Introduction
Oncolytic viruses, characterized by their
preferential infection, replication and destruction of tumor cells
and cancer-associated endothelial cells, have become a major topic
for basic research and consequent clinical applications in novel
tumor therapy strategies (1).
Concomitant antitumor immune responses often exist as well
(2). In recent decades, oncolytic
viral therapy has aroused widespread interest and great progress
has been achieved (3). Various
oncolytic viruses have been studied and some have already been
developed for treatments agents, such as adenovirus, herpes simplex
virus type I, and the vaccinia virus (4,5).
Imlygic (talimogene laherparepvec; T-VEC) is the first oncolytic
virus approved for melanoma therapy by the USFDA for cases where
lesions are in the skin and lymph nodes and cannot be completely
removed by surgery (6). The safety
and effectiveness of oncolytic viruses have been widely accepted
(6).
Orf virus (ORFV) is a prototype member of the genus
Parapoxvirus, belonging to Poxviridae, which can
cause contagious pustular stomatitis, an acute mucocutaneous
infection in sheep and goats, and is usually self-limiting
(7,8). With an increasing number of
comprehensive studies on its molecular, biological and
immunological characteristics, especially given its vigorous and
particular immunomodulatory properties, ORFV has been proposed as a
potential oncolytic virus vector (8–10).
ORFV is known to have a strong immunomodulating
activity against the host. ORFV can rapidly mediate innate and
adaptive immune responses in the body. Upon infection by ORFV,
neutrophils, natural killer (NK) cells and dendritic cells (DC) are
enriched at the infection sites, various types of cells in the
innate immune system are activated, and several key cytokines are
secreted (8). Notably, ORFV induces
mainly the Th1 immune response in the early stages of infection
where the classical cytokines IFN-γ, TNF, IL-6, IL-8, IL-12 and
IL-18 are secreted from the peripheral immune cells (8,11).
Then, the Th2 type immune response appears with the secretion of
IL-4, IL-1 receptor antagonist (IL-1RA), and IL-10 (12).
A live or inactivated ORFV can induce a strong
antitumor immune response in murine models of multiple tumors.
Fiebig et al (9) were the
first to report that inactivated ORFV mediates antitumor effects in
various tumor models, including the murine syngeneic B16F10
melanoma and MDA-MB-231 human breast cancer xenograft, and revealed
that NK cells play an important role in the antitumor effects of
ORFV. Anti-mouse NK-1.1 antibody partially inhibited its antitumor
activity by inhibiting the activity of NK and NKT cells. Moreover,
when IFN-γ was neutralized, the inhibitory effects of ORFV
disappeared (9). Inactivated ORFV
inhibited tumor growth of MDA-MB-231 tumor-bearing NOD/LtSz-scid/j
mice which lacked NK and functional T- and B-lymphocytes (9). Rintoul et al (11) confirmed that live ORFV possessed
antitumor effects through activation of NK cells and by inducing
secretion of cytokines IFN-γ and granzyme B. A previous study has
found that surgery mediates the disfunction of NK cells, but ORFV
injection during surgery can improve the function of NK cells,
thereby reducing intra-operative metastasis and prolonging the
survival time (13). A better
understanding of the molecular mechanisms underlying the antitumor
effects of ORFV will be beneficial to further develop ORFV as
oncolytic vectors for human tumor treatment.
In prior studies, researchers have revealed some
mechanisms through which oncolytic viruses exert oncolytic
functions (14–16): direct tumoricidal cytotoxicity and
activation of host antitumor immune responses. Referring to
functional characteristics of oncolytic viruses and complicated
immunoregulation function of ORFV (17–19),
the present study used high-throughput screening methods to
investigate the ORFV-mediated regulation of cytokine expression. In
the present study, the antitumor activity of ORFV strain NA1/11 was
investigated through in vitro cell experiments and in
vivo animal studies.
Materials and methods
Reagents
The antibodies for cleaved caspase-3 (product no.
9664), cleaved caspase-9 (product no. 7237), Smac (product no.
15108), β-tubulin (product no. 2146), PARP (product no. 9542) were
obtained from Cell Signaling Technology, Inc. Annexin V/7-AAD
Apoptosis Detection Kit was obtained from Nanjing KeyGen Biotech
Co., Ltd. The enhanced chemiluminescence (ECL) detection kit was
acquired from Pierce; Thermo Fisher Scientific, Inc. The Cell
Counting Kit-8 (CCK-8) reagent was purchased from Dojindo Molecular
Technologies, Inc. Polyvinylidene difluoride (PVDF) membranes were
obtained from EMD Millipore. Fetal bovine serum, MEM, DMEM,
streptomycin and penicillin were from Gibco; Thermo Fisher
Scientific, Inc. All other chemicals and solvents were of
analytical grade.
Cell lines and virus
CRC cell lines (LoVo, HCT116, RKO, SW480, SW1116,
Caco-2 and CT26) were purchased from the cell bank of the Chinese
Academy of Sciences. All CRC cells were cultured in DMEM
supplemented with 10% fetal bovine serum (FBS). Ovine fetal
turbinate (OFTu) cells were prepared as previously described
(20), and maintained in MEM
supplemented with 10% FBS. Orf virus (NA1/11) was isolated in our
previous study (21).
NA1/11∆132-GFP is a recombinant Orf virus with the deletion of the
ORFV132 gene by homologous recombination. OFTu cells were infected
with NA1/11 or NA1/11∆132-GFP (MOI =0.1) and were harvested when
approximately 80-90% of the cells exhibited cytopathogenic effects
(CPE). After repeated freezing and thawing, cellular debris was
removed by centrifugation at 800 × g for 5 min at 4°C, and
supernatants were purified by sucrose gradient ultracentrifugation.
Pellets were suspended in PBS (when used for animal research), or
MEM (when used for virus proliferation in cells) aliquots were
frozen at −80°C. Viral titers were obtained by the median tissue
culture infective dose (TCID50) method (22).
Preparation of NA1/11Δ132-GFP
recombinant ORFV
A virus recombinant transfer vector pSPV-EGFP with a
high expression of green fluorescent protein (GFP) was successfully
constructed (23). With pSPV-EGFP,
the plasmids ORFV132F-pSPV-EGFP-ORFV132R were constructed by
molecular cloning and used to transfect OFTu cells using
Lipofectamine 3000 at room temperature (Invitrogen; Thermo Fisher
Scientific, Inc.), after OFTu cells were infected with NA1/11 (MOI
=1) for 2 h. For each well of a 6-well plate, plasmid DNA and lipid
were mixed gently in 200 µl Opti-MEM media by adding 2.5 µg plasmid
DNA and 5 µl Lipofectamine Reagent and incubated for 20 min at room
temperature. It was necessary to replace the medium with fresh
medium 4-6 h after transfection. Screening for fluorescent viral
plaques was performed under a fluorescence microscope (Nikon
Eclipse E400; Nikon Corporation) at a magnification of ×100. After
15 rounds of plaque purification, NA1/11Δ132-GFP was obtained.
Wound-healing assay
Cells were seeded in 6-well plates at a density of
1×106 cells/well and cultured for 24 h. A wound was
scratched in the cell lawn with a sterile 200-µl pipette tip and
the cells were washed with serum-free DMEM three times to remove
debris. The cells were exposed to ORFV at designated multiplicities
of infection (MOI =0, 1, 5, 10). Images of wound healing
progression were captured at 24 h post infection under a light
microscope (Nikon Eclipse E400) at a magnification of ×100.
Crystal violet stain
CT26 and LoVo cells were seeded in 24-well plates at
a density of 1×104 cells/well and infected with ORFV at
designated multiplicities of infection (MOI =0, 1, 5, 10). Living
cells were stained with crystal violet solution (0.5% crystal
violet) at room temperature for 0, 24, 48 and 72 h. Cells were
washed with PBS three times to remove the debris and fixed with 500
µl methanol for 5 min at room temperature. After being washed with
PBS three times to remove methanol, the cells were stained with
crystal violet solution for 10 min at room temperature. The stain
was removed and the plates were washed thrice with PBS. Images of
the crystal violet stain were captured under a light microscope
(Nikon Eclipse E400) at a magnification of ×100.
CCK-8 assay
Cells were seeded at 1×103 cells/well in
a 96-well plate. Cells were exposed to designated multiplicities of
infection (MOI =0, 1, 5, 10) for various time-points (0, 24, 48 and
72 h). The cell supernatant was removed, the CCK-8 solution (10%
CCK-8 reagent diluted in DMEM supplemented with 10% FBS) was added
and the cells were incubated for 2 h at 37°C in an atmosphere of 5%
CO2. The optical density (OD) was measured at 450 nm
using an iMark microplate reader (iMark™; Bio-Rad Laboratories,
Inc.).
