Introduction
Thyroid carcinoma (TC) is the most prevalent
malignant endocrine neoplasm originating from either the thyroid
follicular or parafollicular epithelial cells (1). Notably, it constitutes ~12% of newly
diagnosed cancers in adolescents and has witnessed a steady
increase in incidence rates (2,3).
Predominantly affecting females, TC ranks as the second most common
cancer in young women aged 18–35 years old (4). Multiple carcinogenic factors,
including nutrition/dietary practices, obesity, and radiation
exposure, synergistically contribute to the onset of TC (5). Current therapeutic modalities, which
include radiation, thyroid hormone therapy, and surgery, are
frequently employed in combination (5,6).
Although the treatment of TC is improving, owing to the lack of
precise molecular targets, the overall 5-year survival rate of
patients with TC remains suboptimal, particularly in patients with
advanced clinical stage TC (7).
Therefore, there is an urgent need to improve our understanding of
the underlying mechanisms and to identify novel therapeutic
strategies for the management of TC.
Arginine methyltransferase 1 (PRMT1), a member of
the PRMT family, plays an important role in arginine methylation
(8). PRMT1 can coactivate the
histone code and epigenetic control by methylating histone H4 at
the third arginine residue (H4R3) (9), in particular H4R3me2as, which is a
marker of transcriptional activation (10). PRMT1 modulates epigenetic changes in
physiological and pathophysiological processes including cell
proliferation, survival, and metabolism (11). Increased PRMT1 expression drives
tumorigenesis and progression in various types of cancer, including
lung, breast, and prostate cancer (12–14).
Collectively, these results indicate that PRMT1 contributes to
cancer progression by methylating both histone and non-histone
proteins. A previous study revealed that PRMT1 was aberrantly
expressed in TC tumor tissues compared with normal tissues
(15). However, to the best of the
authors' knowledge, there are no studies assessing the function of
PRMT1 in TC.
Zinc-finger E homeobox-binding 1 (ZEB1) is an
important transcription factor for epithelial-mesenchymal
transition (EMT) (16). It is
aberrantly expressed in several types of cancer, including gastric,
colorectal, and cervical cancer (17–19).
Evidence has shown that PRMT1 modulates EMT by regulating ZEB1
expression in breast cancer (20).
Furthermore, ZEB1 expression is associated with PRMT1 expression in
pancreatic cancer cells (21).
However, it remains unclear whether PRMT1 regulates ZEB1 expression
in TC.
In this study, the effect of PRMT1 on TC cell
proliferation, migration, and apoptosis was assessed by modulating
its expression in vitro using TC cell lines and in
vivo using a xenograft mouse model. This study offers a novel
perspective for potential therapeutic avenues in the management of
TC.
Materials and methods
Cell culture
Human TC cell lines (8505C, CAL62, and BCPAP) and
the normal human thyroid cell line Nthy-ori 3-1 were supplied by
the National Collection of Authenticated Cell Cultures. The BCPAP
cells were authenticated using short tandem repeat profiling (iCell
Bioscience, Inc.). 8505C and CAL62 cells were cultured in DMEM
(Gibco; Thermo Fisher Scientific, Inc.), whereas BCPAP and Nthy-ori
3-1 cells were cultured in RPMI-1640 medium (Gibco; Thermo Fisher
Scientific, Inc.). In both cases, media was supplemented with 10%
FBS (MilliporeSigma) and cells were maintained in a humidified
incubator supplied with 5% CO2 air at 37°C.
Cell transfection and treatment
PRMT1 small interfering RNA (siRNA; si-PRMT1),
negative control (si-NC), si-ZEB1, and a PRMTI overexpression
plasmid (oe-PRMT1) were designed by Tsingke Biotechnology Co., Ltd.
The cells were cultured to 70–80% confluence in six-well plates.
Next, 8505C cells were transfected with 50 nM si-PRMT1 or si-NC and
BCPAP cells with 0.8 µg oe-PRMT1 or co-transfected with si-ZEB1
using the Lipofectamine® 3000 system (Invitrogen; Thermo
Fisher Scientific, Inc.) for 6 h. The sequences of siRNA were:
si-PRMT1, GCCTACTTCAACATCGAGT; si-ZEB1, TGCAGAAAATGAGCAAAACCATG;
and si-NC, TTCTCCGAACGTGTCACG. Cells transfected with si-PRMT1 were
then treated with 10 or 50 µM AMI-1 (a PRMT1 inhibitor; T2352;
TargetMol) for 48 h, or otherwise untreated and used as control
cells.
CCK-8 assay
Cell viability was determined using a CCK-8 assay
kit (Beyotime Institute of Biotechnology), according to the
manufacturer's instructions. The 8505C or BCPAP cells
(5×103 cells/well) were placed into 96-well plates and
cultured for 0, 10, or 24 h for 8505C cells, or 0, 48, and 72 h for
BCPAP, after which they were treated with 10 µl CCK-8 reagent for a
further 2 h. Subsequently, the absorbance at 450 nm was measured
using a microplate reader (Thermo Fisher Scientific, Inc.).
Colony formation assay
BCPAP and 8505C cells (1×103 cells/well)
were incubated for 7 days in six-well plates. The cells were fixed
with 4% paraformaldehyde for 15 min and stained with 0.1% crystal
violet (Sangon Biotech) for 10 min both at room temperature, after
which cells were imaged using a light microscope at ×100
magnification (Olympus Corporation). The number of colonies in five
random, non-overlapping fields of view were counted and
averaged.
Flow cytometry assay
Apoptosis was assessed using an Annexin V-FITC
Apoptosis Detection Kit (MilliporeSigma). BCPCP and 8505C cells
were seeded in six-well plates at a density of 1×105
cells/well. Cells were allowed to adhere overnight after which the
cells were stained with Annexin V/PI for 10 min at room temperature
and the percentage of apoptotic cells was examined using a flow
cytometer (FACSCalibur; BD Biosciences) and analyzed using
CellQuest software 3.3 (BD Biosciences).
Wound healing assay
The cells were incubated until they were 90%
confluent in six-well plates. The monolayer of cells was scratched
using a 100 µl pipette tip. After removing the detached cells using
PBS, the cells were imaged (0 h) and then incubated for 24 h and
imaged again (24 h). Cell images were acquired using an inverted
light microscope (Olympus Corporation) at 0 and 24 h. The wound
width was measured using Image-Pro Plus 6.0 (Media Cybernetics,
Inc.).
Transwell assays
Cell migration was determined using a Corning
BioCoat Matrigel Invasion Chamber (Corning, NY, USA). To the upper
chamber of the Transwell insert, a 200 µl of serum-free cell
suspension (5×104 cells) was added. Medium (600 µl) with
10% FBS was added to the lower chambers. After 24 h of incubation,
the invaded cells were fixed with 4% paraformaldehyde for 30 min
and stained with crystal violet for an additional 30 min both at
room temperature. Cells were counted in five arbitrary optical
fields of view under a light microscope (magnification, ×200;
Olympus Corporation).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from TC tissues and cells
using the TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.). cDNA was synthesized using the FastKing-RT
SuperMix (Tiangen Biotech Co., Ltd.). Next, Super SYBR Green qPCR
Master mix (Life Inc.) was used for PCR amplification using a
real-time PCR system (Bio-Rad Laboratories, Inc.). The
amplification conditions were 95°C for 3 min for denaturation;
followed by 40 cycles of 95°C for 5 sec, and 60°C for 45 sec.
