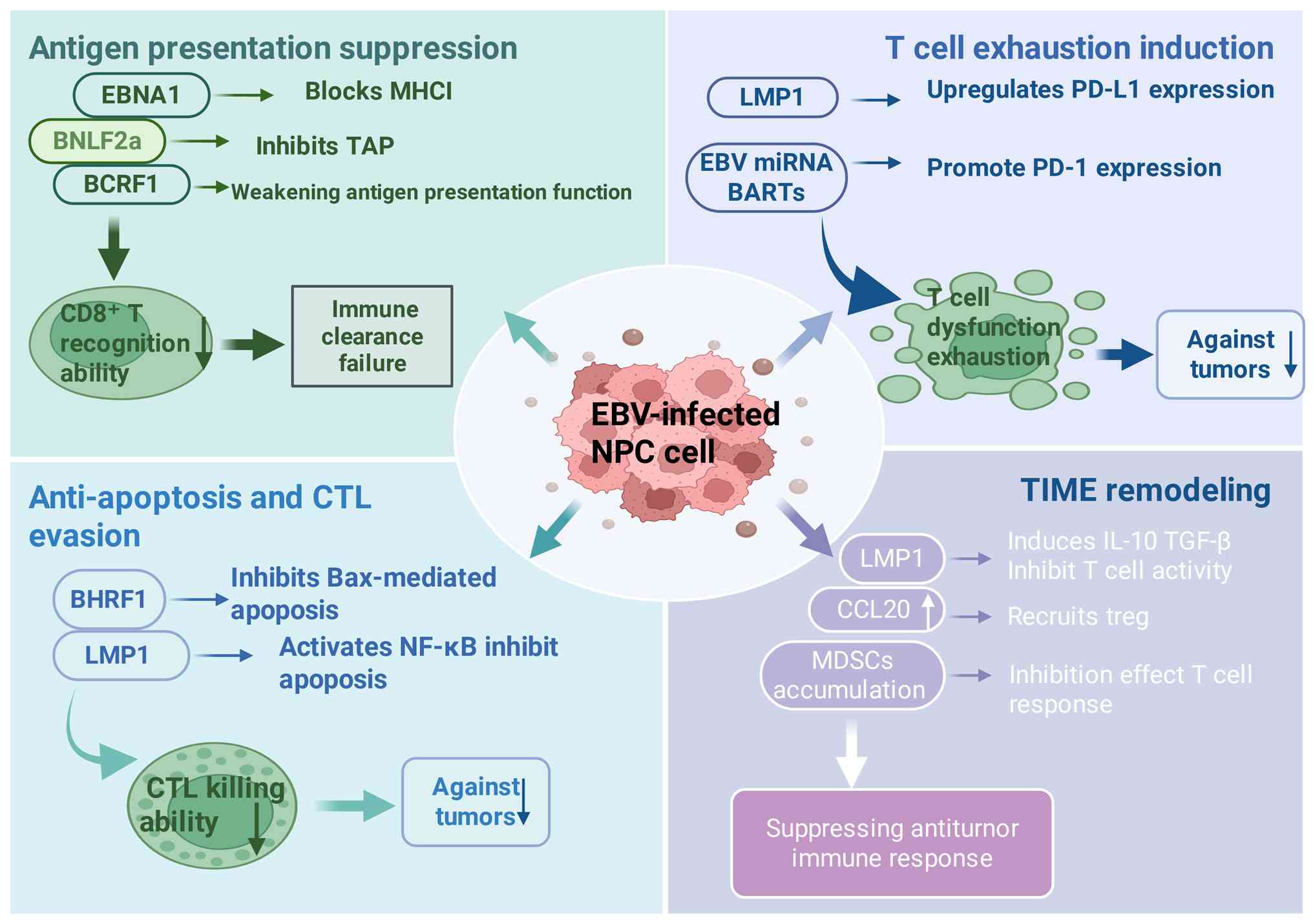
|
1
|
Han S, Tay JK, Loh CJL, Chu AJM, Yeong
JPS, Lim CM and Toh HC: Epstein-barr virus epithelial Cancers-A
comprehensive understanding to drive novel therapies. Front
Immunol. 12:7342932021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jin S, Li R, Chen MY, Yu C, Tang LQ, Liu
YM, Li JP, Liu YN, Luo YL, Zhao Y, et al: Single-cell
transcriptomic analysis defines the interplay between tumor cells,
viral infection, and the microenvironment in nasopharyngeal
carcinoma. Cell Res. 30:950–965. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mahajan S, Bongaerts M, Hardillo J, Tsang
A, Lo KW, Kortleve D, Ma B and Debets R: Transcriptomics of
Epstein-Barr virus aids to the classification of T-cell evasion in
nasopharyngeal carcinoma. Curr Opin Immunol. 83:1023352023.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lo AK, Dawson CW, Lung HL, Wong KL and
Young LS: The Role of EBV-Encoded LMP1 in the NPC tumor
microenvironment: From Function to therapy. Front Oncol.
11:6402072021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li X, Guo Y, Xiao M and Zhang W: The
immune escape mechanism of nasopharyngeal carcinoma. FASEB J.
37:e230552023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li W, Lv S, Liu G, Lu N, Jiang Y, Liang H,
Xia W, Xiang Y, Xie C and He J: Epstein-Barr virus DNA
seropositivity links distinct tumoral heterogeneity and immune
landscape in nasopharyngeal carcinoma. Front Immunol.
14:11240662023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lin Q, Zhou Y, Ma J, Han S, Huang Y, Wu F,
Wang X, Zhang Y, Mei X and Ma L: Single-cell analysis reveals the
multiple patterns of immune escape in the nasopharyngeal carcinoma
microenvironment. Clin Transl Med. 13:e13152023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cao Z, Pan C, Liu Z, Quan Q, Li M, Huang
Y, Liang C, Chen Y, Fan T, Chen P, et al: Co-targeting MRPS7-23
synergistically enhances cisplatin efficacy to suppress
nasopharyngeal carcinoma growth and metastasis. Int J Biol Sci.
22:970–994. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yarza R, Bover M, Agulló-Ortuño MT and
Iglesias-Docampo LC: Current approach and novel perspectives in
nasopharyngeal carcinoma: The role of targeting proteasome
dysregulation as a molecular landmark in nasopharyngeal cancer. J
Exp Clin Cancer Res. 40:2022021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yan C, Zheng L, Jiang S, Yang H, Guo J,
Jiang LY, Li T, Zhang H, Bai Y, Lou Y, et al: Exhaustion-associated
cholesterol deficiency dampens the cytotoxic arm of antitumor
immunity. Cancer Cell. 41:1276–1293.e11. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cai M, Wang Y, Ma H, Yang L and Xu Z:
Advances and challenges in immunotherapy for locally advanced
nasopharyngeal carcinoma. Cancer Treat Rev. 131:1028402024.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Qing D, Lu Z and Lu H: Long-term immune
checkpoint inhibitor therapy in a patient with metastatic
nasopharyngeal carcinoma: A case report. Front Immunol.
16:15858442025. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu D, Liu L, Zhao X, Zhang X, Chen X, Che
X and Wu G: A comprehensive review on targeting diverse immune
cells for anticancer therapy: Beyond immune checkpoint inhibitors.
Crit Rev Oncol Hematol. 210:1047022025. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sun L, Liu C and Peng J: Specific immune
responses and oncolytic effects induced by EBV LMP2A-Armed modified
Ankara-Vaccinia virus vectored vaccines in nasopharyngeal cancer.
Pharmaceutics. 17:522025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cui X and Snapper CM: Epstein barr virus:
Development of vaccines and immune cell therapy for EBV-Associated
diseases. Front Immunol. 12:7344712021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhao GX, Bu GL, Liu GF, Kong XW, Sun C, Li
ZQ, Dai DL, Sun HX, Kang YF, Feng GK, et al: mRNA-based vaccines
targeting the T-cell Epitope-rich domain of epstein barr virus
latent proteins elicit robust Anti-tumor immunity in mice. Adv Sci
(Weinh). 10:e23021162023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lechner M, Schartinger VH, Steele CD, Nei
WL, Ooft ML, Schreiber LM, Pipinikas CP, Chung GT, Chan YY, Wu F,
et al: Somatostatin receptor 2 expression in nasopharyngeal cancer
is induced by Epstein Barr virus infection: Impact on prognosis,
imaging and therapy. Nat Commun. 12:1172021. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Prasad M, Ponnalagu S, Zeng Q, Luu K, Lang
SM, Wong HY, Cheng MS, Wu M, Mallilankaraman K, Sobota RM, et al:
Epstein-Barr virus-induced ectopic CD137 expression helps
nasopharyngeal carcinoma to escape immune surveillance and enables
targeting by chimeric antigen receptors. Cancer Immunol Immunother.
