1. Introduction
The fascinating roots of nanotechnology can be
connected with physicist, Richard Feynman, a great pioneer in the
field. In his groundbreaking 1959 speech, ‘There's Plenty of
Room at the Bottom’, Feynman introduced the noteworthy concept
of regulating and influencing matter at the atomic and molecular
scale (1). He envisioned the
creation of miniaturised devices and materials at the nanoscale,
setting the stage for remarkable advancements. The term
nanotechnology was officially coined later, although its spirit was
adopted by the Japanese scientist, Norio Taniguchi, in 1974, who
examined the precise fabrication of nanoscale materials. The
visionary work of Taniguchi focused on processes that could be
finely tuned at the atomic level (2,3).
Building on this foundation, the American engineer, Eric Drexler,
further advanced the concept of molecular nanotechnology in the
1980s. The influential book of Drexler in 1986 entitled ‘Engines of
Creation’ (4) explored the
possibilities of constructing intricate structures at the atomic
level using molecular machines, inspired by the pioneering theories
of Feynman. Nanoparticles are tiny particles with dimensions
between 1 and 100 nanometres, which possess distinctive
characteristics due to their very small size and outstanding
surface-to-volume ratio (5-7).
These properties, which are often non-uniform compared with those
of bulk materials, render nanoparticles highly valuable in various
fields (8). For example, metal
nanoparticles (MetNPs), such as gold and silver exhibit superior
optical (9) and catalytic
(10) behaviour due to surface
plasmon resonance, while metal oxide nanoparticles (MONPs), such as
titanium dioxide (TiO2) and zinc oxide (ZnO) are widely
used in photocatalysis (11) and
environmental remediation (12).
Carbon-based nanoparticles (CBNPs), including carbon nanotubes
(CNTs) and graphene, exhibit marked electrical conductivity and
mechanical strength, leading to their use in electronics and
materials science. Nanoparticles are synthesised using top-down
approaches, such as mechanical milling or bottom-up methods such as
chemical vapour deposition (13,14).
Their high reactivity and surface energy allow their advanced
utilisation in fields, such as medicine, energy and environmental
protection, where they play key roles in catalysis, drug delivery
and pollutant removal. MetNPs such as gold, silver, selenium and
iron synthesised through green methods have immense potential due
to their ability to be modified with various chemical functional
groups. This functionalisation permits their conjugation with
antibodies, ligands and drugs, opening avenues for targeted drug
delivery, gene therapy, and diagnostic imaging. Additionally, these
nanoparticles play essential roles in biotechnology, particularly
in magnetic separation, pre-concentration of target analytes, and
the development of vehicles for drug and gene delivery (15,16).
There are various properties of nanoparticles. To strengthen the
conceptual organisation of the present review, a
functionality-based classification of nanoparticles is introduced
(Table I), categorising them
according to their primary operational roles rather than
composition alone. This framework classifies nanoparticles into
catalytic, adsorptive, optical, magnetic, mechanical-reinforcement,
electrochemical, antimicrobial, biocompatible and sensor-specific
nanoparticles.
 | Table IFunctionality-based classification of
nanoparticles. |
Table I
Functionality-based classification of
nanoparticles.
| Functionality
category | Types of
nanoparticles | Properties | Typical synthesis
methods | Applications | (Refs.) |
|---|
| Catalytic
nanoparticles | Metal NPs (Pt, Pd,
Au and Ag), metal oxides (TiO2 and MnO2), and
MOF-derived NPs | High surface area,
active sites, and redox activity | Chemical reduction,
sol-gel, thermal decomposition, and MOF-templated synthesis | Photocatalysis,
organic pollutant degradation, fuel cells, and hydrogen
evolution | (119) |
| Adsorptive
nanoparticles | Carbon nanotubes,
graphene oxide, biochar NPs, zeolites and silica NPs | Large surface area,
porosity, and surface functional groups | Hydrothermal, green
synthesis, pyrolysis, and surface functionalisation | Heavy metal
removal, dye adsorption, and water purification | (120) |
| Optical
nanoparticles | Quantum dots,
plasmonic Au/Ag NPs, and upconversion NPs | Fluorescence,
plasmon resonance, and tuneable bandgap | Colloidal
synthesis, solvothermal and microwave synthesis | Imaging, sensing,
optical probes and biosensing | (51) |
| Magnetic
nanoparticles |
Fe3O4,
CoFe2O4 and
MnFe2O4 | Superparamagnetism
and magnetic separation capability | Co-precipitation,
thermal decomposition, and solvothermal | MRI contrast
agents, targeted drug delivery and magnetic separation of
pollutants | (117) |
|
Mechanical-reinforcement
nanomaterials | Carbon nanotubes,
nanofibers, nanoclays and graphene | High tensile
strength, flexibility, and high modulus | Chemical vapor
deposition, exfoliation, and electrospinning | Polymer
nanocomposites, structural reinforcement, and coatings | (121,122) |
|
Electrical/electro-chemical
nanoparticles | Metal selenides
(NiSe2, CoSe2), metal sulphides, conductive
polymers, and carbon nanomaterials | High conductivity,
fast electron transfer, and electrocatalytic activity | Electrodeposition,
solvothermal, hydrothermal, and MOF-derived synthesis | Electrochemical
sensors, super-capacitors and batteries | (123) |
| Antimicrobial
nanoparticles | Ag NPs, ZnO NPs and
CuO NPs | ROS generation,
membrane disruption, and ion release | Green synthesis,
chemical reduction, and precipitation | Antibacterial
coatings, wound dressings and food preservation | (83,124) |
|
Biocompatible/drug-delivery
nanoparticles | Liposomes, polymer
NPs, silica NPs and gold nanoshells | Biocompatibility,
controlled release, and surface modifiability | Self-assembly,
emulsification, and nanoprecipitation | Drug delivery, gene
delivery and targeted therapeutics | (125,126) |
| Sensor-specific
nanoparticles |
NiSe2/MOF composites,
AuNP-based probes and carbon dots | High selectivity,
electrocatalysis, and signal amplification | MOF-derived
synthesis, hydrothermal, and green synthesis | Pharmaceutical
detection, environmental sensing and biosensors | (127) |
Size and surface area
The small size of nanoparticles leads to a large
surface area relative to volume, which enhances reactivity and
interaction with other substances. This makes nanoparticles highly
effective in catalysis and adsorption processes (17,18).
Quantum effects
At the nanoscale, quantum mechanics begins to
dominate material behaviour, leading to unique optical, electronic
and magnetic properties that are not present in larger particles
(19).
Surface energy
Nanoparticles possess higher surface energy, which
can affect their stability and reactivity. As a result, they tend
to agglomerate to reduce surface energy, but can be stabilised
through surface treatments or coatings (20).
Mechanical strength
Some nanoparticles, such as CNTs, exhibit
exceptional mechanical properties, such as high tensile strength
and flexibility (21).
Optical properties
Nanoparticles can exhibit unique optical properties
due to surface plasmon resonance, particularly in metals such as
gold and silver, rendering them useful in imaging, sensing and
photothermal applications (19).
Major developments in the field of MetNPs include a
transition from early physical and chemical synthesis methods to
modern green, biogenic methods that provide sustainable and
eco-friendly production (Table
II). Advances in nanoscale characterisation and mechanistic
understanding have enabled greater control over particle size,
shape and surface chemistry. These breakthroughs have led to rapid
growth in applications such as biomedicine, catalysis, sensing, and
environmental remediation, with recent advances focusing on the
synthesis of hybrid nanocomposites and nanozymes.
 | Table IIMajor developments in the field of
metal nanoparticles. |
Table II
Major developments in the field of
metal nanoparticles.
| Era/phase | Key
developments | Description of
advancement | Representative
metals/examples | (Refs.) |
|---|
| Early phase
(1980-2000) | Physical and
chemical synthesis | Development of
top-down and bottom-up methodssuch as chemical reduction, laser
ablation, sol-gel and thermal decomposition | Au, Ag, Pt and Pd
nanoparticles synthesised by citrate reduction (the Turkevich
method) | (128,129) |
| Characterisation
advancements | Introduction of
TEM, SEM, XRD, UV-Vis and DLS enabling improved size/shape
analysis | Enabled correlation
between structure and optical properties | Au and Ag SPR
studies | (9,130) |
| Green synthesis
emergence (2000-2010) | Plant-based
biosynthesis | Use of plant
extracts (polyphenols, alkaloids, and proteins) for nanoparticle
reduction | AgNPs from
Azadirachta indica and AuNPs from Camellia
sinensis | (101,103) |
| | Microbial
synthesis | Bacteria, fungi,
and algae used as bioreactors for metal ion reduction | AgNPs from
Bacillus subtilis and AuNPs from Fusarium
oxysporum | (82,131) |
| |
Biomolecule-mediated synthesis | Amino acids,
enzymes, and polysaccharides used as stabilising/capping
agents | Chitosan-capped
AgNPs and protein-stabilised AuNPs | (132) |
| Mechanistic
understanding (2010-2018) | Mechanistic
elucidation | Understanding of
bioreduction pathways, enzyme involvement, and electron shuttle
mechanisms | NADH-dependent
enzymatic reduction for Au and AgNPs | (93,133) |
| | Controlled
synthesis | Advancement in
size/shape control: nanorods, nanoflowers, and nanocubes | Au nanorods, Ag
nanocubes and Pt nanoflowers | (134) |
| | Surface
functionalisation | Enhanced
biocompatibility and targeting via PEGylation and ligand
attachment | PEG-AuNPs and
antibody-tagged AgNPs | (135) |
| Application
expansion (2015-2022) | Biomedical
applications | Use in drug
delivery, imaging, hyperthermia, biosensors, and antimicrobial
coatings | AuNPs for cancer
therapy and AgNP wound dressings | (40) |
| | Environmental
applications | Use in pollutant
degradation, sensing, and heavy metal removal |
Fe3O4 NPs for
wastewater treatment and AgNP sensors for Hg2+ | (29) |
| | Catalytic
applications | High catalytic
activity due to surface area and active sites | PdNPs for
hydrogenation and AuNPs for CO oxidation | (136) |
| Advanced
Technologies (2020-present) | Hybrid
nanocomposites | Integration with
polymers, MOFs, graphene, and carbon nanotubes |
NiSe2/MOF hybrids and
AgNP-graphene composites | (137) |
| | Nanozymes | Metal nanoparticles
mimicking enzymatic activity (peroxidase and oxidase) |
Fe3O4 NP peroxidase
mimics and AuNP oxidase mimic | (138) |
| | Precision
biosynthesis | Genome-engineered
microbes and enzymatic pathways to tailor NP morphology | CRISPR-modified
bacteria for AgNPs | (85) |
| | Clinical
translation efforts | Toxicity reduction,
pharmacokinetic studies, and regulatory focus | PEG-AuNPs in
clinical imaging trials | (139) |
| Future directions
(emerging) | AI-guided
synthesis | Machine-learning
models predicting shape, size, and stability | Computational
design of Au, Ag and Pd NPs | (140) |
| | Sustainable
nanotechnology | Zero-waste
synthesis and agro-waste-mediated nanoparticle production | Fruit peel-mediated
AgNPs and biomass-derived AuNPs | (141) |
| | Multifunctional
theranostics | Combining therapy +
imaging in one nanoparticle platform |
Au-Fe3O4 hybrid
nanoplatforms | (142) |
2. Types of nanoparticles
MNPs
MetNPs are metallic particles ranging from 1 to 100
nanometres in size and exhibit unique physical, chemical and
optical properties that differ significantly from their bulk
counterparts. These properties arise from their high
surface-area-to-volume ratio and quantum effects, making them
highly versatile across various fields (22). The size, shape and surface
characteristics of MetNPs can be tailored during synthesis,
influencing reactivity, catalytic activity, and optical behaviour
(23). Common types include gold
nanoparticles, known for their biocompatibility and use in medical
diagnostics and drug delivery; silver nanoparticles, widely used
for antimicrobial properties; and iron nanoparticles, effective in
environmental remediation. Platinum and copper nanoparticles are
valued for their catalytic and electrical properties, respectively.
The applications of MetNPs span diverse areas. In environmental
remediation, they are used for heavy metal removal, the degradation
of organic pollutants and the catalytic breakdown of contaminants
(24). In medicine, they serve as
drug delivery agents, imaging tools and antimicrobial agents. Their
role in energy applications includes use in fuel cells and
batteries, while in sensors and diagnostics they enhance disease
detection and environmental monitoring (25). MetNPs are synthesised using
chemical, physical and biological methods, including chemical
reduction of metal salts, laser ablation and green synthesis using
plant extracts or microorganisms. However, stability issues,
potential toxicity and scalability concerns need to be addressed to
fully harness their potential. Despite these challenges, MetNPs
represent a promising avenue for innovation across multiple
industries.
MONPs
MONPs are a class of nanomaterials consisting of
metal cations bonded to oxygen anions. These nanoparticles exhibit
exceptional properties, including high stability, catalytic
activity, and tuneable electronic, magnetic and optical
characteristics. Their unique properties are attributed to their
small size, high surface-area-to-volume ratio, and quantum
confinement effects, rendering them highly desirable in various
scientific and industrial applications. MONPs are synthesised from
a wide variety of metals, with some of the most common types being
TiO2, ZnO, iron oxide
(Fe2O3/Fe3O4), cerium
oxide (CeO2) and aluminium oxide
(Al2O3). TiO2 is widely used for
its photocatalytic activity in environmental remediation and
self-cleaning surfaces (26). ZnO
is prized for its antimicrobial properties and applications in
sunscreens and sensors. Iron oxide nanoparticles, particularly
magnetite (Fe3O4), are utilised in biomedical
applications, such as magnetic resonance imaging (MRI) and targeted
drug delivery. CeO2 is known for its role as a redox
catalyst, particularly in automotive catalytic converters and as an
antioxidant in biological systems (27). The applications of MONPs span a
range of fields. In environmental science, they are employed for
water and air purification, as well as for the degradation of
organic pollutants through photocatalytic processes (28,29).
