Introduction
Glioblastomas are among the most aggressive
malignant brain tumors in adults, characterized by rapid and
invasive growth and a high recurrence rate, making effective
treatment a major challenge and contributing to their poor
prognosis (1). The current standard
therapy combines surgical resection and adjuvant radio-chemotherapy
(2). However, median survival
remains low at 14.6 months (3).
Despite numerous clinical trials since the introduction of
temozolomide, no new effective drugs have been developed,
highlighting the urgent need for further research (2).
One of the main challenges in targeting
glioblastomas is tumor heterogeneity (4). Based on transcriptional profiles, as
well as genetic and epigenetic alterations, glioblastomas are
highly diverse tumors that can be classified into distinct
subtypes, even among patients with the same tumor grade (5). Differences in tumor composition
between patients define intertumoral heterogeneity, whereas the
presence of multiple subtypes within a single tumor in the same
patient is referred to as intratumoral heterogeneity (6). A key contributor to intratumoral
heterogeneity is the presence of glioma stem-like cells (GSCs), a
subpopulation of cells with self-renewal capacity and
tumor-initiating potential (4,7). GSCs
not only drive tumor progression and recurrence but also interact
dynamically with the tumor microenvironment (TME), which further
supports their maintenance and resistance to therapy (4,7). This
dynamic interaction between GSCs and the TME underscores the
critical role of the TME, which acts as a protective niche for
tumor cells (8). The TME comprises,
for example, immune cells (microglia, T cells and macrophages),
modulatory factors such as cytokines and chemokines, as well as
structural elements including fibroblasts, pericytes, endothelial
cells, and the extracellular matrix (8–10).
Microglia and peripheral macrophages, collectively referred to as
glioma-associated microglia/macrophages, infiltrate tumor regions
and secrete inflammatory mediators, including cytokines and
chemokines (9). These mediators
bind to specific receptors, activating intracellular signaling
pathways through second messengers and triggering diverse cellular
responses that promote tumor growth and invasiveness (11–14).
Chemokines further induce chemotaxis within the TME and play a key
role in tumor metabolism, including cell proliferation, resistance
to apoptosis, and the regulation of genes involved in these
processes (10,11,15).
Given that glioblastoma is a highly heterogeneous
tumor, developing research models that accurately preserve its
complex TME remains a major challenge. Traditional models,
including cell lines and animal models, have significant
limitations (16,17). Cell lines often undergo spontaneous
mutations and, in long-term cultures, also lack oxygen, nutrients,
pH gradients, physiological inputs, and interactions between tumor
cells and TME (18,19). Animal models are more time-consuming
and high-cost models with ethical limitations (17,20).
Furthermore, interspecies genetic differences complicate the
representation of human pathophysiology and make the translation of
data to human outcomes complex (16). Considering the significant
limitations of traditional cell lines and animal models in
accurately representing the complexity of glioblastoma, novel
models are required. In this context, glioblastoma organoids (GBOs)
have recently gained attention, as they preserve tumor
heterogeneity by exhibiting structural and functional
characteristics similar to those of human organs and tissues
(17,21). Organoids can be derived either from
stem cells or directly from tumor tissue (19,21).
These models offer a high success rate in culture, short formation
times, relatively low cost, and reproducible tumor traits (22). Moreover, organoids serve as valuable
tools to study tumor biology, including the TME, and test
radiotherapy, chemotherapy and immunotherapy (18,20–22).
Human brain organoid technology was first introduced by Lancaster
et al (2013) (23), marking
the beginning of rapid advancements in this field. Since then,
numerous brain tumor organoids have been developed and optimized
(24). For example, Hubert et
al (2016) (25) cultured
patient-derived GBOs within two months using Matrigel and EGF/bFGF.
Jakob et al (2020) (26)
further optimized this method by eliminating Matrigel and growth
factors, reducing the formation time of 70 organoid strains from 53
patients to just 1–2 weeks after tumor resection.
However, despite these advancements in cultivation
speed and methodology, it remains unclear whether GBOs maintain the
characteristics of the original tumor over a long-term cultivation
period. This gap raises questions about the suitability of GBOs as
reliable models for studying glioblastoma. The present study aimed
to investigate the long-term preservation of key tumor properties,
including cellular components such as GSCs, immune cells,
endothelial cells, proliferation marker, and inflammatory mediators
such as cytokines and chemokines in long-term cultivated GBOs.
Materials and methods
Cultivation of patient-derived
GBOs
Tumor tissues were obtained directly via surgical
resection (from a total of 10 patients: five women and five men
aged between 53 and 75 years and with a median age of 61 years) by
the Department of Neurosurgery (University Medical Center
Schleswig-Holstein, UKSH Kiel, Germany) after written informed
patient consent in accordance with the Helsinki Declaration of
1975, revised in 2013. Approval was granted by the Ethics Committee
of the University of Kiel (approval no. D524/17; Kiel, Germany).
Histological analysis was performed by the Department of Pathology
at the University Medical Center Hamburg-Eppendorf (UKE, Hamburg,
Germany).
The obtained fresh tumor samples were prepared as
previously described by Jacob et al (2020) (26). The preparation did not involve
classical enzymatic or mechanical tissue dissociation. Instead, the
tumor samples were only roughly cut and placed directly into
culture. In detail, tumor samples were transferred to a sterile
dish within H+GPSA wash medium containing Hibernate A, 1X GlutaMax,
1X PenStrep and 1X Amphotericin B (all from Thermo Fisher
Scientific, Inc.), and then cut into smaller pieces excluding areas
of cell death and blood vessels. Several wash steps were performed
to remove tissue debris. Cutting was continued under a
stereomicroscope Stemi 305 KMA (Carl Zeiss AG) to identify vessels
and areas of cell death in detail, and to finally get pieces of 1–3
mm size. The medium was then removed, and RBC-lysis buffer (Thermo
Fisher Scientific, Inc.) was added to selectively destroy red blood
cells. After shaking at 2 × g at room temperature for 10 min, the
buffer was removed, and another wash step was performed. GBO medium
was prepared as previously described by Hellmold et al
(2025) (27) and added to tissue
pieces. A total of 2 ml of the medium was added to each well of an
ultra-low attachment 6-well plate (Corning, Inc.). A total of ~10
tissue pieces in GBO medium were transferred into each well,
resulting in a total volume of 4 ml. The plates were incubated on
an orbital shaker (cat. no. 8012-1771; Binder GmbH) at 120 U/min at
37°C and 5% CO2. The GBOs were observed regularly, and
medium exchange was performed every second day (Fig. 1).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
To analyze gene expression levels of glial
structural proteins, proliferation marker, specific markers for
GSCs, immune and endothelial cells, as well as cytokines and
chemokines during the in vitro cultivation of 10 different
GBO preparations over time, RT-qPCR was employed. For each GBO
preparation, RNA isolation was performed at five different time
points using TRIzol (Invitrogen; Thermo Fisher Scientific, Inc.),
and cDNA was then synthesized as previously described by Hellmold
et al (2025) (27). Each
cDNA sample was also analyzed for glyceraldehyde-3-phosphate
dehydrogenase (GAPDH), serving as an internal standard. qPCR was
performed using TaqMan primer probes (Applied Biosystems; Thermo
Fisher Scientific, Inc.). Double determination, including positive
and negative controls, was applied. The plate was run in a
real-time PCR cycler (QuantStudio5; Applied Biosystems; Thermo
Fisher Scientific, Inc.) using the following conditions: 50 cycles,
volume of 20 µl, enzyme activation at 95°C for 10 min, denaturation
at 95°C for 15 sec, and annealing at 60°C for 1 min. Cycles of
threshold (CT) were detected, and the ∆CT
values were calculated as CT analyzed gene-CT
GAPDH. The applied gene-specific primers and probes are
listed in Table SI.
Hematoxylin-eosin (H&E) staining
and immunofluorescence double-staining
H&E staining was employed to assess the
long-term preservation of the cellular structure of GBOs over
cultivation time using the H&E Staining Kit (cat. no. 245880;
Abcam).
