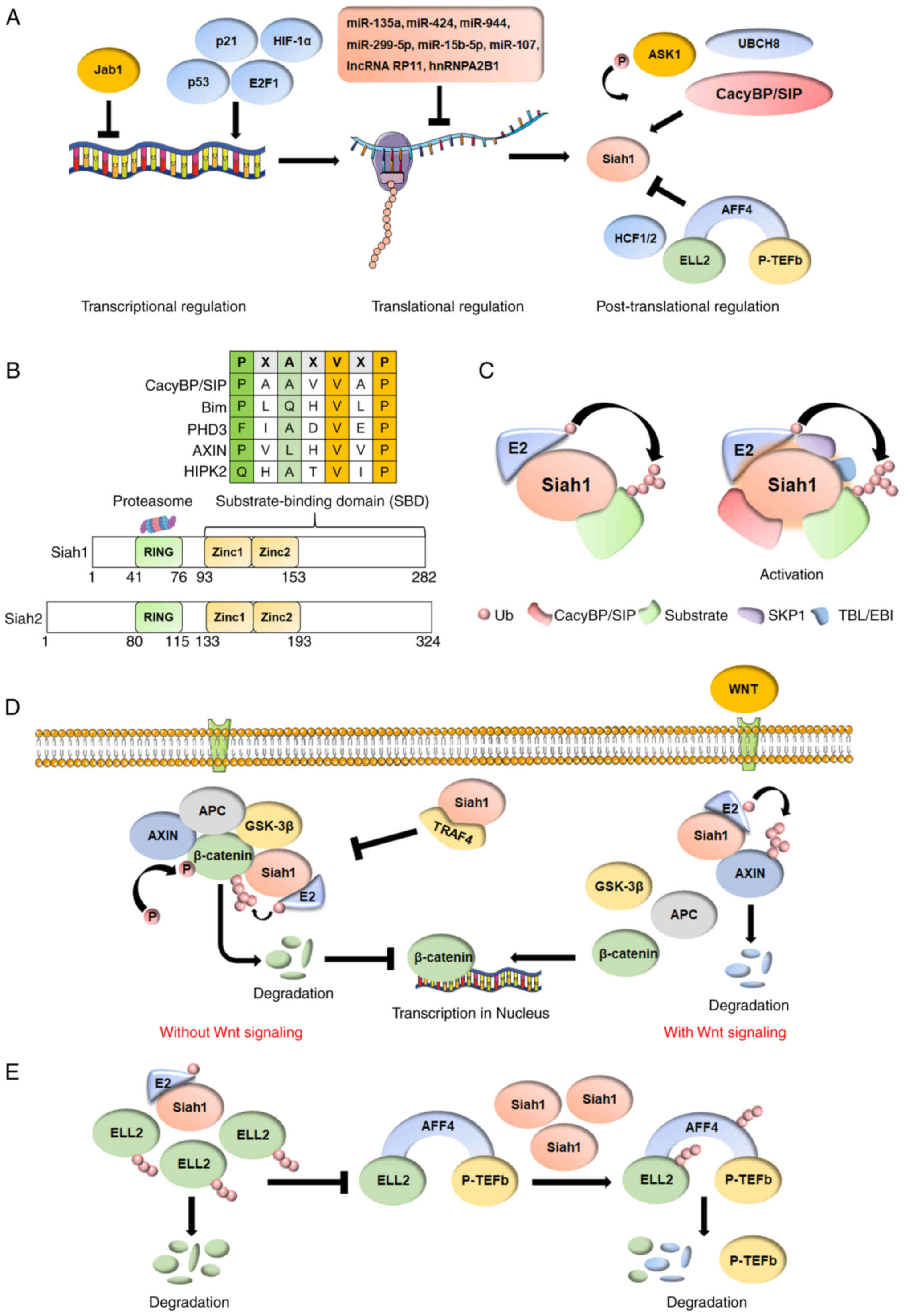
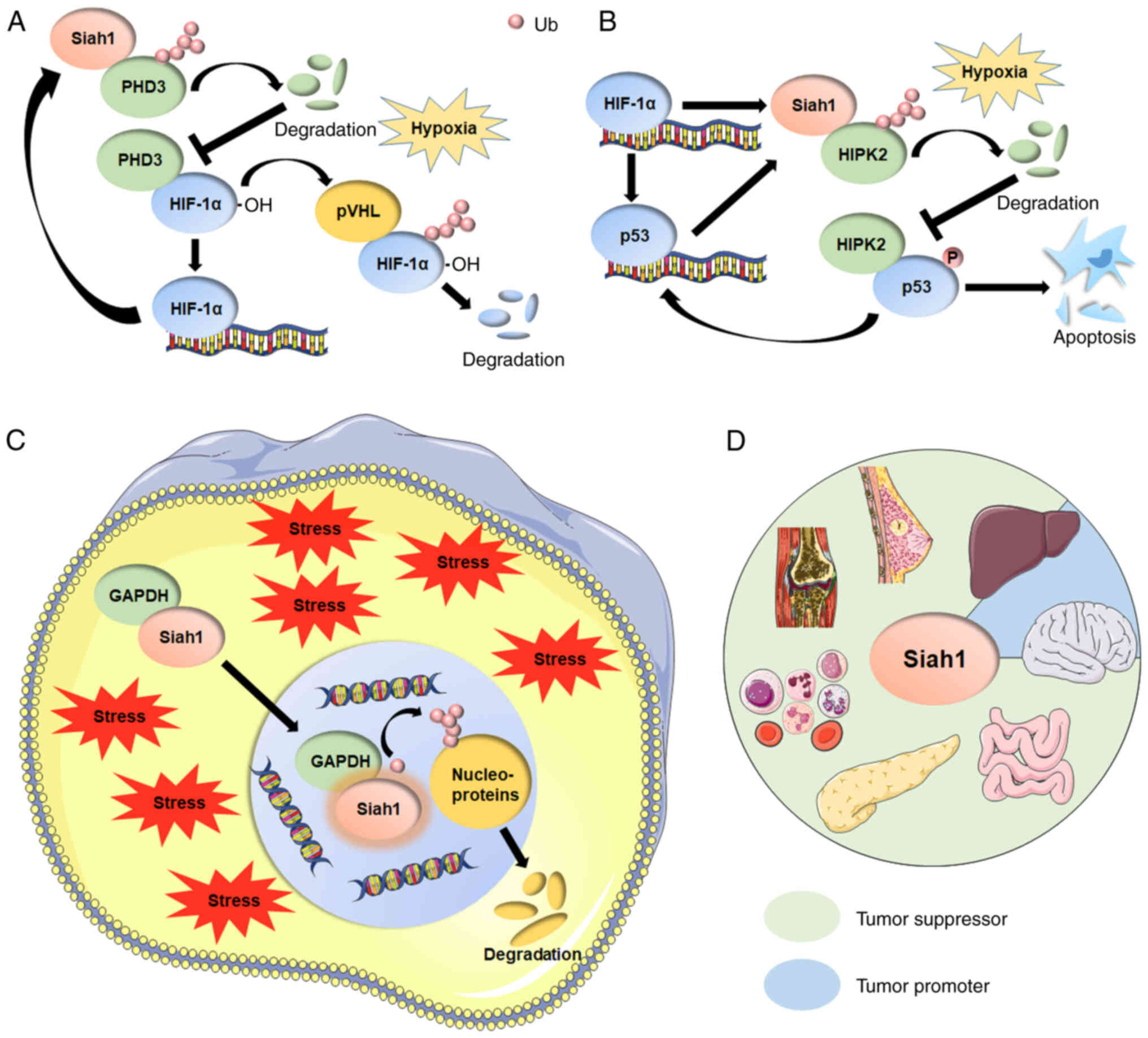
|
1
|
Hershko A and Ciechanover A: The ubiquitin
system. Ann Rev Biochem. 67:425–479. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mani RS: The emerging role of speckle-type
POZ protein (SPOP) in cancer development. Drug Discov Today.
19:1498–1502. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zou T and Zhang J: Diverse and pivotal
roles of neddylation in metabolism and immunity. FEBS J.
288:3884–3912. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen RH: Cullin 3 and its role in
tumorigenesis. Adv Exp Med Biol. 1217:187–210. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nandi D, Tahiliani P, Kumar A and Chandu
D: The ubiquitin-proteasome system. J Biosci. 31:137–155. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cuneo MJ and Mittag T: The ubiquitin
ligase adaptor SPOP in cancer. FEBS J. 286:3946–3958. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang D, Ma L, Wang B, Liu J and Wei W: E3
ubiquitin ligases in cancer and implications for therapies. Cancer
Metastasis Rev. 36:683–702. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang Z, Song Y, Ye M, Dai X, Zhu X and Wei
W: The diverse roles of SPOP in prostate cancer and kidney cancer.
Nat Rev Urol. 17:339–350. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Morreale F and Walden H: Types of
ubiquitin ligases. Cell. 165:248. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yun S, Möller A, Chae SK, Hong WP, Bae YJ,
Bowtell DD, Ryu SH and Suh PG: Siah proteins induce the epidermal
growth factor-dependent degradation of phospholipase Cepsilon. J
Biol Chem. 283:1034–1042. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Qi J, Kim H, Scortegagna M and Ronai ZA:
Regulators and effectors of Siah ubiquitin ligases. Cell Biochem