In the medical field, they play a role in drug delivery, imaging,
and antimicrobial treatments (30). MONPs are also extensively used in
energy storage and conversion devices, such as lithium-ion
batteries, supercapacitors, and fuel cells (31). Their catalytic properties make them
essential in industrial chemical processes, including the
production of fertilisers and the refinement of fuels. Several
synthesis methods are used to produce MONPs, including sol-gel
processes, hydrothermal methods, chemical vapor deposition, and
green synthesis using biological agents (32). These methods allow control over the
size, shape and surface characteristics of the nanoparticles, which
are critical for optimising their performance in specific
applications. Despite their advantages, MONPs pose challenges, such
as potential toxicity, environmental persistence and the need for
cost-effective and scalable production methods (33). Addressing these challenges is
essential for their safe and sustainable use across various
sectors, highlighting their potential to revolutionise industries
ranging from healthcare to environmental management.
CBNPs
CBNPs are nanomaterials composed primarily of carbon
atoms, engineered in various structures and dimensions. These
nanoparticles exhibit notable mechanical, electrical, thermal and
chemical properties, rendering them a cornerstone of nanotechnology
research and applications. Their versatility stems from the unique
bonding nature of carbon, which allows them to form various
allotropes and hybrid structures (34). Key types of CBNPs include CNTs,
fullerenes, graphene, graphene oxide, carbon dots (CDs) and
nanodiamonds. CNTs are cylindrical structures with extraordinary
tensile strength, electrical conductivity, and thermal stability,
making them valuable in electronics, materials reinforcement, and
energy storage (21). Fullerenes,
spherical carbon molecules, are known for their electron-accepting
capabilities and are used in drug delivery, solar cells and
antioxidants. Graphene is a two-dimensional sheet of carbon atoms
arranged in a hexagonal lattice, renowned for its high electrical
and thermal conductivity, flexibility, and strength, leading to
applications in flexible electronics, sensors and advanced
composites (34). CDs are
fluorescent nanoparticles with tuneable optical properties,
commonly used in bioimaging and sensing. Nanodiamonds exhibit
exceptional hardness and biocompatibility, making them ideal for
polishing, drug delivery, and imaging (35). The applications of CBNPs span
multiple disciplines. In medicine, they serve as drug delivery
agents, bioimaging tools and antimicrobial materials. In
electronics, they are employed in transistors, flexible displays
and conductive coatings. Their thermal properties render them
useful in heat management systems, while their mechanical strength
is exploited in the development of advanced composites.
Environmental applications include water purification, pollutant
adsorption and catalysis for pollutant degradation. Additionally,
their role in energy storage and conversion, such as in batteries,
supercapacitors, and fuel cells, highlights their importance in
renewable energy technologies. CBNPs are synthesised through
various techniques, including chemical vapor deposition, laser
ablation, arc discharge and green synthesis using biological
agents. These methods allow control over their size, structure and
functionalisation, which are critical for tailoring them to
specific applications. However, challenges, such as production
scalability, cost, potential toxicity and environmental impact need
to be addressed. Despite these obstacles, CBNPs represent a
transformative class of nanomaterials, with the potential to drive
advancements in science, technology and industry (29,33-35).
Polymeric nanoparticles (PNPs)
PNPs are nano-sized particles composed of polymers,
typically ranging in size from 1 to 1,000 nanometres. These
nanoparticles are highly versatile and can be engineered for a wide
range of applications due to their biocompatibility, chemical
tunability, and ability to encapsulate and protect active agents.
Their structure often includes a polymeric core and shell, which
can be functionalised to enhance stability, target specificity and
controlled-release properties. PNPs are categorised into different
types based on their structure. Nanospheres are solid matrix-like
particles in which the active compound is dispersed or adsorbed
throughout the polymer matrix. Nanocapsules, on the other hand, are
vesicular systems in which the active agent is enclosed within a
polymeric shell (36,37). The polymers used in PNP synthesis
can be natural (e.g., chitosan, gelatin and alginate) or synthetic
[e.g., polylactic acid, polyglycolic acid and
poly(lactic-co-glycolic acid)] and are selected based on the
intended application and required properties (38). The applications of PNPs are vast,
with their most prominent role in the biomedical field. They are
widely used as drug delivery systems, enabling the encapsulation of
therapeutic agents for enhanced solubility, stability and targeted
delivery. Their ability to release drugs in a controlled manner
reduces side-effects and improves therapeutic efficacy. In cancer
therapy, PNPs can be designed to deliver chemotherapeutics directly
to tumour cells, minimising damage to healthy tissues (36,39).
They are also used in gene therapy, vaccine delivery and tissue
engineering. Beyond medicine, PNPs are employed in the food
industry for the controlled release of nutrients and additives, in
agriculture for the delivery of pesticides and fertilisers, and in
environmental applications for pollutant removal. Synthesis methods
for PNPs include emulsion techniques (single or double emulsion),
precipitation, solvent evaporation, and ionic gelation, among
others. These methods allow the precise control over particle size,
shape and surface properties. The functionalisation of the
nanoparticle surface with ligands or coatings further enhances
their ability to target specific cells or tissues, a feature
particularly valuable in medical applications. Despite their
advantages, challenges remain in the use of PNPs, such as scaling
up production, ensuring uniformity, and addressing potential
environmental and biological toxicity. Advances in polymer
chemistry and nanoparticle engineering continue to overcome these
hurdles, rendering PNPs a key player in modern nanotechnology and
materials science. Their adaptability and functionality promise
significant contributions across diverse industries, from
healthcare to environmental sustainability (37,39-42).
Lipid-based nanoparticles (LNPs)
LNPs are nanoscale structures composed of lipids,
typically ranging in size from 10 to 1,000 nanometres. They have
garnered significant attention due to their biocompatibility,
ability to encapsulate hydrophilic and hydrophobic molecules and
potential for targeted delivery. Their unique properties render
them indispensable in drug delivery systems, particularly for
delivering challenging therapeutic agents such as nucleic acids and
poorly soluble drugs. The structure of LNPs generally includes a
lipid bilayer or a core-shell configuration, in which a hydrophobic
core is surrounded by a lipid shell (43-45).
Common types of LNPs include liposomes, solid lipid nanoparticles
(SLNs) and nanostructured lipid carriers (NLCs). Liposomes are
spherical vesicles with one or more lipid bilayers, capable of
encapsulating hydrophilic drugs in their aqueous core and
hydrophobic drugs within their lipid bilayer (46). SLNs are composed of a solid lipid
core stabilised by surfactants, providing a highly stable system
for drug encapsulation. NLCs are an advanced version of SLNs,
incorporating both solid and liquid lipids to improve drug-loading
and release characteristics. LNPs are widely used in the
pharmaceutical and biomedical fields. They play a crucial role in
the delivery of small molecules, peptides, proteins and nucleic
acids, providing protection from enzymatic degradation and
enhancing cellular uptake. LNPs have revolutionised the field of
gene therapy and mRNA vaccines, as demonstrated by their use in
COVID-19 mRNA vaccines (43,46-48).
In cancer therapy, they enable targeted delivery of
chemotherapeutics to tumour cells while minimising systemic
toxicity. Beyond healthcare, LNPs are applied in cosmetics for
enhanced skin delivery, in agriculture for the delivery of
pesticides and fertilisers, and in food technology for
encapsulating nutrients and bioactives. The synthesis of LNPs
involves methods, such as thin-film hydration, microfluidics,
high-pressure homogenisation and solvent evaporation (38). These techniques allow for the
precise control over particle size, surface charge and
encapsulation efficiency. Surface functionalisation with ligands,
such as antibodies or peptides, enables targeted delivery to
specific cells or tissues, enhancing therapeutic efficacy. While
LNPs offer numerous advantages, challenges persist, including
stability issues, scalability for industrial production and
potential immunogenicity (49,50).
Addressing these challenges through advances in lipid chemistry and
nanotechnology will further enhance their efficacy and broaden
their applications. LNPs continue to be a cornerstone of modern
nanomedicine and materials science, with the potential to address
unmet needs in healthcare, agriculture and beyond.
Semiconductor nanoparticles [quantum
dots (QDs)]
Semiconductor nanoparticles, commonly known as QDs,
are nanometre-scale crystals typically ranging from 2 to 10
nanometres in size. These nanoparticles exhibit unique quantum
mechanical properties, particularly quantum confinement, which
arises when the particle size is smaller than the exciton Bohr
radius. This confinement results in discrete energy levels and
size-dependent optical and electronic properties, rendering QDs
highly versatile in various applications. QDs are composed of
semiconductor materials such as cadmium selenide (CdSe), cadmium
telluride (CdTe), indium phosphide (InP) and zinc sulfide (ZnS).
Their most notable property is their size-tuneable fluorescence; by
varying the particle size, QDs can emit light across the visible to
infrared spectrum (9). This
property, combined with their high photostability, broad absorption
spectra, and narrow emission peaks, renders QDs invaluable for
imaging and optoelectronic applications. The applications of QDs
span multiple fields. In biomedicine, they are widely used as
fluorescent probes for bioimaging and biosensing due to their
brightness and resistance to photobleaching. QDs are instrumental
in the development of advanced diagnostics, enabling
high-resolution imaging of cellular and molecular processes. In
optoelectronics, QDs are employed in QD light-emitting diodes
(QD-LEDs), offering vibrant displays with superior colour accuracy
and energy efficiency (9,35). They are also used in solar cells to
improve light absorption and energy conversion efficiency, as well
as in lasers and photodetectors. In environmental science, QDs are
used in sensors for detecting pollutants and toxins, as well as in
photocatalysis for water and air purification. QDs are typically
synthesised using methods such as colloidal synthesis, chemical
vapour deposition, molecular beam epitaxy, and microemulsion
techniques. These methods allow precise control over particle size,
composition, and surface properties, which are crucial for tuning
their optical and electronic characteristics. Surface modification
and functionalisation of QDs are often employed to improve their
stability, biocompatibility, and specificity for targeted
applications (51,52). Despite their advantages, the
widespread use of QDs faces challenges. A number of QDs are made
from heavy metals, such as cadmium, raising concerns about
environmental toxicity and biocompatibility. Research is ongoing to
develop eco-friendly and non-toxic alternatives, such as graphene
and silicon QDs. Additionally, achieving cost-effective and
scalable production remains a focus of current efforts. QDs
represent a transformative class of materials with immense
potential to impact science and technology. Their unique properties
and adaptability make them a cornerstone of advancements in
imaging, electronics, renewable energy and environmental
applications (9,19).
Ceramic nanoparticles
Ceramic nanoparticles are nanoscale materials made
from inorganic, non-metallic solids, such as oxides, carbides,
nitrides, or silicates. Typically ranging in size from 1 to 100
nanometres, these particles are characterised by their high
stability, hardness, chemical inertness, and resistance to heat and
corrosion. Their properties can be finely tuned by controlling
their size, composition and morphology, rendering them highly
versatile in various scientific and industrial applications
(53). Common types of ceramic
nanoparticles include TiO2, Al2O3,
silicon dioxide (SiO2), zirconium dioxide
(ZrO2) and magnesium oxide (MgO). These materials
exhibit diverse properties, such as photocatalytic activity,
thermal conductivity, and electrical insulation. For instance,
TiO2 is widely used for its photocatalytic and
UV-blocking properties, while SiO2 finds applications in
reinforcement, electronics and drug delivery (54,55).
Ceramic nanoparticles are utilised in a broad range of fields. In
medicine, they serve as carriers for drug delivery, imaging agents
in diagnostics, and materials for bone and dental tissue
engineering. Their biocompatibility and ability to be
functionalised make them suitable for these applications. In
environmental science, they are employed for water purification,
pollutant adsorption, and photocatalysis for the degradation of
organic contaminants. In electronics, ceramic nanoparticles enhance
the properties of devices such as capacitors, sensors, and thermal
insulators. In industrial applications, they are used as additives
to improve the mechanical strength, heat resistance, and durability
of materials such as coatings, paints, and polymers (56,57).
Furthermore, they are integral to the development of advanced
ceramics for high-performance applications in the aerospace,
automotive and energy industries. Synthesis methods for ceramic
nanoparticles include sol-gel processes, hydrothermal synthesis,
flame spray pyrolysis and mechanical milling. These methods enable
precise control over particle size, morphology, and composition.
Surface functionalisation techniques are often employed to enhance
their dispersibility and compatibility with specific environments
or applications. Despite their advantages, ceramic nanoparticles
face challenges, such as aggregation, which can reduce their
effectiveness, and concerns regarding their environmental and
biological impacts. Addressing these challenges through improved
synthesis techniques and comprehensive toxicity assessments is
essential for their sustainable and safe use. Ceramic nanoparticles
represent a critical class of materials with wide-ranging
applications due to their unique physical and chemical properties.
As research and technology advance, they continue to play a crucial
role in addressing challenges across healthcare, environmental
management, energy storage and materials engineering (53,58,59)
Magnetic nanoparticles (MNPs)
MNPs are nanoscale particles that exhibit magnetic
properties due to their composition of magnetic materials such as
iron oxide (magnetite Fe3O4 or maghemite
γ-Fe3O4), cobalt, or nickel. These particles
display unique characteristics, including superparamagnetism,
whereby they exhibit strong magnetisation only in the presence of
an external magnetic field and lose their magnetisation when the
field is removed. This property, along with their high
surface-area-to-volume ratio, makes them highly versatile for
various applications. MNPs are often engineered with a core-shell
structure, in which the core consists of a magnetic material and
the shell is coated with substances such as silica, polymers, or
carbon to enhance stability, biocompatibility, and dispersibility.
