Introduction
Cervical cancer is one of the most common
gynecologic malignancies and the fourth leading cause of
cancer-related deaths in women (1).
It is estimated that there were 570,000 cases and 311,000 deaths
worldwide in 2018 (1). Early
screening, surgery, and radiotherapy have significantly improved
the prognosis of patients with cervical cancer; however, tumor
metastasis, which is a major cause of death from cervical cancer,
can still not be completely prevented (2). Therefore, studying the mechanism of
cervical cancer metastasis and understanding the driving factors
behind it are crucial for the development of reasonable
interventions to improve patient prognosis.
Autophagy is an evolutionarily ancient mechanism
which degrades redundant or potentially harmful cytosolic entities
to maintain stable cell metabolism (3). While the role of autophagy in
tumorigenesis is dual and context-specific (4,5),
increasing evidence suggests that autophagy is involved in the
metastasis of tumor cells (6–8). The
role of autophagy in tumors remains unclear, and further
investigation is required.
Kindlin-2 (also known as FERMT2 or MIG-2) is a
member of the kindlin family, which consists of three members:
Kindlin-1, −2, and −3. Members of this family generally contain F1,
F2, and F3 subdomains and characteristically harbor a pleckstrin
homeodomain in the F2 subdomain (9). Kindlin-2 has been shown to be involved
in the progression of pancreatic cancer, breast cancer, and glioma
(10–12). However, to our knowledge, the role
of Kindlin-2 in cervical cancer has not been reported. Moreover,
the role of Kindlin-2 in autophagy is unknown. Autophagy is a
complex process that involves several signaling pathways. AKT/mTOR
is a main pathway regulating cell autophagy, and activation of this
pathway inhibits autophagy (13,14).
However, whether the AKT/mTOR pathway mediates autophagy regulation
by kindlin-2 remains to be studied.
In this study, we compared Kindlin-2 expression in
cervical cancer tissues and healthy cervical tissues. Further, we
found that Kindlin-2 acts as a novel autophagy regulator that
regulates the AKT/mTOR pathway, a major autophagy pathway. In
addition, we found that Kindlin-2 inhibits the migration of
cervical cancer cells by inducing autophagy. Kindlin-2 may be a
tumor migration suppressor gene, and has potential as a tumor
marker and therapeutic target in cervical cancer.
Materials and methods
Clinical tissue specimens
Normal tissue adjacent to the tumor (NAT), which is
usually used as a control in tumor studies, should be collected
>2 cm from the tumor margin (15). In the present study, given the small
size of the cervix and the differences in gene expression between
NAT and normal tissues (15),
normal cervical tissue was used as a control. Forty-two cervical
cancer tissue samples were obtained from patients (age range, 35–70
years; mean age, 51.8±8.7 years) with cervical cancer diagnosed by
histopathology, and 24 normal cervical tissue samples were obtained
from patients with benign uterine lesions, and all patients
underwent hysterectomy at the Guangxi Medical University Cancer
Hospital between April 2017 and September 2017. The study was
approved by the Ethics Committee of Guangxi Medical University
Cancer Hospital, and all patients signed informed consent to
participate in the study.
Cell culture and transfection
The SiHa human cervical cancer cell line was
purchased from Shanghai GeneChem Co., Ltd., and CaSki and C-33A
human cervical cancer cells were purchased from Zhongqiaoxinzhou
Biotech. The cells were cultured in Dulbecco's modified Eagle's
medium (Gibco; Thermo Fisher Scientific, Inc.) containing 10% fetal
bovine serum (FBS) and 100 U/ml penicillin-streptomycin solution
(Beyotime Institute of Biotechnology) in a humidified 5%
CO2 atmosphere at 37°C. To observe the effect of
kindlin-2 expression in cervical cancer cells, SiHa and CaSki cells
were transfected with a lentivirus encoding full-length human
kindlin-2 cDNA or carrying a short hairpin (sh)RNA targeting
kindlin-2, constructed by Shanghai GeneChem Co., Ltd. The siRNA
sequence was as follows: 5′-GCGGACAGTTCTTACAACTTA-3′. Empty
lentiviral vector and lentivirus containing nonsense shRNA
sequences were used as negative controls.
Reagents and antibodies
Earle's balanced salts solution (EBSS; product no.
E2888) was purchased from Sigma-Aldrich (Merck KGaA).
3-Methyladenine (3-MA) and rapamycin were purchased from Shanghai
GeneChem Co., Ltd. The AKT inhibitor MK2206 and the AKT activator
SC79 were purchased from Beyotime Institute of Biotechnology.
Primary antibodies against LC3 A/B (product no. 12741; dilution
1:1,000), SQSTM1/p62 (product no. 88588; dilution 1:1,000),
phospho-mTOR (product no. 5536; dilution 1:1,000) and GAPDH
(product no. 97166; dilution 1:1,000) were obtained from Cell
Signaling Technology, Inc. Antibodies targeting kindlin-2 (cat. no.
11453-1-AP; dilution 1:2,000), mTOR (cat. no. 66888-1-Ig; dilution
1:10,000), phospho-AKT (cat. no. 66444-1-Ig; dilution 1:3,000) were
purchased from ProteinTech Group, Inc. and AKT antibody (cat. no.
WL0003b; dilution 1:500) was purchased from Wanleibio Co., Ltd.
Transwell migration assay
For the Transwell cell migration assay,
3×104 SiHa cells or 6×104 CaSki cells were
seeded in 200 µl of serum-free medium in the upper chamber, whereas
600 µl of medium containing 20% FBS was added to the lower chamber.
After incubation for 16 h, the cells on the upper insert surface
were removed with a cotton swab. Cells that had successfully
migrated to the lower surface of the insert were stained with 0.1%
crystal violet at room temperature for 10 min. Cells were counted
in five randomly selected high-power fields using a microscope
(×20, magnification; IX71; Olympus Corporation).