71:2583–2596. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Argirion I, Pfeiffer RM, Proietti C,
Coghill AE, Yu KJ, Middeldorp JM, Sarathkumara YD, Hsu WL, Chien
YC, Lou PJ, et al: Comparative analysis of the humoral immune
response to the EBV proteome across EBV-related malignancies.
Cancer Epidemiol Biomarkers Prev. 32:687–696. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhao B: Epstein-Barr Virus B cell growth
transformation: The nuclear events. Viruses. 15:8322023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jin J, Sun T, Zhang M, Cheng J, Gu J,
Huang L, Xiao M, Zhou J and Luo H: EBV-Encoded MicroRNA-BART17-3p
targets DDX3X and promotes EBV infection in EBV-Associated
T/Natural Killer-cell lymphoproliferative diseases. Open Forum
Infect Dis. 10:ofad5162023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Deobagkar-Lele M, Crawford G, Crockford
TL, Back J, Hodgson R, Bhandari A, Bull KR and Cornall RJ: B cells
require DOCK8 to elicit and integrate T cell help when antigen is
limiting. Sci Immunol. 9:eadd48742024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gan CP, Kok SY, Lee BKB, Zulaziz N, Cheong
SC, Savelyeva N and Lim KP: Cancer vaccine overcomes immune evasion
of nasopharyngeal carcinoma by restoring MHC-I through
transcriptional regulation of NLRC5. J Transl Med. 23:14142025.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang H, Fan S, Zhan Y, Xu Y, Du Y, Luo J,
Zang H, Peng S and Wang W: Targeting EGFR-binding protein SLC7A11
enhancing antitumor immunity of T cells via inducing MHC-I antigen
presentation in nasopharyngeal carcinoma. Cell Death Dis.
16:212025. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Warner BE and Shair KH: High IgG titers
against EBV glycoprotein 42 correlate with lower risk of
nasopharyngeal carcinoma. J Clin Invest. 135:e1892072025.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang S, Chen R, Wu Y, Song X, Peng X and
Chen M: Fluorinated polyethyleneimine vectors with serum resistance
and adjuvant effect to deliver LMP2 mRNA vaccine for nasopharyngeal
carcinoma therapy. Acta Biomater. 192:340–352. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Guo M, Duan X, Peng X, Jin Z, Huang H,
Xiao W, Zheng Q, Deng Y, Fan N, Chen K and Song X: A lipid-based
LMP2-mRNA vaccine to treat nasopharyngeal carcinoma. Nano Res.
16:5357–5367. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Campos Canesso MC, de Castro TBR,
Nakandakari-Higa S, Lockhart A, Luehr J, Bortolatto J, Parsa R,
Esterházy D, Lyu M, Liu TT, et al: Identification of
antigen-presenting cell-T cell interactions driving immune
responses to food. Science. 387:eado50882025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Xie Y, Wu Q, Jia Y, Cheng L, Tian Y, Yang
X, Wang W, Xu W, Zhao Z and Wei Y: Enhanced antigen presentation by
macrophages promotes human atherosclerosis progression: Therapeutic
implications. Eur Heart J. Nov 11–2025.(Epub ahead of print). doi:
10.1093/eurheartj/ehaf878. View Article : Google Scholar
|
|
30
|
30. He Y, Mohapatra G, Asokan S, Nobs SP
and Elinav E: Microbiome modulation of antigen presentation in
tolerance and inflammation. Curr Opin Immunol. 91:1024712024.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lin M, Zhang XL, You R, Yang Q, Zou X, Yu
K, Liu YP, Zou RH, Hua YJ, Huang PY, et al: Neoantigen landscape in
metastatic nasopharyngeal carcinoma. Theranostics. 11:6427–6444.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yang H, Zeng X, Chen X, Cai S, Li Y, Xu W,
Lai J and Qiu S: The key role of DLX6 in nasopharyngeal carcinoma:
Metastasis, angiogenesis and tumor immune mechanism. Front Immunol.
16:15225802025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sharma S, Mehta NU, Sauer T, Rollins LA,
Dittmer DP and Rooney CM: Cotargeting EBV lytic as well as latent
cycle antigens increases T-cell potency against lymphoma. Blood
Adv. 8:3360–3371. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang S, Zhou Y, Liu Z, Wang Y, Zhou X,
Chen H, Zhang X, Chen Y, Feng Q, Ye X, et al: Immunosequencing
identifies signatures of T cell responses for early detection of
nasopharyngeal carcinoma. Cancer Cell. 43:1423–1441.e10. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Massey J, Artuz C, Dyer Z, Jackson K, Khoo
M, Visweswaran M, Withers B, Moore J, Ma D and Sutton I:
Diversification and expansion of the EBV-reactive cytotoxic T
lymphocyte repertoire following autologous haematopoietic stem cell
transplant for multiple sclerosis. Clin Immunol. 254:1097092023.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rudin CM, Balli D, Lai WV, Richards AL,
Nguyen E, Egger JV, Choudhury NJ, Sen T, Chow A, Poirier JT, et al:
Clinical benefit from immunotherapy in patients with SCLC is
associated with tumor capacity for antigen presentation. J Thorac
Oncol. 18:1222–1232. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Perales-Linares R, Leli NM, Mohei H, Beghi
S, Rivera OD, Kostopoulos N, Giglio A, George SS, Uribe-Herranz M,
Costabile F, et al: Parkin deficiency suppresses antigen
presentation to promote tumor immune evasion and immunotherapy
resistance. Cancer Res. 83:3562–3576. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang X, Chai Y, Quan Y, Wang J, Song J,
Zhou W, Xu X, Xu H, Wang B and Cao X: NPM1 inhibits tumoral antigen
presentation to promote immune evasion and tumor progression. J
Hematol Oncol. 17:972024. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Roberts A, Bentley L, Tang T, Stewart F,
Pallini C, Juvvanapudi J, Wallace GR, Cooper AJ, Scott A, Thickett
D, et al: Ex vivo modelling of PD-1/PD-L1 immune checkpoint
blockade under acute, chronic, and exhaustion-like conditions of
T-cell stimulation. Sci Rep. 11:40302021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fu XH, Guan RY, Huang Z, Li Y, Lu G, Mou
WW and Du J: From multiple myeloma to acute myeloid leukemia: A
case report of a 61-year-old woman after 8 years of chemotherapy
and immunotherapy. Recent Pat Anticancer Drug Discov. 19:396–401.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lu F, Bai X, Lv Z and Zhang W: Immune
archetypes TIME classification system for nasopharyngeal carcinoma:
A new direction for precision immunotherapy. Biochim Biophys Acta
Rev Cancer. 1880:1894872025. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yeo ELL, Hong BH, Tay SH, Neo J, Ong EHW,
Chow WM, Tan KM, Low KP, Sim AYL, Lu T, et al: Tumor immune
microenvironment delineates progression trajectories of distinct
nasopharyngeal carcinoma phenotypes. Cell Rep Med. 6:1021432025.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cammann C, Israel N, Frentzel S, Jeron A,
Topfstedt E, Schüler T, Simeoni L, Zenker M, Fehling HJ, Schraven
B, et al: T cell-specific constitutive active SHP2 enhances T cell
memory formation and reduces T cell activation. Front Immunol.
13:9586162022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tang K, Zhao M, Wu YH, Wu Q, Wang S, Dong
Y, Yu B, Song Y and Liu HM: Structure-based design, synthesis and
biological evaluation of aminopyrazines as highly potent,
selective, and cellularly active allosteric SHP2 inhibitors. Eur J
Med Chem. 230:1141062022. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhao Y and Jiang L: Targeting SHP1 and
SHP2 to suppress tumors and enhance immunosurveillance. Trends Cell
Biol. 35:667–677. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wang Y, Zhang W, Shi R, Luo Y, Feng Z,
Chen Y, Zhang Q, Zhou Y, Liang J, Ye X, et al: Identification of
HLA-A*11:01 and A*02:01-Restricted EBV Peptides Using HLA
Peptidomics. Viruses. 16:6692024. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Chen Y, Ouyang D, Wang Y, Pan Q, Zhao J,
Chen H, Yang X, Tang Y, Wang Q, Li Y, et al: EBV promotes
TCR-T-cell therapy resistance by inducing CD163+M2 macrophage
polarization and MMP9 secretion. J Immunother Cancer.