These features render MNPs particularly suitable for biomedical,
environmental and industrial applications (25). In the biomedical field, MNPs are
widely used for drug delivery, where they function as carriers to
transport drugs to specific sites in the body, guided by an
external magnetic field. They are also employed in hyperthermia
therapy, in which they generate localised heat under an alternating
magnetic field to destroy tumour cells, and as contrast agents in
MRI to improve imaging quality (60). Additionally, MNPs play a role in
biosensing, aiding in the detection of biomolecules in diagnostic
assays. In environmental science, MNPs are effective in water
treatment, as they can remove heavy metals, organic pollutants and
oil spills. They are also used as catalysts for pollutant
degradation. In technology, MNPs are integral to high-density data
storage and spintronics due to their exceptional magnetic
properties. In industrial applications, they are used in magnetic
fluids for seals, damping systems, and actuators, as well as in
catalysis for chemical synthesis (61). MNPs are synthesised using various
methods, such as co-precipitation, thermal decomposition, sol-gel
synthesis and hydrothermal techniques, which allow precise control
over their size, composition and surface properties. The
functionalisation of their surface is often performed to attach
specific ligands, polymers, or biomolecules, enabling targeted
applications. Despite their advantages, challenges such as particle
aggregation, long-term stability, scalability of production and
potential toxicity remain. Addressing these challenges through
advances in synthesis techniques, surface modifications and
comprehensive safety assessments is crucial. Magnetic nanoparticles
represent a transformative class of materials with diverse
applications in medicine, environmental management, and technology,
driven by their unique ability to respond to external magnetic
fields (25,60,62).
Core-shell nanoparticles
Core-shell nanoparticles are a class of
nanostructures comprising a core material surrounded by a shell of
a different composition, typically designed to combine or enhance
the properties of both the core and the shell materials. The core
and shell can be made from various materials, such as metals,
oxides, polymers, or carbon, providing versatility in tailoring
their physical, chemical and optical properties for specific
applications. The core-shell structure provides several advantages,
including improved stability, enhanced functionality and controlled
interaction with the surrounding environment. The core is typically
selected for its unique intrinsic properties, such as magnetic,
optical, or catalytic characteristics, while the shell is
engineered to enhance biocompatibility, prevent core oxidation, or
introduce new functionalities (63). For example, metallic cores such as
gold or silver are often combined with silica or polymer shells to
enhance stability and enable surface functionalisation. Similarly,
magnetic cores, such as iron oxide, are coated with inert shells to
reduce toxicity and improve dispersibility. Core-shell
nanoparticles are widely utilised across various fields. In
medicine, they are employed in drug delivery systems, imaging and
diagnostics. For instance, nanoparticles with a magnetic core and a
biocompatible polymer shell can deliver drugs to targeted sites
under the influence of an external magnetic field while minimising
off-target effects (64). In
environmental applications, core-shell structures are used in
pollutant adsorption, water purification and photocatalysis. In
energy and electronics, they play a critical role in solar cells,
fuel cells, and advanced sensors by enhancing efficiency and
stability. Catalysis is another key area where core-shell
nanoparticles are utilised, with the shell often serving to protect
the catalytic core while allowing reactant access. Core-shell
nanoparticles are synthesised using various techniques, such as
co-precipitation, sol-gel processes, layer-by-layer assembly and
chemical vapour deposition. These methods enable precise control
over the size, morphology and composition of the particles
(61,65). Surface functionalisation is often
employed to introduce specific ligands or coatings that enhance
compatibility with biological systems or improve stability in
various environments. Despite their numerous advantages,
challenges, such as scalability of production, potential
environmental and biological impacts, and cost-effective synthesis
need to be addressed (64,66,67).
Ongoing research is focused on optimising fabrication techniques,
exploring sustainable materials and ensuring safe use (68). Core-shell nanoparticles represent a
versatile and innovative class of materials with significant
potential to advance technology and address critical challenges in
healthcare, energy and environmental sustainability.
Dendrimers
Dendrimers are highly branched, three-dimensional
macromolecules with a well-defined, tree-like structure. These
nanoscale materials, typically ranging from 1 to 10 nanometres in
size, are synthesised in a controlled, stepwise manner to achieve
uniformity in size, shape and surface functionality. The unique
architecture of dendrimers consists of three main components: A
central core, repetitive branching units, and peripheral functional
groups. This structure endows dendrimers with a high degree of
molecular precision, large surface area, and tuneable properties,
making them highly versatile in a variety of applications (67). The interior of dendrimers features
void spaces that can encapsulate guest molecules, while the
terminal groups on their surface can be functionalised to interact
with specific targets. The high density of functional groups on the
surface allows for multivalency, which is particularly valuable in
applications requiring targeted interactions, such as drug
delivery, gene therapy and diagnostics. Dendrimers are classified
by generations, with each successive generation adding more
branching layers and increasing size and functionality (69). In biomedical applications,
dendrimers have shown promise as drug delivery vehicles due to
their ability to encapsulate drugs within their internal cavities
or conjugate drugs to their surface functional groups. This dual
functionality enables controlled drug release, improved solubility
and targeted delivery to specific cells or tissues, reducing
side-effects. They are also used in gene therapy as carriers for
nucleic acids, enhancing stability and delivery efficiency.
Additionally, dendrimers are employed in diagnostics as imaging
agents and biosensors, benefiting from their multivalency and high
surface reactivity (67,70). In environmental science, dendrimers
are utilised for water purification, pollutant removal and as
catalysts or photocatalysts for environmental remediation. Their
functional groups can be tailored to bind specific contaminants,
enabling selective and efficient removal. In materials science,
dendrimers contribute to the development of advanced coatings,
adhesives and nanocomposites, providing improved mechanical,
thermal and optical properties. They are also employed in
electronics and energy applications, such as in the fabrication of
LEDs and fuel cells. Dendrimers are synthesised using two main
approaches: Divergent synthesis, where branching units are added
outward from the core, and convergent synthesis, where dendrons
(branching segments) are built separately and then attached to the
core. Both methods provide precise control over structure and
functionality, although they differ in scalability and complexity
(71). Despite their advantages,
dendrimers face challenges such as high production costs, potential
cytotoxicity, and complex synthesis processes. Advances in
synthetic methodologies, the development of biocompatible
dendrimers, and cost-effective production techniques are crucial
for their broader adoption. Dendrimers represent a cutting-edge
class of nanomaterials with immense potential to transform fields
such as medicine, environmental science and materials engineering
due to their unique architecture and versatile properties (36,67,43,44).
Despite the advantages of MetNPs, conventional
methods of synthesis often pose significant environmental and
health risks. Traditional chemical synthesis typically involves
toxic reagents, hazardous solvents, and high energy inputs, leading
to harmful by-products that can impact both human health and
ecosystems. A number of nanoparticle synthesis methods (e.g.,
chemical reduction, pyrolysis and chemical vapour deposition) rely
on toxic chemicals, high energy inputs and non-renewable resources.
These processes can generate hazardous waste, consume large amounts
of energy and can lead to the release of pollutants, including
toxic solvents used in chemical reactions and by-products that are
harmful to ecosystems. Energy consumption can also lead to
increased carbon emissions. Some conventional synthesis methods use
hazardous precursors and produce toxic by-products. Different
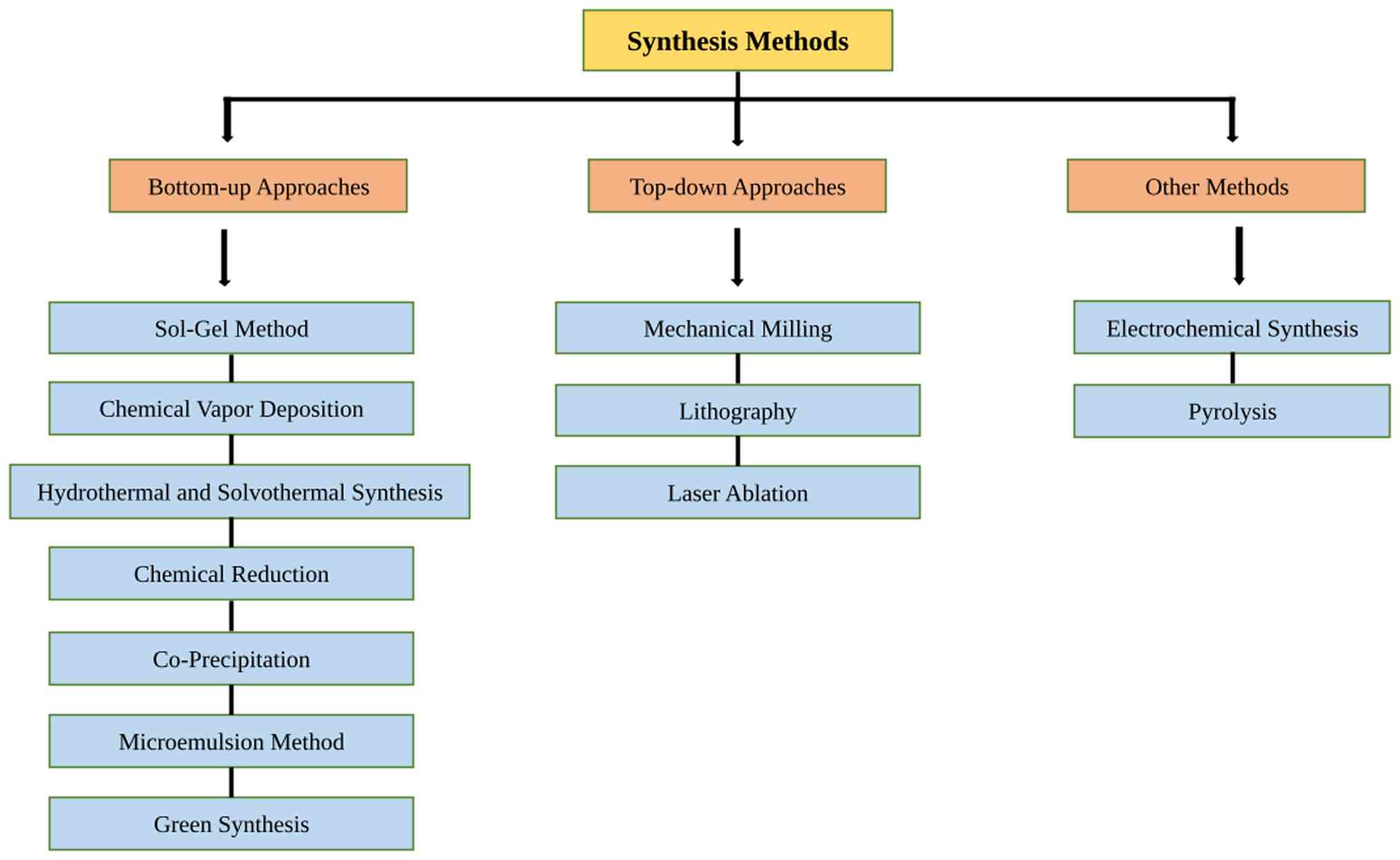
synthesis methods of nanoparticles are illustrated in Fig. 1.
3. Biosynthesis of nanoparticles
Biosynthesis is critical for preventing the
generation of unwanted or harmful by-products by establishing
dependable, sustainable synthesis processes that are eco-friendly.
This objective requires optimal solvent systems and natural
resources. Some basic aspects of green synthesis may thus be
described by numerous components, such as waste minimisation,
pollution reduction, and the use of better (non-toxic) solvents.
The green production of MNPs has been used to accommodate a variety
of biological components, including bacteria, fungi and algae
(3). Green synthesis involves the
use of plant extracts, microorganisms, and enzymes as reducing and
stabilising agents, thereby reducing the need for harmful chemicals
and minimising environmental impact. MetNPs, including those made
from silver, gold, copper and zinc oxide, have special properties
such as high surface-to-volume ratios, quantum effects, and
tuneable optical and catalytic properties, making them highly
valuable in diverse fields such as catalysis, medicine,
environmental remediation and electronics. However, conventional
methods of nanoparticle synthesis often incorporate the use of
toxic chemicals, high energy input and environmentally harmful
by-products (8,3,72).
