Introduction
For decades, transurethral resection of the prostate
(TURP) has been regarded as the definitive surgical treatment for
benign prostatic hyperplasia (BPH) (1,2).
TURP, along with suprapubic enucleation techniques, remains the
gold standard for relieving bladder outlet obstruction in clinical
practice (3).
However, despite its widespread success, 5-10% of
patients undergoing BPH surgery experience severe post-operative
complications, most commonly urinary incontinence, followed by
reoperation, recatheterization, urinary tract infections and
perioperative bleeding (4,5).
Early post-operative incontinence has been reported
in up to 30-40% of cases (6),
underscoring a persistent challenge in optimizing functional
outcomes following TURP. The pathophysiological mechanisms
underlying post-TURP incontinence remain incompletely understood.
It is generally accepted that conventional TURP involves the
resection of the prostatic apex, a region intimately associated
with the external urethral sphincter, which plays a critical role
in passive urinary control (7).
The fundamental rationale for prostatic apex
preservation is that maintaining a greater length of the urethral
sphincter complex may protect more sphincteric muscle fibers,
thereby preserving continence (8).
Active continence is maintained by the levator ani muscle complex
acting on the prostatic apex and membranous urethra (7), while the external urethral sphincter,
located predominantly within the prostate between the apex and the
verumontanum (9-11),
provides essential closure function.
The preservation of the Denonvilliers' fascia, a
tendinous structure extending from the prostate base to its apex,
is also deemed to be vital, as it serves as a supportive fulcrum
for both the prostate and the urethra (11,12).
Moreover, Van der Poel et al (13) and Hoyland et al (14) demonstrated that the innervation of
the prostatic apex and the urethral sphincter is intricately
interconnected, suggesting that the maintenance of the prostatic
urethra during surgery enhances post-operative continence
outcomes.
Recently, several controlled clinical trials have
investigated the efficacy of urethral mucosa preservation at the
prostatic apex in the surgical management of BPH (15-18).
However, individual studies have reported inconsistent results, and
to date, to the best of our knowledge, no systematic review or
meta-analysis has comprehensively compared the outcomes of
apex-preserving versus apex-resecting techniques.
Therefore, the present study aimed to systematically
review and quantitatively synthesize the available evidence on the
clinical efficacy of prostatic apex preservation during BPH
surgery, with particular attention to urinary continence and
perioperative outcomes.
Data and methods
Search strategy
The present systematic review and meta-analysis
adhered strictly to the Preferred Reporting Items for Systematic
Reviews and Meta-Analyses (PRISMA) guidelines (19) and was prospectively registered in
PROSPERO (CRD420250642661).
A comprehensive electronic search was conducted from
database inception to January, 2025 across PubMed, Google Scholar
and the Cochrane Library. Search terms included combinations of
‘prostatic apex preservation’, ‘benign prostatic hyperplasia’ and
‘outcomes’.
Additional manual searches of reference lists and
clinical trial registries were performed to identify any relevant
studies not indexed in the databases. The complete search algorithm
is detailed in Table SI.
Inclusion and exclusion criteria
Study selection was guided by the Population,
Intervention, Comparator, Outcome, and Study Design (PICOS)
framework.
The inclusion criteria were the following: i)
Population: Patients diagnosed with BPH, without restriction by age
or comorbidities; ii) intervention: Surgical procedures that
preserved the prostatic apex; iii) comparator: Conventional
surgical approaches involving prostatic apex dissection; iv)
primary outcome: Post-operative urinary incontinence rate; v)
secondary outcomes: Intraoperative blood loss, volume of prostate
tissue resected, International Prostate Symptom Score (IPSS),
quality of life (QoL), peak urinary flow rate (Qmax) and duration
of the surgery; vi) study design: Randomized controlled trials
(RCTs) and non-randomized controlled clinical studies.
The exclusion criteria included the following:
Studies not involving BPH or human participants, non-interventional
or observational studies (e.g., reviews, editorials and case
reports), non-English publications and studies for which full-text
data were unavailable. The full PICOS schema used for eligibility
assessment is summarized in Table
SII.
Data extraction
Two independent reviewers performed data extraction
using a standardized template. Discrepancies were resolved through
discussion and consensus. The following data were extracted from
each eligible study: First author, publication year and journal,
study design and sample size, mean age of the participants,
post-operative urinary incontinence rate, intraoperative blood
loss, Qmax, IPSS, QoL score and prostate volume resected. When
necessary, corresponding authors were contacted to clarify
incomplete or ambiguous data.