12:e0083752024. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Bhandarkar V, Dinter T and Spranger S:
Architects of immunity: How dendritic cells shape CD8+ T cell fate
in cancer. Sci Immunol. 10:eadf47262025. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wankhede D, Halama N, Kloor M, Edelmann D,
Brenner H and Hoffmeister M: Prognostic Value of CD8+ T cells at
the invasive margin is comparable to the immune score in
nonmetastatic colorectal cancer: A prospective multicentric cohort
study. Clin Cancer Res. 31:1711–1718. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Soeratram TTD, Biesma HD, Egthuijsen JMP,
Meershoek-Klein Kranenbarg E, Hartgrink HH, van de Velde CJH,
Mookhoek A, van Dijk E, Kim Y, Ylstra B, et al: Prognostic Value of
T-Cell density in the tumor center and outer margins in gastric
cancer. Mod Pathol. 36:1002182023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wu R, Horimoto Y, Oshi M, Benesch MGK,
Khoury T, Takabe K and Ishikawa T: Emerging measurements for
tumor-infiltrating lymphocytes in breast cancer. Jpn J Clin Oncol.
54:620–629. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hassan WA, ElBanna AK, Noufal N, El-Assmy
M, Lotfy H and Ali RI: Significance of tumor-associated
neutrophils, lymphocytes, and neutrophil-to-lymphocyte ratio in
non-invasive and invasive bladder urothelial carcinoma. J Pathol
Transl Med. 57:88–94. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Sarnaik AA, Hwu P, Mulé JJ and
Pilon-Thomas S: Tumor-infiltrating lymphocytes: A new hope. Cancer
Cell. 42:1315–1318. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Del M, Illac C, Morisseau M, Angeles MA,
Ducassou A, Betrian S, Bataillon G, Ferron G, Chantalat E, Gabiache
E and Martinez A: Intraepithelial tumor-infiltrating lymphocytes
shape loco-regional PET/CT spread of locally advanced cervical
cancer. Int J Gynecol Cancer. 34:490–496. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Pauken KE and Wherry EJ: Overcoming T cell
exhaustion in infection and cancer. Trends Immunol. 36:265–276.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zinselmeyer BH, Heydari S, Sacristán C,
Nayak D, Cammer M, Herz J, Cheng X, Davis SJ, Dustin ML and
McGavern DB: PD-1 promotes immune exhaustion by inducing antiviral
T cell motility paralysis. J Exp Med. 210:757–774. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Juneja VR, McGuire KA, Manguso RT, LaFleur
MW, Collins N, Haining WN, Freeman GJ and Sharpe AH: PD-L1 on tumor
cells is sufficient for immune evasion in immunogenic tumors and
inhibits CD8 T cell cytotoxicity. J Exp Med. 214:895–904. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wang M: Canagliflozin disrupts T cell
activation. Nat Rev Nephrol. 19:4782023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lao TD and Le TAH: Hypermethylation of
genes on chromosome 3p as a biomarker for nasopharyngeal carcinoma
diagnosis: A Vietnamese case-control study. Int J Biol Markers.
39:201–208. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Beckers D, Jainarayanan AK, Dustin ML and
Capera J: T cell resistance: On the mechanisms of T cell
Non-activation. Immune Netw. 24:e422024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Law SC, Hoang T, O'Rourke K, Tobin JWD,
Gunawardana J, Loo-Oey D, Bednarska K, Merida de Long L, Sabdia MB,
Hapgood G, et al: Successful treatment of Epstein-Barr
virus-associated primary central nervous system lymphoma due to
post-transplantation lymphoproliferative disorder, with ibrutinib
and third-party Epstein-Barr virus-specific T cells. Am J
Transplant. 21:3465–3471. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Hao B, Lin S, Liu H, Xu J, Chen L, Zheng
T, Zhang W, Dang Y, Reiter RJ, Li C, et al: Baicalein tethers
CD274/PD-L1 for autophagic degradation to boost antitumor immunity.
Autophagy. 21:917–933. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Saddozai UAK, Lu Z, Dong S, Khawar MB, Fan
Z, Cai L, Guo X, Akbar MU, Khattak S, Sun H and Wang Y: Prognostic
value of metal-based ferroptosis and cuproptosis genes and score in
lower grade gliomas. Front Immunol. 16:16080772025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Shi H, Chen S and Chi H: Immunometabolism
of CD8+ T cell differentiation in cancer. Trends Cancer.
10:610–626. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Cele S, Karim F, Lustig G, San JE,
Hermanus T, Tegally H, Snyman J, Moyo-Gwete T, Wilkinson E,
Bernstein M, et al: SARS-CoV-2 evolved during advanced HIV disease
immunosuppression has Beta-like escape of vaccine and Delta
infection elicited immunity. medRxiv. Dec 7–2021.(Epub ahead of
print). doi: 10.1101/2021.09.14.21263564.
|
|
66
|
Zagatto AM, Bishop DJ, Antunes BM, Beck
WR, Malta ES, de Poli RAB, Cavaglieri CR, Chacon-Mikahil MPT and
Castro A: Impacts of high-intensity exercise on the metabolomics
profile of human skeletal muscle tissue. Scand J Med Sci Sports.
32:402–413. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Chen Z and Zhang X: The role of metabolic
reprogramming in kidney cancer. Front Oncol. 14:14023512024.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Yang Y, Fan D, Zheng BH and Zhou ST:
Latest findings on the function of immune metabolism in tumor
immunity. Sichuan Da Xue Xue Bao Yi Xue Ban. 54:497–504. 2023.(In
Chinese). PubMed/NCBI
|
|
69
|
Jonker PB and Muir A: Metabolic ripple
effects-deciphering how lipid metabolism in cancer interfaces with
the tumor microenvironment. Dis Model Mech. 17:dmm0508142024.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zhu M, Hu Y, Gu Y, Lin X, Jiang X, Gong C
and Fang Z: Role of amino acid metabolism in tumor immune
microenvironment of colorectal cancer. Am J Cancer Res. 15:233–247.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Borrego-Yaniz G, Márquez A,
Estupiñán-Moreno E, Terrón-Camero LC, González-Gay MA, Castañeda S,
Guggino G, Saadoun D, Lio P, Fontana S, et al: Genome-Wide DNA
methylation study reveals specific signatures in the affected
arterial tissue of patients with giant cell arteritis. Arthritis
Rheumatol. 78:385–393. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Li C, Yuan Y, Jiang X and Wang Q:
Epigenetic regulation of CD8+ T cell exhaustion: Recent advances
and update. Front Immunol. 16:17000392025. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhang M, Hu T, Ma T, Huang W and Wang Y:
Epigenetics and environmental health. Front Med. 18:571–596. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Dutta A, Venkataganesh H and Love PE:
Epigenetic regulation of T cell development. Int Rev Immunol.
42:82–90. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Mi T, Soerens AG, Alli S, Kang TG,
Vasandan AB, Wang Z, Vezys V, Kimura S, Iacobucci I, Baylin SB, et
al: Conserved epigenetic hallmarks of T cell aging during immunity
and malignancy. Nat Aging. 4:1053–1063. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Alves E, Taifour S, Chee J, Nowak AK,
Gaudieri S and Blancafort P: Reprogramming the anti-tumor immune
response via CRISPR genetic and epigenetic editing. Mol Ther
Methods Clin Dev. 21:592–606. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Zhong RQ, Tian H and Gao XJ: Epigenetic
regulation in tooth development. Zhonghua Kou Qiang Yi Xue Za Zhi.
56:288–293. 2021.(In Chinese). PubMed/NCBI
|
|
78
|
Tong J, Zhang W, Chen Y, Yuan Q, Qin NN
and Qu G: The emerging role of RNA modifications in the regulation
of antiviral innate immunity. Front Microbiol. 13:8456252022.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Li J, Wang Y, Shen W, Zhang Z, Su Z, Guo
X, Pei P, Hu L, Liu T, Yang K and Guo L: Mitochondria-modulating
liposomes reverse Radio-resistance for colorectal cancer. Adv Sci
(Weinh). 11:e24008452024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
An J, Guo R, Liu M, Hu H and Zhang H:
Multi-modal Ca2+ nanogenerator via reversing T cell exhaustion for
enhanced chemo-immunotherapy. J Control Release. 372:715–727. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Qian L, Zhang F, Yin M and Lei Q: Cancer
metabolism and dietary interventions. Cancer Biol Med. 19:163–174.
2021.PubMed/NCBI
|
|
82
|
Di Donna A, Masala S, Muto G, Marcia S,
Giordano F and Muto M: Metabolic bone diseases: Recommendations for
interventional radiology. Semin Musculoskelet Radiol. 28:641–650.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Liu H, Zeng L, Yang Y, Huang Z, Guo C,
Huang L, Niu X, Zhang C and Wang H: Bcl-3 regulates the function of
Th17 cells through raptor mediated glycolysis metabolism. Front
Immunol. 13:9297852022. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Jeong DI, Hao Q, Lee SY, Kim S, Karmakar
M, Chu S, Park M and Cho HJ: Cellulose nanocrystal-annealed
hydrogel system for local chemo-metabolic therapy of melanoma. J
Control Release. 377:324–338. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Zhao H and Li Y: Cancer metabolism and
intervention therapy. Mol Biomed. 2:52021. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Li Z, Li X, Ai S, Liu S and Guan W:
Glucose metabolism Intervention-facilitated nanomedicine therapy.