Biophys. 67:15–24. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Garrison JB, Correa RG, Gerlic M, Yip KW,
Krieg A, Tamble CM, Shi R, Welsh K, Duggineni S, Huang Z, et al:
ARTS and Siah collaborate in a pathway for XIAP degradation. Mol
Cell. 41:107–116. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Santelli E, Leone M, Li C, Fukushima T,
Preece NE, Olson AJ, Ely KR, Reed JC, Pellecchia M, Liddington RC
and Matsuzawa SI: Structural analysis of Siah1-Siah-interacting
protein interactions and insights into the assembly of an E3 ligase
multiprotein complex. J Biol Chem. 280:34278–34287. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang Q, Wang Z, Hou F, Harding R, Huang
X, Dong A, Walker JR and Tong Y: The substrate binding domains of
human SIAH E3 ubiquitin ligases are now crystal clear. Biochim
Biophys Acta Gen Subj. 1861:3095–3105. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Knauer SK, Mahendrarajah N, Roos WP and
Krämer OH: The inducible E3 ubiquitin ligases SIAH1 and SIAH2
perform critical roles in breast and prostate cancers. Cytokine
Growth Factor Rev. 26:405–413. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nakayama K and Ronai Z: Siah: New players
in the cellular response to hypoxia. Cell Cycle. 3:1345–1347. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Famulski JK, Trivedi N, Howell D, Yang Y,
Tong Y, Gilbertson R and Solecki DJ: Siah regulation of Pard3A
controls neuronal cell adhesion during germinal zone exit. Science.
330:1834–1838. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wong CS and Möller A: Siah: A promising
anticancer target. Cancer Res. 73:2400–2406. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
House CM, Möller A and Bowtell DD: Siah
proteins: Novel drug targets in the Ras and hypoxia pathways.
