Introduction
Prostate cancer (PCa) is the fifth most common
cancer globally and the leading cause of cancer in men (1). More than 80% of patients with PCa die
due to bone metastases (2).
Androgen deprivation therapy (ADT) is the primary treatment for
advanced PCa. However, most patients with PCa with metastasis
receiving ADT develop castration-resistant prostate cancer (CRPC)
within 1–3 years (3,4).
Despite the lack of circulating androgens in
patients with CRPC, the androgen receptor (AR) signal is considered
the driving force of cancer growth and invasion (3). Thus, drugs reducing the level of
androgen (abiraterone) and inhibiting AR (enzalutamide) activity
have been recommended for CRPC treatment. Unfortunately, acquired
drug resistance targeting the AR ligand-binding domain is a common
clinical problem (5,6). Recently, several studies have
revealed a fundamental relationship between the AR and PI3K/Akt
signaling pathway in CRPC development and progression (6,7). It
has been revealed that loss of PTEN, the key regulator of the
PI3K/Akt signaling pathway, may decrease transcription of AR target
genes by inhibiting the negative regulators of AR activity in PCa
(8). Treatment with the AR
inhibitor enzalutamide in a PTEN loss mouse model enhanced PI3K/Akt
signaling activity by reducing the expression of peptidyl-prolyl
cis-trans isomerase FKBP5 (FKBP5) (9).
Several clinical reports have also revealed that the
AR and PI3K/Akt1 signaling pathways could compensate for each other
in inhibition of either pathway alone in patients with CRPC
(6,10). It is speculated that the PI3K/Akt
signaling pathway that participates in the mechanism of resistance
to ADT is also involved in the AR signaling pathway in CRPC
(10). MicroRNAs (miRNAs/miRs) are
small non-coding RNA molecules that regulate the expression of
target mRNA at a post-transcriptional level by binding to the
complementary sites within the 3′ untranslated region. miR-149 has
been found to serve as a tumor suppressor in various cancers, such
as gastric, lung, breast and renal cell carcinoma (11,12).
Loss of miR-149 has been associated with the development of renal
cell carcinoma, while elevated expression of miR-149 has been
reported to be important in the development of nasopharyngeal
carcinoma (13). Recently, several
studies have also revealed that miR-149 is involved in the
development and progression of CRPC (14,15).
Fujii et al (16) reported
that miR-149 targets syndecan-1 to control cell proliferation and
invasion by regulating the expression of Oct4 and SOX2 in PCa.
In our previous study, we revealed that PCa cells
contain a small subset of cells, called side population (SP). These
cells possess a much greater capacity of colony formation and
tumorigenic potential than non-SP (17). Furthermore, we also revealed that
the expression of miR-149 was significantly different in SP cells
and non-SP cells of the TSU CRPC cell line (17). Yet, the precise mechanism through
which miR-149 regulates PCa progression still remains unclear.
The androgen-independent Lymph Node Carcinoma of the
Prostate (LNCaP) subline, C4-2 cells, were obtained from LNCaP
tumors maintained in castrated hosts. This tumor subline has been
found to acquire metastatic potential targeting the bone when
orthotopically inoculated into both castrated and intact athymic
male nude mice (18). In the
current study, the role of miR-149 in the malignant phenotype and
its molecular mechanism were further investigated in CRPC cell line
C4-2.
Materials and methods
Cell culture, transfection and
reagent
The human prostate cells C4-2 were purchased from
the Tongpai (Shanghai) Biotechnology Co., Ltd., and cultured in
RPMI-1640 (Cytiva) medium supplemented with 10% FBS (Thermo Fisher
Scientific, Inc.), 100 µg/ml streptomycin (Sigma-Aldrich; Merck
KGaA) and 100 U/ml penicillin (Sigma-Aldrich; Merck KGaA) in a
humidified atmosphere containing 5%CO2/95% air at 37°C.
All the miRNAs, including mimics control, inhibitor control,
miR-149-5p mimic and miR-149-5p inhibitor, were acquired from
Tongpai Biotechnology Co., Ltd. (Shanghai, China). The 50 nM mimic
(miR-149-5p mimic and mimics control) and 100 nM inhibitor
(miR-149-5p inhibitor and inhibitor control) were transfected into
C4-2 cells using Lipofectamine® 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.) according to the manufacturer's protocol.
The sequences were as follows: miR-149-5p mimic,
5′-UCUGGCUCCGUGUCUUCACUCCC-3′; mimics control,
5′-UUCUCCGAACGUGUCACGUTT−3′; miR-149-5p inhibitor,
5′-GGGAGUGAAGACACGGAGCCAGA-3′; and inhibitor control,
5′-CAGUACUUUUGUGUAGUACAA−3′. Akt1 specific short hairpin (sh)RNA
against Akt1 and the corresponding negative control were
constructed in the pSUPER vector (provided by Professor He Dalin,
Xi'an Jiaotong University, Xi'an, China), and the sequences for
Akt1 shRNA were as follows:
5′-TCGCGTGACCATGAACGAGTTTTTCAAGAGAAAACTCGTTCATGGTCACGCTTTTTTCTCGAG-3′.
Akt1 shRNA (4 µg) was transfected into C4-2 cells using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol. After
72 h of transfection, cells were subjected to reverse
transcription-quantitative polymerase chain reaction PCR (RT-qPCR),
western blotting and Cell Counting Kit-8 (CCK-8) assay, as
described below.
RT-qPCR
Total RNA of PCa cells was extracted by TRIzol
(Invitrogen; Thermo Fisher Scientific, Inc.) according to the
manufacturer's instructions. To measure the Akt1 and AR mRNA
expression, cDNA synthesis was conducted with the PrimeScript RT
Master Mix (Takara Biotechnology Co., Ltd.), according to the
manufacturer's protocol. The cDNA product of PCa cells was then
subjected to qPCR with SYBR Premix Ex Taq (Takara Biotechnology
Co., Ltd.) and GAPDH served as the control. The qPCR conditions
were as follows: 35–45 cycles of 94°C for 30 sec, followed by
55–63°C for 30 sec. Primer sequences used for qPCR are shown in
Table I. The data were analyzed
using the 2−ΔΔCq method (19).
 | Table I.Primer sequences used for reverse
transcription-quantitative PCR. |
Table I.
