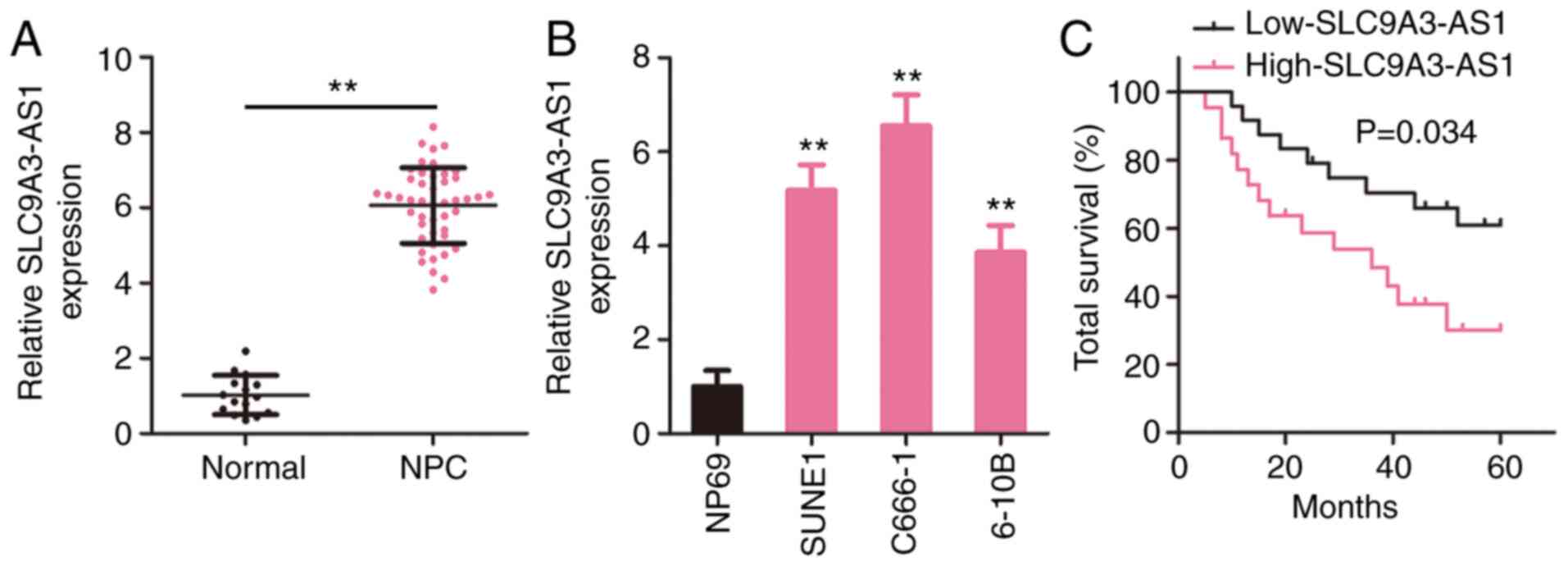
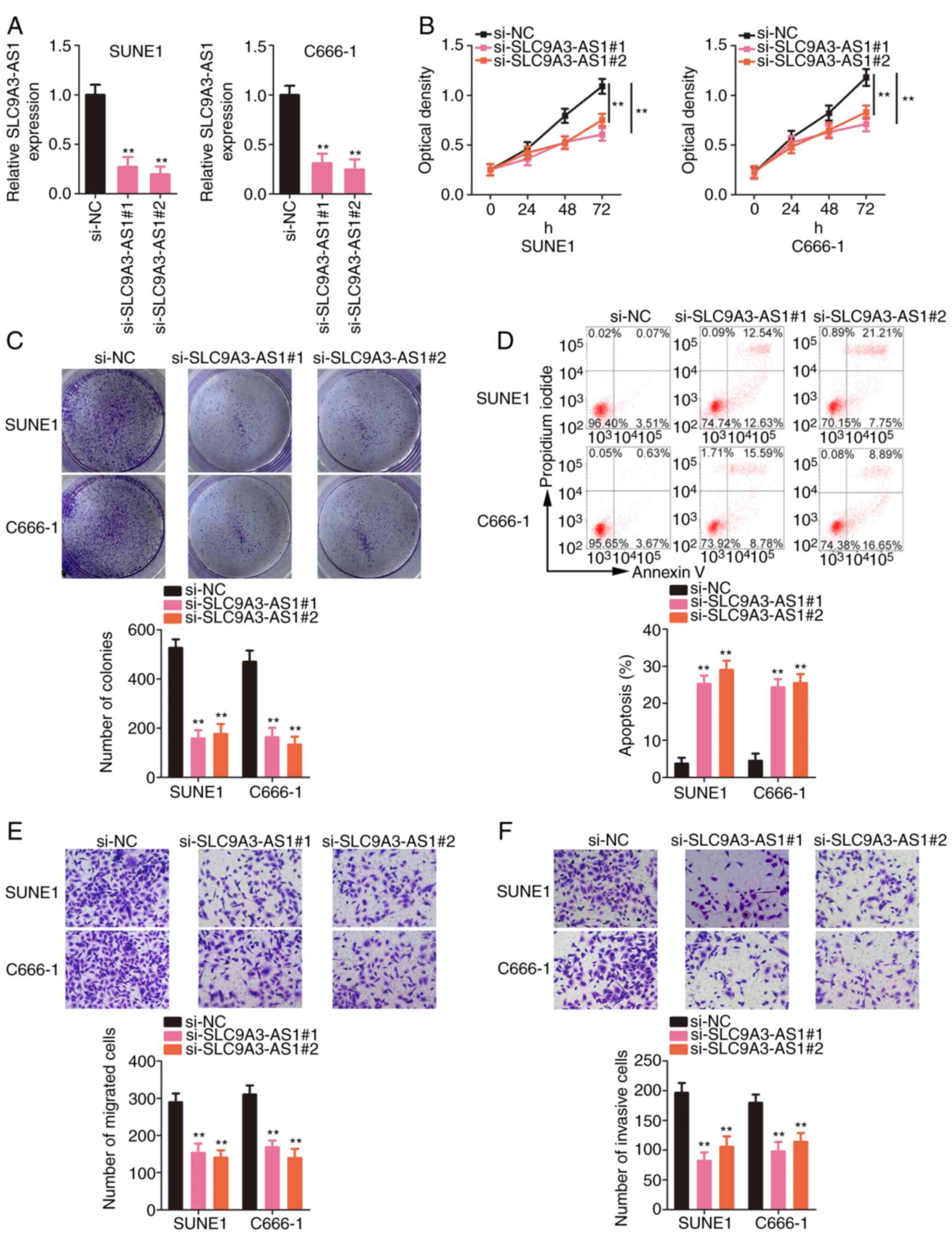
