Introduction
Head and neck squamous cell carcinoma (HNSCC) is a
prevalent malignancy, representing >90% of all head and neck
cases (1,2). While patients with early-stage HNSCC
generally have a favorable prognosis, those with advanced disease
tend to have notably poorer outcomes (3,4).
Although immunotherapy, particularly immune checkpoint blockade
(ICB) targeting the programmed cell death protein 1
(PD-1)/programmed death-ligand 1 (PD-L1) axis, has improved
survival in recurrent/metastatic HNSCC (1,5,6), the
molecular mechanisms underlying ICB resistance remain poorly
understood. Accumulating evidence has implicated the tumor immune
microenvironment as a critical determinant (7).
The tumor microenvironment (TME), particularly
carcinoma-associated fibroblasts (CAFs), drives immune evasion and
therapy resistance (8–10). CAFs influence the infiltration of
immunosuppressive cells, promote CD8+ T-cell exhaustion,
modulate natural killer cell activation and alter macrophage
polarization (11). Moreover, CAFs
contribute to establishing an immunosuppressive TME (12,13).
Emerging evidence has indicated that CAFs may contribute to reduced
responsiveness to ICB treatment (14,15).
CAFs heterogeneity markedly influences the responses
of various types of cancer to ICB. For example, fibroblast
activation protein (FAP)+ CAFs, subdivided into TGF-β
and extracellular matrix (ECM)-driven subclusters, promote
resistance via a PD-1/cytotoxic T-lymphocyte associated protein
4-dependent regulatory T cell loop (16). Conversely, Meflin-expressing CAFs
enhance ICB efficacy in non-small cell lung cancer by promoting
CD4+ T-cell infiltration and vascularization (17). Similarly, leucine rich repeat
containing 15 (LRRC15)+ CAFs mediate resistance by
driving CD8+ T-cell dysfunction (18). A multi-cancer analysis further
consolidated this paradigm by identifying six conserved pan-CAF
subtypes with distinct molecular signatures and roles in
anti-PD-1/PD-L1 resistance (19).
Macrophages and fibroblasts are found ubiquitously
within all tissue types. Tissue-resident macrophages maintain
homeostasis via phagocytosis, orchestrating immune responses and
promoting tissue damage repair. Macrophages are mainly classified
into M1 and M2 phenotypes, based on their functional
differentiation state. Proinflammatory factors (such as TNF-α and
IL-6) or bacterial products (for example, lipopolysaccharide)
induce M1 polarization, whereas immunoregulatory cytokines
(including TGFβ and IL-10) promote M2 polarization. M2 macrophages
secrete proangiogenic factors, such as VEGF and tissue-remodeling
enzymes, including MMPs, to facilitate tumor progression (20,21),
whereas M1 macrophages release antiangiogenic factors; for example,
IL-12 and CXCL10 and exhibit antitumor effects (22).
Periostin (POSTN), a secreted matricellular protein,
promotes tumor progression, chemoresistance and poor prognosis
across various types of cancer (23–28).
POSTN is highly secreted by CAFs and critically shapes the immune
microenvironment. For example, in glioma and ovarian cancer, POSTN
recruits M2-type tumor-associated macrophages (TAMs) and is
associated with poor survival (29,30).
Furthermore, POSTN+ CAFs orchestrate immunotherapy
resistance by forming a spatial unit with secreted phosphoprotein 1
(SPP1)+ macrophages. Through IL-6/STAT3 signaling, this
cellular coalition establishes an immunosuppressive niche
characterized by T-cell exclusion, thereby driving resistance to
ICB (31,32).
Extracellular vesicles (EVs) are a heterogeneous
family of lipid bilayer membrane-delimited nano-to micro-sized
particles naturally released by all cell types (33,34).
EVs comprise prototypic endosome-derived exosomes (endosome-derived
vesicles assembled via the fusion of multivesicular bodies and
released into the extracellular space), microvesicles (cell
membrane-derived submicron-sized particles) and apoptotic bodies
(35). Conceptually, EVs transport
their cargo to target sites, enabling action over distance
(33,35). Size-based EV nomenclature
categorizes the vesicles as follows: EVs < 200 nm in diameter
are classified as small EVs (sEVs), whereas EVs sized 200–1,000 nm
in diameter are classified as large EVs (lEVs) (35). A short POSTN isoform is expressed on
the surface of exosomes secreted by human cardiac explant-derived
progenitor cells (36). Lysyl
oxidase (LOX) is localized on CAF-derived sEV surfaces through its
binding to POSTN (37). However, it
remains unclear how POSTN-enriched CAF sEVs regulate macrophage
polarization.
In the present study, integration of two HNSCC
single-cell RNA sequencing (scRNA-seq) datasets revealed two
distinct fibroblasts subtypes: POSTN− and
POSTN+. Furthermore, the co-localization of FAP and
POSTN in the stromal compartment of HNSCC tissues was revealed.
Notably, FAP and POSTN expression exhibited a markedly positive
correlation with macrophage infiltration. Mechanistically,
POSTN+ CAF-derived sEVs could drive macrophage M2
polarization. Bone morphogenetic protein (BMP) 4 was downregulated
in macrophages treated with POSTN-silenced CAF-derived sEVs,
further demonstrating that BMP4 promoted M2 polarization through
BMP receptor 2 (BMPR2)/Smad signaling. Finally, integrated
scRNA-seq and The Cancer Genome Atlas (TCGA)-HNSCC analyses
elucidated the role of POSTN in modulating macrophage M2
polarization. The current study demonstrated that the
POSTN+ fibroblasts may promote an immunosuppressive
microenvironment in HNSCC by driving sEV-mediated macrophage M2
polarization, directly implicating POSTN as a potential target for
mitigating ICB resistance.
Materials and methods
Data acquisition, quality control and
processing
Single-cell RNA sequencing (scRNA-seq) data were
obtained from the GSE103322 (38)
and GSE139324 (38,39) datasets acquired from the Gene
Expression Omnibus (GEO, http://www.ncbi.nlm.nih.gov/geo/). GSE103322 dataset
was composed of 18 HNSCC patient samples (containing 5,902 single
cells). GSE139324 dataset involved 26 paired samples of peripheral
blood mononuclear cells (PBMCs) and tumor-infiltrating immune cells
from HNSCC patients 18 human papillomavirus (HPV)-negative and
eight HPV-positive), six PBMC samples from healthy donors and five
tissue-resident immune cell samples from healthy tonsils (totaling
131,224 cells). The two datasets were merged, resulting in a total
of 136,206 single cells.
Data processing and analysis were carried out in R
(version 4.4.2; http://www.r-project.org/) using the Seurat package
(version 5.3.1; http://satijalab.org/seurat/). Quality control was
performed by retaining cells with 300–7,000 detected genes
(nFeature_RNA) and less than 10% mitochondrial gene expression
(percent.mt). The top 2,000 variable genes were selected for
downstream analysis. Nonlinear dimensionality reduction was
conducted via Uniform Manifold Approximation and Projection (UMAP),
based on 15 principal components and a clustering resolution of
0.5.
Additionally, transcriptomic FPKM data from The
Cancer Genome Atlas (TCGA)-HNSCC patients were obtained from
cBioPortal (https://www.cbioportal.org/). To correct for batch
effects across subgroups, Harmony (version 0.1.0; http://github.com/immunogenomics/harmony) was applied.
Subsequent subgroup re-analysis adhered to the standard Seurat
workflow. Immune infiltration levels, assessed via single-sample
gene set enrichment analysis (ssGSEA), acquired from the TISIDB
database (http://cis.hku.hk/TISIDB/download.php).
Cell clustering and annotation
Dimensionality reduction was first performed on the
dataset by applying principal component analysis (PCA) to the top
2,000 highly variable genes. Subsequently, cell clustering was
carried out using Seurat's FindClusters function with a resolution
of 0.7. Finally, marker genes for the resulting cell subpopulations
were identified. All cells annotated based on the conventional
markers as previously reported. The following markers were used:
Epithelial makers (EpCAM, SFN, KRT14 and KRT5); Fibroblasts markers
(FN1, DCN, COL1A1, COL1A2, COL3A1 and COL6A1); T cell markers (CD2,
CD3D, CD3E and CD3G); B cell markers (CD19, CD79A, MS4A1, JCHAIN
and MZB1); Myeloid markers (CD68, LYZ, CD14, IL3RA, LAMP3, CLEC4C
and TPSAB1). Subsequently, the fibroblasts (containing 1,761 single
cells) were re-clustered into two subclusters, POSTN+
fibroblasts and POSTN− fibroblasts. Myeloid (containing
30,018 single cells) were re-clustered into two subclusters,
M1-like macrophage and M2-like macrophage for further analysis.
Bioinformatic analysis of the TCGA
database
Gene expression data of TCGA-HNSCC patients
(https://portal.gdc.cancer.gov/projects/TCGA-HNSCC)
were downloaded through the TCGA biolinks R package (version
2.34.1; http://bioconductor.org/packages/TCGAbiolinks/).
Transcripts per million reads (TPM) were transformed to
Log2 (TPM + 1) for further analysis. High and low
expression groups were determined according to the median values of
POSTN and FAP. The potential response to immune checkpoint blockade
ICB was predicted by the tumor immune dysfunction and exclusion
(TIDE) algorithm. The expression levels of POSTN and FAP along with
immune infiltration levels were quantified using ssGSEA. Based on
the ssGSEA enrichment scores of 28 immune signatures, the present
study performed unsupervised hierarchical clustering to identify
immune-related subtypes. TME was further characterized by
calculating the StromalScore, ImmuneScore, ESTIMATEScore using the
ESTIMATE algorithm and tumor purity was inferred accordingly.
Similarly, molecular subtypes were identified by clustering the
entire gene expression dataset. The distinction between
POSTN+ and POSTN− fibroblasts signatures was
visualized using t-distributed stochastic neighbor embedding
(t-SNE).
Immunohistochemistry
Patient inclusion and exclusion criteria were
established to define a homogeneous cohort for analysis. Inclusion
criteria comprised: i) a pathological diagnosis of oral squamous
cell carcinoma (OSCC); ii) consecutive enrollment from patients
undergoing primary surgical resection at Shanghai Stomatological
Hospital; iii) availability of adequately preserved formalin-fixed
paraffin-embedded (FFPE) tumor tissues; and iv) availability of
complete clinical data. Exclusion criteria were: i) Receipt of
neoadjuvant therapy prior to surgery; ii) presence of another
simultaneous active malignancy; iii) insufficient or poor-quality
FFPE blocks unsuitable for sectioning; and iv) incomplete clinical
records.
Based on these criteria, paraffin-embedded tissue
sections (4 µm) from 40 patients with oral squamous cell carcinoma
(OSCC) obtained from Shanghai Stomatological Hospital who underwent
surgical resection were included in this study, with 14 normal oral
mucosa samples serving as controls. The cohort comprised 26 male
and 14 female patients, with 17 patients aged ≤62 years and 23
patients aged ≥62 years. The mean age was 62 years. Clinical and
histopathological characteristics of the cohort are summarized in
Table SIII and all diagnoses were
confirmed by hematoxylin and eosin (H&E) staining.
Clinical and histopathological characteristics of
the cohort are summarized in Table
SIII and all diagnoses were confirmed by H&E staining.
Immunohistochemical staining was conducted using the
SPlink Detection Kit (SP-9000; Beijing Zhongshan Jinqiao
Biotechnology Co., Ltd.). Briefly, tissue sections were
deparaffinized in xylene, rehydrated through a graded ethanol
series and treated with 3% hydrogen peroxide in methanol to quench
endogenous peroxidase activity. After blocking nonspecific binding
with 10% goat serum (cat. no. C0265; Beyotime Biotechnology, Inc.),
the sections were incubated overnight at 4°C with primary
antibodies against FAP (cat. no. ab314075; 1:200; Abcam) and POSTN
(cat. no. ab14041; 1:100; Abcam). Following this, the sections were
incubated with biotinylated goat anti-rabbit IgG (cat. no. ab64256;
1:200; Abcam). at room temperature for 20 min and then washed with
PBS at room temperature for 10 min. Immunoreactivity was visualized
using 3,3′-diaminobenzidine (DAB) as the chromogen detection,
followed by hematoxylin counterstaining at room temperature for 5
min. Negative controls were processed similarly, with the primary
antibody replaced by PBS.
To ensure analytical reliability, a rigorous quality
assessment was implemented. All stained slides were independently
evaluated by two certified pathologists blinded to the
clinicopathological data. Slides were included in the final
analysis only if they met the following pre-defined criteria: i)
adequate control staining (positive/negative controls performing as
expected); ii) optimal morphological preservation of tissue
structure; and iii) low non-specific background interference.
Several slides were excluded during this quality control process
due to failure to meet these standards.
Stained sections were air-dried and examined under a
Leica Thunder DM6B optical microscope (Leica Microsystems GmbH).
Image-Pro Plus software (version 6.0; Media Cybernetics, Inc.) was
used to measure the integrated optical density and the area of
target protein distribution. The mean density per case was
determined by averaging values from at least 10 randomly selected
fields.
Immunofluorescence
A total of four representative HNSCC tissue samples
were used to capture biological heterogeneity while enabling
detailed exploratory analysis. Tissue sections from four HNSCC
cases were deparaffinized, rehydrated and subjected to heat-induced
antigen retrieval in pH 6.0 citrate buffer (cat. no. C1010; Beijing
Solarbio Science & Technology Co., Ltd.) at 95°C for 5 min.
Subsequently, the sections were washed with PBS at room temperature
for 10 min. After blocking nonspecific binding with 10% QuickBlock
Blocking Buffer (cat. no. P0220; Beyotime Biotechnology Inc.), the
sections were incubated overnight at 4°C with the following primary
antibodies: POSTN (cat. no. ab14041; 1:100; Abcam) and CD163 (cat.
no. sc-33715; 1:1,000; Santa Cruz Biotechnology, Inc.). The
following day, sections were washed with 1X TBST (containing 0.1%
Tween-20) at room temperature for 30 min and then incubated at room
temperature for 60 min with species-appropriate fluorescent
secondary antibodies: Goat Anti-Rabbit IgG AF488 (cat. no.
ab150077; 1:200; Abcam) or Goat Anti-Mouse IgG AF594 (cat. no.
ab150116; 1:200; Abcam). The sections were washed with 1X TBST at
room temperature for 10 min. Nuclei were stained with DAPI
(1:3,000; Thermo Fisher Scientific, Inc.) at room temperature for 5
min and then washed with 1X TBST at room temperature for 10 min.
images were acquired using a Leica Thunder DM6B optical microscope
(Leica Microsystems GmbH) for subsequent analysis.