Relative mRNA expression was determined using the 2−ΔΔCq
method (22), with β-actin serving
as an internal control. The primer sequences used were: PRMT1
forward, 5′-ACTGCCTCTTCTACGAGTCCA-3′ and reverse,
5′-TGCACGTAGTCATTCCGCTT-3′; ZEB1 forward, 5′-ACTCTGATTCTACACCGC-3′
and reverse, 5′-TGTCACATTGATAGGGCTT-3′; and β-actin forward,
5′-GACAGGATGCAGAAGGAGATTACT-3′ and reverse,
5′-TGATCCACATCTGCTGGAAGGT-3′.
Xenograft mouse model
All animal experiments were approved by the
Institutional Animal Care and Use Committee of the Affiliated
Hospital of the Zunyi Medical University (approval no.
zyfy-an-2023-0185). BALB/C nude mice (female, 6 weeks, 18–20 g,
n=16) were supplied by GemPharmatech and fed in specific
pathogen-free facilities with a 12/12 h light/dark cycle at
22–24°C. All the mice had ad libitum access to food and
water during the experimental period. To establish TC tumor-bearing
mice, 8505C or PRMT1-knockdown 8505C cell suspensions
(4×106 cells/mouse) were subcutaneously injected every 4
days (n=4 in each group). Tumor volume was monitored every 3 days
and calculated as volume=0.5 × length × width2. To
assess TC pulmonary metastasis in a nude mouse model, cell
suspensions (1×106) were injected into mice through the
tail vein. After 28 days, the mice anesthetized using 2%
pentobarbital sodium (30 mg/kg), followed by euthanasia by cervical
dislocation. The tumors were harvested, weighed, images captured
with a camera (Leica Camera AG), and prepared for subsequent
examination.
Hematoxylin and eosin (H&E)
staining
Thyroid tumor tissues were fixed in 10%
neutral-buffered formalin. Paraffin-embedded tissues were then cut
into 4 µm-thick sections and stained using hematoxylin for 5 min
and eosin for 2 min at room temperature after dewaxing. Finally,
histological changes in the tumor sections were analyzed under a
light microscope (Olympus Corporation).
TUNEL analysis
Cytospin preparations of tumor sections were
examined using a TUNEL assay. Samples were permeabilized with 0.1%
Triton X-100 in 0.1% sodium citrate for 2 min on ice and fixed for
15 min with 4% paraformaldehyde. The TUNEL assay was performed
using an in-situ cell death detection kit according to the
manufacturer's protocol (Fluorescein; Roche Diagnostics GmbH).
Immunofluorescence (IF) analysis
Cells were resuspended in media supplemented with
10% FBS and cultured for 6 h on chamber slides, after which they
were fixed in ice-cold 80% methanol for 10 min. Permeabilization
was performed using 0.5% Triton X-100 (MilliporeSigma) for 10 min.
The cells were then incubated with anti-vimentin (cat. no.
ab286811, 1:100; Abcam) or anti-E-cadherin (cat. no. ab40772,
1:500; Abcam) fluorescent primary antibodies for 60 min at room
temperature. Then, samples were counterstained with 1 µg/ml DAPI
for 5 min at room temperature, and slides were imaged using a
fluorescence microscope (magnification, ×400; Olympus
Corporation).
Western blotting
Total protein was acquired from TC tumor tissues and
cells using RIPA lysis buffer (Beyotime Institute of Biotechnology)
and protein concentration was measured using a BCA kit (Beyotime
Institute of Biotechnology). A total of 20 µg protein was loaded on
a 10% SDS gel, resolved using SDS-PAGE, and transferred to a PVDF
membrane (Beyotime Institute of Biotechnology). After blocking with
5% non-fat milk, the membrane was incubated overnight at 4°C with
one of the following primary antibodies: Anti-PRMT1 (cat. no.
ab73246; 1:500; Abcam), anti-E-cadherin (cat. no. ab40772;
1:10,000; Abcam), anti-vimentin (cat. no. ab137321; 1:1,000;
Abcam), anti-ZEB1 (cat. no. ab81972; 1:2,000; Abcam),
anti-H4R3me2as (cat. no. 39705; 1:1,000; Active Motif), or
anti-GAPDH (cat. no. ab9485; 1:2,500; Abcam). The membrane was then
incubated with a goat anti-rabbit IgG secondary antibody (cat. no.
ab205718; 1:2,000; Abcam) for 60 min. Protein bands were observed
using ECL reagent (Wuhan Servicebio Technology Co., Ltd.) and
quantified using ImageJ version 1.8.0 (National Institutes of
Health).
Statistical analysis
Data are presented as the mean ± SD of at least 3
independent repeats. Differences between ≥3 groups were compared
using a one-way ANOVA followed by a post hoc Tukey's test.
Differences between 2 groups were compared using a Student's
t-test. GraphPad Prism version 8.0 (GraphPad Software, Inc.) was
used for statistical analyses. P<0.05 was considered to indicate
a statistically significant difference.
Results
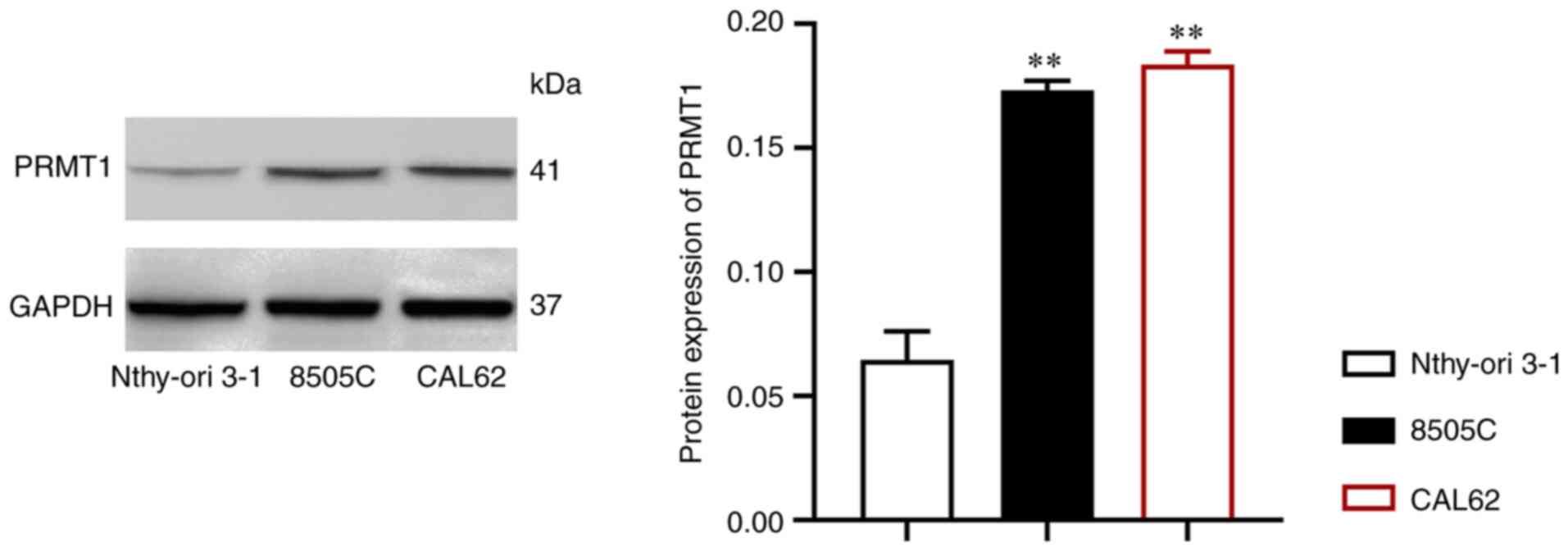
PRMT1 is upregulated in TC
First, PRMT1 expression in the TC cell lines 8505C
and CAL62 was assessed and compared with that in Nthy-ori 3-1
cells. Notably, PRMT1 expression was significantly upregulated in
the TC cell lines (P<0.01; Fig.
