|
1
|
Siegel RL, Kratzer TB, Giaquinto AN, Sung
H and Jemal A: Cancer statistics, 2025. CA Cancer J Clin. 75:10–45.
2025.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Nguyen-Nielsen M and Borre M: Diagnostic
and therapeutic strategies for prostate cancer. Semin Nucl Med.
46:484–490. 2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Padhani AR, Godtman RA and Schoots IG: Key
learning on the promise and limitations of MRI in prostate cancer
screening. Eur Radiol. 34:6168–6174. 2024.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Rapisarda S, Bada M, Crocetto F, Barone B,
Arcaniolo D, Polara A, Imbimbo C and Grosso G: The role of
multiparametric resonance and biopsy in prostate cancer detection:
Comparison with definitive histological report after
laparoscopic/robotic radical prostatectomy. Abdom Radiol (NY).
45:4178–4184. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Serrano MJ, Garrido-Navas MC, Diaz Mochon
JJ, Cristofanilli M, Gil-Bazo I, Pauwels P, Malapelle U, Russo A,
Lorente JA, Ruiz-Rodriguez AJ, et al: Precision prevention and
cancer interception: The new challenges of liquid biopsy. Cancer
Discov. 10:1635–1644. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ozturk EA and Caner A: Liquid biopsy for
promising non-invasive diagnostic biomarkers in parasitic
infections. Acta Parasitol. 67:1–17. 2022.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zainfeld D and Goldkorn A: Liquid biopsy
in prostate cancer: Circulating tumor cells and beyond. Cancer
Treat Res. 175:87–102. 2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zhou Y and Dendukuri N: Statistics for
quantifying heterogeneity in univariate and bivariate meta-analyses
