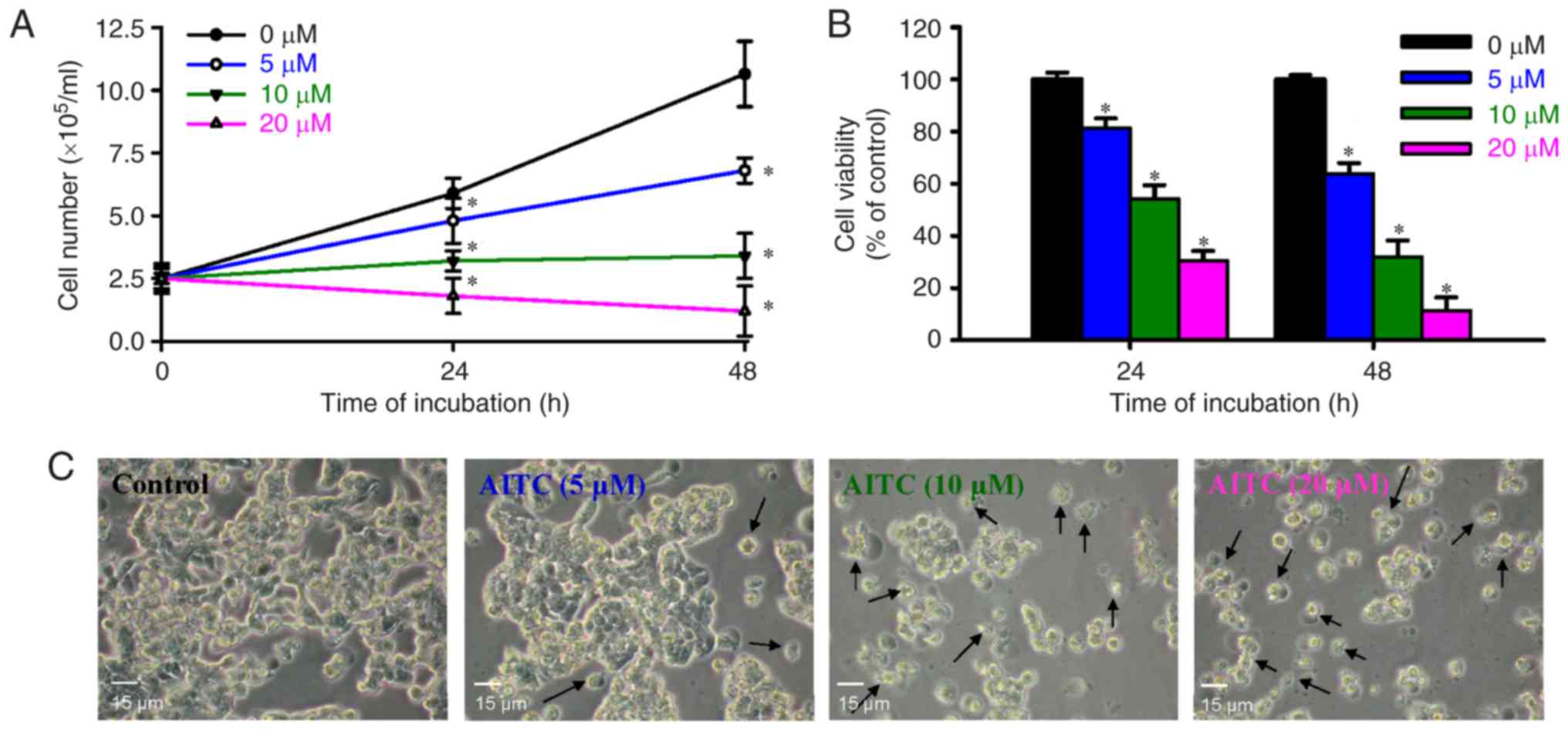
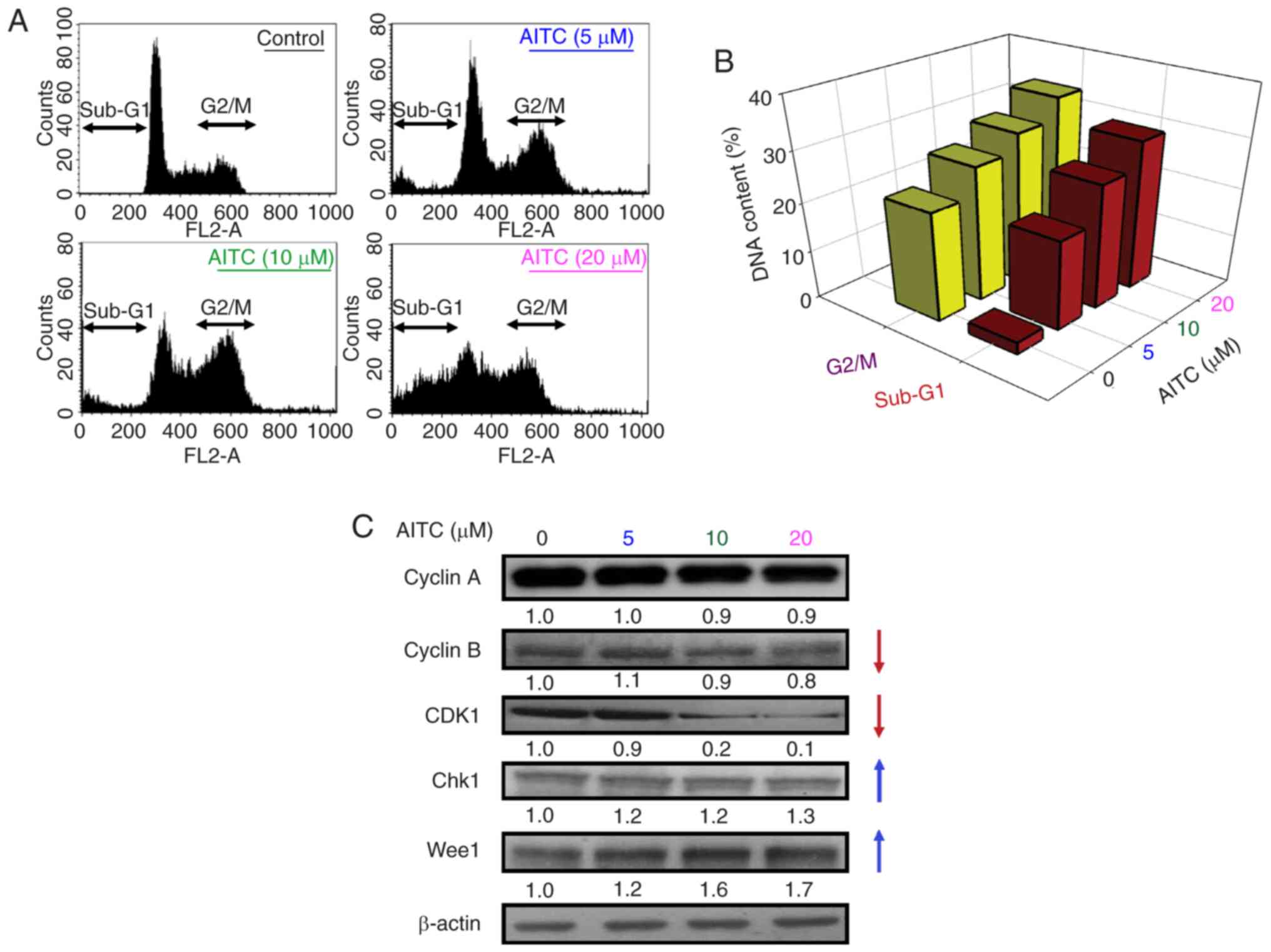
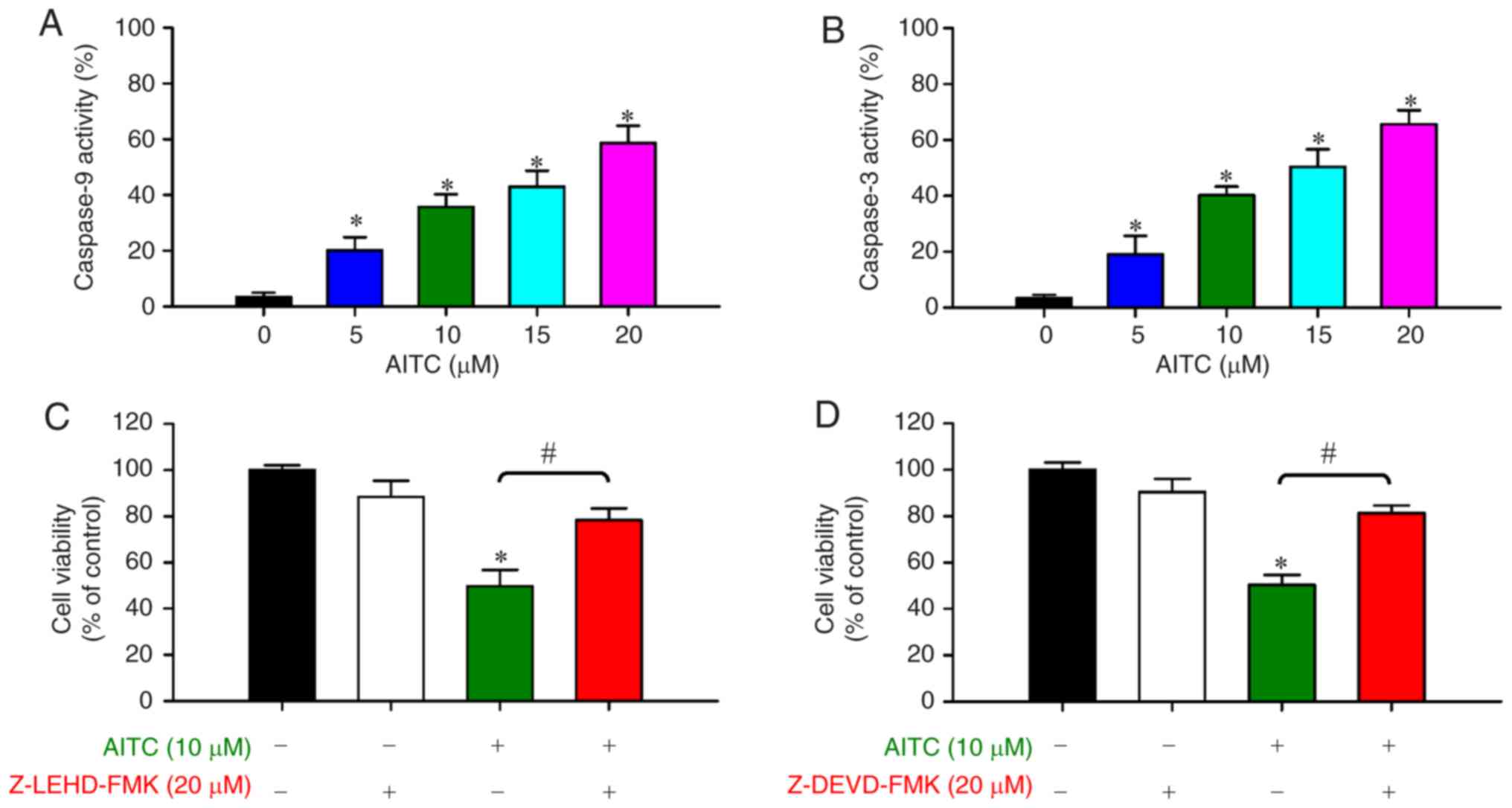
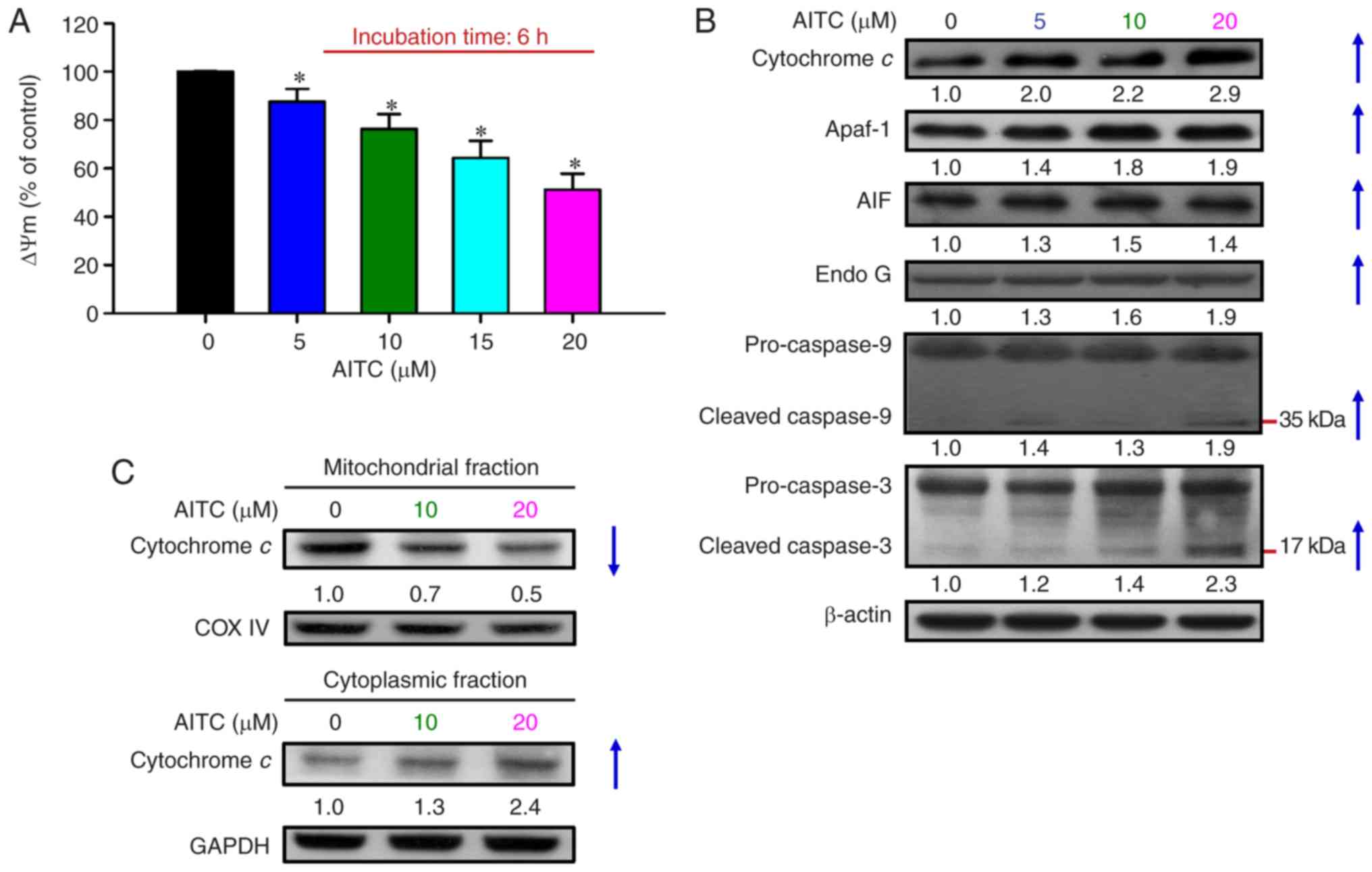
|
1
|
Ferlay J, Colombet M, Soerjomataram I,
Dyba T, Randi G, Bettio M, Gavin A, Visser O and Bray F: Cancer
incidence and mortality patterns in Europe: Estimates for 40
countries and 25 major cancers in 2018. Eur J Cancer. 103:356–387.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shen W, Jin Z, Tong X, Wang H, Zhuang L,
Lu X and Wu S: TRIM14 promotes cell proliferation and inhibits
apoptosis by suppressing PTEN in colorectal cancer. Cancer Manag
Res. 11:5725–5735. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ribeiro Gomes J, Belotto M and D'Alpino
Peixoto R: The role of surgery for unusual sites of metastases from
colorectal cancer: A review of the literature. Eur J Surg Oncol.
43:15–19. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cheng X, Feng H, Wu H, Jin Z, Shen X,
Kuang J, Huo Z, Chen X, Gao H, Ye F, et al: Targeting autophagy
enhances apatinib-induced apoptosis via endoplasmic reticulum
stress for human colorectal cancer. Cancer Lett. 431:105–114. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Afrin S, Giampieri F, Gasparrini M,
Forbes-Hernández TY, Cianciosi D, Reboredo-Rodriguez P, Zhang J,
Manna PP, Daglia M, Atanasov AG and Battino M: Dietary
phytochemicals in colorectal cancer prevention and treatment: A
focus on the molecular mechanisms involved. Biotechnol Adv.
