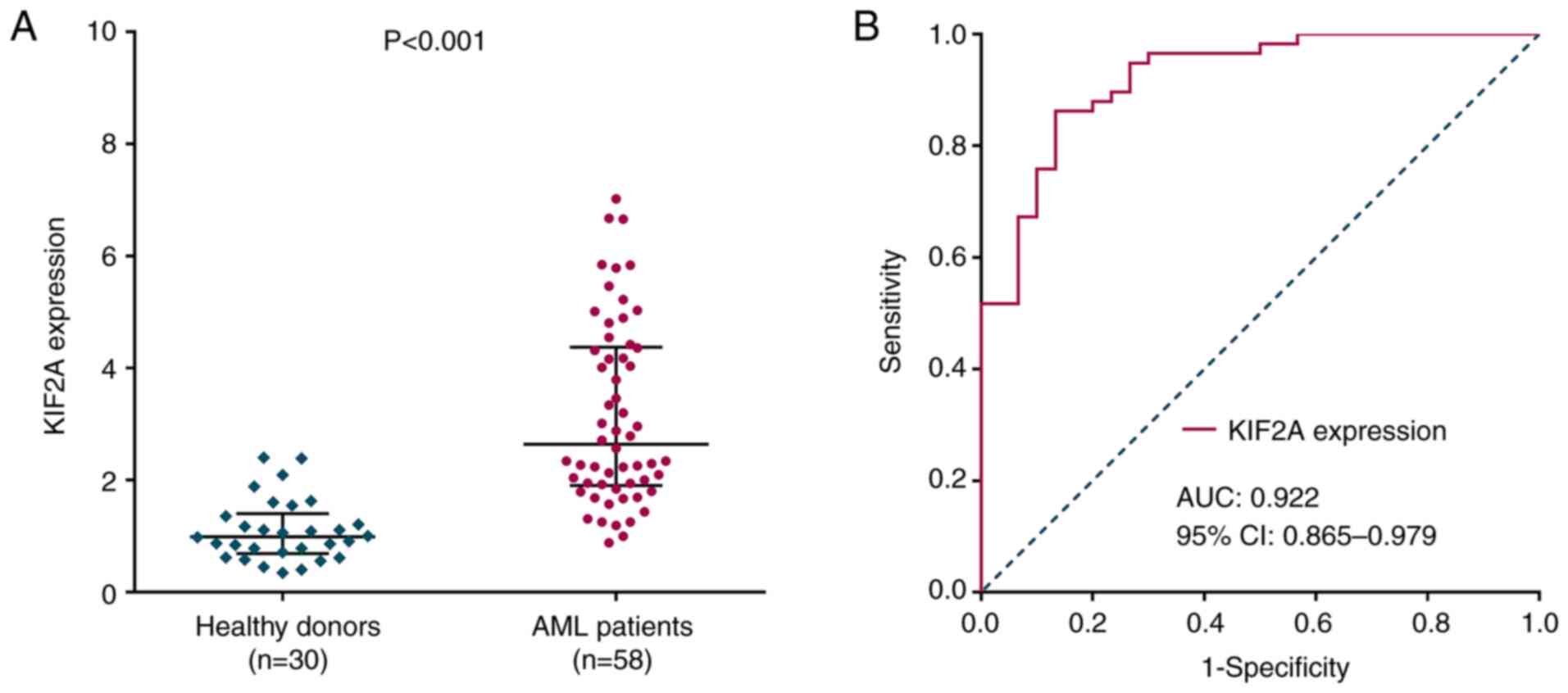
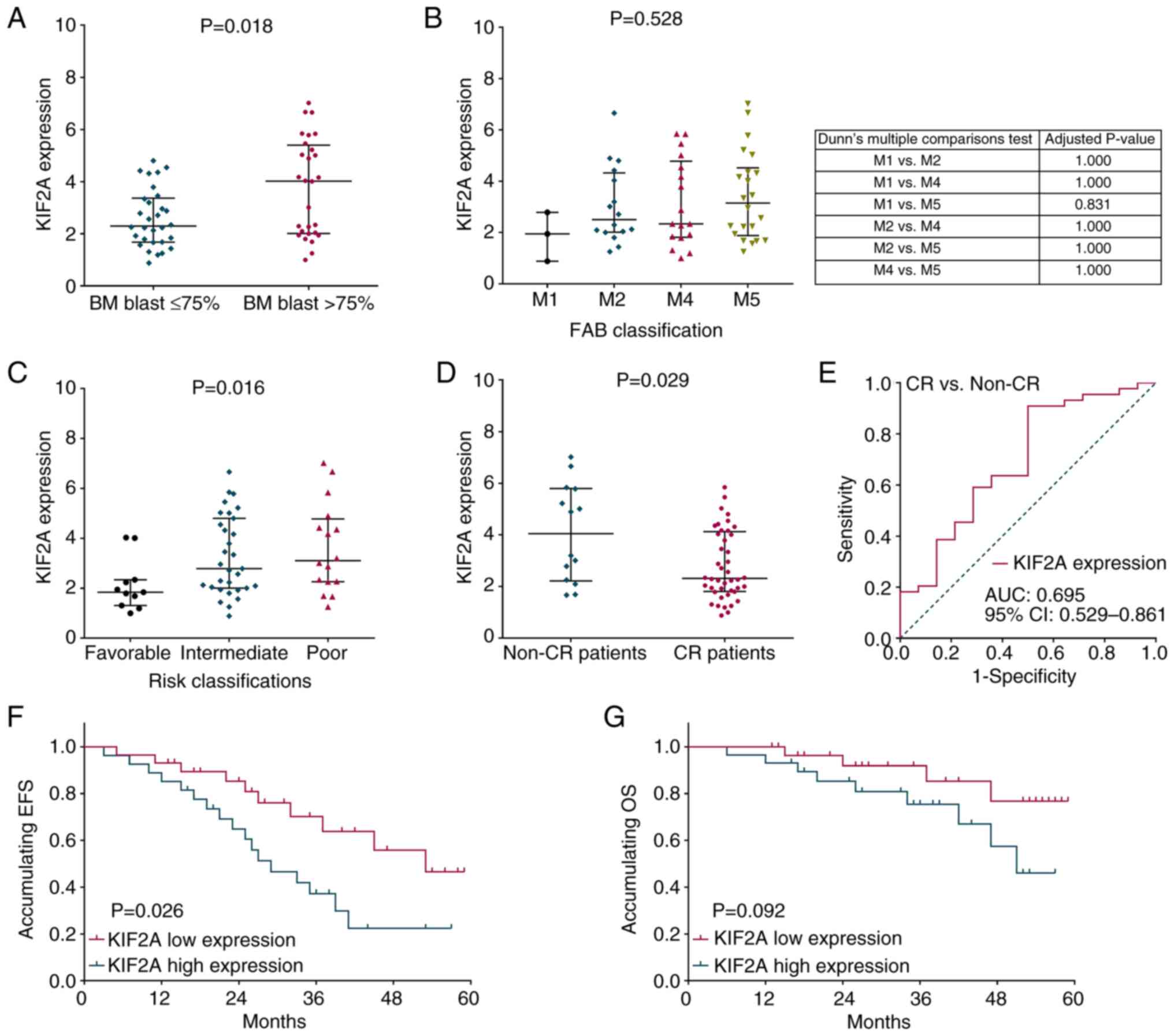
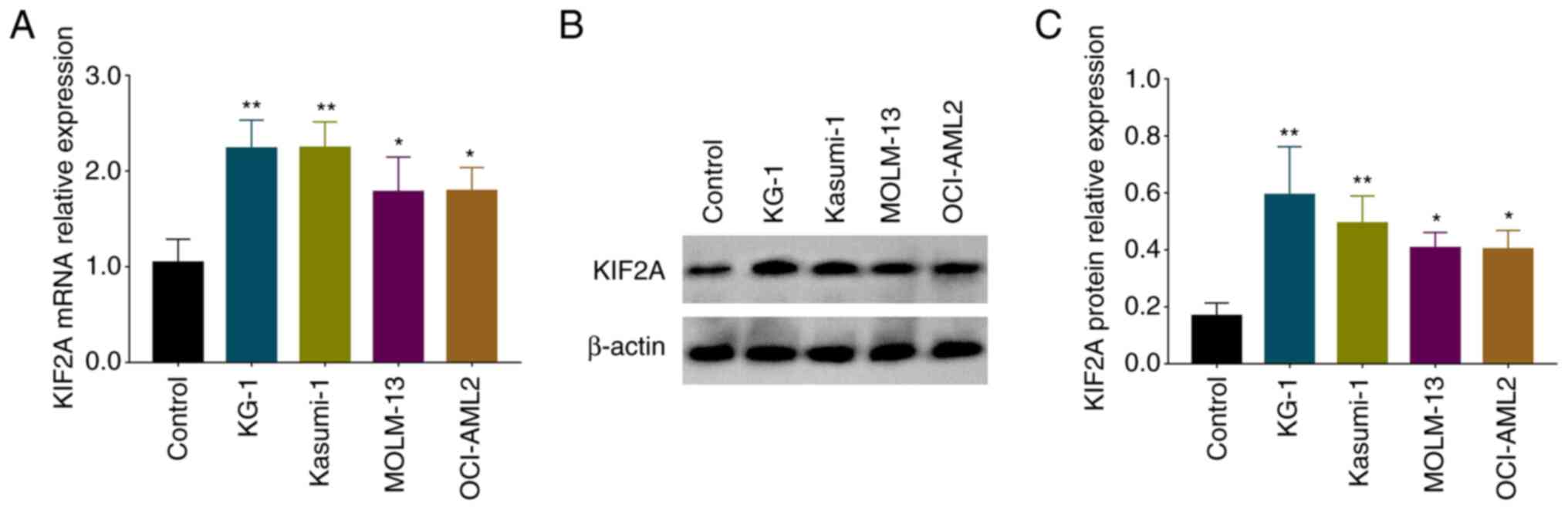
