Introduction
Primary liver cancer is the sixth most common cancer
type worldwide and the third leading cause of cancer-related
mortality (1). It is predicted that
the number of new cases of liver cancer will increase by 55.0%
between 2020 and 2040, with an estimated 1.4 million new cases of
hepatocellular carcinoma (HCC) expected in 2040 (2). With its continuously increasing
incidence and mortality rates, HCC imposes a significant and
challenging health burden globally (3). However, most patients with HCC are
diagnosed with intermediate or advanced stage cancer in the first
instance and only 15–25% are eligible for curative resection
(1). For patients with intermediate
or advanced liver cancer, systemic therapies such as immunotherapy
or targeted therapy are the preferred approaches (4).
Regorafenib, an oral multi-kinase inhibitor, can
inhibit the activation of multiple target kinases, including
rapidly accelerated fibrosarcoma (RAF), vascular endothelial growth
factor receptor (VEGFR) and platelet-derived growth factor receptor
(PDGFR). It functions by inhibiting angiogenesis and tumor cell
proliferation and has become a cornerstone of second-line therapy
for the management of advanced HCC (5,6).
Several clinical trials have evaluated Regorafenib in patients with
previously treated HCC. The REACHIN and RESORCE trials demonstrated
significant improvements in progression-free survival (PFS) and
tumor control benefits with Regorafenib in second or third-line
settings (7,8). However, its clinical application is
still plagued by multiple challenges: The REACHIN, RESORCE and
CORRECT trials showed that the use at the recommended dose (160 mg
daily) is often limited by prominent treatment-related adverse
events, including hand-foot skin reactions, diarrhea and
hypertension, among other adverse events (7–9). In a
clinical trial of Regorafenib combined with nivolumab, the
combination of 80 mg Regorafenib with nivolumab showed improved
safety and effective anti-tumor activity in previously treated
patients with advanced gastric or colorectal cancer (10). More importantly, the
immunomodulatory effects of Regorafenib are dose-dependent: High
doses may induce a hypoxic tumor microenvironment, recruit
immunosuppressive cells such as tumor-associated macrophages (TAMs)
and thereby exacerbate immune evasion (11). By contrast, low doses can promote
favorable macrophage polarization and boost T cell activity.
Researchers have demonstrated that a low concentration of
Regorafenib, combined with JAK/HDAC small-molecule inhibitors or
anti-PD-1 therapy, exerts synergistic anti-tumor efficacy with
higher Regorafenib bioavailability (10,12).
These observations strongly suggest that the optimal
immunomodulatory dose of Regorafenib may be lower than the
recommended monotherapy dose, underscoring the need for combination
therapy.
STAT3 is a member of the signal transducer and
activator of transcription (STAT) family. In response to cytokines
and growth factors, STAT3 is phosphorylated and mediates the
expression of various genes. Sustained activation of STAT3 can
induce cancer through multiple mechanisms, including reducing tumor
cell apoptosis, increasing tumor cell proliferation, promoting
tumor angiogenesis and regulating tumor immune evasion (13). Nifuroxazide is a nitrofuran
derivative with broad-spectrum antibacterial activity (14). Nifuroxazide can reduce the viability
of various cancer cells by inhibiting STAT3 phosphorylation
(15). More importantly,
Nifuroxazide can inhibit programmed death ligand 1 (PD-L1)
expression in HCC, promote T cell activation and enhance anti-tumor
immune responses (16,17). Ye et al (18) also found that Nifuroxazide reduced
the number of myeloid-derived suppressor cells and M2-type
macrophages in tumors, exerting a positive effect on the tumor
immune microenvironment. However, tumor progression is extremely
complex and the efficacy of single-agent therapy is limited,
necessitating more aggressive treatment strategies.
Therefore, the present study was performed to
evaluate the efficacy of Regorafenib combined with Nifuroxazide and
to determine whether this combination therapy enhanced the
therapeutic effects. In the present study, the role of this
combination in enhancing therapeutic efficacy was assessed using
in vitro and in vivo preclinical HCC models. It was
found that Regorafenib combined with Nifuroxazide exerted enhanced
anti-angiogenic effects, promoted tumor cell apoptosis and
inhibited the proliferation and migration of HCC cells.
Additionally, low-dose Regorafenib promoted immune cell
infiltration and polarized macrophages toward an M1 phenotype,
thereby enhancing cytotoxic T cell functions and anti-tumor
immunity, with the combination therapy showing further enhanced
effects.
Materials and methods
Reagents and cell lines
Human HepG2 cells and the mouse H22 hepatoma cells
were obtained from Professor Xuejian Zhao (Department of
Pathophysiology, Prostate Diseases Prevention and Treatment
Research Centre, Norman Bethune College of Medicine, Jilin
University, Changchun, China). All cell lines used in the present
study have been authenticated to ensure identity and purity. Human
HepG2 cells were authenticated by short tandem repeat (STR)
profiling, which matched the reference STR data of ATCC HB-8065.
Mouse H22 hepatoma cells were verified by morphological observation
and functional validation (consistent tumorigenicity in C57BL/6
mice). The two cell lines were tested negative for mycoplasma
contamination using a mycoplasma detection kit (Beyotime
Biotechnology; cat. no. C0301). HepG2 cells, which were initially
reported as a hepatoma cell line in early studies (19) but subsequently classified as a
hepatoblastoma cell line (20,21),
are widely used in liver cancer research. HepG2 cells were cultured
in high-glucose DMEM (Thermo Fisher Scientific Inc.) with 1%
penicillin-streptomycin and 10% FBS (Sunrise Material Co., Ltd.) at
37°C in a humidified atmosphere containing 5% CO2. H22
cells were expanded via ascites in C57BL/6 mice (Henan Skobes
Biotechnology Co., Ltd.). Nifuroxazide (MilliporeSigma) and
Regorafenib (Bayer AG) were used in the present study; the
therapeutic doses of Nifuroxazide used were based on previous
studies for maximal therapeutic benefit (10 µg/ml in HepG2 and 10
mg/kg/day in mouse models) (16,18).
Regorafenib doses (5 µg/ml in HepG2 cells and 5 mg/kg/day in mouse
models) were lower than clinical recommendations, but were based on
a previous study (10).
Cell viability assay
HepG2 cells were seeded at a density of
1×104 cells/well in 96-well plates (Corning, Inc.) and
cultured in high-glucose DMEM supplemented with 10% FBS and 1%
penicillin-streptomycin at 37°C in a humidified atmosphere
containing 5% CO2 for 16 h, after which Regorafenib and
Nifuroxazide were added. After 24 h of treatment, 10 µl CCK-8
solution (Beyotime Biotechnology) was added and cells were
incubated for a further 2 h. Optical density was measured at 450 nm
using a microplate reader (SpectraMax iD3; Molecular Devices,
LLC).
Wound healing assay
HepG2 cells were seeded at a density of
3.5×105 cells/well in six-well plates and cultured for
16 h at 37°C. The cells were then serum-starved (no FBS) for 24 h
at 37°C. Scratches were created in the center of each well using a
200 µl pipette tip, then Regorafenib (5 µg/ml) and Nifuroxazide (10
µg/ml) were added. After 24 and 48 h, scratch widths were measured
and recorded using a light microscope (Nikon Corporation). For
quantification, scratch widths were analyzed via ImageJ software
(version 1.54f; National Institutes of Health); wound healing
rate=(Initial scratch width-width at 24/48 h)/Initial scratch width
×100%, normalized wound healing rate=(wound healing rate of each
group/average wound healing rate of the control group), and each
sample was measured in ≥3 random fields with mean values for
statistical analysis.
Colony formation assay
HepG2 cells were seeded at a density of
2×103 cells/well in six-well plates and cultured for
14–16 h at 37°C, after which Regorafenib (5 µg/ml) and Nifuroxazide
(10 µg/ml) were added. The media was replaced periodically. Cell
colonies visible to the naked eye appeared on day 10, at which
point, they were measured and recorded.
Apoptosis assay
Apoptosis was assessed using an Annexin V-FITC kit.
HepG2 cells were seeded at a density of 3.5×105
cells/well in six-well plates and cultured for 14–16 h at 37°C.
Regorafenib (5 µg/ml) and Nifuroxazide (10 µg/ml) were added and
cells were incubated for 24 or 48 h at 37°C. Cells were harvested,
stained with Annexin V-FITC and PI (Dojindo Laboratories, Inc.) at
room temperature (25±2°C) in the dark for 15 min, resuspended in
400 µl PBS and analyzed using a flow cytometer (Beckman Coulter,
Inc.) with analysis performed using CytExpert Software (version
2.1; Beckman Coulter, Inc.) and the apoptotic rate calculated as
the sum of the percentage of early apoptotic cells and late
apoptotic cells.
Establishment of a mouse HCC
model
Female C57BL/6 mice (6–8 weeks old, weighing 16–20
g, n=24) were obtained from Henan Skobes Biotechnology Co., Ltd.
Female mice were selected because they exhibit more stable
metabolism, stronger resistance to diet-induced obesity and
metabolic imbalances, higher leptin sensitivity and compensatory
hypothalamic Pomc mRNA expression, all reducing experimental
variability and ensuring reliable tumorigenicity of H22 cells
(22). Mice were housed in a
specific pathogen-free grade animal room with individually
ventilated cages at 24±2°C, at 40–50% humidity, with a 12-h
light/dark cycle and ad libitum access to sterile
food/water. H22 cells (2×106 cells/100 µl) were
subcutaneously injected into the right dorsal side of mice. To
minimize animal suffering, humane endpoints were set: Tumor volume
≤2,000 mm3, >20% initial weight loss in 3 consecutive
days, or severe distress (such as labored breathing, inability to
access food/water); mice meeting any endpoint were sacrificed
immediately. All mice were sacrificed in accordance with the preset
experimental protocol at the end of the study.