Due to the limited material, a simple qualitative
visualization of cellular markers was performed over time in
different GBO preparations (n=4) on the protein level, using
immunofluorescence double-staining with cryosections. The first
primary antibody was added and incubated overnight at 4°C. The
first secondary antibody, labeled with Alexa Fluor 488 or Alexa
Fluor 555 (1:1,000; Thermo Fisher Scientific, Inc.), was incubated
for 1 h at 37°C the next day. The second primary antibody was then
incubated overnight at 4°C, followed by the second secondary
antibody labeled with Alexa Fluor 488 or Alexa Fluor 555 (1:1,000;
Thermo Fisher Scientific, Inc.) the next day. The applied primary
antibodies are listed in Table
SII. For negative controls, primary antibodies were omitted.
Nuclei staining was subsequently accomplished with
4′,6-diamidino-2-phenylindole (DAPI) for 30 min at room
temperature. The embedded slides were analyzed using the
fluorescence microscope (AxioObserver.Z1; Carl Zeiss AG) with the
ZEN 3.5 (Blue Edition) software (Carl Zeiss AG).
Statistical analysis
Statistical analyses were accomplished using
GraphPad Prism 8.4® software (GraphPad Software, Inc.
Dotmatics) with a one-way ANOVA with Dunnett's multiple comparisons
post hoc test as indicated for each experiment in the figure
captions. Microsoft Excel was used to perform correlation analyses
by calculating the Pearson correlation index. Statistical
significance in both is outlined according to the P-value:
P<0.05 was considered to indicate a statistically significant
difference. The sample size is stated in the figure captions.
Results
GBO cellular characteristics generally
persist over the course of one month
After obtaining tissue pieces of 1–3 mm in size
(Fig. 1), the cellular GBO
characteristics were addressed in a total of 10 different GBO
preparations during long-term cultivation. For this purpose, the
quantitative gene expression levels of structural proteins,
proliferation, stemness, immune cells, and vessel markers were
analyzed using RT-qPCR, as shown in Figs. 2 and S1, with the respective findings for each
individual GBO preparation traceable. In addition, all RT-qPCR
results were presented as box plots as an alternative
representation to provide an improved overview (Fig. S2).
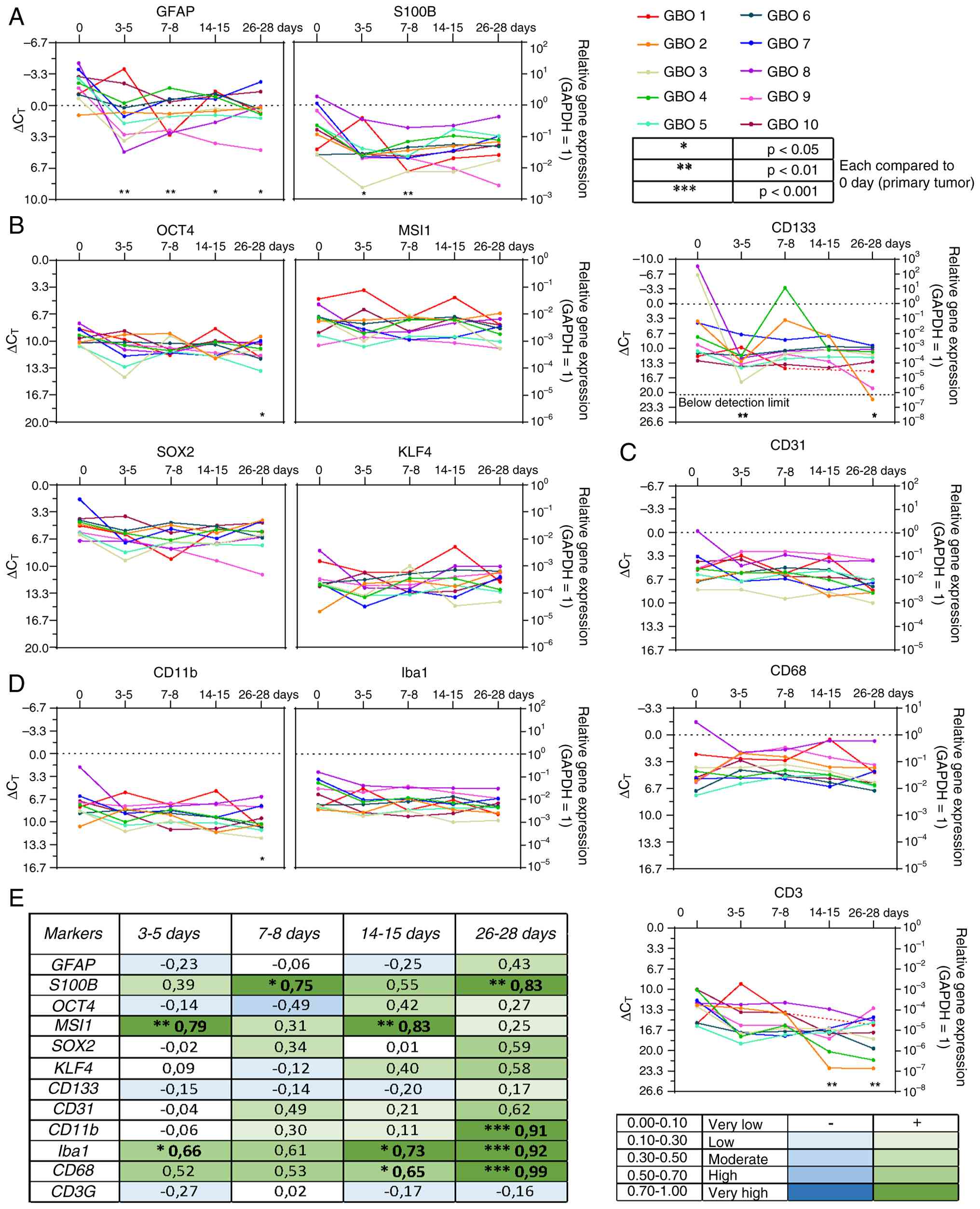 | Figure 2.Reverse transcription-quantitative
PCR and correlation analysis of glial structural proteins,
stemness, vessel, and immune cell markers. Gene expression levels
are shown across the entire cultivation period for 10 independent
GBO preparations (n=10) at five time points: days 0, 3–5, 7–8,
14–15, and 26–28. The x-axis represents days, the left y-axis shows
∆CT values, and the right y-axis displays the
logarithmic scale of expression relative to GAPDH as the
housekeeping gene (GAPDH expression=1). (A) Glial structural
proteins. (B) Stemness markers. (C) Vessel marker. (D) Immune cell
markers. (E) Correlation analysis. Correlations are indicated by a
color gradient: Dark green represents a high positive correlation,
light green a low positive correlation, dark blue a high negative
correlation, and light blue a low negative correlation. Statistical
analysis was performed using one-way ANOVA with Dunnett's multiple
comparisons post hoc test. *P<0.05, **P<0.01 and
***P<0.001. GBO, glioblastoma organoid; GAPDH,
glyceraldehyde-3-phosphate dehydrogenase; GFAP, glial fibrillary
acidic protein; S100B, S100 calcium-binding protein B; OCT4,
octamer-binding transcription factor 4; MSI1, Musashi RNA-binding
protein 1; SOX2, SRY-box transcription factor 2; KLF4, Krüppel-like
factor 4; CD133, cluster of differentiation 133; CD31, cluster of
differentiation 31; CD11b, cluster of differentiation 11b; Iba1,
ionized calcium-binding adapter molecule 1; CD68, cluster of
differentiation 68; CD3, cluster of differentiation 3. |
First, the Ki67 mRNA expression, a well-established
proliferation marker in glioblastoma (28), was analyzed. Although the expression
level decreased at 3–5 days in most GBO preparations, a recovery
was observed (Figs. S1 and
S2B), and expression remained
detectable for up to 28 days, confirming the viability of the
GBOs.