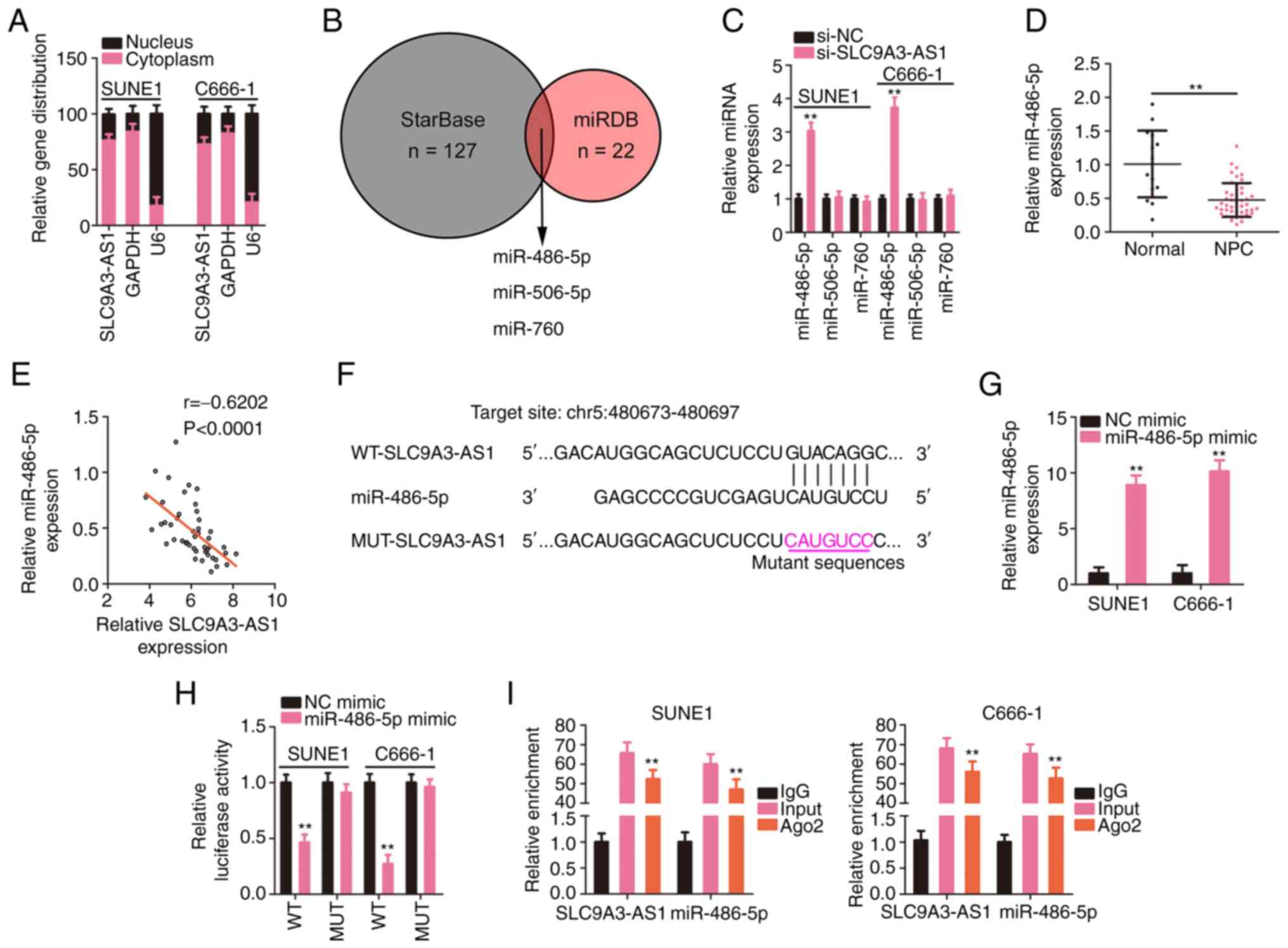
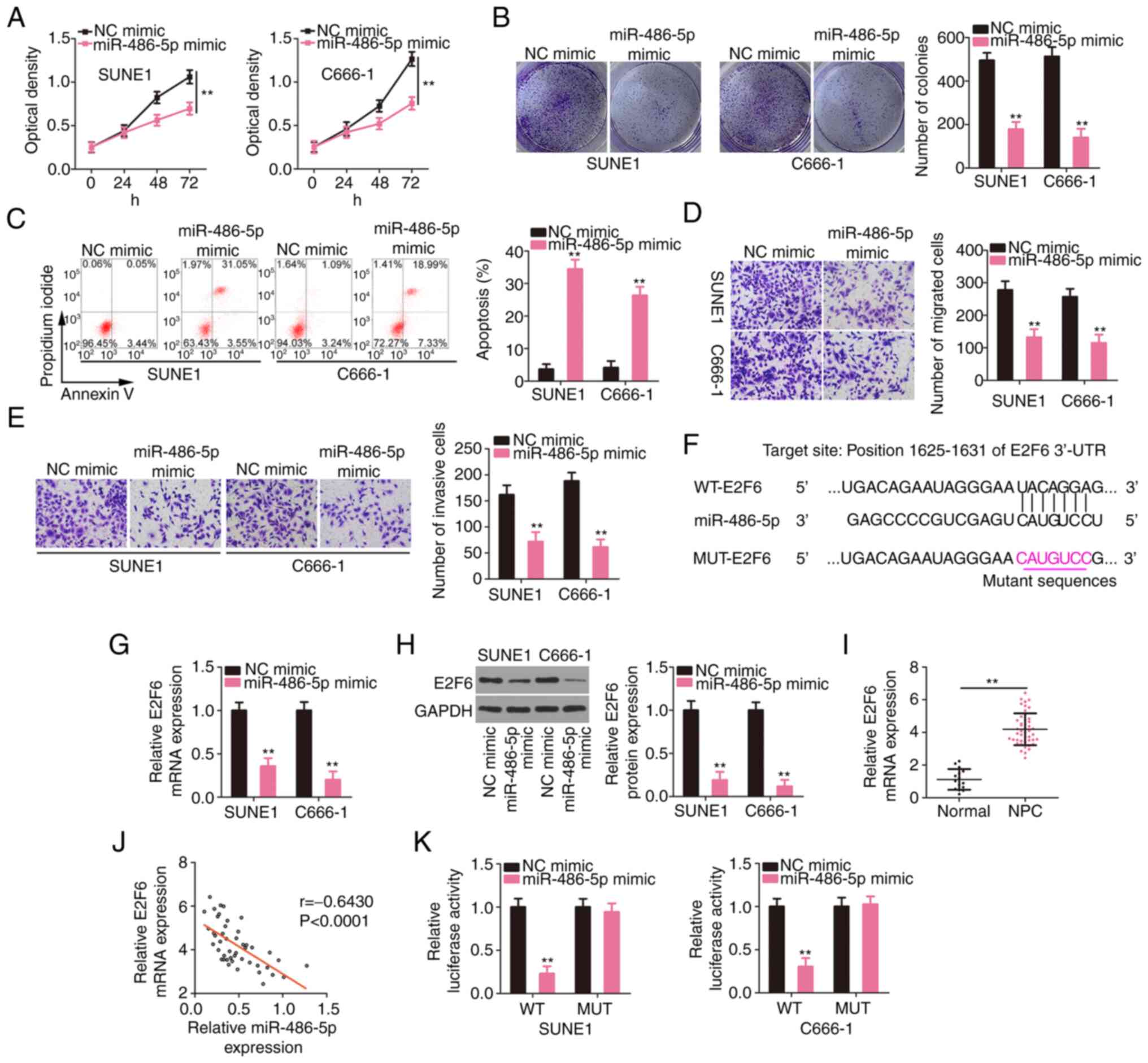
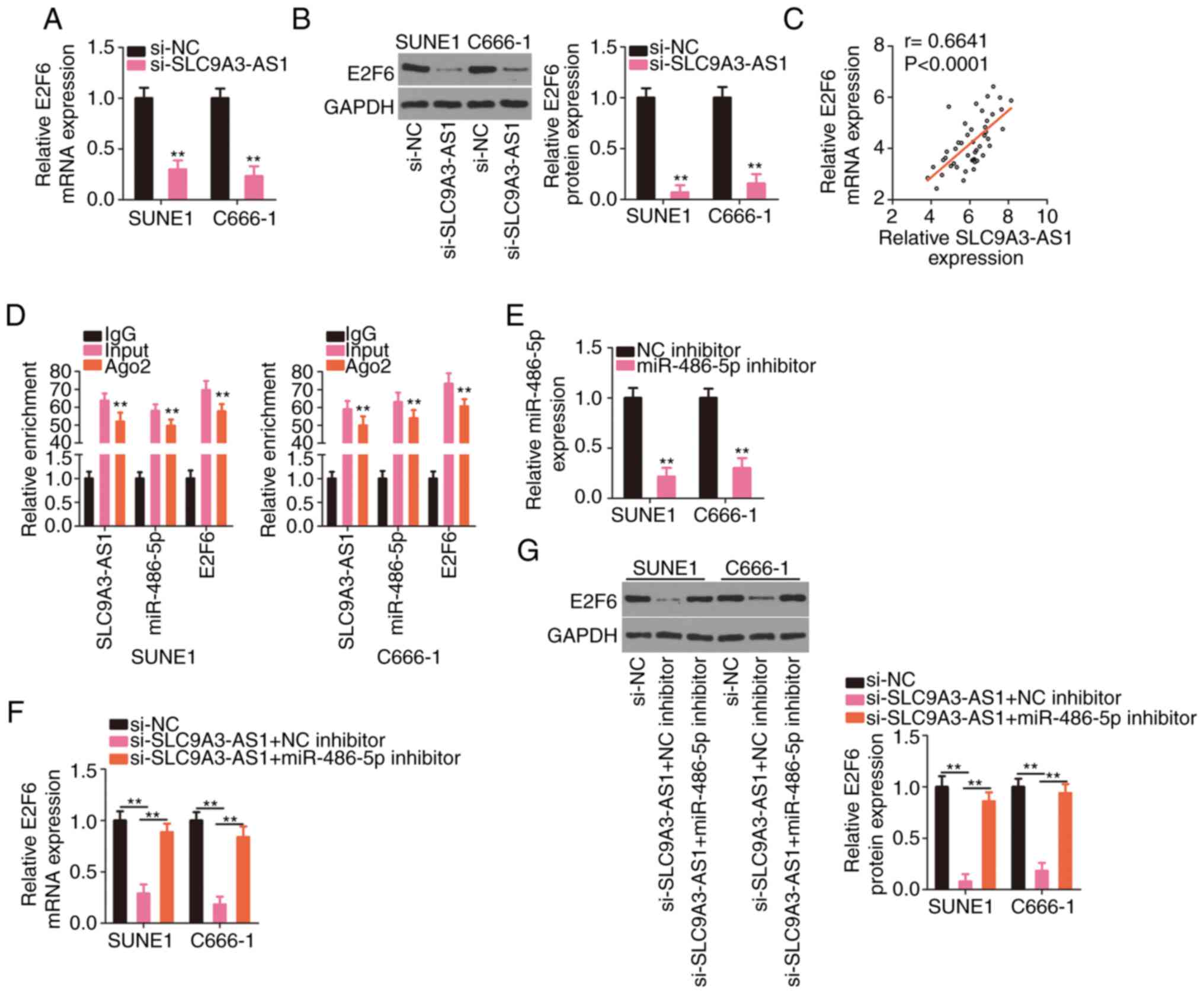
