|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018.PubMed/NCBI
|
|
2
|
GBD 2019 Colorectal Cancer Collaborators,
. Global, regional, and national burden of colorectal cancer and
its risk factors, 1990–2019: A systematic analysis for the Global
Burden of disease study 2019. Lancet Gastroenterol Hepatol.
7:627–647. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mauri G, Patelli G, Crisafulli G, Siena S
and Bardelli A: Tumor ‘age’ in early-onset colorectal cancer. Cell.
188:589–593. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhou J, Yang Q, Zhao S, Sun L, Li R, Wang
J, Wang L and Wang D: Evolving landscape of colorectal cancer:
Global and regional burden, risk factor dynamics, and future
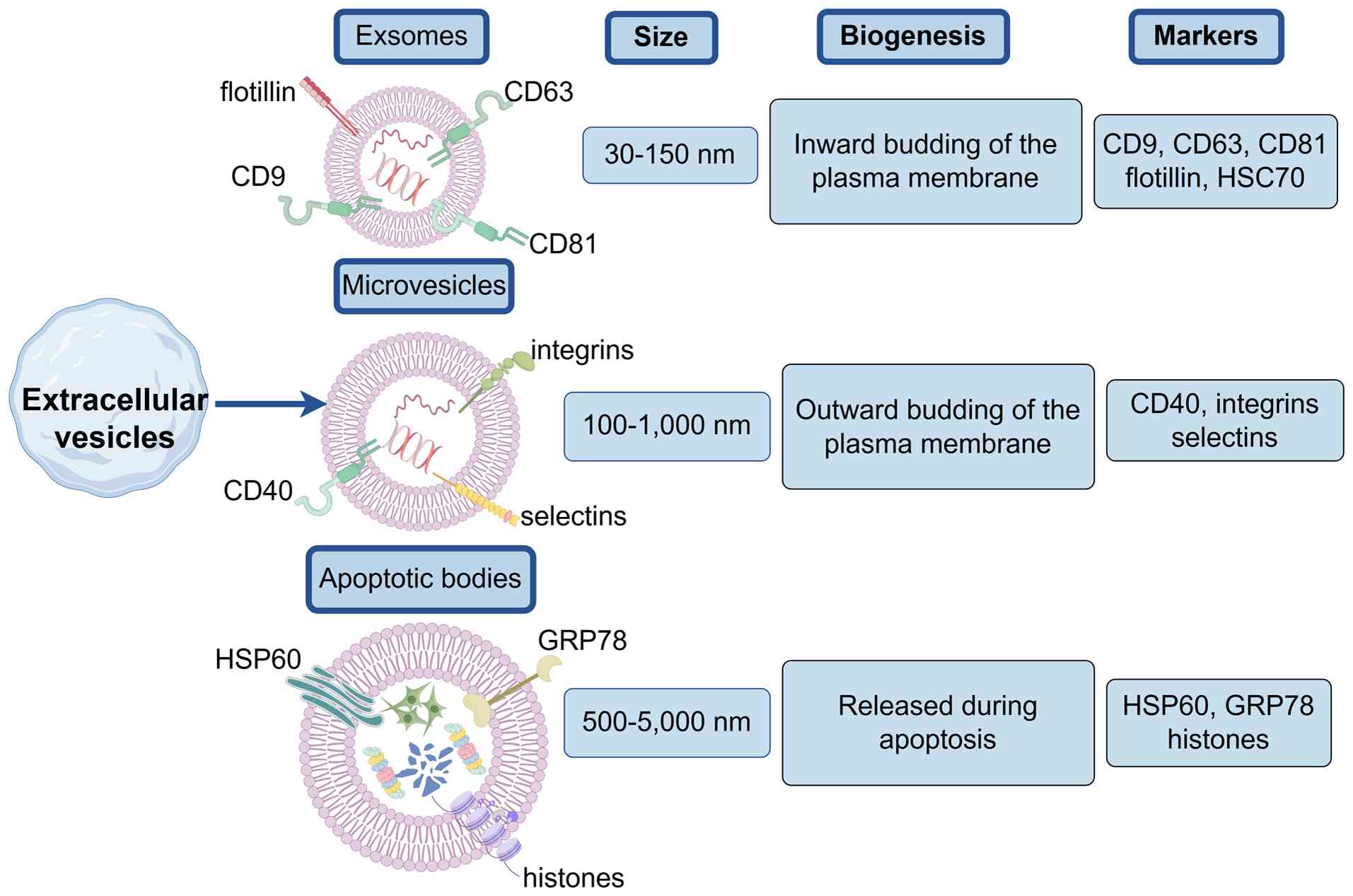
scenarios (the Global Burden of Disease 1990–2050). Ageing Res Rev.
104:1026662025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kim BJ and Hanna MH: Colorectal cancer in
young adults. J Surg Oncol. 127:1247–1251. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bouvard V, Loomis D, Guyton KZ, Grosse Y,
Ghissassi FE, Benbrahim-Tallaa L, Guha N, Mattock H and Straif K;
International Agency for Research on Cancer Monograph Working
Group, : Carcinogenicity of consumption of red and processed meat.
Lancet Oncol. 16:1599–1600. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Johnson CH, Dejea CM, Edler D, Hoang LT,
Santidrian AF, Felding BH, Ivanisevic J, Cho K, Wick EC,
Hechenbleikner EM, et al: Metabolism links bacterial biofilms and
colon carcinogenesis. Cell Metab. 21:891–897. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dejea CM, Wick EC, Hechenbleikner EM,
White JR, Mark Welch JL, Rossetti BJ, Peterson SN, Snesrud EC,
Borisy GG, Lazarev M, et al: Microbiota organization is a distinct
feature of proximal colorectal cancers. Proc Natl Acad Sci USA.
111:18321–18326. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rosato V, Bosetti C, Levi F, Polesel J,
Zucchetto A, Negri E and La Vecchia C: Risk factors for young-onset
colorectal cancer. Cancer Cause Control. 24:335–341.
