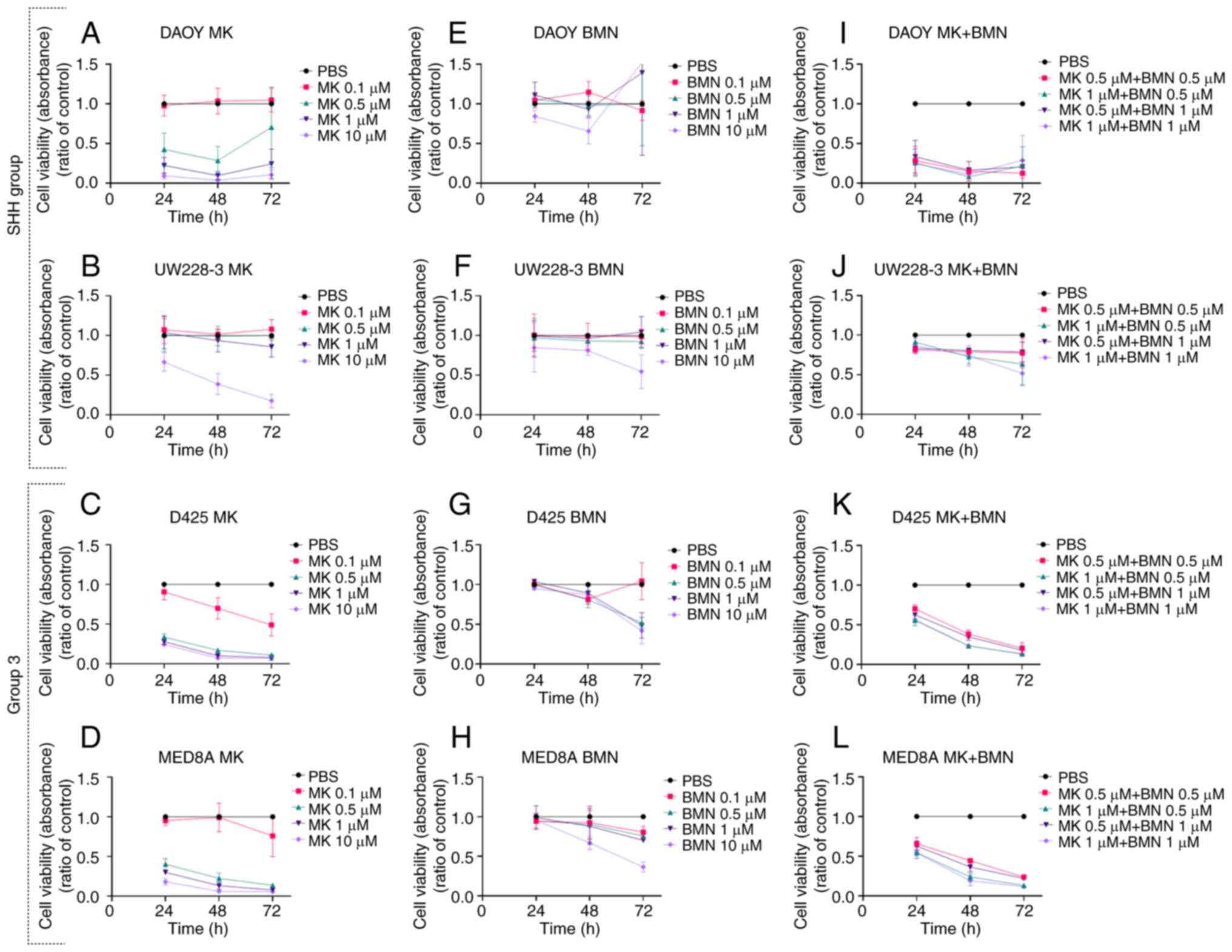
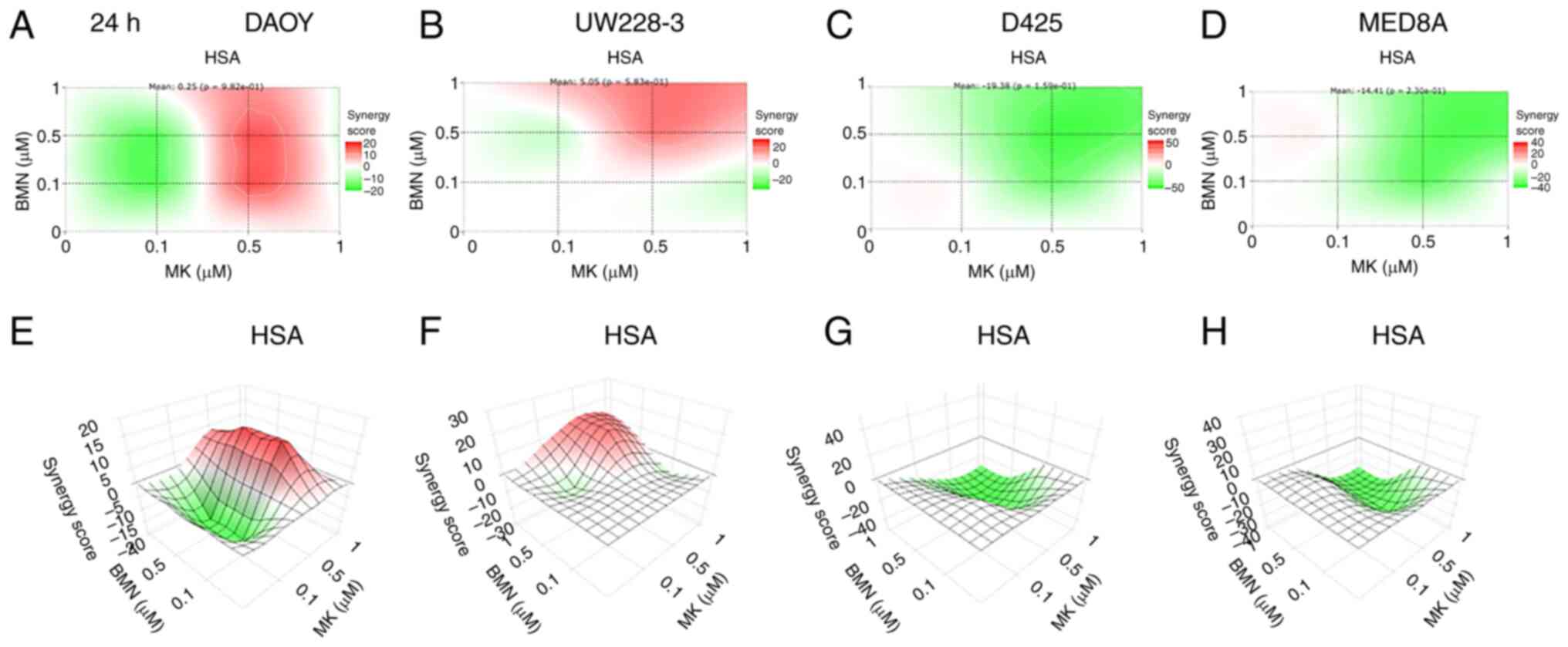
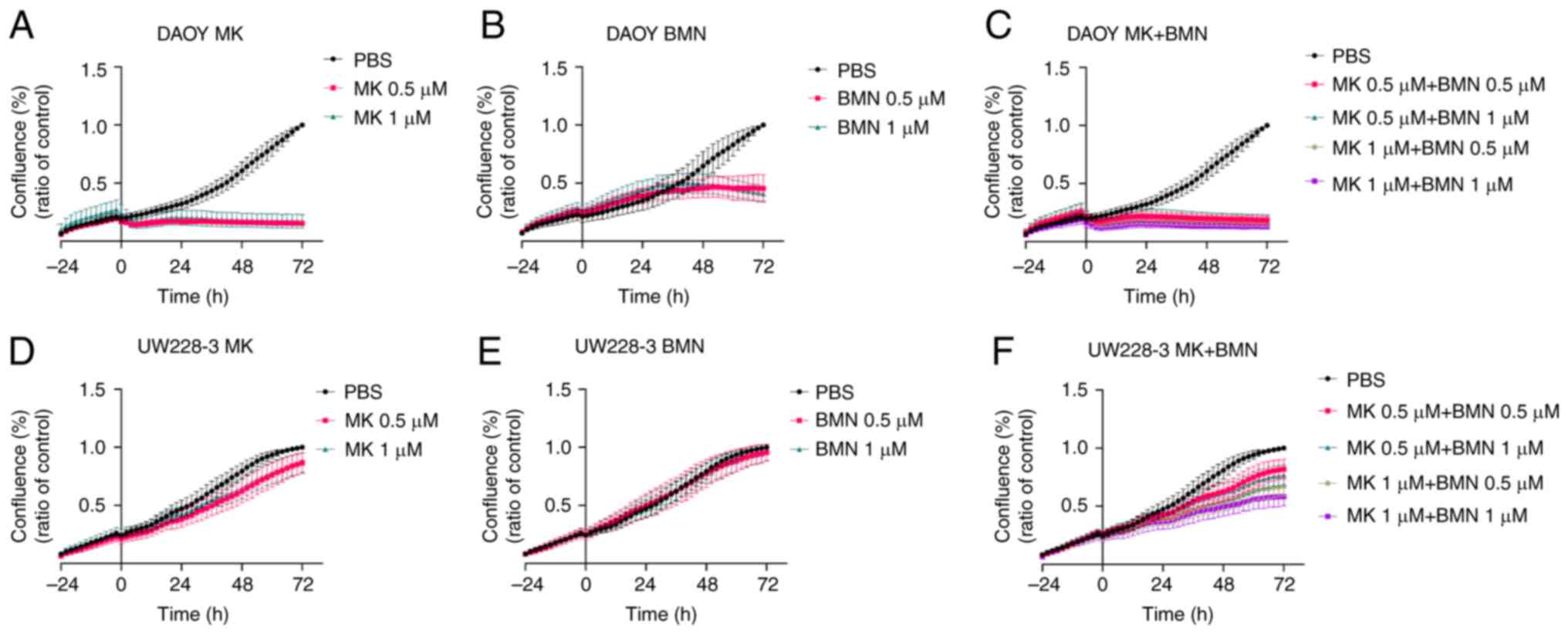
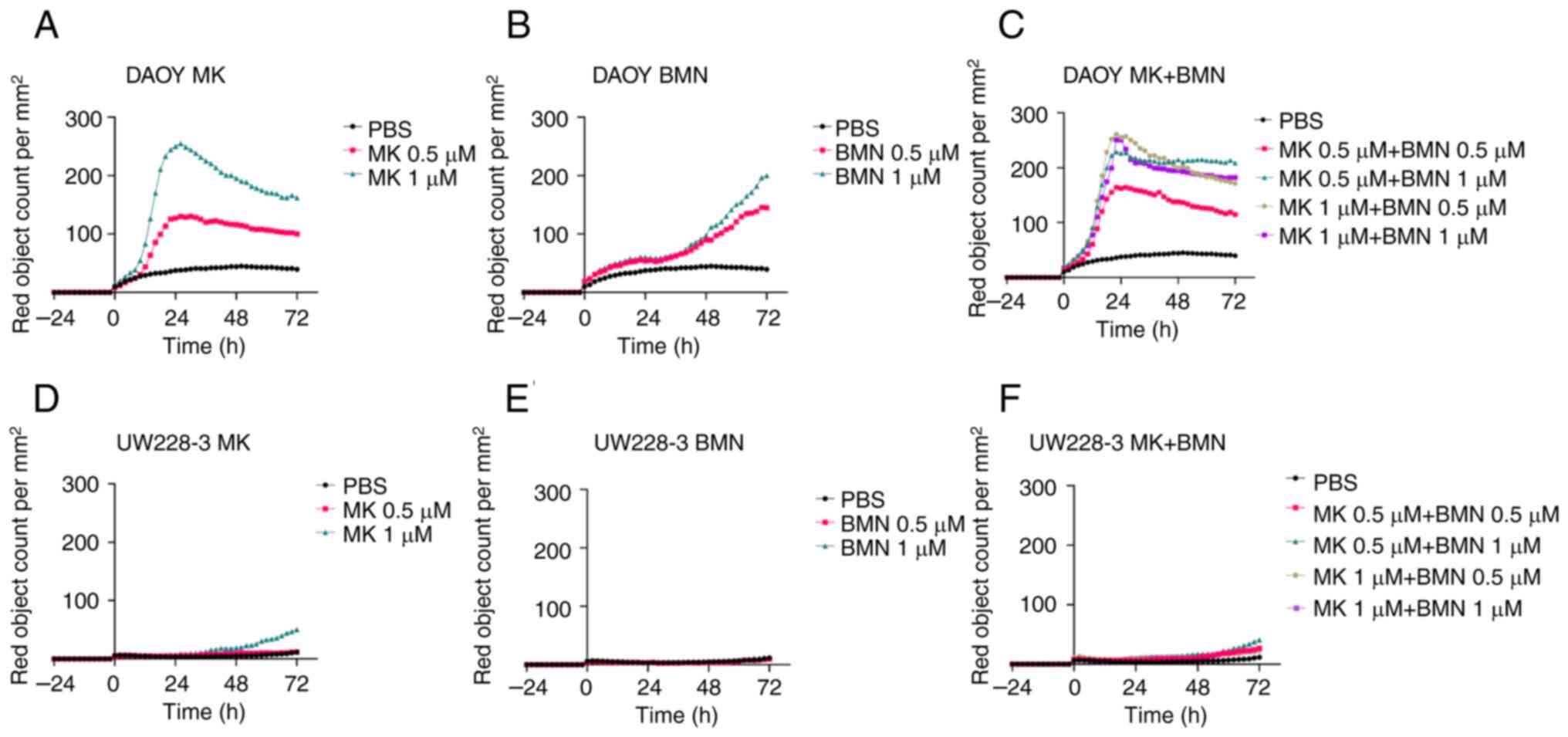
|
1
|
Northcott PA, Robinson GW, Kratz CP,
Mabbott DJ, Pomeroy SL, Clifford SC, Rutkowski S, Ellison DW,
Malkin D, Taylor MD, et al: Medulloblastoma. Nat Rev Dis Primers.
5:112019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mahapatra S and Amsbaugh MJ:
Medulloblastoma. StatPearls Treasure Island (FL): pp. pp12021
|
|
3
|
Northcott PA, Jones DT, Kool M, Robinson
GW, Gilbertson RJ, Cho YJ, Pomeroy SL, Korshunov A, Lichter P,
Taylor MD and Pfister SM: Medulloblastomics: The end of the