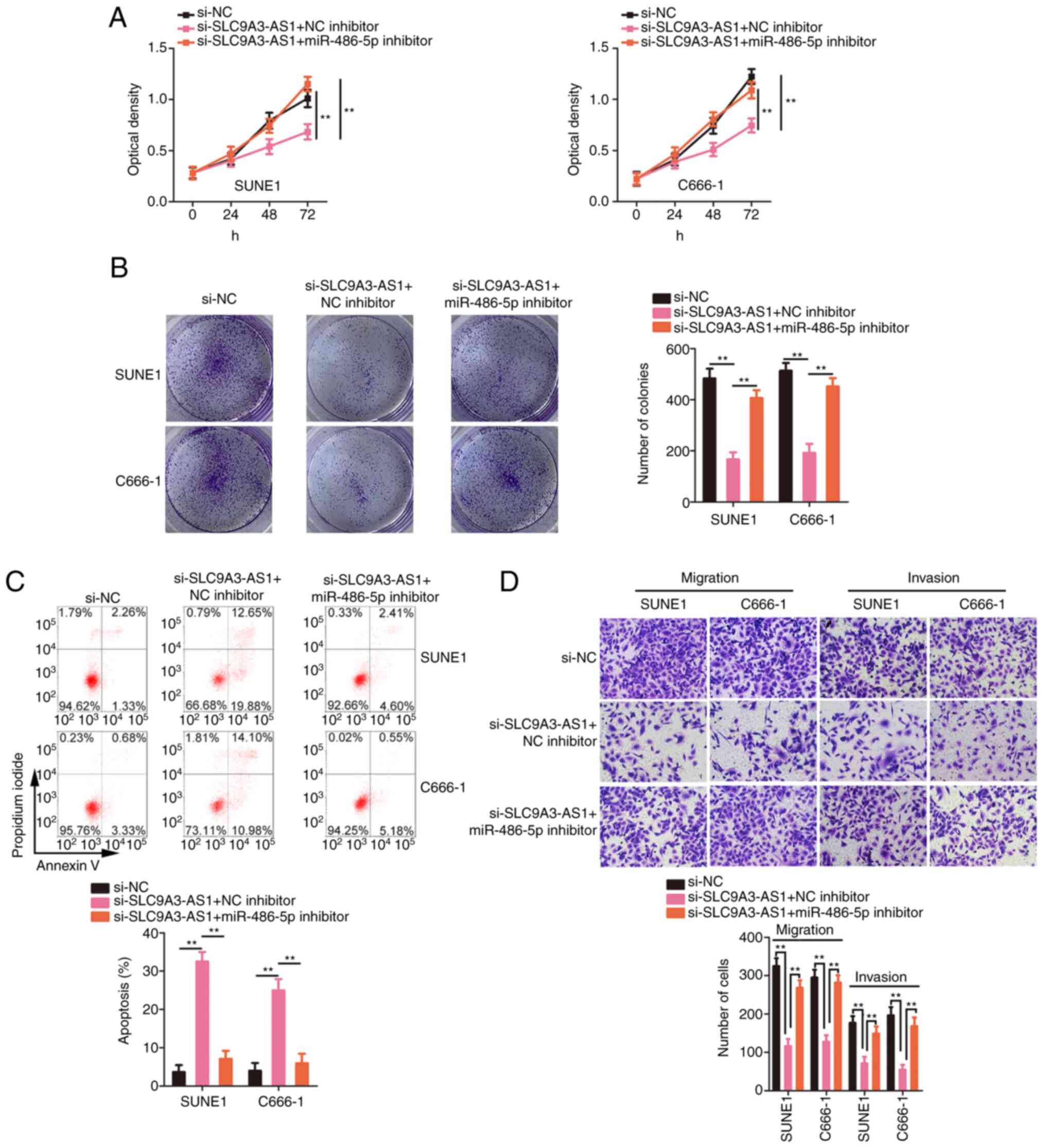
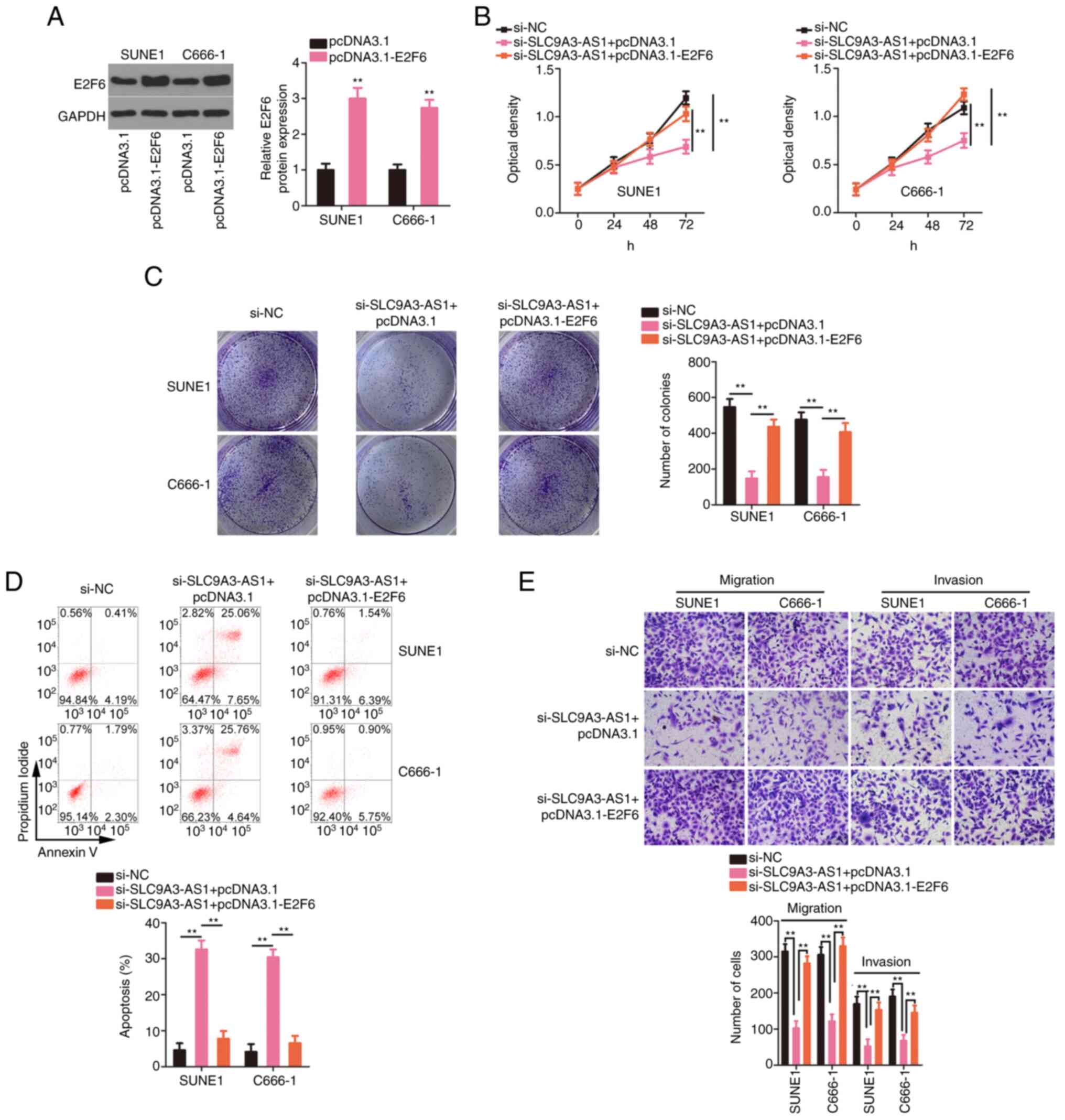