Cell culture
The primary cells used in this
study-carcinoma-associated fibroblasts (CAFs) derived from human
OSCC tissues (CAF-S5 and CAF-S6) and healthy gingival-derived
normal fibroblasts (NFs) are identical to those that were isolated
and characterized in our previous study (37). CAFs and NFs were maintained in
DMEM/F12 medium (Gibco; Thermo Fisher Scientific, Inc.) containing
10% Fetal bovine serum (FBS; ScienCell Research Laboratories,
Inc.). Human immortalized monocytic cell line THP-1 was purchased
from iCell Bioscience (CVCL_0006) and grown in RPMI 1640 medium
(Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10% FBS
(ScienCell Research Laboratories, Inc.). All cultured cells were
supplemented with 100 U/ml penicillin and 100 U/ml streptomycin
(Gibco; Thermo Fisher Scientific, Inc.) and maintained at 37°C in a
humidified atmosphere with 5% CO2.
Stable transfection of POSTN
POSTN was silenced by CAFs with concentrated
lentivirus particles short hairpin (sh)RNA negative control (shNC;
sense:: 5′-GTTCTCCGAACGTGTCACGT-3′; anti-sense:
5′-ACGTGACACGTCGGAGAAC-3′) and POSTN-targeting shRNA (shPOSTN;
sense: 5′-CCCATGGAGAGCCAATTAT-3′; anti-sense:
5′-ATAATTGGCTCTCCATGGG-3′). The lentiviral particles and the shRNA
constructs were designed and provided by Shanghai GenePharma Co.,
Ltd. Specifically, the POSTN-targeting shRNA was cloned into the
lentiviral vector pHBIV-U6-Scramble-ZsGreen-Puro, and the construct
was verified by double enzyme digestion and DNA sequencing.
Human immortalized embryonic kidney cell line 293T
(CVCL_0063) was purchased from Shanghai Fuheng Biology and cultured
in DMEM High medium (Gibco; Thermo Fisher Scientific, Inc.)
supplemented with 10% FBS (ScienCell Research Laboratories, Inc.),
100 U/ml penicillin, and 100 U/ml streptomycin (Gibco; Thermo
Fisher Scientific, Inc.) and maintained at 37°C in a humidified
atmosphere with 5% CO2.
When 293T cells reached 85–90% confluence in 10 cm
dishes were co-transfected with 10 µg of the lentiviral shRNA
vector (pHBIV-U6-Scramble-ZsGreen-Puro), 7.5 µg of the packaging
plasmid pSPAX2, and 2.5 µg of envelope plasmid pMD2G using
Lipofectamine 3000 (L3000015; Thermo Fisher Scientific, Inc.),
following a second-generation system. This combination yielded a
plasmid mass ratio of 4:3:1. Viral supernatant was harvested 48 h
post-transfection, clarified by centrifugation (3,000 rpm at room
temperature for 10 min), and filtered through a 0.45 µm membrane.
The viral particles were then concentrated via ultracentrifugation
(50,000 × g at 4°C for 2 h) and resuspended in 200 µl of DMEM High
medium (Gibco; Thermo Fisher Scientific, Inc.). Subsequently,
CAF-S5 and CAF-S6 were incubated overnight with 8 mg/ml Polybrene
(cat. no. TR-1003; Sigma-Aldrich; Merck KGaA) and either control
lentiviral particles or those carrying POSTN-targeting shRNA at a
multiplicity of infection (MOI) of 100. At 48 h post-transfection,
media containing 2 mg/ml puromycin (Sigma-Aldrich; Merck KGaA) were
used to select stable clones for two weeks.
PMA-induced THP-1 differentiation into
macrophages
The human monocytic leukemia cell line THP-1 was
grown in a T25 culture flask supplemented with RPMI 1640
medium(Gibco; Thermo Fisher Scientific, Inc.) containing 50 ng/ml
of phorbol 12-myristate 13-acetate (PMA; cat. no. P8139;
Sigma-Aldrich; Merck KGaA) for 48 h. Then, they were maintained in
fresh RPMI 1640 culture medium for 24 h to stabilize their
growth.
Cell treatment
To confirm that BMP4 promotes macrophage
polarization toward the M2 phenotype, THP-1 (2×106
cells) were seeded in 6-well plates and differentiated into
macrophages using 50 ng/ml PMA for 48 h. After stabilization,
macrophages were assigned to different groups and then either left
unstimulated or stimulated for an additional 48 h with recombinant
human BMP4 (50 or 100 ng/ml; cat. no. HY-P7007; MedChemExpress).
Macrophages were stained with FITC anti-human CD163 antibody (cat.
no. 333617; 1:50; Biolegend Inc.) and FITC anti-human CD206
antibody (cat. no. 321103; 1:50; Biolegend Inc.) at 4°C for 15 min
and then centrifugation 1,000 × g at 4°C for 5 min. Pellets were
resuspended in 1 ml PBS and analyzed the expression of M2
macrophage polarization biomarkers (CD163 and CD206) was then
analyzed by Flow cytometry (NovoCyte 2040R; ACEA Biosciences)
equipped with NovoExpress software (version 1.6.3; http://explore.agilent.com/ACEA-joins-Agilent). To
further to confirm the role of Smad signaling pathway, 10 µM
LDN193189 inhibitor (cat. no. HY-12071; MedChemExpress) was added
to each well for 48 h during the BMP4 stimulation. Cellular
proteins were then harvested and subjected to western blotting to
evaluate the expression of markers associated with Smad-related and
M2 macrophage polarization.
Isolation, characterization of sEVs
and labeling
sEVs isolation methods were performed as previously
described (37). EV-depleted FBS
was obtained by centrifugation at 100,000 × g at 4°C for 3 h. At
80–90% confluence, CAFs and NFs were washed with PBS three times
and cultured in DMEM/F12 medium with 5% EV-depleted FBS for another
72 h. The resulting medium was collected as conditioned medium
(CM). Then, CM was differentially centrifuged 500 × g for 10 min,
2,500 × g at 4°C for 20 min, 12,000 × g at 4°C for 30 min, filtered
(pore size 0.22 µm), collected by ultracentrifuged at 100,000 × g
at 4°C for 70 min. The pellet was diluted in 20 ml PBS and
ultracentrifuged at 100,000 × g at 4°C for 70 min. The
concentration of sEVs was determined by Nanoflow (N30E; NanoFCM,
Co., Ltd.). Purified sEVs in PBS were placed on a formvar
carbon-coated 200 mesh grid (cat. no. 200M-Cu; EMCN, Inc.) at room
temperature for 20 min, washed twice with PBS and then stained 2%
uranyl acetate at room temperature for 60 sec. Analysis of sEVs
morphology and structure was performed with a transmission electron
microscope (TEM; JEM-2000EX; JEOL, Ltd.) equipped with a CCD camera
(Gatan SC1000, Model 832). Images were acquired and analyzed using
digital Micrograph software (version 3.6.1; Gatan, Inc.). Positive
and negative marker expression of sEVs were confirmed by western
blotting. The particles size and concentration of sEVs were
determined by Nanoflow (N30E, NanoFCM). sEVs were labeled with
PKH26 (cat. no. PKH26PCL, Sigma-Aldrich; Merck KGaA) according to
the company's instructions. Labeled sEVs were re-separated by
ultracentrifugation at 100,000 × g at 4°C for 70 min.
OptiPrep density gradient
purification
Purification of sEVs was performed using an OptiPrep
density gradient, as previously described (40). In brief, 60, 50, 40, 30, 25, 15, 10
and 5% (w/v) iodixanol solutions were prepared by diluting OptiPrep
(60% (w/v) (cat. no. D1556, Sigma-Aldrich; Merck KGaA) with 0.25 M
sucrose/10 mM Tris, pH 7.5 in Ultra-Clear tubes (cat. no. 344059;
Beckman Coulter, Inc.). Purified sEVs were subjected to density
gradient ultracentrifugation. The sample was loaded onto a
continuous gradient and centrifuged at 100,000 × g at 4°C for 18 h
using an Optima XPN-100 ultracentrifuge with a Ti41 rotor (Beckman
Coulter, Inc.), resulting in eight collected fractions. Each
fraction was diluted in PBS, pelleted by ultracentrifugation at
100,000 × g at 4°C for 70 min in Ultra-Clear tubes and finally
washed and resuspended in PBS.
Immunogold labeling of sEVs
Purified sEVs were adsorbed onto formvar
carbon-coated grids for 20 min. After three washes with PBS, free
aldehyde groups were quenched by incubation with 50 mmol/l glycine.
Following nine additional PBS washes, nonspecific sites were
blocked with 5% BSA for 1 h. Grids were then incubated overnight at
4°C with primary antibodies (cat. no. CD9/POSTN; 10 µg/ml; Abcam).
After incubation at room temperature with a 5 nm colloidal
gold-conjugated secondary antibody (cat. no. A31565; 1:50; Thermo
Fisher Scientific, Inc.) and a wash with PBS containing 0.1% BSA,
samples were fixed at room temperature in 2.5% glutaraldehyde for
15 min. Finally, after thorough deionized water rinsing, grids were
counterstained with phosphotungstic acid at room temperature for 5
min and imaged using a JEM-2000EX transmission electron microscope
(JEOL, Ltd.).
Uptake of PKH26-labeled sEVs by
macrophages
The purified sEVs in 50 µl PBS were fluorescently
labeled with 0.4 µl PKH26 Dye (Sigma-Aldrich; Merck KGaA) at room
temperature for 20 min. The labeled sEVs were then centrifuged at
100,000 × g at 4°C for 70 min. After discarding the supernatant,
equal amounts of sEVs were added to the macrophages at 37°C for 2 h
and 6 h, respectively. Macrophages were then fixed in 4%
paraformaldehyde at room temperature for 10 min. After
centrifugation at 1,000 × g at room temperature for 5 min, the
macrophages were resuspended in 1 ml PBS and the percentage of
fluorescence was analyzed using a Flow Cytometer (NovoCyte 2040R;
ACEA Biosciences).
CAF sEVs treatment for macrophages and
flow cytometry analysis
To confirm whether shNC and shPOSTN sEV-derived
CAF-S5/-S6 induce macrophage polarization into M1 and M2
macrophage, sEVs (5×109 particles) were added to
macrophage (2×106 cells) grown in 6-well plate for 48 h.
To test whether CAF-S5/-S6 sEV-POSTN directly induced M2 macrophage
polarization, sEVs (5×109 particles) were incubated
overnight 4°C with a POSTN-neutralizing antibody. These pre-treated
sEVs were then co-incubated with macrophage (2×106
cells) grown in 6-well plate at 37°C for 48 h. For staining, 100 µl
of macrophage (1×106 cells) was incubated with 5 µl of
FITC anti-human CD80 antibody (cat. no. 375405; BioLegend, Inc.),
FITC anti-human CD86 antibody (cat. no. 374203; BioLegend, Inc.),
FITC anti-human CD163 antibody (cat. no. 333617; BioLegend, Inc.)
and FITC anti-human CD206 antibody (cat. no. 321103; BioLegend,
Inc.) on ice for 15 min and washed twice with PBS. The stained
cells resuspended in 1 ml PBS and analyzed on a Flow cytometer
(NovoCyte 2040R, ACEA Biosciences) equipped with NovoExpress
software (version 1.6.3; http://explore.agilent.com/ACEA-joins-Agilent).
RNA extraction and reverse
transcription-quantitative (RT-q) PCR
Total RNA was extracted using TRIzol®
(Thermo Fisher Scientific, Inc.) according to the manufacture's
protocol. Brief, THP-1-derived macrophages (2×106 cells)
treated with PBS (CTRL), shNC sEVs (5×109 particles) and
shPOSTN sEVs (5×109 particles) for 48 h. Total RNA was
precipitated using isopropanol, washed with 100% ethanol and
resuspended in 30 µl of DEPC water. RNA concentration was
quantified using a Nanodrop One spectrophotometer (Thermo Fisher
Scientific, Inc.). Reverse transcription was performed with the
PrimerScript RT reagent Kit (Takara Biotechnology Co., Ltd.)
according to the manufacture's protocol, followed by quantitative
real-time PCR using the SYBR Premix Ex Taq reagent kit (Takara
Biotechnology Co., Ltd.) according to the manufacture's protocol on
a LightCycler 480II system (Roche Diagnostics) with the following
cycling protocol: Initial denaturation at 95°C for 15 min; followed
by 40 cycles of denaturation at 95°C for 10 sec, annealing at
56–60°C for 20 sec and extension at 72°C for 32 sec. GAPDH was used
as an internal control. The data for each sample were
semi-quantitatively analyzed using the 2−ΔΔCq method
(41). The primers for CD68, CD80,
CD86, CD163, CD206, BMP4, TNF-α, IL-6, TGF-β, IL-10 and GAPDH were
purchased from Sangon Biotech Co., Ltd. Primer sequences were as
following: CD68 (forward): 5′-AGCCACAAAACCACCACTCA-3′, (reverse):
5′-CTAGTGGTGGCAGGACTGTG-3′; CD80 (forward):
5′-GCAGGGAACATCACCATCCA-3′, (reverse): 5′-CACTTCCTTGGTCACGTGGA-3′;
CD86 (forward): 5′-GGAAGAAGAAGAAGCGGCCT-3′, (reverse):
5′-CGCTGGGCTTCATCAGATCT-3′; CD163 (forward):
5′-GCGGGAGAGTGGAAGTGAAA-3′; (reverse): 5′-ACCTGCACTGGAATTAGCCC-3′;
CD206 (MRC1) (forward): 5′-AGGATGGGTACTGGGCAGAT-3′; (reverse):
5′-CTGGACCTTGGCTTCGTGAT-3′; BMP4 (forward):
5′-GGAGGAGGAGGAAGAGCAGA-3′, (reverse): 5′-TTCTTCGTGGTGGAAGCTCC-3′;
TNF-α (forward): 5′-CTTCCAGCTGGAGAAGGGTG-3′, (reverse):
5′-CCCAAAGTAGACCTGCCCAG-3′; IL-6 (forward):
5′-AGTGAGGAACAAGCCAGAGC-3′, (reverse): 5′-GGTCAGGGGTGGTTATTGCA-3′;
TGF-β1 (forward): 5′-GCCCTGGACACCAACTATT-3′; (reverse):
5′-AGGCTCCAAATGTAGGGG-3′; IL-10 (forward):
5′-AAGACCCAGACATCAAGGCG-3′, (reverse): 5′-AGGCATTCTTCACCTGCTCC-3′;
GAPDH (forward): 5′-GTGAAGGTCGGAGTCAACG-3′, (reverse):
5′-TGAGGTCAATGAAGGGGTC-3′. Each experiment was repeated at least
three times.