The vast surface area of nanoparticles renders them ideal for a
variety of applications, including medicine (73), cosmetics (74), food chemicals, drug delivery,
biosensors, bioimaging and cancer treatment (75). Nanoparticles have gained popularity
among scientists globally due to their potential applications in
areas of research and technology and are gaining popularity in
medical research due to their small size, vast surface area,
solubility and potential applications. The mechanisms of the
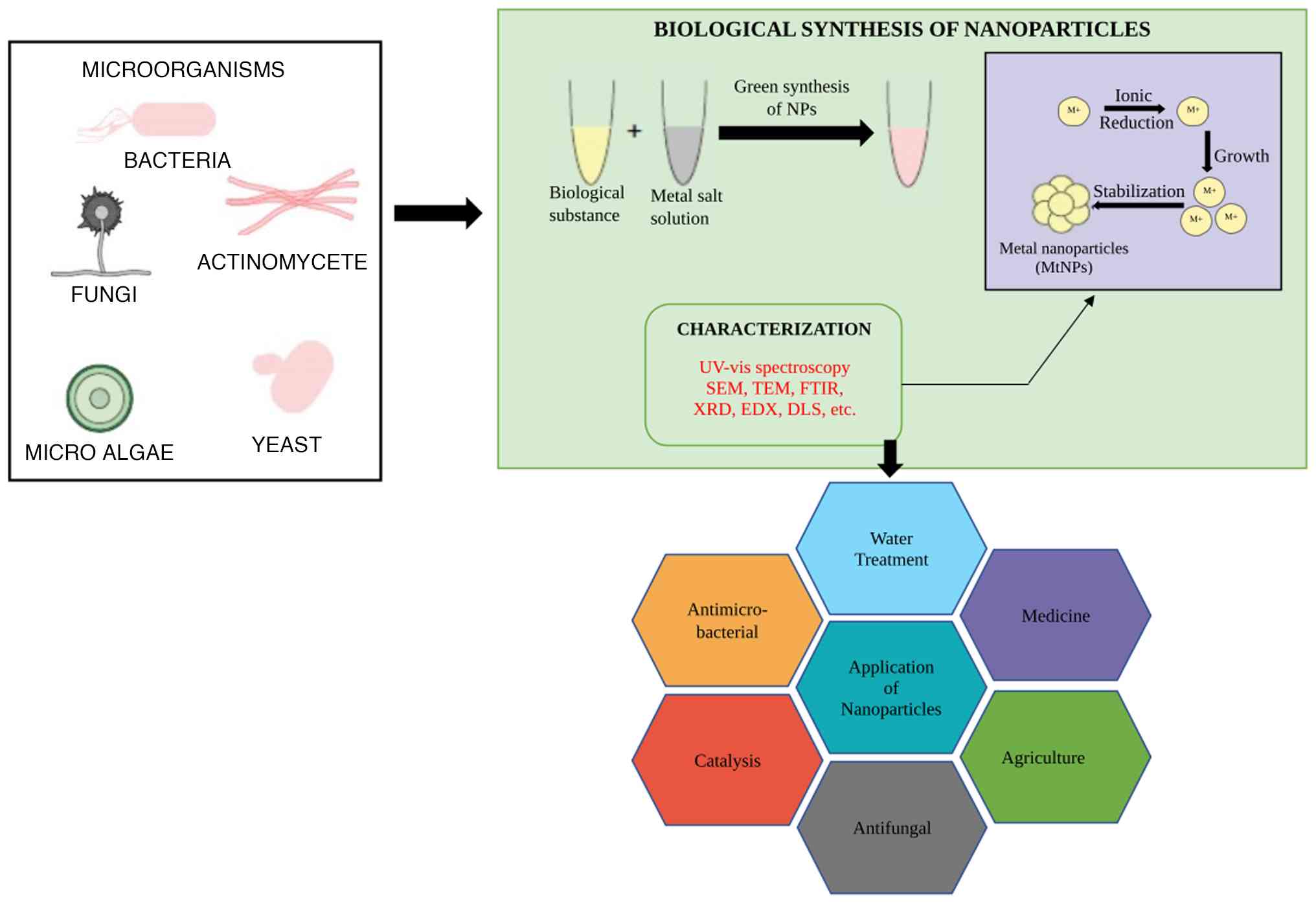
synthesis of nanoparticles using the green method is depicted in
Fig. 2. The mechanisms of green
synthesis for nanoparticles along with their size, characteristics,
applications and synthesis methods are presented in Table III.
 | Table IIISummary of the mechanisms of the
green synthesis for nanoparticles along with their size,
characteristics, applications and synthesis methods. |
Table III
Summary of the mechanisms of the
green synthesis for nanoparticles along with their size,
characteristics, applications and synthesis methods.
| Metal | Synthesis
mechanism | Size range | Characteristic | Application | Synthesis
approach | (Refs.) |
|---|
| Silver (Ag) | Reduction of
Ag+ ions using plant extracts, microbes, or enzymes | 5-100 nm | High antimicrobial
activity, strong surface plasmon resonance, good electrical and
thermal conductivity | Antimicrobial
coatings, wound healing, drug delivery, water purification,
biosensors | Plant extracts such
as Azadirachta indica, Moringa oleifera and
Aloe vera; microbial reduction by Escherichia
coli and Pseudomonas aeruginosa; enzyme
catalysis | (143-145) |
| Gold (Au) | Reduction of
Au3+ ions with natural reducing agents (phytochemicals
and enzymes) | 10-100 nm | Biocompatible,
non-toxic, and excellent optical properties, especially in drug
delivery and diagnostics | Cancer therapy,
drug delivery, biosensing, photothermal therapy, and
diagnostics | Plant extracts such
as Coriandrum sativum; fungal and bacterial reduction
by Fusarium and Bacillus subtilis; and
enzyme-assisted reduction by nitrate reductase | (146,147) |
| Palladium (Pd) | Reduction of
Pd2+ ions by plant compounds or microbial
metabolites | 5-50 nm | Catalytic
properties, biocompatible, can be functionalised for selective
catalysis | Catalysis in
organic reactions (hydrogenation, oxidation), electronics, ad
hydrogen storage | Plant extracts such
as Jatropha curcas; bacterial reduction by
Pseudomonas; enzymatic reduction by laccase | (148) |
| Platinum (Pt) | Reduction of
Pt4+ ions by enzymes, plant metabolites, or microbial
activity | 5-50 nm | High catalytic
activity, biocompatibility, and stability under high temperature
and pressure | Catalysis in fuel
cells, sensors, drug delivery, and hydrogenation reactions | Plant extracts such
as Aloe vera; microbial reduction by Saccharomyces
cerevisiae; enzyme catalysis by nitrate reductase | (147) |
| Iron (Fe) | Reduction of
Fe3+ or Fe2+ ions by biological agents
(bacteria, fungi, and plant extracts) | 10-100 nm | Magnetic
properties, low toxicity, biocompatible, abundant and
cost-effective | Water treatment,
magnetic resonance imaging (MRI), drug delivery, and environmental
remediation | Plant extracts such
as Moringa oleifera; fungal reduction by
Aspergillus; bacterial reduction by Geobacter
sulfurreducens | (88,90) |
| Selenium (Se) | Reduction of
SeO32- ions to Se nanoparticles by plants or
bacteria | 10-200 nm | Antioxidant
properties, biocompatible, and capable of enhancing immune
responses | Antioxidant, cancer
therapy, and environmental cleanup (removal of heavy metals) | Plant extracts such
as Coriandrum sativum; bacterial reduction by
Bacillus subtilis; enzymatic reduction by laccase | (118,149) |
| Copper (Cu) | Reduction of
Cu2+ ions using plant polyphenols or bacterial
enzymes | 10-100 nm | High electrical and
thermal conductivity, antimicrobial activity, and capable of
oxidising organic compounds | Electrical
applications, antimicrobial agents, catalysis, and environmental
remediation | Plant extracts such
as Camellia sinensis; microbial reduction by
Pseudomonas aeruginosa; enzyme-assisted reduction by nitrate
reductase and laccase | (116,150) |
4. Principles of biosynthesis
The biosynthesis of MetNPs involves the reduction of
metal ions to nanoparticles using naturally occurring biological
entities. This method eliminates the need for harsh chemicals,
toxic solvents and energy-intensive processes, which are often
required in traditional chemical synthesis. The main principles
guiding green synthesis are simplicity, sustainability and
environmental safety. MetNPs derived from plant extracts are stable
and readily monodispersed by adjusting pH values, temperature
range, retention period and the ratio of mixing. MNPs formed using
green methods are derived from diverse plant extracts, such as neem
leaves (Azadirachta indica), basil leaves (Ocimum
tenuiflorum), curry leaves (Murraya koenigii), guava
leaves (Psidium guajava) and mango leaves (Mangifera
indica) (72,76,77).
5. Biological agents in green synthesis
Various biological agents have been explored for the
synthesis of green nanoparticles. These include the following:
Plant extracts
The green synthesis of nanoparticles using plant
extracts has emerged as an eco-friendly and sustainable approach to
nanomaterial production. This method leverages the rich diversity
of phytochemicals present in plants, such as flavonoids, alkaloids,
terpenoids, phenols and enzymes, which act as natural reducing and
stabilising agents. When plant extracts are exposed to metal ions,
these biomolecules facilitate the reduction of the ions to their
corresponding nanoparticles and stabilise them to prevent
aggregation (3,72,77).
This process is simple, cost-effective and scalable, making it an
attractive alternative to conventional chemical methods that often
involve toxic reagents and harsh conditions. Plant-mediated
synthesis is versatile and has been successfully employed to
produce a wide range of nanoparticles, including gold, silver,
copper and zinc oxide, with controlled size and morphology.
Extracts from plants such as Azadirachta indica (neem)
(8), Moringa oleifera,
Aloe vera (78) and
Ocimum sanctum (holy basil) have been extensively studied
for their efficacy in nanoparticle synthesis (79,80).
The resulting nanoparticles often exhibit enhanced biocompatibility
and bioactivity, rendering them suitable for applications in
medicine, environmental remediation, and agriculture. For instance,
silver nanoparticles synthesised using plant extracts have
demonstrated potent antimicrobial activity, while gold
nanoparticles have shown promise in drug delivery and cancer
therapy (3,8). This green synthesis approach aligns
with the principles of green chemistry by minimising waste,
reducing energy consumption and avoiding hazardous chemicals.
Despite its advantages, challenges such as achieving
reproducibility, controlling particle size and shape, and scaling
up the synthesis process remain (3,72,77,79,80).
Microorganisms
The green synthesis of nanoparticles using
microorganisms is an innovative and eco-friendly approach that
leverages the natural capabilities of bacteria, fungi and algae to
produce nanoparticles. Microorganisms are particularly effective in
nanoparticle synthesis because they possess enzymes and metabolites
that can reduce metal ions to their nanoparticle forms, while also
stabilising and shaping the nanoparticles during the process. This
biogenic method of synthesis is beneficial for producing
nanoparticles without the need for toxic chemicals, thus ensuring a
more sustainable and environmentally friendly production process.
Bacteria such as Escherichia coli, Pseudomonas
aeruginosa and Bacillus subtilis are commonly used in
the biosynthesis of MetNPs, including silver, gold, and copper.
These bacteria can secrete enzymes, such as nitrate reductase and
cytochrome, which play crucial roles in the reduction of metal
ions. Fungi, particularly species such as Aspergillus and
Fusarium, are also widely utilised for their ability to
synthesise nanoparticles extracellularly. The metabolites and
proteins produced by fungi not only reduce metal ions, but also
function as stabilising agents, controlling the size and shape of
the nanoparticles (81,82). Microalgae, such as Chlorella
and Spirulina, are used to produce nanoparticles such as
silver and selenium due to their high metal ion absorption
capabilities. The microbial synthesis of nanoparticles provides
numerous advantages, including the ability to precisely control the
size, shape, and surface properties of nanoparticles, which are
crucial for their functionality in various applications (83). These nanoparticles are often highly
biocompatible, making them suitable for biomedical applications
such as drug delivery, cancer therapy and biosensing, and find
applications in environmental remediation, where they can be used
for water purification, pollutant degradation, and heavy metal
removal. Despite its advantages, microbial synthesis of
nanoparticles does face challenges, such as scalability for
industrial production, the variability of microbial strains, and
the need for optimisation of culture conditions. Additionally,
ensuring safety and minimising the toxicity of biogenic
nanoparticles is an ongoing area of research. However, the use of
microorganisms in green nanoparticle synthesis represents a
promising, sustainable alternative to traditional chemical methods,
offering both environmental and economic benefits in nanotechnology
(32,83).
Algae and enzymes
The green synthesis of nanoparticles using algae and
enzymes is an emerging and eco-friendly approach that combines the
unique capabilities of biological systems to produce nanoparticles
in a sustainable manner. Algae, both microalgae and macroalgae, are
increasingly being explored for nanoparticle synthesis due to their
high metal ion absorption capacity and ability to secrete
biomolecules, including proteins, polysaccharides and secondary
metabolites, which serve as reducing and stabilising agents.
Enzymes, on the other hand, are natural catalysts that facilitate
the reduction of metal ions to nanoparticles, providing precise
control over the size and shape of the particles. This approach
avoids the use of toxic chemicals, making it more environmentally
friendly compared with conventional chemical synthesis methods.
Algae such as Chlorella, Spirulina, Dunaliella
and Phaeodactylum have been used for synthesising various
MetNPs such as silver, gold and copper (61). These algae possess the ability to
accumulate and reduce metal ions through their biochemical
pathways, producing nanoparticles that are often biocompatible and
environmentally safe. The synthesis process involves the exposure
of algae to metal salt solutions, where enzymes and other cellular
components facilitate the reduction of metal ions to their
nanoparticle forms. The biogenic nanoparticles formed are
stabilised by the natural biomolecules secreted by the algae, which
also help control their size, shape and surface properties. Enzymes
play a crucial role in the green synthesis of nanoparticles due to
their high specificity and catalytic efficiency (84). Enzymes such as nitrate reductase,
dehydrogenases and laccase are often used to reduce metal ions to
their nanoparticle forms and are capable of selectively reducing
metal ions, allowing for the controlled synthesis of nanoparticles
with well-defined characteristics. In addition to their reducing
ability, enzymes can stabilise the nanoparticles by coating them
with functional groups that enhance their solubility and
biocompatibility. The use of algae and enzymes in nanoparticle
synthesis provides several advantages. This method is
cost-effective, environmentally friendly and scalable, with algae
being abundant and easy to culture. The nanoparticles produced are
often highly biocompatible, making them ideal for applications in
medicine, such as drug delivery, imaging and antimicrobial
treatments. In environmental applications, algae-based
nanoparticles are used for water purification, pollutant removal
and the remediation of heavy metals (85). Additionally, enzyme-mediated
nanoparticle synthesis is employed in catalysis and the development
of advanced materials. Despite the benefits, challenges remain in
optimising the synthesis process, such as improving the
reproducibility of nanoparticle production, controlling particle
size distribution and scaling up for industrial applications
(86). Furthermore, research is
ongoing to better understand the mechanisms involved in
algae-mediated and enzyme-mediated synthesis to enhance efficiency
and ensure the safety of the biogenic nanoparticles (87). Overall, the green synthesis of
nanoparticles using algae and enzymes is a promising and
sustainable method that holds great potential for applications in
diverse fields, from environmental protection to biomedical
engineering.