Int J Nanomedicine. 17:2707–2731. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Mathis BJ, Tanaka K and Hiramatsu Y:
Metabolically healthy obesity: Are interventions useful? Curr Obes
Rep. 12:36–60. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Pan X, Wang J, Zhang L, Li G and Huang B:
Metabolic plasticity of T cell fate decision. Chin Med J (Engl).
137:762–775. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Liu Y, Wu Z, Li Y, Chen Y, Zhao X, Wu M
and Xia Y: Metabolic reprogramming and interventions in
angiogenesis. J Adv Res. 70:323–338. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Chen B, Zhang Y, Dai S, Zhou P, Luo W,
Wang Z, Chen X, Cheng P, Zheng G and Ren J: Molecular
characteristics of primary pulmonary lymphoepithelioma-like
carcinoma based on integrated genomic analyses. Signal Transduct
Target Ther. 6:62021. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Wang H, Zhan Y, Luo J, Wang W and Fan S:
Unveiling immune resistance mechanisms in nasopharyngeal carcinoma
and emerging targets for antitumor immune response: Tertiary
lymphoid structures. J Transl Med. 23:382025. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Li JP, Wu CY, Chen MY, Liu SX, Yan SM,
Kang YF, Sun C, Grandis JR, Zeng MS and Zhong Q: PD-1+CXCR5-CD4+
Th-CXCL13 cell subset drives B cells into tertiary lymphoid
structures of nasopharyngeal carcinoma. J Immunother Cancer.
9:e0021012021. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Deng M, Liu X, Jiang Y, Luo R, Xu L, Zhang
X, Su J, Xu C and Hou Y: Tertiary lymphoid structures' pattern and
prognostic value in primary adenocarcinoma of jejunum and ileum.
World J Surg Oncol. 22:2612024. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Edmonds NL, Gradecki SE, Katyal P, Lynch
KT, Stowman AM, Gru AA, Engelhard VH, Slingluff CL Jr and Mauldin
IS: Tertiary lymphoid structures in desmoplastic melanoma have
increased lymphocyte density, lymphocyte proliferation, and immune
cross talk with tumor when compared to non-desmoplastic melanomas.
Oncoimmunology. 12:21644762023. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Huang Y, Xiao Z, Wu S, Zhang X, Wang J and
Huangfu X: Biochemical transformation and bioremediation of
thallium in the environment. Sci Total Environ. 953:1760282024.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Xiaoxu D, Min X and Chengcheng C: Immature
central tumor tertiary lymphoid structures are associated with
better prognosis in non-small cell lung cancer. BMC Pulm Med.
24:1552024. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wu S, Pan J, Pan Q, Zeng L, Liang R and Li
Y: Multi-omics profiling and experimental verification of tertiary
lymphoid structure-related genes: Molecular subgroups, immune
infiltration, and prognostic implications in lung adenocarcinoma.
Front Immunol. 15:14532202024. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Fujihara J and Nishimoto N:
Thallium-poisoner's poison: An overview and review of current
knowledge on the toxicological effects and mechanisms. Curr Res
Toxicol. 6:1001572024. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
MacFawn IP, Magnon G, Gorecki G, Kunning
S, Rashid R, Kaiza ME, Atiya H, Ruffin AT, Taylor S, Soong TR, et
al: The activity of tertiary lymphoid structures in high grade
serous ovarian cancer is governed by site, stroma, and cellular
interactions. Cancer Cell. 42:1864–1881.e5. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Wang Y, Lu J, Wu C, Fei F, Chu Z and Lu P:
Clinical markers predict the efficacy of several immune checkpoint
inhibitors in patients with non-small cell lung cancer in China.
Front Immunol. 14:12761072023. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Chen Z, Wang X, Jin Z, Li B, Jiang D, Wang
Y, Jiang M, Zhang D, Yuan P, Zhao Y, et al: Deep learning on
tertiary lymphoid structures in hematoxylin-eosin predicts cancer
prognosis and immunotherapy response. NPJ Precis Oncol. 8:732024.
View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Liu R, Huang X, Yang S, Du W, Chen X and
Li H: Discovery of an independent poor-prognosis subtype associated
with tertiary lymphoid structures in breast cancer. Front Immunol.
15:13645062024. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Teillaud JL, Houel A, Panouillot M,
Riffard C and Dieu-Nosjean MC: Tertiary lymphoid structures in
anticancer immunity. Nat Rev Cancer. 24:629–646. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Tang Z, Bai Y, Fang Q, Yuan Y, Zeng Q,
Chen S, Xu T, Chen J, Tan L, Wang C, et al: Spatial transcriptomics
reveals tryptophan metabolism restricting maturation of
intratumoral tertiary lymphoid structures. Cancer Cell.
43:1025–1044.e14. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Li H, Zhu SW, Zhou JJ, Chen DR, Liu J, Wu
ZZ, Wang WY, Zhang MJ and Sun ZJ: Tertiary Lymphoid Structure
Raises Survival and Immunotherapy in HPV-HNSCC. J Dent Res.
102:678–688. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Peyraud F, Guegan JP, Vanhersecke L,
Brunet M, Teyssonneau D, Palmieri LJ, Bessede A and Italiano A:
Tertiary lymphoid structures and cancer immunotherapy: From bench
to bedside. Med. 6:1005462025. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Xu W, Lu J, Tian X, Ye S, Wei S, Wang J,
Anwaier A, Qu Y, Liu W, Chang K, et al: Unveiling the impact of
tertiary lymphoid structures on immunotherapeutic responses of
clear cell renal cell carcinoma. MedComm (2020). 5:e4612024.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Yang YL, Zhou C, Chen Q, Shen SZ, Li JD,
Wang XL and Wang PR: YAP1/Piezo1 involve in the dynamic changes of
lymphatic vessels in UVR-induced photoaging progress to squamous
cell carcinoma. J Transl Med. 21:8202023. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Chelvanambi M, Fecek RJ, Taylor JL and
Storkus WJ: STING agonist-based treatment promotes vascular
normalization and tertiary lymphoid structure formation in the
therapeutic melanoma microenvironment. J Immunother Cancer.
9:e0019062021. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Vion R, Roulleaux-Dugage M, Flippot R,
Ouali K, Rouanne M, Clatot F, Sellars M, Champiat S, Chaput N,
Massard C and Danlos FX: Induction of tertiary lymphoid structures
in tumor microenvironment to improve anti-tumoral immune checkpoint
blockade efficacy. Eur J Cancer. 225:1155722025. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Zou Q, Liao K, Li G, Huang X, Zheng Y,
Yang G, Luo M, Xue EY, Lan C, Wang S, et al:
Photo-metallo-immunotherapy: Fabricating Chromium-based
nanocomposites to enhance CAR-T cell infiltration and cytotoxicity
against solid tumors. Adv Mater. 37:e24074252025. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Liu Y, Ye SY, He S, Chi DM, Wang XZ, Wen
YF, Ma D, Nie RC, Xiang P, Zhou Y, et al: Single-cell and spatial
transcriptome analyses reveal tertiary lymphoid structures linked
to tumour progression and immunotherapy response in nasopharyngeal
carcinoma. Nat Commun. 15:77132024. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Liu SX, Wu TW, Luo DH, Zhang LL, Zhou L,
Luo YL, Du WT, Huang TT, Jiang S, Zhang Z, et al:
Interferon-responsive HEVs drive tumor tertiary lymphoid structure
formation and predict immunotherapy response in nasopharyngeal
carcinoma. Cell Rep Med. 6:1022002025. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Yuan L, Jia GD, Lv XF, Xie SY, Guo SS, Lin
DF, Liu LT, Luo DH, Li YF, Deng SW, et al: Camrelizumab combined
with apatinib in patients with first-line platinum-resistant or
PD-1 inhibitor resistant recurrent/metastatic nasopharyngeal
carcinoma: A single-arm, phase 2 trial. Nat Commun. 14:48932023.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Ahmed MM, Gebriel MG, Morad EA, Saber IM,
Elwan A, Salah M, Fakhr AE, Shalaby AM and Alabiad MA: Expression
of immune checkpoint regulators, cytotoxic T-Lymphocyte Antigen-4,
and programmed Death-Ligand 1 in Epstein-Barr Virus-associated
nasopharyngeal carcinoma. Appl Immunohistochem Mol Morphol.
29:401–408. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Lim DW, Kao HF, Suteja L, Li CH, Quah HS,
Tan DS, Tan SH, Tan EH, Tan WL, Lee JN, et al: Clinical efficacy
and biomarker analysis of dual PD-1/CTLA-4 blockade in
recurrent/metastatic EBV-associated nasopharyngeal carcinoma. Nat
Commun. 14:27812023. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Nguyen K, Alsaati N, Le Coz C and Romberg
N: Genetic obstacles to developing and tolerizing human B cells.
