1. Introduction
Natural bioactive compounds that originate from
plants, animals, fungi and their bioactive compounds have been
extensively examined and researched for their use as treatments for
multiple conditions, such as neurological disorders, cardiovascular
diabetes, hypertension, reproductive disorders and cancer (1). A number of studies have
comprehensively covered a variety of different natural bioactive
components utilized effectively for neurodegenerative disorders
(NDDs) such as memory loss, Huntington's disease, Parkinson's
disease and amyotrophic lateral sclerosis (1,2).
Natural substances are being studied as possible neuroprotective
agents for the treatment of NDDs (3). Since natural treatments are less
costly and more culturally appropriate, and may aid in reducing the
risk of developing neurodegenerative disorders at an early stage,
millions of individuals with age-related NDDs may benefit from
using natural bioactive compounds instead of traditional
pharmaceutical products. Several investigations using animals and
cells have demonstrated the neuroprotective benefits of a
polyphenol-rich diet (3-5).
One of the most natural methods used to raise dopamine levels and
increase dopamine release, is by the use of bioactive compounds
such as, naringenin, cyanidin, uridine; rutin, naringin, caffeine,
vitamin D and amino acids. There continues to be a shortage of
established procedures for examining the effects of bioactive
compounds on dopamine levels in the brain. Some of the current
studies systematically cover a number of different natural
bioactive compounds which enhance dopamine levels, such as
β-phenylethylamine (β-PEA), omega-3 fatty acids and others that
have been shown to enhance dopamine levels in the brain (6).
2. Dopamine
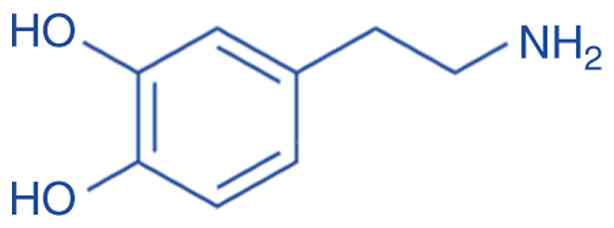
Dopamine (3,4-dihydroxyphenethylamine) is an amine
formed by removing a carboxyl group from the L-DOPA
(C8H11NO2) molecule (Fig. 1); it is an essential particle
produced in the brain, kidneys and in plants, and in the majority
of animals. Dopamine functions as a neurotransmitter, a substance
produced by neurons to transmit information to other neurons. A
recent study focused on how aromatherapy influences the treatment
of neuro pathologies, such as Alzheimer's disease (7). The brain chemical, dopamine, affects
behavior driven by rewards. The pituitary gland release of
prolactin is suppressed by hypothalamic dopamine. There are five
known subtypes of dopamine receptors in mammals, namely D1 D2, D3,
D4 and D5; there are two groups into which these receptors are
separated. In humans, the trace amine-associated receptor 1 has a
high affinity for dopamine. Abnormal dopamine synthesis or
metabolism has been related to addiction, bipolar disease and
attention deficit hyperactivity disorder (8). In a previous study, an in
vitro neuronal cell line system was employed to examine the
possible neuroprotective properties of Aloysia citrovorum-derived
essential oils against oxidative stress and amyloid-induced
neurotoxicity (9). The main enzyme
that converts DOPA into dopamine is known as DOPA decarboxylase.
Tyrosine hydroxylase, the rate-limiting enzyme in the synthesis of
dopamine, transforms tyrosine into DOPA (9). Considering the intimate association
between the hippocampus and memory/learning processes, essential
oils may change NDDs via altering the olfactory system. The process
of converting L-tyrosine to L-DOPA is facilitated by the enzyme
tyrosine hydroxylase (9).
Furthermore, essential oils may function directly as therapies by
modulating a variety of oxidative stress and inflammatory processes
linked to NDDs.
In certain cell types, such as neurons and cells
found in the adrenal medulla, dopamine synthesis is achieved
through a two-step process (10).
The synthesis of dopamine is under the control of homeostasis,
which can increase or decrease production depending on the levels
of extracellular dopamine (11).
The essential components for this reaction are iron
(Fe2+), diatomic oxygen (O2) and
tetrahydrobiopterin (BH4).
The conversion of L-DOPA to dopamine is catalyzed by
aromatic L-amino acid decarboxylase during neurotransmitter
synthesis (12). The rate at which
dopamine is synthesized is usually determined by the speed at which
TH converts tyrosine into L-DOPA. The function of TH is carefully
monitored by different methods, including phosphorylation at four
serine residues by several kinases (12):
Dephosphorylation by two phosphatases: The
regulation of enzyme activity through response inhibition by
catecholamine neurotransmitters, such as dopamine occurs through
protein-protein interactions with other enzymes and structural
proteins. To create dopamine effectively, the body needs tyrosine,
which can be obtained from dietary proteins or converted from
phenylalanine. Even though dopamine can be found in certain foods,
it is incapable of crossing the brain barrier, requiring the brain
to synthesize it for proper neuronal functioning (9).
Degradation: Dopamine undergoes a process of
breakdown by specific enzymes to form inactive metabolites. The
degradation of dopamine in the striatum is mostly carried out by
MAO-A, although both MAO-A and MAO-B are involved in its metabolism
(13).
Catechol-O-methyl transferase (COMT). In order to
degrade catecholamine neurotransmitters, such as dopamine,
norepinephrine and epinephrine, the enzyme COMT transfers a methyl
group from s-adenylyl methionine to catechol substrates. The
formation of O-methylated metabolites of these neurotransmitters
aids in stopping their signaling, particularly in areas of the
brain with relatively low dopamine transporter expression, such as
the prefrontal cortex (14).
Aldehyde dehydrogenase (ALDH): The primary result of
dopamine degradation is Homovanillic acid, which is discharged
through urine (9).
As regards dopamine receptors, these are classified
into two main families: D1-like receptors (D1 and D5) and D2-like
receptors (D2, D3 and D4). These receptors are distributed
throughout the brain and play crucial roles in various
physiological processes. As regards functions, the brain is filled
with receptors that are vital for many physiological functions
(11): Learning and motivation,
memory and focus, pleasure and reward, mood and attention, movement
and coordination, sleep regulation, pain processing and
cardiovascular function.
As regards diseases and disorders, there is a
correlation between dopamine dysfunction and multiple medical
conditions (10). i) Parkinson's
disease: Due to the degeneration of dopaminergic neurons in the
substantia nigra, motor symptoms and impulsivity control disorders
manifest (15). ii) Schizophrenia:
The dysregulation of dopamine signaling affects the pathways of
both the mesolimbic and mesocortical regions. iii) Attention
deficit hyperactivity disorder: Related to changes in dopamine
signaling that impact focus and self-control. iv) Addiction: The
presence of dopamine is essential for the brain to engage in
rewarding activities that can become addictive. v) Tourette
syndrome: Related to disruptions in dopamine signaling influencing
motor function. vi) Bipolar disorder: Results in an imbalance of
dopamine and other neurotransmitters impacting mood and behavior.
Exploring the complex functions of dopamine in the brain and body
is a vital research goal, potentially paving the way for
groundbreaking therapies for various neurological and psychiatric
conditions (15).
3. Bioactive compounds that enhance dopamine
levels in the brain
Naringenin
Naringenin (molecular formula:
C15H12O5) is the glycoside portion
of the molecule Naringin that protects the nigrostriatal
dopaminergic projection in neurotoxin models of Parkinson's
disease, and it has a typical chemical structure (16). These characteristics are a basic
flavonoid structure with 15 carbon atoms and three rings, two of
which are benzene rings that connect the three carbon chains
(17). Naringenin, chemically
known as 4,5,7-trihydroxyflavone, has a molar mass of 272.3 and a
melting point of 251°C (18). In nature, naringenin is solid and
nearly insoluble in water, although it is soluble in organic
solvents such as ethanol, ether, dimethyl form amide and dimethyl
sulfoxide. Naringenin occurs primarily in citrus fruits such
grapes, oranges, blood oranges, lemons and grapefruit, with some
research demonstrating a high concentration in citrus peel
(19) (Fig. 2A).
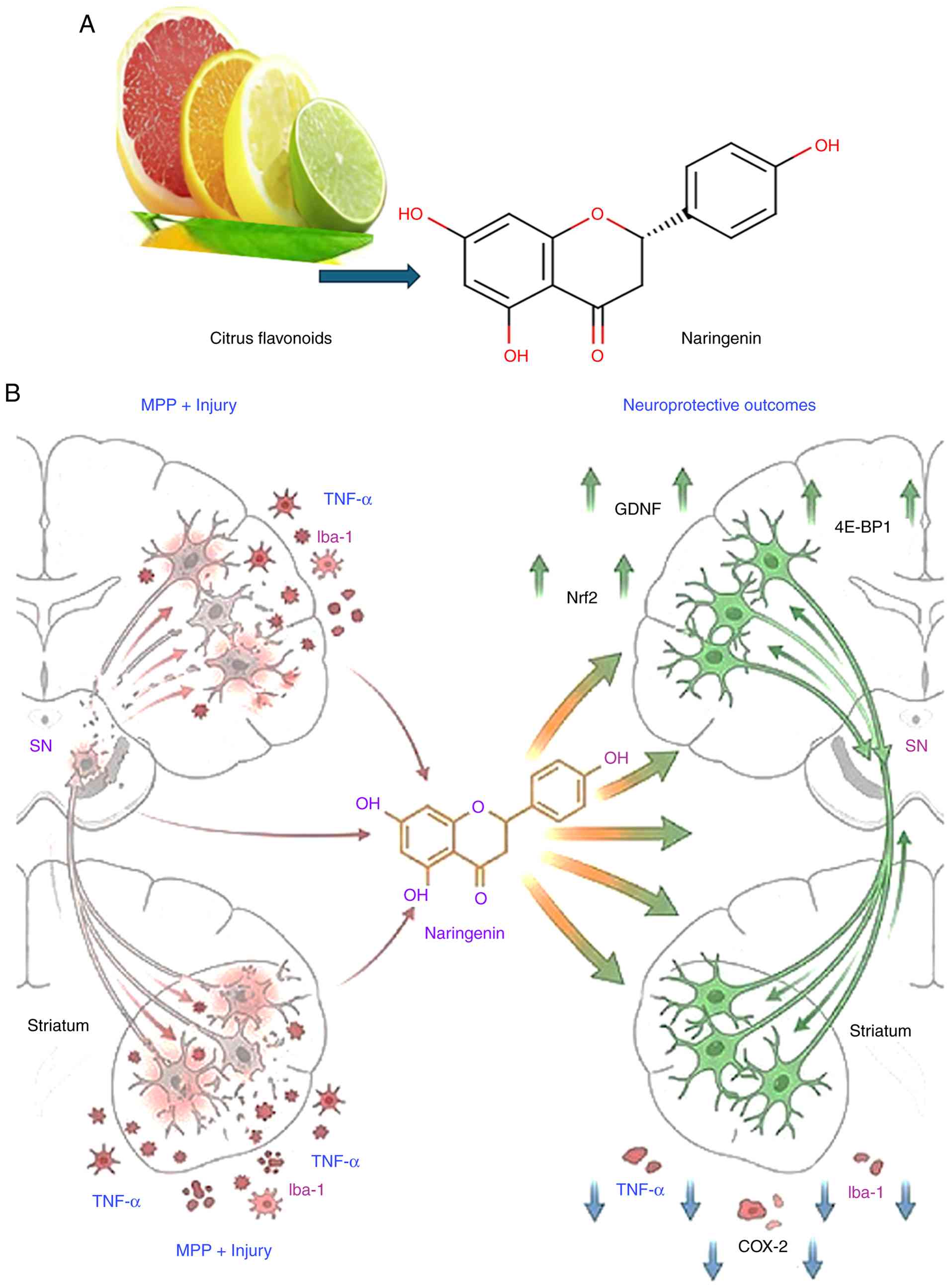 | Figure 2(A) Naringenin (molecular formula:
C15H12O5) in citrus fruits such
grapes, oranges, oranges, lemons and grapefruit. (B)
Neuroprotective mechanisms of naringenin
(C15H12O5) in an experimental
model of Parkinson's disease: Regulation of dopaminergic survival,
GDNF expression, and TNF-α/COX-2-driven neuro inflammation. GDNF,
glial cell line-derived neurotrophic factor;
1-methyl-4-phenylpyridinium (MPP+) selectively targets dopaminergic
neurons by uptake via the dopamine transporter (DAT), inhibiting
mitochondrial complex I (NADH dehydrogenase) to disrupt electron
transport, deplete ATP, and generate reactive oxygen species (ROS).