6. Mechanisms of green synthesis
The green synthesis process typically follows three
key steps:
Reduction of metal ions
The biological agents reduce the metal ions (e.g.,
Ag+, Au3+, Cu2+) to their
corresponding nanoparticles. The reduction of metal ions is a
central mechanism in the green synthesis of nanoparticles, where
metal ions are reduced to their elemental or nanoparticulate state
with the aid of biological agents, such as plant extracts,
microorganisms and enzymes. In this process, metal ions, often in
their higher oxidation states (e.g., Ag+,
Au3+, Cu2+), are converted into their
zero-oxidation state (e.g., Ag0, Au0,
Cu0) through electron transfer facilitated by natural
reducing agents (18).
Phytochemicals in plant extracts, such as polyphenols, flavonoids
and terpenoids, function as electron donors, reducing metal ions
while being oxidised themselves. Similarly, microorganisms,
including bacteria and fungi, produce enzymes such as nitrate
reductase and laccase that catalyse the reduction of metal ions.
These enzymes typically transfer electrons from biological
substrates to metal ions, promoting nanoparticle formation. The
reduction of metal ions can also be accelerated by light in certain
cases, a process known as photoreduction, where light energy
induces the production of reactive oxygen species that aid in the
reduction. This reduction results in the formation of nanoparticles
that are stabilised by the biomolecules present in the biological
agents. These biogenic nanoparticles often exhibit enhanced
biocompatibility and tuneable properties, making them ideal for
applications in medicine, environmental science, and materials
engineering (18,88).
Stabilisation of nanoparticles
Biomolecules from the biological agents stabilise
the synthesised nanoparticles, preventing agglomeration and
controlling size and shape. In the green synthesis of
nanoparticles, stabilisation is a crucial mechanism that ensures
the nanoparticles retain their size, shape, and functionality
without agglomerating or degrading over time. Once metal ions are
reduced to form nanoparticles, the biological agents involved in
the synthesis, such as plant extracts, microorganisms, or enzymes,
play an essential role in stabilising the nanoparticles (89). This stabilisation is typically
achieved through the adsorption of biomolecules onto the surface of
the nanoparticles. Phytochemicals such as polyphenols, flavonoids
and proteins, which are naturally present in plants, provide
functional groups such as hydroxyl, carboxyl and amino groups that
form a protective layer around the nanoparticles. This surface
coating prevents the nanoparticles from aggregating by hindering
interactions between particles through electrostatic repulsion or
steric hindrance (90). In
microbial systems, proteins, enzymes and polysaccharides secreted
by bacteria, fungi and algae can serve as capping agents that not
only stabilise the nanoparticles but also impart specific
properties, such as increased biocompatibility or targeted
functionality. These biomolecules function by binding to the
surface of the nanoparticles, reducing surface energy and
preventing the particles from clumping together. Furthermore, the
size and shape of the nanoparticles can be controlled by adjusting
the concentration and type of stabilising agents used, which is
essential for tailoring the properties of the nanoparticles for
specific applications. This stabilisation mechanism also helps in
maintaining the dispersion of nanoparticles in solution, preventing
agglomeration and enhancing their stability under varying
environmental conditions. The biocompatible nature of the
stabilising agents makes these nanoparticles suitable for
applications in fields such as medicine, environmental remediation
and sensor technology, where prolonged stability and safe
interaction with biological systems are paramount (3,72,80,91).
Purification and characterisation
The synthesised nanoparticles are purified and
characterised using techniques, such as scanning electron
microscopy (SEM), UV-visible spectroscopy, X-ray diffraction (XRD)
and transmission electron microscopy (TEM). In the green synthesis
of nanoparticles, purification and characterisation are essential
steps that ensure the quality, size, shape and functionality of the
synthesised nanoparticles. After the nanoparticles are formed
through the reduction of metal ions by biological agents (such as
plant extracts, microorganisms, or enzymes), they must be purified
to remove any residual reactants, by-products, or unreacted metal
ions. This purification is typically achieved through methods, such
as centrifugation, filtration, dialysis, or gel electrophoresis,
which help separate the nanoparticles from unwanted materials.
These methods rely on differences in particle size, charge, or
solubility to isolate the nanoparticles in their purest form,
ensuring that they are free from contaminants that could affect
their properties or hinder their intended applications (6,80,92,93).
Once purified, the nanoparticles are characterised
to determine their physical, chemical and structural properties.
Characterisation techniques provide insight into the size, shape,
morphology, surface charge and composition of the nanoparticles,
which are critical for assessing their suitability for specific
applications. Common techniques used in the characterisation of
green-synthesised nanoparticles include the following:
i) TEM and SEM. These techniques provide
detailed images of the size, shape and surface morphology of the
nanoparticles, enabling researchers to assess their uniformity and
dispersion (6).
ii) XRD. XRD aids in the determination of the
crystalline structure of the nanoparticles and can provide
information about their phase composition (80).
iii) UV-Vis spectroscopy. UV-Vis spectroscopy
is used to monitor the optical properties of nanoparticles, such as
their absorption spectra, which are influenced by particle size and
shape. It can also confirm the formation of nanoparticles by
detecting the characteristic plasmon resonance peaks (92).
iv) Dynamic light scattering (DLS). DLS
measures the size distribution and zeta potential of nanoparticles,
providing information on their stability and surface charge
(93).
v) Fourier transform infrared spectroscopy
(FTIR). FTIR is used to analyse the functional groups present
on the surface of the nanoparticles, which helps identify the
biomolecules responsible for stabilisation and capping (94).
vi) Energy-dispersive X-ray spectroscopy
(EDX). EDX, often coupled with SEM or TEM, provides elemental
analysis of the nanoparticles, confirming their composition and
verifying the successful incorporation of metal ions (95).
Through purification and characterisation,
researchers can ensure that the green-synthesised nanoparticles
possess the desired properties, are free of contaminants, and are
suitable for various applications in medicine, environmental
remediation, catalysis and materials science. These steps are
crucial in ensuring that the nanoparticles meet the necessary
quality standards for their intended uses.
7. MetNPs synthesised using the green
method
Various types of nanoparticles can be synthesised
using biological sources, including plants, fungi and bacteria,
which serve as reducing and stabilising agents. Silver
nanoparticles may be readily synthesised using a silver metal ion
solution and a reducing biological agent. Silver nanoparticles have
been reported to be synthesised from a variety of medicinal plants,
including Cinnamomum camphora (96), Oryza sativa and Zea
mays. Silver ion reduction is one of the simplest and most
cost-effective processes for producing silver nanoparticles. Kumar
et al (97) synthesised
silver nanoparticles by reducing an AgNO3 solution with
Nelumbo nucifera plant extract. Philip (98) produced silver nanoparticles from
the leaf extract of Hibiscus rosa-sinensis. In 2009, Bar
et al synthesised silver nanoparticles using Jatropha
curcas seed extract after heating an aqueous solution at 80˚C;
the appearance of a crimson colour suggested the formation of
silver nanoparticles (99).
Gold nanoparticles have a variety of uses in
biomedical research, including the rapid detection and
identification of heart disorders, cancer and infectious pathogens
(100). Shankar et al
(101) also stated that the
synthesised nanoparticles were of different forms, including
spherical, decahedral, triangular and icosahedral structures. The
same group produced gold nanoparticles from neem extract (102). According to Song et al
(103), temperature plays an
essential role in the development of specific shapes and sizes of
synthesised gold nanoparticles. Gold nanoparticles were also
synthesised using a variety of biological sources, including
Bacillus marisflavi, Coffea arabica (104), Aeromonas hydrophila
(105,106) and Croton sparsiflorus leaf
extract (107). Both palladium
and platinum are highly valuable metals that appear silvery white.
Several plant species, including Cinnamomum zeylanicum,
Anogeissus latifolia, Curcuma longa, Diospyros
kaki, Gardenia jasminoides, Cinnamomum camphora,
Glycine max and Musa paradisiaca, have been used to
prepare palladium and platinum nanoparticles (91). Ahmed et al (8) discovered that proteins were
responsible for converting chloroplatinic ions into platinum
nanoparticles. Ascorbic acid, terpenoids, amino acids, specific
proteins and gallic acid found in basil leaf extract all played
major roles in reducing platinum ions. The green synthesis method
for the development of zero-valent iron nanoparticles has been a
major approach for the treatment of brominated organic compounds,
pesticides, azo dyes, alkaline-earth metals, malachite green,
nitrate, monochlorobenzene, antibiotics, and for converting metals
such as chromium, cobalt, and copper (89,108,109). Green-synthesised iron
nanoparticles have been produced from several plants, including
common lantana (Lantana camara), water hyacinth
(Eichhornia crassipes), and sensitive plants (Mimosa
pudica) (110). The green
synthesis of selenium nanoparticles using plants is an
environmentally friendly, cost-effective and non-toxic technology.
Garlic (Allium sativum) bud extract, for example, has been
utilised to produce selenium nanoparticles with high antioxidant
activity, as validated using ferric reducing antioxidant power
(FRAP), 2,2'-azinobis (3-ethylbenzothiazoline-6-sulfonic acid and
2,2-diphenyl-1-picrylhydrazyl assays (111). Tea extract (Camellia
sinensis) has also been used to produce selenium nanoparticles
with antioxidant properties. Furthermore, horseshoe geranium
(Pelargonium zonale) leaf extract has produced selenium
nanoparticles with substantial antibacterial and antifungal
activity against pathogens (112). These nanoparticles also assist in
the removal of heavy metals from the environment. Copper
nanoparticles exhibit therapeutic potential, including
antimicrobial, antifungal and antiviral activities, and have been
synthesised using plant-mediated methods from a variety of plants,
including fire lily (Gloriosa superba L.), common grape
(Vitis vinifera), oleander (Nerium oleander), Ceylon
caper (Capparis zeylanica), and jackfruit champa
(Artabotrys odoratissimus) (113). Copper nanoparticles synthesised
from fire lily (Gloriosa superba L.) leaf extract and
pomegranate (Punica granatum) bark extract have been shown
to exhibit enhanced reduction and capping properties (114,115). Copper nanoparticles have also
been synthesised using various plant extracts, with sizes ranging
from 4 to 100 nm, including Japanese magnolia (Magnolia
kobus) and angel's trumpet (Datura innoxia) (113).
8. Characterisation techniques
Characterisation approaches are critical for
understanding the characteristics, structure and behaviour of
nanoparticles, and aid in determining shape, particle size, surface
charge, chemical composition, crystallinity and other essential
properties. The various characterisation techniques used for
nanoparticles, as well as a description of each, is presented in
Table IV.
 | Table IVTypes of characterisation techniques
for nanoparticles. |
Table IV
Types of characterisation techniques
for nanoparticles.
| Techniques |
Characteristics | Typical output | (Refs.) |
|---|
| Transmission
electron microscopy: | Provides accurate
images for determining nanoparticle size, shape, and structure. A
beam of electrons passes through the nanoparticle sample, and the
interaction of electrons with the atoms in the sample produces a
highly detailed image. Used to study morphology, particle size
distribution, and structural details down to the atomic level. | High-resolution
images | (151) |
| Scanning electron
microscopy; | Provides surface
images and information about the morphology and topography of
nanoparticles. Electrons are focused on the surface of the
nanoparticles, and secondary electrons emitted from the surface
create a detailed image. Used to analyse surface texture and
particle shapes with a slightly lower resolution than TEM. | Surface images | (152) |
| Dynamic light
scattering; | Measures the
hydrodynamic size and size distribution of nanoparticles in
suspension. Measures the scattering of light caused by the Brownian
motion of nanoparticles in a liquid medium, providing size
distribution. Commonly used for determining particle size in
colloidal suspensions and solutions. | Size distribution
graph | (153) |
| X-ray
diffraction; | Determines the
crystalline structure and phase composition of nanoparticles.
X-rays are directed at the sample, and the diffraction pattern of
the rays reveals the crystal structure. Used to identify
crystallinity, lattice structure, and particle size (through the
Debye-Scherrer equation). | Diffraction
patterns | (130) |
| Fourier-transform
infrared spectroscopy; | Identifies chemical
bonds and functional groups on the surface of nanoparticles.
Infrared radiation is passed through the sample, and the absorption
spectra are analysed to determine chemical bonds. Used to identify
surface modifications, coatings, and interactions between
nanoparticles and other molecules. | IR spectra
(functional groups) | (154) |
| Ultraviolet-visible
spectroscopy; | Measures the
optical properties, such as absorption and scattering, of
nanoparticles. Light in the ultraviolet and visible range is passed
through the sample, and the absorbance is measured. Commonly used
to confirm the formation of nanoparticles (e.g., for metal
nanoparticles like gold and silver) and study their optical
properties. | Absorption peak
(SPR band) | (9) |
| Atomic force
microscopy: | Provides 3D
topographical information and surface details at the nanoscale. A
cantilever with a sharp tip scans the surface of the nanoparticles,
and the interaction between the tip and sample generates a 3D
surface map. Used to examine surface roughness, particle height,
and nanoscale surface interactions. | 2D/3D surface
images | (155) |
| Energy dispersive
X-ray spectroscopy; | Determines the
elemental composition of nanoparticles. Attached to an SEM or TEM,
EDX detects X-rays emitted from the sample when bombarded by
electrons, identifying the elements present. Used for elemental
analysis and mapping, useful for confirming the composition of
nanoparticles. | Elemental
spectra | (152) |
| Thermo-gravimetric
analysis; | Measures changes in
the physical and chemical properties of nanoparticles as a function
of temperature. A sample is heated, and changes in weight are
monitored to study thermal stability and decomposition. Used to
determine thermal stability, degradation temperatures, and the
presence of organic or inorganic components on the nanoparticle
surface. | Weight-loss
curves | (156) |
| Zeta potential
analysis; | Determines the
surface charge and stability of nanoparticles in suspension.