1).
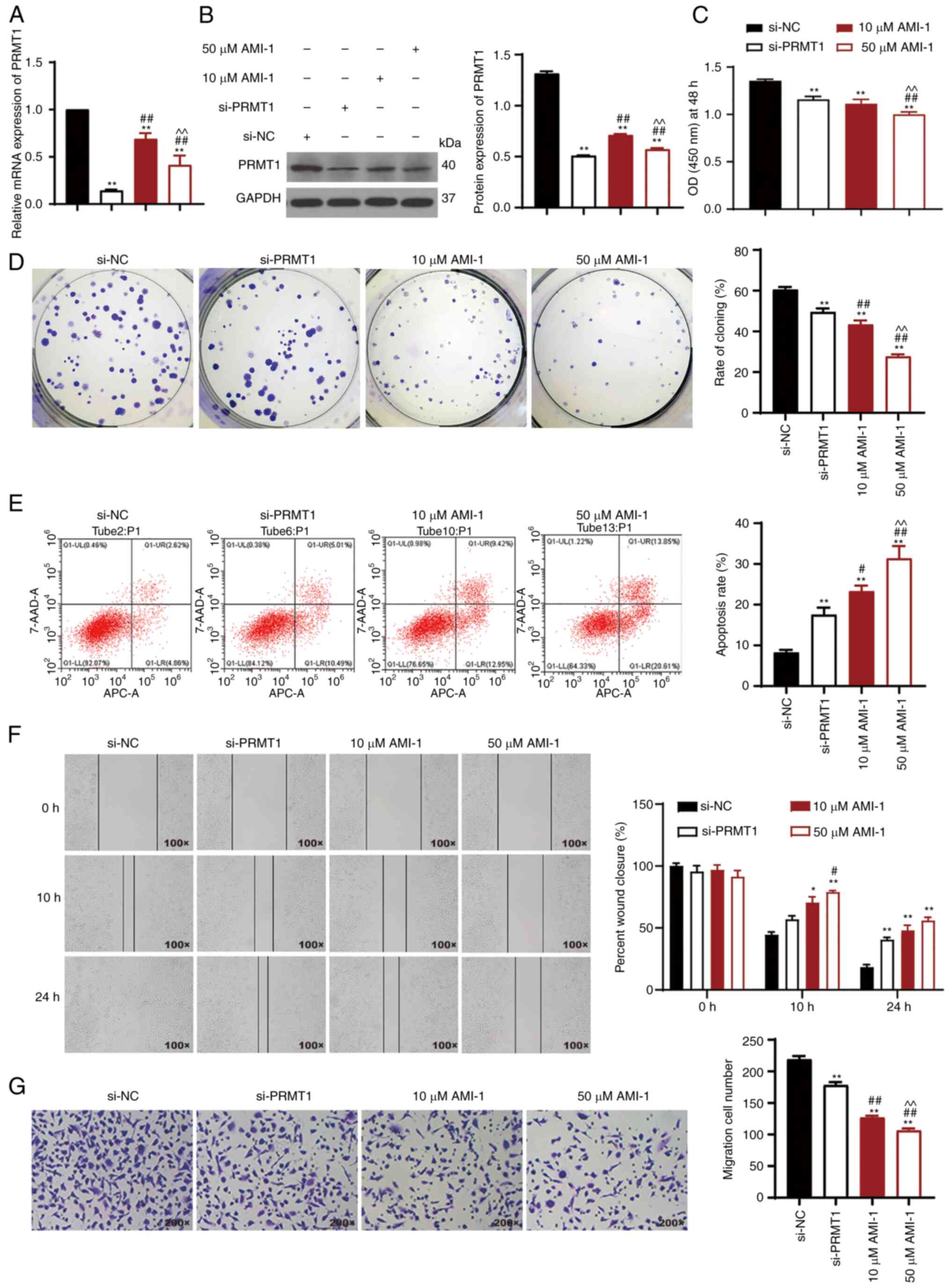
PRMT1 knockdown inhibits cell
proliferation and migration and promotes cell apoptosis in TC
8505C cells are a mesenchymal TC cell line, and it
has been shown that the upregulated genes in the mesenchymal TC
cell line are primarily involved in DNA replication and exhibit
increased proliferative and migratory capacity compared with other
cell lines (23). Therefore,
si-PRMT1 was transfected into 8505C cells, and cells treated with
PRMT1 inhibitor (10 or 50 µM AMI-1) were used as positive controls.
The results showed that the expression of PRMT1 mRNA and protein
was reduced in 8505C cells after treatment with AMI-1 or
transfected with si-PRMT1 (P<0.01; Fig. 2A and B). si-PRMT1 transfection
exhibited a better ability in reducing PRMT1 expression than did
treatment with AMI-1 (P<0.01; Fig.
2A and B). The CCK-8 assays showed a marked decrease in cell
viability following PRMT1 knockdown (P<0.01; Fig. 2C). Additionally, the colony
formation rate decreased upon PRMT1 knockdown (P<0.01; Fig. 2D). Apoptosis was increased following
knockdown of PRMT1 (P<0.01; Fig.
2E). Wound healing and Transwell assays were used to further
verify the effect of PRMT1 knockdown on cell migration (P<0.01;
Fig. 2F and G). The levels of PRMT1
expression and cell proliferation, colony formation, and migration
of 8505C cells treated with 50 µM AMI-1 were significantly lower
than those of cells treated with 10 µM AMI-1 (P0.05; Fig. 2A-G). These results demonstrated that
PRMT1 knockdown/inhibition reduced the metastatic potential and
increased the apoptosis of TC cells.
PRMT1 knockdown suppresses cell
metastasis and downregulates the expression of ZEB1 and H4R3me2as
in TC
Subsequently, the inhibitory mechanism of PRMT1
knockdown on TC progression was assessed by determining the
expression of E-cadherin, vimentin, ZEB1, and H4R3me2as in 8505C
cells treated with or without si-PRMT1 or AMI-1. IF results
indicated that E-cadherin expression increased, whereas vimentin
expression decreased in 8505C cells treated with AMI-1 or si-PRMT1
compared to si-NC cells (Fig. 3A).
Western blotting was used to further verify that PRMT1
downregulation promoted E-cadherin expression (P<0.01) and
reduced vimentin expression (P<0.01; Fig. 3B). AMI-1 exerted its effect on the
levels of E-cadherin and vimentin in a dose-dependent manner, and
50 µM AMI-1 showed a better efficiency in regulating the expression
of E-cadherin and vimentin than si-PRMT1 (P<0.01; Fig. 3B).