WIREs Mech Dis. 14:e15542022. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Zhu Z, Ding R, Yu W, Liu Y, Zhou Z and Liu
CY: YAP/TEAD4/SP1-induced VISTA expression as a tumor
cell-intrinsic mechanism of immunosuppression in colorectal cancer.
Cell Death Differ. 32:911–925. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Chen Y, Sun J, Liu J, Wei Y, Wang X, Fang
H, Du H, Huang J, Li Q, Ren G, et al: Aldehyde dehydrogenase
2-mediated aldehyde metabolism promotes tumor immune evasion by
regulating the NOD/VISTA axis. J Immunother Cancer. 11:e0074872023.
View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Kim JY, Kim Y, Woo SY, Kim JO, Kim H, Son
SR, Jang DS and Choi JH: Hydnocarpin, a natural flavonolignan,
induces the ROS-Mediated apoptosis of ovarian cancer cells and
reprograms Tumor-Associated immune cells. Antioxidants (Basel).
14:8462025. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Jeong M and Collins N: Nutritional
modulation of antitumor immunity. Curr Opin Immunol. 87:1024222024.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Spina CS and Drake CG: Mechanisms of
immune modulation by radiation. Semin Radiat Oncol. 31:205–216.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Sun LL, Zhao LN, Sun J, Yuan HF, Wang YF,
Hou CY, Lv P, Zhang HH, Yang G, Zhang NN, et al: Inhibition of USP7
enhances CD8+ T cell activity in liver cancer by suppressing
PRDM1-mediated FGL1 upregulation. Acta Pharmacol Sin. 45:1686–1700.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Zahraeifard S, Xiao Z, So JY, Ahad A,
Montoya S, Park WY, Sornapudi T, Andohkow T, Read A, Kedei N, et
al: Loss of tumor suppressors promotes inflammatory tumor
microenvironment and enhances LAG3+T cell mediated immune
suppression. Nat Commun. 15:58732024. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Murphy EJ, Fehrenbach GW, Abidin IZ,
Buckley C, Montgomery T, Pogue R, Murray P, Major I and Rezoagli E:
Polysaccharides-naturally occurring immune modulators. Polymers
(Basel). 15:23732023. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Posner M: Curative application of
immunotherapy in untreated head and neck and nasopharynx cancer.
JAMA. 333:1584–1585. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Jaiswal AR, Liu AJ, Pudakalakatti S, Dutta
P, Jayaprakash P, Bartkowiak T, Ager CR, Wang ZQ, Reuben A, Cooper
ZA, et al: Melanoma evolves complete immunotherapy resistance
through the acquisition of a hypermetabolic phenotype. Cancer
Immunol Res. 8:1365–1380. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Bernardo M, Tolstykh T, Zhang YA, Bangari
DS, Cao H, Heyl KA, Lee JS, Malkova NV, Malley K, Marquez E, et al:
An experimental model of anti-PD-1 resistance exhibits activation
of TGFß and Notch pathways and is sensitive to local mRNA
immunotherapy. Oncoimmunology. 10:18812682021. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Zhou B, Gao Y, Zhang P and Chu Q: Acquired
resistance to immune checkpoint blockades: The underlying
mechanisms and potential strategies. Front Immunol. 12:6936092021.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Zhang W, Wang M, Ji C, Liu X, Gu B and
Dong T: Macrophage polarization in the tumor microenvironment:
Emerging roles and therapeutic potentials. Biomed Pharmacother.
177:1169302024. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Chen W, Bao L, Ren Q, Zhang Z, Yi L, Lei
W, Yang Z, Lu Y, You B, You Y and Gu M: SCARB1 in extracellular
vesicles promotes NPC metastasis by co-regulating M1 and M2
macrophage function. Cell Death Discov. 9:3232023. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Tang Q, Li L, Ge J, Wang D, Qu H, Wu J,
Wang Q, Peng Z, Mo Y, Wang Y, et al: m6A modification-dependent
upregulation of WNT2 facilitates M2-like macrophage polarization
and perpetuates malignant progression of nasopharyngeal carcinoma.
Oncogene. 44:2730–2745. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Boutilier AJ and Elsawa SF: Macrophage
polarization states in the tumor microenvironment. Int J Mol Sci.
22:69952021. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Saeed AF: Tumor-associated macrophages:
Polarization, immunoregulation, and immunotherapy. Cells.
14:7412025. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Zou Z, Lin H, Li M and Lin B:
Tumor-associated macrophage polarization in the inflammatory tumor
microenvironment. Front Oncol. 13:11031492023. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Sun Z, Xu Y, Shao B, Dang P, Hu S, Sun H,
Chen C, Wang C, Liu J, Liu Y and Hu J: Exosomal circPOLQ promotes
macrophage M2 polarization via activating IL-10/STAT3 axis in a
colorectal cancer model. J Immunother Cancer. 12:e0084912024.
View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Wei B, Pan J, Yuan R, Shao B, Wang Y, Guo
X and Zhou S: Polarization of Tumor-Associated macrophages by
Nanoparticle-loaded escherichia coli combined with immunogenic cell
death for cancer immunotherapy. Nano Lett. 21:4231–4240. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Zhang J, Tang K, Yang Y, Yang D and Fan W:
Advanced nanoprobe strategies for imaging macrophage polarization
in cancer immunology. Research (Wash DC). 8:06222025.PubMed/NCBI
|
|
139
|
Nagatani Y, Funakoshi Y, Suto H, Imamura
Y, Toyoda M, Kiyota N, Yamashita K and Minami H: Immunosuppressive
effects and mechanisms of three myeloid-derived suppressor cells
subsets including monocytic-myeloid-derived suppressor cells,
granulocytic-myeloid-derived suppressor cells, and
immature-myeloid-derived suppressor cells. J Cancer Res Ther.
17:1093–1100. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Bhardwaj V and Ansell SM: Modulation of
T-cell function by myeloid-derived suppressor cells in
hematological malignancies. Front Cell Dev Biol. 11:11293432023.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Zhang W, Li X, Jiang M, Ji C, Chen G,
Zhang Q, Liu P, Zhang R, Ren X, Yu W and Yu J: SOCS3
deficiency-dependent autophagy repression promotes the survival of
early-stage myeloid-derived suppressor cells in breast cancer by
activating the Wnt/mTOR pathway. J Leukoc Biol. 113:445–460. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Wong SW, McCarroll J, Hsu K, Geczy CL and
Tedla N: Intranasal delivery of recombinant S100A8 protein delays
lung cancer growth by remodeling the lung immune microenvironment.
Front Immunol. 13:8263912022. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Zang J, Yang Y, Zheng X, Yang Y, Zhao Y,
Miao Z, Zhang T, Gu J, Liu Y, Yin W, et al: Dynamic tagging to
drive arginine nano-assembly to metabolically potentiate immune
checkpoint blockade therapy. Biomaterials. 292:1219382023.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Zhao Z, Qin J, Qian Y, Huang C, Liu X,
Wang N, Li L, Chao Y, Tan B, Zhang N, et al: FFAR2 expressing
myeloid-derived suppressor cells drive cancer immunoevasion. J
Hematol Oncol. 17:92024. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Bertrand BP, Heim CE, Koepsell SA and
Kielian T: Elucidating granulocytic myeloid-derived suppressor cell
heterogeneity during Staphylococcus aureus biofilm infection. J
Leukoc Biol. 115:620–632. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Genova C, Dellepiane C, Carrega P,
Sommariva S, Ferlazzo G, Pronzato P, Gangemi R, Filaci G, Coco S
and Croce M: Therapeutic implications of tumor microenvironment in
lung cancer: Focus on immune checkpoint blockade. Front Immunol.
12:7994552021. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Qin G, Liu S, Liu J, Hu H, Yang L, Zhao Q,
Li C, Zhang B and Zhang Y: Overcoming resistance to immunotherapy
by targeting GPR84 in myeloid-derived suppressor cells. Signal
Transduct Target Ther. 8:1642023. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Yamamoto S, Kato M, Takeyama Y, Azuma Y,
Yukimatsu N, Hirayama Y, Otoshi T, Yamasaki T, Fujioka M, Gi M, et
al: Irradiation plus myeloid-derived suppressor cell-targeted
therapy for overcoming treatment resistance in immunologically cold
urothelial carcinoma. Br J Cancer. 128:2197–2205. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Liu J, Piranlioglu R, Ye F, Shu K, Lei T
and Nakashima H: Immunosuppressive cells in oncolytic virotherapy
for glioma: Challenges and solutions. Front Cell Infect Microbiol.