This produces oxidative stress, apoptosis, and energy failure,
emulating PD nigrostriatal degeneration. |
Recent experimental evidence indicates that naringin
and naringenin demonstrate extensive pharmacological activity, with
specific focus on neurodegenerative diseases including Alzheimer's
and Parkinson's, along with other neurological disorders, such as
anxiety, depression, schizophrenia and chronic hyperglycemic
peripheral neuropathy. Neurodegeneration is a complex process
influenced by various factors, including oxidative stress, dopamine
depletion, and neuro inflammation. Naringenin, a compound found in
citrus fruits, has been shown to boost dopamine levels and possess
anti-inflammatory properties that can aid in reducing inflammation
within the brain (20).
Naringenin may aid in the recovery of dopaminergic
neurons following injury if it is administered shortly after the
damage has occurred. Dopaminergic neurons are preserved, and glial
cell line-derived neurotrophic factor (GDNF) is restored in the
substantia nigra (SN), and the number of neurons immune reactive
for calcium-binding adaptor molecule 1 (Iba-1) and necrosis factor
α (TNF-α) are also resorted (21).
In the striatum was reduced as indicators of inflammation in the
brain after pre-treatment with naringenin in rats with unilateral
1-methyl-4-phenylpyridinium (MPP+) injury. Eukaryotic initiation
factor 4E-binding protein 1 (4E-BP1), and growth differentiation
factor and GDNF were upregulated in the cerebellum following a
single injection of naringin. Naringin then decreases TNF-α and
cyclooxygenase-2 (COX-2) levels and raises the transcription factor
nuclear factor 2 (Nrf2) (20).
The potential of naringin as an antioxidant shows
promise in the treatment of neurology and diabetes. The decline of
nerve cells and dopamine levels in the striatum and substantia
nigra pars compacta eliminates dopamine-producing cells, paving the
way for the development of Parkinson's disease (Fig. 2B).
Changes in the brain's dendritic arborization and
synaptic architecture have been shown to be associated with
psychological and neurological disorders such as depression,
anxiety and memory loss. Naringenin, a dietary flavanone found in
citrus fruits, vegetables, berries and nuts, may play a significant
role in addressing these issues (22).
Cyanidin
Cyanidin has been shown to protect SH-SY5Y human
neuroblastoma cells from MPP+-induced toxicity, which serves as a
model for Parkinson's disease. This finding highlights the
potential therapeutic benefits of cyanidin in combating
neurodegenerative diseases (23).
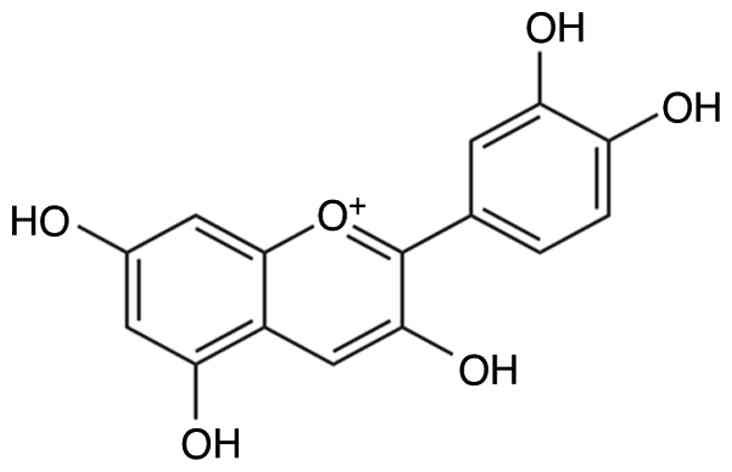
Cyanidin belongs to the flavonoid family of
polyphenolic natural compounds. The molecular structure of cyanidin
is illustrated in Fig. 3. It is
commonly found in fruits and vegetables, as well as in leaves,
petals, flowers and red fruits. Several vegetables and fruits
including blackberries, red onions, grapes, cherries, apples,
raspberries, peaches, plums, beans, red cabbage and cranberries
contain cyanidin (24).
Cyanidin have been shown to have potential
therapeutic effects on a variety of disorders and are commonly
recommended as medicines in several countries (25). Consumption cyanidins provide health
benefits against the development of obesity and diabetes, as well
as suppressing inflammatory. Cyanidin and its metabolites have
higher absorption and bioavailability, and interaction with gut
microbes may enhance their health benefits. Several in vitro
studies have demonstrated the ability of cyanidin to reduce
reactive oxygen species (26).
Despite current reports indicating the various health benefits of
cyanidin, its antioxidant potential through modulation of the Nrf2
pathway is still less defined in modulating oxidative stress
against DNA damage, apoptosis, carcinogenic toxicity and
inflammatory conditions (26). The
administration of cyanidin shows promise as a potential
pharmacological or functional food therapy for combating oxidative
stress and protecting against Alzheimer's disease. Cyanidin
possesses potent antioxidant properties and unique characteristics
that render it a promising candidate for further research and
development as a therapeutic agent. Therefore, it is imperative to
conduct further in vivo studies to explore its potential
benefits in greater detail (27).
To further elucidate the molecular mechanisms underlying the
neuroprotective effects of cyanidin, a previous study investigated
the effects of cyanidin on neuroinflammation in human neuroblastoma
cells (27). Although these
findings indicate the potential benefits of cyanidin for
neurological health, further research is necessary to establish a
direct association between cyanidin consumption and dopamine
production or regulation in humans.
Rutin
Rutin, a flavonoid quercetin glycoside and the
disaccharide rutinose (α-L-rhamnopyranosyl-(1→6)-β-D-glucopyranose)
(Fig. 4), is found in numerous
plants and fruits, partiuclarly buckwheat, apricots, cherries,
grapes, grapefruit, peaches and oranges (28). Pharmacological studies have
reported the beneficial effects of rutin in several disease states,
and its therapeutic potential in several models of non-communicable
diseases has aroused great enthusiasm. The present review
summarized the currently available knowledge on the mechanisms of
action of rutin in different experimental models of diseases of the
central nervous system. Rutin has been shown to upregulate the
tyrosine hydroxylase (TH) gene, a crucial component in the
biosynthesis of dopamine (28) The
mechanisms of action reviewed herein include reducing
pro-inflammatory cytokines, improving antioxidant enzyme
activities, activating mitogen-activated protein kinase cascade,
downregulating mRNA expression of Multiple Sclerosis (MS)-related
and pro-apoptotic genes, upregulating ion transport and
anti-apoptotic genes and restoring mitochondrial complex enzyme
activities. These findings suggest that rutin may be a promising
neuroprotective compound for the treatment of atypical neurological
disorders (29).
Rutin has been found to upregulate the TH gene, a
crucial component in dopamine biosynthesis (28). This discovery suggests that rutin
may play a role in increasing dopamine production within the body.
In a study utilizing a rat model of 6-hydroxydopamine
(6-OHDA)-induced Parkinson's disease, pre-treatment with rutin was
shown to protect against the reduction in dopamine content and its
metabolite 3,4-dihydroxyphenyl acetic acid (30). These results indicate that rutin
could potentially help in maintaining optimal dopamine levels in
the brain. Furthermore, rutin has exhibited neuroprotective
properties on dopaminergic neurons. In a previous study, in an
animal model of Parkinson's disease, rutin was able to shield
neurons in the substantia nigra from the detrimental effects of
6-OHDA, indirectly supporting dopamine function (30). When combined with levodopa, a
common medication used for Parkinson's disease, rutin was shown to
mitigate the peripheral adverse effects of dopamine, such as nausea
and hypotension (28). This
suggests that rutin may enhance the efficacy of dopamine-related
treatments. Moreover, rutin has demonstrated antioxidant and
anti-inflammatory characteristics, which could contribute to its
neuroprotective effects (31). By
reducing oxidative stress and inflammation, rutin may aid in the
preservation of dopaminergic neurons and the maintenance of
dopamine levels. While these findings are promising, it is
important to acknowledge that the majority of studies have been
conducted using animal models or in vitro. Further research,
particularly human clinical trials, is necessary to fully
comprehend the effects of rutin on dopamine levels and its
potential therapeutic applications in neurodegenerative disorders.
Rutin, a flavonoid present in numerous plants and fruits, has
demonstrated promising effects on dopamine levels and
neuroprotection in several studies (31).
