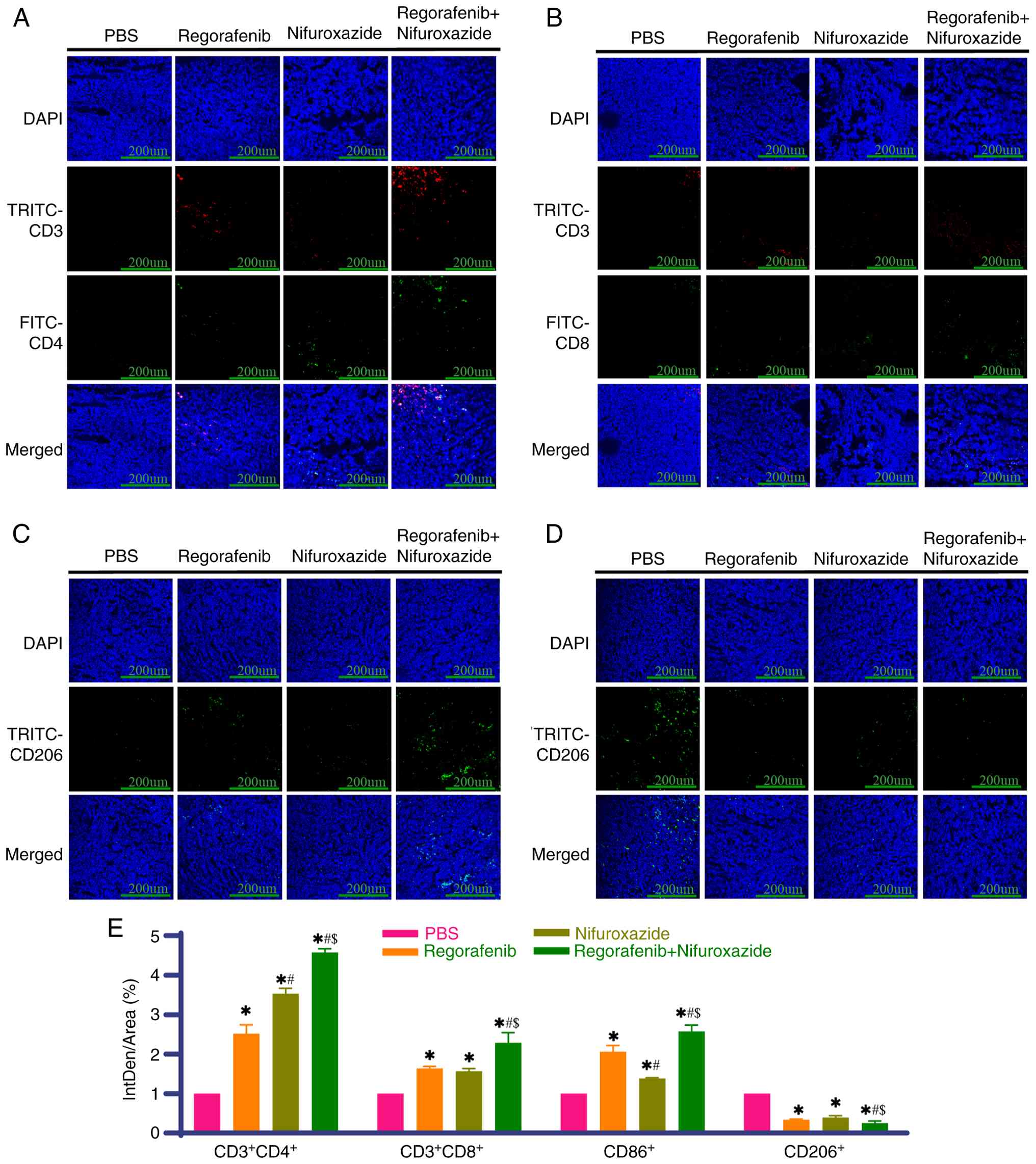
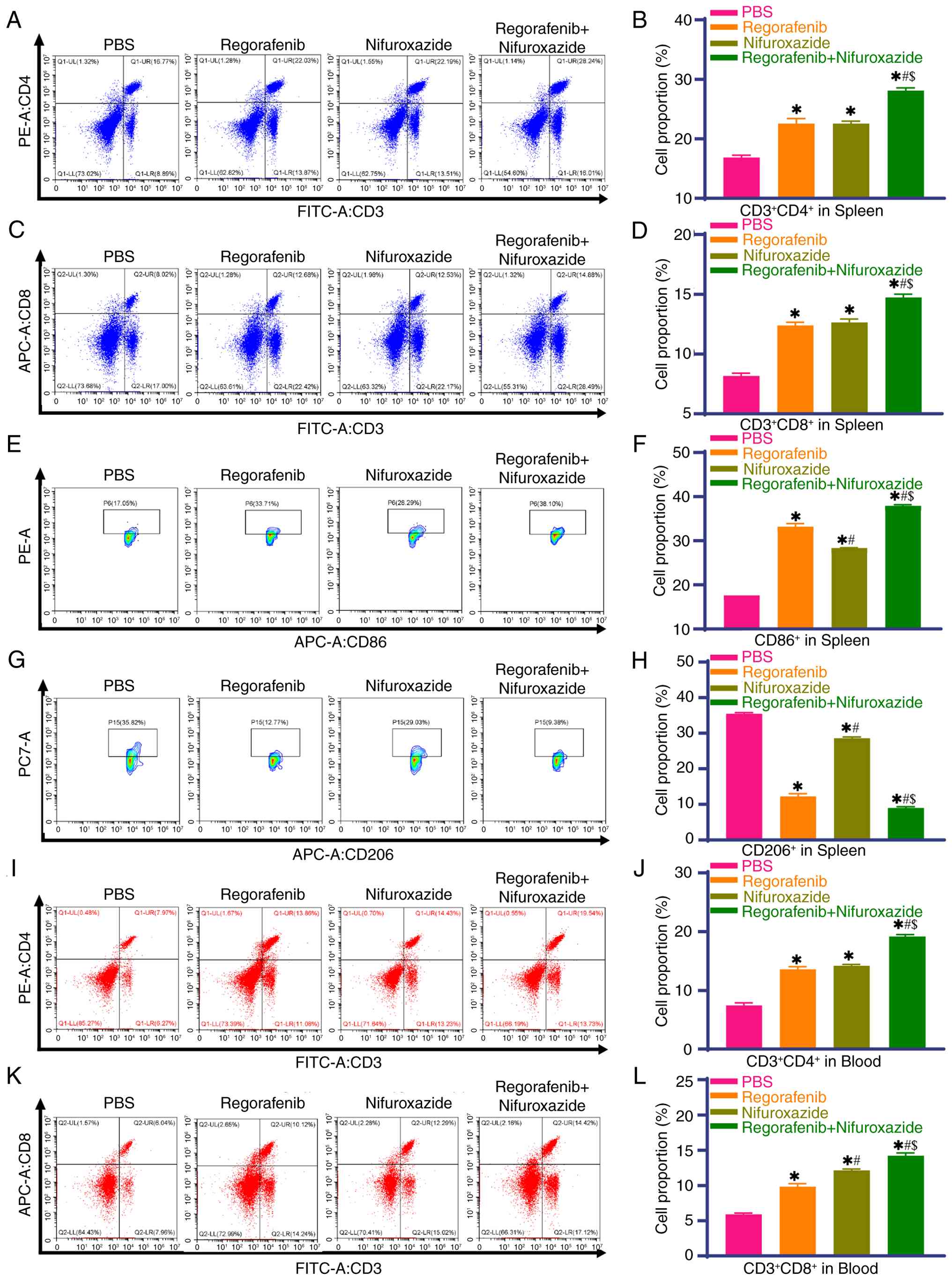
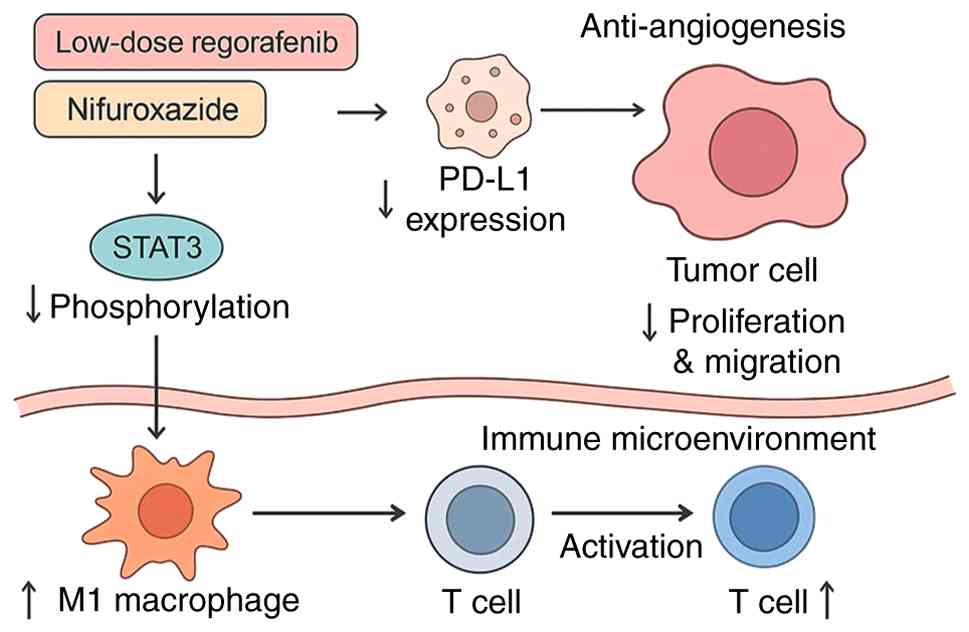
|
1
|
Anwanwan D, Singh SK, Singh S, Saikam V
and Singh R: Challenges in liver cancer and possible treatment
approaches. Biochim Biophys Acta Rev Cancer. 1873:1883142020.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rumgay H, Arnold M, Ferlay J, Lesi O,
Cabasag CJ, Vignat J, Laversanne M, McGlynn KA and Soerjomataram I:
Global burden of primary liver cancer in 2020 and predictions to
2040. J Hepatol. 77:1598–1606. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Marrero JA, Kulik LM, Sirlin CB, Zhu AX,
Finn RS, Abecassis MM, Roberts LR and Heimbach JK: Diagnosis,
staging and management of hepatocellular carcinoma: 2018 practice
guidance by the american association for the study of liver
diseases. Hepatology. 68:723–750. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Brown ZJ, Tsilimigras DI, Ruff SM, Mohseni
A, Kamel IR, Cloyd JM and Pawlik TM: Management of hepatocellular
carcinoma: A review. JAMA Surg. 158:410–420. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kudo M: Targeted and immune therapies for
hepatocellular carcinoma: Predictions for 2019 and beyond. World J
Gastroenterol. 25:789–807. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Guo X, Zhao Z, Zhu L, Liu S, Zhou L, Wu F,
