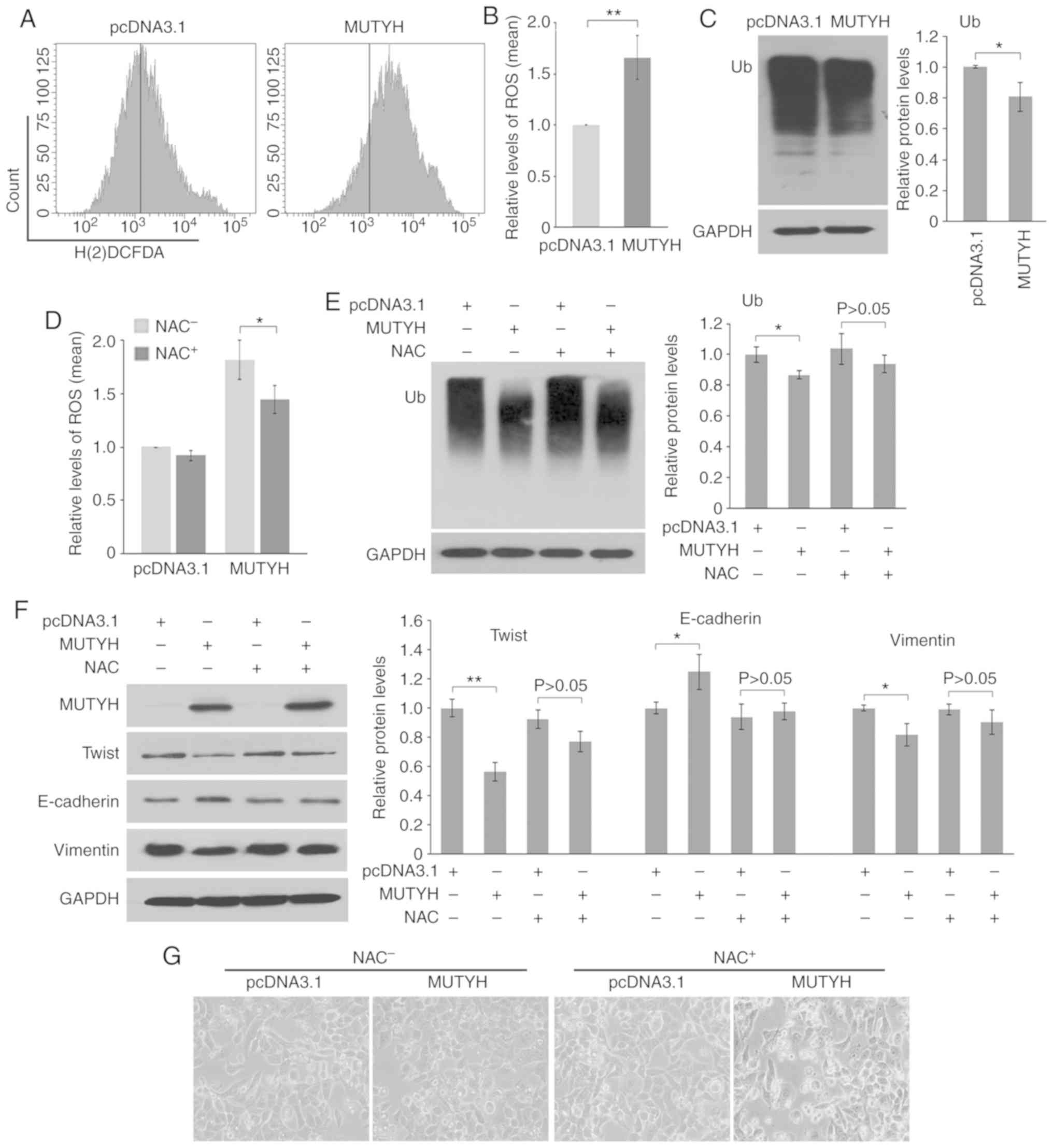
|
1
|
Murphy G, McCormack V, Abedi-Ardekani B,
Arnold M, Camargo MC, Dar NA, Dawsey SM, Etemadi A, Fitzgerald RC,
Fleischer DE, et al: International cancer seminars: A focus on
esophageal squamous cell carcinoma. Ann Oncol. 28:2086–2093. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liu J, Wei Z, Zhang J, Hu W, Ma Z and Liu
Q: Which factors are associated with extremely short-term survival
after surgery in patients with esophageal squamous cell carcinoma?
Asia Pac J Clin Oncol. 12:308–313. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Baba Y, Watanabe M, Yoshida N and Baba H:
Neoadjuvant treatment for esophageal squamous cell carcinoma. World
J Gastrointest Oncol. 6:121–128. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Galluzzi L, Senovilla L, Vitale I, Michels
J, Martins I, Kepp O, Castedo M and Kroemer G: Molecular mechanisms
of cisplatin resistance. Oncogene. 31:1869–1883. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kartalou M and Essigmann JM: Mechanisms of
resistance to cisplatin. Mutat Res. 478:23–43. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
De Koning P, Neijt JP, Jennekens FG and
Gispen WH: Evaluation of cis-diamminedichloroplatinum (II)
(cisplatin) neurotoxicity in rats. Toxicol Appl Pharmacol.
89:81–87. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Marullo R, Werner E, Degtyareva N, Moore
B, Altavilla G, Ramalingam SS and Doetsch PW: Cisplatin induces a
mitochondrial-ROS response that contributes to cytotoxicity
depending on mitochondrial redox status and bioenergetic functions.
PLoS One. 8:e811622013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Santos NA, Catão CS, Martins NM, Curti C,
Bianchi ML and Santos AC: Cisplatin-induced nephrotoxicity is
associated with oxidative stress, redox state unbalance, impairment
of energetic metabolism and apoptosis in rat kidney mitochondria.
