Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumor
associated with Epstein-Barr virus (EBV) infection, and its
distinct tumor immune microenvironment (TIME) makes it an ideal
model for studying tumor immune editing. EBV carries out a key role
in NPC pathogenesis and modulates the TIME (1). During NPC development, EBV infection
drives multiple immune evasion mechanisms, for example, tumor cells
can upregulate coinhibitory receptors to suppress IFN-γ production
by CD8+ tumor-infiltrating lymphocytes (TILs), thereby enhancing
immunosuppressive capacity (2).
Moreover, latent and lytic EBV genes, such as latent membrane
protein 1 (LMP1) and EB nuclear antigen 1 (EBNA1), have been shown
to contribute markedly to tumor cell proliferation, survival and
immune evasion (3,4).
T cell fate in NPC immune editing is orchestrated by
a variety of factors, including aberrant EBV antigen presentation,
T cell metabolic exhaustion and dynamic remodeling of tertiary
lymphoid structures (TLSs) within the tumor microenvironment (TME).
Tumor cells infected with EBV can escape immune surveillance by
inducing metabolic exhaustion in T cells, a process associated with
metabolic reprogramming in the TIME (5). Single-cell RNA sequencing analyses
have revealed the existence of diverse cellular subsets in NPC,
which either promote or suppress antitumor immunity through
intricate cellular interactions (6–8).
In terms of microenvironmental remodeling, NPC is
characterized by abundant lymphocyte infiltration, indicating its
potential immunogenicity. However, this also underscores the
presence of sophisticated immunosuppressive mechanisms.
Tumor-associated fibroblasts and tumor-associated macrophages
(TAMs) modulate T cell function through cytokine secretion and
extracellular matrix (ECM) remodeling, thereby reshaping immune
responses (9,10). In this context, therapeutic
approaches targeting the TIME, such as immune checkpoint inhibitors
(ICIs) and chimeric antigen receptor-T cell therapy, have shown
promising potential but still require further refinement to enhance
clinical efficacy (11–13).
In summary, immune editing in NPC is a multifaceted
process involving EBV antigen presentation, T cell metabolic
exhaustion and microenvironmental remodeling. These interconnected
factors collectively determine T cell fate and carry out key roles
in antitumor immune responses. The present review systematically
explores the key molecular checkpoints, cellular interactions and
structural dynamics that influence T cell fate decisions during NPC
immune editing, highlighting their potential clinical implications.
Rather than acting as isolated mechanisms, EBV antigen
presentation, T cell metabolic exhaustion and TLS remodeling
constitute a temporally ordered and mechanistically interconnected
immune editing process in NPC. Persistent viral antigen exposure
initiates chronic T cell activation, which progressively drives
metabolic dysfunction and ultimately reshapes local immune
architecture. The present review therefore emphasizes not only
individual mechanisms, but also their causal relationships and
dynamic progression during EBV-associated immune editing.
EBV latent infection and aberrant antigen
presentation
Expression profile of EBV latent
proteins
In NPC, the expression patterns of EBV latent
proteins hold notable clinical relevance. Key EBV-encoded latent
proteins, including LMP1 and latent membrane protein 2A (LMP2A), as
well as EBNA1, carry out key roles in the pathogenesis and
progression of NPC. LMP1 is considered the primary oncogenic driver
of EBV, capable of activating multiple intracellular signaling
pathways to promote cellular proliferation, survival and immune
evasion (4). LMP2A contributes to
the maintenance of EBV latency by inhibiting B cell differentiation
and proliferation, thereby enabling the virus to evade host immune
surveillance (14). Studies have
demonstrated that the expression levels of these latent proteins
are markedly elevated in NPC tissues compared with normal tissues,
underscoring their potential as therapeutic targets (15–18).
Beyond modulating tumor cell behavior, LMP1 and
LMP2A also influence host immune responses by altering the
expression of major histocompatibility complex (MHC) molecules.
LMP1 has been shown to downregulate MHC class I molecule
expression, thereby reducing T cell-mediated tumor recognition
(19). Similarly, LMP2A is
implicated in the suppression of MHC class II molecule expression,
hindering T cell activation and proliferation (20). Through interference with the antigen
presentation machinery, these latent proteins enable EBV-infected
cells to escape immune detection, facilitating persistent viral
infection and tumorigenesis.
Additionally, EBV-encoded microRNAs (miRNAs) are key
modulators of host antigen presentation pathways. These viral
miRNAs can suppress host antiviral responses by targeting specific
host genes. For example, EBV-miR-BART17-3p has been shown to impair
host immunity by targeting DDX3X, thereby promoting EBV persistence
and progression (21). The
expression of EBV miRNAs is associated with the development of
EBV-related malignancies, highlighting their functional importance
in shaping the TIME.
Collectively, the expression profile of EBV latent
proteins not only serves as a key factor in understanding NPC
pathogenesis but also represents a promising foundation for the
development of targeted therapeutic strategies. Further exploration
of the functional roles of these proteins and their interactions
with host immunity may provide novel directions and targets for
clinical interventions.
Mechanisms of defective antigen
processing and presentation
Defective antigen processing and presentation in NPC
cells involve multiple molecular and cellular disruptions. Among
them, the transporter associated with antigen processing (TAP)
plays a key role in MHC class I-mediated antigen presentation. TAP
translocates peptides derived from intracellular protein
degradation into the endoplasmic reticulum, where they bind to MHC
class I molecules and are subsequently presented on the cell
surface for recognition by CD8+ T cells. Studies have
shown that TAP dysfunction in NPC cells frequently results in
reduced MHC class I expression, thereby compromising antigen
presentation and enabling tumor cells to evade immune detection.
For example, TAP deficiency has been associated with impaired
recognition and cytotoxicity by CD8+ T cells,
contributing to tumor progression (22,23).
EBV latent proteins also directly interfere with
antigen processing and presentation pathways. LMP1 and LMP2 are
highly expressed in NPC cells and disrupt the expression of MHC
class I molecules and the overall antigen processing machinery.
LMP1, for example, activates the NF-κB signaling pathway to
suppress components of the antigen presentation system,
destabilizing MHC class I molecules and promoting immune evasion by
tumor cells (24). These findings
suggest that EBV infection is not only a causal factor in
oncogenesis but also a key mechanism through which tumor cells
escape immune surveillance.
Dysfunction in the cross-presentation pathway is
another key aspect of defective antigen presentation in NPC.
Cross-presentation refers to the process by which
antigen-presenting cells (APCs) internalize exogenous antigens and
present them via the endogenous pathway to CD8+ T cells.
In NPC, the immunosuppressive TME and impaired APC function lead to
decreased cross-presentation efficiency. Studies in EBV-positive
NPC models have shown that APCs exhibit reduced cross-presentation
of tumor antigens, resulting in inadequate T cell activation and
further enhancing immune evasion (25–27).
Therefore, impairments in the cross-presentation pathway are
associated with tumor immune escape and should be considered a
vital component in the study of NPC immunobiology.
In summary, the mechanisms of defective antigen
processing and presentation in NPC include TAP dysfunction, EBV
protein-induced suppression of antigen presentation and impaired
cross-presentation (Fig. 1). These
mechanisms synergistically enable NPC cells to effectively escape
immune surveillance, thereby facilitating tumor progression.
Understanding these defects provides key insights into the
immunoevasive strategies of NPC and may inform the development of
novel immunotherapeutic approaches targeting these pathways.
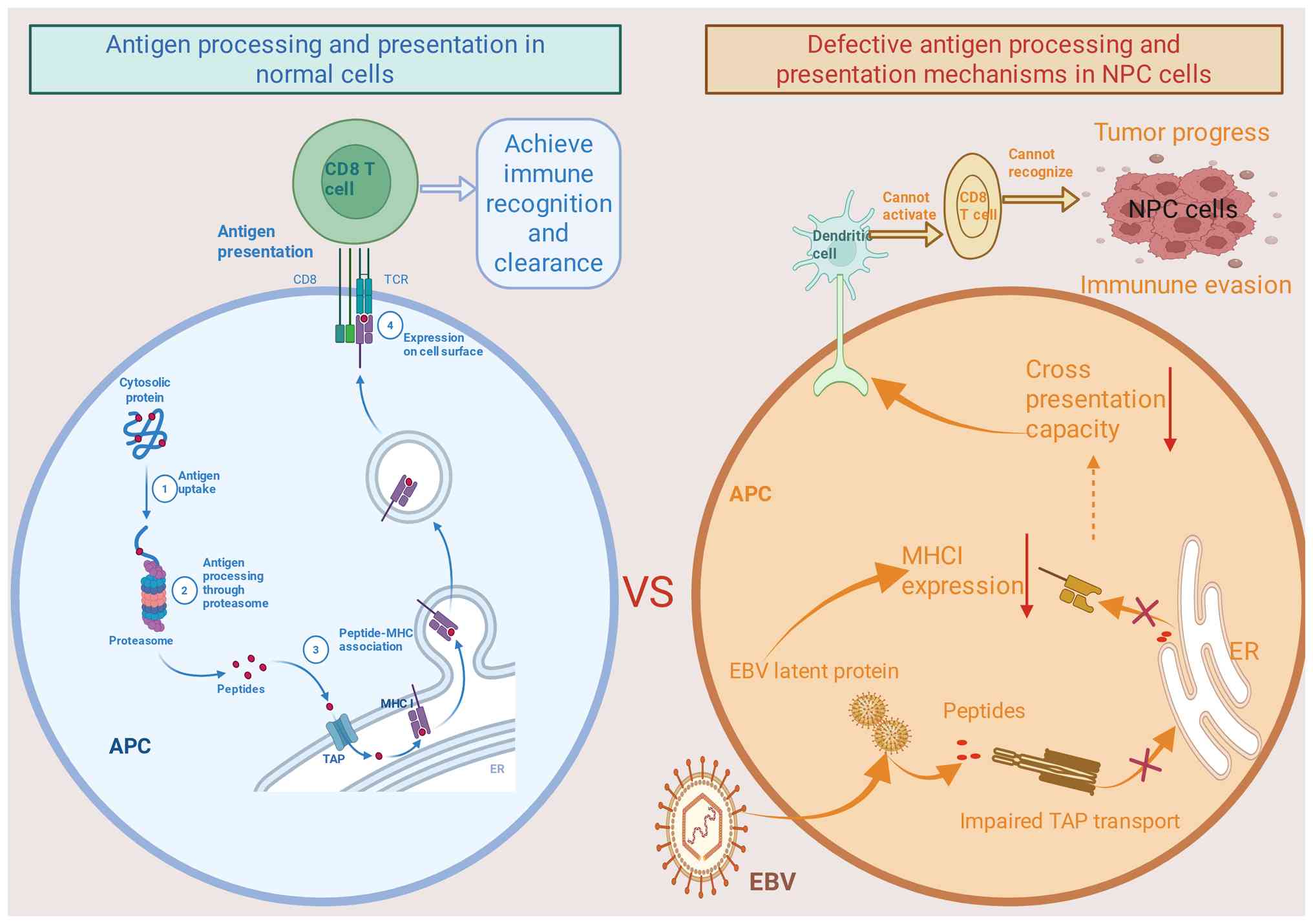 | Figure 1.Schematic of defective antigen
processing and presentation mechanisms in NPC cells. In normal
cells, antigen processing occurs through the degradation of
endogenous proteins via the proteasome. The resulting peptides are
then transported to the ER, where they bind to MHC-I molecules and
are subsequently displayed on the cell surface. This process
ensures the recognition and clearance of abnormal cells by CD8+ T
cells, thereby achieving immune surveillance and elimination. By
contrast, in NPC cells, EBV latent infection leads to defects in
antigen presentation. The expression of EBV latent proteins impairs
the function of the TAP transporter, preventing efficient peptide
transport to the ER and thereby inhibiting MHC-I expression. These
defects reduce cross-presentation capacity, ultimately resulting in
the inability of T cells to recognize and activate NPC cells,
leading to immune evasion and promoting tumor progression. MHC-I,
major histocompatibility complex class I; TAP, transporter
associated with antigen processing; ER, endoplasmic reticulum; APC,
antigen-presenting cell; EBV, Epstein-Barr virus; NPC,
nasopharyngeal carcinoma; CD8+ T cell, CD8-positive T
lymphocyte. |
Clinical relevance of defective
antigen presentation
Antigen presentation plays a key role in
orchestrating immune responses, and its impairment is associated
with the pathogenesis and progression of malignancies such as
melanoma, lung cancer, colorectal cancer and nasopharyngeal
carcinoma (28–30). In NPC, defects in antigen
presentation are associated with disease staging and progression. A
decline in antigen-presenting efficiency allows tumor cells to
escape immune surveillance, particularly in the context of
EBV-associated antigens. Studies suggest that patients with
early-stage NPC often retain relatively intact antigen-presenting
function, while in patients at the advanced-stage, particularly
during metastasis, this ability is markedly diminished (31,32).
Tumor cells may downregulate MHC molecule expression to evade
recognition by T cells (33,34).
These findings underscore the potential association between
defective antigen presentation and NPC progression, emphasizing the
need to pay clinical attention to these mechanisms for improved
disease assessment and prognostic evaluation.
Moreover, the efficiency of antigen presentation has
been shown to associate with patient prognosis. Patients with
stronger antigen-presenting capacity generally exhibit more
favorable clinical outcomes. In NPC, the responsiveness of T cell
subsets is tightly associated with prognosis (35). Several studies have reported that
patients with efficient antigen presentation tend to have improved
responses to immunotherapies and demonstrate notably prolonged
survival (36–38). This indicates the potential value of
strategies aimed at enhancing antigen presentation to improve
clinical outcomes in NPC.
To address the impairment of antigen presentation,
several therapeutic approaches have been proposed. For example,
ICIs [such as anti-programmed cell death (PD)-1/PD-Ligand 1 (PD-L1)
antibodies] have shown efficacy in restoring T cell-mediated
antitumor responses in multiple clinical trials (39). Additionally, targeting the TME to
enhance the function of APCs has emerged as a viable strategy.
Small molecules or biologics that improve APC functionality can
enhance T cell recognition and killing of tumor cells, offering new
therapeutic opportunities for patients with NPC (40).
In summary, defective antigen presentation has
clinical implications for NPC. Understanding the underlying
mechanisms not only facilitates improved diagnosis and staging but
also supports the development of immunotherapeutic strategies that
restore antigen presentation and strengthen antitumor immunity.
Importantly, persistent EBV antigen presentation represents more
than an initiating immune event. In the context of incomplete viral
clearance, continuous presentation of EBV-derived antigens sustains
prolonged T cell receptor engagement and chronic immune activation.
This persistent activation pressure establishes the immunological
conditions under which downstream metabolic stress and functional
exhaustion of T cells are likely to emerge. However, it should be
noted that the majority of these findings are derived from in
vitro systems or EBV-transformed cell lines, which may not
fully recapitulate the complex TIME of NPC in vivo (41,42).
Spatiotemporal dynamics of T cell
activation
Molecular switches in initial T cell
activation
The initiation of T cell activation relies heavily
on the strength of T cell receptor (TCR) signaling and the
synergistic engagement of costimulatory molecules. Upon recognition
of peptide-MHC complexes by the TCR, a cascade of intracellular
signaling events is triggered, regulating T cell fate and function.
Protein tyrosine kinases and protein tyrosine phosphatases play
opposing but coordinated roles in modulating these signals. Among
them, Src homology region 2 domain-containing phosphatase (SHP) 1
and SHP2, two key protein tyrosine phosphatases, have attracted
increasing attention (43–45). SHP1 generally acts as a negative
regulator of T cell signaling, whereas SHP2 exhibits more complex,
context-dependent roles (43).
Concurrently, costimulatory molecules such as CD28 and 4-1BB
augment TCR signaling and promote T cell proliferation and
differentiation. Thus, the interplay between TCR signal strength
and costimulation is important for effective T cell activation and
function.
In the context of EBV infection, the activation
threshold of virus-specific T cells plays a key role. The
activation of EBV-specific T cells depends not only on TCR-antigen
interactions but also on the cytokine milieu. Studies have shown
that chronic EBV infection can elevate the activation threshold of
T cells, leading to functional exhaustion and impaired antiviral
responses (46,47). Furthermore, the clonal expansion of
virus-specific T cells is directly influenced by their activation
state. The quality and quantity of these T cell clones determine
the effectiveness of immune responses against EBV. Investigating
the activation thresholds of EBV-specific T cell clones can
therefore provide valuable insights into immune escape mechanisms
in EBV-associated diseases.
The cytokine microenvironment also exerts a profound
influence on T cell polarization. Cytokines such as IL-2, IL-4 and
IL-6 drive the differentiation of naïve T cells into various
effector subsets including T helper (Th) 1, Th2 and Th17 cells. In
EBV-infected settings, dysregulation of the cytokine milieu may
skew T cell polarization, impairing effector function and
durability (48). For example,
excessive IL-6 production has been associated with T cell
exhaustion and diminished antiviral capacity. Modulating the
cytokine environment may therefore represent a viable strategy to
enhance T cell functionality, particularly in the treatment of
EBV-associated malignancies. Therefore, while persistent EBV
antigen exposure is widely proposed to contribute to chronic T cell
activation, direct causal evidence associating antigen persistence
to specific T cell fate outcomes in patients with NPC remains
limited.
Spatiotemporal distribution of
TILs
TILs exhibit distinct spatial and temporal
distribution patterns within the TME, with considerable
heterogeneity in T cell subsets across different tumor regions.
Studies in various cancer types, including breast and colorectal
cancer, have revealed differences in the density and composition of
T cells between the tumor core and invasive margins (49,50).
For example, CD8+ T cells are often more abundant at the
tumor periphery than in the tumor core in breast cancer (51). In colorectal cancer, the
distribution of CD4+ and CD8+ T cells is
closely associated with tumor grade and clinical prognosis, with
higher-grade tumors typically showing increased CD8+ T
cell infiltration (52).
This regional variation reflects the complexity of
the TME and may contribute to immune evasion. Tumor cells can
secrete immunosuppressive factors and alter metabolic conditions,
thereby inhibiting T cell activity and infiltration. Moreover, the
immune infiltration pattern is influenced by molecular features of
the tumor, such as PD-L1 expression, which has been positively
associated with CD8+ T cell density and may predict
responses to ICIs (53).
Understanding the spatial organization of T cell subsets within the
tumor provides key insights into TME characteristics and supports
the development of personalized immunotherapy strategies.
T cell trafficking and positioning are important for
mounting effective antitumor immune responses and the chemokine
network carries out a central role in directing T cell migration.
Chemokines secreted by various cell types bind to specific
receptors on T cells, guiding their homing to tumor sites. The
expression profiles of chemokines vary considerably across
different TMEs, directly affecting T cell infiltration patterns. In
breast and colorectal cancer, chemokines such as CXCL9 and CXCL10
associate positively with CD8+ T cell infiltration,
functioning through CXCR3 receptor signaling to facilitate T cell
recruitment (54). Additionally,
other cells within the TME, including TAMs and endothelial cells,
contribute to T cell recruitment and activation through the
secretion of cytokines such as IL-6 and IL-8 (52).
Beyond migration, chemokines also influence T cell
effector function. For example, inhibitory signals within the TME,
such as PD-L1 expression, can modulate T cell sensitivity to
chemokines, thereby affecting their motility and survival (55–57).
These findings suggest that targeting chemokine signaling may
improve T cell localization and functionality, offering potential
avenues for enhancing immunotherapeutic efficacy.
The spatial positioning of T cells within the tumor
directly impacts their cytotoxic potential. TILs that localize at
the invasive margin or in direct contact with tumor cells are
generally more effective in exerting antitumor activity. By
contrast, T cells trapped in poorly vascularized or hypoxic tumor
cores often display diminished functionality. For example, in lung
cancer, a higher density of CD8+ T cells at the tumor
margin associates with improved survival, whereas sparse
CD8+ infiltration in the tumor core is associated with
poor prognosis (51). Furthermore,
immunosuppressive components of the TME, such as TAMs, can secrete
inhibitory mediators that impair T cell positioning and
function.
Optimizing T cell localization within tumors is thus
a key objective in enhancing the efficacy of immunotherapy.
Strategies such as adoptive T cell therapy and cancer vaccines aim
to improve T cell infiltration and activity within tumors. A deeper
understanding of the dynamic interactions between T cells and the
TME will inform the rational design of novel immunotherapies.
Mechanisms of activation-induced cell
death (AICD)
The Fas/FasL pathway plays a key role in AICD, which
is essential for the contraction phase of immune responses. Fas
(CD95), a death receptor expressed on the surface of several cell
types, binds to its ligand FasL to trigger downstream signaling
cascades that activate caspases and induce programmed cell death
(58). In T cells, the Fas/FasL
pathway not only regulates immune homeostasis but also influences T
cell fate within the TME. Tumor cells can downregulate Fas
expression to escape FasL-mediated killing by cytotoxic T cells,
thus promoting their survival (59). Dysregulation of this pathway is
implicated in the development of autoimmunity and cancer,
suggesting that modulation of Fas/FasL signaling may offer
therapeutic benefit in immuno-oncology.