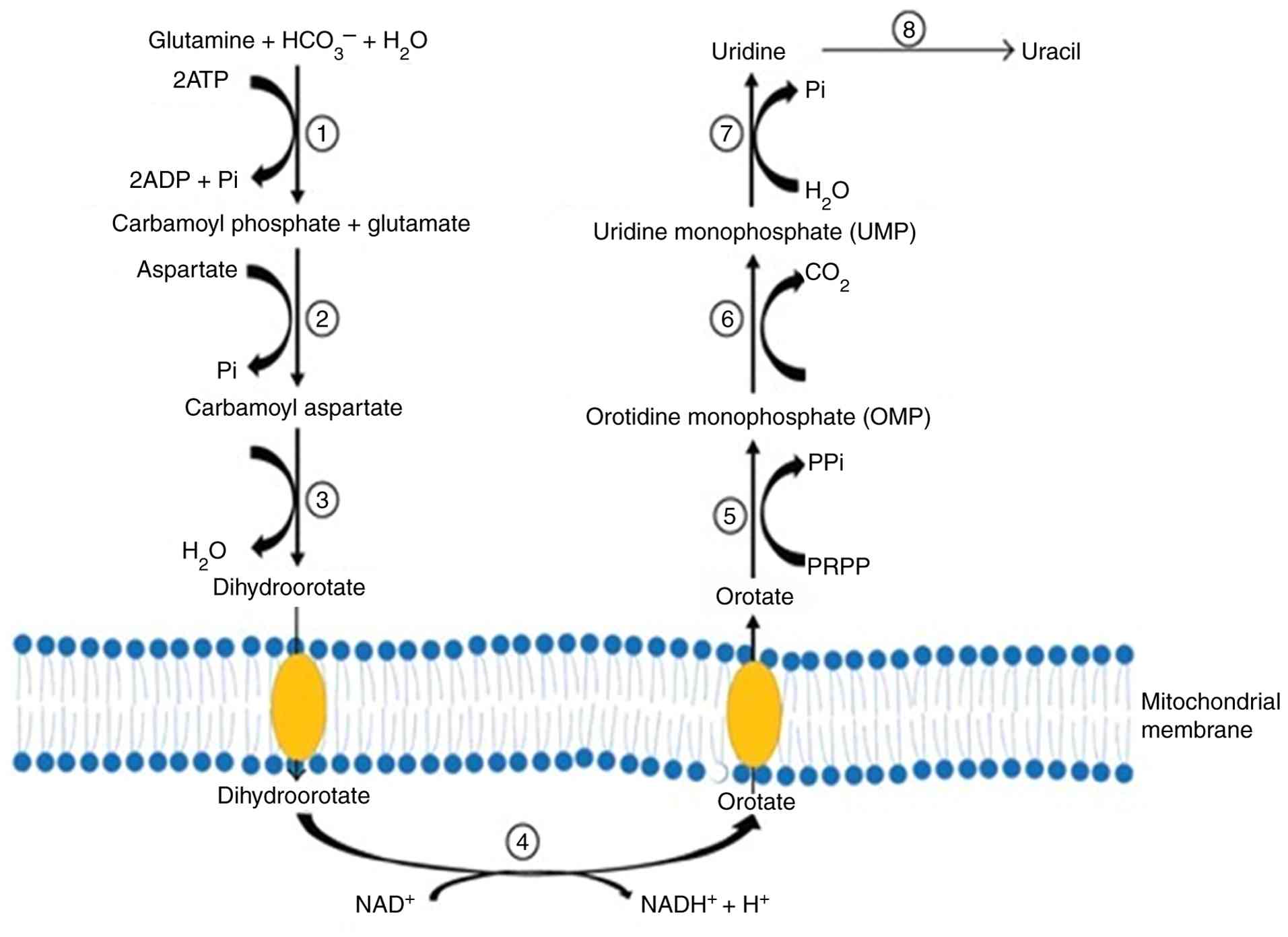
Uridine
Uridine occurs primarily in blood and brain fluid,
where it helps to maintain fundamental cellular activities that are
influenced by enzyme activity, dietary patterns and ATP depletion.
Certain mushrooms may have distinct synergistic effects with
uridine, and antioxidants, potentially improving brain function
(32,33). Uridine is contained in infant
formulae as its monophosphate, UMP, which is bioavailable (34,35).
Uridine is known to play a crucial role in
modulating neurotransmitter systems, specifically dopamine and
serotonin. This nucleoside has been shown to potentially produce an
anti-epileptic effect by disrupting the dopaminergic system. Giant
oyster mushrooms, lion's mane and tiger milk mushroom (a
lesser-known variation) promote neurite outgrowth, which
regenerates nerve cells. They boosted nerve growth factor in both
cells and animals.
Several foods contain uridine in the form of RNA
(36). Although it is claimed that
virtually none of the uridine in this form is bioavailable, as
demonstrated by Gasser et al (36) in 1981, it is destroyed in the liver
and gastrointestinal tract, and no food, when consumed, has ever
been reliably shown to elevate blood uridine levels. A previous
study found that plasma uridine levels increased 1.8-fold at 30 min
following the consumption of beer, indicating at the very least
inconsistent evidence (37). On
the other hand, ethanol which is found in beer raises uridine
levels, which may explain the increased Uridine plasma
concentration rises due to pyrimidine breakdown after purine
degradation by ethanol and fructose (38).
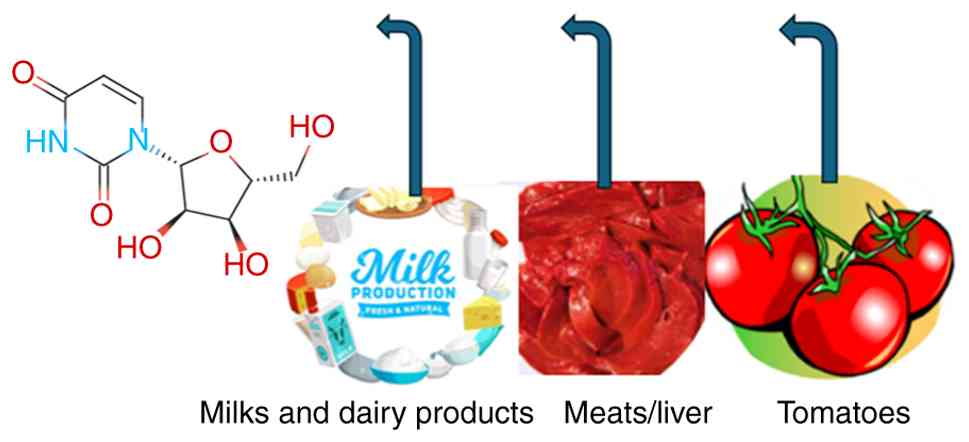
A number of natural products rich in uridine
(Fig. 5), such as milk and dairy
products from goats and sheep, tomatoes (0.5-1.0 g uridine per
kg/dry weight) (39), sugarcane
extract, broccoli and beer (40),
meats (such as the liver and pancreas) and Brewer's yeast (1.7%
uridine, dry weight) (39).
Uridine metabolism consists of three stages: De novo
synthesis, salvage synthesis path and catabolism and homeostasis,
which is closely related to glucose homeostasis, and the breakdown
of amino acids (40). Uridine
contributes to the glycolysis pathway of galactose (41). There is no metabolic mechanism that
converts galactose. As a result, galactose is converted to glucose
and metabolized by the common glucose pathway. Uridine is absorbed
by the brain to generate CDP-choline, a well-known memory enhancer,
as well as other phospholipids. In a study on healthy individuals
who underwent brain imaging, uridine supplements boosted their
brain levels of phosphor ethanolamine (PE), a key
phospholipid-building component (42). Uridine is produced by the liver and
is released during regular cell and RNA breakdown in humans,
particularly during fasting from fat cells. As a result, uridine is
constantly present in the circulation (43). Recently produced or recycled
uridine engages in several critical activities in the brain and
nervous system, including maintaining the lipid membranes of neural
cells and creating connections between neurons, as it can pass the
blood-brain barrier (44).
Additionally, it appears to bind to P2Y2, P2Y4, P2Y6 and P2Y14
receptors in the brain. However, it may also potentially be
attached to other, yet unidentified receptors (45). Uridine is deemed to be a promising
molecule for further research; however, there are presently few
human clinical trials evaluating its effects and there is
insufficient information available on its metabolism (Fig. 6) and its function in the brain
(45).
Vitamin D and sunlight
Human skin produces vitamin D, a fat-soluble
vitamin, when it is exposed to sunlight. However, the majority of
individuals do not receive sufficient levels of sun-induced vitamin
D. Researchers estimate that 50% of individuals may not have
sufficient levels vitamin D (46).
Vitamin D is produced in the skin following exposure to UVB rays
but without burns. In addition to the liver and kidneys, where
1,25(OH)2D is produced in a paracrine/autocrine function,
additional tissues also receive vitamin D subsequent metabolism to
produce its primary circulating form, 25(OH)D, and hormonal form,
1,25(OH)2D. The immune system, intestinal epithelium, parathyroid
gland, prostate gland and breast are a few types of these tissues
(46).
Lower dopamine levels are caused by a vitamin D
deficiency, while dopamine release is enhanced by vitamin D3
therapy (47,48). One of the most natural methods to
raise dopamine levels in the brain is to spend time in the sun.
Studies have revealed that exposure to sunshine increases dopamine
release (49,50).
Dopamine levels in the brain can rise following
exposure to sunlight. To support dopamine, release, exposure to
sunlight during the spring and summer seasons is mandatory. It is
vital to expose the top of the face and head to sunlight to promote
dopamine levels. It is therefore recommended that no head and face
cover or sunglasses be used to increase the level of dopamine in
the brain (51).
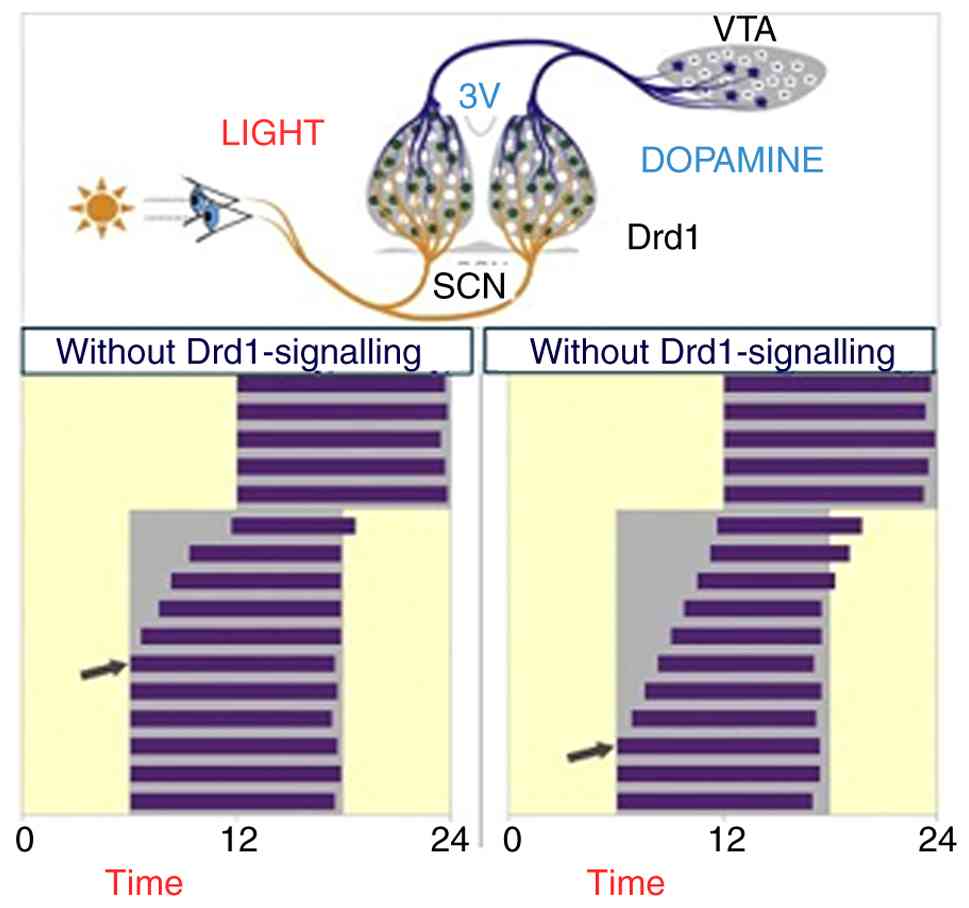
Dopamine D1 receptor (Drd1) signaling within the
superchiasmatic nucleus (SCN) is essential for resynchronizing
activity rhythms in time with phase-shifting light-dark cycles and
that raising the Dopamine (DA) tone through the selective
activation of dopaminergic neurons in the ventral tegmental area
VTA accelerates photic stimulation (51). (Fig.