Cancer Res. 69:8835–8838. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Matsuzawa S, Li C, Ni CZ, Takayama S, Reed
JC and Ely KR: Structural analysis of Siah1 and its interactions
with Siah-interacting protein (SIP). J Biol Chem. 278:1837–1840.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Topolska-Woś AM, Chazin WJ and Filipek A:
CacyBP/SIP-structure and variety of functions. Biochim Biophys
Acta. 1860:79–85. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yan S, Li A and Liu Y: CacyBP/SIP inhibits
the migration and invasion behaviors of glioblastoma cells through
activating Siah1 mediated ubiquitination and degradation of
cytoplasmic p27. Cell Biol Int. 42:216–226. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wen YY, Yang ZQ, Song M, Li BL, Yao XH,
Chen XL, Zhao J, Lu YY, Zhu JJ and Wang EH: The expression of SIAH1
is downregulated and associated with Bim and apoptosis in human
breast cancer tissues and cells. Mol Carcinog. 49:440–449. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Briant DJ, Ceccarelli DF and Sicheri F: I
Siah substrate! Structure. 14:627–628. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Czechowicz JS, Nagel CH, Voges M, Spohn M,
Eibl MM and Hauber J: Interaction between the cellular E3 ubiquitin
ligase SIAH-1 and the viral immediate-early protein ICP0 enables
efficient replication of herpes simplex virus type 2 in vivo. PLoS
One. 13:e02018802018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shi H, Zheng B, Wu Y, Tang Y, Wang L, Gao
Y, Gong H, Du J and Yu R: Ubiquitin ligase Siah1 promotes the
migration and invasion of human glioma cells by regulating HIF-1α
signaling under hypoxia. Oncol Rep. 33:1185–1190. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Krämer OH, Stauber RH, Bug G, Hartkamp J
and Knauer SK: SIAH proteins: Critical roles in leukemogenesis.
Leukemia. 27:792–802. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sim HW and Knox J: Hepatocellular
carcinoma in the era of immunotherapy. Curr Probl Cancer. 42:40–48.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Matsuo K, Satoh S, Okabe H, Nomura A,
Maeda T, Yamaoka Y and Ikai I: SIAH1 inactivation correlates with
tumor progression in hepatocellular carcinomas. Genes Chromosomes
Cancer. 36:283–291. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yoshibayashi H, Okabe H, Satoh S, Hida K,
Kawashima K, Hamasu S, Nomura A, Hasegawa S, Ikai I and Sakai Y:
SIAH1 causes growth arrest and apoptosis in hepatoma cells through
beta-catenin degradation-dependent and -independent mechanisms.
Oncol Rep. 17:549–556. 2007.PubMed/NCBI
|
|
31
|
Yao H, Ashihara E and Maekawa T: Targeting
the Wnt/β-catenin signaling pathway in human cancers. Expert Opin
Ther Targets. 15:873–887. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Clevers H and Nusse R: Wnt/β-catenin
signaling and disease. Cell. 149:1192–1205. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Arend RC, Londoño-Joshi AI, Straughn JM Jr
and Buchsbaum DJ: The Wnt/β-catenin pathway in ovarian cancer: A
review. Gynecol Oncol. 131:772–779. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Taciak B, Pruszynska I, Kiraga L, Bialasek
M and Krol M: Wnt signaling pathway in development and cancer. J
Physiol Pharmacol. 69:doi: 10.26402. 2018.PubMed/NCBI
|
|
35
|
Steinhart Z and Angers S: Wnt signaling in
development and tissue homeostasis. Development. 145:dev1465892018.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Huang P, Yan R, Zhang X, Wang L, Ke X and
Qu Y: Activating Wnt/β-catenin signaling pathway for disease
therapy: Challenges and opportunities. Pharmacol Ther. 196:79–90.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kim E, Lisby A, Ma C, Lo N, Ehmer U, Hayer
KE, Furth EE and Viatour P: Promotion of growth factor signaling as
a critical function of β-catenin during HCC progression. Nat
Commun. 10:19092019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ji L, Jiang B, Jiang X, Charlat O, Chen A,
Mickanin C, Bauer A, Xu W, Yan X and Cong F: The SIAH E3 ubiquitin
ligases promote Wnt/β-catenin signaling through mediating
wnt-induced axin degradation. Genes Dev. 31:904–915. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yang Y, Zhang J, Wu T, Xu X, Cao G, Li H
and Chen X: Histone deacetylase 2 regulates the doxorubicin (Dox)
resistance of hepatocarcinoma cells and transcription of ABCB1.
Life Sci. 216:200–206. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Cheng C, Li C, Zhu X, Han W, Li J and Lv
Y: Doxorubicin-loaded Fe(3)O(4)-ZIF-8 nano-composites for
hepatocellular carcinoma therapy. J Biomater Appl. 33:1373–1381.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Long L, Xiang H, Liu J, Zhang Z and Sun L:
ZEB1 mediates doxorubicin (Dox) resistance and mesenchymal
characteristics of hepatocarcinoma cells. Exp Mol Pathol.
106:116–122. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Li LY, Yang CC, Yang JF, Li HD, Zhang BY,
Zhou H, Hu S, Wang K, Huang C, Meng XM, et al: ZEB1 regulates the
activation of hepatic stellate cells through Wnt/β-catenin
signaling pathway. Eur J Pharmacol. 865:1727872019. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Qin Y, Yu J, Zhang M, Qin F and Lan X:
ZEB1 promotes tumorigenesis and metastasis in hepatocellular
carcinoma by regulating the expression of vimentin. Mol Med Rep.