PD-1 is another key immune checkpoint that regulates
T cell activation and survival. Expressed primarily on activated T
cells, PD-1 binds to its ligands (PD-L1/PD-L2) to attenuate T cell
responses and promote immune tolerance. The PD-1 pathway has been
shown to contribute to AICD, especially in settings of chronic
infection and cancer, where prolonged antigen exposure leads to T
cell exhaustion (60). Activation
of PD-1 signaling not only suppresses effector functions but also
upregulates pro-apoptotic genes, thereby accelerating T cell
attrition (61). Therapeutic
blockade of PD-1/PD-L1 interactions has emerged as an effective
strategy in cancer immunotherapy by restoring T cell function and
enhancing antitumor immunity (62,63).
Mitochondrial dysfunction is another hallmark of
exhausted T cells in the TME. Persistent antigen stimulation,
coupled with nutrient deprivation and oxidative stress, leads to
impaired mitochondrial metabolism and diminished T cell
functionality. Studies have demonstrated that exhausted T cells
exhibit reduced mitochondrial membrane potential and ATP
production, along with elevated reactive oxygen species (ROS)
levels (64). Excess levels of ROS
induce oxidative damage and activate apoptotic pathways,
exacerbating T cell exhaustion (65). Interventions that restore
mitochondrial function and metabolic fitness may therefore
rejuvenate T cell responses. For example, pharmacological agents
that enhance mitochondrial bioenergetics have shown potential in
improving T cell-mediated antitumor activity.
Together, these mechanisms highlight the
vulnerability of T cells under chronic stimulation and within
immunosuppressive TMEs. AICD involves multiple mechanisms, such as
Fas/FasL, PD-1/PD-L1 and ROS accumulation, leading to T cell
dysfunction and death, thereby allowing tumor cells to evade immune
clearance (Fig. 2). A comprehensive
understanding of AICD pathways is key for developing strategies to
preserve T cell functionality and improve outcomes in
immunotherapy.
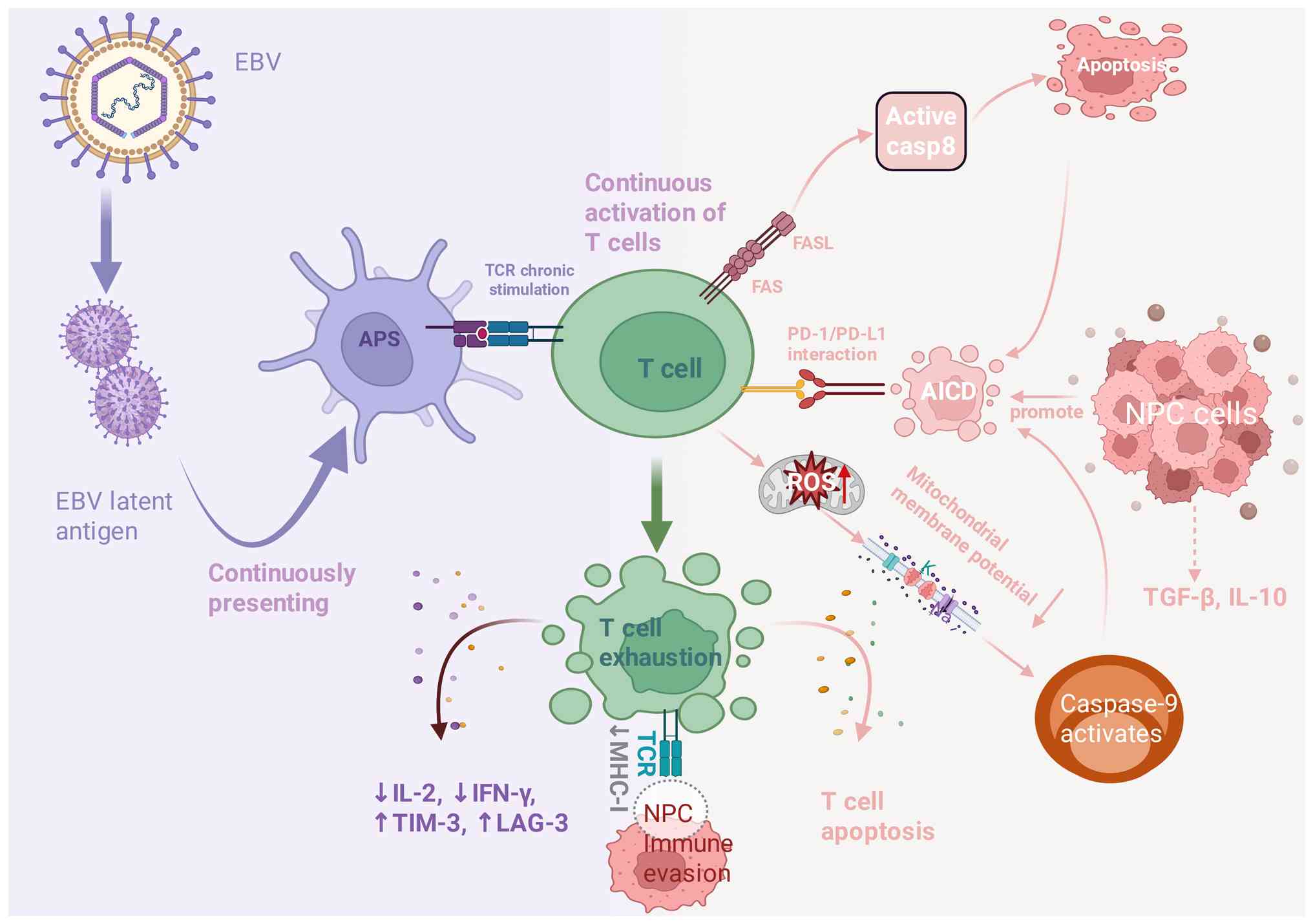 | Figure 2.Schematic of AICD in T cells in NPC.
EBV latent antigens are continuously presented by APCs, leading to
sustained activation and exhaustion of T cells. Chronic TCR
activation triggers immunosuppressive signals, resulting in T cell
dysfunction (T cell exhaustion) and reduced antitumor efficacy.
Activation of the PD-1/PD-L1 pathway, accumulation of ROS and
changes in mitochondrial membrane potential promote AICD. These
processes further enhance immune evasion by NPC cells, which
secrete immunosuppressive factors such as TGF-β and IL-10,
inhibiting T cell function and promoting tumor growth. EBV,
Epstein-Barr virus; APS, antigen-presenting cell; TCR, T cell
receptor; AICD, activation-induced cell death; PD-1/PD-L1,
programmed cell death protein 1/programmed cell death ligand 1;
ROS, reactive oxygen species; NPC, nasopharyngeal carcinoma. |
Molecular basis of T cell metabolic
exhaustion
Reprogramming of energy
metabolism
Chronic antigen-driven activation is increasingly
recognized as a primary upstream driver of metabolic reprogramming
and exhaustion in TILs. Within the TME, exhausted T cells undergo
profound metabolic reprogramming, particularly in glycolysis and
oxidative phosphorylation (OXPHOS). These cells often exhibit a
glycolysis-dependent metabolic profile, relying less on
mitochondrial OXPHOS. This metabolic shift allows T cells to
maintain ATP production under hypoxic conditions, sustaining their
basic survival and minimal functionality. However, due to nutrient
competition between tumor and immune cells, essential substrates
become scarce in the TME, forcing T cells to adopt glycolysis as
their predominant energy source. Exhausted T cells commonly produce
elevated levels of lactate, while their mitochondrial OXPHOS
capacity is markedly diminished. This reprogramming not only
reduces energy production but also impairs T cell proliferation and
promotes apoptotic susceptibility. Key signaling pathways,
including mTOR and AMPK, are involved in regulating this glycolytic
switch, ultimately contributing to impaired T cell effector
function and antitumor activity (66,67).
Mitochondria play a central role in T cell
metabolism, particularly in maintaining viability and effector
function. In exhausted T cells, mitochondrial dysfunction is
frequently observed, characterized by reduced membrane potential
and impaired OXPHOS activity. Mitochondrial deficits contribute to
energy insufficiency and facilitate progression to exhaustion.
Moreover, these T cells accumulate high levels of ROS, which not
only impair mitochondrial integrity but also disrupt intracellular
signaling cascades. This metabolic dysfunction negatively affects
cytokine production and clonal expansion, further weakening the
antitumor immune response. Enhancing mitochondrial bioenergetics
has been shown to restore T cell function; studies indicate that
interventions targeting mitochondrial metabolism can improve T cell
activity and increase antitumor immunity (64,68).
Moreover, it remains challenging to disentangle whether metabolic
dysfunction is a primary driver of T cell exhaustion or a
downstream consequence of prolonged activation and inhibitory
signaling.
Amino acid metabolism is also essential for T cell
survival and function, particularly under the nutrient-limited
conditions of the TME. Tumor cells can deplete specific amino
acids, such as glutamine and arginine, thereby impairing T cell
proliferation and cytokine secretion. Amino acid deprivation
compromises metabolic fitness and alters T cell fate decisions.
Furthermore, abnormal amino acid metabolism can affect epigenetic
modifications, leading to transcriptional reprogramming and reduced
immune competence. Strategies that replenish or modulate amino acid
availability may help restore T cell function, offering novel
avenues for improving the efficacy of cancer immunotherapy,
especially when combined with conventional treatments (69,70).
Importantly, the majority of studies examining T cell metabolic
exhaustion rely on static or endpoint measurements, which may not
adequately capture the dynamic and reversible nature of metabolic
states during disease progression.
Epigenetic regulatory networks
DNA methylation is a key epigenetic modification
that influences gene expression by adding methyl groups to cytosine
residues, typically within CpG islands. Recent studies have
revealed that T cell exhaustion is closely associated with aberrant
DNA methylation patterns (71,72).
Chronic antigen stimulation in infections or cancer can induce
exhaustion in CD8+ T cells, accompanied by increased
methylation of key regulatory genes such as PD-1, cytotoxic T
lymphocyte-associated protein 4 (CTLA-4) and T cell immunoglobulin
and mucin-domain containing-3 (TIM-3). Hypermethylation of promoter
regions suppresses the expression of these genes, leading to
reduced effector function (73).
Moreover, the extent of DNA methylation in exhausted
T cells associates with impaired proliferation, survival and memory
formation. In some cases, methylation-driven upregulation of
inhibitory receptors forms a negative feedback loop that deepens
exhaustion and further weakens antitumor immunity (74). Reversing these epigenetic changes,
using DNA methylation inhibitors or epigenetic editing tools, has
emerged as a promising strategy to restore T cell
functionality.
Histone modifications are another key layer of
epigenetic regulation. Specific histone marks, such as H3K27me3
(associated with gene repression) and H3K4me3 (associated with gene
activation), maintain transcriptional balance. In exhausted T
cells, this balance is often disrupted. Downregulation of key
transcription factors such as Basic Leucine Zipper ATF-Like
Transcription Factor(BATF)leads to altered histone modification
patterns at loci associated with exhaustion, impairing the gene
expression necessary for effector function (75). Upregulation of histone deacetylases
results in hypoacetylation of histones, while overactivation of
histone methyltransferases such as EZH2 promotes repressive
chromatin states, both contributing to T cell dysfunction (76). Thus, targeting histone-modifying
enzymes presents another opportunity for epigenetic reprogramming
in exhausted T cells.
Non-coding RNAs have emerged as important regulators
of gene expression and metabolic reprogramming in T cells. Long
non-coding RNAs, in particular, can modulate the expression of
transcription factors and metabolic enzymes by binding to
regulatory DNA elements or protein complexes (77). During exhaustion, the expression of
various long non-coding RNAs is altered, and several are implicated
in controlling glycolytic and OXPHOS-related genes. These
non-coding RNAs influence T cell viability, cytokine production and
cell fate decisions (78).
In summary, epigenetic regulation plays a pivotal
role in the development and maintenance of T cell exhaustion
(Fig. 3). Understanding the
mechanisms involved may provide insight into potential therapeutic
targets for reactivating exhausted T cells and enhancing
immunotherapeutic outcomes.
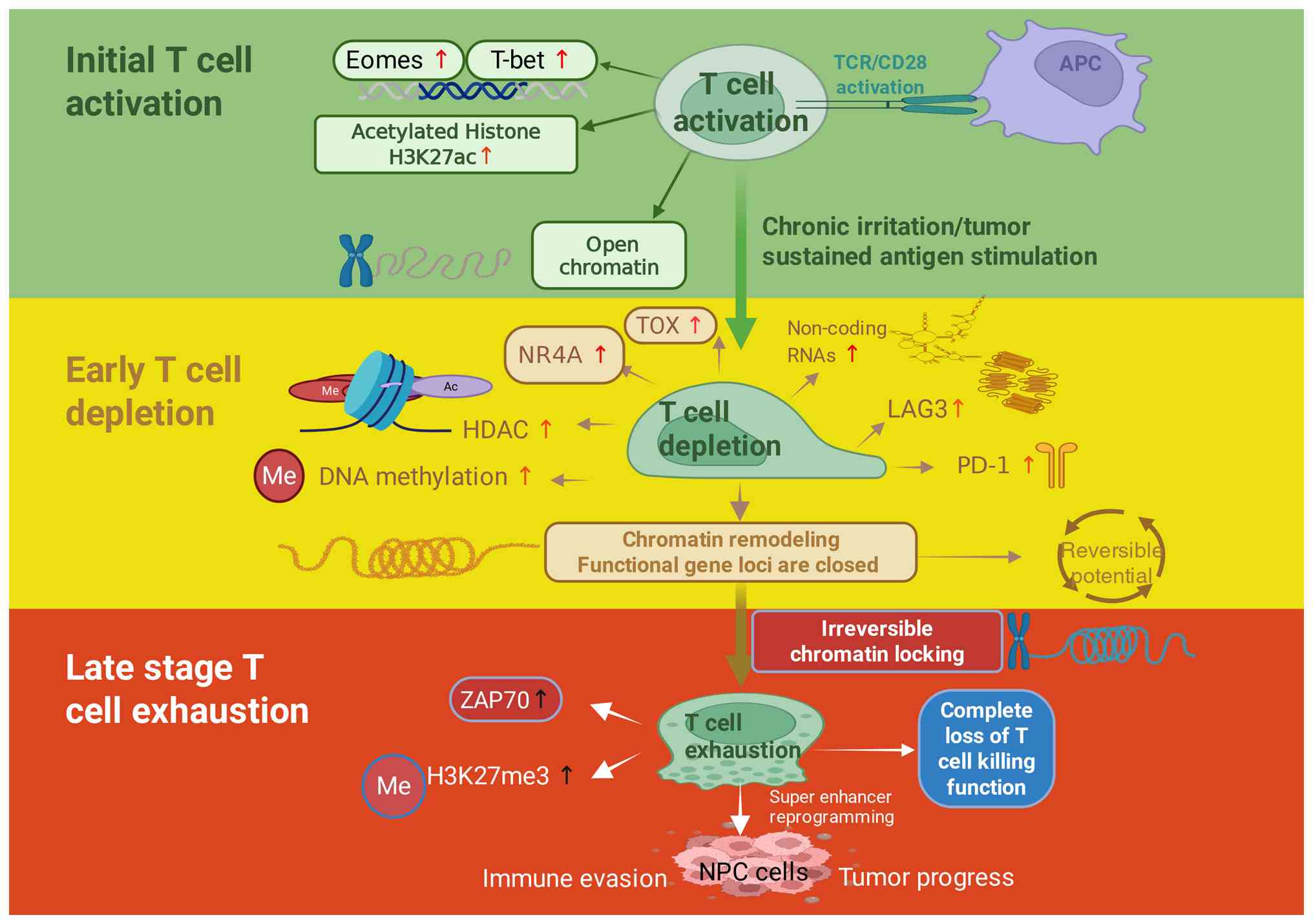 | Figure 3.Schematic of the epigenetic
regulatory network in the initiation and progression of T cell
exhaustion. T cells undergo distinct stages in response to
continuous antigen stimulation, including initial activation, early
depletion and late exhaustion, with accompanying epigenetic
regulatory changes. In the initial activation phase, T cells are
activated through TCR/CD28 and stimulated by APCs, leading to
chromatin opening and upregulation of transcription factors such as
Eomes and T-bet. Chronic stimulation thereafter drives T cells into
early depletion, characterized by increased expression of NR4A, TOX
and non-coding RNAs, along with DNA methylation and histone
modifications that result in the closure of functional gene loci.
Finally, during the late exhaustion stage, chromatin becomes
irreversibly locked, with increased H3K27me3, leading to a complete
loss of T cell killing function and contributing to tumor
progression and immune evasion. TCR, T cell receptor; CD28,
co-stimulatory receptor on T cells; APC, antigen-presenting cell;
Eomes, eomesodermin; T-bet, T-box transcription factor; H3K27ac,
acetylation of histone H3 on lysine 27; TOX, thymocyte
selection-associated high mobility group box; NR4A, nuclear
receptor subfamily 4 group A; LAG3, lymphocyte-activation gene 3;
PD-1, programmed cell death protein 1; HDAC, histone deacetylase;
me, methylation; Ac, acetylation; ZAP70, zeta-chain-associated
protein kinase 70; H3K27me3, trimethylation of histone H3 on lysine
27; NPC, nasopharyngeal carcinoma; Super enhancer, a cluster of
enhancers with the potential to drive gene expression at a high
level. |
Metabolic intervention strategies
Glycolysis inhibitors have garnered increasing
attention as potential tools to reverse T cell exhaustion (79,80).
Tumor cells rely heavily on glycolysis for energy production and
biosynthesis, while T cells in the TME often experience metabolic
exhaustion and impaired function (81). In chronic infections and cancer,
elevated glycolytic activity can hinder T cell proliferation and
effector cytokine secretion (82).
Therefore, targeting glycolysis to alleviate metabolic stress in T
cells represents a promising therapeutic strategy.
Several studies have demonstrated that glycolysis
inhibitors can enhance T cell function by reducing lactate
accumulation and mitigating extracellular acidification (83,84).
This metabolic rebalancing facilitates improved T cell
proliferation and cytokine production (85). Furthermore, glycolysis inhibition
may improve energy efficiency, shifting T cells toward more
sustainable mitochondrial metabolism and enhancing their cytotoxic
activity. These findings support the clinical potential of
glycolytic modulation, especially when used in combination with
immunotherapy to improve patient outcomes.
However, challenges remain, such as ensuring drug
selectivity and minimizing resistance (86). Future research should focus on
optimizing glycolysis inhibitor regimens to maximize T cell
recovery while minimizing off-target effects.
Mitochondria-targeted drugs offer another promising
avenue for enhancing T cell function. As central regulators of
energy metabolism and apoptosis, mitochondria are key for T cell
survival. In exhausted T cells, mitochondrial dysfunction severely
limits antitumor responses (87).
Agents such as MitoTEMPO, a mitochondria-targeted antioxidant, have
been shown to improve mitochondrial function, reduce ROS levels and
restore T cell energy metabolism (88). These interventions can promote T
cell proliferation and effector function, supporting their use in
immunotherapy.
Despite their promise, mitochondria-targeted
therapies face challenges related to delivery, tissue distribution
and toxicity. Continued development of safer, more effective
compounds is necessary. Notably, combining these drugs with immune
checkpoint blockade may yield synergistic benefits, improving both
metabolic fitness and antitumor efficacy. Metabolic reprogramming
represents an emerging paradigm in cancer therapy. Tumor cells
often evade immune detection by altering their metabolism, which
contributes to T cell exhaustion and dysfunction (82). By reprogramming T cell metabolism,
researchers aim to reinvigorate immune responses against
tumors.
Evidence suggests that combining metabolic
reprogramming with immunotherapy enhances treatment efficacy. For
example, interventions that restore T cell energy production and
reduce TME immunosuppression improve cytokine production,
proliferation and cytotoxicity. These effects can also potentiate
the efficacy of ICIs, offering synergistic therapeutic outcomes
(89).
Nevertheless, challenges remain in selecting optimal
metabolic targets and integrating interventions with existing
treatment protocols. Future research should aim to unravel the
mechanistic basis of metabolic reprogramming and identify effective
combinations with immunotherapy, ultimately offering more
personalized and effective cancer treatment strategies.
Dynamic remodeling of TLSs
Composition and functional features of
TLSs
In the TME of NPC, TLSs represent organized
aggregates of immune cells typically found at the invasive margins
and within the tumor core. From a temporal and mechanistic
perspective, TLS remodeling emerges as a downstream consequence of
sustained immune activation and T cell dysfunction during
EBV-associated immune editing. Chronic inflammation, together with
metabolically exhausted T cells, reshape local cytokine milieus and
affect stromal cell activation and antigen-presenting niches,
thereby driving alterations in TLS organization, composition and
functionality (90).
Histologically, TLSs resemble secondary lymphoid organs, comprising
densely clustered B and T lymphocytes. Increasing evidence suggests
that the presence of TLSs associates positively with clinical
prognosis in NPC (91,92). Their characteristic features include
high levels of lymphocytic infiltration, mature B cell follicles
and active T cell responses (93).
In various cancer types, the maturity, spatial distribution and
cellular heterogeneity of TLSs are associated with patient survival
and antitumor immune responses. Notably, in NPC, TLSs are often
accompanied by the presence of high endothelial venules (HEVs),
which play a key role in lymphocyte recruitment and TLS formation
(94).
