|
1
|
Zheng M: Classification and pathology of
lung cancer. Surg Oncol Clin N Am. 25:447–468. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mithoowani H and Febbraro M:
Non-small-cell lung cancer in 2022: A review for general
practitioners in oncology. Curr Oncol. 29:1828–1839. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hirsch FR, Scagliotti GV, Mulshine JL,
Kwon R, Curran WJ, Wu YL and Paz-Ares L: Lung cancer: Current
therapies and new targeted treatments. Lancet. 389:299–311. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jonna S and Subramaniam DS: Molecular
diagnostics and targeted therapies in non-small cell lung cancer
(NSCLC): An update. Discov Med. 27:167–170. 2019.PubMed/NCBI
|
|
5
|
Zhang Y, Chen M, Fang X, Han Y and Li Y:
Progression and metastasis of lung cancer: Clinical features,
molecular mechanisms, and clinical managements. Medcomm.
6:e704772025. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Manfioletti G and Fedele M:
Epithelial-mesenchymal transition (EMT). Int J Mol Sci.
24:113862023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Manfioletti G and Fedele M:
Epithelial-mesenchymal transition (EMT) 2021. Int J Mol Sci.
23:58482022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jiang Y, Han Q, Zhao H and Zhang J:
Promotion of epithelial-mesenchymal transformation by
hepatocellular carcinoma-educated macrophages through
Wnt2b/β-catenin/c-Myc signaling and reprogramming glycolysis. J Exp
Clin Canc Res. 40:132021. View Article : Google Scholar
|
|
9
|
Lin YT and Wu KJ: Epigenetic regulation of
epithelial-mesenchymal transition: Focusing on hypoxia and TGF-β
signaling. J Biomed Sci. 27:392020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shi Q, Xue C, Zeng Y, Yuan X, Chu Q, Jiang
S, Wang J, Zhang Y, Zhu D and Li L: Notch signaling pathway in
cancer: From mechanistic insights to targeted therapies. Signal
Transduct Tar. 9:1282024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ang HL, Mohan CD, Shanmugam MK, Leong HC,
Makvandi P, Rangappa KS, Bishayee A, Kumar AP and Sethi G:
Mechanism of Epithelial-mesenchymal transition in cancer and its
regulation by natural compounds. Med Res Rev. 43:1141–1200. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xue W, Yang L, Chen C, Ashrafizadeh M,
Tian Y and Sun R: Wnt/β-catenin-driven EMT regulation in human
cancers. Cell Mol Life Sci. 81:792024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wei P, Zhang N, Wang Y, Li D, Wang L, Sun
X, Shen C, Yang Y, Zhou X and Du X: FOXM1 promotes lung
adenocarcinoma invasion and metastasis by upregulating SNAIL. Int J
Biol Sci. 11:186–198. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pan J, Fang S, Tian H, Zhou C, Zhao X,
Tian H, He J, Shen W, Meng X, Jin X and Gong Z: lncRNA
JPX/miR-33a-5p/Twist1 axis regulates tumorigenesis and metastasis
of lung cancer by activating Wnt/β-catenin signaling. Mol Cancer.
19:92020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Clarhaut J, Gemmill RM, Potiron VA,
Ait-Si-Ali S, Imbert J, Drabkin HA and Roche J: ZEB-1, a repressor
of the semaphorin 3F tumor suppressor gene in lung cancer cells.
Neoplasia. 11:157–166. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang X, Li M, Yin J, Fang J, Ying Y, Ye T,
Zhang F, Ma S, Qin H and Liu X: Emetine dihydrochloride alleviated
radiation-induced lung injury through inhibiting EMT. J Cell Mol
Med. 27:3839–3850. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chanvorachote P, Petsri K and Thongsom S:
Epithelial to mesenchymal transition in lung cancer: Potential
EMT-Targeting natural product-derived compounds. Anticancer Res.
42:4237–4246. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Al-Ward H, Liu CY, Liu N, Shaher F,
Al-Nusaif M, Mao J and Xu H: Voltage-gated sodium channel β1 Gene:
An overview. Hum Hered. 85:101–109. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lewis AH and Raman IM: Resurgent current
of voltage-gated Na(+) channels. J Physiol. 592:4825–4838. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ślęczkowska M, Almomani R, Marchi M, Salvi
E, de Greef B, Sopacua M, Hoeijmakers J, Lindsey P, Waxman SG,
Lauria G, et al: Peripheral ion channel genes screening in painful
small fiber neuropathy. Int J Mol Sci. 23:140952022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lu Y, Yu W, Xi Z, Xiao Z, Kou X and Wang
XF: Mutational analysis of SCN2B, SCN3B and SCN4B in a large
Chinese Han family with generalized tonic-clonic seizure. Neurol
Sci. 31:675–677. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dai W, Zhou J, Wang H, Zhang M, Yang X and
Song W: miR-424-5p promotes the proliferation and metastasis of
colorectal cancer by directly targeting SCN4B. Pathol Res Pract.