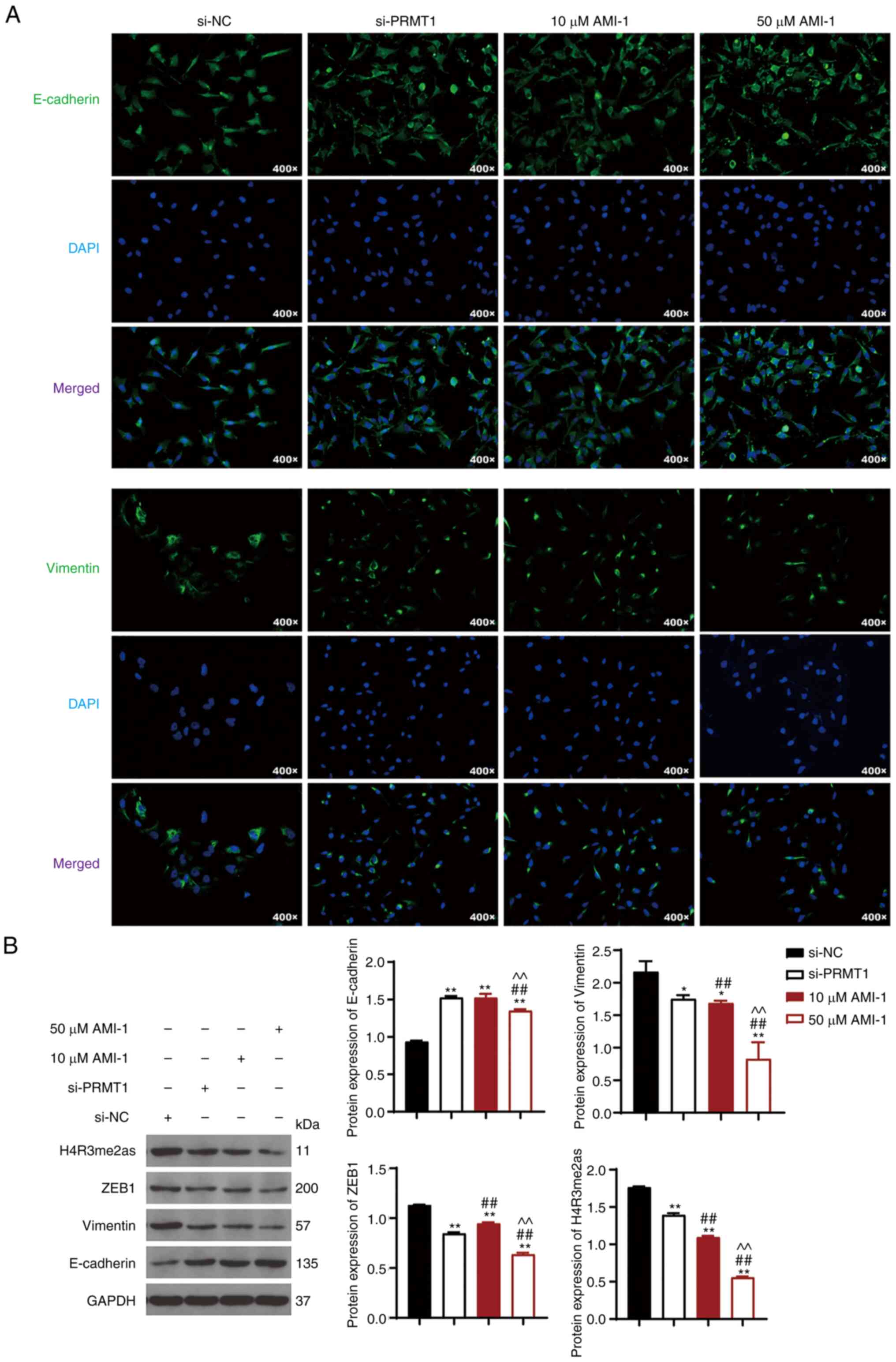 | Figure 3.PRMT1 knockdown suppresses cell
metastasis and downregulates expression of ZEB1 and H4R3me2as in
TC. (A) IF analyzed the contents of E-cadherin and vimentin in
8505C cells, magnification=400×. (B) Western blotting was used to
assess the protein expression levels of E-cadherin, vimentin,
H4R3me2as, and ZEB1 in 8505C cells. The 8505C cells were
transfected with si-NC or si-PRMT1. *P<0.05, **P<0.01
compared with si-NC; ##P<0.01 compared with si-PRMT1;
^^P<0.01 compared with 10 µM AMI-1. Each experiment
was conducted at least thrice. PRMT1, protein arginine
methyltransferase 1; ZEB1, zinc finger e-box binding homeobox1,
H4R3me2as, asymmetric demethylation of H4 at the third arginine
residue; TC, thyroid carcinoma; si-NC, small interfering RNA
negative control. |
PRMT1 downregulation also decreased the protein
expression levels of ZEB1 and H4R3me2as (P<0.01; Fig. 3B). Notably, si-PRMT1 exhibited a
better effect on reversing ZEB1 expression compared with 10 µM
AMI-1 (P<0.01; Fig. 3B).
PRMT1 overexpression accelerates TC
progression by downregulating the expression of ZEB1 and
H4R3me2as
To further verify the role of PRMT1 on the malignant
characteristics of TC, PRMT1 was overexpressed in BCPAP cells, a TC
cell line that has been previously used as a control of 8505C cells
(24). PRMT1 expression was
successfully upregulated after transfection with oe-PRMT1
(P<0.01; Fig. 4A and B).
Compared with the control cells, PRMT1 overexpression promoted cell
proliferation and migration (P<0.05) but did not significantly
affect apoptosis (P=0.06; Fig.
4C-G). Additionally, E-cadherin expression was downregulated,
and vimentin expression was upregulated in BCPAP cells upon PRMT1
overexpression (P<0.05; Fig. 4H and
I). PRMT1 overexpression increased the expression of ZEB1 and
H4R3me2as in BCPAP cells (P<0.01; Fig. 4I). This study further demonstrates
that PRMT1 promotes TC progression by upregulating the expression
of ZEB1 and H4R3me2as.
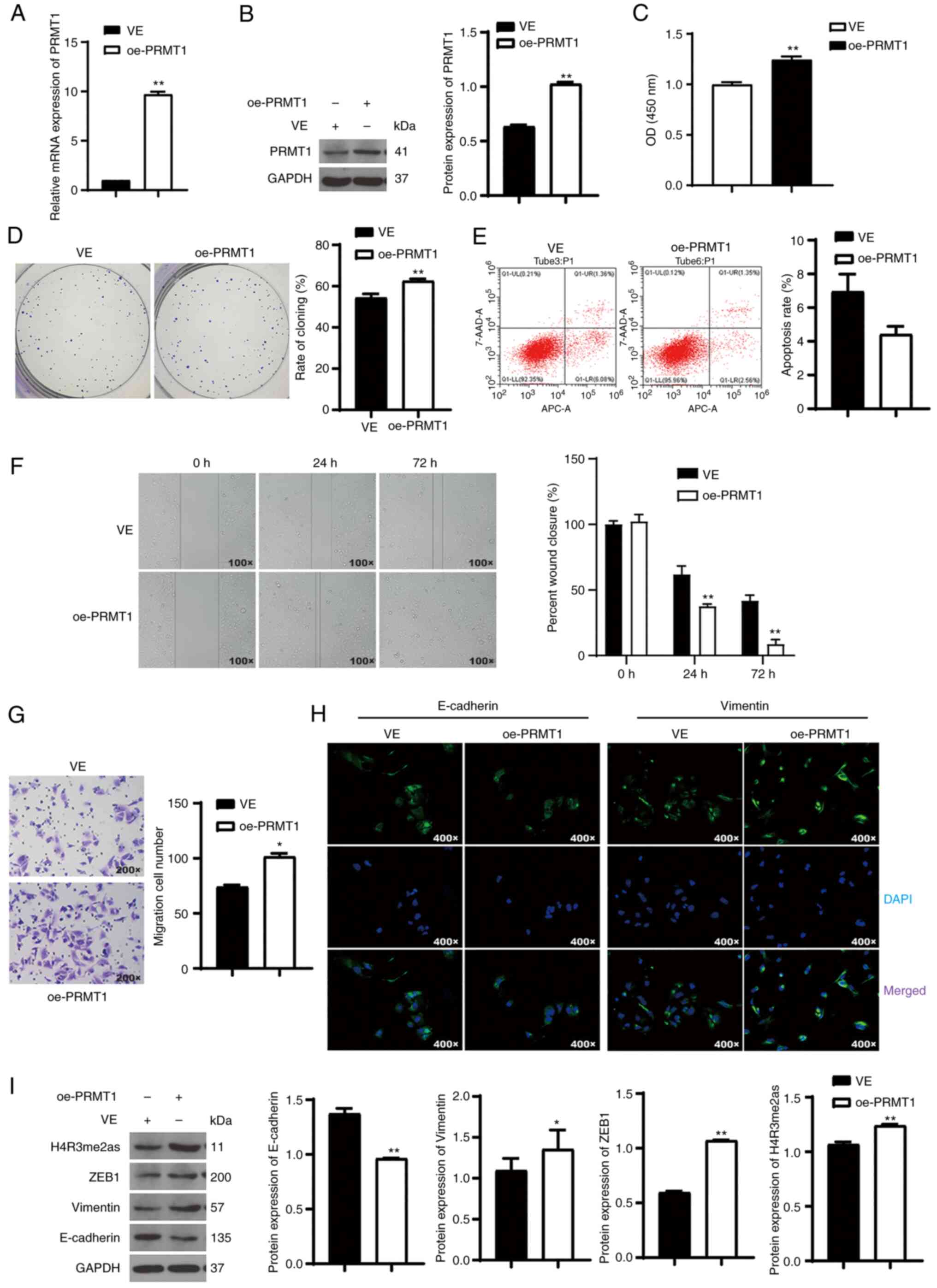 | Figure 4.PRMT1 overexpression accelerates TC
progression and upregulates the expression of ZEB1 and H4R3me2as.
