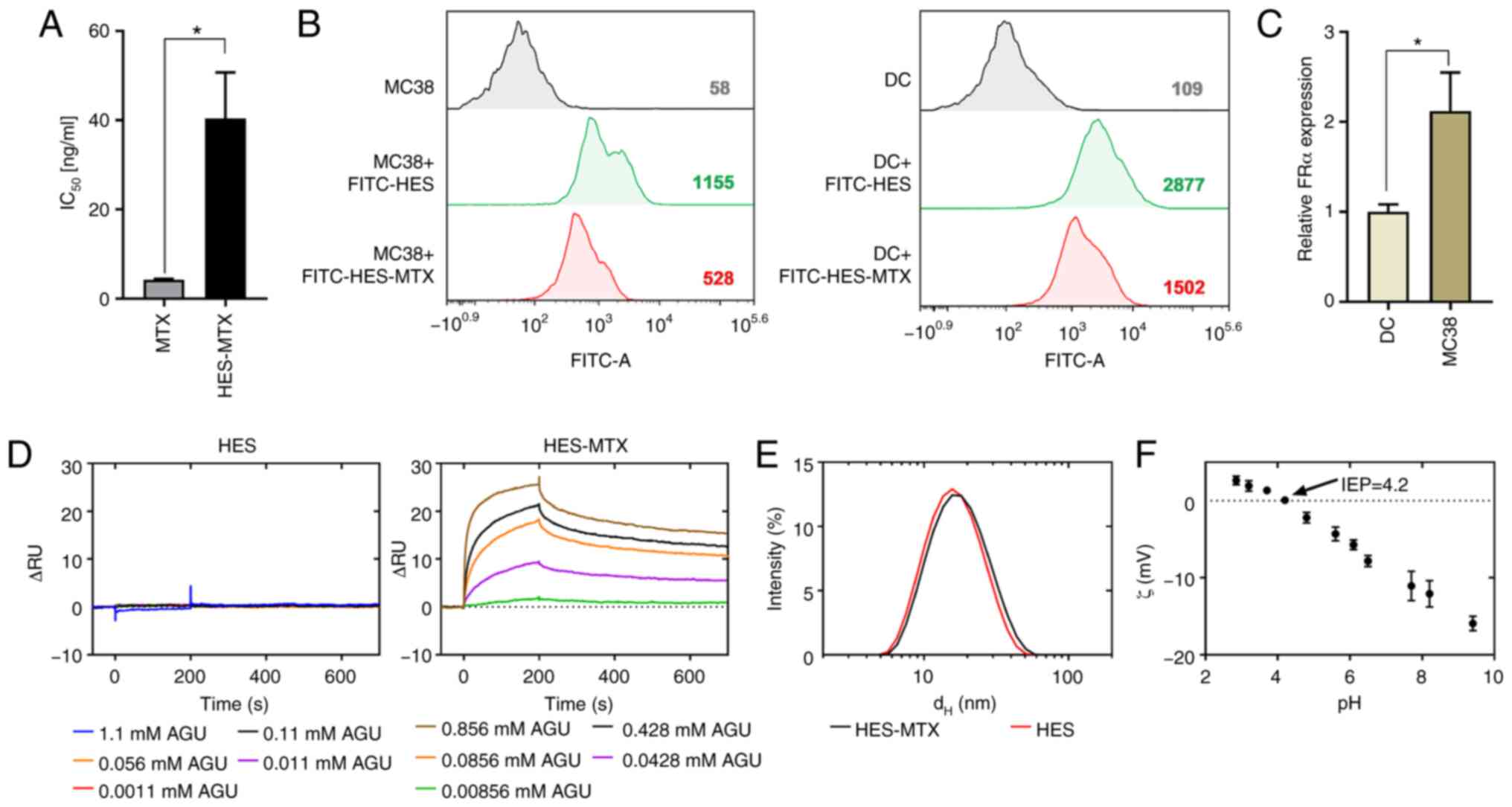
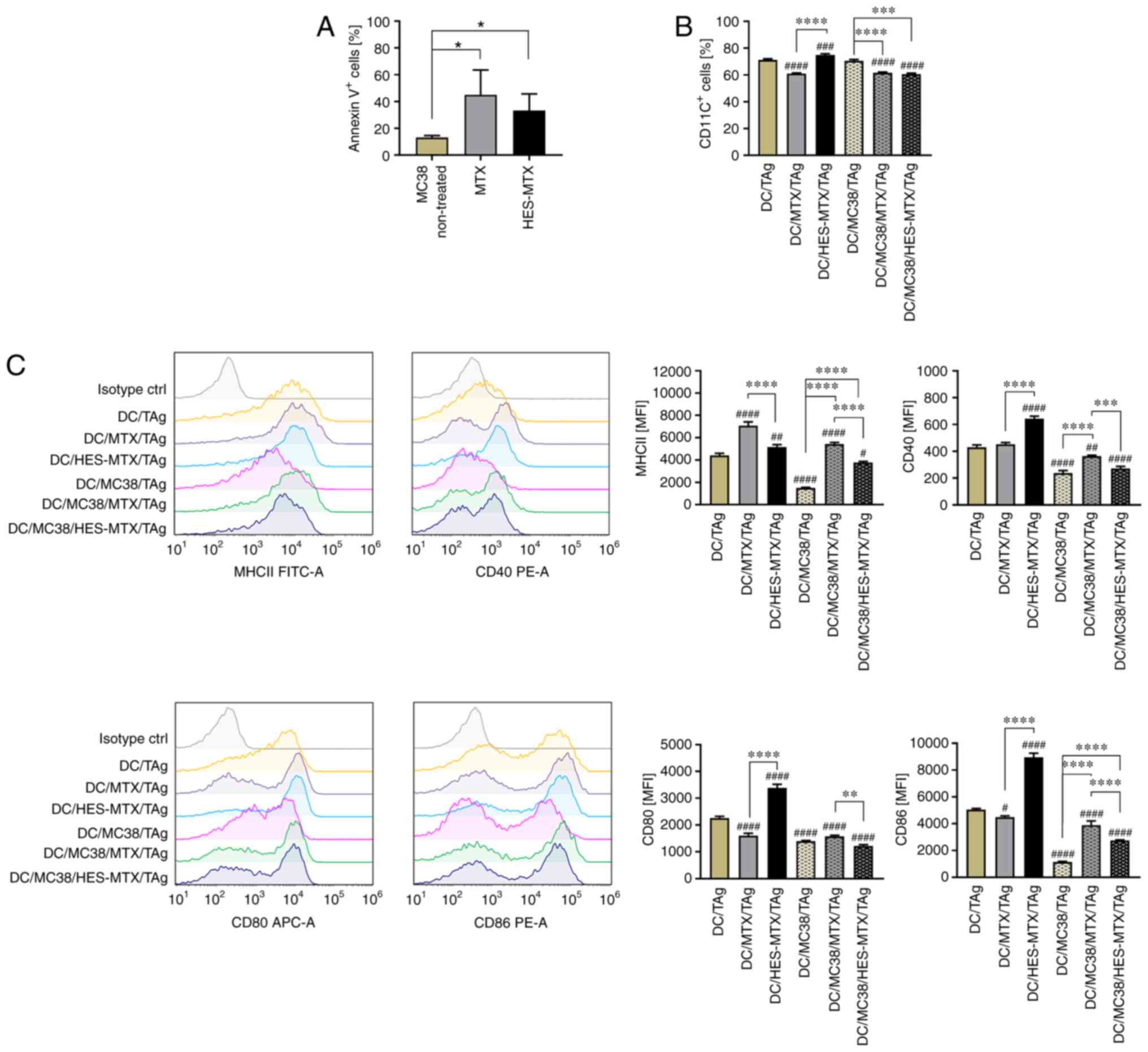
|
1
|
Nars MS and Kaneno R: Immunomodulatory
effects of low dose chemotherapy and perspectives of its
combination with immunotherapy. Int J Cancer. 132:2471–2478. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Golombek SK, May JN, Theek B, Appold L,
Drude N, Kiessling F and Lammers T: Tumor targeting via EPR:
Strategies to enhance patient responses. Adv Drug Deliv Rev.