A total of 7 days post-injection, mice were randomly
divided into four groups (n=6 per group): PBS group (daily gavage
of 100 µl PBS); Regorafenib group (daily gavage, 5 mg/kg);
Nifuroxazide group (daily gavage, 10 mg/kg); combination group
(daily Regorafenib gavage 5 mg/kg + Nifuroxazide gavage 10 mg/kg).
Treatments were administered daily for 7 days. Mice were sacrificed
via intraperitoneal injection of 2% tribromoethanol (20 ml/kg, deep
anesthesia confirmed by absent pedal reflex) followed by overdose
sodium pentobarbital (150 mg/kg) 1 day after completion of the
treatment regimen. Mortality was confirmed by cessation of
respiration/heartbeat, absent corneal reflex and persistent muscle
flaccidity. Subsequently, tumor tissues were excised, weighed and
images captured. All procedures were approved by the Animal Ethics
Committee of the First Affiliated Hospital of Zhengzhou University
(approval no. 2023-KY-1333) and adhered to the international 3R
Principles (replacement, reduction, refinement).
Flow cytometry
Spleen and peripheral blood cell suspensions were
prepared and treated with red blood cell lysis buffer (Beyotime
Biotechnology) to remove red cells. Cell density was adjusted to
1×107 cells/ml. A total of 100 µl suspension per sample
was stained with fluorescently-labeled antibodies against CD3
(BioLegend, Inc.; cat. no. 100204; 1:200), CD4 (BioLegend, Inc.;
cat. no. 100406; 1:200), CD8 (BioLegend, Inc.; cat. no. 100706;
1:200), CD45 (BD Biosciences; cat. no. 553080; 1:200), CD11b
(BioLegend, Inc.; cat. no. 101206; 1:200), CD206 (BioLegend, Inc.;
cat. no. 141708; 1:200), CD86 (BioLegend, Inc.; cat. no. 105006;
1:200), or F4/80 (BioLegend, Inc.; cat. no. 123116; 1:200)
according to the manufacturer's protocol. Samples were stained with
the aforementioned fluorescently-labeled antibodies incubated in
the dark at 4°C for 30 min, washed with pre-cooled PBS three times
and resuspended in 400 µl PBS. The fluorescence intensity was
measured using a CytoFLEX flow cytometer (Beckman Coulter,
Inc.).
Western blotting
Proteins were extracted using RIPA lysis buffer
(Beyotime Biotechnology, cat. no. P0013B) supplemented with 1%
protease inhibitor cocktail and 1% phosphatase inhibitor cocktail.
Protein concentration was determined via the BCA Protein Assay Kit
(Beyotime Biotechnology; cat. no. P0010) following the
manufacturer's protocol. A total of 10 µg protein per lane was
separated via SDS-PAGE using 10% Tris-Gly SDS-PAGE precast gels
(Beyotime Biotechnology; cat. no. P0052A), followed by transfer to
PVDF membranes (MilliporeSigma) at 220 mA. Membranes were blocked
with 5% skimmed milk in 0.05% Tween-20 at room temperature for 1 h,
then incubated overnight at 4°C with one of the following primary
antibodies: anti-PD-L1 (1:1,000; Shanghai Abways Biotechnology Co.,
Ltd.; cat. no. CY5980); anti-VEGF (1:2,000, Affinity Biosciences,
Ltd.; cat. no. AF5131); anti-p-STAT3 (1:1,000, Cell Signaling
Technology, Inc.; cat. no. 9145); anti-STAT3 (1:1,000, Cell
Signaling Technology, Inc.; cat. no. 9139); anti-Bcl-2 (1:2,000,
Shanghai Abways Biotechnology Co., Ltd.; cat. no. CY6717); anti-Bax
(1:2,000, Shanghai Abways Biotechnology Co., Ltd.; cat. no.
CY5059); anti-MMP2 (1:1,000, Cell Signaling Technology, Inc.; cat.
no. 40994); anti-cyclin D1 (1:2,000, Santa Cruz Biotechnology,
Inc.; cat. no. sc-8396); anti-cleaved-caspase 3 (1:2,000, Cell
Signaling Technology; cat. no. 9664); anti-caspase 3 (1:2,000, Cell
Signaling Technology; cat. no. 9662); phospho-JAK2 (1:1,000;
UpingBio technology Co, Ltd.; cat. no. YP-Ab-14423); JAK2 (1:1,000;
UpingBio technology Co, Ltd.; cat. no. YP-Ab-14800); SHP-1
(1:1,000; UpingBio technology Co, Ltd.; cat. no. YP-mAb-14236);
anti-Tubulin (1:4,000, MilliporeSigma; cat. no. T9026); and
anti-GAPDH (1:4,000, MilliporeSigma; cat. no. G8795) according to
the manufacturer's protocol. Following washing, membranes were
incubated with a horseradish peroxidase (HRP)-conjugated secondary
antibody (1:5,000; OriGene Technologies, Inc.; cat. nos.
TA373082/TA373083) at room temperature for 60 min. Signals were
visualized using an enhanced chemiluminescence kit (Beyotime
Biotechnology; cat. no. P0018) and imaged using a Fusion FX spectra
imaging system (Vilber Lourmat). Densitometric analysis was
performed using ImageJ software (version 1.54f; National Institutes
of Health, USA).
Immunofluorescence imaging
Immunofluorescence imaging was used to detect
CD4+ and CD8+ T cells and macrophage
infiltration in tumor tissues. Tumor sections were prepared as
follows: Freshly excised tumor tissues were fixed in 4%
paraformaldehyde (room temperature, 4–6 h), rinsed with PBS
(Beyotime Biotechnology), then dehydrated via gradient ethanol and
transparentized with xylene. Tissues were embedded in paraffin
(56–58°C), sectioned into 4-µm slices with a rotary microtome,
mounted on poly-L-lysine-coated slides and baked at 60°C for 2–3 h
to secure slices. Prior to primary antibody incubation, the
prepared tumor sections were dewaxed, rehydrated through a reversed
graded ethanol series, and rinsed thoroughly with PBS. Sections
were then blocked with 5% normal goat serum (Beyotime
Biotechnology, cat. no. C0265) for 30 min at room temperature to
eliminate non-specific antibody binding. The prepared tumor
sections were incubated with the following primary antibodies:
anti-CD3 (1:200; OmnimAbs; cat. no. OM244830); anti-CD4 (1:200;
Cell Signaling Technology, Inc.; cat. no. 25229); anti-CD8 (1:400;
Cell Signaling Technology, Inc.; cat. no. 98941); anti-CD206
(1:800; Cell Signaling Technology, Inc.; cat. no. 24595S);
anti-CD86 (1:200, Novus Biologicals LLC; cat. no. NBP2-25208); or
anti-Ki67 (1:100, Bioworld Technology, Inc.; cat. no. BS90769)
according to the manufacturer's protocol, overnight at 4°C. After
washing with PBS, sections were incubated with the
fluorescent-tagged secondary antibody (1:200; Shanghai Abways
Biotechnology Co., Ltd.; cat. no. CY3101) for 30 min at room
temperature. Following DAPI (Beyotime Biotechnology) staining at
room temperature (25±2°C) for 5 min and washing with PBS, slides
were mounted using an anti-fade solution, visualized and imaged
using a confocal microscope (AR1; Nikon Corporation) using a ×20
objective lens.
TUNEL assay
Apoptosis in tissues was assessed using a TUNEL
assay kit (Beyotime Biotechnology). Tumor sections were prepared,
treated with DNase-free Proteinase K, incubated at 37°C for 20 min,
washed with PBS and treated with TUNEL solution at 37°C for 60 min.
Following DAPI (Beyotime Biotechnology) counterstaining at room
temperature (25±2°C) for 5 min and washing with PBS, slides were
mounted using an anti-fade solution, visualized and imaged using a
confocal microscope (AR1; Nikon Corporation) using a ×20 objective
lens.
Clustering and Gene Set Enrichment
Analysis (GSEA)
All analyzed datasets were retrieved from the Gene
Expression Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/) under the accession
number GSE148947 (10). RNA-seq
data were processed using R software (version 4.1.0, https://www.r-project.org/), with the DESeq2, limma
and clusterProfiler packages employed to identify differential gene
expression (DGE) and perform Gene Ontology (GO), Kyoto Encyclopedia
of Genes and Genomes (KEGG) and Gene Set Enrichment Analysis
(GSEA). Gene sets for GSEA were obtained from the Molecular
Signatures Database (https://www.gsea-msigdb.org/gsea/msigdb) (23) and no custom (unique) gene sets were
applied in the analysis.
Statistical analysis
All experimental data were statistically analyzed
using SPSS 24.0 software (IBM Corp.) and GraphPad Prism 8 software
(Dotmatics). Results are presented as the mean ± standard deviation
(SD), with each experiment was repeated independently at least
three times to ensure reproducibility. Differences between multiple
groups were assessed using one-way analysis of variance (ANOVA)
followed by Tukey's HSD post hoc test. P<0.05 was considered to
indicate a statistically significant difference.