beginning. Nat Rev Cancer. 12:818–834. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kumar LP, Deepa SF, Moinca I, Suresh P and
Naidu KV: Medulloblastoma: A common pediatric tumor: Prognostic
factors and predictors of outcome. Asian J Neurosurg. 10:502015.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Martin AM, Raabe E, Eberhart C and Cohen
KJ: Management of pediatric and adult patients with
medulloblastoma. Curr Treat Options Oncol. 15:581–594. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu X, Ding C, Tan W and Zhang A:
Medulloblastoma: Molecular understanding, treatment evolution, and
new developments. Pharmacol Ther. 210:1075162020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Leary SE and Olson JM: The molecular
classification of medulloblastoma: Driving the next generation
clinical trials. Curr Opin Pediatr. 24:33–39. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mulhern RK, Palmer SL, Merchant TE,
Wallace D, Kocak M, Brouwers P, Krull K, Chintagumpala M, Stargatt
R, Ashley DM, et al: Neurocognitive consequences of risk-adapted
therapy for childhood medulloblastoma. J Clin Oncol. 23:5511–5519.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Salloum R, Chen Y, Yasui Y, Packer R,
Leisenring W, Wells E, King A, Howell R, Gibson TM, Krull KR, et
al: Late morbidity and mortality among medulloblastoma survivors
diagnosed across three decades: A report from the childhood cancer
survivor study. J Clin Oncol. 37:731–740. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cotter JA and Hawkins C: Medulloblastoma:
WHO 2021 and beyond. Pediatr Dev Pathol. 25:23–33. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Louis DN, Perry A, Wesseling P, Brat DJ,
Cree IA, Figarella-Branger D, Hawkins C, Ng HK, Pfister SM,
Reifenberger G, et al: The 2021 WHO classification of tumors of the
central nervous system: A summary. Neuro Oncol. 23:1231–1251. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Remke M, Ramaswamy V and Taylor M:
Medulloblastoma molecular dissection: The way toward targeted
therapy. Curr Opin Oncol. 25:674–681. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kijima N and Kanemura Y: Molecular
classification of medulloblastoma. Neurol Med Chir (Tokyo).