Within NPC-associated TLSs, B cell follicles and T
cell zones exhibit a distinct spatial organization. B cells are
typically concentrated within the follicular core, whereas T cells
are predominantly distributed in the surrounding areas, forming a
functional immunological interface. This spatial arrangement
facilitates coordinated interactions between immune cell subsets.
For example, T cells can activate B cells via cytokine secretion,
thereby enhancing antigen-specific immune responses (94). T cells within TLSs often display a
highly activated phenotype, supporting tumor-specific immunity. The
structured interaction between B and T cells not only boosts immune
efficiency but may also establish local immunological memory,
contributing to improved clinical outcomes in patients with NPC
(95). Notably, evidence regarding
the functional role of TLSs in NPC is largely extrapolated from
other tumor types, and their immunological impact may vary
substantially depending on maturation status and cellular
composition.
HEVs play a key role in the formation and
maintenance of TLSs within the TME. These specialized vessels
enable efficient transmigration of lymphocytes from the bloodstream
into tumor tissues. Studies have demonstrated a positive
association between HEV density and TLS presence in NPC, suggesting
that HEVs facilitate immune cell infiltration and contribute to
effective antitumor immunity (93,96).
Furthermore, HEVs regulate immune cell trafficking by expressing
specific chemokines that attract both T and B cells, thereby
enhancing the immunological activity of TLSs. As such, HEVs
represent not only structural components of TLSs but also potential
therapeutic targets in immuno-oncology (94). In turn, remodeled TLSs may further
modulate antigen presentation efficiency and T cell activation
thresholds, forming a feedback loop that reinforces immune
exhaustion or, in specific contexts, sustains localized immune
responses.
Regulatory networks governing TLS
formation
The lymphotoxin (LT) and LIGHT (Lymphotoxin-like,
exhibits inducible expression, and competes with HSV glycoprotein D
for HVEM, a receptor expressed by T lymphocytes) signaling pathways
play essential roles in the initiation and maturation of TLSs. LT,
a cytokine primarily secreted by CD4+ T cells and other
immune cells, binds to the LTβ receptor to promote lymphoid tissue
development and maintenance. Studies have shown that elevated
expression of LT enhances lymphocyte proliferation and
differentiation and facilitates the organization of TLSs within
tumors. In the TME, increased LT signaling contributes to the
maturation of TLSs and enhances tumor-specific immune responses
(97). LIGHT, a TNF superfamily
member, can engage its receptors to activate downstream signaling
cascades that promote immune cell migration and TLS formation.
Overexpression of LIGHT has been shown to increase the number and
function of TLSs in tumors, thereby amplifying antitumor immunity
(98). Thus, targeting the LT and
LIGHT pathways holds potential for modulating TLS formation in
cancer immunotherapy.
The C-X-C motif chemokine ligand (CXCL) 13/C-X-C
motif chemokine receptor (CXCR) 5 axis is another key regulatory
pathway in TLS development and function. CXCL13, a chemokine mainly
secreted by plasma cells and dendritic cells (DCs), guides the
migration of B cells and certain T cell subsets toward TLS regions
by binding to its receptor, CXCR5, expressed on these immune cells.
Elevated expression of CXCL13 in the TME is associated with
increased TLS formation. High levels of CXCL13 promote immune cell
aggregation and activation within the tumor and facilitate the
germinal center reaction, thereby enhancing the humoral immune
response (99). This mechanism has
been confirmed in multiple types of cancer, including breast and
lung cancer, where high CXCL13 expression associates with improved
clinical outcomes (100–102). The CXCL13/CXCR5 signaling axis not
only supports TLS formation but also amplifies their immunological
function, making it a promising focus for future translational
research and clinical application.
Stromal cells provide essential structural and
functional support during TLS formation and maintenance. These
cells, including fibroblasts and endothelial cells, produce
cytokines and chemokines that regulate immune cell recruitment and
activation. In the TME, stromal remodeling and activation are key
for maintaining the structural integrity of TLSs. Specific stromal
cell subsets have been identified that secrete chemokines such as
CXCL12 and CCL21, which promote lymphocyte migration and survival,
thereby facilitating TLS development (103). In addition, stromal cells interact
with immune cells to regulate their activation and proliferation,
thus influencing the efficacy of antitumor immunity. In several
types of cancer, stromal cell phenotypes have been associated with
TLS maturity, well-structured TLSs often coincide with high stromal
activity and cytokine production, which are predictive of improved
prognosis. Therefore, as shown in Fig.
4, stromal cells not only serve as a scaffold for TLSs but also
act as dynamic regulators of their immunological function, offering
potential targets for therapeutic intervention.
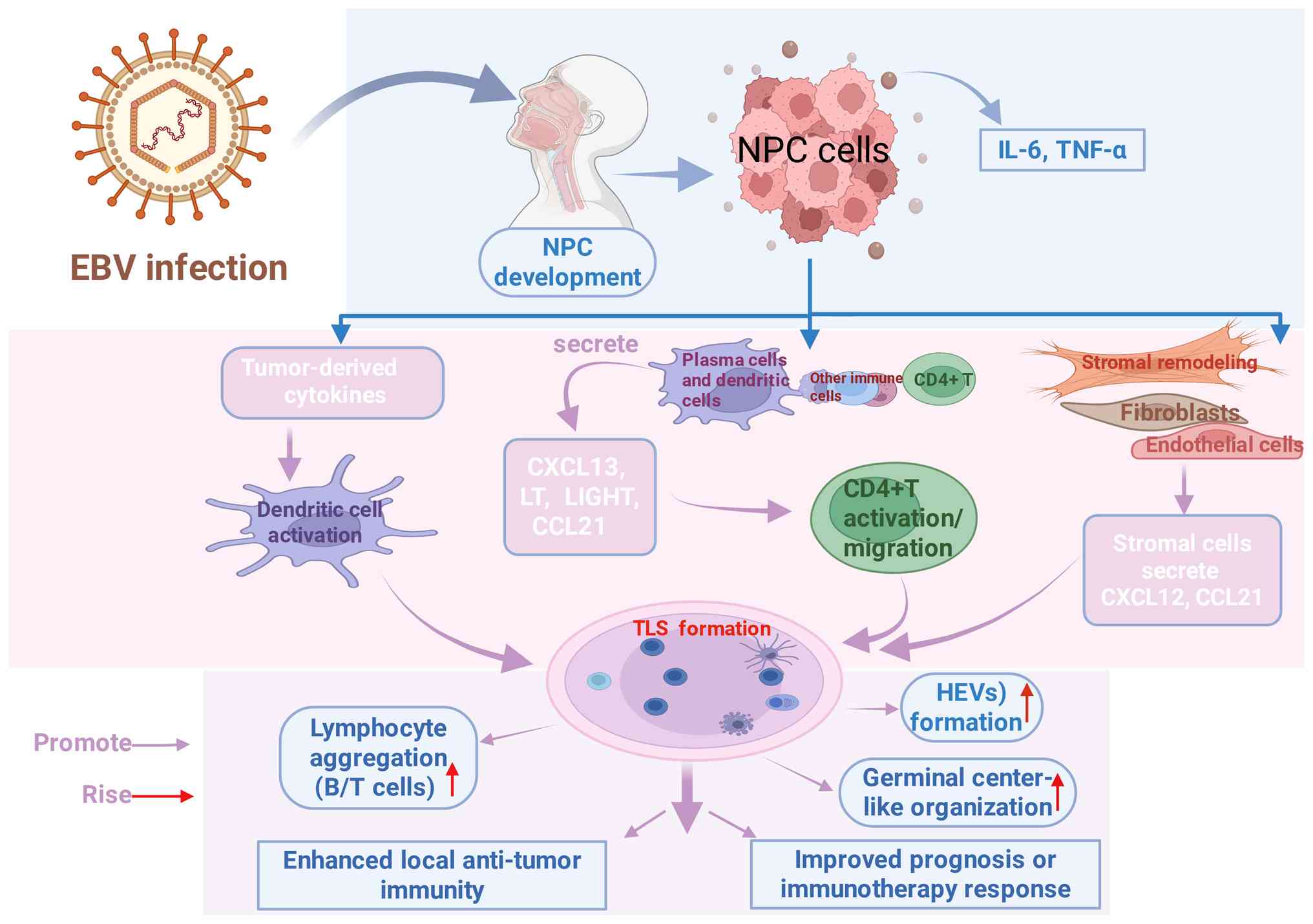 | Figure 4.Schematic of the regulatory network
formed by TLSs in NPC. Following EBV infection, tumor-derived
cytokines activate dendritic cells, promoting the aggregation of B
cells and T cells, thereby enhancing local antitumor immunity.
During the progression of NPC, various immune cells (for example,
plasma cells, dendritic cells) and stromal cells (for example,
fibroblasts, endothelial cells) participate in the activation and
migration of CD4+ T cells by secreting chemokines such
as CXCL12 and CCL21. This process promotes the formation of HEVs
and the organization of germinal center-like structures, ultimately
improving prognosis or enhancing responses to immunotherapy. TLSs,
tertiary lymphoid structures; EBV, Epstein-Barr virus; NPC,
nasopharyngeal carcinoma; B/T cells, B lymphocytes and T
lymphocytes; CXCL12, C-X-C motif chemokine ligand 12; CCL21, C-C
motif chemokine ligand 21; HEVs, high endothelial venules;
CD4+ T, CD4-positive T lymphocyte. |
Association of TLSs with therapeutic
responses
Recent studies have revealed a strong association
between TLSs and clinical responses to immunotherapy across various
cancer types (104–106). The density of TLSs within the TME
has emerged as a promising biomarker for predicting immunotherapy
efficacy. For example, in melanoma, the presence of TLSs associates
positively with intratumoral immune responses and overall patient
survival (94). Furthermore, TLS
density is associated with increased infiltration of
CD8+ T cells and B cells, suggesting that TLSs may
enhance immune responses by supporting effector cell accumulation.
High TLS density has also been associated with improved responses
to ICIs, particularly anti-PD-1 therapy, in renal cell carcinoma
and non-small cell lung cancer (103). As such, TLSs serve not only as
prognostic indicators but also as a potential basis for
personalized treatment strategies.
The maturation status of TLSs is another key factor
influencing tumor prognosis and immunotherapeutic responsiveness.
Mature TLSs are typically characterized by well-organized germinal
centers, dense lymphocyte aggregates and active immune signaling.
In clear cell renal cell carcinoma, mature TLSs have been
associated with improved survival and enhanced responsiveness to
immunotherapy (107). Moreover,
mature TLSs often promote plasma cell differentiation and antibody
production, features associated with favorable outcomes in several
cancer types (103). Evaluating
TLS maturity may thus provide valuable insights for clinical
prognosis and guide treatment selection.
Artificial induction of TLS formation has emerged as
a novel strategy to enhance antitumor immunity. Through the use of
immune-stimulating agents or genetic engineering techniques, TLSs
can be generated or amplified within tumor tissues to recruit and
activate immune cells. For example, viral vectors and specific
pharmacologic compounds have been shown to induce TLS formation,
thereby improving T cell infiltration and the response to immune
checkpoint blockade (103).
Preclinical studies have also demonstrated that pharmacologic
agents enhancing TLS formation can reshape the TME and boost the
efficacy of immunotherapy (108–111). Thus, therapeutic induction of TLSs
offers a promising approach to augment immune responses and may
represent a novel avenue for clinical cancer treatment.
Recent studies have begun to provide NPC-specific
evidence associating TLSs to the immunotherapy response (112,113). Analyses of NPC tumor specimens
have shown that higher TLS density and more mature TLS organization
are associated with increased infiltration of CD8+ T cells and B
cells, enhanced antigen presentation signatures and an inflamed
tumor immune phenotype. Importantly, emerging clinical observations
suggest that patients with NPC with TLS-enriched tumors tend to
exhibit improved responses to ICIs, including higher objective
response rates and prolonged progression-free survival, although
these findings are primarily derived from retrospective cohorts and
exploratory analyses (113,114).
However, it should be noted that current
NPC-specific data remain limited in scale, and the majority of
studies have not yet established a definitive quantitative
association between TLS density and immunotherapy response rate.
Therefore, while available evidence supports a potential
association, further prospective validation is required before TLSs
can be considered robust predictive biomarkers for immunotherapy
responsiveness in NPC.
Importantly, accumulating evidence suggests that
TLSs may exert a context-dependent, dual role in NPC. Mature TLSs
characterized by organized B cell follicles, follicular DCs and
germinal center-like structures are generally associated with
effective antitumor immunity. By contrast, immature or dysregulated
TLSs may harbor increased regulatory T (Treg) cells, exhausted T
cells or immunosuppressive cytokine signals, potentially
contributing to immune tolerance rather than immune activation. In
NPC, where chronic EBV-driven inflammation persists, TLSs may
dynamically shift between immune-supportive and immune-suppressive
states, underscoring the need to evaluate TLS quality, maturation
status and cellular composition rather than density alone (90,91).
Coordinated regulation of the immune
checkpoint network
Expression characteristics of
classical checkpoint molecules
In NPC, classical immune checkpoint molecules,
including PD-1, CTLA-4 and TIM-3, are highly expressed and play key
roles in mediating immune suppression. Studies have shown that PD-1
is expressed in ≤46.2% of NPC tumor tissues, while CTLA-4
expression is detected in 88.4% of cases (115,116). PD-1 is predominantly expressed on
TILs and is associated with immune evasion mechanisms, particularly
in EBV-positive NPC, highlighting its relevance in the TME
(115). Additionally, TIM-3
expression has been reported in NPC and is considered to contribute
to T cell exhaustion within the TME (117).
A complex interplay and synergistic effect exists
among different checkpoint molecules. Co-expression of PD-1 and
CTLA-4 has been identified as a major driver of immunosuppression
in NPC. Their combined inhibitory effects lead to severe functional
impairment of T cells in the TME (115). Moreover, high levels of TIM-3
expression, in conjunction with PD-1 and CTLA-4, further exacerbate
immune exhaustion, forming a robust suppressive network that
inhibits effector T cell function and promotes tumor progression
and metastasis (117). These
synergistic interactions not only shape the immune landscape of the
tumor but also present potential targets for immunotherapeutic
intervention. Notably, combinatorial blockade of these checkpoints
may restore T cell function and enhance antitumor immunity.
The expression of checkpoint molecules is associated
with the functional status of T cells. In the NPC microenvironment,
elevated PD-1 expression often associates with T cell dysfunction
and exhaustion (115). For
example, TILs frequently co-express PD-1 and TIM-3 while exhibiting
reduced levels of effector cytokines such as IFN-γ and TNF-α
(117). This expression profile
reflects a dysfunctional T cell phenotype with impaired cytotoxic
capacity. Therefore, checkpoint molecule expression not only serves
as a biomarker of the immunological landscape in NPC but also
reflects the degree of T cell impairment, providing a rationale for
targeted immunotherapeutic strategies aimed at reinvigorating
antitumor T cell responses and improving clinical outcomes.
Emerging immune regulatory
molecules
The discovery of novel immune checkpoints has
expanded the therapeutic landscape of cancer immunotherapy. Among
these, V-domain Ig suppressor of T cell activation (VISTA) and
lymphocyte-activation gene 3 (LAG-3) have garnered increasing
attention. VISTA primarily functions by inhibiting T cell
activation and proliferation, thereby promoting immune evasion in
the TME. High VISTA expression is associated with poor prognosis in
various malignancies, including melanoma, non-small cell lung
cancer, pancreatic cancer, colorectal cancer, ovarian cancer and
acute myeloid leukemia (118–120). Several anti-VISTA therapeutic
agents have entered clinical trials, showing promising preliminary
results (121).
LAG-3 is another negative regulatory molecule that
plays a key role in modulating T cell activity. It binds to MHC
class II molecules and suppresses T cell proliferation and cytokine
production. Numerous studies have associated LAG-3 expression with
immunosuppressive states in the TME, and blockade of LAG-3
signaling has been shown to enhance T cell effector function and
antitumor activity (122–124). As a result, monoclonal antibodies
targeting LAG-3 have demonstrated therapeutic potential in clinical
trials across various cancer types.
Members of the B7 family are also key regulators of
immune responses in NPC. This family includes costimulatory
molecules such as B7-1 (CD80) and B7-2 (CD86), as well as
inhibitory molecules such as PD-L1. Within the NPC
microenvironment, B7 molecules interact with receptors on T cells
to modulate their activity and function, influencing the immune
evasion capacity of tumor cells. It has been reported that NPC
cells upregulate CD80 and CD86 expression to enhance costimulatory
signaling, which paradoxically may promote tumor progression by
inducing chronic T cell stimulation and subsequent exhaustion
(125).
Furthermore, inhibitory B7 family members such as
PD-L1 suppress T cell activation and proliferation, thereby
enhancing tumor immune evasion. In NPC, elevated PD-L1 expression
has been associated with tumor aggressiveness and poor prognosis.
ICIs targeting the PD-1/PD-L1 axis have demonstrated encouraging
efficacy in clinical trials, offering new hope for NPC treatment
(121). CD28 is a key
costimulatory receptor on T cells that interacts with CD80 and CD86
to promote T cell activation and expansion. Alterations in the
CD28/CD80/CD86 axis in NPC may considerably influence immune
responses within the TME. Studies have shown that NPC cells can
upregulate CD80 and CD86 to enhance costimulatory signaling, which
may paradoxically contribute to immune exhaustion and tumor
progression. This dysregulation can impair T cell proliferation and
contribute to a dysfunctional immune phenotype (122,126).
Moreover, changes in CD28 expression have been
observed in patients with NPC, with low CD28 expression being
associated with tumor aggressiveness and poor clinical outcomes.
Targeting the CD28/CD80/CD86 network may therefore represent a
promising strategy to enhance antitumor immune responses in NPC.
Novel therapeutic approaches aiming to modulate this axis are
currently under development to improve the efficacy of
immunotherapies in this context (121).
Mechanisms of resistance to checkpoint
blockade
The emergence of antigen-loss variants is a major
mechanism of resistance to immune checkpoint blockade therapy.
Tumor cells may acquire mutations or deletions that result in the
loss of surface antigens, rendering them invisible to T cells. For
example, in melanoma, treatment with anti-PD-1 antibodies has been
shown to select for tumor cells with MHC class I mutations or
deletions, impairing antigen presentation and facilitating immune
escape (127). In addition, tumors
may selectively retain variants that are poorly recognized by T
cells, further compounding resistance to immune checkpoint
inhibition.
Adaptive changes in TCR signaling also contribute to
therapeutic resistance. Tumor cells can suppress TCR signaling
pathways through metabolic reprogramming, leading to impaired T
cell function. For example, enhanced tumor metabolism may induce a
high metabolic burden in T cells, rendering them unable to
proliferate or function effectively in the hypoxic and
nutrient-deprived TME. These impairments are often associated with
downregulation of TCR signaling components (128). Therapeutic strategies that restore
or modulate TCR signaling may therefore improve responses to
checkpoint inhibitors.
The compensatory expansion of immunosuppressive cell
populations, such as Treg cells and TAMs, also plays a key role in
mediating resistance. These cells secrete suppressive cytokines
such as IL-10 and TGF-β, which inhibit effector T cell activity and
facilitate immune evasion. In some cancer types, a notable increase
in Treg frequency has been observed following checkpoint blockade,
thereby limiting therapeutic efficacy (129). Targeting these immunosuppressive
populations, through Treg depletion or TAM inhibition, may offer a
viable strategy to overcome resistance and improve the efficacy of
immune checkpoint blockade.
Immunoregulatory functions of myeloid
cells
Polarization of TAMs
In the TME of NPC, TAMs exist primarily in two
phenotypic states: M1 and M2. The dynamic balance between these
subtypes plays a key role in tumor progression. M1 macrophages are
generally considered antitumorigenic due to their ability to
produce proinflammatory cytokines such as TNF-α and IFN-γ, thereby
promoting effective antitumor immune responses. By contrast, M2
macrophages support tumor growth and metastasis by secreting
anti-inflammatory cytokines (for example, IL-10) and promoting
angiogenesis, which contribute to an immunosuppressive TME
(130). Studies have shown that
the ratio of M1 to M2 macrophages in NPC is regulated by various
signals within the TME (131,132). As the tumor progresses, this
balance often shifts toward the M2 phenotype, thereby facilitating
immune evasion and accelerating tumor development (133). Understanding the dynamic
plasticity of TAMs in NPC is important for elucidating immune
escape mechanisms and may provide potential targets for novel
immunotherapeutic strategies.
Colony-stimulating factor 1 (CSF1) and its receptor
CSF1R play key roles in the recruitment and polarization of
macrophages in the TME. In NPC, tumor cells secrete CSF1 to recruit
surrounding macrophages and promote their differentiation into the
M2 phenotype, thereby enhancing tumor growth and dissemination
(134). Activation of the
CSF1/CSF1R signaling pathway has been directly associated with an
increase in TAMs, which not only contributes to immune suppression
within the TME but also associates with poor clinical outcomes
(135). Thus, inhibition of CSF1R
signaling may serve as an effective strategy to reprogram
macrophage polarization and boost antitumor immunity.
The interaction between macrophages and T cells is
another key regulatory mechanism in the NPC microenvironment. M1
macrophages activate T cells by producing proinflammatory cytokines
and presenting antigens, thereby enhancing CTL responses.