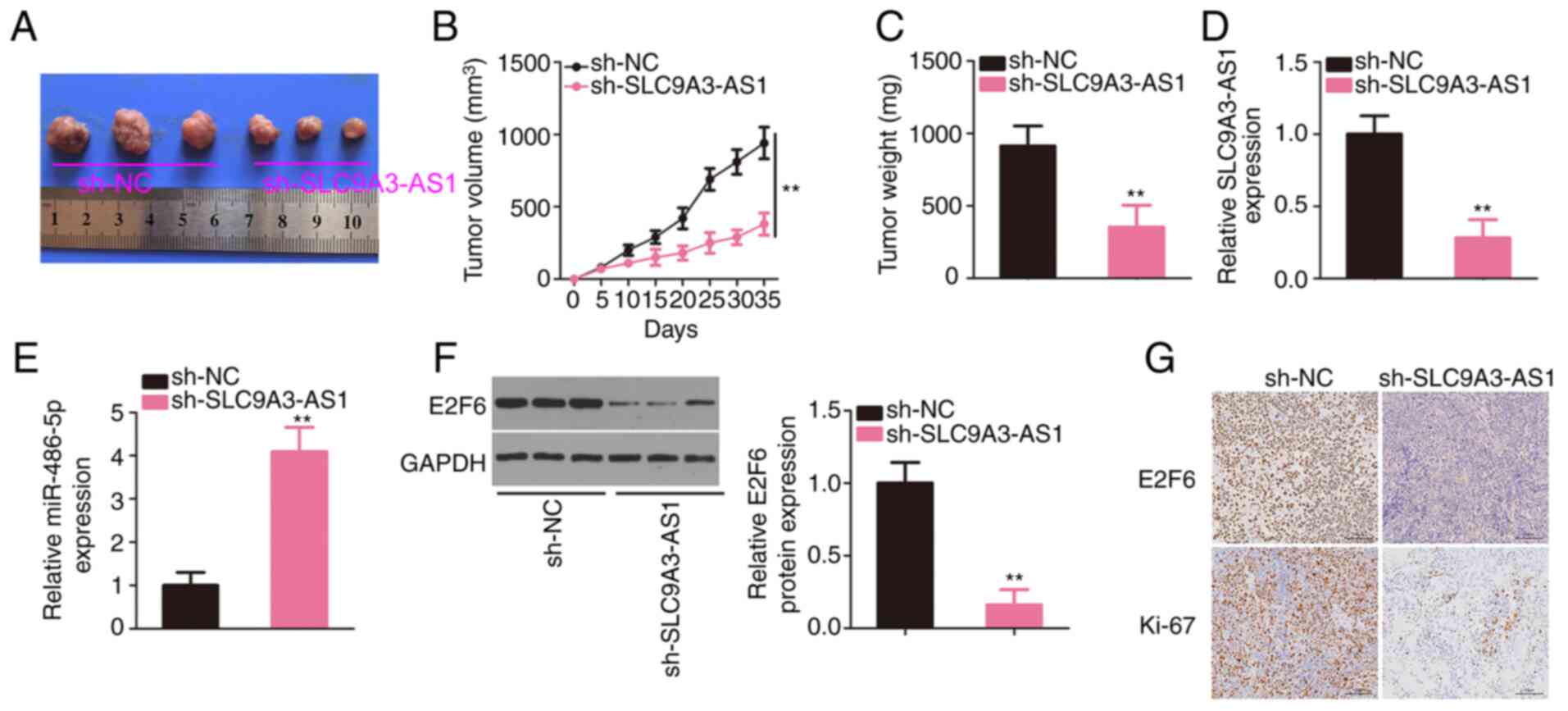
|
1
|
Chua ML, Wee JT, Hui EP and Chan AT:
Nasopharyngeal carcinoma. Lancet. 387:1012–1024. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lee AW, Ng WT, Chan YH, Sze H, Chan C and
Lam TH: The battle against nasopharyngeal cancer. Radiother Oncol.