Results
Transcriptome and functional analyses
of the effects of Regorafenib and Nifuroxazide in HCC
Transcriptome profiling of Regorafenib-treated HCC
cells (P<0.05, log2FC >1) revealed differential
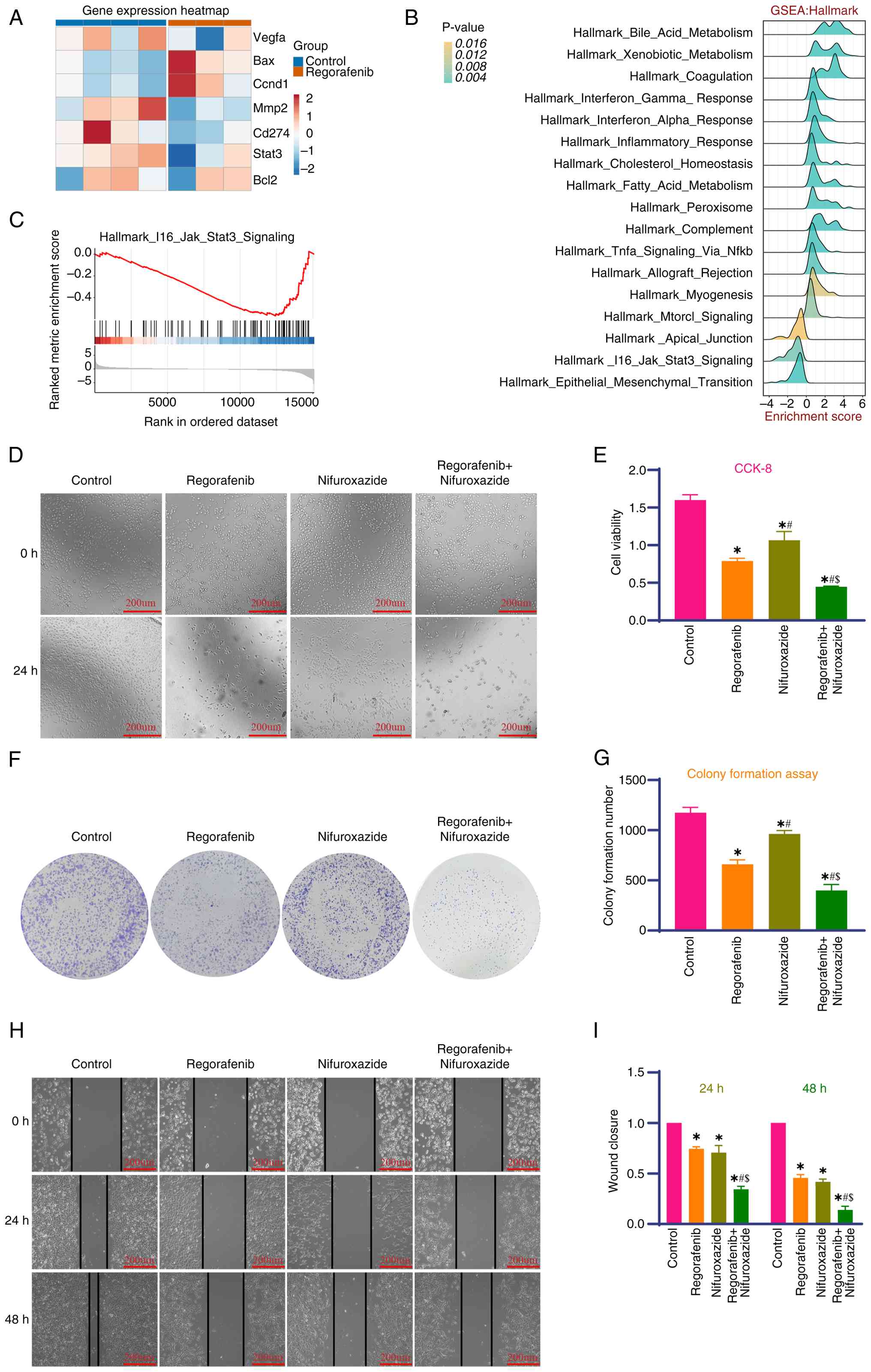
regulation of key genes; the results are shown as a heatmap
(Fig. 1A), with GSEA (Fig. 1B) identifying immune pathway
enrichment and inhibitory IL6-JAK-STAT3 signaling (Fig. 1C), indicative of STAT3-mediated
immune remodeling. Kyoto Encyclopedia of Genes and Genomes (KEGG)
and Gene Ontology (GO) enrichment analyses (Fig. S1) further delineated the following
functional implications: Upregulated differentially expressed genes
(DEGs) were clustered in immune-trafficking pathways (Fig. S1C) and GO terms for suppressed cell
motility (Fig. S1D), while
downregulated DEGs were associated with metabolic reprogramming
(Fig. S1E and F). In vitro,
CCK-8 (Fig. 1D and E) and colony
formation assays (Fig. 1F and G)
showed reduced proliferation and colony formation in HepG2 cells
when treated with a combination of Regorafenib and Nifuroxazide,
while wound-healing assays (Fig. 1H and
I) showed the combination group exhibited reduced migration at
both 24 and 48 h.
Effects of Nifuroxazide combined with
Regorafenib on apoptosis in HepG2 cells
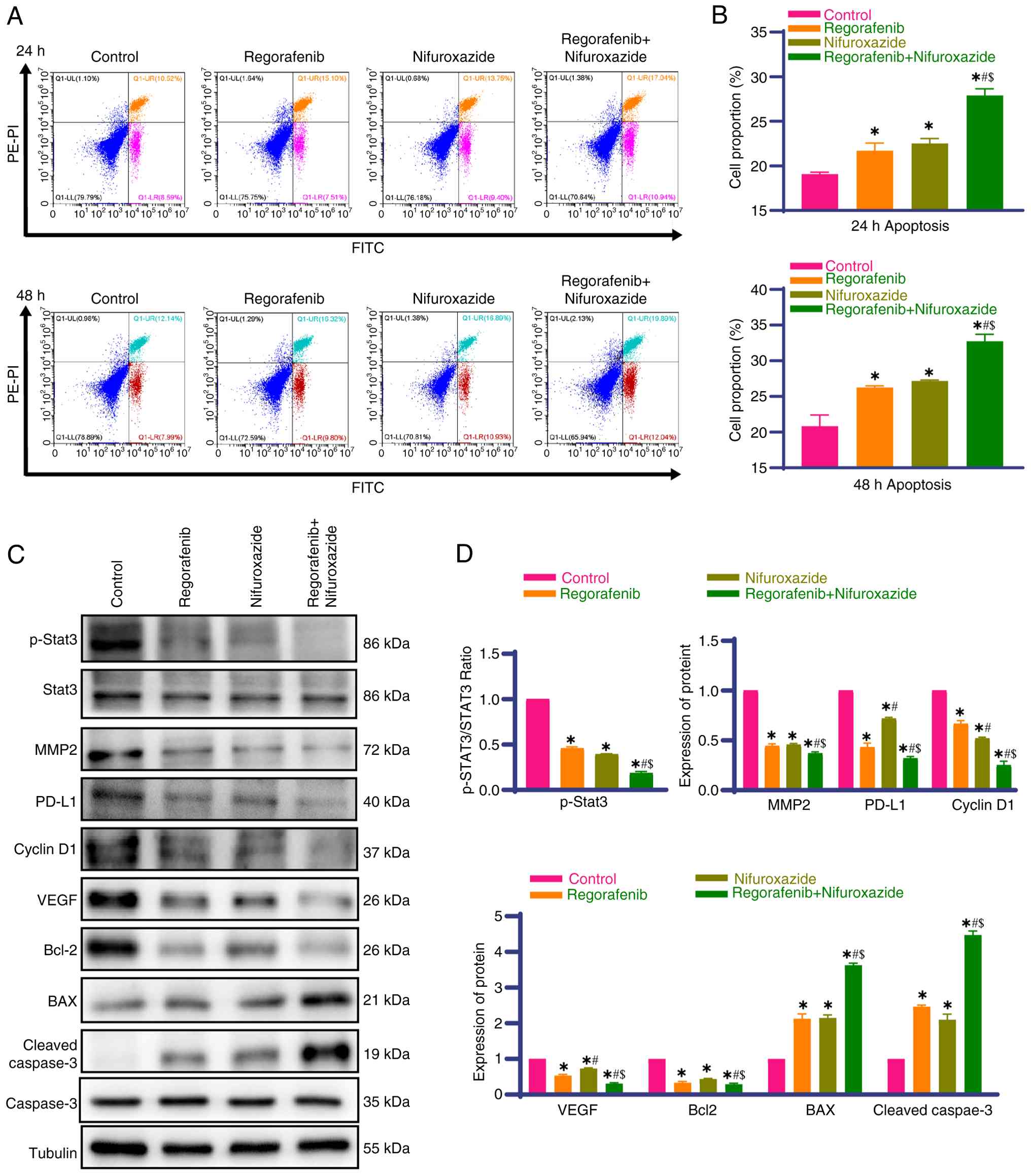
To clarify the pro-apoptotic effect and molecular
mechanism of combination therapy, flow cytometry showed that
monotherapy with Nifuroxazide and Regorafenib markedly induced
HepG2 apoptosis and the apoptotic rate was further increased with
combination treatment (Fig. 2A and
B). Western blot analysis confirmed that both drugs reduced
p-STAT3 expression, consistent with the IL6-JAK-STAT3 pathway
inhibition based on GSEA and MMP2 expression and this reduction was
more prominent in the combination treatment group (Fig. 2C and D; the original uncropped
western blot images for the detections in this section are in
Fig. S2). The present study
further detected key molecules in the JAK2/SHP-1/STAT3 pathway:
Compared with the control group, monotherapy with either drug
markedly downregulated the expression of p-JAK2 and upregulated
SHP-1 expression, while total JAK2 expression remained unchanged
across all groups. The combination group exhibited a more
pronounced downregulation of p-JAK2 and upregulation of SHP-1,
which further supports the targeted inhibition of the STAT3 pathway
by the combination therapy (Fig.
S3A-D). Monotherapy with both drugs increased the expression of
pro-apoptotic proteins (BAX and cleaved caspase-3) and decreased
the expression of anti-apoptotic proteins (Bcl-2 and
proliferation-related Cyclin-D1) and this effect was more
pronounced in the combination group (Fig. 2C and D), indicative of increased
apoptosis via the mitochondrial pathway. Additionally, monotherapy
and combination therapy markedly reduced immune checkpoint PD-L1
and pro-angiogenic VEGF expression (both key in an
immunosuppressive microenvironment), with a greater degree of
downregulation in the combination group (Fig. 2C and D), a finding that was
consistent with the findings of the transcriptomic analysis and
GSEA results showing modulation of immune
activation/anti-angiogenic regulatory characteristics.
