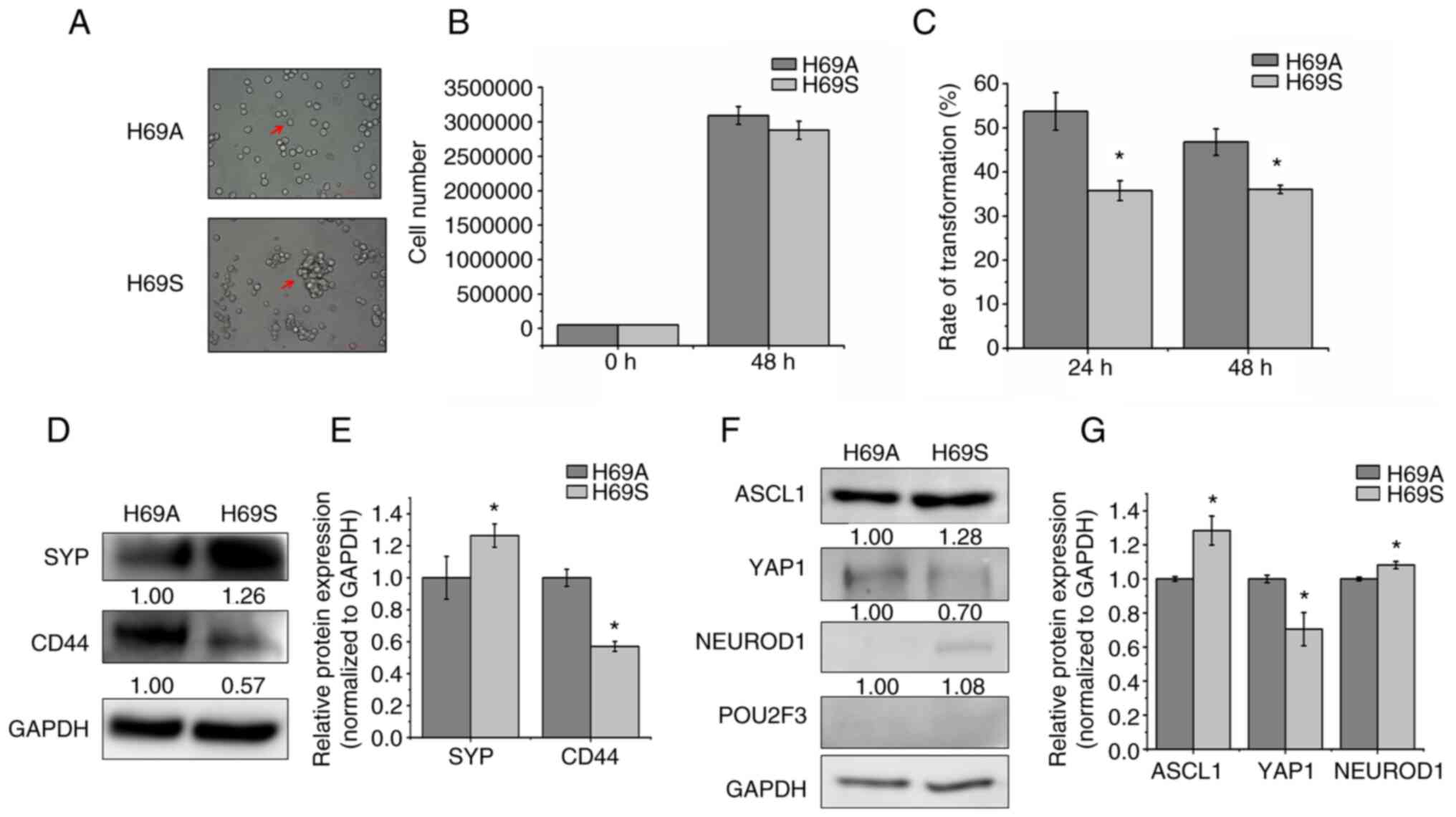
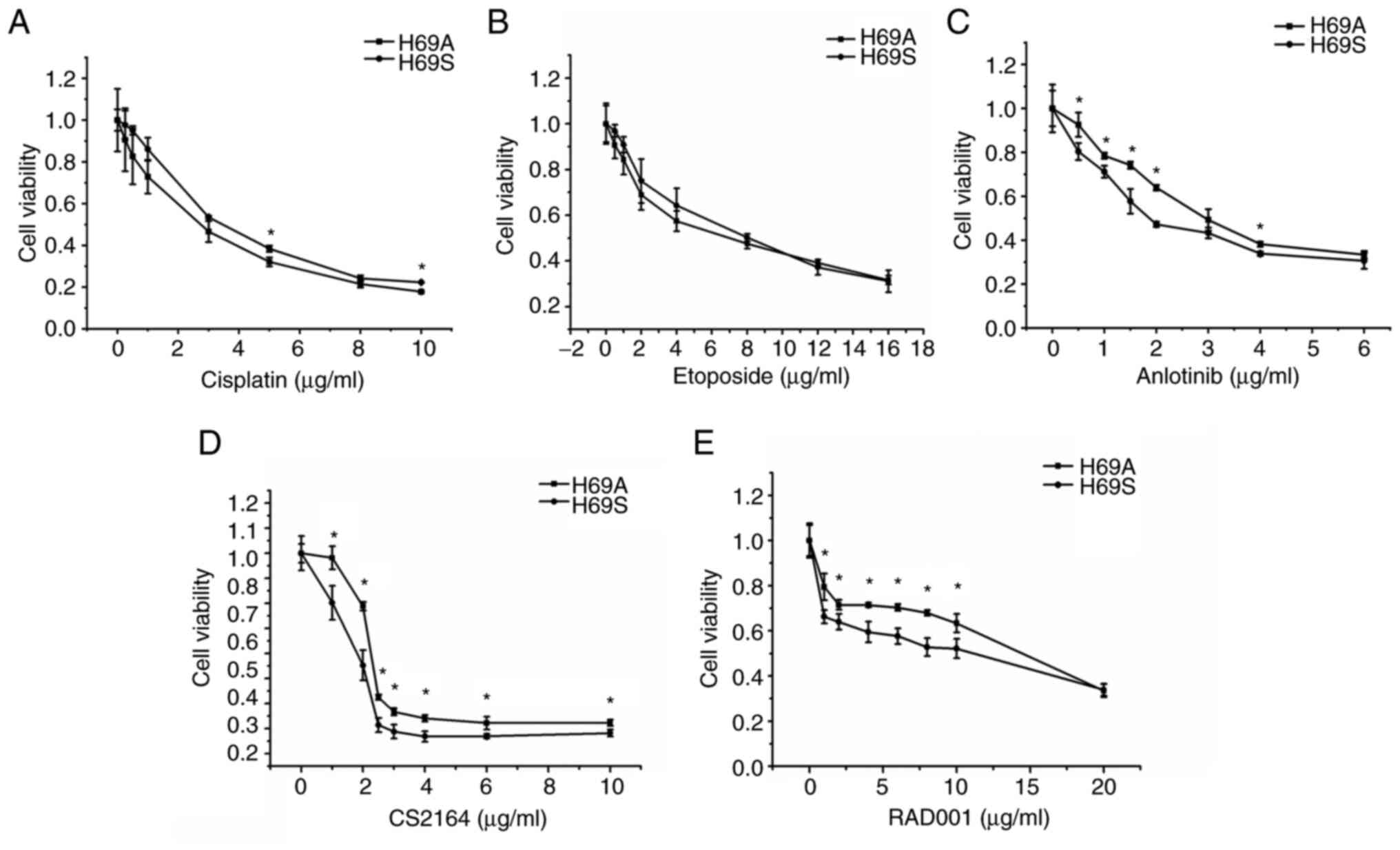
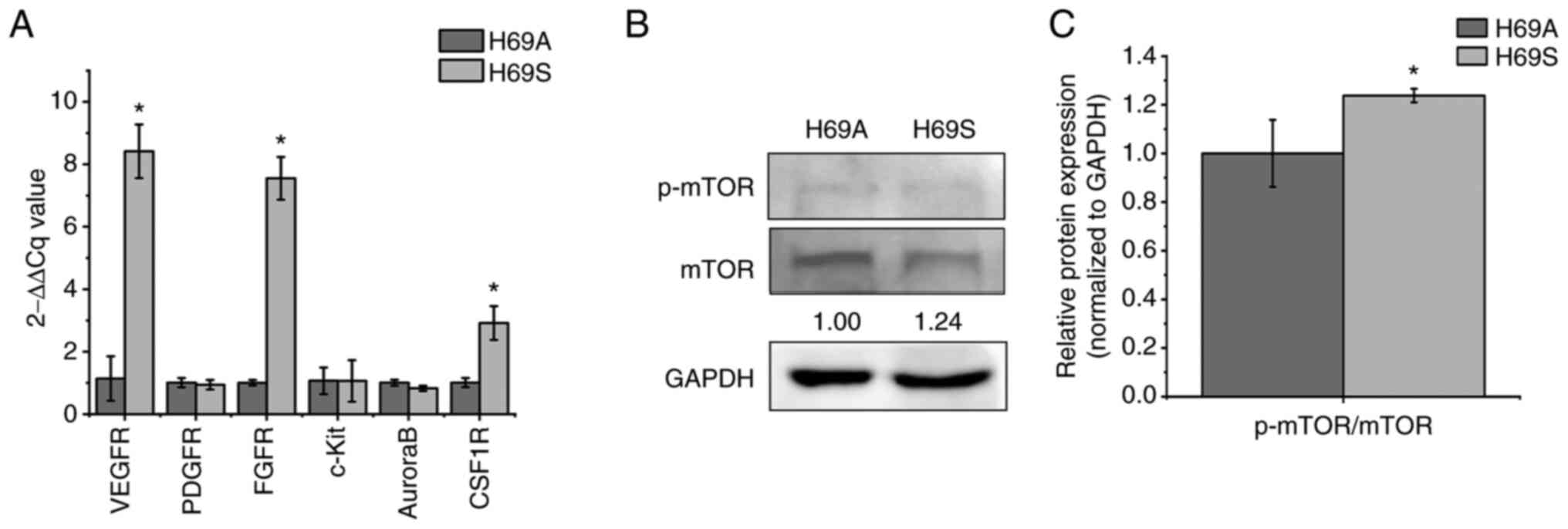
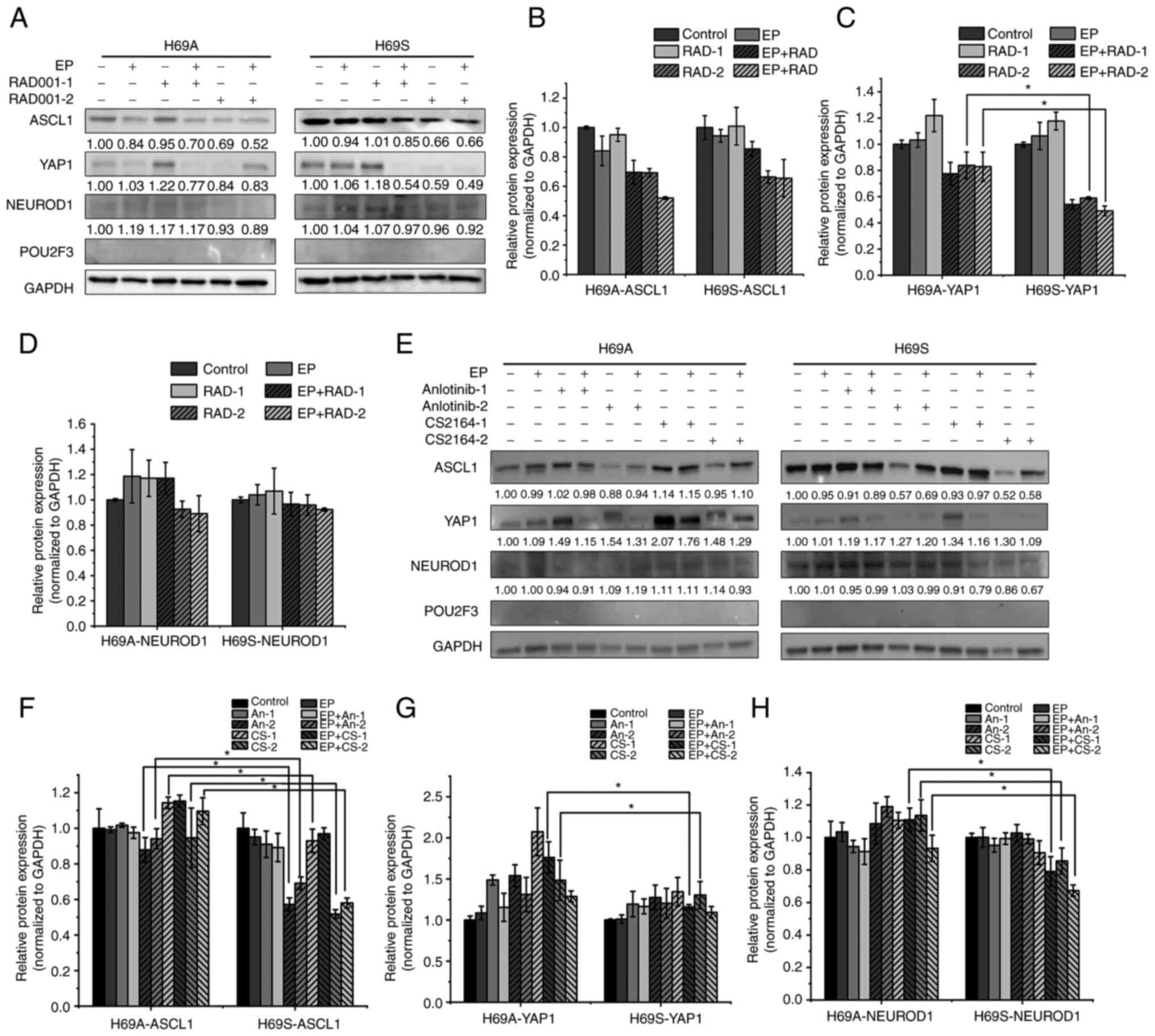
|
1
|
Oronsky B, Reid TR, Oronsky A and Carter
CA: What's new in sclc? A review. Neoplasia. 19:842–847. 2017.
View Article : Google Scholar
|
|
2
|
Jahchan NS, Lim JS, Bola B, Morris K,
Seitz G, Tran KQ, Xu L, Trapani F, Morrow CJ, Cristea S, et al:
Identification and targeting of long-term tumor-propagating cells
in small cell lung cancer. Cell Rep. 16:644–656. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nicholson AG, Chansky K, Crowley J,
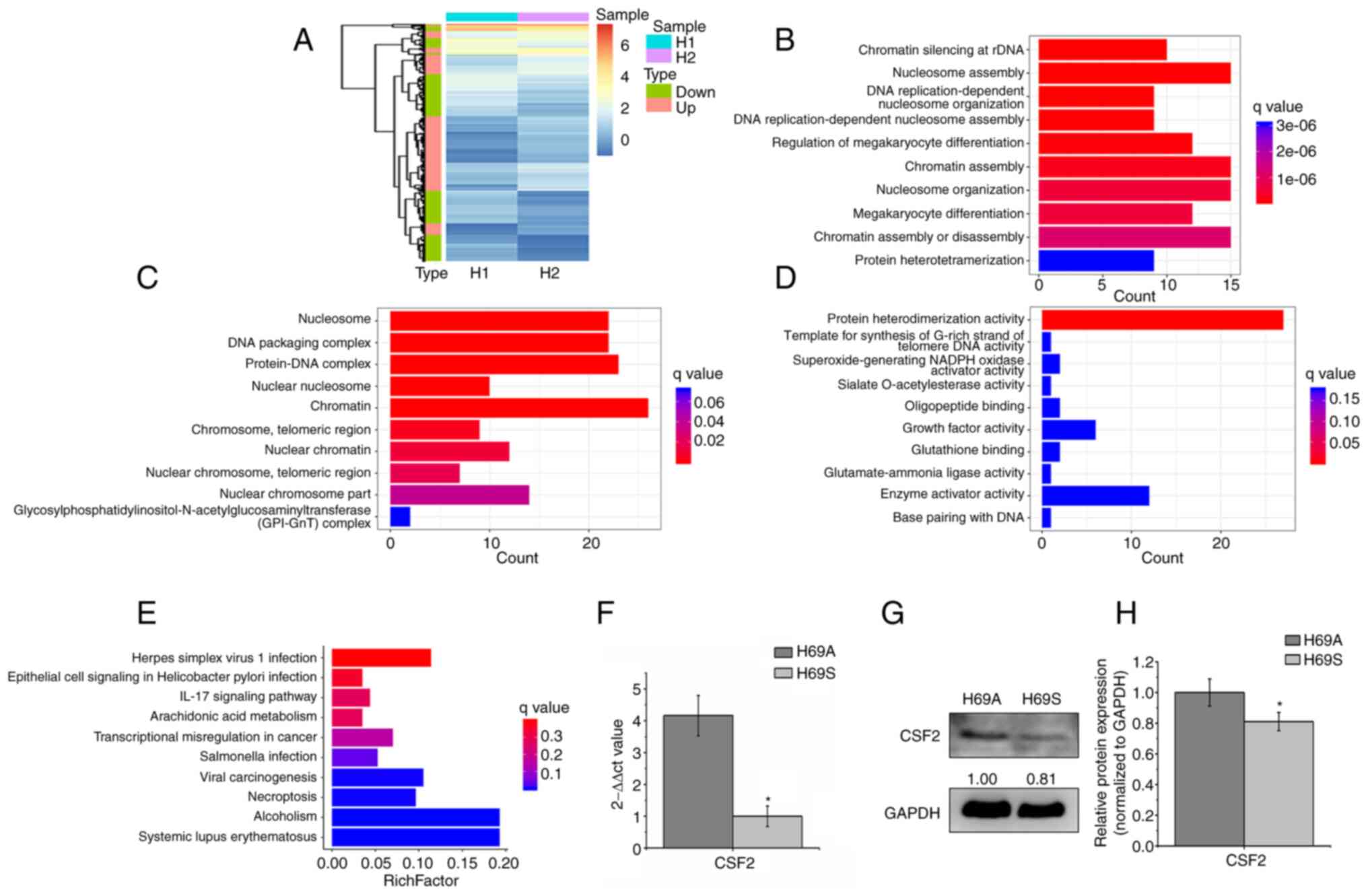
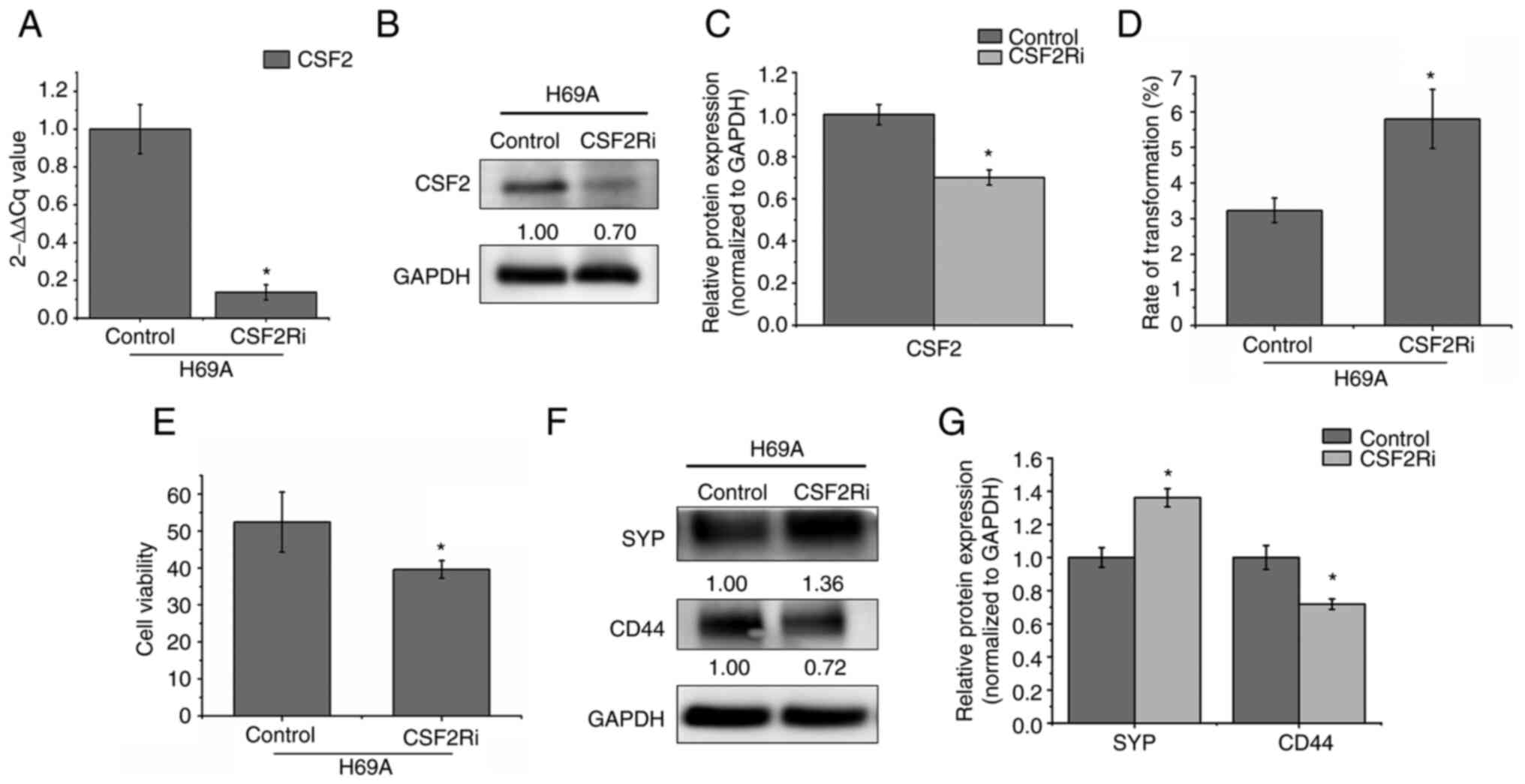
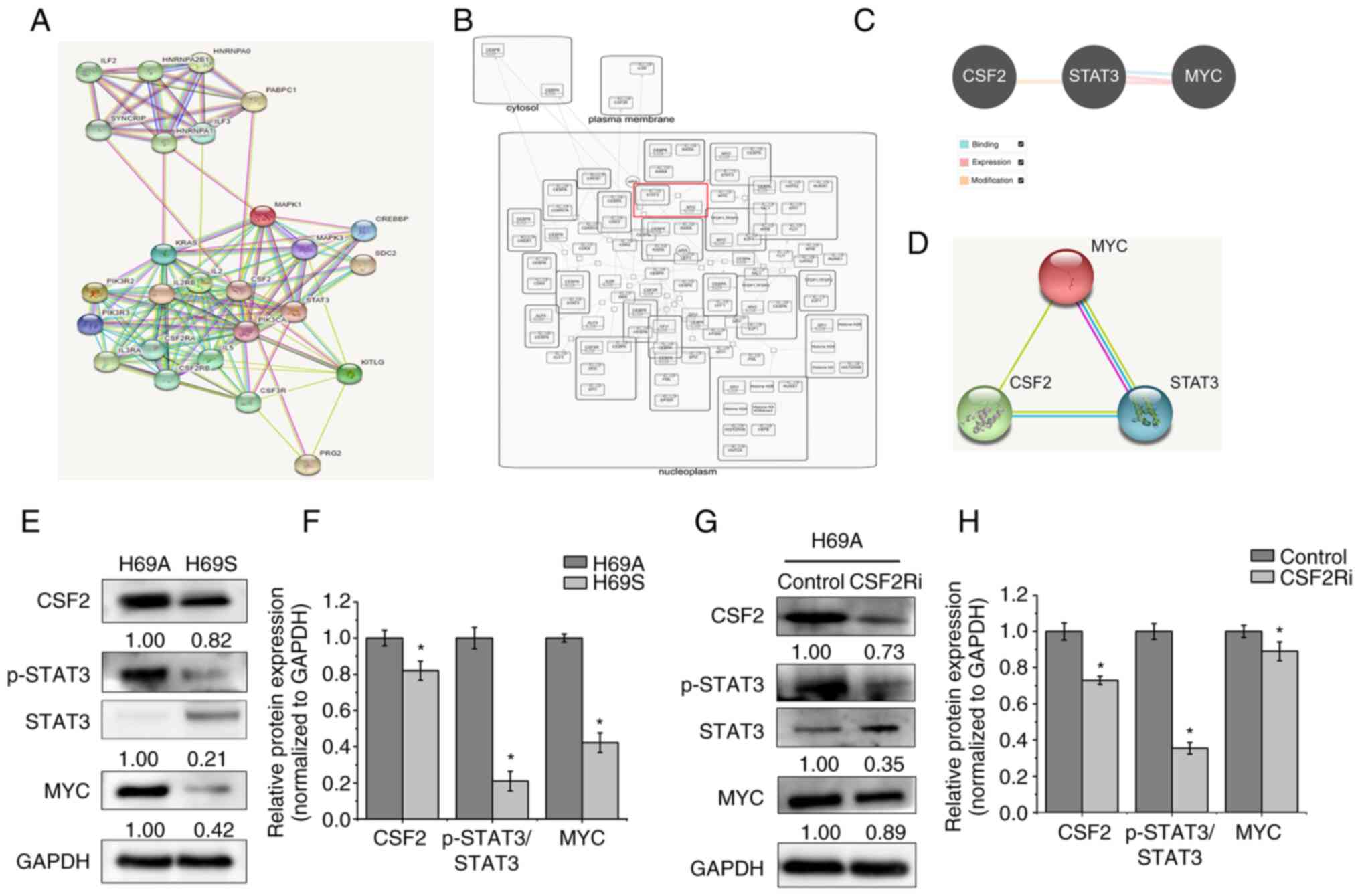
Beyruti R, Kubota K, Turrisi A, Eberhardt WE, van Meerbeeck J,
Rami-Porta R; Staging, Prognostic Factors Committee, et al: The
international association for the study of lung cancer lung cancer
staging project: Proposals for the revision of the clinical and
pathologic staging of small cell lung cancer in the forthcoming