56:687–697. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lospinoso Severini L, Ghirga F, Bufalieri
F, Quaglio D, Infante P and Di Marcotullio L: The SHH/GLI signaling
pathway: A therapeutic target for medulloblastoma. Expert Opin Ther
Targets. 24:1159–1181. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gajjar AJ and Robinson GW:
Medulloblastoma-translating discoveries from the bench to the
bedside. Nat Rev Clin Oncol. 11:714–722. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Northcott PA, Korshunov A, Pfister SM and
Taylor MD: The clinical implications of medulloblastoma subgroups.
Nat Rev Neurol. 8:340–351. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kool M, Korshunov A, Remke M, Jones DT,
Schlanstein M, Northcott PA, Cho YJ, Koster J, Schouten-van
Meeteren A, van Vuurden D, et al: Molecular subgroups of
medulloblastoma: An international meta-analysis of transcriptome,
genetic aberrations, and clinical data of WNT, SHH, group 3, and
group 4 medulloblastomas. Acta Neuropathol. 123:473–484. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rouleau M, Patel A, Hendzel MJ, Kaufmann
SH and Poirier GG: PARP inhibition: PARP1 and beyond. Nat Rev
Cancer. 10:293–301. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Beck C, Robert I, Reina-San-Martin B,
Schreiber V and Dantzer F: Poly(ADP-ribose) polymerases in
double-strand break repair: Focus on PARP1, PARP2 and PARP3. Exp
Cell Res. 329:18–25. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mladenov E and Iliakis G: Induction and
repair of DNA double strand breaks: The increasing spectrum of
non-homologous end joining pathways. Mutat Res. 711:61–72. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Benafif S and Hall M: An update on PARP
inhibitors for the treatment of cancer. Onco Targets Ther.
8:519–528. 2015.PubMed/NCBI
|
|
22
|
Ray Chaudhuri A and Nussenzweig A: The
multifaceted roles of PARP1 in DNA repair and chromatin
remodelling. Nat Rev Mol Cell Biol. 18:610–621. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang Z, Wang F, Tang T and Guo C: The role
of PARP1 in the DNA damage response and its application in tumor
therapy. Front Med. 6:156–164. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shen Y, Rehman FL, Feng Y, Boshuizen J,
Bajrami I, Elliott R, Wang B, Lord CJ, Post LE and Ashworth A: BMN
673, a novel and highly potent PARP1/2 inhibitor for the treatment
of human cancers with DNA repair deficiency. Clin Cancer Res.
19:5003–5015. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Murai J, Huang SY, Renaud A, Zhang Y, Ji
J, Takeda S, Morris J, Teicher B, Doroshow JH and Pommier Y:
Stereospecific PARP trapping by BMN 673 and comparison with
olaparib and rucaparib. Mol Cancer Ther. 13:433–443. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hoy SM: Talazoparib: First global
approval. Drugs. 78:1939–1946. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Buck J, Dyer PJC, Hii H, Carline B,
Kuchibhotla M, Byrne J, Howlett M, Whitehouse J, Ebert MA, McDonald
KL, et al: Veliparib is an effective radiosensitizing agent in a
preclinical model of medulloblastoma. Front Mol Biosci.
8:6333442021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
van Vuurden DG, Hulleman E, Meijer OL,
Wedekind LE, Kool M, Witt H, Vandertop PW, Würdinger T, Noske DP,
Kaspers GJ and Cloos J: PARP inhibition sensitizes childhood high
grade glioma, medulloblastoma and ependymoma to radiation.
Oncotarget. 2:984–996. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Daniel RA, Rozanska AL, Mulligan EA, Drew
Y, Thomas HD, Castelbuono DJ, Hostomsky Z, Plummer ER, Tweddle DA,
Boddy AV, et al: Central nervous system penetration and enhancement
of temozolomide activity in childhood medulloblastoma models by
poly(ADP-ribose) polymerase inhibitor AG-014699. Br J Cancer.
103:1588–1596. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Garcia TB, Snedeker JC, Baturin D, Gardner
L, Fosmire SP, Zhou C, Jordan CT, Venkataraman S, Vibhakar R and
Porter CC: A small-molecule inhibitor of WEE1, AZD1775, synergizes
with olaparib by impairing homologous recombination and enhancing
DNA damage and apoptosis in acute leukemia. Mol Cancer Ther.