Conversely, M2 macrophages suppress T cell activity and facilitate
immune escape (136). In NPC, an
increase in M2 macrophages has been associated with reduced
infiltration and functional impairment of CD8+ T cells, suggesting
an association between TAM polarization and T cell efficacy
(137). Furthermore, macrophages
secrete immunosuppressive cytokines such as IL-10 and TGF-β, which
promote the differentiation of Treg cells, thereby further
inhibiting CD8+ T cell function (138). Enhancing the functional crosstalk
between macrophages and T cells may therefore offer a promising
strategy for improving the efficacy of immunotherapy in NPC.
Functions of myeloid-derived
suppressor cells (MDSCs)
MDSCs are key immunosuppressive components of the
TME and have gained increasing attention in the context of NPC.
Composed of immature myeloid cells, MDSCs exhibit potent
immunosuppressive activity. In patients with NPC, MDSCs are notably
expanded, and their presence is associated with tumor progression
and poor prognosis (139). Based
on surface markers, MDSCs can be divided into two main subsets:
Monocytic MDSCs and granulocytic or polymorphonuclear MDSCs.
Monocytic MDSCs are particularly abundant in the peripheral blood
of patients with NPC and suppress T cell activity via the secretion
of inhibitory cytokines such as IL-10 and TGF-β, thereby promoting
tumor growth and metastasis (140). Additionally, MDSCs contribute to
immunosuppression by depleting L-arginine and generating ROS,
impairing effective antitumor immune responses (141).
In NPC, MDSC accumulation is associated with tumor
metabolic states. MDSCs begin to accumulate early during tumor
development, and both their numbers and suppressive capacity
increase as the tumor progresses, further exacerbating immune
evasion and leading to worse clinical outcomes (140). Therefore, understanding the
phenotypic and functional characteristics of MDSCs is important for
the development of targeted therapeutic strategies.
L-arginine metabolism plays a pivotal role in
MDSC-mediated immunosuppression. In the NPC microenvironment, MDSCs
express high levels of arginase-1, which depletes local L-arginine,
thereby impairing T cell proliferation and activation. Studies have
shown that arginine metabolism not only impacts T cell function but
also supports the survival and suppressive function of MDSCs
(139,142–144). Arginine depletion disrupts amino
acid synthesis in T cells, weakening their immune responses and
facilitating immune evasion by tumor cells.
Moreover, L-arginine metabolism is associated with
MDSC phenotype and function. MDSCs activate downstream signaling
pathways such as STAT3 and NF-κB to promote their immunosuppressive
activity. They also secrete proinflammatory cytokines such as IL-6,
which sustain the immunosuppressive nature of the TME (141). Thus, targeting arginine metabolism
represents a promising avenue to enhance the efficacy of
immunotherapy in NPC.
Therapeutic strategies targeting MDSCs have
potential in cancer immunotherapy. By eliminating or functionally
inhibiting MDSCs, researchers aim to reduce immunosuppression and
potentiate antitumor immune responses. Several studies have
demonstrated that MDSC-targeted therapies can improve responses to
immune checkpoint blockade (145–148).
One approach involves the use of specific drugs to
inhibit MDSC development and function. Small-molecule inhibitors
targeting MDSC metabolic pathways, such as arginine and fatty acid
metabolism, have been shown to reduce MDSC numbers and restore T
cell function (149). Furthermore,
clinical trials suggest that combining ICIs with MDSC-targeting
agents may notably enhance patient survival and quality of life.
Collectively, therapies aimed at modulating MDSCs offer a promising
strategy for improving outcomes in NPC and future research should
focus on optimizing these approaches to maximize their clinical
benefit.
Functional impairment of DCs
DCs are APCs that play a central role in initiating
and regulating immune responses. Impaired DC maturation is
frequently associated with disease progression, particularly in the
context of tumors and chronic viral infections (150,151). For example, EBV infection has been
shown to downregulate key molecules involved in DC maturation,
resulting in impaired antigen presentation and weakened T cell
responses (152). Moreover, DC
maturation is dependent on several signaling pathways, including
Toll-like receptor (TLR) and IFN pathways. Although TLR activation
enhances DC maturation and functionality, chronic inflammation
often suppresses these pathways, leading to DC dysfunction
(153,154). Additionally, metabolic
alterations, such as reduced expression of lactate dehydrogenase,
can impair the energy metabolism and antigen-presenting capacity of
DCs, contributing to immune evasion in the TME (155).
DCs are a heterogeneous population composed of
conventional, plasmacytoid and monocyte-derived DCs, each
exhibiting distinct antigen presentation and T cell activation
capacities. Conventional DC1s are particularly efficient in
cross-presenting tumor antigens to CD8+ T cells, whereas
conventional DC2s are primarily involved in CD4+ T cell priming and
regulation (156,157). Plasmacytoid DCs play a key role in
antiviral immunity by producing type I IFNs, but they often exhibit
functional exhaustion in the TME, leading to diminished antigen
presentation (158). This suggests
that the composition and function of DC subsets considerably
influence antigen presentation and immune responses in different
immunological contexts. In NPC and other conditions such as chronic
viral infection, DC dysfunction can hinder T cell activation and
limit the overall effectiveness of antitumor immunity (152,159).
In NPC therapy, DC-based vaccines have garnered
attention as a promising immunotherapeutic approach. Given the
central role of EBV in NPC, DC vaccines can stimulate
antigen-specific immune responses against viral antigens. For
example, a CD137L-expressing DC vaccine has demonstrated favorable
tolerability and partial tumor responses in clinical trials, with
some patients achieving measurable tumor regression (160,161). Recent studies also suggest that
combining optimized DC vaccines with other immunotherapeutic
modalities, such as ICIs, may further enhance therapeutic efficacy
(162–165). However, clinical application of DC
vaccines in NPC remains in the early stages and further large-scale
trials are required to evaluate their efficacy, safety and optimal
combination strategies to offer more effective immunotherapeutic
options for patients with NPC.
Metabolic features of the TME
Effects of hypoxia and
acidification
Hypoxia is a common feature of the TME and markedly
influences T cell function through the regulation of
hypoxia-inducible factor 1α (HIF-1α). As a central regulator of
cellular responses to hypoxia, HIF-1α is associated with T cell
survival and function. Although HIF-1α activation under hypoxic
conditions promotes T cell survival, it may also impair their
effector function. Hypoxia can inhibit T cell proliferation and
cytotoxicity, with HIF-1α altering cellular metabolism by
regulating glycolytic pathways and mitochondrial activity.
Specifically, HIF-1α activation reduces intracellular NADPH levels,
compromising antioxidant capacity and contributing to T cell
dysfunction (166). Furthermore,
HIF-1α modulates apoptotic signaling pathways, further affecting T
cell survival and function. Thus, HIF-1α not only facilitates T
cell adaptation to hypoxia but may also contribute to immune
evasion in the TME (167,168).
Lactate accumulation, a result of aerobic
glycolysis by tumor cells, leads to acidification of the TME and
suppresses immune cell function. Studies have demonstrated that
lactate notably reduces T cell cytotoxicity and cytokine production
and promotes T cell exhaustion (169–171). Mechanistically, lactate impairs
intracellular energy metabolism and increases oxidative stress,
leading to diminished T cell activity. Additionally, lactate
inhibits immune cell migration and infiltration into the tumor
site, further limiting their effectiveness. The suppressive effects
of lactate extend beyond T cells to macrophages and other immune
cells, highlighting its role as a key factor in tumor-induced
immunosuppression. Therefore, targeting lactate metabolism may
represent a promising strategy to improve the efficacy of cancer
immunotherapy.
The acidic TME also influences the expression of
immune checkpoints, thereby modulating T cell responses. Acidic
conditions have been shown to upregulate the expression of
checkpoint molecules such as PD-L1, enhancing immune evasion by
tumor cells (172). The increased
expression of PD-L1 under acidic conditions is associated with
HIF-1α stabilization, which further reinforces immunosuppression.
An acidic pH also impairs T cell activation, further limiting
antitumor immunity. Thus, modulating the acid-base balance of the
TME, particularly by neutralizing acidosis to suppress immune
checkpoint expression, may improve responses to immunotherapy.
Notably, metabolic exhaustion is not merely a passive consequence
of chronic activation but actively constrains T cell effector
function, survival capacity and migratory behavior. Metabolically
compromised T cells exhibit diminished cytokine production and
altered chemokine responsiveness, which in turn influence their
spatial distribution and persistence within the TME. These
functional impairments provide a mechanistic association between
metabolic stress and subsequent immune architectural remodeling
(173–175).
Nutrient competition and metabolic
suppression
Tryptophan metabolism plays an important role in
immune regulation, and alterations in this pathway can directly
affect T cell function and fate. Tryptophan catabolism via
metabolites such as kynurenine and quinolinic acid exerts
immunomodulatory effects, particularly in the TME where tumor cells
actively deplete tryptophan to inhibit T cell proliferation and
activity (176). Abnormal
tryptophan metabolism has been associated with T cell exhaustion
and reduced antitumor efficacy, ultimately impairing immune
responses in patients with cancer (177). Moreover, tryptophan metabolites
can modulate immune checkpoint expression, promoting T cell
exhaustion. These findings suggest that targeting tryptophan
metabolism may provide novel strategies for enhancing cancer
immunotherapy.
The adenosine signaling pathway is another key
regulator of T cell metabolism and is commonly associated with
immunosuppression in the TME. Adenosine, generated by tumor
metabolism, suppresses T cell proliferation and cytokine secretion
through activation of downstream pathways such as cAMP and AMPK
(178). Elevated adenosine levels
in the TME contribute to T cell dysfunction and facilitate immune
escape by tumor cells (179).
Therefore, targeting the adenosine signaling axis offers a
promising approach to reinvigorate exhausted T cells and restore
effective antitumor responses.
Glutamine is an essential nutrient for T cell
activation and function. It supports T cell proliferation, cytokine
production and differentiation. In the TME, competition for
glutamine between tumor and immune cells results in reduced
availability, particularly impairing T cell metabolic capacity in
chronic tumors and infections (180,181). Glutamine metabolism also
influences systemic energy metabolism and intracellular signaling
pathways, affecting T cell development and function (182,183). Thus, therapeutic strategies
targeting glutamine metabolism may enhance T cell-mediated
antitumor immunity.
Combination strategies for metabolic
intervention
Indoleamine 2,3-dioxygenase (IDO) inhibitors have
emerged as promising agents in cancer immunotherapy, particularly
for reversing immune suppression in the TME by blocking tryptophan
metabolism (184–186). IDO inhibitors reduce Treg activity
and enhance effector T cell function. Preclinical studies have
shown that agents such as D-1-methyl-tryptophan and INCB024360
notably enhance antitumor immune responses in mice, especially when
combined with ICIs (85,187,188). Clinical trials have further
suggested that IDO inhibitors may improve outcomes in patients with
treatment-resistant tumors, highlighting their potential value as
components of combinatorial immunotherapy.
CD73, an ectoenzyme involved in adenosine
generation, plays a central role in mediating immunosuppression in
the TME. Therapeutic strategies targeting CD73 aim to inhibit
adenosine production, thereby enhancing T cell cytotoxicity and
reversing immune suppression. CD73 inhibitors such as AB928 have
been shown to promote T cell proliferation and effector function,
particularly when used in combination with PD-1/PD-L1 blockade,
resulting in improved antitumor responses (81). In addition to boosting immune
responses, CD73 targeting also reduces tumor metastasis and
recurrence, suggesting broad therapeutic potential across multiple
types of cancer.
Combining metabolic modulation with immune
checkpoint blockade has demonstrated synergistic effects in cancer
therapy. Tumor cells often reprogram their metabolism to suppress T
cell function, leading to immune exhaustion. Intervening in
metabolic pathways, through administration of metabolic
intermediates or inhibition of key enzymes, can restore T cell
activity and enhance the efficacy of ICIs (189). This combined approach may overcome
the immunosuppressive barriers of the TME and improve clinical
outcomes. Future studies should focus on elucidating the precise
mechanisms underlying various metabolic pathways and optimizing
combinatorial strategies with checkpoint inhibitors to develop more
effective immunotherapies.
Characteristics of EBV-specific immune
responses
Clonal dynamics of EBV
antigen-specific T cells
In NPC, the clonal dynamics of EBV-specific T cells
are important to understanding the TIME and the therapeutic
response. EBV infection triggers antigen-specific T cell responses
with distinct phenotypic features. Studies have demonstrated that
EBV-specific CD8+ and CD4+ T cells exhibit altered proportions and
often show an exhausted phenotype, with ISG15+ CD8+ T cells being
highly enriched in EBV-positive tumors. The infiltration of
antigen-specific T cells in the TME is associated with disease
progression (190).
Analysis of T cell clonal dynamics has revealed a
notable association between TCR repertoire diversity and disease
status. Decreased TCR diversity is commonly observed during tumor
progression and is associated with increased tumor burden and
immune evasion. High-throughput TCR sequencing enables precise
monitoring of TCR repertoire shifts, revealing the expansion or
contraction of specific clones before and after treatment (191). Moreover, the clonal expansion of
EBV-specific T cells reflects dynamic changes in the TME,
highlighting their role in tumor immune surveillance.
In terms of memory T cell maintenance, EBV-specific
memory T cells are often sustained at high frequencies following
primary infection, particularly in healthy EBV carriers. This
indicates their key role in controlling EBV persistence and
EBV-related malignancies (192).
Notably, EBV has evolved mechanisms to modulate T cell metabolism
and activation, thereby facilitating immune evasion. These findings
suggest that therapeutic strategies aimed at enhancing EBV-specific
memory T cell responses may improve antitumor immunity.
In summary, the clonal dynamics of EBV
antigen-specific T cells not only reflect their phenotypic
adaptations to latent viral antigens but also reveal the
association between TCR repertoire diversity and disease
progression. These insights provide potential biomarkers and
therapeutic targets for NPC and deepen understanding of immune
evasion mechanisms in EBV-associated malignancies.
Role of humoral immune responses
Humoral immunity plays a pivotal role in
controlling EBV infection. Changes in EBV-specific antibody
profiles have been associated with tumorigenesis, disease
progression and patient prognosis. In patients with NPC, the
presence and titers of EBV-specific antibodies, such as viral
capsid antigen IgA/IgG and EBNA IgA/IgG, serve as important
biomarkers for disease monitoring (193). Elevated antibody levels are
frequently observed in patients with NPC, particularly in those
with disease recurrence, indicating their potential as early
warning indicators for disease progression and the therapeutic
response (194). Furthermore, the
diversity of EBV-specific antibody profiles may reflect the immune
status of the host and may be influenced by changes in the TME
(195).
Neutralizing antibodies play an important
protective role during EBV infection by recognizing and binding
viral envelope proteins, thereby preventing viral entry and
replication. In NPC, the generation of EBV-specific neutralizing
antibodies not only aids viral clearance but also enhances
antitumor immune responses by promoting CD8+ T cell activation and
cytotoxicity (196). High levels
of neutralizing antibodies have been associated with favorable
clinical outcomes, suggesting their importance in limiting
EBV-associated tumor progression (16).
B cells, as the primary source of antibodies, also
contribute to immune regulation within the TME. In NPC, B cells
exert multiple functions through antibody production, cytokine
secretion and T cell modulation. EBV infection promotes B cell
activation and expansion, enhancing antiviral immunity. Within the
TME, B cells can form germinal center-like structures that
facilitate affinity maturation and the production of tumor-specific
antibodies, potentially suppressing tumor growth and metastasis
(197). Additionally, B cells
interact with T cells via antigen presentation and secretion of
immunoregulatory factors, further enhancing antitumor immunity
(198). Therefore, B cell activity
and abundance are key determinants of NPC prognosis.
Virological mechanisms of immune
evasion
EBV is a major etiological factor in NPC, and viral
genetic variation plays a key role in immune evasion. Mutations in
several regions of the EBV genome have been implicated in the
ability of the virus to evade host immune surveillance, including
alterations in the PD-1/PD-L1 signaling pathway. In EBV-positive
tumors, upregulation of PD-L1 on tumor cells suppresses the
function of EBV-specific T cells, allowing for immune escape
(199). Additionally, EBV
mutations can affect antigen presentation, reducing TCR recognition
of viral epitopes and impairing effective immune responses
(200). Thus, viral genetic
variation contributes to both oncogenicity and immune evasion.
EBV-encoded miRNAs also play a central role in
modulating host immunity. EBV miRNAs, such as those from the BART
and BHRF1 families, downregulate host immune-related gene
expression, suppressing cytokine production and immune cell
activation. For example, BART10-3p targets multiple host genes to
inhibit antiviral responses and promote tumor cell survival
(201). These mechanisms not only
facilitate immune evasion but may also promote tumorigenesis within
the EBV-positive microenvironment.
Latency is another immune evasion strategy used by
EBV. During latency, the virus remains hidden in B cells with
limited antigen expression, meaning that infected cells are
difficult to detect and eliminate. Latent-to-lytic reactivation can
trigger acute immune responses and, in some cases, immune-mediated
pathology such as EBV-associated lymphoproliferative disorders
(202). Furthermore, reactivation
may contribute to tumor progression by impairing immune cell
function within the TME. Understanding the immunological
consequences of EBV latency and reactivation is key for developing
novel therapeutic strategies against EBV-associated
malignancies.
In summary, the virological mechanisms underlying
EBV-mediated immune evasion are complex and multifaceted (as
detailed in Fig. 5). Future studies
are warranted to further elucidate these pathways, which may
uncover novel targets and strategies for cancer immunotherapy.
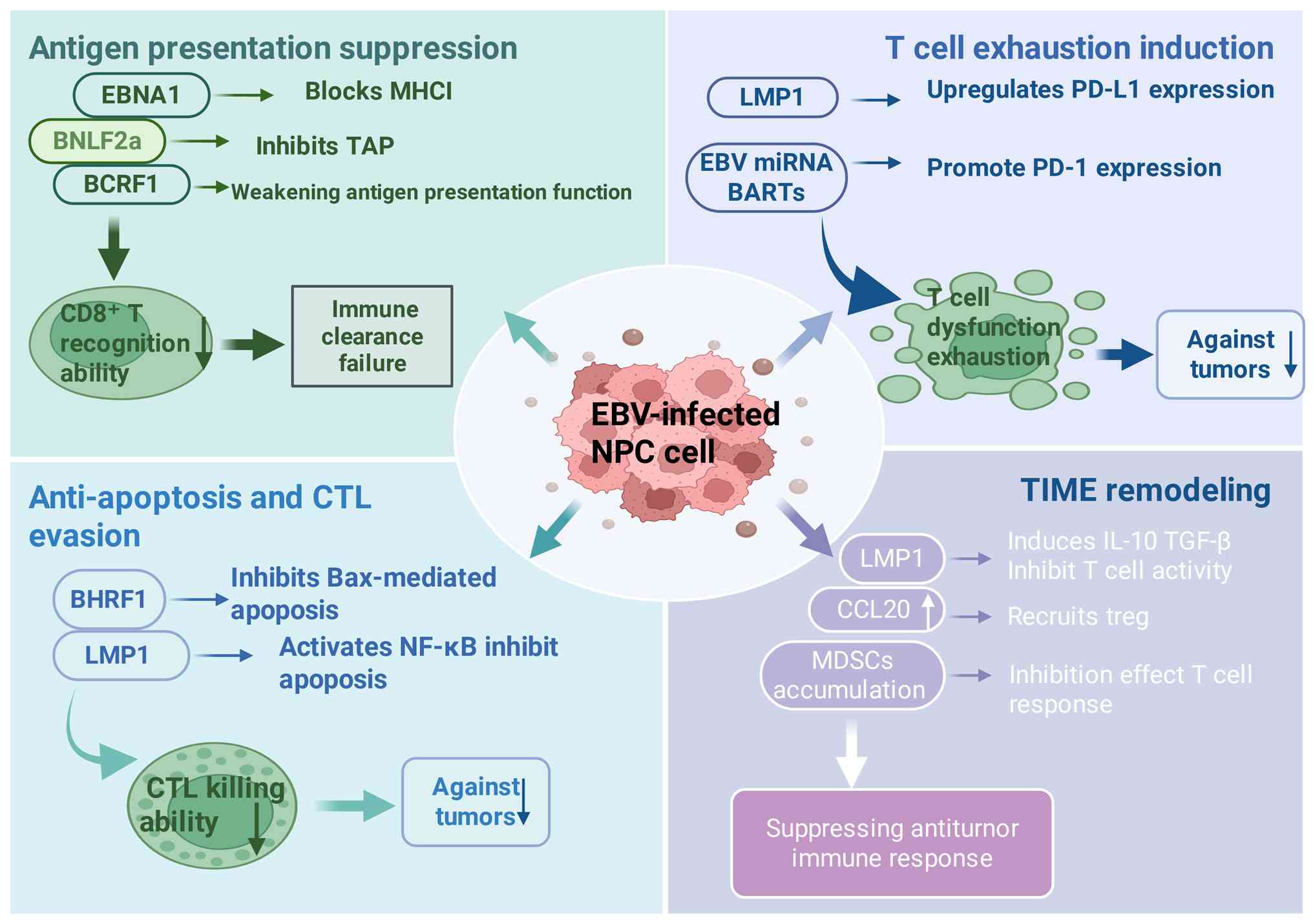 | Figure 5.Schematic diagram of the virological
mechanisms of immune evasion by EBV. After EBV infection of NPC
cells, immune evasion is achieved through multiple mechanisms that
suppress antitumor immune responses. The molecular mechanisms
include: i) Antigen presentation inhibition: EBV proteins (EBNA1,
BNLF2a and BCRF1) block the function of MHC class I molecules and
TAP transport, weakening the recognition capacity of
CD8+ T cells. ii) Anti-apoptosis and CTL evasion: BHRF1
and LMP1 inhibit apoptosis (for example, by blocking Bax) or
activate the NF-κB pathway, assisting tumor cells in evading
CTL-mediated killing. iii) Induction of T cell exhaustion: LMP1 and
BARTs mRNA upregulate the PD-L1/PD-1 signaling pathway, leading to
T cell dysfunction. iv) Remodeling of the TIME: Through the
secretion of factors such as IL-10, TGF-β and CCL20, EBV recruits
Tregs and MDSCs, further suppressing T cell activity and creating
an immunosuppressive microenvironment. EBV, Epstein-Barr virus;
NPC, nasopharyngeal carcinoma; EBNA1, Epstein-Barr nuclear antigen
1; BNLF2a, BamN-terminal latent protein family member 2a; BCRF1,
BamC-terminal reading frame 1; BHRF1, BamHI rightward reading frame
1; LMP1, latent membrane protein 1; BARTs, BamHI-A rightward
transcripts; MHC-I, major histocompatibility complex class I; TAP,
transporter associated with antigen processing; CTL, cytotoxic T
lymphocyte; PD-L1, programmed cell death ligand 1; PD-1, programmed
cell death protein 1; Treg cell, regulatory T cell; MDSCs,
myeloid-derived suppressor cells; CCL20, C-C motif chemokine ligand
20; TIME, tumor immune microenvironment. |
Dynamic process of immunoediting
Characteristics of the elimination
phase
During the elimination phase of NPC, effective
early immune surveillance plays an important role. This early
immune response primarily depends on specific immune cells, such as
CD8+ T cells and natural killer (NK) cells, which are key for
recognizing and eliminating tumor cells. For example, in studies of
chronic hepatitis B virus infection, effective immune surveillance
in the acute phase was marked by rapid activation of virus-specific
T cells and efficient viral clearance (203). Similarly, in NPC, EBV infection
triggers the expression of viral antigens by tumor cells, thereby
activating host immune responses.