104:272–278. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wei KR, Zheng RS, Zhang SW, Liang ZH, Ou
ZX and Chen WQ: Nasopharyngeal carcinoma incidence and mortality in
China in 2010. Chin J Cancer. 33:381–387. 2014.PubMed/NCBI
|
|
4
|
Lee AW, Tung SY, Ngan RK, Chappell R, Chua
DT, Lu TX, Siu L, Tan T, Chan LK, Ng WT, et al: Factors
contributing to the efficacy of concurrent-adjuvant chemotherapy
for locoregionally advanced nasopharyngeal carcinoma: Combined
analyses of NPC-9901 and NPC-9902 Trials. Eur J Cancer. 47:656–666.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tan WL, Tan EH, Lim DW, Ng QS, Tan DS,
Jain A and Ang MK: Advances in systemic treatment for
nasopharyngeal carcinoma. Chin Clin Oncol. 5:212016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tao Q and Chan AT: Nasopharyngeal
carcinoma: Molecular pathogenesis and therapeutic developments.
Expert Rev Mol Med. 9:1–24. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lam WK and Chan JY: Recent advances in the
management of nasopharyngeal carcinoma. F1000Res. 7:F1000 Faculty
Rev-1829. 2018. View Article : Google Scholar
|
|
8
|
Quinn JJ and Chang HY: Unique features of
long non-coding RNA biogenesis and function. Nat Rev Genet.
17:47–62. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Peng WX, Koirala P and Mo YY:
LncRNA-mediated regulation of cell signaling in cancer. Oncogene.
36:5661–5667. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Spizzo R, Almeida MI, Colombatti A and
Calin GA: Long non-coding RNAs and cancer: A new frontier of
translational research? Oncogene. 31:4577–4587. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sanchez Calle A, Kawamura Y, Yamamoto Y,
Takeshita F and Ochiya T: Emerging roles of long non-coding RNA in
cancer. Cancer Sci. 109:2093–2100. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu C, Zhang H and Liu H: Long noncoding
RNA UCA1 accelerates nasopharyngeal carcinoma cell progression by
modulating miR-124-3p/ITGB1 axis. Onco Targets Ther. 12:8455–8466.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhong Q, Chen Y and Chen Z: LncRNA MINCR
regulates irradiation resistance in nasopharyngeal carcinoma cells
via the microRNA-223/ZEB1 axis. Cell Cycle. 19:53–66. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhao CX, Zhu W, Ba ZQ, Xu HJ, Liu WD, Zhu
B, Wang L, Song YJ, Yuan S and Ren CP: The regulatory network of
nasopharyngeal carcinoma metastasis with a focus on EBV, lncRNAs
and miRNAs. Am J Cancer Res. 8:2185–2209. 2018.PubMed/NCBI
|
|
15
|
Guo H, Huang S, Li S, Yu H, Wu S and Zhou
X: Prognostic significance of the long noncoding RNAs in
nasopharyngeal carcinoma: A systematic review and meta-analysis.
Cancer Manag Res. 10:1763–1779. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu H, Lei C, He Q, Pan Z, Xiao D and Tao
Y: Nuclear functions of mammalian MicroRNAs in gene regulation,
immunity and cancer. Mol Cancer. 17:642018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ji W, Sun B and Su C: Targeting microRNAs
in cancer gene therapy. Genes (Basel). 8:212017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tian Y, Tang L, Yi P, Pan Q, Han Y, Shi Y,
Rao S, Tan S, Xia L, Lin J, et al: MiRNAs in radiotherapy
resistance of nasopharyngeal carcinoma. J Cancer. 11:3976–3985.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang S, Claret FX and Wu W: MicroRNAs as
therapeutic targets in nasopharyngeal carcinoma. Front Oncol.
9:7562019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lee KT, Tan JK, Lam AK and Gan SY:
MicroRNAs serving as potential biomarkers and therapeutic targets
in nasopharyngeal carcinoma: A critical review. Crit Rev Oncol
Hematol. 103:1–9. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Salmena L, Poliseno L, Tay Y, Kats L and
Pandolfi PP: A ceRNA hypothesis: The Rosetta Stone of a hidden RNA
language? Cell. 146:353–358. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bai Y, Qu Y, Wu Z, Ren Y, Cheng Z, Lu Y,
Hu J, Lou J, Zhao J, Chen C and Mao H: Absolute quantification and
analysis of extracellular vesicle lncRNAs from the peripheral blood
of patients with lung cancer based on multi-colour fluorescence
chip-based digital PCR. Biosens Bioelectron. 142:1115232019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ghafouri-Fard S, Shoorei H, Anamag FT and
Taheri M: The role of non-coding RNAs in controlling cell cycle
related proteins in cancer cells. Front Oncol. 10:6089752020.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Qian Y, Shi L and Luo Z: Long Non-coding
RNAs in cancer: Implications for diagnosis, prognosis, and therapy.