Wound healing assay
SiHa and CaSki cell migration was evaluated by a
wound healing assay. When cells reached 100% confluence, they were
treated with mitomycin C (5 µg/ml; cat. no. M4287; Sigma-Aldrich;
Merck KGaA) for 1 h. Then, the cell monolayer was scratched using a
200-µl pipette tip, washed with PBS, and supplemented with
serum-free medium. The plates were further incubated and images
were captured using a microscope at 0, 24 and 48 h after treatment.
Migration ability was assessed by measuring the gap area using the
ImageJ software (version 1.52a; NIH).
Western blotting
Cells were lysed using RIPA lysis buffer
supplemented with 0.1 mM PMSF. The proteins were quantified using
BCA protein assay kit. Next, 30 µg total protein were separated by
8–12% sodium dodecyl sulfate polyacrylamide gel electrophoresis and
transferred onto a polyvinylidene fluoride membrane. The membrane
was blocked using 5% skimmed milk in Tris-buffered saline
containing 0.1% Tween-20 at room temperature for 1 h. The membrane
was incubated with a primary antibody at 4°C overnight, and then
with horseradish peroxidase (HRP)-conjugated anti-rabbit (cat. no.
A0208) or anti-mouse (cat. no. A0216) secondary antibodies
(dilution 1:1,000; Beyotime Institute of Bioetchnology) at room
temperature for 1 h. Specific bands were detected by
chemiluminescence using ECL detection reagent (Biokits Tech, Inc.).
GAPDH was probed to serve as a control. The protein expression
levels were quantified using Image Lab software (version 6.0;
Bio-Rad Laboratories, Inc.).
Autophagic flux analysis
To detect autophagic flux, SiHa and CaSki cells were
transfected with mRFP-GFP-LC3 lentivirus (Shanghai GeneChem Co.,
Ltd.). After 72 h, the cells were seeded in confocal dishes, and
GFP and RFP puncta were observed using a laser confocal microscope
(TCS SP8; Leica Microsystems GmbH). In merged images, yellow spots
indicated autophagosomes and red spots indicated autolysosomes. In
each group, 30 cells were randomly selected for counting.
Immunohistochemistry
Formalin-fixed, paraffin-embedded tissues were cut
into 4-µm sections. Tissue sections were deparaffinized with xylene
and rehydrated, subjected to antigen retrieval in EDTA (pH 9.0),
and incubated in a 3% hydrogen peroxide solution to block
endogenous peroxidase at room temperature for 25 min. The sections
were blocked with 3% bovine serum albumin (cat. no. G5001;
Servicebio, Wuhan, China) at room temperature for 30 min, incubated
with kindlin-2 antibody (cat. no. 11453-1-AP; dilution 1:100;
ProteinTech Group, Inc.) at 4°C overnight, washed with PBS, and
incubated with HRP-conjugated secondary antibody (cat. no. A0208;
dilution 1:50; Beyotime Institute of Biotechnology) at room
temperature for 50 min. Then, the sections were immersed in
3,3′-diaminobenzidine for color development. Nuclei were
counterstained with hematoxylin at room temperature for 3 min.