Western blotting
Total protein was extracted with RIPA buffer (cat.
no. R0010; Beijing Solarbio Science & Technology Co., Ltd.)
supplemented with protease (cat. no. HY-K0010; MedChemExpress) and
phosphatase inhibitors (cat. no. HY-K0021; MedChemExpress) at 4°C
for 30 min. The lysates were then subjected to protein
concentration measurement with a BCA kit (cat. no. P0009; Beyotime
Biotechnology). Proteins (20 µg per lane) were separated by 4–20%
SDS-PAGE and subsequently transferred to a PVDF membrane
(MilliporeSigma). The membrane was then blocked with 5% fat-free
milk for 1 h at room temperature to prevent non-specific binding,
followed by an overnight incubation with primary antibodies at 4°C.
The primary antibodies included POSTN antibody (cat. no. ab14041;
1:500; Abcam), CD9 (cat. no. ab236630; 1:1,000; Abcam), CD81 (cat.
no. ab79559; 1:1,000; Abcam), Calnexin (10427-2-AP; 1:1,000,
Proteintech, Wuhan, China), CD80 (cat. no. ab134120; 1:1,000;
Abcam), CD86 (cat. no. abs115477; 1:1,000; Absin Bioscience), CD163
(sc-33715; 1:1,000, Santa Cruz Biotechnology), CD206 (cat. no.
ab64693; 1:1,000; Abcam), BMPR2 (cat. no. abs147034; 1:1,000; Absin
Bioscience), Phospho-Smad 1
(Ser463/465)/Smad5(Ser463/465)/Smad9(Ser465/467) (cat. no. 13820;
1:1,000; Cell Signaling Technology, Inc.), Smad5 (12534; 1:1,000;
Cell Signaling Technology, Inc.), Smad9 (cat. no. abs131190;
1:1,000; Absin Bioscience) and GAPDH (cat. no. 10494-1-AP; 1:5,000;
Proteintech Group, Inc.), followed by incubation with a
HRP-conjugated Goat anti-Rabbit IgG (H+L) as the secondary antibody
(cat. no. SA00001-2; 1:3,000; Proteintech Group, Inc.) for 2 h.
After washing the membrane three times with 1X TBST, the protein
bands were visualized using an ECL detection system. Amersham
ImageQuant 800 (Cytiva) and the intensity of the band was
quantified using ImageJ software (version 1.54k; National
Institutes of Health).
Enzyme-linked immunosorbent assay
(ELISA)
To confirm the presence of POSTN on the surface of
CAF sEVs, Human POSTN ELISA kits was purchased (cat. no. H2452c,
Elabscience). To assess the content of BMP4 on the surface of
sEV-derived CAF-S5/-S6 transfected with shNC and shPOSTN, Human
BMP4 ELISA kits was purchased (cat. no. CSB-E17298h; Cusabio
Technology, LLC). Serially diluted CAF-derived sEVs were added to
each well (100 µl/well) and incubated for 90 min. Subsequently,
biotinylated POSTN and BMP4 antibody was introduced and incubated
at 37°C for 1 h, followed by sequential additions of horseradish
peroxidase-conjugated streptavidin (100 µl, 37°C for 30 min) and
3,3′,5,5′-tetramethylbenzidine substrate (37°C for 15 min). The
reaction was terminated with 50 µl of stop solution and absorbance
at 450 nm was measured using a Thermo Fisher Scientific, Inc.
microplate reader.
For POSTN blocking on the sEV surface, CAF sEVs were
incubated overnight at 4°C with a POSTN-blocking antibody (10 µg/ml
in 100 µl PBS; Abcam). Non-bound antibodies were removed by washing
with 20 ml PBS and ultracentrifugation at 100,000 × g at 4°C for 70
min. All experiments were performed in at least triplicate.
RNA sequencing analysis
Total RNA was extracted from macrophages treated
with CAF sEV-derived shNC and shPOSTN for 48 h using
TRIzol® Reagent (Thermo Fisher Scientific, Inc.) per the
manufacturer's protocol. RNA integrity was assessed by Agilent 5300
Bioanalyser and concentration was measured using a Nanodrop
ND-2000. High-quality RNA samples were used for library
preparation. Subsequently, RNA purification, reverse transcription,
library construction and sequencing were conducted by Majorbio
Biotechnology on an Illumina NovaSeq/HiSeq Xten (Illumina, Inc.)
platform with NovaSeq reagents kits (Illumina, Inc.) according to
standard protocol. Raw paired-end reads were processed with Fastp
(version 1.0.1; http://github.com/OpenGene/fastp) for adapter trimming
and quality control. HISAT2 was used to align the cleaned reads to
the reference genome and transcript assembly was performed with
StringTie (version 3.0.3; http://ccb.jhu.edu/software/stringtie). Gene
expression was quantified in TPM and differential expression
analysis was carried out to identify differentially expressed genes
(DEGs) between comparative groups. RSEM was used to quantify gene
abundances. Essentially, differential expression analysis was
performed using the DEGseq (42).
Functional-enrichment analysis including Gene Ontology (GO) and
Kyoto Encyclopedia of Genes and Genomes (KEGG) were performed to
identify which DEGs were significantly enriched in GO terms and
metabolic pathways at Bonferroni-corrected P-value ≤0.05 compared
with the whole-transcriptome background. GO functional enrichment
and KEGG pathway analysis were carried out by Goatools and KOBAS
(43), respectively.
Statistical analysis
Single-cell sequencing was performed using RStudio
software (version 4.1.1). Data distributions were visualized via
histograms generated in R software. Categorical variables were
compared using the Chi-squared test. For continuous variables,
comparisons between two groups used the unpaired two-tailed
Student's t-test or the Wilcoxon rank-sum test. Comparisons among
three or more groups were performed using one-way ANOVA test
followed by Tukey's post hoc test. Inter-variable relationships
were examined using Spearman's rank correlation. Statistical
analysis and quantification of western blotting bands were
performed using GraphPad prism software (version 7.0; Dotmatics)
and ImageJ (version 1.6.0; National Institutes of Health). Results
are expressed as mean ± SEM from a minimum of three independent
experiments. P<0.05 was considered to indicate a statistically
significant difference.
Results
POSTN+FAP+
fibroblasts are associated with a poor ICB response in HNSCC
An integrated analysis of the HNSCC single-cell
profiling data was conducted using sequencing datasets (Fig. 1A), revealing five clusters that were
annotated via Seurat clustering according to specific markers. The
gene lists associated with cluster annotation markers are included
in Table SI. Existing studies have
revealed that POSTN is markedly elevated in tumor-derived
fibroblasts compared with in normal tissues (37,44,45).
The vlnplot demonstrated that POSTN was mainly expressed in the
fibroblasts cluster (Fig. 1B).
Subsequently, the fibroblasts were further re-clustered into nine
clusters (Fig. 1C) and it was
revealed that POSTN was expressed in subpopulations 1, 4 and 5,
with its expression was predominantly enriched in subpopulation 1
(Fig. 1D). To determine whether
POSTN was expressed in M1/M2 macrophages, the myeloid cluster was
further re-clustered into 14 subtypes. Low POSTN expression was
detected in five of these subpopulations and it was negligible in
the others (Fig. S1A). Based on
established biomarkers (Table
SII), these subpopulations were categorized into M1-like and
M2-like macrophages (Fig. S1B).
Notably, low expression of POSTN was present in M1-like macrophages
and it was barely detectably in M2-like macrophages (Fig. S1C). Based on UMAP clustering
analysis, the fibroblast subpopulations were categorized into
POSTN− and POSTN+ fibroblasts (Fig. 1E). Heatmap analysis revealed that
myofibroblastic CAF (myCAF)-related proteins (ACTA2, MYH11, TAGLN,
PDGFA, MCAM and MYLK) and inflammatory CAF (iCAF)-related proteins
(IL6, CXCL12, C3 and CFD) were predominantly expressed in
POSTN− fibroblasts. By contrast, ECM CAF (eCAF)-related
proteins (FAP, FN1, SFRP2, THY1, COL1A1, FBLN1 and LUM) and antigen
presenting CAF (apCAF)-related proteins (CD74, HLA-DRA, HLA-DRB5,
HLA-DRB1 and HLA-DPA1) were predominantly expressed in
POSTN+ fibroblasts (Fig.
1F). Vnplots further illustrated the expression levels of eCAF
and apCAF-associated proteins in POSTN+ fibroblasts and
POSTN− fibroblasts (Fig.
S1D). Spearman's correlation analysis revealed a significant
correlation between the expression levels of POSTN and FAP
(Fig. 1G). Moreover, patients with
POSTNhigh and FAPhigh HNSCC exhibited a high
poor response to ICB (Fig. 1H). The
clinical parameters of 40 OSCC cases and 14 normal controls are
listed in Table SIII. H&E and
immunohistochemical staining identified higher expression levels of
FAP and POSTN in the stroma of OSCC cases compared with those in
the normal control samples (Fig.
1I). These findings indicated that
POSTN+FAP+ fibroblasts are localized in the
stromal region and associated with a poor response to ICB in
patients with HNSCC.
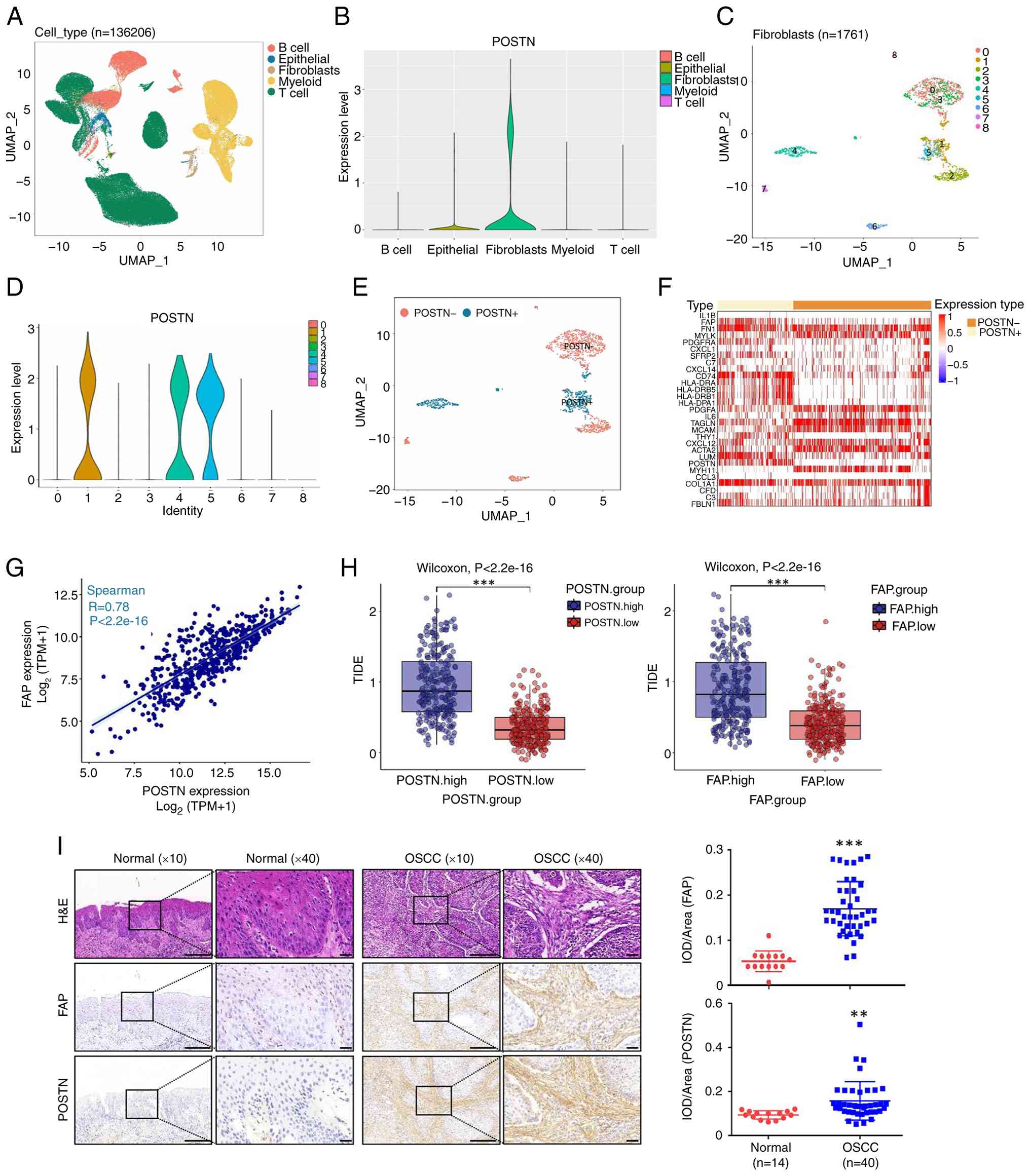 | Figure 1.POSTN+FAP+
fibroblasts contributed to the resistance against ICB in HNSCC. (A)
The datasets of GSE103322 and GSE139324 were integrated for further
analysis. A total of five clusters were identified previously based
on reported biomarkers, including B cell, epithelial, fibroblasts,
myeloid and T cells. (B) Vlnplots demonstrated the expression
levels of POSTN across five clusters. (C) Fibroblasts re-clustered
into nine clusters. (D) Vlnplots visualized the expression levels
of POSTN in the nine clusters. (E) POSTN− fibroblasts
and POSTN+ fibroblasts were annotated based on POSTN
expression levels in fibroblasts subtypes. (F) Heatmap displayed
the expression levels of cell-type specific biomarkers (myCAF,
iCAF, apCAF and eCAF) in POSTN− fibroblasts and
POSTN+ fibroblasts. (G) POSTN showed a significant
correlation with FAP in the TCGA-HNSCC (***P<0.001; Spearman's
rank correlation). (H) High expression of POSTN and FAP was
associated with elevated TIDE scores. Blue and red bars
representing higher and lower expression levels, respectively. (I)
H&E and IHC analysis of FAP and POSTN expression in OSCC cases
(n=40) and normal controls (n=14). Scale bar, 100 and 20 µm. Right:
IOD/Area of FAP and POSTN in the normal and OSCC tissues were
quantified. Statistical significance was determined by unpaired
two-tailed Student's t-test, **P<0.01; ***P<0.001. Error bars
represent the mean ± SEM. POSTN, perostin; FAP, fibroblast
activation protein; ICB, immune checkpoint blockade; HNSCC, head
and neck squamous cell carcinoma; TIDE, tumor immune dysfunction
and exclusion; H&E, hematoxylin and eosin; IHC,
immunohistochemical; OSCC, oral squamous cell carcinoma; IOD,
integrated option density. |
Association of
POSTN+FAP+ fibroblasts with macrophage
infiltration and contributing to an immunosuppressive
microenvironment in HNSCC
Recent studies have provided evidence for
intercellular communication between
POSTN+FAP+ fibroblasts and macrophages
(46–48). Additionally, the macrophage
infiltration rate is a critical factor in responses to ICB. The
ssGSEA algorithm (Fig. 2A)
demonstrated that the expression levels of POSTN and FAP were
markedly associated with the immune infiltration of macrophages in
TCGA-HNSCC. The expression levels of POSTN and FAP exhibited strong
positive correlations with markers of M1 (CD80 and CD86) and M2
(CD163 and CD206) macrophages, respectively. However, the
correlations with the M2 macrophage markers (CD163,
P<2.2×10−16; CD206, P<2.2×10−16) were
markedly stronger (Figs. 2B,
S2A and B). Immunofluorescence
staining revealed the co-expression of POSTN and CD163 in the
stromal area in HNSCC cases (Fig.