Reverse transcription-quantitative
(RT-q)PCR
Total RNA was extracted using TRIzol reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) and
reverse-transcribed to cDNA using the High Capacity cDNA Reverse
Transcription Kit (Thermo Fisher Scientific, Inc.). RT-qPCR was
carried out with Fast SYBR™ Master Mix (Thermo Fisher Scientific,
Inc.) on ABI StepOne Real-Time PCR platform according to the
manufacturer's instructions (Thermo Fisher Scientific, Inc.). The
primers used were as follows: E-cadherin forward,
5′-TACACTGCCCAGGAGCCAGA-3′ and reverse, 5′-TGGCACCAGTGTCCGGATTA-3′;
N-cadherin forward, 5′-ACCTGAACGACTGGGGGCCA-3′ and reverse,
5′-TGCCAAAGCCTCCAGCAAGCA-3′; β-actin forward,
5′-TGAAGGTGACAGCAGTCGGTTG-3′ and reverse,
5′-GGCTTTTAGGATGGCAAGGGAC-3′. The reaction protocol was 30 sec at
94°C, 43 cycles of 5 sec at 94°C and 34 sec at 60°C. Data were
analyzed as Relative Quantitation (RQ) with respect to a calibrator
sample using the 2−ΔΔCq method (24). The results were presented as the
mean ± SD from three experiments.
In vivo tumor model
In total, 12 healthy male Balb/c mice, aged from 6
to 8 weeks (body weight of 18±1.3 g), were purchased from the
Laboratory Animal Center of the Southern Medical University.
Animals were housed 6 per cage in the animal facility. The room
temperature was maintained constant at 22±1°C and the humidity was
controlled at 50-60% on a 12-h light/dark cycle. The animals had
access to food and water ad libitum. In the CRC mouse model,
CT26 cells (5×105 in 50 µl PBS) were subcutaneously
injected into Balb/c mice. When the tumor volume reached 25-30
mm3 [tumor volume (mm3)=(1/6) × π ×
a2 × b (a=width, b=length)], twelve mice were randomly
divided into two groups and administrated with the following agents
by intratumoral injection for a total of 4 injections in 12 days:
i) PBS control; and ii) ORFV (1×108 TCID50).
At the end of the experiment, animals were sacrificed by cervical
dislocation. Excised tumors were measured. The organs and tumor
tissues were fixed in 4% paraformaldehyde for subsequent IHC assay
at room temperature for 24 h. For mouse lung metastasis models,
5×105 CT26 cells were injected intravenously (i.v.) into
12 Balb/c mice. Mice were then treated i.v. with ORFV
(1×107 TCID50) in 100 µl of PBS, or
control-treated (100 µl PBS), on days 1, 3 and 8. At 11 days after
CT26 injection, mice were anesthetized with 60-120 µg/g Euthanyl
(Sigma-Aldrich; Merck KGaA) and their blood and lungs were
harvested. The serum was stored at −20°C for cytokine antibody
array. The lung tissues were fixed in 4% paraformaldehyde at room
temperature for 24 h for hematoxylin and eosin (H&E) staining,
respectively. All animal procedures were reviewed and approved by
the Institutional Animal Care and Use Committee at Foshan
University (Foshan, China) (approval no. 20170311).
Immunohistochemistry
Tumor tissues and organs isolated from the mice were
fixed with 4% paraformaldehyde at room temperature for 24 h,
embedded in paraffin, and sliced (5-µm thick). The slides of the
tumor tissues were immunohistochemically stained with cleaved
caspase-3 (1:2,000 dilution in antibody diluent) according to the
immunohistochemical (IHC) detection system kit manufacturer's
instructions (product no. BD5001; Bioworld Technology, Inc.).
Briefly, slides were heated at 55°C for 40 min and the dewaxing,
rehydration, antigen retrieval and blocking were performed. The
primary antibody was incubated at 4°C overnight. Immunostaining
continued with HRP-conjugated secondary antibody (product no.
BD5001; Bioworld Technology, Inc.) incubated at room temperature
for 10 min according to the manufacturer's instructions. The DAB
chomogen (product no. BD5001; Bioworld Technology, Inc.) was used
for incubation at room temperature for 4 min followed by washing.
The slides of the organs were stained with hematoxylin for 2 min
and dehydrated with ethanol and xylenes, mounted and cover slipped.
The images were obtained under a light microscope.
Flow cytometry
The flow cytometric assay was performed to detect
apoptosis in tumor cells exposed to ORFV. CT26 tumor cells were
seeded in a 6-well plate at a density of 1×106
cells/well and exposed to ORFV (MOI =10) or PBS for 24 h. The
collected cells were processed using Annexin V-APC/7-AAD Apoptosis
Detection Kit according to the manufacturer's instructions.
Apoptosis of the stained cells was detected using a LSRFortessa
flow cytometer (BD Biosciences). BD FACSDiva software 4.1 (BD
Biosciences) was used for analysis.
Western blot analysis
Cells exposed to ORFV were lysed in RIPA lysis
buffer (cat. no. P0013C; Beyotime Institute of Biotechnology)
containing 50 mM Tris (pH 7.4), 150 mM NaCl, 1% NP-40, 0.5% sodium
deoxycholate, 0.1% SDS and protein inhibitors, such as sodium
orthovanadate, sodium fluoride, EDTA and leupeptin. Levels of
proteins were measured by bicinchoninic acid assay. The lysis was
mixed with 1X SDS protein loading buffer and boiled for 15 min. For
each individual sample, 30 µg of protein were loaded per lane on
12% SDS polyacrylamide gels. Proteins were electro-transferred to
PVDF membranes at a constant 100 V for 60 min. The PVDF membranes
were blocked with 5% non-fat milk for 2 h at room temperature and
incubated with primary antibody (with 1:1000 dilution, according to
the manufacturer's protocol) overnight at 4°C. The PVDF membranes
were washed with TBST five times for 5 min per wash and incubated
with HRP-conjugated antibodies, including goat anti-rabbit IgG
H&L (HRP) (product code ab6721; 1:10,000 dilution) and goat
anti-mouse IgG H&L (HRP) (product code ab6789; 1:2,000
dilution; both from Abcam) for 1 h at room temperature. The
membranes were washed with TBST again and visualized with ECL
chemiluminescence reagent (Tanon Science and Technology Co., Ltd.)
using photographic film.
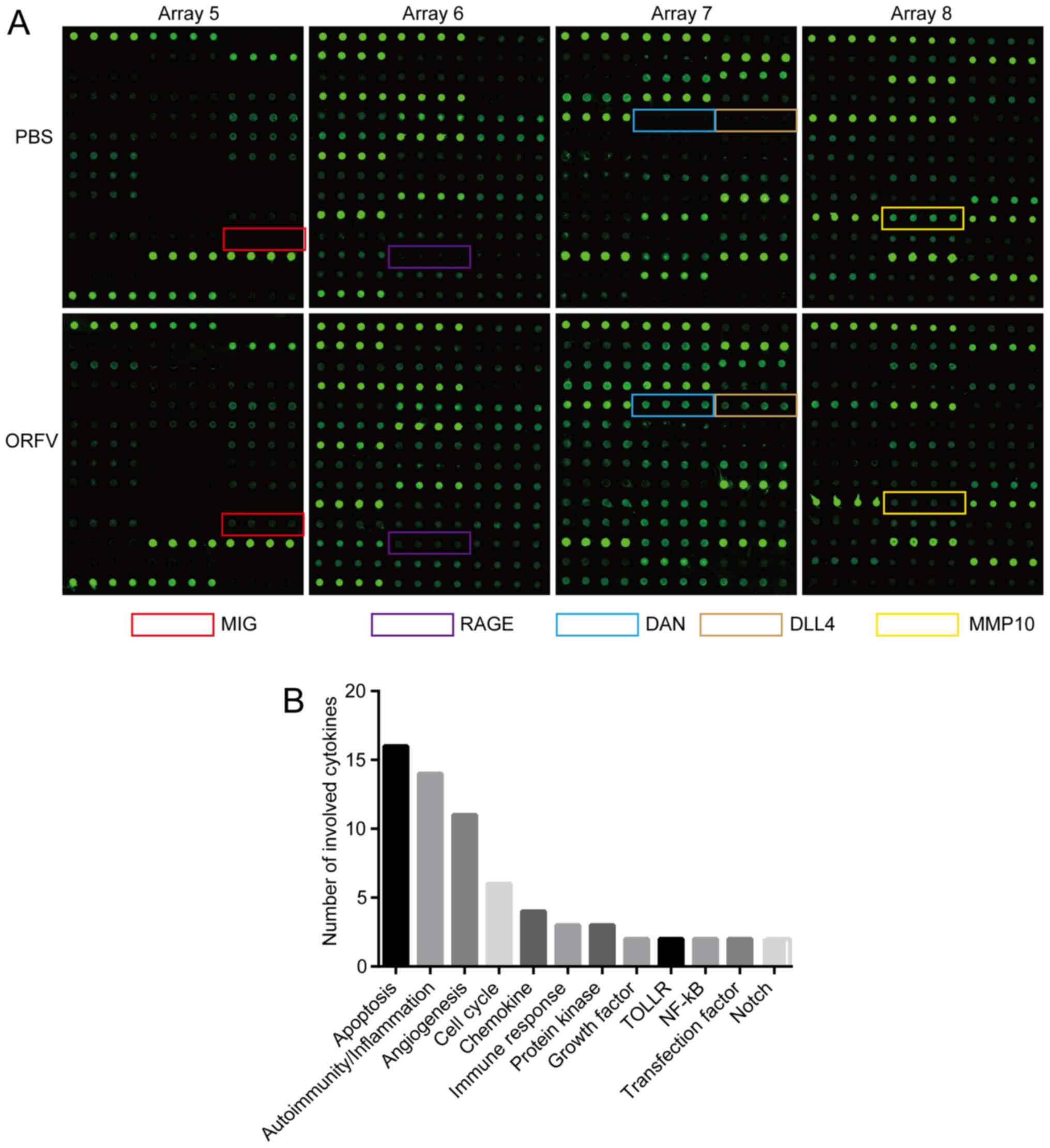
Cytokine antibody array
Mouse sera of the ORFV-treated and PBS-treated
subjects were collected and measured, according to the
manufacturer's instructions, with a semi-quantitative mouse
cytokine antibody array (RayBio Human Cytokine Antibody Array G
series 4000; RayBiotech, Inc.) which detects 200 cytokines in one
experiment. Briefly, the samples were added to the cytokine
antibody chips, then incubated with the biotinylated antibody. Cy3
Equivalent Dye-Streptavidin was added, and the fluorescence signals
were visualized through a laser scanner equipped with a Cy3
wavelength (InnoScan 300 Microarray Scanner; Innopsys). The mean
fluorescent signals of each group containing three repeats were
analyzed with specific software Q-Analyzer Tool (code:
QAM-CAA-4000) provided by RayBiotech, Inc. The cytokines (FC ≥2 or
FC ≤0.5; Table I) were noted by
functional category, comprehensively referring to the COSMIC
database (http://cancer.sanger.ac.uk/cosmic/), GeneCards
database (http://www.genecards.org/), NCBI Gene
database (https://www.ncbi.nlm.nih.gov/gene/), and references
(25–31).