eighth edition of the tnm classification for lung cancer. J Thorac
Oncol. 11:300–311. 2016. View Article : Google Scholar
|
|
4
|
Song Y, Sun Y, Lei Y, Yang K and Tang R:
YAP1 promotes multidrug resistance of small cell lung cancer by
CD74-related signaling pathways. Cancer Med. 9:259–268. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Prabavathy D and Ramadoss N: Heterogeneity
of small cell lung cancer stem cells. Adv Exp Med Biol. 1139:41–57.
2019. View Article : Google Scholar
|
|
6
|
Gazdar AF, Carney DN, Nau MM and Minna JD:
Characterization of variant subclasses of cell lines derived from
small cell lung cancer having distinctive biochemical,
morphological, and growth properties. Cancer Res. 45:2924–2930.
1985.PubMed/NCBI
|
|
7
|
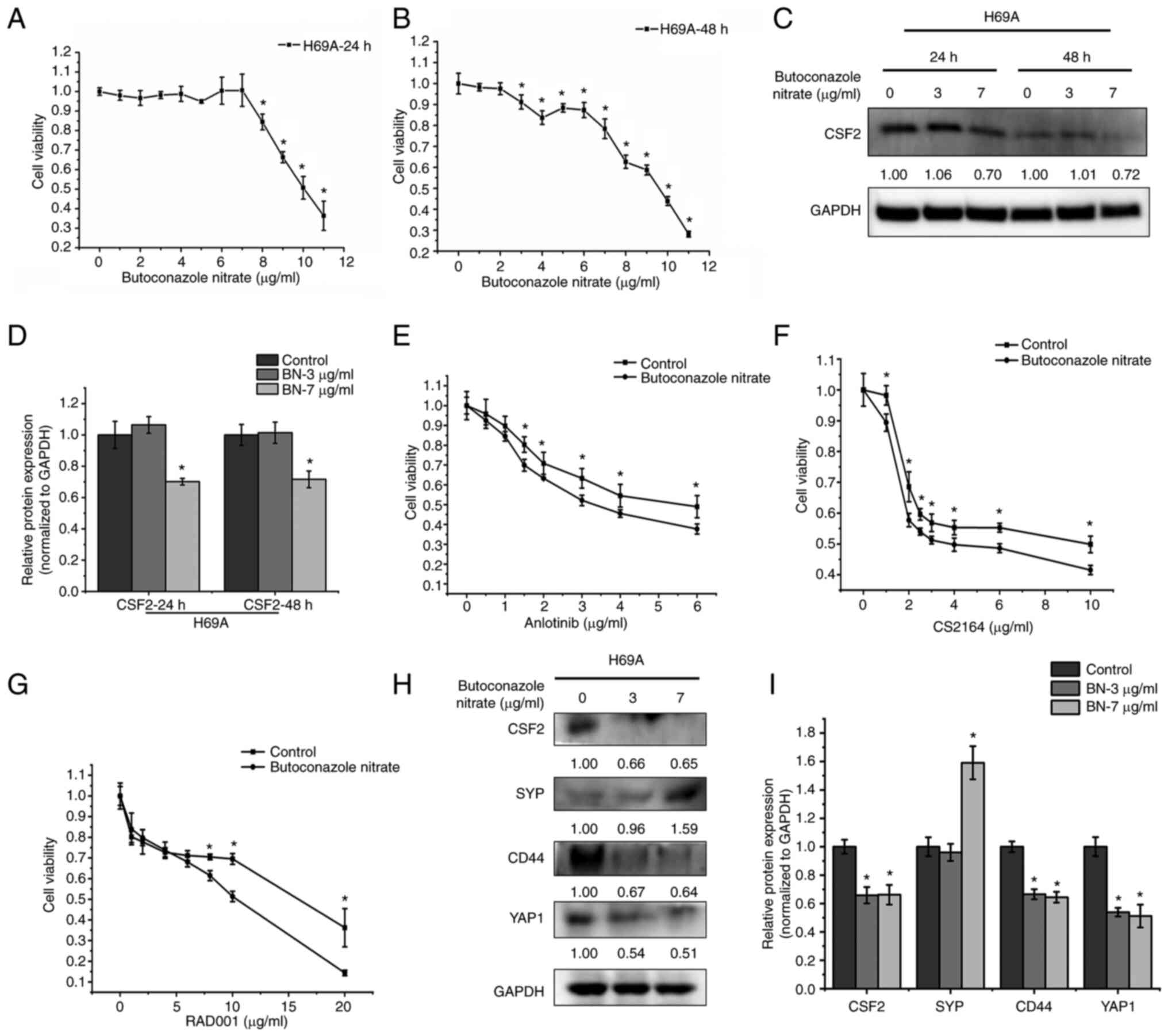
Zhang W, Girard L, Zhang YA, Haruki T,
Papari-Zareei M, Stastny V, Ghayee HK, Pacak K, Oliver TG, Minna JD
and Gazdar AF: Small cell lung cancer tumors and preclinical models
display heterogeneity of neuroendocrine phenotypes. Transl Lung
