1. Introduction
Sepsis. Sepsis is a severe condition
characterized by a dysregulated host inflammatory response to
infection, resulting in organ damage. The latest Global Burden of
Disease 2021 analysis estimated 166 million cases of sepsis
worldwide in 2021 and ~21.4 million sepsis-related deaths,
collectively accounting for almost 31.5% of the total global
mortality (1). A constant decline
in sepsis-related mortality between 1990 and 2019 was followed by a
sharp trend reversal in 2020 and 2021 globally, which was
significantly attributed to the coronavirus disease 2019 (COVID-19)
pandemic and increased susceptibility in elderly populations. The
incidence of sepsis increased by 230% and mortality increased by
26.3% since 1990 among adults aged ≥15 years, with the highest
mortality rates being reported among the oldest age group (≥70
years; 9.28 million deaths in 2021). Although the incidence of
sepsis-related deaths from infectious conditions, such as diarrheal
diseases, tuberculosis, measles and lower respiratory infections
has considerably decreased over the past three decades, fatalities
related to non-infectious underlying conditions such as stroke,
chronic obstructive pulmonary disease, cirrhosis, and ischemic
heart disease have increased, highlighting the shift in sepsis
epidemiology toward complications of chronic diseases (1). A previous study found that children
aged <5 years constituted 26.4% (2.9 million) of the global
sepsis death toll and 41.5% (20.3 million) of the cases of sepsis.
The incidence of sepsis was lower among children and the younger
population aged 5-19 years, accounting for 10% (4.9 million) and
4.1% (0.45 million associated deaths), respectively. Adults aged
≥20 years accounted for the majority of incident cases of sepsis,
representing 48.5% (23.7 million), and for 70% (7.7 million) of
associated deaths. Men had a higher sepsis-related mortality rate
than women (164 vs. 134 per 100,000, respectively), whereas women
had a higher incidence of sepsis globally at 717 vs. 643 cases per
100,000(2). Moreover, significant
geographical and economic disparities have been reported, with high
rates in low- and middle-income countries or countries with an
intermediate sociodemographic index, such as those located in
sub-Saharan Africa and South-East Asia (3). The frequency of sepsis greatly varies
by location. The aforementioned scenario supports the view that the
incidence of sepsis has decreased in children and is no longer
mainly driven by infections; however, the overall global burden of
sepsis is high and mostly driven by aging populations, chronic
diseases, and new infections such as COVID-19(1). It poses a major challenge globally,
owing to its increasing incidence and high mortality rates, and
also poses a tremendous financial burden on healthcare systems. The
higher rates in low- and middle-income countries are attributable
to inferior medical facilities that are devoid of the necessary
tools and resources required for the diagnosis, prevention and
treatment of sepsis (Fig. 1).
Furthermore, age is a critical determinant wherein the
proportionate mortality from sepsis is the highest in neonates,
declines during middle adulthood, and then re-increases in the
older age groups (3).
Curcumin, a principal constituent of turmeric
rhizomes, demonstrates a broad spectrum of physiological and
pharmacological properties, and has long been used in traditional
medicine. Although turmeric has been used for thousands of years
for its medicinal benefits, research related to its precise
mechanisms of action and elucidation of its bioactive components is
relatively recent (4). Curcumin is
used to treat various chronic diseases owing to its potent
anti-inflammatory and antioxidant properties.
For the purposes of the present review, search
engines, including Scopus and PubMed, were used to identify
relevant literature. Original research and review articles with key
words such as curcumin, sepsis, hyperinflammation, cytokine storm,
anti-inflammatory, oxidative stress, clinical translation and organ
protection have been included in the present review. Only studies
published in the English language were considered for this review.
All identified publications were assessed, but only those that were
relevant to curcumin and its potential role in mitigating sepsis
were included.
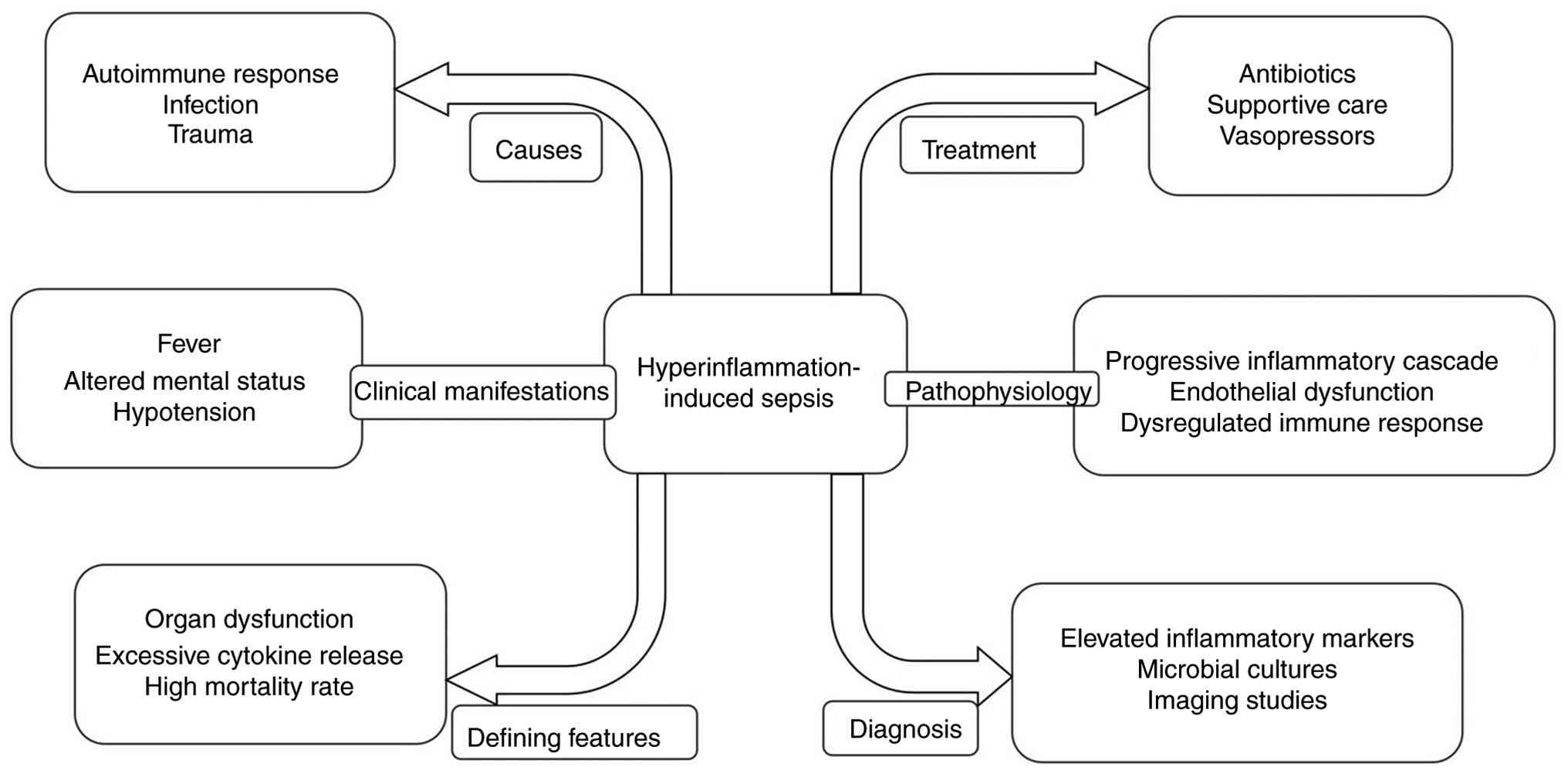
Pathophysiology of
hyperinflammation-induced sepsis
Sepsis represents a dynamic immune dysregulation,
where an initial hyperinflammatory phase driven by excessive
cytokine release leads to tissue injury and organ dysfunction. This
is followed by a dysfunctional immunosuppressive phase
characterized by lymphocyte apoptosis and impaired immune
responses, increasing the propensity to secondary infections and
poor clinical outcomes (5).