130:17–38. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Roma-Rodrigues C, Mendes R, Baptista PV
and Fernandes AR: Targeting tumor microenvironment for cancer
therapy. Int J Mol Sci. 20:8402019. View Article : Google Scholar
|
|
4
|
Hanahan D and Coussens LM: Accessories to
the crime: Functions of cells recruited to the tumor
microenvironment. Cancer Cell. 21:309–322. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Whiteside TL: The tumor microenvironment
and its role in promoting tumor growth. Oncogene. 27:5904–5912.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jahchan NS, Mujal AM, Pollack JL,
Binnewies M, Sriram V, Reyno L and Krummel MF: Tuning the tumor
myeloid microenvironment to fight cancer. Front Immunol.
10:16112019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Netea-Maier RT, Smit JWA and Netea MG:
Metabolic changes in tumor cells and tumor-associated macrophages:
A mutual relationship. Cancer Lett. 413:102–109. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Veglia F and Gabrilovich DI: Dendritic
cells in cancer: The role revisited. Curr Opin Immunol. 45:43–51.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Szczygieł A and Pajtasz-Piasecka E:
Between biology and medicine: Perspectives on the use of dendritic
cells in anticancer therapy. Postepy Hig Med Dosw (Online).
71:921–941. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Constantino J, Gomes C, Falcão A, Cruz MT
and Neves BM: Antitumor dendritic cell-based vaccines: Lessons from
20 years of clinical trials and future perspectives. Transl Res.
168:74–95. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sabado RL, Balan S and Bhardwaj N:
Dendritic cell-based immunotherapy. Cell Res. 27:74–95. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Palucka K and Banchereau J:
Dendritic-cell-based therapeutic cancer vaccines. Immunity.
39:38–48. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bol KF, Schreibelt G, Gerritsen WR, de
Vries IJM and Figdor CG: Dendritic cell-based immunotherapy: State
of the art and beyond. Clin Cancer Res. 22:1897–1906. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Goszczyński T, Nevozhay D, Wietrzyk J,
Omar MS and Boratyński J: The antileukemic activity of modified
fibrinogen-methotrexate conjugate. Biochim Biophys Acta.
1830:2526–2530. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hao F, Lee RJ, Yang C, Zhong L, Sun Y,
Dong S, Cheng Z, Teng L, Meng Q, Lu J, et al: Targeted Co-delivery
of siRNA and methotrexate for tumor therapy via mixed micelles.
Pharmaceutics. 11:922019. View Article : Google Scholar
|
|
16
|
Chen Y, Zhang W, Huang Y, Gao F, Sha X,
Lou K and Fang X: The therapeutic effect of methotrexate-conjugated
Pluronic-based polymeric micelles on the folate receptor-rich
tumors treatment. Int J Nanomedicine. 10:4043–4057. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Thomas TP, Huang B, Choi SK, Silpe JE,
Kotlyar A, Desai AM, Zong H, Gam J, Joice M and Baker JR Jr:
Polyvalent dendrimer-methotrexate as a folate receptor-targeted
cancer therapeutic. Mol Pharm. 9:2669–2676. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li MH, Choi SK, Thomas TP, Desai A, Lee
KH, Kotlyar A, Banaszak Holl MM and Baker JR Jr: Dendrimer-based
multivalent methotrexates as dual acting nanoconjugates for cancer
cell targeting. Eur J Med Chem. 47:560–572. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Figueiró F, de Oliveira CP, Bergamin LS,
Rockenbach L, Mendes FB, Jandrey EH, Moritz CE, Pettenuzzo LF,
Sévigny J, Guterres SS, et al: Methotrexate up-regulates
ecto-5′-nucleotidase/CD73 and reduces the frequency of T
lymphocytes in the glioblastoma microenvironment. Purinergic
Signal. 12:303–312. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Goszczyński TM, Filip-Psurska B, Kempińska
K, Wietrzyk J and Boratyński J: Hydroxyethyl starch as an effective
methotrexate carrier in anticancer therapy. Pharmacol Res Perspect.
2:e000472014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Goszczyński T, Boratyński J, Wietrzyk J,
Filip-Psurska B and Kempińska K: Patent WO/2013/127885; A conjugate
of methotrexate and Hydroxyethyl starch for use in the treatment
cancer. 2013.
|
|
22
|
Koźmiński P, Halik PK, Chesori R and
Gniazdowska E: Overview of dual-acting drug methotrexate in
different neurological diseases, autoimmune pathologies and
cancers. Int J Mol Sci. 21:34832020. View Article : Google Scholar
|
|
23
|
Abolmaali SS, Tamaddon AM and Dinarvand R:
A review of therapeutic challenges and achievements of methotrexate
delivery systems for treatment of cancer and rheumatoid arthritis.