13:11410342023. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Martín-Portugués M, Esteban-Sampedro J, de
Mendoza C, Ruiz-Irastorza G, Royuela A, Puente AO, de la
Cruz-Echeandía M, Blanco-Valencia XP, Soriano V and Moreno-Torres
V: Hospitalisations with Cryoglobulin-related diseases in spain
over 25 Years. Liver Int. 45:e701952025. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Bhatt K, Nukovic A, Colombani T and
Bencherif SA: Biomaterial-assisted local oxygenation safeguards the
prostimulatory phenotype and functions of human dendritic cells in
hypoxia. Front Immunol. 14:12783972023. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Kawahara K, Mukai T, Iseki M, Nagasu A,
Nagasu H, Akagi T, Tsuji S, Hiramatsu-Asano S, Ueki Y, Ishihara K,
et al: SH3BP2 deficiency ameliorates murine systemic lupus
erythematosus. Int J Mol Sci. 22:41692021. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Schneble D, El-Gazzar A, Kargarpour Z,
Kramer M, Metekol S, Stoshikj S and Idzko M: Cell-type-specific
role of P2Y2 receptor in HDM-driven model of allergic airway
inflammation. Front Immunol. 14:12090972023. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Wen Y, Rudemiller NP, Zhang J, Lu X, Ren
J, Privratsky JR, Griffiths R, Zhang JJ, Hammer GE and Crowley SD:
C-C motif chemokine receptor 7 exacerbates hypertension through
effects on T lymphocyte trafficking. Hypertension. 75:869–876.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Greene TT, Jo Y, Chiale C, Macal M, Fang
Z, Khatri FS, Codrington AL, Kazane KR, Akbulut E, Swaminathan S,
et al: Metabolic deficiencies underlie reduced plasmacytoid
dendritic cell IFN-I production following viral infection. Nat
Commun. 16:14602025. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Kremer KN, Khammash HA, Miranda AM, Rutt
LN, Twardy SM, Anton PE, Campbell ML, Garza-Ortiz C, Orlicky DJ,
Pelanda R, et al: Liver sinusoidal endothelial cells regulate the
balance between hepatic immunosuppression and immunosurveillance.
Front Immunol. 15:14977882024. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Smilovic D, Rietsche M, Drakew A, Vuksic M
and Deller T: Constitutive tumor necrosis factor (TNF)-deficiency
causes a reduction in spine density in mouse dentate granule cells
accompanied by homeostatic adaptations of spine head size. J Comp
Neurol. 530:656–669. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Nickles E, Xia R, Sun R and Schwarz H:
Methods for generating the CD137L-DC-EBV–VAX anti-cancer vaccine.
Methods Cell Biol. 183:187–202. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Tischer-Zimmermann S, Bonifacius A,
Santamorena MM, Mausberg P, Stoll S, Döring M, Kalinke U, Blasczyk
R, Maecker-Kolhoff B and Eiz-Vesper B: Reinforcement of
cell-mediated immunity driven by tumor-associated Epstein-Barr
virus (EBV)-specific T cells during targeted B-cell therapy with
rituximab. Front Immunol. 14:8789532023. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Huang J, Harris E and Lorch J: Vaccination
as a therapeutic strategy for Nasopharyngeal carcinoma. Oral Oncol.
135:1060832022. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Nickles E, Dharmadhikari B, Yating L,
Walsh RJ, Koh LP, Poon M, Tan LK, Wang LZ, Ang Y, Asokumaran Y, et
al: Dendritic cell therapy with CD137L-DC-EBV–VAX in locally
recurrent or metastatic nasopharyngeal carcinoma is safe and
confers clinical benefit. Cancer Immunol Immunother. 71:1531–1543.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Liao L, Liu Y, Li X, Jiang Z, Jiang Z and
Yao J: Dual-regulated biomimetic nanocomposites for promoted tumor
photodynamic immunotherapy. ACS Appl Mater Interfaces.
17:20919–20931. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Yang H, Xiong Z, Heng X, Niu X, Wang Y,
Yao L, Sun L, Liu Z and Chen H: Click-Chemistry-mediated cell
membrane glycopolymer engineering to potentiate dendritic cell
vaccines. Angew Chem Int Ed Engl. 63:e2023157822024. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Najafi S and Mortezaee K: Advances in
dendritic cell vaccination therapy of cancer. Biomed Pharmacother.
164:1149542023. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
van Willigen WW, Bloemendal M,
Boers-Sonderen MJ, de Groot JWB, Koornstra RHT, van der Veldt AAM,
Haanen JBAG, Boudewijns S, Schreibelt G, Gerritsen WR, et al:
Response and survival of metastatic melanoma patients treated with
immune checkpoint inhibition for recurrent disease on adjuvant
dendritic cell vaccination. Oncoimmunology. 9:17388142020.
View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Wang D, Liu R, Ding Y, Pei Q, Sun T, Shao
X, Jiang M, Liu J, Huang J, Yan Z, et al: Whole-body vibration
protects against chronic high-altitude hypoxic bone loss by
regulating the nitric oxide/HIF-1α axis in osteoblasts. FASEB J.
39:e703852025. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Wu Q, You L, Nepovimova E, Heger Z, Wu W,
Kuca K and Adam V: Hypoxia-inducible factors: Master regulators of
hypoxic tumor immune escape. J Hematol Oncol. 15:772022. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Cheng X, Wang P, Lyu H, Lee Y, Yoon J and
Dong H: Targeting the hypoxia signaling pathway with nanomedicine
to reverse immunotherapy resistance. Cancer Drug Resist.
8:462025.PubMed/NCBI
|
|
169
|
Schulz MC, Kopf M and Gekle M: Crosstalk
with renal proximal tubule cells drives acidosis-induced
inflammatory response and dedifferentiation of fibroblasts via
p38-singaling. Cell Commun Signal. 22:1482024. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Li K, Niu L, Zhang X, Li T, Zhou X, Wang
L, Han J and Wang Z: NEAT1 is a therapeutic target for reversing
T-cell exhaustion in bladder cancer. J Immunother Cancer.
13:e0137332025. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Chen Y, Gao J, Ma M, Wang K, Liu F, Yang
F, Zou X, Cheng Z and Wu D: The potential role of CMC1 as an
immunometabolic checkpoint in T cell immunity. Oncoimmunology.
13:23449052024. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Fowler EA, Farias Amorim C, Mostacada K,
Yan A, Amorim Sacramento L, Stanco RA, Hales ED, Varkey A, Zong W,
Wu GD, et al: Neutrophil-mediated hypoxia drives pathogenic CD8+ T
cell responses in cutaneous leishmaniasis. J Clin Invest.
134:e1779922024. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Knopf P, Stowbur D, Hoffmann SHL, Hermann
N, Maurer A, Bucher V, Poxleitner M, Tako B, Sonanini D,
Krishnamachary B, et al: Acidosis-mediated increase in
IFN-γ-induced PD-L1 expression on cancer cells as an immune escape
mechanism in solid tumors. Mol Cancer. 22:2072023. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Babl N, Decking SM, Voll F, Althammer M,
Sala-Hojman A, Ferretti R, Korf C, Schmidl C, Schmidleithner L,
Nerb B, et al: MCT4 blockade increases the efficacy of immune
checkpoint blockade. J Immunother Cancer. 11:e0073492023.
View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Nicolini A and Ferrari P: Involvement of
tumor immune microenvironment metabolic reprogramming in colorectal
cancer progression, immune escape, and response to immunotherapy.
Front Immunol. 15:13537872024. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Huang Y, Qin G, Cui T, Zhao C, Ren J and
Qu X: A bimetallic nanoplatform for STING activation and CRISPR/Cas
mediated depletion of the methionine transporter in cancer cells
restores anti-tumor immune responses. Nat Commun. 14:46472023.
View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Madden MZ, Ye X, Chi C, Fisher EL, Wolf
MM, Needle GA, Bader JE, Patterson AR, Reinfeld BI, Landis MD, et
al: Differential effects of glutamine inhibition strategies on
antitumor CD8 T Cells. J Immunol. 211:563–575. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Xiao J, Liu T, Zeng F and Zhang J: New
insights into T cell metabolism in liver cancer: From mechanism to
therapy. Cell Death Discov. 11:1182025. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Xu C, Jiang H, Feng LJ, Jiang MZ, Wang YL
and Liu SJ: Christensenella minuta interacts with multiple gut
bacteria. Front Microbiol. 15:13010732024. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Wang R, Liu Z, Fan Z and Zhan H: Lipid
metabolism reprogramming of CD8+ T cell and therapeutic
implications in cancer. Cancer Lett. 567:2162672023. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Wang J, Jia W, Zhou X, Ma Z, Liu J and Lan
P: CBX4 suppresses CD8+ T cell antitumor immunity by reprogramming
glycolytic metabolism. Theranostics. 14:3793–3809. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Silva C, Andrade N, Rodrigues I, Ferreira
AC, Soares ML and Martel F: The pro-proliferative effect of
interferon-γ in breast cancer cell lines is dependent on
stimulation of ASCT2-mediated glutamine cellular uptake. Life Sci.