The TME in early NPC harbors a variety of immune
cells that secrete cytokines and express activation markers to
enhance T cell-mediated surveillance. The effectiveness of this
early immune response not only determines the extent of tumor cell
clearance but also influences the formation of immune memory. Thus,
enhancing early immune surveillance may represent a promising
strategy for NPC treatment.
Immunogenic cell death (ICD) is a key component of
immune-mediated tumor clearance, particularly in NPC. ICD promotes
the release of tumor antigens through specific death signals,
thereby triggering robust immune activation. Studies have shown
that chemotherapeutic or radiotherapeutic stress can induce ICD via
the release of molecules such as HMGB1 and ATP, which recruit DCs
and activate T cells (204–207).
In chronic hepatitis B virus models, CD3+ macrophages are activated
during viral clearance and produce large amounts of TNF-α and other
proinflammatory mediators, enhancing ICD and immune activation
(208). Similarly, in NPC,
ICD-inducing chemotherapeutic agents have been shown to improve
tumor immunogenicity and promote T cell activation and
expansion.
The activation of innate immunity during the
elimination phase involves complex multilayered mechanisms.
Tumor-derived danger signals (for example, damage-associated
molecular patterns) are recognized by innate immune cells such as
macrophages and DCs via pattern recognition receptors (for example,
TLRs), leading to the production of inflammatory cytokines and the
recruitment of additional immune effectors (204). Moreover, innate cells such as NK
and NK T cells are rapidly activated and secrete interferons and
TNF-α, which boost T cell-mediated responses. This innate immune
activation not only modulates the TME but also lays the groundwork
for adaptive immunity (209).
Overall, the rapid and effective activation of the innate immune
system is essential for successful tumor elimination and long-term
immune memory formation in NPC.
Transition of the equilibrium
phase
The equilibrium phase represents a key transitional
stage in NPC development, where immune selection pressure shapes
the dynamic interactions between tumor cells and immune cells.
Initially, host immunity attempts to control tumor growth via CTLs
and NK cells. However, as tumors evolve, immune pressure leads to
the emergence of escape mechanisms, such as the upregulation of
immune checkpoint molecules (for example, PD-1 and CTLA-4), which
suppress T cell activity and facilitate immune evasion (210). Furthermore, cytokines such as IL-6
and TGF-β within the TME promote T cell exhaustion, weakening
antitumor responses (211).
Tumor heterogeneity, a hallmark of cancer, is
particularly pronounced in NPC. It arises not only from genetic
variation within tumor cells but also from the diverse composition
and function of immune cells in the TME. Various immune
subpopulations (for example, M1 vs. M2 macrophages, effector vs.
Treg cells and B cell subsets) exert dual roles in tumor
progression. M1 macrophages support antitumor immunity, whereas M2
macrophages promote immunosuppression and tumor growth (212). These diverse immune cell
interactions, along with differential cytokine secretion and
antigen expression, lead to TIME remodeling and increased tumor
heterogeneity (213).
Immunoediting is a key mechanism by which tumors
modulate antigen expression in response to immune pressure. Tumor
cells may selectively lose or downregulate specific antigens to
reduce their immunogenicity and avoid recognition by T cells
(214). This process contributes
to the emergence of immune-evasive tumor subclones with enhanced
survival and suppressive capabilities. In NPC, changes in
EBV-associated antigen expression may impair T cell recognition and
hinder effective immune responses (215). These dynamic alterations in the
antigenic landscape influence disease progression and therapeutic
efficacy, underscoring the need to consider immunoediting features
when designing treatment strategies.
Establishment of the escape phase
Immune escape is a defining feature of advanced
tumor progression, particularly in EBV-associated types of cancer
such as NPC. The evolving TME plays a key role in establishing
immunosuppression. EBV-infected tumor cells upregulate immune
checkpoint molecules such as PD-L1 to inhibit T cell responses,
thereby facilitating immune evasion (216).
The interactions between tumor cells and
infiltrating immune cells within the TME are central to immune
escape. EBV-positive tumor cells secrete both proinflammatory and
immunosuppressive cytokines that reprogram surrounding immune
cells. TAMs and Treg cells are often enriched in the TME, secreting
immunosuppressive cytokines such as IL-10 and TGF-β to reinforce
local immunosuppression (217).
Metabolic reprogramming is another mechanism by
which tumors escape immune surveillance. Tumor-derived metabolites
such as lactate inhibit T cell function and promote a suppressive
microenvironment conducive to tumor survival and growth (218). Chronic EBV antigen exposure leads
to progressive CD8+ T cell dysfunction, characterized by high
expression of inhibitory molecules (for example, PD-1, LAG-3) and
diminished cytokine secretion and proliferative capacity (180).
T cell exhaustion, a hallmark of chronic infection
and cancer, is often irreversible. It results from persistent
antigen stimulation, suppressive signaling and metabolic stress in
the TME (219). In NPC, nutrient
competition within the TME, particularly involving glucose and
glutamine, exacerbates T cell exhaustion and impairs antitumor
immunity.
Stromal remodeling also contributes to immune
escape and the establishment of immune privilege. Tumor-associated
stromal cells such as fibroblasts and myeloid cells interact with
tumor cells to reshape the ECM and modulate immune cell
infiltration (220). In NPC,
EBV-infected cells secrete cytokines that recruit and activate Treg
cells, which secrete TGF-β and IL-10 to suppress effector T cell
functions and maintain immune privilege (217). Additionally, tumor cells secrete
matrix metalloproteinases that degrade ECM components, promoting
tumor invasion and altering immune cell localization and activity
(221). Therefore, targeting
stromal remodeling and reversing T cell exhaustion may represent
promising strategies for overcoming immune escape in NPC and
enhancing immunotherapy efficacy.
Therapeutic strategies and clinical
translation
Limitations of current
immunotherapies
Immunotherapy has emerged as a promising treatment
modality for NPC, yet pronounced limitations remain. Notably, the
response rate to anti-PD-1 inhibitors in patients with NPC is
relatively low. Clinical trials have revealed that despite
substantial infiltration of CD8+ T cells in the TME, therapeutic
efficacy remains suboptimal. This discrepancy is largely attributed
to the prominent immunosuppressive signals within the TME, which
restrict the effectiveness of PD-1 blockade (222). Single-cell omics analysis of 50
patient samples demonstrated that NPC cells enhance the development
and suppressive function of Treg cells via the CD70-CD27
interaction, leading to the inhibition of CD8+ T cell activity
(222). Such immunosuppressive
mechanisms highlight the complexity of the TME and the challenges
of monotherapy targeting PD-1.
Regarding treatment resistance, tumor cells employ
diverse immune evasion strategies. Overexpression of CD70 in NPC
cells impairs CD4+ T cell metabolism and function, driving T cell
exhaustion (222). Moreover,
metabolic reprogramming within the TME has been identified as a key
contributor to T cell dysfunction, thereby promoting tumor
progression and metastasis (223).
Comprehensive investigation into these resistance mechanisms is
important to inform the development of novel therapeutic approaches
that can enhance immunotherapy outcomes.
The development of reliable biomarkers for
predicting the immunotherapy response remains challenging. Although
PD-L1 expression is widely used as a predictive marker, its
prognostic accuracy is contentious (224). Radiomic features derived from
magnetic resonance imaging have demonstrated superior predictive
performance compared with PD-L1 scoring in patients with NPC,
indicating the need to explore more robust and prospective
biomarkers (224). Additionally,
the composition of the gut microbiome has emerged as a potential
modulator of immunotherapy efficacy, with specific microbial
communities associating with treatment responses in NPC (225). Future research should focus on
multi-dimensional biomarker integration to improve response
prediction and facilitate personalized treatment.
In summary, despite the potential of current
immunotherapies in NPC, low response rates, complex resistance
mechanisms and biomarker limitations underscore the urgent need for
comprehensive strategies that consider the TME, metabolic pathways
and novel biomarkers to improve therapeutic efficacy and patient
outcomes.
Novel combination therapeutic
strategies
Combining immunotherapy with radiotherapy has shown
promising synergistic effects in NPC treatment. Radiotherapy
enhances tumor immunogenicity by inducing ICD, leading to the
release of tumor antigens and the activation of antitumor immune
responses. This promotes the infiltration and activation of immune
cells, particularly T lymphocytes (226). Studies have demonstrated that the
combination of anti-PD-1 antibodies with radiotherapy improves the
TME by facilitating T cell recruitment and activation, resulting in
improved clinical responses (227–230). Furthermore, radiotherapy can
alleviate immunosuppressive elements in the TME, thereby enhancing
the efficacy of ICIs and offering more effective therapeutic
options for patients with NPC (231). Nonetheless, further clinical
trials are warranted to optimize combination regimens and assess
safety profiles.
Epigenetic modulators have gained increasing
attention in augmenting immunotherapy. In NPC, epigenetic drugs can
modulate interactions between tumor and immune cells, thereby
influencing immune responses. Certain epigenetic agents suppress
immunosuppressive cytokines within the TME, enhancing immune cell
activity and boosting immunotherapy efficacy (232). Additionally, epigenetic regulation
of T cells can restore their functionality and reverse exhaustion
states, strengthening antitumor immunity (233). These advances suggest that
combining epigenetic therapy with immunotherapy may improve
clinical outcomes in NPC.
Targeting metabolic pathways in combination
regimens offers another promising avenue. Metabolic reprogramming
underpins tumor cell survival and proliferation; thus, inhibiting
these pathways can suppress tumor growth and enhance immune
responses. Studies indicate that metabolic interventions can
remodel the TME, improving T cell function and augmenting their
antitumor activity (234–237). Moreover, metabolic inhibitors
combined with immune checkpoint blockade have shown potential in
increasing tumor immunogenicity and immune cell infiltration,
leading to superior therapeutic outcomes (238). Successful implementation of these
strategies may provide novel treatment options and markedly improve
survival and quality of life for patients with NPC.
Prospects for personalized
therapy
Personalized therapeutic strategies based on TCR
repertoire analysis hold potential for NPC treatment. TCR profiling
enables the identification of tumor-specific antigens and
stratification of patients according to their T cell reactivity,
thereby guiding optimal therapy selection. The diversity of the TCR
repertoire is associated with immune escape mechanisms and can
predict potential responsiveness to immunotherapy (239). Furthermore, TCR-based approaches
can be integrated with other modalities, such as ICIs, to amplify
overall antitumor immunity and improve clinical efficacy. This
individualized approach enhances treatment precision and
potentially reduces adverse effects, thereby improving patient
quality of life.
The development of neoantigen vaccines also
shows-considerable potential in personalized NPC therapy. These
vaccines aim to elicit robust immune responses against
tumor-specific neoantigens, facilitating the immune recognition and
elimination of cancer cells. Neoantigen vaccines have been shown to
enhance CD8+ T cell responses and improve tumor immune
surveillance, contributing to prolonged patient survival (239). Ongoing research focuses on
optimizing neoantigen identification and vaccine delivery systems
to enhance immunogenicity and safety (240). Incorporating genomic data allows
for the design of tailored vaccines that further augment
therapeutic efficacy and immune responsiveness (241).
The microbiome plays a key role in modulating host
immunity, particularly in the context of NPC immunotherapy. The
composition of the microbiome influences treatment responses,
including sensitivity to immune checkpoint blockade (239). Modulation of the microbiota may
enhance T cell function and strengthen antitumor immunity. Specific
microbial communities have been reported to promote proliferation
and activation of immune effector cells, thereby improving
treatment outcomes (242). Future
studies should explore interventions such as dietary modifications,
probiotics or other microbiome-targeted therapies to optimize
microbial composition and enhance survival in patients with NPC
(243). Consequently, the majority
of proposed therapeutic strategies discussed in the present review
should be considered hypothesis-generating, and their clinical
applicability requires validation in well-designed prospective
studies.
Despite increasing insight into immune
dysregulation in EBV-associated NPC, the clinical translation of
these findings remains limited. ICIs, particularly targeting the
PD-1/PD-L1 axis, have demonstrated clinical benefit in a subset of
patients; however, primary resistance and heterogeneous response
rates remain important therapeutic challenges. These limitations
suggest that immune checkpoint blockade alone may be insufficient
to fully reverse the complex and dynamic immune editing processes
occurring in this disease.
One major therapeutic bottleneck is the persistent
antigenic pressure imposed by EBV (244). Chronic EBV antigen presentation
sustains prolonged TCR signaling, which drives continuous immune
activation and ultimately promotes T cell dysfunction. Current
immunotherapeutic strategies largely focus on downstream
reinvigoration of exhausted T cells, while the upstream viral
antigen burden is rarely addressed. Future therapeutic approaches
may benefit from targeting EBV latency programs, modulating viral
antigen processing and presentation, or integrating antiviral
strategies to reduce chronic immune stimulation and delay or
prevent T cell exhaustion.
A second important limitation lies in the metabolic
exhaustion of TILs. Accumulating evidence indicates that
mitochondrial dysfunction, redox imbalance and impaired glucose and
lipid metabolism fundamentally constrain T cell effector capacity
and persistence within the TME. However, metabolic fitness is not
routinely evaluated in clinical practice, and biomarkers reflecting
T cell metabolic status are lacking. Future research should
prioritize the identification of metabolic signatures predictive of
immunotherapy responsiveness and explore metabolic interventions,
such as modulation of mitochondrial function or antioxidant
pathways, as rational combinatorial strategies alongside ICIs.
TLS remodeling further complicates therapeutic
decision-making. Although the presence of TLSs has been associated
with improved outcomes in several types of cancer, their functional
heterogeneity in NPC remains poorly characterized. Emerging
evidence suggests that TLSs may exhibit both immunostimulatory and
immunosuppressive properties depending on their maturation state,
cellular composition and spatial organization. Therefore, simply
promoting TLS formation may not uniformly enhance antitumor
immunity. Future studies should focus on stratifying TLS phenotypes
and defining their functional relevance in NPC, which may inform
patient selection and therapeutic timing.
Collectively, these observations highlight the need
for stage-adapted and mechanism-guided therapeutic strategies.
Rather than targeting isolated immune components, effective
clinical translation may require combinatorial approaches that
simultaneously address persistent antigen exposure, metabolic
exhaustion and immune architectural remodeling. Longitudinal
studies integrating immune profiling, metabolic assessment and
spatial transcriptomic analysis of TLS dynamics may be important in
translating mechanistic insights into precision immunotherapy for
EBV-associated NPC.
Conclusion
NPC, which is characterized by high incidence and
mortality rates, according to the latest GLOBOCAN 2022 estimates,
nasopharyngeal carcinoma accounts for 120,000-130,000 new cases and
more than 70,000 mortalities worldwide each year, with a marked
geographic predominance in East and Southeast Asia, has long
attracted attention due to the complexity of its immune evasion
mechanisms (245). The present
review has comprehensively examined the intricate molecular
networks involved in T cell fate decisions during immune editing,
with particular focus on the interplay among aberrant EBV antigen
presentation, T cell metabolic exhaustion and TLS remodeling. These
key processes not only deepen understanding of NPC immune escape
but also lay a foundation for future immunotherapeutic
strategies.
First, as a pivotal oncogenic factor in NPC, EBV
considerably disrupts antigen presentation, thereby impairing T
cell activation and function. Detailed analysis of EBV antigens
reveals multiple immune evasion mechanisms within the TME,
including altered antigen presentation and secretion of
immunosuppressive factors, collectively diminishing T cell
recognition of tumor cells. Insight into these processes enhances
comprehension of NPC pathogenesis and offers valuable guidance for
vaccine development targeting EBV-associated antigens.
Second, T cell metabolic exhaustion represents
another key aspect. Within the metabolically hostile TME, T cells
endure profound metabolic stress that compromises their effector
functions. Emerging evidence suggests that metabolic reprogramming
is important for sustaining T cell survival and activity in tumors.
Thus, future research should prioritize strategies to modulate T
cell metabolic states to restore their antitumor functionality and
improve immunotherapy efficacy.
Furthermore, remodeling of TLSs in the TME has
garnered increasing interest (91,112).
TLS formation not only affects T cell aggregation and activation
but may also play a key role in tumor progression. Investigating
the mechanisms underlying TLS development and their specific
functions in NPC may offer more comprehensive therapeutic
insights.
In summary, immune evasion in NPC is governed by a
complex and interdependent network. Future studies should focus on
dynamically dissecting these processes to facilitate the design of
combination therapies that concurrently target multiple immune
escape pathways. Additionally, establishing robust predictive
models for treatment responses may aid the development of
personalized precision immunotherapy, ultimately enhancing patient
survival and quality of life. By integrating multidisciplinary
findings, a more holistic understanding of NPC immune evasion can
be achieved and clinically actionable frameworks to drive advances
in NPC treatment can be provided.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Foundation of Natural
Science of China (grant no. 81660171) and the Hainan Provincial
Natural Science Foundation of China (grant no. 820RC627). This work
was supported by innovative Research Project for Graduate Students
in Hainan Provincial Universities (grant no. Qhys2023-429).
Availability of data and materials
Not applicable.
Authors' contributions
LZ, SD and DC wrote the manuscript and created the
figures. DC collected and organized literature. LZ and SD proofread
the manuscript. ZM and BC are responsible for the study design,
research fields, drafting and finalizing the manuscript. All
authors read and approved the final version of the manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Han S, Tay JK, Loh CJL, Chu AJM, Yeong
JPS, Lim CM and Toh HC: Epstein-barr virus epithelial Cancers-A
comprehensive understanding to drive novel therapies. Front
Immunol. 12:7342932021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jin S, Li R, Chen MY, Yu C, Tang LQ, Liu
YM, Li JP, Liu YN, Luo YL, Zhao Y, et al: Single-cell
transcriptomic analysis defines the interplay between tumor cells,
viral infection, and the microenvironment in nasopharyngeal
carcinoma. Cell Res. 30:950–965. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mahajan S, Bongaerts M, Hardillo J, Tsang
A, Lo KW, Kortleve D, Ma B and Debets R: Transcriptomics of
Epstein-Barr virus aids to the classification of T-cell evasion in
nasopharyngeal carcinoma. Curr Opin Immunol. 83:1023352023.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lo AK, Dawson CW, Lung HL, Wong KL and
Young LS: The Role of EBV-Encoded LMP1 in the NPC tumor
microenvironment: From Function to therapy. Front Oncol.
11:6402072021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li X, Guo Y, Xiao M and Zhang W: The
immune escape mechanism of nasopharyngeal carcinoma. FASEB J.
37:e230552023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li W, Lv S, Liu G, Lu N, Jiang Y, Liang H,
Xia W, Xiang Y, Xie C and He J: Epstein-Barr virus DNA
seropositivity links distinct tumoral heterogeneity and immune
landscape in nasopharyngeal carcinoma. Front Immunol.
