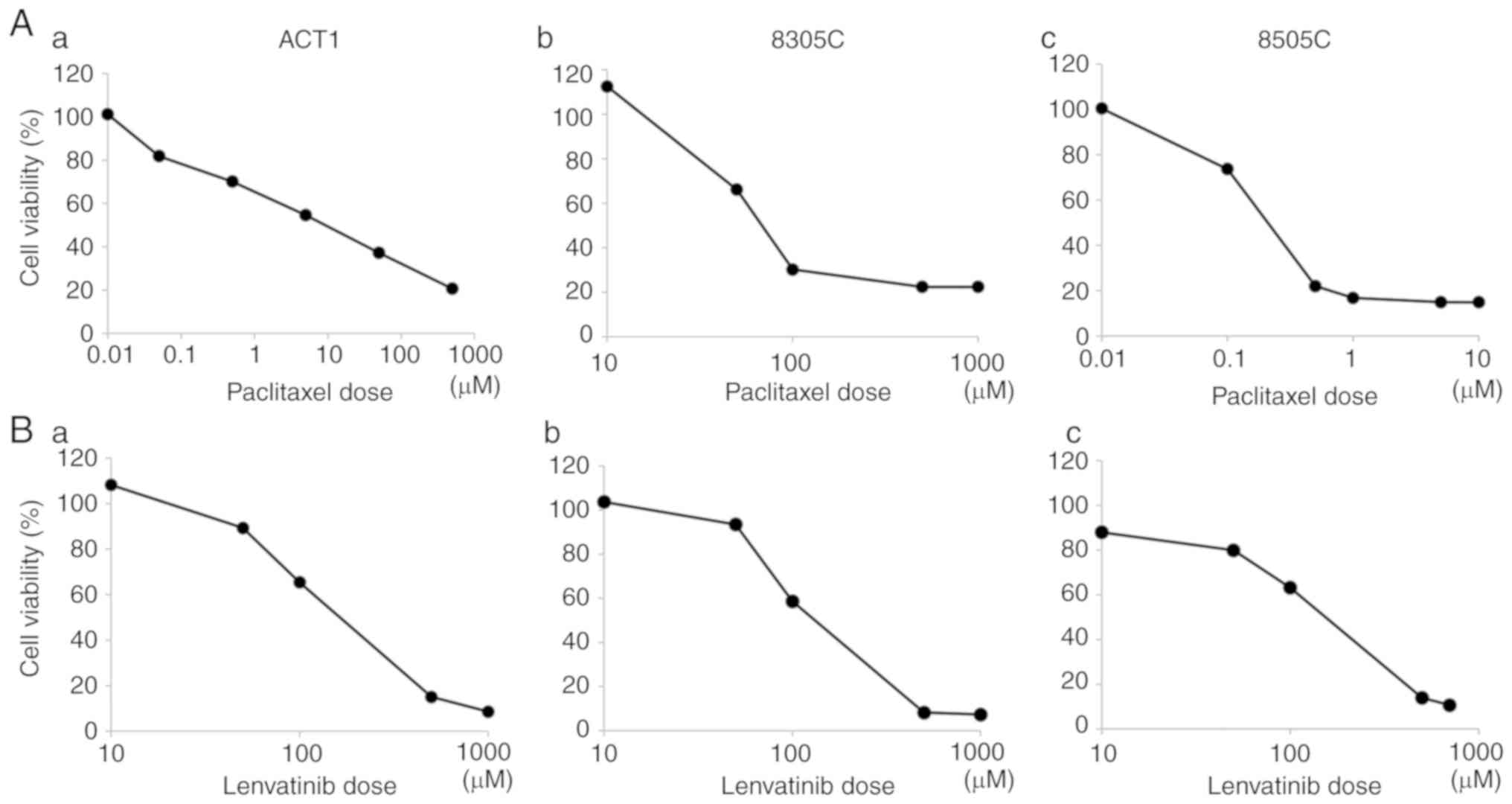
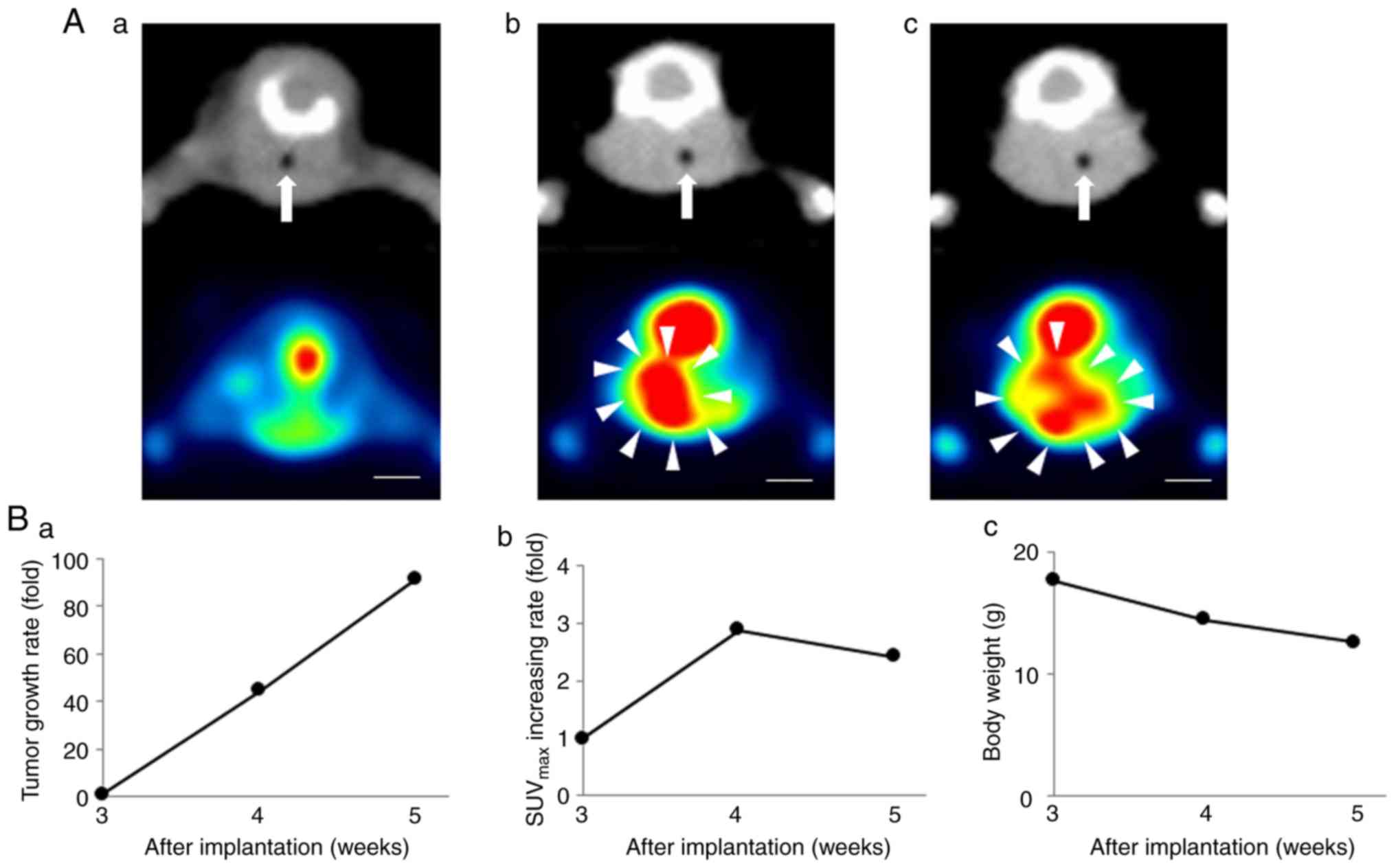
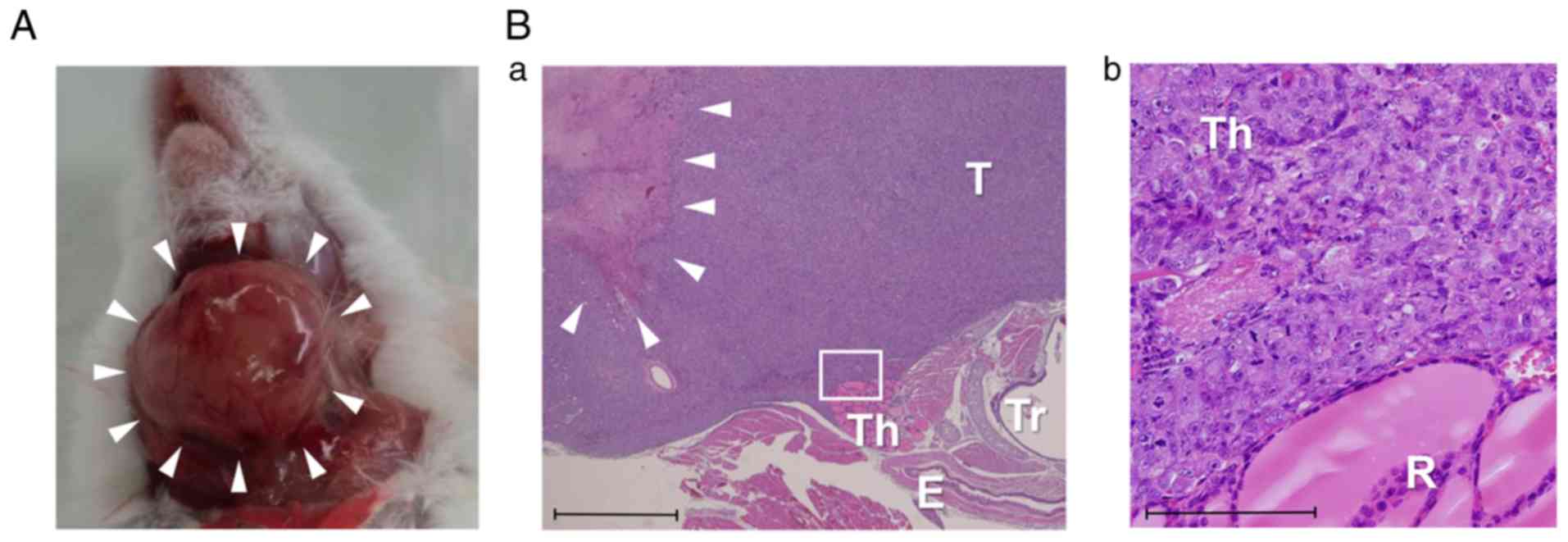
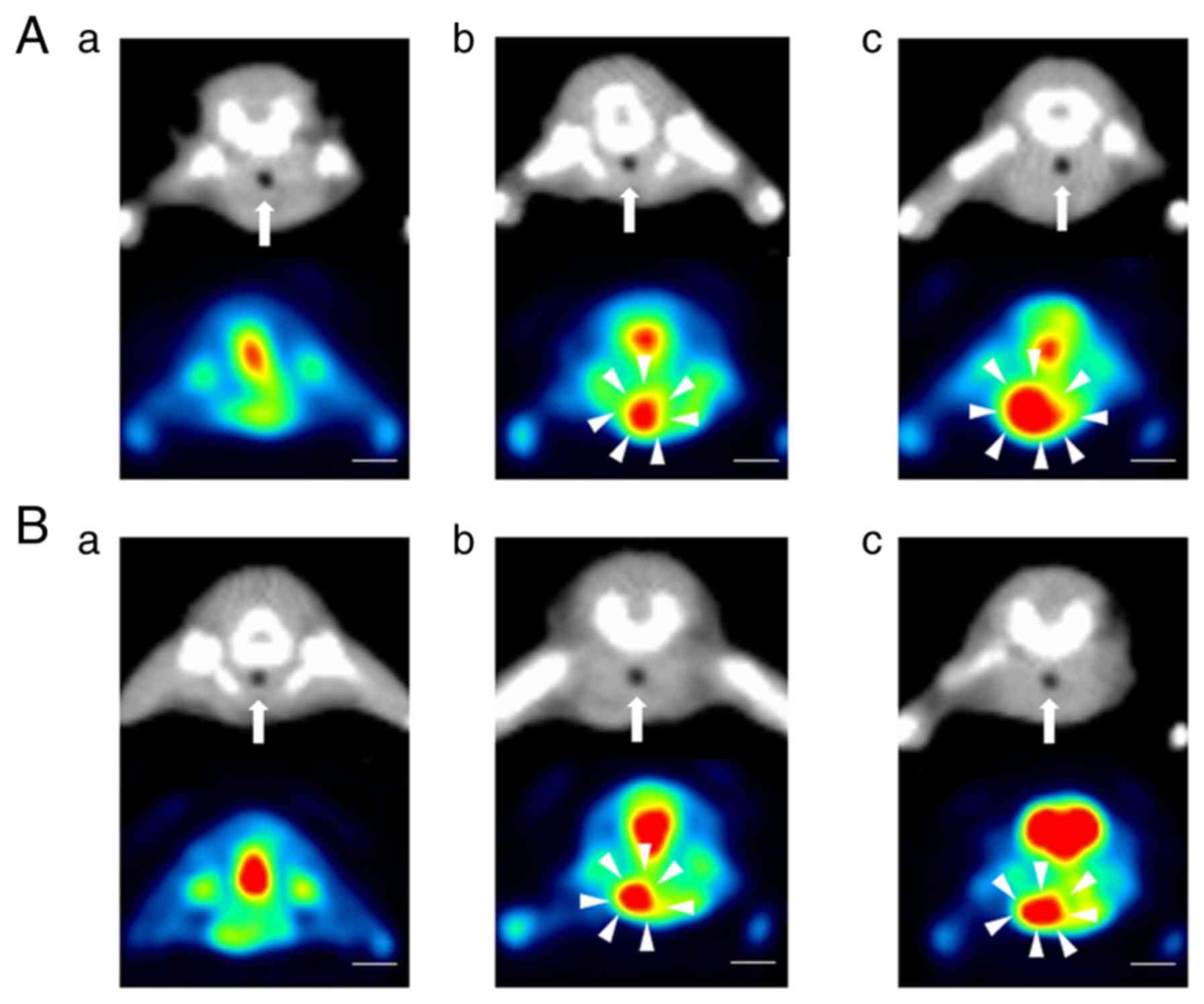
|
1
|
Kebebew E, Dreenspan FS, Clark OH, Woeber
KA and McMillan A: Anaplastic thyroid carcinoma. Treatment outcome
and prognostic factors. Cancer. 103:1330–1335. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tennvall J, Lundell G, Wahlberg P,
Bergenfelz A, Grimelius L, Akerman M, Hjelm Skog AL and Wallin G:
Anaplastic thyroid carcinoma: Three protocols combining
doxorubicin, hyperfractionated radiotherapy and surgery. Br J
Cancer. 12:1848–1853. 2002. View Article : Google Scholar
|
|
3
|
Sugitani I, Onoda N, Ito K and Sugitabi S:
Management of anaplastic thyroid carcinoma: Fruits from ATC
Research Consortium of Japan. J Nippon Med Sch. 85:18–27. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shimaoka K, Schoenfeld DA, DeWys WD,