2013.PubMed/NCBI
|
|
10
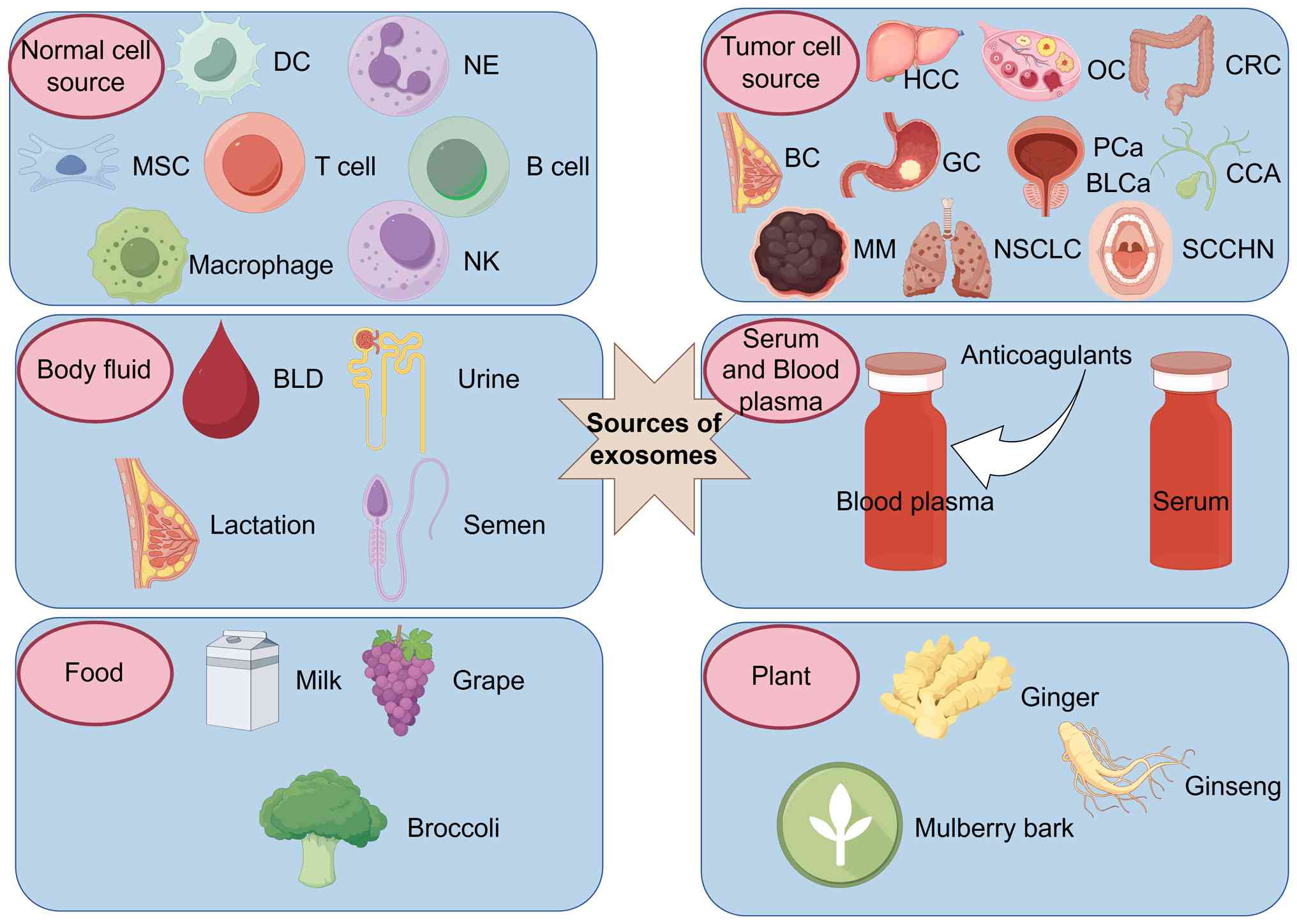
|
Zhang Y, Luo J, Yang W and Ye WC: CircRNAs
in colorectal cancer: Potential biomarkers and therapeutic targets.
Cell Death Dis. 14:3532023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang W, Zheng H, Lv W and Zhu Y: Current
status and prospect of immunotherapy for colorectal cancer. Int J
Colorectal Dis. 38:2662023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Abedizadeh R, Majidi F, Khorasani HR,
Abedi H and Sabour D: Colorectal cancer: A comprehensive review of
carcinogenesis, diagnosis, and novel strategies for classified
treatments. Cancer Metastasis Rev. 43:729–753. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Arjmand B, Alavi-Moghadam S, Faraji Z,
Aghajanpoor-Pasha M, Jalaeikhoo H, Rajaeinejad M, Nikandish M,
Faridfar A, Rezazadeh-Mafi A, Rezaei-Tavirani M and Irompour A: The
potential role of intestinal stem cells and microbiota for the
treatment of colorectal cancer. Adv Exp Med Biol. 1470:115–128.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Logozzi M, Di Raimo R, Mizzoni D and Fais
S: What we know on the potential use of exosomes for nanodelivery.
Semin Cancer Biol. 86:13–25. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lai RC, Yeo RW, Tan KH and Lim SK:
Exosomes for drug delivery-a novel application for the mesenchymal
stem cell. Biotechnol Adv. 31:543–551. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lai RC, Chen TS and Lim SK: Mesenchymal
stem cell exosome: A novel stem cell-based therapy for
cardiovascular disease. Regener Med. 6:481–492. 2011. View Article : Google Scholar
|
|
17
|
Nabariya DK, Pallu R and Yenuganti VR:
Exosomes: The protagonists in the tale of colorectal cancer?
Biochim Biophys Acta Rev Cancer. 1874:1884262020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang M, Hu S, Liu L, Dang P, Liu Y, Sun
Z, Qiao B and Wang C: Engineered exosomes from different sources
for cancer-targeted therapy. Signal Transduct Target Ther.
8:1242023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Guo G, Tan Z, Liu Y, Shi F and She J: The
therapeutic potential of stem cell-derived exosomes in the
ulcerative colitis and colorectal cancer. Stem Cell Res Ther.
13:1382022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hu JL, Wang W, Lan XL, Zeng ZC, Liang YS,
Yan YR, Song FY, Wang FF, Zhu XH, Liao WJ, et al: CAFs secreted
exosomes promote metastasis and chemotherapy resistance by
enhancing cell stemness and epithelial-mesenchymal transition in
colorectal cancer. Mol Cancer. 18:912019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Brennan CW, Verhaak RG, McKenna A, Campos
B, Noushmehr H, Salama SR, Zheng S, Chakravarty D, Sanborn JZ,
Berman SH, et al: The somatic genomic landscape of glioblastoma.
Cell. 155:462–477. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Grady WM and Pritchard CC: Molecular
alterations and biomarkers in colorectal cancer. Toxicol Pathol.
42:124–139. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Parsons DW, Wang TL, Samuels Y, Bardelli
A, Cummins JM, DeLong L, Silliman N, Ptak J, Szabo S, Willson JK,
et al: Colorectal cancer: mutations in a signalling pathway.