7), illustrates the functional connection between dopaminergic
neurons in the ventral tegmental area (VTA) and neurons in the SCN
that express Drd1 receptors, demonstrating their direct
neuroanatomical and functional link. Stimulating these neurons
while recording SCN activity allows for functional assessment and
real-time study of the VTA-SCN circuit dynamics, accelerating the
response to light stimulation.
There are several natural products rich in vitamin
D. According to studies, 25(OH)D appears to be roughly 5-fold more
potent than the parent vitamin in terms of increasing blood 25(OH)D
concentration (52,53). Further research discovered that
when the 25(OH)D content of beef, pig, chicken, turkey and eggs is
taken into consideration, the overall quantity of vitamin D in the
meal is 2- to 18-fold greater than the amount in the parent vitamin
alone, depending on the food (52).
Vitamin B9 and magnesium (Mg)
Vitamin B9 (folate) is a primary vitamin B that
plays a key role in methyl-group transfer reactions and is one of
the most critical vitamins required for the optimal performance of
energy and nervous system functions. The connection between mental
health signs and B vitamins must be understood. Mental symptoms can
be suppressed, and the disease duration may decrease with the use
of vitamins B6, B8 and B12; individuals with low folate levels in
their blood are at an increased risk of suffering from depression
(54). One of the reasons for this
is that folate is necessary for the production and enhancement of
dopamine in the brain (54,55).
When there are low folate blood levels, there will also be lower
levels of dopamine as the body cannot produce dopamine efficiently,
contributing to depression (56).
Another condition among depressed individuals is folate
deficiencies; aproximately one-third of depressive patients have an
absolute deficiency. Water-soluble vitamin folate is required for
the correct production of dopamine, adrenaline and serotonin
neurotransmitters (55). Natural
folate sources include leafy greens, asparagus, brussels sprouts,
avocado, beef liver, seeds and nuts (57). Natural compounds, such as magnesium
are an essential mineral. Unfortunately, many individuals are
deficient in it. This is unfortunate as it plays a role in >300
biochemical reactions in our body and is essential for optimal
neurotransmitter activity. Magnesium has antidepressant effects,
one reason being that it increases dopamine activity in the brain
(58).
There are ample foods that are high in magnesium and
the consumption of these foods on a regular basis can ensure
adequate levels. Such foods include spinach, watercress, pumpkin
seeds, almonds, avocados and nuts (58).
Amino acids. Phenylalanine (Phe) and
tyrosine (Tyr)
The only other known activities of aromatic amino
acids in the brain, previously being parts of protein, are as
precursors to the catecholamine (dopamine, norepinephrine and
adrenaline) and the monoamine neurotransmitter, serotonin. This
latter biochemical association is critical due to brain
concentrations of catecholamine [tryptophan as (5HT), Phe and Tyr]
and other precursor amino acids, which are in turn regulate the
synthesis and release of these neurotransmitters (59).
The large neutral amino acids and other amino acids
that compete with them for a shared transporter across the blood
brain barrier, as well as physiological and pathophysiological
variables that affect the blood concentrations of these amino acids
predictably alter the concentrations of aromatic amino acids in the
brain, the production and release of certain monoamine
transmitters, and subsequently the way in which the brain functions
(59,60).
With an emphasis on the Tyr-catecholamine
association, the present review takes into account the research
demonstrating that precursor availability affects monoamine
synthesis, as well as the physiological variables that affect the
connection. The hypothesis that Phe functions as a substrate with
Tyr for Tyr hydroxylase and is not an inhibitor of this enzyme
in vivo at normal and even high concentrations, is supported
by the consideration of the role of Phe in catecholamine
production. The present review also discusses how Tyr-mediated
modifications to catecholamine production affect brain activities
(61).
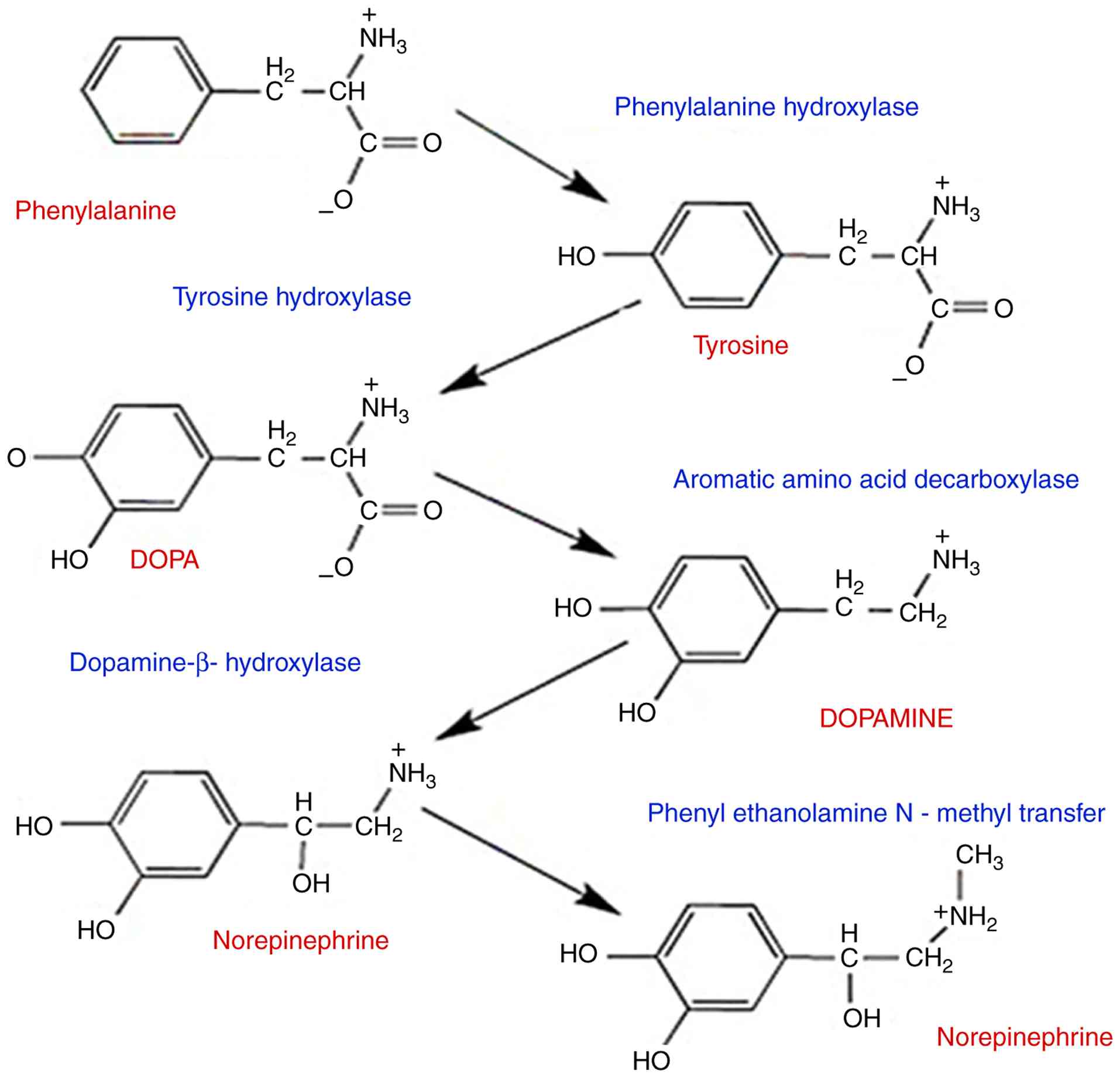
The enzyme TH catalyzes the first step, which is
hydroxylation to DOPA. Aromatic L-amino acid decarboxylase rapidly
converts DOPA to dopamine after it is produced. In neurons that
employ dopamine as a transmitter, enzymatic alteration stops there.
An extra enzyme called dopamine-by-hydroxylase is present in
neurons that employ norepinephrine as a transmitter, and it
transforms dopamine into norepinephrine. Phenylethanolamine
N-methyltransferase is an extra enzyme found in neurons that use
epinephrine as a transmitter. It catalyzes the conversion of
norepinephrine to epinephrine. Tyr hydroxylation, the first step in
the route, is rate limiting, which means that it regulates the rate
of synthesis throughout the whole pathway (61,62).
Initial research has revealed that the enzyme is susceptible to
end-product inhibition (63).
A previous in vivo study tracked the rate at
which 14C-Tyr converted to 14C catecholamine in the brains of rats
given a medication to increase endogenous catecholamine
concentrations (such as a monoamine oxidase inhibitor) or in
vitro using enzyme preparations (64). Subsequent research revealed that TH
was susceptible to several other regulators, such as an enzyme that
is rapidly activated in response to an increase in neuronal
activity (i.e., synthesis rises during times of greater neuronal
demand (65). Additional research
revealed that administering the amino acid to increase brain Tyr
concentrations can rapidly accelerate the production of DOPA in rat
brains (66). This result implied
that Tyr concentrations are at much below saturation levels in the
area around TH. This discovery was further refined to demonstrate
that neuronal activity was necessary, at least in DA neurons, to
detect a precursor-linked stimulation of DA production. For
instance, Tyr administration may increase the synthesis of dopamine
in the corpus striatum, which is home to a significant terminal
projection from DA cell bodies in the substantia nigra. However, to
activate the neurons in these rats, pre-treatment with a DA
receptor antagonist was necessary (67).
DA neurons in the rat retina provide a physiological
illustration of the requirements for neuronal activity. The retina
has a large amount of light-sensitive dopamine interneurons, which
are quiescent in darkness, but become active in light. Neuronal
activity is associated with TH activation (68). If activation is typically necessary
for Tyr administration to raise Tyr hydroxylation rate, Tyr
injection should increase hydroxylation rate in retinas during the
day but not at night, (69,70).
DOPA concentrations typically accumulate linearly for 30 min after
drug administration, providing an accurate estimate of the
hydroxylation rate and overall rate of catecholamine synthesis,
phenylethanolamine N-methyltransferase is a key enzyme in the
biosynthesis of catecholamines, specifically catalyzing the
conversion of norepinephrine (noradrenaline) into epinephrine
(adrenaline), the mechanism of biosynthesis for catecholamine
neurotransmitter appearances on (Fig.
8).
This reaction represents the terminal step in
catecholamine biosynthesis and is crucial for stress responses and
various physiological functions as the hydroxylation step is
rate-limited in the pathway (70,71).
A previous study examined how L-Tyrosine (TYR), a biological
precursor of dopamine believed to improve creativity and cognitive
regulation, promotes creativity in both divergent and convergent
thinking (72). While TYR did
promote convergent (or ‘deep’) thinking, no evidence could be found
that it had any influence on divergent (or ‘brainstorming’). That
study indicated that TYR may support control-hungry creative
processes, since convergent thinking may require more cognitive
top-down control. The meals consumed may thus have an impact on
thought processes (72). Tyrosine
is hydroxylase to L-DOPA by tyrosine hydroxylase, whereas
phenylalanine hydroxylase converts tyrosine to tyrosine. The enzyme
aromatic amino acid decarboxylase transforms DOPA into dopamine.
