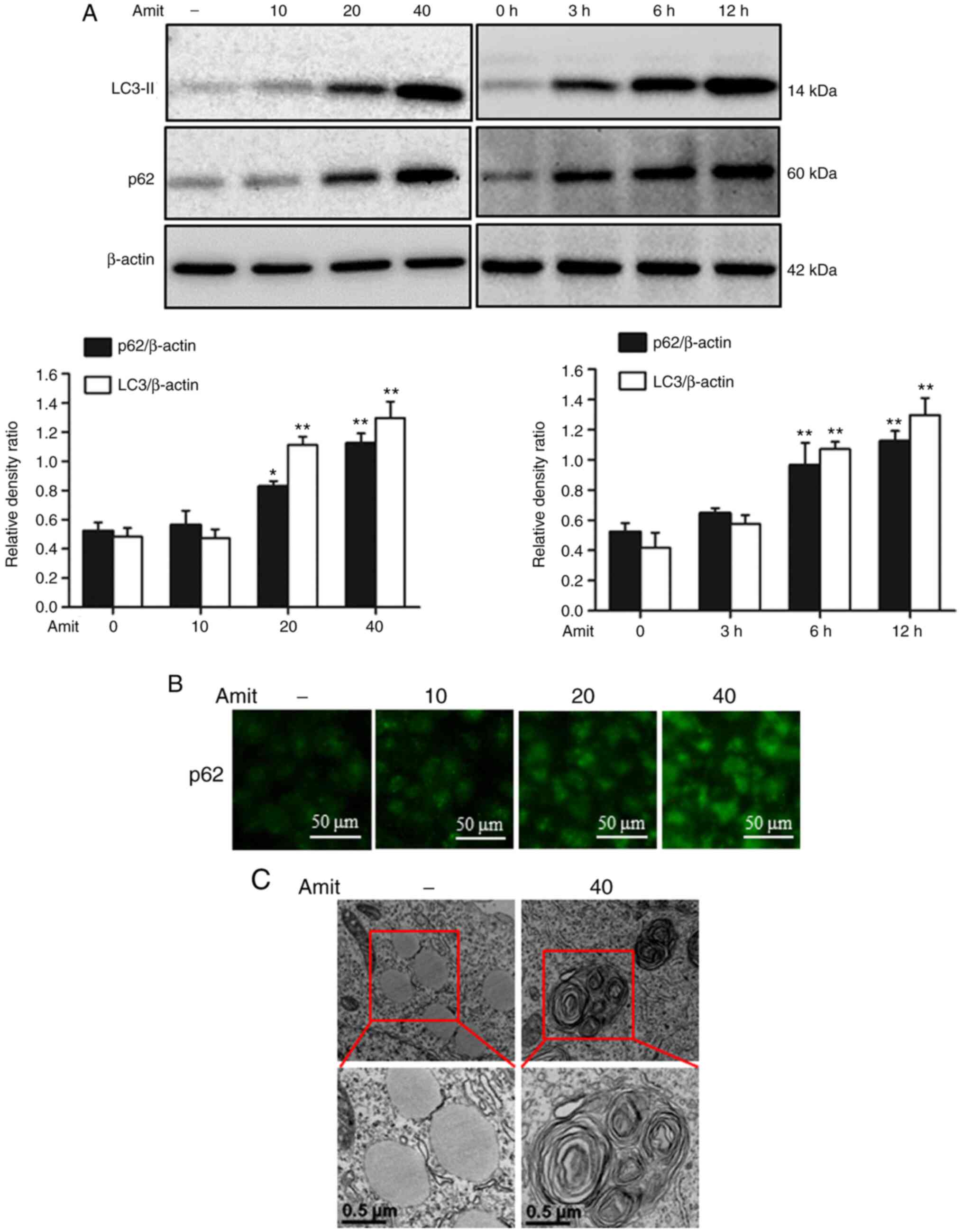
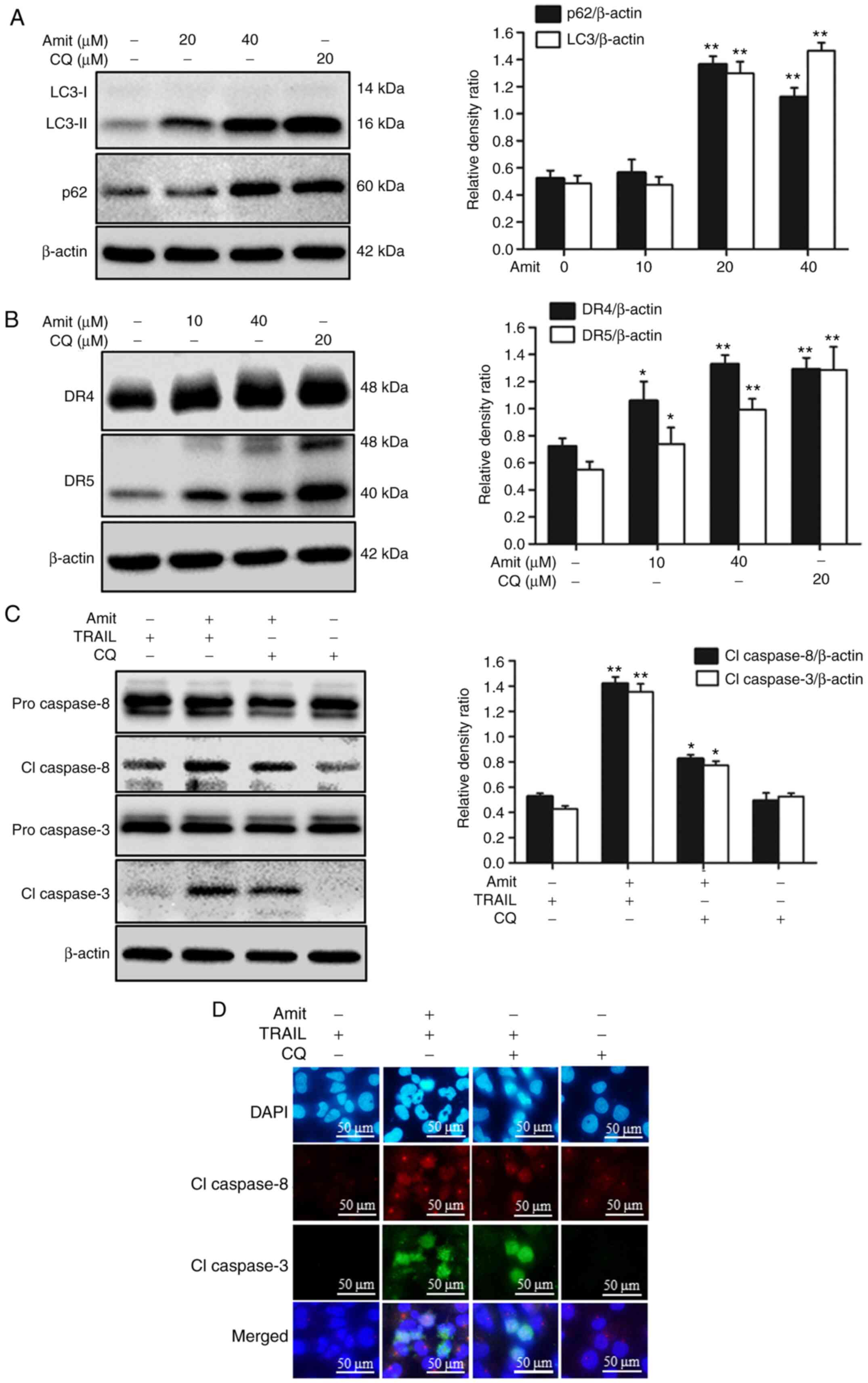
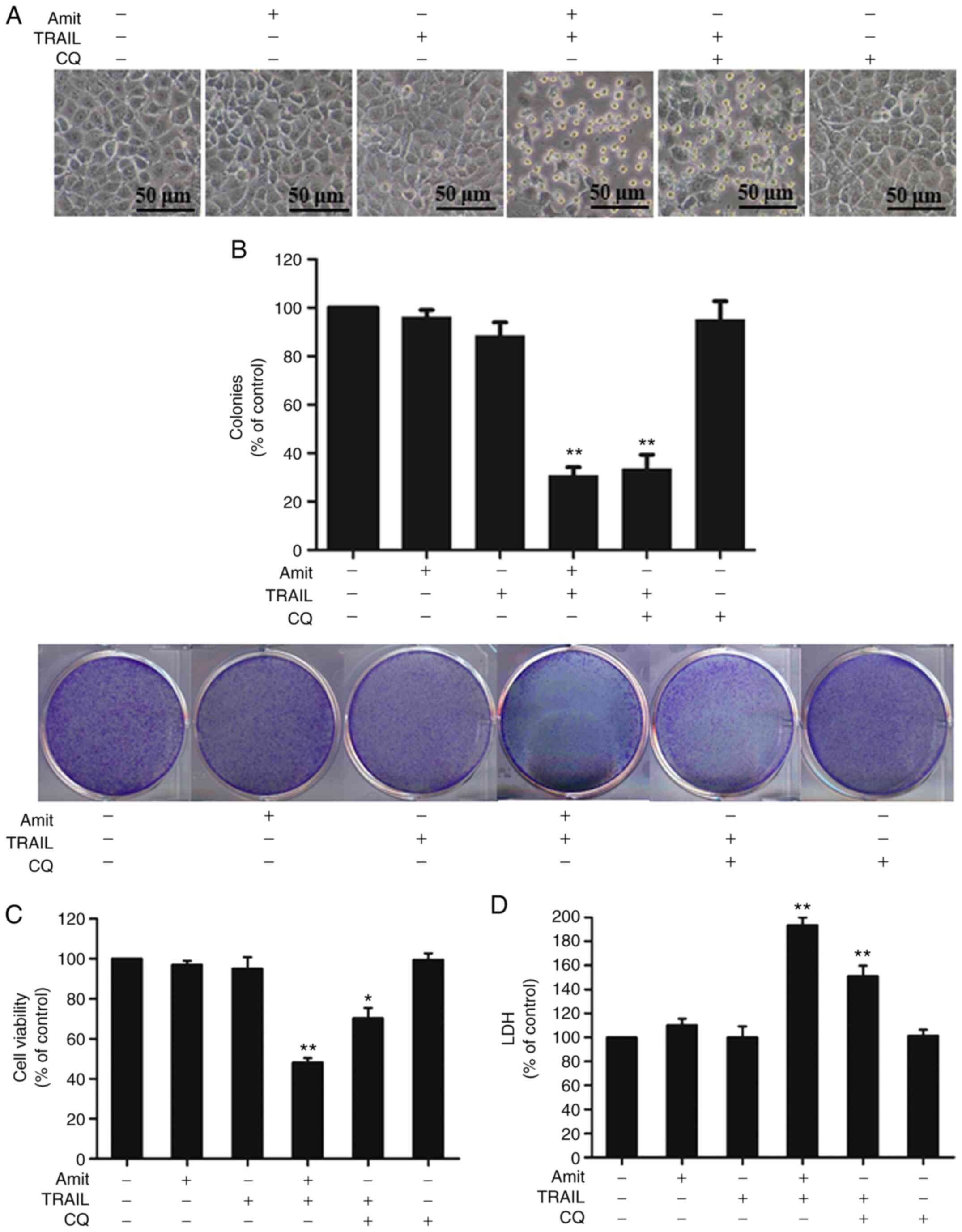
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2018. CA Cancer J Clin. 68:7–30. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ettinger DS, Akerley W, Borghaei H, Chang
AC, Cheney RT, Chirieac LR, D'Amico TA, Demmy TL, Ganti AK,
Govindan R, et al: Non-Small cell lung cancer. J Natl Compr Canc
Netw. 10:1236–1271. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kanitkar AA, Schwartz AG, George J and
Soubani AO: Causes of death in long-term survivors of non-small
cell lung cancer: A regional surveillance, epidemiology, and end
results study. Ann Thorac Med. 13:76–81. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Heinzmann K, Nguyen QD, Honess D, Smith
DM, Stribbling S, Brickute D, Barnes C, Griffiths J and Aboagye E:
Depicting changes in tumor biology in response to cetuximab
monotherapy or combination therapy by apoptosis and proliferation
imaging using 18 F-ICMT-11 and 18 F-FLT PET.
J Nucl Med. 59:1558–1565. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Thomas PA: Stage IIIA N2 non-small-cell
lung cancer: Current controversies in combined-modality therapy.