Nature. 436:7922005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bardelli A, Parsons DW, Silliman N, Ptak
J, Szabo S, Saha S, Markowitz S, Willson JK, Parmigiani G, Kinzler
KW, et al: Mutational analysis of the tyrosine kinome in colorectal
cancers. Science. 300:9492003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kuipers EJ, Grady WM, Lieberman D,
Seufferlein T, Sung JJ, Boelens PG, van de Velde CJ and Watanabe T:
Colorectal cancer. Nat Rev Dis Primers. 1:150652015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
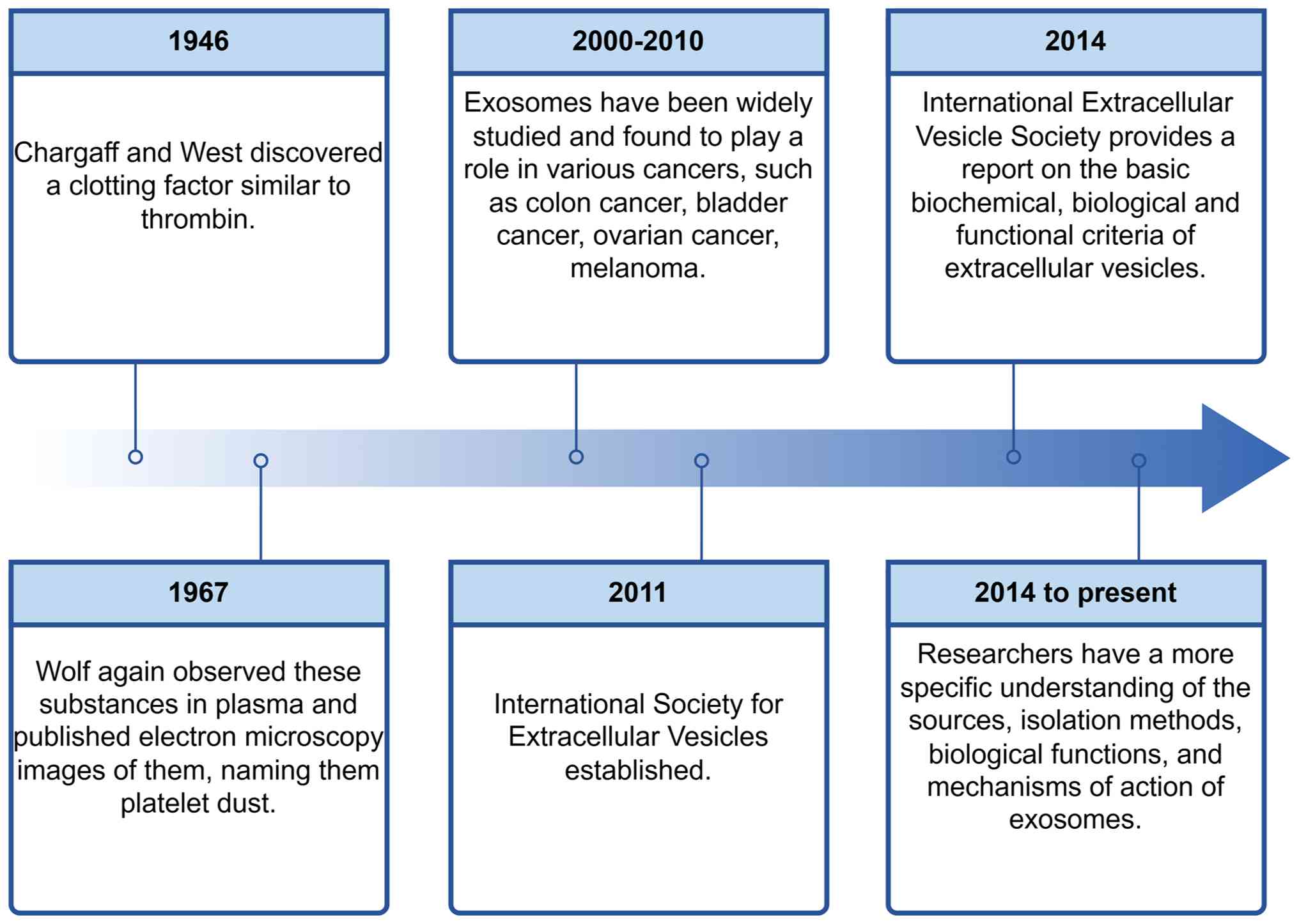
Chargaff E and West R: The biological
significance of the thromboplastic protein of blood. J Biol Chem.
166:189–197. 1946. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wolf P: The nature and significance of
platelet products in human plasma. Br J Haematol. 13:269–288. 1967.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Harding C, Heuser J and Stahl P:
Receptor-mediated endocytosis of transferrin and recycling of the
transferrin receptor in rat reticulocytes. J Cell Biol. 97:329–339.
1983. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pan BT, Teng K, Wu C, Adam M and Johnstone
RM: Electron microscopic evidence for externalization of the
transferrin receptor in vesicular form in sheep reticulocytes. J
Cell Biol. 101:942–948. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Alzhrani GN, Alanazi ST, Alsharif SY,
Albalawi AM, Alsharif AA, Abdel-Maksoud MS and Elsherbiny N:
Exosomes: Isolation, characterization, and biomedical applications.
Cell Biol Int. 45:1807–1831. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Simpson RJ, Kalra H and Mathivanan S:
ExoCarta as a resource for exosomal research. J Extracell Vesicles.
1:183742012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mathivanan S, Lim JWE, Tauro BJ, Ji H,
Moritz RL and Simpson RJ: Proteomics analysis of A33
immunoaffinity-purified exosomes released from the human colon
tumor cell line LIM1215 reveals a tissue-specific protein
signature. Mol Cell Proteomics. 9:197–208. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Welton JL, Khanna S, Giles PJ, Brennan P,
Brewis IA, Staffurth J, Mason MD and Clayton A: Proteomics analysis
of bladder cancer exosomes. Mol Cell Proteomics. 9:1324–1338. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Runz S, Keller S, Rupp C, Stoeck A, Issa
Y, Koensgen D, Mustea A, Sehouli J, Kristiansen G and Altevogt P:
Malignant ascites-derived exosomes of ovarian carcinoma patients
contain CD24 and EpCAM. Gynecol Oncol. 107:563–571. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mears R, Craven RA, Hanrahan S, Totty N,
Upton C, Young SL, Patel P, Selby PJ and Banks RE: Proteomic
analysis of melanoma-derived exosomes by two-dimensional
polyacrylamide gel electrophoresis and mass spectrometry.
Proteomics. 4:4019–4031. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sivalingam AM and Sureshkumar DD: Exosomes
in regulating miRNAs for biomarkers of neurodegenerative disorders.
Mol Neurobiol. 62:7576–7596. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gao S, Dong Y, Yan C, Yu T and Cao H: The
role of exosomes and exosomal microRNA in diabetic cardiomyopathy.
