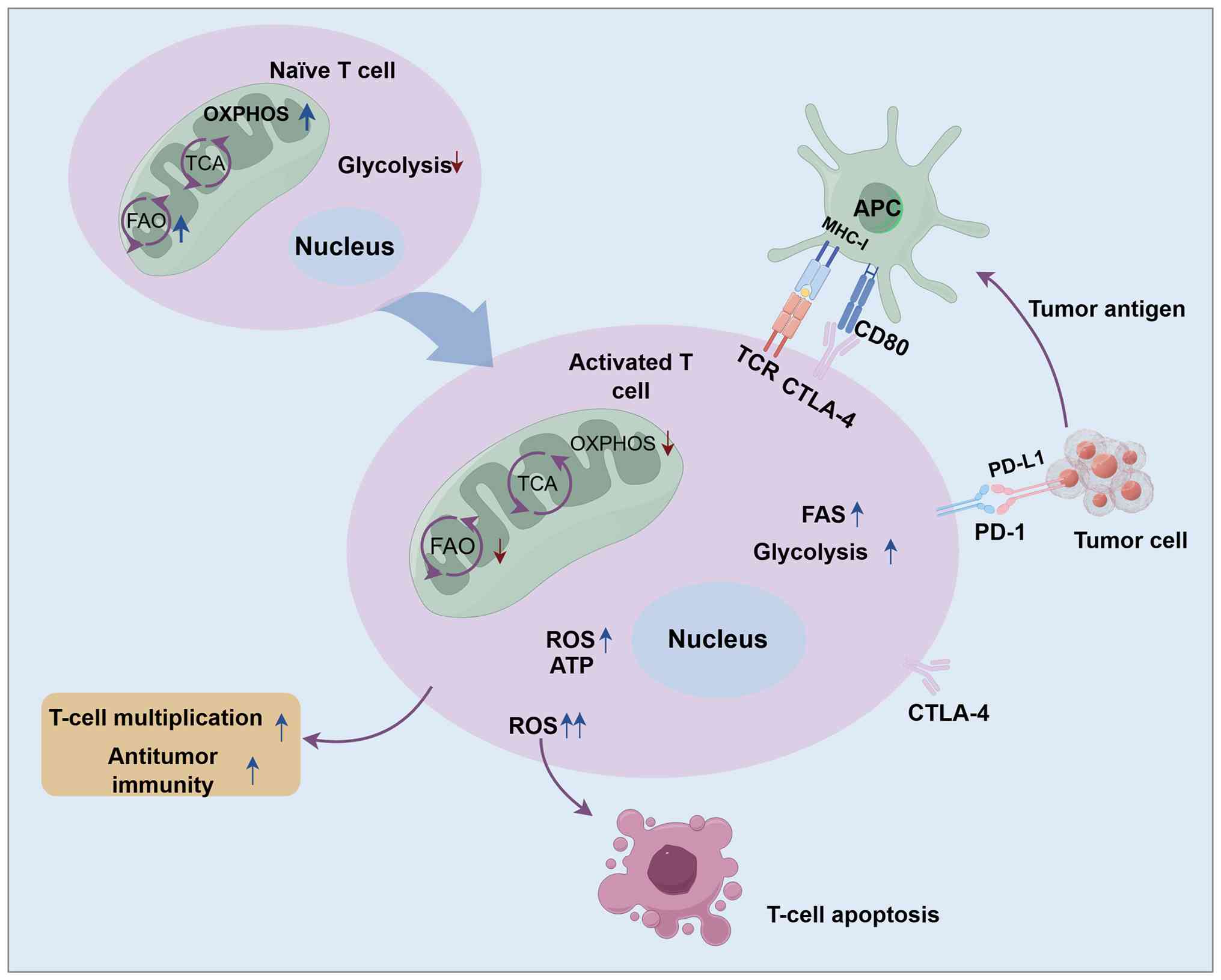
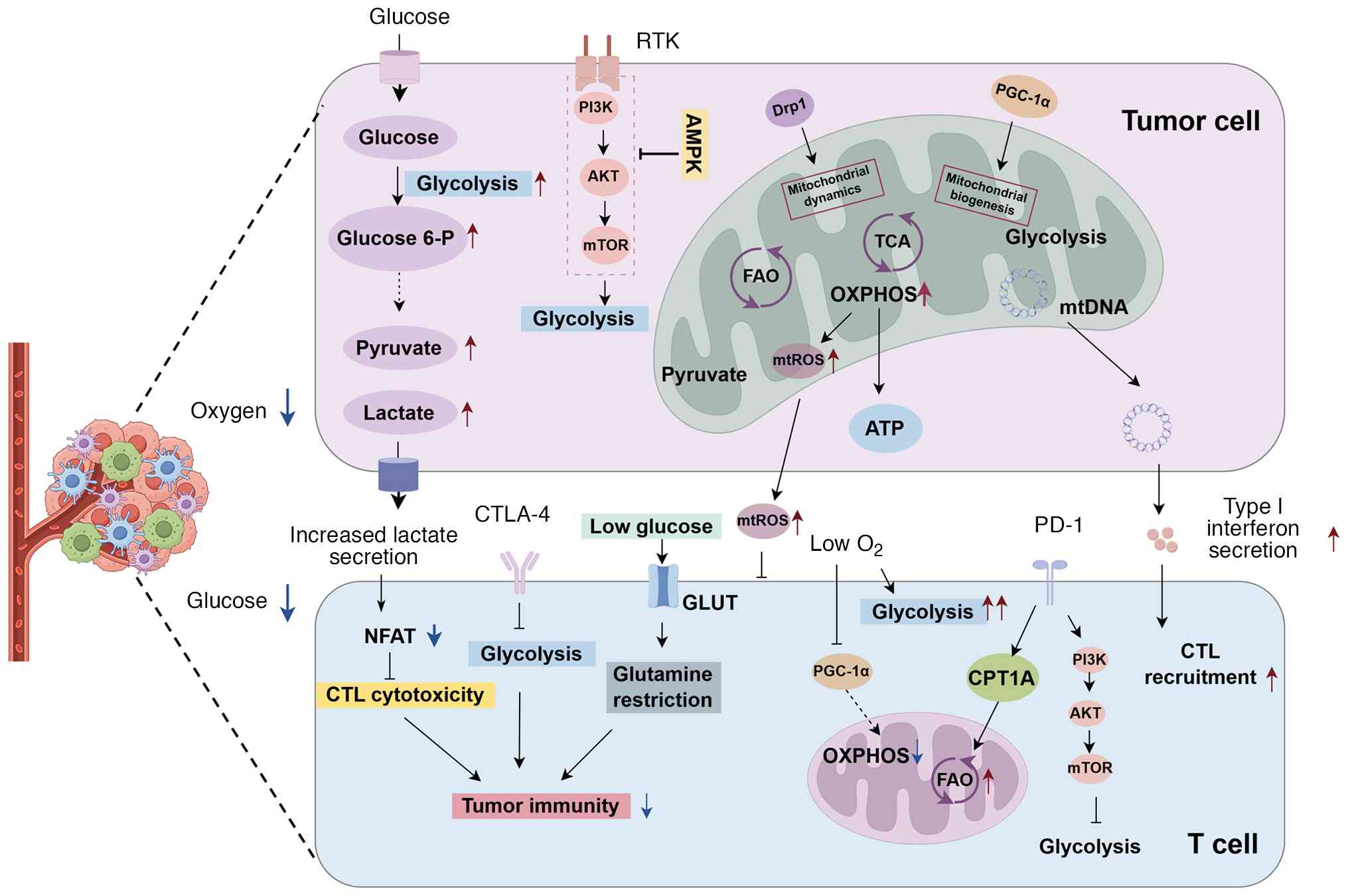
|
1
|
Bereiter-Hahn J: Behavior of mitochondria
in the living cell. Int Rev Cytol. 122:1–63. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chan DC: Mitochondrial dynamics and its
involvement in disease. Annu Rev Pathol. 15:235–259. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mills EL, Kelly B and O'Neill LAJ:
Mitochondria are the powerhouses of immunity. Nat Immunol.
18:488–498. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chandel NS: Mitochondria. Cold Spring
Harbor Perspect Biol. 13:a0405432021. View Article : Google Scholar
|
|
5
|
Seo JH, Agarwal E, Bryant KG, Caino MC,
Kim ET, Kossenkov AV, Tang HY, Languino LR, Gabrilovich DI, Cohen
AR, et al: Syntaphilin ubiquitination regulates mitochondrial
dynamics and tumor cell movements. Cancer Res. 78:4215–4228. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wellen KE and Thompson CB: A two-way
street: Reciprocal regulation of metabolism and signalling. Nat Rev
Mol Cell Biol. 13:270–276. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chan DC: Mitochondrial fusion and fission
in mammals. Annu Rev Cell Dev Biol. 22:79–99. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ikeda H, Kawase K, Nishi T, Watanabe T,
Takenaga K, Inozume T, Ishino T, Aki S, Lin J, Kawashima S, et al:
Immune evasion through mitochondrial transfer in the tumour
microenvironment. Nature. 638:225–236. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Suzuki J, Yamada T, Inoue K, Nabe S,
Kuwahara M, Takemori N, Takemori A, Matsuda S, Kanoh M, Imai Y, et
al: The tumor suppressor menin prevents effector CD8 T-cell
dysfunction by targeting mTORC1-dependent metabolic activation. Nat
Commun. 9:32962018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Harrington JS, Ryter SW, Plataki M, Price
DR and Choi AMK: Mitochondria in health, disease, and aging.
Physiol Rev. 103:2349–2422. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
El Hiani Y, Ahidouch A, Lehen'kyi V, Hague
F, Gouilleux F, Mentaverri R, Kamel S, Lassoued K, Brûlé G and