Effect of Nifuroxazide combined with
Regorafenib on tumor growth in a mouse model of HCC
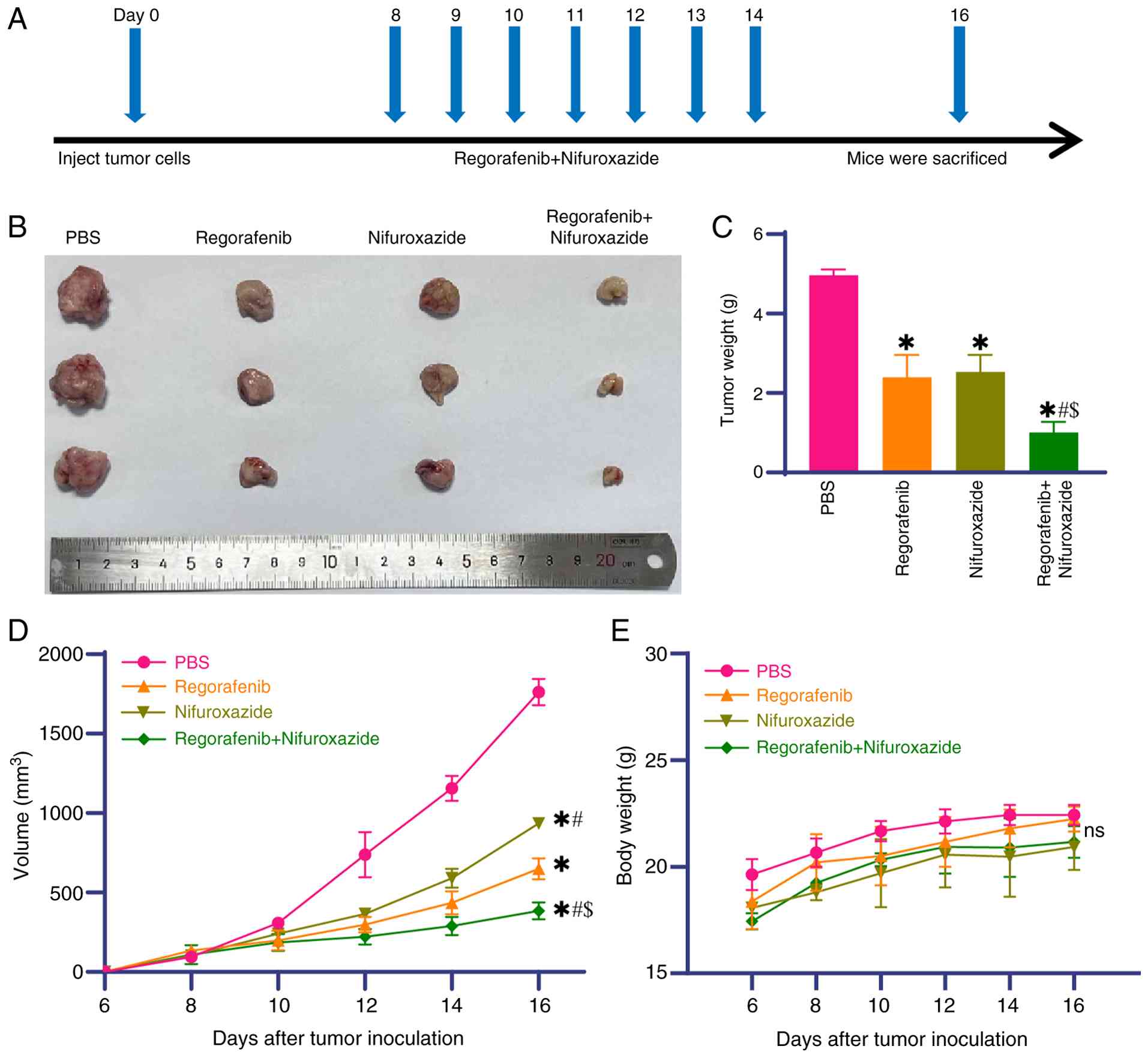
At seven days after model establishment, mice
underwent 7 consecutive days of treatment. One day post-treatment,
mice were sacrificed and tumor tissues were excised, weighed and
images captured (Fig. 3A). The
results showed that monotherapy with Nifuroxazide or Regorafenib
markedly inhibited tumor growth compared with the PBS group and the
combination group showed the greatest reduction in tumor growth
(Fig. 3B-D). Body weight monitoring
revealed no significant group differences despite variations in
tumor volumes (Fig. 3E), suggesting
no obvious systemic toxicity from the combination treatment.
Effects of Nifuroxazide and
Regorafenib on apoptosis, proliferation, migration and PDL1 and
VEGF expression in the mouse model of HCC
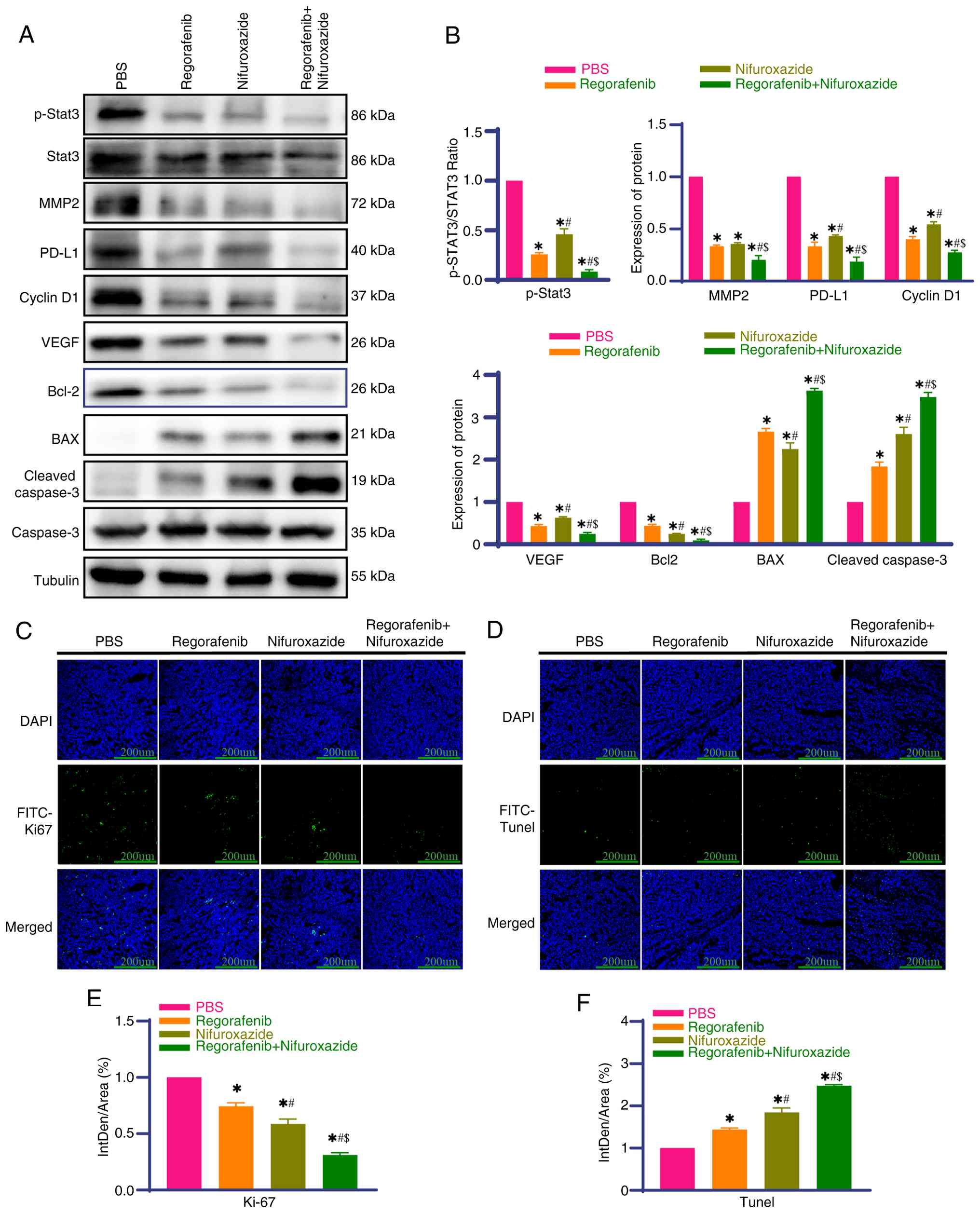
Tumor tissues were isolated for detection 1 day
after the final treatment. Western blotting showed that
Nifuroxazide and Regorafenib monotherapy reduced p-STAT3 and MMP2
expression compared with the PBS group and the reductions in both
were more pronounced in the combination group (Fig. 4A and B; the original uncropped
western blot gel images for the related protein detections are
Fig. S4). Consistent with the
in vitro results, similar changes in the JAK2/SHP-1/STAT3
pathway were detected in tumor tissues: Compared with the PBS
group, monotherapy groups showed markedly decreased p-JAK2
expression and increased SHP-1 expression, while total JAK2 levels
were unchanged. The combination group had the most marked p-JAK2
downregulation and SHP-1 upregulation, confirming the in
vivo targeting of the STAT3 pathway by the combination therapy
(Fig. S2E-H). Monotherapy with
each drug upregulated the expression of pro-apoptotic BAX and
cleaved-caspase-3, whilst downregulating the expression of
anti-apoptotic Bcl-2 and proliferation-related CyclinD1, with a
pronounced effect in the combination group (Fig. 4A and B). Similarly, monotherapy with
either drug reduced PD-L1 and VEGF expression and the effect was
more pronounced in the combination group (Fig. 4A and B), consistent with the in
vitro experiments and transcriptomic results.
Immunofluorescence analysis showed lower Ki-67 expression
(associated with proliferation) when treated with either drug alone
compared with the PBS group and the decrease in expression was
larger in the combination group (Fig.