Primer sequences used for reverse
transcription-quantitative PCR.
| Gene | Forward
(5′→3′) | Reverse
(5′→3′) |
|---|
| miR-149 |
GCGCTCTGGCTCCGTGTCTTC |
GTGCAGGGTCCGAGGT |
| U6 |
CTCGCTTCGGCAGCACATATACT |
ACGCTTCACGAATTTGCGTGTC |
| AR |
CTTCCCTCCCTATCTAACCCTC |
TCTAAACTTCCCGTGGCATAA |
| Akt1 |
TCCTCCTCAAGAATGATGGCA |
GTGCGTTCGATGACAGTGGT |
| mTOR |
GCGAACCTCAGGGCAAGAT |
TGACTCATCTCTCGGAGTTCCA |
| GAPDH |
AAGGTGAAGGTCGGAGTCAAC |
GGGGTCATTGATGGCAACAATA |
Western blotting
Total protein was obtained from cultured cells using
ice-cold lysis buffer containing 0.1% sodium dodecyl sulfate, 1%
NP-40, 0.1% sodium dodecyl sulfate and 200 µg/ml
phenylmethanesulfonyl fluoride. Insoluble materials were removed by
ultracentrifugation at 15,000 × g for 30 min at 4°C. The
concentration of the extracted protein was measured
spectrophotometrically with Coomassie G-250. Total protein (50 µg)
was loaded in each lane and resolved on a 12% SDS-PAGE gel. The
proteins were transferred onto PVDF membranes using a wet transfer
method following polyacrylamide gel electrophoresis, and then
blocked with 3% bovine serum albumin (Santa Cruz Biotechnology,
Inc.) for 1 h at room temperature and washed with Tris-buffered
saline and 0.1% Tween-20 three times. Samples were incubated at 4°C
overnight with the following primary antibodies: Akt1 (1:1,000;
cat. no. 2938S; Cell Signaling Technology, Inc.), p-Akt1 S473
(1:1,000; cat. no. 9018S; Cell Signaling Technology, Inc.), AR
(1:1,000; cat. no. 5153S; Cell Signaling Technology, Inc.), PTEN
(1:1,000; cat. no. 9559S; Cell Signaling Technology, Inc.), p-PI3K
(1:1,000; cat. no. 3821S; Cell Signaling Technology, Inc.), total
PI3K (1:1,000; cat. no. 4249S; Cell Signaling Technology, Inc.),
mTOR (1:1,000; cat. no. 2983S; Cell Signaling Technology, Inc.),
p-mTOR (1:1,000; cat. no. 2974S; Cell Signaling Technology, Inc.),
PSA (1:1,000; cat. no. 5365; Cell Signaling Technology, Inc.),
FKBP5 (1:1,000; cat. no. 12210; Cell Signaling Technology, Inc.),
E-cadherin (1:1,000; cat. no. sc-8426; Santa Cruz Biotechnology,
Inc.), Snail (1:1,000; cat. no. sc-271977; Santa Cruz
Biotechnology, Inc.), Vimentin (1:4,000; cat. no. sc-6260; Santa
Cruz Biotechnology, Inc.) and β-actin (1:4,000; cat. no. sc-47778;
Santa Cruz Biotechnology, Inc.). Then, membranes were incubated
with horseradish peroxidase-conjugated secondary antibodies
(1:4,000; cat. nos. A0208 and A0216; Beyotime Institute of
Biotechnology) at room temperature for 2 h. The western bands were
visualized using ECL detection reagent (Pierce; Thermo Fisher
Scientific, Inc.). For semi-quantification of band intensity,
appropriate films were scanned and band densities were determined
using Quantity One software (version 4.6.6; Bio-Rad Laboratories,
Inc.), normalized against GAPDH, and presented as a ratio of
control.
CCK-8 assay
The PCa cells were incubated for 24 h and then
collected for the preparation of a cell suspension. Cells were then
transferred into a 96-well plate (2,000 cells/well); group
contained three replicate wells. The proliferation potency of PCa
was evaluated at four time points: 0, 1, 2 and 3 days after the
implantation. After each time point, cells were treated with the
CCK-8 solution (Dojindo Molecular Technologies, Inc.) for 2 h. The
absorbance at 450 nm wavelength was determined using a Multilabel
Reader (Thermo Fisher Scientific, Inc.).
Transwell migration and invasion
assays
The PCa cells were resuspended in DMEM (Gibco;
Thermo Fisher Scientific, Inc.) without FBS, and the cell
suspension containing 1×105 cells was transferred into
the upper compartment of Transwell inserts (Costar; Corning, Inc.)
in 24-well plates, while the lower compartment was covered with
DMEM that was supplemented with 10% FBS. After incubation at 37°C
for 24 h, non-migratory cells were removed by cotton swab, while
the migratory cells were fixed in 4% paraformaldehyde and stained
with 0.5% crystal violet at room temperature for 10 min. Cell
migration was analyzed by counting the migratory cells in five
random visual fields per plate using a light microscope (Olympus
Corporation). The Transwell invasion assay was performed using the
same procedures as those for the migration assay, except the
24-well plates were precoated with Matrigel (37°C for 30 min).
Cell cycle assay
Cells were collected 36 h after transfection and
then centrifuged at 160 × g for 5 min. Subsequently, cells were
fixed with 500 µl 70% cold ethanol for 4°C at 2 h, stained with 500
µl PI/RNase staining solution, and incubated at 37°C for 30 min in
the dark. The cell samples were analyzed by flow cytometry using a
FACScan flow cytometer (BD Biosciences). All the results were
analyzed using FlowJo software (version 10; FlowJo LLC).
Apoptosis assay
The early apoptosis assay of the PCa cells was
performed using an Annexin V-FITC/PI staining kit (Nanjing
Jiancheng Bioengineering Research Institute), according to the
manufacturer's instructions. The PCa cells were cultured in 12-well
plates and then resuspended in binding buffer 36 h after
transfection, followed by staining with Annexin V-FITC/PI in the
dark at room temperature for 15 min.
Statistical analysis
All experiments were independently performed in
triplicate. Data are presented as the mean ± standard deviation.
Data analysis was performed using SPSS software version 13.0 for
Windows (SPSS, Inc.). Results were compared by one-way ANOVA
followed by post hoc Tukey's tests. P<0.05 was considered to
indicate a statistically significant difference.
Results
Akt1 is regulated by miR-149 in CRPC
cell line C4-2
To clarify the biological role of miR-149 in PCa,
miR-149 mimic and miR-149 inhibitor were transfected into C4-2
cells, after which the transfection efficiency was confirmed by
RT-qPCR (Fig. 1A). The results
showed that miR-149 mimic significantly increased the expression
levels of miR-149, while miR-149 inhibitor significantly decreased
the expression levels of miR-149 (P<0.05). Furthermore, RT-qPCR
assay was carried out to investigate the influence of miR-149 on
the expression of Akt1 and mTOR in C4-2 cells. As shown in Fig. 1B and C, transfection of miR-149
mimics significantly reduced the mRNA expression levels of Akt1 and
mTOR in C4-2 cells, while the miR-149 inhibitor exerted the
opposite effect (P<0.05). The effect of miR-149 mimic and
miR-149 inhibitor on the protein expression levels of Akt1, p-Akt1,
p-PI3K, PI3K, p-mTOR, mTOR and PTEN were also investigated by
western blotting (Fig. 1G). As a
result, the expression levels of Akt1, p-Akt1 and p-mTOR were
significantly decreased in C4-2 CRPC cells transfected with miR-149
mimic, while miR-149 inhibitor exerted the opposite effects
compared with the NC groups (P<0.05; Fig. 1D-F). In addition, while p-Akt and
p-mTOR expression was altered when compared with β-actin, p-PI3K,
p-Akt and p-mTOR expression did not differ when compared with total
protein in these groups (P>0.05; Fig. 1H-J). Moreover, the expression of
PI3K, p-PI3K and PTEN did not significantly differ in these groups.
The present results revealed that miR-149 overexpression did not
affect the protein expression of PI3K, p-PI3K and PTEN, which
regulate Akt1 expression in the PI3K/Akt signaling pathway (data
not shown). Collectively, these results suggested that miR-149
targeted Akt1 and negatively regulated the PI3K/AKT signaling
pathway in PCa cells.