Ouadid-Ahidouch H: Extracellular signal-regulated kinases 1 and 2
and TRPC1 channels are required for calcium-sensing
receptor-stimulated MCF-7 breast cancer cell proliferation. Cell
Physiol Biochem. 23:335–346. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
DeBerardinis RJ and Chandel NS:
Fundamentals of cancer metabolism. Sci Adv. 2:e16002002016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu BH, Xu CZ, Liu Y, Lu ZL, Fu TL, Li GR,
Deng Y, Luo GQ, Ding S, Li N and Geng Q: Mitochondrial quality
control in human health and disease. Mil Med Res.
11:322024.PubMed/NCBI
|
|
14
|
Vodnala SK, Eil R, Kishton RJ, Sukumar M,
Yamamoto TN, Ha NH, Lee PH, Shin M, Patel SJ, Yu Z, et al: T cell
stemness and dysfunction in tumors are triggered by a common
mechanism. Science. 363:eaau01352019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lyssiotis CA and Kimmelman AC: Metabolic
interactions in the tumor microenvironment. Trends Cell Biol.
27:863–875. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Scharping NE, Rivadeneira DB, Menk AV,
Vignali PDA, Ford BR, Rittenhouse NL, Peralta R, Wang Y, Wang Y,
DePeaux K, et al: Mitochondrial stress induced by continuous
stimulation under hypoxia rapidly drives T cell exhaustion. Nat
Immunol. 22:205–215. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yu YR, Imrichova H, Wang H, Chao T, Xiao
Z, Gao M, Rincon-Restrepo M, Franco F, Genolet R, Cheng WC, et al:
Disturbed mitochondrial dynamics in CD8+ TILs reinforce
T cell exhaustion. Nat Immunol. 21:1540–1551. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Vardhana SA, Hwee MA, Berisa M, Wells DK,
Yost KE, King B, Smith M, Herrera PS, Chang HY, Satpathy AT, et al:
Impaired mitochondrial oxidative phosphorylation limits the
self-renewal of T cells exposed to persistent antigen. Nat Immunol.
21:1022–1033. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yang MQ, Zhang SL, Sun L, Huang LT, Yu J,
Zhang JH, Tian Y, Han CB and Ma JT: Targeting mitochondria:
Restoring the antitumor efficacy of exhausted T cells. Mol Cancer.