4C and E). TUNEL assays showed fewer apoptotic cells in the PBS
group, increased apoptosis in the monotherapy groups (either drug)
and the highest number of apoptotic cells in the combination group
(Fig. 4D and F). These results
suggested that the combination treatment enhanced the anti-tumor
effects of each drug in vivo by enhancing apoptosis and
inhibiting proliferation/migration.
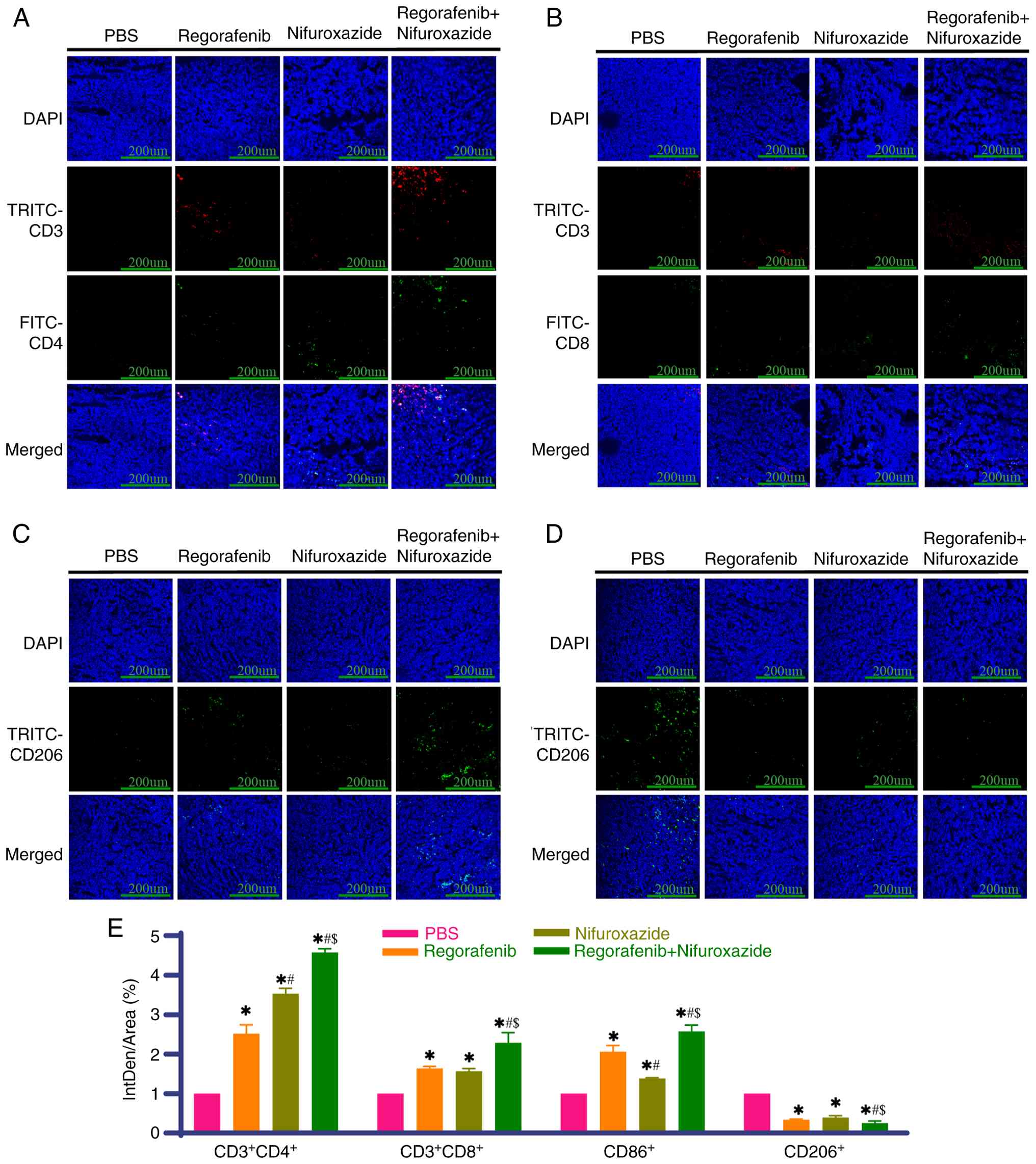
Nifuroxazide combined with regorafenib
promotes the activation of tumor-infiltrating lymphocytes
To investigate the effects of combination therapy on
the tumor immune microenvironment, immunofluorescence was used to
detect immune cell infiltration in tumor tissues. The results
showed that compared with the PBS group, the monotherapy groups
exhibited markedly increased proportions of
CD3+CD4+ and CD3+CD8+ T
cells in tumor tissues and the increase was even higher in the
combination group (Fig. 5A, B and
E). Monotherapy with either drug increased the proportion of M1
macrophages and decreased the proportion of M2 macrophages, with a
more pronounced effect in the combination group (Fig. 5C-E). Notably, activation of the
PD-1/PD-L1 pathway impairs anti-tumor immunity by inhibiting T
lymphocyte function and mediating tumor immune escape. Therefore,
the results of the present study suggested that the combination
treatment activated anti-tumor immunity and abrogated immune escape
by promoting effector T cell infiltration and regulating
macrophages toward a pro-inflammatory phenotype.
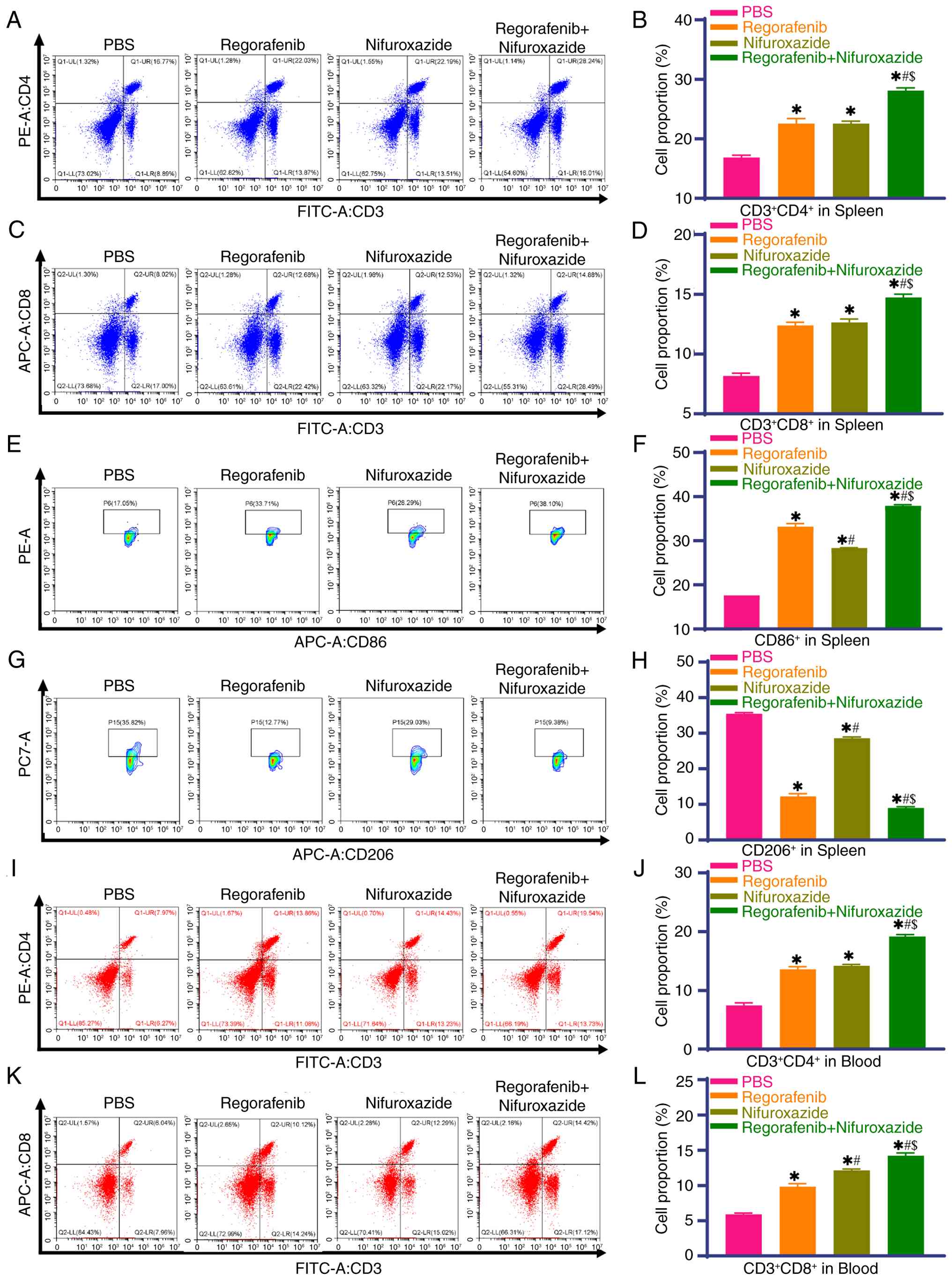
Nifuroxazide combined with regorafenib
regulates the proportion of immune cells in the spleen and
peripheral blood
The spleen, a core peripheral immune organ, is key
in systemic immune responses. Flow cytometry showed that compared
with the PBS group, Nifuroxazide and Regorafenib monotherapy groups
had markedly higher proportions of splenic
CD3+CD4+ T lymphocytes,
CD3+CD8+ T lymphocytes and CD86+
cells and lower CD206+ cells and this effect was more
prominent in the combination group (Fig. 6A-H). Further analysis of peripheral
blood circulating T lymphocytes revealed that the monotherapy and
combination groups had a markedly higher proportion of CD3+CD4+ and
CD3+CD8+ T lymphocytes than the PBS group, with a more pronounced
increase in the combination group (Fig.