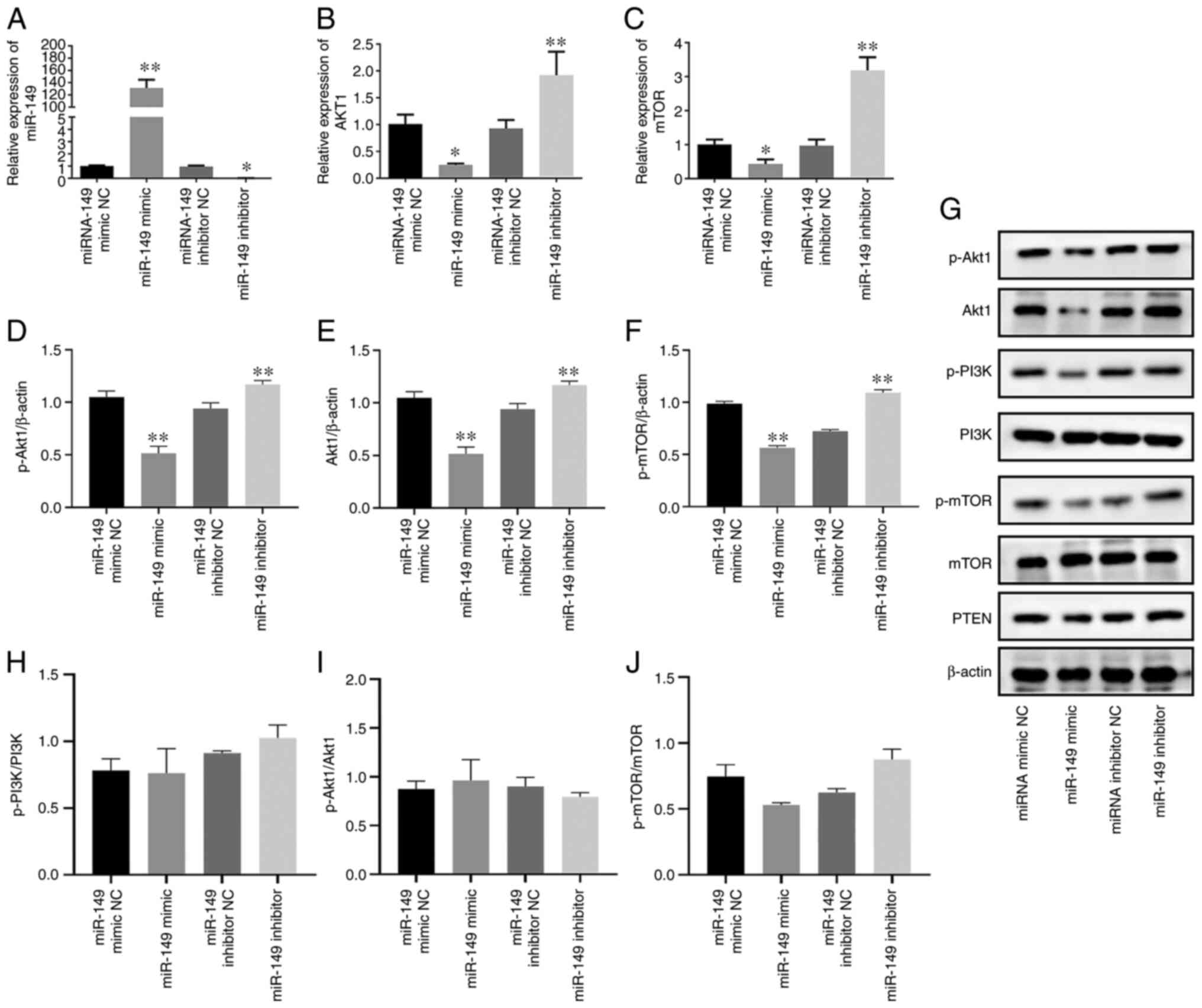 | Figure 1.Akt1 is regulated by miR-149 in
castration-resistant prostate cancer cells. Reverse
transcription-quantitative PCR analysis of (A) miR-149, (B) Akt1
and (C) mTOR mRNA expression levels in C4-2 cells transfected with
miR-149 mimic and miR-149 inhibitor. Semi-quantification of western
blotting results demonstrated the expression of (D) p-Akt1/β-actin,
(E) Akt1/β-actin and (F) p-mTOR/β-actin. (G) Western blot analysis
of Akt, p-Akt, PI3K, p-PI3K, p-mTOR, mTOR and PTEN expression in
C4-2 cells transfected with miR-149 mimic and miR-149 inhibitor.
Semi-quantification of western blotting results demonstrated the
expression of (H) p-PI3K/PI3K, (I) p-Akt1/Akt1 and (J) p-mTOR/mTOR.
Data are expressed as the mean ± standard deviation (n=3); each bar
represents the mean of three independent experiments carried out in
triplicate. *P<0.05, **P<0.01 vs. NC group. miR, microRNA;
p-, phosphorylated; NC, negative control. |
AR is regulated by Akt1 in CRPC cell
line C4-2
As AR has an important role in PCa initiation and
progress (3), it was examined
whether Akt1 participates in the AR signaling pathway and thereby
regulates carcinogenesis, including cancer progression. Initially,
Akt1 was knocked down in C4-2 cells using RNA interference and
proof of transfection was confirmed (Fig. S1). Western blotting results
indicated that the protein expression of p-Akt1 was significantly
decreased in the Akt1 KD + miR-149 mimic NC group compared with the
Akr1 NC + miR-149 mimic NC group (P<0.01). Furthermore, the
effects of Akt1 shRNA and miR-149 mimics on the expression levels
of AR and AR-driven proteins were investigated. As a result, the
protein expression levels of AR, PSA and FKBP5 were significantly
decreased in the Akt1 KD + miR-149 mimic NC, Akt1 NC + miR-149
mimic and Akt1 KD + miR-149 mimic groups compared with the Akt1 NC
+ miR-149 mimic NC group (P<0.01; Fig. 2A and B). Collectively, these
results suggested that miR-149 targeted Akt1 and negatively
regulated the AR signaling pathway in PCa cells.