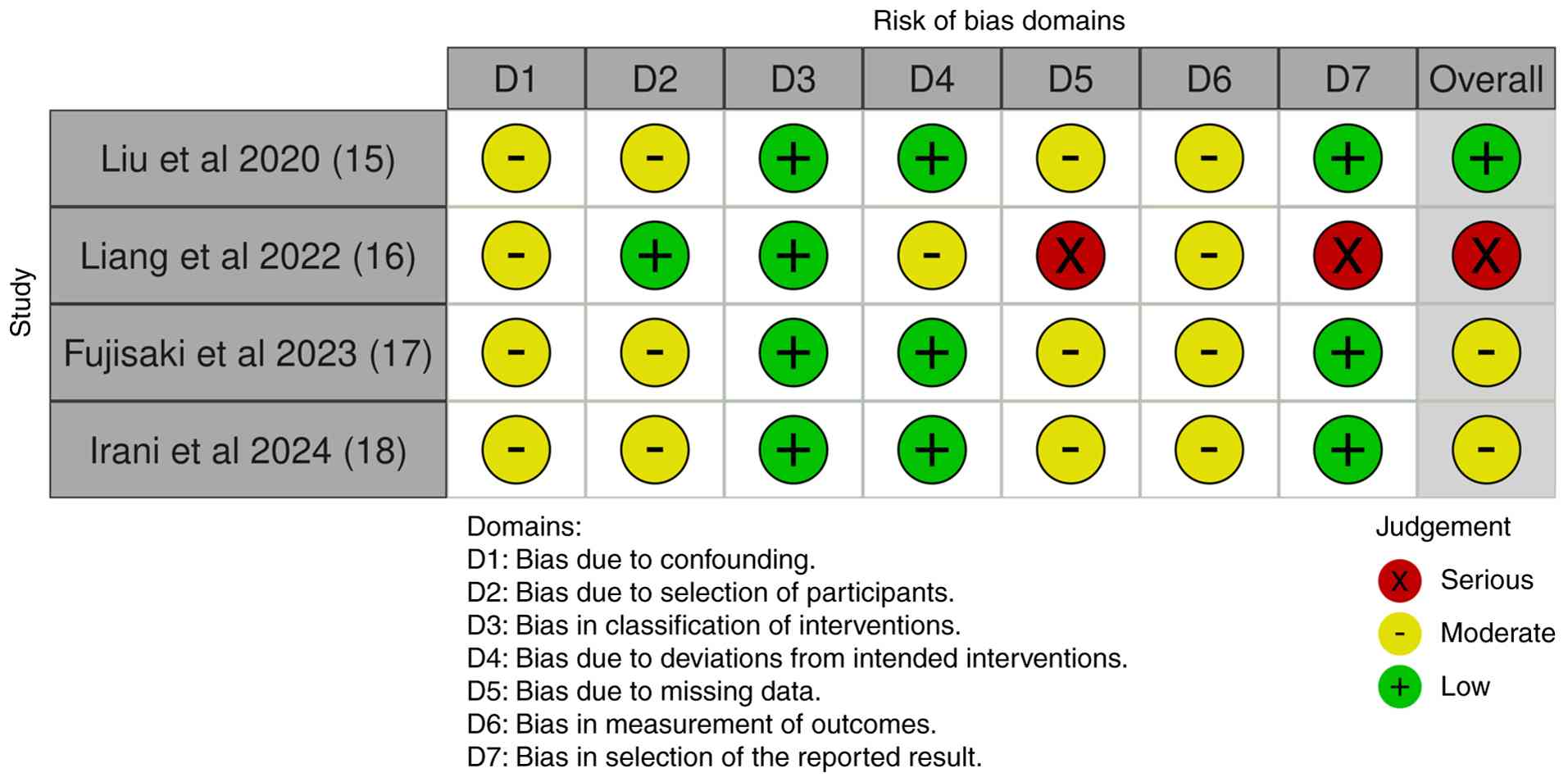
Risk of bias assessment
The methodological quality of the included non-RCTs
was evaluated using the Risk of Bias in Non-randomized Studies of
Interventions (ROBINS-I) tool (20). In total, four studies met the
inclusion criteria and were assessed in detail (15-18).
The results of the ROBINS-I assessment are presented in Fig. 1.
Overall, studies demonstrated a low-to-moderate to
moderate-to-severe risk of bias, primarily due to missing outcome
data and selective reporting. Specifically, Liu and Yang (15) and Fujisaki et al (17) exhibited a low-to-moderate risk,
Liang et al (16) had a
moderate-to-severe risk due to incomplete data and selective
outcome reporting, while Irani et al (18) was rated low-to-moderate.
Statistical analysis
All statistical analyses were conducted using Review
Manager (RevMan) version 5.4(21).
For continuous variables, outcomes are expressed as the mean
difference (MD) with 95% confidence intervals (CIs). For
dichotomous outcomes, the relative risk (RR) with 95% CI was
calculated. Heterogeneity was evaluated using the Chi-squared
(χ2) test and quantified by the I2 statistic.
An I2 value >50% indicated substantial heterogeneity,
and a P-value <0.05 was considered to indicate a statistically
significant difference. The random effects model was used for the
present study as a small number of studies and clinical
heterogeneity are expected (21,22).
Results
Study selection and
characteristics
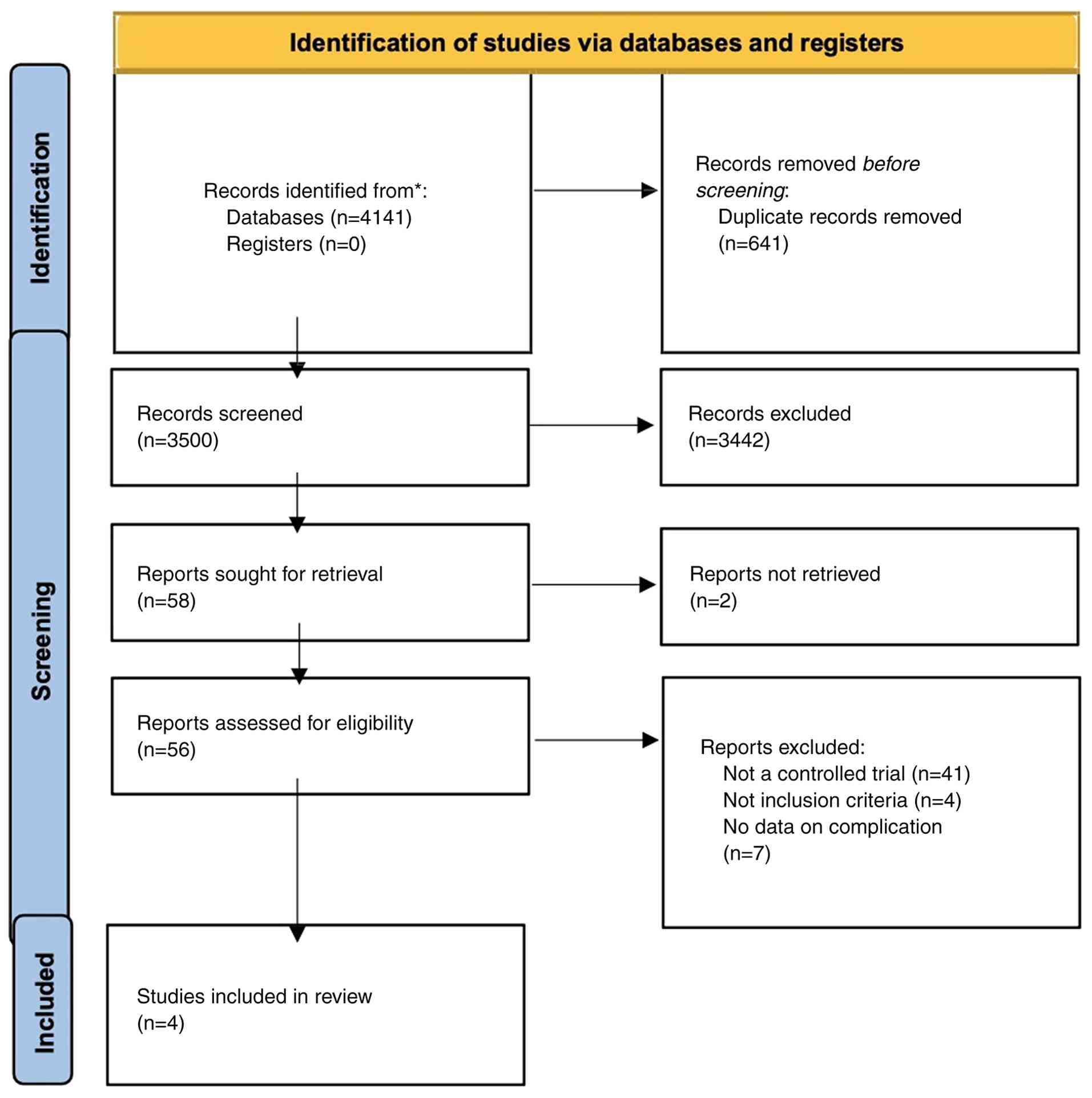
A total of 4,141 studies were identified via the
databases. A total of 641 duplicate studies were detected.