Cancer Chemother Pharmacol. 71:1115–1130. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Visentin M, Zhao R and Goldman ID: The
antifolates. Hematol Oncol Clin North Am. 26629–648. (ix)2012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Brecher ME, Owen HG and Bandarenko N:
Alternatives to albumin: Starch replacement for plasma exchange. J
Clin Apher. 12:146–153. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hoffmann S, Caysa H, Kuntsche J,
Kreideweiss P, Leimert A, Mueller T and Mäder K: Carbohydrate
plasma expanders for passive tumor targeting: In vitro and in vivo
studies. Carbohydr Polym. 95:404–413. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nogueira E, Sárria MP, Azoia NG, Antunes
E, Loureiro A, Guimarães D, Noro J, Rollett A, Guebitz G and
Cavaco-Paulo A: Internalization of methotrexate conjugates by
folate receptor-α. Biochemistry. 57:6780–6786. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Thomas TP, Goonewardena SN, Majoros IJ,
Kotlyar A, Cao Z, Leroueil PR and Baker JR: Folate-targeted
nanoparticles show efficacy in the treatment of inflammatory
arthritis. Arthritis Rheum. 63:2671–2680. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang S and Dormidontova EE: Nanoparticle
targeting using multivalent ligands: Computer modeling. Soft
Matter. 7:4435–4445. 2011. View Article : Google Scholar
|
|
30
|
Fang J, Nakamura H and Maeda H: The EPR
effect: Unique features of tumor blood vessels for drug delivery,
factors involved, and limitations and augmentation of the effect.
Adv Drug Deliv Rev. 63:136–151. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Maeda H: Tumor-selective delivery of
macromolecular drugs via the EPR effect: Background and future
prospects. Bioconjug Chem. 21:797–802. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Maeda H and Matsumura Y: EPR effect based
drug design and clinical outlook for enhanced cancer chemotherapy.
Adv Drug Deliv Rev. 63:129–130. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shurin GV, Tourkova IL, Kaneno R and
Shurin MR: Chemotherapeutic agents in noncytotoxic concentrations
increase antigen presentation by dendritic cells via an
IL-12-dependent mechanism. J Immunol. 183:137–144. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kaneno R, Shurin GV, Tourkova IL and
Shurin MR: Chemomodulation of human dendritic cell function by
antineoplastic agents in low noncytotoxic concentrations. J Transl
Med. 7:582009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kaneno R, Shurin GV, Kaneno FM, Naiditch
H, Luo J and Shurin MR: Chemotherapeutic agents in low noncytotoxic
concentrations increase immunogenicity of human colon cancer cells.
Cell Oncol (Dordr). 34:97–106. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang L, Dermawan K, Jin M, Liu R, Zheng
H, Xu L, Zhang Y, Cai Y, Chu Y and Xiong S: Differential impairment
of regulatory T cells rather than effector T cells by
paclitaxel-based chemotherapy. Clin Immunol. 129:219–229. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sistigu A, Viaud S, Chaput N, Bracci L,
Proietti E and Zitvogel L: Immunomodulatory effects of
cyclophosphamide and implementations for vaccine design. Semin
Immunopathol. 33:369–383. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu JY, Wu Y, Zhang XS, Yang JL, Li HL,
Mao YQ, Wang Y, Cheng X, Li YQ, Xia JC, et al: Single
administration of low dose cyclophosphamide augments the antitumor
effect of dendritic cell vaccine. Cancer Immunol Immunother.