23:2602024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
König T, Nolte H, Aaltonen MJ, Tatsuta T,
Krols M, Stroh T, Langer T and McBride HM: MIROs and DRP1 drive
mitochondrial-derived vesicle biogenesis and promote quality
control. Nat Cell Biol. 23:1271–1286. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mangalhara KC, Varanasi SK, Johnson MA,
Burns MJ, Rojas GR, Esparza Moltó PB, Sainz AG, Tadepalle N, Abbott
KL, Mendiratta G, et al: Manipulating mitochondrial electron flow
enhances tumor immunogenicity. Science. 381:1316–1323. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sullivan LB, Gui DY and Vander Heiden MG:
Altered metabolite levels in cancer: Implications for tumour
biology and cancer therapy. Nat Rev Cancer. 16:680–693. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
De Martino M, Rathmell JC, Galluzzi L and
Vanpouille-Box C: Cancer cell metabolism and antitumour immunity.
Nat Rev Immunol. 24:654–669. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tian W, Liu Y, Cao C, Zeng Y, Pan Y, Liu
X, Peng Y and Wu F: Chronic stress: Impacts on tumor
microenvironment and implications for anti-cancer treatments. Front
Cell Dev Biol. 9:7770182021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lei Y, Liao F, Tian Y, Wang Y, Xia F and
Wang J: Investigating the crosstalk between chronic stress and
immune cells: Implications for enhanced cancer therapy. Front
Neurosci. 17:13211762023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu Y, Tian S, Ning B, Huang T, Li Y and
Wei Y: Stress and cancer: The mechanisms of immune dysregulation
and management. Front Immunol. 13:10322942022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kawano I, Bazila B, Ježek P and Dlasková
A: Mitochondrial dynamics and cristae shape changes during
metabolic reprogramming. Antioxid Redox Signaling. 39:684–707.
2023. View Article : Google Scholar
|
|
28
|
Xu X, Araki K, Li S, Han JH, Ye L, Tan WG,
Konieczny BT, Bruinsma MW, Martinez J, Pearce EL, et al: Autophagy
is essential for effector CD8(+) T cell survival and memory
formation. Nat Immunol. 15:1152–1161. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wu H, Zhao X, Hochrein SM, Eckstein M,
Gubert GF, Knöpper K, Mansilla AM, Öner A, Doucet-Ladevèze R,
Schmitz W, et al: Mitochondrial dysfunction promotes the transition
of precursor to terminally exhausted T cells through
HIF-1α-mediated glycolytic reprogramming. Nat Commun. 14:68582023.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chang CH, Curtis JD, Maggi LB Jr, Faubert
B, Villarino AV, O'Sullivan D, Huang SC, van der Windt GJ, Blagih
J, Qiu J, et al: Posttranscriptional control of T cell effector
function by aerobic glycolysis. Cell. 153:1239–1251. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ron-Harel N, Santos D, Ghergurovich JM,
Sage PT, Reddy A, Lovitch SB, Dephoure N, Satterstrom FK, Sheffer
M, Spinelli JB, et al: Mitochondrial biogenesis and proteome
remodeling promote one-carbon metabolism for T cell activation.
Cell Metab. 24:104–117. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Guerrero JA, Klysz DD, Chen Y,
Malipatlolla M, Lone J, Fowler C, Stuani L, May A, Bashti M, Xu P,
et al: GLUT1 overexpression in CAR-T cells induces metabolic
reprogramming and enhances potency. Nat Commun. 15:86582024.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ramstead AG, Wallace JA, Lee SH, Bauer KM,
Tang WW, Ekiz HA, Lane TE, Cluntun AA, Bettini ML, Round JL, et al:
Mitochondrial pyruvate carrier 1 promotes peripheral T cell
homeostasis through metabolic regulation of thymic development.
Cell Rep. 30:2889–2899.e6. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Christie DA, Kirchhof MG, Vardhana S,
Dustin ML and Madrenas J: Mitochondrial and plasma membrane pools
of stomatin-like protein 2 coalesce at the immunological synapse
during T cell activation. PLoS One. 7:e371442012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Fan X, Yang M, Lang Y, Lu S, Kong Z, Gao
Y, Shen N, Zhang D and Lv Z: Mitochondrial metabolic reprogramming
in diabetic kidney disease. Cell Death Dis. 15:4422024. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
MacIver NJ, Michalek RD and Rathmell JC:
Metabolic regulation of T lymphocytes. Annu Rev Immunol.
31:259–283. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chapman NM and Chi H: Hallmarks of T-cell
exit from quiescence. Cancer Immunol Res. 6:502–508. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mescher MF, Curtsinger JM, Agarwal P,
Casey KA, Gerner M, Hammerbeck CD, Popescu F and Xiao Z: Signals
required for programming effector and memory development by CD8+ T
cells. Immunol Rev. 211:81–92. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Makowski L, Chaib M and Rathmell JC:
Immunometabolism: From basic mechanisms to translation. Immunol
Rev. 295:5–14. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang R, Dillon CP, Shi LZ, Milasta S,
Carter R, Finkelstein D, McCormick LL, Fitzgerald P, Chi H, Munger
J and Green DR: The transcription factor Myc controls metabolic
reprogramming upon T lymphocyte activation. Immunity. 35:871–882.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Vaeth M, Maus M, Klein-Hessling S,
Freinkman E, Yang J, Eckstein M, Cameron S, Turvey SE, Serfling E,
Berberich-Siebelt F, et al: Store-operated Ca2+ entry
controls clonal expansion of T cells through metabolic
reprogramming. Immunity. 47:664–679.e6. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Stirnweiss A, Hartig R, Gieseler S,
Lindquist JA, Reichardt P, Philipsen L, Simeoni L, Poltorak M,
Merten C, Zuschratter W, et al: T cell activation results in
conformational changes in the Src family kinase Lck to induce its
activation. Sci Signaling. 6:ra132013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Schwindling C, Quintana A, Krause E and
Hoth M: Mitochondria positioning controls local calcium influx in T
cells. J Immunol. 184:184–190. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Velnati S, Massarotti A, Antona A, Talmon
M, Fresu LG, Galetto AS, Capello D, Bertoni A, Mercalli V, Graziani
A, et al: Structure activity relationship studies on Amb639752:
toward the identification of a common pharmacophoric structure for
DGKα inhibitors. J Enzyme Inhib Med Chem. 35:96–108. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Araki K, Turner AP, Shaffer VO, Gangappa
S, Keller SA, Bachmann MF, Larsen CP and Ahmed R: mTOR regulates
memory CD8 T-cell differentiation. Nature. 460:108–112. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Klemm F, Maas RR, Bowman RL, Kornete M,
Soukup K, Nassiri S, Brouland JP, Iacobuzio-Donahue CA, Brennan C,
Tabar V, et al: Interrogation of the microenvironmental landscape
in brain tumors reveals disease-specific alterations of immune
cells. Cell. 181:1643–1660.e17. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Cabrita R, Lauss M, Sanna A, Donia M,
Skaarup Larsen M, Mitra S, Johansson I, Phung B, Harbst K,
Vallon-Christersson J, et al: Tertiary lymphoid structures improve
immunotherapy and survival in melanoma. Nature. 577:561–565. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Connolly KA, Kuchroo M, Venkat A, Khatun
A, Wang J, William I, Hornick NI, Fitzgerald BL, Damo M, Kasmani
MY, et al: A reservoir of stem-like CD8+ T cells in the
tumor-draining lymph node preserves the ongoing antitumor immune
response. Sci Immunol. 6:eabg78362021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Prokhnevska N, Cardenas MA, Valanparambil
RM, Sobierajska E, Barwick BG, Jansen C, Reyes Moon A, Gregorova P,
delBalzo L, Greenwald R, et al: CD8+ T cell activation
in cancer comprises an initial activation phase in lymph nodes
followed by effector differentiation within the tumor. Immunity.