Following deduplication, 3,500 records were filtered. Subsequently,
3,442 were removed as they were not related to apex preservation.
No automation tools were used in the screening part. The remaining
58 studies were refiltered for retrieval. A total of two studies
were not retrieved for full text, and out of the 56 studies that
were retrieved, 41 studies were excluded as they were not
controlled trials, four studies were not related to the inclusion
criteria, and seven studies did not cover the complication. Thus,
four studies were included (15-18).
Of these four studies, 638 participants were identified. The PRISMA
flow chart (23) reflects the
identification of studies via the databases and registers,
rescreening and reassessment of the qualified studies (Fig. 2).
Of the total 638 participants, 452 were in the
prostatic apex preservation group and 186 were in the apex
dissection group. The characteristics of the studies included in
the present systematic review are presented in Table I.
 | Table ICharacteristics of the studies
included in the present systematic review and meta-analysis. |
Table I
Characteristics of the studies
included in the present systematic review and meta-analysis.
| Authors, year of
publication | Study | No. of
participants | Country | Study period | Study design | Outcome and
follow-up | (Refs.) |
|---|
| Liu and Yang,
2020 | Comparison of the
transurethral resection of the prostate by traditional versus
preserved urethral mucosa of the prostatic apex | 40 (preserved
group) 40 (dissection group) | China | 2015-2016 | Single center
prospective trial with mean age were 73 years for the APUMP and 75
years for no-APUMP group | Urinary function
with 1 month of follow-up and surgical indicator (surgery time,
intraoperative blood loss, gram of excised prostate, and
incontinence rate) | (15) |
| Liang et al,
2022 | Clinical study on
the application of preserved urethral mucosa at the prostatic apex
in transurethral plasma kinetic resection of the prostate | 45 (preserved
group) 45 (dissection group) | China | 2018-2021 | Single center
prospective study with mean age were 69.3 years for the APUMP and
68.6 years for no- APUMP group | Urinary function
with 1 and 3 months of follow-up and surgical indicator (surgery
time, intraoperative blood loss, gram of excised prostate, and
incontinence rate) | (16) |
| Fujisaki et
al, 2023 | Use of the anterior
prostatic mucosa preservation technique during holmium laser
enucleation of the prostate can reduce postoperative stress urinary
incontinence | 340 (preserved
group) 75 (dissection group) | Japan | 2018 | Single center
retrospective study performed by two surgeons with mean age were 75
years for the APUMP and 73 years for no-APUMP group | Urinary function
with 1 month of follow-up and surgical indicator (enucleation time,
incontinence rate, and gram of excised prostate) | (17) |
| Irani et al,
2024 | The impact of
urethral mucosa preservation of prostatic apex during monopolar
transurethral resection of the prostate on postoperative function
outcomes: a retrospective comparative study | 27 (preserved
group) 26 (dissection group) | Iran | 2018-2020 | Single center
retrospective cross sectional study mean age were 69 years for the
APUMP and 67 years for no-APUMP group | Urinary function
with 6 months of follow-up and surgical indicator (surgery time,
intraoperative blood loss, gram of excised prostate, and
incontinence rate) | (18) |
The characteristics of outcomes from each study,
such as the duration of surgery, incontinence rate, intraoperative
blood loss, prostate volume excised, IPSS, QoL and Qmax are
presented in Table II.
 | Table IIOutcomes of the included studies. |
Table II
Outcomes of the included studies.
| | Outcome | |
|---|
| Authors, year of
publication | Age, years | Duration of
surgery | Urine
incontinence | Intra operative
blood loss | Gram of prostate
excised | IPSS | QoL | Qmax | (Refs.) |
|---|
| Liu and Yang,
2020 | A: 75±8.3 B: 73±1
0.5 | A: 65±185 B:
50±16.5 | A: 9 (7 recovered
in 1 week and 2 cases recovered in 3 weeks) B: 0 | A: 280±33 B:
190±35 | - | A: 5±1.2 B: 4.9±
1.1 | A: 1.3±0.9 B:
1.1±5 | A :22.1±5 B:
21.8±4.5 | (15) |
| Liang et al,
2022 | A: 68.6 ±8.22 B:
69.27±6.15 | A: 53.87±17.48 B:
4.11±14.18 | A: 7 (4 cases
recovered in 1 week and 4 cases recovered in 3 weeks) B: 0 | A: 85.2 7±34.0 B:
68.78±27.05 | A: 46.56±18.44 B:
47.62±18.38 | A: 6.51±1.04 B:
6.18±1.24 | A: 1.56±0.55 B:
1.51±0.51 | A: 19.92 ±1.3 B:
20.08±1.17 | (16) |
| Fujisaki et
al, 2023 | A: 73±11.25 B:
75±8.25 | A: 46.5±43.25 B:
33±31.5 | A: 11/75(14.7%) B:
14/340 (4.1%) | - | A: 35±4.25 B:
34.5±57 | - | - | A: 9.9±10.1 B:
10.5±14 | (17) |
| Irani et al,
2024 | A: 67.7±9.12 B:
69.8±8.47 | A: 40.59±5.2 B:
33.84±4.44 | A: 6 (4 were healed
within 1 week and two within 3 months) B: 0 | A: 305±63.4 B: 212.