19:2297–2306. 2019.PubMed/NCBI
|
|
44
|
Han X, Liu F, Zhang C, Ren Z, Li L and
Wang G: SIAH1/ZEB1/IL-6 axis is involved in doxorubicin (Dox)
resistance of osteosarcoma cells. Biol Chem. 400:545–553. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Abshire CF, Carroll JL and Dragoi AM:
FLASH protects ZEB1 from degradation and supports cancer cells'
epithelial-to-mesenchymal transition. Oncogenesis. 5:e2542016.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wu Y, Yang X, Chen Z, Tian L, Jiang G,
Chen F, Li J, An P, Lu L, Luo N, et al: m6 A-induced
lncRNA RP11 triggers the dissemination of colorectal cancer cells
via upregulation of Zeb1. Mol Cancer. 18:872019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Brauckhoff A, Malz M, Tschaharganeh D,
Malek N, Weber A, Riener MO, Soll C, Samarin J, Bissinger M and
Schmidt J: Nuclear expression of the ubiquitin ligase seven in
absentia homolog (SIAH)-1 induces proliferation and migration of
liver cancer cells. J Hepatol. 55:1049–1057. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Veronesi U, Boyle P, Goldhirsch A,
Orecchia R and Viale G: Breast cancer. Lancet. 365:1727–1741. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Woolston C: Breast cancer. Nature.
527:S1012015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
DeSantis C, Siegel R, Bandi P and Jemal A:
Breast cancer statistics, 2011. CA Cancer J Clin. 61:409–418. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ullah MF: Breast cancer: Current
perspectives on the disease status. Adv Exp Med Biol. 1152:51–64.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Tsang JYS and Tse GM: Molecular
classification of breast cancer. Adv Anat Pathol. 27:27–35. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Barzaman K, Karami J, Zarei Z,
Hosseinzadeh A, Kazemi MH, Moradi-Kalbolandi S, Safari E and
Farahmand L: Breast cancer: Biology, biomarkers, and treatments.
Int Immunopharmacol. 84:1065352020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bruzzoni-Giovanelli H, Faille A,
Linares-Cruz G, Nemani M, Deist FL, Germani A, Chassoux D, Millot
G, Roperch JP, Amson R, et al: SIAH-1 inhibits cell growth by
altering the mitotic process. Oncogene. 18:7101–7109. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Medhioub M, Vaury C, Hamelin R and Thomas
G: Lack of somatic mutation in the coding sequence of SIAH1 in
tumors hemizygous for this candidate tumor suppressor gene. Int J
Cancer. 87:794–797. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Germani A, Bruzzoni-Giovanelli H, Fellous
A, Gisselbrecht S, Varin-Blank N and Calvo F: SIAH-1 interacts with
alpha-tubulin and degrades the kinesin Kid by the proteasome
pathway during mitosis. Oncogene. 19:5997–6006. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wen YY, Yang ZQ, Song M, Li BL, Zhu JJ and
Wang EH: SIAH1 induced apoptosis by activation of the JNK pathway
and inhibited invasion by inactivation of the ERK pathway in breast
cancer cells. Cancer Sci. 101:73–79. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Nedeljković M and Damjanović A: Mechanisms
of chemotherapy resistance in triple-negative breast cancer-how we
can rise to the challenge. Cells. 8:9572019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Zhang L, Ma P, Sun LM, Han YC, Li BL, Mi
XY, Wang EH and Song M: MiR-107 down-regulates SIAH1 expression in
human breast cancer cells and silencing of miR-107 inhibits tumor
growth in a nude mouse model of triple-negative breast cancer. Mol
Carcinog. 55:768–777. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Hong HC, Chuang CH, Huang WC, Weng SL,
Chen CH, Chang KH, Liao KW and Huang HD: A panel of eight microRNAs
is a good predictive parameter for triple-negative breast cancer
relapse. Theranostics. 10:8771–8789. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Sahlberg KK, Bottai G, Naume B, Burwinkel
B, Calin GA, Børresen-Dale AL and Santarpia L: A serum microRNA
signature predicts tumor relapse and survival in triple-negative
breast cancer patients. Clin Cancer Res. 21:1207–1214. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Flores-Pérez A, Marchat LA,
Rodríguez-Cuevas S, Bautista VP, Fuentes-Mera L, Romero-Zamora D,
Maciel-Dominguez A, de la Cruz OH, Fonseca-Sánchez M, Ruíz-García
E, et al: Suppression of cell migration is promoted by miR-944
through targeting of SIAH1 and PTP4A1 in breast cancer cells. BMC
Cancer. 16:3792016. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ren H, Mi X, Zhao P, Zhao X, Wei N, Huang
H, Meng Z, Kou J, Sun M, Liu Y, et al: TRAF4, a new substrate of
SIAH1, participates in chemotherapy resistance of breast cancer
cell by counteracting SIAH1-mediated downregulation of β-catenin.