Fang S, Chen M, Zheng L and Ji J: The evolving landscape of
biomarkers for systemic therapy in advanced hepatocellular
carcinoma. Biomark Res. 13:602025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Demols A, Borbath I, Van den Eynde M,
Houbiers G, Peeters M, Marechal R, Delaunoit T, Goemine JC, Laurent
S, Holbrechts S, et al: Regorafenib after failure of gemcitabine
and platinum-based chemotherapy for locally advanced/metastatic
biliary tumors: REACHIN, a randomized, double-blind, phase II
trial. Ann Oncol. 31:1169–1177. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bruix J, Qin S, Merle P, Granito A, Huang
YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, et al:
Regorafenib for patients with hepatocellular carcinoma who
progressed on sorafenib treatment (RESORCE): A randomised,
double-blind, placebo-controlled, phase 3 trial. Lancet. 389:56–66.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Grothey A, Van Cutsem E, Sobrero A, Siena
S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C, et
al: Regorafenib monotherapy for previously treated metastatic
colorectal cancer (CORRECT): An international, multicentre,
randomised, placebo-controlled, phase 3 trial. Lancet. 381:303–312.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ou DL, Chen CW, Hsu CL, Chung CH, Feng ZR,
Lee BS, Cheng AL, Yang MH and Hsu C: Regorafenib enhances antitumor