 | Table I.Differentially expressed cytokines
(fold change ≥2 or ≤0.5) were associated with regulation of
apoptosis, angiogenesis, autoimmunity/inflammation, and the cell
cycle after the ORFV infection. |
Table I.
Differentially expressed cytokines
(fold change ≥2 or ≤0.5) were associated with regulation of
apoptosis, angiogenesis, autoimmunity/inflammation, and the cell
cycle after the ORFV infection.
| Cytokine | F-value | P-value | Fold change | Mean
(ORFV-infected, n=3) | Mean (control,
n=3) | Functional
classification |
|---|
| IL-15 | 0.000 | 0.184 | 2,189.610 | 827.6 | 0.4 | Apoptosis,
angiogenesis, and autoimmunity/inflammation |
| IL-13 | 0.000 | 0.342 | 1,869.543 | 402.6 | 0.2 |
Autoimmunity/inflammation |
| RAGE | 0.000 | 0.190 | 562.576 | 49.2 | 0.1 | MAPK signaling,
ERK1/2 and p53/TP53 signaling and NF-κB |
| EDAR | 0.000 | 0.282 | 372.656 | 3,807.4 | 10.2 | Apoptosis and
NF-κB |
| MIG | 0.000 | 0.114 | 353.760 | 359.9 | 1.0 | Transfection factor
and TOLLR |
| TRAIL | 0.000 | 0.250 | 289.072 | 321.7 | 1.1 | Apoptosis,
angiogenesis, and cell cycle |
| BLC (CXCL13) | 0.000 | 0.133 | 219.855 | 111.9 | 0.5 | Apoptosis and
autoimmunity/inflammation |
| IL-7 | 0.000 | 0.306 | 200.395 | 185.2 | 0.9 | Apoptosis and
autoimmunity/inflammation |
| CD27 | 0.000 | 0.080 | 185.302 | 471.6 | 2.5 | Apoptosis,
Autoimmunity/inflammation, and cell cycle |
| Tryptase ε | 0.000 | 0.197 | 180.294 | 1,374.2 | 7.6 | Immune
response |
| CCL6 | 0.000 | 0.117 | 156.765 | 2,374.0 | 15.1 | Chemokine |
| DLL4 | 0.002 | 0.018 | 64.517 | 6,938.8 | 107.6 | Angiogenesis,
Notch |
| Fractalkine | 0.001 | 0.370 | 48.519 | 15,760.2 | 324.8 | Immune
response |
| TARC | 0.001 | 0.198 | 36.512 | 56.8 | 1.6 |
Autoimmunity/inflammation |
| DAN | 0.000 | 0.184 | 34.164 | 2,324.7 | 68.0 |
Autoimmunity/inflammation and cell
cycle |
| VEGF-D | 0.001 | 0.317 | 32.318 | 46.4 | 1.4 | Notch,
angiogenesis, and cell cycle |
| OX40 ligand | 0.002 | 0.205 | 25.443 | 245.5 | 9.6 | Apoptosis,
autoimmunity/inflammation |
| Prostasin | 0.013 | 0.258 | 20.023 | 2,087.1 | 104.2 | Trypsin-like
cleavage specificity |
| I-TAC | 0.063 | 0.071 | 11.777 | 96.6 | 8.2 | Transfection
factor, TOLLR, apoptosis, angiogenesis, and
autoimmunity/inflammation |
| Leptin | 0.006 | 0.429 | 11.208 | 960.1 | 85.7 | Apoptosis,
autoimmunity/inflammation, and angiogenesis |
| TIM-1 | 0.071 | 0.192 | 9.311 | 111.9 | 12.0 | Immune
response |
| IL-21 | 0.299 | 0.002 | 9.142 | 91.7 | 10.0 |
Autoimmunity/inflammation |
| TREM-1 | 0.272 | 0.021 | 9.061 | 42.5 | 4.7 | Chemokine |
| H60 | 0.089 | 0.044 | 7.084 | 95.2 | 13.4 | MHC I |
| TPO | 0.370 | 0.017 | 6.500 | 192.7 | 29.6 | Growth factor |
| KC | 0.551 | 0.002 | 5.892 | 234.5 | 39.8 | Angiogenesis,
autoimmunity/inflammation |
| TACI | 0.006 | 0.150 | 5.619 | 78.9 | 14.0 | Apoptosis |
| ALK-1 | 0.638 | 0.005 | 3.444 | 67.2 | 19.5 | Protein kinase |
| BAFF R | 0.058 | 0.004 | 3.272 | 100.8 | 30.8 | Immune
response |
| Osteoactivin | 0.291 | 0.035 | 3.211 | 1,234.1 | 384.4 | Growth factor |
| Pentraxin 3 | 0.012 | 0.164 | 3.115 | 5,956.9 | 1,912.4 | Apoptosis and
autoimmunity/inflammation |
| Granzyme B | 0.131 | 0.096 | 2.767 | 406.0 | 146.7 | Apoptosis and
autoimmunity/inflammation |
| Prolactin | 0.805 | 0.069 | 2.419 | 2,615.6 | 1,081.4 | Insulin1 and
apoptosis |
| VEGF R1 | 0.841 | 0.007 | 2.236 | 942.3 | 421.4 | Angiogenesis and
protein kinase |
| Neprilysin | 0.698 | 0.081 | 2.184 | 1,059.8 | 485.2 | Apoptosis and
angiogenesis |
| CD30 | 0.049 | 0.190 | 2.152 | 110.5 | 51.4 | Apoptosis and
angiogenesis |
| Axl | 0.874 | 0.093 | 0.469 | 682.5 | 1,455.8 | Protein kinase,
apoptosis, and cell cycle |
| CXCL16 | 0.252 | 0.010 | 0.389 | 28.8 | 74.2 | Chemokine |
| ANG-3 | 0.967 | 0.031 | 0.369 | 120.1 | 325.4 | Tyrosine kinase
signaling pathway |
| MMP10 | 0.630 | 0.035 | 0.369 | 39.6 | 107.4 | Tumor
metastasis |
| IFN-γ R1 | 0.225 | 0.034 | 0.316 | 5.3 | 16.8 | The JAK/STAT
signaling pathway |
| VEGF-B | 0.063 | 0.027 | 0.080 | 9.1 | 113.0 | Angiogenesis and
cell cycle |
Statistical analysis
SPSS software version 17.0 (SPSS, Inc.) was used for
all analysis. Results are expressed as the means ± SEM. One-way
ANOVA was performed to test the null hypothesis of group
differences with post hoc tests (Dunnett's test) and unpaired
Student's t-test was also performed. P<0.05 was considered to
indicate a statistically significant difference. The F-value, which
represents the significance of the whole fitting equation and was
used to assess the differences between groups, was analyzed using
One-way ANOVA.