Measures the electrophoretic mobility of particles in an applied
electric field, providing information about surface charge and
dispersion stability. Used to evaluate colloidal stability and
predict nanoparticle aggregation behaviour. | Zeta potential
values (mV) | (157) |
9. Applications of biosynthesised
MetNPs
Green-synthesised MetNPs derived from
environmentally friendly biological agents, such as plants,
bacteria, fungi, or algae have a wide range of applications due to
their unique properties, biocompatibility and a low environmental
impact (75). In biomedicine,
silver and gold nanoparticles exhibit potent antibacterial
properties, rendering them useful in wound dressings, medical
device coatings and disinfectants (97). They also serve as effective drug
delivery systems, enhancing treatment outcomes, while minimising
adverse effects. Additionally, these nanoparticles are being
investigated for cancer therapy and antioxidant applications, such
as reducing oxidative stress-related disorders (75,105). In environmental remediation,
nanoparticles such as iron, silver and selenium remove heavy metals
and degrade contaminants from polluted water and soil. They are
also utilised in air purification systems to break down hazardous
substances (66). In agriculture,
green-synthesised nanoparticles act as nanopesticides and
fertilisers, stimulating plant growth and enhancing crop yield.
Silver nanoparticles are used to extend the shelf life of food
through antimicrobial packaging, whereas selenium nanoparticles are
employed as nutritional supplements due to their increased
bioavailability (11,78,111). These nanoparticles also provide
benefits in skincare products, sunscreens, and antimicrobial
fabrics. Furthermore, they play crucial roles in catalysis,
biosensing and imaging for medical diagnostics and environmental
monitoring, demonstrating their adaptability across multiple
sectors (69).
10. Current challenges and future
prospects
Despite recent advancements, green-synthesised
MetNPs face challenges related to scalability and consistency
during the transition from laboratory-scale to industrial-scale
production. The diversity of biological agents, such as plants and
bacteria, can lead to variations in nanoparticle characteristics.
Furthermore, the mechanisms underlying nanoparticle formation
remain insufficiently understood, limiting optimisation. Concerns
regarding toxicity and long-term environmental effects require
further investigation (68,87).
While biosynthesised MetNPs demonstrate substantial
promise for biomedical, environmental and industrial applications,
several critical obstacles remain for their practical translation,
with toxicity being one of the most significant challenges.
Biological molecules involved in synthesis (proteins,
polysaccharides, secondary metabolites, etc.) can unpredictably
alter nanoparticle surface chemistry, affecting cellular uptake and
potentially leading to variable genotoxicity and long-term
environmental effects (116).
Additionally, variations in size, shape and capping agents can
result in markedly different biological responses, complicating
risk assessment. Scalability represents another major bottleneck. A
number of biosynthetic processes are limited by slow reaction
rates, poor metal-ion conversion efficiency and strong dependence
on biological sources (plant extracts, microbial cultures, etc.),
making it difficult to achieve reproducible industrial-scale
production. The lack of standardised purification processes can
introduce impurities or residual biomolecules, contributing to
inconsistencies in stability and performance. Furthermore,
regulatory challenges continue to hinder commercialisation due to
the absence of harmonised global guidelines for assessing safety,
environmental impact, and long-term biodegradation of
biosynthesised nanomaterials. Regulatory agencies also require
extensive datasets to differentiate laboratory-based findings from
real-world exposure scenarios, which are currently limited
(105,117,118).
Looking ahead, addressing these challenges will
require a multi-pronged strategy. Advances in the mechanistic
understanding of biosynthesis, using omics tools, real-time
spectroscopy and molecular-level modelling, may allow improved
control over nanoparticle morphology and stability. The development
of high-throughput, reactor-based green synthesis systems could
overcome yield limitations and enable consistent large-scale
production. Establishing standardised international protocols for
toxicity testing, quality control and environmental risk assessment
will be essential for regulatory approval (94). Finally, future research should
emphasise application-specific surface functionalisation, hybrid
biochemical synthesis approaches and life-cycle assessment to
ensure that biosynthesised MNPs are deployed safely, sustainably
and at a commercial scale.
Future advances in nanotechnology and biotechnology
may further enhance scalability and precision. Standardised
methodologies and comprehensive safety evaluations will be critical
for widespread adoption. Future research is likely to focus on
improving sustainability and expanding applications in industries
such as energy, agriculture and medicine.
11. Conclusion
The purpose of this review aligns with the growing
emphasis on sustainable nanotechnology, mechanistic insight and
translational potential. MetNPs, particularly those synthesised
through biogenic and green routes, are a rapidly evolving class of
functional materials with major implications for healthcare,
catalysis, sensing and environmental remediation. However, the
existing literature is fragmented across individual studies,
lacking an integrated approach that connects biosynthesis
mechanisms with application-driven performance and safety
considerations. The present review aimed to address this gap by
providing a comprehensive, critically evaluated overview of recent
advances, highlighting sustainable synthesis innovations,
structure-property relationships, challenges in toxicity and
scalability and future research directions.
The journey of nanotechnology, begun by visionaries
such, as Richard Feynman and Norio Taniguchi, has resulted in the
revolutionary invention of MetNPs. These small entities, having
unique features arising from their nanoscale size, have several
uses in health, environmental remediation, and electronics. The
green synthesis of MetNPs provides a viable alternative to
traditional approaches, since it employs biological agents that
reduce environmental effects while promoting sustainability.
Despite the potential benefits, issues such as scalability,
consistency and safety persist. Addressing these difficulties is
critical for moving green nanotechnology beyond laboratory research
and into practical applications. Future studies are thus warranted
to focus on optimising synthesis procedures, understanding the
mechanisms underlying nanoparticle creation and conducting safety
studies for wider deployment. As the discipline advances,
incorporating nanotechnology into common applications holds immense
promise for improving human health, safeguarding the environment
and driving technological innovation.
Acknowledgements
The authors would like to extend their sincere
gratitude to Shri Guru Ram Rai University, Dehradun, for providing
guidance, the facilities and the support that were required for the
completion of the present review.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
RV designed the scope of the study, AK edited the
manuscript, and RBA was involved in the literature review and
writing of the manuscript. RS designed the structure of the review.
NG, BSR and IR guided the direction of the review. RV and RS
supervised the overall preparation of the review. All the authors
have read and approved the final version of the manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kim DH, Gopal J and Sivanesan I:
Nanomaterials in plant tissue culture: The disclosed and
undisclosed. RSC Adv. 7:36492–36505. 2017.
|
|
2
|
Taniguchi N: On the basic concept of
nanotechnology. Proceedings of the International Conference on
Production Engineering, Tokyo, Japan, pp18-23, 1974.
|
|
3
|
Mukherjee A: Green synthesis of metal
nanoparticles. Int J Multidiscip Res. 6:1–8. 2024.
|
|
4
|
Eric DK: Engines of creation: The coming
era of nanotechnology. New York: Anchor, 1986.
|
|
5
|
Thrall JH: Nanotechnology and medicine.
Radiology. 230:315–318. 2004.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rauwel P, Küünal S, Ferdov S and Rauwel E:
A review on the green synthesis of silver nanoparticles and their
morphologies studied via TEM. Adv Mater Sci Eng. 2015:1–9.
2015.
|
|
7
|
Monica RC and Cremonini R: Nanoparticles
and higher plants. Caryologia. 62:161–165. 2009.
|
|
8
|
Ahmed S, Saifullah Ahmad M, Swami BL and
Ikram S: Green synthesis of silver nanoparticles using
Azadirachta indica aqueous leaf extract. J Radiat Res Appl
Sci. 9:1–7. 2016.
|
|
9
|
Kelly KL, Coronado E, Zhao LL and Schatz
GC: The optical properties of metal nanoparticles: The influence of
size, shape, and dielectric environment. J Phys Chem B.
107:668–677. 2003.
|
|
10
|
Zeng J, Zhang Q, Chen J and Xia Y: A
comparison study of the catalytic properties of Au-based nanocages,
nanoboxes, and nanoparticles. Nano Lett. 10:30–35. 2010.
|
|
11
|
Chaudhry N, Dwivedi S, Chaudhry V, Singh
A, Saquib Q, Azam A and Musarrat J: Bio-inspired nanomaterials in
agriculture and food: Current status, foreseen applications and
challenges. Microb Pathog. 123:196–200. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Kolahalam LA, Kasi Viswanath IV, Diwakar
BS, Govindh B, Reddy V and Murthy YLN: Review on nanomaterials:
Synthesis and applications. Mater Today Proc. 18:2182–2190.
2019.
|
|
13
|
Meyer RA, Sunshine JC and Green JJ:
Biomimetic particles as therapeutics. Trends Biotechnol.
33:514–524. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Singh R, Altaee A and Gautam S:
Nanomaterials in the advancement of hydrogen energy storage.
Heliyon. 6(e04487)2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Liu Y, Liu X, Wang X and Jiang H:
AI-empowered electrochemical sensors for biomedical applications:
Technological advances and future challenges. Biosensors (Basel).
15(487)2025.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zhang B, Biswal BK, Zhang J and
Balasubramanian R: Hydrothermal treatment of biomass feedstocks for
sustainable production of chemicals, fuels, and materials: Progress
and perspectives. Chem Rev. 123:7193–7294. 2023.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Roduner E: Size matters: Why nanomaterials
are different. Chem Soc Rev. 35:583–592. 2006.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Aijaz A, Fujiwara N and Xu Q: From
metal-organic framework to nitrogen-decorated nanoporous carbons:
High CO2 uptake and efficient catalytic oxygen
reduction. J Am Chem Soc. 136:6790–6793. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Gabiam EN, Erk N, Genc AA, Sert B,
Harputlu E and Lotfy HM: Development of a highly sensitive
electrochemical sensor for detecting the antipsychotic agent
cariprazine in pharmaceutical capsules and biological fluids using
boron nitride quantum dots. ChemistrySelect. 10(e00548)2025.
|
|
20
|
Gabiam EN, Erk N, Genc AA, Bouali W,
Soylak M, Salamat Q and Ahmed HEH: Synthesize of NiSe2/Mo-MOF
nanocomposite for sensitive determination of antiretroviral agent
dolutegravir in pharmaceutical formulations and biological fluids.
J Electroanal Chem. 986(119096)2025.
|
|
21
|
Wei C, Cho K and Srivastava D: Tensile
strength of carbon nanotubes under realistic temperature and strain
rate. Phys Rev B. 67(115407)2003.
|
|
22
|
Abdelgadir A, Adnan M, Patel M, Saxena J,
Alam MJ, Alshahrani MM, Singh R, Sachidanandan M, Badraoui R and
Siddiqui AJ: Probiotic Lactobacillus salivarius mediated synthesis
of silver nanoparticles (AgNPs-LS): A sustainable approach and
multifaceted biomedical application. Heliyon.
10(e37987)2024.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Singh P, Singh KR, Verma R, Singh J and
Singh RP: Efficient electro-optical characteristics of bioinspired
iron oxide nanoparticles synthesized by Terminalia chebula dried
seed extract. Mater Lett. 307(131053)2022.
|
|
24
|
Ling D, Lee N and Hyeon T: Chemical
synthesis and assembly of uniformly sized iron oxide nanoparticles
for medical applications. Acc Chem Res. 48:1276–1285.
2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Gubin SP, Koksharov YA, Khomutov GB and
Yurkov GY: Magnetic nanoparticles: Preparation, structure and
properties. Russ Chem Rev. 74:489–520. 2005.
|
|
26
|
Tai CY, Tai CT, Chang MH and Liu HS:
Synthesis of Magnesium Hydroxide and Oxide Nanoparticles Using a
Spinning Disk Reactor. Ind Eng Chem Res. 46:5536–5541. 2007.
|
|
27
|
Raghupathi KR, Koodali RT and Manna AC:
Size-dependent bacterial growth inhibition and mechanism of
antibacterial activity of zinc oxide nanoparticles. Langmuir.
27:4020–4028. 2011.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Fujishima A, Zhang X and Tryk DA:
TiO2 photocatalysis and related surface phenomena. Surf
Sci Rep. 63:515–582. 2008.
|
|
29
|
Qu X, Alvarez PJJ and Li Q: Applications
of nanotechnology in water and wastewater treatment. Water Res.
47:3931–3946. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Gupta AK and Gupta M: Synthesis and
surface engineering of iron oxide nanoparticles for biomedical
applications. Biomaterials. 26:3995–4021. 2005.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Armand M and Tarascon JM: Building better
batteries. Nature. 451:652–657. 2008.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Singh P, Kim YJ, Zhang D and Yang DC:
Biological synthesis of nanoparticles from plants and
microorganisms. Trends Biotechnol. 34:588–599. 2016.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Kahru A and Dubourguier HC: From
ecotoxicology to nanoecotoxicology. Toxicology. 269:105–119.