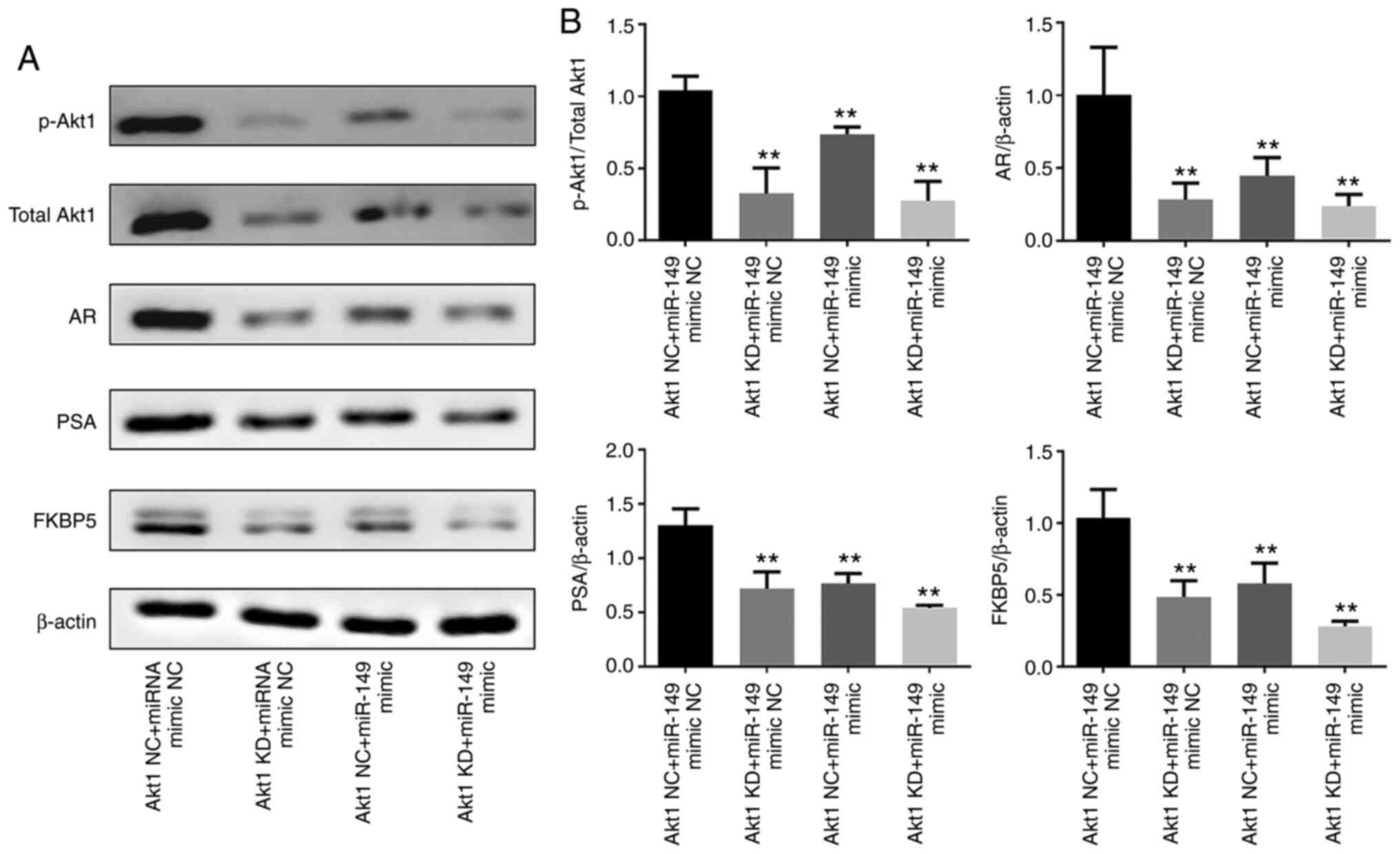 | Figure 2.AR is regulated by Akt1 in the
castration-resistant prostate cancer cell line. (A) Western blot
analysis of p-Akt1, total Akt1, AR, PSA and FKBP5 expression in
C4-2 cells transfected with miR-149 mimic and miR-149 inhibitor.
(B) Semi-quantification of western blotting results demonstrated
the expression of p-Akt1/total Akt1, AR/β-actin, PSA/β-actin and
FKBP5/β-actin. Data are expressed as the mean ± standard deviation
(n=3). **P<0.01 vs. Akt1 NC + miR-149 mimic NC group. AR,
androgen receptor; miR, microRNA; p-, phosphorylated; NC, negative
control; FKBP5, peptidyl-prolyl cis-trans isomerase FKBP5; KD,
knockdown. |
Effects of miR-149 and Akt1-shRNA on
cell proliferation and apoptosis in CRPC cell line C4-2
To confirm the tumor-suppressive role of miR-149,
ectopic expression assays were carried out by miRNA and shRNA
transfection into PCa cells. The CRPC cell line C4-2 was
transfected with miR-149 mimic and Akt1-shRNA. CCK-8 assay was
performed to determine the influence of miR-149 and Akt1-shRNA on
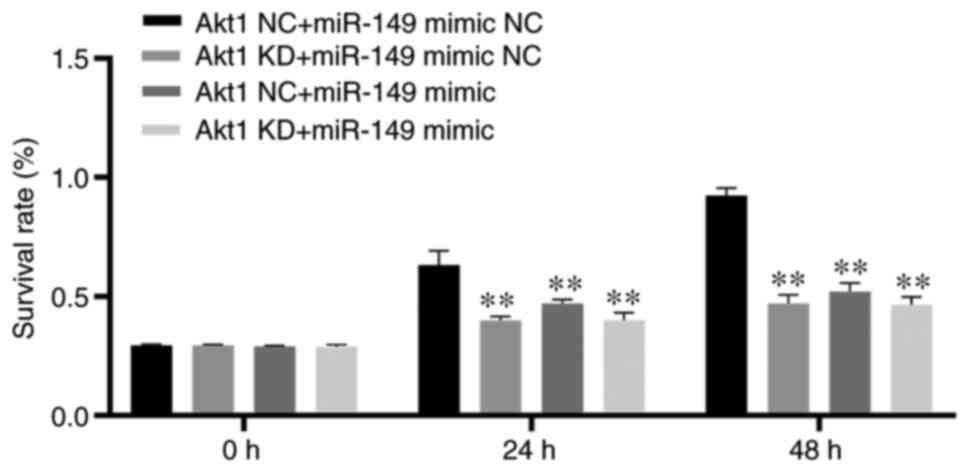
CRPC cell proliferation. As a result, the CCK-8 assay showed that
proliferation was significantly decreased in the Akt1 KD + miR-149
mimic NC, Akt1 NC + miR-149 mimic and Akt1 KD + miR-149 mimic
groups compared with the Akt1 NC + miR-149 mimic NC group
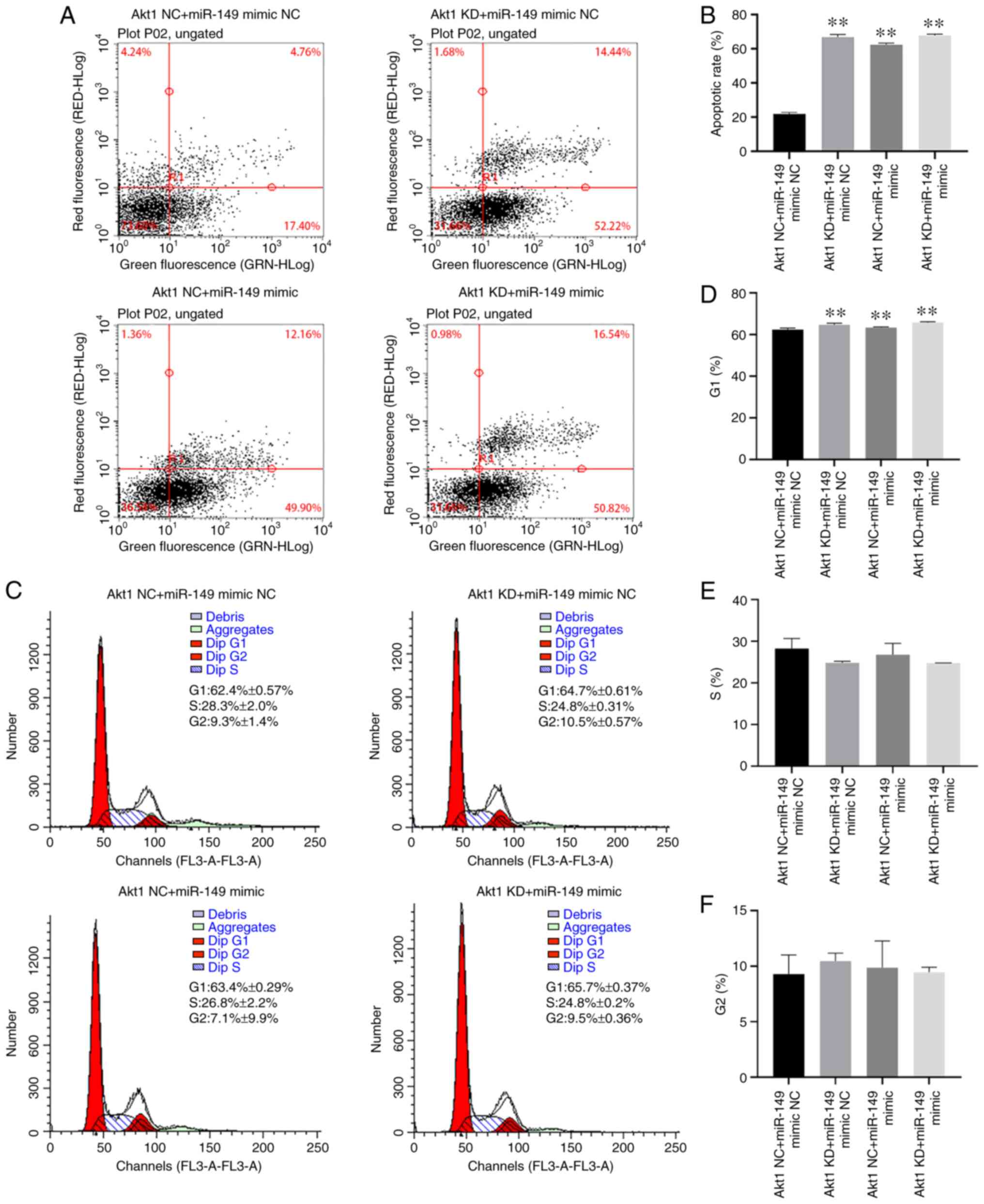
(P<0.01; Fig. 3). FACS analysis