(A and B) RT-qPCR and western bolting were used to assess the
expression of PRMT1 in BCPAP cells. (C and D) The CCK-8 and colony
formation analyses were used to determine BCPAP cell viability and
proliferation, respectively. (E) Flow cytometry was used to analyze
BCPAP cell apoptosis. (F and G) Wound healing (magnification, ×100)
and Transwell (magnification, ×200) assays were used to assess cell
migration of BCPAP cells. (H) IF was used to assess E-cadherin and
vimentin expression in BCPAP cells. Magnification, ×400. (I)
Western blotting was used to assess the protein expression of
E-cadherin, vimentin, H4R3me2as, and ZEB1 in BCPAP cells. BCPAP
cells were transfected with oe-PRMT1. *P<0.05, **P<0.01 vs.
with VE. PRMT1, protein arginine methyltransferase 1; ZEB1, zinc
finger e-box binding homeobox1; H4R3me2as, asymmetric demethylation
of H4 at the third arginine residue; TC, thyroid carcinoma;
oe-PRMT1, overexpression-PRMT1; VE, vector. |
Subsequently, BCPAP cells were co-transfected with
oe-PRMT1 and si-ZEB1. As shown in Fig.
5A and B, ZEB1 expression was successfully knocked down
following si-ZEB1 transfection, and PRMT1 expression was elevated
in BCPAP cells (P<0.01). ZEB1 knockdown reversed the effects of
PRMT1 overexpression on promoting cell proliferation, migration,
and expression of vimentin and E-cadherin (P<0.01; Fig. 5C-G). Additionally, ZEB1 inhibition
downregulated the protein expression of H4R3me2as induced by
oe-PRMT1 (P<0.01; Fig. 5G).
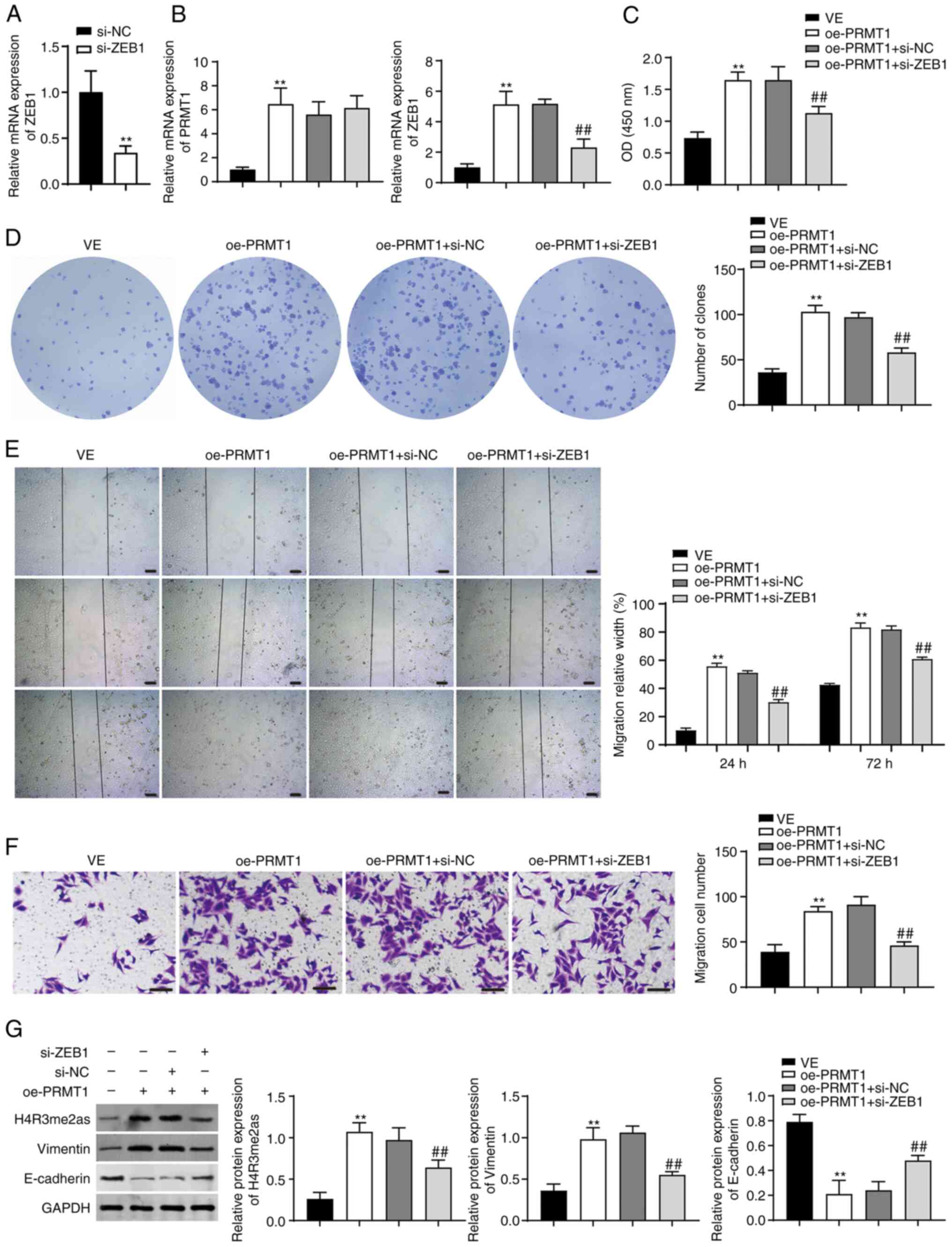 | Figure 5.PRMT1 overexpression accelerates TC
progression through the activation of ZEB1/H4R3me2as. (A) RT-qPCR
was used to assess the expression of ZEB1 in BCPAP cells
transfected with si-ZEB1. (B) mRNA expression of PRMT1 and ZEB1 in
multiple group of cells. (C and D) CCK-8 and colony formation
assays were used to test BCPAP cell viability and proliferation,
respectively. (E and F) Wound healing and Transwell assays were
used to assess cell migration of BCPAP cells. Scale bar, 50 µM. (G)
Western blotting was used to measure the protein expression levels
of E-cadherin, vimentin, and H4R3me2as in BCPAP cells. BCPAP cells
were transfected with oe-PRMT1 and/or si-ZEB1. **P<0.01 vs. VE
group, ##P<0.01 vs. oe-PRMT1 + si-NC group. PRMT1,
protein arginine methyltransferase 1; ZEB1, zinc finger e-box
binding homeobox1; H4R3me2as, asymmetric demethylation of H4 at the
third arginine residue; TC, thyroid carcinoma; oe-PRMT1,
overexpression-PRMT1; VE, vector. |
PRMT1 knockdown reduces tumor growth
and metastasis by downregulating ZEB1/H4R3me2as in a xenograft
mouse model of TC
A TC xenograft mouse model was established to
explore the effects of PRMT1 on tumor growth. PRMT1 protein levels
were reduced in TC tumor tissues from tumor-bearing mice treated
with si-PRMT1 or AMI-1 compared with those in the control group
(P<0.01; Fig. 6A). The data
showed that PRMT1 downregulation inhibited tumor growth in mice
(P<0.01; Fig. 6B). Cells in the
si-NC group exhibited apparent ablation, and the nuclei showed
nuclear pleomorphism and small nucleoli; however, PRMT1
downregulation ameliorated these phenomena (Fig. 6C). Moreover, there were pulmonary
nodules in the lungs of mice with TC, and the extent of these
nodules was reduced in the mice injected with the PRMT1 knockdown
cells (P<0.01; Fig. 6D). TUNEL
apoptosis assays showed that the number of positive cells increased
following PRMT1 knockdown (Fig.