56:107–124.e5. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Nagasaki J, Inozume T, Sax N, Ariyasu R,
Ishikawa M, Yamashita K, Kawazu M, Ueno T, Irie T, Tanji E, et al:
PD-1 blockade therapy promotes infiltration of tumor-attacking
exhausted T cell clonotypes. Cell Rep. 38:1103312022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Pai JA, Hellmann MD, Sauter JL, Mattar M,
Rizvi H, Woo HJ, Shah N, Nguyen EM, Uddin FZ, Quintanal-Villalonga
A, et al: Lineage tracing reveals clonal progenitors and long-term
persistence of tumor-specific T cells during immune checkpoint
blockade. Cancer Cell. 41:776–790.e7. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Liang H, Huang J, Li H, He W, Ao X, Xie Z,
Chen Y, Lv Z, Zhang L, Zhong Y, et al: Spatial proximity of
CD8+ T cells to tumor cells predicts neoadjuvant therapy
efficacy in breast cancer. NPJ Breast Cancer. 11:132025. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Galon J, Costes A, Sanchez-Cabo F,
Kirilovsky A, Mlecnik B, Lagorce-Pagès C, Tosolini M, Camus M,
Berger A, Wind P, et al: Type, density, and location of immune
cells within human colorectal tumors predict clinical outcome.
Science. 313:1960–1964. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Pagès F, Mlecnik B, Marliot F, Bindea G,
Ou FS, Bifulco C, Lugli A, Zlobec I, Rau TT, Berger MD, et al:
International validation of the consensus Immunoscore for the
classification of colon cancer: A prognostic and accuracy study.
Lancet. 391:2128–2139. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Angell HK, Bruni D, Barrett JC, Herbst R
and Galon J: The immunoscore: Colon cancer and beyond. Clin Cancer
Res. 26:332–339. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Sen DR, Kaminski J, Barnitz RA, Kurachi M,
Gerdemann U, Yates KB, Tsao HW, Godec J, LaFleur MW, Brown FD, et
al: The epigenetic landscape of T cell exhaustion. Science.
354:1165–1169. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Reina-Campos M, Scharping NE and Goldrath
AW: CD8+ T cell metabolism in infection and cancer. Nat
Rev Immunol. 21:718–738. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zhu J and Thompson CB: Metabolic
regulation of cell growth and proliferation. Nat Rev Mol Cell Biol.
20:436–450. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Brand A, Singer K, Koehl GE, Kolitzus M,
Schoenhammer G, Thiel A, Matos C, Bruss C, Klobuch S, Peter K, et
al: LDHA-associated lactic acid production blunts tumor
immunosurveillance by T and NK cells. Cell Metab. 24:657–671. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Vigano S, Alatzoglou D, Irving M,
Ménétrier-Caux C, Caux C, Romero P and Coukos G: Targeting
adenosine in cancer immunotherapy to enhance T-cell function. Front
Immunol. 10:9252019. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Qin M, Hamanishi J, Ukita M, Yamanoi K,
Takamatsu S, Abiko K, Murakami R, Miyamoto T, Suzuki H, Ueda A, et
al: Tertiary lymphoid structures are associated with favorable
survival outcomes in patients with endometrial cancer. Cancer
Immunol Immunother. 71:1431–1442. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Zhang MJ, Wen Y and Sun ZJ: The impact of
metabolic reprogramming on tertiary lymphoid structure formation:
Enhancing cancer immunotherapy. BMC Med. 23:2172025. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Jia D, Wang Q, Qi Y, Jiang Y, He J, Lin Y,
Sun Y, Xu J, Chen W, Fan L, et al: Microbial metabolite enhances
immunotherapy efficacy by modulating T cell stemness in pan-cancer.
Cell. 187:1651–1665.e21. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Guo Y, Xie YQ, Gao M, Zhao Y, Franco F,
Wenes M, Siddiqui I, Bevilacqua A, Wang H, Yang H, et al: Metabolic
reprogramming of terminally exhausted CD8+ T cells by
IL-10 enhances anti-tumor immunity. Nat Immunol. 22:746–756. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Sukumar M, Kishton RJ and Restifo NP:
Metabolic repro-graming of anti-tumor immunity. Curr Opin Immunol.
46:14–22. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Scharping NE, Menk AV, Moreci RS,
Whetstone RD, Dadey RE, Watkins SC, Ferris RL and Delgoffe GM: The
tumor microenvironment represses T cell mitochondrial biogenesis to
drive intratumoral T cell metabolic insufficiency and dysfunction.