286:1200542021. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Yang Y, Chen Q, Fan S, Lu Y, Huang Q, Liu
X and Peng X: Glutamine sustains energy metabolism and alleviates
liver injury in burn sepsis by promoting the assembly of
mitochondrial HSP60-HSP10 complex via SIRT4 dependent protein
deacetylation. Redox Rep. 29:23123202024. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Zhang Y, Hu Z, Zhang J, Ren C and Wang Y:
Dual-target inhibitors of indoleamine 2, 3 dioxygenase 1 (Ido1): A
promising direction in cancer immunotherapy. Eur J Med Chem.
238:1145242022. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Fujiwara Y, Kato S, Nesline MK, Conroy JM,
DePietro P, Pabla S and Kurzrock R: Indoleamine 2,3-dioxygenase
(IDO) inhibitors and cancer immunotherapy. Cancer Treat Rev.
110:1024612022. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Kim M and Tomek P: Tryptophan: A rheostat
of cancer immune escape mediated by immunosuppressive enzymes IDO1
and TDO. Front Immunol. 12:6360812021. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Wang M, Liu Y, Li Y, Lu T, Wang M, Cheng
Z, Chen L, Wen T, Pan M and Hu G: Tumor Microenvironment-responsive
nanoparticles enhance IDO1 blockade immunotherapy by remodeling
metabolic immunosuppression. Adv Sci (Weinh). 12:e24058452025.
View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Hu C, Liu Y, Cao W, Li N, Gao S, Wang Z
and Gu F: Efficacy and mechanism of a biomimetic nanosystem
carrying doxorubicin and an IDO inhibitor for treatment of advanced
Triple-negative breast cancer. Int J Nanomedicine. 19:507–526.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Li J, Yang H, Zhang L, Zhang S and Dai Y:
Metabolic reprogramming and interventions in endometrial carcinoma.
Biomed Pharmacother. 161:1145262023. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Qiu MZ, Wang C, Wu Z, Zhao Q, Zhao Z,
Huang CY, Wu W, Yang LQ, Zhou ZW, Zheng Y, et al: Dynamic
single-cell mapping unveils Epstein-Barr virus-imprinted T-cell
exhaustion and on-treatment response. Signal Transduct Target Ther.
8:3702023. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Pogorelyy MV, Kirk AM, Adhikari S,
Minervina AA, Sundararaman B, Vegesana K, Brice DC, Scott ZB; SJTRC
Study Team, ; Thomas PG: TIRTL-seq: Deep, quantitative, and
affordable paired TCR repertoire sequencing. bioRxiv. Oct
31–2024.doi: 10.1101/2024.09.16.613345.
|
|
192
|
Zhang Q and Xu M: EBV-induced T-cell
responses in EBV-specific and nonspecific cancers. Front Immunol.
14:12509462023. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Young J, Asaoka M, Ghasemi F, Chida K, Roy
AM, Yan L, Hakamada K and Takabe K: The American Joint Committee on
Cancer (AJCC) Breast Cancer Staging, Eighth Edition, is more
Reflective of Cancer Biology than the Seventh Edition. Ann Surg
Oncol. 32:3268–3277. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Ando M: Induced pluripotent stem
cell-derived rejuvenated cytotoxic T lymphocyte therapy for
Epstein-Barr virus-associated lymphomas: Application to clinical
practice. Rinsho Ketsueki. 63:1310–1315. 2022.(In Japanese).
PubMed/NCBI
|
|
195
|
Ning XH, Liu J, Hu L, Lu HY, Liu Q and
Wang DH: Effects of NOX4 on radiosensitivity of nasopharyngeal
carcinoma cells by activating the PI3K/AKT pathway. Zhonghua Er Bi
Yan Hou Tou Jing Wai Ke Za Zhi. 55:514–519. 2020.(In Chinese).
PubMed/NCBI
|
|
196
|
Hang W, Guo HM, Wu QL, Yan H, Liu G and
Gao M: MicroRNA-203a-3p affects the biological characteristics of
nasopharyngeal carcinoma by targeting vascular endothelial growth
factor-C. J Physiol Pharmacol. Jan 21–2021.(Epub ahead of print).
doi: 10.26402/jpp.2021.4.12. PubMed/NCBI
|
|
197
|
Mouquet H: Humoral immunity in HIV-1
post-treatment controllers. Curr Opin HIV AIDS. 20:80–85. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
198
|
La Civita E, Zannella C, Brusa S, Romano
P, Schettino E, Salemi F, Carrano R, Gentile L, Punziano A, Lagnese
G, et al: BNT162b2 Elicited an Efficient Cell-Mediated Response
against SARS-CoV-2 in kidney transplant recipients and common
variable immunodeficiency patients. Viruses. 15:16592023.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Satou A and Nakamura S: EBV-positive
B-cell lymphomas and lymphoproliferative disorders: Review from the
perspective of immune escape and immunodeficiency. Cancer Med.
10:6777–6785. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Gou Q, Yan B, Duan Y, Guo Y, Qian J, Shi J
and Hou Y: Ubiquitination of CD47 Regulates innate Anti-Tumor
immune response. Adv Sci (Weinh). 12:e24122052025. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Wang Y, Peng Z, Wang Y, Yang Y, Fan R, Gao
K, Zhang H, Xie Z and Jiang W: Immune Microenvironment change and
involvement of circular RNAs in TIL cells of recurrent
nasopharyngeal carcinoma. Front Cell Dev Biol. 9:7222242021.
View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Senft D: Programming immune escape. Nat
Rev Cancer. 24:2942024. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Liang Y, Yang H, Li Q, Zhao P, Li H, Zhang
Y, Cai W, Ma X and Duan Y: Novel biomimetic dual-mode nanodroplets
as ultrasound contrast agents with potential ability of precise
detection and photothermal ablation of tumors. Cancer Chemother
Pharmacol. 86:405–418. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Wang Y, Guo L, Fan G, Han Y, Zhang Q, Ren
L, Zhang H, Wang G, Zhang X, Huang T, et al: Impact of
corticosteroids on initiation and half-year durability of humoral
response in COVID-19 survivors. Chin Med J Pulm Crit Care Med.
2:48–55. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Wu X, Zhang J, Deng Z, Sun X, Zhang Y,
Zhang C, Wang J, Yu X and Yang G: Bacteria-based biohybrids for
remodeling adenosine-mediated immunosuppression to boost
radiotherapy-triggered antitumor immune response. Biomaterials.
316:1230002025. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Florêncio KGD, Edson EA, Fernandes KSDS,
Luiz JPM, Pinto FDCL, Pessoa ODL, Cunha FQ, Machado-Neto JA and
Wilke DV: Chromomycin A5 induces bona fide immunogenic cell death
in melanoma. Front Immunol. 13:9417572022. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Ding Q, Tang W, Li X, Ding Y, Chen X, Cao
W, Wang X, Mo W, Su Z, Zhang Q and Guo H: Mitochondrial-targeted
brequinar liposome boosted mitochondrial-related ferroptosis for
promoting checkpoint blockade immunotherapy in bladder cancer. J
Control Release. 363:221–234. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Wang C, Zai W, Zhao K, Li Y, Shi B, Wu M,
Zhou X, Kozlowski M, Zhang X, Fang Z and Yuan Z: Potential role of
liver-resident CD3+ macrophages in HBV clearance in a mouse
hepatitis B model. JHEP Rep. 7:1013232025. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Gu Y, Lian Y, Zheng Q, Huang Z, Gu L, Bi
Y, Li J and Huang Y, Wu Y, Chen L and Huang Y: Association among
cytokine profiles of innate and adaptive immune responses and
clinical-virological features in untreated patients with chronic
hepatitis B. BMC Infect Dis. 20:5092020. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Kunst C, Schmid S, Michalski M, Tümen D,
Buttenschön J, Müller M and Gülow K: The influence of gut
microbiota on oxidative stress and the immune system. Biomedicines.
11:13882023. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Avila-Ponce de León U, Vázquez-Jiménez A,
Matadamas-Guzman M, Pelayo R and Resendis-Antonio O:
Transcriptional and microenvironmental landscape of macrophage
transition in cancer: A boolean analysis. Front Immunol.
12:6428422021. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
El Saftawy E, Aboulhoda BE, Alghamdi MA,
Abd Elkhalek MA and AlHariry NS: Heterogeneity of modulatory immune
microenvironment in bladder cancer. Tissue Cell. 93:1026792025.