Dopamine-hydroxylase catalyzes the conversion of dopamine to
norepinephrine, and is then methylated to epinephrine by
phenylethanolamine N-methyltransferase. The enzyme that limits the
rate of the pathway is tyrosine hydroxylase (72). Monoterpene hydrocarbons,
particularly pinene, have been shown to be critical components in
essential oils, responsible for effects such as metal chelation,
free radical scavenging, enhancing reducing power and inhibiting
certain enzyme functions (73).
Since acute β-PEA administration increased the active psychomotor
behaviors of β-PEA-administered mice and produced a positive
emotional state, the dopamine concentration and dopamine-related
protein expression of rats administered β-PEA was investigated to
determine whether β-PEA changes dopaminergic neurotransmission
(74). The results revealed that
the rats administered acute β-PEA had significantly higher levels
of dopamine than the saline control group (74). Moreover, when rats received β-PEA
immediately, it significantly improved their tyrosine hydroxylase
levels and immune responses relative to the saline control group.
The immunological responses of DAT and vesicular monoamine
transporter 2 (VMAT-2) levels, however, did not change between the
β-PEA-administered rats and the saline control group (74). Acute β-PEA treatment in mice
enhanced the level of striatal dopamine, as well as the expression
of p-DAT and TH, an enzyme that limits the production of dopamine.
According to the findings, acute β-PEA injection is expected to
cause p-DAT, reverse DAT function, and ultimately boost dopamine
neurotransmission in the striatum. Notably, it was discovered that
locomotor activity remained unaltered even after acutely
administering 50 mg/kg β-PEA, which increased the striatal dopamine
concentration It was revealed that mice treated with β-PEA had
higher amounts of striatal dopamine and greater locomotor activity,
as well as stereotyped behaviors such as grooming. (74). However, in an alternative
investigation, β-PEA at an elevated dose of 50 mg/kg heightened
circling, which is recognized as stereotyped behavior, and the
head-twitching reaction, which is deemed to be a behavioral
indicator of the hallucinogenic effect in humans through the hyper
stimulation of dopaminergic neurotransmission (75).
Natural products rich in Phe and Tyr. The
following foods are excellent providers of Phe and Tyr: Meats
(lamb, venison, hog and beef), fowl (duck, goose, turkey and
chicken), seafood (shrimp, lobster, mackerel, salmon, trout and
tuna), eggs, dairy products (yoghurt, cheese and milk), nuts
(cashews, walnuts, macadamia nuts, pistachios and almonds),
pumpkin, squash, hemp and sunflower seeds, nut butters (such as
cashew, almond and peanut butter), legumes (beans, black beans,
chickpeas and lentils), whole grains (wheat, barley, rye, oats and
quinoa), soy goods (edamame, tempeh, tofu, soybeans) and protein
supplements (76). Phe is present
in a wide variety of high-protein plant and animal foods, such as
meat, fish, poultry and legumes. Aspartame, an artificial sweetener
frequently added to diet soda and other sugar-free meals, also
contains Phe (76). Natural
protein or dietary Phe intake is limited by the European
Phenylketonuria Guidelines to no >25% of daily intake to
maintain target blood Phe concentrations (77). Phenylketonuria (PKU) is an
autosomal recessive genetic disorder of phenylalanine metabolism
caused by a deficiency of the enzyme hydroxylase, which catalyzes
the conversion of phenylalanine to tyrosine (76). The enzyme which regulates the rate
of dopamine production is known as tyrosine hydroxylase, or Tyr H.
The hydroxylation of tyrosine into L-DOPA is catalyzed through;
hormones and neurotransmitters in the central and peripheral
nervous systems, catecholamine dopamine, epinephrine and
norepinephrine are the end products of the route. In the latter
case, the adrenal medulla synthesizes them (78,79).
Several brain processes, including attention (80), memory cognition (81) and emotion, are influenced by these
catechol monoamines (82,83). The hormone known as the
fight-or-flight response, epinephrine, which is generated by the
adrenal gland, has an impact on several bodily tissues (84). As a result, variations in
catecholamine levels can have a variety of effects, possibly
including elevated blood pressure, addiction, dystonia and bipolar
disorder (85,86). Tyr H is the slowest enzyme in the
process; therefore, its activity is highly relevant to a number of
biomedical research disciplines. The complex nature of Tyr H
regulation is logical considering the significance of its function.
Research on transcription processes controlling its production, as
well as the relatively recent topic of its destruction in the
proteasome over degradation is extremely active (87).
Omega-3 fatty acids
The main sources of omega-3 fatty acids are cold
water fish, such as Salmon, dark-water cod, sailfish, sardines and
herring. In a study that nurtured rats with omega-3 adipose acids
and a control group that did not receive these acids, the animals
fed the omega-3 adipose acids had 40% more dopamine in their
frontal brain than rats on the control diet, and more advanced
situations of dopamine in the brain than the animals that did not
receive the omega-3 adipose acids (88). That study also noted a decrease in
the enzyme that breaks down dopamine and increased dopamine binding
to dopamine receptors (88).
Research also shows that omega-3 adipose acids can help restore
normal dopamine release following traumatic brain injury (89). Considering the widespread relevance
of marine omega-3 fatty acids, it is recommended to consume fish or
other seafood 1-2 times per week, particularly fatty (dark flesh)
fish high in eicosatetraenoic acid (EPA) and docosahexaenoic acid
(DHA) (90). This is paraticularly
crucial for women who are pregnant or who wish to become pregnant,
as well as nursing mothers. A developing child also needs a
consistent supply of DHA from the third trimester to the second
year of life to create the brain and other elements of the nervous
system, as DHA is the most abundant fatty acid in the brain. While
the evidence for harm from a lack of omega-3 fats is significantly
more consistent and there is a balance of benefit vs. risk, a
number of women avoid eating fish out of fear that mercury and
other potential pollutants would harm their future children
(91).
The strongest evidence yet for the beneficial
effects of omega-3 fats comes from heart disease. These fats appear
to help maintain a regular heartbeat as opposed to an irregular
one, which may be fatal (92).
These arrhythmias account for the majority of the >500,000
cardiac-related deaths that occur in the USA annually. Furthermore,
omega-3 fats lower blood pressure and heart rate, while improving
blood vessel health. Increased doses lower triglycerides and
inflammation, both of which are factors in the development of
atherosclerosis (92). Omega-3
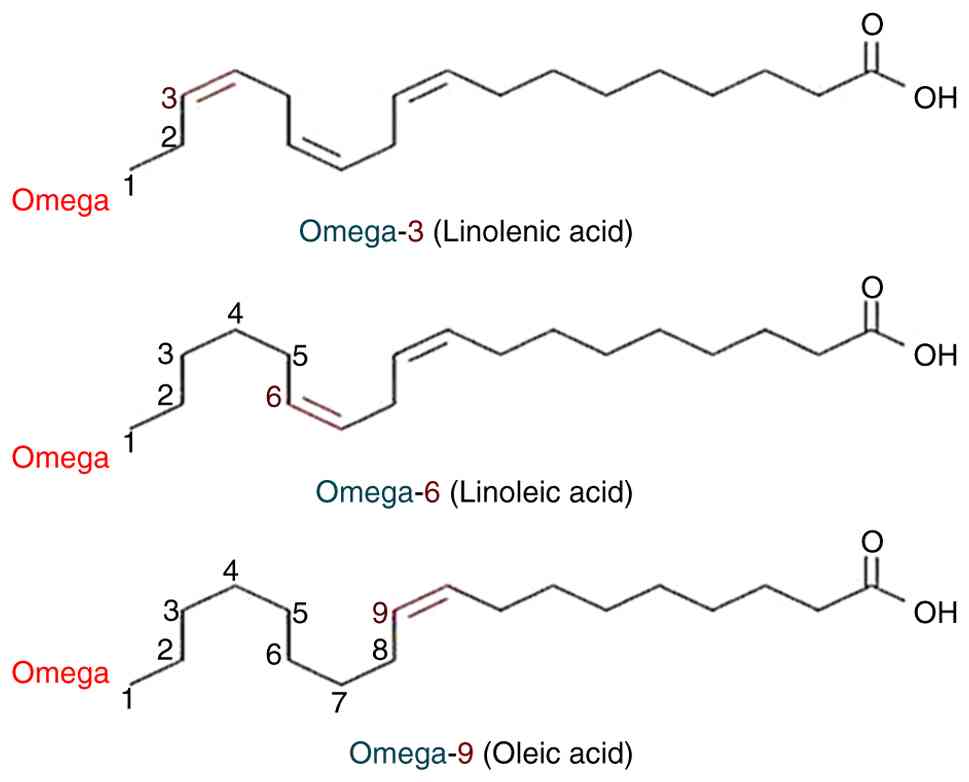
fatty acids (omega-3s) include a carbon-carbon double bond that is
located three carbons from the methyl end of the chain. Foods high
in omega-3s, or n-3s, include fish oil and flaxseed, as well as
dietary supplements. The majority of the scientific research
focuses on three types of omega-3 fatty acids (Fig. 9): Alpha-linoleic acid (ALA), EPA
and DHA. With 20 and 22 carbon atoms, respectively, EPA and DHA are
considered long-chain (LC) omega-3s, although ALA only possesses 18
carbon atoms. As a result, linoleic acid and ALA are considered as
essential fatty acids, which indicates that they can only be
received via food (93). Only a
minimal amount of conversion (<15%) has been reported between
ALA and EPA and DHA, which occurs mostly in the liver (92). Therefore, the only feasible option
to raise levels of these fatty acids in the body is to consume EPA
and DHA directly from meals and/or dietary supplements.
Natural products rich in omega-3s
Variations exist in fish's omega-3 content. High
concentrations of long-chain omega-3 fatty acids (LC omega-3s) are
found in cold-water fatty fish, such salmon, mackerel, tuna,
herring, and sardines; low-fat fish, like cod, tilapia, and bass,
and shellfish, have small concentrations (92). The association between the omega-3
concentration in fish and the makeup of their diet is also evident
(94). DHA and EPA levels in
farmed fish are typically greater than in wild fish, but this
varies depending on the diet (94). The plant oils canola, soybean and
flaxseed (linseed) oils are among those that contain ALA, (92,93).
Additionally, high in ALA are walnuts and chia seeds. Certain foods
are fortified with DHA and other omega-3s, including several types
of yogurts, eggs, milk, juices and soy drinks. In the USA, the
majority of newborn formulae on the market since 2002 have included
DHA and arachidonic acid, which are the two most common LC PUFAs in
the brain (95).
Caffeine
Caffeine is the most frequently utilized
psychoactive substance (96).
Caffeine maintains dopamine levels higher, particularly in brain
regions associated with ‘attention’. Caffeine enhances sustained
attention and attentiveness through this neurochemical interaction
and lowers tiredness feelings (97,98).