216:1527312020. View Article : Google Scholar : PubMed/NCBI
|
|
23
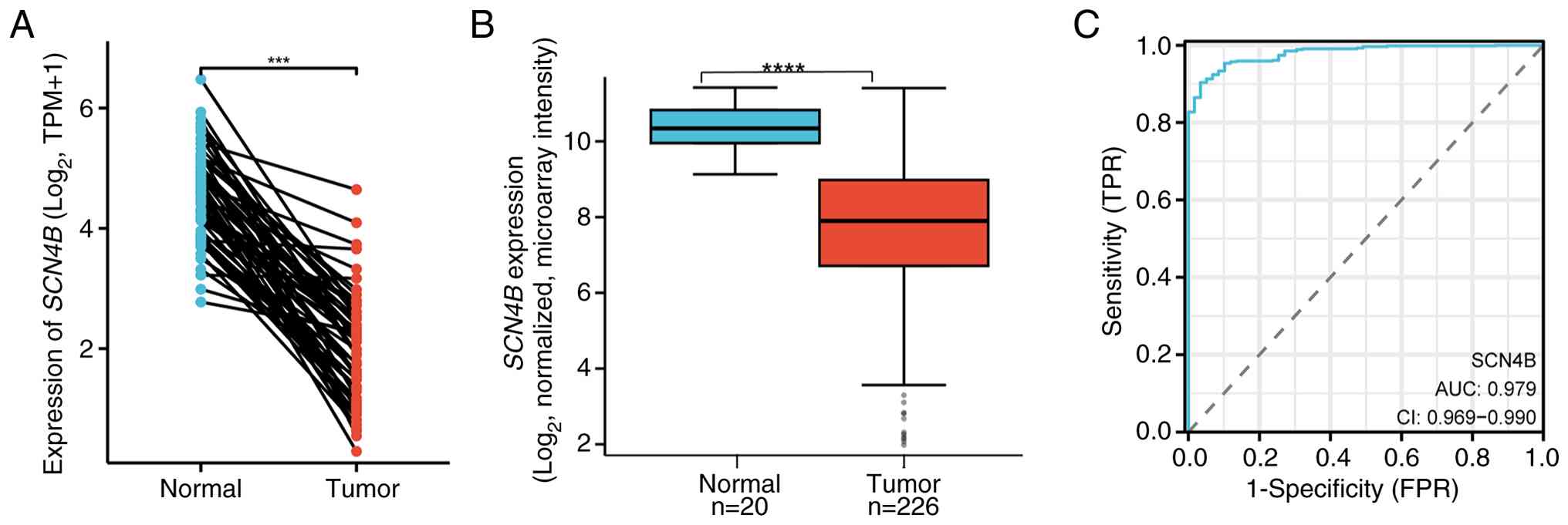
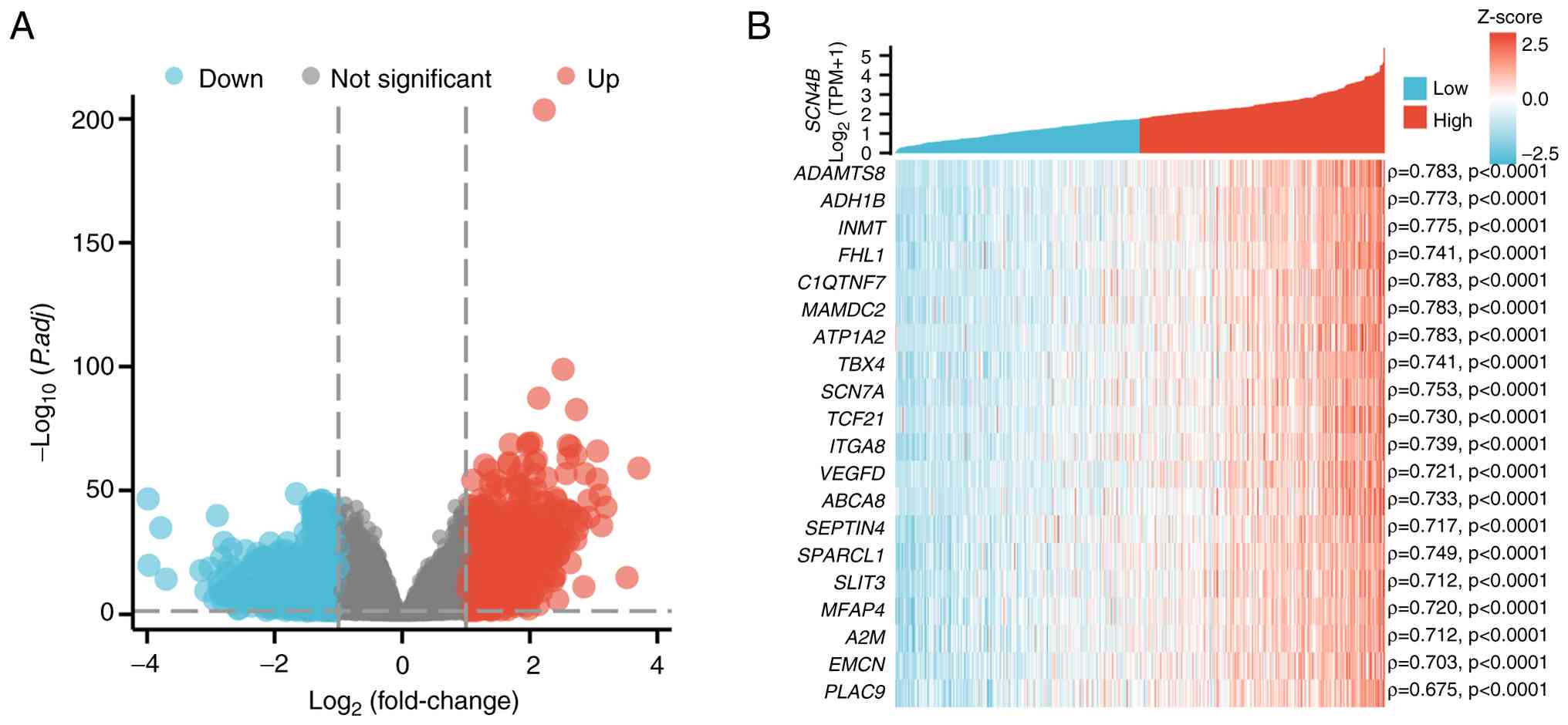
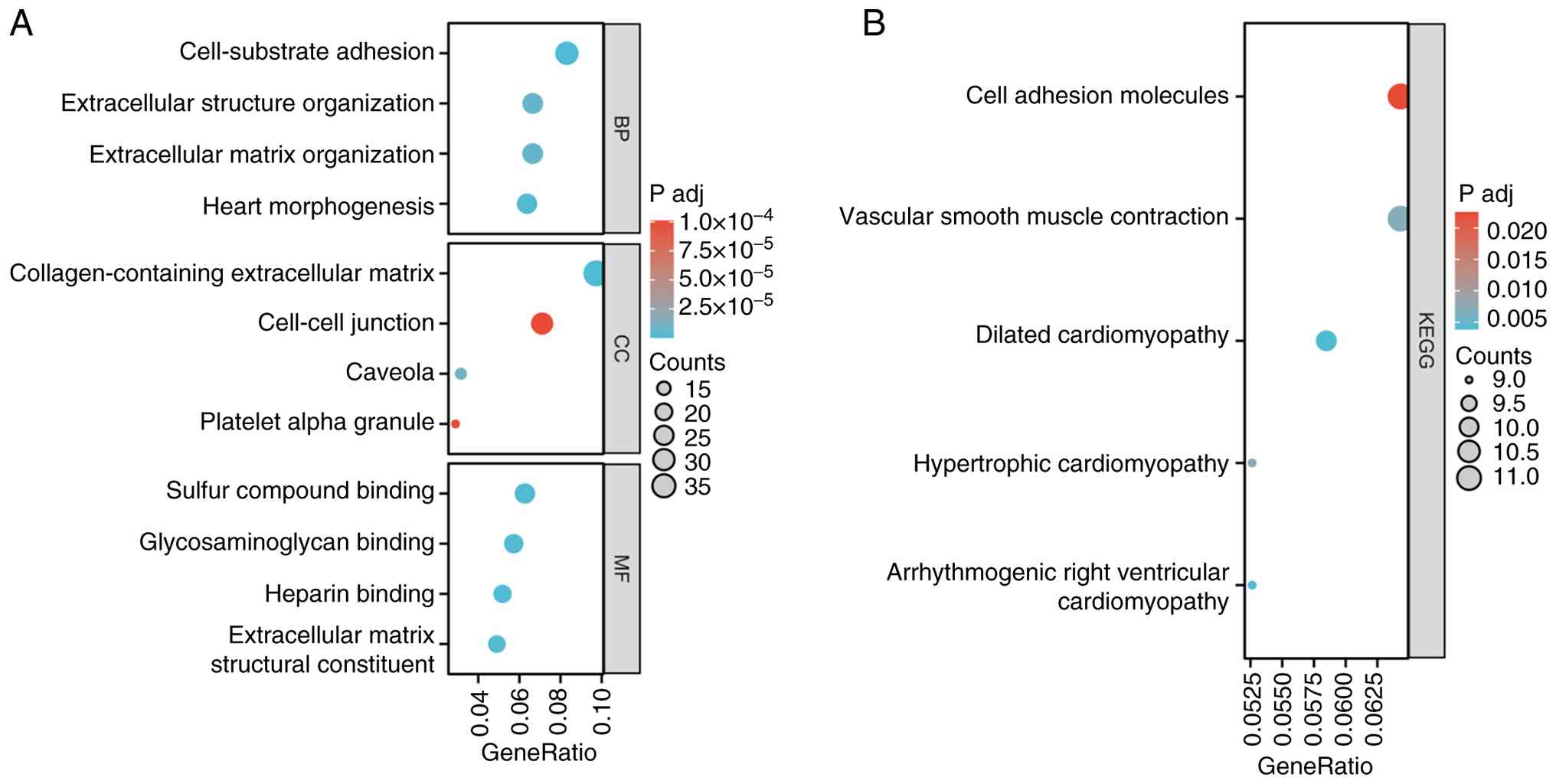
|
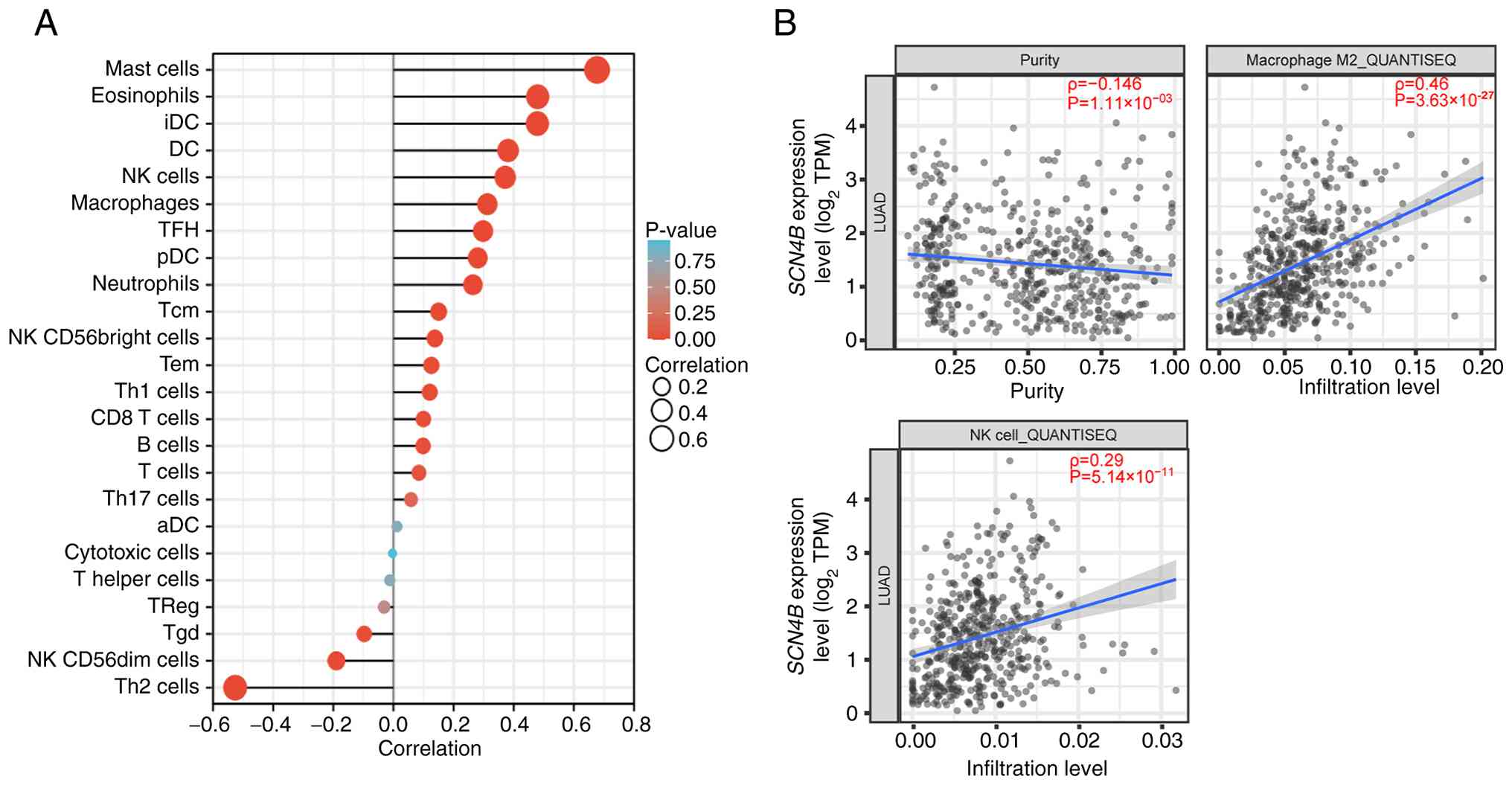
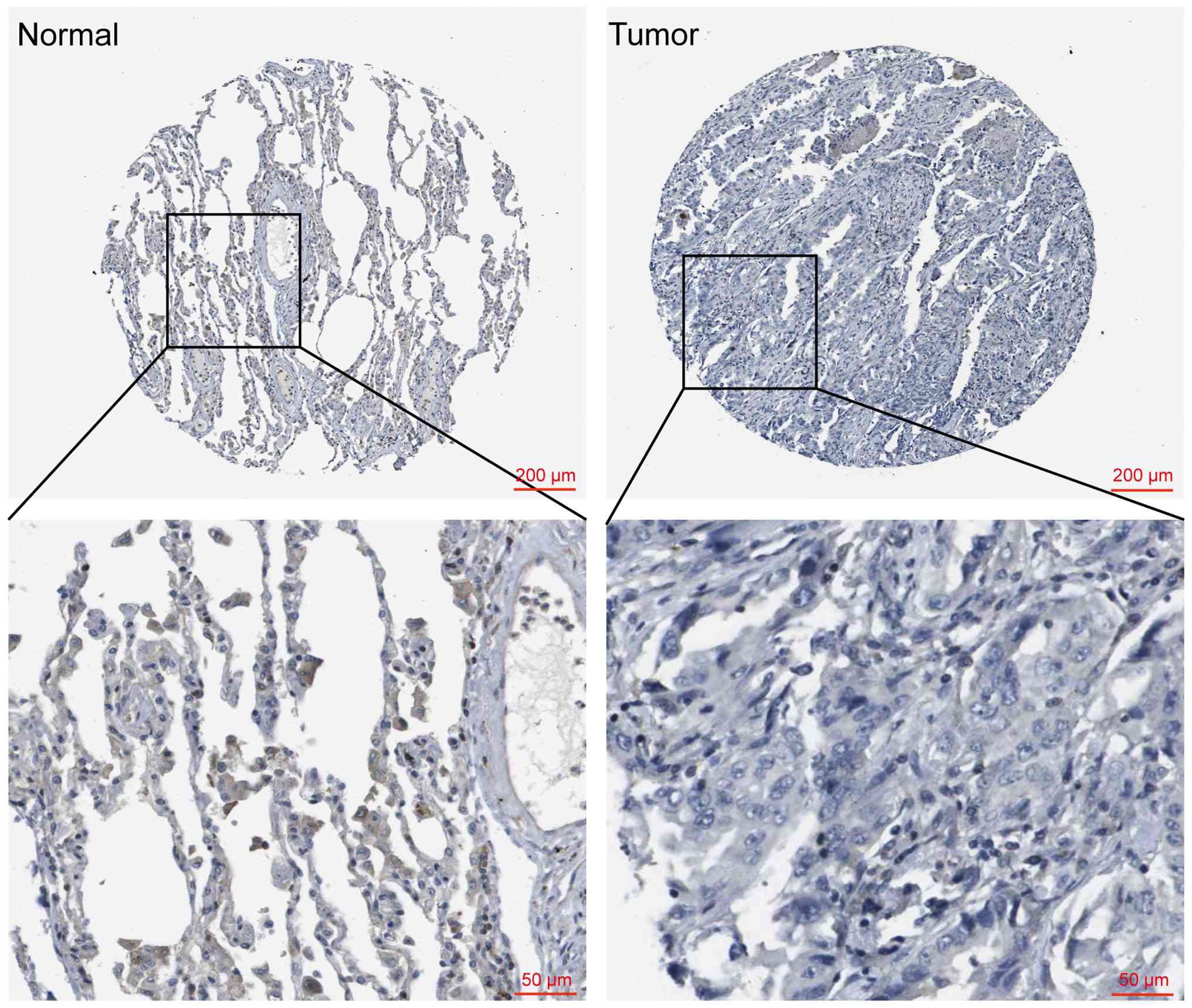
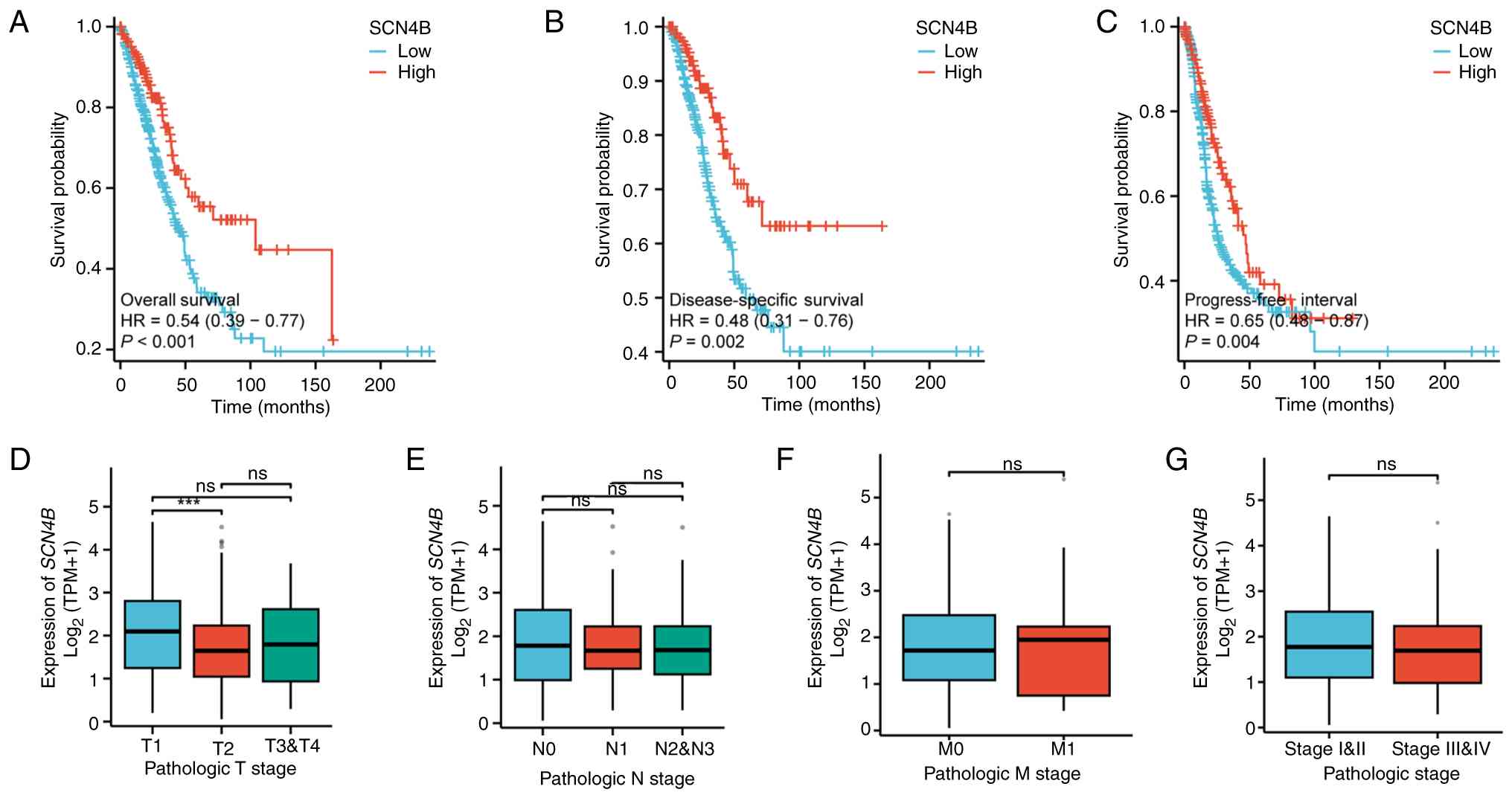
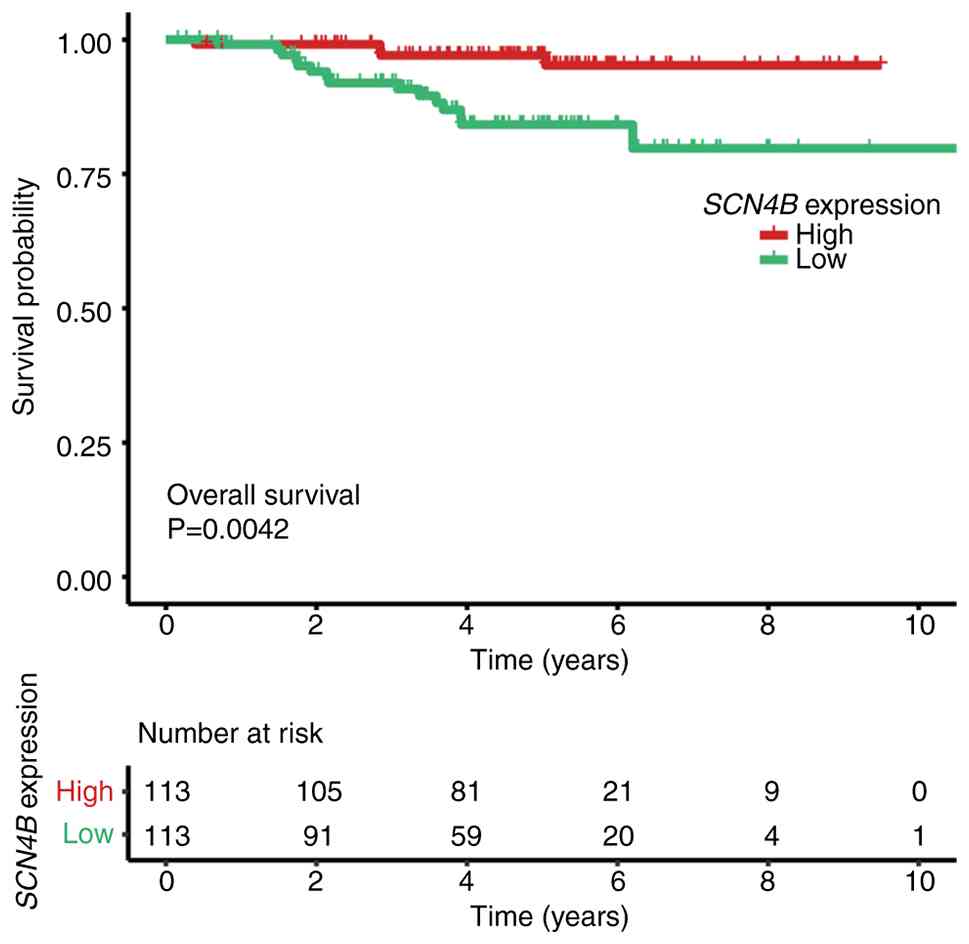
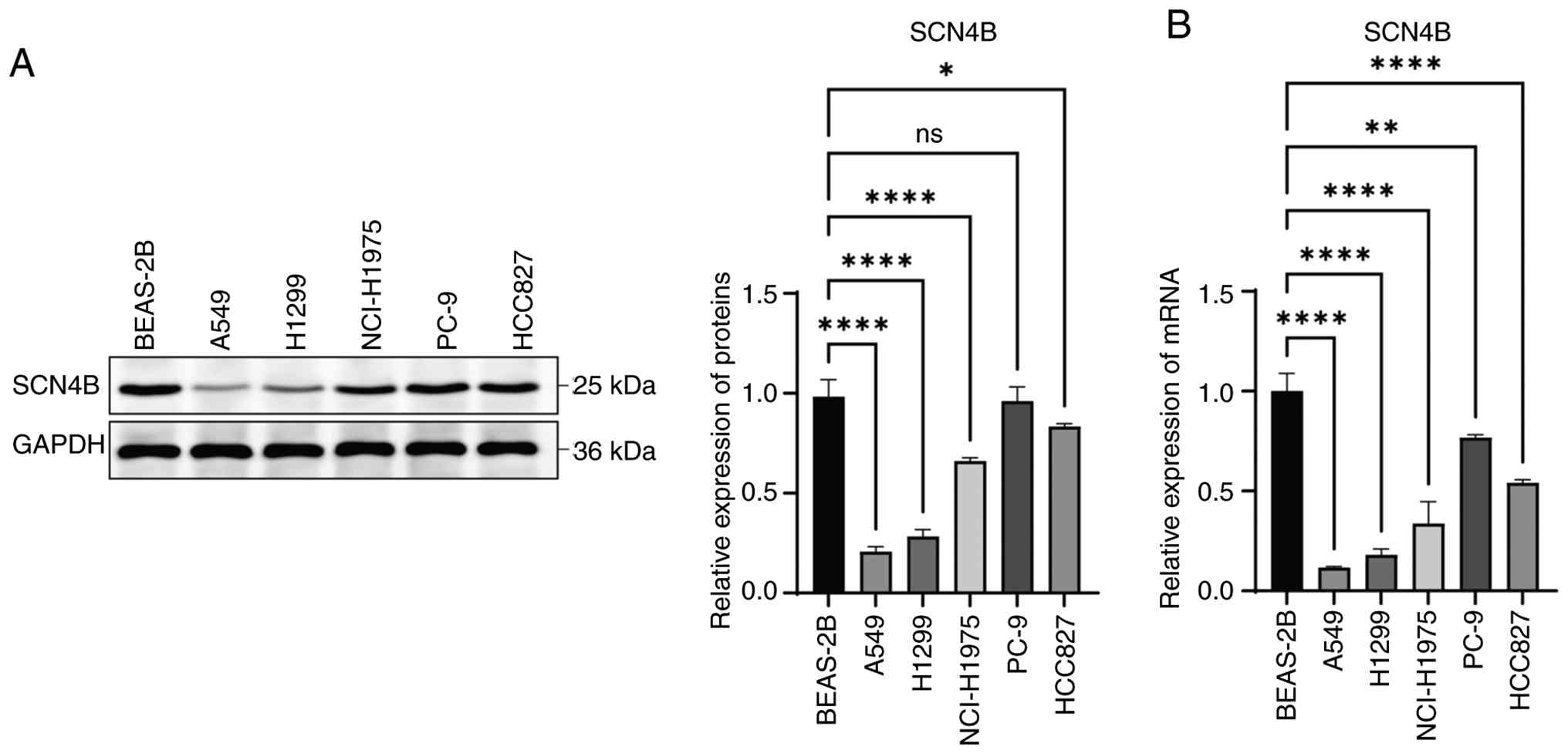