Results
ORFV strain NA1/11 infects CRC cells
and inhibits proliferation
We used the ORFV strain NA1/11, isolated from an
outbreak in northeast China, to infect human CRC cells (LoVo,
Caco-2, HCT116, RKO, SW480 and SW1116) with multiplicities of
infection (MOI) of 0 (mock), 1, 5, 10. After 24 h, cytopathic
lesions were observed in some CRC cells, including rounding and
swelling (Fig. S1). A recombinant
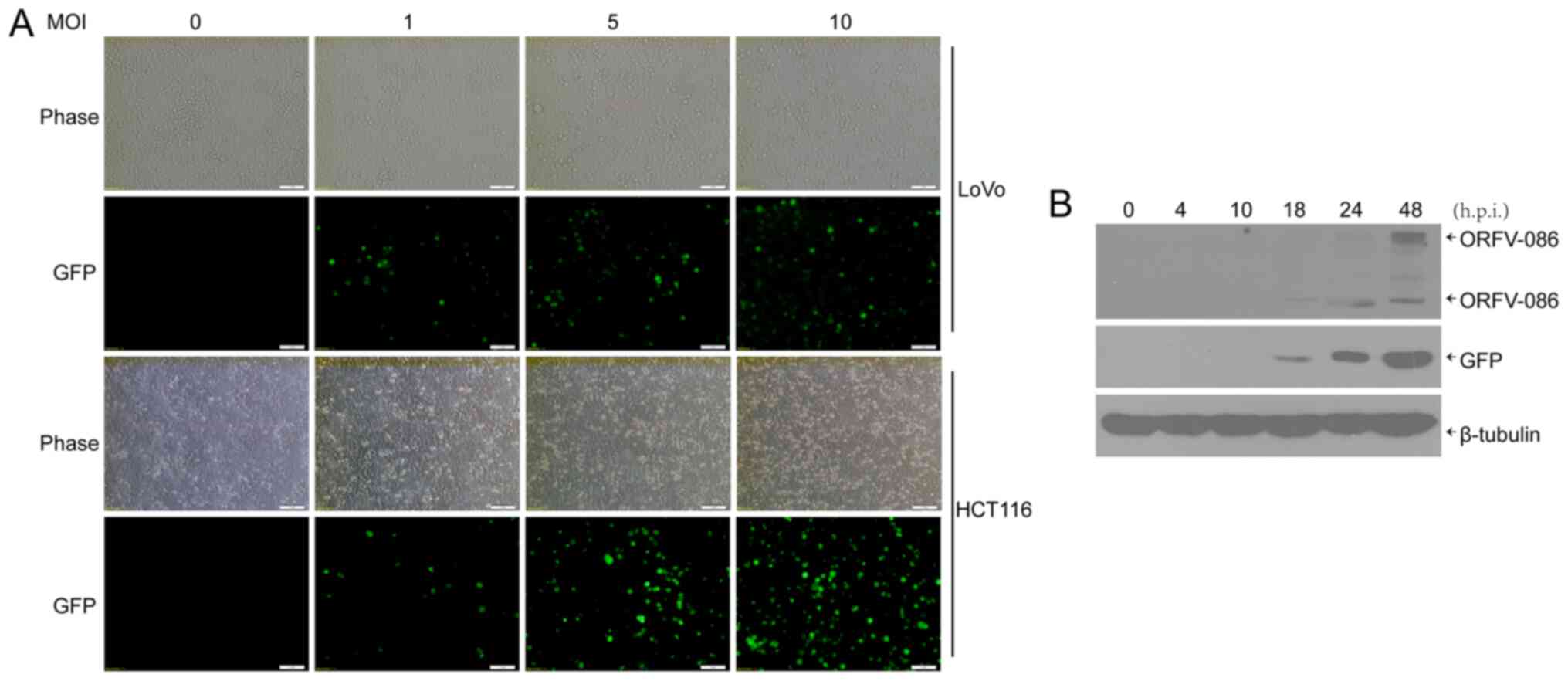
orf virus, NA1/11∆132-GFP, was used to infect human CRC cells.
Then, 24 h post infection (h.p.i.), GFP was visible in the CRC
cells (Fig. 1A). In addition, it
was determined, via western blotting, that both viral protein
ORFV086, which is a proteolytic protein, and GFP reporter protein
were expressed at 18 h.p.i in the CRC cells (Fig. 1B).
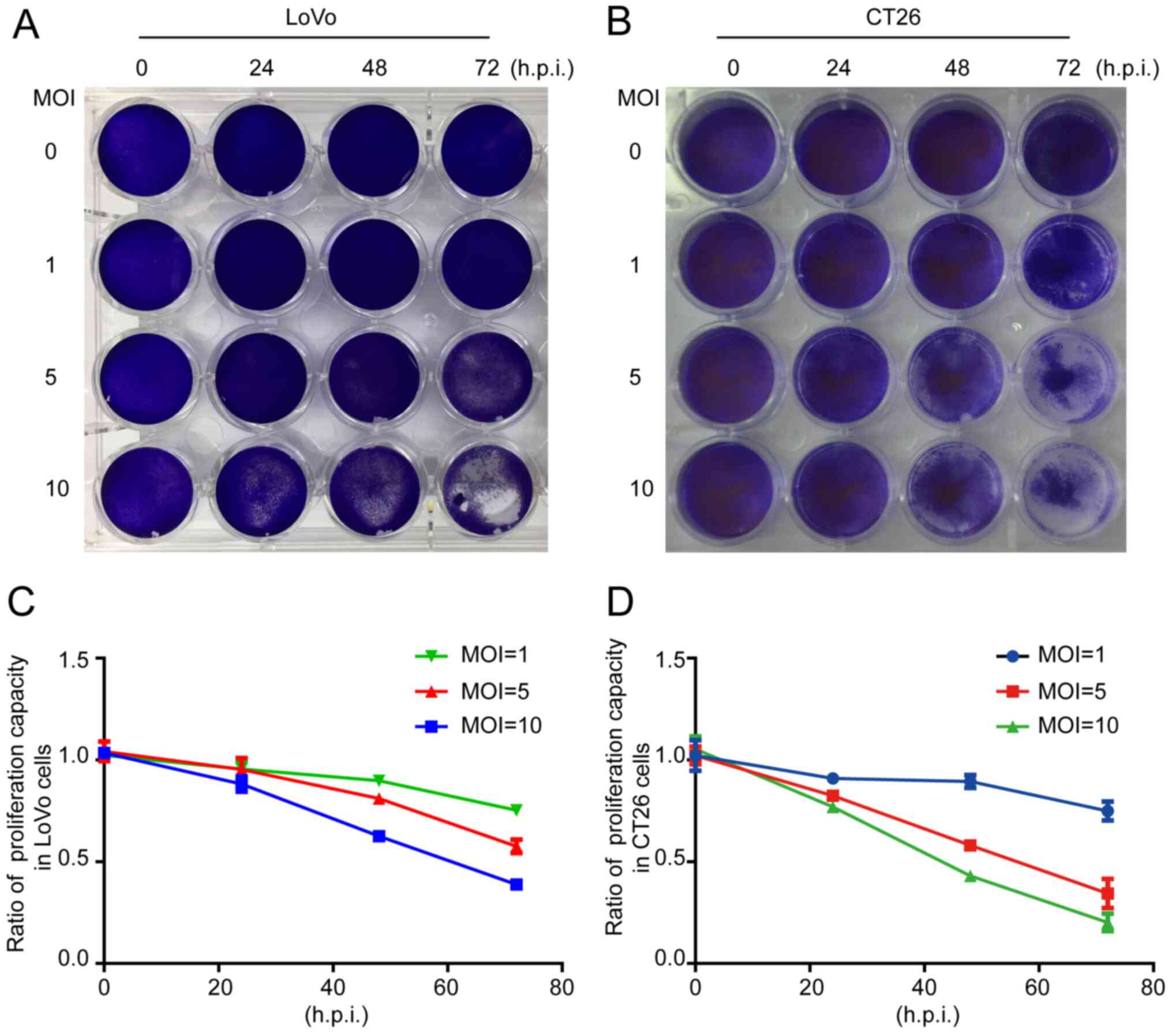
The crystal violet assay revealed that the total
number of cells was decreased when LoVo or CT26 cells were infected
by ORFV (Fig. 2A and B). To
investigate whether inhibition of proliferation was involved in
response to ORFV, CCK-8 assays were performed (Fig. 2C and D). ORFV inhibited the
proliferation of LoVo and CT26 in a dose-dependent manner. Notably,
the inhibitory effect was not obvious at 24 h.p.i., but
increasingly significant at 48 and 72 h.p.i. ORFV also inhibited
the proliferation of Caco-2, HCT116, RKO, SW480 and SW1116 cells
(Fig. S2).