The clinical manifestations of sepsis include fever,
tachycardia, tachypnea and organ dysfunction. These processes are
driven by a state of hyperinflammation, which involves complex
interactions between immune cells and vascular cells, including
leukocytes, cytokines, reactive oxygen species (ROS), endothelial
cells, complement and the coagulation system (Fig. 2). An imbalance in the immune
response of the host during sepsis is the core of persistent
inflammation. Prolonged exposure to pathogen-associated molecular
patterns and damage-associated molecular patterns may stimulate a
cascade of events, leading to leukocyte infiltration and the
activation of the endothelium and the complement system. The
accelerated synthesis and the secretion of acute-phase proteins and
pro-inflammatory cytokines in the early stages of the progression
of sepsis reinforce the immune response further into a cycle of
self-sustaining inflammation. The participation of interleukin
(IL)-17 by T-helper and innate lymphoid cells increases the release
of tumor necrosis factor (TNF)-a and IL-1β, potentiating the
inflammatory response (5,6). High-mobility group box 1 is a
molecule that functions both as a cytokine and damage-associated
molecular pattern. It is highly upregulated in sepsis and mediates
inflammation via the activation of multiple pathogen recognition
receptors (PRRs) (8). As it
exhibits promising therapeutic potential, its clinical evaluation
is in the preclinical stages.
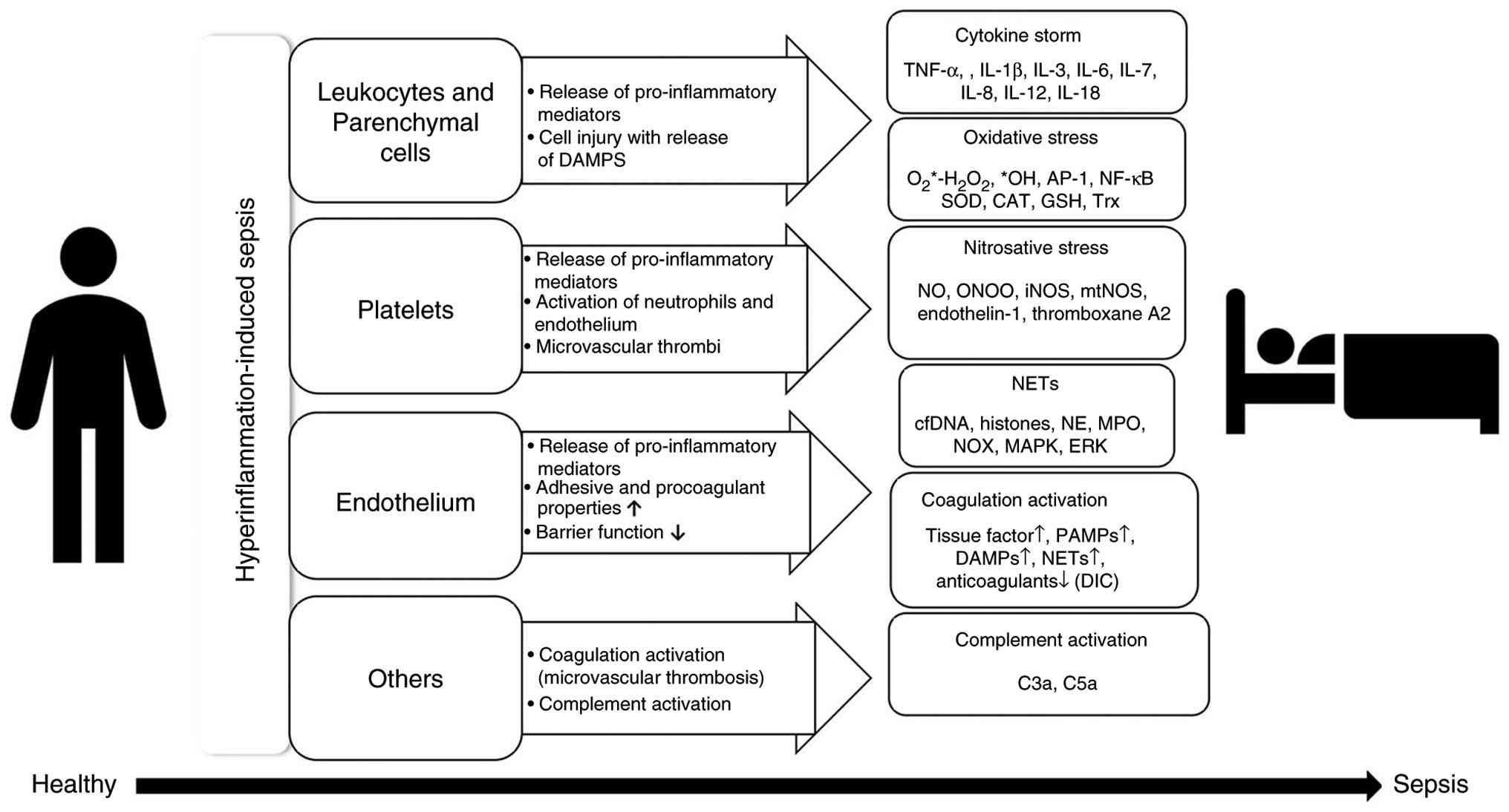 | Figure 2Hyperinflammation-induced sepsis
pathophysiology. The image depicts the series of cellular and
molecular events occurring during hyperinflammation-induced sepsis
and highlights the involvement of leukocytes, platelets, and
endothelial cells in the cascade of reactions that include cytokine
release, oxidative and nitrosative stress, NET formation,
complement and coagulation activation. DAMPS, damage-associated
molecular patterns; NET, neutrophil extracellular trap; SOD,
superoxide dismutase; CAT, catalase; GSH, glutathione; NO, nitric
oxide; iNOS, inducible nitric oxide synthase; mtNOS, mitochondrial
nitric oxide synthase; cfDNA, cell-free DNA; NE, neutrophil
elastase; MPO, myeloperoxidase; NOX, NADPH oxidase. |
Neutrophils constitute the first line of defense in
the immune system. A recent study found that a population of
elderly neutrophils may effectively deliver antigens to T-cells,
which, in turn, trigger interferon (IFN)-γ production (9). Neutrophils form neutrophil
extracellular traps (NETs) consisting of DNA, histones,
myeloperoxidase and elastase, which help neutralize pathogens. NETs
are generated by neutrophils upon stimulation by bacteria, viruses,
or cancer cells (10,11), and they can occur as
self-destructive or vital processes (12). Although extracellular traps are
also produced by macrophages, their role in sepsis remains unclear
(13). The uncontrolled formation
and improper clearance of NETs shift their function from tissue
protection to tissue damage, causing hyperinflammation and
thrombosis (10,14).
The complement system is vital in the innate immune
response (15) and is activated
via the following three pathways: The classical pathway initiated
by C1 binding to antibodies, an alternative pathway through the
activation of the hydrolysed C3 on microbial surfaces, and the
lectin pathway, wherein the mannose-binding lectin binds to
pathogen carbohydrates. The secretion of chemotactic agents, such
as C3a and C5a, leads to the recruitment of leukocytes and changes
in vascular flow, permeability and adhesion. Moreover, the terminal
complement complex facilitated by the complement system causes
bacterial lysis (16). During
sepsis, the activation of the complement contributes to
hyperinflammation (17), and
increased C5a levels are associated with worse outcomes (18). Although preclinical studies have
shown promise, complement-targeted therapies, including C5a
inhibitors, have not yet been translated to clinical use for sepsis
(19).