Front Endocrinol (Lausanne). 14:13274952024. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mei R, Qin W, Zheng Y, Wan Z and Liu L:
Role of adipose tissue derived exosomes in metabolic disease. Front
Endocrinol (Lausanne). 13:8738652022. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li Y, Tang Y and Yang GY: Therapeutic
application of exosomes in ischaemic stroke. Stroke Vasc Neurol.
6:483–495. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lötvall J, Hill AF, Hochberg F, Buzás EI,
Di Vizio D, Gardiner C, Gho YS, Kurochkin IV, Mathivanan S,
Quesenberry P, et al: Minimal experimental requirements for
definition of extracellular vesicles and their functions: A
position statement from the International society for extracellular
vesicles. J Extracell Vesicles. 3:269132014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang Y, Bi J, Huang J, Tang Y, Du S and
Li P: Exosome: A review of its classification, isolation
techniques, storage, diagnostic and targeted therapy applications.
Int J Nanomedicine. 15:6917–6934. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kalluri R and LeBleu VS: The biology,
function, and biomedical applications of exosomes. Science.
367:eaau69772020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chia BS, Low YP, Wang Q, Li P and Gao Z:
Advances in exosome quantification techniques. Trends Anal Chem.
86:93–106. 2017. View Article : Google Scholar
|
|
44
|
Langevin SM, Kuhnell D, Orr-Asman MA,
Biesiada J, Zhang X, Medvedovic M and Thomas HE: Balancing yield,
purity and practicality: A modified differential
ultracentrifugation protocol for efficient isolation of small
extracellular vesicles from human serum. RNA Biol. 16:5–12. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Gardiner C, Di Vizio D, Sahoo S, Théry C,
Witwer KW, Wauben M and Hill AF: Techniques used for the isolation
and characterization of extracellular vesicles: Results of a
worldwide survey. J Extracell Vesicles. 5:329452016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Karimi N, Cvjetkovic A, Jang SC,
Crescitelli R, Hosseinpour Feizi MA, Nieuwland R, Lötvall J and
Lässer C: Detailed analysis of the plasma extracellular vesicle
proteome after separation from lipoproteins. Cell Mol Life Sci.
75:2873–2886. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Navajas R, Corrales FJ and Paradela A:
Serum exosome isolation by size-exclusion chromatography for the
discovery and validation of preeclampsia-associated biomarkers.
Methods Mol Biol. 1959:39–50. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Witwer KW, Buzás EI, Bemis LT, Bora A,
Lässer C, Lötvall J, Nolte-'t Hoen EN, Piper MG, Sivaraman S, Skog
J, et al: Standardization of sample collection, isolation and
analysis methods in extracellular vesicle research. J Extracell
Vesicles. 2:2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Cheng H, Fang H, Xu RD, Fu MQ, Chen L,
Song XY, Qian JY, Zou YZ, Ma JY and Ge JB: Development of a rinsing
separation method for exosome isolation and comparison to
conventional methods. Eur Rev Med Pharmacol Sci. 23:5074–5083.
2019.PubMed/NCBI
|
|
50
|
Soares Martins T, Catita J, Martins Rosa
I, A B da Cruz E Silva O and Henriques AG: Exosome isolation from
distinct biofluids using precipitation and column-based approaches.
PLoS One. 13:e01988202018. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
García-Romero N, Madurga R, Rackov G,
Palacín-Aliana I, Núñez-Torres R, Asensi-Puig A, Carrión-Navarro J,
Esteban-Rubio S, Peinado H, González-Neira A, et al: Polyethylene
glycol improves current methods for circulating extracellular
vesicle-derived DNA isolation. J Transl Med. 17:752019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
He L, Zhu D, Wang J and Wu X: A highly
efficient method for isolating urinary exosomes. Int J Mol Med.
43:83–90. 2019.PubMed/NCBI
|
|
53
|
Konoshenko MY, Lekchnov EA, Vlassov AV and
Laktionov PP: Isolation of extracellular vesicles: General
methodologies and latest trends. Biomed Res Int. 2018:85453472018.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Lobb RJ, Becker M, Wen SW, Wong CSF,
Wiegmans AP, Leimgruber A and Möller A: Optimized exosome isolation
protocol for cell culture supernatant and human plasma. J Extracell
Vesicles. 4:270312015. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Fitzgerald J, Leonard P, Darcy E, Sharma S
and O'Kennedy R: Immunoaffinity Chromatography: Concepts and
Applications. Methods Mol Biol. 1485:27–51. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Li P, Kaslan M, Lee SH, Yao J and Gao Z:
Progress in exosome isolation techniques. Theranostics. 7:789–804.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zarovni N, Corrado A, Guazzi P, Zocco D,
Lari E, Radano G, Muhhina J, Fondelli C, Gavrilova J and Chiesi A:
Integrated isolation and quantitative analysis of exosome shuttled
proteins and nucleic acids using immunocapture approaches. Methods.
87:46–58. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Jackson EL and Lu H: Advances in
microfluidic cell separation and manipulation. Curr Opin Chem Eng.
2:398–404. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Gholizadeh S, Shehata Draz M, Zarghooni M,
Sanati-Nezhad A, Ghavami S, Shafiee H and Akbari M: Microfluidic
approaches for isolation, detection, and characterization of
extracellular vesicles: Current status and future directions.
Biosens Bioelectron. 91:588–605. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Johnstone RM, Bianchini A and Teng K:
Reticulocyte maturation and exosome release: Transferrin receptor
containing exosomes shows multiple plasma membrane functions.
Blood. 74:1844–1851. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Johnstone RM: Revisiting the road to the
discovery of exosomes. Blood Cells Mol Dis. 34:214–219. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Lathe GH and Ruthven CR: The separation of
substances on the basis of their molecular weights, using columns
of starch and water. Biochem J. 60:xxxiv1955.PubMed/NCBI
|
|
63
|
Gámez-Valero A, Monguió-Tortajada M,
Carreras-Planella L, Franquesa MI, Beyer K and Borràs FE:
Size-Exclusion Chromatography-based isolation minimally alters
Extracellular Vesicles' characteristics compared to precipitating
agents. Sci Rep. 6:336412016. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Yu LL, Zhu J, Liu JX, Jiang F, Ni WK, Qu
LS, Ni RZ, Lu CH and Xiao MB: A comparison of traditional and novel
methods for the separation of exosomes from human samples. Biomed
Res Int. 2018:36345632018. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Greening DW, Xu R, Ji H, Tauro BJ and
Simpson RJ: A protocol for exosome isolation and characterization:
evaluation of ultracentrifugation, density-gradient separation, and
immunoaffinity capture methods. Methods Mol Biol. 1295:179–209.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Salieb-Beugelaar GB, Simone G, Arora A,
Philippi A and Manz A: Latest developments in microfluidic cell
biology and analysis systems. Anal Chem. 82:4848–4864. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Koh YQ, Almughlliq FB, Vaswani K, Peiris
HN and Mitchell MD: Exosome enrichment by ultracentrifugation and
size exclusion chromatography. Front Biosci (Landmark Ed).