was also performed to evaluate the effect of miR-149 mimic and
Akt1-shRNA transfection on the apoptosis of CRPC cells. The data
indicated that apoptosis of C4-2 cells was significantly enhanced
in the Akt1 KD + miR-149 mimic NC, Akt1 NC + miR-149 mimic and Akt1
KD + miR-149 mimic groups compared with the Akt1 NC + miR-149 mimic
NC group (P<0.01; Fig. 4A and
B). In addition, with the cell cycle assay results, there was a
significant increase of the G1 cell proportion in the Akt1 KD +
miR-149 mimic NC, Akt1 NC + miR-149 mimic and Akt1 KD + miR-149
mimic groups compared with the Akt1 NC + miR-149 mimic NC group
(P<0.01; Fig. 4C-F).
Effects of miR-149 and Akt1-shRNA on
cell migration and invasion in CRPC cell line C4-2
To investigate the biological function of miR-149 on
the aggressiveness of CRPC cells, Transwell assays were performed
to investigate the effect of miR-149 and Akt1-shRNA on CRPC cell
migration and invasion. Results of the Transwell assay with or
without Matrigel showed that migration and invasion of C4-2 cells
were significantly inhibited in the Akt1 KD + miR-149 mimic NC,
Akt1 NC + miR-149 mimic and Akt1 KD + miR-149 mimic groups compared
with the Akt1 NC + miR-149 mimic NC group (P<0.05; Fig. 5A-D).
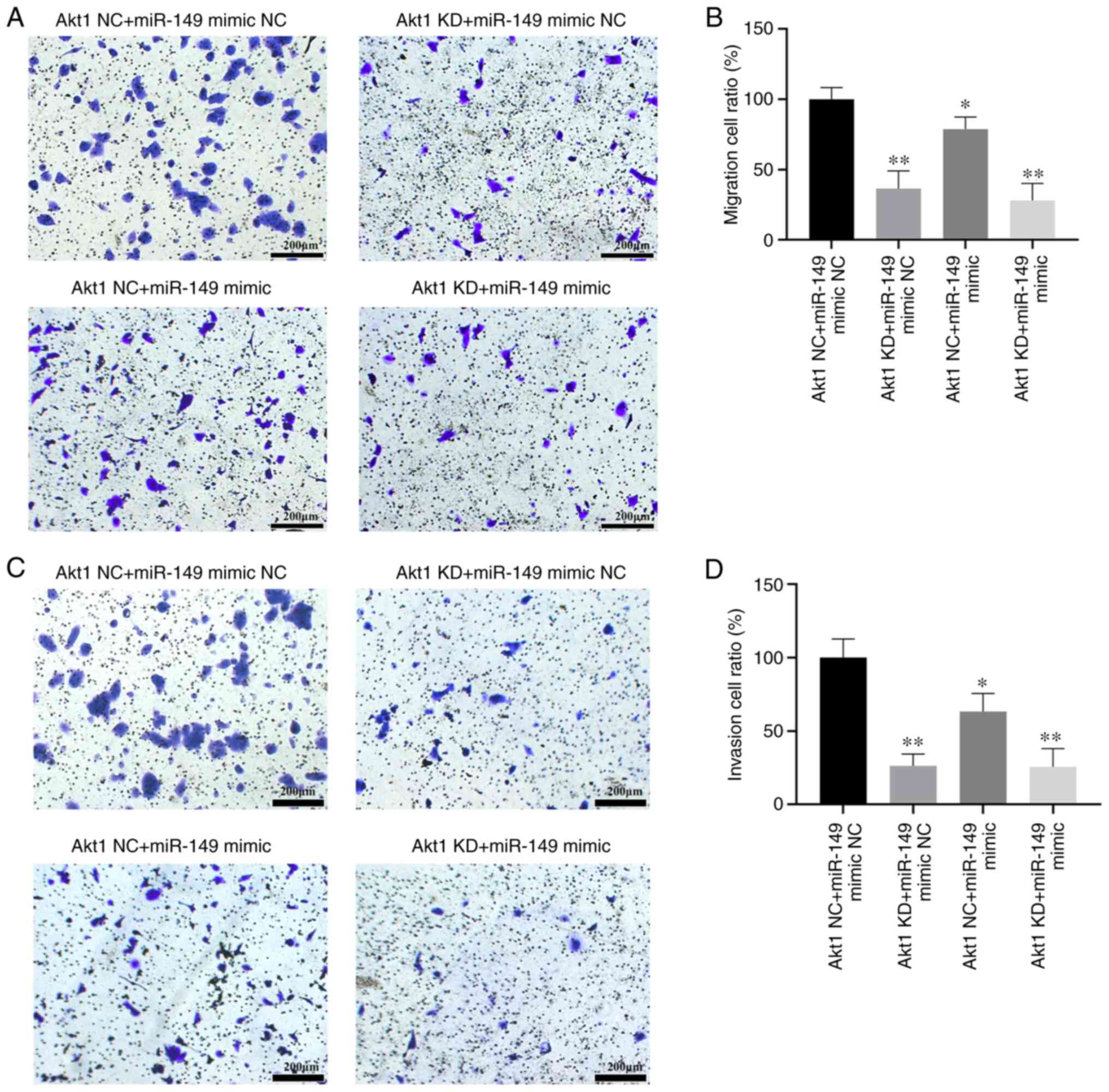 | Figure 5.Effects of miR-149 on cell migration
and invasion in CRPC cell line. CRPC cells were seeded onto
Matrigel-coated or non-Matrigel-coated Transwell chambers for 48 h.