6I-L). These results indicated that the combination treatment
altered the systemic immune status in tumor-bearing mice by
regulating splenic immune cell composition and the proportions of
peripheral blood T lymphocytes, thereby enhancing systemic
anti-tumor immunity, which is related to local tumor immune
microenvironment activation.
Discussion
As a typically highly vascular tumor, HCC exhibits
abnormally active angiogenesis, which not only provides nutritional
support for tumor proliferation but also promotes immune evasion by
constructing physical barriers and an immunosuppressive
microenvironment. This characteristic makes anti-angiogenic therapy
one of the core strategies for systemic treatment of HCC (24). Currently, first-line multi-kinase
inhibitors (MKIs) such as sorafenib and lenvatinib, as well as
second-line agents such as Regorafenib and cabozantinib, exert
anti-angiogenic effects by inhibiting the VEGF pathway. Moreover,
combination regimens of such MKIs with immune checkpoint inhibitors
(ICIs) have become a research hotspot in HCC treatment (25). However, such combination strategies
face significant challenges in clinical practice: On the one hand,
MKIs exhibit significant dose-dependent toxicity and dose
reductions or treatment interruptions due to adverse events are
extremely common, even halting the development of some
ICI-anti-angiogenesis combinations (26–28);
on the other hand, preclinical studies have confirmed that higher
doses of MKIs may induce a hypoxic tumor microenvironment and
recruit immunosuppressive cells such as TAMs, thereby exacerbating
immune evasion and weakening the synergistic effect of combination
therapy (11). Therefore, exploring
the optimal dose of MKIs to balance efficacy, safety and
immunomodulatory effects and identifying effective combination
therapies are crucial for optimizing treatment regimens. The
selected doses of Regorafenib (5 µg/ml in vitro, 5 mg/kg
in vivo) and Nifuroxazide (10 µg/ml in vitro, 10
mg/kg in vivo) were supported by literature evidence,
preliminary tests and experimental validation in the present study.
For Regorafenib, the clinical recommended dose (160 mg/day) is
limited by significant dose-dependent toxicity (such as hand-foot
skin reactions, hypertension) (8,9,29,30).
Preclinical studies consistently confirm that low doses of
Regorafenib (5–10 µg/ml in vitro, 5–10 mg/kg/day in
vivo) avoid systemic adverse effects while retaining key
biological activities, including inhibiting VEGF-mediated
angiogenesis, promoting M1 macrophage polarization and enhancing T
cell infiltration, without inducing hypoxic immunosuppression
(11,12). For Nifuroxazide, 10 µg/ml (in
vitro) and 10 mg/kg/day (in vivo) are well-validated
doses in HCC research, shown to effectively inhibit STAT3
phosphorylation, downregulate PD-L1 expression and reduce M2
macrophage infiltration in tumors (16,18).
As an oral agent with decades of clinical use for gastrointestinal
infections, Nifuroxazide exhibits favorable liver tissue
penetration (31) and a
well-documented safety profile (14). Although dedicated dose optimization
procedures or full pharmacokinetic (PK) data were not collected in
the present study, the selected doses are consistent with
established pharmacokinetic-pharmacodynamic (PK-PD) relationships
in preclinical HCC models and their rationality is directly
verified by the significant anti-tumor efficacy, target inhibition
and absence of toxicity in the present study. In vivo, this
combination maintained mouse body weight and was safe. Although
pharmacokinetic data were not fully collected, the doses used
balanced efficacy and safety and were supported by the preclinical
evidence. In addition, all drugs in the present study were
administered via oral gavage, a route that closely mimics clinical
oral medication. As an orally available agent with decades of
clinical use, Nifuroxazide is absorbed and metabolized in the
liver, with well-documented liver tissue penetration. Importantly,
it has been validated as a promising anticancer candidate by
inhibiting STAT3 signaling and selectively eradicating ALDH1 high
cancer-initiating cells (31).
Regorafenib is also clinically administered orally for advanced
liver cancer treatment (7). Thus,
the optimized doses and clinically relevant administration route
used in the present study are directly translatable to clinical
practice, jointly supporting the potential clinical application of
this combination therapy. The present study is the first, to the
best of the authors' knowledge, to confirm that a combination of
low-dose Regorafenib and Nifuroxazide exerts a markedly enhanced
anti-tumor effect in liver cancer models, with its core mechanism
lying in the dual actions of targeted inhibition of the STAT3
signaling pathway and remodeling of the tumor immune
microenvironment.
Enhanced inhibition of the STAT3 pathway is the core
molecular basis (32). As a key
driver of tumor progression, sustained STAT3 activation can
exacerbate tumor angiogenesis and immune evasion by promoting VEGF
expression, upregulating PD-L1 and recruiting immunosuppressive
cells (33). The results of the
present study showed that combination therapy markedly reduced
p-STAT3 expression in HepG2 cells and in tumor tissues from H22
liver cancer-bearing mice. Regorafenib inhibits p-STAT3-mediated
signaling by directly targeting the self-inhibited SHP-1 (34), while Nifuroxazide directly targets
STAT3 phosphorylation. The two block the STAT3 pathway at different
levels, jointly downregulating the expression of its downstream
target genes, thereby exerting enhanced inhibition of tumor cell
proliferation and migration and promoting apoptosis.
PD-L1, as a key immune checkpoint molecule, is
widely expressed on the surface of tumor cells and various immune
cells (35,36). Upon binding to the PD-1 receptor on
T cells, it directly inhibits the activation and proliferation of
effector T cells and also impairs the antigen-presenting function
of macrophages in the tumor microenvironment through paracrine
mechanisms (37), forming a core
regulatory axis of tumor immune evasion. In HCC, abnormal
activation of the PD-1/PD-L1 signaling pathway is an important
pathological basis for the formation of an immunosuppressive
microenvironment; blocking this pathway can relieve the functional
inhibition of effector T cells and markedly enhance their specific
killing of tumor cells (38).
Previous studies have shown that increased T cell infiltration in
tumor tissues can improve DFS (39–41).
According to the results of the present study, combination therapy
with low-dose Regorafenib and Nifuroxazide markedly enhanced T cell
infiltration in tumor tissues and inhibited STAT3 activation,
consistent with transcriptomic findings of immune pathway
enrichment and further supporting its role in remodeling anti-tumor
immunity. Furthermore, it was demonstrated that the combination of
Regorafenib and Nifuroxazide not only markedly enhanced T cell
infiltration in tumor tissues but also increased the proportion of
T cells in the spleen of tumor-bearing mice.
Macrophages are closely associated with the
progression of various diseases (42,43).
They have become key therapeutic targets for a range of diseases,
including cancer. As core effector cells of the innate immune
system, macrophages exhibit high plasticity, enabling them to
dynamically polarize in the TME and form a complex regulatory
network with PD-L1 and VEGF (44,45).
Given their pro-inflammatory phenotype, M1 macrophages can activate
anti-tumor immune responses by secreting cytokines such as IFN-γ
and TNF-α. M1 macrophages exhibit low surface PD-L1 expression but
high levels of MHC class II molecules and co-stimulatory molecules,
thereby enhancing antigen-presenting function (44,46).
By contrast, M2 macrophages not only secrete large quantities of
immunosuppressive cytokines, such as IL-6 and IL-10, but also bind
to PD-1 on the surface of T cells through high PD-L1 expression,
thereby inhibiting the activation of effector T cells and inducing
the recruitment of regulatory T cells (47). Additionally, M2 macrophages release
pro-angiogenic factors such as VEGF-A and MMPs and collaborate with
tumor cells to form abnormal neovascular networks (48,49).
This imbalance in macrophage phenotypes is particularly prominent
in HCC: VEGF can hinder immune cell infiltration by promoting
peritumoral collagen production and tumor angiogenesis, leading to
a ‘cold immune phenotype’ in tumors (50,51);
meanwhile, sustained activation of PD-L1 further promotes the
polarization of TAMs toward the M2 phenotype (52), forming a vicious cycle of
synergistic progression between immune evasion and angiogenesis. In
the present study, Regorafenib and Nifuroxazide induced the
polarization and recruitment of M1 macrophages in tumor tissues,
while promoting the infiltration of CD8+ T cells. By
downregulating PD-L1 expression, the drugs relieve the inhibitory
effect of TAMs on T cells, thereby establishing a positive immune
cycle by promoting M1 macrophage infiltration and T cell
activation.
The present study had certain limitations. First, it
was based on previously used low concentrations of Regorafenib and
Nifuroxazide. This concentration range may not fully cover the
dose-effect relationship of the drugs under specific in
vitro experimental conditions or in vivo models,
potentially failing to comprehensively reflect their mechanisms of
action and potential off-target effects in a broader biological
context. Second, the present study relies solely on the H22
homologous model, lacking validation in xenograft models derived
from human cell lines or patient-derived tissues. The H22 model was
selected primarily because it retains a fully functional immune
system, which is essential for investigating the immune remodeling
mechanism (the core focus of the present study), but this single
model may limit the generalizability and clinical translatability
of the results. To address this gap, future studies will supplement
two additional in vivo models: i) Human HCC cell
line-derived xenograft (CDX) models using well-characterized human
HCC cell lines (such as HepG2, Huh7) in NOD-SCID immunodeficient
mice to validate direct anti-tumor effects independent of the mouse
immune system; ii) Patient-derived xenograft models established
from freshly isolated human HCC tissues to confirm efficacy across
diverse clinical HCC subtypes. Third, the present study only used
female C57BL/6 mice and the efficacy of the combination therapy in
male mice remains unvalidated. The authors acknowledge restricting
to female mice as a limitation; future studies will include male
mice to validate efficacy across sexes. Finally, the effect of
combination therapy on other immune cells was not explored in depth
and the mechanism of immunomodulation remains incompletely
understood. Notably, the current results only demonstrated
phenotypic alterations of immune cells without functional
validation. Future studies will supplement detection of immune
activation markers (such as CD69, CD107a on T cells) and key
functional cytokines (such as IFN-γ, TNF-α, IL-10) in tumor tissues
and peripheral blood. This will further confirm whether the
observed phenotypic changes translate to enhanced anti-tumor immune
activity, thereby strengthening the evidence for immune remodeling
mediated by the combination therapy.