Creech RH and DeConti R: A randomized trial of doxorubicin versus
doxorubicin plus cisplatin in patients with advancer thyroid
carcinoma. Cancer. 56:2155–2160. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ahuja S and Emst H: Chemotherapy of
thyroid carcinoma. J Endocrinol Invest. 3:303–310. 1987. View Article : Google Scholar
|
|
6
|
Ain KB, Egorin MJ and DeSimone PA:
Treatment of anaplastic thyroid carcinoma with paclitaxel: Phase 2
trial using ninety-six-hour infusion. Collaborative Thyroid Cancer
Health Intervention Trials (CATCHIT) Group. Thyroid. 10:587–594.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Onoda N, Sugino K, Higashiyama T, Kammor
i, Toda K, Ito K, Yoshida A, Suganuma N, Nakashima N, Suzuki S, et
al: The safety and efficacy of weekly paclitaxel administration for
anaplastic thyroid cancer patients: A nationwide prospective study.
Thyroid. 26:1293–1299. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tahara M, Kiyota N, Yamazaki T, Chayara N,
Nakano K, Inagaki L, Toda K, Enokida T, Minami H, Imamura Y, et al:
Lenvatinib for anaplastic thyroid cancer. Front Oncol. 7:252017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hundahl SA, Fleming ID, Fremgen AM and
Menck HR: A national cancer data base report on 53,856 cases of
thyroid carcinoma treated in the U.S., 1985–1995. Cancer.
83:2638–2648. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kitamura Y, Shimizu K, Hagahama M, Sugino
K, Osaki O, Mimura T, Ito K, Ito K and Tanaka S: Immediate causes
of death in thyroid carcinoma: Clinicopathological analysis of 161
fetal cases. J Clin Endocrinol Metab. 84:4034–4039. 1999.
View Article : Google Scholar
|
|
11
|
Kim S, Park YW, Schiff BA, Doan DD, Yazici
Y, Jasser SA, Younes M, Mandal M, Bekele BN and Myers JN: An
orthotopic model of anaplastic thyroid carcinoma in athymic nude
mice. Clin Cancer Res. 11:1713–1721. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nucera C, Nehs MA, Mekel M, Zhang X, Hodin
R, Lawler J, Nose V and Parangi S: A novel orthotopic mouse model
of human anaplastic thyroid carcinoma. Thyroid. 19:1077–1084. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sewell W, Reeb A and Lin RY: An orthotopic
mouse model of anaplastic thyroid carcinoma. J Vis Exp.
74:500972013.
|
|
14
|
Paget S: The distribution of secondary
growths in cancer of the breast. Lancet. 133:571–573. 1889.
View Article : Google Scholar
|
|
15
|
Bibby MC: Orthotopic models of cancer for
preclinical drug evaluation: Advantages and disadvantages. Eur J
Cancer. 40:852–857. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nehas MA, Nucera C, Nagarkatti SS, Sado w,
Morales-Garcia D, Hodin RA and Parangi S: Late intervention with
anti-BRAFV600E therapy induces tumor regression in an orthotopic
mouse model of anaplastic thyroid cancer. Endocrinology.