38:1073222020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pan JH, Abernathy B, Kim YJ, Lee JH, Kim
JH, Shin EC and Kim JK: Cruciferous vegetables and colorectal
cancer prevention through microRNA regulation: A review. Crit Rev
Food Sci Nutr. 58:2026–2038. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vanduchova A, Anzenbacher P and
Anzenbacherova E: Isothiocyanate from broccoli, sulforaphane, and
its properties. J Med Food. 22:121–126. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mitsiogianni M, Mantso T, Trafalis DT,
Vasantha Rupasinghe HP, Zoumpourlis V, Franco R, Botaitis S, Pappa
A and Panayiotidis MI: Allyl isothiocyanate regulates lysine
acetylation and methylation marks in an experimental model of
malignant melanoma. Eur J Nutr. 59:557–569. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wu CL, Huang AC, Yang JS, Liao CL, Lu HF,
Chou ST, Ma CY, Hsia TC, Ko YC and Chung JG: Benzyl isothiocyanate
(BITC) and phenethyl isothiocyanate (PEITC)-mediated generation of
reactive oxygen species causes cell cycle arrest and induces
apoptosis via activation of caspase-3, mitochondria dysfunction and
nitric oxide (NO) in human osteogenic sarcoma U-2 OS cells. J
Orthop Res. 29:1199–1209. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kumar G, Tuli HS, Mittal S, Shandilya JK,
Tiwari A and Sandhu SS: Isothiocyanates: A class of bioactive
metabolites with chemopreventive potential. Tumour Biol.
36:4005–4016. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Abbaoui B, Lucas CR, Riedl KM, Clinton SK
and Mortazavi A: Cruciferous vegetables, isothiocyanates, and
bladder cancer prevention. Mol Nutr Food Res. 62:e18000792018.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tsai SC, Huang WW, Huang WC, Lu CC, Chiang
JH, Peng SF, Chung JG, Lin YH, Hsu YM, Amagaya S, et al:
ERK-modulated intrinsic signaling and G(2)/M phase arrest
contribute to the induction of apoptotic death by allyl
isothiocyanate in MDA-MB-468 human breast adenocarcinoma cells. Int
J Oncol. 41:2065–2072. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu P, Behray M, Wang Q, Wang W, Zhou Z,
Chao Y and Bao Y: Anti-cancer activities of allyl isothiocyanate
and its conjugated silicon quantum dots. Sci Rep. 8:10842018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang Y: Allyl isothiocyanate as a cancer
chemopreventive phytochemical. Mol Nutr Food Res. 54:127–135. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Qin G, Li P and Xue Z: Effect of allyl
isothiocyanate on the viability and apoptosis of the human cervical
cancer HeLa cell line in vitro. Oncol Lett. 15:8756–8760.
2018.PubMed/NCBI
|
|
16
|
Jiang Z, Liu X, Chang K, Liu X and Xiong
J: Allyl isothiocyanate inhibits the proliferation of renal
carcinoma cell line GRC-1 by inducing an imbalance between Bcl2 and
Bax. Med Sci Monit. 22:4283–4288. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bo P, Lien JC, Chen YY, Yu FS, Lu HF, Yu
CS, Chou YC, Yu CC and Chung JG: Allyl isothiocyanate induces cell
toxicity by multiple pathways in human breast cancer cells. Am J
Chin Med. 44:415–437. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rakariyatham K, Yang X, Gao Z, Song M, Han
Y, Chen X and Xiao H: Synergistic chemopreventive effect of allyl
isothiocyanate and sulforaphane on non-small cell lung carcinoma
cells. Food Funct. 10:893–902. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sávio AL, da Silva GN and Salvadori DM:
Inhibition of bladder cancer cell proliferation by allyl
isothiocyanate (mustard essential oil). Mutat Res. 771:29–35. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lau WS, Chen T and Wong YS: Allyl
isothiocyanate induces G2/M arrest in human colorectal
adenocarcinoma SW620 cells through down-regulation of Cdc25B and
Cdc25C. Mol Med Rep. 3:1023–1030. 2010.PubMed/NCBI
|
|
21
|
Chen HE, Lin JF, Tsai TF, Lin YC, Chou KY
and Hwang TI: Allyl isothiocyanate induces autophagy through the
up-regulation of Beclin-1 in human prostate cancer cells. Am J Chin
Med. Oct 4–2018.(Epub ahead of print). doi:
10.1142/S0192415X18500830. View Article : Google Scholar
|
|
22
|
Chen NG, Chen KT, Lu CC, Lan YH, Lai CH,
Chung YT, Yang JS and Lin YC: Allyl isothiocyanate triggers G2/M
phase arrest and apoptosis in human brain malignant glioma GBM 8401
cells through a mitochondria-dependent pathway. Oncol Rep.