In sepsis, the activation of the coagulation system
is also very common, ranging from mild activation to disseminated
intravascular coagulation (DIC) (20). DIC may lead to defective hemostasis
due to the consumption of clotting factors and platelets (21). The term immunothrombosis describes
a coordinated association between the immune and coagulation
systems to trap pathogens through a combination of fibrin,
neutrophils, monocytes and platelets (22). However, it may become dangerous
when microvascular thrombosis is uncontrolled and could lead to
hypoxia and organ failure (23).
NETs engage the coagulation cascade via factor XII and
transcription factor and cleave antithrombotic proteins, while
further activating platelets along with cell-free DNA and histones,
supporting fibrin generation and promoting the amplification and
further generation of NETs (24,25).
However, high levels of NETs, as noted in the case of severe
sepsis, can lead to tissue deterioration and a hypercoagulant
response, causing complications such as DIC, thrombosis and organ
failure (10,14).
The complement and coagulation systems are also
functionally redundant and function synergistically. Complement
factors facilitate the expression of tissue factor on leukocytes,
activate platelets and release von Willebrand factor from
endothelial cells (26). This
relationship was established using a primate model of
Escherichia coli-induced sepsis, where the administration of
compstatin, a C3 inhibitor, significantly reduced microvascular
thrombosis (27). Pyroptosis is a
pro-inflammatory form of programmed cell death that is also
connected with immunothrombosis. Pyroptosis is activated by PRRs,
and it triggers the formation of the inflammasome and the cleavage
of gasdermin D, which forms pore-releasing inflammatory cytokines
and tissue factors (28).
Understanding these interactions will help further identify novel
therapeutic strategies that would help reduce the severity of
hyperinflammation-related complications in sepsis.
The current management of sepsis includes
controlling the underlying infection, stabilizing hemodynamic
factors and modulating the host immune response. Significant
challenges still exist despite the success of antibiotics in
treating sepsis. These include the development of antibiotic
resistance when several antibiotics are used incorrectly, further
complicating the treatment (29).
Antibiotics have a negative effect on the beneficial microflora,
which may lead to diarrhea and the occurrence of secondary
infections (30). Moreover,
treatment outcomes achieved using immunomodulators, including
hydrocortisone, are not always favorable (31,32).
Several side-effects have been reported in response to vasopressin
(33).
2. Curcumin
Curcumin
[(1E,6E)-1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione]
is the major polyphenol present in the turmeric plant (Curcuma
longa), which is one of the oldest known medicinal spices.
Curcumin is also present in other Curcuma species, including
Curcuma domestica (34),
Curcuma aromatica and Curcuma xanthorrhiza (35). Curcuma longa, a rhizome from
the Zingiberaceae family, is largely cultivated in India (36,37).
Upon consumption, curcumin is biotransformed into major biliary
metabolites, including dihydrocurcumin, tetrahydrocurcumin and
hexahydrocurcumin, which are further metabolized to monoglucuronide
conjugates (37-39).
Curcumin exerts several biological and pharmacological effects,
including antispasmodic (37),
anticancer, antibacterial and antirheumatic effects (36,40).
Curcumin also shows benefits in treating anxiety, arthritis,
metabolic syndrome, inflammatory diseases and hyperlipidemia.
Furthermore, curcumin may control post-exercise inflammation and
muscular pain, enhancing recuperation and, in turn, performance in
athletes (41). Chronic
inflammatory diseases, such as rheumatism, atherosclerosis, type II
diabetes, Alzheimer's disease and inflammatory bowel disease have
also been treated or managed with curcumin (42-44).
Curcumin exerts these health benefits by modulating various
signaling pathways and altering gene expression. It is a partial
inhibitor of protein kinase and is known to affect the activity of
protein kinase C (45), protein
tyrosine kinase (46),
cyclooxygenase (COX)-1 and COX-2 (43,47),
inhibiting lipoxygenase, TNF-α, IFN-γ, inducible nitric oxide
synthase and transcriptional nuclear factor κB (NF-κB) (43). Curcumin is also a potent ROS
scavenger that protects hemoglobin from nitrite-induced oxidation
to methemoglobin and is also known to inhibit lipid peroxidation
(48). Treatment with curcumin can
increase fibronectin and collagen expression, and it is accompanied
by the infiltration of numerous cells such as macrophages,
neutrophils and fibroblasts (49).
The presence of myofibroblasts facilitates more rapid wound
contraction (48). The
pharmacological derivatives of curcumin, its molecular weight and
formula are presented in Table
SI.
Chemistry of curcumin
Curcuminoids constitute ~1-6% of the dry weight of
turmeric. The three major curcuminoids in turmeric are curcumin
(60-70% of the crude extract), demethoxycurcumin (20-27%) and
bisdemethoxycurcumin (10-15%) (50,51).
Curcumin has been extensively studied by researchers in recent
times owing to its attractive characteristics, particularly in
biological applications. It is considered a good lead molecule in
drug discovery programs. However, its poor pharmacokinetic and
pharmacodynamic properties restrict its progression to a drug
candidate (52). It is water
insoluble at room temperature and has a neutral pH (Log P-value,
2.3-3.2); however, it is soluble in solvents such as acetone,
methanol and ethanol. Moreover, as it forms phenolates at an
alkaline pH, it degrades rapidly in neutral and alkaline conditions
via solvolysis and oxidative degradation pathways, thereby not
fulfilling the basic stability requirement under physiological
conditions. Its chemical instability is the major challenge posed
during its development, as it undergoes keto-enol tautomerism due
to the presence of a β-diketone moiety. This instability restricts
the reproducibility of in vivo and in vitro findings
and further constrains computational models. The chemical
instability of curcumin is also responsible for its poor
bioavailability (<1%), which further affects studies on the
Absorption, Distribution, Metabolism, Excretion, and Toxicity of
drug candidates. Several approaches are currently being explored,
particularly formulation development, nanoformulation approaches
and the use of other additives (e.g., piperine, a known
bioavailability enhancer) to improve the stability of curcumin
under physiological conditions and enhance bioavailability
(53-55).
Moreover, curcumin can combine with several biopolymers to enhance
wound healing (56). The
incorporation of curcumin into nanocarrier systems can enhance its
penetration into tissues (57).
Its structural components, including a diketone moiety and two
phenolic groups, facilitate key reactions including hydrogen
donation, nucleophilic addition and hydrolysis. These reactions
underpin the diverse biological applications of curcumin, including
its potent ROS-scavenging activity, anti-inflammatory effects and
immunomodulatory properties, which constitute core mechanisms
supporting its potential use in the treatment of sepsis (31,36,51).
3. Pharmacological potential of curcumin in
sepsis
The phenolic compound, curcumin, exerts a range of
pharmacological effects, including ameliorating hyperinflammation,
modulating the immune response and scavenging free radicals
(Fig. 3). Curcumin attenuates
cytokine storms, excessive inflammation and acute respiratory
distress syndrome both in vitro and in vivo (32,58).
Furthermore, curcumin exerts an anti-inflammatory effect by
inhibiting ROS formation and normalizing cytokine secretion to
block the oxidation pathway. It also prevents the production of
inflammatory cytokines and oxidative stress-related proteins,
thereby improving the survival rate and reducing alveolar
exudation, degeneration and necrotic cell death (58-62).