16:2058–2068. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
McGowan CH and Russell P: Cell cycle
regulation of human WEE1. EMBO J. 14:2166–2175. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ghelli Luserna di Rorà A, Cerchione C,
Martinelli G and Simonetti G: A WEE1 family business: Regulation of
mitosis, cancer progression, and therapeutic target. J Hematol
Oncol. 13:1262020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Do K, Doroshow JH and Kummar S: WEE1
kinase as a target for cancer therapy. Cell Cycle. 12:3159–3164.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jin P, Gu Y and Morgan DO: Role of
inhibitory CDC2 phosphorylation in radiation-induced G2 arrest in
human cells. J Cell Biol. 134:963–970. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mueller S, Hashizume R, Yang X, Kolkowitz
I, Olow AK, Phillips J, Smirnov I, Tom MW, Prados MD, James CD, et
al: Targeting Wee1 for the treatment of pediatric high-grade
gliomas. Neuro Oncol. 16:352–360. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Caretti V, Hiddingh L, Lagerweij T,
Schellen P, Koken PW, Hulleman E, van Vuurden DG, Vandertop WP,
Kaspers GJ, Noske DP and Wurdinger T: WEE1 kinase inhibition
enhances the radiation response of diffuse intrinsic pontine
gliomas. Mol Cancer Ther. 12:141–150. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Magnussen GI, Holm R, Emilsen E, Rosnes
AK, Slipicevic A and Flørenes VA: High expression of Wee1 is
associated with poor disease-free survival in malignant melanoma:
Potential for targeted therapy. PLoS One. 7:e382542012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Iorns E, Lord CJ, Grigoriadis A, McDonald
S, Fenwick K, Mackay A, Mein CA, Natrajan R, Savage K, Tamber N, et
al: Integrated functional, gene expression and genomic analysis for
the identification of cancer targets. PLoS One. 4:e51202009.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mir SE, De Witt Hamer PC, Krawczyk PM,
Balaj L, Claes A, Niers JM, Van Tilborg AA, Zwinderman AH, Geerts
D, Kaspers GJ, et al: In silico analysis of kinase expression
identifies WEE1 as a gatekeeper against mitotic catastrophe in
glioblastoma. Cancer Cell. 18:244–257. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hirai H, Arai T, Okada M, Nishibata T,
Kobayashi M, Sakai N, Imagaki K, Ohtani J, Sakai T, Yoshizumi T, et
al: MK-1775, a small molecule Wee1 inhibitor, enhances anti-tumor
efficacy of various DNA-damaging agents, including 5-fluorouracil.
Cancer Biol Ther. 9:514–522. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wang G, Niu X, Zhang W, Caldwell JT,
Edwards H, Chen W, Taub JW, Zhao L and Ge Y: Synergistic antitumor
interactions between MK-1775 and panobinostat in preclinical models
of pancreatic cancer. Cancer Lett. 356:656–668. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ghiasi N, Habibagahi M, Rosli R, Ghaderi
A, Yusoff K, Hosseini A, Abdullah S and Jaberipour M: Tumour
suppressive effects of WEE1 gene silencing in breast cancer cells.
Asian Pac J Cancer Prev. 14:6605–6611. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Leijen S, Beijnen JH and Schellens JH:
Abrogation of the G2 checkpoint by inhibition of Wee-1 kinase
results in sensitization of p53-deficient tumor cells to
DNA-damaging agents. Curr Clin Pharmacol. 5:186–191. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Hirai H, Iwasawa Y, Okada M, Arai T,
Nishibata T, Kobayashi M, Kimura T, Kaneko N, Ohtani J, Yamanaka K,
et al: Small-molecule inhibition of Wee1 kinase by MK-1775
selectively sensitizes p53-deficient tumor cells to DNA-damaging
agents. Mol Cancer Ther. 8:2992–3000. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Rajeshkumar NV, De Oliveira E, Ottenhof N,
Watters J, Brooks D, Demuth T, Shumway SD, Mizuarai S, Hirai H,
Maitra A and Hidalgo M: MK-1775, a potent Wee1 inhibitor,
synergizes with gemcitabine to achieve tumor regressions,
selectively in p53-deficient pancreatic cancer xenografts. Clin
Cancer Res. 17:2799–2806. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Harris PS, Venkataraman S, Alimova I,
Birks DK, Balakrishnan I, Cristiano B, Donson AM, Dubuc AM, Taylor
MD, Foreman NK, et al: Integrated genomic analysis identifies the
mitotic checkpoint kinase WEE1 as a novel therapeutic target in
medulloblastoma. Mol Cancer. 13:722014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Holzhauser S, Lukoseviciute M, Andonova T,
Ursu RG, Dalianis T, Wickström M and Kostopoulou ON: Targeting
fibroblast growth factor receptor (FGFR) and phosphoinositide
3-kinase (PI3K) signaling pathways in medulloblastoma cell lines.