Its pharmacological effects on behavior are comparable to those of
stimulants (methylphenidate and amphetamine) and 2-phenyl
methyl-sulfinyl acetamide, is the standard IUPAC chemical name for
Modafinil, which boost dopamine signaling by augmenting dopamine
release from the terminals or inhibiting dopamine transporters
(99,101). The stimulating, relaxing and
reinforcing effects of these medications are due to their
dopamine-enhancing properties (100,102-104).
On the other hand, On the other hand, preclinical research suggests
that the antagonistic actions of caffeine on adenosine receptors
(A1 and A2A subtypes) improves their pharmacological effects
(104). Its antagonistic action
on striatal A2A receptor (A2AR) has been specifically linked to
dopamine effects (105).
Similarly, as A2AR mutant animals lack these responses,
caffeine-induced increases in locomotor activity and emotion appear
to be mediated by A2AR (106,107). Additionally, the effects of
caffeine on wakefulness are disrupted when A2AR expression is
silenced in the nucleus accumbency using short-hairpin RNA
(108).
Other studies have demonstrated that caffeine causes
the brain dopamine levels to rise, (109,110); additionally an excellent approach
to raising dopamine levels is to drink coffee. It also increases
dopamine receptors and improves dopamine signaling (111). Dopamine levels in the brain are
increased by coffee and other stimulants. This may be due to the
good taste of coffee. However, coffee and caffeine may interfere
with sleep; thus, their consumption should be avoided in the
evening and before bed. Finally, attempting to consume all the
coffee fruit as opposed to simply coffee or pure caffeine may be
beneficial. The coffee bean is traditionally removed from the
coffee fruit to be roasted. The fruit that surrounds it is thrown
away. However, this is problematic since the coffee fruit has
several beneficial ingredients that are absent from coffee beans.
Furthermore, using whole coffee fruit concentrate has been shown to
considerably improve cognitive performance (112). It has also been demonstrated that
tea raises dopamine levels in the brain. This includes black tea,
as well as green tea (113). The
amino acid threonine is present in both black and green tea.
Additionally, it has been demonstrated that threonine crosses the
blood-brain barrier and markedly increases dopamine release in the
brain (114-116).
Caffeine naturally occurs in plant leaves, seeds and fruits,
substitute, where it acts as a natural herbicide, insect repellent
and pollinator attractants Caffeine naturally occurs in plant
leaves, seeds and fruits, (117,118). This botanically consequent
chemical is the most widely used stimulant globally (119). Caffeine enters the human diet
through plant-based foods (Fig.
10), such as coffee beans, tea leaves, guarana, cocoa beans,
and kola nuts (120). Coffee is a
key caffeine source in the diet (121).
4. Challenges and future prospects
The focus of the present review was to summarize the
significance of natural bioactive compounds that contribute to
boosting dopamine levels in the brain by influencing the amount of
central nerve dopamine. Numerous direct investigations have been
conducted in which nerve dopamine levels were gradually boosted, as
well as dopamine generation utilizing numerous well-studied plants.
Natural bioactive chemicals can enhance neuronal dopamine levels.
The study of natural and bioactive compounds has attracted
increasing attention due to their direct influence on brain health,
physiological activities and medicinal and edible properties.
Researchers have conducted numerous studies on bioactive compounds
and their biological efficacy for living organisms. However, the
extraction process, structure determination, activity and mechanism
of these bioactive compounds have not formed systematic theoretical
and technical system, which greatly limits their functional
application. Therefore, the majority of commercially available
natural products are functional foods containing bioactive
compounds that affect dopamine levels in the brain. The main
reasons for this situation are as follows:
The composition, structure and activity of bioactive
compounds in increasing dopamine levels in the culture medium are
relatively different, and the association between the source,
chemical structure and biological activity is not yet clear. Even
for well-studied plant compounds, there are only some preliminary
discussions on the relationship of structure to biological activity
in humans. However, the majority of studies on biological activity
are at the cell and/or animal level; only a limited number of
clinical or preclinical studies have been conducted, and the
mechanism of biological activity still needs further confirmation.
These issues impede the translation of biologically active
ingredients that regulate dopamine levels in the human brain.
The industrial production of bioactive chemicals in
the human brain suffers by the absence of efficient techniques for
extraction, purification, or synthesis, and several small molecular
active ingredients remain in the phase of structural elucidation
and in vitro activity validation.
The advancement of technological systems to maximize
the utilization of bioactive compounds in food, healthcare products
and clinical medicine is essential for improving brain dopamine
levels.
5. Conclusion
In order to develop an effective preventive and
therapeutic strategy, it is crucial to possess a thorough
understanding of the bioactive compounds that impact brain dopamine
levels. These compounds play a vital role in preventing
neurodegenerative diseases linked to fluctuations in brain dopamine
levels, improving cognitive functions such as memory and focus,
heightening feelings of pleasure, regulating mood and attention and
managing psychiatric disorders. Researchers need to transition from
investigating the antioxidant properties of natural bioactive
compounds to determining the ideal brain dopamine levels based on
factors such as age, gender, and developmental stages.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
The author (MOE) has confirmed the authenticity of
all the raw data and have reviewed and approved the final version
of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript or
to generate images, and subsequently, the authors revised and
edited the content produced by the AI tools as necessary, taking
full responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Sairazi NS and Sirajudeen KNS: Natural
products and their bioactive compounds: Neuroprotective potentials
against neurodegenerative diseases. Evid Based Complement Alternat
Med. 2020(6565396)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Liu Z, Zhou T, Ziegler AC, Dimitrion P and
Zuo L: Oxidative stress in neurodegenerative Diseases: From
molecular mechanisms to clinical applications. Oxid Med Cell
Longev. 2017(2525967)2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Sharifi-Rad M, Lankatillake C, Dias DA,
Docea AO, Mahomoodally MF, Lobine D, Chazot PL, Kurt B, Tumer TB,
Moreira AC, et al: Impact of natural compounds on neurodegenerative
disorders: From preclinical to pharmacotherapeutics. J Clin Med.
9(1061)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Naomi R, Yazid MD, Teoh SH, Balan SS,
Shariff H, Kumar J, Bahari H and Embong H: Dietary polyphenols as a
protection against cognitive decline: Evidence from animal
experiments; mechanisms and limitations. Antioxidants (Basel).
12(1054)2023.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ramis MR, Sarubbo F, Moranta D, Tejada S,
Lladó J, Miralles A and Esteban S: Cognitive and neurochemical
changes following polyphenol-Enriched diet in rats. Nutrients.
13(59)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Grosso C, Santos M and Barroso MF: From
plants to Psycho-neurology: Unravelling the therapeutic benefits of
bioactive compounds in brain disorders. Antioxidants (Basel).
12(1603)2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Bonacaro A, Arosio M, Svelto A and
Tettamanti M: The effects of essential oil and massage on patients
affected by Alzheimer's disease. J Gerontological Nurs. 48:39–47.
2022.
|
|
8
|
Xu H and Yang F: The interplay of dopamine
metabolism abnormalities and mitochondrial defects in the
pathogenesis of schizophrenia. Transl Psychiatry.
12(464)2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Daubner SC, Le T and Wang S: Tyrosine
hydroxylase and regulation of dopamine synthesis. Arch Biochem
Biophys. 508:1–12. 2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Cruickshank L, Kennedy AR and Shankland N:
‘CSD Entry TIRZAX: 5-(2 Ammonioethyl)-2-hydroxyphenolate,
Dopamine’. Cambridge Structural Database: Access Structures.
Cambridge Crystallographic Data Centre, 2013
doi:10.5517/cc10m9nl.
|
|
11
|
Best JA, Nijhout HF and Reed MC:
Homeostatic mechanisms in dopamine synthesis and release: A
mathematical model. Theor Biol Med Model. 6(21)2009.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Berry MD, Juorio AV and Li XM: Aromatic
L-amino acid decarboxylase: A neglected and misunderstood enzyme.
Neurochem Res. 21:1075–1087. 1996.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Cho HU, Kim S and Sim J: Redefining
differential roles of MAO-A in dopamine degradation and MAO-B in
tonic GABA synthesis. Exp Mol Med. 53:1148–1158. 2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Akil M, Kolachana BS, Rothmond DA, Hyde
TM, Weinberger DR and Kleinman JE: Catechol-O-methyl transferase
genotype and dopamine regulation in the human brain. J Neurosci.
23:2008–2013. 2003.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Franco R, Reyes-Resina I and Navarro G:
Dopamine in health and disease: Much more than a neurotransmitter.
Biomedicines. 9(109)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Joshi R, Kulkarni YA and Wairkar S:
Pharmacokinetic, pharmacodynamic and formulations aspects of
Naringenin: An update. Life Sci. 215:43–56. 2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kumar S and Pandey AK: Chemistry and
biological activities of flavonoids: An overview.
ScientificWorldJournal. 2013(162750)2013.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Gattuso G, Barreca D, Gargiulli C, Leuzzi
U and Caristi C: Flavonoid composition of citrus juices. Molecules.
12:1641–1673. 2007.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Shilpa VS, Shams R, Dash KK, Pandey VK,
Dar AH, Ayaz Mukarram S, Harsányi E and Kovács B: Phytochemical
properties, extraction, and pharmacological benefits of naringin: A
review. Molecules. 28(5623)2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Emran TB, Islam F, Nath N, Sutradhar H,
Das R, Mitra S, Alshahrani MM, Alhasaniah AH and Sharma R: Naringin
and naringenin polyphenols in neurological diseases: Understandings
from a therapeutic viewpoint. Life (Basel). 13(99)2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Chen C, Wei YZ, He XM, Li DD, Wang GQ, Li
JJ and Zhang F: Naringenin produces neuroprotection against
LPS-Induced dopamine neurotoxicity via the inhibition of microglial
NLRP3 inflammasome activation. Front Immunol.
10(936)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yilmaz E, Acar G, Onal U, Erdogan E,
Baltaci AK and Mogulkoc R: Effect of 2-Week naringin
supplementation on neurogenesis and BDNF levels in
ischemia-reperfusion model of rats. Neuromolecular Med.
26(4)2024.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chen J, Sun J, Jiang J and Zhou J:
Cyanidin protects SH-SY5Y human neuroblastoma cells from
1-Methyl-4-Phenylpyridinium-induced neurotoxicity. Pharmacology.
102:126–132. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
de Araújo FF, de Paulo Farias D, Neri-Numa
IA and Pastore GM: Polyphenols and their applications: An approach
in food chemistry and innovation potential. Food Chem.
338(127535)2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Cao G, Muccitelli HU, Sánchez-Moreno C and
Prior RL: Anthocyanins are absorbed in glycated forms in elderly
women: A pharmacokinetic study. Am J Clin Nutr. 73:920–906.
2001.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Rahman S, Mathew S, Nair P, Ramadan WS and
Vazhappilly CG: Health benefits of cyanidin-3-glucoside as a potent
modulator of Nrf2-mediated oxidative stress. Inflammopharmacology.
29:907–923. 2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Thummayot S, Tocharus C, Jumnongprakhon P,
Suksamrarn A and Tocharus J: Cyanidin attenuates Aβ25-35-induced
neuroinflammation by suppressing NF-κB activity downstream of
TLR4/NOX4 in human neuroblastoma cells. Acta Pharmacol Sin.