Finally, the tissue sections were dehydrated and mounted. The
images were captured using a light microscope (XSP-C204; Chongqing
Optical Instrument Co., Ltd.). Kindlin-2 expression was measured
based on the average optical density, using ImageJ version
1.52a.
Bioinformatics and statistical
analysis
RNA-seq data and survival data for patients with
cervical cancer were downloaded from the Gene Expression Omnibus
(GEO) database (GSE44001 and GSE52904) (16,17)
and Gene Expression Profiling Interactive Analysis (GEPIA), an
online analysis tool based on The Cancer Genome Atlas (TCGA) and
the Genotype-Tissue Expression (GTEx) database (18). R (version 3.5.3) and related
packages were used for statistical analysis. Evaluate Cutpoints
(http://wnbikp.umed.lodz.pl/Evaluate-Cutpoints/) was
used to determine the optimal cut-off for survival analysis
(19). All data are reported as the
mean ± SD. Student's t-test was used to compare means
between two groups. For multiple comparisons, ANOVA and Dunnett's
post hoc analysis were used. Disease-free survival time was
analyzed using Kaplan-Meier curves, and any differences in survival
were evaluated with a log-rank test. Differences were considered
statistically significant at P<0.05.
Results
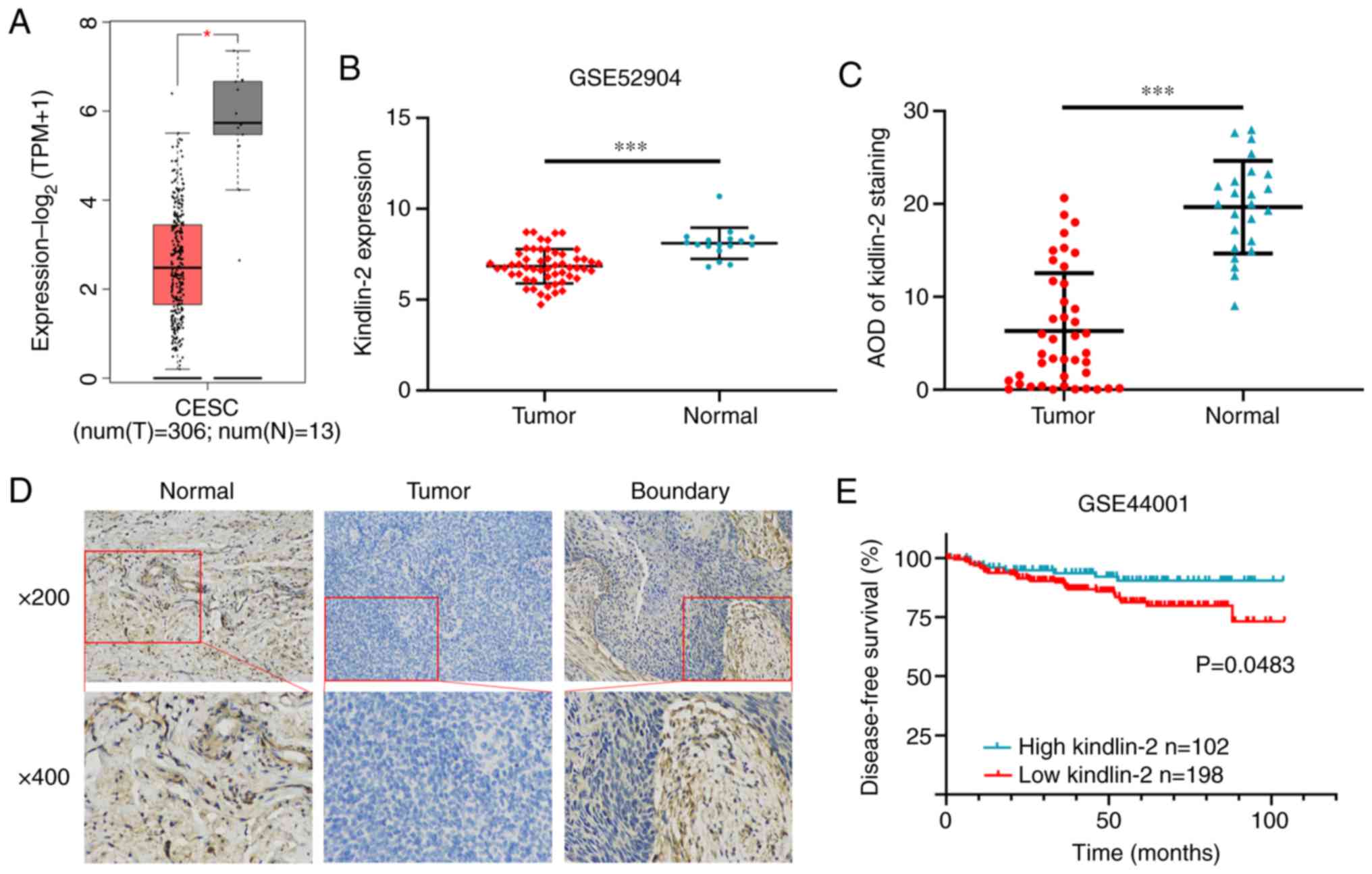
Kindlin-2 is downregulated in cervical
cancer, and low Kindlin-2 expression is correlated with poor
disease-free survival
First, it was determined whether kindlin-2 is
differentially expressed between healthy cervical tissues and
cervical cancer tissues. To this end, we used Gene Expression
Profiling Interactive Analysis (GEPIA) tools to analyze kindlin-2
mRNA expression in the TCGA and GTEx databases, including 306
cervical cancer tissues and 13 healthy cervical tissues. The
results revealed that kindlin-2 expression was significantly
downregulated in cervical cancer tissues (P<0.05, Fig. 1A). In addition, 55 cervical cancer
tissues and 17 healthy cervical tissues in the GSE52904 dataset
(GPL 6244) were analyzed, and similar results were obtained
(P<0.001, Fig. 1B). To validate
the expression of kindlin-2 in clinical specimens, the expression
of kindlin-2 in 42 cervical cancer tissues and 24 healthy cervical
tissues was detected using immunohistochemistry. The results
revealed that kindlin-2 expression in cancer tissues was lower than
that in healthy tissues (P<0.001, Fig. 1C and D). Kaplan-Meier survival
analysis was performed using the GSE44001 dataset in the GEO
database. The results revealed that disease-free survival was
shorter in the low-kindlin-2 expression group than in the
high-expression group (P<0.05, Fig.
1E).
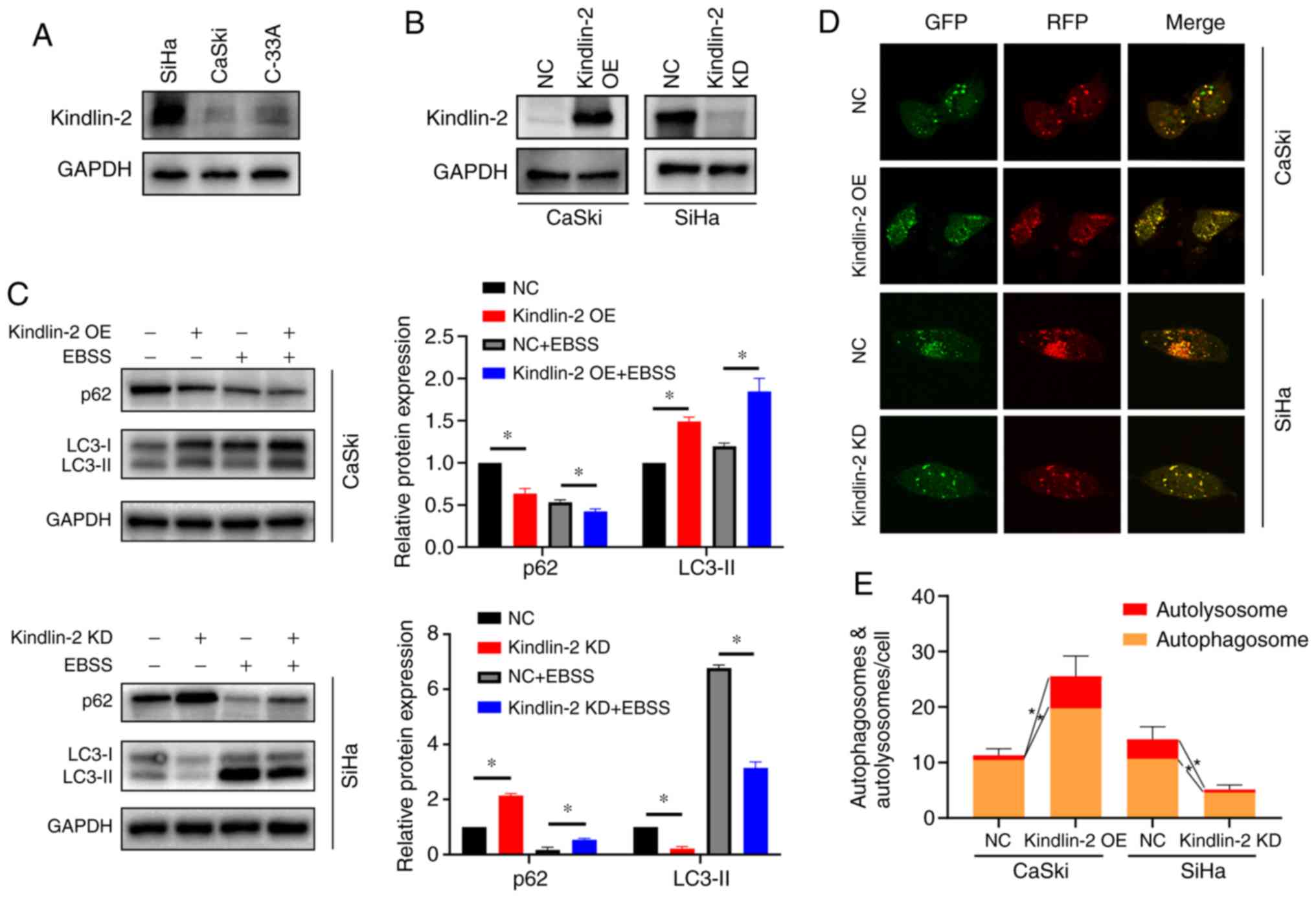
Kindlin-2 induces autophagy of
cervical cancer cells
To explore the role of kindlin-2 in cervical cancer
cells, kindlin-2 expression was assessed in three cervical cancer
cell lines. Kindlin-2 expression was the highest in SiHa cells and
the lowest in CaSki cells (Fig.