2C). To delineate the role of POSTN+ fibroblasts in
the TME, we analyzed 28 immune-related signatures, two distinct
immunity subtypes were identified through unsupervised hierarchical
clustering: high and low-immunity subtypes (Fig. 2D). The high-immunity subtype was
characterized by high stromal, immune, ESTIMATE and POSTN scores
coupled with low tumor purity (Fig.
2E). Then, two molecular subtypes of HNSCC-TCGA were identified
through unsupervised hierarchical clustering (Fig. S2C and D). Further analysis of the
TME revealed that POSTN+ fibroblasts were characterized
by high stromal, ESTIMATE, immune and POSTN scores, as well as low
tumor purity (Fig. S2E).
Interestingly, M0 macrophages, M2 macrophages and CD4+ T
memory cells also highly infiltrated the POSTN+
fibroblasts, highlighting the strong positive correlation between
the POSTN+ fibroblasts and M2 macrophages in HNSCC
(Fig. 2F). Collectively, these
findings strongly suggest that POSTN+ fibroblasts are
associated with an immunosuppressive TME in HNSCC, potentially
through their strong correlation with and spatial proximity to M2
macrophages.
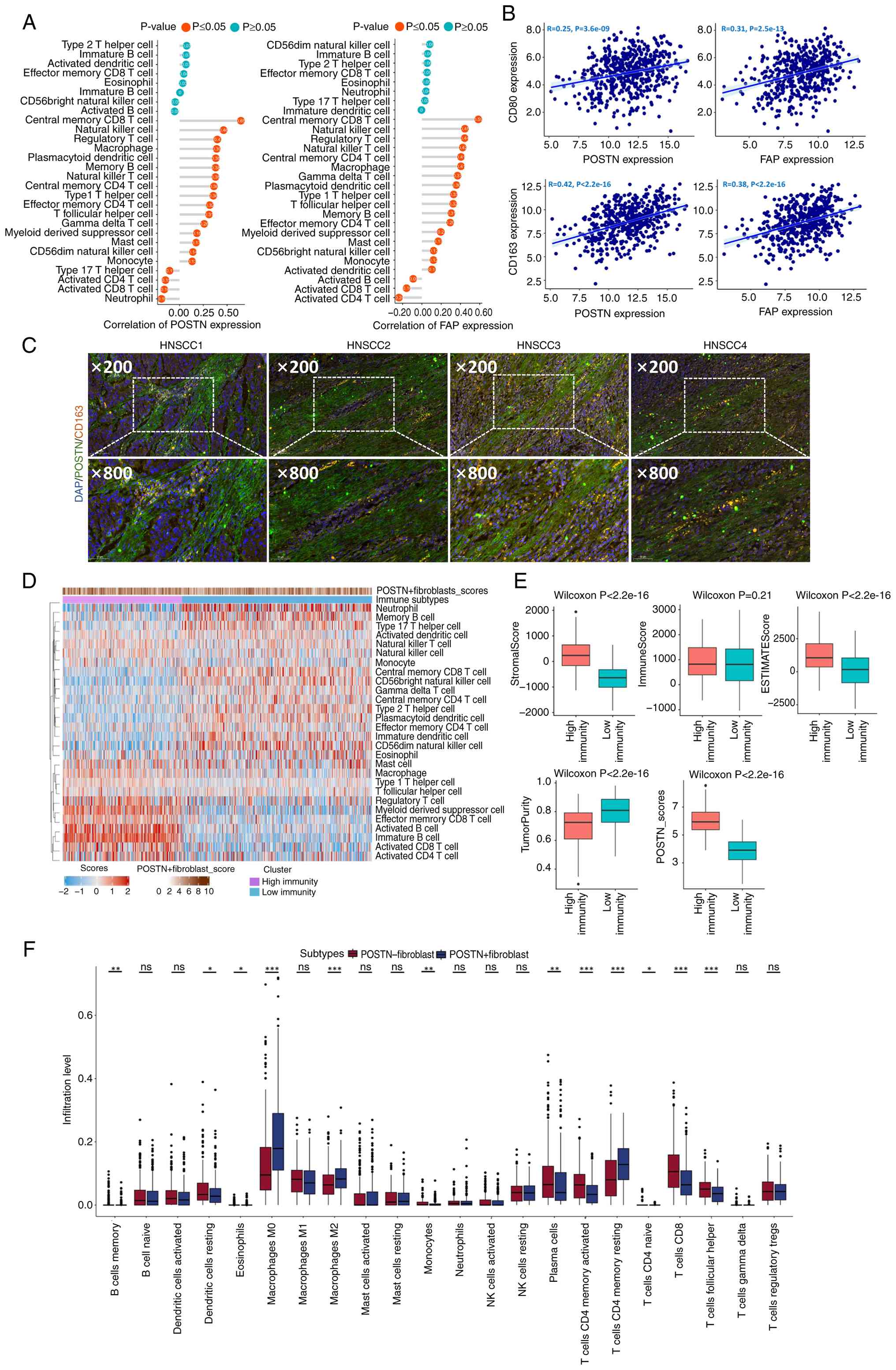 | Figure 2.Association of
POSTN+FAP+ fibroblasts with the immune
landscape in HNSCC. (A) POSTN and FAP expression associated with
the level of immune cell infiltration in the TCGA-HNSCC (Spearman's
rank correlation). (B) A significant positive correlation was
observed between the expression of POSTN/FAP and CD80/CD163 in the
TCGA-HNSCC (***P<0.001; Spearman's rank correlation). (C)
Co-expression of POSTN (green) and CD163 (orange) in HNSCC cases
(n=4). Top: images of HNSCC tissues (Scale bar, 100 µm). Bottom:
images of HNSCC tissues (Scale bar, 50 µm). (D) High and low
immunity subtypes were identified by unsupervised hierarchical
clustering. (E) The distribution of stromal, immune, ESTIMATE,
TumorPurity and POSTN scores in high and low immunity subtypes. (F)
The infiltration abundances of 22 immune cells across
POSTN−/POSTN+ fibroblasts. Statistical
significance was determined by unpaired two-tailed Student's
t-test, ns, not significant; *P<0.05; **P<0.01;
***P<0.001. Error bars represent the mean ± SEM. POSTN,
perostin; FAP, fibroblast activation protein; TCGA, The Cancer
Genome Atlas; HNSCC, head and neck squamous cell carcinoma. |
POSTN is present on the surface of CAF
sEVs
To further confirm the expression levels of POSTN in
CAFs and their sEVs, human primary OSCC-derived CAF-S5/-S6 and
healthy gingival-derived NFs were successfully extracted and
validated, as previously described (37). The sEVs were isolated from the
respective CM of CAF-S5/-S6 and NFs using differential
ultracentrifugation (Fig. 3A) and
they exhibited characteristic exosomal morphology in TEM images
(Fig. 3B). These sEVs tested
positive for exosomal markers, including CD9, CD81 and ALIX and
tested negative for Calnexin (Fig.
3C). The size distribution and particle concentration of sEVs
were examined using a Nanoflow cytometer, revealing an mean size of
~100 nm and a concentration of ~1×1010 sEVs/1ml of CM
(Fig. 3D). The expression levels of
POSTN in CAF-S5/-S6 and their sEVs were markedly higher than those
in NFs and their sEVs (Fig. 3E).
Furthermore, density gradient fractionation analysis revealed the
presence of POSTN on CAF-derived sEVs (Fig. 3F). Immunogold labeling confirmed the
presence of POSTN on the surface of CAF-S5/-S6 sEVs (Fig. 3G). ELISA further demonstrated that
the level of sEV-POSTN from these cells increased in an sEV
dose-dependent manner (Fig. 3H), a
signal that was markedly reduced by a POSTN-blocking antibody
(Fig. 3H). Taken together, these
findings identified POSTN as a surface component of CAF-S5/-S6
sEVs.
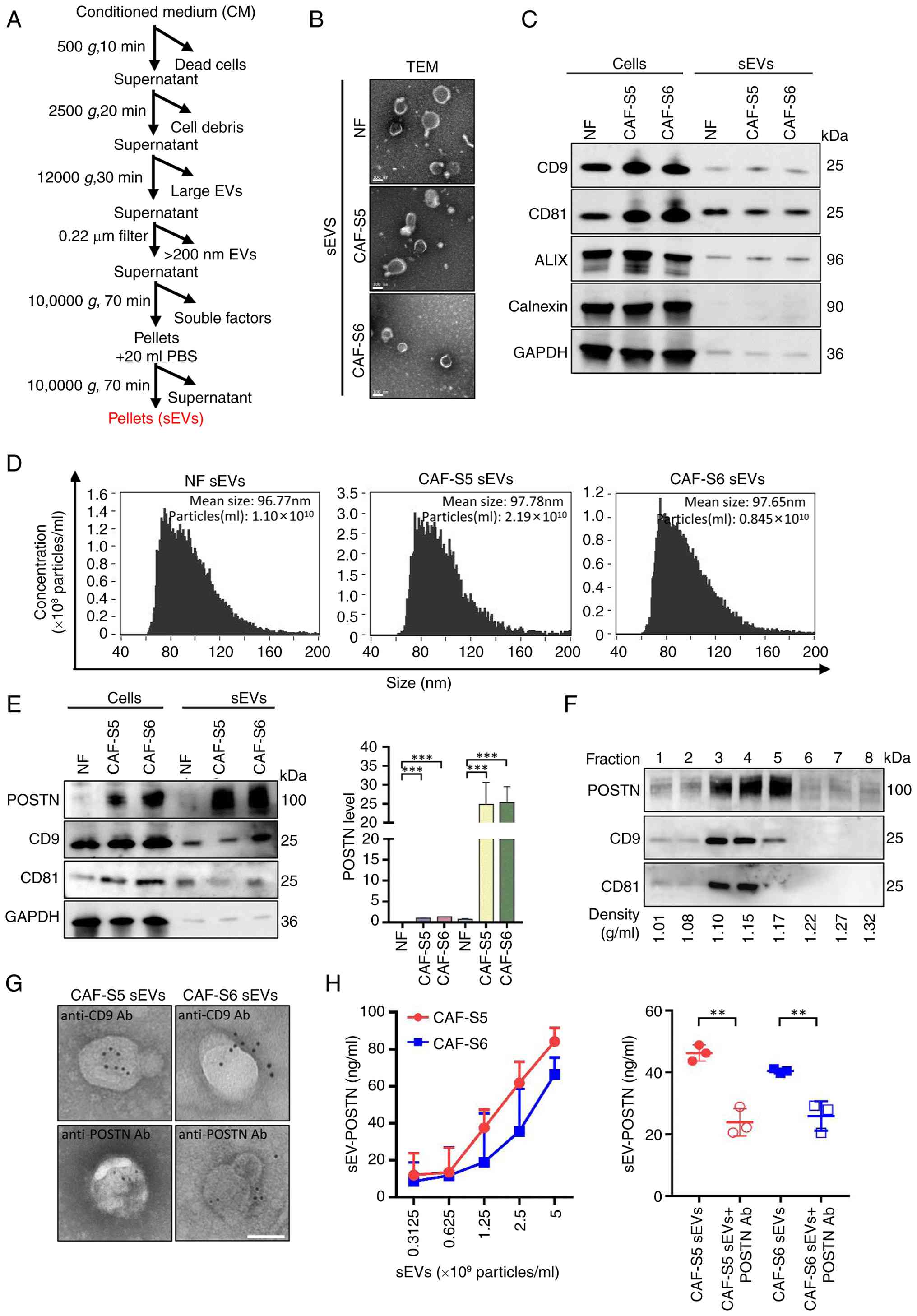 | Figure 3.POSTN is located on the surface of
sEV-derived from CAFs. (A) Flowchart of sEVs isolation process. (B)
The morphology and structure of sEVs examined by TEM. Scale bar,
100 nm. (C) The expression of biomarkers of sEVs were examined by
western blotting. CD9, CD81 and ALIX were used as positive
biomarkers for sEVs, while Calnexin was used as a negative
biomarker. GAPDH was used as internal control. (D) Nanoflow
cytometer examined the particles concentration and mean size
distribution of sEVs. (E) The expression levels of POSTN in CAFs
and their sEVs were measured. CD9 and CD81 were used as positive
markers of sEVs (n=3 per group). GAPDH was used as an internal
control. (F) Western blotting analysis was performed the expression
levels of POSTN in density gradient fractionation of sEV-derived
CAF-S6. CD9 and CD81 were used as positive markers of sEVs. (G) TEM
images of immunogold-labeled POSTN on CAF-S5/-S6 sEVs (Scale bar,
100 nm). (H) ELISA examination of POSTN in CAF-S5/-S6 sEVs (n=3 per
group). Statistical significance was determined by unpaired
two-tailed Student's t-test, **P<0.01; ***P<0.001. Error bars
represent the mean ± SEM. For source data for blotting assays, see
Fig. S5. POSTN, perostin; sEV,
small extracellular vesicles; CAFs, carcinoma-associated
fibroblasts; TEM, transmission electron microscopy. |
CAF-derived sEV-POSTN induces M2
polarization of THP-1-derived macrophages
To determine whether sEV-POSTN can induce macrophage
polarization, CAF-S5/-S6 stably transfected with shNC and shPOSTN
were established and sEVs were isolated by differential
ultracentrifugation. The expression of POSTN in CAF-S5/-S6 and
their sEVs was markedly reduced by shPOSTN compared with that in
the shNC group (Fig. 4A). The mean
sizes of sEVs derived from shNC/shPOSTN-transfected CAF-S5/-S6 were
~95 and 80 nm, respectively and their particles concentration were
~1.9×1010 and 2.7×1010 sEV/1 ml CM,
respectively (Fig. 4B). Human THP-1
cells were incubated with PMA (50 ng/ml) for 48 h to induce
differentiation into M0 macrophages, which exhibited the
corresponding adherent morphology (Fig.