Cancer Res. 7:32–49. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Calbo J, van Montfort E, Proost N, van
Drunen E, Beverloo HB, Meuwissen R and Berns A: A functional role
for tumor cell heterogeneity in a mouse model of small cell lung
cancer. Cancer Cell. 19:244–256. 2011. View Article : Google Scholar
|
|
9
|
Sutherland KD, Proost N, Brouns I,
Adriaensen D, Song JY and Berns A: Cell of origin of small cell
lung cancer: Inactivation of Trp53 and Rb1 in distinct cell types
of adult mouse lung. Cancer Cell. 19:754–764. 2011. View Article : Google Scholar
|
|
10
|
Quintanal-Villalonga Á, Chan JM, Yu HA,
Pe'er D, Sawyers CL, Sen T and Rudin CM: Lineage plasticity in
cancer: A shared pathway of therapeutic resistance. Nat Rev Clin
Oncol. 17:360–371. 2020. View Article : Google Scholar
|
|
11
|
Baldwin GC, Golde DW, Widhopf GF, Economou
J and Gasson JC: Identification and characterization of a
low-affinity granulocyte-macrophage colony-stimulating factor
receptor on primary and cultured human melanoma cells. Blood.
78:609–615. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar
|
|
13
|
Chen S, Chen X, Li W, Shan T, Lin WR, Ma
J, Cui X, Yang W, Cao G, Li Y, et al: Conversion of
epithelial-to-mesenchymal transition to mesenchymal-to-epithelial
transition is mediated by oxygen concentration in pancreatic cancer
cells. Oncol Lett. 15:7144–7152. 2018.
|
|
14
|
Chaffer CL, Marjanovic ND, Lee T, Bell G,
Kleer CG, Reinhardt F, D'Alessio AC, Young RA and Weinberg RA:
Poised chromatin at the ZEB1 promoter enables breast cancer cell
plasticity and enhances tumorigenicity. Cell. 154:61–74. 2013.
View Article : Google Scholar
|
|
15
|
Ireland AS, Micinski AM, Kastner DW, Guo
B, Wait SJ, Spainhower KB, Conley CC, Chen OS, Guthrie MR, Soltero
D, et al: MYC drives temporal evolution of small cell lung cancer
subtypes by reprogramming neuroendocrine fate. Cancer Cell.
38:60–78.e12. 2020. View Article : Google Scholar
|
|
16
|
Hong IS: Stimulatory versus suppressive
effects of GM-CSF on tumor progression in multiple cancer types.