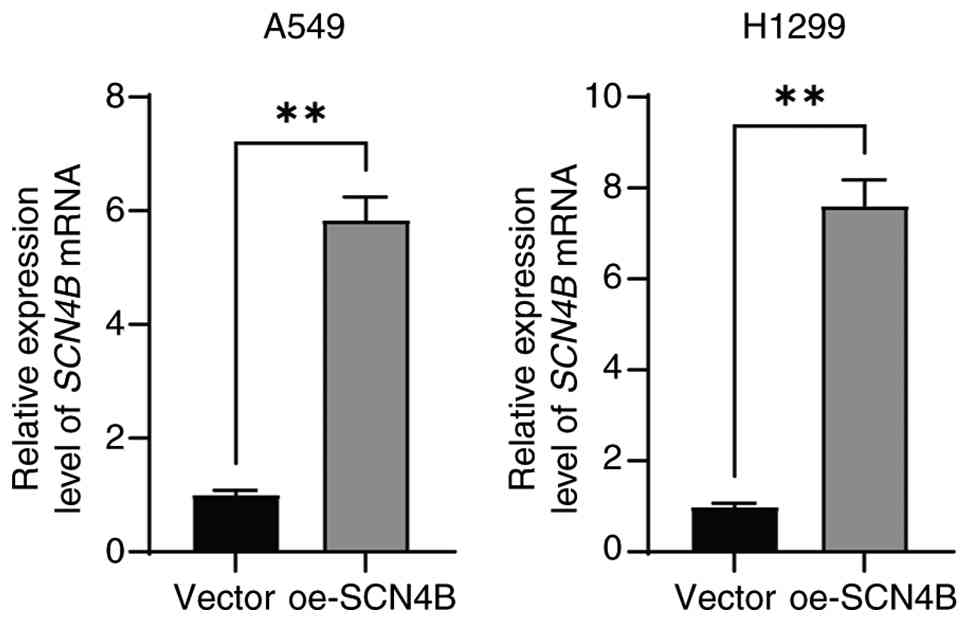
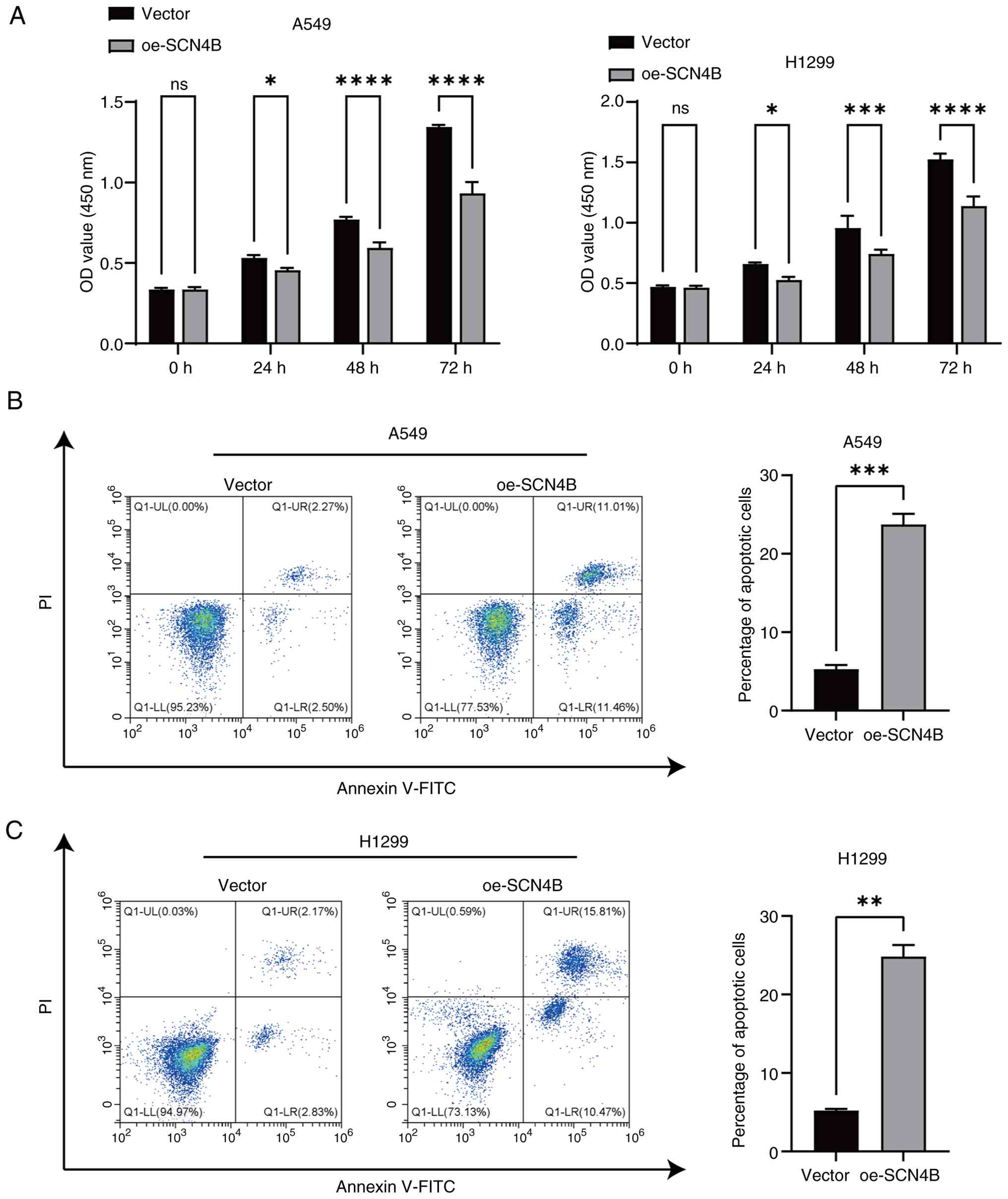
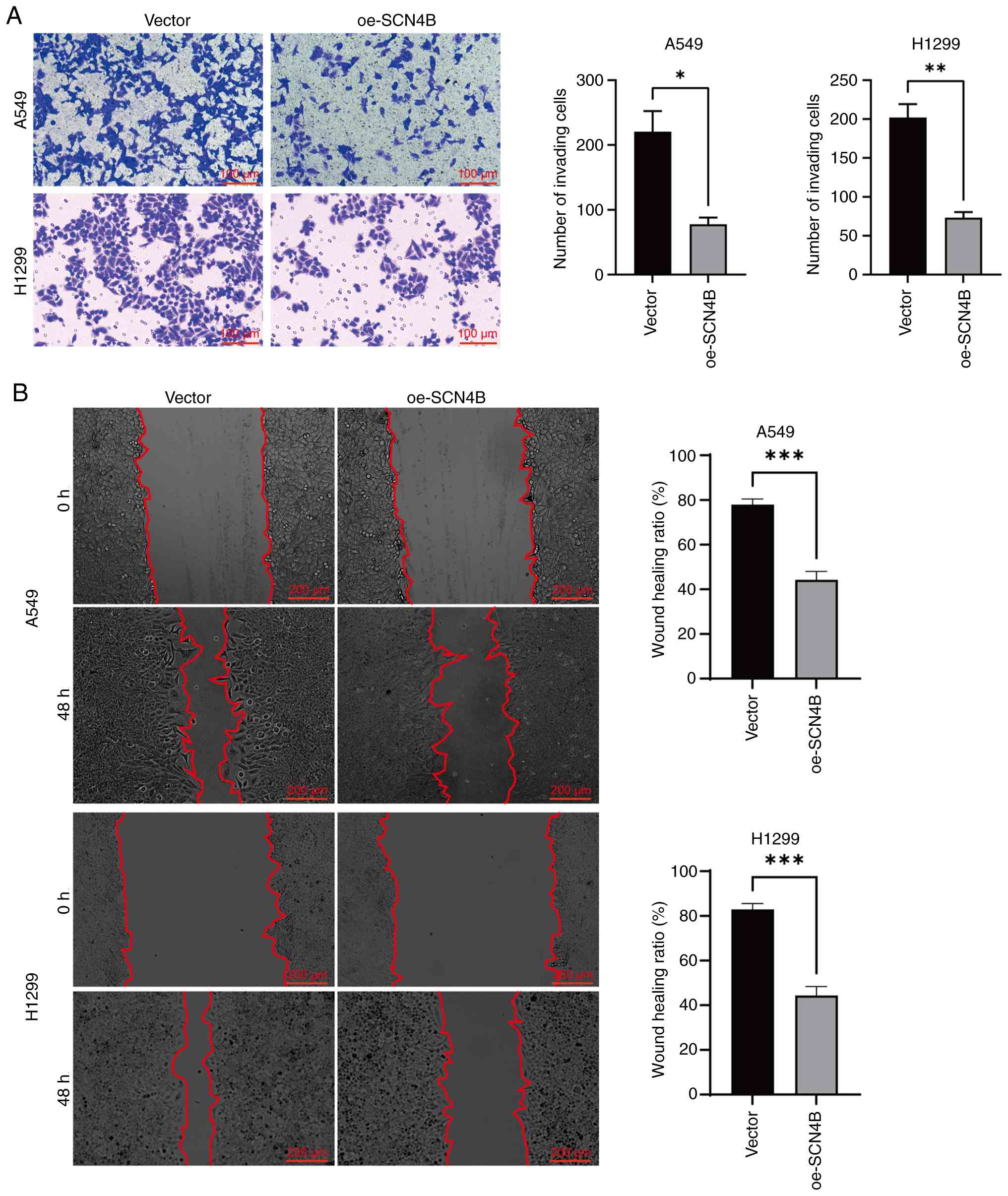
Li X, Chen W, Jiang S, Zhang L, Huang H,
Ji Y, Ni Q and Ling C: Low expression of SCN4B predicts poor
prognosis in Non-small cell lung cancer. Curr Cancer Drug Targets.
25:445–466. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
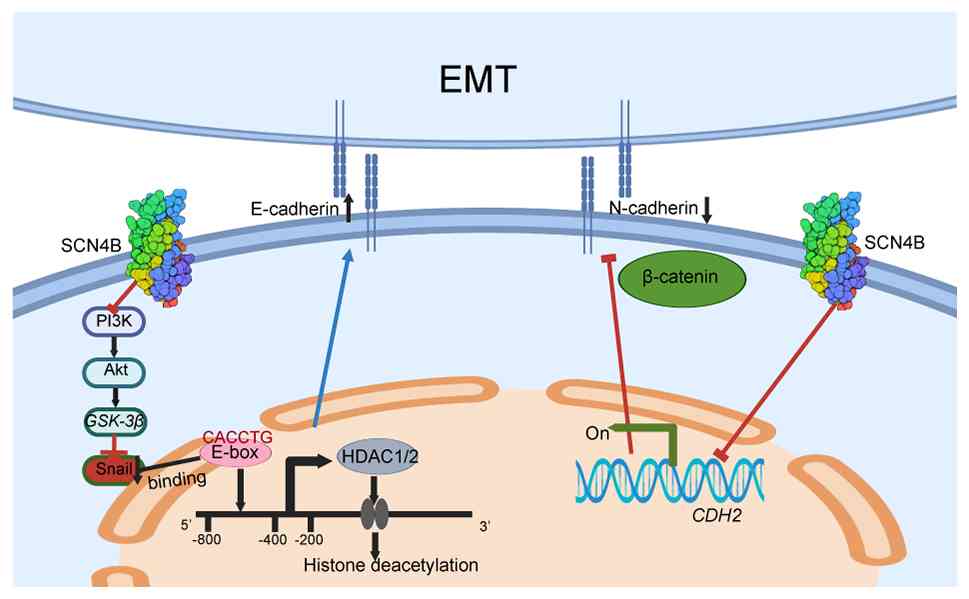
Peinado H, Ballestar E, Esteller M and