5±65 | - | A: 7.59±3.23 B:
9.44±3.71 | - | A: 18.3±7.99 B:
16.37±3.96 | (18) |
Quality of evidence
The Grading of Assessment, Development, and
Evaluation (GRADE) approach was used to assess the overall evidence
concerning individual outcomes. The GRADEpro tool was applied to
evaluate the certainty of the evidence (26,27).
The summary assessment, development and evaluation of the findings
according to the GRADE guidelines for the included studies
(outcomes on urine incontinence and other outcomes that are
moderate-quality evidence) are presented in Table III.
 | Table IIISummary of the findings according to
the GRADE guidelines for included studies. |
Table III
Summary of the findings according to
the GRADE guidelines for included studies.
| Certainty
assessment | No. of
patients | Effect | |
|---|
| No. of studies | Study design | Risk of bias | Inconsistency | Indirectness | Imprecision | Other
considerations | Preserved urethral
mucosa method | Non-preserved
urethral mucosa method | Relative (95%
CI) | Absolute (95%
CI) | Certainty | Importance |
|---|
| Surgery time |
| 4 | Non-randomized
studies |
Seriousa | Not serious | Not serious |
Seriousb | None | 452 | 186 | - | MD 8.14 lower
(10.39 lower to 5.88 lower) | ⨁⨁��
Lowa,b | Important |
| Urine
incontinence |
| 4 | Non-randomized
studies |
Seriousa | Not serious | Not serious |
Seriousb | None | 14/452 (3.1%) | 33/186 (17.7%) | RR 0.16 (0.08 to
0.33) | 149 fewer 1,000 per
(from 163 fewer to 119 fewer) | ⨁⨁��
Lowa,b | Critical |
| Intra operative
blood loss |
| 3 | Non-randomized
studies |
Seriousa | Not serious | Not serious |
Seriousb | None | 112 | 111 | - | MD 50.7 lower
(60.01 lower to 41.39 lower) | ⨁⨁ ��
Lowa,b | Important |
| IPSS |
| 3 | Non-randomized
studies |
Seriousa | Not serious | Not serious |
Seriousb | None | 112 | 111 | - | MD 0.15 lower (0.49
lower to 0.19 higher) | ⨁⨁��
Lowa,b | Important |
| QoL |
| 2 | Non-randomized
studies |
Seriousa | Not serious | Not serious |
Seriousb | None | 85 | 85 | - | MD 0.3 lower (0.5
lower to 0.11 lower) | ⨁⨁��
Lowa,b | Not important |
| Qmax |
| 4 | Non-randomized
studies |
Seriousa | Not serious | Not serious |
Seriousb | None | 452 | 186 | - | MD 0.11 higher
(0.38 lower to 0.59 higher) | ⨁⨁��
Lowa,b | Critical |
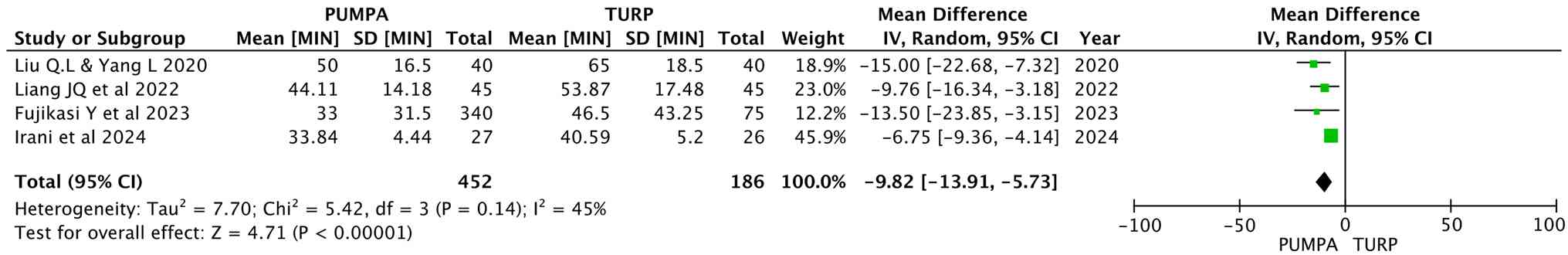
The duration of surgery between the
two groups
All studies analyzed the duration of surgery,
involving 638 participants (452 in the prostatic apex preservation
group and 186 in the apex dissection group). Random effects were
used, and the results revealed that the surgery duration of the
preserving technique was significantly shorter than that of the
non-preserving technique (MD, -9.82; 95% CI, -13.91 to -5.73;
P<0.00001) (Fig. 3).
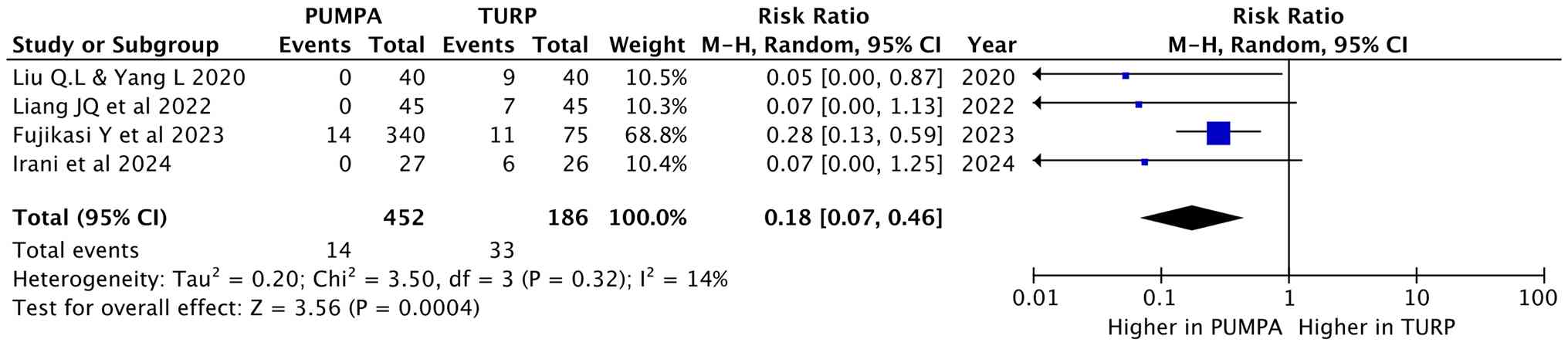
Urinary incontinence
All studies analyzed incontinence post-operatively,
involving 638 participants (452 in the prostatic apex preservation
group and 186 in the apex dissection group). Random effects were
used, and the results revealed that the preserving group had a
lower incontinence rate compared to the non-preserving group (RR,
0.18; 95% CI, 0.07 to 0.46; P=0.0004) (Fig. 4).