2A). Therefore, kindlin-2 was overexpressed and knocked down in
CaSki and SiHa cells, respectively (Fig. 2B). To explore the role of kindlin-2
in autophagy, autophagic protein marker expression was first
assessed using immunoblotting. p62/SQSTM1 and LC3 are considered to
play important roles in autophagy (20,21).
The amount of LC3-II is generally associated with the number of
autophagosomes (22). p62 is a
selective autophagy substrate and thus, p62 protein levels are
generally inversely correlated with the level of autophagy
(22). In CaSki cells
overexpressing kindlin-2, the p62 and LC3-II levels were decreased
and increased, respectively, whereas in kindlin-2-knockdown SiHa
cells, p62 and LC3-II levels were increased and decreased,
respectively, compared to the NC group. To verify whether kindlin-2
promotes starvation-induced autophagy, cells were cultured with
EBSS-containing medium for 6 h to induce autophagy. The results
indicated that kindlin-2 enhanced the level of LC3-II and
suppressed p62 accumulation under starvation compared to the
NC+EBSS group in CaSki cells while the opposite results were
obtained in SiHa cells (Fig.
2C).
Autophagy is a dynamic process. Thus, an increase in
autophagosomes (as indicated by an increase in the autophagosome
marker LC3-II) may indicate an increase in autophagy, blockage of
autophagolysosome formation, or both (22). Therefore, to determine the cause of
autophagosome accumulation, we transfected cells with an
mRFP-GFP-LC3 tandem construct to detect autophagic flux based on
the fact that in the acidic environment of autophagolysosomes, GFP
fluorescence is quenched while RFP fluorescence remains stable. The
results revealed that the numbers of yellow spots (GFP+
and RFP+, indicating autophagosomes) and red spots
(GFP– and RFP+, indicating autolysosomes)
were increased in kindlin-2-overexpressing CaSki cells and
decreased in kindlin-2-knockdown SiHa cells when compared with the
respective controls, indicating that kindlin-2 in fact promoted
autophagic flux and thus increased the number of autophagosomes
(P<0.05, Fig. 2D and E).
Kindlin-2 inactivates AKT/mTOR
signaling
The AKT/mTOR pathway is the main pathway associated
with autophagy (23,24). Therefore, it was surmised that
kindlin-2 may promote autophagy by inactivating the AKT/mTOR
signaling pathway. To test this hypothesis, the expression of
AKT/mTOR-related proteins was examined by western blotting.
Phosphorylated AKT and phosphorylated mTOR were significantly
decreased and increased in kindlin-2-overexpressing CaSki cells and
kindlin-2-knockdown SiHa cells, respectively, when compared with
the respective control cells. Total AKT and total mTOR levels were
not affected (Fig. 3A). These
findings indicated that kindlin-2 inactivated AKT/mTOR
signaling.