Arch Toxicol. 81:495–504. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jiang Y, Guo C, Vasko MR and Kelley MR:
Implications of apurinic/ apyrimidinic endonuclease in reactive
oxygen signaling response after cisplatin treatment of dorsal root
ganglion neurons. Cancer Res. 68:6425–6434. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Markkanen E, Dorn J and Hübscher U: MUTYH
DNA glycosylase: The rationale for removing undamaged bases from
the DNA. Front Genet. 4:182013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ruggieri V, Pin E, Russo MT, Barone F,
Degan P, Sanchez M, Quaia M, Minoprio A, Turco E, Mazzei F, et al:
Loss of MUTYH function in human cells leads to accumulation of
oxidative damage and genetic instability. Oncogene. 32:4500–4508.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nascimento EFR, Ribeiro ML, Magro DO,
Carvalho J, Kanno DT, Martinez CAR and Coy CSR: Tissue expresion of
the genes MUTYH and OGG1 in patients with sporadic colorectal
cancer. Arq Bras Cir Dig. 30:98–102. 2017.(In English, Portuguese).
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dong J, Wang X, Yu Y, Yan X and Cui JW:
Association of base excision repair gene polymorphisms with the
response to chemotherapy in advanced non-small cell lung cancer.
Chin Med J (Engl). 131:1904–1908. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Santos LS, Branco SC, Silva SN, Azevedo
AP, Gil OM, Manita I, Ferreira TC, Limbert E, Rueff J and Gaspar
JF: Polymorphisms in base excision repair genes and thyroid cancer
risk. Oncol Rep. 28:1859–1868. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kong F, Han XY, Luan Y, Qi TG, Sun C, Wang
J, Hou HY, Jiang YH, Zhao JJ and Cheng GH: MUTYH Association with
esophageal adenocarcinoma in a Han Chinese population. Asian Pac J
Cancer Prev. 14:6411–6413. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sliwinski T, Markiewicz L, Rusin P,
Pietruszewska W, Olszewski J, Morawiec-Sztandera A, Mlynarski W and
Majsterek I: Polymorphisms of the DNA base excision repair gene
MUTYH in head and neck cancer. Exp Oncol. 31:57–59. 2009.PubMed/NCBI
|
|
17
|
Przybylowska K, Kabzinski J, Sygut A,
Dziki L, Dziki A and Majsterek I: An association selected
polymorphisms of XRCC1, OGG1 and MUTYH gene and the level of
efficiency oxidative DNA damage repair with a risk of colorectal
cancer. Mutat Res. 745-746:6–15. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Al-Tassan N, Chmiel NH, Maynard J, Fleming
N, Livingston AL, Williams GT, Hodges AK, Davies DR, David SS,
Sampson JR and Cheadle JP: Inherited variants of MYH associated
with somatic G:C->T:A mutations in colorectal tumors. Nat Genet.
30:227–232. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Slyskova J, Naccarati A, Pardini B,
Polakova V, Vodickova L, Smerhovsky Z, Levy M, Lipska L, Liska V
and Vodicka P: Differences in nucleotide excision repair capacity
between newly diagnosed colorectal cancer patients and healthy
controls. Mutagenesis. 27:225–232. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu YQ, Wang SK, Xu QQ, Yuan HQ, Guo YX,
Wang Q, Kong F, Lin ZM, Sun DQ, Wang RM and Lou HX:
Acetyl-11-keto-β-boswellic acid suppresses docetaxel-resistant
prostate cancer cells in vitro and in vivo by blocking Akt and
Stat3 signaling, thus suppressing chemoresistant stem cell-like
properties. Acta Pharmacol Sin. 40:689–698. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Smith BN and Bhowmick NA: Role of EMT in
metastasis and therapy resistance. J Clin Med. 5:E172016.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zheng X, Carstens JL, Kim J, Scheible M,
Kaye J, Sugimoto H, Wu CC, LeBleu VS and Kalluri R:
Epithelial-to-mesenchymal transition is dispensable for metastasis
but induces chemoresistance in pancreatic cancer. Nature.
527:525–530. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shang F and Taylor A: Ubiquitin–proteasome
pathway and cellular responses to oxidative stress. Free Radic Biol
Med. 51:5–16. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dudek EJ, Shang F, Valverde P, Liu Q,
Hobbs M and Taylor A: Selectivity of the ubiquitin pathway for
oxidatively modified proteins: Relevance to protein precipitation
diseases. FASEB J. 19:1707–1709. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shibue T and Weinberg RA: EMT, CSCs, and
drug resistance: The mechanistic link and clinical implications.
Nat Rev Clin Oncol. 14:611–629. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Saxena M, Stephens MA, Pathak H and
Rangarajan A: Transcription factors that mediate
epithelial-mesenchymal transition lead to multidrug resistance by
upregulating ABC transporters. Cell Death Dis. 2:e1792011.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Díaz VM, Viñas-Castells R and García de
Herreros A: Regulation of the protein stability of EMT
transcription factors. Cell Adh Migr. 8:418–428. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Goldberg AL: Protein degradation and
protection against misfolded or damaged proteins. Nature.
426:895–899. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Glickman MH and Ciechanover A: The
ubiquitin–proteasome proteolytic pathway: Destruction for the sake
of construction. Physiol Rev. 82:373–428. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shang F, Gong X and Taylor A: Activity of
ubiquitin-dependent pathway in response to oxidative stress.
Ubiquitin-activating enzyme is transiently upregulated. J Biol
Chem. 272:23086–23093. 1997. View Article : Google Scholar : PubMed/NCBI
|