The number of cells on the underside of the filter was determined
after 48 h. (A) The effect of transfection with Akt1 NC + miR-149
mimic NC, Akt1 KD + miR-149 mimic NC, Akt1 NC + miR-149 mimic or
Akt1 KD + miR-149 mimic on cell migration. (B) Transfection with
miR-149 mimics or Akt1 shRNA resulted in significant inhibition of
C4-2 cell migration. (C) The effect of transfection with Akt1 NC +
miR-149 mimic NC, Akt1 KD + miR-149 mimic NC, Akt1 NC + miR-149
mimic or Akt1 KD + miR-149 mimic on cell invasion. (D) Transfection
with miR-149 mimics or Akt1 shRNA resulted in significant
inhibition of C4-2 cell invasion. Scale bar, 200 µm. Data are
expressed as the mean ± standard deviation (n=3). *P<0.05,
**P<0.01 vs. Akt1 NC + miR-149 mimic NC group. miR, microRNA;
NC, negative control; KD, knockdown; CRPC, castration-resistant
prostate cancer; shRNA, short hairpin RNA. |
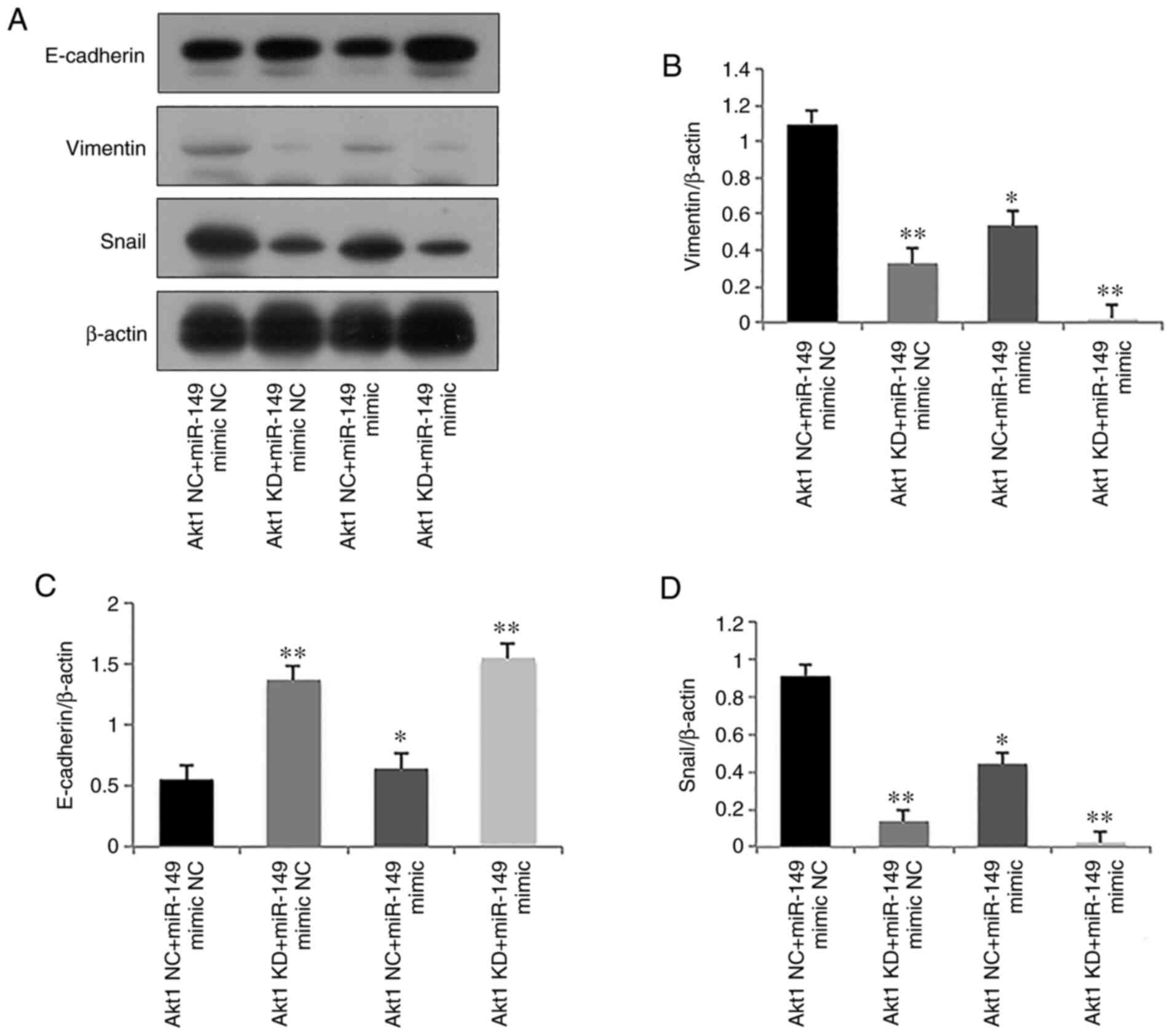
EMT has an important role during tumor cell invasion
(20). To assess the effect of
miR-149 on EMT, E-cadherin (epithelial marker), Snail and Vimentin
(mesenchymal markers) expression levels were analyzed in C4-2
cells. Significantly elevated expression of E-cadherin, and
significantly reduced expression levels of Vimentin and Snail were
observed in the Akt1 KD + miR-149 mimic NC, Akt1 NC + miR-149 mimic
and Akt1 KD + miR-149 mimic groups compared with the Akt1 NC +
miR-149 mimic NC group (P<0.05, Fig. 6A-D), which indicated that
transfection with miR-149 mimics and Akt1-shRNA inhibited the
migratory and invasive abilities of C4-2 cells by repressing
EMT.
Discussion
miR-149 has been implicated in tumor progression by
regulating cell proliferation, migration, invasion and
chemoresistance (21). It has been
reported that downregulation of miR-149 is associated with advanced
tumor progression and poor prognosis of human hepatocellular
carcinoma (HCC). Furthermore, a multivariate analysis performed in
a previous study found that miR-149 expression was an independent
prognostic factor for poor 5-year disease-free survival and 5-year
overall survival in HCC (22). In
addition, miR-149-3p has a high capacity to discriminate between
patients with melanoma and healthy controls, making it suitable to
be used in early melanoma diagnosis (23).