Eur J Cardiothorac Surg. 36:431–432. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nowak-Sliwinska P, Scapozza L and Altaba
AR: Drug repurposing in oncology: Compounds, pathways, phenotypes
and computational approaches for colorectal cancer. Biochim Biophys
Acta Rev Cancer. 1871:434–454. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jia Y, Yun CH, Park E, Ercan D, Manuia M,
Juarez J, Xu C, Rhee K, Chen T, Zhang H, et al: Overcoming
EGFR(T790M) and EGFR(C797S) resistance with mutant-selective
allosteric inhibitors. Nature. 534:129–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nesterov A, Ivashchenko Y and Kraft AS:
Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)
triggers apoptosis in normal prostate epithelial cells. Oncogene.
21:1135–1140. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Trivedi R and Mishra DP: Trailing TRAIL
resistance: Novel targets for TRAIL sensitization in cancer Cells.
Front Oncol. 5:692015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Marsters SA, Sheridan JP, Pitti RM, Huang
A, Skubatch M, Baldwin D, Yuan J, Gurney A, Goddard AD, Godowski P
and Ashkenazi A: A novel receptor for Apo2L/TRAIL contains a
truncated death domain. Curr Biol. 7:1003–1006. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jin CY, Moon DO, Lee JD, Heo MS, Choi YH,
Lee CM, Park YM and Kim GY: Sulforaphane sensitizes tumor necrosis
factor-related apoptosis-inducing ligand-mediated apoptosis through
downregulation of ERK and akt in lung adenocarcinoma A549 cells.
Carcinogenesis. 28:1058–1066. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Thorburn A, Behbakht K and Ford H: TRAIL
receptor-targeted therapeutics: Resistance mechanisms and
strategies to avoid them. Drug Resist Updat. 11:17–24. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mérino D, Lalaoui N, Morizot A, Solary E
and Micheau O: TRAIL in cancer therapy: Present and future
challenges. Expert Opin Ther Targets. 11:1299–1314. 2007.
View Article : Google Scholar
|
|
15
|
Hale AN, Ledbetter DJ, Gawriluk TR and
Rucker EB III: Autophagy: Regulation and role in development.
Autophagy. 9:951–972. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rouschop KM and Wouters BG: Regulation of
autophagy through multiple independent hypoxic signaling pathways.
Curr Mol Med. 9:417–424. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Thorburn A, Thamm DH and Gustafson DL:
Autophagy and cancer therapy. Mol Pharmacol. 85:830–838. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sui X, Chen R, Wang Z, Huang Z, Kong N,
Zhang M, Han W, Lou F, Yang J, Zhang Q, et al: Autophagy and
chemotherapy resistance: A promising therapeutic target for cancer
treatment. Cell Death Dis. 4:e838. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zinnah KMA and Park SY: Duloxetine
enhances TRAIL-mediated apoptosis via AMPK-mediated inhibition of
autophagy flux in lung cancer cells. Anticancer Res. 39:6621–6633.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Di Rosso ME, Sterle HA, Cremaschi GA and
Genaro AM: Beneficial effect of fluoxetine and sertraline on
chronic stress-induced tumor growth and cell dissemination in a
mouse model of lymphoma: Crucial role of antitumor immunity. Front
Immunol. 9:13412018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Thaker PH, Han LY, Kamat AA, Arevalo JM,
Takahashi R, Lu C, Jennings NB, Armaiz-Pena G, Bankson JA, Ravoori
M, et al: Chronic stress promotes tumor growth and angiogenesis in
a mouse model of ovarian carcinoma. Nat Med. 12:939–944. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim-Fuchs C, Le CP, Pimentel MA,
Shackleford D, Ferrari D, Angst E, Hollande F and Sloan EK: Chronic
stress accelerates pancreatic cancer growth and invasion: A
critical role for beta-adrenergic signaling in the pancreatic
microenvironment. Brain Behav Immun. 40:40–47. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hasegawa H and Saiki I: Psychosocial
stress augments tumor development through beta-adrenergic
activation in mice. Jpn J Cancer Res. 93:729–735. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fann JR, Fan MY and Unützer J: Improving
primary care for older adults with cancer and depression. J Gen
Intern Med. 24 (Suppl 2):S417–S424. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Laird B, Colvin L and Fallon M: Management
of cancer pain: Basic principles and neuropathic cancer pain. Eur J
Cancer. 44:1078–1082. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Frick LR and Rapanelli M: Antidepressants:
Influence on cancer and immunity? Life Sci. 92:525–532. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang Z, Du X, Zhao C, Cao B, Zhao Y and
Mao X: The antidepressant amitriptyline shows potent therapeutic
activity against multiple myeloma. Anticancer Drugs. 24:792–798.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cordero MD, Sánchez-Alcázar JA,
Bautista-Ferrufino MR, Carmona-López MI, Illanes M, Ríos MJ,
Garrido-Maraver J, Alcudia A, Navas P and de Miguel M: Acute
oxidant damage promoted on cancer cells by amitriptyline in
comparison with some common chemotherapeutic drugs. Anticancer
Drugs. 21:932–944. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yuan X, Gajan A, Chu Q, Xiong H, Wu K and
Wu GS: Developing TRAIL/TRAIL death receptor-based cancer
therapies. Cancer Metastasis Rev. 37:733–748. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Islam MA, Sooro MA and Zhang P: Autophagic
regulation of p62 is critical for cancer therapy. Int J Mol Sci.