56:1597–1604. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Rossowska J, Anger N, Kicielińska J,
Pajtasz-Piasecka E, Bielawska-Pohl A, Wojas-Turek J and Duś D:
Temporary elimination of IL-10 enhanced the effectiveness of
cyclophosphamide and BMDC-based therapy by decrease of the
suppressor activity of MDSCs and activation of antitumour immune
response. Immunobiology. 220:389–398. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Rossowska J, Pajtasz-Piasecka E, Anger N,
Wojas-Turek J, Kicielińska J, Piasecki E and Duś D:
Cyclophosphamide and IL-12-transduced DCs enhance the antitumor
activity of tumor antigen-stimulated DCs and reduce Tregs and MDSCs
number. J Immunother. 37:427–439. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Pajtasz-Piasecka E, Szyda A, Rossowska J,
Krawczenko A, Indrová M, Grabarczyk P, Wysocki P, Mackiewicz A and
Duś D: Loss of tumorigenicity of murine colon carcinoma MC38/0 cell
line after transduction with a retroviral vector carrying murine
IL-12 genes. Folia Biol (Praha). 50:7–14. 2004.PubMed/NCBI
|
|
42
|
Rossowska J, Pajtasz-Piasecka E, Szyda A,
Krawczenko A, Zietara N and Dus D: Tumour antigen-loaded mouse
dendritic cells maturing in the presence of inflammatory cytokines
are potent activators of immune response in vitro but not
in vivo. Oncol Rep. 21:1539–1549. 2009.PubMed/NCBI
|
|
43
|
Thomas E, Jones G, de Souza P, Wardrop C
and Wusteman F: Measuring blood volume with fluorescent-labeled
hydroxyethyl starch. Crit Care Med. 28:627–631. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Nevozhay D, Budzynska R, Kanska U,
Jagiello M, Omar MS, Boratynski J and Opolski A: Antitumor
properties and toxicity of dextran-methotrexate conjugates are
dependent on the molecular weight of the carrier. Anticancer Res.
26:1135–1143. 2006.PubMed/NCBI
|
|
45
|
Ciekot J, Goszczyński T and Boratyńskit J:
Methods for methotrexate determination in macromolecular conjugates
drug carrier. Acta Pol Pharm. 69:1342–1346. 2012.PubMed/NCBI
|
|
46
|
Rossowska J, Anger N, Szczygieł A,
Mierzejewska J and Pajtasz-Piasecka E: Reprogramming the murine
colon cancer microenvironment using lentivectors encoding shRNA
against IL-10 as a component of a potent DC-based
chemoimmunotherapy. J Exp Clin Cancer Res. 37:1262018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Pajtasz-Piasecka E, Rossowska J, Szyda A,
Krawczenko A and Dus D: Generation of anti-tumor response by JAWS
II mouse dendritic cells transduced with murine interleukin 12
genes. Oncol Rep. 17:1249–1257. 2007.PubMed/NCBI
|
|
48
|
Ji X, Guo H, Tang Q, Ma D and Xue W: A
targeted nanocarrier based on polyspermine for the effective
delivery of methotrexate in nasopharyngeal carcinoma. Mater Sci Eng
C Mater Biol Appl. 81:48–56. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Goos JACM, Cho A, Carter LM, Dilling TR,
Davydova M, Mandleywala K, Puttick S, Gupta A, Price WS, Quinn JF,
et al: Delivery of polymeric nanostars for molecular imaging and
endoradiotherapy through the enhanced permeability and retention
(EPR) effect. Theranostics. 10:567–584. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kobayashi H, Watanabe R and Choyke PL:
Improving conventional enhanced permeability and retention (EPR)
effects; what is the appropriate target? Theranostics. 4:81–89.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Pajtasz-Piasecka E and Indrová M:
Dendritic cell-based vaccines for the therapy of experimental
tumors. Immunotherapy. 2:257–268. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zong J, Keskinov AA, Shurin GV and Shurin
MR: Tumor-derived factors modulating dendritic cell function.
Cancer Immunol Immunother. 65:821–833. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Zhong H, Han B, Tourkova IL, Lokshin A,
Rosenbloom A, Shurin MR and Shurin GV: Low-dose paclitaxel prior to
intratumoral dendritic cell vaccine modulates intratumoral cytokine
network and lung cancer growth. Clin Cancer Res. 13:5455–5462.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Rossowska J, Anger N, Szczygieł A,
Mierzejewska J and Pajtasz-Piasecka E: Intratumoral
lentivector-mediated TGF-β1 gene downregulation as a potent
strategy for enhancing the antitumor effect of therapy composed of
cyclophosphamide and dendritic cells. Front Immunol. 8:7132017.
View Article : Google Scholar : PubMed/NCBI
|