153:985–994. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cha W, Kim DW, Kim SD, Jeon EH, Jeong WJ
and Ahn SH: Effect of perioperative treatment with a
hypoxia-inducible factor-1-alpha inhibitor in an orthotopic
surgical mouse models of thyroid cancer. Anticancer Res.
35:2049–2054. 2015.PubMed/NCBI
|
|
18
|
Nehs MA, Nagarkatti S, Nucera C, Hodin RA
and Parangi S: Thyroidectomy with neoadjuvant PLX4720 extends
survival and decreases tumor burden in an orthotopic mouse models
of anaplastic thyroid cancer. Surgery. 148:1154–1162. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mokhtar M, Kondo K, Takizawa H, Ohtani T,
Otsuka H, Kubo H, Kajiura K, Nakagawa Y, Kawanaka Y, Yoshida M, et
al: Non-invasive monitoring of anticancer effects of cisplatin on
lung cancer in an orthotopic SICD mouse model using [18F] FDG
PET-CT. Oncol Rep. 31:2007–2014. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Otani T, Kondo K, Takizawa H, Kajiura K,
Fujino H, Otsuka H and Miyoshi H: Non-invasive monitoring of
cisplatin and erlotinib efficacy against lung cancer in orthotopic
SCID mouse models by small animal FDG-PET/CT and CT. Oncol Rep.
41:447–454. 2019.PubMed/NCBI
|
|
21
|
Marcus C, Whitworth PW, Surasi DS, Pai SI
and Subramaniam RM: PET/CT in the Management of thyroid cancers.
AJR Am J Roentgenol. 202:1316–1329. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Poisson T, Deandreis D, Leboulleux S,
Bidault F, Bonniaud G, Baillot S, Auperin A, AI Ghuzlan A, Travagli
JP and Lumbroso J: 18F-fluorodeoxyglucose positron emission
tomography and computed tomography in anaplastic thyroid cancer.
Eur J Nucl Med Mol Imaging. 37:2277–2285. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Killkenny C, Browne WJ, Cuthill IC,
Emerson M and Altman DG: Improving bioscience research reporting:
The ARRIVE guidelines for reporting animal research. Osteoarthritis
Cartilage. 20:256–260. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jing C, Gao Z, Wang R, Yang Z, Shi B and
Hou P: Lenvatinib enhances the antitumor effects of paclitaxel in
anaplastic thyroid cancer. Am J Cancer Res. 4:903–912. 2017.
|
|
25
|
Ahmed S, Ghazarian MP, Cabanillas ME,
Zafereo ME, Williams MD, Vu T, Schomer DF and Debnam JM: Imaging of
anaplastic thyroid carcinoma. AJNR AM J Neuroradiol. 3:547–551.
2018. View Article : Google Scholar
|
|
26
|
Deeken-Draisey A, Yang GY, Gao J and
Alexiev BA: Anaplastic thyroid carcinoma: An epidemiologic,
histologic, immunohistochemical, and molecular single-institution
study. Hum Pathol. 82:140–148. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kostakoglu L and Chauvie S: Metabolic
tumor volume metrics in lymphoma. Semin Nucl Med. 48:50–66. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dibble EH, Alvarez AC, Truong MT, Mercier
G, Cook EF and Subramaniam RM: 18F-FDG metabolic tumor volume and
total glycolytic activity of oral cavity and oropharyngeal squamous
cell cancer: Adding value to clinical staging. J Nucl Med.
53:709–715. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Manohar PM, Beesley LJ, Bellile EL, Worden
FP and Avram AM: Prognostic value of FDG-PET/CT metabolic
parameters in metastatic radioiodine-refractory differentiated
thyroid cancer. Clin Nucl Med. 43:641–647. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tohyama O, Matsui J, Kodama K, Hata-Sugi
N, Kimura T, Okamoto K, Minoshima Y, Iwata M and Funahashi Y:
Antitumor activity of lenvatinib (e7080): An angiogenesis inhibitor
that targets multiple receptor tyrosine kinases in preclinical
human thyroid cancer models. J Thyroid Res. 2014:132014. View Article : Google Scholar
|
|
31
|
Miller K, Wang M, Gralow J, Dickler M,
Cobleigh M, Perez EA, Shenkiier T, Cella D and Davidson NA:
Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic
breast cancer. N Engl J Med. 26:2666–2676. 2007. View Article : Google Scholar
|