2010.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Geim AK and Novoselov KS: The rise of
graphene. Nat Mater. 6:183–191. 2007.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Baker SN and Baker GA: Luminescent carbon
nanodots: Emergent nanolights. Angew Chem Int Ed Engl.
49:6726–6744. 2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kumari A, Yadav SK and Yadav SC:
Biodegradable polymeric nanoparticles based drug delivery systems.
Colloids Surf B Biointerfaces. 75:1–18. 2010.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Vauthier C and Bouchemal K: Methods for
the preparation and manufacture of polymeric nanoparticles. Pharm
Res. 26:1025–1058. 2009.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Wong HL, Rauth AM, Bendayan R and Wu XY:
In vivo evaluation of a new polymer-lipid hybrid nanoparticle (PLN)
formulation of doxorubicin in a murine solid tumor model. Eur J
Pharm Biopharm. 65:300–308. 2007.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Hans ML and Lowman AM: Biodegradable
nanoparticles for drug delivery and targeting. Curr Opin Solid
State Mater Sci. 6:319–327. 2002.
|
|
40
|
Panyam J and Labhasetwar V: Biodegradable
nanoparticles for drug and gene delivery to cells and tissue. Adv
Drug Deliv Rev. 55:329–347. 2003.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Dash TK and Konkimalla VB:
Poly-є-caprolactone based formulations for drug delivery and tissue
engineering: A review. J Control Release. 158:15–33.
2012.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Kah M, Beulke S, Tiede K and Hofmann T:
Nanopesticides: State of knowledge, environmental fate, and
exposure modeling. Crit Rev Environ Sci Technol. 43:1823–1867.
2013.
|
|
43
|
Allen TM and Cullis PR: Liposomal drug
delivery systems: From concept to clinical applications. Adv Drug
Deliv Rev. 65:36–48. 2013.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Mehnert W and Mäder K: Solid lipid
nanoparticles: Production, characterization and applications. Adv
Drug Deliv Rev. 64 (Suppl):S83–S101. 2012.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Müller RH, Radtke M and Wissing SA: Solid
lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC)
in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 54
(Suppl 1):S131–S155. 2002.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Hou X, Zaks T, Langer R and Dong Y: Lipid
nanoparticles for mRNA delivery. Nat Rev Mater. 6:1078–1094.
2021.
|
|
47
|
Pardi N, Hogan MJ, Porter FW and Weissman
D: mRNA vaccines-a new era in vaccinology. Nat Rev Drug Discov.
17:261–279. 2018.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Polack FP, Thomas SJ, Kitchin N, Absalon
J, Gurtman A, Lockhart S, Perez JL, Pérez Marc G, Moreira ED,
Zerbini C, et al: Safety and efficacy of the BNT162b2 mRNA Covid-19
vaccine. N Engl J Med. 383:2603–2615. 2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Cullis PR and Hope MJ: Lipid nanoparticle
systems for enabling gene therapies. Mol Ther. 25:1467–1475.
2017.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Buschmann MD, Carrasco MJ, Alishetty S,
Paige M, Alameh MG and Weissman D: Nanomaterial delivery systems
for mRNA vaccines. Vaccines (Basel). 9(65)2021.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Resch-Genger U, Grabolle M,
Cavaliere-Jaricot S, Nitschke R and Nann T: Quantum dots versus
organic dyes as fluorescent labels. Nat Methods. 5:763–775.
2008.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Dhariwal J, Rao GK and Vaya D: Recent
advancements towards the green synthesis of carbon quantum dots as
an innovative and eco-friendly solution for metal ion sensing and
monitoring. RSC Sustainability. 2:11–36. 2024.
|
|
53
|
Carter CB and Norton MG: Ceramic
materials: Science and engineering. 2nd edition. Springer, New
York, 2013.
|
|
54
|
Diebold U: The surface science of titanium
dioxide. Surf Sci Rep. 48:53–229. 2003.
|
|
55
|
Bergna HE and Roberts WO: Colloidal
silica: Fundamentals and applications. 1st edition. CRC Press, Boca
Raton, 2005.
|
|
56
|
Vallet-Regí M, Balas F and Arcos D:
Mesoporous materials for drug delivery. Angew Chem Int Ed Engl.
46:7548–7558. 2007.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Dorozhkin SV: Calcium orthophosphate-based
bioceramics. Materials. 2:399–498. 2009.
|
|
58
|
Roco MC, Mirkin CA and Hersam MC:
Nanotechnology research directions for societal needs in 2020.
Dordrecht, Heidelberg, London, New York: Springer, pp690, 2011.
|
|
59
|
Barsoum MW: Fundamentals of ceramics. 2nd
Edition. CRC press, 2019. https://doi.org/10.1201/9781498708166.
|
|
60
|
Pankhurst QA, Connolly J, Jones SK and
Dobson JJ: Applications of magnetic nanoparticles in biomedicine. J
Phys D Appl Phys. 36(R167)2003.
|
|
61
|
Gao J, Gu H and Xu B: Multifunctional
magnetic nanoparticles: Design, synthesis, and biomedical
applications. Acc Chem Res. 42:1097–1107. 2009.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Fert A: Nobel lecture: Origin,
development, and future of spintronics. Rev Mod Phys. 80:1517–1530.
2008.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Huang X, Wu H, Pu S, Zhang W, Liao X and
Shi B: One-step room-temperature synthesis of Au@Pd core-shell
nanoparticles with tunable structure using plant tannin as
reductant and stabilizer. Green Chem. 13:950–957. 2011.
|
|
64
|
Caruso F: Nanoengineering of particle
surfaces. Adv Mater. 13:11–22. 2001.
|
|
65
|
Astruc D, Lu F and Aranzaes JR:
Nanoparticles as recyclable catalysts: The frontier between
homogeneous and heterogeneous catalysis. Angew Chem Int Ed Engl.
44:7852–7872. 2005.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Mondal K and Sharma A: Recent advances in
the synthesis and application of photocatalytic metal-metal oxide
core-shell nanoparticles for environmental remediation and their
recycling process. RSC Advances. 6:83589–83612. 2016.
|
|
67
|
Zhang L, Gu FX, Chan JM, Wang AZ, Langer
RS and Farokhzad OC: Nanoparticles in medicine: Therapeutic
applications and developments. Clin Pharmacol Ther. 83:761–769.
2008.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Shaker K, Jabbar M, Awais H, Abbas A,
Nordin AH, Ilyas RA, San Khoo P and Suryanegara L: Synthesis,
properties, and environmental applications of magnetic
nanocellulose composites. J Environ Chem Eng. 13(119494)2025.
|
|
69
|
Astruc D, Boisselier E and Ornelas C:
Dendrimers designed for functions: From physical, photophysical,
and supramolecular properties to applications in sensing,
catalysis, molecular electronics, photonics, and nanomedicine. Chem
Rev. 110:1857–1959. 2010.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Cheng Y, Xu Z, Ma M and Xu T: Dendrimers
as drug carriers: Applications in different routes of drug
administration. J Pharm Sci. 97:123–143. 2008.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Tomalia DA, Naylor AM and Goddard WA:
Starburst dendrimers: Molecular-level control of size, shape,
surface chemistry, topology, and flexibility from atoms to
macroscopic matter. Angew Chem Int Ed. 29:138–175. 1990.
|
|
72
|
Devatha CP and Thalla AK: Green synthesis
of nanomaterials. In: Synthesis of Inorganic Nanomaterials.
Elsevier, pp169-184, 2018.
|
|
73
|
Azzazy HME, Mansour MMH, Samir TM and
Franco R: Gold nanoparticles in the clinical laboratory: Principles
of preparation and applications. Clin Chem Lab Med. 50:193–209.
2011.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Alanazi FK, Radwan AA and Alsarra IA:
Biopharmaceutical applications of nanogold. Saudi Pharm J.
18:179–193. 2010.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Shi J, Kantoff PW, Wooster R and Farokhzad
OC: Cancer nanomedicine: Progress, challenges and opportunities.
Nat Rev Cancer. 17:20–37. 2017.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Li G, Li J and Yang Z: An electrochemical
sensor based on graphene-chitosan-cyclodextrin modification for the
detection of Staphylococcus aureus. Carbon Lett. 34:495–504.
2024.
|
|
77
|
Chowdhury MA, Iqbal MZ, Rana MM, Hossain
N, Shahin M, Islam MA and Rahman MM: Green synthesis of novel green
ceramic-based nanoparticles prepared by sol-gel technique for
diverse industrial application. Results Surf Interf.
14(100178)2024.
|
|
78
|
Fardsadegh B and Jafarizadeh-Malmiri H:
Aloe vera leaf extract mediated green synthesis of selenium
nanoparticles and assessment of their In vitro antimicrobial
activity against spoilage fungi and pathogenic bacteria strains.
Green Process Synth. 8:399–407. 2019.
|
|
79
|
Rana A, Yadav K and Jagadevan S: A
comprehensive review on green synthesis of nature-inspired metal
nanoparticles: Mechanism, application and toxicity. J Clean Prod.
272(122880)2020.
|
|
80
|
Rahuman HBF, Dhandapani R, Narayanan S,
Palanivel V, Paramasivam R, Subbarayalu R, Thangavelu S and
Muthupandian S: Medicinal plants mediated the green synthesis of
silver nanoparticles and their biomedical applications. IET
Nanobiotechnol. 16:115–144. 2022.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Syed A and Ahmad A: Extracellular
biosynthesis of platinum nanoparticles using the fungus Fusarium
oxysporum. Colloids Surf B Biointerfaces. 97:27–31.
2012.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Mukherjee P, Ahmad A, Mandal D, Senapati
S, Sainkar SR, Khan MI, Parishcha R, Ajaykumar PV, Alam M, Kumar R
and Sastry M: Fungus-mediated synthesis of silver nanoparticles and
their immobilization in the mycelial matrix: A novel biological
approach to nanoparticle synthesis. Nano Lett. 1:515–519. 2001.
|
|
83
|
Jones N, Ray B, Ranjit KT and Manna AC:
Antibacterial activity of ZnO nanoparticle suspensions on a broad
spectrum of microorganisms. FEMS Microbiol Lett. 279:71–76.
2008.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Gericke M and Pinches A: Microbial
production of gold nanoparticles. Gold Bull. 39:22–28. 2006.
|
|
85
|
Durán N, Marcato PD, Alves OL, De Souza
GIH and Esposito E: Mechanistic aspects of biosynthesis of silver
nanoparticles by several Fusarium oxysporum strains. J
Nanobiotechnology. 3(8)2005.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Kalimuthu K, Babu RS, Venkataraman D,
Bilal M and Gurunathan S: Biosynthesis of silver nanocrystals by
Bacillus licheniformis. Colloids and surfaces B:
Biointerfaces. 65:150–153. 2008.
|
|
87
|
Seth R and Meena A: Enzymes-based
nanomaterial synthesis: An eco-friendly and green synthesis
approach. Clean Technol Environ Policy. 27:5775–5798. 2024.
|
|
88
|
Sallam SA, El-Subruiti GM and Eltaweil AS:
Facile synthesis of Ag-γ-Fe2O3 superior nanocomposite for catalytic
reduction of nitroaromatic compounds and catalytic degradation of
methyl orange. Catal Lett. 148:3701–3714. 2018.
|
|
89
|
Ningthoujam R, Sahoo B, Ghosh P, Shivani
A, Ganguli P and Chaudhuri S: Green production of zero-valent iron
nanoparticles using pomegranate peel extracts and its use in
lindane degradation. Nanotechnol Environ Eng. 8:581–589. 2023.
|
|
90
|
Rocchetti G, Lucini L, Ahmed SR and Saber
FR: In vitro cytotoxic activity of six Syzygium leaf extracts as
related to their phenolic profiles: An untargeted UHPLC-QTOF-MS
approach. Food Res Int. 126(108715)2019.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Siddiqi KS and Husen A: Green synthesis,
characterization and uses of palladium/platinum nanoparticles.
Nanoscale Res Lett. 11(482)2016.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Khan MQ, Kumar P, Khan RA, Ahmad K and Kim
H: Fabrication of sulfur-doped reduced graphene oxide modified
glassy carbon electrode (S@rGO/GCE) based acetaminophen sensor.
Inorganics. 10(218)2022.
|
|
93
|
Iravani S: Green synthesis of metal
nanoparticles using plants. Green Chem. 13:2638–2650. 2011.
|
|
94
|
Rozali NL, Azizan KA, Singh R, Syed Jaafar
SN, Othman A, Weckwerth W and Ramli U: Fourier transform infrared
(FTIR) spectroscopy approach combined with discriminant analysis
and prediction model for crude palm oil authentication of different
geographical and temporal origins. Food Control.
146(109509)2023.
|
|
95
|
Naganthran A, Verasoundarapandian G,
Khalid FE, Masarudin MJ, Zulkharnain A, Nawawi NM, Karim M,
Abdullah CAC and Ahmad AS: Synthesis, characterization and
biomedical application of silver nanoparticles. Materials.
15(427)2022.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Huang J, Lin L, Li Q, Sun D, Wang Y, Lu Y,
He N, Yang K, Yang X, Wang H, et al: Continuous-flow biosynthesis
of silver nanoparticles by lixivium of sundried Cinnamomum
camphora leaf in tubular microreactors. Ind Eng Chem Res.