23:865–874. 2018. View
Article : Google Scholar : PubMed/NCBI
|
|
68
|
Ryu KJ, Lee JY, Park C, Cho D and Kim SJ:
Isolation of small extracellular vesicles from human serum using a
combination of ultracentrifugation with polymer-based
precipitation. Ann Lab Med. 40:253–258. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Zhang S, Duan Z, Liu F, Wu Q, Sun X and Ma
H: The impact of exosomes derived from distinct sources on
rheumatoid arthritis. Front Immunol. 14:12407472023. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Xu F, Zhang Q, Liu Y, Tang R, Li H, Yang H
and Lin L: The role of exosomes derived from various sources in
facilitating the healing of chronic refractory wounds. Pharmacol
Res. 216:1077532025. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Si C, Gao J and Ma X: Natural killer
cell-derived exosome-based cancer therapy: From biological roles to
clinical significance and implications. Mol Cancer. 23:1342024.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Dai X, Ye Y and He F: Emerging innovations
on exosome-based onco-therapeutics. Front Immunol. 13:8652452022.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Liang B, Peng P, Chen S, Li L, Zhang M,
Cao D, Yang J, Li H, Gui T, Li X and Shen K: Characterization and
proteomic analysis of ovarian cancer-derived exosomes. J
Proteomics. 80:171–182. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Panfoli I, Granata S, Candiano G, Verlato
A, Lombardi G, Bruschi M and Zaza G: Analysis of urinary exosomes
applications for rare kidney disorders. Expert Rev Proteomics.
17:735–749. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Di SJ, Cui XW, Liu TJ and Shi YY:
Therapeutic potential of human breast milk-derived exosomes in
necrotizing enterocolitis. Mol Med. 30:2432024. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Cai H, Pang Y, Wang Q, Qin W, Wei C, Li Y,
Li T, Li F, Wang Q, Li Y, et al: Proteomic profiling of circulating
plasma exosomes reveals novel biomarkers of Alzheimer's disease.
Alzheimers Res Ther. 14:1812022. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Muller L, Hong CS, Stolz DB, Watkins SC
and Whiteside TL: Isolation of biologically-active exosomes from
human plasma. J Immunol Methods. 411:55–65. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Ibsen SD, Wright J, Lewis JM, Kim S, Ko
SY, Ong J, Manouchehri S, Vyas A, Akers J, Chen CC, et al: Rapid
isolation and detection of exosomes and associated biomarkers from
plasma. ACS Nano. 11:6641–6651. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Kim NH, Kim J, Lee JY, Bae HA and Kim CY:
Application of milk exosomes for musculoskeletal health: talking
points in recent outcomes. Nutrients. 15:46452023. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Karabay AZ, Barar J, Hekmatshoar Y and
Rahbar Saadat Y: Multifaceted therapeutic potential of
plant-derived exosomes: Immunomodulation, anticancer, anti-aging,
anti-melanogenesis, detoxification, and drug delivery.
Biomolecules. 15:3942025. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Cao M, Diao N, Cai X, Chen X, Xiao Y, Guo
C, Chen D and Zhang X: Plant exosome nanovesicles (PENs): Green
delivery platforms. Mater Horiz. 10:3879–3894. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Yi Q, Xu Z, Thakur A, Zhang K, Liang Q,
Liu Y and Yan Y: Current understanding of plant-derived
exosome-like nanoparticles in regulating the inflammatory response
and immune system microenvironment. Pharmacol Res. 190:1067332023.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Li Q, Zhang Y, Shi B, Lin C, Feng Q, Zhou
H, Hao M, Ding Y, Ma C, Mu J and Wang D: Galangin exosomes induce
cell apoptosis through miR-10b/P53 axis in gastric cancer. Sci Rep.
15:128762025. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Zhang Q, Deng T, Zhang H, Zuo D, Zhu Q,
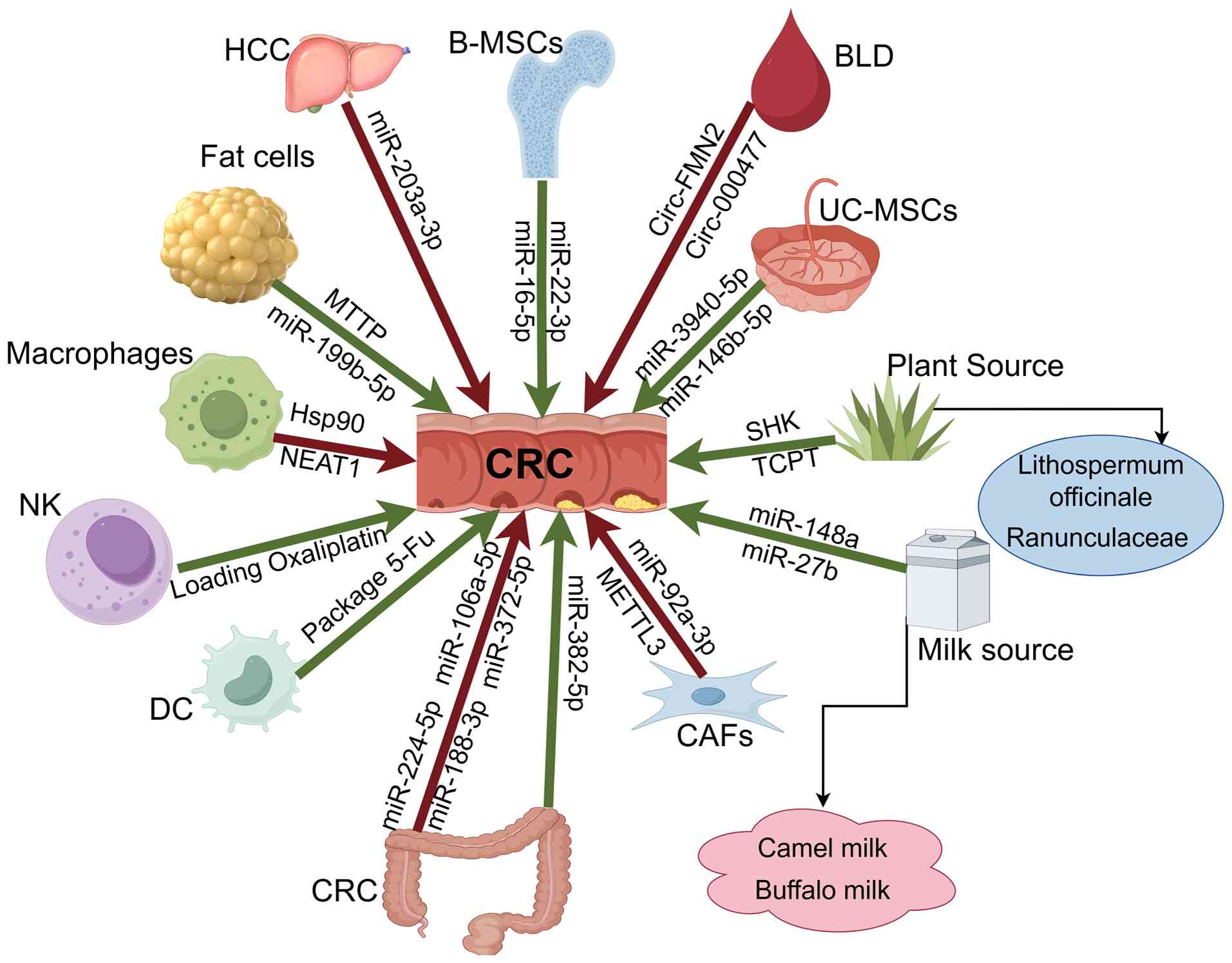
Bai M, Liu R, Ning T, Zhang L, Yu Z, et al: Adipocyte-derived
exosomal MTTP suppresses ferroptosis and promotes chemoresistance
in colorectal cancer. Adv Sci (Weinh). 9:e22033572022. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Lv X, Li Z, Dai Y, Xiao Y, Shen F, Wang J,
Cao J, Wang L, Peng Q and Jiao Y: The mir-199b-5p encapsulated in
adipocyte-derived exosomes mediates radioresistance of colorectal