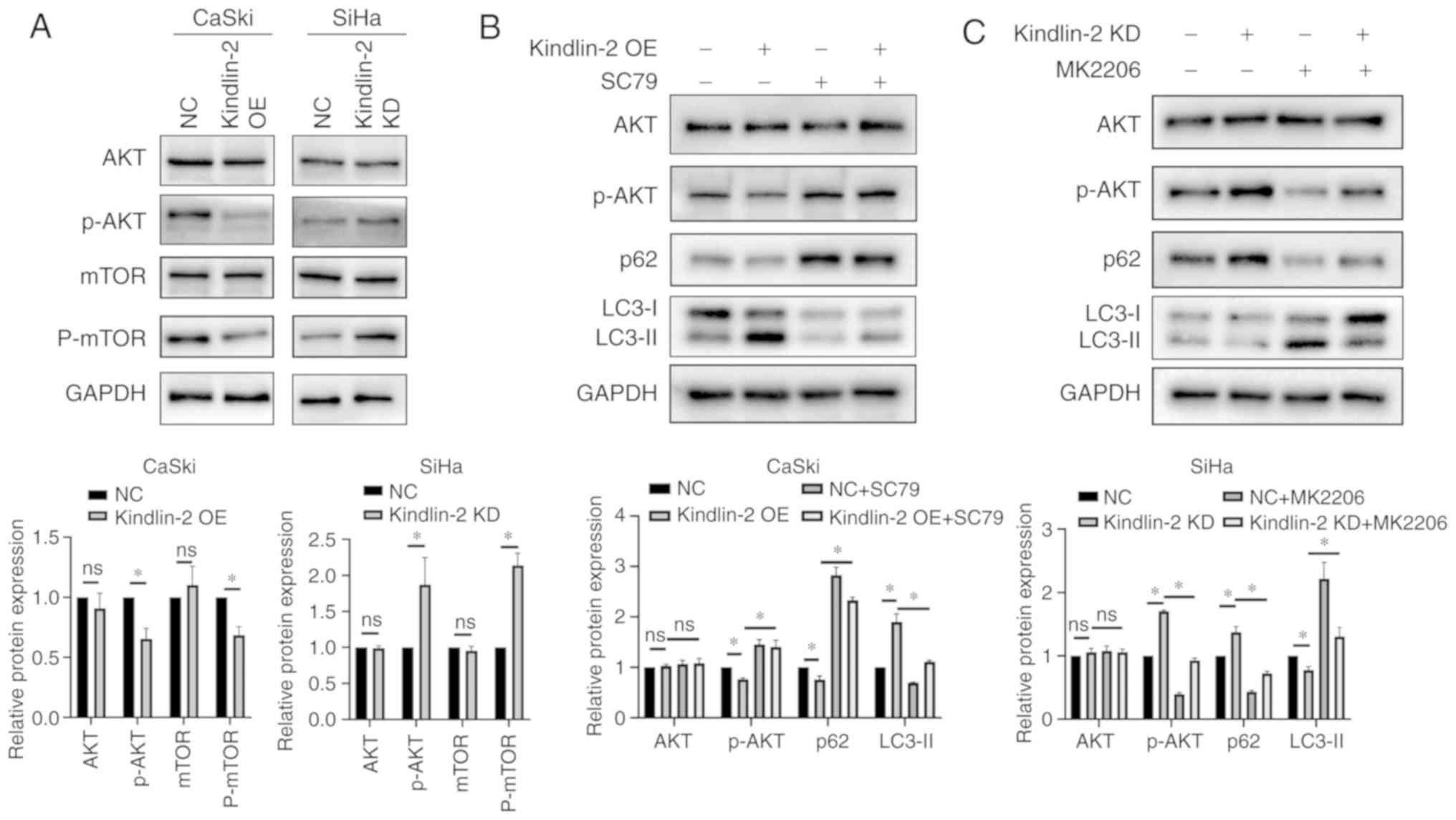 | Figure 3.Kindlin-2 induces autophagy by
inactivating the AKT/mTOR pathway. (A) Protein levels of AKT,
p-AKT, mTOR, p-mTOR, and GAPDH in the indicated CaSki and SiHa
cells as detected by western blotting. (B) The indicated CaSki
cells were treated with SC79 (20 µM) for 24 h and the protein
levels of AKT, p-AKT, p62, LC3, and GAPDH were determined by
western blotting. (C) The indicated SiHa cells were treated with
MK2206 (5 µM) for 24 h and protein levels of AKT, p-AKT, p62, LC3
and GAPDH were detected by western blotting. *P<0.05. NC,
negative control; OE, overexpression; KD, knockdown; ns, not
significant. |
Kindlin-2 induces autophagy via the
AKT/mTOR pathway
To confirm whether kindlin-2 induces autophagy via
the AKT/mTOR pathway, kindlin-2-overexpressing CaSki cells were
treated with the AKT activator SC79 (20 µM) and then autophagy
marker expression was assessed. The autophagy of the kindlin-2
OE+SC79 group was significantly attenuated as compared to the
kindlin-2 OE group, demonstrating that this activator can attenuate
autophagy induction mediated by kindlin-2 overexpression (Fig. 3B). In addition, the AKT inhibitor
MK2206 (5 µM) was added to kindlin-2-knockdown SiHa cells. The
autophagy of the kindlin-2 KD+MK2206 group was significantly
promoted when compared with kindlin-2 KD group, demonstrating that
this inhibitor can reverse the inhibition of autophagy in
Kindlin-2-knockdown SiHa cells (Fig.
3C). Collectively, these results indicated that kindlin-2
regulates autophagy via inhibition of AKT/mTOR signaling.
Kindlin-2 inhibits cervical cancer
cell migration by promoting autophagy
Wound healing assays revealed that wound closure was
slower in kindlin-2-overexpressing CaSki cells than in
Lv-NC-infected CaSki cells (Fig. 4A and
B), whereas the opposite results were observed in
kindlin-2-knockdown SiHa cells (Fig. 4C
and D). Transwell assays revealed that kindlin-2 overexpression
suppressed migration in CaSki cells, whereas kindlin-2 knockdown
enhanced migration in SiHa cells when compared with the respective
controls (Fig. 4E-G). To
investigate whether autophagy is involved in the regulatory effect
of kindlin-2 on cell migration, the autophagy inhibitor 3-MA (5 mM)
was added to kindlin-2-overexpressing CaSki cells, which attenuated
the inhibitory effect of kindlin-2 on cell migration (Fig. 4A, B, E, and F). Addition of the
autophagy activator rapamycin (20 nM) reversed the promoting effect
of kindlin-2 knockdown on SiHa cell migration (Fig. 4C-E and G). Collectively, these
results indicated that kindlin-2 inhibited cell migration and that
this effect at least partly depended on autophagy.