immunity via inhibition of p38 kinase/Creb1/Klf4 axis in
tumor-associated macrophages. J Immunother Cancer. 9:e0016572021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin YY, Tan CT, Chen CW, Ou DL, Cheng AL
and Hsu C: Immunomodulatory effects of current targeted therapies
on hepatocellular carcinoma: Implication for the future of
immunotherapy. Semin Liver Dis. 38:379–388. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bajpai P, Agarwal S, Afaq F, Al Diffalha
S, Chandrashekar DS, Kim HG, Shelton A, Miller CR, Singh SK, Singh
R, et al: Combination of dual JAK/HDAC inhibitor with regorafenib
synergistically reduces tumor growth, metastasis and
regorafenib-induced toxicity in colorectal cancer. J Exp Clin
Cancer Res. 43:1922024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Al Zaid Siddiquee K and Turkson J: STAT3
as a target for inducing apoptosis in solid and hematological
tumors. Cell Res. 18:254–267. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Begovic B, Ahmedtagic S, Calkic L,
Vehabović M, Kovacevic SB, Catic T and Mehic M: Open Clinical trial
on using nifuroxazide compared to probiotics in treating acute
diarrhoeas in adults. Mater Sociomed. 28:454–458. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Luo L, Xu F, Peng H, Luo Y, Tian X,
Battaglia G, Zhang H, Gong Q, Gu Z and Luo K: Stimuli-responsive
polymeric prodrug-based nanomedicine delivering nifuroxazide and