of binary data: The case of meta-analyses of diagnostic accuracy.
Stat Med. 33:2701–2717. 2014.PubMed/NCBI View
Article : Google Scholar
|
|
9
|
Holling H, Böhning W, Masoudi E, Böhning D
and Sangnawakij P: Evaluation of a new version of
I2 with emphasis on diagnostic problems. Commun
Stat Simul Comput. 9:942–972. 2020.
|
|
10
|
Reitsma JB, Glas AS, Rutjes AW, Scholten
RJ, Bossuyt PM and Zwinderman AH: Bivariate analysis of sensitivity
and specificity produces informative summary measures in diagnostic
reviews. J Clin Epidemiol. 58:982–990. 2005.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Takwoingi Y, Guo B, Riley RD and Deeks JJ:
Performance of methods for meta-analysis of diagnostic test
accuracy with few studies or sparse data. Stat Methods Med Res.
26:1896–1911. 2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Rosenberger KJ, Chu H and Lin L: Empirical
comparisons of meta-analysis methods for diagnostic studies: A
meta-epidemiological study. BMJ Open. 12(e055336)2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
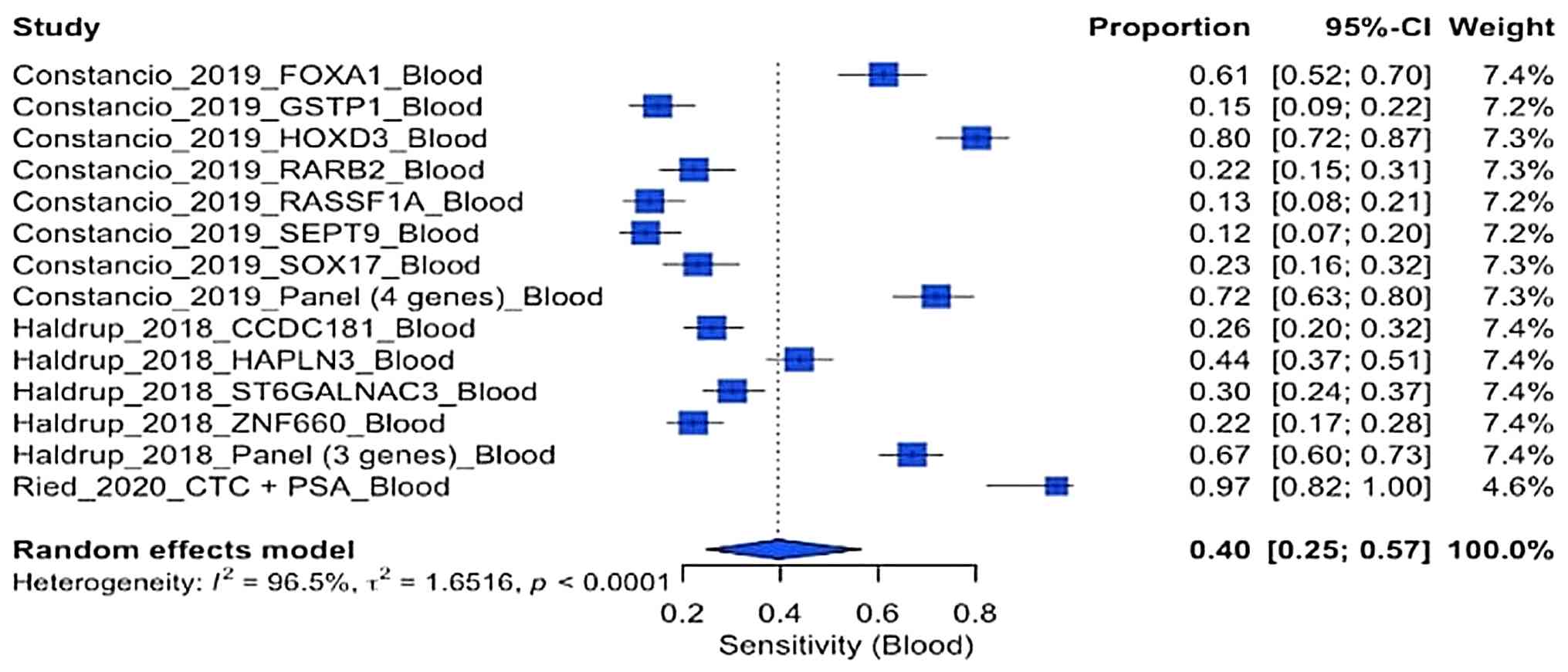
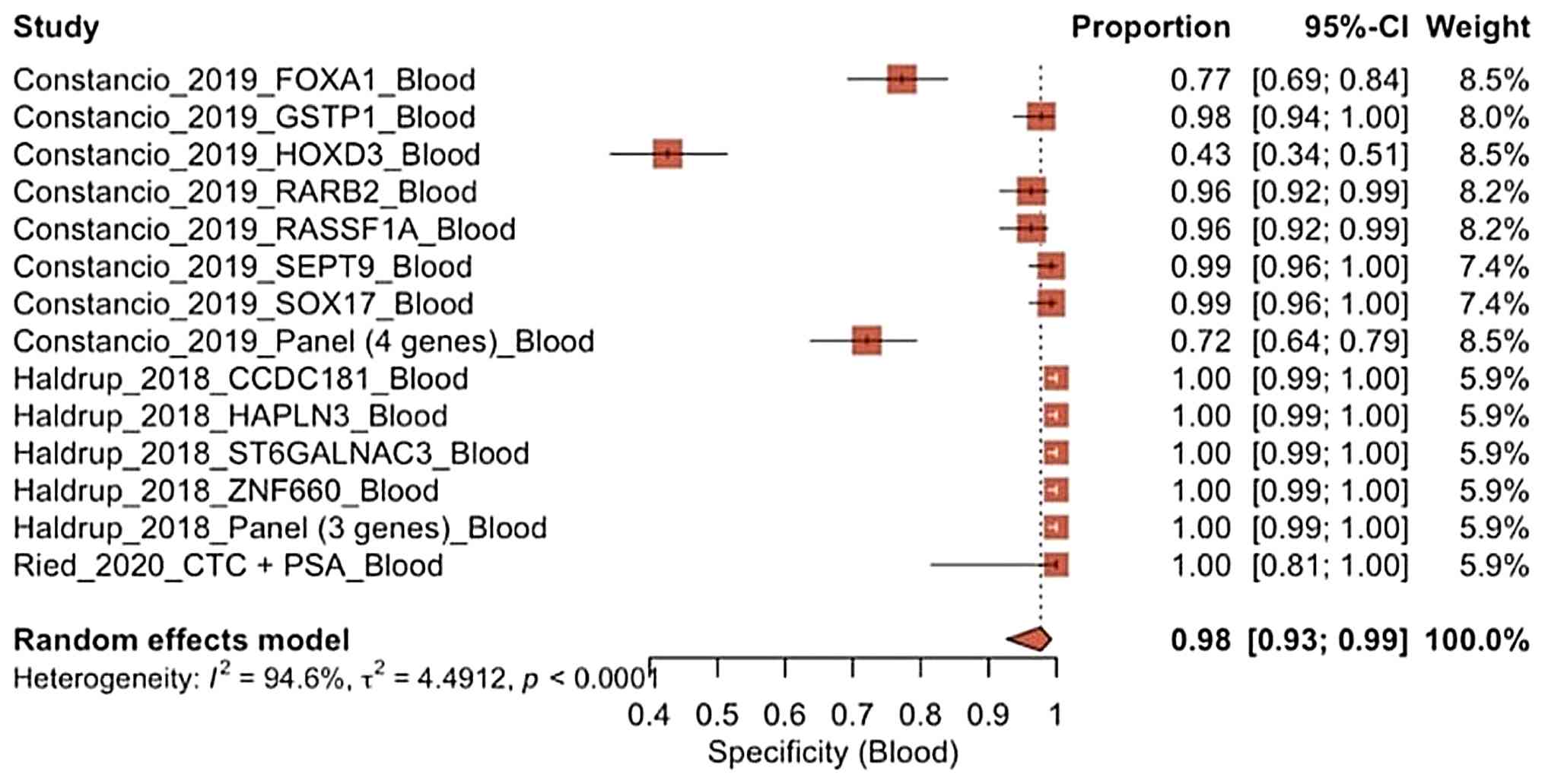
Constâncio V, Nunes SP, Moreira-Barbosa C,
Freitas R, Oliveira J, Pousa I, Oliveira J, Soares M, Dias CG, Dias
T, et al: Early detection of the major male cancer types in
blood-based liquid biopsies using a DNA methylation panel. Clin
Epigenetics. 11(175)2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Haldrup C, Pedersen AL, Øgaard N, Strand
SH, Høyer S, Borre M, Ørntoft TF and Sørensen KD: Biomarker
potential of ST6GALNAC3 and ZNF660 promoter hypermethylation in
prostate cancer tissue and liquid biopsies. Mol Oncol. 12:545–560.