Immunity. 45:374–388. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Dumauthioz N, Tschumi B, Wenes M, Marti B,
Wang H, Franco F, Li W, Lopez-Mejia IC, Fajas L, Ho PC, et al:
Enforced PGC-1α expression promotes CD8 T cell fitness, memory
formation and antitumor immunity. Cell Mol Immunol. 18:1761–1771.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Song M, Sandoval TA, Chae CS, Chopra S,
Tan C, Rutkowski MR, Raundhal M, Chaurio RA, Payne KK, Konrad C, et
al: IRE1α-XBP1 controls T cell function in ovarian cancer by
regulating mitochondrial activity. Nature. 562:423–428. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Cham CM, Driessens G, O'Keefe JP and
Gajewski TF: Glucose deprivation inhibits multiple key gene
expression events and effector functions in CD8+ T cells. Eur J
Immunol. 38:2438–2450. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Wenes M, Jaccard A, Wyss T,
Maldonado-Pérez N, Teoh ST, Lepez A, Renaud F, Franco F, Waridel P,
Yacoub Maroun C, et al: The mitochondrial pyruvate carrier
regulates memory T cell differentiation and antitumor function.
Cell Metab. 34:731–746.e9. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Nava Lauson CB, Tiberti S, Corsetto PA,
Conte F, Tyagi P, Machwirth M, Ebert S, Loffreda A, Scheller L,
Sheta D, et al: Linoleic acid potentiates CD8+ T cell
metabolic fitness and antitumor immunity. Cell Metab.
35:633–650.e9. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Tang Y, Chen Z, Zuo Q and Kang Y:
Regulation of CD8+ T cells by lipid metabolism in cancer
progression. Cell Mol Immunol. 21:1215–1230. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Pauken KE and Wherry EJ: Overcoming T cell
exhaustion in infection and cancer. Trends Immunol. 36:265–276.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Greenwald RJ, Freeman GJ and Sharpe AH:
The B7 family revisited. Annu Rev Immunol. 23:515–548. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Patsoukis N, Bardhan K, Chatterjee P, Sari
D, Liu B, Bell LN, Karoly ED, Freeman GJ, Petkova V, Seth P, et al:
PD-1 alters T-cell metabolic reprogramming by inhibiting glycolysis
and promoting lipolysis and fatty acid oxidation. Nat Commun.
6:66922015. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Boussiotis VA and Patsoukis N: Effects of
PD-1 signaling on immunometabolic reprogramming. Immunometabolism.
4:e2200072022. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Baldanzi G: Immune checkpoint receptors
signaling in T cells. Int J Mol Sci. 23:35292022. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Zhao M, Yuan H, Yang G, Wang Y, Bu Y,
Zhang H, Zhao L, Lv P, Yun H, Geng Y, et al: Tumour cell-expressed
PD-L1 reprograms lipid metabolism via EGFR/ITGB4/SREBP1c signalling
in liver cancer. JHEP Rep. 6:1010092024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Ma F, Liu X, Zhang Y, Tao Y, Zhao L,
Abusalamah H, Huffman C, Harbison RA, Puram SV, Wang Y and Peng G:
Tumor extracellular vesicle-derived PD-L1 promotes T cell
senescence through lipid metabolism reprogramming. Sci Transl Med.
17:eadm72692025. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Ping Y, Shan J, Qin H, Li F, Qu J, Guo R,
Han D, Jing W, Liu Y, Liu J, et al: PD-1 signaling limits
expression of phospholipid phosphatase 1 and promotes intratumoral
CD8+ T cell ferroptosis. Immunity. 57:2122–2139.e9.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Alsayyah C, Singh MK, Morcillo-Parra MA,
Cavellini L, Shai N, Schmitt C, Schuldiner M, Zalckvar E, Mallet A,
Belgareh-Touzé N, et al: Mitofusin-mediated contacts between
mitochondria and peroxisomes regulate mitochondrial fusion. PLoS
Biol. 22:e30026022024. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Sandoval H, Thiagarajan P, Dasgupta SK,
Schumacher A, Prchal JT, Chen M and Wang J: Essential role for Nix
in autophagic maturation of erythroid cells. Nature. 454:232–235.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Lu Y, Li Z, Zhang S, Zhang T, Liu Y and
Zhang L: Cellular mitophagy: Mechanism, roles in diseases and small
molecule pharmacological regulation. Theranostics. 13:736–766.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Wang S, Long H, Hou L, Feng B, Ma Z, Wu Y,
Zeng Y, Cai J, Zhang DW and Zhao G: The mitophagy pathway and its
implications in human diseases. Signal Transduction Targeted Ther.
8:3042023. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
White E, Lattime EC and Guo JY: Autophagy
regulates stress responses, metabolism, and anticancer immunity.
Trends Cancer. 7:778–789. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Silwal P, Kim JK, Kim YJ and Jo EK:
Mitochondrial reactive oxygen species: Double-edged weapon in host
defense and pathological inflammation during infection. Front
Immunol. 11:16492020. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Mougiakakos D, Johansson CC, Jitschin R,
Böttcher M and Kiessling R: Increased thioredoxin-1 production in
human naturally occurring regulatory T cells confers enhanced
tolerance to oxidative stress. Blood. 117:857–861. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Mougiakakos D, Johansson CC and Kiessling
R: Naturally occurring regulatory T cells show reduced sensitivity
toward oxidative stress-induced cell death. Blood. 113:3542–3545.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Seledtsov VI, Goncharov AG and Seledtsova
GV: Clinically feasible approaches to potentiating cancer
cell-based immunotherapies. Hum Vaccin Immunother. 11:851–869.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Weiskopf D, Schwanninger A, Weinberger B,
Almanzar G, Parson W, Buus S, Lindner H and Grubeck-Loebenstein B:
Oxidative stress can alter the antigenicity of immunodominant
peptides. J Leukocyte Biol. 87:165–172. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Cheung EC and Vousden KH: The role of ROS
in tumour development and progression. Nat Rev Cancer. 22:280–297.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Hildeman DA, Mitchell T, Teague TK, Henson
P, Day BJ, Kappler J and Marrack PC: Reactive oxygen species
regulate activation-induced T cell apoptosis. Immunity. 10:735–744.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Bailly C: Regulation of PD-L1 expression
on cancer cells with ROS-modulating drugs. Life Sci.