39:1439–1452. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Enogieru AB, Haylett W, Hiss DC, Bardien S
and Ekpo OE: Rutin as a potent antioxidant: Implications for
neurodegenerative disorders. Oxid Med Cell Longev.
2018(6241017)2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Chaudhuri A: Multiple sclerosis is
primarily a neurodegenerative disease. J Neural Transm (Vienna).
120:1463–1466. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Khan MM, Raza SS, Javed H, Ahmad A, Khan A
and Islam F, Safhi MM and Islam F: Rutin protects dopaminergic
neurons from oxidative stress in an animal model of Parkinson's
disease. Neurotox Res. 22:1–5. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Budzynska B, Faggio C, Kruk-Slomka M,
Samec D, Nabavi SF, Sureda A, Devi KP and Nabavi SM: Rutin as
neuroprotective agent: From bench to bedside. Curr Med Chem.
26:5152–5164. 2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Phan CW, David P, Wong KH, Naidu M and
Sabaratnam V: Uridine from pleurotus giganteus and its neurite
outgrowth stimulatory effects with underlying mechanism. PLoS One.
10(e0143004)2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Phan CW, Wong WL, David P, Naidu M and
Sabaratnam V: Pleurotus giganteus (Berk.) Karunarathna & K.D.
Hyde: Nutritional value and in vitro neurite outgrowth activity in
rat pheochromocytoma cells. BMC Complement Altern Med.
12(102)2012.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wurtman RJ: A nutrient combination that
can affect synapse formation. Nutrients. 6:1701–1710.
2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Carver JD: Advances in nutritional
modifications of infant formulas. Am J Clin Nutr. 77
(Suppl):1550S–1554S. 2003.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Gasser T, Moyer JD and Handschumacher RE:
Novel Single-pass exchange of circulating uridine in rat liver.
Science. 213:777–778. 1981.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Yamamoto T, Moriwaki Y, Takahashi S,
Tsutsumi Z, Ka T, Fukuchi M and Hada T: Effect of beer on the
plasma concentrations of uridine and purine bases. Metabolism.
51:1317–1323. 2002.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Yamamoto T, Moriwaki Y, Takahashi S,
Yamakita J, Tsutsumi Z, Ohata H, Hiroishi K, Nakano T and Higashino
K: Effect of ethanol and fructose on plasma uridine and purine
bases. Metabolism. 46:544–547. 1997.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Hidalgo A, Pompei C, Galli A and Cazzola
S: Uracil as an index of lactic acid bacteria contamination of
tomato products. J Agric Food Chem. 53:349–355. 2005.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Zhang Y, Guo S, Xie C and Fang J: Uridine
metabolism and its role in glucose, lipid, and amino acid
homeostasis. Biomed Res Int. 2020(7091718)2020.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Stryer B and T: Glycolysis Is an
Energy-Conversion Pathway in Many Organisms. In: Stryer B, Tymoczko
JL, (eds.) Glycolysis Is an Energy-Conversion Pathway in Many
Organisms. 5th edition, New York, W H Freeman, 2002.
|
|
42
|
Agarwal N, Sung YH, Jensen JE, daCunha G,
Harper D, Olson D and Renshaw PF: Short-term administration of
uridine increases brain membrane phospholipid precursors in healthy
adults: A 31-phosphorus magnetic resonance spectroscopy study at
4T. Bipolar Discord. 12:825–833. 2010.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Yamamoto T, Koyama H, Kurajoh M, Shoji T,
Tsutsumi Z and Moriwaki Y: Biochemistry of uridine in plasma. Clin
Chim Acta. 412:1712–1724. 2011.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Cansev M: Uridine and cytidine in the
brain: Their transport and utilization. Brain Res Rev. 52:389–397.
2006.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Dobolyi A, Juhasz G, Kovacs Z and Kardos
J: Uridine function in the central nervous system. Curr Top Med
Chem. 11:1058–1067. 2011.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Bikle DD: Feingold KR ABBM. Vitamin D:
Production, Metabolism and Mechanisms of Action. In: Vitamin D:
Production, Metabolism and Mechanisms of Action. South Dartmouth
(MA), 2021.
|
|
47
|
Trinko JR, Land BB, Solecki WB, Wickham
RJ, Tellez LA, Maldonado-Aviles J, de Araujo IE, A Addy N and
DiLeone RJ: Vitamin D3: A role in dopamine circuit regulation,
diet-induced obesity, and drug consumption. eNeuro 3:
ENEURO.0122-15.2016, 2016.
|
|
48
|
Kesby JP, Cui X, Ko P, McGrath JJ, Burne
THJ and Eyles DW: Developmental vitamin D deficiency alters
dopamine turnover in neonatal rat forebrain. Neurosci Lett.
461:155–158. 2009.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Aubert PM, Seibyl JP, Price JL, Harris TS,
Filbey FM, Jacobe H, Devous MD Sr and Adinoff B: Dopamine efflux in
response to ultraviolet radiation in addicted sunbed users.
Psychiatry Res Neuroimaging. 251:7–14. 2016.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Tsai HY, Chen KC, Yang YK, Chen PS, Yeh
TL, Chiu NT and Lee IH: Sunshine-exposure variation of human
striatal dopamine D2/D3 receptor availability in healthy
volunteers. Prog Neuropsychopharmacol Biol Psychiatry. 35:107–110.
2011.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Grippo RM, Purohit AM, Zhang Q, Zweifel LS
and Güler AD: Direct midbrain dopamine input to the suprachiasmatic
nucleus accelerates circadian entrainment. Curr Biol.
27:2465–2475.e3. 2017.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Taylor CL, Patterson KY, Roseland JM, Wise
SA, Merkel JM, Pehrsson PR and Yetley EA: Including food
25-hydroxyvitamin D in intake estimates may reduce the discrepancy
between dietary and serum measures of vitamin D status. J Nutri.
144:654–659. 2014.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Cashman KD, Seamans KM, Lucey AJ, Stöcklin
E, Weber P, Kiely M and Hill TR: Relative effectiveness of oral
25-hydroxyvitamin D3 and vitamin D3 in raising wintertime serum
25-hydroxyvitamin D in older adults. J Clin Nutri. 95:1350–1356.
2012.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Leahy LG: Vitamin B Supplementation:
What's the right choice for your patients? J Psychosoc Nurs Ment
Health Serv. 55:7–11. 2017.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Miller AL: The methylation,
neurotransmitter, and antioxidant connections between folate and
depression. Altern Med Rev. 13:216–226. 2008.PubMed/NCBI
|
|
56
|
Bottiglieri T, Laundy M, Crellin R, Toone
BK, Carney MW and Reynolds EH: Homocysteine, Folate, methylation,
and monoamine metabolism in depression. J Neurol Neurosurg
Psychiatry. 69:228–232. 2000.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Cuskelly GJ, McNulty H and Scott JM:
Effect of increasing dietary folate on red-cell folate:
Implications for prevention of neural tube defects. Lancet.
347:657–659. 1996.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Cardoso CC, Lobato KR, Binfaré RW,
Ferreira PK, Rosa AO, Santos AR and Rodrigues AL: Evidence for the
involvement of the monoaminergic system in the Antidepressant-like
effect of magnesium. Prog Neuropsychopharmacol Biol Psychiatry.
33:235–242. 2009.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Fernstrom JD: Aromatic amino acids and
monoamine synthesis in the central nervous system: Influence of the
diet. J Nutr Biochem. 1:508–517. 1990.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Fernstrom JD: Role of precursor
availability in control of monoamine biosynthesis in brain. Physiol
Rev. 63:484–546. 1983.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Kaufman S: Tyrosine Hydroxylase. In: John
Wiley and Sons, 2: 251. pp103-220, 1995.
|
|
62
|
Nagatsu T, Levitt M and Udenfriend S:
Tyrosine hydroxylase. The initial step in norepinephrine
biosynthesis. J Biol Chem. 239:2910–2917. 1964.PubMed/NCBI
|
|
63
|
Spector S, Gordon R, Sjoerdsma A and
Udenfriend S: End-product inhibition of tyrosine hydroxylase as a
possible mechanism for regulation of norepinephrine synthesis. Mol
Pharmacol. 3:549–555. 1967.PubMed/NCBI
|
|
64
|
Neff NH, Ngai SH, Wang CT and Costa E:
Calculation of the rate of catecholamine synthesis from the rate of
conversion of tyrosine-14C to catecholamines. Effect of adrenal
demedullation on synthesis rates. Mol Pharmacol. 5:90–99.
1969.PubMed/NCBI
|
|
65
|
Iuvone PM, Rauch AL, Marshburn PB, Glass
DB and Neff NH: Activation of retinal tyrosine hydroxylase in vitro
by cyclic AMP-dependent protein kinase: Characterization and
comparison to activation in vivo by photic stimulation. J
Neurochem. 39:1632–1640. 1982.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Carlsson A and Lindqvist M: Dependence of
5-HT and catecholamine synthesis on concentrations of precursor
amino-acids in rat brain. Naunyn Schmiedebergs Arch Pharmacol.
303:157–164. 1978.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Scally MC, Ulus I and Wurtman RJ: Brain
tyrosine level controls striatal dopamine synthesis in
haloperidol-treated rats. J Neural Transm. 41:1–6. 1977.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Iuvone PM, Galli CL, Garrison-Gund CK and
Neff NH: Light stimulates tyrosine hydroxylase activity and
dopamine synthesis in retinal amacrine neurons. Science.
202:901–902. 1978.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Fernstrom MH, Volk EA, Fernstrom JD and
Iuvone PM: Effect of tyrosine administration on dopa accumulation
in light- and dark-adapted retinas from normal and diabetic rats.
Life Sci. 39:2049–2057. 1986.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Wurtman RJ, Larin F, Mostafapour S and
Fernstrom JD: Brain catechol synthesis: Control by train tyrosine
concentration. Science. 185:183–184. 1974.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Carlsson A, Davis JN, Kehr W, Lindqvist M
and Atack CV: Simultaneous measurement of tyrosine and tryptophan
hydroxylase activities in brain in vivo using an inhibitor of the
aromatic amino acid decarboxylase. Naunyn Schmiedebergs Arch
Pharmacol. 275:153–168. 1972.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Colzato LS, de Haan AM and Hommel B: Food
for creativity: Tyrosine promotes deep thinking. Psychol Res.
79:709–714. 2015.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Abuhamdah S, Abuhamdah R, Howes MJ,
Al-Olimat S, Ennaceur A and Chazot PL: Pharmacological and
neuroprotective profile of an essential oil derived from leaves of
Aloysia citrodora Palau. J Pharm Pharmacal. 67:1306–1315.
2015.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Ryu IS, Kim OH, Kim JS, Sohn S, Choe ES,
Lim RN, Kim TW, Seo JW and Jang EY: Effects of β-Phenylethylamine
on psychomotor, rewarding, and reinforcing behaviors and affective
state: The role of dopamine D1 receptors. Int J Mol Sci.
22(9485)2021.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Dourish CT: An observational analysis of
the behavioural effects of beta-phenylethylamine in isolated and
grouped mice. Prog Neuropsychopharmacol Biol Psychiatry.