Glial fibrillary acidic protein (GFAP) and S100
calcium-binding protein B (S100B) were used as markers for
structural proteins. GFAP is involved in glial differentiation and
serves as a classical marker for glial tumor cells (29), whereas S100B is overexpressed in
malignant gliomas and modulates microglia/macrophage activation
(30). In the data of the present
study, GFAP mRNA expression also exhibited a drop at 3–5 days in
most GBO preparations, reaching its lowest point, as reflected by a
negative correlation when comparing the GFAP expression data
between the primary tumor tissue and GBOs cultured for 3–5 days.
This drop was followed by a gradual recovery with a positive
correlation between the primary tumor tissue and GBOs cultured for
26–28 days (Figs. 2A and E, and
S2A). However, the overall GFAP
expression level remained lower at the end of the observation
period compared with the beginning. S100B showed a similar drop at
3–5 days, followed by a slight recovery. However, in the case of
S100B, no negative correlation was observed between the primary
tumor tissue and GBOs cultured for 3–5 days. In general, the
baseline expression of S100B was lower than that of GFAP.
Stemness markers such as octamer-binding
transcription factor 4 (OCT4), Musashi RNA-binding protein 1
(MSI1), SRY-box transcription factor 2 (SOX2), Krüppel-like factor
4 (KLF4) and cluster of differentiation 133 (CD133) are associated
with tumor growth, invasiveness and poor prognosis (31–33).
Therefore, these markers were selected to investigate their
presence in the different GBO preparations over cultivation time.
The expression of OCT4, SOX2 and KLF4 was relatively homogeneous in
the analyzed GBO preparations, resulting in no significant
correlation between the primary tumor tissues and GBOs cultured for
all investigated time points (Figs. 2B
and E, and S2C). MSI1 showed a
similar pattern but tended to display a mild positive correlation
around GBO cultivation days 3–5 and 14–15, forming a subtle
V-shaped trend. However, when considering the overall MSI1-positive
population, which was more heterogeneous than OCT4, SOX2 and KLF4,
no significant differences were detected. CD133 expression was
highly heterogeneous, showing initially high values that
subsequently declined to lower levels, becoming undetectable in one
GBO preparation. This expression pattern led to a slight,
insignificant negative correlation between the primary tumor
tissues and GBOs cultured for most investigated time points.
However, toward the end, a slight positive correlation emerged,
with more homogeneous expression patterns approaching a plateau.
Regarding baseline expression of stemness markers, SOX2 showed the
highest level. OCT4 and MSI1 exhibited similar profiles, while KLF4
showed lower expression, and CD133 displayed the lowest baseline
expression.
Immune cells such as microglia, macrophages and
microglia-associated cells represent a key component of the TME and
play a critical role in tumor infiltration (9). Ionized calcium-binding adapter
molecule 1 (Iba1) was analyzed as a microglia marker, which is
upregulated in glioblastomas (33),
cluster of differentiation 3 (CD3) as a marker for T lymphocytes
and NK cells (34), as well as
cluster of differentiation 68 (CD68) and cluster of differentiation
11b (CD11b), which are expressed in macrophages and microglia
(35). In the current analysis,
Iba1, CD68 and CD11b exhibited relatively homogeneous mRNA
expression patterns with significant positive correlations between
the primary tumor tissues and GBOs cultured for up to 28 days
(Figs. 2D and E, S2E). By contrast, CD3 expression
displayed greater heterogeneity, with some GBO preparations showing
lower expression levels. Overall, CD3 predominantly revealed a
negative correlation between the primary tumor tissues and GBOs,
characterized by a downward trend. Iba1 and CD68 exhibited the
highest baseline expression, followed by CD11b, whereas CD3 showed
the lowest.
To address the presence of tumor vessels in GBOs,
cluster of differentiation 31 (CD31) was used (36). Depending on the analyzed GBO
preparations, CD31 expression was generally similar in magnitude
with slight variations (Figs. 2C and
E, and S2D). With some
exceptions, the baseline expression level was comparable to that of
the immune cell marker CD68, with no significant correlations when
comparing the expression between primary tumor tissues and GBOs
cultured for all investigated time points.
To illustrate the persistence of the cellular
structure of the investigated GBO preparations over cultivation
time, exemplary H&E staining was performed as shown in Fig. 3A with whole-organism and
corresponding high-magnification inset images. The GBOs presented
no significant visual differences, indicating that cellular
structures remained unchanged once a GBO had been formed.
Additionally, blood vessels were observed up to 28 days, indicating
that the vascular structure of organoids was preserved during a
one-month cultivation period.
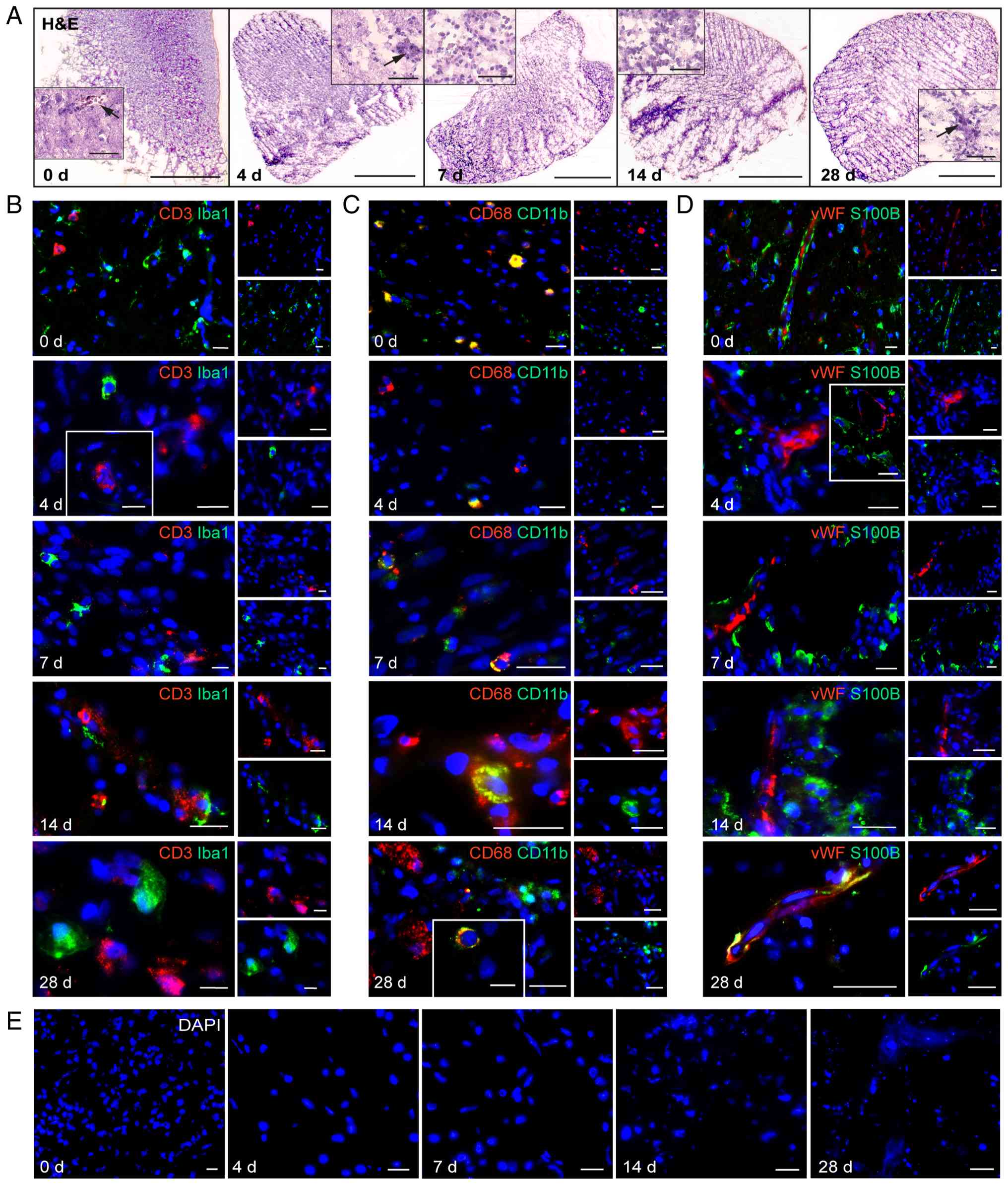 | Figure 3.H&E staining and
immunofluorescence double-staining images of an exemplary GBO
preparation at five time points: days 0, 4, 7, 14, and 28. Selected
insets were taken from two additional GBO preparations (total n=3).