Breast Cancer Res Treat. 183:275–289. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Butti R, Gunasekaran VP, Kumar TVS,
Banerjee P and Kundu GC: Breast cancer stem cells: Biology and
therapeutic implications. Int J Biochem Cell Biol. 107:38–52. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Fisusi FA and Akala EO: Drug combinations
in breast cancer therapy. Pharm Nanotechnol. 7:3–23. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Tang Y, Wang Y, Kiani MF and Wang B:
Classification, treatment strategy, and associated drug resistance
in breast cancer. Clin Breast Cancer. 16:335–343. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Zhou J, Li W, Ming J, Yang W, Lu L, Zhang
Q, Ruan S and Huang T: High expression of TRAF4 predicts poor
prognosis in tamoxifen-treated breast cancer and promotes tamoxifen
resistance. Anticancer Drugs. 31:558–566. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Zhang X, Wen Z and Mi X: Expression and
anti-apoptotic function of TRAF4 in human breast cancer MCF-7
cells. Oncol Lett. 7:411–414. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Wang A, Wang J, Ren H, Yang F, Sun L, Diao
K, Zhao Z, Song M, Cui Z, Wang E, et al: TRAF4 participates in
wnt/β-catenin signaling in breast cancer by upregulating β-catenin
and mediating its translocation to the nucleus. Mol Cell Biochem.
395:211–219. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Liu M, Hsu J, Chan C, Li Z and Zhou Q: The
ubiquitin ligase Siah1 controls ELL2 stability and formation of
super elongation complexes to modulate gene transcription. Mol
Cell. 46:325–334. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
de Thé H, Pandolfi PP and Chen Z: Acute
promyelocytic leukemia: A paradigm for oncoprotein-targeted cure.
Cancer Cell. 32:552–560. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
De Braekeleer E, Douet-Guilbert N and De
Braekeleer M: RARA fusion genes in acute promyelocytic leukemia: A
review. Expert Rev Hematol. 7:347–357. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Liquori A, Ibañez M, Sargas C, Sanz MA,
Barragán E and Cervera J: Acute promyelocytic leukemia: A
constellation of molecular events around a single PML-RARA Fusion
Gene. Cancers (Basel). 12:6242020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Pietschmann K, Buchwald M, Müller S,
Knauer SK, Kögl M, Heinzel T and Krämer OH: Differential regulation
of PML-RARα stability by the ubiquitin ligases SIAH1/SIAH2 and
TRIAD1. Int J Biochem Cell Biol. 44:132–138. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Bug G, Ritter M, Wassmann B, Schoch C,
Heinzel T, Schwarz K, Romanski A, Kramer OH, Kampfmann M, Hoelzer
D, et al: Clinical trial of valproic acid and all-trans retinoic
acid in patients with poor-risk acute myeloid leukemia. Cancer.
104:2717–2725. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Bursen A, Moritz S, Gaussmann A, Moritz S,
Dingermann T and Marschalek R: Interaction of AF4 wild-type and
AF4.MLL fusion protein with SIAH proteins: Indication for t(4;11)
pathobiology? Oncogene. 23:6237–6249. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Krämer OH, Müller S, Buchwald M, Reichardt
S and Heinzel T: Mechanism for ubiquitylation of the leukemia
fusion proteins AML1-ETO and PML-RARalpha. Faseb J. 22:1369–1379.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Luo Z, Lin C and Shilatifard A: The super
elongation complex (SEC) family in transcriptional control. Nat Rev
Mol Cell Biol. 13:543–547. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Dahl NA, Danis E, Balakrishnan I, Wang D,
Pierce A, Walker FM, Gilani A, Serkova NJ, Madhavan K, Fosmire S,
et al: Super elongation complex as a targetable dependency in
diffuse midline glioma. Cell Rep. 31:1074852020. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Yu D, Liu R, Yang G and Zhou Q: The
PARP1-Siah1 axis controls HIV-1 transcription and expression of
Siah1 substrates. Cell Rep. 23:3741–3749. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wu J, Xue Y, Gao X and Zhou Q: Host cell
factors stimulate HIV-1 transcription by antagonizing
substrate-binding function of Siah1 ubiquitin ligase to stabilize
transcription elongation factor ELL2. Nucleic Acids Res.
48:7321–7332. 2020.PubMed/NCBI
|
|
82
|
Nagel CH, Albrecht N, Milovic-Holm K,
Mariyanna L, Keyser B, Abel B, Weseloh B, Hofmann TG, Eibl MM and
Hauber J: Herpes simplex virus immediate-early protein ICP0 is
targeted by SIAH-1 for proteasomal degradation. J Virol.
85:7644–7657. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Abe T, Umeki I, Kanno SI, Inoue SI,
Niihori T and Aoki Y: LZTR1 facilitates polyubiquitination and
degradation of RAS-GTPases. Cell Death Differ. 27:1023–1035. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Frattini V, Trifonov V, Chan JM, Castano
A, Lia M, Abate F, Keir ST, Ji AX, Zoppoli P, Niola F, et al: The
integrated landscape of driver genomic alterations in glioblastoma.
Nat Genet. 45:1141–1149. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Wirsching HG, Galanis E and Weller M:
Glioblastoma. Handb Clin Neurol. 134:381–397. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
He Y, Roos WP, Wu Q, Hofmann TG and Kaina
B: The SIAH1-HIPK2-p53ser46 damage response pathway is involved in
temozolomide-induced glioblastoma cell death. Mol Cancer Res.