cancer cells by targeting JAG1. Heliyon. 10:e244122024. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Wang Y and Lin C: Exosomes miR-22-3p
derived from mesenchymal stem cells suppress colorectal cancer cell
proliferation and invasion by regulating RAP2B and PI3K/AKT
pathway. J Oncol. 2021:38744782021.PubMed/NCBI
|
|
87
|
Xu Y, Shen L, Li F, Yang J, Wan X and
Ouyang M: microRNA-16-5p-containing exosomes derived from bone
marrow-derived mesenchymal stem cells inhibit proliferation,
migration, and invasion, while promoting apoptosis of colorectal
cancer cells by downregulating ITGA2. J Cell Physiol.
234:21380–21394. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Ning S, Chen Y, Li S, Liu M, Liu H, Ye M,
Wang C, Pan J, Wei W, Li J and Zhang L: Exosomal miR-99b-5p
secreted from mesenchymal stem cells can retard the progression of
colorectal cancer by targeting FGFR3. Stem Cell Rev Rep.
19:2901–2917. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Li T, Wan Y, Su Z, Li J, Han M and Zhou C:
Mesenchymal stem cell-derived exosomal microRNA-3940-5p inhibits
colorectal cancer metastasis by targeting integrin α6. Dig Dis Sci.
66:1916–1927. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Yu S, Liao R, Bai L, Guo M, Zhang Y, Zhang
Y, Yang Q, Song Y, Li Z, Meng Q, et al: Anticancer effect of
hUC-MSC-derived exosome-mediated delivery of PMO-miR-146b-5p in
colorectal cancer. Drug Deliv Transl Res. 14:1352–1369. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Han Y, Zheng W, Zhang Y, Tong C, Song P,
Qi Z and Zhang S: Oxaliplatin-loaded natural killer cell-derived
exosomes for a safe and efficient chemoimmunotherapy of colorectal
cancer. J Pharm Sci. 114:1037832025. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Xu M, Chen Q, Li J, Peng L and Ding L:
Dendritic cell-derived exosome-entrapped fluorouracil can enhance
its anti-colon cancer effect. J BUON. 25:1413–1422. 2020.PubMed/NCBI
|
|
93
|
Jiang J, Wang W, Zhu L, Shi B, Chen Y, Xia
Y, Feng W, Yao W, Lu A and Zhang H: Unveiling the role of hypoxic
macrophage-derived exosomes in driving colorectal cancer
progression. Front Immunol. 14:12606382023. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Liu F, Ai F, Tang A, Yang Z, Li Z and Liu
S: Macrophage-derived exosomes promoted the development and
stemness of inflammatory bowel disease-related colorectal cancer
via nuclear paraspeckle assembly transcript 1-mediated
miRNA-34a-5p/phosphoprotein enriched in astrocytes 15 axis. Inflamm
Bowel Dis. 31:524–538. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhao S, Mi Y, Guan B, Zheng B, Wei P, Gu
Y, Zhang Z, Cai S, Xu Y, Li X, et al: Tumor-derived exosomal
miR-934 induces macrophage M2 polarization to promote liver
metastasis of colorectal cancer. J Hematol Oncol. 13:1562020.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Wu Y, Xiao Y, Ding Y, Ran R, Wei K, Tao S,
Mao H, Wang J, Pang S, Shi J, et al: Colorectal cancer cell-derived
exosomal miRNA-372-5p induces immune escape from colorectal cancer
via PTEN/AKT/NF-κB/PD-L1 pathway. Int Immunopharmacol.
143:1132612024. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zhang C, Wang XY, Zhang P, He TC, Han JH,
Zhang R, Lin J, Fan J, Lu L, Zhu WW, et al: Cancer-derived exosomal
HSPC111 promotes colorectal cancer liver metastasis by
reprogramming lipid metabolism in cancer-associated fibroblasts.
Cell Death Dis. 13:572022. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Meng Q, Xiang H, Wang Y, Hu K, Luo X, Wang
J, Chen E, Zhang W, Chen J, Chen X, et al: Exosomes containing
circSCP2 in colorectal cancer promote metastasis via sponging
miR-92a-1-5p and interacting with PTBP1 to stabilize IGF2BP1. Biol
Direct. 19:1302024. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Li T, Li T, Liang Y, Yuan Y, Liu Y, Yao Y
and Lei X: Colorectal cancer cells-derived exosomal miR-188-3p
promotes liver metastasis by creating a pre-metastatic niche via
activation of hepatic stellate cells. J Transl Med. 23:3692025.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Feng CZ, Zhong SQ, Ye SW, Zheng Z, Sun H
and Zhou SH: Tumor-derived exosomal miR-425-5p and miR-135b-3p
enhance colorectal cancer progression through immune suppression
and vascular permeability promotion. World J Gastrointest Oncol.