View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Huang S: Tuning the rheostat of immune
gene translation. Stress Biol. 3:72023. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Chen Y, Qiu Y, Chen M, Huang L, Lin X, Qiu
X, Wei Y and Gao L: Transition from secukinumab to adalimumab in
COVID-19-induced psoriasis flare-up treatment: A case report. Clin
Case Rep. 11:e80472023. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Fang S, Jiang M, Jiao J, Zhao H, Liu D,
Gao D, Wang T, Yang Z and Yuan H: Unraveling the
ROS-Inflammation-immune balance: A new perspective on aging and
disease. Aging Dis. 17:305–325. 2025.PubMed/NCBI
|
|
216
|
Jiang XN, Yu BH, Yan WH, Lee J, Zhou XY
and Li XQ: Epstein-Barr virus-positive diffuse large B-cell
lymphoma features disrupted antigen capture/presentation and
hijacked T-cell suppression. Oncoimmunology. 9:16833462020.
View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Huo S, Luo Y, Deng R, Liu X, Wang J, Wang
L, Zhang B, Wang F, Lu J and Li X: EBV-EBNA1 constructs an
immunosuppressive microenvironment for nasopharyngeal carcinoma by
promoting the chemoattraction of Treg cells. J Immunother Cancer.
8:e0015882020. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Kang K, Lin X, Chen P, Liu H, Liu F, Xiong
W, Li G, Yi M, Li X, Wang H and Xiang B: T cell exhaustion in human
cancers. Biochim Biophys Acta Rev Cancer. 1879:1891622024.
View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Wetzel TJ, Erfan SC, Figueroa LD, Wheeler
LM and Ananieva EA: Crosstalk between arginine, glutamine, and the
branched chain amino acid metabolism in the tumor microenvironment.
Front Oncol. 13:11865392023. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Sharma S, Bhatnagar R and Gaur D:
Complement evasion strategies of human pathogenic bacteria. Indian
J Microbiol. 60:283–296. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Wang H, Tsung A, Mishra L and Huang H:
Regulatory T cell: A double-edged sword from
metabolic-dysfunction-associated steatohepatitis to hepatocellular
carcinoma. EBioMedicine. 101:1050312024. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Gong L, Luo J, Zhang Y, Yang Y, Li S, Fang
X, Zhang B, Huang J, Chow LK, Chung D, et al: Nasopharyngeal
carcinoma cells promote regulatory T cell development and
suppressive activity via CD70-CD27 interaction. Nat Commun.
14:19122023. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Liu X, Shen H, Zhang L, Huang W, Zhang S
and Zhang B: Immunotherapy for recurrent or metastatic
nasopharyngeal carcinoma. NPJ Precis Oncol. 8:1012024. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Mai H, Li L, Xin X, Jiang Z, Tang Y, Huang
J, Lei Y, Chen L, Dong T and Zhong X: Prediction of immunotherapy
response in nasopharyngeal carcinoma: A comparative study using
MRI-based radiomics signature and programmed cell death ligand 1
expression score. Eur Radiol. 35:4403–4414. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Yu Z, Wang Q, Wang Z, Liu S, Xia T, Duan
C, Liu Y, Ding X, Chen S, Yu T, et al: Lachnoclostridium intestinal
flora is associated with immunotherapy efficacy in nasopharyngeal
carcinoma. Head Neck. 47:269–281. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Piao Y, Yang Y, Wu S and Han L:
Toripalimab plus cetuximab combined with radiotherapy in a locally
advanced platinum-based chemotherapy-insensitive nasopharyngeal
carcinoma patient: A case report. Front Oncol. 14:13832502024.
View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Peng Y, Yan H, Mei W, Zhang P and Zeng C:
Combining radiotherapy with immunotherapy in cervical cancer: Where
do we stand and where are we going? Curr Treat Options Oncol.
24:1378–1391. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Mukherjee D, Romano E, Walshaw R, Zeef
LAH, Banyard A, Kitcatt SJ, Cheadle EJ, Tuomela K, Pendharkar S,
Al-Deka A, et al: Reprogramming the immunosuppressive tumor
microenvironment results in successful clearance of tumors
resistant to radiation therapy and anti-PD-1/PD-L1. Oncoimmunology.
12:22230942023. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Wang J, Gai J, Zhang T, Niu N, Qi H,
Thomas DL II, Li K, Xia T, Rodriguez C, Parkinson R, et al:
Neoadjuvant radioimmunotherapy in pancreatic cancer enhances
effector T cell infiltration and shortens their distances to tumor
cells. Sci Adv. 10:eadk18272024. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Saylor PJ, Kozin SV, Matsui A, Goldberg
SI, Aoki S, Shigeta K, Mamessier E, Smith MR, Michaelson MD, Lee RJ
and Duda DG: The radiopharmaceutical radium-223 has
immunomodulatory effects in patients and facilitates
anti-programmed death receptor-1 therapy in murine models of bone
metastatic prostate cancer. Radiother Oncol. 192:1100912024.
View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Tan JW and Hu JR: Fever with atypical
lymphocytosis: Pearls and pitfalls in Epstein-Barr virus serology.
BMJ Case Rep. 16:e2500812023. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Zapata L, Caravagna G, Williams MJ,
Lakatos E, AbdulJabbar K, Werner B, Chowell D, James C, Gourmet L,
Milite S, et al: Immune selection determines tumor antigenicity and
influences response to checkpoint inhibitors. Nat Genet.
55:451–460. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Ren Y, Okazaki T, Ngamsnae P, Hashimoto H,
Ikeda R, Honkura Y, Suzuki J and Izumi SI: Anatomy and function of
the lymphatic vessels in the parietal pleura and their plasticity
under inflammation in mice. Microvasc Res. 148:1045462023.
View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Yeo YH, Liang J, Lauzon M, Luu M,
Noureddin M, Ayoub W, Kuo A, Sankar K, Gong J, Hendifar A, et al:
Immunotherapy and transarterial radioembolization combination
treatment for advanced hepatocellular carcinoma. Am J
Gastroenterol. 118:2201–2211. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Zheng Y, Wang X and Huang M: Metabolic
regulation of CD8+ T cells: From mechanism to therapy. Antioxid
Redox Signal. 37:1234–1253. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Koda S, Hu J, Ju X, Sun G, Shao S, Tang
RX, Zheng KY and Yan J: The role of glutamate receptors in the
regulation of the tumor microenvironment. Front Immunol.
14:11238412023. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Sun H, Li S, Wang Q, Luo C, Zhong L, Wan
G, Li Z, Zhao G, Bu X, Zeng M and Feng G: Formyl peptide enhances
cancer immunotherapy by activating antitumoral neutrophils, and T
cells. Biomed Pharmacother. 175:1166702024. View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Wu H, Zhao X, Hochrein SM, Eckstein M,
Gubert GF, Knöpper K, Mansilla AM, Öner A, Doucet-Ladevèze R,
Schmitz W, et al: Mitochondrial dysfunction promotes the transition
of precursor to terminally exhausted T cells through
HIF-1α-mediated glycolytic reprogramming. Nat Commun. 14:68582023.
View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Jiang C, Feng D, Zhang Y, Yang K, Hu X and
Xie Q: SCAT8/miR-125b-5p axis triggers malignant progression of
nasopharyngeal carcinoma through SCARB1. BMC Mol Cell Biol.
24:152023. View Article : Google Scholar : PubMed/NCBI
|
|
240
|
Xu H, Liu J, Zhang Y, Zhou Y, Zhang L,
Kang J, Ning C, He Z and Song S: KIF23, under regulation by
androgen receptor, contributes to nasopharyngeal carcinoma
deterioration by activating the Wnt/β-catenin signaling pathway.
Funct Integr Genomics. 23:1162023. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Qiu Q, Yang X, Li X, Ren Y and Huang N:
Viral IL-10 can promote the proliferation, migration and invasion
of nasopharyngeal carcinoma cells and inhibit their apoptosis.
Discov Oncol. 16:1992025. View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Chen P, Liu B, Xia X, Huang P and Zhao J:
Current progress in immunotherapy of nasopharyngeal carcinoma. Am J
Cancer Res. 13:1140–1147. 2023.PubMed/NCBI
|
|
243
|
De Boer RJ and Yates AJ: Modeling T cell
fate. Annu Rev Immunol. 41:513–532. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Feng X, Meng M, Li H, Gao Y, Song W, Di R,
Li Z, Zhang X and Zhang M: T-cell dysfunction in natural
killer/T-cell lymphoma. Oncoimmunology. 12:22125322023. View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Liu H, Tang L, Li Y, Xie W, Zhang L, Tang
H, Xiao T, Yang H, Gu W, Wang H and Chen P: Nasopharyngeal
carcinoma: Current views on the tumor microenvironment's impact on
drug resistance and clinical outcomes. Mol Cancer. 23:202024.
View Article : Google Scholar : PubMed/NCBI
|