17:1129–1141. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Fan Z, Li Z, Yang Y, Liu S, Guo J and Xu
Y: HIF-1α coordinates epigenetic activation of SIAH1 in hepatocytes
in response to nutritional stress. Biochim Biophys Acta Gene Regul
Mech. 1860:1037–1046. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Matsui-Hasumi A, Sato Y, Uto-Konomi A,
Yamashita S, Uehori J, Yoshimura A, Yamashita M, Asahara H, Suzuki
S and Kubo M: E3 ubiquitin ligases SIAH1/2 regulate
hypoxia-inducible factor-1 (HIF-1)-mediated Th17 cell
differentiation. Int Immunol. 29:133–143. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Ke Q and Costa M: Hypoxia-inducible
factor-1 (HIF-1). Mol Pharmacol. 70:1469–1480. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Semenza GL: Targeting HIF-1 for cancer
therapy. Nat Rev Cancer. 3:721–732. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
You L, Wu W, Wang X, Fang L, Adam V,
Nepovimova E, Wu Q and Kuca K: The role of hypoxia-inducible factor
1 in tumor immune evasion. Med Res Rev. 41:1622–1643. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Peng S, Zhang J, Tan X, Huang Y, Xu J,
Silk N, Zhang D, Liu Q and Jiang J: The VHL/HIF axis in the
development and treatment of pheochromocytoma/paraganglioma. Front
Endocrinol (Lausanne). 11:5868572020. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Gopalsamy A, Hagen T and Swaminathan K:
Investigating the molecular basis of Siah1 and Siah2 E3 ubiquitin
ligase substrate specificity. PLoS One. 9:e1065472014. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Kim SY, Choi DW, Kim EA and Choi CY:
Stabilization of HIPK2 by escape from proteasomal degradation
mediated by the E3 ubiquitin ligase Siah1. Cancer Lett.
279:177–184. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Choi DW and Choi CY: HIPK2 modification
code for cell death and survival. Mol Cell Oncol. 1:e9559992014.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Feng Y, Zhou L, Sun X and Li Q:
Homeodomain-interacting protein kinase 2 (HIPK2): A promising
target for anti-cancer therapies. Oncotarget. 8:20452–20461. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Moll UM and Petrenko O: The MDM2-p53
interaction. Mol Cancer Res. 1:1001–1008. 2003.PubMed/NCBI
|
|
98
|
Krastev DB and Buchholz F: Ribosome
biogenesis and p53: Who is regulating whom? Cell Cycle.
10:3417–3418. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Chao CC: Mechanisms of p53 degradation.
Clin Chim Acta. 438:139–147. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Kanapathipillai M: Treating p53 mutant
aggregation-associated cancer. Cancers (Basel). 10:1542018.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Feng L, Hollstein M and Xu Y: Ser46
phosphorylation regulates p53-dependent apoptosis and replicative
senescence. Cell Cycle. 5:2812–2819. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Liebl MC and Hofmann TG: Cell fate
regulation upon DNA damage: p53 serine 46 kinases pave the cell
death road. Bioessays. 41:e19001272019. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Winter M, Sombroek D, Dauth I,
Moehlenbrink J, Scheuermann K, Crone J and Hofmann TG: Control of
HIPK2 stability by ubiquitin ligase Siah-1 and checkpoint kinases
ATM and ATR. Nat Cell Biol. 10:812–824. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Moehlenbrink J, Bitomsky N and Hofmann TG:
Hypoxia suppresses chemotherapeutic drug-induced p53 Serine 46
phosphorylation by triggering HIPK2 degradation. Cancer Lett.
292:119–124. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Chen D, Li M, Luo J and Gu W: Direct
interactions between HIF-1 alpha and Mdm2 modulate p53 function. J
Biol Chem. 278:13595–13598. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Wang P, Guan D, Zhang XP, Liu F and Wang
W: Modeling the regulation of p53 activation by HIF-1 upon hypoxia.
FEBS Lett. 593:2596–2611. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Fukuba H, Yamashita H, Nagano Y, Jin HG,
Hiji M, Ohtsuki T, Takahashi T, Kohriyama T and Matsumoto M: Siah-1
facilitates ubiquitination and degradation of factor inhibiting
HIF-1alpha (FIH). Biochem Biophys Res Commun. 353:324–329. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Xiao Z, Wei Z, Deng D, Zheng Z, Zhao Y,
Jiang S, Zhang D, Zhang LJ, Fan M, Chen S, et al: Downregulation of
Siah1 promotes colorectal cancer cell proliferation and migration
by regulating AKT and YAP ubiquitylation and proteasome
degradation. Cancer Cell Int. 20:502020. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Wu W, Liu X, Wei L, Li T, Zang Y, Qian Y,
Bai T, Li J, Xie M, Zhu Y, et al: Tp53 mutation inhibits
ubiquitination and degradation of WISP1 via down-regulation of
siah1 in pancreatic carcinogenesis. Front Pharmacol. 9:8572018.
View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Ascherio A and Schwarzschild M: The
epidemiology of Parkinson's disease: Risk factors and prevention.
Lancet Neurol. 15:1257–1272. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Bloem BR, Okun MS and Klein C: Parkinson's
disease. Lancet. 397:2284–2303. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Lees A, Hardy J and Revesz T: Parkinson's
disease. Lancet. 373:2055–2066. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Franck T, Krueger R, Woitalla D, Müller T,
Engelender S and Riess O: Mutation analysis of the seven in
absentia homolog 1 (SIAH1) gene in Parkinson's disease. J Neural
Transm (Vienna). 113:1903–1908. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Nagano Y, Yamashita H, Takahashi T,
Kishida S, Nakamura T, Iseki E, Hattori N, Mizuno Y, Kikuchi A and
Matsumoto M: Siah-1 facilitates ubiquitination and degradation of
synphilin-1. J Biol Chem. 278:51504–51514. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Liani E, Eyal A, Avraham E, Shemer R,
Szargel R, Berg D, Bornemann A, Riess O, Ross CA, Rott R and
Engelender S: Ubiquitylation of synphilin-1 and alpha-synuclein by
SIAH and its presence in cellular inclusions and Lewy bodies imply
a role in Parkinson's disease. Proc Natl Acad Sci USA.