To date, only a limited number of studies have
reported on the effect of miR-149 on the malignant characteristics
of PCa. In our previous study, it was revealed that the ectopic
expression of miR-149 mimic could significantly decrease the
capacity of proliferation and colony formation in TSU-derived SP
cells, promote apoptosis and inhibit the growth rate of tumors
in vitro (17). In the
current study, the potential role and functional mechanism of
miR-149 in the development of CRPC was further investigated. These
results revealed that ectopic expression of miR-149 mimic could
induce cell cycle arrest and apoptosis of CRPC cells. As
malignancies are associated with increased cancer cell
proliferation and invasion, the present study also demonstrated
that ectopic miR-149 mimic resulted in an inhibition of PCa
proliferation and invasion. EMT is an essential process in cancer
progression and metastasis, characterized by changes in epithelial
and mesenchymal marker gene expression and changes in cell
morphology (20). Emerging
evidence has indicated that miRNAs have a vital role in the
regulation of progression and EMT in numerous types of cancer cells
(24). In the current study, it
was observed that transfection with the miR-149 mimic could
increase the expression of epithelial markers and decrease the
expression of mesenchymal markers, which suggested that miR-149
regulated migration and invasion by repressing EMT. The present
results suggested that miR-149 attenuates the aggressiveness of
C4-2 cells and acts as a tumor suppressor gene in CRPC cells.
The PI3K/Akt signaling pathway is one of the most
commonly dysregulated pathways in malignant cells. Aberrant
activity of the PI3K/Akt signaling pathway has been found at early
and advanced stages of PCa (25).
Akt1, a serine/threonine-protein kinase, phosphorylates the key
molecules, leading to increased cellular growth and survival
(26). Over previous years,
several studies have indicated that the PI3K/Akt signaling pathway
is regulated by miRNAs in various types of cancer (27–29).
It has been reported that miR-26a can activate the PI3K/Akt
signaling pathway by inhibiting the expression of PTEN, thus
suggesting that miR-26a may be a potential therapeutic target for
lung cancer (30). Furthermore, a
number of previous studies have reported that miR-149 can suppress
cancer cell malignant phenotypes by regulation of Akt1 in a number
of different types of human cancers (22,31,32).
Zhang et al (22) also
reported that miR-149 plays a tumor-suppressive role by targeting
Akt1 expression in human HCC. This study also identified that the
increased expression of miR-149 significantly inhibited HCC cell
proliferation and invasion by regulating the PI3K/Akt signaling
pathway (22).
Consistent with our previous study, the current
study found that the PI3K/Akt signaling pathway is regulated by
miR-149 in CRPC cells. Mechanistically, miR-149 could inhibit the
expression of Akt1 at the mRNA level, reducing the expression of
total Akt1 and p-Akt1 protein, thus inhibiting the expression of
mTOR, the target gene of the PI3K/Akt signaling pathway. PTEN is a
critical regulator of PI3K/Akt signaling, which can inhibit the
phosphorylation of Akt1 protein, thus inhibiting the functional
activity of the PI3K/Akt signal pathway (29). These results indicated that miR-149
regulated Akt1 expression via targeting Akt1 mRNA rather than
regulating PI3K, p-PI3K and PTEN expression.
In addition, Akt1-shRNA was also used to investigate
the effect of the PI3K/Akt signaling pathway on the aggressiveness
of CRPC cells. As a result, it was found that knockdown of Akt1 had
a similar tumor suppressive effect as miR-149 mimics, which could
inhibit cell invasion and proliferation, and induce cell cycle
arrest and apoptosis of CRPC cells.
AR belongs to the steroid hormone subfamily of
nuclear hormone receptors and mediates the signaling of androgens
(3). Despite failures in treating
recurrent CRPC, it is widely accepted that AR signaling is a
driving force in CRPC progression; thus, drugs reducing AR levels
and AR activity are likely to be effective in CRPC treatment
(33,34). Unfortunately, acquired drug
resistance is an increasing clinical problem, so novel biomarkers
and treatments for PCa and CRPC are urgently needed (35). The PI3K/Akt and AR signaling
pathways have been shown to cross-regulate through reciprocal
inhibitory activity (8).
Consequently, the PI3K/Akt signaling pathway can be activated in
response to androgen or AR blockade therapy in human PCa,
facilitating CRPC growth; vice versa, inhibition of the PI3K/Akt
signaling pathway can augment AR signaling, thus leading to
therapeutic resistance (6,8). In addition, it has also been reported
that AR is an additional substrate for Akt, which can phosphorylate
AR and inhibit AR target genes from inducing CRPC apoptosis
(36).
The present study demonstrated that AR expression
was regulated by miR-149 and the PI3K/Akt signaling pathway.
Silencing of Akt1 by Akt1-shRNA inhibited the expression of AR,
attenuated the proliferation and invasion, and induced apoptosis of
CRPC cells. These results furthered the understanding of the roles
of the PI3K/Akt1 signaling pathway involved in AR-regulated PCa
progression and the classic roles of AR in PI3K/Akt-mediated cancer
cell apoptosis. The association between the PI3K/Akt and AR
signaling pathways could provide another perspective for
investigating cancer cell growth and apoptosis.
The present study has several limitations. First,
only one type of CRPC cell was used for the in vitro cell
assay. In addition, the effect of miR-149 on the malignant
phenotypes in CRPC was not investigated in vivo in this
study. Further experiments are still required to explore the role
of the miR-149/Akt1 signaling pathway in the progression of
CRPC.
In conclusion, it was demonstrated that ectopic
miR-149 could inhibit the expression levels of AR, PSA and
AR-driven genes in C4-2 cells in association with a blockade of
Akt1 inactivation, thus leading to alterations of gene expression
involved in cell cycle progression, migration, invasion and
apoptosis. These findings uncovered the potential role of the
miR-149/Akt1 signaling pathway in the progression of CRPC, which
indicated that inhibition of Akt1 expression via miR-149
overexpression could be a novel therapeutic target for the
treatment of CRPC. Inhibition of oncogenic miRNAs or delivery of
tumor-suppressive miRNAs could become a novel treatment strategy
for PCa.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of Beijing (grant no. 7192053).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YJ and YL made substantial contributions to the
conception and design of the present study. QL, JZ, BF, DW, YH, YW
and ML conducted data acquisition, analysis and interpretation. JZ
and QL drafted the article and critically revised it for important
intellectual content. All authors have read and approved the final
manuscript. YL, JZ and QL agreed to be accountable for all aspects
of the work in ensuring that questions related to the accuracy or
integrity of the work are appropriately investigated and resolved.
All authors provided critical feedback and helped shape the
research, analysis, and manuscript. JZ, QL, YJ and YL confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Siegel RL, Ward EM and Jemal A:
Global cancer incidence and mortality rates and trends-an update.
Cancer Epidemiol Biomarkers Prev. 25:16–27. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ritch C and Cookson M: Recent trends in
the management of advanced prostate cancer. F1000Res. 7:15132018.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dai C, Heemers H and Sharifi N: Androgen
signaling in prostate cancer. Cold Spring Harb Perspect Med.
7:a0304522017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Teo MY, Rathkopf DE and Kantoff P:
Treatment of advanced prostate cancer. Annu Rev Med. 70:479–499.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bastos D and Antonarakis E: CTC-derived
AR-V7 detection as a prognostic and predictive biomarker in
advanced prostate cancer. Expert Rev Mol Diagn. 18:155–163. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shorning BY, Dass MS, Smalley MJ and
Pearson HB: The PI3K-AKT-mTOR pathway and prostate cancer: At the
crossroads of AR, MAPK, and WNT signaling. Int J Mol Sci.