19:14052018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wiley SR, Schooley K, Smolak PJ, Din WS,
Huang CP, Nicholl JK, Sutherland GR, Smith TD, Rauch C, Smith CA,
et al: Identification and characterization of a new member of the
TNF family that induces apoptosis. Immunity. 3:673–682. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Walczak H, Miller RE, Ariail K, Gliniak B,
Griffith TS, Kubin M, Chin W, Jones J, Woodward A, Le T, et al:
Tumoricidal activity of tumor necrosis factor-related
apoptosis-inducing ligand in vivo. Nat Med. 5:157–163. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Aggarwal BB, Bhardwaj U and Takada Y:
Regulation of TRAIL-induced apoptosis by ectopic expression of
antiapoptotic factors. Vitam Horm. 67:453–483. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang S: TRAIL: A sword for killing tumors.
Curr Med Chem. 17:3309–3317. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chaudhary PM, Eby M, Jasmin A, Bookwalter
A, Murray J and Hood L: Death receptor 5, a new member of the TNFR
family, and DR4 induce FADD-dependent apoptosis and activate the
NF-kappaB pathway. Immunity. 7:821–830. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pan G, O'Rourke K, Chinnaiyan AM, Gentz R,
Ebner R, Ni J and Dixit VM: The receptor for the cytotoxic ligand
TRAIL. Science. 276:111–113. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cretney E, Takeda K and Smyth MJ: Cancer:
Novel therapeutic strategies that exploit the TNF-related
apoptosis-inducing ligand (TRAIL)/TRAIL receptor pathway. Int J
Biochem Cell Biol. 39:280–286. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Danial NN and Korsmeyer SJ: Cell death:
Critical control points. Cell. 116:205–219. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Plummer R, Attard G, Pacey S, Li L, Razak
A, Perrett R, Barrett M, Judson I, Kaye S, Fox NL, et al: Phase 1
and pharmacokinetic study of lexatumumab in patients with advanced
cancers. Clin Cancer Res. 13:6187–6194. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hotte SJ, Hirte HW, Chen EX, Siu LL, Le
LH, Corey A, Iacobucci A, MacLean M, Lo L, Fox NL and Oza AM: A
phase 1 study of mapatumumab (fully human monoclonal antibody to
TRAIL-R1) in patients with advanced solid malignancies. Clin Cancer
Res. 14:3450–3455. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Cheng H, Hong B, Zhou L, Allen JE, Tai G,
Humphreys R, Dicker DT, Liu YY and El-Deiry WS: Mitomycin C
potentiates TRAIL-induced apoptosis through p53-independent
upregulation of death receptors: Evidence for the role of c-Jun
N-terminal kinase activation. Cell Cycle. 11:3312–3323. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Dolloff NG, Mayes PA, Hart LS, Dicker DT,
Humphreys R and El-Deiry WS: Off-target lapatinib activity
sensitizes colon cancer cells through TRAIL death receptor
up-regulation. Sci Transl Med. 3:86ra502011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Maiuri MC, Zalckvar E, Kimchi A and
Kroemer G: Self-Eating and self-killing: Crosstalk between
autophagy and apoptosis. Nat Rev Mol Cell Biol. 8:741–752. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Mizushima N, Levine B, Cuervo AM and
Klionsky DJ: Autophagy fights disease through cellular
self-digestion. Nature. 451:1069–1075. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Amin A, Bajbouj K, Koch A, Gandesiri M and
Schneider-Stock R: Defective autophagosome formation in p53-null
colorectal cancer reinforces crocin-induced apoptosis. Int J Mol
Sci. 16:1544–1561. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wu YT, Tan HL, Huang Q, Kim YS, Pan N, Ong
WY, Liu ZG, Ong CN and Shen HM: Autophagy plays a protective role
during zVAD-induced necrotic cell death. Autophagy. 4:457–466.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
White E: Autophagic cell death unraveled:
Pharmacological inhibition of apoptosis and autophagy enables
necrosis. Autophagy. 4:399–401. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Vucicevic L, Misirkic M, Janjetovic K,
Vilimanovich U, Sudar E, Isenovic E, Prica M, Harhaji-Trajkovic L,
Kravic-Stevovic T, Bumbasirevic V and Trajkovic V: Compound C
induces protective autophagy in cancer cells through AMPK
inhibition-independent blockade of Akt/mTOR pathway. Autophagy.