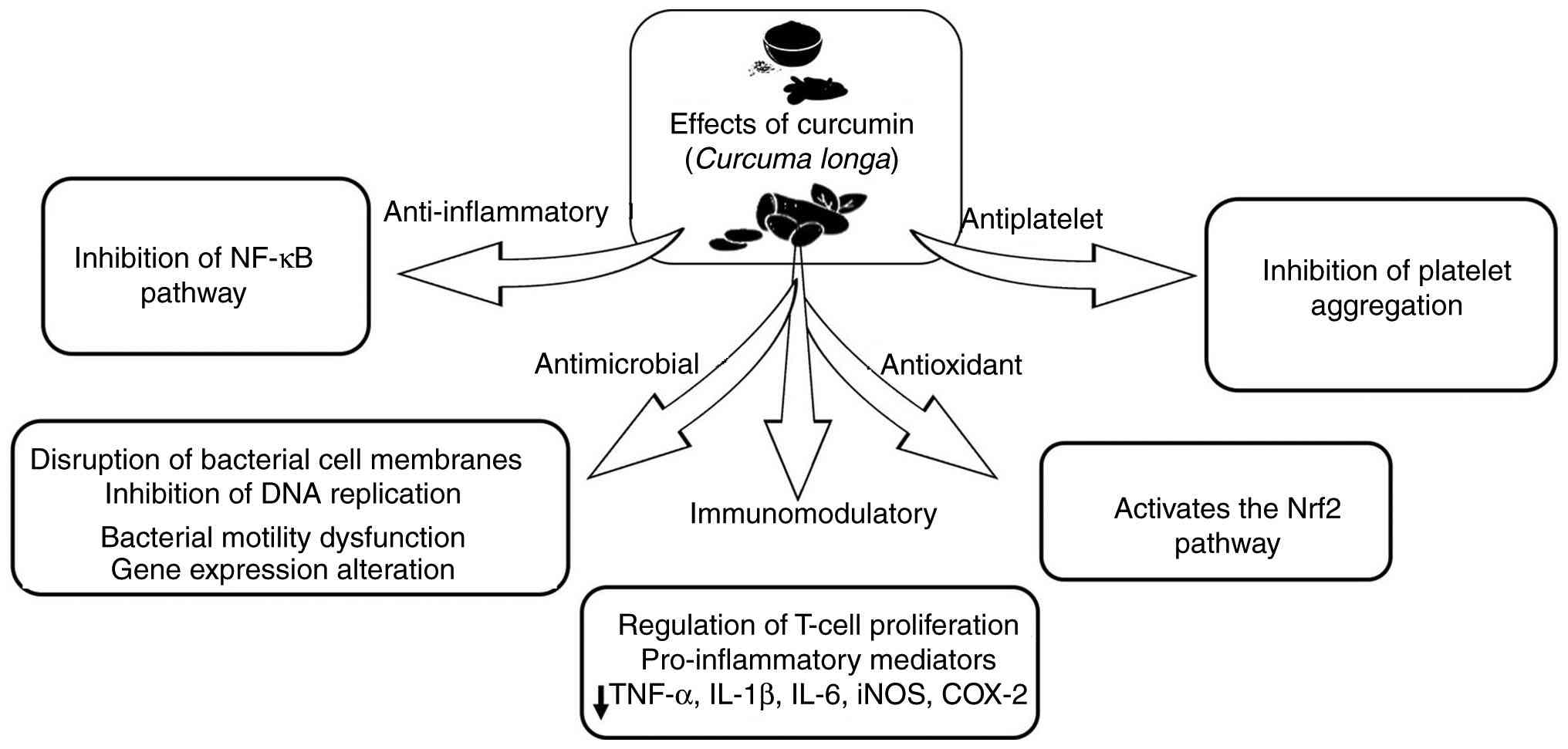 | Figure 3Multifaceted effects of curcumin. The
image depicts the biological activities of curcumin, including its
anti-inflammatory, antioxidant, antimicrobial, antiplatelet, and
immunomodulatory effects, all of which are considered pleiotropic.
The synergistic effect of the inhibition of the NF-κB pathway,
activation of the Nrf2 pathway, modulation of the gut microbiota,
and pro-inflammation mediator suppression by curcumin is shown.
Nrf2, nuclear factor erythroid 2-related factor 2; iNOS, inducible
nitric oxide synthase; COX-2, cyclooxygenase 2. |
The antioxidant activity of curcumin is due to its
ability as a free radical scavenger in reducing oxidative damage
(60). The pleiotropic nature of
curcumin allows it to modulate multiple signaling pathways,
rendering it a potent candidate for treating a wide array of
chronic conditions. Experimental evidence from various in
vivo and in vitro disease models ranging from metabolic
disorders to neurodegenerative conditions demonstrates its
versatile therapeutic efficacy (62-75).
A comprehensive summary of these therapeutic effects across
different disease models and the pharmacological potential of
curcumin is presented Table
SII.
Curcumin exerts antimicrobial effects against
several pathogens incriminated in sepsis by disrupting bacterial
cell membranes, inhibiting DNA replication, inducing bacterial
motility dysfunction, and altering gene expression (76,77).
Curcumin has shown activity against Staphylococcus aureus,
Escherichia coli, Salmonella paratyphi, Toxoplasma
gondii, Bacillus subtilis, Paenibacillus
macerans, B. licheniformis and Azotobacter
(78) and also against 20
Candida species (79,80).
Curcumin nanoformulations have been shown to be more effective
against Escherichia coli, Staphylococcus aureus, and
Pseudomonas aeruginosa, maintaining activity even following
1 month of storage and exhibiting no toxicity, in contrast to
antibiotics such as chloramphenicol and gentamicin (76,78).
The broad-spectrum antibacterial potential of curcumin has been
extensively documented in recent literature (79,80).
Specifically, its differential efficacy against various bacterial
cell wall structures has been a primary focus of investigation. A
comprehensive summary of the inhibitory effects of curcumin and its
derivatives against a wide range of Gram-negative and Gram-positive
bacteria (29,81-90)
is provided in Table SIII.
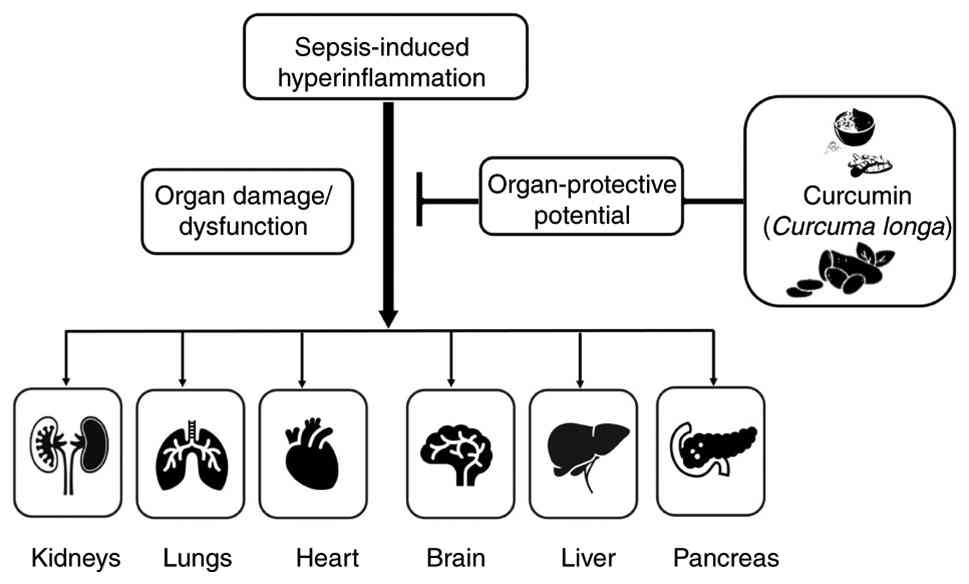
4. Organ-protective potential of
curcumin
Sepsis is a lethal condition that affects one or
multiple organs due to hyperinflammation. It is characterized by
the overactivation of the host response to an infection, which
triggers a complex process through hyperimmune activation,
dysregulated immune responses exaggerated ROS production, and
finally, NET formation. The factors acting in an integrated manner
lead to severe tissue damage and organ failure (36,40),
with the lungs, liver, heart, brain and kidneys being the major
organs affected due to sepsis (Fig.