In conclusion, the present study is the first, to
the best of the authors' knowledge, to confirm that low-dose
Regorafenib and Nifuroxazide exert significant anti-tumor effects
in liver cancer models by enhancing STAT3 inhibition,
downregulating PD-L1 expression, promoting M1 macrophage
polarization and activating T cells. These results provided
preclinical experimental evidence for the development of combined
immunotherapeutic strategies for HCC.
Based on the findings of the present study, it was
hypothesized that the enhanced anti-tumor effects of this
combination originate from the two drugs' complementary mechanisms:
Nifuroxazide inhibits STAT3 phosphorylation, drives macrophage
polarization towards the pro-inflammatory M1 phenotype and promotes
CD8+ T cell activation; meanwhile, low-dose Regorafenib
reduces VEGF expression and downregulates PD-L1 expression in HCC
cells. A schematic diagram illustrating the hypothesized molecular
mechanisms of the combination treatment is shown in Fig. 7.
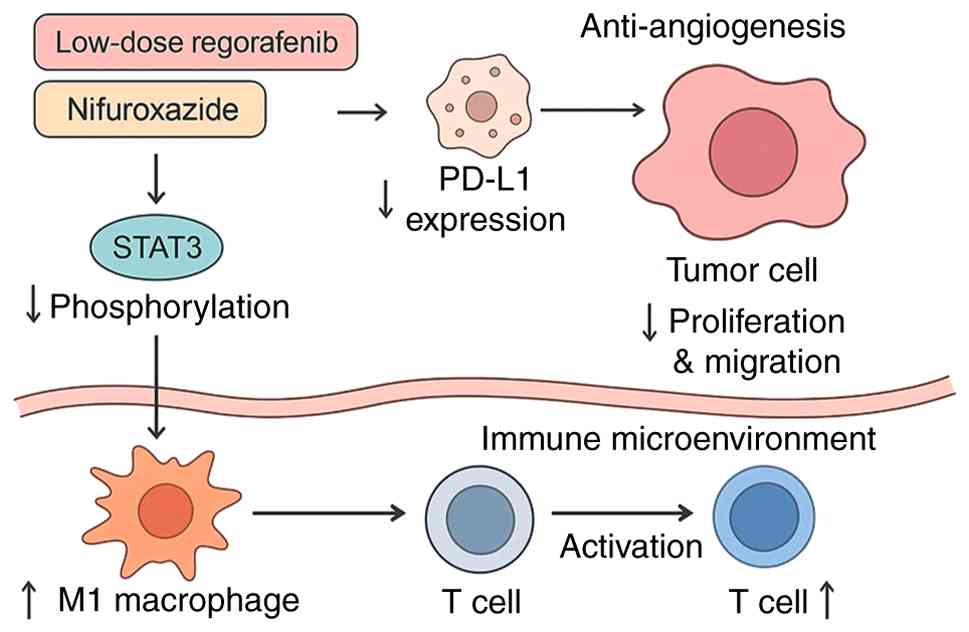 | Figure 7.Proposed mechanism by which
Regorafenib and Nifuroxazide exert enhanced suppression of
hepatocellular carcinoma by inhibiting STAT3 and immune remodeling.
Low-dose Regorafenib combined with Nifuroxazide exerts enhanced
anti-tumor effects through complementary mechanisms: Nifuroxazide
inhibits STAT3 phosphorylation, drives macrophage polarization
toward the pro-inflammatory M1 phenotype and promotes
CD8+ T cell activation, thereby reversing the
immunosuppressive tumor microenvironment. Concurrently, low-dose
Regorafenib exerts anti-angiogenic effects by suppressing VEGF
expression and downregulates PD-L1 expression on HCC cells,
directly impeding tumor cell proliferation and migration.
Collectively, this dual-action enhanced effect enhances immune
surveillance, disrupts tumor vascular supply and amplifies the
killing efficacy against HCC cells. STAT3, signal transducer and
activator of transcription 3; VEGF, vascular endothelial growth
factor; PD-L1, programmed death ligand 1; HCC, hepatocellular
carcinoma. |
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the Key Scientific Research
Projects in Higher Education of Henan Province (grant no.
25A320041), the Henan Provincial Science and Technology Tackle Plan
(grant no. 242102311143) and the Henan Provincial Medical Science
and Technology Tackle Plan (grant no. SBCJ202103064).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
KL and JC contributed to investigation and writing
the original draft. ZZ, YG, JZ, WD, TY, XD, TZ and HJ contributed
to the implementation of experiments. PC was responsible for
project administration, supervision and writing the original draft.
JR was responsible for conceptualization, funding acquisition,
supervision and writing, reviewing and editing. All authors have
read and approved the final manuscript and confirm the authenticity
of all the raw data.
Ethics approval and consent to
participate
All animal experiments were reviewed and approved by
the Animal Ethics Committee of the First Affiliated Hospital of
Zhengzhou University (approval no. 2023-KY-1333). This committee
operates independently, free from undue influence by researchers or
sponsors and is duly qualified to oversee animal research ethics.
All experiments were conducted in strict compliance with the
General Requirements for Biosafety in Animal Experiments of
Laboratory Animals in China (GB/T 43051-2023) (53), which is consistent with
internationally recognized animal welfare guidelines, including the
basic principles outlined in the National Institutes of Health
Guide for the Care and Use of Laboratory Animals. Every effort was
made to minimize animal suffering and ensure animal welfare.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
HCC
|
hepatocellular carcinoma
|
|
JAK2
|
Janus kinase 2
|
|
p-JAK2
|
phosphorylated Janus kinase 2
|
|
STAT3
|
signal transducer and activator of
transcription 3
|
|
PD-L1
|
programmed death ligand 1
|
|
SHP-1
|
Src homology region 2
domain-containing phosphatase 1
|
|
VEGF
|
vascular endothelial growth factor
|
|
CCK-8
|
Cell Counting Kit-8
|
|
TUNEL
|
terminal deoxynucleotidyl transferase
dUTP nick end labeling
|
|
GO
|
Gene Ontology
|
|
KEGG
|
Kyoto Encyclopedia of Genes and
Genomes
|
|
GSEA
|
Gene Set Enrichment Analysis
|
References
|
1
|
Anwanwan D, Singh SK, Singh S, Saikam V
and Singh R: Challenges in liver cancer and possible treatment
approaches. Biochim Biophys Acta Rev Cancer. 1873:1883142020.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rumgay H, Arnold M, Ferlay J, Lesi O,
Cabasag CJ, Vignat J, Laversanne M, McGlynn KA and Soerjomataram I:
Global burden of primary liver cancer in 2020 and predictions to
2040. J Hepatol. 77:1598–1606. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Marrero JA, Kulik LM, Sirlin CB, Zhu AX,
Finn RS, Abecassis MM, Roberts LR and Heimbach JK: Diagnosis,
staging and management of hepatocellular carcinoma: 2018 practice
guidance by the american association for the study of liver
diseases. Hepatology. 68:723–750. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Brown ZJ, Tsilimigras DI, Ruff SM, Mohseni
A, Kamel IR, Cloyd JM and Pawlik TM: Management of hepatocellular
carcinoma: A review. JAMA Surg. 158:410–420. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kudo M: Targeted and immune therapies for
hepatocellular carcinoma: Predictions for 2019 and beyond. World J
Gastroenterol. 25:789–807. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Guo X, Zhao Z, Zhu L, Liu S, Zhou L, Wu F,
Fang S, Chen M, Zheng L and Ji J: The evolving landscape of
biomarkers for systemic therapy in advanced hepatocellular
carcinoma. Biomark Res. 13:602025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Demols A, Borbath I, Van den Eynde M,
Houbiers G, Peeters M, Marechal R, Delaunoit T, Goemine JC, Laurent
S, Holbrechts S, et al: Regorafenib after failure of gemcitabine
and platinum-based chemotherapy for locally advanced/metastatic
biliary tumors: REACHIN, a randomized, double-blind, phase II
trial. Ann Oncol. 31:1169–1177. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bruix J, Qin S, Merle P, Granito A, Huang
YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, et al:
Regorafenib for patients with hepatocellular carcinoma who
progressed on sorafenib treatment (RESORCE): A randomised,
double-blind, placebo-controlled, phase 3 trial. Lancet. 389:56–66.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Grothey A, Van Cutsem E, Sobrero A, Siena
S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C, et
al: Regorafenib monotherapy for previously treated metastatic
colorectal cancer (CORRECT): An international, multicentre,
randomised, placebo-controlled, phase 3 trial. Lancet. 381:303–312.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ou DL, Chen CW, Hsu CL, Chung CH, Feng ZR,
Lee BS, Cheng AL, Yang MH and Hsu C: Regorafenib enhances antitumor
immunity via inhibition of p38 kinase/Creb1/Klf4 axis in
tumor-associated macrophages. J Immunother Cancer. 9:e0016572021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin YY, Tan CT, Chen CW, Ou DL, Cheng AL
and Hsu C: Immunomodulatory effects of current targeted therapies
on hepatocellular carcinoma: Implication for the future of
immunotherapy. Semin Liver Dis. 38:379–388. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bajpai P, Agarwal S, Afaq F, Al Diffalha
S, Chandrashekar DS, Kim HG, Shelton A, Miller CR, Singh SK, Singh
R, et al: Combination of dual JAK/HDAC inhibitor with regorafenib
synergistically reduces tumor growth, metastasis and
regorafenib-induced toxicity in colorectal cancer. J Exp Clin
Cancer Res. 43:1922024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Al Zaid Siddiquee K and Turkson J: STAT3
as a target for inducing apoptosis in solid and hematological
tumors. Cell Res. 18:254–267. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Begovic B, Ahmedtagic S, Calkic L,
Vehabović M, Kovacevic SB, Catic T and Mehic M: Open Clinical trial
on using nifuroxazide compared to probiotics in treating acute
diarrhoeas in adults. Mater Sociomed. 28:454–458. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Luo L, Xu F, Peng H, Luo Y, Tian X,
Battaglia G, Zhang H, Gong Q, Gu Z and Luo K: Stimuli-responsive
polymeric prodrug-based nanomedicine delivering nifuroxazide and
doxorubicin against primary breast cancer and pulmonary metastasis.