4C) and highly expressed the macrophage marker CD68
(P<0.001; Fig. 4D). Flow
cytometry revealed that macrophages exhibited no significant
difference in their uptake of PKH26-labeled sEVs derived from
shNC/shPOSTN-transfected CAF-S5/-S6 (Fig. 4E). RT-qPCR assay revealed that the
M2 macrophage markers CD163 and CD206 were markedly downregulated,
whereas the M1 macrophage markers CD80 and CD86 remained unchanged,
after co-incubation with sEVs derived from shPOSTN-transfected
CAF-S5/-S6 compared with those from sEVs derived from
shNC-transfected CAF-S5/-S6 (P<0.01; Fig. 4F). Flow cytometry confirmed that the
expression levels of M2 macrophage markers (CD163 and CD206) were
markedly reduced in macrophages treated with sEVs derived from
POSTN-knockdown CAF-S5/-S6 (Fig.
4G). Furthermore, the expression levels of M2 macrophage
markers (CD163 and CD206) were markedly downregulated in
macrophages treated with sEVs derived from shPOSTN-transfected
CAF-S5/-S6 compared with those treated with sEVs derived from
shNC-transfected CAF-S5/S6 (Fig.
4H). However, the expression of M1 macrophage markers (CD80 and
CD86) showed no significant difference in macrophages treated with
sEVs derived from shNC/shPOSTN-transfected CAF-S5/-S6 (Fig. 4H).
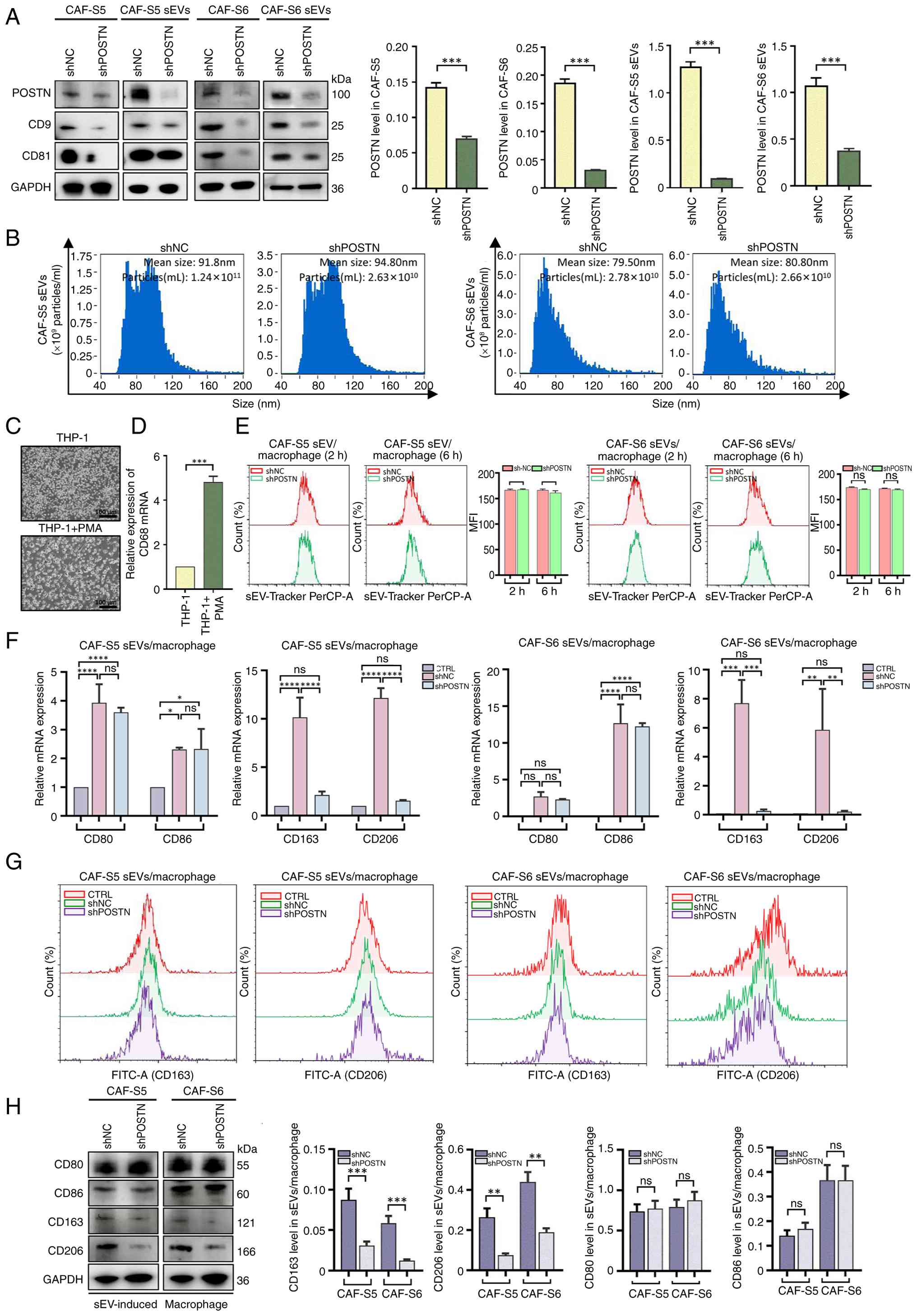 | Figure 4.sEV-derived CAFs induced macrophage
M2 polarization. (A) The expression levels of POSTN in CAF-S5/-S6
and their sEVs transfected with shNC and shPOSTN were examined by
western blotting (n=3 per group). CD9 and CD81 were used as
positive markers of sEVs. Left: Representative images. Right:
Quantitative analysis of POSTN in different groups. (B) Particles
concentration and size distribution of sEV-derived CAF-S5/-S6
transfected with shNC and shPOSTN were analyzed by Nanoflow
cytometer. (C) Representative image of macrophages derived from
THP-1 treated with PMA (50 ng/ml; scale bar, 100 µm). (D) The mRNA
expression of CD68 was analyzed by RT-qPCR. (E) Internalization of
PKH26-labeled sEV-derived CAF-S5/-S6 transfected with shNC and
shPOSTN by macrophages was examined by flow cytometer at 2 and 6 h
after incubation (n=3 per group). Quantitative analysis of the MFI
of PKH26-labeled sEVs internalized by macrophages for 2 and 6 h,
respectively. The mRNA expression of CD80, CD86, CD163 and CD206
expression in macrophages were treated with sEV-derived CAF-S5/-S6
transfected with shNC and shPOSTN were analyzed by (F) RT-qPCR, (G)
flow cytometry and (H) western blotting. Statistical significance
was determined using a one-way ANOVA test. ns, not significant;
*P<0.05; **P<0.01; ***P<0.001; ****P<0.0001. Error bars
represent the mean ± SEM. For source data for blotting assays, see
Fig. S5. sEV, small extracellular
vesicles; CAFs, carcinoma-associated fibroblasts; POSTN, perostin;
sh, short hairpin; NC, negative control; RT-qPCR, reverse
transcription-quantitative PCR; MFI, mean fluorescence
intensity. |
RT-qPCR and western blotting analysis of macrophages
also showed that the expression levels of M2 macrophage markers
(CD163 and CD206) were markedly downregulated in macrophages
treated with CAF-S5/-S6 sEVs pre-incubated with a POSTN-blocking
antibody, compared with those treated with CAF-S5/-S6 sEVs
(Fig. S3A and B). Flow cytometry
confirmed that CAF-S5/-S6 sEVs pre-incubated with a POSTN-blocking
antibody markedly reduced the expression levels of M2 macrophage
markers (CD163 and CD206) (Fig. S3C
and D). These results demonstrated that sEVs secreted by
POSTN-knockdown CAF-S5/-S6 may inhibit M2 macrophage
polarization.
BMP4 drives macrophage M2 polarization
by activating BMPR2/Smad signaling
To further investigate the mechanism by which POSTN
regulates THP-1-derived macrophages M2 polarization, RNA sequencing
was performed on THP-1-derived macrophages stimulated with sEVs
derived from CAF-S6 transfected with shPOSTN and shNC. PCA was
employed to assess transcriptomics profiles, revealing a clear
distinction between macrophages treated with shPOSTN sEVs and CTRL
or shNC sEVs (Fig. 5A). A Venn
diagram revealed 15 shared genes and 242 unique genes between
macrophages treated with shPOSTN sEVs and shNC sEVs (Fig. 5B). Furthermore, a heatmap showed
significant upregulation and downregulation of genes in macrophages
stimulated with sEVs derived from CAF-S6 transfected with shPOSTN
compared with those treated with sEVs derived from CAF-S6
transfected with shNC (Fig. 5C). A
volcano plot identified 443 DEGs, including 262 upregulated and 181
downregulated genes, in macrophages treated with shPOSTN sEVs
compared with those treated with shNC sEVs (Fig. 5D). Among the top DEGs, BMP4 was
identified as a critical growth factor that fosters a pro-tumoral
immune environment by polarizing macrophages toward an M2-like
phenotype, thereby promoting tumor growth (49).
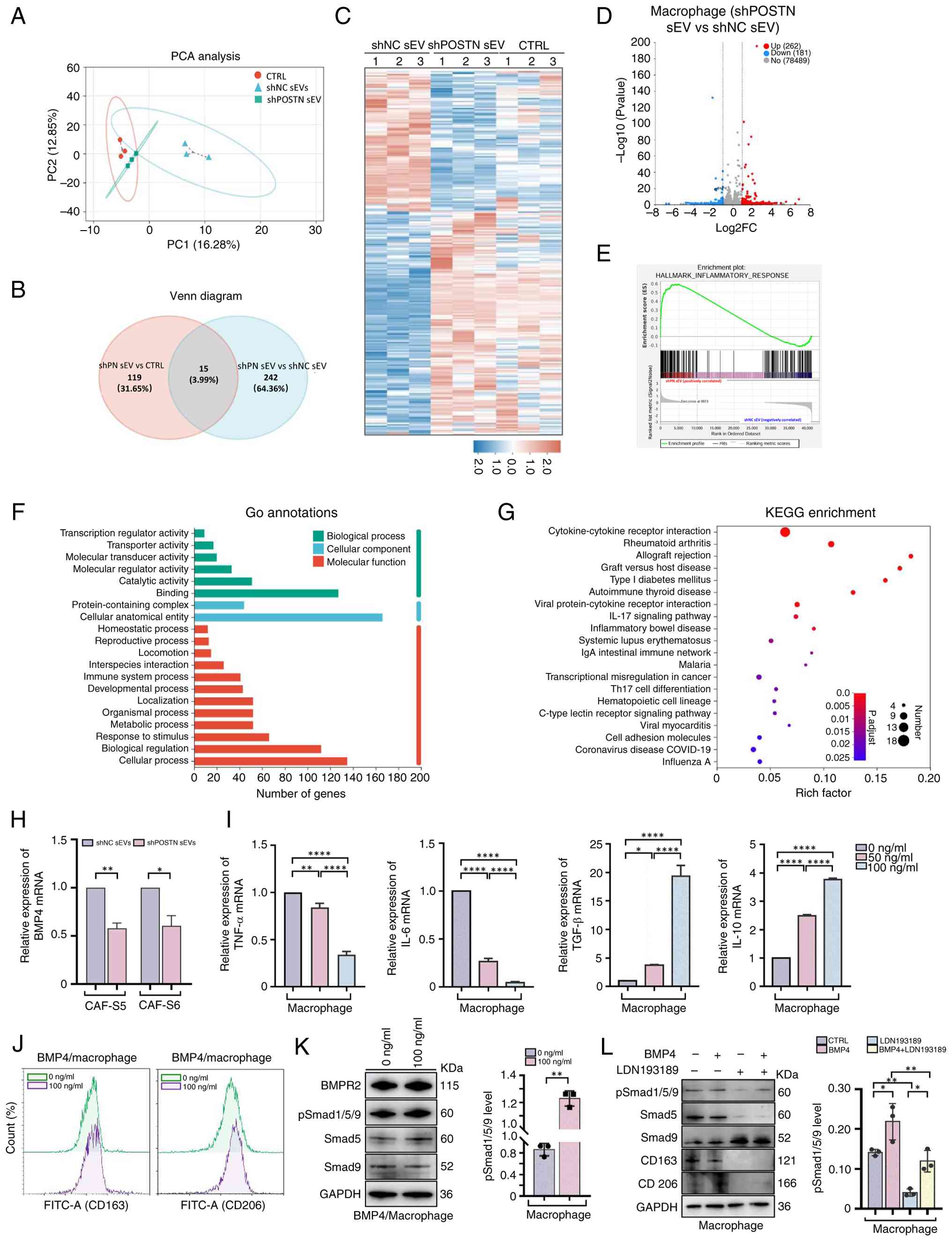 | Figure 5.BMP4 activates BMPR2/Smad signaling
to induce macrophage M2 polarization. (A) PCA analysis of
macrophages were treated with sEV-derived CAF-S6 transfected with
shNC and shPOSTN in three replicate times. (B) Venn-diagram of DEGs
between shPOSTN sEV vs. CTRL and shPOSTN sEVs vs. shNC sEVs. (C)
Heatmap showing the top DEGs in each group. (D) Volcano plot
showing BMP4 downregulation in macrophages treated with shPOSTN
sEVs vs. shNC sEVs. (E) GSEA analysis of hallmark pathways in
macrophages treated with sEV-derived CAF-S6 transfected with either
shPOSTN or shNC. (F) GO enrichment analysis was performed to
identify pathways associated with the representative DEGs. (G) The
bubble plot displays the top 20 markedly enriched KEGG pathways for
the DEGs. (H) The mRNA expression of BMP4 in macrophages induced
sEVs derived from CAF-S5/-S6 transfected with shNC and shPOSTN were
analyzed by RT-qPCR. (I) The mRNA expression of TNF-α, IL-6, TGF-β
and IL-10 in macrophages stimulated with BMP4 at concentrations of
0, 50 and 100 ng/ml. (J) The expression of CD163 and CD206 in
macrophages treated with BMP4 at concentrations of 0, 100 ng/ml
examined by flow cytometry. (K) The level of BMPR2, pSmad1/5/9,
Smad 5 and Smad 9 in macrophages treated with BMP4 (100 ng/ml) for
48 h were examined by western blotting (n=3 per group). (L) The
level of pSmad1/5/9, Smad5, Smad9, CD163 and CD206 in macrophages
treatment with or without LDN193189 inhibitor (n=3 per group).