4).
Curcumin reduces the initial inflammatory cell
infiltration in various organs and tissues, including the lungs,
liver, kidneys, brain, heart, spleen and intestines (66,91-109).
Curcumin has demonstrated outstanding protective benefits against
cardiac ischemia and reperfusion, hyperuricemia and renal
endothelial dysfunction, as it inhibits the Janus kinase
(JNK)2/signal transducer and activator of transcription 3 pathway
(91) and the extracellular
signal-regulated kinase/plasmacytoma variant translocation 1/c-Jun
N-terminal kinase/NF-kB pathway (92). Curcumin can protect against renal
ischemia-reperfusion injury-induced acute kidney injury by
upregulating the expression of DCC-interacting protein 13-alpha,
which additionally blocks the Akt signaling pathway (93). The anti-apoptotic effects of
tetrahydrocurcumin (THC) significantly decreased the expression of
Bax and cleaved caspase-3 and increased that of Bcl2, which further
decreased the development of diabetic cardiomyopathy by reducing
oxidative stress and fibrosis by stimulating the induction of the
SIRT1-DRP1/PGC-1α signaling pathway. Some key markers, including
serum creatinine, blood urea nitrogen, kidney injury molecule-1 and
urine microalbumin/creatinine, were used to assess the decline in
renal function (94).
Curcumin has been reported to decrease inflammation;
normalize the levels of hepatic enzymes such as alkaline
phosphatase, aspartate transaminase and alanine transaminase
(66,95); attenuate hepatocyte damage
(96,97) in in vitro models; and
decrease the extent of cell degeneration and necrosis in an in
vivo model of lipopolysaccharide-induced sepsis (98). Treatment with curcumin was shown to
reduce histological damage, inflammation, degeneration and necrosis
in the glomeruli and renal tubules of the kidneys and increase the
survival rate by up to 90% in a rat model of cecal
ligation/puncture (CLP)-induced sepsis (96-98).
Moreover, curcumin can suppress the levels of pro-inflammatory
cytokines (IL-1β and IL-6) and increase the expression of the
anti-inflammatory cytokine IL-10, thereby attenuating liver
dysfunction in an in vivo model of CLP-induced and
lipopolysaccharide (LPS)-induced endotoxemia model of sepsis
(98,99). Furthermore, curcumin plays a
crucial role in attenuating the expression of various proteins,
including inhibitory κB kinase β, inhibitor κBα, phosphorylated
NF-κB, TNF-α, IL-1β and IL-18, in a mouse model of LPS-induced
acute liver failure and sepsis (66,97).
It was also found to exert a cytoprotective effect on hepatic
microvascular inflammatory responses in endotoxemia in an in
vivo model by inhibiting Kupffer cell activation, reducing the
adhesion of neutrophils, and controlling endothelial cell edema
(66,100). Furthermore, in an in vitro
model, some of the hydrogenated metabolites of curcumin, such as
THC and octahydrocurcumin, demonstrated hepatoprotective effects
against acetaminophen-induced hepatic injury (101). Another study reported the
neuroprotective and neurotrophic effects of the curcumin analog,
J147, both in vivo and in vitro. J147 improves memory
and prevents depressive-like behavior by modulating
neuroinflammation by suppressing the TLR4/NF-κB signaling pathway
in the microglia of mice with sepsis. Pre-treatment with J147
significantly reduced the levels of IL-6, IL-1β, TNF-α and ionized
calcium-binding adapter protein 1 in microglia (102). Additionally, curcumin was found
to attenuate the activation of transcription factors, including
NF-κB and activator protein 1, in an in vivo model, thereby
alleviating hemorrhage (103).
Curcumin can also suppress hypoxia-induced mRNA synthesis and the
protein levels of hypoxia-inducible factor 1a, interfering with the
secretion of vascular endothelial growth factor A in GH3 cells
(104). Additionally, curcumin
reduces the blood-brain barrier impairment, decreases the severity
of edema and apoptosis, and minimizes mitochondrial damage in the
brains of mice with sepsis (105).
L48H37 (1-ethyl-3,5-bis(3,4,5-trimethoxybenzylidene)
piperidin-4-one) is a selective autophagy inhibitor and an analog
of curcumin that can suppress LPS-induced inflammation by reducing
TNF-α and IL-6 production in mouse macrophages, thereby improving
survival and protecting lung injury in LPS-induced mice with sepsis
(106).
Curcumin also mitigates cardiac dysfunction
associated with sepsis. It can reduce the decline of cardiac
contractility in sepsis, attenuate cardiac inflammation, and
decrease the extent of structural damage to myocardial cells in a
rat model of CLP surgery-induced sepsis. Curcumin intervention can
significantly decrease the levels of the cardiac injury markers,
troponin and malondialdehyde, while restoring superoxide dismutase
activity in the plasma of rats with sepsis. It also induces
contractility and decreases inflammation and structural damage in
the heart. Apart from these effects, it decreases the extent of
myocardial inflammation while attenuating structural injury to
cardiomyocytes (107). It also
alleviates LPS-induced cardiac dysfunction in an LPS-induced mouse
model of sepsis by attenuating oxidative stress and inflammation by
regulating the JNK/ERK signaling pathway (108).
Multiple organ dysfunction syndrome caused by sepsis
involves sepsis-induced myocardial dysfunction as a crucial
component. In the CLP-induced model of sepsis, treatment with free
curcumin and nanocurcumin was found to preserve the structure of
the mitochondria and cardiac myofibrils, decrease severe
sepsis-induced cardiac lesions, and alter the components of the
mTOR pathway, including the mechanistic target of rapamycin complex
(mTORC)1, Raptor, mTORC2 and Rictor in the hearts of mice with
sepsis (109). Beyond its
antimicrobial properties, curcumin exhibits significant multi-organ
protective effects across various physiological systems. Current
research has highlighted its efficacy in mitigating oxidative
stress and inflammatory damage in the liver, kidneys, heart and
lungs (66,110-125).
A detailed summary of these organ-protective mechanisms and the
specific experimental models used is provided in Table SIV.
5. Limitations of curcumin as a therapeutic
agent
In addition to its numerous benefits, curcumin also
has some drawbacks, such as chemical instability, poor water
solubility, rapid clearance and poor absorption, which limit its
clinical applications (38). While
there is encouraging evidence highlighting the curative effects of
curcumin in sepsis-induced complications, the poor bioavailability
of curcumin is a key obstacle to the clinical evolution of this
captivating chemical, notwithstanding the facts described above
that suggest its rational and useful implementation in treating
different complications. Curcumin is highly distributed in tissues
and undergoes rapid metabolism. The hydrophobicity of curcumin is
responsible for its poor bioavailability. Following oral intake,
curcumin undergoes conjugation in the liver and intestinal walls
and is metabolized into curcumin glucuronide and sulfates (38,126). Several options are currently
being explored to enhance the bioavailability of curcumin. One such
approach is the use of the bioavailability enhancer piperine from
black pepper (38,126,127), an inhibitor of intestinal and
hepatic glucuronidation, which increases the bioavailability of
curcumin both in animals and humans when co-administered with
curcumin. Initiatives such as the design of curcumin-phospholipid
complexes, liposomal curcumin and curcumin nanoparticles are
attempts that could improve the bioavailability of curcumin
(38,128). In the event that the
bioavailability of curcumin is successfully enhanced without
compromising safety in humans, this naturally occurring polyphenol
could be elevated to the forefront of therapeutic medicine to treat
a range of illnesses, including malaria (128,129).