246:1174032020. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Kuo CL, Ponneri Babuharisankar A, Lin YC,
Lien HW, Lo YK, Chou HY, Tangeda V, Cheng LC, Cheng AN and Lee AY:
Mitochondrial oxidative stress in the tumor microenvironment and
cancer immunoescape: Foe or friend? J Biomed Sci. 29:742022.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Deng H, Yang W, Zhou Z, Tian R, Lin L, Ma
Y, Song J and Chen X: Targeted scavenging of extracellular ROS
relieves suppressive immunogenic cell death. Nat Commun.
11:49512020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Kurniawan H, Franchina DG, Guerra L,
Bonetti L, -Baguet LS, Grusdat M, Schlicker L, Hunewald O, Dostert
C, Merz MP, et al: Glutathione restricts serine metabolism to
preserve regulatory T cell function. Cell Metab. 31:920–936.e7.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Kyriakoudi S, Drousiotou A and Petrou PP:
When the balance tips: Dysregulation of mitochondrial dynamics as a
culprit in disease. Int J Mol Sci. 22:46172021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
de Brito OM and Scorrano L: Mitofusin 2
tethers endoplasmic reticulum to mitochondria. Nature. 456:605–610.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Buck MD, O'Sullivan D, Klein Geltink RI,
Curtis JD, Chang CH, Sanin DE, Qiu J, Kretz O, Braas D, van der
Windt GJ, et al: Mitochondrial dynamics controls T cell fate
through metabolic programming. Cell. 166:63–76. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Picca A, Mankowski RT, Burman JL, Donisi
L, Kim JS, Marzetti E and Leeuwenburgh C: Mitochondrial quality
control mechanisms as molecular targets in cardiac ageing. Nat Rev
Cardiol. 15:543–554. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Yang JF, Xing X, Luo L, Zhou XW, Feng JX,
Huang KB, Liu H, Jin S, Liu YN, Zhang SH, et al: Mitochondria-ER
contact mediated by MFN2-SERCA2 interaction supports
CD8+ T cell metabolic fitness and function in tumors.
Sci Immunol. 8:eabq24242023. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Tilokani L, Nagashima S, Paupe V and
Prudent J: Mitochondrial dynamics: Overview of molecular
mechanisms. Essays Biochem. 62:341–360. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Menk AV, Scharping NE, Rivadeneira DB,
Calderon MJ, Watson MJ, Dunstane D, Watkins SC and Delgoffe GM:
4-1BB costimulation induces T cell mitochondrial function and
biogenesis enabling cancer immunotherapeutic responses. J Exp Med.
215:1091–1100. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Hu C, Huang Y and Li L: Drp1-dependent
mitochondrial fission plays critical roles in physiological and
pathological progresses in mammals. Int J Mol Sci. 18:1442017.
View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Cereghetti GM, Stangherlin A, Martins De
Brito O, Chang CR, Blackstone C, Bernardi P and Scorrano L:
Dephosphorylation by calcineurin regulates translocation of Drp1 to
mitochondria. Proc Natl Acad Sci USA. 105:15803–15808. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Smith-Garvin JE, Koretzky GA and Jordan
MS: T cell activation. Annu Rev Immunol. 27:591–619. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Woehrle T, Ledderose C, Rink J, Slubowski
C and Junger WG: Autocrine stimulation of P2Y1 receptors is part of
the purinergic signaling mechanism that regulates T cell
activation. Purinergic Signalling. 15:127–137. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Ledderose C, Bromberger S, Slubowski CJ,
Sueyoshi K and Junger WG: Frontline science: P2Y11 receptors
support T cell activation by directing mitochondrial trafficking to
the immune synapse. J Leukocyte Biol. 109:497–508. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Quintana A, Schwindling C, Wenning AS,
Becherer U, Rettig J, Schwarz EC and Hoth M: T cell activation
requires mitochondrial translocation to the immunological synapse.
Proc Natl Acad Sci USA. 104:14418–14423. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Simula L, Pacella I, Colamatteo A,
Procaccini C, Cancila V, Bordi M, Tregnago C, Corrado M, Pigazzi M,
Barnaba V, et al: Drp1 controls effective T cell
immune-surveillance by regulating T cell migration, proliferation,
and cMyc-dependent metabolic reprogramming. Cell Rep.
25:3059–3073.e10. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Mohammadalipour A, Dumbali SP and Wenzel
PL: Mitochondrial transfer and regulators of mesenchymal stromal
cell function and therapeutic efficacy. Front Cell Dev Biol.
8:6032922020. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Lambeth DO: What is the function of GTP
produced in the Krebs citric acid cycle? IUBMB Life. 54:143–144.
2002.PubMed/NCBI
|
|
113
|
Saha T, Dash C, Jayabalan R, Khiste S,
Kulkarni A, Kurmi K, Mondal J, Majumder PK, Bardia A, Jang HL and
Sengupta S: Intercellular nanotubes mediate mitochondrial
trafficking between cancer and immune cells. Nat Nanotechnol.
17:98–106. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Baldwin JG, Heuser-Loy C, Saha T, Schelker
RC, Slavkovic-Lukic D, Strieder N, Hernandez-Lopez I, Rana N,
Barden M, Mastrogiovanni F, et al: Intercellular nanotube-mediated
mitochondrial transfer enhances T cell metabolic fitness and
antitumor efficacy. Cell. 187:6614–6630.e21. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Monaghan RM and Whitmarsh AJ:
Mitochondrial proteins moonlighting in the nucleus. Trends Biochem
Sci. 40:728–735. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
English J, Son JM, Cardamone MD, Lee C and
Perissi V: Decoding the rosetta stone of mitonuclear communication.
Pharmacol Res. 161:1051612020. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Quirós PM, Mottis A and Auwerx J:
Mitonuclear communication in homeostasis and stress. Nat Rev Mol
Cell Biol. 17:213–226. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Whelan SP and Zuckerbraun BS:
Mitochondrial signaling: Forwards, backwards, and in between. Oxid
Med Cell Longev. 2013:3516132013. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Jazwinski SM: The retrograde response: A
conserved compensatory reaction to damage from within and from
without. Prog Mol Biol Transl Sci. 127:133–154. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Walker BR and Moraes CT:
Nuclear-mitochondrial interactions. Biomolecules. 12:4272022.