doxorubicin against primary breast cancer and pulmonary metastasis.
J Control Release. 318:124–135. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhao T, Wei P, Zhang C, Zhou S, Liang L,
Guo S, Yin Z, Cheng S, Gan Z, Xia Y, et al: Nifuroxazide suppresses
PD-L1 expression and enhances the efficacy of radiotherapy in
hepatocellular carcinoma. Elife. 12:RP909112024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen P, Li K, Chen J, Hei H, Geng J, Huang
N, Lei M, Jia H, Ren J and Jin C: Enhanced effect of radiofrequency
ablation on HCC by siRNA-PD-L1-endostatin Co-expression plasmid
delivered. Transl Oncol. 53:1023192025. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ye TH, Yang FF, Zhu YX, Li YL, Lei Q, Song
XJ, Xia Y, Xiong Y, Zhang LD, Wang NY, et al: Inhibition of STAT3
signaling pathway by nifuroxazide improves antitumor immunity and
impairs colorectal carcinoma metastasis. Cell Death Dis.
8:e25342017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Aden DP, Fogel A, Plotkin S, Damjanov I
and Knowles BB: Controlled synthesis of HBsAg in a differentiated
human liver carcinoma-derived cell line. Nature. 282:615–616. 1979.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Knowles BB, Howe CC and Aden DP: Human
hepatocellular carcinoma cell lines secrete the major plasma
proteins and hepatitis B surface antigen. Science. 209:497–499.