6E). Western blotting indicated that E-cadherin expression was
upregulated, whereas the expression of vimentin, ZEB1, and
H4R3me2as was decreased following PRMT1 knockdown (P<0.05;
Fig. 6F). Additionally, AMI-1
regulated tumor growth and the expression these proteins in a
dose-dependent manner, and 50 µM AMI-1 exhibited a more potent
effect than si-PRMT1 (P<0.05; Fig.
6A-F).
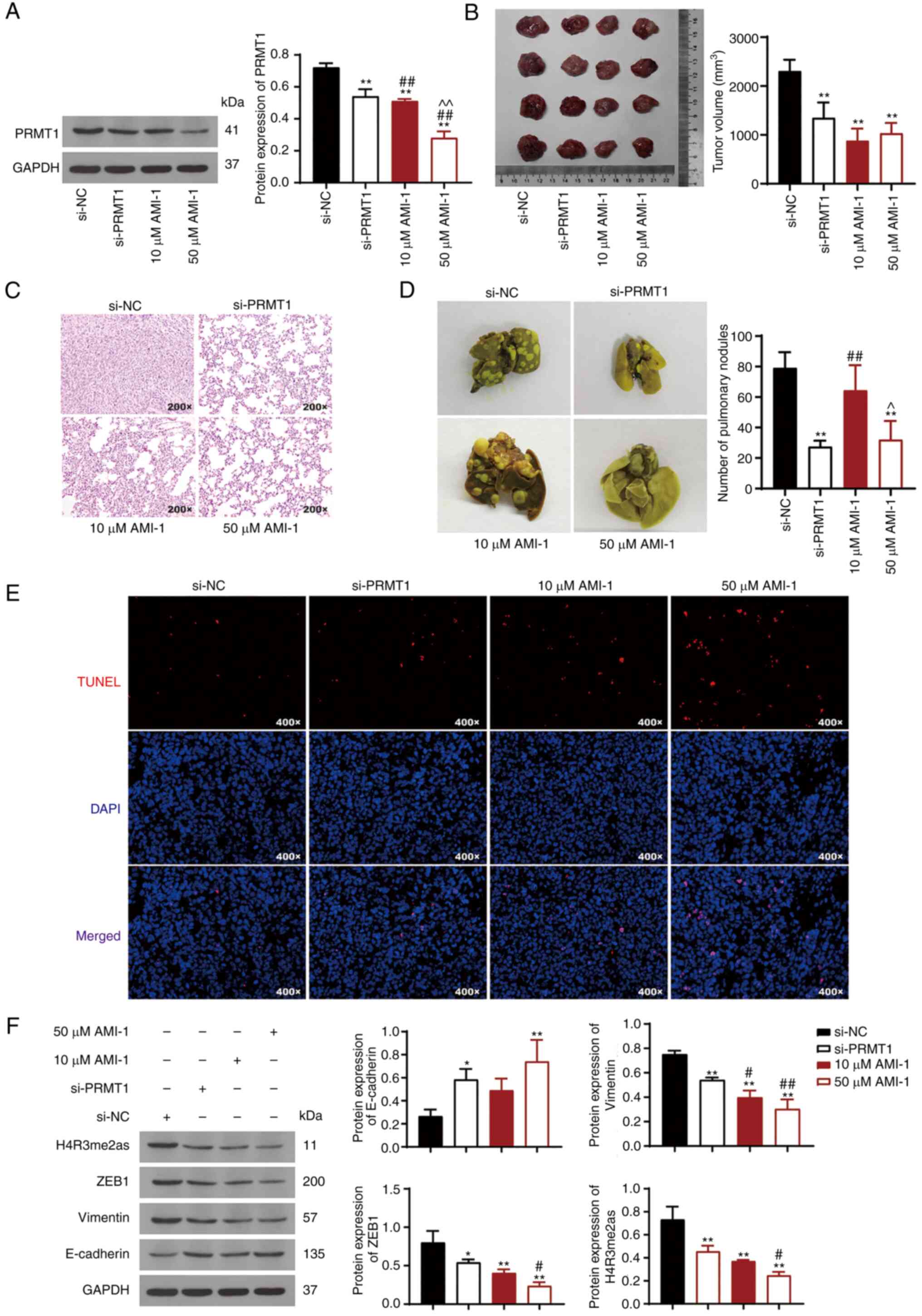 | Figure 6.PRMT1 knockdown reduces tumor growth
and metastasis by inhibiting ZEB1/H4R3me2as in a xenograft model.
(A) PRMT1 protein expression in TC tissues (B) Excised tumors from
the mouse model of TC. (C) H&E staining of TC tissues.
Magnification, ×200. (D) Image of pulmonary nodules and number of
pulmonary nodules. (E) TUNEL assays were used to assess apoptosis
in xenograft model mice. Magnification, ×400. (F) Western blotting
was to measure the protein expression levels of E-cadherin,
vimentin, H4R3me2as, and ZEB1 in TC tissues. BALB/C nude mice were
subcutaneously injected with treated 8505C cells. *P<0.05,
**P<0.01 vs. si-NC; #P<0.05,
##P<0.01 vs. si-PRMT1; ^P<0.05;
^^P<0.01 vs. 10 µM AMI-1. PRMT1, protein arginine
methyltransferase 1; ZEB1, zinc finger e-box binding homeobox1;
H4R3me2as, asymmetric demethylation of H4 at the third arginine
residue; TC, thyroid carcinoma; si-NC, small interfering RNA
negative control. |
Discussion
TC is the most prevalent endocrine neoplasm
(1). PRMT1, which catalyzes the
protein methylation of arginine residues, plays a crucial role in
tumorigenesis (25). In the present
study, PRMT1 expression was increased in TC, and its downregulation
inhibited cell proliferation and migration, as well as tumor growth
and metastasis. Furthermore, the expression levels of ZEB1 and
H4R3me2as were determined to be associated with those of PRMT1,
suggesting a novel PRMT1/ZEB1/H4R3me2as axis that is involved in
the malignant progression of TC.
PRMT1 is the primary type I PRMT, and accounts for
85% of the activity of type I PRMTs in mammals. PRMT1 participates
in various cellular processes due to the diversity of its histone
and non-histone substrates (26).
Previous studies have implicated PRMT1 in various cancers, as it
regulates post-translational modifications that are crucial for
cancer pathophysiology (27,28).