(A) H&E staining images with low (×2,5)- and high
(×40)-magnification; black arrows indicate blood vessels. (B and C)
Immune cell markers. (D) Vessel marker. (E) Areas with cell death
in one GBO preparation during the cultivation period. Scale bars: 1
mm for whole-organism H&E images, 50 µm for the corresponding
H&E-insets, 20 µm for immunofluorescence images. GBO,
glioblastoma organoid; DAPI, 4′,6-diamidino-2-phenylindole; vWF,
von Willebrand factor; CD11b, cluster of differentiation 11b; Iba1,
ionized calcium-binding adapter molecule 1; CD68, cluster of
differentiation 68; CD3, cluster of differentiation 3; S100B, S100
calcium-binding protein B. |
After the evaluation of the markers at the RNA level
to ensure precise scientific quantification, immunofluorescence
double-staining was performed to visualize stable marker expression
and typical co-staining of cell-type-specific markers at the
protein level throughout the culture period. Exemplary images are
demonstrated in Figs. 3 and
4, highlighting typical
constellations. Due to the limited materials for valid quantitative
analyses, these images were included to support the validity of the
RNA-based findings using a total of three GBO preparations without
suggesting any quantitative assessment.
 | Figure 4.Immunofluorescence double-staining
images of an exemplary GBO preparation at five time points: Days 0,
4, 7, 14, and 28. Selected insets were taken from two additional
GBO preparations (total n=3). (A-D) Stemness markers. (E) Exemplary
Apotome image of KLF4 at day 28. GBO, glioblastoma organoid; GFAP,
glial fibrillary acidic protein; S100B, S100 calcium-binding
protein B; OCT4, octamer-binding transcription factor 4; MSI1,
Musashi RNA-binding protein 1; SOX2, SRY-box transcription factor
2; CD133, cluster of differentiation 133. |
CD3/Iba1 were stained to label T cells and
microglia, CD11b/CD68 to assess macrophages, and vWF/S100B to
visualize vasculature and glial tissue (Fig. 3B-D). The images revealed that all
markers remained detectable at the protein level over a cultivation
period of one month. In some cases, co-staining was observed as
expected, for example, with CD11b and CD68 (Fig. 3C).
To identify the presence of GSCs during the GBO
cultivation period, typical stemness markers were combined,
including SOX2, OCT4, MSI1, CD133 and KLF4, with typical glial
structural markers such as GFAP and S100B (Fig. 4A-E). The staining results revealed
that stem-like cells persist over a cultivation period of one month
alongside glial cells. Co-staining of stemness and glial structural
markers was rarely observed; in most cases, cells positive for
stemness markers were found as single cells or small groups near
cells positive for GFAP or S100B during the whole GBO cultivation
period. Especially, SOX2 and KLF4 (Fig.
4C, day 14 and E) were also observed in the nucleus, in
addition to their cytoplasmic localization, illustrating their role
as transcription factors. This is also highlighted by the apotome
images of KLF4 (Fig. 4E, whole
visualization of the marker is provided in Fig. S4).
Notably, some GBO preparations exhibited a
progressive cell death over time (exemplary data shown in Fig. 3E), highlighting the heterogeneity of
organoids and probably suggesting that the presence of certain
cellular markers may vary depending on the microenvironmental
conditions.
Inflammatory features are retained in
GBOs for up to one month
Inflammation plays a significant role in the
pathogenesis of glioblastoma, contributing to tumor proliferation,
progression, invasion and therapeutic resistance (11,15).
Therefore, it is of interest to determine whether the previously
mentioned existing cells in GBOs exhibited their characteristic
features during cultivation time, such as the expression of
inflammatory mediators and their receptors. Focus was addressed on
the chemokines C-X-C motif chemokine ligand 12 (CXCL12), C-X-C
motif chemokine ligand 16 (CXCL16) and C-X3-C motif chemokine
ligand 1 (CX3CL1), as well as interleukin-6 (IL-6) and
interleukin-1 beta (IL-1β), together with their respective
receptors. C-X-C motif chemokine receptor 4 (CXCR4), one of the
receptors for CXCL12, is overexpressed in gliomas and, together
with its ligand, promotes migration and invasion (12). Moreover, the CXCL12-CXCR4 axis
facilitates temozolomide resistance, highlighting its central role
in glioblastoma treatment (37).
C-X-C motif chemokine receptor 7 (CXCR7), another receptor for
CXCL12, is known for its anti-apoptotic effects contributing to
enhanced tumor cell survival (38).
It can serve as a prognostic marker, correlating with patient
outcomes (39). The CXCL16-C-X-C
motif chemokine receptor 6 (CXCR6) axis enhances migration via
reverse signaling mechanisms (40)
and modulates T cell activity within the glioblastoma
microenvironment, suggesting a dual role in tumor progression and
immune regulation (41). Finally,
the CX3CL1-C-X3-C motif chemokine receptor 1 (CX3CR1) axis
participates in tumor-immune interactions by accumulating
tumor-associated microglia/macrophages, thereby influencing tumor
growth and progression through modulation of the TME (42).
The results are demonstrated in Fig. 5, with the respective findings for
each individual GBO preparation traceable. In addition, all qPCR
results were presented as box plots as an alternative
representation to provide an improved overview (Fig. S3).