101:5500–5505. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Burchell VS, Nelson DE, Sanchez-Martinez
A, Delgado-Camprubi M, Ivatt RM, Pogson JH, Randle SJ, Wray S,
Lewis PA, Houlden H, et al: The Parkinson's disease-linked proteins
Fbxo7 and Parkin interact to mediate mitophagy. Nat Neurosci.
16:1257–1265. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Yamamura Y, Kuzuhara S, Kondo K, Yanagi T,
Uchida M, Matsumine H and Mizuno Y: Clinical, pathologic and
genetic studies on autosomal recessive early-onset parkinsonism
with diurnal fluctuation. Parkinsonism Relat Disord. 4:65–72. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Shahnawaz M, Mukherjee A, Pritzkow S,
Mendez N, Rabadia P, Liu X, Hu B, Schmeichel A, Singer W, Wu G, et
al: Discriminating α-synuclein strains in Parkinson's disease and
multiple system atrophy. Nature. 578:273–277. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Lövestam S, Schweighauser M, Matsubara T,
Murayama S, Tomita T, Ando T, Hasegawa K, Yoshida M, Tarutani A,
Hasegawa M, et al: Seeded assembly in vitro does not replicate the
structures of α-synuclein filaments from multiple system atrophy.
FEBS Open Bio. 11:999–1013. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Schweighauser M, Shi Y, Tarutani A,
Kametani F, Murzin AG, Ghetti B, Matsubara T, Tomita T, Ando T,
Hasegawa K, et al: Structures of α-synuclein filaments from
multiple system atrophy. Nature. 585:464–469. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Szargel R, Shani V, Elghani FA, Mekies LN,
Liani E, Rott R and Engelender S: The PINK1, synphilin-1 and SIAH-1
complex constitutes a novel mitophagy pathway. Hum Mol Genet.
25:3476–3490. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Subramaniam SR and Chesselet MF:
Mitochondrial dysfunction and oxidative stress in Parkinson's
disease. Prog Neurobiol. 106–107. 17–32. 2013.PubMed/NCBI
|
|
123
|
Pickrell AM and Youle RJ: The roles of
PINK1, parkin, and mitochondrial fidelity in Parkinson's disease.
Neuron. 85:257–273. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Ganguly G, Chakrabarti S, Chatterjee U and
Saso L: Proteinopathy, oxidative stress and mitochondrial
dysfunction: Cross talk in Alzheimer's disease and Parkinson's
disease. Drug Des Devel Ther. 11:797–810. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Rocha EM, De Miranda B and Sanders LH:
Alpha-synuclein: Pathology, mitochondrial dysfunction and
neuroinflammation in Parkinson's disease. Neurobiol Dis.
109:249–257. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Macdonald R, Barnes K, Hastings C and
Mortiboys H: Mitochondrial abnormalities in Parkinson's disease and
Alzheimer's disease: Can mitochondria be targeted therapeutically?
Biochem Soc Trans. 46:891–909. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Wang T, Zhang J, Hu M, Zhang Y, Cui P, Li
X, Li J, Vestin E, Brännström M, Shao LR and Billig H: Differential
expression patterns of glycolytic enzymes and
mitochondria-dependent apoptosis in PCOS patients with endometrial
hyperplasia, an early hallmark of endometrial cancer, in vivo and
the impact of metformin in vitro. Int J Biol Sci. 15:714–725. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Buratti J, Ji L, Keren B, Lee Y, Booke S,
Erdin S, Kim SY, Palculict TB, Meiner V, Chae JH, et al: De novo
variants in SIAH1, encoding an E3 ubiquitin ligase, are associated
with developmental delay, hypotonia and dysmorphic features. J Med
Genet. 58:205–212. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Khan I and Leventhal BL: Developmental
delay. StatPearls StatPearls Publishing. Copyright © 2021,
StatPearls Publishing LLC.; Treasure Island, FL: 2021
|
|
130
|
Oberklaid F and Efron D: Developmental
delay - identification and management. Aust Fam Physician.
34:739–742. 2005.PubMed/NCBI
|
|
131
|
Martiniuk AL, Vujovich-Dunn C, Park M, Yu
W and Lucas BR: Plagiocephaly and developmental delay: A systematic
review. J Dev Behav Pediatr. 38:67–78. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Zhou T, Chen Y, Zhao B, Hu S, Li J, Liu M,
Liang S, Bao Z and Wu X: Characterization and functional analysis
of SIAH1 during skin and hair follicle development in the angora
rabbit (Oryctolagus cuniculus). Hereditas. 157:102020.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Farmanullah Hosseini SM, Liang A, Hua G,
Rehman ZU, Talpur HS, Salim M, Ahmad S, Abulaiti A, Khan M, et al:
Adaptive molecular evolution of AKT3 gene for positive diversifying
selection in mammals. Biomed Res Int. 2020:25846272020. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Alcantara D, Timms AE, Gripp K, Baker L,
Park K, Collins S, Cheng C, Stewart F, Mehta SG, Saggar A, et al:
Mutations of AKT3 are associated with a wide spectrum of
developmental disorders including extreme megalencephaly. Brain.
140:2610–2622. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Ko HR, Jin E, Lee SB, Kim CK, Yun T, Cho
SW, Park KW and Ahn JY: SIAH1 ubiquitin ligase mediates
ubiquitination and degradation of Akt3 in neural development. J
Biol Chem. 294:15435–15445. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Taoufik E and Probert L: Ischemic neuronal
damage. Curr Pharm Des. 14:3565–3573. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Bruce-Keller AJ: Microglial-neuronal
interactions in synaptic damage and recovery. J Neurosci Res.