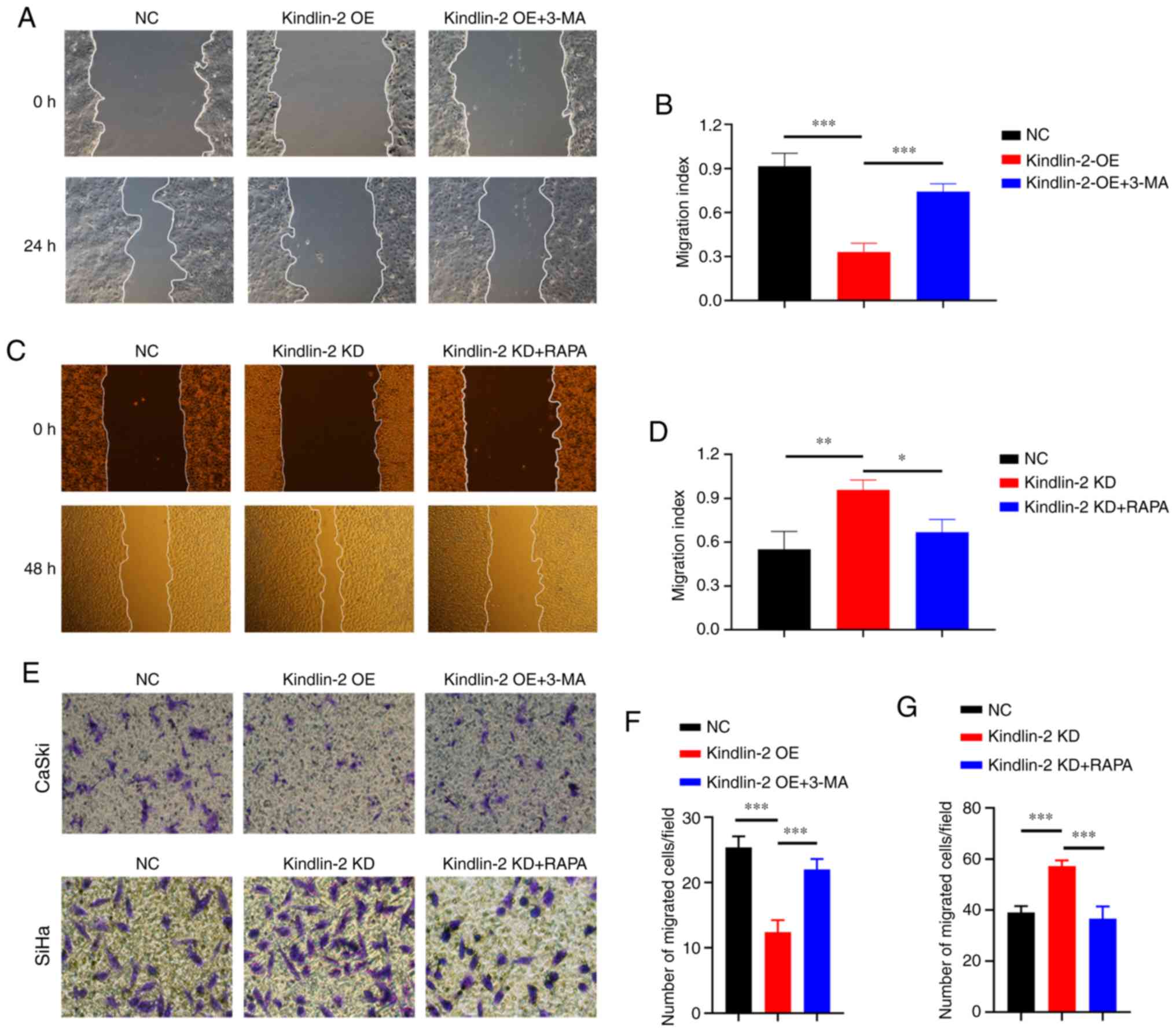 | Figure 4.Kindlin-2 impairs the migration of
cervical cancer cells by inducing autophagy. CaSki cells were
transfected with kindlin-2 lentivirus or NC lentivirus and then
treated with 5 mM 3-MA. SiHa cells were transfected with kindlin-2
RNAi or NC lentivirus and then treated with 20 nM RAPA. Wound
healing and Transwell migration assays were performed to detect
cell migration under different treatments. (A and B) Representative
images (magnification, ×100) and quantification of CaSki cells
subjected to the indicated treatments after scratch wounding. (C
and D) Representative images (magnification, ×100) and
quantification of SiHa cells subjected to the indicated treatments
after scratch wounding. The wound area was evaluated using ImageJ
software. (E) Representative images (magnification, ×200) and
quantification of migrated (F) CaSki and (G) SiHa cells. Migrated
cells were counted in five random fields. *P<0.05, **P<0.01,
***P<0.001. NC, negative control; OE, overexpression; KD,
knockdown; 3-MA, 3-methyladenine; RAPA, rapamycin. |
Discussion
In most types of cancer other than cervical cancer,
kindlin-2 is expressed at high levels in tumors and acts to promote
tumor progression (25–27). In the present study, it was revealed
that kindlin-2 plays a different role in cervical cancer. Kindlin-2
was expressed at low levels in cervical cancer, induced autophagy
by inhibiting the AKT/mTOR pathway, and inhibited the migration of
cervical cancer cells by promoting autophagy. The present study
revealed the role of kindlin-2 in autophagy and migration of
cervical cancer cells.
Kindlin-2 exerts multiple and even opposite effects
in different tumors, of which tumorigenicity is dominant. For
example, kindlin-2 was revealed to interact with epidermal growth
factor receptor to promote the migration of breast cancer cells
(25) and to promote gastric cancer
cell invasion by phosphorylating integrins β1 and β3 (26). However, kindlin-2 can also exert a
tumor-suppressive effect. For example, it was revealed to inhibit
tumor peritoneal dissemination in serous epithelial ovarian cancer
(28) and tumor cell invasion in
mesenchymal cancer (29). The role
of kindlin-2 in cervical cancer remained unclear. In the present
study, kindlin-2 overexpression suppressed the migration ability of
cervical cancer cells, whereas downregulation of kindlin-2 promoted
cell migration, indicating that kindlin-2 inhibits cervical cancer
cell migration. These contradictory findings suggest that kindlin-2
acts as either an oncoprotein or a tumor suppressor depending on
the cancer type and context.
Autophagy plays an important role in tumor
progression (30,31). In cervical cancer cells, hispolon
was revealed to inhibit cell migration by activating autophagy
(32), and spermidine inhibited
cell growth and promoted apoptosis by activating autophagy
(33). However, the relationship
between kindlin-2 and autophagy remained unclear. In the present
study, autophagy-promoting effects were observed after
overexpression of kindlin-2 and the opposite effects were observed
after knockdown of kindlin-2. This indicated that kindlin-2 can
induce autophagy in cervical cancer cells, confirming the
relationship between kindlin-2 and autophagy in cervical cancer
cells.