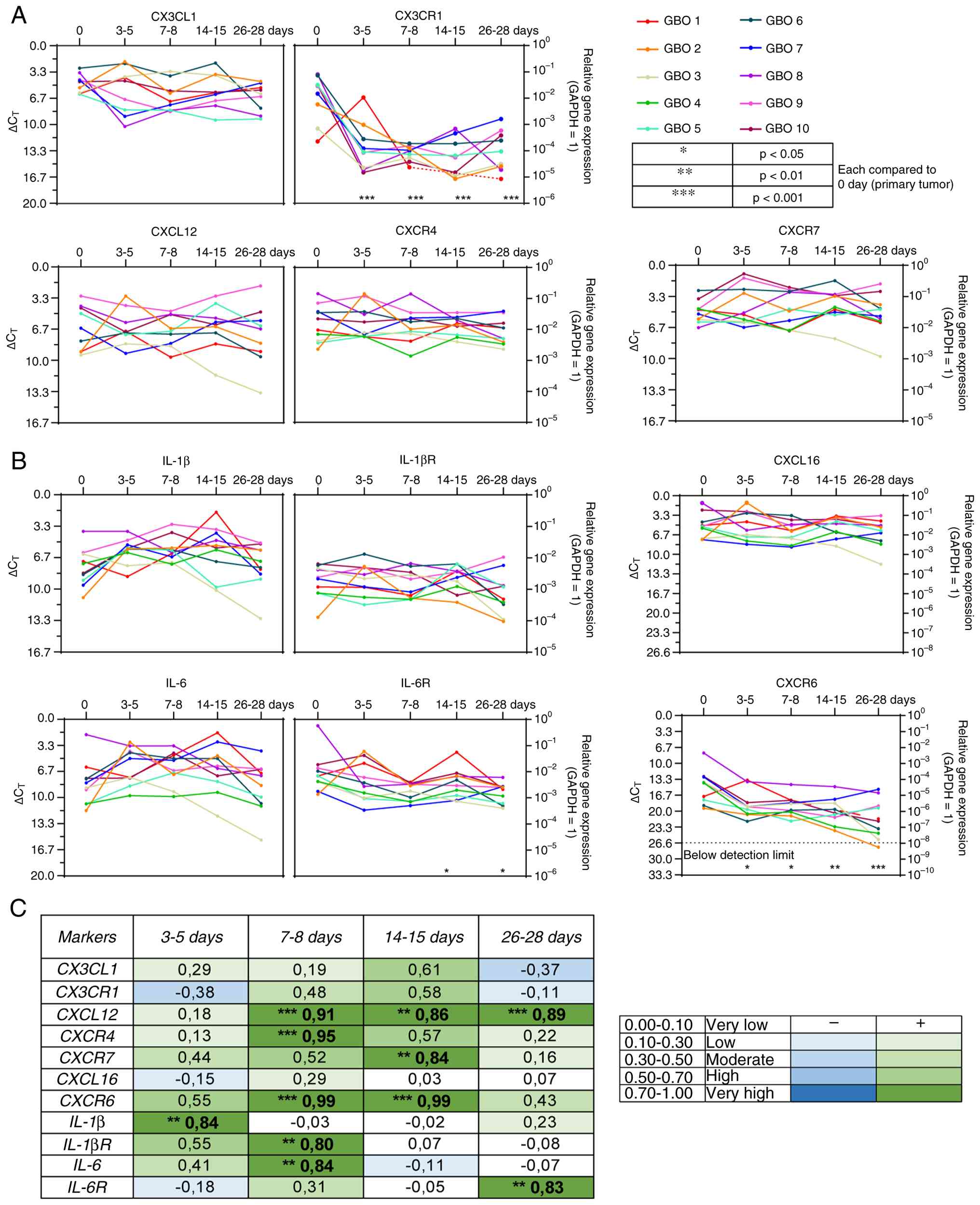 | Figure 5.Reverse transcription-quantitative
PCR and correlation analysis of chemokines and cytokines with their
respective receptors. Gene expression levels are shown across the
entire cultivation period for 10 different glioblastoma organoid
preparations (n=10) at five time points: days 0, 3–5, 7–8, 14–15,
and 26–28. The x-axis represents days, the left y-axis shows
∆CT values, and the right y-axis displays the
logarithmic scale of expression relative to GAPDH as the
housekeeping gene (GAPDH expression=1). (A) Chemokines and
chemokine receptors. (B) Cytokines and cytokine receptors. (C)
Correlation analysis. Correlations are indicated by a color
gradient: Dark green represents a high positive correlation, light
green a low positive correlation, dark blue a high negative
correlation, and light blue a low negative correlation. Statistical
analysis was performed using one-way ANOVA with Dunnett's multiple
comparisons post hoc test. *P<0.05, **P<0.01 and
***P<0.001. GAPDH, glyceraldehyde-3-phosphate dehydrogenase;
CX3CL1, C-X3-C motif chemokine ligand 1; CX3CR1, C-X3-C motif
chemokine receptor 1; CXCL12, C-X-C motif chemokine ligand 12;
CXCR4, C-X-C motif chemokine receptor 4; CXCR7, C-X-C motif
chemokine receptor 7; CXCL16, C-X-C motif chemokine ligand 16;
CXCR6, C-X-C motif chemokine receptor 6; IL-1β, interleukin-1 beta;
IL-1βR, interleukin-1 beta receptor; IL-6, interleukin-6; IL-6R,
interleukin-6 receptor. |
In the experiments of the present study, CXCL12 mRNA
expression showed some heterogeneity in the investigated GBO
preparations but exhibited, over time, a highly significant
positive correlation from day 7 onwards when comparing the
expression between the primary tumor tissues and GBOs cultured for
the respective time points (Figs. 5A
and C, and S3A). CXCL16
expression was more homogeneous in the investigated GBO
preparations and consistent over time, resulting in no significant
differences, while CX3CL1 displayed a very homogeneous mRNA
expression at the beginning of the cultivation procedure and then
developed a certain heterogeneity, resulting in a negative
correlation between the primary tumor tissues and GBOs cultured for
28 days (Figs. 5A and C, and
S3A). The baseline expression
levels were similar in magnitude for all three ligands (Figs. 5A and S3A). The mRNA expression of the chemokine
receptor CXCR4 still revealed a statistically significant positive
correlation on days 7–8 and remained unchanged at other time points
(Figs. 5A and C, and S3A). CXCR7 displayed similar patterns
with some exceptions, showing relatively consistent expression,
exhibiting a positive correlation between the primary tumor tissues
and GBOs cultured for days 14–15. CXCR6 presented the lowest
baseline expression of all investigated chemokine receptors and
exhibited a positive correlation between the primary tumor tissues
and GBOs at two time points. There was a tendency toward
downregulation over time, and one GBO preparation became
undetectable at the latest time point; however, this was not
clearly reflected in the correlation analysis due to the
heterogeneity of the expression patterns in the different GBO
preparations (Figs. 5A and C,
S3A). By contrast, CX3CR1
exhibited a highly heterogeneous mRNA expression with a significant
decline at the end of the GBO cultivation period, resulting in a
negative correlation between the primary tumor tissues and GBOs
cultured for 28 days (Figs. 5A and
C, and S3A).
Proinflammatory cytokines such as IL-6 and IL-1β,
along with their receptors, promote tumor cell proliferation,
invasiveness, and survival in glioblastoma (13,14).
In the present study, the interleukins presented some heterogeneity
within the various GBO preparations, exhibiting similar baseline
mRNA expression levels (Figs. 5B and
C, and S3B). Moreover, the
interleukins, particularly IL-6, showed a downward trend in the
correlation analysis during cultivation time when comparing primary
tumor tissues and GBOs. IL-1β initially displayed a positive
correlation, which shifted to negative or weakly positive values
over time before becoming slightly more positive at the end
(Figs. 5B and C and S3B). In general, IL-6 receptor (IL-6R)
showed a higher baseline mRNA expression than IL-1β receptor
(IL-1βR). IL-1βR also exhibited a positive correlation at the
beginning, which then became slightly negative. IL-6R displayed a
relatively homogeneous expression pattern across the different time
points, with a positive correlation observed at the end of the GBO
cultivation period (Figs. 5B and C,
and S3B).
Cytokines and chemokines, along with their
respective cellular sources, were also visualized qualitatively at
the protein level using immunofluorescence double-staining.
Representative images are shown in Figs. 6 and 7. The stemness markers SOX2 and MSI1 were
combined with CXCR4 and CXCR6 (Fig. 6B
and E), as both receptors are known to be expressed in
glioblastoma progenitor cells (43,44).
S100B was stained together with CXCR7 and CX3CL1, which are
reported to be expressed in glioma cells (38,45).
CX3CR1 was combined with Iba1, reflecting its expression in
microglia (46). Finally, CXCL12
and CXCL16 were stained in combination with GFAP, as both
chemokines are expressed in glial cells (38,47).
In numerous staining constellations, co-staining was observed
during the whole GBO cultivation period, for example, CXCL12 and
CXCL16 with GFAP (Fig. 6A and D),
CX3CL1 with S100B (Fig. 6F), and
CXCR4 with MSI1 (Fig. 6B).