24:449–455. 2010.PubMed/NCBI
|
|
23
|
Lai KC, Lu CC, Tang YJ, Chiang JH, Kuo DH,
Chen FA, Chen IL and Yang JS: Allyl isothiocyanate inhibits cell
metastasis through suppression of the MAPK pathways in epidermal
growth factor-stimulated HT29 human colorectal adenocarcinoma
cells. Oncol Rep. 31:189–196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hwang ES and Lee HJ: Allyl isothiocyanate
and its N-acetylcysteine conjugate suppress metastasis via
inhibition of invasion, migration, and matrix
metalloproteinase-2/-9 activities in SK-Hep 1 human hepatoma cells.
Exp Biol Med (Maywood). 231:421–430. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Thejass P and Kuttan G: Inhibition of
endothelial cell differentiation and proinflammatory cytokine
production during angiogenesis by allyl isothiocyanate and phenyl
isothiocyanate. Integr Cancer Ther. 6:389–399. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Smith TK, Lund EK, Parker ML, Clarke RG
and Johnson IT: Allyl-isothiocyanate causes mitotic block, loss of
cell adhesion and disrupted cytoskeletal structure in HT29 cells.
Carcinogenesis. 25:1409–1415. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Smith T, Musk SR and Johnson IT: Allyl
isothiocyanate selectively kills undifferentiated HT29 cells in
vitro and suppresses aberrant crypt foci in the colonic mucosa of
rats. Biochem Soc Trans. 24:381S1996. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Musk SR and Johnson IT: Allyl
isothiocyanate is selectively toxic to transformed cells of the
human colorectal tumour line HT29. Carcinogenesis. 14:2079–2083.
1993. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lu CC, Yang JS, Huang AC, Hsia TC, Chou
ST, Kuo CL, Lu HF, Lee TH, Wood WG and Chung JG: Chrysophanol
induces necrosis through the production of ROS and alteration of
ATP levels in J5 human liver cancer cells. Mol Nutr Food Res.
54:967–976. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee HP, Chen PC, Wang SW, Fong YC, Tsai
CH, Tsai FJ, Chung JG, Huang CY, Yang JS, Hsu YM, et al: Plumbagin
suppresses endothelial progenitor cell-related angiogenesis in
vitro and in vivo. J Funct Foods. 52:537–544. 2019. View Article : Google Scholar
|
|
31
|
Chang LC, Hsieh MT, Yang JS, Lu CC, Tsai
FJ, Tsao JW, Chiu YJ, Kuo SC and Lee KH: Effect of
bis(hydroxymethyl) alkanoate curcuminoid derivative MTH-3 on cell
cycle arrest, apoptotic and autophagic pathway in triple-negative
breast adenocarcinoma MDA-MB-231 cells: An in vitro study.
Int J Oncol. 52:67–76. 2018.PubMed/NCBI
|
|
32
|
Chen YA, Shih HW, Lin YC, Hsu HY, Wu TF,
Tsai CH, Wu CL, Wu HY, Hsieh JT, Tang CH and Lai CH: Simvastatin
sensitizes radioresistant prostate cancer cells by compromising DNA
double-strand break repair. Front Pharmacol. 9:6002018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lee CF, Chiang NN, Lu YH, Huang YS, Yang
JS, Tsai SC, Lu CC and Chen FA: Benzyl isothiocyanate (BITC)
triggers mitochondria-mediated apoptotic machinery in human
cisplatin-resistant oral cancer CAR cells. Biomedicine (Taipei).
8:152018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lin CC, Chen KB, Tsai CH, Tsai FJ, Huang
CY, Tang CH, Yang JS, Hsu YM, Peng SF and Chung JG: Casticin
inhibits human prostate cancer DU 145 cell migration and invasion
via Ras/Akt/NF-κB signaling pathways. J Food Biochem.