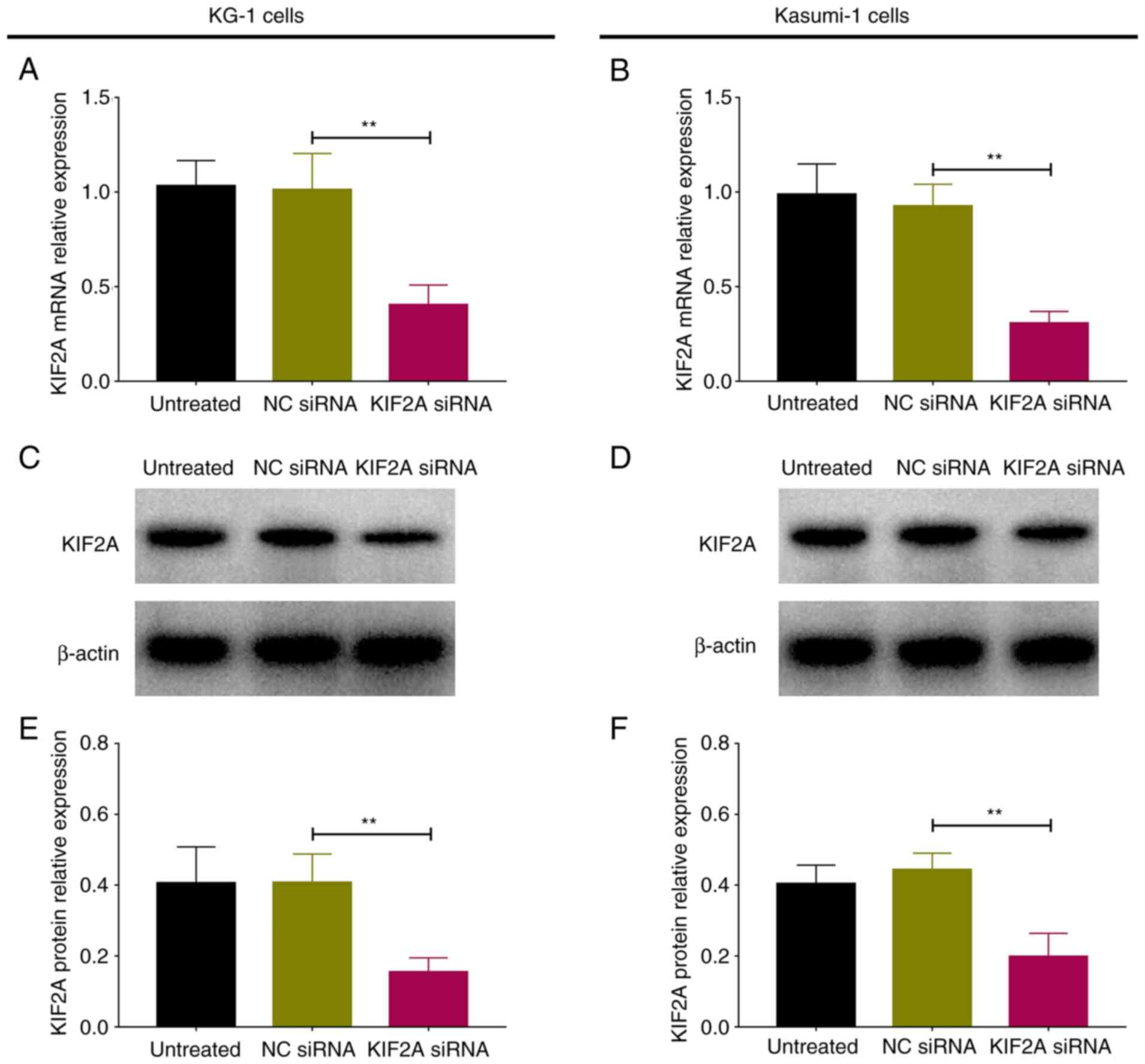
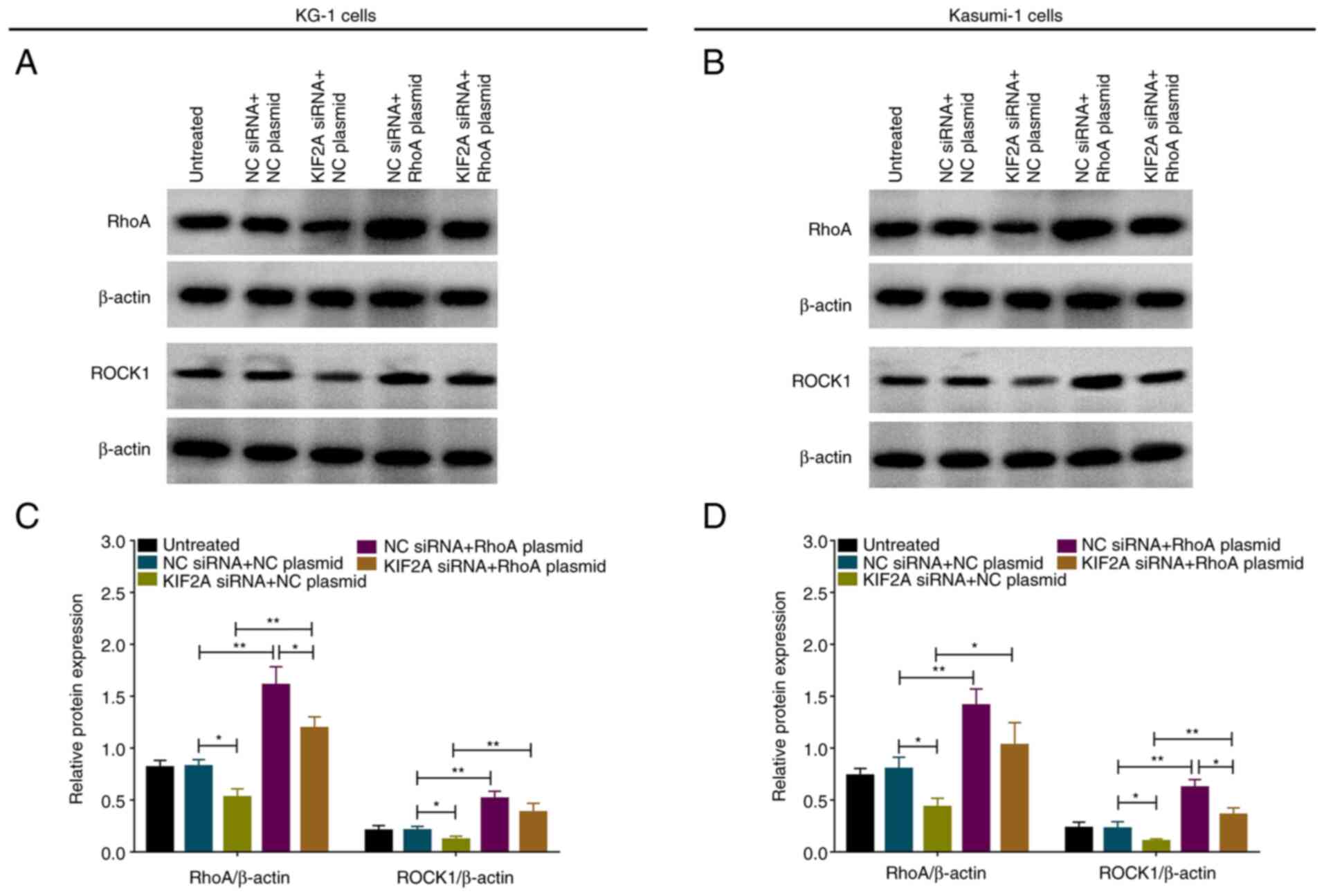
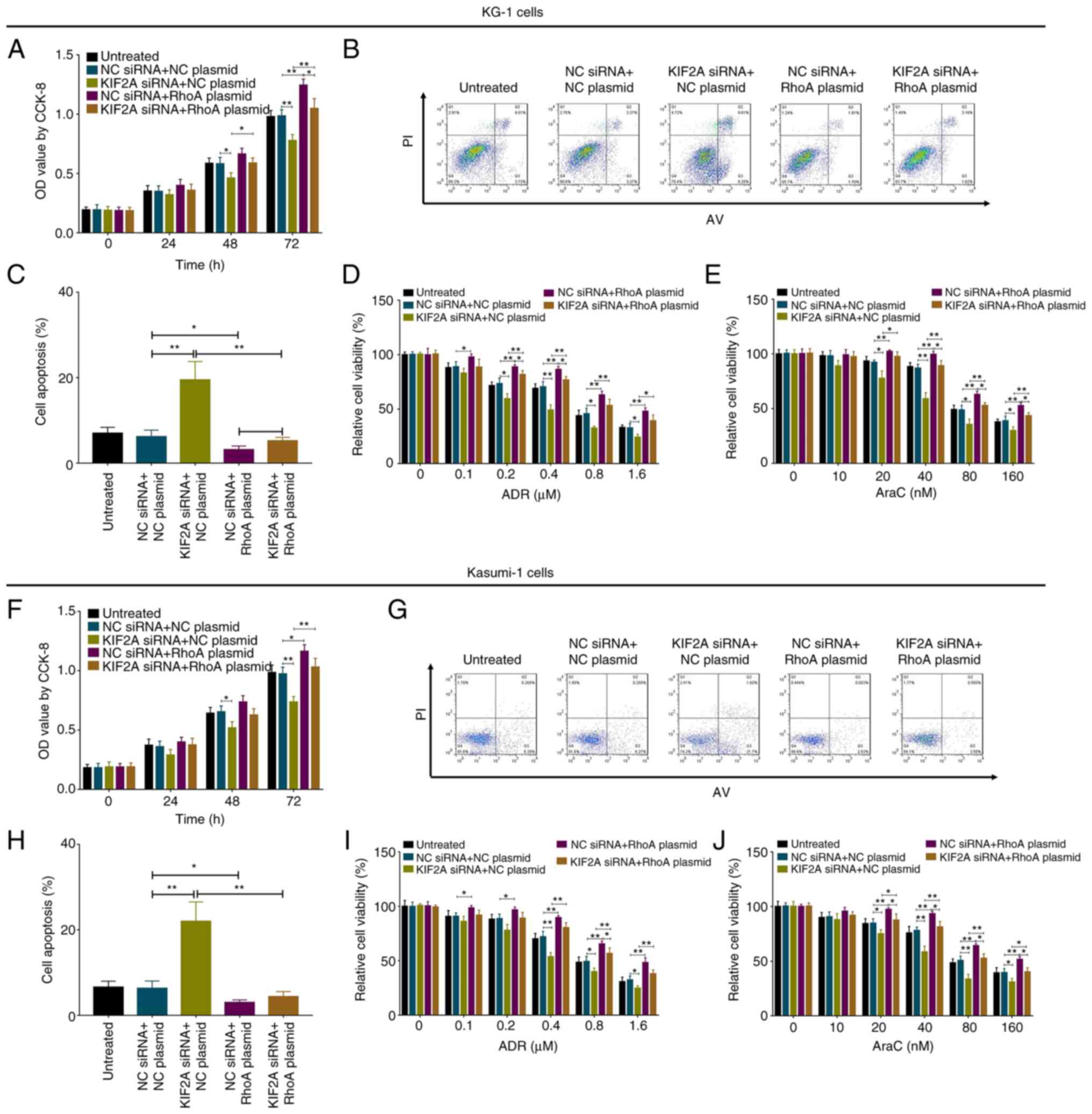
|
1
|
Kuykendall A, Duployez N, Boissel N,
Lancet JE and Welch JS: Acute myeloid leukemia: The good, the bad,
and the ugly. Am Soc Clin Oncol Educ Book. 38:555–573. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jung J, Cho BS, Kim HJ, Han E, Jang W, Han
K, Lee JW, Chung NG, Cho B, Kim M and Kim Y: Reclassification of
acute myeloid leukemia according to the 2016 WHO classification.