Translational challenges related to
curcumin
The clinical use of curcumin, which is supported by
robust preclinical evidence of its multifaceted properties, still
faces some challenges in addition to its poor oral bioavailability.
One of the primary challenges is the chemical instability of
curcumin and its rapid degradation in the body (e.g., pH-dependent
breakdown, sensitivity to light), which reduces its therapeutic
availability and complicates formulation stability and storage,
thereby limiting the reproducibility of in vivo results
(130).
Another challenge is the complexity of the
pharmacodynamics of curcumin and its biological activity, which has
raised concerns about assay interference and nonspecific
interactions and, consequently, has categorized this compound as a
pan-assay interference compound. Such complexity may not only
hinder mechanistic studies, but may also reduce confidence in
distinguishing true target engagement, thereby undermining the
translational validity of the research (131).
Furthermore, variability in preclinical and clinical
trial design (variable dosing regimens, lack of standardized
formulations, inconsistent endpoints, and limited statistical
power, making direct comparison across studies difficult) has been
a reason for mixed or inconclusive results pertaining to its
efficacy. Moreover, the interaction of curcumin with
drug-metabolizing enzymes and transporters (e.g., inhibition of
cytochrome P450 and P-glycoprotein) adds to the problems of
polypharmacy, as it may alter the pharmacokinetics of concurrently
administered drugs, thereby complicating dose optimization and
safety in patients taking multiple medications (132).
6. Conclusions and future perspectives
Sepsis is a severe, life-threatening condition
resulting from an overwhelming host response to an infection. It
continues to pose a challenge in critical care due to its high
morbidity and mortality rates. Sepsis is associated with systemic
inflammation, immune dysregulation, and diffuse organ malfunction
and requires immediate and appropriate therapeutic interventions.
Curcumin has been considered as a supplement in the management of
sepsis owing to its anti-inflammatory, antioxidant and
immune-modulating effects. As aforementioned, preclinical and early
clinical studies have shown that curcumin influences the key
pathways of the sepsis cascade by inhibiting proinflammatory
cytokines, reducing oxidative stress, and modulating immune
responses. Given its multifaceted effects, curcumin may reduce the
severity of sepsis, improve outcomes, and reduce serious
complications such as organ failure. These results, although
encouraging, warrant further studies in terms of their activity and
safety profiles to define their optimal role in the management of
sepsis, especially in larger clinical trials. In-depth studies are
therefore warranted to fully elucidate the therapeutic potential of
curcumin in the management of sepsis. Optimization of the
bioavailability and delivery methods for curcumin should be the
primary focus, as poor absorption and rapid metabolism limit its
clinical applications. Improvements in nanoparticle-based delivery
systems or formulation advancements may enhance the potency of
curcumin. Clinical trials of curcumin may be necessary to define
its optimal dosage and duration of therapy and to identify possible
interactions with standard sepsis therapies. Furthermore, the
synergistic effect of curcumin can be increased when used
concurrently with other therapeutic agents, thereby providing a
more effective and multidimensional treatment approach. The
therapeutic incorporation of curcumin in sepsis management will
depend on a deeper understanding of its pharmacokinetics and
mechanisms of action, ultimately enabling clinical translation and
fostering innovative strategies for more effective treatment.
Supplementary Material
Curcumin-based metabolites.
Curcumin and its therapeutic effects
in various disease models.
Effects of curcumin on Gram-negative
and Gram-positive bacteria.
yH2AX
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
GA conceptualized the study, searched the
literature, and wrote the original draft of the manuscript. SD and
AK prepared the tables and figures, and were involved in the
literature search. JS, ER and PKV wrote a section of the manuscript
and edited the manuscript. AJ conceptualized and supervised the
study and edited the manuscript. All authors have read and approved
the final version of the manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
GBD 2021 Global Sepsis Collaborators.
Global, Regional, and national sepsis incidence and mortality,
1990-2021: A systematic analysis. Lancet Global Health.
13:e2013–e2026. 2025.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gottlieb M, Wusterbarth E, Hlavin R,
Bernard K and Moyer E: Epidemiology of sepsis presentations and
management among United States emergency departments from 2016 to
2023. Acad Emerg Med. 32:467–470. 2025.PubMed/NCBI View Article : Google Scholar
|
|
3
|
La Via L, Sangiorgio G, Stefani S, Marino
A, Nunnari G, Cocuzza S, La Mantia I, Cacopardo B, Stracquadanio S,
Spampinato S, et al: The global burden of sepsis and septic shock.
Epidemiologia (Basel). 5:456–478. 2024.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Guo Y, An B, Lang Z, Zhou F, Zhang X and
Wang H: Effects of curcumin on inhibiting the proliferation of
pulmonary artery smooth muscle cells and relieving pulmonary
arterial hypertension. Farmacia. 68:307–312. 2020.
|
|
5
|
Vella R, Panci D, Carini F, Malta G, Vieni
S, David S, Albano GD, Puntarello M, Zerbo S and Argo A: Cytokines
in sepsis: A critical review of the literature on systemic
inflammation and multiple organ dysfunction. Front Immunol.
16(1682306)2025.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Li LL, Dai B, Sun YH and Zhang TT: The
activation of IL-17 signaling pathway promotes pyroptosis in
pneumonia-induced sepsis. Ann Transl Med. 8(674)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Sherwood ER, Burelbach KR, McBride MA,
Stothers CL, Owen AM, Hernandez A, Patil NK, Williams DL and
Bohannon JK: Innate immune memory and the host response to
infection. J Immunol. 208:785–792. 2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Chan JK, Roth J, Oppenheim JJ, Tracey KJ,
Vogl T and Feldmann M: Alarmins: Awaiting a clinical response. J
Clin Invest. 122:2711–2719. 2012.PubMed/NCBI View
Article : Google Scholar
|
|
9
|
Jin H, Aziz M, Murao A, Kobritz M, Shih AJ
and Adelson RP: Antigen-presenting aged neutrophils induce CD4+ T
cells to exacerbate inflammation in sepsis. J Clin Invest.
133(e164585)2023.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chen Z, Zhang H, Qu M, Nan K, Cao H and
Cata JP: Review: The emerging role of neutrophil extracellular
traps in sepsis and Sepsis-associated thrombosis. Front Cell Infect
Microbiol. 11(653228)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Wang H, Kim SJ, Lei Y, Wang S, Wang H,
Huang H, Zhang H and Tsung A: Neutrophil extracellular traps in
homeostasis and disease. Sig Transduct Target Ther.
9(235)2024.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Denning NL, Aziz M, Gurien SD and Wang P:
Damps and nets in sepsis. Front Immunol. 10(2536)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Weng W, Hu Z and Pan Y: Macrophage
extracellular traps: Current opinions and the state of research
regarding various diseases. J Immunol Res.
2022(7050807)2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Delabranche X, Stiel L, Severac F, Galoisy
AC, Mauvieux L and Zobairi F: Evidence of netosis in septic
shock-induced disseminated intravascular coagulation. Shock.
47:313–317. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Sahu SK, Kulkarni DH, Ozanturk AN, Ma L
and Kulkarni HS: Emerging roles of the complement system in
host-pathogen interactions. Trends in Microbiol. 30:390–402.
2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Jayaraman A, Walachowski S and Bosmann M:
The complement system: A key player in the host response to
infections. Eur J Immunol. 54(2350814)2024.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Abe T, Kubo K, Izumoto S, Shimazu S, Goan
A and Tanaka T: Complement activation in human sepsis is related to
Sepsis-induced disseminated intravascular coagulation. Shock.