Autophagy has both metastasis-promoting and
-suppressive effects (34,35). It was determined, in the present
study, that inhibition of autophagy rescued the decrease in cell
migration ability caused by kindlin-2 overexpression, whereas
promotion of autophagy attenuated the increase in cell migration
ability induced by kindlin-2 knockdown. This indicated that
kindlin-2 inhibited cell migration by inducing autophagy. Similar
results have been reported in other types of tumors, including
hepatocellular carcinoma, renal cell cancer, and colorectal cancer
(36–38). The present results also provide new
evidence for the role of autophagy in the migration of cervical
cancer cells.
The AKT/mTOR pathway negatively regulates autophagy
(23,24). Moreover, AKT/mTOR pathway activation
has been revealed to promote tumor growth, metastasis,
angiogenesis, and therapeutic resistance (39), and this pathway was revealed to
serve as a therapeutic target in various tumors (40–42).
In the present study, it was revealed that overexpression of
kindlin-2 resulted in inhibition of the AKT/mTOR pathway, whereas
downregulation of kindlin-2 had the opposite effect, suggesting
that kindlin-2 negatively regulates this pathway. In further
experiments, it was revealed that treatment with an AKT activator
or inhibitor reversed the effect of overexpression or knockdown of
kindlin-2 on autophagy. These results indicated that kindlin-2
regulated autophagy via the AKT/mTOR pathway.
In conclusion, the present study revealed that
kindlin-2 impairs the migration of cervical cancer cells by
promoting autophagy, which is mediated by the AKT/mTOR pathway.
Autophagy plays an important role in the migration of cervical
cancer cells. Unfortunately, a mouse model of cervical cancer
metastasis has not been established yet, and further studies are
required to confirm the effect of kindlin-2 on tumor metastasis
in vivo. Another limitation is the small sample size used
for the detection of the expression of kindlin-2. A larger sample
size is required to confirm the results. Collectively, the present
study findings increased our understanding of the role of kindlin-2
in tumors and provides a novel potential target for the treatment
of cervical cancer metastasis.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Natural
Science Foundation of China (nos. 81760466 and 81460398), Natural
Science Foundation of Guangxi (no. 2015GXNSFAA139159) and Guangxi
Zhuang Autonomous Region Clinical Key Specialized Subject
Construction project funds.
Availability of data and materials
All data generated or used during this study are
included in this published article, still, further details are
available from the corresponding author on reasonable request.
Authors
GW, YaL and DY conceived and designed this study.
GW, XX, YF, XY preformed experimentation and data analysis. YiL
collected the clinical samples and preformed experimentation. GW
wrote the draft manuscript. DY and YaL reviewed and edited the
manuscript. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Guangxi Medical University Cancer Hospital, and all patients signed
an informed consent to participate in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wang J, Wang T, Yang YY, Chai YL, Shi F
and Liu ZI: Patient age, tumor appearance and tumor size are risk
factors for early recurrence of cervical cancer. Mol Clin Oncol.
3:363–366. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Galluzzi L and Green DR:
Autophagy-independent functions of the autophagy machinery. Cell.
177:1682–1699. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
White E: Deconvoluting the
context-dependent role for autophagy in cancer. Nat Rev Cancer.
12:401–410. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Amaravadi R, Kimmelman AC and White E:
Recent insights into the function of autophagy in cancer. Genes
Dev. 30:1913–1930. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Guo W, Wang H, Yang Y, Guo S, Zhang W, Liu
Y, Yi X, Ma J, Zhao T, Liu L, et al: Down-regulated miR-23a
contributes to the metastasis of cutaneous melanoma by promoting
autophagy. Theranostics. 7:2231–2249. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gao L, Lv G, Li R, Liu WT, Zong C, Ye F,
Li XY, Yang X, Jiang JH, Hou XJ, et al: Glycochenodeoxycholate
promotes hepatocellular carcinoma invasion and migration by
AMPK/mTOR dependent autophagy activation. Cancer Lett. 454:215–223.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang S, Zhu M, Wang Q, Hou Y, Li L, Weng
H, Zhao Y, Chen D, Ding H, Guo J and Li M: Alpha-fetoprotein
inhibits autophagy to promote malignant behaviour in hepatocellular
carcinoma cells by activating PI3K/AKT/mTOR signalling. Cell Death
Dis. 9:10272018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhan J and Zhang H: Kindlins: Roles in
development and cancer progression. Int J Biochem Cell Biol.
98:93–103. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ou YW, Zhao ZT, Wu CY, Xu BN, Song YM and
Zhan QM: Mig-2 attenuates cisplatin-induced apoptosis of human
glioma cells in vitro through AKT/JNK and AKT/p38 signaling
pathways. Acta Pharmacol Sin. 35:1199–1206. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sossey-Alaoui K, Pluskota E, Bialkowska K,
Szpak D, Parker Y, Morrison CD, Lindner DJ, Schiemann WP and Plow
EF: Kindlin-2 regulates the growth of breast cancer tumors by
activating CSF-1-mediated macrophage infiltration. Cancer Res.
77:5129–5141. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yoshida N, Masamune A, Hamada S, Kikuta K,
Takikawa T, Motoi F, Unno M and Shimosegawa T: Kindlin-2 in
pancreatic stellate cells promotes the progression of pancreatic
cancer. Cancer Lett. 390:103–114. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Heras-Sandoval D, Pérez-Rojas JM,
Hernández-Damián J and Pedraza-Chaverri J: The role of
PI3K/AKT/mTOR pathway in the modulation of autophagy and the
clearance of protein aggregates in neurodegeneration. Cell Signal.
26:2694–2701. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Rabanal-Ruiz Y, Otten EG and Korolchuk VI:
mTORC1 as the main gateway to autophagy. Essays Biochem.
61:565–584. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Aran D, Camarda R, Odegaard J, Paik H,
Oskotsky B, Krings G, Goga A, Sirota M and Butte AJ: Comprehensive
analysis of normal adjacent to tumor transcriptomes. Nat Commun.