J Control Release. 318:124–135. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhao T, Wei P, Zhang C, Zhou S, Liang L,
Guo S, Yin Z, Cheng S, Gan Z, Xia Y, et al: Nifuroxazide suppresses
PD-L1 expression and enhances the efficacy of radiotherapy in
hepatocellular carcinoma. Elife. 12:RP909112024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen P, Li K, Chen J, Hei H, Geng J, Huang
N, Lei M, Jia H, Ren J and Jin C: Enhanced effect of radiofrequency
ablation on HCC by siRNA-PD-L1-endostatin Co-expression plasmid
delivered. Transl Oncol. 53:1023192025. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ye TH, Yang FF, Zhu YX, Li YL, Lei Q, Song
XJ, Xia Y, Xiong Y, Zhang LD, Wang NY, et al: Inhibition of STAT3
signaling pathway by nifuroxazide improves antitumor immunity and
impairs colorectal carcinoma metastasis. Cell Death Dis.
8:e25342017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Aden DP, Fogel A, Plotkin S, Damjanov I
and Knowles BB: Controlled synthesis of HBsAg in a differentiated
human liver carcinoma-derived cell line. Nature. 282:615–616. 1979.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Knowles BB, Howe CC and Aden DP: Human
hepatocellular carcinoma cell lines secrete the major plasma
proteins and hepatitis B surface antigen. Science. 209:497–499.
1980. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
López-Terrada D, Cheung SW, Finegold MJ
and Knowles BB: Hep G2 is a hepatoblastoma-derived cell line. Hum
Pathol. 40:1512–1515. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
de Souza GO, Wasinski F and Donato J:
Characterization of the metabolic differences between male and
female C57BL/6 mice. Life Sci. 301:1206362022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Subramanian A, Tamayo P, Mootha VK,
Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub
TR, Lander ES and Mesirov JP: Gene set enrichment analysis: A
knowledge-based approach for interpreting genome-wide expression
profiles. Proc Natl Acad Sci USA. 102:15545–15550. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Poon RTP, Fan ST and Wong J: Clinical
significance of angiogenesis in gastrointestinal cancers: A target
for novel prognostic and therapeutic approaches. Ann Surg.
238:9–28. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cheng AL, Hsu C, Chan SL, Choo SP and Kudo
M: Challenges of combination therapy with immune checkpoint
inhibitors for hepatocellular carcinoma. J Hepatol. 72:307–319.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Amin A, Plimack ER, Ernstoff MS, Lewis LD,
Bauer TM, McDermott DF, Carducci M, Kollmannsberger C, Rini BI,
Heng DYC, et al: Safety and efficacy of nivolumab in combination
with sunitinib or pazopanib in advanced or metastatic renal cell
carcinoma: The CheckMate 016 study. J Immunother Cancer. 6:1092018.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xu J, Zhang Y, Jia R, Yue C, Chang L, Liu
R, Zhang G, Zhao C, Zhang Y, Chen C, et al: Anti-PD-1 antibody
SHR-1210 combined with apatinib for advanced hepatocellular
carcinoma, gastric, or esophagogastric junction cancer: An
open-label, dose escalation and expansion study. Clin Cancer Res.
25:515–523. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Finn RS, Ikeda M, Zhu AX, Sung MW, Baron
AD, Kudo M, Okusaka T, Kobayashi M, Kumada H, Kaneko S, et al:
Phase Ib study of lenvatinib plus pembrolizumab in patients with
unresectable hepatocellular carcinoma. J Clin Oncol. 38:2960–2970.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Killock D: Liver cancer: Regorafenib-a new
RESORCE in HCC. Nat Rev Clin Oncol. 14:70–71. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hecht JR, Park YS, Tabernero J, Lee MA,
Lee S, Virgili AC, Van den Eynde M, Fontana E, Fakih M, Asghari G,
et al: Zanzalintinib plus atezolizumab versus regorafenib in
refractory colorectal cancer (STELLAR-303): A randomised,
open-label, phase 3 trial. Lancet. 406:2360–2370. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bailly C: Toward a repositioning of the
antibacterial drug nifuroxazide for cancer treatment. Drug Discov
Today. 24:1930–1936. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hu B, Zou T, Qin W, Shen X, Su Y, Li J,
Chen Y, Zhang Z, Sun H, Zheng Y, et al: Inhibition of EGFR
overcomes acquired lenvatinib resistance driven by STAT3-ABCB1
signaling in hepatocellular carcinoma. Cancer Res. 82:3845–3857.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zou S, Tong Q, Liu B, Huang W, Tian Y and
Fu X: Targeting STAT3 in cancer immunotherapy. Mol Cancer.
19:1452020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tai WT, Chu PY, Shiau CW, Chen YL, Li YS,
Hung MH, Chen LJ, Chen PL, Su JC, Lin PY, et al: STAT3 mediates
regorafenib-induced apoptosis in hepatocellular carcinoma. Clin
Cancer Res. 20:5768–5776. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Voli F, Valli E, Lerra L, Kimpton K,
Saletta F, Giorgi FM, Mercatelli D, Rouaen JRC, Shen S, Murray JE,
et al: Intratumoral copper modulates PD-L1 expression and
influences tumor immune evasion. Cancer Res. 80:4129–4144. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang X, Yang X, Zhang C, Wang Y, Cheng T,
Duan L, Tong Z, Tan S, Zhang H, Saw PE, et al: Tumor cell-intrinsic
PD-1 receptor is a tumor suppressor and mediates resistance to PD-1
blockade therapy. Proc Natl Acad Sci USA. 117:6640–6650. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xiong H, Mittman S, Rodriguez R,
Moskalenko M, Pacheco-Sanchez P, Yang Y, Nickles D and Cubas R:
Anti-PD-L1 treatment results in functional remodeling of the
macrophage compartment. Cancer Res. 79:1493–1506. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Tabrizian P, Abdelrahim M and Schwartz M:
Immunotherapy and transplantation for hepatocellular carcinoma. J
Hepatol. 80:822–825. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wu TD, Madireddi S, de Almeida PE,
Banchereau R, Chen YJ, Chitre AS, Chiang EY, Iftikhar H, O'Gorman
WE, Au-Yeung A, et al: Peripheral T cell expansion predicts tumour
infiltration and clinical response. Nature. 579:274–278. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Sun L, Su Y, Jiao A, Wang X and Zhang B: T
cells in health and disease. Signal Transduct Target Ther.
8:2352023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fridman WH, Pagès F, Sautès-Fridman C and
Galon J: The immune contexture in human tumours: Impact on clinical
outcome. Nat Rev Cancer. 12:298–306. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Varol C, Mildner A and Jung S:
Macrophages: Development and tissue specialization. Annu Rev
Immunol. 33:643–675. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Lavin Y, Mortha A, Rahman A and Merad M:
Regulation of macrophage development and function in peripheral
tissues. Nat Rev Immunol. 15:731–744. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sharma A, Seow JJW, Dutertre CA, Pai R,
Blériot C, Mishra A, Wong RMM, Singh GSN, Sudhagar S, Khalilnezhad
S, et al: Onco-fetal reprogramming of endothelial cells drives
immunosuppressive macrophages in hepatocellular carcinoma. Cell.
183:377–394.e21. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lu LG, Zhou ZL, Wang XY, Liu BY, Lu JY,
Liu S, Zhang GB, Zhan MX and Chen Y: PD-L1 blockade liberates
intrinsic antitumourigenic properties of glycolytic macrophages in
hepatocellular carcinoma. Gut. 71:2551–2560. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cheng K, Cai N, Zhu J, Yang X, Liang H and
Zhang W: Tumor-associated macrophages in liver cancer: From
mechanisms to therapy. Cancer Commun (Lond). 42:1112–1140. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Locati M, Curtale G and Mantovani A:
Diversity, mechanisms and significance of macrophage plasticity.
Annu Rev Pathol. 15:123–147. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yeung OW, Lo CM, Ling CC, Qi X, Geng W, Li
CX, Ng KT, Forbes SJ, Guan XY, Poon RT, et al: Alternatively
activated (M2) macrophages promote tumour growth and invasiveness
in hepatocellular carcinoma. J Hepatol. 62:607–616. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kadioglu E and De Palma M: Cancer
metastasis: Perivascular macrophages under watch. Cancer Discov.
5:906–908. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gabrilovich DI, Chen HL, Girgis KR,
Cunningham HT, Meny GM, Nadaf S, Kavanaugh D and Carbone DP:
Production of vascular endothelial growth factor by human tumors
inhibits the functional maturation of dendritic cells. Nat Med.
2:1096–1103. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Rahma OE and Hodi FS: The intersection
between tumor angiogenesis and immune suppression. Clin Cancer Res.
25:5449–5457. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhang H, Liu L, Liu J, Dang P, Hu S, Yuan
W, Sun Z, Liu Y and Wang C: Roles of tumor-associated macrophages
in anti-PD-1/PD-L1 immunotherapy for solid cancers. Mol Cancer.
22:582023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
National Standard of the People's Republic
of China, . Laboratory animal - General requirements for biosafety
in animal experiment (GB/T 43051-2023) [S]. National Standard of
the People's Republic of China; Beijing: 2023
|