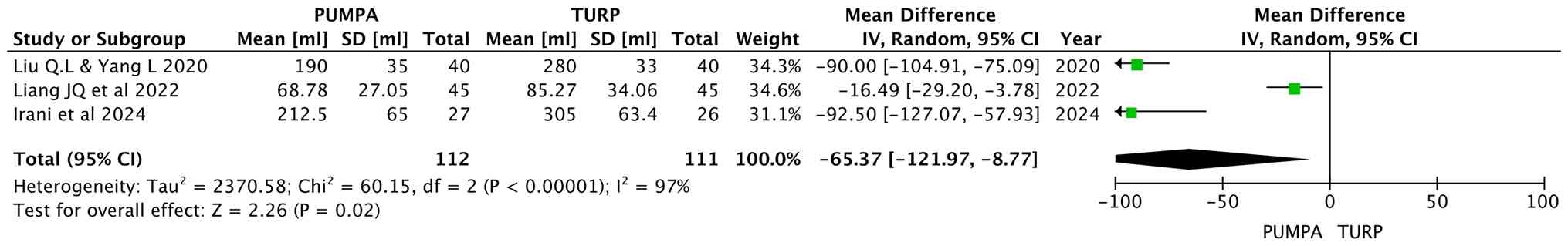
Intraoperative blood loss
Liu and Yang (15),
Liang et al (16), and
Irani et al (17) analyzed
the blood loss during surgery, involving 223 participants (112 in
the prostatic apex preservation group and 111 in the apex
dissection group). Random effects were used, and the results
revealed that blood loss in the preserving group was also less than
that in the non-preserving group (MD, -65.37; 95% CI, -121.97 to
-8.77; P=0.02; Fig. 5).
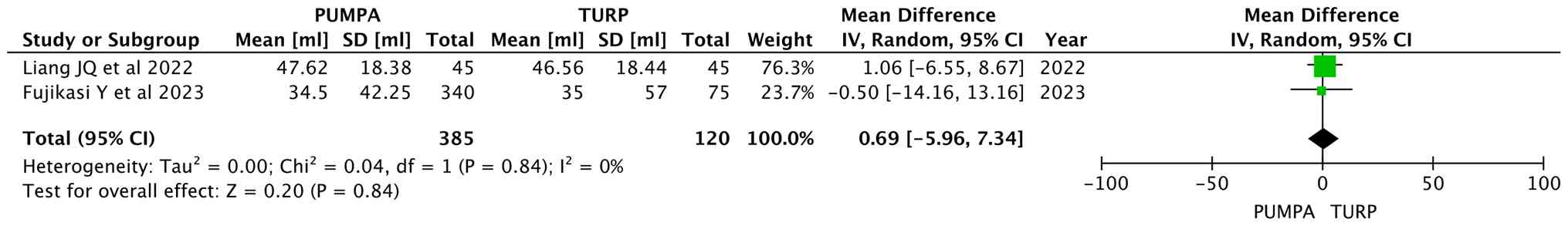
Prostate excised volume
Liang et al (16) and Fujisaki et al (17) analyzed the excised prostate volume,
involving 505 participants (385 in the prostatic apex preservation
group and 120 in the apex dissection group). Random effects models
were used, and the results revealed similar volumes of excised
tissue between the two groups (MD, 0.69; 95% CI, -5.96 to 7.34;
P=0.84) (Fig. 6).
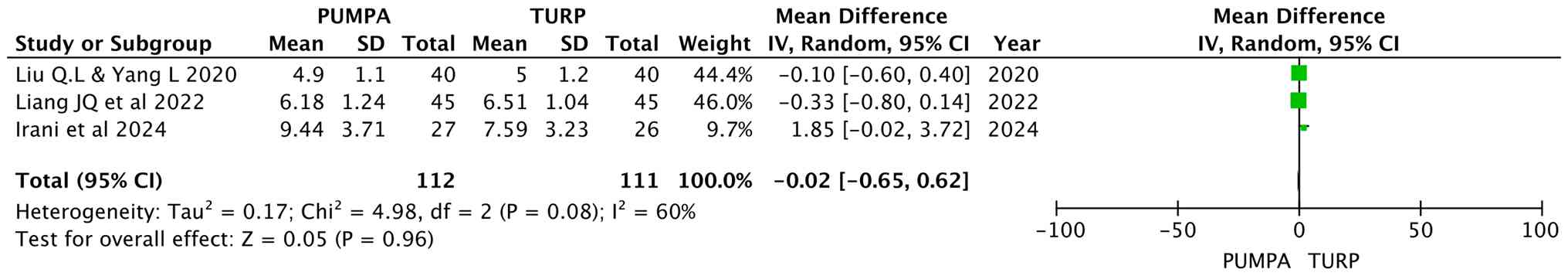
Post-operative IPSS
Liu and Yang (15)
and Liang et al (16)
analyzed the IPSS at 1 month post-operatively, while Irani et
al (18) analyzed the IPSS at
6 months post-operatively, involving 223 participants (112 in the
prostatic apex preservation group and 111 in the apex dissection
group). Random effects were used, and the results revealed similar
IPSS between the two groups (MD, -0.02; 95% CI, -0.65 to 0.62;
P=0.96) (Fig. 7).
QoL score post-operatively
Liu and Yang (15)
and Liang et al (16)
analyzed the QoL post-operatively, involving 170 participants (85
in the prostatic apex preservation group and 85 in the apex
dissection group). Random effects were used, and the results
revealed similar QoL scores between the two groups (MD, -0.30; 95%
CI, -0.50 to 0.11; P=0.003; Fig.
8).