ORFV strain NA1/11 inhibits the
migration of CRC cells
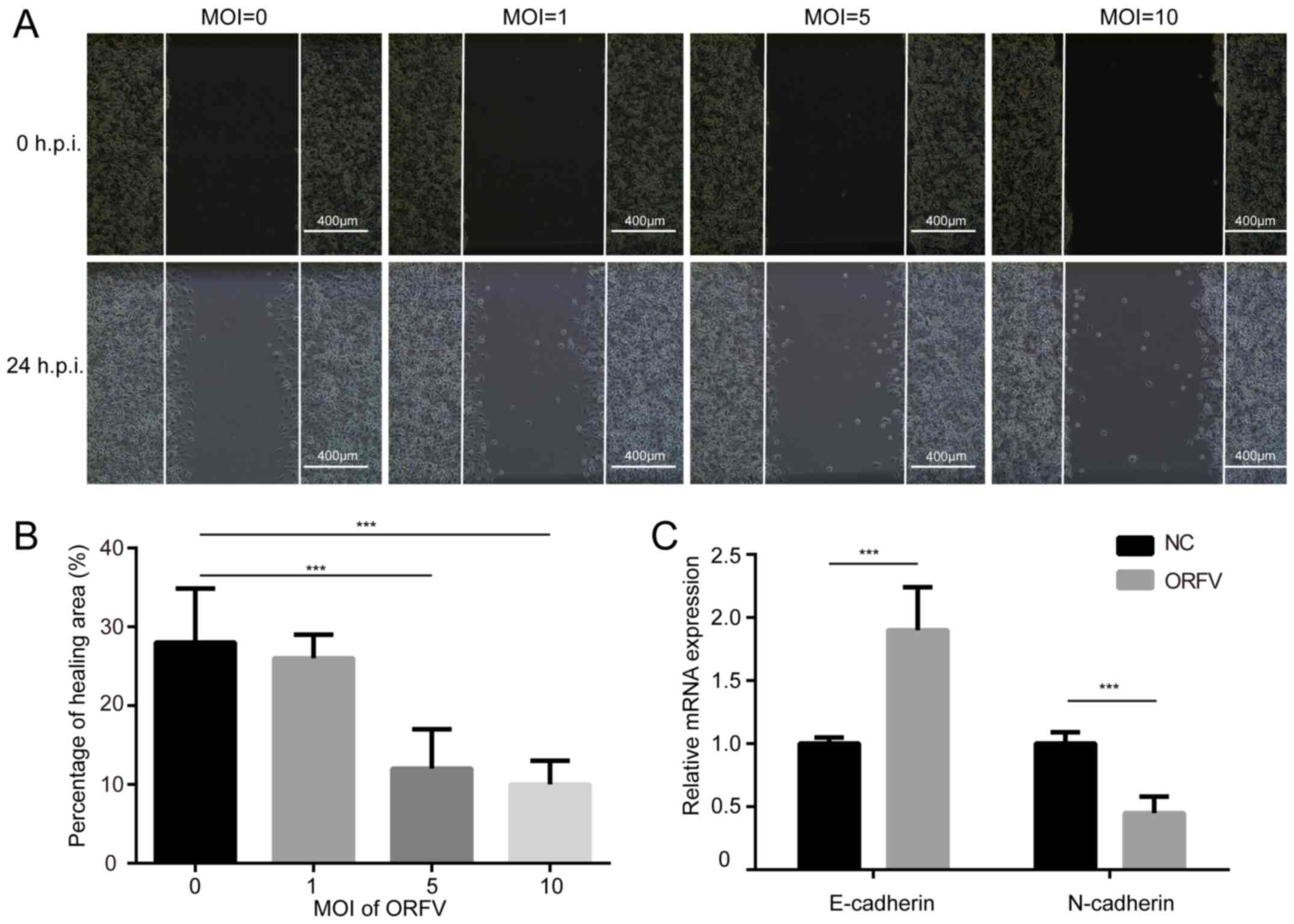
The influence of ORFV on the migration of LoVo and
CT26 cells was assessed via a wound healing assay. The cells were
cultured at high density in medium containing 10% FBS. After 24 h
of serum-starvation, a scratch was made in the confluent cells and
the cells incubated for an additional 24 h in medium containing 10%
FBS, with or without ORFV treatment. ORFV decreased the migration
of the cells into the denuded area, compared to the control
(P<0.05; Figs. 3A, B and
S3A, B). However, the
morphological changes and wound healing assay differences among
multiple MOIs of virus infection were small, probably due to
insufficient infection time.
Cadherin is associated with tumor progression and
metastasis (32). E-cadherin
homophilic binding can lead to contact-mediated inhibition of
growth, thus the loss of E-cadherin expression is often associated
with tumor metastasis (33). On the
contrary, N-cadherin promotes the metastatic behavior of tumor
cells by directly mediating cell-cell adhesion (34). Thus, it was examined whether
E-cadherin and N-cadherin are involved in increased cytotoxicity of
ORFV. As anticipated, ORFV treatment caused a significant increase
in the expression of E-cadherin. A reduction of N-cadherin was
observed in CRC cells incubated with ORFV (Figs. 3C and S3C), indicating that ORFV regulated the
expression of cadherins to inhibit the migration of CRC cells.
ORFV strain NA1/11 reduces in vivo
tumor growth and metastasis
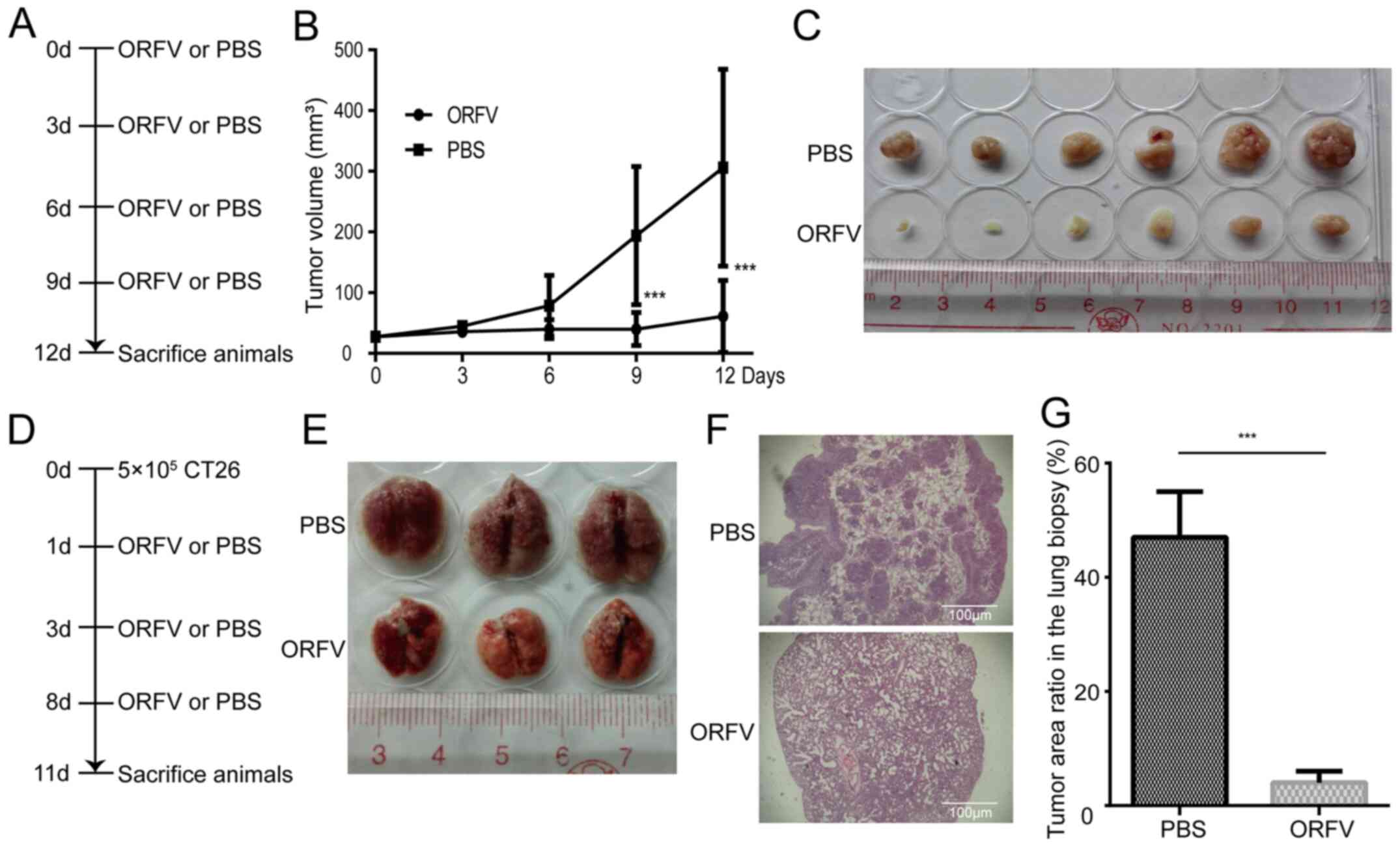
To investigate the tumoricidal activity of ORFV
strain NA1/11 in CRC cells, CT26 cells were injected subcutaneously
into BALB/c mice for the development of transplanted tumors. When
the tumor size was 25-30 mm3, the mice were treated with
ORFV or PBS (Fig. 4A). Consistent
with the in vitro results, ORFV strain NA1/11 significantly
inhibited in vivo tumor growth (Fig. 4B) and reduced the tumor size
(Fig. 4C). Compared to the
PBS-treated group, the average tumor volume decreased significantly
in the ORFV-treated group. In addition, CT26 cells were injected
into the tail vein of Balb/c mice for the development of a CRC lung
metastasis model (Fig. 4D). After
three ORFV or PBS treatments, the lungs were harvested (Fig. 4E) and subjected to H&E staining.
The average tumor area ratio in lung biopsies of the ORFV-treated
group significantly decreased in comparison with those from
PBS-treated group (Fig. 4F and
G).
ORFV strain NA1/11 alters cytokine
secretion
To explore the mechanism of ORFV inhibition of
cancer cell growth and metastasis, the ORFV-induced cytokine
expression was detected via cytokine antibody chip analysis. After
three ORFV or PBS treatments, mice sera were collected from lung
metastasis models on the 11th day (Fig.
4D) and subjected to semi-quantitative cytokine antibody chip
assays with 200 cytokines.