2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ried K, Tamanna T, Matthews S, Eng P and
Sali A: New screening test improves detection of prostate cancer
using circulating tumor cells and prostate-specific markers. Front
Oncol. 10(582)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kim J, Shim JS, Han BH, Kim HJ, Park J,
Cho IJ, Kang SG, Kang JY, Bong KW and Choi N: Hydrogel-based
hybridization chain reaction (HCR) for detection of urinary
exosomal miRNAs as a diagnostic tool of prostate cancer. Biosens
Bioelectron. 192(113504)2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
McKiernan J, Donovan MJ, Margolis E,
Partin A, Carter B, Brown G, Torkler P, Noerholm M, Skog J, Shore
N, et al: A prospective adaptive utility trial to validate
performance of a novel urine exosome gene expression assay to
predict high-grade prostate cancer in patients with
prostate-specific antigen 2-10 ng/ml at initial biopsy. Eur Urol.
74:731–738. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Van Neste L, Hendriks RJ, Dijkstra S,
Trooskens G, Cornel EB, Jannink SA, de Jong H, Hessels D, Smit FP,
Melchers WJ, et al: Detection of high-grade prostate cancer using a
urinary molecular biomarker-based risk score. Eur Urol. 70:740–748.
2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ochiai A, Okihara K, Kamoi K, Oikawa T,
Shimazui T, Murayama S, Tomita K, Umekawa T, Uemura H and Miki T:
Clinical utility of the prostate cancer gene 3 (PCA3) urine assay
in Japanese men undergoing prostate biopsy. BJU Int. 111:928–933.
2013.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wang L, Skotland T, Berge V, Sandvig K and
Llorente A: Exosomal proteins as prostate cancer biomarkers in
urine: From mass spectrometry discovery to immunoassay-based
validation. Eur J Pharm Sci. 98:80–85. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Yu J, Yu C, Jiang K, Yang G, Yang S, Tan
S, Li T, Liang H, He Q, Wei F, et al: Unveiling potential: Urinary
exosomal mRNAs as non-invasive biomarkers for early prostate cancer
diagnosis. BMC Urol. 24(163)2024.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Opoku Mensah B, Fondjo LA, Owiredu WKBA
and Adusei B: Urinary PCA3 a superior diagnostic biomarker for
prostate cancer among Ghanaian men. Dis Markers.
2022(1686991)2022.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Cheng B, Luo T, Wu Y, Hu J, Yang C, Wu J,
Luo Y, Shangguan W, Li W, Yang L, et al: Urinary exosomal
FAM153C-RPL19 chimeric RNA as a diagnostic and prognostic biomarker
for prostate cancer in Chinese patients. Cancer Lett.
631(217938)2025.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Davey M, Benzina S, Savoie M, Breault G,
Ghosh A and Ouellette RJ: Affinity captured urinary extracellular
vesicles provide mRNA and miRNA biomarkers for improved accuracy of
prostate cancer detection: A pilot study. Int J Mol Sci.
21(8330)2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Trujillo B, Wu A, Wetterskog D and Attard
G: Blood-based liquid biopsies for prostate cancer: Clinical
opportunities and challenges. Br J Cancer. 127:1394–1402.
2022.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Prensner JR, Rubin MA, Wei JT and
Chinnaiyan AM: Beyond PSA: The next generation of prostate cancer
biomarkers. Sci Transl Med. 4(127rv3)2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Chi KR: The dark side of the human genome.
Nature. 538:275–277. 2016.PubMed/NCBI View
Article : Google Scholar
|
|
28
|
Minciacchi VR, Zijlstra A, Rubin MA and Di
Vizio D: Extracellular vesicles for liquid biopsy in prostate
cancer: Where are we and where are we headed? Prostate Cancer
Prostatic Dis. 20:251–258. 2017.PubMed/NCBI View Article : Google Scholar
|