Exp Mol Med. 48:e2422016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mueller MM and Fusenig NE: Constitutive
expression of G-CSF and GM-CSF in human skin carcinoma cells with
functional consequence for tumor progression. Int J Cancer.
83:780–789. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Trutmann M, Terracciano L, Noppen C, Kloth
J, Kaspar M, Peterli R, Tondelli P, Schaeffer C, Zajac P, Heberer M
and Spagnoli GC: GM-CSF gene expression and protein production in
human colorectal cancer cell lines and clinical tumor specimens.
Int J Cancer. 77:378–385. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li X, Wang J, Wu W, Gao H, Liu N, Zhan G,
Li L, Han L and Guo X: Myeloid-derived suppressor cells promote
epithelial ovarian cancer cell stemness by inducing the
CSF2/p-STAT3 signalling pathway. FEBS J. 287:5218–5235. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Qin W, Tian Y, Zhang J, Liu W, Zhou Q, Hu
S, Yang F, Lu L, Lu H, Cui S, et al: The double inhibition of PDK1
and STAT3-Y705 prevents liver metastasis in colorectal cancer. Sci
Rep. 9:129732019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lin CH, Chiang MC and Chen YJ: STAT3
mediates resistance to anoikis and promotes invasiveness of
nasopharyngeal cancer cells. Int J Mol Med. 40:1549–1556. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li X, Brock GN, Rouchka EC, Cooper NGF, Wu
D, O'Toole TE, Gill RS, Eteleeb AM, O'Brien L and Rai SN: A
comparison of per sample global scaling and per gene normalization
methods for differential expression analysis of RNA-seq data. PLoS
One. 12:e01761852017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sahraeian SME, Mohiyuddin M, Sebra R,
Tilgner H, Afshar PT, Au KF, Bani Asadi N, Gerstein MB, Wong WH,
Snyder MP, et al: Gaining comprehensive biological insight into the
transcriptome by performing a broad-spectrum RNA-seq analysis. Nat
Commun. 8:592017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Fukutomi T, Kohno M, Izumi Y, Watanabe M,
Hayashi Y and Nomori H: Pulmonary pleomorphic carcinoma producing
granulocyte-macrophage colony-stimulating factor: Report of a case.