Statistical significance was determined using a one-way ANOVA test,
ns, not significant *P<0.05; **P<0.01; ****P<0.0001. Error
bars represent the mean ± SEM. Source data for blotting assays, see
Fig. S5. PCA, principal component
analysis; sEV, small extracellular vesicles; sh, short hairpin; NC,
negative control; DEGs, differentially expressed genes; POSTN,
perostin; sEV, small extracellular vesicles; GSEA, gene set
enrichment analysis; GO, Gene Ontology; KEGG, Kyoto Encyclopedia of
Genes and Genomes; RT-qPCR, reverse transcription-quantitative PCR;
pSmad, phosphorylated Smad. |
GSEA revealed that inflammatory response was
upregulated in the macrophages treated with shPOSTN sEVs compared
with in those treated with shNC sEVs (Fig. 5E). GO analysis revealed that 20
biological processes were markedly altered in macrophages treated
with shPOSTN sEVs compared with shNC sEVs, with notable changes in
‘cellular anatomical entity’, ‘binding’ and ‘cellular processes’
(Fig. 5F). Additionally, KEGG
pathway analysis revealed significant enrichment of pathways, such
as the ‘cytokine-cytokine receptor interaction’ and ‘viral
protein-cytokine receptor interaction’, which are closely
associated with the inflammatory response during macrophage M2
polarization (50,51) (Fig.
5G). The levels of BMP4 in sEV-derived CAF-S5/-S6 transfected
with shNC were higher than those in the shPOSTN group (Fig. S4), average BMP4 level was 2.6 pg/ml
in the shNC group and 1.9 pg/ml in the shPOSTN group. Furthermore,
RT-qPCR analysis showed markedly higher levels of BMP4 in
macrophages treated with sEVs derived from shNC-transfected
CAF-S5/-S6 compared with in those treated with sEVs derived from
shPOSTN-transfected CAF-S5/-S6 (Fig.
5H). Moreover, the mRNA expression levels of pro-inflammatory
factors (TNF-α and IL-6) were markedly downregulated in macrophages
with 50 and 100 ng/ml of BMP4 (Fig.
5I). By contrast, the mRNA expression levels of
anti-inflammatory factors (TGF-β and IL-10) were markedly
upregulated in macrophages with 50 and 100 ng/ml of BMP4 (Fig. 5I). In addition, flow cytometry
showed BMP4-induced upregulation of M2 macrophage markers CD163 and
CD206 (Fig. 5J). Moreover, the
levels of BMPR2 and phospho-Smad1/5/9 were increased in macrophages
treated with 100 ng/ml BMP4 for 48 h (Fig. 5K). To assess the role of Smad
signaling in BMP4-induced M2 polarization, the BMP type I receptor
was inhibited with LDN193189. This treatment suppressed
BMP4-triggered Smad1/5/9 phosphorylation and the expression of
CD163/CD206 (Fig. 5L). These
findings suggested that POSTN-deficient CAF sEVs may decrease BMP4
secretion, which otherwise activates the BMPR2/Smad signaling to
drive macrophage M2 polarization, which could offer a therapeutic
strategy for targeting TAMs.
Discussion
Researchers have increasingly favored employing
scRNA-seq technology to investigate the heterogeneous
characteristics of CAFs in solid tumors (52,53).
The analytical outcomes of scRNA-seq are subject to notable
variability depending on the specific experimental methodology,
with each approach exhibiting unique advantages and technical
constraints (54). Despite its
technical limitations, such as the loss of spatial information and
under-representation of certain cell types due to isolation
challenges (55), scRNA-seq has
been instrumental in identifying distinct CAF subsets, including
myCAF, iCAF and apCAF. These subsets possess unique gene expression
profiles and functional properties, influencing tumor progression
and immune responses through various mechanisms, such as the
secretion of cytokines, chemokines and ECM components (56–58).
Although ICB targeting PD-1/PD-L1 has improved survival in
recurrent or metastatic HNSCC (59,60),
its underlying mechanisms require further elucidation. Notably,
differences in ICB efficacy associated with CAF subgroups have been
reported across diverse solid tumors. Among these,
POSTN+ fibroblasts are characterized by high α-smooth
muscle actin (αSMA) expression and serve crucial roles in ECM
regulation and tissue architecture maintenance (61,62).
Previous studies have revealed that POSTN+ fibroblasts
exhibited characteristics of myCAF, as they expressed
ECM-associated signature genes (POSTN, MMP14 and MMP11) (31,63,64).
To comprehensively characterize the tumor immune
microenvironment in HNSCC, the present study integrated an
PBMC-derived scRNA-seq dataset (GSE139324) alongside primary tumor
data. This approach was necessitated by the limited availability of
HNSCC-specific scRNA-seq resources at the time of analysis and was
implemented to bolster the robustness and diversity of the immune
cell profiling, following rigorous batch effect correction.
Importantly, the inclusion of the PBMC dataset served as a critical
negative control. As expected, fibroblasts markers were absent in
PBMC-derived cells. Specifically, POSTN expression was exclusively
confined to fibroblasts clusters within the tumor samples and was
completely undetectable across all immune cell populations.
The current study deciphered the heterogeneity of
fibroblasts and identified distinct populations of
POSTN− and POSTN+ fibroblasts. Moreover, it
was demonstrated that eCAF and apCAF signatures were highly
expressed in POSTN+ fibroblasts, whereas myCAF and iCAF
signatures were enriched in POSTN− fibroblasts.
Functionally, apCAF serve an essential role in promoting the tumor
immunosuppressive environment by modulating T-cell activity
(65), whereas POSTN+
fibroblasts suppress CD8+ T-cell infiltration, impair
antitumor immunity and foster an immunosuppressive
microenvironment, affecting the response to immunotherapy. Hence,
the potential interaction between POSTN+ apCAF and
macrophages plays a key role in shaping the immunosuppressive
microenvironment of HNSCC. The epithelial-mesenchymal transition
(EMT)-related gene signature serves as a novel mechanistic
explanation for the underlying prognosis of hepatocellular
carcinoma (HCC) (66). EMT is a
crucial step in tumor invasion and metastasis and CAFs serve as the
primary drivers inducing tumor EMT (66). The POSTN+ fibroblasts
identified in the current study inherently possess a robust
mesenchymal phenotype and potent ECM remodeling capacity. These
POSTN+ apCAF may not only directly act on tumor cells by
secreting EMT-inducing factors (such as TGF-β), but could also
indirectly create favorable conditions for tumor cells that have
undergone EMT to evade immune surveillance by constructing an
immunosuppressive microenvironment. At the molecular level, future
investigations should explore between the crosstalk between
POSTN+ apCAF and established oncogenic pathways. A
previous study demonstrated that CLSPN drives HCC progression via
the Wnt/β-catenin pathway (67) and
existing evidence indicates that Wnt signaling is closely
associated with the activation of CAFs and that β-catenin
activation can regulate the expression of multiple chemokines
(67). Therefore, a possible
hypothesis is that aberrant Wnt signaling in tumor cells may
reprogram fibroblasts into a POSTN+ apCAF phenotype,
thereby initiating the macrophage recruitment and activation. This
may represent an important research direction for linking
intracellular oncogenic signaling with systemic remodeling of the
TME. However, it should be noted that the use of integrated public
scRNA-seq datasets may introduce inherent limitations, such as
batch effects or variations in sample processing, which could
affect the interpretation of fibroblast heterogeneity.
According to functional enrichment analysis,
POSTN+ fibroblasts were primarily enriched in genes
regulating collagen metabolism and ECM assembly. Although their
specific mechanisms in driving ICB resistance require full
elucidation, current evidence indicates that they contribute to
immunotherapy resistance by upregulating PD-1 and CTLA4 protein
expression on CD4+CD25+ T lymphocytes
(17). Furthermore,
FAP+αSMA+ CAFs promote collagen production,
forming multi-layered physical barriers that shield tumor cells
from T-cell contact (68), whereas
high levels of LRRC15+ CAFs are associated with poor
immunotherapy outcomes (69).
Furthermore, the current study revealed that patients with HNSCC
and high expression levels of POSTN and FAP, exhibited markedly
poorer responses to ICB therapy. The co-expression of FAP and POSTN
in HNSCC is markedly associated with poor prognosis, supporting
their potential as prognostic biomarkers. The present study
revealed that POSTN and FAP expression levels were positively
associated with macrophage infiltration in TCGA-HNSCC. Further
integrated analysis of TCGA-HNSCC demonstrated that
POSTNhigh/FAPhigh HNSCC tumors exhibited
characteristic immune evasion signatures and poorer ICB response
rates, potentially indicating an immune-resistant phenotype.
H&E and immunohistochemical (IHC) staining identified higher
expression levels of FAP and POSTN in the stroma of OSCC cases
compared with normal controls. However, it is important to note the
inherent limitations of the IHC methodology employed. While
instrumental for validating expression and spatial distribution,
IHC is a semi-quantitative technique susceptible to subjective
interpretation. Moreover, its static nature cannot directly
demonstrate dynamic cellular interactions. For instance, although
antigen-presenting fibroblasts were observed proximal to immune
cells, IHC alone cannot confirm their functional role in antigen
presentation or establish causality within communication networks.
These functional insights warrant future investigation using
techniques such as multiplex fluorescence IHC, transmission
electron microscopy, co-culture models and in vivo
loss-of-function experiments.
Several studies have investigated POSTN+
fibroblasts and SPP1+ macrophages in HNSCC. These
studies have primarily reported associations between individual
gene expression and patient survival, lacking deep insight into
their interactions and effects on the TME (70,71).
Notably, this crosstalk was investigated from multiple aspects in
the current study, including single-cell transcriptomics,
immunofluorescent labeling of clinical specimens and bioinformatics
analysis of published datasets. In the HNSCC cohort from TCGA and
in other scRNA-seq datasets, SPP1+ macrophages have been
shown to interact with POSTN+ fibroblasts, with the
interaction intensity being positively associated with poor overall
survival (72,73). Using integrative single-cell and
spatial transcriptomics, researchers have identified a significant
colocalization of POSTN+ fibroblasts and
SPP1+ macrophages. This finding suggests that their
interaction may serve a critical role in establishing a hypoxic,
TNF-α-rich microenvironment conducive to tumor progression via
NF-κB signaling (74). Furthermore,
the present study highlighted the potential value of
POSTN+ fibroblasts in HNSCC in facilitating an
immunosuppressive microenvironment by inducing macrophage M2
polarization via sEV-POSTN. The robust positive association between
POSTN expression and macrophage infiltration, supported by the
experimental evidence that POSTN+ sEVs may directly
steer M2 polarization through the BMP4/BMPR2/Smad pathway,
underscores a distinct mechanism of immune regulation. To spatially
validate the interplay between POSTN+ fibroblasts and
macrophages, spatial transcriptomics will be further employed to
delineate this precise interplay in HNSCC.
The prominence of POSTN+
fibroblast-macrophage interactions in HNSCC is likely driven by the
distinct etiology and anatomical location of the disease. The
persistent exposure of head and neck mucosa to inflammatory
triggers and microbiota fosters a unique microenvironment that
favors dominant stromal-immune crosstalk (75). Consequently, targeting POSTN or its
downstream signaling in macrophages represents a promising,
disease-specific therapeutic strategy. This approach to alleviate
immunosuppression and overcome ICB resistance may hold greater
value in HNSCC than in malignancies where POSTN+
fibroblasts primarily fulfill other biological roles.