Cano A: Snail mediates E-cadherin repression by the recruitment of
the Sin3A/histone deacetylase 1 (HDAC1)/HDAC2 complex. Mol Cell
Biol. 24:306–319. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ma Q, Ye S, Liu H, Zhao Y, Mao Y and Zhang
W: HMGA2 promotes cancer metastasis by regulating
epithelial-mesenchymal transition. Front Oncol. 14:13208872024.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
R Core Team R, . A language and
environment for statistical computing. R Foundation for Statistical
Computing; Vienna, Austria: 2025, URL. http://www.R-project.org/
|
|
27
|
Hughes G, Kopetzky J and McRoberts N:
Mutual information as a performance measure for binary predictors
characterized by both ROC curve and PROC curve analysis. Entropy
(Basel). 22:9382020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gustavsson EK, Zhang D, Reynolds RH,
Garcia-Ruiz S and Ryten M: Ggtranscript: An R package for the
visualization and interpretation of transcript isoforms using
ggplot2. Bioinformatics. 38:3844–3846. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wu X, Sui Z, Zhang H, Wang Y and Yu Z:
Integrated analysis of lncRNA-mediated ceRNA network in lung
adenocarcinoma. Front Oncol. 10:5547592020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hess AS and Hess JR: Kaplan-Meier survival
curves. Transfusion. 60:670–672. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yamauchi M, Yamaguchi R, Nakata A, Kohno
T, Nagasaki M, Shimamura T, Imoto S, Saito A, Ueno K, Hatanaka Y,
et al: Epidermal growth factor receptor tyrosine kinase defines
critical prognostic genes of stage I lung adenocarcinoma. PLoS One.