6:143–1458. 1982.PubMed/NCBI View Article : Google Scholar
|
|
76
|
MacDonald A, van Wegberg AMJ, Ahring K,
Beblo S, Bélanger-Quintana A, Burlina A, Campistol J, Coşkun T,
Feillet F, Giżewska M, et al: PKU dietary handbook to accompany PKU
guidelines. Orphanet J Rare Dis. 15(171)2020.PubMed/NCBI View Article : Google Scholar
|
|
77
|
van Wegberg AMJ, MacDonald A, Ahring K,
Bélanger-Quintana A, Blau N, Bosch AM, Burlina A, Campistol J,
Feillet F, Giżewska M, et al: The complete European guidelines on
phenylketonuria: Diagnosis and treatment. Orphanet J Rare Dis.
12(162)2017.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Molinoff PB and Axelrod J: Biochemistry of
catecholamines. Annu Rev Biochem. 40:465–500. 1971.
|
|
79
|
Weiner N: Tyrosine-3-monooxygenase
(tyrosine hydroxylase) In: Aromatic amino acid hydroxylases and
mental disease. John Wiley & Sons, Ltd, New York, pp141-190,
1979.
|
|
80
|
Tripp G and Wickens JR: Neurobiology of
ADHD. Neuropharmacology. 57:579–589. 2009.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Arnsten AF: Catecholamine regulation of
the prefrontal cortex. J Psychopharmacol. 11:151–162.
1997.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Anisman H and Zacharko RM: Behavioral and
neurochemical consequences associated with stressors. Ann N Y Acad
Sci. 467:205–225. 1986.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Brooks DJ and Piccini P: Imaging in
Parkinson's disease: The role of monoamines in behavior. Biol
Psychiatry. 59:908–918. 2006.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Weiner N: Regulation of norepinephrine
biosynthesis. Annu Rev Pharmacol. 10:273–290. 1970.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Cousins DA, Butts K and Young AH: The role
of dopamine in bipolar disorder. Bipolar Disord. 11:787–806.
2009.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Koob GF and Volkow ND: Neurocircuitry of
addiction. Neuropsychopharmacology. 35:217–238. 2010.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Døskeland AP and Flatmark T:
Ubiquitination of soluble and membrane-bound tyrosine hydroxylase
and degradation of the soluble form. Eur J Biochem. 269:1561–1569.
2002.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Harris WS: Coates PM BJBMR. Omega-3 fatty
acids. In: Encyclopedia of Dietary Supplements. 2nd edition. Vol
86. London and New York, Informa Healthcare, 2010.
|
|
89
|
Shin SS and Dixon CE: Oral fish oil
restores striatal dopamine release after traumatic brain injury.
Neurosci Lett. 496:168–171. 2011.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Rimm EB, Appel LJ, Chiuve SE, Djoussé L,
Engler MB, Kris-Etherton PM, Mozaffarian D, Siscovick DS and
Lichtenstein AH: American Heart Association Nutrition Committee of
the Council on Lifestyle and Cardiometabolic Health et al.
Seafood Long-Chain n-3 Polyunsaturated Fatty Acids and
Cardiovascular Disease: A Science Advisory From the American Heart
Association. Circulation. 138:e35–e47. 2018.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Oken E, Kleinman KP, Berland WE, Simon SR,
Rich-Edwards JW and Gillman MW: Decline in fish consumption among
pregnant women after a national mercury advisory. Obstet Gynecol.
102:346–351. 2003.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Leaf A: Prevention of sudden cardiac death
by n-3 polyunsaturated fatty acids. J Cardiovasc Med (Hagerstown).
8 (Suppl 1):S27–S29. 2007.PubMed/NCBI View Article : Google Scholar
|
|
93
|
JH Jones P and Papamandjaris AA: Lipids:
Cellular Metabolism. In: Present Knowledge in Nutrition. 10th
edition. Wiley, pp132-148, 2012.
|
|
94
|
Miller MR, Nichols PD and Carter CG: n-3
Oil sources for use in aquaculture-alternatives to the
unsustainable harvest of wild fish. Nutr Res Rev. 21:85–96.
2008.PubMed/NCBI View Article : Google Scholar
|
|
95
|
U.S. Food, Drug Administration. Questions
& answers for consumers concerning infant formulas, 2015.
|
|
96
|
Mitchell DC, Knight CA, Hockenberry J,
Teplansky R and Hartman TJ: Beverage caffeine intakes in the U.S.
Food Chem Toxicol. 63:136–142. 2014.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Volkow ND, Tomasi D, Wang GJ, Telang F,
Fowler JS, Logan J, Benveniste H, Kim R, Thanos PK and Ferré S:
Evidence that sleep deprivation downregulates dopamine D2R in
ventral striatum in the human brain. J Neurosci. 32:6711–6717.
2012.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Wisor JP, Nishino S, Sora I, Uhl GH,
Mignot E and Edgar DM: Dopaminergic role in stimulant-induced
wakefulness. J Neurosci. 21:1787–1794. 2001.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Volkow ND, Wang G, Fowler JS, Logan J,
Gerasimov M, Maynard L, Ding Y, Gatley SJ, Gifford A and Franceschi
D: Therapeutic doses of oral methylphenidate significantly increase
extracellular dopamine in the human brain. J Neurosci.
21(RC121)2001.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Abi-Dargham A, Kegeles LS, Martinez D,
Innis RB and Laruelle M: Dopamine mediation of positive reinforcing
effects of amphetamine in stimulant naïve healthy volunteers:
Results from a large cohort. Eur Neuropsychopharmacol. 13:459–468.
2003.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Volkow ND, Fowler JS, Logan J, Alexoff D,
Zhu W, Telang F, Wang GJ, Jayne M, Hooker JM, Wong C, et al:
Effects of modafinil on dopamine and dopamine transporters in the
male human brain: Clinical implications. JAMA. 301:1148–1154.
2009.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Nguyen TL, Tian YH, You IJ, Lee SY and
Jang CG: Modafinil-induced conditioned place preference via
dopaminergic system in mice. Synapse. 65:733–741. 2011.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Volkow ND, Wang GJ, Fowler JS, Logan J,
Gatley SJ, Wong C, Hitzemann R and Pappas NR: Reinforcing effects
of psychostimulants in humans are associated with increases in
brain dopamine and occupancy of D (2) receptors. J Pharmacol Exp
Ther. 291:409–415. 1999.PubMed/NCBI
|
|
104
|
Wuo-Silva R, Fukushiro DF, Borçoi AR,
Fernandes HA, Procópio-Souza R, Hollais AW, Santos R, Ribeiro LT,
Corrêa JM, Talhati F, et al: Addictive potential of modafinil and
cross-sensitization with cocaine: A pre-clinical study. Addict
Biol. 16:565–579. 2011.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Chen JF, Xu K, Petzer JP, Staal R, Xu YH,
Beilstein M, Sonsalla PK, Castagnoli K, Castagnoli N Jr and
Schwarzschild MA: Neuroprotection by caffeine and A(2A) adenosine
receptor inactivation in a model of Parkinson's disease. J
Neurosci. 21(RC143)2001.PubMed/NCBI View Article : Google Scholar
|
|
106
|
El Yacoubi M, Ledent C, Ménard JF,
Parmentier M, Costentin J and Vaugeois JM: The stimulant effects of
caffeine on locomotor behavior in mice are mediated through its
blockade of adenosine A(2A) receptors. Br J Pharmacol.
129:1465–1473. 2000.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Huang ZL, Qu WM, Eguchi N, Chen JF,
Schwarzschild MA, Fredholm BB, Urade Y and Hayaishi O: Adenosine
A2A, but not A1, receptors mediate the arousal effect of caffeine.
Nat Neurosci. 8:858–859. 2005.PubMed/NCBI View
Article : Google Scholar
|
|
108
|
Lazarus M, Shen HY, Cherasse Y, Qu WM,
Huang ZL, Bass CE, Winsky-Sommerer R, Semba K, Fredholm BB, Boison
D, et al: Arousal effect of caffeine depends on adenosine A2A
receptors in the shell of the nucleus accumbens. J Neurosci.
31:10067–10075. 2011.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Solinas M, Ferré S, You ZB, Karcz-Kubicha
M, Popoli P and Goldberg SR: Caffeine induces dopamine and
glutamate release in the shell of the nucleus accumbens. J
Neurosci. 22:6321–6324. 2002.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Zheng X, Takatsu S, Wang H and Hasegawa H:
Acute intraperitoneal injection of caffeine improves endurance
exercise performance in association with increasing brain dopamine
release during exercise. Pharmacol Biochem Behav. 122:136–143.
2014.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Volkow ND, Wang GJ, Logan J, Alexoff D,
Fowler JS, Thanos PK, Wong C, Casado V, Ferre S and Tomasi D:
Caffeine increases striatal dopamine D2/D3 receptor availability in
the human brain. Transl Psychiatry. 5(e549)2015.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Heimbach JT, Marone PA, Hunter JM, Nemzer
BV, Stanley SM and Kennepohl E: Safety studies on products from
whole coffee fruit. Food Chem Toxicol. 48:2517–2525.
2010.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Chaturvedi RK, Shukla S, Seth K, Chauhan
S, Sinha C, Shukla Y and Agrawal AK: Neuroprotective and
neurorescue effect of black tea extract in
6-hydroxydopamine-lesioned rat model of Parkinson's disease.
Neurobiol Dis. 22:421–434. 2006.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Alramadhan E, Hanna MS, Hanna MS,
Goldstein TG, Avila SM and Weeks BS: Dietary and botanical
anxiolytics. Med Sci Monit. 18:RA40–RA48. 2012.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Yokogoshi H, Kobayashi M, Mochizuki M and
Terashima T: Effect of theanine, r-glutamyl ethyl amide, on brain
monoamines and striatal dopamine release in conscious rats.
Neurochem Res. 23:667–673. 1998.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Yao J, Shen XN, Shen H and Wu M: Effects
of theanine on monoamine neurotransmitters and related genes in
cerebral ischemia-reperfusion injury rats. Zhonghua Yu Fang Yi Xue
Za Zhi. 46:635–639. 2012.PubMed/NCBI(In Chinese).
|
|
117
|
Lee Y, Moon SJ and Montell C: Multiple
gustatory receptors required for the caffeine response in
Drosophila. Proc Natl Acad Sci USA. 106:4495–500. 2009.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Wright GA, Baker DD, Palmer MJ, Stabler D,
Mustard JA, Power EF, Borland AM and Stevenson PC: Caffeine in
floral nectar enhances a pollinator's memory of reward. Science.
339:1202–1204. 2013.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Fredholm BB, Bättig K, Holmén J, Nehlig A
and Zvartau EE: Actions of caffeine in the brain with special
reference to factors that contribute to its widespread use.
Pharmacol Rev. 51:83–133. 1999.PubMed/NCBI
|
|
120
|
Barone JJ and Roberts HR: Caffeine
consumption. Food Chem Toxicol. 34:119–129. 1996.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Mitchell DC, Hockenberry J, Teplansky R
and Hartman TJ: Assessing dietary exposure to caffeine from
beverages in the U.S. population using brand-specific versus
category-specific caffeine values. Food Chem Toxicol. 80:247–252.
2015.PubMed/NCBI View Article : Google Scholar
|