8:10772017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lee YY, Kim TJ, Kim JY, Choi CH, Do IG,
Song SY, Sohn I, Jung SH, Bae DS, Lee JW and Kim BG: Genetic
profiling to predict recurrence of early cervical cancer. Gynecol
Oncol. 131:650–654. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Medina-Martinez I, Barrón V,
Roman-Bassaure E, Juárez-Torres E, Guardado-Estrada M, Espinosa AM,
Bermudez M, Fernández F, Venegas-Vega C, Orozco L, et al: Impact of
gene dosage on gene expression, biological processes and survival
in cervical cancer: A genome-wide follow-up study. PLoS One.
9:e978422014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tang Z, Li C, Kang B, Gao G, Li C and
Zhang Z: GEPIA: A web server for cancer and normal gene expression
profiling and interactive analyses. Nucleic Acids Res. 45:W98–W102.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ogłuszka M, Orzechowska M, Jędroszka D,
Witas P and Bednarek AK: Evaluate Cutpoints: Adaptable continuous
data distribution system for determining survival in kaplan-meier
estimator. Comput Methods Programs Biomed. 177:133–139. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tanida I, Ueno T and Kominami E: LC3 and
autophagy. Methods Mol Biol. 445:77–88. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Moscat J and Diaz-Meco MT: p62 at the
crossroads of autophagy, apoptosis, and cancer. Cell.
137:1001–1004. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mizushima N, Yoshimori T and Levine B:
Methods in mammalian autophagy research. Cell. 140:313–326. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Janku F, McConkey DJ, Hong DS and Kurzrock
R: Autophagy as a target for anticancer therapy. Nat Rev Clin
Oncol. 8:528–539. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Saiki S, Sasazawa Y, Imamichi Y, Kawajiri
S, Fujimaki T, Tanida I, Kobayashi H, Sato F, Sato S, Ishikawa K,
et al: Caffeine induces apoptosis by enhancement of autophagy via
PI3K/Akt/mTOR/p70S6K inhibition. Autophagy. 7:176–187. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Guo B, Gao J, Zhan J and Zhang H:
Kindlin-2 interacts with and stabilizes EGFR and is required for
EGF-induced breast cancer cell migration. Cancer Lett. 361:271–281.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shen Z, Ye Y, Kauttu T, Seppänen H,
Vainionpää S, Wang S, Mustonen H and Puolakkainen P: Novel focal
adhesion protein kindlin-2 promotes the invasion of gastric cancer
cells through phosphorylation of integrin β1 and β3. J Surg Oncol.
108:106–112. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
An Z, Dobra K, Lock JG, Strömblad S,
Hjerpe A and Zhang H: Kindlin-2 is expressed in malignant
mesothelioma and is required for tumor cell adhesion and migration.
Int J Cancer. 127:1999–2008. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ren C, Du J, Xi C, Yu Y, Hu A, Zhan J, Guo
H, Fang W, Liu C and Zhang H: Kindlin-2 inhibits serous epithelial
ovarian cancer peritoneal dissemination and predicts patient
outcomes. Biochem Biophys Res Commun. 446:187–194. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shi X and Wu C: A suppressive role of
mitogen inducible gene-2 in mesenchymal cancer cell invasion. Mol
Cancer Res. 6:715–724. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
White E, Mehnert JM and Chan CS:
Autophagy, metabolism, and cancer. Clin Cancer Res. 21:5037–5046.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
White E: The role for autophagy in cancer.
J Clin Invest. 125:42–46. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hsin MC, Hsieh YH, Wang PH, Ko JL, Hsin IL
and Yang SF: Hispolon suppresses metastasis via autophagic
degradation of cathepsin S in cervical cancer cells. Cell Death
Dis. 8:e30892017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chen Y, Zhuang H, Chen X, Shi Z and Wang
X: Spermidine-induced growth inhibition and apoptosis via
autophagic activation in cervical cancer. Oncol Rep. 39:2845–2854.
2018.PubMed/NCBI
|
|
34
|
Kenific CM, Thorburn A and Debnath J:
Autophagy and metastasis: Another double-edged sword. Curr Opin
Cell Biol. 22:241–245. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dower CM, Wills CA, Frisch SM and Wang HG:
Mechanisms and context underlying the role of autophagy in cancer
metastasis. Autophagy. 14:1110–1128. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang M, Liu S, Chua MS, Li H, Luo D, Wang
S, Zhang S, Han B and Sun C: SOCS5 inhibition induces autophagy to
impair metastasis in hepatocellular carcinoma cells via the
PI3K/Akt/mTOR pathway. Cell Death Dis. 10:6122019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhang Y, Fan Y, Huang S, Wang G, Han R,
Lei F, Luo A, Jing X, Zhao L, Gu S and Zhao X: Thymoquinone
inhibits the metastasis of renal cell cancer cells by inducing
autophagy via AMPK/mTOR signaling pathway. Cancer Sci.
109:3865–3873. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chu CA, Lee CT, Lee JC, Wang YW, Huang CT,
Lan SH, Lin PC, Lin BW, Tian YF, Liu HS and Chow NH: MiR-338-5p
promotes metastasis of colorectal cancer by inhibition of
phosphatidylinositol 3-kinase, catalytic subunit type 3-mediated
autophagy pathway. EBioMedicine. 43:270–281. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Porta C, Paglino C and Mosca A: Targeting
PI3K/Akt/mTOR signaling in cancer. Front Oncol. 4:642014.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Mabuchi S, Kuroda H, Takahashi R and
Sasano T: The PI3K/AKT/mTOR pathway as a therapeutic target in
ovarian cancer. Gynecol Oncol. 137:173–179. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Slomovitz BM and Coleman RL: The
PI3K/AKT/mTOR pathway as a therapeutic target in endometrial
cancer. Clin Cancer Res. 18:5856–5864. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lee JJ, Loh K and Yap YS: PI3K/Akt/mTOR
inhibitors in breast cancer. Cancer Biol Med. 12:342–354.
2015.PubMed/NCBI
|