PRMT1 expression can be used to predict the outcomes of neoadjuvant
treatments for locally advanced uterine cervical cancer (29). Li et al (30) reported that PRMT1 promoted breast
cancer cell proliferation and tumorigenesis. In the present study,
PRMT1 upregulation was observed in a TC cell line. Subsequent
experiments revealed that PRMT1 knockdown suppressed TC cell
proliferation and migration, whereas its overexpression accelerated
these processes. In addition, PRMT1 delayed TC tumor growth and
metastasis in a xenograft tumor mouse model. These results
indicated that PRMT1 was associated with TC tumorigenesis.
EMT is a morphogenetic reversible process related to
tumor initiation, invasion, metastasis, and resistance to therapy
(31). EMT is orchestrated by a
collection of transcription factors, including ZEB1, ZEB2, and zinc
finger proteins (32). E-cadherin
and vimentin are important mesenchymal biomarkers (33). E-cadherin is a tumor suppressor
protein, and its downregulation in association with EMT frequently
occurs during tumor metastasis (34). Vimentin is a filamentous protein
that is confirmed to be upregulated during cancer metastasis
(35). In the present study,
E-cadherin expression was elevated, and vimentin expression was
reduced following PRMT1 knockdown. Furthermore, the metastasis of
TC cells to the lungs was observed, as evidenced by pulmonary
nodule formation in mice. However, PRMT1 downregulation inhibited
this pulmonary nodule formation. Collectively, these results
revealed that PRMT1 participates in TC tumor progression and that
its knockdown reduces metastasis.
H4R3me2as, a specific substrate of PRMT1, is a
transcriptional activation marker (26). A previous study reported that the
methylation of H4R3 by PRMT1 is necessary for the activation of its
expression (36). PRMT1 regulates
tumor-initiating properties through H4 arginine methylation in
esophageal squamous cell carcinoma (37). The results of the present study
revealed that H4R3me2as expression was regulated by PRMT1 and was
related to TC progression. Previous studies demonstrated that PRMT1
participates in pancreatic cancer progression by targeting ZEB1
(21). ZEB1 is an EMT-related
transcription factor (38).
Abnormal ZEB1 expression has been confirmed in multiple types of
cancer, including breast (39),
colorectal (40), hepatocellular
(41), and ovarian cancer (42). Morillo-Bernal et al (43) demonstrated that ZEB1 is a target of
FOXE1 in regulating TC progression. In the present study, ZEB1 was
upregulated in TC and tumor tissues. PRMT1 has been reported to
function as an upstream mediator of the ZEB family of proteins
(44). This upregulation also
promotes EMT (45). These results
also revealed that PRMT1 knockdown reduced ZEB1 expression in TC
and tumor tissues. Based on these data, it is hypothesized that
PRMT1 knockdown may suppress TC progression by downregulating
ZEB1/H4R3me2as.
In conclusion, the present study showed that PRMT1
was upregulated in both TC and tumor-bearing mice. PRMT1 knockdown
inhibited cell proliferation and migration and promoted apoptosis
in TC. Importantly, it was shown that PRMT1 participated in TC
progression by upregulating ZEB1 and H4R3me2as expression levels.
These results provide critical insights into PRMT1-driven TC
pathophysiology and treatment as well as identifying novel
potential therapeutic targets and diagnostic biomarkers for the
management of TC. However, further studies are required to confirm
whether PRMT1 can influence the phenotypes of normal cells and
uncover its clinical applicability. In addition, studies are
required to verify the role of PRMT1 in the regulating of ZEB1
expression in TC progression.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
GF conceived the study, collected the data, analyzed
the data, and wrote and revised the manuscript. CC and YL designed
the study and collected the data.
Ethics approval and consent to
participate
All animal experiments were approved by the
Institutional Animal Care and Use Committee of the Affiliated
Hospital of the Zunyi Medical University (approval no.
zyfy-an-2023-0185).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
TC
|
thyroid carcinoma
|
|
PRMT1
|
protein arginine methyltransferase
1
|
|
ZEB1
|
zinc-finger E homeobox-binding 1
|
|
EMT
|
epithelial-mesenchymal transition
|
References
|
1
|
Kleinschmidt-DeMasters B and Marshall C:
Thyroid carcinoma metastases to central nervous system and
vertebrae. Folia Neuropathol. 60:292–300. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pan Z, Xu T, Bao L, Hu X, Jin T, Chen J,
Chen J, Qian Y, Lu X, Li L, et al: CREB3L1 promotes tumor growth
and metastasis of anaplastic thyroid carcinoma by remodeling the
tumor microenvironment. Mol Cancer. 21:1902022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Deng Y, Li X, Jiang W and Tang J: SNRPB
promotes cell cycle progression in thyroid carcinoma via inhibiting
p53. Open Med (Wars). 17:1623–1631. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Franchini F, Palatucci G, Colao A, Ungaro
P, Macchia PE and Nettore IC: Obesity and thyroid cancer risk: An
update. Int J Environ Res Public Health. 19:11162022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pan Y, Wu L, He S, Wu J, Wang T and Zang
H: Identification of hub genes in thyroid carcinoma to predict
prognosis by integrated bioinformatics analysis. Bioengineered.
12:2928–2940. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sun R, Yang L, Wang Y, Zhang Y, Ke J and
Zhao D: DNAJB11 predicts a poor prognosis and is associated with
immune infiltration in thyroid carcinoma: A bioinformatics
analysis. J Int Med Res. 49:30006052110537222021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Geng X, Sun Y, Fu J, Cao L and Li Y:
MicroRNA-17-5p inhibits thyroid cancer progression by suppressing
Early growth response 2 (EGR2). Bioengineered. 12:2713–2722. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lv L, Wang X, Shen J, Cao Y and Zhang Q:
MiR-574-3p inhibits glucose toxicity-induced pancreatic β-cell
dysfunction by suppressing PRMT1. Diabetol Metab Syndr. 14:992022.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang YC, Wang CW, Lin WC, Tsai YJ, Chang
CP, Lee YJ, Lin MJ and Li C: Identification, chromosomal
arrangements and expression analyses of the evolutionarily
conserved prmt1 gene in chicken in comparison with its vertebrate
paralogue prmt8. PLoS One. 12:e01850422017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yao B, Gui T, Zeng X, Deng Y, Wang Z, Wang
Y, Yang D, Li Q, Xu P, Hu R, et al: PRMT1-mediated H4R3me2a
recruits SMARCA4 to promote colorectal cancer progression by
enhancing EGFR signaling. Genome Med. 13:582021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim E, Jang J, Park JG, Kim KH, Yoon K,
Yoo BC and Cho JY: Protein Arginine methyltransferase 1 (PRMT1)
selective inhibitor, TC-E 5003, has anti-inflammatory properties in
TLR4 Signaling. Int J Mol Sci. 21:30582020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Matsubara H, Fukuda T, Awazu Y, Nanno S,
Shimomura M, Inoue Y, Yamauchi M, Yasui T and Sumi T: PRMT1
expression predicts sensitivity to platinum-based chemotherapy in
patients with ovarian serous carcinoma. Oncol Lett. 21:1622021.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang C, Dong L, Zhao Z, Zhang Z, Sun Y, Li
C, Li G, You X, Yang X, Wang H and Hong W: Design and Synthesis of
Novel PRMT1 inhibitors and investigation of their effects on the
migration of cancer cell. Front Chem. 10:8887272022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gu X, He M, Lebedev T, Lin CH, Hua ZY,
Zheng YG, Li ZJ, Yang JY and Li XG: PRMT1 is an important factor
for medulloblastoma cell proliferation and survival. Biochem
Biophys Rep. 32:1013642022.PubMed/NCBI
|
|
15
|
Vezzalini M, Aletta JM, Beghelli S,
Moratti E, Della Peruta M, Mafficini A, Mojica WD, Mombello A,
Scarpa A and Sorio C: Immunohistochemical detection of arginine
methylated proteins (MeRP) in archival tissues. Histopathology.