1980. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
López-Terrada D, Cheung SW, Finegold MJ
and Knowles BB: Hep G2 is a hepatoblastoma-derived cell line. Hum
Pathol. 40:1512–1515. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
de Souza GO, Wasinski F and Donato J:
Characterization of the metabolic differences between male and
female C57BL/6 mice. Life Sci. 301:1206362022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Subramanian A, Tamayo P, Mootha VK,
Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub
TR, Lander ES and Mesirov JP: Gene set enrichment analysis: A
knowledge-based approach for interpreting genome-wide expression
profiles. Proc Natl Acad Sci USA. 102:15545–15550. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Poon RTP, Fan ST and Wong J: Clinical
significance of angiogenesis in gastrointestinal cancers: A target
for novel prognostic and therapeutic approaches. Ann Surg.
238:9–28. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cheng AL, Hsu C, Chan SL, Choo SP and Kudo
M: Challenges of combination therapy with immune checkpoint
inhibitors for hepatocellular carcinoma. J Hepatol. 72:307–319.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Amin A, Plimack ER, Ernstoff MS, Lewis LD,
Bauer TM, McDermott DF, Carducci M, Kollmannsberger C, Rini BI,
Heng DYC, et al: Safety and efficacy of nivolumab in combination
with sunitinib or pazopanib in advanced or metastatic renal cell
carcinoma: The CheckMate 016 study. J Immunother Cancer. 6:1092018.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xu J, Zhang Y, Jia R, Yue C, Chang L, Liu
R, Zhang G, Zhao C, Zhang Y, Chen C, et al: Anti-PD-1 antibody
SHR-1210 combined with apatinib for advanced hepatocellular
carcinoma, gastric, or esophagogastric junction cancer: An
open-label, dose escalation and expansion study. Clin Cancer Res.
25:515–523. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Finn RS, Ikeda M, Zhu AX, Sung MW, Baron
AD, Kudo M, Okusaka T, Kobayashi M, Kumada H, Kaneko S, et al:
Phase Ib study of lenvatinib plus pembrolizumab in patients with
unresectable hepatocellular carcinoma. J Clin Oncol. 38:2960–2970.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Killock D: Liver cancer: Regorafenib-a new
RESORCE in HCC. Nat Rev Clin Oncol. 14:70–71. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hecht JR, Park YS, Tabernero J, Lee MA,
Lee S, Virgili AC, Van den Eynde M, Fontana E, Fakih M, Asghari G,
et al: Zanzalintinib plus atezolizumab versus regorafenib in
refractory colorectal cancer (STELLAR-303): A randomised,
open-label, phase 3 trial. Lancet. 406:2360–2370. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bailly C: Toward a repositioning of the
antibacterial drug nifuroxazide for cancer treatment. Drug Discov
Today. 24:1930–1936. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hu B, Zou T, Qin W, Shen X, Su Y, Li J,
Chen Y, Zhang Z, Sun H, Zheng Y, et al: Inhibition of EGFR
overcomes acquired lenvatinib resistance driven by STAT3-ABCB1
signaling in hepatocellular carcinoma. Cancer Res. 82:3845–3857.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zou S, Tong Q, Liu B, Huang W, Tian Y and
Fu X: Targeting STAT3 in cancer immunotherapy. Mol Cancer.