43:e129022019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lin CW, Chin HK, Lee SL, Chiu CF, Chung
JG, Lin ZY, Wu CY, Liu YC, Hsiao YT, Feng CH, et al: Ursolic acid
induces apoptosis and autophagy in oral cancer cells. Environ
Toxicol. 34:983–991. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chiang JH, Yang JS, Ma CY, Yang MD, Huang
HY, Hsia TC, Kuo HM, Wu PP, Lee TH and Chung JG: Danthron, an
anthraquinone derivative, induces DNA damage and caspase
cascades-mediated apoptosis in SNU-1 human gastric cancer cells
through mitochondrial permeability transition pores and
Bax-triggered pathways. Chem Res Toxicol. 24:20–29. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lu CC, Yang JS, Chiang JH, Hour MJ, Lin
KL, Lee TH and Chung JG: Cell death caused by quinazolinone HMJ-38
challenge in oral carcinoma CAL 27 cells: Dissections of
endoplasmic reticulum stress, mitochondrial dysfunction and tumor
xenografts. Biochim Biophys Acta. 1840:2310–2320. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Huang WW, Chiu YJ, Fan MJ, Lu HF, Yeh HF,
Li KH, Chen PY, Chung JG and Yang JS: Kaempferol induced apoptosis
via endoplasmic reticulum stress and mitochondria-dependent pathway
in human osteosarcoma U-2 OS cells. Mol Nutr Food Res.
54:1585–1595. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Van der Jeught K, Xu HC, Li YJ, Lu XB and
Ji G: Drug resistance and new therapies in colorectal cancer. World
J Gastroenterol. 24:3834–3848. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhang Y, Xu J, Zhang N, Chen M, Wang H and
Zhu D: Targeting the tumour immune microenvironment for cancer
therapy in human gastrointestinal malignancies. Cancer Lett.
458:123–135. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Johnson IT: Cruciferous vegetables and
risk of cancers of the gastrointestinal tract. Mol Nutr Food Res.
62:e17010002018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chang WJ, Chen BH, Inbaraj BS and Chien
JT: Preparation of allyl isothiocyanate nanoparticles, their
anti-inflammatory activity towards RAW 264.7 macrophage cells and
anti-proliferative effect on HT1376 bladder cancer cells. J Sci
Food Agric. 99:3106–3116. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Löbrich M and Jeggo PA: The impact of a
negligent G2/M checkpoint on genomic instability and cancer
induction. Nat Rev Cancer. 7:861–869. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Geng F, Tang L, Li Y, Yang L, Choi KS,
Kazim AL and Zhang Y: Allyl isothiocyanate arrests cancer cells in
mitosis, and mitotic arrest in turn leads to apoptosis via Bcl-2
protein phosphorylation. J Biol Chem. 286:32259–32267. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Bhattacharya A, Li Y, Geng F, Munday R and
Zhang Y: The principal urinary metabolite of allyl isothiocyanate,
N-acetyl-S-(N-allylthiocarbamoyl)cysteine, inhibits the growth and
muscle invasion of bladder cancer. Carcinogenesis. 33:394–398.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kim C and Kim B: Anti-cancer natural
products and their bioactive compounds inducing ER stress-mediated
apoptosis: A Review. Nutrients. 10:10212018. View Article : Google Scholar
|
|
47
|
Park KW, Eun Kim G, Morales R, Moda F,
Moreno-Gonzalez I, Concha-Marambio L, Lee AS, Hetz C and Soto C:
The endoplasmic reticulum chaperone GRP78/BiP modulates prion
propagation in vitro and in vivo. Sci Rep. 7:447232017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang C, Song X, Shang M, Zou W, Zhang M,
Wei H and Shao H: Curcumin exerts cytotoxicity dependent on
reactive oxygen species accumulation in non-small-cell lung cancer
cells. Future Oncol. 15:1243–1253. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Cheng MH, Huang HL, Lin YY, Tsui KH, Chen
PC, Cheng SY, Chong IW, Sung PJ, Tai MH, Wen ZH, et al: BA6 Induces
apoptosis via stimulation of reactive oxygen species and inhibition
of oxidative phosphorylation in human lung cancer cells. Oxid Med
Cell Longev. 2019:63421042019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Liao HY, Kao CM, Yao CL, Chiu PW, Yao CC
and Chen SC: 2,4,6-Trinitrotoluene induces apoptosis via
ROS-regulated mitochondrial dysfunction and endoplasmic reticulum
stress in HepG2 and Hep3B cells. Sci Rep. 7:81482017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yang F, Tang J, Dai K and Huang Y:
Metallic wear debris collected from patients induces apoptosis in
rat primary osteoblasts via reactive oxygen species-mediated
mitochondrial dysfunction and endoplasmic reticulum stress. Mol Med
Rep. 19:1629–1637. 2019.PubMed/NCBI
|