17:1061612025. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Li K, Xue W, Lu Z, Wang S, Zheng J, Lu K,
Li M, Zong Y, Xu F, Dai J, et al: Tumor-derived exosomal ADAM17
promotes pre-metastatic niche formation by enhancing vascular
permeability in colorectal cancer. J Exp Clin Cancer Res.
43:592024. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Liang Y, Li J, Yuan Y, Ju H, Liao H, Li M,
Liu Y, Yao Y, Yang L, Li T and Lei X: Exosomal miR-106a-5p from
highly metastatic colorectal cancer cells drives liver metastasis
by inducing macrophage M2 polarization in the tumor
microenvironment. J Exp Clin Cancer Res. 43:2812024. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Li S, Fu X, Ning D, Liu Q, Zhao J, Cheng
Q, Chen X and Jiang L: Colon cancer exosome-associated HSP90B1
initiates pre-metastatic niche formation in the liver by polarizing
M1 macrophage into M2 phenotype. Biol Direct. 20:522025. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Sun J, Luo J, Liu J, Wu H, Li Y, Xu Y, Liu
L, Liu X and Zhang Q: Cancer-secreted exosomal miR-1825 induces
angiogenesis to promote colorectal cancer metastasis. Cancer Cell
Int. 25:632025. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Yan YY, Deng ZF, Wu XT, Lu Y, Zhu ZY, Wen
Q, Zhang W, Zhang HY, Chen XZ, Wu YS, et al: Low miR-224-5p in
exosomes confers colorectal cancer 5-FU resistance by upregulating
S100A4. Drug Resist Updat. 79:1012112025. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Ren H, Wang M, Ma X, An L, Guo Y and Ma H:
METTL3 in cancer-associated fibroblasts-derived exosomes promotes
the proliferation and metastasis and suppresses ferroptosis in
colorectal cancer by eliciting ACSL3 m6A modification. Biol Direct.
19:682024. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Yang C, Zhang Y, Yan M, Wang J, Wang J,
Wang M, Xuan Y, Cheng H, Ma J, Chai C, et al: Exosomes derived from
cancer-associated fibroblasts promote tumorigenesis, metastasis and
chemoresistance of colorectal cancer by upregulating circ_0067557
to target Lin28. BMC Cancer. 24:642024. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Liu Y, Wang Y, Yu Z and Wang Z: Impacts of
TP53TG1 in cancer-associated fibroblasts-derived exosomes on
epithelial-mesenchymal transition capacity of colorectal carcinoma
cells by targeting miR-330-3p. Heliyon. 10:e303012024. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Xu H, Lan Q, Huang Y, Zhang Y, Zeng Y, Su
P and Chu Z, Lai W and Chu Z: The mechanisms of colorectal cancer
cell mesenchymal-epithelial transition induced by hepatocyte
exosome-derived miR-203a-3p. BMC Cancer. 21:7182021. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Yu Q, Zhang Y, Tian Y, Peng A, Cui X, Ding
B, Yang L, Liu Y, Ju Y and Gao C: Exosomal Circ_FMN2 derived from
the serum of colorectal cancer patients promotes cancer progression
by miR-338-3p/MSI1 Axis. Appl Biochem Biotechnol. 195:7322–7337.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Qiao XX, Shi HB and Xiao L: Serum exosomal
hsa-circ-0004771 modulates the resistance of colorectal cancer to
5-fluorouracil via regulating miR-653/ZEB2 signaling pathway.
Cancer Cell Int. 23:2432023. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Karbasi S, Erfanian N, Dehghan H, Zarban
A, Namaei MH, Hanafi-Bojd MY and Nasseri S: Assessment of the
anti-cancer effects of camel milk exosomes (CMEXOs) on murine
colorectal cancer cell line (CT-26). Iran J Allergy Asthma Immunol.
23:321–329. 2024.PubMed/NCBI
|
|
113
|
Babaker MA, Aljoud FA, Alkhilaiwi F,
Algarni A, Ahmed A, Khan MI, Saadeldin IM and Alzahrani FA: The
therapeutic potential of milk extracellular vesicles on colorectal
cancer. Int J Mol Sci. 23:68122022. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Martino E, Balestrieri A, Mele L, Sardu C,
Marfella R, D'Onofrio N, Campanile G and Balestrieri ML: Milk
exosomal miR-27b worsen endoplasmic reticulum stress mediated
colorectal cancer cell death. Nutrients. 14:50812022. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Lu Y, Zhou H, Han C, Gong Y, Li Y, Xia Y,
Liang B, Yang H and Wang Z: Enhanced therapeutic impact of
Shikonin-encapsulated exosomes in the inhibition of colorectal
cancer progression. Nanotechnology. 35:2024. View Article : Google Scholar
|
|
116
|
Liu Y, Cheng DH, Su ZY, Lv JH, Wang L,
Deng YY and Li L: Effects of total coumarins from Pileostegia
tomentella on exosomal miRNA expression and angiogenesis in
colorectal cancer cells. Pharm Biol. 62:153–161. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Jin Y, Sun L, Chen Y and Lu Y: The
homologous tumor-derived-exosomes loaded with miR-1270 selectively
enhanced the suppression effect for colorectal cancer cells. Cancer
Med. 13:e69362024. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Liu M, Li TZ and Xu C: The role of
tumor-associated fibroblast-derived exosomes in chemotherapy
resistance of colorectal cancer and its application prospect.
Biochim Biophys Acta Gen Subj. 1869:1307962025. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Wang Z, Kim SY, Tu W, Kim J, Xu A, Yang
YM, Matsuda M, Reolizo L, Tsuchiya T, Billet S, et al:
Extracellular vesicles in fatty liver promote a metastatic tumor
microenvironment. Cell Metab. 35:1209–1226.e13. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI
|
|
121
|
Arnold M, Sierra MS, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global patterns and trends in
colorectal cancer incidence and mortality. Gut. 66:683–691. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Liu SC and Zhang H: Early diagnostic
strategies for colorectal cancer. World J Gastroenterol.
30:3818–3822. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Yin H, Xie J, Xing S, Lu X, Yu Y, Ren Y,
Tao J, He G, Zhang L, Yuan X, et al: Machine learning-based
analysis identifies and validates serum exosomal proteomic
signatures for the diagnosis of colorectal cancer. Cell Rep Med.
5:1016892024. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Hui J, Zhou M, An G, Zhang H, Lu Y, Wang X
and Zhao X: Regulatory role of exosomes in colorectal cancer
progression and potential as biomarkers. Cancer Biol Med.