14:11240662023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lin Q, Zhou Y, Ma J, Han S, Huang Y, Wu F,
Wang X, Zhang Y, Mei X and Ma L: Single-cell analysis reveals the
multiple patterns of immune escape in the nasopharyngeal carcinoma
microenvironment. Clin Transl Med. 13:e13152023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cao Z, Pan C, Liu Z, Quan Q, Li M, Huang
Y, Liang C, Chen Y, Fan T, Chen P, et al: Co-targeting MRPS7-23
synergistically enhances cisplatin efficacy to suppress
nasopharyngeal carcinoma growth and metastasis. Int J Biol Sci.
22:970–994. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yarza R, Bover M, Agulló-Ortuño MT and
Iglesias-Docampo LC: Current approach and novel perspectives in
nasopharyngeal carcinoma: The role of targeting proteasome
dysregulation as a molecular landmark in nasopharyngeal cancer. J
Exp Clin Cancer Res. 40:2022021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yan C, Zheng L, Jiang S, Yang H, Guo J,
Jiang LY, Li T, Zhang H, Bai Y, Lou Y, et al: Exhaustion-associated
cholesterol deficiency dampens the cytotoxic arm of antitumor
immunity. Cancer Cell. 41:1276–1293.e11. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cai M, Wang Y, Ma H, Yang L and Xu Z:
Advances and challenges in immunotherapy for locally advanced
nasopharyngeal carcinoma. Cancer Treat Rev. 131:1028402024.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Qing D, Lu Z and Lu H: Long-term immune
checkpoint inhibitor therapy in a patient with metastatic
nasopharyngeal carcinoma: A case report. Front Immunol.
16:15858442025. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu D, Liu L, Zhao X, Zhang X, Chen X, Che
X and Wu G: A comprehensive review on targeting diverse immune
cells for anticancer therapy: Beyond immune checkpoint inhibitors.
Crit Rev Oncol Hematol. 210:1047022025. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sun L, Liu C and Peng J: Specific immune
responses and oncolytic effects induced by EBV LMP2A-Armed modified
Ankara-Vaccinia virus vectored vaccines in nasopharyngeal cancer.
Pharmaceutics. 17:522025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cui X and Snapper CM: Epstein barr virus:
Development of vaccines and immune cell therapy for EBV-Associated
diseases. Front Immunol. 12:7344712021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhao GX, Bu GL, Liu GF, Kong XW, Sun C, Li
ZQ, Dai DL, Sun HX, Kang YF, Feng GK, et al: mRNA-based vaccines
targeting the T-cell Epitope-rich domain of epstein barr virus
latent proteins elicit robust Anti-tumor immunity in mice. Adv Sci
(Weinh). 10:e23021162023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lechner M, Schartinger VH, Steele CD, Nei
WL, Ooft ML, Schreiber LM, Pipinikas CP, Chung GT, Chan YY, Wu F,
et al: Somatostatin receptor 2 expression in nasopharyngeal cancer
is induced by Epstein Barr virus infection: Impact on prognosis,
imaging and therapy. Nat Commun. 12:1172021. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Prasad M, Ponnalagu S, Zeng Q, Luu K, Lang
SM, Wong HY, Cheng MS, Wu M, Mallilankaraman K, Sobota RM, et al:
Epstein-Barr virus-induced ectopic CD137 expression helps
nasopharyngeal carcinoma to escape immune surveillance and enables
targeting by chimeric antigen receptors. Cancer Immunol Immunother.
71:2583–2596. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Argirion I, Pfeiffer RM, Proietti C,
Coghill AE, Yu KJ, Middeldorp JM, Sarathkumara YD, Hsu WL, Chien
YC, Lou PJ, et al: Comparative analysis of the humoral immune
response to the EBV proteome across EBV-related malignancies.
Cancer Epidemiol Biomarkers Prev. 32:687–696. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhao B: Epstein-Barr Virus B cell growth
transformation: The nuclear events. Viruses. 15:8322023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jin J, Sun T, Zhang M, Cheng J, Gu J,
Huang L, Xiao M, Zhou J and Luo H: EBV-Encoded MicroRNA-BART17-3p
targets DDX3X and promotes EBV infection in EBV-Associated
T/Natural Killer-cell lymphoproliferative diseases. Open Forum
Infect Dis. 10:ofad5162023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Deobagkar-Lele M, Crawford G, Crockford
TL, Back J, Hodgson R, Bhandari A, Bull KR and Cornall RJ: B cells
require DOCK8 to elicit and integrate T cell help when antigen is
limiting. Sci Immunol. 9:eadd48742024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gan CP, Kok SY, Lee BKB, Zulaziz N, Cheong
SC, Savelyeva N and Lim KP: Cancer vaccine overcomes immune evasion
of nasopharyngeal carcinoma by restoring MHC-I through
transcriptional regulation of NLRC5. J Transl Med. 23:14142025.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang H, Fan S, Zhan Y, Xu Y, Du Y, Luo J,
Zang H, Peng S and Wang W: Targeting EGFR-binding protein SLC7A11
enhancing antitumor immunity of T cells via inducing MHC-I antigen
presentation in nasopharyngeal carcinoma. Cell Death Dis.
16:212025. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Warner BE and Shair KH: High IgG titers
against EBV glycoprotein 42 correlate with lower risk of
nasopharyngeal carcinoma. J Clin Invest. 135:e1892072025.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang S, Chen R, Wu Y, Song X, Peng X and
Chen M: Fluorinated polyethyleneimine vectors with serum resistance
and adjuvant effect to deliver LMP2 mRNA vaccine for nasopharyngeal
carcinoma therapy. Acta Biomater. 192:340–352. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Guo M, Duan X, Peng X, Jin Z, Huang H,
Xiao W, Zheng Q, Deng Y, Fan N, Chen K and Song X: A lipid-based
LMP2-mRNA vaccine to treat nasopharyngeal carcinoma. Nano Res.
16:5357–5367. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Campos Canesso MC, de Castro TBR,
Nakandakari-Higa S, Lockhart A, Luehr J, Bortolatto J, Parsa R,
Esterházy D, Lyu M, Liu TT, et al: Identification of
antigen-presenting cell-T cell interactions driving immune
responses to food. Science. 387:eado50882025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Xie Y, Wu Q, Jia Y, Cheng L, Tian Y, Yang
X, Wang W, Xu W, Zhao Z and Wei Y: Enhanced antigen presentation by
macrophages promotes human atherosclerosis progression: Therapeutic
implications. Eur Heart J. Nov 11–2025.(Epub ahead of print). doi:
10.1093/eurheartj/ehaf878. View Article : Google Scholar
|
|
30
|
30. He Y, Mohapatra G, Asokan S, Nobs SP
and Elinav E: Microbiome modulation of antigen presentation in
tolerance and inflammation. Curr Opin Immunol. 91:1024712024.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lin M, Zhang XL, You R, Yang Q, Zou X, Yu
K, Liu YP, Zou RH, Hua YJ, Huang PY, et al: Neoantigen landscape in
metastatic nasopharyngeal carcinoma. Theranostics. 11:6427–6444.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yang H, Zeng X, Chen X, Cai S, Li Y, Xu W,
Lai J and Qiu S: The key role of DLX6 in nasopharyngeal carcinoma:
Metastasis, angiogenesis and tumor immune mechanism. Front Immunol.
16:15225802025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sharma S, Mehta NU, Sauer T, Rollins LA,
Dittmer DP and Rooney CM: Cotargeting EBV lytic as well as latent
cycle antigens increases T-cell potency against lymphoma. Blood
Adv. 8:3360–3371. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang S, Zhou Y, Liu Z, Wang Y, Zhou X,
Chen H, Zhang X, Chen Y, Feng Q, Ye X, et al: Immunosequencing
identifies signatures of T cell responses for early detection of
nasopharyngeal carcinoma. Cancer Cell. 43:1423–1441.e10. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Massey J, Artuz C, Dyer Z, Jackson K, Khoo
M, Visweswaran M, Withers B, Moore J, Ma D and Sutton I:
Diversification and expansion of the EBV-reactive cytotoxic T
lymphocyte repertoire following autologous haematopoietic stem cell
transplant for multiple sclerosis. Clin Immunol. 254:1097092023.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rudin CM, Balli D, Lai WV, Richards AL,
Nguyen E, Egger JV, Choudhury NJ, Sen T, Chow A, Poirier JT, et al:
Clinical benefit from immunotherapy in patients with SCLC is
associated with tumor capacity for antigen presentation. J Thorac
Oncol. 18:1222–1232. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Perales-Linares R, Leli NM, Mohei H, Beghi
S, Rivera OD, Kostopoulos N, Giglio A, George SS, Uribe-Herranz M,
Costabile F, et al: Parkin deficiency suppresses antigen
presentation to promote tumor immune evasion and immunotherapy
resistance. Cancer Res. 83:3562–3576. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang X, Chai Y, Quan Y, Wang J, Song J,
Zhou W, Xu X, Xu H, Wang B and Cao X: NPM1 inhibits tumoral antigen
presentation to promote immune evasion and tumor progression. J
Hematol Oncol. 17:972024. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Roberts A, Bentley L, Tang T, Stewart F,
Pallini C, Juvvanapudi J, Wallace GR, Cooper AJ, Scott A, Thickett
D, et al: Ex vivo modelling of PD-1/PD-L1 immune checkpoint
blockade under acute, chronic, and exhaustion-like conditions of
T-cell stimulation. Sci Rep. 11:40302021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fu XH, Guan RY, Huang Z, Li Y, Lu G, Mou
WW and Du J: From multiple myeloma to acute myeloid leukemia: A
case report of a 61-year-old woman after 8 years of chemotherapy
and immunotherapy. Recent Pat Anticancer Drug Discov. 19:396–401.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lu F, Bai X, Lv Z and Zhang W: Immune
archetypes TIME classification system for nasopharyngeal carcinoma:
A new direction for precision immunotherapy. Biochim Biophys Acta
Rev Cancer. 1880:1894872025. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yeo ELL, Hong BH, Tay SH, Neo J, Ong EHW,
Chow WM, Tan KM, Low KP, Sim AYL, Lu T, et al: Tumor immune
microenvironment delineates progression trajectories of distinct
nasopharyngeal carcinoma phenotypes. Cell Rep Med. 6:1021432025.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cammann C, Israel N, Frentzel S, Jeron A,
Topfstedt E, Schüler T, Simeoni L, Zenker M, Fehling HJ, Schraven
B, et al: T cell-specific constitutive active SHP2 enhances T cell
memory formation and reduces T cell activation. Front Immunol.
13:9586162022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tang K, Zhao M, Wu YH, Wu Q, Wang S, Dong
Y, Yu B, Song Y and Liu HM: Structure-based design, synthesis and
biological evaluation of aminopyrazines as highly potent,
selective, and cellularly active allosteric SHP2 inhibitors. Eur J
Med Chem. 230:1141062022. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhao Y and Jiang L: Targeting SHP1 and
SHP2 to suppress tumors and enhance immunosurveillance. Trends Cell
Biol. 35:667–677. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wang Y, Zhang W, Shi R, Luo Y, Feng Z,
Chen Y, Zhang Q, Zhou Y, Liang J, Ye X, et al: Identification of
HLA-A*11:01 and A*02:01-Restricted EBV Peptides Using HLA
Peptidomics. Viruses. 16:6692024. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Chen Y, Ouyang D, Wang Y, Pan Q, Zhao J,
Chen H, Yang X, Tang Y, Wang Q, Li Y, et al: EBV promotes
TCR-T-cell therapy resistance by inducing CD163+M2 macrophage
polarization and MMP9 secretion. J Immunother Cancer.
12:e0083752024. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Bhandarkar V, Dinter T and Spranger S:
Architects of immunity: How dendritic cells shape CD8+ T cell fate
in cancer. Sci Immunol. 10:eadf47262025. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wankhede D, Halama N, Kloor M, Edelmann D,
Brenner H and Hoffmeister M: Prognostic Value of CD8+ T cells at
the invasive margin is comparable to the immune score in
nonmetastatic colorectal cancer: A prospective multicentric cohort
study. Clin Cancer Res. 31:1711–1718. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Soeratram TTD, Biesma HD, Egthuijsen JMP,
Meershoek-Klein Kranenbarg E, Hartgrink HH, van de Velde CJH,
Mookhoek A, van Dijk E, Kim Y, Ylstra B, et al: Prognostic Value of
T-Cell density in the tumor center and outer margins in gastric
cancer. Mod Pathol. 36:1002182023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wu R, Horimoto Y, Oshi M, Benesch MGK,
Khoury T, Takabe K and Ishikawa T: Emerging measurements for
tumor-infiltrating lymphocytes in breast cancer. Jpn J Clin Oncol.
54:620–629. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hassan WA, ElBanna AK, Noufal N, El-Assmy
M, Lotfy H and Ali RI: Significance of tumor-associated
neutrophils, lymphocytes, and neutrophil-to-lymphocyte ratio in
non-invasive and invasive bladder urothelial carcinoma. J Pathol
Transl Med. 57:88–94. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Sarnaik AA, Hwu P, Mulé JJ and
Pilon-Thomas S: Tumor-infiltrating lymphocytes: A new hope. Cancer
Cell. 42:1315–1318. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Del M, Illac C, Morisseau M, Angeles MA,
Ducassou A, Betrian S, Bataillon G, Ferron G, Chantalat E, Gabiache
E and Martinez A: Intraepithelial tumor-infiltrating lymphocytes
shape loco-regional PET/CT spread of locally advanced cervical
cancer. Int J Gynecol Cancer. 34:490–496. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Pauken KE and Wherry EJ: Overcoming T cell
exhaustion in infection and cancer. Trends Immunol. 36:265–276.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zinselmeyer BH, Heydari S, Sacristán C,
Nayak D, Cammer M, Herz J, Cheng X, Davis SJ, Dustin ML and
McGavern DB: PD-1 promotes immune exhaustion by inducing antiviral
T cell motility paralysis. J Exp Med. 210:757–774. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Juneja VR, McGuire KA, Manguso RT, LaFleur
MW, Collins N, Haining WN, Freeman GJ and Sharpe AH: PD-L1 on tumor
cells is sufficient for immune evasion in immunogenic tumors and
inhibits CD8 T cell cytotoxicity. J Exp Med. 214:895–904. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wang M: Canagliflozin disrupts T cell
activation. Nat Rev Nephrol. 19:4782023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lao TD and Le TAH: Hypermethylation of
genes on chromosome 3p as a biomarker for nasopharyngeal carcinoma
diagnosis: A Vietnamese case-control study. Int J Biol Markers.
39:201–208. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Beckers D, Jainarayanan AK, Dustin ML and
Capera J: T cell resistance: On the mechanisms of T cell
Non-activation. Immune Netw. 24:e422024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Law SC, Hoang T, O'Rourke K, Tobin JWD,
Gunawardana J, Loo-Oey D, Bednarska K, Merida de Long L, Sabdia MB,
Hapgood G, et al: Successful treatment of Epstein-Barr
virus-associated primary central nervous system lymphoma due to
post-transplantation lymphoproliferative disorder, with ibrutinib
and third-party Epstein-Barr virus-specific T cells. Am J
Transplant. 21:3465–3471. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Hao B, Lin S, Liu H, Xu J, Chen L, Zheng
T, Zhang W, Dang Y, Reiter RJ, Li C, et al: Baicalein tethers
CD274/PD-L1 for autophagic degradation to boost antitumor immunity.
Autophagy. 21:917–933. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Saddozai UAK, Lu Z, Dong S, Khawar MB, Fan
Z, Cai L, Guo X, Akbar MU, Khattak S, Sun H and Wang Y: Prognostic
value of metal-based ferroptosis and cuproptosis genes and score in
lower grade gliomas. Front Immunol. 16:16080772025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Shi H, Chen S and Chi H: Immunometabolism
of CD8+ T cell differentiation in cancer. Trends Cancer.
10:610–626. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Cele S, Karim F, Lustig G, San JE,
Hermanus T, Tegally H, Snyman J, Moyo-Gwete T, Wilkinson E,
Bernstein M, et al: SARS-CoV-2 evolved during advanced HIV disease
immunosuppression has Beta-like escape of vaccine and Delta
infection elicited immunity. medRxiv. Dec 7–2021.(Epub ahead of
print). doi: 10.1101/2021.09.14.21263564.
|
|
66
|
Zagatto AM, Bishop DJ, Antunes BM, Beck
WR, Malta ES, de Poli RAB, Cavaglieri CR, Chacon-Mikahil MPT and
Castro A: Impacts of high-intensity exercise on the metabolomics
profile of human skeletal muscle tissue. Scand J Med Sci Sports.
32:402–413. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Chen Z and Zhang X: The role of metabolic
reprogramming in kidney cancer. Front Oncol. 14:14023512024.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Yang Y, Fan D, Zheng BH and Zhou ST:
Latest findings on the function of immune metabolism in tumor
immunity. Sichuan Da Xue Xue Bao Yi Xue Ban. 54:497–504. 2023.(In
Chinese). PubMed/NCBI
|
|
69
|
Jonker PB and Muir A: Metabolic ripple
effects-deciphering how lipid metabolism in cancer interfaces with
the tumor microenvironment. Dis Model Mech. 17:dmm0508142024.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zhu M, Hu Y, Gu Y, Lin X, Jiang X, Gong C
and Fang Z: Role of amino acid metabolism in tumor immune
microenvironment of colorectal cancer. Am J Cancer Res. 15:233–247.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Borrego-Yaniz G, Márquez A,
Estupiñán-Moreno E, Terrón-Camero LC, González-Gay MA, Castañeda S,
Guggino G, Saadoun D, Lio P, Fontana S, et al: Genome-Wide DNA
methylation study reveals specific signatures in the affected
arterial tissue of patients with giant cell arteritis. Arthritis
Rheumatol. 78:385–393. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Li C, Yuan Y, Jiang X and Wang Q:
Epigenetic regulation of CD8+ T cell exhaustion: Recent advances
and update. Front Immunol. 16:17000392025. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhang M, Hu T, Ma T, Huang W and Wang Y:
Epigenetics and environmental health. Front Med. 18:571–596. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Dutta A, Venkataganesh H and Love PE:
Epigenetic regulation of T cell development. Int Rev Immunol.
42:82–90. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Mi T, Soerens AG, Alli S, Kang TG,
Vasandan AB, Wang Z, Vezys V, Kimura S, Iacobucci I, Baylin SB, et
al: Conserved epigenetic hallmarks of T cell aging during immunity
and malignancy. Nat Aging. 4:1053–1063. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Alves E, Taifour S, Chee J, Nowak AK,
Gaudieri S and Blancafort P: Reprogramming the anti-tumor immune
response via CRISPR genetic and epigenetic editing. Mol Ther
Methods Clin Dev. 21:592–606. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Zhong RQ, Tian H and Gao XJ: Epigenetic
regulation in tooth development. Zhonghua Kou Qiang Yi Xue Za Zhi.
56:288–293. 2021.(In Chinese). PubMed/NCBI
|
|
78
|
Tong J, Zhang W, Chen Y, Yuan Q, Qin NN
and Qu G: The emerging role of RNA modifications in the regulation
of antiviral innate immunity. Front Microbiol. 13:8456252022.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Li J, Wang Y, Shen W, Zhang Z, Su Z, Guo
X, Pei P, Hu L, Liu T, Yang K and Guo L: Mitochondria-modulating
liposomes reverse Radio-resistance for colorectal cancer. Adv Sci
(Weinh). 11:e24008452024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
An J, Guo R, Liu M, Hu H and Zhang H:
Multi-modal Ca2+ nanogenerator via reversing T cell exhaustion for
enhanced chemo-immunotherapy. J Control Release. 372:715–727. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Qian L, Zhang F, Yin M and Lei Q: Cancer
metabolism and dietary interventions. Cancer Biol Med. 19:163–174.
2021.PubMed/NCBI
|
|
82
|
Di Donna A, Masala S, Muto G, Marcia S,
Giordano F and Muto M: Metabolic bone diseases: Recommendations for
interventional radiology. Semin Musculoskelet Radiol. 28:641–650.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Liu H, Zeng L, Yang Y, Huang Z, Guo C,
Huang L, Niu X, Zhang C and Wang H: Bcl-3 regulates the function of
Th17 cells through raptor mediated glycolysis metabolism. Front
Immunol. 13:9297852022. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Jeong DI, Hao Q, Lee SY, Kim S, Karmakar
M, Chu S, Park M and Cho HJ: Cellulose nanocrystal-annealed
hydrogel system for local chemo-metabolic therapy of melanoma. J
Control Release. 377:324–338. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Zhao H and Li Y: Cancer metabolism and
intervention therapy. Mol Biomed. 2:52021. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Li Z, Li X, Ai S, Liu S and Guan W:
Glucose metabolism Intervention-facilitated nanomedicine therapy.
Int J Nanomedicine. 17:2707–2731. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Mathis BJ, Tanaka K and Hiramatsu Y:
Metabolically healthy obesity: Are interventions useful? Curr Obes
Rep. 12:36–60. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Pan X, Wang J, Zhang L, Li G and Huang B:
Metabolic plasticity of T cell fate decision. Chin Med J (Engl).