Ann Lab Med. 39:311–316. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
National Cancer Institute: SEER Cancer
stat facts: Acute myeloid leukemia (AML). https://seer.cancer.gov/statfacts/html/amyl.html/December
17–2018.
|
|
4
|
National Cancer Institute: SEER Cancer
Statistics Review, 1975-2016. https://seer.cancer.gov/csr/1975_2016/April
18–2019
|
|
5
|
Jetani H, Navarro-Bailón A, Maucher M,
Frenz S, Verbruggen CM, Yeguas A, Vidriales MB, González M,
Saborido JR, Kraus S, et al: Siglec-6 is a novel target for CAR
T-cell therapy in acute myeloid leukemia (AML). Blood. Jul
21–2021.(Epub ahead of print). doi: 10.1182/blood.2020009192.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Michelozzi IM, Kirtsios E and Giustacchini
A: Driving CAR T stem cell targeting in acute myeloid leukemia: The
roads to success. Cancers (Basel). 13:28162021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Song MK, Park BB and Uhm JE: Targeted
therapeutic approach based on understanding of aberrant molecular
pathways leading to leukemic proliferation in patients with acute
myeloid leukemia. Int J Mol Sci. 22:57892021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhao M, Wang J, Qu M, Zhao Y, Wang H, Ke
Y, Liu Y, Lei ZN, Liu HM, Hu Z, et al: OGP46 induces
differentiation of acute myeloid leukemia cells via different
optimal signaling pathways. Front Cell Dev Biol. 9:6529722021.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lu W and Gelfand VI: Moonlighting motors:
Kinesin, dynein, and cell polarity. Trends Cell Biol. 27:505–514.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen K, Nam W and Epureanu BI: Collective
intracellular cargo transport by multiple kinesins on multiple
microtubules. Phys Rev E. 101:0524132020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Scherer J, Yi J and Vallee RB: Role of
cytoplasmic dynein and kinesins in adenovirus transport. FEBS Lett.
594:1838–1847. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ganem NJ and Compton DA: The KinI kinesin
Kif2a is required for bipolar spindle assembly through a functional
relationship with MCAK. J Cell Biol. 166:473–478. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang Q, Lu D, Liu W, Ye S, Guo H, Liao T
and Chen C: Effects of KIF2A on the prognosis of nasopharyngeal
carcinoma and nasopharyngeal carcinoma cells. Oncol Lett.
18:2718–2723. 2019.PubMed/NCBI
|
|
14
|
Wang G, Wang Z and Yu H: Kinesin family
member 2A high expression correlates with advanced tumor stages and
worse prognosis in non-small cell lung cancer patients. J Clin Lab
Anal. 34:e231352020.PubMed/NCBI
|
|
15
|
Ding T, Li J, Sun J, Fan X, Shi C, Zhou D
and Deng R: Association of kinesin family member 2A with increased
disease risk, deteriorative clinical characteristics, and shorter
survival profiles in acute myeloid leukemia. Braz J Med Biol Res.
54:e91732020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang X, Wang Y, Liu X, Zhao A, Yang Z,
Kong F, Sun L, Yu Y and Jiang L: KIF2A promotes the progression via
AKT signaling pathway and is upregulated by transcription factor
ETV4 in human gastric cancer. Biomed Pharmacother. 125:1098402020.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li X, Shu K, Wang Z and Ding D: Prognostic
significance of KIF2A and KIF20A expression in human cancer: A
systematic review and meta-analysis. Medicine (Baltimore).