20:575–598. 2023.PubMed/NCBI
|
|
125
|
Lin WC, Lin CC, Lin YY, Yang WH, Twu YC,
Teng HW and Hwang WL: Molecular actions of exosomes and their
theragnostics in colorectal cancer: Current findings and
limitations. Cell Oncol (Dordr). 45:1043–1052. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Rizk NI, Kassem DH, Abulsoud AI,
AbdelHalim S, Yasser MB, Kamal MM and Hamdy NM: Revealing the role
of serum exosomal novel long non-coding RNA NAMPT-AS as a promising
diagnostic/prognostic biomarker in colorectal cancer patients. Life
Sci. 352:1228502024. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Zhao Y, Du T, Du L, Li P, Li J, Duan W,
Wang Y and Wang C: Long noncoding RNA LINC02418 regulates MELK
expression by acting as a ceRNA and may serve as a diagnostic
marker for colorectal cancer. Cell Death Dis. 10:5682019.
View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Fabijanec M, Hulina-Tomašković A,
Štefanović M, Verbanac D, Ćelap I, Somborac-Bačura A, Grdić
Rajković M, Demirović A, Ramić S, Krušlin B, et al:
MicroRNA-193a-3p as a valuable biomarker for discriminating between
colorectal cancer and colorectal adenoma patients. Int J Mol Sci.
25:81562024. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Xu Y and Zhu M: Novel exosomal miR-46146
transfer oxaliplatin chemoresistance in colorectal cancer. Clin
Transl Oncol. 22:1105–1116. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Zhao Y, Zhao Y, Liu L, Li G, Wu Y, Cui Y
and Xie L: Tumor-exosomal miR-205-5p as a diagnostic biomarker for
colorectal cancer. Clin Transl Oncol. 27:1185–1197. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Yan S, Jiang Y, Liang C, Cheng M, Jin C,
Duan Q, Xu D, Yang L, Zhang X, Ren B and Jin P: Exosomal
miR-6803-5p as potential diagnostic and prognostic marker in
colorectal cancer. J Cell Biochem. 119:4113–4119. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Liu X, Pan B, Sun L, Chen X, Zeng K, Hu X,
Xu T, Xu M and Wang S: Circulating exosomal miR-27a and miR-130a
act as novel diagnostic and prognostic biomarkers of colorectal
cancer. Cancer Epidemiol Biomarkers Prev. 27:746–754. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Wang L, Song X, Yu M, Niu L, Zhao Y, Tang
Y, Zheng B, Song X and Xie L: Serum exosomal miR-377-3p and
miR-381-3p as diagnostic biomarkers in colorectal cancer. Future
Oncol. 18:793–805. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Liu W, Yang D, Chen L, Liu Q, Wang W, Yang
Z, Shang A, Quan W and Li D: Plasma exosomal miRNA-139-3p is a
novel biomarker of colorectal cancer. J Cancer. 11:4899–4906. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Zhao YJ, Song X, Niu L, Tang Y, Song X and
Xie L: Circulating exosomal miR-150-5p and miR-99b-5p as diagnostic
biomarkers for colorectal cancer. Front Oncol. 9:11292019.
View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Wang J, Yan F, Zhao Q, Zhan F, Wang R,
Wang L, Zhang Y and Huang X: Circulating exosomal miR-125a-3p as a
novel biomarker for early-stage colon cancer. Sci Rep. 7:41502017.
View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Li T, Zhou T, Wu J, Lv H, Zhou H, Du M,
Zhang X, Wu N, Gong S, Ren Z, et al: Plasma exosome-derived
circGAPVD1 as a potential diagnostic marker for colorectal cancer.
Transl Oncol. 31:1016522023. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Xie Y, Li J, Li P, Li N, Zhang Y, Binang
H, Zhao Y, Duan W, Chen Y, Wang Y, et al: RNA-Seq profiling of
serum exosomal circular RNAs reveals circ-PNN as a potential
biomarker for human colorectal cancer. Front Oncol. 10:9822020.
View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Choi DS, Park JO, Jang SC, Yoon YJ, Jung
JW, Choi DY, Kim JW, Kang JS, Park J, Hwang D, et al: Proteomic
analysis of microvesicles derived from human colorectal cancer
ascites. Proteomics. 11:2745–2751. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Erozenci LA, Böttger F, Bijnsdorp IV and
Jimenez CR: Urinary exosomal proteins as (pan-)cancer biomarkers:
Insights from the proteome. FEBS Lett. 593:1580–1597. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Ma L, Yu H, Zhu Y, Xu K, Zhao A, Ding L,
Gao H and Zhang M: Isolation and proteomic profiling of urinary
exosomes from patients with colorectal cancer. Proteome Sci.
21:32023. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Yao J, Chen Y and Lin Z: Exosomes:
Mediators in microenvironment of colorectal cancer. Int J Cancer.
153:904–917. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Huang C, Zhou Y, Feng X, Wang J, Li Y and
Yao X: Delivery of engineered primary tumor-derived exosomes
effectively suppressed the colorectal cancer chemoresistance and
liver metastasis. ACS Nano. 17:10313–10326. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Shekh R, Ahmad A, Tiwari RK, Saeed M,
Shukla R, Al-Thubiani WS, Ansari IA, Ashfaque M and Bajpai P: High
therapeutic efficacy of 5-fluorouracil-loaded exosomes against
colon cancer cells. Chem Biol Drug Des. 101:962–976. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Li Y, Gao Y, Gong C, Wang Z, Xia Q, Gu F,
Hu C, Zhang L, Guo H and Gao S: A33 antibody-functionalized
exosomes for targeted delivery of doxorubicin against colorectal
cancer. Nanomedicine. 14:1973–1985. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Liu T, Zhang X, Du L, Wang Y, Liu X, Tian
H, Wang L, Li P, Zhao Y, Duan W, et al: Correction to:
Exosome-transmitted miR-128-3p increase chemosensitivity of
oxaliplatin-resistant colorectal cancer. Mol Cancer. 19:892020.
View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Bagheri E, Abnous K, Farzad SA, Taghdisi
SM, Ramezani M and Alibolandi M: Targeted doxorubicin-loaded
mesenchymal stem cells-derived exosomes as a versatile platform for
fighting against colorectal cancer. Life Sci. 261:1183692020.
View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Wu S, Yun J, Tang W, Familiari G,
Relucenti M, Wu J, Li X, Chen H and Chen R: Therapeutic m6A Eraser
ALKBH5 mRNA-loaded exosome-liposome hybrid nanoparticles inhibit
progression of colorectal cancer in preclinical tumor models. ACS
Nano. 17:11838–11854. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Shakerian N, Darzi-Eslam E, Afsharnoori F,
Bana N, Noorabad Ghahroodi F, Tarin M, Mard-Soltani M, Khalesi B,
Hashemi ZS and Khalili S: Therapeutic and diagnostic applications
of exosomes in colorectal cancer. Med Oncol. 41:2032024. View Article : Google Scholar : PubMed/NCBI
|