21:45072020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Edlind MP and Hsieh AC: PI3K-AKT-mTOR
signaling in prostate cancer progression and androgen deprivation
therapy resistance. Asian J Androl. 16:378–386. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lee SH, Johnson D, Luong R and Sun Z:
Crosstalking between androgen and PI3K/AKT signaling pathways in
prostate cancer cells. J Biol Chem. 290:2759–2768. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang K, Ruan H, Xu T, Liu L, Liu D, Yang
H, Zhang X and Chen K: Recent advances on the progressive mechanism
and therapy in castration-resistant prostate cancer. Onco Targets
Ther. 11:3167–3178. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rafiei S, Gui B, Wu J, Liu XS, Kibel AS
and Jia L: Targeting the MIF/CXCR7/AKT signaling pathway in
castration-resistant prostate cancer. Mol Cancer Res. 17:263–276.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
He Y, Yu D, Zhu L, Zhong S, Zhao J and
Tang J: miR-149 in human cancer: A systemic review. J Cancer.
9:375–388. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xie Z, Xu J, Peng L, Gao Y, Zhao H and Qu
Y: miR-149 promotes human osteocarcinoma progression via targeting
bone morphogenetic protein 9 (BMP9). Biotechnol Lett. 40:47–55.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li Y, Ju K, Wang W, Liu Z, Xie H, Jiang Y,
Jiang G, Lu J, Dong Z and Tang F: Dinitrosopiperazine-decreased
PKP3 through upregulating miR-149 participates in nasopharyngeal
carcinoma metastasis. Mol Carcinog. 57:1763–1779. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ma J, Wei H, Li X and Qu X: hsa-miR-149-5p
suppresses prostate carcinoma malignancy by suppressing RGS17.
Cancer Manag Res. 13:2773–2783. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang C, Ding T, Yang D, Zhang P, Hu X, Qin
W and Zheng J: The lncRNA OGFRP1/miR-149-5p/IL-6 axis regulates
prostate cancer chemoresistance. Pathol Res Pract. 224:1535352021.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fujii T, Shimada K, Tatsumi Y, Fujimoto K
and Konishi N: Syndecan-1 responsive microRNA-126 and 149 regulate
cell proliferation in prostate cancer. Biochem Biophys Res Commun.
456:183–189. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen Y, Zhao J, Luo Y, Wang Y and Jiang Y:
Downregulated expression of miRNA-149 promotes apoptosis in side
population cells sorted from the TSU prostate cancer cell line.
Oncol Rep. 36:2587–2600. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu HC, Hsieh JT, Gleave ME, Brown NM,
Pathak S and Chung LW: Derivation of androgen-independent human
LNCaP prostatic cancer cell sublines: Role of bone stromal cells.
Int J Cancer. 57:406–412. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Odero-Marah V, Hawsawi O, Henderson V and
Sweeney J: Epithelial-mesenchymal transition (EMT) and prostate
cancer. Adv Exp Med Biol. 1095:101–110. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhi Y, Zhou H, Mubalake A, Chen Y, Zhang
B, Zhang K, Chu X and Wang R: Regulation and functions of
MicroRNA-149 in human cancers. Cell Prolif. 51:e124652018.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang Y, Guo X, Xiong L, Yu L, Li Z, Guo
Q, Li Z, Li B and Lin N: Comprehensive analysis of
microRNA-regulated protein interaction network reveals the tumor
suppressive role of microRNA-149 in human hepatocellular carcinoma
via targeting AKT-mTOR pathway. Mol Cancer. 13:2532014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fogli S, Polini B, Carpi S, Pardini B,
Naccarati A, Dubbini N, Lanza M, Breschi MC, Romanini A and Nieri
P: Identification of plasma microRNAs as new potential biomarkers
with high diagnostic power in human cutaneous melanoma. Tumour
Biol. 39:10104283177016462017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Montanari M, Rossetti S, Cavaliere C,
D'Aniello C, Malzone MG, Vanacore D, Di Franco R, La Mantia E,
Iovane G, Piscitelli R, et al: Epithelial-mesenchymal transition in
prostate cancer: An overview. Oncotarget. 8:35376–35389. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen H, Zhou L, Wu X, Li R, Wen J, Sha J
and Wen X: The PI3K/AKT pathway in the pathogenesis of prostate
cancer. Front Biosci (Landmark Ed). 21:1084–1091. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nóbrega M, Cilião HL, Souza MF, Souza MR,
Serpeloni JM, Fuganti PE and Cólus IMS: Association of
polymorphisms of PTEN, AKT1, PI3K, AR, and AMACR genes in patients
with prostate cancer. Genet Mol Biol. 43:e201803292020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hu M, Zhu S, Xiong S, Xue X and Zhou X:
MicroRNAs and the PTEN/PI3K/Akt pathway in gastric cancer (Review).
Oncol Rep. 41:1439–1454. 2019.PubMed/NCBI
|
|
28
|
Ghafouri-Fard S, Shoorei H and Taheri M:
miRNA profile in ovarian cancer. Exp Mol Pathol. 113:1043812020.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wise HM, Hermida MA and Leslie NR:
Prostate cancer, PI3K, PTEN and prognosis. Clin Sci (Lond).
131:197–210. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chen J, Xu Y, Tao L, Pan Y, Zhang K, Wang
R, Chen LB and Chu X: miRNA-26a contributes to the acquisition of
malignant behaviors of doctaxel-resistant lung adenocarcinoma cells
through targeting EZH2. Cell Physiol Biochem. 41:583–597. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Niu Y, Tang G, Wu X and Wu C: LncRNA NEAT1
modulates sorafenib resistance in hepatocellular carcinoma through
regulating the miR-149-5p/AKT1 axis. Saudi J Gastroenterol.
26:194–203. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pan SJ, Zhan SK, Pei BG, Sun QF, Bian LG
and Sun BM: MicroRNA-149 inhibits proliferation and invasion of
glioma cells via blockade of AKT1 signaling. Int J Immunopathol
Pharmacol. 25:871–881. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
De Nunzio C, Presicce F, Giacinti S,
Bassanelli M and Tubaro A: Castration-resistance prostate cancer:
What is in the pipeline? Minerva Urol Nefrol. 70:22–41.
2018.PubMed/NCBI
|
|
34
|
Galletti G, Leach BI, Lam L and Tagawa ST:
Mechanisms of resistance to systemic therapy in metastatic
castration-resistant prostate cancer. Cancer Treat Rev. 57:16–27.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
James ND, de Bono JS, Spears MR, Clarke
NW, Mason MD, Dearnaley DP, Ritchie AWS, Amos CL, Gilson C, Jones
RJ, et al: Abiraterone for prostate cancer not previously treated
with hormone therapy. N Engl J Med. 377:338–351. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yan Y and Huang H: Interplay among
PI3K/AKT, PTEN/FOXO and AR signaling in prostate cancer. Adv Exp
Med Biol. 1210:319–331. 2019. View Article : Google Scholar : PubMed/NCBI
|