58:191–201. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Huo J, Zhu XL, Ma R, Dong HL and Su BX:
GAPDH/Siah1 cascade is involved in traumatic spinal cord injury and
could be attenuated by sivelestat sodium. Neuroscience.
330:171–180. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Huo J, Ma R, Chai X, Liang HJ, Jiang P,
Zhu XL, Chen X and Su BX: Inhibiting a spinal cord signaling
pathway protects against ischemia injury in rats. J Thoracic
Cardiovascular Surg. 157:494–503. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Hara MR, Thomas B, Cascio MB, Bae BI,
Hester LD, Dawson VL, Dawson TM, Sawa A and Snyder SH:
Neuroprotection by pharmacologic blockade of the GAPDH death
cascade. Proc Natl Acad Sci USA. 103:3887–3889. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Bangaru ML, Park F, Hudmon A, McCallum JB
and Hogan QH: Quantification of gene expression after painful nerve
injury: Validation of optimal reference genes. J Mol Neurosci.
46:497–504. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Chuang DM and Ishitani R: A role for GAPDH
in apoptosis and neurodegeneration. Nat Med. 2:609–610. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Su BX, Chen X, Huo J, Guo SY, Ma R and Liu
YW: The synthetic cannabinoid WIN55212-2 ameliorates traumatic
spinal cord injury via inhibition of GAPDH/Siah1 in a CB2-receptor
dependent manner. Brain Res. 1671:85–92. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Zhai D, Chin K, Wang M and Liu F:
Disruption of the nuclear p53-GAPDH complex protects against
ischemia-induced neuronal damage. Mol Brain. 7:202014. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Li C, Feng JJ, Wu YP and Zhang GY:
Cerebral ischemia-reperfusion induces GAPDH S-nitrosylation and
nuclear translocation. Biochemistry (Mosc). 77:671–678. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Jiang X and Shen X: Knockdown of
miR-299-5p inhibits the progression of hepatocellular carcinoma by
targeting SIAH1. Bull Cancer. 105:873–883. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Hui Z, Zhijun Y, Yushan Y, Liping C,
Yiying Z, Difan Z, Chunglit CT and Wei C: The combination of
acyclovir and dexamethasone protects against Alzheimer's
disease-related cognitive impairments in mice. Psychopharmacology
(Berl). 237:1851–1860. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Vogel J, Young A, Oxtoby N, Smith R,
Ossenkoppele R, Strandberg OT, Joie RL, Aksman LM, Grothe MJ,
Iturria-Medina Y, et al: Four distinct trajectories of tau
deposition identified in Alzheimer's disease. Nat Med. 27:871–881.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Knopman DS, Amieva H, Petersen RC,
Chételat G, Holtzman DM, Hyman BT, Nixon RA and Jones DT: Alzheimer
disease. Nat Rev Dis Primers. 7:332021. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Wasik U, Schneider G, Mietelska-Porowska
A, Mazurkiewicz M, Fabczak H, Weis S, Zabke C, Harrington CR,
Filipek A and Niewiadomska G: Calcyclin binding protein and Siah-1
interacting protein in Alzheimer's disease pathology: Neuronal
localization and possible function. Neurobiol Aging. 34:1380–1388.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Chen Y, Lian YJ, Ma YQ, Wu CJ, Zheng YK
and Xie NC: LncRNA SNHG1 promotes α-synuclein aggregation and
toxicity by targeting miR-15b-5p to activate SIAH1 in human
neuroblastoma SH-SY5Y cells. Neurotoxicology. 68:212–221. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Pang RT, Liu WM, Leung CO, Ye TM, Kwan PC,
Lee KF and Yeung WS: miR-135A regulates preimplantation embryo
development through down-regulation of E3 ubiquitin ligase seven in
absentia homolog 1A (SIAH1A) expression. PLoS One. 6:e278782011.
View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Schafer AR, Smith JL, Pryke KM, DeFilippis
VR and Hirsch AJ: The E3 ubiquitin ligase SIAH1 targets MyD88 for
proteasomal degradation during dengue virus infection. Front
Microbiol. 11:242020. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Zhou Y, Li L, Liu Q, Xing G, Kuai X, Sun
J, Yin X, Wang J, Zhang L and He F: E3 ubiquitin ligase SIAH1
mediates ubiquitination and degradation of TRB3. Cell Signal.
20:942–948. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Nagano Y, Fukushima T, Okemoto K, Tanaka
K, Bowtell DD, Ronai Z, Reed JC and Matsuzawa SI: Siah1/SIP
regulates p27(kip1) stability and cell migration under metabolic
stress. Cell Cycle. 10:2592–2602. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Hara MR and Snyder SH: Nitric
oxide-GAPDH-Siah: A novel cell death cascade. Cell Mol Neurobiol.
26:527–538. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Song Y, Xu Y, Pan C, Yan L, Wang ZW and
Zhu X: The emerging role of SPOP protein in tumorigenesis and
cancer therapy. Mol Cancer. 19:22020. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Shah M, Stebbins JL, Dewing A, Qi J,
Pellecchia M and Ronai ZA: Inhibition of Siah2 ubiquitin ligase by
vitamin K3 (menadione) attenuates hypoxia and MAPK signaling and
blocks melanoma tumorigenesis. Pigment Cell Melanoma Res.
22:799–808. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Zhang H, Cao X, Wang J, Li Q, Zhao Y and
Jin X: LZTR1: A promising adaptor of the CUL3 family. Oncol Lett.
22:5642021. View Article : Google Scholar : PubMed/NCBI
|