CAFs recruit monocytes and promote their
differentiation into M2-like TAMs through various factors,
including cytokines (CXCL12, CCL5, IL-6 and IL-33) and the
glycoprotein chitinase 3-like-1 (76–78).
In particular, POSTN+ fibroblasts attract and polarize
macrophages into an SPP1+ TAM phenotype via the
IL-6/STAT3 signaling pathway. This process hinders effective T-cell
infiltration and diminishes immunotherapy response (31). Consequently, patients exhibiting
high co-expression of POSTN+ fibroblasts and
SPP1+ macrophages demonstrate poorer outcomes in
immunotherapy cohorts (31). The
present analysis of TCGA-HNSCC data revealed that the expression of
POSTN and FAP was markedly positively associated with M1 macrophage
markers (CD80 and CD86) and M2 macrophage markers (CD163 and
CD206). This notable finding led to the hypothesis that POSTN may
actively modulate M1 macrophages polarization, raising a critical
question for further research. Immunofluorescence staining revealed
the co-expression of POSTN and CD163 in the stromal area of HNSCC
cases, suggesting a potential interaction within the TME. It should
be noted that preliminary analysis was performed on four
representative samples. Future studies with larger sample sizes are
warranted to confirm and extend these findings.
sEVs may also be useful adjuncts to existing
immunotherapeutics. Despite the profound success of ICB, a notable
percentage of patients treated with these agents develop treatment
resistance. PD-L1+ sEVs are used as a potential target,
which impair anti-PD-L1 therapy by binding and targeting
therapeutic antibodies, thereby enhancing their clearance by
macrophages (79). It is expected
that blocking PD-L1 expression or release in EVs may enhance ICB
efficacy and overcome resistance in patients. Human CPC-derived
exosomal vesicles carrying a short POSTN isoform (aa 22–669), have
been shown to promote cardiomyocyte proliferation through
activation of phosphorylation-induced activation of focal adhesion
kinase, actin polymerization and nuclear translocation of
Yes-associated protein signaling (36). Our previous study revealed that LOX
binds the FN/POSTN/BMP4 complex located on the surface of sEVs
(37). Immunogold labeling
localized POSTN to the membrane surface in the present study.
Furthermore, ELISA confirmed that the levels of sEV-POSTN from
CAF-S5/-S6 increased in a concentration-dependent manner and this
signal was markedly reduced by pretreatment with a POSTN-blocking
antibody. Given the membrane localization and functional importance
of sEV-POSTN, a critical next step is to investigate how POSTN
knockdown alters sEVs cargoes and how such alterations subsequently
influence macrophage phenotype and function.
The current study also demonstrated that CAF sEVs
derived POSTN promoted the M2 macrophage polarization by activating
BMP4-induced BMPR2/Smad signaling. These findings suggested that
sEVs are crucial mediators of the crosstalk between CAFs and
macrophages. CAFs have emerged as promising targets for anti-tumor
therapy through the modulation of key signaling pathways (10). To improve chemotherapy and
immunotherapy outcomes, researchers are currently investigating
FAP+ CAF-directed therapies, including antibody-drug
conjugates and CAR-T cells in preclinical and clinical studies
(80,81). However, the clinical benefits of
these approaches for patients with HNSCC remain unclear. The
present study highlighted the role of POSTN+ fibroblasts
in predicting immunotherapy response in HNSCC and identifies
potential targets for enhancing treatment efficacy. Future work
should incorporate retrospective analyses of immunotherapy-treated
HNSCC cohorts and large-scale prospective trials to validate these
findings. Additionally, further investigation is needed to assess
the clinical feasibility of targeting POSTN+
fibroblast-macrophage interactions as a therapeutic strategy.
In conclusion, the present study demonstrated that
POSTN+ fibroblasts may promote an immunosuppressive
microenvironment and ICB resistance in HNSCC by secreting
POSTN+ sEVs that drive M2 macrophages polarization via
BMP4/BMPR2/Smad signaling, highlighting POSTN as a promising
therapeutic potential target.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study received support from the Natural Science
Foundation of China (grant no. 82301087 to Wanqi Lv), General
Program (grant no. SSH-2024-A07 to Xue Liu; SSH-2022-07 to Yanjin
Wang) in Shanghai Stomatological Hospital, Shanghai Stomatological
Hospital's Talent Introduction Startup Project (grant no.
SSH-2025-RC04 to Yuqiong Wu), Outstanding Young Medical Talent
Project of Shanghai Municipal Health Commission (grant no.
2022YQ046 to Si Chen), Outstanding Young Talent Project of Shanghai
Stomatological Hospital (grant no. SSH-2022-KJCX-C02 to Si Chen)
and National Key Clinical Program on Orthodontics (grant no.
GJLCZDZK).
Availability of data and materials
The data generated in the present study are
included in the figures and/or tables of this article, with
additional raw data available from the corresponding author upon
request. The HNSCC scRNA-seq datasets are publicly available from
the GEO repository (GSE103322; http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE103322)
and (GSE139324; http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE139324).
The analysis can be assessed R software. Gene expression data of
TCGA-Head and neck squamous cell carcinoma (HNSCC) patients can be
downloaded at https://www.cbioportal.org/. Immune infiltration
levels were quantified using the single-sample gene set enrichment
analysis (ssGSEA) method. The relevant data and resources can be
downloaded from http://cis.hku.hk/TISIDB/download.php. The raw RNA
sequencing data reported in this paper have been deposited in the
Genome Sequence Archive (82) in
National Genomics Data Center (83), China National Center for
Bioinformation/Beijing Institute of Genomics, Chinese Academy of
Sciences (GSA-Human: HRA015291) that are publicly accessible
at https://ngdc.cncb.ac.cn/gsa-human/submit/hra/submit.
Authors' contributions
XL, CZ and SC were responsible for
conceptualization, visualization, funding acquisition and writing
the original draft. CQ was responsible for methodology and data
curation. YWa and WL were responsible for visualization, formal
analysis, funding acquisition and investigation. QZ and YWu were
responsible for funding acquisition, date curation and formal
analysis. XL and CZ confirm the authenticity of all the raw data.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
All procedures involving human participants were
approved by the Ethics Committee of Shanghai Stomatological
Hospital, Fudan University (approval no. 2024-023). The use of
HNSCC tissue sections was additionally approved by the same
committee (approval no. 2024-005). Written informed consent was
obtained from all individual participants included in the
study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Cohen EEW, Bell RB, Bifulco CB, Burtness
B, Gillison ML, Harrington KJ, Le QT, Lee NY, Leidner R, Lewis RL,
et al: The society for immunotherapy of cancer consensus statement
on immunotherapy for the treatment of squamous cell carcinoma of
the head and neck (HNSCC). J Immunother Cancer. 7:1842019.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
El-Naggar AK, Chan JKC, Grandis JR, Takata
T and Slootweg PJ: WHO Classification of Head and Neck Tumors. 4th
edition. IARC; Lyon: 2017
|
|
3
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI
|
|
4
|
Liu JC, Bhayani M, Kuchta K, Galloway T
and Fundakowski C: Patterns of distant metastasis in head and neck
cancer at presentation: Implications for initial evaluation. Oral
Oncol. 88:131–136. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ferris RL, Blumenschein G Jr, Fayette J,
Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE,
Even C, et al: Nivolumab for recurrent squamous-cell carcinoma of
the head and neck. N Engl J Med. 375:1856–1867. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Harrington KL, Burtness B, Greil R,
Soulières D, Tahara M, de Castro G Jr, Psyrri A, Brana I, Basté N,
Neupane P, et al: Pembrolizumab with or without chemotherapy in
recurrent or metastatic head and neck squamous cell carcinoma:
Updated results of the phase III KEYNOTE-048 study. J Clin Oncol.
41:790–802. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Morad G, Helmink BA, Sharma P and Wargo
JA: Hallmarks of response, resistance, and toxicity to immune
checkpoint blockade. Cell. 184:5309–5337. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Elhanani O, Ben-Uri R and Keren L: Spatial
profiling technologies illuminate the tumor microenvironment.
Cancer Cell. 41:404–420. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang K, Wang X, Song C, He Z, Wang R, Xu
Y, Jiang Y, Wan Y, Mei J and Mao W: The role of lipid metabolic
reprogramming in tumor microenvironment. Theranostics.
13:1774–1808. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen X and Song E: Turning foes to
friends: Targeting cancer- associated fibroblasts. Nat Rev Drug
Discov. 18:99–115. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mao X, Xu J, Wang W, Liang C, Hua J, Liu
J, Zhang B, Meng Q, Yu X and Shi S: Crosstalk between
cancer-associated fibroblasts and immune cells in the tumor
microenvironment: New findings and future perspectives. Mol Cancer.
20:1312021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Barrett R and Puré E: Cancer-associated
fibroblasts: Key determinants of tumor immunity and immunotherapy.
Curr Opin Immunol. 64:80–87. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Deng Y, Cheng J, Fu B, Liu W, Chen G,
Zhang Q and Yang Y: Hepatic carcinoma-associated fibroblasts
enhance immune suppression by facilitating the generation of
myeloid-derived suppressor cells. Oncogene. 36:1090–1101. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hsu CY, Saleh RO, Mohammed JS, Mansuri N,
Rekha MM, Anand A, Sahoo S, Zwamel SS, Zwamel AH and Hulail HM: The
dynamic interplay between melanoma cells and CAFs: Implications
drug resistance and immune evasion and possible therapeutics. Exp
Cell Res. 449:1145812025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wein L, Luen SJ, Savas P, Salgado R and
Loi S: Checkpoint blockade in the treatment of breast cancer:
Current status and future directions. Br J Cancer. 119:4–11. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Loi S, Michiels S, Adams S, Loibl S,
Budczies J, Denkert C and Salgado R: The journey of
tumor-infiltrating lymphocytes as a biomarker in breast cancer:
Clinical utility in an era of checkpoint inhibition. Ann Oncol.
32:1236–1244. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kieffer Y, Hocine HR, Gentric G, Pelon F,
Bernard C, Bourachot B, Lameiras S, Albergante L, Bonneau C, Guyard
A, et al: Single-cell analysis reveals fibroblast clusters linked
to immunotherapy resistance in cancer. Cancer Discov. 10:1330–1351.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Miyai Y, Sugiyama D, Hase T, Asai N, Taki
T, Nishida K, Fukui T, Chen-Yoshikawa TF, Kobayashi H, Mii S, et
al: Meflin-positive cancer-associated fibroblasts enhance tumor
response to immune checkpoint blockade. Life Sci Alliance.
5:e2021012302022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Krishnamurty AT, Shyer JA, Thai M, Gandham
V, Buechler MB, Yang YA, Pradhan RN, Wang AW, Sanchez PL, Qu Y, et
al: LRRC15+ myofibroblasts dictate the stromal setpoint
to suppress tumour immunity. Nature. 611:148–154. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Galbo PM Jr, Zang X and Zheng D: Molecular
features of cancer-associated fibroblast subtypes and their
implication on cancer pathogenesis, prognosis, and immunotherapy
resistance. Clin Cancer Res. 27:2636–2647. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang LX, Zhang SX, Wu HJ, Rong XL and Guo
J: M2b macrophage polarization and its roles in diseases. J Leukoc
Biol. 106:345–358. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Funes SC, Rios M, Escobar-Vera J and
Kalergis AM: Implications of macrophage polarization in
autoimmunity. Immunology. 154:186–195. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mehraj U, Dar AH, Wani NA and Mir MA:
Tumor microenvironment promotes breast cancer chemoresistance.
Cancer Chemother Pharmacol. 87:147–158. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jin X, Deng Q, Ye S, Liu S, Fu Y, Liu Y,
Wu G, Ouyang G and Wu T: Cancer-associated fibroblast-derived
periostin promotes papillary thyroid tumor growth through
integrin-FAK-STAT3 signaling. Theranostics. 14:3014–3028. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhu M, Fejzo MS, Anderson L, Dering J,
Ginther C, Ramos L, Gasson JC, Karlan BY and Slamon DJ: Periostin
promotes ovarian cancer angiogenesis and metastasis. Gynecol Oncol.
119:337–344. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Contié S, Voorzanger-Rousselot N, Litvin
J, Clézardin P and Garnero P: Increased expression and serum levels
of the stromal cellsecreted protein periostin in breast cancer bone
metastases. Int J Cancer. 128:352–360. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ryner L, Guan Y, Firestein R, Xiao Y, Choi
Y, Rabe C, Lu S, Fuentes E, Huw LY, Lackner MR, et al: Upregulation
of periostin and reactive stroma is associated with primary
chemoresistance and predicts clinical outcomes in epithelial
ovarian cancer. Clin Cancer Res. 21:2941–2951. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jeong JY, Jeong W and Kim HJ: Promotion of
chondrosarcoma cell survival, migration and lymphangiogenesis by
periostin. Anticancer Res. 40:5463–5469. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhou W, Ke SQ, Huang Z, Flavahan W, Fang
X, Paul J, Wu L, Sloan AE, McLendon RE, Li X, et al: Periostin
secreted by glioblastoma stem cells recruits M2 tumourassociated
macrophages and promotes malignant growth. Nat Cell Biol.
17:170–182. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tang M, Liu B, Bu X and Zhao P: Crosstalk
between ovarian cancer cells and macrophages through periostin
promotes macrophage recruitment. Cancer Sci. 109:1309–1318. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang H, Liang Y, Liu Z, Zhang R, Chao J,
Wang M, Liu M, Qiao L, Xuan Z, Zhao H and Lu L: POSTN+
cancer-associated fibroblasts determine the efficacy of
immunotherapy in hepatocellular carcinoma. J Immunother Cancer.
12:e0087212024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen C, Guo Q, Liu Y, Hou Q, Liao M, Guo
Y, Zang Y, Wang F, Liu H, Luan X, et al: Single-cell and spatial
transcriptomics reveal POSTN+ cancer-associated
fibroblasts correlated with immune suppression and tumour
progression in non-small cell lung cancer. Clin Transl Med.