137:762–775. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Liu Y, Wu Z, Li Y, Chen Y, Zhao X, Wu M
and Xia Y: Metabolic reprogramming and interventions in
angiogenesis. J Adv Res. 70:323–338. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Chen B, Zhang Y, Dai S, Zhou P, Luo W,
Wang Z, Chen X, Cheng P, Zheng G and Ren J: Molecular
characteristics of primary pulmonary lymphoepithelioma-like
carcinoma based on integrated genomic analyses. Signal Transduct
Target Ther. 6:62021. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Wang H, Zhan Y, Luo J, Wang W and Fan S:
Unveiling immune resistance mechanisms in nasopharyngeal carcinoma
and emerging targets for antitumor immune response: Tertiary
lymphoid structures. J Transl Med. 23:382025. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Li JP, Wu CY, Chen MY, Liu SX, Yan SM,
Kang YF, Sun C, Grandis JR, Zeng MS and Zhong Q: PD-1+CXCR5-CD4+
Th-CXCL13 cell subset drives B cells into tertiary lymphoid
structures of nasopharyngeal carcinoma. J Immunother Cancer.
9:e0021012021. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Deng M, Liu X, Jiang Y, Luo R, Xu L, Zhang
X, Su J, Xu C and Hou Y: Tertiary lymphoid structures' pattern and
prognostic value in primary adenocarcinoma of jejunum and ileum.
World J Surg Oncol. 22:2612024. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Edmonds NL, Gradecki SE, Katyal P, Lynch
KT, Stowman AM, Gru AA, Engelhard VH, Slingluff CL Jr and Mauldin
IS: Tertiary lymphoid structures in desmoplastic melanoma have
increased lymphocyte density, lymphocyte proliferation, and immune
cross talk with tumor when compared to non-desmoplastic melanomas.
Oncoimmunology. 12:21644762023. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Huang Y, Xiao Z, Wu S, Zhang X, Wang J and
Huangfu X: Biochemical transformation and bioremediation of
thallium in the environment. Sci Total Environ. 953:1760282024.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Xiaoxu D, Min X and Chengcheng C: Immature
central tumor tertiary lymphoid structures are associated with
better prognosis in non-small cell lung cancer. BMC Pulm Med.
24:1552024. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wu S, Pan J, Pan Q, Zeng L, Liang R and Li
Y: Multi-omics profiling and experimental verification of tertiary
lymphoid structure-related genes: Molecular subgroups, immune
infiltration, and prognostic implications in lung adenocarcinoma.
Front Immunol. 15:14532202024. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Fujihara J and Nishimoto N:
Thallium-poisoner's poison: An overview and review of current
knowledge on the toxicological effects and mechanisms. Curr Res
Toxicol. 6:1001572024. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
MacFawn IP, Magnon G, Gorecki G, Kunning
S, Rashid R, Kaiza ME, Atiya H, Ruffin AT, Taylor S, Soong TR, et
al: The activity of tertiary lymphoid structures in high grade
serous ovarian cancer is governed by site, stroma, and cellular
interactions. Cancer Cell. 42:1864–1881.e5. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Wang Y, Lu J, Wu C, Fei F, Chu Z and Lu P:
Clinical markers predict the efficacy of several immune checkpoint
inhibitors in patients with non-small cell lung cancer in China.
Front Immunol. 14:12761072023. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Chen Z, Wang X, Jin Z, Li B, Jiang D, Wang
Y, Jiang M, Zhang D, Yuan P, Zhao Y, et al: Deep learning on
tertiary lymphoid structures in hematoxylin-eosin predicts cancer
prognosis and immunotherapy response. NPJ Precis Oncol. 8:732024.
View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Liu R, Huang X, Yang S, Du W, Chen X and
Li H: Discovery of an independent poor-prognosis subtype associated
with tertiary lymphoid structures in breast cancer. Front Immunol.
15:13645062024. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Teillaud JL, Houel A, Panouillot M,
Riffard C and Dieu-Nosjean MC: Tertiary lymphoid structures in
anticancer immunity. Nat Rev Cancer. 24:629–646. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Tang Z, Bai Y, Fang Q, Yuan Y, Zeng Q,
Chen S, Xu T, Chen J, Tan L, Wang C, et al: Spatial transcriptomics
reveals tryptophan metabolism restricting maturation of
intratumoral tertiary lymphoid structures. Cancer Cell.
43:1025–1044.e14. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Li H, Zhu SW, Zhou JJ, Chen DR, Liu J, Wu
ZZ, Wang WY, Zhang MJ and Sun ZJ: Tertiary Lymphoid Structure
Raises Survival and Immunotherapy in HPV-HNSCC. J Dent Res.
102:678–688. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Peyraud F, Guegan JP, Vanhersecke L,
Brunet M, Teyssonneau D, Palmieri LJ, Bessede A and Italiano A:
Tertiary lymphoid structures and cancer immunotherapy: From bench
to bedside. Med. 6:1005462025. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Xu W, Lu J, Tian X, Ye S, Wei S, Wang J,
Anwaier A, Qu Y, Liu W, Chang K, et al: Unveiling the impact of
tertiary lymphoid structures on immunotherapeutic responses of
clear cell renal cell carcinoma. MedComm (2020). 5:e4612024.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Yang YL, Zhou C, Chen Q, Shen SZ, Li JD,
Wang XL and Wang PR: YAP1/Piezo1 involve in the dynamic changes of
lymphatic vessels in UVR-induced photoaging progress to squamous
cell carcinoma. J Transl Med. 21:8202023. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Chelvanambi M, Fecek RJ, Taylor JL and
Storkus WJ: STING agonist-based treatment promotes vascular
normalization and tertiary lymphoid structure formation in the
therapeutic melanoma microenvironment. J Immunother Cancer.
9:e0019062021. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Vion R, Roulleaux-Dugage M, Flippot R,
Ouali K, Rouanne M, Clatot F, Sellars M, Champiat S, Chaput N,
Massard C and Danlos FX: Induction of tertiary lymphoid structures
in tumor microenvironment to improve anti-tumoral immune checkpoint
blockade efficacy. Eur J Cancer. 225:1155722025. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Zou Q, Liao K, Li G, Huang X, Zheng Y,
Yang G, Luo M, Xue EY, Lan C, Wang S, et al:
Photo-metallo-immunotherapy: Fabricating Chromium-based
nanocomposites to enhance CAR-T cell infiltration and cytotoxicity
against solid tumors. Adv Mater. 37:e24074252025. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Liu Y, Ye SY, He S, Chi DM, Wang XZ, Wen
YF, Ma D, Nie RC, Xiang P, Zhou Y, et al: Single-cell and spatial
transcriptome analyses reveal tertiary lymphoid structures linked
to tumour progression and immunotherapy response in nasopharyngeal
carcinoma. Nat Commun. 15:77132024. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Liu SX, Wu TW, Luo DH, Zhang LL, Zhou L,
Luo YL, Du WT, Huang TT, Jiang S, Zhang Z, et al:
Interferon-responsive HEVs drive tumor tertiary lymphoid structure
formation and predict immunotherapy response in nasopharyngeal
carcinoma. Cell Rep Med. 6:1022002025. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Yuan L, Jia GD, Lv XF, Xie SY, Guo SS, Lin
DF, Liu LT, Luo DH, Li YF, Deng SW, et al: Camrelizumab combined
with apatinib in patients with first-line platinum-resistant or
PD-1 inhibitor resistant recurrent/metastatic nasopharyngeal
carcinoma: A single-arm, phase 2 trial. Nat Commun. 14:48932023.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Ahmed MM, Gebriel MG, Morad EA, Saber IM,
Elwan A, Salah M, Fakhr AE, Shalaby AM and Alabiad MA: Expression
of immune checkpoint regulators, cytotoxic T-Lymphocyte Antigen-4,
and programmed Death-Ligand 1 in Epstein-Barr Virus-associated
nasopharyngeal carcinoma. Appl Immunohistochem Mol Morphol.
29:401–408. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Lim DW, Kao HF, Suteja L, Li CH, Quah HS,
Tan DS, Tan SH, Tan EH, Tan WL, Lee JN, et al: Clinical efficacy
and biomarker analysis of dual PD-1/CTLA-4 blockade in
recurrent/metastatic EBV-associated nasopharyngeal carcinoma. Nat
Commun. 14:27812023. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Nguyen K, Alsaati N, Le Coz C and Romberg
N: Genetic obstacles to developing and tolerizing human B cells.
WIREs Mech Dis. 14:e15542022. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Zhu Z, Ding R, Yu W, Liu Y, Zhou Z and Liu
CY: YAP/TEAD4/SP1-induced VISTA expression as a tumor
cell-intrinsic mechanism of immunosuppression in colorectal cancer.
Cell Death Differ. 32:911–925. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Chen Y, Sun J, Liu J, Wei Y, Wang X, Fang
H, Du H, Huang J, Li Q, Ren G, et al: Aldehyde dehydrogenase
2-mediated aldehyde metabolism promotes tumor immune evasion by
regulating the NOD/VISTA axis. J Immunother Cancer. 11:e0074872023.
View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Kim JY, Kim Y, Woo SY, Kim JO, Kim H, Son
SR, Jang DS and Choi JH: Hydnocarpin, a natural flavonolignan,
induces the ROS-Mediated apoptosis of ovarian cancer cells and
reprograms Tumor-Associated immune cells. Antioxidants (Basel).
14:8462025. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Jeong M and Collins N: Nutritional
modulation of antitumor immunity. Curr Opin Immunol. 87:1024222024.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Spina CS and Drake CG: Mechanisms of
immune modulation by radiation. Semin Radiat Oncol. 31:205–216.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Sun LL, Zhao LN, Sun J, Yuan HF, Wang YF,
Hou CY, Lv P, Zhang HH, Yang G, Zhang NN, et al: Inhibition of USP7
enhances CD8+ T cell activity in liver cancer by suppressing
PRDM1-mediated FGL1 upregulation. Acta Pharmacol Sin. 45:1686–1700.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Zahraeifard S, Xiao Z, So JY, Ahad A,
Montoya S, Park WY, Sornapudi T, Andohkow T, Read A, Kedei N, et
al: Loss of tumor suppressors promotes inflammatory tumor
microenvironment and enhances LAG3+T cell mediated immune
suppression. Nat Commun. 15:58732024. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Murphy EJ, Fehrenbach GW, Abidin IZ,
Buckley C, Montgomery T, Pogue R, Murray P, Major I and Rezoagli E:
Polysaccharides-naturally occurring immune modulators. Polymers
(Basel). 15:23732023. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Posner M: Curative application of
immunotherapy in untreated head and neck and nasopharynx cancer.
JAMA. 333:1584–1585. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Jaiswal AR, Liu AJ, Pudakalakatti S, Dutta
P, Jayaprakash P, Bartkowiak T, Ager CR, Wang ZQ, Reuben A, Cooper
ZA, et al: Melanoma evolves complete immunotherapy resistance
through the acquisition of a hypermetabolic phenotype. Cancer
Immunol Res. 8:1365–1380. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Bernardo M, Tolstykh T, Zhang YA, Bangari
DS, Cao H, Heyl KA, Lee JS, Malkova NV, Malley K, Marquez E, et al:
An experimental model of anti-PD-1 resistance exhibits activation
of TGFß and Notch pathways and is sensitive to local mRNA
immunotherapy. Oncoimmunology. 10:18812682021. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Zhou B, Gao Y, Zhang P and Chu Q: Acquired
resistance to immune checkpoint blockades: The underlying
mechanisms and potential strategies. Front Immunol. 12:6936092021.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Zhang W, Wang M, Ji C, Liu X, Gu B and
Dong T: Macrophage polarization in the tumor microenvironment:
Emerging roles and therapeutic potentials. Biomed Pharmacother.
177:1169302024. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Chen W, Bao L, Ren Q, Zhang Z, Yi L, Lei
W, Yang Z, Lu Y, You B, You Y and Gu M: SCARB1 in extracellular
vesicles promotes NPC metastasis by co-regulating M1 and M2
macrophage function. Cell Death Discov. 9:3232023. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Tang Q, Li L, Ge J, Wang D, Qu H, Wu J,
Wang Q, Peng Z, Mo Y, Wang Y, et al: m6A modification-dependent
upregulation of WNT2 facilitates M2-like macrophage polarization
and perpetuates malignant progression of nasopharyngeal carcinoma.
Oncogene. 44:2730–2745. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Boutilier AJ and Elsawa SF: Macrophage
polarization states in the tumor microenvironment. Int J Mol Sci.
22:69952021. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Saeed AF: Tumor-associated macrophages:
Polarization, immunoregulation, and immunotherapy. Cells.
14:7412025. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Zou Z, Lin H, Li M and Lin B:
Tumor-associated macrophage polarization in the inflammatory tumor
microenvironment. Front Oncol. 13:11031492023. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Sun Z, Xu Y, Shao B, Dang P, Hu S, Sun H,
Chen C, Wang C, Liu J, Liu Y and Hu J: Exosomal circPOLQ promotes
macrophage M2 polarization via activating IL-10/STAT3 axis in a
colorectal cancer model. J Immunother Cancer. 12:e0084912024.
View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Wei B, Pan J, Yuan R, Shao B, Wang Y, Guo
X and Zhou S: Polarization of Tumor-Associated macrophages by
Nanoparticle-loaded escherichia coli combined with immunogenic cell
death for cancer immunotherapy. Nano Lett. 21:4231–4240. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Zhang J, Tang K, Yang Y, Yang D and Fan W:
Advanced nanoprobe strategies for imaging macrophage polarization
in cancer immunology. Research (Wash DC). 8:06222025.PubMed/NCBI
|
|
139
|
Nagatani Y, Funakoshi Y, Suto H, Imamura
Y, Toyoda M, Kiyota N, Yamashita K and Minami H: Immunosuppressive
effects and mechanisms of three myeloid-derived suppressor cells
subsets including monocytic-myeloid-derived suppressor cells,
granulocytic-myeloid-derived suppressor cells, and
immature-myeloid-derived suppressor cells. J Cancer Res Ther.
17:1093–1100. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Bhardwaj V and Ansell SM: Modulation of
T-cell function by myeloid-derived suppressor cells in
hematological malignancies. Front Cell Dev Biol. 11:11293432023.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Zhang W, Li X, Jiang M, Ji C, Chen G,
Zhang Q, Liu P, Zhang R, Ren X, Yu W and Yu J: SOCS3
deficiency-dependent autophagy repression promotes the survival of
early-stage myeloid-derived suppressor cells in breast cancer by
activating the Wnt/mTOR pathway. J Leukoc Biol. 113:445–460. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Wong SW, McCarroll J, Hsu K, Geczy CL and
Tedla N: Intranasal delivery of recombinant S100A8 protein delays
lung cancer growth by remodeling the lung immune microenvironment.
Front Immunol. 13:8263912022. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Zang J, Yang Y, Zheng X, Yang Y, Zhao Y,
Miao Z, Zhang T, Gu J, Liu Y, Yin W, et al: Dynamic tagging to
drive arginine nano-assembly to metabolically potentiate immune
checkpoint blockade therapy. Biomaterials. 292:1219382023.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Zhao Z, Qin J, Qian Y, Huang C, Liu X,
Wang N, Li L, Chao Y, Tan B, Zhang N, et al: FFAR2 expressing
myeloid-derived suppressor cells drive cancer immunoevasion. J
Hematol Oncol. 17:92024. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Bertrand BP, Heim CE, Koepsell SA and
Kielian T: Elucidating granulocytic myeloid-derived suppressor cell
heterogeneity during Staphylococcus aureus biofilm infection. J
Leukoc Biol. 115:620–632. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Genova C, Dellepiane C, Carrega P,
Sommariva S, Ferlazzo G, Pronzato P, Gangemi R, Filaci G, Coco S
and Croce M: Therapeutic implications of tumor microenvironment in
lung cancer: Focus on immune checkpoint blockade. Front Immunol.
12:7994552021. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Qin G, Liu S, Liu J, Hu H, Yang L, Zhao Q,
Li C, Zhang B and Zhang Y: Overcoming resistance to immunotherapy
by targeting GPR84 in myeloid-derived suppressor cells. Signal
Transduct Target Ther. 8:1642023. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Yamamoto S, Kato M, Takeyama Y, Azuma Y,
Yukimatsu N, Hirayama Y, Otoshi T, Yamasaki T, Fujioka M, Gi M, et
al: Irradiation plus myeloid-derived suppressor cell-targeted
therapy for overcoming treatment resistance in immunologically cold
urothelial carcinoma. Br J Cancer. 128:2197–2205. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Liu J, Piranlioglu R, Ye F, Shu K, Lei T
and Nakashima H: Immunosuppressive cells in oncolytic virotherapy
for glioma: Challenges and solutions. Front Cell Infect Microbiol.
13:11410342023. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Martín-Portugués M, Esteban-Sampedro J, de
Mendoza C, Ruiz-Irastorza G, Royuela A, Puente AO, de la
Cruz-Echeandía M, Blanco-Valencia XP, Soriano V and Moreno-Torres
V: Hospitalisations with Cryoglobulin-related diseases in spain
over 25 Years. Liver Int. 45:e701952025. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Bhatt K, Nukovic A, Colombani T and
Bencherif SA: Biomaterial-assisted local oxygenation safeguards the
prostimulatory phenotype and functions of human dendritic cells in
hypoxia. Front Immunol. 14:12783972023. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Kawahara K, Mukai T, Iseki M, Nagasu A,
Nagasu H, Akagi T, Tsuji S, Hiramatsu-Asano S, Ueki Y, Ishihara K,
et al: SH3BP2 deficiency ameliorates murine systemic lupus
erythematosus. Int J Mol Sci. 22:41692021. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Schneble D, El-Gazzar A, Kargarpour Z,
Kramer M, Metekol S, Stoshikj S and Idzko M: Cell-type-specific
role of P2Y2 receptor in HDM-driven model of allergic airway
inflammation. Front Immunol. 14:12090972023. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Wen Y, Rudemiller NP, Zhang J, Lu X, Ren
J, Privratsky JR, Griffiths R, Zhang JJ, Hammer GE and Crowley SD:
C-C motif chemokine receptor 7 exacerbates hypertension through
effects on T lymphocyte trafficking. Hypertension. 75:869–876.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Greene TT, Jo Y, Chiale C, Macal M, Fang
Z, Khatri FS, Codrington AL, Kazane KR, Akbulut E, Swaminathan S,
et al: Metabolic deficiencies underlie reduced plasmacytoid
dendritic cell IFN-I production following viral infection. Nat
Commun. 16:14602025. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Kremer KN, Khammash HA, Miranda AM, Rutt
LN, Twardy SM, Anton PE, Campbell ML, Garza-Ortiz C, Orlicky DJ,
Pelanda R, et al: Liver sinusoidal endothelial cells regulate the
balance between hepatic immunosuppression and immunosurveillance.
Front Immunol. 15:14977882024. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Smilovic D, Rietsche M, Drakew A, Vuksic M
and Deller T: Constitutive tumor necrosis factor (TNF)-deficiency
causes a reduction in spine density in mouse dentate granule cells
accompanied by homeostatic adaptations of spine head size. J Comp
Neurol. 530:656–669. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Nickles E, Xia R, Sun R and Schwarz H:
Methods for generating the CD137L-DC-EBV–VAX anti-cancer vaccine.
Methods Cell Biol. 183:187–202. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Tischer-Zimmermann S, Bonifacius A,
Santamorena MM, Mausberg P, Stoll S, Döring M, Kalinke U, Blasczyk
R, Maecker-Kolhoff B and Eiz-Vesper B: Reinforcement of
cell-mediated immunity driven by tumor-associated Epstein-Barr
virus (EBV)-specific T cells during targeted B-cell therapy with
rituximab. Front Immunol. 14:8789532023. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Huang J, Harris E and Lorch J: Vaccination
as a therapeutic strategy for Nasopharyngeal carcinoma. Oral Oncol.
135:1060832022. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Nickles E, Dharmadhikari B, Yating L,
Walsh RJ, Koh LP, Poon M, Tan LK, Wang LZ, Ang Y, Asokumaran Y, et
al: Dendritic cell therapy with CD137L-DC-EBV–VAX in locally
recurrent or metastatic nasopharyngeal carcinoma is safe and
confers clinical benefit. Cancer Immunol Immunother. 71:1531–1543.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Liao L, Liu Y, Li X, Jiang Z, Jiang Z and
Yao J: Dual-regulated biomimetic nanocomposites for promoted tumor
photodynamic immunotherapy. ACS Appl Mater Interfaces.
17:20919–20931. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Yang H, Xiong Z, Heng X, Niu X, Wang Y,
Yao L, Sun L, Liu Z and Chen H: Click-Chemistry-mediated cell
membrane glycopolymer engineering to potentiate dendritic cell
vaccines. Angew Chem Int Ed Engl. 63:e2023157822024. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Najafi S and Mortezaee K: Advances in
dendritic cell vaccination therapy of cancer. Biomed Pharmacother.
164:1149542023. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
van Willigen WW, Bloemendal M,
Boers-Sonderen MJ, de Groot JWB, Koornstra RHT, van der Veldt AAM,
Haanen JBAG, Boudewijns S, Schreibelt G, Gerritsen WR, et al:
Response and survival of metastatic melanoma patients treated with
immune checkpoint inhibition for recurrent disease on adjuvant
dendritic cell vaccination. Oncoimmunology. 9:17388142020.