Surg Today. 42:288–291. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rudin CM, Ismaila N, Hann CL, Malhotra N,
Movsas B, Norris K, Pietanza MC, Ramalingam SS, Turrisi AT III and
Giaccone G: Treatment of small-cell lung cancer: American society
of clinical oncology endorsement of the american college of chest
physicians guideline. J Clin Oncol. 33:4106–4111. 2015. View Article : Google Scholar
|
|
27
|
Giercksky HE, Thorstensen L, Qvist H,
Nesland JM and Lothe RA: Comparison of genetic changes in frozen
biopsies and microdissected archival material from the same
colorectal liver metastases. Diagn Mol Pathol. 6:318–325. 1997.
View Article : Google Scholar
|
|
28
|
Friedmann-Morvinski D: Glioblastoma
heterogeneity and cancer cell plasticity. Crit Rev Oncog.
19:327–336. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Maynard A, McCoach CE, Rotow JK, Harris L,
Haderk F, Kerr DL, Yu EA, Schenk EL, Tan W, Zee A, et al:
Therapy-induced evolution of human lung cancer revealed by
single-cell RNA sequencing. Cell. 182:1232–1251.e22. 2020.
View Article : Google Scholar
|
|
30
|
Lin CA, Yu SL, Chen HY, Chen HW, Lin SU,
Chang CC, Yu CJ, Yang PC and Ho CC: EGFR-mutant SCLC exhibits
heterogeneous phenotypes and resistance to common antineoplastic
drugs. J Thorac Oncol. 14:513–526. 2019. View Article : Google Scholar
|
|
31
|
Herbst RS, Morgensztern D and Boshoff C:
The biology and management of non-small cell lung cancer. Nature.
553:446–454. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Raso MG, Bota-Rabassedas N and Wistuba II:
Pathology and classification of SCLC. Cancers (Basel). 13:8202021.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rudin CM, Poirier JT, Byers LA, Dive C,
Dowlati A, George J, Heymach JV, Johnson JE, Lehman JM, MacPherson
D, et al: Molecular subtypes of small cell lung cancer: A synthesis
of human and mouse model data. Nat Rev Cancer. 19:289–297. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
George J, Lim JS, Jang SJ, Cun Y, Ozretić
L, Kong G, Leenders F, Lu X, Fernández-Cuesta L, Bosco G, et al:
Comprehensive genomic profiles of small cell lung cancer. Nature.
524:47–53. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Rudin CM, Brambilla E, Faivre-Finn C and
Sage J: Small-cell lung cancer. Nat Rev Dis Primers. 7:32021.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gay CM, Stewart CA, Park EM, Diao L,
Groves SM, Heeke S, Nabet BY, Fujimoto J, Solis LM, Lu W, et al:
Patterns of transcription factor programs and immune pathway
activation define four major subtypes of SCLC with distinct
therapeutic vulnerabilities. Cancer Cell. 39:346–360.e7. 2021.
View Article : Google Scholar
|
|
37
|
Huang YH, Klingbeil O, He XY, Wu XS, Arun
G, Lu B, Somerville TDD, Milazzo JP, Wilkinson JE, Demerdash OE, et
al: POU2F3 is a master regulator of a tuft cell-like variant of
small cell lung cancer. Genes Dev. 32:915–928. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chalishazar MD, Wait SJ, Huang F, Ireland
AS, Mukhopadhyay A, Lee Y, Schuman SS, Guthrie MR, Berrett KC,
Vahrenkamp JM, et al: MYC-driven small-cell lung cancer is
metabolically distinct and vulnerable to arginine depletion. Clin
Cancer Res. 25:5107–5121. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Huang F, Ni M, Chalishazar MD, Huffman KE,
Kim J, Cai L, Shi X, Cai F, Zacharias LG, Ireland AS, et al:
Inosine monophosphate dehydrogenase dependence in a subset of small
cell lung cancers. Cell Metab. 28:369–382.e5. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Mollaoglu G, Guthrie MR, Böhm S,
Brägelmann J, Can I, Ballieu PM, Marx A, George J, Heinen C,
Chalishazar MD, et al: MYC drives progression of small cell lung
cancer to a variant neuroendocrine subtype with vulnerability to
aurora kinase inhibition. Cancer Cell. 31:270–285. 2017. View Article : Google Scholar
|
|
41
|
Chen C, Duckworth CA, Zhao Q, Pritchard
DM, Rhodes JM and Yu LG: Increased circulation of galectin-3 in
cancer induces secretion of metastasis-promoting cytokines from
blood vascular endothelium. Clin Cancer Res. 19:1693–1704. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Valdembri D, Serini G, Vacca A, Ribatti D
and Bussolino F: In vivo activation of JAK2/STAT-3 pathway during
angiogenesis induced by GM-CSF. FASEB J. 16:225–227. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zgheib A, Lamy S and Annabi B:
Epigallocatechin gallate targeting of membrane type 1 matrix
metalloproteinase-mediated Src and Janus kinase/signal transducers
and activators of transcription 3 signaling inhibits transcription
of colony-stimulating factors 2 and 3 in mesenchymal stromal cells.