The detection images (Fig. 5A) were scanned and converted to a
concentration unit. Fold-changes between the ORFV-treated and
PBS-treated groups were calculated. A total of 42 cytokines (FC ≥2
or FC ≤0.5; Table I) were sorted
into different clusters according to functional annotation, and the
forehead clusters of cytokines with the largest numbers were
autoimmunity/inflammation, apoptosis, angiogenesis, and the cell
cycle. The fold-change sorting of these cytokines and brief
functional annotations are presented in Fig. 5B. Notably, for the individual
differences in the mice, the results of cytokine antibody chips
array revealed that a variety of cytokines with large fold changes
were not statistically significant (Table I). The results of cytokine antibody
chip assay indicated that NA1/11 may play roles in those cytokines
and direct the preliminary direction for our future research. These
cytokines must be further confirmed in additional studies.
ORFV strain NA1/11 induces CRC cell
apoptosis
Among differentially expressed cytokines screened by
cytokine antibody chip analysis, apoptosis-related cytokines were
in the majority. There were 15 upregulated cytokines and 1
downregulated cytokine related to apoptosis in the ORFV-treated
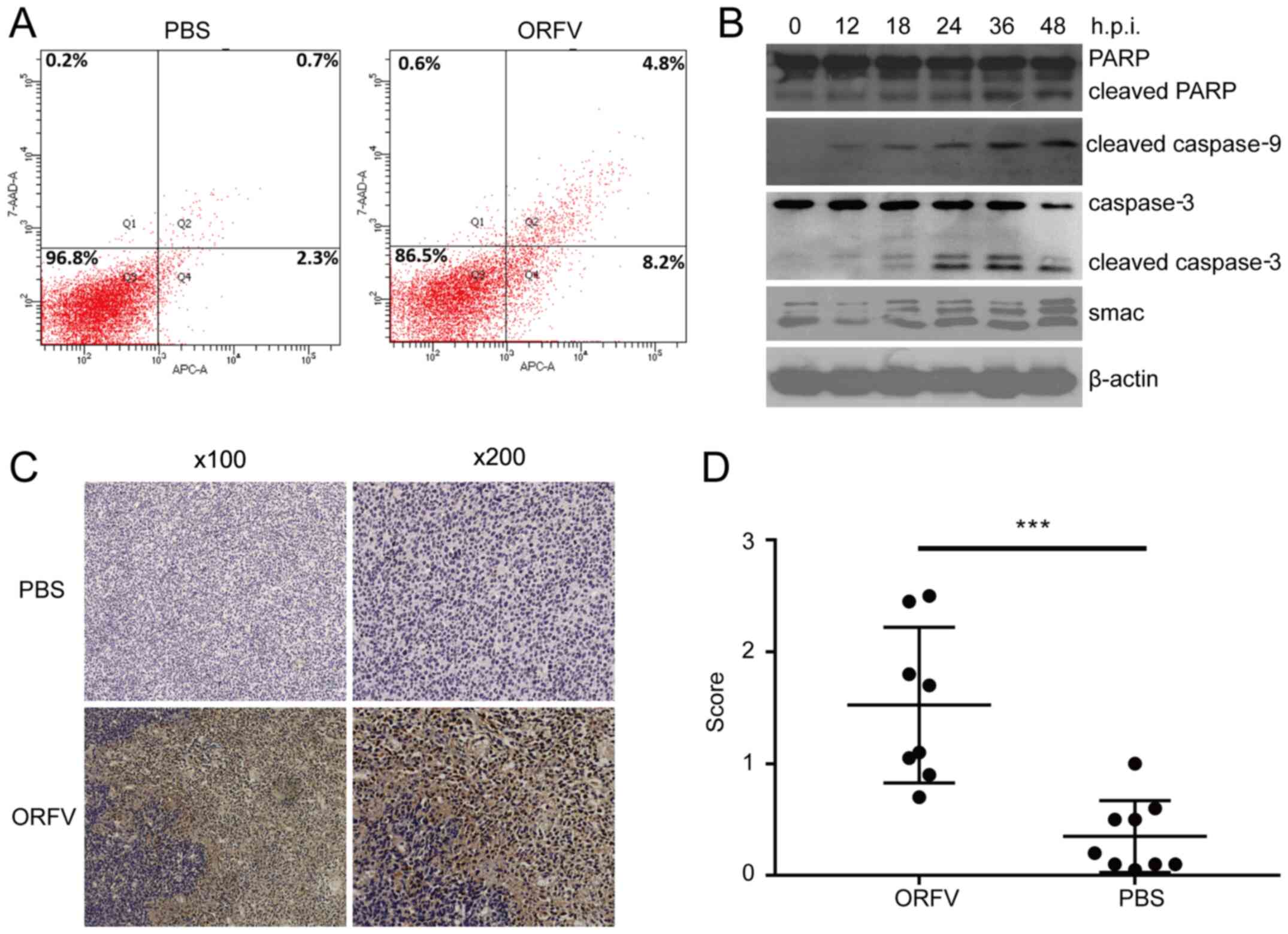
group. Thus, apoptosis was examined by three distinct approaches.
Using flow cytometry, apoptotic cells were revealed to be
significantly increased in the ORFV-stimulated group compared to
the PBS-treated group (Fig. 6A).
Western blot analysis revealed that apoptosis-regulated proteins
Smac, cleaved caspase-3, cleaved caspase-9 and cleaved PARP were
significantly enhanced in LoVo cells incubated with ORFV virus from
0 to 48 h.p.i. (Fig. 6B).
To examine whether the antitumor activity of ORFV
was due to the potentiation of apoptosis in vivo, the tumor
tissues from the mouse model were analyzed by IHC assay. Consistent
with the in vitro results, the expression of cleaved
caspase-3 in tumor tissues from the ORFV-treated group was
significantly increased compared to that of tumor tissues from the
PBS-treated groups (Fig. 6C and
D).
Discussion
There are some unique biological characteristics
which make ORFV an attractive oncolytic virus vector. First, the
ORFV genes are replicated and transcribed in the cytoplasm rather
than integrated into the host genome, and thus ORFV is considered
relatively safe to develop as an oncolytic virus. Second, the host
range of ORFV infection is constricted, its damage is limited
mainly to the skin that can recover quickly, and the virus has low
toxicity and shows little evidence of systemic transmission. Third,
ORFV infection can rapidly mediate humoral and adaptive immune
responses, but little neutralizing serum antibody is produced after
stimulation by ORFV. Furthermore, attenuated virus strains can be
constructed by knockout of the virulence genes. As a viral vaccine
vector, the gene compatibility is large, and it can replicate and
express exogenous genes. Finally, ORFV infection can cause the
release of immunoregulatory molecules, including IFN, IL-2, and
TNF. Therefore, ORFV has the potential to be an ideal oncolytic
virus vector (9,10,17,19,35,36).
Although ORFV strain NZ2 has been reported to be
able to induce anticancer effects in murine cancer models (11), it is necessary to verify the
antitumor effect of the ORFV strain NA1/11 for its high variability
in the terminal regions of the genome among different strains
(37). In the present study, it was
first confirmed that ORFV strain NA1/11 could infect and cause
typical cytopathic lesions in CRC cell lines. Then, CCK-8 assay
indicated that ORFV strain NA1/11 could suppress the proliferation
of CRC cells. Furthermore, it was revealed that ORFV strain NA1/11
significantly inhibited the migration of CRC cells. ORFV strain
NA1/11 inhibited tumor proliferation and metastasis in vivo.
A cytokine antibody chip assay was performed to explore the
potential mechanism of ORFV, which revealed that apoptosis may play
an important role in ORFV-induced cytotoxicity.
Cytokine antibody chip assay revealed the general
profile of cytokine regulation. According to functional
classification, four groups containing the most numerous cytokines
were apoptosis, autoimmunity/inflammation, angiogenesis, and the
cell cycle. Some of the cytokines, including IL-15, IL-21, CD27,
VEGF-B, delta-like canonical Notch ligand 4 (DLL4), are pleiotropic
cytokines that likely play important roles in apoptosis and
autoimmunity/inflammation. It is possible that cytokines, such as
CCL6, TREM-1, CXCL16, could affect chemotactic activity, inhibition
of tumor metastasis or regulation of the NF-κB pathway. More
detailed studies are required to depict an integrated regulation
network of the ORFV-mediated oncolysis.
Interleukins (ILs) were first discovered to be
expressed by leukocytes, which extensively influence the functions
of the immune system, such as promoting the development and
differentiation of T and B lymphocytes and hematopoietic cells
(26). In the present study, IL-15
was the cytokine most markedly altered in the microarray assay,
with a fold-change of more than 2000. This cytokine and IL-2 share
numerous common functional characteristics, including regulating
the activation and proliferation of T cells and NK cells (27,31).