7:e439232012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Feng X, Zahed H, Onwuka J, Callister M,
Johansson M, Etzioni R and Robbins HA: Cancer stage compared with
mortality as end points in randomized clinical trials of cancer
screening: A systematic review and Meta-analysis. JAMA.
331:1910–1917. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Qin H, Lu H, Qin C, Huang X, Peng K, Li Y,
Lan C, Bi A, Huang Z, Wei Y, et al: Pan-cancer analysis suggests
that LY6H is a potential biomarker of diagnosis,
immunoinfiltration, and prognosis. J Cancer. 15:5515–5539. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wang Y, Wang H, Pan T, Li L, Li J and Yang
H: STIM1 silencing inhibits the migration and invasion of A549
cells. Mol Med Rep. 16:3283–3289. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Albuquerque C, Manguinhas R, Costa JG, Gil
N, Codony-Servat J, Castro M, Miranda JP, Fernandes AS, Rosell R
and Oliveira NG: A narrative review of the migration and invasion
features of non-small cell lung cancer cells upon xenobiotic
exposure: Insights from in vitro studies. Transl Lung Cancer Res.
10:2698–2714. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Guo Q, Yu W, Tan J, Zhang J, Chen J, Rao
S, Guo X and Cai K: Remodelin delays non-small cell lung cancer
progression by inhibiting NAT10 via the EMT pathway. Cancer Med.
13:e72832024. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu X, Chen Y, Li Y, Petersen RB and Huang
K: Targeting mitosis exit: A brake for cancer cell proliferation.
Biochim Biophys Acta Rev Cancer. 1871:179–191. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Venugopal N, Yeh J, Kodeboyina SK, Lee TJ,
Sharma S, Patel N and Sharma A: Differences in the early stage gene
expression profiles of lung adenocarcinoma and lung squamous cell
carcinoma. Oncol Lett. 18:6572–6582. 2019.PubMed/NCBI
|
|
40
|
Seetharaman S and Etienne-Manneville S:
Cytoskeletal crosstalk in cell migration. Trends Cell Biol.
30:720–735. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Biswas KH: Molecular Mobility-mediated
regulation of E-cadherin adhesion. Trends Biochem Sci. 45:163–173.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Morana O, Wood W and Gregory CD: The
apoptosis paradox in cancer. Int J Mol Sci. 23:13282022. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Moyer A, Tanaka K and Cheng EH: Apoptosis
in cancer biology and therapy. Annu Rev Pathol. 20:303–328. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yang S, Zhang D, Sun Q, Nie H, Zhang Y,
Wang X, Huang Y and Sun Y: Single-cell and spatial transcriptome
profiling identifies the transcription factor BHLHE40 as a driver
of EMT in metastatic colorectal cancer. Cancer Res. 84:2202–2217.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Corso G, Figueiredo J, De Angelis SP,
Corso F, Girardi A, Pereira J, Seruca R, Bonanni B, Carneiro P,
Pravettoni G, et al: E-cadherin deregulation in breast cancer. J
Cell Mol Med. 24:5930–5936. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Na TY, Schecterson L, Mendonsa AM and
Gumbiner BM: The functional activity of E-cadherin controls tumor
cell metastasis at multiple steps. Proc Natl Acad Sci USA.
117:5931–5937. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Osuka S, Zhu D, Zhang Z, Li C, Stackhouse
CT, Sampetrean O, Olson JJ, Gillespie GY, Saya H, Willey CD and Van
Meir EG: N-cadherin upregulation mediates adaptive radioresistance
in glioblastoma. J Clin Invest. 131:e1360982021. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Gerber TS, Ridder DA, Goeppert B, Brobeil
A, Stenzel P, Zimmer S, Jakel J, Metzig MO, Schwab R, Martin SZ, et
al: N-cadherin: A diagnostic marker to help discriminate primary
liver carcinomas from extrahepatic carcinomas. Int J Cancer.
154:1857–1868. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ridge KM, Eriksson JE, Pekny M and Goldman
RD: Roles of vimentin in health and disease. Gene Dev. 36:391–407.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Cui H, Huang J, Lei Y, Chen Q, Hu Z, Niu
J, Wei R, Yang K, Li H, Lu T, et al: Design and synthesis of dual
inhibitors targeting snail and histone deacetylase for the
treatment of solid tumour cancer. Eur J Med Chem. 229:1140822022.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Huang Y, Hong W and Wei X: The molecular
mechanisms and therapeutic strategies of EMT in tumor progression
and metastasis. J Hematol Oncol. 15:1292022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hao Y, Baker D and Ten DP: TGF-β-Mediated
Epithelial-mesenchymal transition and cancer metastasis. Int J Mol
Sci. 20:27672019. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Brackenbury WJ and Isom LL: Na Channel β
Subunits: Overachievers of the ion channel family. Front Pharmacol.
2:532011. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Peng L, Gao R, Han L, Fu T and Bian C:
Roles of β-catenin protein in non-small cell lung cancer (Review).
Oncol Lett. 30:5652025. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yu F, Yu C, Li F, Zuo Y, Wang Y, Yao L, Wu
C, Wang C and Ye L: Wnt/β-catenin signaling in cancers and targeted
therapies. Signal Transduct Tar. 6:3072021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Fontana R, Mestre-Farrera A and Yang J:
Update on Epithelial-mesenchymal plasticity in cancer progression.
Annu Rev Pathol. 19:133–156. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Haddadin L and Sun X: Stem cells in
cancer: From mechanisms to therapeutic strategies. Cells.
14:5382025. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Hu Q, Saleem K, Pandey J, Charania AN,
Zhou Y and He C: Cell adhesion molecules in fibrotic diseases.
Biomedicines. 11:19952023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lin W, Fang J, Wei S, He G, Liu J, Li X,
Peng X, Li D, Yang S, Li X, et al: Extracellular vesicle-cell
adhesion molecules in tumours: Biofunctions and clinical
applications. Cell Commun Signal. 21:2462023. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Morla S: Glycosaminoglycans and
glycosaminoglycan mimetics in cancer and inflammation. Int J Mol
Sci. 20:19362019. View Article : Google Scholar
|
|
61
|
Xiao Y and Yu D: Tumor microenvironment as
a therapeutic target in cancer. Pharmacol Ther. 221:1077532021.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Du T, Gao J, Li P, Wang Y, Qi Q, Liu X, Li
J, Wang C and Du L: Pyroptosis, metabolism, and tumor immune
microenvironment. Clin Transl Med. 11:e4922021. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Elhanani O, Ben-Uri R and Keren L: Spatial
profiling technologies illuminate the tumor microenvironment.
Cancer Cell. 41:404–420. 2023. View Article : Google Scholar : PubMed/NCBI
|