View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Ju YS, Alexandrov LB, Gerstung M,
Martincorena I, Nik-Zainal S, Ramakrishna M, Davies HR,
Papaemmanuil E, Gundem G, Shlien A, et al: Origins and functional
consequences of somatic mitochondrial DNA mutations in human
cancer. Elife. 3:e029352014. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Chen S, Liao Z and Xu P: Mitochondrial
control of innate immune responses. Front Immunol. 14:11662142023.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Riley JS and Tait SW: Mitochondrial DNA in
inflammation and immunity. EMBO Rep. 21:e497992020. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Lin Y, Yang B, Huang Y, Zhang Y, Jiang Y,
Ma L and Shen YQ: Mitochondrial DNA-targeted therapy: A novel
approach to combat cancer. Cell Insight. 2:1001132023. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
West AP, Shadel GS and Ghosh S:
Mitochondria in innate immune responses. Nat Rev Immunol.
11:389–402. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
West AP, Khoury-Hanold W, Staron M, Tal
MC, Pineda CM, Lang SM, Bestwick M, Duguay BA, Raimundo N, MacDuff
DA, et al: Mitochondrial DNA stress primes the antiviral innate
immune response. Nature. 520:553–557. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Shimada K, Crother TR, Karlin J, Dagvadorj
J, Chiba N, Chen S, Ramanujan VK, Wolf AJ, Vergnes L, Ojcius DM, et
al: Oxidized mitochondrial DNA activates the NLRP3 inflammasome
during apoptosis. Immunity. 36:401–414. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
De Gaetano A, Solodka K, Zanini G, Selleri
V, Mattioli AV, Nasi M and Pinti M: Molecular mechanisms of
mtDNA-mediated inflammation. Cells. 10:28982021. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Wang X, Zhang H, Wang Y, Bramasole L, Guo
K, Mourtada F, Meul T, Hu Q, Viteri V, Kammerl I, et al: DNA
sensing via the cGAS/STING pathway activates the immunoproteasome
and adaptive T-cell immunity. EMBO J. 42:e1105972023. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Cheng AN, Cheng LC, Kuo CL, Lo YK, Chou
HY, Chen CH, Wang YH, Chuang TH, Cheng SJ and Lee AY: Mitochondrial
lon-induced mtDNA leakage contributes to PD-L1-mediated
immunoescape via STING-IFN signaling and extracellular vesicles. J
Immunother Cancer. 8:e0013722020. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Mahmood M, Liu EM, Shergold AL, Tolla E,
Tait-Mulder J, Huerta-Uribe A, Shokry E, Young AL, Lilla S, Kim M,
et al: Mitochondrial DNA mutations drive aerobic glycolysis to
enhance checkpoint blockade response in melanoma. Nat Cancer.
5:659–672. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Corrado M, Samardžić D, Giacomello M, Rana
N, Pearce EL and Scorrano L: Deletion of the mitochondria-shaping
protein Opa1 during early thymocyte maturation impacts mature
memory T cell metabolism. Cell Death Differ. 28:2194–2206. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Peng J, Hu X, Fan S, Zhou J, Ren S, Sun R
and Chen Y, Shen X and Chen Y: Inhibition of mitochondrial
biosynthesis using a ‘right-side-out’ membrane-camouflaged micelle
to facilitate the therapeutic effects of shikonin on
triple-negative breast cancer. Adv Healthc Mater. 11:e22007422022.
View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Gemta LF, Siska PJ, Nelson ME, Gao X, Liu
X, Locasale JW, Yagita H, Slingluff CL Jr, Hoehn KL, Rathmell JC
and Bullock TNJ: Impaired enolase 1 glycolytic activity restrains
effector functions of tumor-infiltrating CD8+ T cells.
Sci Immunol. 4:eaap95202019. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Al-Habsi M, Chamoto K, Matsumoto K, Nomura
N, Zhang B, Sugiura Y, Sonomura K, Maharani A, Nakajima Y, Wu Y, et
al: Spermidine activates mitochondrial trifunctional protein and
improves antitumor immunity in mice. Science. 378:eabj35102022.
View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Cronin SJF, Seehus C, Weidinger A, Talbot
S, Reissig S, Seifert M, Pierson Y, McNeill E, Longhi MS, Turnes
BL, et al: The metabolite BH4 controls T cell proliferation in
autoimmunity and cancer. Nature. 563:564–568. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Malinee M, Pandian GN and Sugiyama H:
Targeted epigenetic induction of mitochondrial biogenesis enhances
antitumor immunity in mouse model. Cell Chem Biol. 29:463–475.e6.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Chamoto K, Chowdhury PS, Kumar A, Sonomura
K, Matsuda F, Fagarasan S and Honjo T: Mitochondrial activation
chemicals synergize with surface receptor PD-1 blockade for T
cell-dependent antitumor activity. Proc Natl Acad Sci USA.
114:E761–E770. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Zhang Y, Kurupati R, Liu L, Zhou XY, Zhang
G, Hudaihed A, Filisio F, Giles-Davis W, Xu X, Karakousis GC, et
al: Enhancing CD8+ T cell fatty acid catabolism within a
metabolically challenging tumor microenvironment increases the
efficacy of melanoma immunotherapy. Cancer Cell. 32:377–391.e9.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
He J, Shangguan X, Zhou W, Cao Y, Zheng Q,
Tu J, Hu G, Liang Z, Jiang C, Deng L, et al: Glucose limitation
activates AMPK coupled SENP1-Sirt3 signalling in mitochondria for T
cell memory development. Nat Commun. 12:43712021. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Yuan S, Sun R, Shi H, Chapman NM, Hu H,
Guy C, Rankin S, Kc A, Palacios G, Meng X, et al: VDAC2 loss
elicits tumour destruction and inflammation for cancer therapy.
Nature. 640:1062–1071. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Liu YN, Yang JF, Huang DJ, Ni HH, Zhang
CX, Zhang L, He J, Gu JM, Chen HX, Mai HQ, et al: Hypoxia induces
mitochondrial defect that promotes T cell exhaustion in tumor
microenvironment through MYC-regulated pathways. Front Immunol.