47:6081–6090. 2008.
|
|
97
|
Kumar S, Singh M, Halder D and Mitra A:
Mechanistic study of antibacterial activity of biologically
synthesized silver nanocolloids. Colloids Surf A Physicochem Eng
Asp. 449:82–86. 2014.
|
|
98
|
Philip D: Green synthesis of gold and
silver nanoparticles using Hibiscus rosa sinensis. Phys E:
Low-Dimens Syst Nanostruct. 42:1417–1424. 2010.
|
|
99
|
Bar H, Bhui DK, Sahoo GP, Sarkar P, Pyne S
and Misra A: Green synthesis of silver nanoparticles using seed
extract of Jatropha curcas. Colloids Surf A: Physicochem Eng
Asp. 348 (20 Suppl):S212–S216. 2009.
|
|
100
|
Aminabad NS, Farshbaf M and Akbarzadeh A:
Recent advances of gold nanoparticles in biomedical applications:
State of the Art. Cell Biochem Biophys. 77:123–137. 2019.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Shankar SS, Rai A, Ankamwar B, Singh A,
Ahmad A and Sastry M: Biological synthesis of triangular gold
nanoprisms. Nat Mater. 3:482–488. 2004.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Shiv Shankar S, Rai A, Ahmad A and Sastry
M: Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell
nanoparticles using Neem (Azadirachta indica) leaf broth. J
Colloid Interface Sci. 275:496–502. 2004.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Song JY, Jang HK and Kim BS: Biological
synthesis of gold nanoparticles using Magnolia kobus and
Diopyros kaki leaf extracts. Process Biochem. 44:1133–1138.
2009.
|
|
104
|
Keijok WJ, Pereira RHA, Alvarez LAC, Prado
AR, da Silva AR, Ribeiro J, Oliveira JP and Guimaraes M: Controlled
biosynthesis of gold nanoparticles with Coffea arabica using
factorial design. Sci Rep. 9(16019)2019.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Blanco E, Shen H and Ferrari M: Principles
of nanoparticle design for overcoming biological barriers to drug
delivery. Nat Biotechnol. 33:941–951. 2015.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Fernando S, Gunasekara T and Holton J:
Antimicrobial nanoparticles: Applications and mechanisms of action.
Sri Lankan J Infect Dis. 8(2)2018.
|
|
107
|
Yetisgin AA, Cetinel S, Zuvin M, Kosar A
and Kutlu O: Therapeutic nanoparticles and their targeted delivery
applications. Molecules. 25(2193)2020.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Du C, Xu N, Yao Z, Bai X, Gao Y, Peng L,
Gu B and Zhao J: Mechanistic insights into sulfate and
phosphate-mediated hexavalent chromium removal by tea polyphenols
wrapped nano-zero-valent iron. Sci Total Environ.
850(157996)2022.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Modwi A, Idriss H, Khezami L, Albadri A,
Ismail M, Assadi AA and Nguyen-Tri P: Ba2+ removal from aquatic
medium via TiY2O5@g-C3N4 nanocomposites. Diam Relat Mater.
135(109830)2023.
|
|
110
|
Kharissova OV, Dias R, Kharisov BI, Pérez
BO and Pérez VMJ: The greener synthesis of nanoparticles. Trends
Biotechnol. 31:P240–P248. 2013.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Vyas JV and Rana S: Antioxidant activity
and green synthesis of selenium nanoparticles using Allium
sativum extract. Int J Phytomed. 9(634)2017.
|
|
112
|
Hariharan S, Chauhan S, Velu K, Dharmaraj
S, CM VK and Ganesan S: Biological activities of selenium
nanoparticles synthesized from Camellia sinensis (L) Kuntze
leaves. Appl Biochem Biotechnol. 195:5823–5837. 2023.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Din MI, Arshad F, Hussain Z and Mukhtar M:
Green adeptness in the synthesis and stabilization of copper
nanoparticles: Catalytic, antibacterial, cytotoxicity, and
antioxidant activities. Nanoscale Res Lett. 12(638)2017.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Naika HR, Lingaraju K, Manjunath K, Kumar
D, Nagaraju G, Suresh D and Nagabhushana H: Green synthesis of CuO
nanoparticles using Gloriosa superba L. extract and their
antibacterial activity. J Taibah Univ Sci. 9:7–12. 2015.
|
|
115
|
Padma PN, Banu ST and Kumari SC: Studies
on green synthesis of copper nanoparticles using Punica
granatum. Ann Res Rev Biol. 23:1–0. 2018.
|
|
116
|
Alahdal FAM, Qashqoosh MTA, Manea YK,
Mohammed RKA and Naqvi S: Green synthesis and characterization of
copper nanoparticles using Phragmanthera austroarabica extract and
their biological/environmental applications. Sustain Mater Technol.
35(e00540)2023.
|
|
117
|
Laurent S, Forge D, Port M, Roch A, Robic
C, Vander Elst L and Muller RN: Magnetic Iron Oxide Nanoparticles:
Synthesis, Stabilization, Vectorization, Physicochemical
Characterizations, and Biological Applications. Chem Rev.
108:2064–2110. 2008.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Narnoliya LK, Jadaun JS and Singh SP: The
phytochemical composition, biological effects and biotechnological
approaches to the production of high-value essential oil from
geranium. In: Essential Oil Research. Cham: Springer International
Publishing, pp327-352, 2019.
|
|
119
|
Chen X, Shen S, Guo L and Mao SS:
Semiconductor-based photocatalytic hydrogen generation. Chem Rev.
110:6503–6570. 2010.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Gupta VK and Nayak A: Cadmium removal and
recovery from aqueous solutions by novel adsorbents prepared from
orange peel and Fe2O3 nanoparticles. Chem Eng
J. 180:81–90. 2012.
|
|
121
|
Yu MF, Files BS, Arepalli S and Ruoff RS:
Tensile loading of ropes of single wall carbon nanotubes and their
mechanical properties. Phys Rev Lett. 84:5552–5555. 2000.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Reif J, Rafiee J, Wang Z, Song H, Yu ZZ
and Koratkar N: Enhanced mechanical properties of nanocomposites at
low graphene content. ACS Nano. 3:3884–3890. 2009.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Zhang Y, Zhou Q, Zhu J, Yan Q, Dou SX and
Sun W: Nanostructured metal chalcogenides for energy storage and
electrocatalysis. Adv Funct Mater. 27(1702317)2017.
|
|
124
|
Rai M, Yadav A and Gade A: Silver
nanoparticles as a new generation of antimicrobials. Biotechnol
Adv. 27:76–83. 2009.PubMed/NCBI View Article : Google Scholar
|
|
125
|
Peer D, Karp JM, Hong S, Farokhzad OC,
Margalit R and Langer R: Nanocarriers as an emerging platform for
cancer therapy. Nat Nanotechnol. 2:751–760. 2007.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Riehemann K, Schneider SW, Luger TA, Godin
B, Ferrari M and Fuchs H: Nanomedicine-challenge and perspectives.
Angew Chem Int Ed Engl. 48:872–897. 2009.
|
|
127
|
Chaudhary M, Kumar A, Devi A, Singh BP,
Malhotra BD, Singhal K, Shukla S, Ponnada S, Sharma RK,
Vega-Olivencia CA, et al: Prospects of nanostructure-based
electrochemical sensors for drug detection: A review. Mater Adv.
4:432–457. 2023.
|
|
128
|
Turkevich J, Stevenson PC and Hillier J: A
study of the nucleation and growth processes in the synthesis of
colloidal gold. Discuss Faraday Soc. 11:55–75. 1951.
|
|
129
|
Brust M, Walker M, Bethell D, Schiffrin DJ
and Whyman R: Synthesis of thiol-derivatised gold nanoparticles in
a two-phase liquid-liquid system. J Chem Soc Chem Commun.
7:801–802. 1994.
|
|
130
|
Cullity BD and Stock SR: Elements of X-ray
diffraction. 3rd edition. Prentice Hall, Upper Saddle River,
2001.
|
|
131
|
Ahmad A, Mukherjee P, Senapati S, Mandal
D, Khan MI, Kumar R and Sastry M: Extracellular biosynthesis of
silver nanoparticles using the fungus Fusarium oxysporum.
Colloids Surf B Biointerfaces. 28:313–318. 2003.
|
|
132
|
Raveendran P, Fu J and Wallen SL:
Completely ‘green’ synthesis and stabilization of metal
nanoparticles. J Am Chem Soc. 125:13940–13941. 2003.PubMed/NCBI View Article : Google Scholar
|
|
133
|
Narayanan KB and Sakthivel N: Biological
synthesis of metal nanoparticles by microbes. Adv Colloid Interface
Sci. 156:1–3. 2010.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Xia Y, Xiong Y, Lim B and Skrabalak SE:
Shape-controlled synthesis of metal nanocrystals: Simple chemistry
meets complex physics? Angew Chem Int Ed Engl. 48:60–103.
2009.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Dreaden EC, Alkilany AM, Huang X, Murphy
CJ and El-Sayed MA: The golden age: Gold nanoparticles for
biomedicine. Chem Soc Rev. 41:2740–2779. 2012.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Haruta M: Gold as a novel catalyst in the
21st century: Preparation, working mechanism and applications. Gold
Bull. 37:27–36. 2004.
|
|
137
|
Khan I, Saeed K and Khan I: Nanoparticles:
Properties, applications and toxicities. Arab J Chem. 12:908–931.
2019.
|
|
138
|
Wei H and Wang E: Nanomaterials with
enzyme-like characteristics (nanozymes): Next-generation artificial
enzymes†. Chem Soc Rev. 42:6060–6093. 2013.PubMed/NCBI View Article : Google Scholar
|
|
139
|
Bobo D, Robinson K, Islam J, Thurecht K
and Corrie S: Nanoparticle-based medicines: A review of
FDA-approved materials and clinical trials to date. Pharm Res.
33:2373–2387. 2016.PubMed/NCBI View Article : Google Scholar
|
|
140
|
Butler KT, Davies DW, Cartwright H, Isayev
O and Walsh A: Machine learning for molecular and materials
science. Nature. 559:547–555. 2018.PubMed/NCBI View Article : Google Scholar
|
|
141
|
Iravani S and Varma RS: Greener synthesis
of lignin nanoparticles and their applications. Green Chem.
22:612–636. 2020.
|
|
142
|
Cheng L, Wang C, Feng L, Yang K and Liu Z:
Functional nanomaterials for phototherapies of cancer. Chem Rev.
114:10869–10939. 2014.PubMed/NCBI View Article : Google Scholar
|
|
143
|
Ilhan S and Çamli Pulat Ç: Biogenic silver
nanoparticles synthesized from Piper longum fruit extract inhibit
HIF-1α/VEGF mediated angiogenesis in prostate cancer cells.
Cumhuriyet Sci J. 42:236–244. 2021.
|
|
144
|
Soltani L and Darbemamieh M: Biosynthesis
of silver nanoparticles using hydroethanolic extract of Cucurbita
pepo L. Fruit and their anti-proliferative and apoptotic activity
against breast cancer cell line (MCF-7). Multidiscip Cancer
Investig. 5:1–10. 2021.
|
|
145
|
Goel M, Sharma A and Sharma B: Recent
advances in biogenic silver nanoparticles for their biomedical
applications. Sustain Chem. 4:61–94. 2023.
|
|
146
|
Vijayaram S, Razafindralambo H, Sun YZ,
Vasantharaj S, Ghafarifarsani H, Hoseinifar SH and Raeeszadeh M:
Applications of green syntheized metal nanoparticles-a review. Biol
Trace Elem Res. 202:360–386. 2024.PubMed/NCBI View Article : Google Scholar
|
|
147
|
Magangana TP, Makunga NP, Fawole OA and
Opara UL: Processing factors affecting the phytochemical and
nutritional properties of pomegranate (Punica granatum L.)
peel waste: A review. Molecules. 25(4690)2020.PubMed/NCBI View Article : Google Scholar
|
|
148
|
Jia L, Zhang Q, Li Q and Song H: The
biosynthesis of palladium nanoparticles by antioxidants in
Gardenia jasminoides Ellis: Long lifetime nanocatalysts for
p-nitrotoluene hydrogenation. Nanotechnology.
20(385601)2009.PubMed/NCBI View Article : Google Scholar
|
|
149
|
Puri A and Patil S: Biogenic synthesis of
selenium nanoparticles using diospyros montana bark extract:
Characterization, antioxidant, antibacterial, and antiproliferative
activity. Biosci Biotechnol Res Asia. 19:423–441. 2022.
|
|
150
|
Younus M, Hasan MM, Ali S, Saddq B, Sarwar
G, Ullah MI, Maqsood A, Ahmar S, Mora-Poblete F, Hassan F, et al:
Extracts of Euphorbia nivulia Buch.-Ham. showed both phytotoxic and
insecticidal capacities against Lemna minor L. and Oxycarenus
hyalinipennis Costa. PLoS One. 16(e0250118)2021.PubMed/NCBI View Article : Google Scholar
|
|
151
|
Williams DB and Carter CB: Transmission
electron microscopy: A textbook for materials science. Springer,
2009.
|
|
152
|
Goldstein JI, Newbury DE, Michael JR,
Ritchie NWM, Scott JHJ and Joy DC: Scanning electron microscopy and
X-ray microanalysis. 4th edition. Springer, 2018.
|
|
153
|
Malvern Instruments: Dynamic light
scattering: An introduction in 30 minutes. Malvern Instruments
Limited, Worcestershire, 2017.
|
|
154
|
Coates J: Interpretation of infrared
spectra, a practical approach. In: Meyers RA, McKelvy ML (eds),
Encyclopedia of Analytical Chemistry. John Wiley & Sons Ltd.
Chichester, pp10815-10837, 2000.
|
|
155
|
Meyer E, Hug HJ and Bennewitz R: Scanning
probe microscopy. Springer, 2004.
|
|
156
|
Brown ME: Introduction to thermal
analysis: Techniques and applications. Springer, 2001.
|
|
157
|
Hunter RJ: Zeta potential in colloid
science. Academic Press, San Diego, 1981.
|