Anticancer Res. 40:53–66. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Lukoseviciute M, Maier H,
Poulou-Sidiropoulou E, Rosendahl E, Holzhauser S, Dalianis T and
Kostopoulou ON: Targeting PI3K, FGFR, CDK4/6 signaling pathways
together with cytostatics and radiotherapy in two medulloblastoma
cell lines. Front Oncol. 11:7486572021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kostopoulou ON, Holzhauser S, Lange BKA,
Ohmayer A, Andonova T, Bersani C, Wickström M and Dalianis T:
Analyses of FGFR3 and PIK3CA mutations in neuroblastomas and the
effects of the corresponding inhibitors on neuroblastoma cell
lines. Int J Oncol. 55:1372–1384. 2019.PubMed/NCBI
|
|
50
|
Zheng S, Wang W, Aldahdooh J, Malyutina A,
Shadbahr T, Tanoli Z, Pessia A and Jing T: SynergyFinder Plus:
Toward better interpretation and annotation of drug combination
screening datasets. Genomics Proteomics Bioinformatics. 20:587–596.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Smith MA, Hampton OA, Reynolds CP, Kang
MH, Maris JM, Gorlick R, Kolb EA, Lock R, Carol H, Keir ST, et al:
Initial testing (stage 1) of the PARP inhibitor BMN 673 by the
pediatric preclinical testing program: PALB2 mutation predicts
exceptional in vivo response to BMN 673. Pediatr Blood Cancer.
62:91–98. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Moreira DC, Venkataraman S, Subramanian A,
Desisto J, Balakrishnan I, Prince E, Pierce A, Griesinger A, Green
A, Eberhardt CG, et al: Targeting MYC-driven replication stress in
medulloblastoma with AZD1775 and gemcitabine. J Neurooncol.
147:531–545. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kreahling JM, Gemmer JY, Reed D, Letson D,
Bui M and Altiok S: MK1775, a selective Wee1 inhibitor, shows
single-agent antitumor activity against sarcoma cells. Mol Cancer
Ther. 11:174–182. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Guertin AD, Li J, Liu Y, Hurd MS, Schuller
AG, Long B, Hirsch HA, Feldman I, Benita Y, Toniatti C, et al:
Preclinical evaluation of the WEE1 inhibitor MK-1775 as
single-agent anticancer therapy. Mol Cancer Ther. 12:1442–1452.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Byskata K, Lukoseviciute M, Tuti F,
Zupancic M, Kostopoulou ON, Holzhauser S and Dalianis T: Targeted
therapy with PI3K, PARP, and WEE1 inhibitors and radiotherapy in
HPV positive and negative tonsillar squamous cell carcinoma cell
lines reveals synergy while effects with APR-246 are limited.
Cancers (Basel). 15:932022. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Heijink AM, Blomen VA, Bisteau X, Degener
F, Matsushita FY, Kaldis P, Foijer F and van Vugt MA: A haploid
genetic screen identifies the G1/S regulatory machinery as a
determinant of Wee1 inhibitor sensitivity. Proc Natl Acad Sci USA.
112:15160–15165. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Guney Eskiler G, Cecener G, Egeli U and
Tunca B: Synthetically lethal BMN 673 (talazoparib) loaded solid
lipid nanoparticles for BRCA1 mutant triple negative breast cancer.
Pharm Res. 35:2182018. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Bucher N and Britten CD: G2 checkpoint
abrogation and checkpoint kinase-1 targeting in the treatment of
cancer. Br J Cancer. 98:523–528. 2008. View Article : Google Scholar : PubMed/NCBI
|