Similarly, co-staining of CX3CR1 and Iba1 was detected (Fig. 6G), consistent with known findings
aforementioned.
 | Figure 6.Immunofluorescence double-staining
images of three glioblastoma organoid preparations at three time
points (days 0, 4, and 28). (A-G) Chemokines and chemokine
receptors. (A-C) CXCL12-CXCR4-CXCR7 axis. (D and E) CXCL16-CXCR6
axis. (F and G) CX3CL1-CX3CR1 axis. Scale bars: 20 µm. CXCL12,
C-X-C motif chemokine ligand 12; CXCR4, C-X-C motif chemokine
receptor 4; CXCR7, C-X-C motif chemokine receptor 7; CXCL16, C-X-C
motif chemokine ligand 16; CXCR6, C-X-C motif chemokine receptor 6;
CX3CL1, C-X3-C motif chemokine ligand 1; CX3CR1, C-X3-C motif
chemokine receptor 1; GFAP, glial fibrillary acidic protein; S100B,
S100 calcium-binding protein B; MSI1, Musashi RNA-binding protein
1; SOX2, SRY-box transcription factor 2; Iba1, ionized
calcium-binding adapter molecule 1. |
 | Figure 7.Cytokines and cytokine receptors were
visualized using immunofluorescence double-staining at three time
points (days 0, 4, and 28), exemplified for one glioblastoma
organoid preparation. (A and B) IL-1β and IL-1βR. (C and D) IL-6
and IL-6R. Scale bars, 20 µm. IL-1β, interleukin-1 beta; IL-1βR,
IL-1β receptor; IL-6, interleukin-6; IL-6R, IL-6 receptor; Iba1,
ionized calcium-binding adapter molecule 1; CD11b, cluster of
differentiation 11b. |
To assess cytokine secretion in immune cells, IL-1β
and its receptor were stained in combination with Iba1 (Fig. 7A and B) to visualize both the
secreted cytokine and its receptor on macrophages and microglia
(48). Additionally, CD11b was
stained with IL-6 and IL-6R (Fig. 7C
and D), given the positive correlation between cytokine
expression and immune cell infiltration reported in glioblastoma
(49). Similar to the chemokines,
the cytokines remained detectable at the protein level during 1
month of cultivation. Moreover, co-staining of, for example, Iba1
and IL-1β, and CD11b and IL-6R was detectable in GBOs up to 28 days
of cultivation time (Fig. 6B and
D). Collectively, all fluorescence images demonstrated that
inflammatory features persisted throughout the 1-month cultivation
period.
In summary, structural proteins, proliferation,
stemness and inflammatory markers remain preserved at the RNA level
during the cultivation method over the course of one month, with
some exceptions (for example, reduction of CXCR6, CX3CR1, and CD3
expression during the cultivation period, and presence of a GFAP
and S100B mRNA expression drop after a 3-5-day cultivation period).
All fluorescence images confirmed that structural proteins,
stemness, and inflammatory markers remained present and detectable
at the protein level even after 28 days of GBO cultivation
time.
Discussion
The present study aimed to analyze the cellular
characteristics and inflammatory features of patient-derived GBOs
during a 1-month cultivation period. The present findings
demonstrated that glial structural proteins, GSCs, immune cells,
vessels, proliferation marker, and inflammatory mediators remained
detectable up to the final time point in most cases. H&E
staining revealed no major structural alterations over time, and
immunofluorescence confirmed the continued presence of nearly all
analyzed markers at the protein level after 28 days. GSCs were
distinct from glial cells, occasionally forming small clusters near
GFAP/S100B-positive cells. Co-staining could be observed as
expected, such as CXCL12/CXCL16 with GFAP, CXCR4 with MSI1, and
CX3CR1 with Iba1, highlighting the preservation of cellular
diversity and functional integrity over time. Both RNA and protein
levels confirmed long-term stability of the GBO preparations, as
most markers remained largely preserved with minor fluctuations in
most cases.
The maintenance of cellular architecture and
characteristic features, including blood vessels, immune cells, and
inflammatory mediators, is essential for preserving an intact TME
in glioblastoma. Jacob et al (2020) (26) successfully optimized patient-derived
GBOs with a particular investigation of cellular characteristics.
They established a biobank and documented the viability of
organoids following cryopreservation. However, at that time, the
long-term stability of the GBOs and the maintenance of key TME
features, including inflammatory mediators, remained
uninvestigated, potentially limiting the translation of data into
patient care. The present study aimed to address this gap by
conducting quantitative analyses of different cell components and
inflammatory mediators over a 1-month cultivation. Despite some
exceptions over a prolonged cultivation period, the observed
stability in the current experiments supports the reliability of
GBOs as a physiologically relevant preclinical model. The
persistence of GSCs within GBO preparations also aligns with
previous findings suggesting that GSCs contribute to tumor
recurrence and therapy resistance (4,7). Their
continued presence alongside glial structural proteins indicates
that GBOs can, in principle, recapitulate the intratumoral
heterogeneity typical of primary glioblastoma tissue.
Nevertheless, although most markers in
patient-derived GBOs showed stable expression, not all remained
consistent throughout the cultivation period. In the present study,
the proliferation marker Ki67 and the glial structural proteins
GFAP and S100B displayed an early, transient drop between days 3–5,
followed by gradual recovery. This may be due to glial tumor cells
experiencing stress in the cultured GBOs, temporarily reducing
marker expression. The stemness marker CD133, however, exhibited
high heterogeneity and declined over time in some GBO preparations.
This heterogeneity can be attributed to the coexistence of
CD133-positive and CD133-negative GSCs (50) and may also be due to population
shifts favoring CD133-negative cells over time. Similarly, CXCR6,
expressed in glioblastoma progenitor cells (43,44),
showed low expression and declined at later time points. This may
result from transcriptional downregulation or epigenetic silencing,
or from loss of the CXCR6-positive subset, while overall stemness
markers remained. These findings align with Hattermann et al
(2013) (43), where CXCR6 was
restricted to a small subset of proliferating glioblastoma cells
co-expressing MSI1, SOX2 and OCT4. A decline in CD3, a marker for T
cells and NK cells (34), was
observed after 14–15 days, likely due to the absence of survival or
activation signals in the organoid environment. CX3CR1, expressed
in microglia (46), decreased
significantly over time, whereas Iba1 expression as a microglia
marker remained stable, suggesting that the CX3CR1 decline reflects
reduced microenvironmental signaling rather than microglial loss.
Notably, CXCR6 and CD133 became undetectable at day 28 in one GBO
preparation, and CD3 decline was most substantial in one GBO
preparation. Comparing these results, all changes were pronounced
in GBO 2, highlighting heterogeneity between GBO preparations.
In summary, the observations of the present study
indicate that key components of the TME fluctuate depending on the
marker, tumor material, and cultivation time. The current results
suggest that the optimal time window for studying patient-derived
GBOs is likely between 7–14 days, when cellular composition is most
stable. Over time, some GBOs developed progressive cell death,
indicating that GBO quality depends on the original tumor material
and reflects both heterogeneity and microenvironment-dependent
variation. Therefore, careful evaluation of GBOs for structural
abnormalities is essential before experimental use to ensure
reproducibility and reliability.
Based on the present findings, future research on
the GBO model should aim to further optimize its application as a
preclinical model. Future studies could routinely implement
intraoperative diagnostics, such as frozen section or detailed
sequencing analyses. This would enable the cultivation of organoids
separately according to their specific genetic and epigenetic
profiles, which could be crucial for testing individualized patient
treatments. Furthermore, correlations between treatment responses
observed in patient-derived GBOs and those of the corresponding
patients could be systematically evaluated.
The scientific significance of the GBOs lies in
their ability to preserve tumor heterogeneity by exhibiting
structural and functional characteristics similar to those of human
organs and tissues (17,21). GBOs offer a high success rate in
culture, short formation times, relatively low cost, and
reproducible tumor traits (22).
Moreover, organoids serve as valuable tools for studying tumor
biology, including the TME, and for testing radiotherapy,
chemotherapy and immunotherapy (18,20–22).