11:19062020. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Teijeira A, Labiano S, Garasa S,
Etxeberria I, Santamaría E, Rouzaut A, Enamorado M, Azpilikueta A,
Inoges S, Bolaños E, et al: Mitochondrial morphological and
functional reprogramming following CD137 (4-1BB) costimulation.
Cancer Immunol Res. 6:798–811. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Wan H, Xu B, Zhu N and Ren B: PGC-1α
activator-induced fatty acid oxidation in tumor-infiltrating CTLs
enhances effects of PD-1 blockade therapy in lung cancer. Tumori.
106:55–63. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Chowdhury PS, Chamoto K, Kumar A and Honjo
T: PPAR-induced fatty acid oxidation in T cells increases the
number of tumor-reactive CD8+ T cells and facilitates
anti-PD-1 therapy. Cancer Immunol Res. 6:1375–1387. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Bukoreshtliev NV, Wang X, Hodneland E,
Gurke S, Barroso JFV and Gerdes HH: Selective block of tunneling
nanotube (TNT) formation inhibits intercellular organelle transfer
between PC12 cells. FEBS Lett. 583:1481–1488. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Liu K, Ji K, Guo L, Wu W, Lu H, Shan P and
Yan C: Mesenchymal stem cells rescue injured endothelial cells in
an in vitro ischemia-reperfusion model via tunneling nanotube like
structure-mediated mitochondrial transfer. Microvasc Res. 92:10–18.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Onfelt B, Nedvetzki S, Benninger RKP,
Purbhoo MA, Sowinski S, Hume AN, Seabra MC, Neil MA, French PM and
Davis DM: Structurally distinct membrane nanotubes between human
macrophages support long-distance vesicular traffic or surfing of
bacteria. J Immunol. 177:8476–8483. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Zhang H, Yu X, Ye J, Li H, Hu J, Tan Y,
Fang Y, Akbay E, Yu F, Weng C, et al: Systematic investigation of
mitochondrial transfer between cancer cells and T cells at
single-cell resolution. Cancer Cell. 41:1788–1802.e10. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Pierini S, Fang C, Rafail S, Facciponte
JG, Huang J, De Sanctis F, Morgan MA, Uribe-Herranz M, Tanyi JL and
Facciabene A: A tumor mitochondria vaccine protects against
experimental renal cell carcinoma. J Immunol. 195:4020–4027. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Shang L, Jiang X, Zhao X, Huang X, Wang X,
Jiang X, Kong X, Yao M, Jiang S and Wong PP: Mitochondrial
DNA-boosted dendritic cell-based nanovaccination triggers antitumor
immunity in lung and pancreatic cancers. Cell Rep Med.
5:1016482024. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Mok BY, de Moraes MH, Zeng J, Bosch DE,
Kotrys AV, Raguram A, Hsu F, Radey MC, Peterson SB, Mootha VK, et
al: A bacterial cytidine deaminase toxin enables CRISPR-free
mitochondrial base editing. Nature. 583:631–637. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Fan Y, Xu W, Gao BQ, Qin H, Wu X, Wei J,
Ni Q, Zhou L, Xiang J, Wu J, et al: Leveraging base excision repair
for efficient adenine base editing of mitochondrial DNA. Nat
Biotechnol. Mar 25–2025.(Epub ahead of print). View Article : Google Scholar
|
|
154
|
Guo Y, Zhang H, Yan C, Shen B, Zhang Y,
Guo X, Sun S, Yu F, Yan J, Liu R, et al: Small molecule agonist of
mitochondrial fusion repairs mitochondrial dysfunction. Nat Chem
Biol. 19:468–477. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Chen L, Fan Y, Jiang N, Huang X, Yu M,
Zhang H, Xu Z, He D, Wang Y, Ding C, et al: An energy
metabolism-engaged nanomedicine maintains mitochondrial homeostasis
to alleviate cellular ageing. Nat Nanotechnol. 20:1332–1344. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Pan X, Wang Z, Tan M, Fu Z, Nie G and Wang
H: Nanoinducer-mediated mitochondria-selective degradation enhances
T cell immunotherapy against multiple cancers. Nat Nanotechnol.
20:947–958. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Lv Y, Song B, Yang G, Wang Y, Wu Z, Si M,
Yang Z, Chen H, Liu C, Li M, et al: In situ transformable
nanoparticle effectively suppresses bladder cancer by damaging
mitochondria and blocking mitochondrial autophagy flux. Adv Sci
(Weinh). 12:e24094252025. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Wang W, Yao SY, Luo J, Ding C, Huang Q,
Yang Y, Shi Z, Lin J, Pan YC, Zeng X, et al: Engineered
hypoxia-responsive albumin nanoparticles mediating mitophagy
regulation for cancer therapy. Nat Commun. 16:5962025. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Liu J, Yan Y, Zhang Y, Pan X, Xia H, Zhou
J, Wan F, Huang X, Zhang W, Zhang Q, et al: Lysosome-mitochondria
cascade targeting nanoparticle drives robust pyroptosis for cancer
immunotherapy. J Am Chem Soc. 146:34568–34582. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Pei D and Buyanova M: Overcoming endosomal
entrapment in drug delivery. Bioconjug Chem. 30:273–283. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Goswami R, Jeon T, Nagaraj H, Zhai S and
Rotello VM: Accessing intracellular targets through
nanocarrier-mediated cytosolic protein delivery. Trends Pharmacol
Sci. 41:743–754. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Kang H, Han M, Xue J, Baek Y, Chang J, Hu
S, Nam H, Jo MJ, El Fakhri G, Hutchens MP, et al: Renal clearable
nanochelators for iron overload therapy. Nat Commun. 10:51342019.
View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Feng K, Xu Z, Wang Y, Wu X, Xiong F, Ruan
Y, Wu X, Ye L, Su D, Yu J and Sun X: Renal-clearable porous hollow
copper iron oxide nanoparticles for trimodal
chemodynamic-photothermal-chemo anti-tumor therapy. Nanoscale.
15:3188–3198. 2023. View Article : Google Scholar : PubMed/NCBI
|