7:40–50. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Shen S, Zhang Y, Wang Z, Zhang R and Gong
X: Bufalin induces the interplay between apoptosis and autophagy in
glioma cells through endoplasmic reticulum stress. Int J Biol Sci.
10:212–224. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Klionsky DJ, Abdelmohsen K, Abe A, Abedin
MJ, Abeliovich H, Arozena AA, Adachi H, Adams CM, Adams PD and
Adeli K: Guidelines for the use and interpretation of assays for
monitoring autophagy (3rd edition). Autophagy. 12:1–222. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Gómez-Sánchez R, Yakhine-Diop SMS,
Rodríguez-Arribas M, Bravo-San Pedro JM, Martínez-Chacón G,
Uribe-Carretero E, de Castro DC, Pizarro-Estrella E, Fuentes JM and
González-Polo RA: mRNA and protein dataset of autophagy markers
(LC3 and p62) in several cell lines. Data Brief. 7:641–647. 2016.
View Article : Google Scholar
|
|
53
|
Mauthe M, Orhon I, Rocchi C, Zhou X, Luhr
M, Hijlkema KJ, Coppes RP, Engedal N, Mari M and Reggiori F:
Chloroquine inhibits autophagic flux by decreasing
autophagosome-lysosome fusion. Autophagy. 14:1435–1455. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Nordstrøm LU, Sironi J, Aranda E, Maisonet
J, Perez-Soler R, Wu P and Schwartz EL: Discovery of autophagy
inhibitors with antiproliferative activity in lung and pancreatic
cancer cells. ACS Med Chem Lett. 6:134–139. 2015. View Article : Google Scholar
|
|
55
|
Pan H, Wang Y, Na K, Wang Y, Wang L, Li Z,
Guo C, Guo D and Wang X: Autophagic flux disruption contributes to
Ganoderma lucidum polysaccharide-induced apoptosis in human
colorectal cancer cells via MAPK/ERK activation. Cell Death Dis.
10:4562019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Gąsiorkiewicz BM, Koczurkiewicz-Adamczyk
P, Piska K and Pękala E: Autophagy modulating agents as
chemosensitizers for cisplatin therapy in cancer. Invest New Drugs.
39:538–563. 2020. View Article : Google Scholar
|
|
57
|
Nazim UM, Yin H and Park SY:
Downregulation of c-FLIP and upregulation of DR-5 by cantharidin
sensitizes TRAIL-mediated apoptosis in prostate cancer cells via
autophagy flux. Int J Mol Med. 46:280–288. 2020.PubMed/NCBI
|
|
58
|
Park EJ, Min Kj, Choi KS, Kubatka P,
Kruzliak P, Kim DE and Kwon TK: Chloroquine enhances TRAIL-mediated
apoptosis through up-regulation of DR5 by stabilization of mRNA and
protein in cancer cells. Sci Rep. 6:229212016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Shin GC, Kang HS, Lee AR and Kim KH:
Hepatitis B virus-triggered autophagy targets TNFRSF10B/death
receptor 5 for degradation to limit TNFSF10/TRAIL response.
Autophagy. 12:2451–2466. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Twomey JD and Zhang B: Circulating tumor
cells develop resistance to TRAIL-induced apoptosis through
autophagic removal of death receptor 5: Evidence from an in vitro
model. Cancers (Basel). 11:942019. View Article : Google Scholar : PubMed/NCBI
|