13:e15152023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
van Niel G, D'Angelo G and Raposo G:
Shedding light on the cell biology of extracellular vesicles. Nat
Rev Mol Cell Biol. 19:213–228. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Welsh JA, Goberdhan DCI, O'Driscoll L,
Buzas EI, Blenkiron C, Bussolati B, Cai H, Di Vizio D, Deiedonks
TAP, Erdbrügger U, et al: Minimal information for studies of
extracellular vesicles (MISEV2023): From basic to advanced
approaches. J Extracell Vesicles. 13:e124042024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Théry C, Witwer KW, Aikawa E, Alcaraz MJ,
Anderson JD, Andriantsitohaina R, Antoniou A, Arab T, Archer F,
Atkin-Smith GK, et al: Minimal information for studies of
extracellular vesicles 2018 (MISEV2018): A position statement of
the international society for extracellular vesicles and update of
the MISEV2014 guidelines. J Extracell Vesicles. 7:15357502018.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
You T, Tang H, Wu W, Gao J, Li X, Li N, Xu
X, Xing J, Ge H, Xiao Y, et al: POSTN secretion by extracellular
matrix cancer-associated fibroblasts (eCAFs) correlates with poor
ICB response via macrophage chemotaxis activation of Akt signaling
pathway in gastric cancer. Aging Dis. 14:2177–2192. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Liu X, Li H, Wang Y, Zhang Q, Liu Y and
Liu T: LOX+ iCAFs in HNSCC have the potential to predict
prognosis and immunotherapy responses revealed by single cell RNA
sequencing analysis. Sci Rep. 15:70282025. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ruffin AT, Cillo AR, Tabib T, Liu A, Onkar
S, Kunning SR, Lampenfeld C, Atiya HI, Abecassis I, Kürten CHL, et
al: B cell signatures and tertiary lymphoid structures contribute
to outcome in head and neck squamous cell carcinoma. Nat Commun.
12:33492021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Cillo AR, Kürten CHL, Tabib T, Qi Z, Onkar
S, Wang T, Liu A, Duvvuri U, Kim S, Soose RJ, et al: Immune
landscape of viral- and carcinogen-driven head and neck cancer.
Immunity. 52:183–199.e9. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Sung BH, Ketova T, Hoshino D, Zijlstra A
and Weaver AM: Directional cell movement through tissues is
controlled by exosome secretion. Nat Commun. 6:71642015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-delta delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wang L, Feng Z, Wang X, Wang X and Zhang
X: DEGseq: An R package for identifying differentially expressed
genes from RNA-seq data. Bioinformatics. 26:136–138. 2019.
View Article : Google Scholar
|
|
43
|
Xie C, Mao X, Huang J, Ding Y, Wu J, Dong
S, Kong L, Gao G, Li CY and Wei L: KOBAS 2.0: A web server for
annotation and identification of enriched pathways and diseases.
Nucleic Acids Res. 39:W316–W322. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Hong HY, Hwang DY, Yang CC, Cheng SM, Chen
PC, Aala WJ, I-Chen Harn H, Evans ST, Onoufriadis A, Liu SL, et al:
Profibrotic subsets of SPP1+ macrophages and
POSTN+ fibroblasts contribute to fibrotic scarring in
acne keloidalis. J Invest Dernatol. 144:1491–1504.e10. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wu S, Liu M, Zhang M, Ye X, Gu H, Jiang C,
Zhu H, Ye X, Li Q, Huang X and Cao M: The gene expression of CALD1,
CDH2, and POSTN in fibroblast are related to idiopathic pulmonary
fibrosis. Front Immunol. 15:12750642024. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Puram SV, Tirosh I, Parikh AS, Patel AP,
Yizhak K, Gillespie S, Rodman C, Luo CL, Mroz EA, Emerick KS, et
al: Single-cell transcriptomic analysis of primary and metastatic
tumor ecosystems in head and neck cancer. Cell. 171:1611–1624.e24.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Buechler MB, Fu W and Turley SJ:
Fibroblast-macrophage reciprocal interactions in health, fibrosis,
and cancer. Immunity. 54:903–915. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yang AT, Kim YO, Yan XZ, Abe H, Aslam M,
Park KS, Zhao XY, Jia JD, Klein T, You H and Schuppan D: Fibroblast
activation protein activates macrophages and promotes parenchymal
liver inflammation and fibrosis. Cell Mol Gastroenterol Hepatol.
15:841–867. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Martínez VG, Rubio C, Martínez-Fernández
M, Segovia C, López-Calderón F, Garín MI, Teijeira A,
Munera-Maravilla E, Varas A, Sacedón R, et al: BMP4 induces M2
macrophage polarization and favors tumor progression in bladder
cancer. Clin Cancer Res. 23:7388–7399. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wang Y, Liu S, Li L, Li L, Zhou X, Wan M,
Lou P, Zhao M, Lv K, Yuan Y, et al: Peritoneal M2
macrophage-derived extracellular vesicles as natural multitarget
nanotherapeutics to attenuate cytokine storms after severe
infections. J Control Release. 349:118–132. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Su D, Zhu S, Xu K, Hou Z, Hao F, Xu F, Lin
Y, Zhu Y, Liu D, Duan Q, et al: Phosphoproteomic analysis reveals
changes in A-Raf-related protein phosphorylation in response to
toxoplasma gondii infection in porcine macrophages. Parasit
Vectors. 17:1912024. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Friedman G, Levi-Galibov O, David E,
Bornstein C, Giladi A, Dadiani M, Mayo A, Halperin C,
Pevsner-Fischer M, Lavon H, et al: Cancer-associated fibroblast
compositions change with breast cancer progression linking the
ratio of S100A4+ and PDPN+ CAFs to clinical
outcome. Nat Cancer. 1:692–708. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Bartoschek M, Oskolkov N, Bocci M, Lövrot
J, Larsson C, Sommarin M, Madsen CD, Lindgren D, Pekar G, Karlsson
G, et al: Spatially and functionally distinct subclasses of breast
cancer-associated fibroblasts revealed by single cell RNA
sequencing. Nat Commun. 9:51502018. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Hwang B, Lee JH and Bang D: Single-cell
RNA sequencing technologies and bioinformatics pipelines. Exp Mol
Med. 50:1–14. 2018. View Article : Google Scholar
|
|
55
|
Moncada R, Barkley D, Wagner F, Chiodin M,
Devlin JC, Baron M, Hajdu CH, Simeone DM and Yanai I: Integrating
microarray-based spatial transcriptomics and single-cell RNA-seq
reveals tissue architecture in pancreatic ductal adenocarcinomas.
Nat Biotechnol. 38:333–342. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Öhlund D, Handly-Santana A, Biffi G,
Elyada E, Almeida AS, Ponz-Sarvise M, Corbo V, Oni TE, Hearn SA,
Lee EJ, et al: Distinct populations of inflammatory fibroblasts and
myofibroblasts in pancreatic cancer. J Exp Med. 214:579–596. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Song J, Wei R, Liu C, Zhao Z and Liu X,
Wang Y, Liu F and Liu X: Antigen-presenting cancer associated
fibroblasts enhance antitumor immunity and predict immunotherapy
response. Nat Commun. 16:21752025. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Maia A and Wiemann S: Cancer-associated
fibroblasts: Implications for cancer therapy. Cancers (Basel).
13:35262021. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Daste A, Larroquette M, Gibson N, Lasserre
M and Domblides C: Immunotherapy for head and neck squamous cell
carcinoma: Current status and perspectives. Immunotherapy.
16:187–197. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Aboaid H, Khalid T, Hussain A, Myat YM,
Nanda RK, Srinivasmurthy R, Nguyen K, Jones DT, BigcasJ L and Thein
KZ: Advances and challenges in immunotherapy in head and neck
cancer. Front Immunol. 16:15965832025. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Xing F, Saidou J and Watabe K: Cancer
associated fibroblasts (CAFs) in tumor microenvironment. Front
Biosci (Landmark Ed). 15:166–179. 2021. View Article : Google Scholar
|
|
62
|
Kim I, Choi S, Yoo S, Lee M and Kim IS:
Cancer-associated fibroblasts in the hypoxic tumor
microenvironment. Cancers (Basel). 14:33212022. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Wu Y, Li S, Yu H, Zhang S, Yan L, Guan X,
Xu W, Wang Z, Lv A, Tian X, et al: Integrative single-cell and
spatial transcriptomics analysis reveals ECM-remodeling
cancer-associated fibroblast-derived POSTN as a key mediator in
pancreatic ductal adenocarcinoma progression. Int J Biol Sci.
21:3573–3596. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Liu Y, Dong G, Yu J and Liang P:
Integration of single-cell and spatial transcriptomics reveals
fibroblast subtypes in hepatocellular carcinoma: Spatial
distribution, differentiation trajectories, and therapeutic
potential. J Transl Med. 23:1982025. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Thomas ME, Jie E, Kim AM, Mayberry TG,
Cowan BC, Luechtefeld HD, Wakefield MR and Fang Y: Exploring the
role of antigen-presenting cancer-associated fibroblasts and CD74
on the pancreatic ductal adenocarcinoma tumor microenvironment. Med
Oncol. 42:152024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Shi Y, Wang J, Huang G, Zhu J, Jian H, Xia
G, Wei Q, Li Y and Yu H: A novel epithelial-mesenchymal transition
gene signature for the immune status and prognosis of
hepatocellular carcinoma. Hepatol Int. 16:906–917. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Shi Y, Wang Y, Niu K, Zhang W, Lv Q and
Zhang Y: How CLSPN could demystify its prognostic value and
potential molecular mechanism for hepatocellular carcinoma: A
crosstalk study. Comput Biol Med. 172:1082602024. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Grout JA, Sirven P, Leader AM, Maskey S,
Hector E, Puisieux I, Steffan F, Cheng E, Tung N, Maurin M, et al:
Spatial positioning and matrix programs of cancer-associated
fibroblasts promote T cell exclusion in human lung tumors. Cancer
Discov. 12:2606–2625. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Dominguez CX, Müller S, Keerthivasan S,
Koeppen H, Hung J, Gierke S, Breart B, Foreman O, Bainbridge TW,
Castiglioni A, et al: Single-cell RNA sequencing reveals stromal
evolution into LRRC15+ myofibroblasts as a determinant
of patient response to cancer immunotherapy. Cancer Discov.
10:232–253. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Wenhua S, Tsunematsu T, Umeda M, Tawara H,
Fujiwara N, Mouri Y, Arakaki R, Ishimaru N and Kudo Y: Cancer
cell-derived novel periostin isoform promotes invasion in head and
neck squamous cell carcinoma. Cancer Med. 12:8510–8525. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Bie T and Zhang X: Higher expression of
SPP1 predicts poorer survival outcomes in head and neck cancer. J
Immunol Res. 2021:85695752021. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Qi J, Sun H, Zhang Y, Wang Z, Xun Z, Li Z,
Ding X, Bao R, Hong L, Jia W, et al: Single-cell and spatial
analysis reveal interaction of FAP+ fibroblasts and
SPP1+ macrophages in colorectal cancer. Nat Commun.
13:17422022. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Khaliq AM, Erdogan C, Kurt Z, Turgut SS,
Grunvald MW, Rand T, Khare S, Borgia JA, Hayden DM, Pappas SG, et
al: Refining colorectal cancer classification and clinical
stratification through a single-cell atlas. Genome Biol.
23:1132022. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Wu J, Dai T, Li Z, Pan M, Zhang W, Chen H,
Qiao L, Lian Q, Liu Y and Chen J: Integrating multi-modal
transcriptomics identifies cellular subtypes with distinct roles in
PDAC progression. Cell Oncol (Dordr). 48:1571–1592. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Oppel F, Gendreizig S, Martinez-Ruiz L,
Florido J, López-Rodríguez A, Pabla H, Loganathan L, Hose L, Kühnel
P, Schmidt P, et al: Mucosa-like differentiation of head and neck
cancer cells is inducible and drives the epigenetic loss of cell
malignancy. Cell Death Dis. 15:7242024. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Tan B, Shi X, Zhang J, Qin J, Zhang N, Ren
H, Qian M, Siwko S, Carmon K, Liu Q, et al: Inhibition of Rspo-Lgr4
facilitates checkpoint blockade therapy by switching macrophage
polarization. Cancer Res. 7817:4929–4942. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Ksiazkiewicz M, Gottfried E, Kreutz M,
Mack M, Hofstaedter F and Kunz-Schughart LA: Importance of
CCL2-CCR2A/2B signaling for monocyte migration into spheroids of
breast cancer-derived fibro blasts. Immunobiology. 215:737–747.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Cohen N, Shani O, Raz Y, Sharon Y, Hoffman
D, Abramovitz L and Erez N: Fibroblasts drive an immunosuppressive
and growth promoting microenvironment in breast cancer via
secretion of Chitinase 3-like 1. Oncogene. 36:4457–4468. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Najafi S, Majidpoor J and Mortezaee K:
Extracellular vesicle-based drug delivery in cancer immunotherapy.
Drug Deliv Transl Res. 13:2790–2806. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Fabre M, Ferrer C, Dominguez-Hormaetxe S,
Bockorny B, Murias L, Seifert O, Eisler SA, Kontermann RE,
Pfizenmaier K, Lee SY, et al: OMTX705, a novel FAP-targeting ADC
demonstrates activity in chemotherapy and pembrolizumab-resistant
solid tumor models. Clin Cancer Res. 26:3420–3430. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wang LC, Lo A, Scholler J, Sun J, Majumdar
RS, Kapoor V, Antzis M, Cotner CE, Johnson LA, Durham AC, et al:
Targeting fibroblast activation protein in tumor stroma with
chimeric antigen receptor T cells can inhibit tumor growth and
augment host immunity without severe toxicity. Cancer Immunol Res.
2:154–166. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
CNCB-NGDC Members and Partners, . Database
resources of the national genomics data center, China national
center for bioinformation in 2025. Nucleic Acids Res. 53:D30–D44.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Zhang S, Chen X, Jin E, Wang A, Chen T,
Zhang X, Zhu J, Dong L, Sun Y, Yu C, et al: The GSA family in 2025:
A broadened sharing platform for multi-omics and multimodal data.
Genomics Proteomics Bioinformatics. 23:qzaf0722025. View Article : Google Scholar : PubMed/NCBI
|



