IL-15 can also induce the activation of JAK kinases. IL-21 is
another interleukin whose expression levels were upregulated in
this assay, with a fold-change of 9.142, and plays a role in both
the innate and adaptive immune responses. It can induce the
differentiation, proliferation, and activity of various immune
cells, including NK cells, macrophages, cytotoxic T cells and B
cells, and functions in synergy with IL-15 in some of these
regulatory roles (31). IL-21 can
stimulate IFN production in T cells and NK cells, and inhibits
activation and maturation of DC within T-cell mediated immune
responses (28). Analogously, IL-7
may facilitate the functions of the immune system through a similar
mechanism (29). These results
indicated that interleukins may play a crucial role in the
oncolytic effect of ORFV, possibly through regulating the
proliferation and activation of T cells, NK cells and other immune
cells. CD27, a member of the TNF-receptor superfamily, plays a
crucial role in regulating B-cell activation and immunoglobulin
synthesis via binding to ligand CD70, and can exert
apoptosis-promoting effects through association with the
proapoptotic CD27-binding protein (SIVA) (25, 30).
Angiogenesis plays an important role in tumor tissue
development and thus has become a key target in various tumor
molecular therapy strategies (38,39).
The most downregulated cytokine in the microarray assay was VEGF-B,
a member of the vascular endothelial growth factor (VEGF) family,
which can regulate the formation of blood vessels (angiogenesis)
and is involved in the growth of endothelial cells (40). DLL4 is related to the Notch
signaling pathway and can negatively regulate the proliferation,
migration, and angiogenic sprouting of endothelial cells (41).
Chemokines are a family of small cytokines able to
induce directed chemotaxis in adjacent responsive cells, and are
classified into four main subfamilies: CXC, CC, CX3C and XC
(42). In the present study, the
microarray assay results indicated three chemokines, CCL6, TREM-1
and CXCL16, that may take part in ORFV-mediated oncolysis in cancer
cells (43–45). For instance, TREM-1 can strengthen
neutrophil and monocyte-mediated inflammatory responses, which are
triggered by promoting the release of pro-inflammatory chemokines
and other cytokines (44). The
receptor of advanced glycosylation product (RAGE), a multi-ligand
cell surface receptor, was markedly upregulated when cancer cells
were treated with ORFV. It can also interact with various molecules
involved in homeostasis, development, and inflammation. The MAPK
signaling, ERK1/2 signaling, p53/TP53 signaling and NF-κB signaling
pathways are important pathways in which RAGE functions (46,47).
The cytokine matrix metallopeptidase 10 (MMP10), involved in tissue
remodeling and in certain disease processes, is the only cytokine
identified to be related to tumor metastasis (48).
Apoptosis is an important cellular process induced
or inhibited by viral infection. Apoptosis plays an important role
in the antitumor effect (49), and
previous studies suggested that apoptosis contributes to the
oncolytic effect induced by oncolytic viruses (1,50,51).
In the present study, there were 15 upregulated cytokines and 1
downregulated cytokine related to apoptosis in the ORFV strain
NA1/11-treated group, consistent with literature studies (30,52–55).
In conclusion, the present results indicated that
ORFV strain NA1/11 could inhibit CRC growth and metastasis by
inducing apoptosis of CRC cells. In addition, ORFV NA1/11s could
upregulate serum levels of IL-7, IL-13, IL-15, CD27, CD30,
pentraxin 3, and B lymphocyte chemoattractant (BLC or CXCL13) and
downregulate Axl, CXCL16, ANG-3, MMP10, IFN-γ R1 and VEGF-B,
leading to enhanced oncolytic activity.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the
National Natural Science Foundation of China (NSFC) (grant nos.
81773271 and 31672536), the Key Projects of Basic and Applied
Research (Natural Science Class), the Department of Education,
Guangdong Province (grant nos. 2017KZDXM088 and 2018KQNCX284), the
Joint Fund of Basic and Applied Basic Research Fund of Guangdong
Province (grant no. 2019A1515110689), the Foshan University
High-level University Fund (grant no. 20170131020) and the Foshan
University Senior Talent Start Fund (grant no. 20161110004). The
funders had no role in the study design, data collection and
analysis, decision to publish or preparation of the manuscript.
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
WH, DC and SL conceived and designed the study. DC,
RW, ML, WL, BX, HD, KW, DG and FL performed the experiments. DC,
RW, ML, and SL analyzed the data. DC, RW, WH and SL wrote the
manuscript. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
All animal procedures were reviewed and approved by
the Institutional Animal Care and Use Committee at Foshan
University (Foshan, China) (approval no. 20170311).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CRC
|
colorectal cancer
|
|
ORFV
|
Orf virus
|
|
NK
|
natural killer
|
|
DC
|
dendritic cells
|
|
IL-1RA
|
IL-1 receptor antagonist
|
|
MOI
|
multiplicities of infection
|
|
OD
|
optical density
|
|
RAGE
|
receptor of advanced glycosylation end
product
|
|
MMP10
|
matrix metallopeptidase 10
|
|
ECL
|
enhanced chemiluminescence
|
|
PVDF
|
polyvinylidene difluoride
|
|
FBS
|
fetal bovine serum
|
|
OFTu
|
ovine fetal turbinate
|
|
VEGF
|
vascular endothelial growth factor
|
|
DLL4
|
delta-like canonical Notch ligand
4
|
|
BLC
|
B lymphocyte chemoattractant
|
References
|
1
|
Lawler SE, Speranza MC, Cho CF and Chiocca
EA: Oncolytic viruses in cancer treatment: A review. JAMA Oncol.
3:841–849. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kaufman HL, Kohlhapp FJ and Zloza A:
Oncolytic viruses: A new class of immunotherapy drugs. Nat Rev Drug
Discov. 14:642–662. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mondal M, Guo J, He P and Zhou D: Recent
advances of oncolytic virus in cancer therapy. Hum Vaccin
Immunother. 16:2389–2402. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Raja J, Ludwig JM, Gettinger SN, Schalper
KA and Kim HS: Oncolytic virus immunotherapy: Future prospects for
oncology. J Immunother Cancer. 6:1402018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu Z, Ravindranathan R, Kalinski P, Guo
ZS and Bartlett DL: Rational combination of oncolytic vaccinia
virus and PD-L1 blockade works synergistically to enhance
therapeutic efficacy. Nat Commun. 8:147542017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Andtbacka RH, Kaufman HL, Collichio F,
Amatruda T, Senzer N, Chesney J, Delman KA, Spitler LE, Puzanov I,
Agarwala SS, et al: Talimogene laherparepvec improves durable
response rate in patients with advanced melanoma. J Clin Oncol.
33:2780–2788. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hosamani M, Scagliarini A, Bhanuprakash V,
McInnes CJ and Singh RK: Orf: An update on current research and
future perspectives. Expert Rev Anti Infect Ther. 7:879–893. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang R, Wang Y, Liu F and Luo S: Orf
virus: A promising new therapeutic agent. Rev Med Virol.
29:e20132019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fiebig HH, Siegling A, Volk HD, Friebe A,
Knolle P, Limmer A and Weber O: Inactivated orf virus
(Parapoxvirus ovis) induces antitumoral activity in
transplantable tumor models. Anticancer Res. 31:4185–4190.
2011.PubMed/NCBI
|
|
10
|
van Rooij EM, Rijsewijk FA, Moonen-Leusen
HW, Bianchi AT and Rziha HJ: Comparison of different prime-boost
regimes with DNA and recombinant Orf virus based vaccines
expressing glycoprotein D of pseudorabies virus in pigs. Vaccine.
28:1808–1813. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rintoul JL, Lemay CG, Tai LH, Stanford MM,
Falls TJ, de Souza CT, Bridle BW, Daneshmand M, Ohashi PS, Wan Y,
et al: ORFV: A novel oncolytic and immune stimulating parapoxvirus
therapeutic. Mol Ther. 20:1148–1157. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Friebe A, Friederichs S, Scholz K, Janssen
U, Scholz C, Schlapp T, Mercer A, Siegling A, Volk HD and Weber O:
Characterization of immunostimulatory components of orf virus
(parapoxvirus ovis). J Gen Virol. 92:1571–1584. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tai LH, de Souza CT, Bélanger S, Ly L,
Alkayyal AA, Zhang J, Rintoul JL, Ananth AA, Lam T, Breitbach CJ,
et al: Preventing postoperative metastatic disease by inhibiting
surgery-induced dysfunction in natural killer cells. Cancer Res.
73:97–107. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Breitbach CJ, Paterson JM, Lemay CG, Falls
TJ, McGuire A, Parato KA, Stojdl DF, Daneshmand M, Speth K, Kirn D,
et al: Targeted inflammation during oncolytic virus therapy
severely compromises tumor blood flow. Mol Ther. 15:1686–1693.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cai J, Lin Y, Zhang H, Liang J, Tan Y,
Cavenee WK and Yan G: Selective replication of oncolytic virus M1
results in a bystander killing effect that is potentiated by Smac
mimetics. Proc Natl Acad Sci USA. 114:6812–6817. 2017.PubMed/NCBI
|
|
16
|
Warner SG, Haddad D, Au J, Carson JS,
O'Leary MP, Lewis C, Monette S and Fong Y: Oncolytic herpes simplex
virus kills stem-like tumor-initiating colon cancer cells. Mol Ther
Oncolytics. 3:160132016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Weber O, Mercer AA, Friebe A, Knolle P and
Volk HD: Therapeutic immunomodulation using a virus - the potential
of inactivated orf virus. Eur J Clin Microbiol Infect Dis.
32:451–460. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Haig DM and McInnes CJ: Immunity and
counter-immunity during infection with the parapoxvirus orf virus.