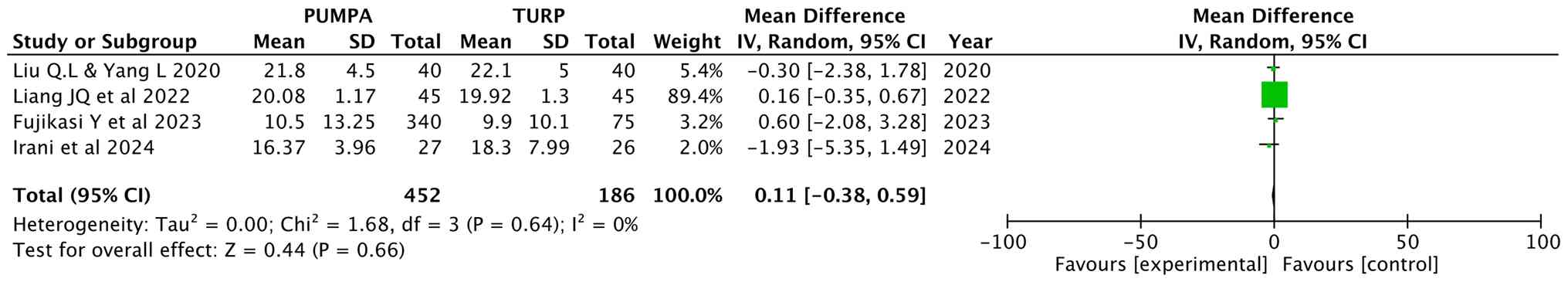
Qmax post-operatively
Liu and Yang (15),
Liang et al (16) and
Fujisaki et al (17)
analyzed the Qmax at 1 month post-operatively, while Irani et
al (18) analyzed the Qmax at
6 months post-operatively, involving 638 participants (452 in the
prostatic apex preservation group and 186 in the apex dissection
group). Random effects were used, and the results revealed a
similar Qmax score between the two groups (MD, 0.11; 95% CI, -0.38
to 0.59; P=0.66) (Fig. 9).
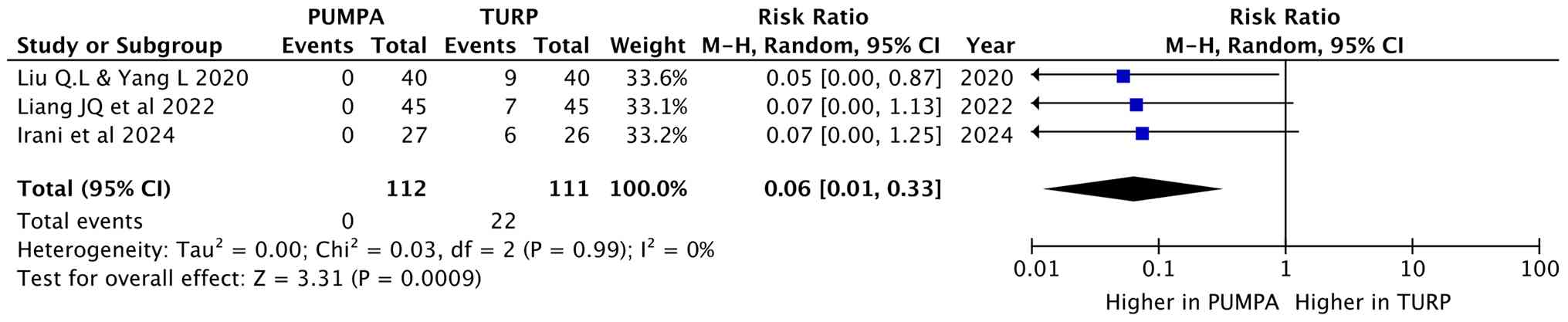
Leave-one-out analysis
Leave-one-out analysis is a method used to evaluate
the robustness of pooled estimation by individually excluding one
study at a time and reanalyzing the data. In the present study,
moderate to high heterogeneity was observed when pooling the effect
sizes for urinary incontinence (I2=14% and P=0.32). The
analysis revealed that the study by Fujisaki et al (17) had a significant influence on the
pooled effect of the urinary incontinence results. When excluding
the study by Fujisaki et al (17), the pooled urinary incontinence rate
across all studies was 0.06 (I2=0%, P=0.99), as
illustrated in the forest plot in Fig. 10. The forest plot, the study by
Fujisaki et al (17),
indicates that the urinary incontinence rate was 0.06 (95% CI, 0.01
to 0.33), which was lower than the first pooled rate of 0.18 (95%
CI, 0.07 to 0.46), although not statistically significant. This
result proposed that this meta-analysis is generally
consistent.
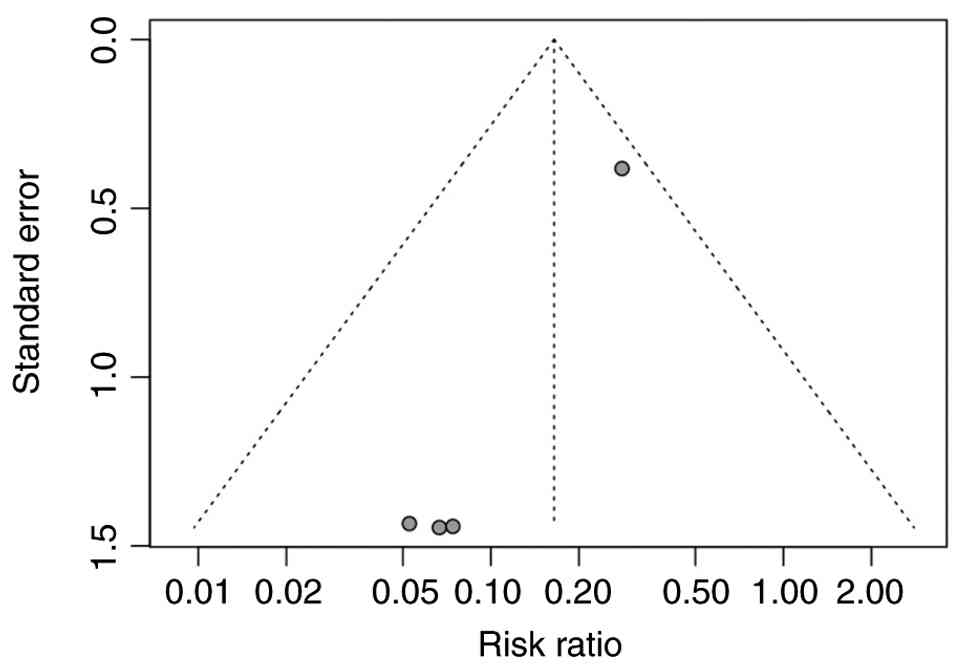
Publication of bias
The publication bias in urinary incontinence was
assessed, as illustrated in the funnel plot in Fig. 11. The plot demonstrates the
standard error and relative risk of urinary incontinence for each
study. While it is hoped that a symmetrical distribution is
achieved in the absence of publication bias, the funnel plot
revealed some asymmetry, focusing on small studies in the left
side, indicating a low prevalence due to the lack of data. Although
the funnel plot visually revealed a potential risk of publication
bias, Egger's regression test indicated potential small-study
effects (bias estimate=-1.40; SE=0.11; P=0.0061). Due to the small
number of studies (k=4), the results should be interpreted with
caution, as Egger's regression test does not provide much value if
the number of studies is <10.