57:725–733. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang Y, Liu X, Liang W, Dean DC, Zhang L
and Liu Y: Expression and Function of ZEB1 in the Cornea. Cells.
10:9252021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang X, Lai Q, He J, Li Q, Ding J, Lan Z,
Gu C, Yan Q, Fang Y, Zhao X and Liu S: LncRNA SNHG6 promotes
proliferation, invasion and migration in colorectal cancer cells by
activating TGF-β/Smad signaling pathway via targeting UPF1 and
inducing EMT via regulation of ZEB1. Int J Med Sci. 16:51–59. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chen XJ, Deng YR, Wang ZC, Wei WF, Zhou
CF, Zhang YM, Yan RM, Liang LJ, Zhong M, Liang L, et al:
Hypoxia-induced ZEB1 promotes cervical cancer progression via
CCL8-dependent tumour-associated macrophage recruitment. Cell Death
Dis. 10:5082019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shen H, Zhu H, Chen Y, Shen Z, Qiu W, Qian
C and Zhang J: ZEB1-induced LINC01559 expedites cell proliferation,
migration and EMT process in gastric cancer through recruiting
IGF2BP2 to stabilize ZEB1 expression. Cell Death Dis. 12:3492021.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Filipović J, Bosić M, Ćirović S, Životić
M, Dunđerović D, Đorđević D, Živković-Perišić S, Lipkovski A and
Marković-Lipkovski J: PRMT1 expression in renal cell
tumors-application in differential diagnosis and prognostic
relevance. Diagn Pathol. 14:1202019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lin Z, Chen Y, Lin Z, Chen C and Dong Y:
Overexpressing PRMT1 inhibits proliferation and invasion in
pancreatic cancer by inverse correlation of ZEB1. IUBMB Life.
70:1032–1039. 2018. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Saiselet M, Floor S, Tarabichi M, Dom G,
Hébrant A, van Staveren WC and Maenhaut C: Thyroid cancer cell
lines: An overview. Front Endocrinol (Lausanne). 3:1332012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pozdeyev N, Berlinberg A, Zhou Q, Wuensch
K, Shibata H, Wood WM and Haugen BR: Targeting the NF-κB pathway as
a combination therapy for advanced thyroid cancer. PLoS One.
10:e01349012015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sung BY, Lin YH, Kong Q, Shah PD, Glick
Bieler J, Palmer S, Weinhold KJ, Chang HR, Huang H, Avery RK, et
al: Wnt activation promotes memory T cell polyfunctionality via
epigenetic regulator PRMT1. J Clin Invest. 132:e1405082022.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Thiebaut C, Eve L, Poulard C and Le
Romancer M: Structure, activity, and function of PRMT1. Life
(Basel). 11:11472021.PubMed/NCBI
|
|
27
|
Yin XK, Wang YL, Wang F, Feng WX, Bai SM,
Zhao WW, Feng LL, Wei MB, Qin CL, Wang F, et al: PRMT1 enhances
oncogenic arginine methylation of NONO in colorectal cancer.
Oncogene. 40:1375–1389. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Giuliani V, Miller MA, Liu CY, Hartono SR,
Class CA, Bristow CA, Suzuki E, Sanz LA, Gao G, Gay JP, et al:
PRMT1-dependent regulation of RNA metabolism and DNA damage
response sustains pancreatic ductal adenocarcinoma. Nat Commun.
12:46262021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shimomura M, Fukuda T, Awazu Y, Nanno S,
Inoue Y, Matsubara H, Yamauchi M, Yasui T and Sumi T: PRMT1
expression predicts response to neoadjuvant chemotherapy for
locally advanced uterine cervical cancer. Oncol Lett. 21:1502021.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li Z, Wang D, Chen X, Wang W, Wang P, Hou
P, Li M, Chu S, Qiao S, Zheng J and Bai J: PRMT1-mediated EZH2
methylation promotes breast cancer cell proliferation and
tumorigenesis. Cell Death Dis. 12:10802021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pastushenko I and Blanpain C: EMT
transition states during tumor progression and metastasis. Trends
Cell Biol. 29:212–226. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pan G, Liu Y, Shang L, Zhou F and Yang S:
EMT-associated microRNAs and their roles in cancer stemness and
drug resistance. Cancer Commun (Lond). 41:199–217. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Huang X, Xiang L, Wang B, Hu J, Liu C, Ren
A, Du K, Ye G, Liang Y, Tang Y, et al: CMTM6 promotes migration,
invasion, and EMT by interacting with and stabilizing vimentin in
hepatocellular carcinoma cells. J Transl Med. 19:1202021.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Na TY, Schecterson L, Mendonsa AM and
Gumbiner BM: The functional activity of E-cadherin controls tumor
cell metastasis at multiple steps. Proc Natl Acad Sci USA.
117:5931–5937. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Usman S, Waseem NH, Nguyen TKN, Mohsin S,
Jamal A, The MT and Waseem A: Vimentin is at the heart of
epithelial mesenchymal transition (EMT) mediated metastasis.
Cancers (Basel). 13:49852021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wagner S, Weber S, Kleinschmidt MA, Nagata
K and Bauer UM: SET-mediated promoter hypoacetylation is a
prerequisite for coactivation of the estrogen-responsive pS2 gene
by PRMT1. J Biol Chem. 281:27242–27250. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhao Y, Lu Q, Li C, Wang X, Jiang L, Huang
L, Wang C and Chen H: PRMT1 regulates the tumour-initiating
properties of esophageal squamous cell carcinoma through histone H4
arginine methylation coupled with transcriptional activation. Cell
Death Dis. 10:3592019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ramundo V, Zanirato G and Aldieri E: The
epithelial-to-mesenchymal transition (EMT) in the development and
metastasis of malignant pleural mesothelioma. Int J Mol Sci.
22:122162021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wu HT, Zhong HT, Li GW, Shen JX, Ye QQ,
Zhang ML and Liu J: Oncogenic functions of the EMT-related
transcription factor ZEB1 in breast cancer. J Transl Med.
18:512020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jin Z and Chen B: LncRNA ZEB1-AS1
regulates colorectal cancer cells by MiR-205/YAP1 Axis. Open Med
(Wars). 15:175–184. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Liu W, Zheng L, Zhang R, Hou P, Wang J, Wu
L and Li J: Circ-ZEB1 promotes PIK3CA expression by silencing
miR-199a-3p and affects the proliferation and apoptosis of
hepatocellular carcinoma. Mol Cancer. 21:722022. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang J, Guan W, Xu X, Wang F, Li X and Xu
G: A novel homeostatic loop of sorcin drives paclitaxel-resistance
and malignant progression via Smad4/ZEB1/miR-142-5p in human
ovarian cancer. Oncogene. 40:4906–4918. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Morillo-Bernal J, Fernández LP and
Santisteban P: FOXE1 regulates migration and invasion in thyroid
cancer cells and targets ZEB1. Endocr Relat Cancer. 27:137–151.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Soleymani L, Zarrabi A, Hashemi F, Hashemi
F, Zabolian A, Banihashemi SM, Moghadam SS, Hushmandi K,
Samarghandian S, Ashrafizadeh M and Khan H: Role of ZEB Family
Members in Proliferation, Metastasis, and Chemoresistance of
Prostate Cancer Cells: Revealing Signaling Networks. Curr Cancer
Drug Targets. 21:749–767. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Iderzorig T, Kellen J, Osude C, Singh S,
Woodman JA, Garcia C and Puri N: Comparison of EMT mediated
tyrosine kinase inhibitor resistance in NSCLC. Biochem Biophys Res
Commun. 496:770–777. 2018. View Article : Google Scholar : PubMed/NCBI
|