Virus Res. 88:3–16. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen D, Long M, Xiao B, Xiong Y, Chen H,
Chen Y, Kuang Z, Li M, Wu Y, Rock DL, et al: Transcriptomic
profiles of human foreskin fibroblast cells in response to orf
virus. Oncotarget. 8:58668–58685. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang Y, Tong S, Li W, Gao F, Ning Z and
Luo S: In vitro cultivation of primary ovine fetal turbinate cells
and its application in researches of ovine orf virus. Zhongguo
Shouyi Kexue. 43:470–475. 2013.(In Chinese).
|
|
21
|
Li W, Ning Z, Hao W, Song D, Gao F, Zhao
K, Liao X, Li M, Rock DL and Luo S: Isolation and phylogenetic
analysis of orf virus from the sheep herd outbreak in northeast
China. BMC Vet Res. 8:2292012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
LaBarre DD and Lowy RJ: Improvements in
methods for calculating virus titer estimates from
TCID50 and plaque assays. J Virol Methods. 96:107–126.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ning Z, Peng Y, Hao W, Duan C, Rock DL and
Luo S: Generation of recombinant Orf virus using an enhanced green
fluorescent protein reporter gene as a selectable marker. BMC Vet
Res. 7:802011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Akiba H, Nakano H, Nishinaka S, Shindo M,
Kobata T, Atsuta M, Morimoto C, Ware CF, Malinin NL, Wallach D, et
al: CD27, a member of the tumor necrosis factor receptor
superfamily, activates NF-kappaB and stress-activated protein
kinase/c-Jun N-terminal kinase via TRAF2, TRAF5, and
NF-kappaB-inducing kinase. J Biol Chem. 273:13353–13358. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Brocker C, Thompson D, Matsumoto A, Nebert
DW and Vasiliou V: Evolutionary divergence and functions of the
human interleukin (IL) gene family. Hum Genomics. 5:30–55. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gaffen SL and Liu KD: Overview of
interleukin-2 function, production and clinical applications.
Cytokine. 28:109–123. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kuchen S, Robbins R, Sims GP, Sheng C,
Phillips TM, Lipsky PE and Ettinger R: Essential role of IL-21 in B
cell activation, expansion, and plasma cell generation during
CD4+ T cell-B cell collaboration. J Immunol.
179:5886–5896. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Or R, Abdul-Hai A and Ben-Yehuda A:
Reviewing the potential utility of interleukin-7 as a promoter of
thymopoiesis and immune recovery. Cytokines Cell Mol Ther.
4:287–294. 1998.PubMed/NCBI
|
|
30
|
Prasad KV, Ao Z, Yoon Y, Wu MX, Rizk M,
Jacquot S and Schlossman SF: CD27, a member of the tumor necrosis
factor receptor family, induces apoptosis and binds to Siva, a
proapoptotic protein. Proc Natl Acad Sci USA. 94:6346–6351. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Steel JC, Waldmann TA and Morris JC:
Interleukin-15 biology and its therapeutic implications in cancer.
Trends Pharmacol Sci. 33:35–41. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang B, Tan Z and Guan F: Tumor-derived
exosomes mediate the instability of cadherins and promote tumor
progression. Int J Mol Sci. 20:36522019. View Article : Google Scholar
|
|
33
|
Petrova YI, Schecterson L and Gumbiner BM:
Roles for E-cadherin cell surface regulation in cancer. Mol Biol
Cell. 27:3233–3244. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mrozik KM, Blaschuk OW, Cheong CM,
Zannettino AC and Vandyke K: N-cadherin in cancer metastasis, its
emerging role in haematological malignancies and potential as a
therapeutic target in cancer. BMC Cancer. 18:9392018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Voigt H, Merant C, Wienhold D, Braun A,
Hutet E, Le Potier MF, Saalmüller A, Pfaff E and Büttner M:
Efficient priming against classical swine fever with a safe
glycoprotein E2 expressing Orf virus recombinant (ORFV VrV-E2).
Vaccine. 25:5915–5926. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Fischer T, Planz O, Stitz L and Rziha HJ:
Novel recombinant parapoxvirus vectors induce protective humoral
and cellular immunity against lethal herpesvirus challenge
infection in mice. J Virol. 77:9312–9323. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li W, Hao W, Peng Y, Duan C, Tong C, Song
D, Gao F, Li M, Rock DL and Luo S: Comparative genomic sequence
analysis of Chinese orf virus strain NA1/11 with other
parapoxviruses. Arch Virol. 160:253–266. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Aruga A: Current status and future
perspective of cancer immunotherapy - clinical application and
development. Nihon Geka Gakkai Zasshi. 114:327–331. 2013.(In
Japanese). PubMed/NCBI
|
|
39
|
Lapeyre-Prost A, Terme M, Pernot S,
Pointet AL, Voron T, Tartour E and Taieb J: Immunomodulatory
activity of VEGF in cancer. Int Rev Cell Mol Biol. 330:295–342.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Poesen K, Lambrechts D, Van Damme P,
Dhondt J, Bender F, Frank N, Bogaert E, Claes B, Heylen L, Verheyen
A, et al: Novel role for vascular endothelial growth factor (VEGF)
receptor-1 and its ligand VEGF-B in motor neuron degeneration. J
Neurosci. 28:10451–10459. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Shutter JR, Scully S, Fan W, Richards WG,
Kitajewski J, Deblandre GA, Kintner CR and Stark KL: Dll4, a novel
Notch ligand expressed in arterial endothelium. Genes Dev.
14:1313–1318. 2000.PubMed/NCBI
|
|
42
|
Fernandez EJ and Lolis E: Structure,
function, and inhibition of chemokines. Annu Rev Pharmacol Toxicol.
42:469–499. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Abel S, Hundhausen C, Mentlein R, Schulte
A, Berkhout TA, Broadway N, Hartmann D, Sedlacek R, Dietrich S,
Muetze B, et al: The transmembrane CXC-chemokine ligand 16 is
induced by IFN-gamma and TNF-alpha and shed by the activity of the
disintegrin-like metalloproteinase ADAM10. J Immunol.
172:6362–6372. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bouchon A, Dietrich J and Colonna M:
Cutting edge: Inflammatory responses can be triggered by TREM-1, a
novel receptor expressed on neutrophils and monocytes. J Immunol.
164:4991–4995. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ma B, Zhu Z, Homer RJ, Gerard C, Strieter
R and Elias JA: The C10/CCL6 chemokine and CCR1 play critical roles
in the pathogenesis of IL-13-induced inflammation and remodeling. J
Immunol. 172:1872–1881. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hermani A, De Servi B, Medunjanin S,
Tessier PA and Mayer D: S100A8 and S100A9 activate MAP kinase and
NF-kappaB signaling pathways and trigger translocation of RAGE in
human prostate cancer cells. Exp Cell Res. 312:184–197. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Dahlmann M, Okhrimenko A, Marcinkowski P,
Osterland M, Herrmann P, Smith J, Heizmann CW, Schlag PM and Stein
U: RAGE mediates S100A4-induced cell motility via MAPK/ERK and
hypoxia signaling and is a prognostic biomarker for human
colorectal cancer metastasis. Oncotarget. 5:3220–3233. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Upadhyay P, Gardi N, Desai S, Chandrani P,
Joshi A, Dharavath B, Arora P, Bal M, Nair S and Dutt A: Genomic
characterization of tobacco/nut chewing HPV-negative early stage
tongue tumors identify MMP10 asa candidate to predict metastases.
Oral Oncol. 73:56–64. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Croce CM and Reed JC: Finally, an
apoptosis-targeting therapeutic for cancer. Cancer Res.
76:5914–5920. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Loya SM and Zhang X: Enhancing the
bystander killing effect of an oncolytic HSV by arming it with a
secretable apoptosis activator. Gene Ther. 22:237–246. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wei X, Liu L, Wang G, Li W, Xu K, Qi H,
Liu H, Shen J, Li Z and Shao J: Potent antitumor activity of the
Ad5/11 chimeric oncolytic adenovirus combined with interleukin-24
for acute myeloid leukemia via induction of apoptosis. Oncol Rep.
33:111–118. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Raeber ME, Zurbuchen Y, Impellizzieri D
and Boyman O: The role of cytokines in T-cell memory in health and
disease. Immunol Rev. 283:176–193. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Vial J, Royet A, Cassier P, Tortereau A,
Dinvaut S, Maillet D, Gratadou-Hupon L, Creveaux M, Sadier A,
Tondeur G, et al: The ectodysplasin receptor EDAR acts as a tumor
suppressor in melanoma by conditionally inducing cell death. Cell
Death Differ. 26:443–454. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Huang ZM, Du SH, Huang LG, Li JH, Xiao L
and Tong P: Leptin promotes apoptosis and inhibits autophagy of
chondrocytes through upregulating lysyl oxidase-like 3 during
osteoarthritis pathogenesis. Osteoarthritis Cartilage.
24:1246–1253. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yuan X, Gajan A, Chu Q, Xiong H, Wu K and
Wu GS: Developing TRAIL/TRAIL death receptor-based cancer
therapies. Cancer Metastasis Rev. 37:733–748. 2018. View Article : Google Scholar : PubMed/NCBI
|