Discussion
Previous studies have highlighted the value of
preserving the prostatic apex in reducing post-operative
complications, particularly urinary incontinence following BPH
surgery (15-18).
Partin et al (7) suggested
that the resection of the prostatic apex, which houses the external
urethral sphincter responsible for passive urinary control, can
compromise continence mechanisms and predispose patients to
post-operative incontinence. Similarly, Zinner et al
(24) emphasized the critical role
of the urethral mucosa in maintaining urinary continence.
The surgical principle of preserving or
reconstructing the urethral mucosa at the prostatic apex is to
extend the mucosal length and create additional mucosal folds
adjacent to the external urethral sphincter. This configuration
provides a ‘sealing pad’ effect, improving urethral closure
pressure and facilitating early post-operative continence (16).
In early research, transient urinary incontinence
was reported in 4 of 15 patients (26.7%) undergoing conventional
TURP, with all cases resolving within 6 months (25). Late incontinence has been linked to
post-operative urinary tract infection, further reinforcing the
importance of mucosal integrity in preserving urinary control
(18).
Across studies, apex-preserving techniques have
consistently achieved higher rates of immediate continence compared
to conventional dissection (15-18).
In the study by Irani et al (18), 6 patients (23%) in the control
group developed immediate post-operative urge incontinence (4 cases
recovered within 1 week and 2 cases within 3 months), whereas no
cases occurred in the preservation group.
Liang et al (16) reported a similar pattern: In total,
7 patients (15%) in the control group experienced immediate
incontinence (4 patients recovered in 1 week and 3 patients in 3
weeks); again there were no cases in the intervention group. Liu
and Yang (15) observed 9 cases
(22.5%) of transient incontinence in the control group, all
resolving within 3 months, while the preservation group
demonstrated 100% immediate continence. Fujisaki et al
(17) documented an incontinence
rate of 4.1% in the preservation group vs. 14.7% in controls
immediately following catheter removal.
Across the included studies, the duration of the
surgery was shorter in the apex-preservation group (15-18),
and intraoperative blood loss was reduced in the majority of
studies (15,16,18).
In total, two studies reported comparable prostate resection
volumes between the two approaches (16,17),
and IPSS values at 3 months were similar across all cohorts
(15,16,18).
QoL metrics, assessed in two studies, exhibited
promising improvements in both groups with Qol scores comparable to
each other (16,17), while Qmax, an indicator of
functional recovery, was consistently satisfactory in all reports
(15,16,18).
To the best of our knowledge, although several
meta-analyses have examined ejaculatory function following
prostatic apex preservation, the present study is the first to
focus primarily on urinary functional outcomes. The findings
presented herein indicate that preserving the prostatic apex during
BPH surgery provides a clinically meaningful advantage in terms of
early urinary continence, shorter operative duration and reduced
intraoperative blood loss, without compromising long-term voiding
efficacy.
Nevertheless, the present study has a few
limitations which should be acknowledged. First, high heterogeneity
was observed in outcomes, such as intraoperative blood loss
(I2=97%) and IPSS (I2=60%), partly due to
incomplete outcome reporting across studies. Second, surgical
expertise and patient characteristics likely contributed to
variability in results. Third, due to limited data availability,
not all functional and perioperative parameters could be included
in the pooled analysis. Fourth, the impact of ethnicity, lifestyle
factors and patient comorbidities could not be evaluated. Lastly,
reoperation outcomes could not be evaluated in the present
meta-analysis as the included randomized controlled trials did not
report reoperation data in a consistent, standardized, or
extractable format. Finally, the majority of the included studies
were small, single-center trials, emphasizing the need for larger,
multicenter randomized studies with longer follow-up.
In conclusion, preserving the prostatic apex during
BPH surgery appears to provide tangible benefits, including the
earlier recovery of urinary continence, reduced intraoperative
bleeding and a shorter surgery duration, while maintaining
comparable postoperative urinary function. However, future
large-scale, high-quality randomized trials with extended follow-up
are warranted to validate these findings and establish the clinical
superiority and safety of apex-preserving techniques in
contemporary BPH management.
Supplementary Material
Fullsearch strategy.
PICO framework.
Acknowledgements
The authors would like to express their sincere
gratitude to Professor Syah Mirsya Warli and Dr Bungaran Sihombing
from Rumah Sakit Haji Adam Malik Medan City for the invaluable
feedback and clinical support for this study. The present
systematic review and meta-analysis has been registered in Prospero
with the registration no. CRD420250642661.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
All authors (DDK, BT and KAWY) contributed to the
conception and design of the study. Material preparation, data
collection and analysis were performed by DDK, BT and KAWY. The
first draft of the manuscript was written by KAWY and BT, and all
authors commented on previous versions of the manuscript. DDK, KAWY
and BT confirm the authenticity of all the raw data. All authors
(DDK, BT and KAWY) have read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Michalak J, Tzou D and Funk J: HoLEP: The
gold standard for the surgical management of BPH in the 21(st)
century. Am J Clin Exp Urol. 3:36–42. 2015.PubMed/NCBI
|
|
2
|
Shvero A, Calio B, Humphreys MR and Das
AK: HoLEP: The new gold standard for surgical treatment of benign
prostatic hyperplasia. Can J Urol. 28:6–10. 2021.PubMed/NCBI
|
|
3
|
Miernik A and Gratzke C: Current treatment
for benign prostatic hyperplasia. Dtsch Arztebl Int. 117:843–854.