J Biol Chem. 288:13378–13386. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Brantley EC, Nabors LB, Gillespie GY, Choi
YH, Palmer CA, Harrison K, Roarty K and Benveniste EN: Loss of
protein inhibitors of activated STAT-3 expression in glioblastoma
multiforme tumors: Implications for STAT-3 activation and gene
expression. Clin Cancer Res. 14:4694–4704. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Weissenberger J, Loeffler S, Kappeler A,
Kopf M, Lukes A, Afanasieva TA, Aguzzi A and Weis J: Il-6 is
required for glioma development in a mouse model. Oncogene.
23:3308–3316. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Niola F, Evangelisti C, Campagnolo L,
Massalini S, Buè MC, Mangiola A, Masotti A, Maira G, Farace MG and
Ciafrè SA: A plasmid-encoded VEGF siRNA reduces glioblastoma
angiogenesis and its combination with interleukin-4 blocks tumor
growth in a xenograft mouse model. Cancer Biol Ther. 5:174–179.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Jung KH, Chu K, Lee ST, Kim SJ, Sinn DI,
Kim SU, Kim M and Roh JK: Granulocyte colony-stimulating factor
stimulates neurogenesis via vascular endothelial growth factor with
STAT activation. Brain Res. 1073–1074. 190–201. 2006.
|
|
48
|
Ohki Y, Heissig B, Sato Y, Akiyama H, Zhu
Z, Hicklin DJ, Shimada K, Ogawa H, Daida H, Hattori K and Ohsaka A:
Granulocyte colony-stimulating factor promotes neovascularization
by releasing vascular endothelial growth factor from neutrophils.
FASEB J. 19:2005–2007. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lammel V, Stoeckle C, Padberg B, Zweifel
R, Kienle DL, Reinhart WH and Simon HU: Hypereosinophilia driven by
GM-CSF in large-cell carcinoma of the lung. Lung Cancer.
76:493–495. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Bahar B, Acedil Ayc Iota B, Çoşkun U,
Büyükberber S, Benekli M and Yildiz R: Granulocyte colony
stimulating factor (G-CSF) and macrophage colony stimulating factor
(M-CSF) as potential tumor markers in non small cell lung cancer
diagnosis. Asian Pac J Cancer Prev. 11:709–712. 2010.PubMed/NCBI
|
|
51
|
Shalom G, Sion-Vardy N, Dudnik J and Ariad
S: Leukemoid reaction in lung cancer patients. Isr Med Assoc J.
12:255–256. 2010.PubMed/NCBI
|
|
52
|
Orr-Asman MA, Chu Z, Jiang M, Worley M,
LaSance K, Koch SE, Carreira VS, Dahche HM, Plas DR, Komurov K, et
al: mTOR kinase inhibition effectively decreases progression of a
subset of neuroendocrine tumors that progress on rapalog therapy
and delays cardiac impairment. Mol Cancer Ther. 16:2432–2441. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Besse B, Heist RS, Papadmitrakopoulou VA,
Camidge DR, Beck JT, Schmid P, Mulatero C, Miller N, Dimitrijevic
S, Urva S, et al: A phase Ib dose-escalation study of everolimus
combined with cisplatin and etoposide as first-line therapy in
patients with extensive-stage small-cell lung cancer. Ann Oncol.
25:505–511. 2014. View Article : Google Scholar
|
|
54
|
Tarhini A, Kotsakis A, Gooding W, Shuai Y,
Petro D, Friedland D, Belani CP, Dacic S and Argiris A: Phase II
study of everolimus (RAD001) in previously treated small cell lung
cancer. Clin Cancer Res. 16:5900–5907. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Lin B, Song X, Yang D, Bai D, Yao Y and Lu
N: Anlotinib inhibits angiogenesis via suppressing the activation
of VEGFR2, PDGFRβ and FGFR1. Gene. 654:77–86. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Xie C, Wan X, Quan H, Zheng M, Fu L, Li Y
and Lou L: Preclinical characterization of anlotinib, a highly
potent and selective vascular endothelial growth factor receptor-2
inhibitor. Cancer Sci. 109:1207–1219. 2018. View Article : Google Scholar
|
|
57
|
Cheng Y, Wang Q, Li K, Shi J, Liu Y, Wu L,
Han B, Chen G, He J, Wang J, et al: Anlotinib vs placebo as third-
or further-line treatment for patients with small cell lung cancer:
A randomised, double-blind, placebo-controlled phase 2 study. Br J
Cancer. 125:366–371. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zhou Y, Shan S, Li ZB, Xin LJ, Pan DS,
Yang QJ, Liu YP, Yue XP, Liu XR, Gao JZ, et al: CS2164, a novel
multi-target inhibitor against tumor angiogenesis, mitosis and
chronic inflammation with anti-tumor potency. Cancer Sci.
108:469–477. 2017. View Article : Google Scholar
|
|
59
|
Sun Y, Yang L, Hao X, Liu Y, Zhang J, Ning
Z and Shi Y: Phase I dose-escalation study of chiauranib, a novel
angiogenic, mitotic, and chronic inflammation inhibitor, in
patients with advanced solid tumors. J Hematol Oncol. 12:92019.
View Article : Google Scholar
|
|
60
|
Pearsall SM, Humphrey S, Revill M, Morgan
D, Frese KK, Galvin M, Kerr A, Carter M, Priest L, Blackhall F, et
al: The rare YAP1 subtype of SCLC revisited in a biobank of 39
circulating tumor cell patient derived explant models: A brief
report. J Thorac Oncol. 15:1836–1843. 2020. View Article : Google Scholar
|
|
61
|
Owonikoko TK, Dwivedi B, Chen Z, Zhang C,
Barwick B, Ernani V, Zhang G, Gilbert-Ross M, Carlisle J, Khuri FR,
et al: YAP1 expression in SCLC defines a distinct subtype with
T-cell-inflamed phenotype. J Thorac Oncol. 16:464–476. 2021.
View Article : Google Scholar
|
|
62
|
Hamilton JA: GM-CSF in inflammation. J Exp
Med. 217:e201909452020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Mehta HM, Malandra M and Corey SJ: G-CSF
and GM-CSF in neutropenia. J Immunol. 195:1341–1349. 2015.
View Article : Google Scholar
|