98:e180402019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li D, Sun H, Meng L and Li D: The
Overexpression of kinesin superfamily protein 2A (KIF2A) was
associated with the proliferation and prognosis of esophageal
squamous cell carcinoma. Cancer Manag Res. 12:3731–3739. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang S, Huang F, Wang Y, Song Q, Yang X
and Wu H: KIF2A overexpression and its association with
clinicopathologic characteristics and poor prognoses in patients
with gastric cancer. Dis Markers. 2016:74845162016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang ZX, Ren SC, Chang ZS and Ren J:
Identification of kinesin family member 2A (KIF2A) as a promising
therapeutic target for osteosarcoma. Biomed Res Int.
2020:71027572020.PubMed/NCBI
|
|
22
|
Zhao P, Lan F, Zhang H, Zeng G and Liu D:
Down-regulation of KIF2A inhibits gastric cancer cell invasion via
suppressing MT1-MMP. Clin Exp Pharmacol Physiol. 45:1010–1018.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang X, Ma C, Wang Q, Liu J, Tian M, Yuan
Y, Li X and Qu X: Role of KIF2A in the progression and metastasis
of human glioma. Mol Med Rep. 13:1781–1787. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Xie T, Li X, Ye F, Lu C, Huang H, Wang F,
Cao X and Zhong C: High KIF2A expression promotes proliferation,
migration and predicts poor prognosis in lung adenocarcinoma.
Biochem Biophys Res Commun. 497:65–72. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Darici S, Alkhaldi H, Horne G, Jørgensen
HG, Marmiroli S and Huang X: Targeting PI3K/Akt/mTOR in AML:
Rationale and clinical evidence. J Clin Med. 9:29342020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nepstad I, Hatfield KJ, Grønningsæter IS
and Reikvam H: The PI3K-Akt-mTOR signaling pathway in human acute
myeloid leukemia (AML) cells. Int J Mol Sci. 21:29072020.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li X, Su Y, Hege K, Madlambayan G, Edwards
H, Knight T, Polin L, Kushner J, Dzinic SH, White K, et al: The
HDAC and PI3K dual inhibitor CUDC-907 synergistically enhances the
antileukemic activity of venetoclax in preclinical models of acute
myeloid leukemia. Haematologica. 106:1262–1277. 2021.PubMed/NCBI
|
|
28
|
Hao Y, Zhang N, Wei N, Yin H, Zhang Y, Xu
H, Zhu C and Li D: Matrine induces apoptosis in acute myeloid
leukemia cells by inhibiting the PI3K/Akt/mTOR signaling pathway.
Oncol Lett. 18:2891–2896. 2019.PubMed/NCBI
|
|
29
|
Annageldiyev C, Tan SF, Thakur S,
Dhanyamraju PK, Ramisetti SR, Bhadauria P, Schick J, Zeng Z, Sharma
V, Dunton W, et al: The PI3K/AKT pathway inhibitor ISC-4 induces
apoptosis and inhibits growth of leukemia in preclinical models of
acute myeloid leukemia. Front Oncol. 10:3932020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xu Y, Li K, Wang SB and Yang SG: MiR-592
functions as a tumor suppressor in acute myeloid leukemia by
targeting ROCK1 and predicts patients' prognosis. Eur Rev Med
Pharmacol Sci. 23:1610–1619. 2019.PubMed/NCBI
|
|
31
|
Liu Y, Gao X and Tian X: High expression
of long intergenic non-coding RNA LINC00662 contributes to
malignant growth of acute myeloid leukemia cells by upregulating
ROCK1 via sponging microRNA-340-5p. Eur J Pharmacol.
859:1725352019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang K, Lin C, Wang C, Shao Q, Gao W, Song
B, Wang L, Song X, Qu X and Wei F: Silencing Kif2a induces
apoptosis in squamous cell carcinoma of the oral tongue through
inhibition of the PI3K/Akt signaling pathway. Mol Med Rep.
9:273–278. 2014. View Article : Google Scholar : PubMed/NCBI
|