2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Qian X, Liu H and Xu D, Xu L, Huang F, He
W, Qi J, Zhu Y and Xu D: Functional outcomes and complications
following B-TURP versus HoLEP for the treatment of benign prostatic
hyperplasia: A review of the literature and meta-analysis. Aging
Male. 20:184–191. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Licari LC, Bologna E, Manfredi C, Franco
A, Ditonno F, DE Nunzio C, Franco G, Cindolo L, Leonardo C,
Adelstein SA, et al: Postoperative urinary incontinence following
BPH surgery: Insights from a comprehensive national database
analysis. Minerva Urol Nephrol. 76:618–624. 2024.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rassweiler J, Teber D, Kuntz R and Hofmann
R: Complications of trans urethral resection of the prostate
(TURP)-incidence, management, and prevention. Eur Urol. 50:969–979;
discussion 980. 2006.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Partin AW, Peters CA, Kavoussi LR,
Dmochowski RR and Wein AJ: The prostate. In: Campbell-Walsh
Urology. Wein AJ, Kavoussi LR, Peters CA, Partin AW, Dmochowski RR
(Eds). 12th edition.: Elsevier, Philadelphia, PA, pp3410-3416,
2020.
|
|
8
|
Valenzi FM, Fuschi A, Al Salhi Y, Sequi
MB, Suraci PP, Pacini M, Scalzo S, Rera OA, Antonioni A, Graziani
D, et al: Is early continence recovery related to the length of the
spared urethra? A prospective multicenter study comparing
preoperative MRI and histologic specimen measurements after robotic
radical prostatectomy. Eur J Surg Oncol. 50(108319)2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Walz J, Burnett AL, Costello AJ, Eastham
JA, Graefen M, Guillonneau B, Menon M, Montorsi F, Myers RP, Rocco
B and Villers A: A critical analysis of the current knowledge of
surgical anatomy related to optimization of cancer control and
preservation of continence and erection in candidates for radical
prostatectomy. Eur Urol. 57:179–192. 2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Walz J, Epstein JI, Ganzer R, Graefen M,
Guazzoni G, Kaouk J, Menon M, Mottrie A, Myers RP, Patel V, et al:
A critical analysis of the current knowledge of surgical anatomy of
the prostate related to optimization of cancer control and
preservation of continence and erection in candidates for radical
prostatectomy: An update. Eur Urol. 70:301–311. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Katsimperis S, Juliebø-Jones P, Ta A,
Tandogdu Z, Al-Bermani O, Bellos T, Esperto F, Tonyali S,
Mitsogiannis I, Skolarikos A, et al: Surgical techniques to
preserve continence after robot-assisted radical prostatectomy.
Front Surg. 10(1289765)2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Dalpiaz O and Anderhuber F: The fascial
suspension of the prostate: A cadaveric study. Neurourol Urodyn.
36:1131–1135. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Van der Poel HG, de Blok W, Joshi N and
van Muilekom E: Preservation of lateral prostatic fascia is
associated with urine continence after robotic-assisted
prostatectomy. Eur Urol. 55:892–900. 2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hoyland K, Vasdev N, Abrof A and Boustead
G: Post-radical prostatectomy incontinence: Etiology and
prevention. Rev Urol. 16:181–188. 2014.PubMed/NCBI
|
|
15
|
Liu QL and Yang L: Comparison of the
transurethral resection of the prostate by traditional versus
preserved urethral mucosa of the prostatic apex. J Endourol.
34:482–486. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Liang JQ, Ma WT, Lu BW, Dai L, Zhao YM,
Zhang JD, Tian B and Liu QL: Clinical study on the application of
preserved urethral mucosa at the prostatic apex in transurethral
plasmakinetic resection of the prostate. Front Surg.
9(922479)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Fujisaki Y, Otsuka I, Kobayashi T, Miyake
N, Ito K, Terada N, Kamoto T and Iwamoto H: Use of the anterior
prostatic urethral mucosa preservation technique during holmium
laser enucleation of the prostate can reduce postoperative stress
urinary incontinence. Asian J Endosc Surg.
17(e13256)2024.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Irani D, Saadatfar A, Aminsharifi A, Ahmed
F, Mirzakhanlouei A, Afrasiabi M, Adib A and Shamohammadi I:
Influence of urethral mucosa preservation of prostatic apex during
monopolar transurethral resection of the prostate on postoperative
functional outcomes: A retrospective comparative study. Urol Sci.
35:75–79. 2024.
|
|
19
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: The PRISMA 2020 statement: An updated guideline for
reporting systematic reviews. BMJ. 372(n71)2021.PubMed/NCBI View
Article : Google Scholar
|
|
20
|
Higgins J, Savović J, Page MJ and Sterne
JAC: RoB 2: A revised Cochrane risk-of-bias tool for randomized
trials. Br Med J. 366(l4898)2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cumpston M, Li T, Page MJ, Chandler J,
Welch VA, Higgins JP and Thomas J: Updated guidance for trusted
systematic reviews: A new edition of the cochrane handbook for
systematic reviews of interventions. Cochrane Database Syst Rev.
10(ED000142)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
DerSimonian R and Laird N: Meta-analysis
in clinical trials revisited. Contemp Clin Trials. 45:139–145.
2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: The PRISMA 2020 statement: An updated guideline for
reporting systematic reviews. BMJ. 372(n71)2021.PubMed/NCBI View
Article : Google Scholar
|
|
24
|
Zinner NR, Sterling AM and Ritter RC: Role
of inner urethral softness in urinary continence. Urology.
16:115–117. 1980.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Carnevale FC, Iscaife A, Yoshinaga EM,
Moreira AM, Antunes AA and Srougi M: Transurethral resection of the
prostate (TURP) versus original and PErFecTED prostate artery
embolization (PAE) due to benign prostatic hyperplasia (BPH):
Preliminary results of a single center, prospective,
urodynamic-controlled analysis. Cardiovasc Intervent Radiol.
39:44–52. 2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Guyatt GH, Oxman AD, Schünemann HJ,
Tugwell P and Knottnerus A: GRADE guidelines: A new series of
articles in the journal of clinical epidemiology. J Clin Epidemiol.
64:380–382. 2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Deng T, Wang Y, Wang Y, Li B, Jin Y, Ren X
and Wang X: Methodology for clinical practice
guidelines-Application of GRADEpro GDT in Evidence Grading of
Systematic Reviews of Interventional trial. Chin J Evid-Based
Cardiovasc Med. 11:1–5. 2019.(In Chinese).
|