54:198–204. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Cavaillon JM: During sepsis and COVID-19,
the pro-inflammatory and anti-inflammatory responses are
concomitant. Clin Rev Allerg Immu. 65:183–187. 2023.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Sommerfeld O, Medyukhina A, Neugebauer S,
Ghait M, Ulferts S, Lupp A, König R, Wetzker R, Schulz S, Figge MT,
et al: Targeting complement C5a Receptor 1 for the treatment of
immunosuppression in sepsis. Mol Ther. 29:338–346. 2021.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Levi M and van der Poll T: Coagulation and
sepsis. Thromb Res. 149:38–44. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Iba T, Watanabe E, Umemura Y, Wada T,
Hayashida K and Kushimoto S: Japanese Surviving Sepsis Campaign
Guideline Working Group for disseminated intravascular coagulation.
Wada H: Sepsis-associated disseminated intravascular coagulation
and its differential diagnoses. J Intensive Care.
7(32)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Engelmann B and Massberg S: Thrombosis as
an intravascular effector of innate immunity. Nat Rev Immunol.
13:34–45. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Perdomo J and Leung HH: Immune thrombosis:
Exploring the significance of immune complexes and NETosis. Biology
(Basel). 12(1332)2023.PubMed/NCBI View Article : Google Scholar
|
|
24
|
de Stoppelaar SF, van't Veer C and van der
Poll T: The role of platelets in sepsis. Thromb Haemost.
112:666–677. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
McDonald B, Davis RP, Kim SJ, Tse M, Esmon
CT, Kolaczkowska E and Jenne CN: Platelets and neutrophil
extracellular traps collaborate to promote intravascular
coagulation during sepsis in mice. Blood. 129:1357–1367.
2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Keragala CB, Draxler DF, McQuilten ZK and
Medcalf RL: Haemostasis and innate immunity-a complementary
relationship: A review of the intricate relationship between
coagulation and complement pathways. Br J Haematol. 180:782–798.
2018.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Silasi-Mansat R, Zhu H, Popescu NI, Peer
G, Sfyroera G, Magotti P, Ivanciu L, Lupu C, Mollnes TE, Taylor FB,
et al: Complement inhibition decreases the procoagulant response
and confers organ protection in a baboon model of Escherichia
coli sepsis. Blood. 116:1002–1010. 2010.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Tsuchiya K: Inflammasome-associated cell
death: Pyroptosis, apoptosis, and physiological implications.
Microbiol Immunol. 64:252–269. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Bahari S, Zeighami H, Mirshahabi H,
Roudashti S and Haghi F: Inhibition of Pseudomonas aeruginosa
quorum sensing by subinhibitory concentrations of curcumin with
gentamicin and azithromycin. J Glob Antimicrob Resist. 10:21–28.
2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Newcomb D, Bolgos G, Green L and Remick
DG: Antibiotic treatment influences outcome in murine sepsis.
Shock. 10:110–117. 1998.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Allegra A, Mirabile G, Ettari R, Pioggia G
and Gangemi S: The impact of curcumin on immune response: An
immunomodulatory strategy to treat sepsis. Int J Mol Sci.
23(14710)2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Mimche PN, Taramelli D and Vivas L: The
plant-based immunomodulator curcumin as a potential candidate for
the development of an adjunctive therapy for cerebral malaria.
Malar J. 10 (Suppl 1)(S10)2011.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Jentzer JC, Coons JC, Link CB and
Schmidhofer M: Pharmacotherapy update on the use of vasopressors
and inotropes in the intensive care unit. J Cardiovasc Pharmacol
Ther. 20:249–260. 2015.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Anjusha S and Gangaprasad A: Phytochemical
and antibacterial analysis of two important curcuma species,
Curcuma aromatica salisb. and Curcuma xanthorrhiza
roxb. (Zingiberaceae). J Pharmacognosy Phytochemistry. 3:50–53.
2014.
|
|
35
|
Kuptniratsaikul V, Dajpratham P,
Taechaarpornkul W, Buntragulpoontawee M, Lukkanapichonchut P,
Chootip C, Saengsuwan J, Tantayakom K and Laongpech S: Efficacy and
safety of Curcuma domestica extracts compared with ibuprofen
in patients with knee osteoarthritis: A multicenter study. Clin
Interv Aging. 9:451–458. 2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Maheshwari RK, Singh AK, Gaddipati J and
Srimal RC: Multiple biological activities of curcumin: A short
review. Life Sci. 78:2081–2087. 2006.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Ammon H and Wahl M: Pharmacology of
Curcuma longa. Planta Med. 57:1–7. 1991.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Pan MH, Huang TM and Lin JK:
Biotransformation of curcumin through reduction and glucuronidation
in mice. Drug Metab Dispos. 27:486–494. 1999.PubMed/NCBI
|
|
39
|
Holder GM, Plummer JL and Ryan AJ: The
metabolism and excretion of curcumin
(1,7-bis-(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,s-dione) in
the rat. Xenobiotica. 8:761–768. 1978.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Basnet P and Skalko-Basnet N: Curcumin: An
anti-inflammatory molecule from a curry spice on the path to cancer
treatment. Molecules. 16:4567–4598. 2011.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Hewlings SJ and Kalman DS: Curcumin: A
review of its effects on human health. Foods. 6(92)2017.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Srivastava R, Dikshit M, Srimal RC and
Dhawan BN: Antithrombotic effect of curcumin. Thromb Res.
40:413–417. 1985.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Hanai H and Sugimoto K: Curcumin has
bright prospects for the treatment of inflammatory bowel disease.
Curr Pharm Des. 15:2087–2094. 2009.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wang J, Wang H, Zhu R, Liu Q, Fei J and
Wang S: Anti-inflammatory activity of curcumin-loaded solid lipid
nanoparticles in IL-1β transgenic mice subjected to the
lipopolysaccharide-induced sepsis. Biomaterials. 53:475–483.
2015.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Liu JY, Lin SJ and Lin JK: Inhibitory
effects of curcumin on protein kinase C activity induced by
12-0-tetradecanoyl-phorbol-13-acetate in NIH 3T3 cells.
Carcinogenesis. 14:857–861. 1993.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Chen H and Huang H: Effect of curcumin on
cell cycle progression and apoptosis in vascular smooth muscle
cells. Br J Pharmacol. 124:1029–1040. 1998.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Zhang F, Altorki NK, Mestre JR,
Subbaramaiah K and Dannenberg AJ: Curcumin inhibits
cyclooxygenase-2 transcription in bile acid- and phorbol
ester-treated human gastrointestinal epithelial cells.
Carcinogenesis. 20:445–451. 1999.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Reddy S and Aggarwal BB: Curcumin is a
non-competitive and selective inhibitor of phosphorylase kinase.
FEBS Lett. 341:19–22. 1994.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Sidhu GS, Singh AK, Thaloor D, Banaudha
KK, Patnaik GK, Srimal RC and Maheshwari RK: Enhancement of wound
healing by curcumin in animals. Wound Repair Regen. 6:167–177.
1998.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Niranjan A, Singh S, Dhiman M and Tewari
SK: Biochemical composition of Curcuma longa L. accessions.
Anal Lett. 46:1069–1083. 2012.
|
|
51
|
Priyadarsini K: The chemistry of curcumin:
From extraction to therapeutic agent. Molecules. 19:20091–20112.
2014.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Workman P and Collins I: Probing the
probes: Fitness factors for small molecule tools. Chem Biol.