However, several limitations should be considered: First, the key
TME components can fluctuate across different markers, depending on
the marker and tumor material (batch-to-batch variability). Second,
during long-term cultivation, some characteristics changed (reduced
expression of specific immune markers and loss of chemokine
receptors in some GBO preparations), as also observed in the
present study. Third, due to the limited material, the fluorescence
images in the present study were included only to visualize stable
marker expression and typical co-staining of cell-type-specific
markers at the protein level throughout the culture period, without
suggesting quantitative assessment. Nevertheless, while the
patient-derived GBO model of the present study preserves tumor
heterogeneity and TME in general, and can therefore serve as a
valuable preclinical model, it should be used for exploratory and
hypothesis-generating studies. Therefore, its application may be
limited to providing biological and mechanistic insights and should
be used to a limited extent to determine clinical efficacy or
therapeutic decision-making without independent in vivo or
clinical validation.
In summary, the long-term preservation of cellular
and inflammatory characteristics underscores the robustness of
patient-derived GBOs as glioblastoma models. By maintaining the key
components of the TME and the heterogeneity of the original tumor,
GBOs provide a powerful platform for experimental studies and
preclinical drug testing. Their ability to bridge the gap between
experimental research and clinical application highlights their
value in accelerating translational glioblastoma research and
advancing the development of effective, individualized treatment
strategies. However, its application may be limited to providing
biological and mechanistic insights and should be used to a limited
extent to determine clinical efficacy or therapeutic
decision-making, without independent in vivo or clinical
validation. Moreover, time-dependent variations in some marker
expression determine an optimum time window with these organoids,
between 7–14 days, which may limit their use for certain
experiments.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
The authors would like to thank Mrs Corinna Keller
[Department of Neurosurgery, University Medical Center
Schleswig-Holstein (UKSH), Campus Kiel, 24105 Kiel, Germany] for
her expert technical assistance.
Funding
The present study was supported by the Department of
Neurosurgery of Kiel (grant no. NCH 2025).
Availability of data and materials
The data generated in the present study are included
in the figures and/or tables of this article.
Authors' contributions
JHF conceptualized the study. JHF, NOS, JH, JC and
JN developed methodology. NOS, JN and JC performed software
analysis and investigation. NOS and JN validated data. NOS and JN
confirm the authenticity of all the raw data. HA and MS provided
resources. NOS, JN, JC, HA, MS and JHF curated data. JN prepared
the original draft of the manuscript. All authors wrote, reviewed
and edited the manuscript. JN visualized data. JHF supervised the
study and conducted project administration. MS acquired funding.
All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The present study was performed in accordance with
the Declaration of Helsinki and received approval from the Ethics
Committee of the University of Kiel (approval no D524/17; Kiel,
Germany). Written informed consent was obtained from all
participants involved in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, artificial
intelligence tools were used to improve the readability and
language of the manuscript or to generate images, and subsequently,
the authors revised and edited the content produced by the
artificial intelligence tools as necessary, taking full
responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Schaff LR and Mellinghoff IK: Glioblastoma
and other primary brain malignancies in adults: A review. JAMA.
329:5742023. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tan AC, Ashley DM, López GY, Malinzak M,
Friedman HS and Khasraw M: Management of glioblastoma: State of the
art and future directions. CA Cancer J Clin. 70:299–312.
2020.PubMed/NCBI
|
|
3
|
Stupp R, Weller M, Belanger K, Bogdahn U,
Ludwin SK, Lacombe D and Mirimanoff RO: Radiotherapy plus
concomitant and adjuvant temozolomide for glioblastoma. N Engl J
Med. 352:987–996. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Alves ALV, Gomes INF, Carloni AC, Rosa MN,
da Silva LS, Evangelista AF, Reis RM and Silva VAO: Role of
glioblastoma stem cells in cancer therapeutic resistance: A
perspective on antineoplastic agents from natural sources and
chemical derivatives. Stem Cell Res Ther. 12:2062021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang P, Xia Q, Liu L, Li S and Dong L:
Current opinion on molecular characterization for GBM
classification in guiding clinical diagnosis, prognosis, and
therapy. Front Mol Biosci. 7:5627982020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sottoriva A, Spiteri I, Piccirillo SG,
Touloumis A, Collins VP, Marioni JC, Curtis C, Watts C and Tavaré
S: Intratumor heterogeneity in human glioblastoma reflects cancer
evolutionary dynamics. Proc Natl Acad Sci. 110:4009–4014. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Dirkse A, Golebiewska A, Buder T, Nazarov
PV, Muller A, Poovathingal S, Brons NHC, Leite S, Sauvageot N,
Sarkisjan D, et al: Stem cell-associated heterogeneity in
Glioblastoma results from intrinsic tumor plasticity shaped by the
microenvironment. Nat Commun. 10:17872019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Barthel L, Hadamitzky M, Dammann P,
Schedlowski M, Sure U, Thakur BK and Hetze S: Glioma: Molecular
signature and crossroads with tumor microenvironment. Cancer
Metastasis Rev. 41:53–75. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gieryng A, Pszczolkowska D, Walentynowicz
KA, Rajan WD and Kaminska B: Immune microenvironment of gliomas.
Lab Invest. 97:498–518. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nagarsheth N, Wicha MS and Zou W:
Chemokines in the cancer microenvironment and their relevance in
cancer immunotherapy. Nat Rev Immunol. 17:559–572. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Corsaro A, Tremonti B, Bajetto A, Barbieri
F, Thellung S and Florio T: Chemokine signaling in tumors:
Potential role of CXC chemokines and their receptors as
glioblastoma therapeutic targets. Expert Opin Ther Targets.
28:937–952. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dai Y, Yu C, Zhou L, Cheng L, Ni H and
Liang W: Chemokine receptor CXCR4 interacts with nuclear receptor
Nur77 and promote glioma invasion and progression. Brain Res.
1822:1486472024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Detchou D and Barrie U: Interleukin 6 and
cancer resistance in glioblastoma multiforme. Neurosurg Rev.
47:5412024. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Narasimhappagari J, Liu L, Balasubramaniam
M, Ayyadevara S, Aboud O and Griffin WST: The seminal role of the
proinflammatory cytokine IL-1β and its signaling cascade in
glioblastoma pathogenesis and the therapeutic effect of
Interleukin-1β receptor antagonist (IL-1RA) and tolcapone. Int J
Mol Sci. 26:68932025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mentlein R, Hattermann K and Held-Feindt
J: Migration, metastasis, and more: The role of chemokines in the
proliferation, spreading, and metastasis of tumors. Trends in Stem
Cell Proliferation and Cancer Research. Resende RR and Ulrich H:
Springer Netherlands; Dordrecht: pp. 339–358. 2013, View Article : Google Scholar
|
|
16
|
Klein E, Hau AC, Oudin A, Golebiewska A
and Niclou SP: Glioblastoma organoids: Pre-Clinical applications
and challenges in the context of immunotherapy. Front Oncol.
10:6041212020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pernik MN, Bird CE, Traylor JI, Shi DD,
Richardson TE, McBrayer SK and Abdullah KG: Patient-derived cancer
organoids for precision oncology treatment. J Pers Med. 11:4232021.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang C, Jin M, Zhao J, Chen J and Jin W:
Organoid models of glioblastoma: Advances, applications and
challenges. Am J Cancer Res. 10:2242–2257. 2020.PubMed/NCBI
|
|
19
|
Xu X, Li L, Luo L, Shu L, Si X, Chen Z,
Xia W, Huang J, Liu Y, Shao A and Ke Y: Opportunities and
challenges of glioma organoids. Cell Commun Signal. 19:1022021.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pawlowski KD, Duffy JT, Babak MV and
Balyasnikova IV: Modeling glioblastoma complexity with organoids
for personalized treatments. Trends Mol Med. 29:282–296. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang X, Sun Y, Zhang DY, Ming G and Song
H: Glioblastoma modeling with 3D organoids: Progress and
challenges. Oxf Open Neurosci. 2:kvad0082023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu C, Yuan X, Hou P, Li Z, Wang C, Fang C
and Tan Y: Development of glioblastoma organoids and their
applications in personalized therapy. Cancer Biol Med. 20:353–368.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lancaster MA, Renner M, Martin CA, Wenzel
D, Bicknell LS, Hurles ME, Homfray T, Penninger JM, Jackson AP and
Knoblich JA: Cerebral organoids model human brain development and
microcephaly. Nature. 501:373–379. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Soubéran A and Tchoghandjian A: Practical
review on preclinical human 3D glioblastoma models: Advances and
challenges for clinical translation. Cancers. 12:23472020.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hubert CG, Rivera M, Spangler LC, Wu Q,
Mack SC, Prager BC, Couce M, McLendon RE, Sloan AE and Rich JN: A
Three-dimensional organoid culture system derived from human
glioblastomas recapitulates the hypoxic gradients and cancer stem
cell heterogeneity of tumors found in vivo. Cancer Res.