Front Med. 7:6123932020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
McCabe EM and Rasmussen TP: LncRNA
involvement in cancer stem cell function and epithelial-mesenchymal
transitions. Semin Cancer Biol. Dec 17–2020.(Epub ahead of print).
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao J, Liu D, Yang H, Yu S and He H: Long
noncoding RNAs in head and neck squamous cell carcinoma: Biological
functions and mechanisms. Mol Biol Rep. 47:8075–8090. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wu J and Hann SS: Functions and roles of
long-non-coding RNAs in human nasopharyngeal carcinoma. Cell
Physiol Biochem. 45:1191–1204. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Dong Q, Zhou L, Liu F, Ao F, Gong X, Jiang
C, Yuan Z and Li J: Long non-coding RNAs in the development,
diagnosis and prognosis of nasopharyngeal carcinoma. Int J Clin Exp
Pathol. 10:8098–8105. 2017.PubMed/NCBI
|
|
30
|
Xu J, Bai J, Zhang X, Lv Y, Gong Y, Liu L,
Zhao H, Yu F, Ping Y, Zhang G, et al: A comprehensive overview of
lncRNA annotation resources. Brief Bioinform. 18:236–249.
2017.PubMed/NCBI
|
|
31
|
Zhang K, Shi ZM, Chang YN, Hu ZM, Qi HX
and Hong W: The ways of action of long non-coding RNAs in cytoplasm
and nucleus. Gene. 547:1–9. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Rashid F, Shah A and Shan G: Long
non-coding RNAs in the cytoplasm. Genomics Proteomics
Bioinformatics. 14:73–80. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lin L, Liu X and Lv B: Long non-coding RNA
MEG3 promotes autophagy and apoptosis of nasopharyngeal carcinoma
cells via PTEN up-regulation by binding to microRNA-21. J Cell Mol
Med. 25:61–72. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sen R, Ghosal S, Das S, Balti S and
Chakrabarti J: Competing endogenous RNA: The key to
posttranscriptional regulation. ScientificWorldJournal.
2014:8962062014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gao ZJ, Yuan WD, Yuan JQ, Yuan K and Wang
Y: miR-486-5p functions as an oncogene by targeting PTEN in
non-small cell lung cancer. Pathol Res Pract. 214:700–705. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yang Y, Ji C, Guo S, Su X, Zhao X, Zhang
S, Liu G, Qiu X, Zhang Q, Guo H and Chen H: The miR-486-5p plays a
causative role in prostate cancer through negative regulation of
multiple tumor suppressor pathways. Oncotarget. 8:72835–72846.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xia L, Song M, Sun M, Chen W and Yang C:
miR-486 promotes capan-2 pancreatic cancer cell proliferation by
targeting phosphatase and tensin homolog deleted on chromosome 10
(PTEN). Front Genet. 10:5412019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
He M, Wang G, Jiang L, Qiu C, Li B, Wang J
and Fu Y: miR-486 suppresses the development of osteosarcoma by
regulating PKC-δ pathway. Int J Oncol. 50:1590–1600. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liu C, Li M, Hu Y, Shi N, Yu H, Liu H and
Lian H: miR-486-5p attenuates tumor growth and lymphangiogenesis by
targeting neuropilin-2 in colorectal carcinoma. Onco Targets Ther.
9:2865–2871. 2016.PubMed/NCBI
|
|
40
|
Ma X, Wei J, Zhang L, Deng D, Liu L, Mei
X, He X and Tian J: miR-486-5p inhibits cell growth of papillary
thyroid carcinoma by targeting fibrillin-1. Biomed Pharmacother.
80:220–226. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
An Y, Zhang J, Cheng X, Li B, Tian Y,
Zhang X and Zhao F: miR-454 suppresses the proliferation and
invasion of ovarian cancer by targeting E2F6. Cancer Cell Int.
20:2372020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Cai Q, Zhao A, Ren LG, Chen J, Liao KS,
Wang ZS and Zhang W: MiR-425 involves in the development and
progression of renal cell carcinoma by inhibiting E2F6. Eur Rev Med
Pharmacol Sci. 22:6300–6307. 2018.PubMed/NCBI
|
|
43
|
Lu Z, Nian Z, Jingjing Z, Tao L and Quan
L: MicroRNA-424/E2F6 feedback loop modulates cell invasion,
migration and EMT in endometrial carcinoma. Oncotarget.
8:114281–114291. 2017. View Article : Google Scholar : PubMed/NCBI
|