View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Wang D, Liu R, Ding Y, Pei Q, Sun T, Shao
X, Jiang M, Liu J, Huang J, Yan Z, et al: Whole-body vibration
protects against chronic high-altitude hypoxic bone loss by
regulating the nitric oxide/HIF-1α axis in osteoblasts. FASEB J.
39:e703852025. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Wu Q, You L, Nepovimova E, Heger Z, Wu W,
Kuca K and Adam V: Hypoxia-inducible factors: Master regulators of
hypoxic tumor immune escape. J Hematol Oncol. 15:772022. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Cheng X, Wang P, Lyu H, Lee Y, Yoon J and
Dong H: Targeting the hypoxia signaling pathway with nanomedicine
to reverse immunotherapy resistance. Cancer Drug Resist.
8:462025.PubMed/NCBI
|
|
169
|
Schulz MC, Kopf M and Gekle M: Crosstalk
with renal proximal tubule cells drives acidosis-induced
inflammatory response and dedifferentiation of fibroblasts via
p38-singaling. Cell Commun Signal. 22:1482024. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Li K, Niu L, Zhang X, Li T, Zhou X, Wang
L, Han J and Wang Z: NEAT1 is a therapeutic target for reversing
T-cell exhaustion in bladder cancer. J Immunother Cancer.
13:e0137332025. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Chen Y, Gao J, Ma M, Wang K, Liu F, Yang
F, Zou X, Cheng Z and Wu D: The potential role of CMC1 as an
immunometabolic checkpoint in T cell immunity. Oncoimmunology.
13:23449052024. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Fowler EA, Farias Amorim C, Mostacada K,
Yan A, Amorim Sacramento L, Stanco RA, Hales ED, Varkey A, Zong W,
Wu GD, et al: Neutrophil-mediated hypoxia drives pathogenic CD8+ T
cell responses in cutaneous leishmaniasis. J Clin Invest.
134:e1779922024. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Knopf P, Stowbur D, Hoffmann SHL, Hermann
N, Maurer A, Bucher V, Poxleitner M, Tako B, Sonanini D,
Krishnamachary B, et al: Acidosis-mediated increase in
IFN-γ-induced PD-L1 expression on cancer cells as an immune escape
mechanism in solid tumors. Mol Cancer. 22:2072023. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Babl N, Decking SM, Voll F, Althammer M,
Sala-Hojman A, Ferretti R, Korf C, Schmidl C, Schmidleithner L,
Nerb B, et al: MCT4 blockade increases the efficacy of immune
checkpoint blockade. J Immunother Cancer. 11:e0073492023.
View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Nicolini A and Ferrari P: Involvement of
tumor immune microenvironment metabolic reprogramming in colorectal
cancer progression, immune escape, and response to immunotherapy.
Front Immunol. 15:13537872024. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Huang Y, Qin G, Cui T, Zhao C, Ren J and
Qu X: A bimetallic nanoplatform for STING activation and CRISPR/Cas
mediated depletion of the methionine transporter in cancer cells
restores anti-tumor immune responses. Nat Commun. 14:46472023.
View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Madden MZ, Ye X, Chi C, Fisher EL, Wolf
MM, Needle GA, Bader JE, Patterson AR, Reinfeld BI, Landis MD, et
al: Differential effects of glutamine inhibition strategies on
antitumor CD8 T Cells. J Immunol. 211:563–575. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Xiao J, Liu T, Zeng F and Zhang J: New
insights into T cell metabolism in liver cancer: From mechanism to
therapy. Cell Death Discov. 11:1182025. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Xu C, Jiang H, Feng LJ, Jiang MZ, Wang YL
and Liu SJ: Christensenella minuta interacts with multiple gut
bacteria. Front Microbiol. 15:13010732024. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Wang R, Liu Z, Fan Z and Zhan H: Lipid
metabolism reprogramming of CD8+ T cell and therapeutic
implications in cancer. Cancer Lett. 567:2162672023. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Wang J, Jia W, Zhou X, Ma Z, Liu J and Lan
P: CBX4 suppresses CD8+ T cell antitumor immunity by reprogramming
glycolytic metabolism. Theranostics. 14:3793–3809. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Silva C, Andrade N, Rodrigues I, Ferreira
AC, Soares ML and Martel F: The pro-proliferative effect of
interferon-γ in breast cancer cell lines is dependent on
stimulation of ASCT2-mediated glutamine cellular uptake. Life Sci.
286:1200542021. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Yang Y, Chen Q, Fan S, Lu Y, Huang Q, Liu
X and Peng X: Glutamine sustains energy metabolism and alleviates
liver injury in burn sepsis by promoting the assembly of
mitochondrial HSP60-HSP10 complex via SIRT4 dependent protein
deacetylation. Redox Rep. 29:23123202024. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Zhang Y, Hu Z, Zhang J, Ren C and Wang Y:
Dual-target inhibitors of indoleamine 2, 3 dioxygenase 1 (Ido1): A
promising direction in cancer immunotherapy. Eur J Med Chem.
238:1145242022. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Fujiwara Y, Kato S, Nesline MK, Conroy JM,
DePietro P, Pabla S and Kurzrock R: Indoleamine 2,3-dioxygenase
(IDO) inhibitors and cancer immunotherapy. Cancer Treat Rev.
110:1024612022. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Kim M and Tomek P: Tryptophan: A rheostat
of cancer immune escape mediated by immunosuppressive enzymes IDO1
and TDO. Front Immunol. 12:6360812021. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Wang M, Liu Y, Li Y, Lu T, Wang M, Cheng
Z, Chen L, Wen T, Pan M and Hu G: Tumor Microenvironment-responsive
nanoparticles enhance IDO1 blockade immunotherapy by remodeling
metabolic immunosuppression. Adv Sci (Weinh). 12:e24058452025.
View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Hu C, Liu Y, Cao W, Li N, Gao S, Wang Z
and Gu F: Efficacy and mechanism of a biomimetic nanosystem
carrying doxorubicin and an IDO inhibitor for treatment of advanced
Triple-negative breast cancer. Int J Nanomedicine. 19:507–526.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Li J, Yang H, Zhang L, Zhang S and Dai Y:
Metabolic reprogramming and interventions in endometrial carcinoma.
Biomed Pharmacother. 161:1145262023. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Qiu MZ, Wang C, Wu Z, Zhao Q, Zhao Z,
Huang CY, Wu W, Yang LQ, Zhou ZW, Zheng Y, et al: Dynamic
single-cell mapping unveils Epstein-Barr virus-imprinted T-cell
exhaustion and on-treatment response. Signal Transduct Target Ther.
8:3702023. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Pogorelyy MV, Kirk AM, Adhikari S,
Minervina AA, Sundararaman B, Vegesana K, Brice DC, Scott ZB; SJTRC
Study Team, ; Thomas PG: TIRTL-seq: Deep, quantitative, and
affordable paired TCR repertoire sequencing. bioRxiv. Oct
31–2024.doi: 10.1101/2024.09.16.613345.
|
|
192
|
Zhang Q and Xu M: EBV-induced T-cell
responses in EBV-specific and nonspecific cancers. Front Immunol.
14:12509462023. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Young J, Asaoka M, Ghasemi F, Chida K, Roy
AM, Yan L, Hakamada K and Takabe K: The American Joint Committee on
Cancer (AJCC) Breast Cancer Staging, Eighth Edition, is more
Reflective of Cancer Biology than the Seventh Edition. Ann Surg
Oncol. 32:3268–3277. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Ando M: Induced pluripotent stem
cell-derived rejuvenated cytotoxic T lymphocyte therapy for
Epstein-Barr virus-associated lymphomas: Application to clinical
practice. Rinsho Ketsueki. 63:1310–1315. 2022.(In Japanese).
PubMed/NCBI
|
|
195
|
Ning XH, Liu J, Hu L, Lu HY, Liu Q and
Wang DH: Effects of NOX4 on radiosensitivity of nasopharyngeal
carcinoma cells by activating the PI3K/AKT pathway. Zhonghua Er Bi
Yan Hou Tou Jing Wai Ke Za Zhi. 55:514–519. 2020.(In Chinese).
PubMed/NCBI
|
|
196
|
Hang W, Guo HM, Wu QL, Yan H, Liu G and
Gao M: MicroRNA-203a-3p affects the biological characteristics of
nasopharyngeal carcinoma by targeting vascular endothelial growth
factor-C. J Physiol Pharmacol. Jan 21–2021.(Epub ahead of print).
doi: 10.26402/jpp.2021.4.12. PubMed/NCBI
|
|
197
|
Mouquet H: Humoral immunity in HIV-1
post-treatment controllers. Curr Opin HIV AIDS. 20:80–85. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
198
|
La Civita E, Zannella C, Brusa S, Romano
P, Schettino E, Salemi F, Carrano R, Gentile L, Punziano A, Lagnese
G, et al: BNT162b2 Elicited an Efficient Cell-Mediated Response
against SARS-CoV-2 in kidney transplant recipients and common
variable immunodeficiency patients. Viruses. 15:16592023.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Satou A and Nakamura S: EBV-positive
B-cell lymphomas and lymphoproliferative disorders: Review from the
perspective of immune escape and immunodeficiency. Cancer Med.
10:6777–6785. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Gou Q, Yan B, Duan Y, Guo Y, Qian J, Shi J
and Hou Y: Ubiquitination of CD47 Regulates innate Anti-Tumor
immune response. Adv Sci (Weinh). 12:e24122052025. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Wang Y, Peng Z, Wang Y, Yang Y, Fan R, Gao
K, Zhang H, Xie Z and Jiang W: Immune Microenvironment change and
involvement of circular RNAs in TIL cells of recurrent
nasopharyngeal carcinoma. Front Cell Dev Biol. 9:7222242021.
View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Senft D: Programming immune escape. Nat
Rev Cancer. 24:2942024. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Liang Y, Yang H, Li Q, Zhao P, Li H, Zhang
Y, Cai W, Ma X and Duan Y: Novel biomimetic dual-mode nanodroplets
as ultrasound contrast agents with potential ability of precise
detection and photothermal ablation of tumors. Cancer Chemother
Pharmacol. 86:405–418. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Wang Y, Guo L, Fan G, Han Y, Zhang Q, Ren
L, Zhang H, Wang G, Zhang X, Huang T, et al: Impact of
corticosteroids on initiation and half-year durability of humoral
response in COVID-19 survivors. Chin Med J Pulm Crit Care Med.
2:48–55. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Wu X, Zhang J, Deng Z, Sun X, Zhang Y,
Zhang C, Wang J, Yu X and Yang G: Bacteria-based biohybrids for
remodeling adenosine-mediated immunosuppression to boost
radiotherapy-triggered antitumor immune response. Biomaterials.
316:1230002025. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Florêncio KGD, Edson EA, Fernandes KSDS,
Luiz JPM, Pinto FDCL, Pessoa ODL, Cunha FQ, Machado-Neto JA and
Wilke DV: Chromomycin A5 induces bona fide immunogenic cell death
in melanoma. Front Immunol. 13:9417572022. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Ding Q, Tang W, Li X, Ding Y, Chen X, Cao
W, Wang X, Mo W, Su Z, Zhang Q and Guo H: Mitochondrial-targeted
brequinar liposome boosted mitochondrial-related ferroptosis for
promoting checkpoint blockade immunotherapy in bladder cancer. J
Control Release. 363:221–234. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Wang C, Zai W, Zhao K, Li Y, Shi B, Wu M,
Zhou X, Kozlowski M, Zhang X, Fang Z and Yuan Z: Potential role of
liver-resident CD3+ macrophages in HBV clearance in a mouse
hepatitis B model. JHEP Rep. 7:1013232025. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Gu Y, Lian Y, Zheng Q, Huang Z, Gu L, Bi
Y, Li J and Huang Y, Wu Y, Chen L and Huang Y: Association among
cytokine profiles of innate and adaptive immune responses and
clinical-virological features in untreated patients with chronic
hepatitis B. BMC Infect Dis. 20:5092020. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Kunst C, Schmid S, Michalski M, Tümen D,
Buttenschön J, Müller M and Gülow K: The influence of gut
microbiota on oxidative stress and the immune system. Biomedicines.
11:13882023. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Avila-Ponce de León U, Vázquez-Jiménez A,
Matadamas-Guzman M, Pelayo R and Resendis-Antonio O:
Transcriptional and microenvironmental landscape of macrophage
transition in cancer: A boolean analysis. Front Immunol.
12:6428422021. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
El Saftawy E, Aboulhoda BE, Alghamdi MA,
Abd Elkhalek MA and AlHariry NS: Heterogeneity of modulatory immune
microenvironment in bladder cancer. Tissue Cell. 93:1026792025.
View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Huang S: Tuning the rheostat of immune
gene translation. Stress Biol. 3:72023. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Chen Y, Qiu Y, Chen M, Huang L, Lin X, Qiu
X, Wei Y and Gao L: Transition from secukinumab to adalimumab in
COVID-19-induced psoriasis flare-up treatment: A case report. Clin
Case Rep. 11:e80472023. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Fang S, Jiang M, Jiao J, Zhao H, Liu D,
Gao D, Wang T, Yang Z and Yuan H: Unraveling the
ROS-Inflammation-immune balance: A new perspective on aging and
disease. Aging Dis. 17:305–325. 2025.PubMed/NCBI
|
|
216
|
Jiang XN, Yu BH, Yan WH, Lee J, Zhou XY
and Li XQ: Epstein-Barr virus-positive diffuse large B-cell
lymphoma features disrupted antigen capture/presentation and
hijacked T-cell suppression. Oncoimmunology. 9:16833462020.
View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Huo S, Luo Y, Deng R, Liu X, Wang J, Wang
L, Zhang B, Wang F, Lu J and Li X: EBV-EBNA1 constructs an
immunosuppressive microenvironment for nasopharyngeal carcinoma by
promoting the chemoattraction of Treg cells. J Immunother Cancer.
8:e0015882020. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Kang K, Lin X, Chen P, Liu H, Liu F, Xiong
W, Li G, Yi M, Li X, Wang H and Xiang B: T cell exhaustion in human
cancers. Biochim Biophys Acta Rev Cancer. 1879:1891622024.
View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Wetzel TJ, Erfan SC, Figueroa LD, Wheeler
LM and Ananieva EA: Crosstalk between arginine, glutamine, and the
branched chain amino acid metabolism in the tumor microenvironment.
Front Oncol. 13:11865392023. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Sharma S, Bhatnagar R and Gaur D:
Complement evasion strategies of human pathogenic bacteria. Indian
J Microbiol. 60:283–296. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Wang H, Tsung A, Mishra L and Huang H:
Regulatory T cell: A double-edged sword from
metabolic-dysfunction-associated steatohepatitis to hepatocellular
carcinoma. EBioMedicine. 101:1050312024. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Gong L, Luo J, Zhang Y, Yang Y, Li S, Fang
X, Zhang B, Huang J, Chow LK, Chung D, et al: Nasopharyngeal
carcinoma cells promote regulatory T cell development and
suppressive activity via CD70-CD27 interaction. Nat Commun.
14:19122023. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Liu X, Shen H, Zhang L, Huang W, Zhang S
and Zhang B: Immunotherapy for recurrent or metastatic
nasopharyngeal carcinoma. NPJ Precis Oncol. 8:1012024. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Mai H, Li L, Xin X, Jiang Z, Tang Y, Huang
J, Lei Y, Chen L, Dong T and Zhong X: Prediction of immunotherapy
response in nasopharyngeal carcinoma: A comparative study using
MRI-based radiomics signature and programmed cell death ligand 1
expression score. Eur Radiol. 35:4403–4414. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Yu Z, Wang Q, Wang Z, Liu S, Xia T, Duan
C, Liu Y, Ding X, Chen S, Yu T, et al: Lachnoclostridium intestinal
flora is associated with immunotherapy efficacy in nasopharyngeal
carcinoma. Head Neck. 47:269–281. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Piao Y, Yang Y, Wu S and Han L:
Toripalimab plus cetuximab combined with radiotherapy in a locally
advanced platinum-based chemotherapy-insensitive nasopharyngeal
carcinoma patient: A case report. Front Oncol. 14:13832502024.
View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Peng Y, Yan H, Mei W, Zhang P and Zeng C:
Combining radiotherapy with immunotherapy in cervical cancer: Where
do we stand and where are we going? Curr Treat Options Oncol.
24:1378–1391. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Mukherjee D, Romano E, Walshaw R, Zeef
LAH, Banyard A, Kitcatt SJ, Cheadle EJ, Tuomela K, Pendharkar S,
Al-Deka A, et al: Reprogramming the immunosuppressive tumor
microenvironment results in successful clearance of tumors
resistant to radiation therapy and anti-PD-1/PD-L1. Oncoimmunology.
12:22230942023. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Wang J, Gai J, Zhang T, Niu N, Qi H,
Thomas DL II, Li K, Xia T, Rodriguez C, Parkinson R, et al:
Neoadjuvant radioimmunotherapy in pancreatic cancer enhances
effector T cell infiltration and shortens their distances to tumor
cells. Sci Adv. 10:eadk18272024. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Saylor PJ, Kozin SV, Matsui A, Goldberg
SI, Aoki S, Shigeta K, Mamessier E, Smith MR, Michaelson MD, Lee RJ
and Duda DG: The radiopharmaceutical radium-223 has
immunomodulatory effects in patients and facilitates
anti-programmed death receptor-1 therapy in murine models of bone
metastatic prostate cancer. Radiother Oncol. 192:1100912024.
View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Tan JW and Hu JR: Fever with atypical
lymphocytosis: Pearls and pitfalls in Epstein-Barr virus serology.
BMJ Case Rep. 16:e2500812023. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Zapata L, Caravagna G, Williams MJ,
Lakatos E, AbdulJabbar K, Werner B, Chowell D, James C, Gourmet L,
Milite S, et al: Immune selection determines tumor antigenicity and
influences response to checkpoint inhibitors. Nat Genet.
55:451–460. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Ren Y, Okazaki T, Ngamsnae P, Hashimoto H,
Ikeda R, Honkura Y, Suzuki J and Izumi SI: Anatomy and function of
the lymphatic vessels in the parietal pleura and their plasticity
under inflammation in mice. Microvasc Res. 148:1045462023.
View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Yeo YH, Liang J, Lauzon M, Luu M,
Noureddin M, Ayoub W, Kuo A, Sankar K, Gong J, Hendifar A, et al:
Immunotherapy and transarterial radioembolization combination
treatment for advanced hepatocellular carcinoma. Am J
Gastroenterol. 118:2201–2211. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Zheng Y, Wang X and Huang M: Metabolic
regulation of CD8+ T cells: From mechanism to therapy. Antioxid
Redox Signal. 37:1234–1253. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Koda S, Hu J, Ju X, Sun G, Shao S, Tang
RX, Zheng KY and Yan J: The role of glutamate receptors in the
regulation of the tumor microenvironment. Front Immunol.
14:11238412023. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Sun H, Li S, Wang Q, Luo C, Zhong L, Wan
G, Li Z, Zhao G, Bu X, Zeng M and Feng G: Formyl peptide enhances
cancer immunotherapy by activating antitumoral neutrophils, and T
cells. Biomed Pharmacother. 175:1166702024. View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Wu H, Zhao X, Hochrein SM, Eckstein M,
Gubert GF, Knöpper K, Mansilla AM, Öner A, Doucet-Ladevèze R,
Schmitz W, et al: Mitochondrial dysfunction promotes the transition
of precursor to terminally exhausted T cells through
HIF-1α-mediated glycolytic reprogramming. Nat Commun. 14:68582023.
View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Jiang C, Feng D, Zhang Y, Yang K, Hu X and
Xie Q: SCAT8/miR-125b-5p axis triggers malignant progression of
nasopharyngeal carcinoma through SCARB1. BMC Mol Cell Biol.
24:152023. View Article : Google Scholar : PubMed/NCBI
|
|
240
|
Xu H, Liu J, Zhang Y, Zhou Y, Zhang L,
Kang J, Ning C, He Z and Song S: KIF23, under regulation by
androgen receptor, contributes to nasopharyngeal carcinoma
deterioration by activating the Wnt/β-catenin signaling pathway.
Funct Integr Genomics. 23:1162023. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Qiu Q, Yang X, Li X, Ren Y and Huang N:
Viral IL-10 can promote the proliferation, migration and invasion
of nasopharyngeal carcinoma cells and inhibit their apoptosis.
Discov Oncol. 16:1992025. View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Chen P, Liu B, Xia X, Huang P and Zhao J:
Current progress in immunotherapy of nasopharyngeal carcinoma. Am J
Cancer Res. 13:1140–1147. 2023.PubMed/NCBI
|
|
243
|
De Boer RJ and Yates AJ: Modeling T cell
fate. Annu Rev Immunol. 41:513–532. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Feng X, Meng M, Li H, Gao Y, Song W, Di R,
Li Z, Zhang X and Zhang M: T-cell dysfunction in natural
killer/T-cell lymphoma. Oncoimmunology. 12:22125322023. View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Liu H, Tang L, Li Y, Xie W, Zhang L, Tang
H, Xiao T, Yang H, Gu W, Wang H and Chen P: Nasopharyngeal
carcinoma: Current views on the tumor microenvironment's impact on
drug resistance and clinical outcomes. Mol Cancer. 23:202024.
View Article : Google Scholar : PubMed/NCBI
|



