76:2465–2477. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jacob F, Salinas RD, Zhang DY, Nguyen PTT,
Schnoll JG, Wong SZH, Thokala R, Sheikh S, Saxena D, Prokop S, et
al: A Patient-derived glioblastoma organoid model and biobank
recapitulates Inter- and Intra-tumoral heterogeneity. Cell.
180:188–204.e22. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hellmold D, Johanning L, Clüver J, Holler
J, Schröder NO, Bayler F, Ahmeti H, Kubelt-Kwamin C, Wieker S,
Helmers AK, et al: Effect of focused ultrasound-induced mechanical
ablation on stemness and dormancy properties of
residual/peri-focally localized glioblastoma cells. Neurooncol Adv.
7:vdaf1842025.PubMed/NCBI
|
|
28
|
Qiang J, Wei Z, Xiao-guang Q and Wei Y:
Gene expression profiling reveals Ki-67 associated proliferation
signature in human glioblastoma. Chin Med J (Engl). 124:2584–2588.
2022.PubMed/NCBI
|
|
29
|
Jung CS, Foerch C, Schanzer A, Heck A,
Plate KH, Seifert V, Steinmetz H, Raabe A and Sitzer M: Serum GFAP
is a diagnostic marker for glioblastoma multiforme. Brain.
130:3336–3341. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang H, Zhang L, Zhang IY, Chen X, Da
Fonseca A, Wu S, Ren H, Badie S, Sadeghi S, Ouyang M, et al: S100B
promotes glioma growth through chemoattraction of Myeloid-derived
macrophages. Clin Cancer Res. 19:3764–3775. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang P, Zhao L, Gong S, Xiong S, Wang J,
Zou D, Pan J, Deng Y, Yan Q, Wu N and Liao B: HIF1α/HIF2α-Sox2/Klf4
promotes the malignant progression of glioblastoma via the
EGFR-PI3K/AKT signalling pathway with positive feedback under
hypoxia. Cell Death Dis. 12:3122021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Polat B, Wohlleben G, Kosmala R, Lisowski
D, Mantel F, Lewitzki V, Löhr M, Blum R, Herud P, Flentje M and
Monoranu CM: Differences in stem cell marker and osteopontin
expression in primary and recurrent glioblastoma. Cancer Cell Int.
22:872022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Noorani I, Petty G, Grundy PL, Sharpe G,
Willaime-Morawek S, Harris S, Thomas GJ, Nicoll JA and Boche D:
Novel association between microglia and stem cells in human
gliomas: A contributor to tumour proliferation? J Pathol Clin Res.
1:67–75. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kmiecik J, Poli A, Brons NH, Waha A, Eide
GE, Enger PØ, Zimmer J and Chekenya M: Elevated CD3+ and CD8+
tumor-infiltrating immune cells correlate with prolonged survival
in glioblastoma patients despite integrated immunosuppressive
mechanisms in the tumor microenvironment and at the systemic level.
J Neuroimmunol. 264:71–83. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lu-Emerson C, Snuderl M, Kirkpatrick ND,
Goveia J, Davidson C, Huang Y, Riedemann L, Taylor J, Ivy P, Duda
DG, et al: Increase in tumor-associated macrophages after
antiangiogenic therapy is associated with poor survival among
patients with recurrent glioblastoma. Neuro Oncol. 15:1079–1087.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mei X, Chen YS, Chen FR, Xi SY and Chen
ZP: Glioblastoma stem cell differentiation into endothelial cells
evidenced through live-cell imaging. Neuro-Oncol. 19:1109–1118.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang S, Chen C, Li J, Xu X, Chen W and Li
F: The CXCL12/ CXCR4 axis confers temozolomide resistance to human
glioblastoma cells via up-regulation of FOXM1. J Neurol Sci.
414:1168372020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hattermann K, Held-Feindt J, Lucius R,
Müerköster SS, Penfold MET, Schall TJ and Mentlein R: The chemokine
receptor CXCR7 is highly expressed in human glioma cells and
mediates antiapoptotic effects. Cancer Res. 70:3299–3308. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Deng L, Zheng W, Dong X, Liu J, Zhu C, Lu
D, Zhang J, Song L, Wang Y and Deng D: Chemokine receptor CXCR7 is
an independent prognostic biomarker in glioblastoma. Cancer
Biomark. 20:1–6. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Adamski V, Mentlein R, Lucius R, Synowitz
M, Held-Feindt J and Hattermann K: The chemokine receptor CXCR6
evokes reverse signaling via the transmembrane chemokine CXCL16.
Int J Mol Sci. 18:14682017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chia TY, Billingham LK, Boland L, Katz JL,
Arrieta VA, Shireman J, Rosas AL, DeLay SL, Zillinger K, Geng Y, et
al: The CXCL16-CXCR6 axis in glioblastoma modulates T-cell activity
in a spatiotemporal context. Front Immunol. 14:13312872024.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lee S, Latha K, Manyam G, Yang Y, Rao A
and Rao G: Role of CX3CR1 signaling in malignant transformation of
gliomas. Neuro Oncol. 22:1463–1473. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hattermann K, Held-Feindt J, Ludwig A and
Mentlein R: The CXCL16-CXCR6 chemokine axis in glial tumors. J
Neuroimmunol. 260:47–54. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ehtesham M, Mapara KY, Stevenson CB and
Thompson RC: CXCR4 mediates the proliferation of glioblastoma
progenitor cells. Cancer Lett. 274:305–312. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Sciumè G, Soriani A, Piccoli M, Frati L,
Santoni A and Bernardini G: CX3CR1/CX3CL1 axis negatively controls
glioma cell invasion and is modulated by transforming growth
factor-beta1. Neuro Oncol. 12:701–710. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hattermann K, Sebens S, Helm O, Schmitt
AD, Mentlein R, Mehdorn HM and Held-Feindt J: Chemokine expression
profile of freshly isolated human glioblastoma-associated
macrophages/microglia. Oncol Rep. 32:270–276. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ludwig A, Schulte A, Schnack C, Hundhausen
C, Reiss K, Brodway N, Held-Feindt J and Mentlein R: Enhanced
expression and shedding of the transmembrane chemokine CXCL16 by
reactive astrocytes and glioma cells. J Neurochem. 93:1293–1303.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kai K, Komohara Y, Esumi S, Fujiwara Y,
Yamamoto T, Uekawa K, Ohta K, Takezaki T, Kuroda J, Shinojima N, et
al: Macrophage/microglia-derived IL-1β induces glioblastoma growth
via the STAT3/NF-κB pathway. Hum Cell. 35:226–237. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kim S, Kim KH, Jung H, Jeong EO, Lee HJ,
Kwon J, Kwon HJ, Choi SW, Koh HS and Kim SH: Elevated serum IL-6 as
a negative prognostic biomarker in glioblastoma: Integrating
bioinformatics and clinical validation. J Cancer. 16:802–811. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Chen R, Nishimura MC, Bumbaca SM,
Kharbanda S, Forrest WF, Kasman IM, Greve JM, Soriano RH, Gilmour
LL and Rivers CS: A hierarchy of Self-renewing Tumor-initiating
cell types in glioblastoma. Cancer Cell. 17:362–375. 2010.
View Article : Google Scholar : PubMed/NCBI
|





