19:1452020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tai WT, Chu PY, Shiau CW, Chen YL, Li YS,
Hung MH, Chen LJ, Chen PL, Su JC, Lin PY, et al: STAT3 mediates
regorafenib-induced apoptosis in hepatocellular carcinoma. Clin
Cancer Res. 20:5768–5776. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Voli F, Valli E, Lerra L, Kimpton K,
Saletta F, Giorgi FM, Mercatelli D, Rouaen JRC, Shen S, Murray JE,
et al: Intratumoral copper modulates PD-L1 expression and
influences tumor immune evasion. Cancer Res. 80:4129–4144. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang X, Yang X, Zhang C, Wang Y, Cheng T,
Duan L, Tong Z, Tan S, Zhang H, Saw PE, et al: Tumor cell-intrinsic
PD-1 receptor is a tumor suppressor and mediates resistance to PD-1
blockade therapy. Proc Natl Acad Sci USA. 117:6640–6650. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xiong H, Mittman S, Rodriguez R,
Moskalenko M, Pacheco-Sanchez P, Yang Y, Nickles D and Cubas R:
Anti-PD-L1 treatment results in functional remodeling of the
macrophage compartment. Cancer Res. 79:1493–1506. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Tabrizian P, Abdelrahim M and Schwartz M:
Immunotherapy and transplantation for hepatocellular carcinoma. J
Hepatol. 80:822–825. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wu TD, Madireddi S, de Almeida PE,
Banchereau R, Chen YJ, Chitre AS, Chiang EY, Iftikhar H, O'Gorman
WE, Au-Yeung A, et al: Peripheral T cell expansion predicts tumour
infiltration and clinical response. Nature. 579:274–278. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Sun L, Su Y, Jiao A, Wang X and Zhang B: T
cells in health and disease. Signal Transduct Target Ther.
8:2352023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fridman WH, Pagès F, Sautès-Fridman C and
Galon J: The immune contexture in human tumours: Impact on clinical
outcome. Nat Rev Cancer. 12:298–306. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Varol C, Mildner A and Jung S:
Macrophages: Development and tissue specialization. Annu Rev
Immunol. 33:643–675. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Lavin Y, Mortha A, Rahman A and Merad M:
Regulation of macrophage development and function in peripheral
tissues. Nat Rev Immunol. 15:731–744. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sharma A, Seow JJW, Dutertre CA, Pai R,
Blériot C, Mishra A, Wong RMM, Singh GSN, Sudhagar S, Khalilnezhad
S, et al: Onco-fetal reprogramming of endothelial cells drives
immunosuppressive macrophages in hepatocellular carcinoma. Cell.
183:377–394.e21. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lu LG, Zhou ZL, Wang XY, Liu BY, Lu JY,
Liu S, Zhang GB, Zhan MX and Chen Y: PD-L1 blockade liberates
intrinsic antitumourigenic properties of glycolytic macrophages in
hepatocellular carcinoma. Gut. 71:2551–2560. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cheng K, Cai N, Zhu J, Yang X, Liang H and
Zhang W: Tumor-associated macrophages in liver cancer: From
mechanisms to therapy. Cancer Commun (Lond). 42:1112–1140. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Locati M, Curtale G and Mantovani A:
Diversity, mechanisms and significance of macrophage plasticity.
Annu Rev Pathol. 15:123–147. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yeung OW, Lo CM, Ling CC, Qi X, Geng W, Li
CX, Ng KT, Forbes SJ, Guan XY, Poon RT, et al: Alternatively
activated (M2) macrophages promote tumour growth and invasiveness
in hepatocellular carcinoma. J Hepatol. 62:607–616. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kadioglu E and De Palma M: Cancer
metastasis: Perivascular macrophages under watch. Cancer Discov.
5:906–908. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gabrilovich DI, Chen HL, Girgis KR,
Cunningham HT, Meny GM, Nadaf S, Kavanaugh D and Carbone DP:
Production of vascular endothelial growth factor by human tumors
inhibits the functional maturation of dendritic cells. Nat Med.
2:1096–1103. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Rahma OE and Hodi FS: The intersection
between tumor angiogenesis and immune suppression. Clin Cancer Res.
25:5449–5457. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhang H, Liu L, Liu J, Dang P, Hu S, Yuan
W, Sun Z, Liu Y and Wang C: Roles of tumor-associated macrophages
in anti-PD-1/PD-L1 immunotherapy for solid cancers. Mol Cancer.
22:582023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
National Standard of the People's Republic
of China, . Laboratory animal - General requirements for biosafety
in animal experiment (GB/T 43051-2023) [S]. National Standard of
the People's Republic of China; Beijing: 2023
|