17:561–577. 2010.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Nelson KM, Dahlin JL, Bisson J, Graham J,
Pauli GF and Walters MA: The essential medicinal chemistry of
curcumin. J Med Chem. 60:1620–1637. 2017.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Parcha V, Kumar P, Farswan M and Maithani
A: Individual and combined effect of aqueous extract of Gymnema
sylvestre, Tinospora cordifolia and Piper longum on carrageenan
induced inflamed rats. Indian Drugs. 47:65–67. 2010.
|
|
55
|
Barua N and Buragohain AK: Therapeutic
potential of curcumin as an antimycobacterial agent. Biomolecules.
11(1278)2021.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Thi Sinh Vo, Vo Tran Thi Bich Chau, Tran
Thi Thu Ngoc Vo and Thi Ngoc Huyen Lai: Turmeric (Curcuma
longa L.): Chemical components and their effective clinical
applications. JOTCSA Chemistry. 8:883–898. 2021.
|
|
57
|
Ahmad RS, Hussain MB, Sultan MT, Arshad M,
Waheed M, Shariati MA, Plygun S and Hashempur MH: Biochemistry,
safety, pharmacological activities, and clinical applications of
turmeric: A mechanistic review. Evid Based Complement Alternat Med.
2020(7656919)2020.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Peter AE, Sandeep BV, Rao BG and Kalpana
VL: Calming the storm: Natural immunosuppressants as adjuvants to
target the cytokine storm in COVID-19. Front Pharmacol.
11(583777)2021.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Xu F, Lin SH, Yang YZ, Guo R, Cao J and
Liu Q: The effect of curcumin on sepsis-induced acute lung injury
in a rat model through the inhibition of the TGF-β1/SMAD3 pathway.
Int Immunopharmacol. 16:1–6. 2013.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Vacek JC, Behera J, George AK, Kamat PK,
Kalani A and Tyagi N: Tetrahydrocurcumin ameliorates
homocysteine-mediated mitochondrial remodeling in brain endothelial
cells. J Cell Physiol. 233:3080–3092. 2018.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Hu W, Cai M, Qi D, Ying X, Huang C and
Xing C: β-Ionone-derived curcumin analogs as potent
anti-inflammatory agents. Pharm Chem J. 51:902–906. 2018.
|
|
62
|
Zhang Y, Liu Z, Wu J, Bai B, Chen H, Xiao
Z, Chen L, Zhao Y, Lum H, Wang Y, et al: New MD2 inhibitors derived
from curcumin with improved anti-inflammatory activity. Eur J Med
Chem. 148:291–305. 2018.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Wu Y, Liu Z, Wu W, Lin S, Zhang N, Wang H,
Tan S, Lin P, Chen X, Wu L and Xu J: Effects of FM0807, a novel
curcumin derivative, on lipopolysaccharide-induced inflammatory
factor release via the ROS/JNK/p53 pathway in RAW264.7 cells.
Biosci Rep. 38(BSR20180849)2018.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Liu W, Guo W, Zhu Y, Peng S, Zheng W,
Zhang C, Shao F, Zhu Y, Hang N, Kong L, et al: Targeting
peroxiredoxin 1 by a curcumin analogue, AI-44, inhibits NLRP3
inflammasome activation and attenuates lipopolysaccharide-induced
sepsis in mice. J Immunol. 201:2403–2413. 2018.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Ahn MY, Hwang JS, Lee SB, Ham SA, Hur J,
Kim JT and Seo HG: Curcuma longa extract-loaded nanoemulsion
improves the survival of endotoxemic mice by inhibiting nitric
oxide-dependent HMGB1 release. PeerJ. 5(e3808)2017.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Zhong W, Qian K, Xiong J, Ma K, Wang A and
Zou Y: Curcumin alleviates lipopolysaccharide-induced sepsis and
liver failure by suppression of oxidative stress-related
inflammation via PI3K/AKT and NF-κB related signaling. Biomed
Pharmacother. 83:302–313. 2016.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Rana M, Maurya P, Reddy SS, Singh V, Ahmad
H, Dwivedi AK, Dikshit M and Barthwal MK: A standardized chemically
modified Curcuma longa extract modulates IRAK-MAPK signaling
in inflammation and potentiates cytotoxicity. Front Pharmacol.
7(223)2016.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Tham CL, Lam KW, Rajajendram R, Cheah YK,
Sulaiman MR, Lajis NH, Kim MK and Israf DA: The effects of a
synthetic curcuminoid analogue,
2,6-bis-(4-hydroxyl-3-methoxybenzylidine)cyclohexanone on
proinflammatory signaling pathways and CLP-induced lethal sepsis in
mice. Eur J Pharmacol. 652:136–144. 2011.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Zhang Y, Liang D, Dong L, Ge X, Xu F, Chen
W, Dai Y, Li H, Zou P, Yang S and Liang G: Anti-inflammatory
effects of novel curcumin analogs in experimental acute lung
injury. Respir Res. 16(43)2015.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Gong Z, Zhou J, Li H, Gao Y, Xu C, Zhao S,
Chen Y, Cai W and Wu J: Curcumin suppresses NLRP3 inflammasome
activation and protects against LPS-induced septic shock. Mol Nutr
Food Res. 59:2132–2142. 2015.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Zhao C, Zhang Y, Zou P, Wang J, He W, Shi
D, Li H, Liang G and Yang S: Synthesis and biological evaluation of
a novel class of curcumin analogs as anti-inflammatory agents for
prevention and treatment of sepsis in mouse model. Drug Des Devel
Ther. 9:1663–1678. 2015.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Shukla P, Verma AK, Dewangan J, Rath SK
and Mishra PR: Chitosan coated curcumin nanocrystals augment
pharmacotherapy via improved pharmacokinetics and interplay of
NFκB, Keap1 and Nrf2 expression in Gram negative sepsis†. RSC Adv.
5:57006–57020. 2015.
|
|
73
|
Shukla P, Dwivedi P, Gupta PK and Mishra
PR: Optimization of novel tocopheryl acetate nanoemulsions for
parenteral delivery of curcumin for therapeutic intervention of
sepsis. Expert Opin Drug Deliv. 11:1697–1712. 2014.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Zhang Y, Jiang X, Peng K, Chen C, Fu L,
Wang Z, Feng J, Liu Z, Zhang H, Liang G and Pan Z: Discovery and
evaluation of novel anti-inflammatory derivatives of natural
bioactive curcumin. Drug Des Devel Ther. 8:2161–2171.
2014.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Wu J, Zhang Y, Cai Y, Wang J, Weng B, Tang
Q, Chen X, Pan Z, Liang G and Yang S: Discovery and evaluation of
piperid-4-one-containing mono-carbonyl analogs of curcumin as
anti-inflammatory agents. Bioorg Med Chem. 21:3058–3065.
2013.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Sharifi S, Fathi N, Memar MY, Hosseiniyan
Khatibi SM, Khalilov R, Negahdari R, Zununi Vahed S and Maleki
Dizaj S: Anti-microbial activity of curcumin nanoformulations: New
trends and future perspectives. Phytother Res. 34:1926–1946.
2020.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Ali Raza Naqvi S, Nadeem S, Komal S, Naqvi
A, Samee Mubarik M, Yaqub Qureshi S, Ahmad S, Zahid M, Khan
Naeem-Ul-Haq, Raza SS and Aslam N: Antioxidants: Natural
antibiotics. In: Antioxidants. IntechOpen, 2019.
|
|
78
|
Pandit RS, Gaikwad SC, Agarkar GA, Gade AK
and Rai M: Curcumin nanoparticles: Physico-chemical fabrication and
its in vitro efficacy against human pathogens. 3 Biotech.
5:991–997. 2015.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Martins CVB, da Silva DL, Neres ATM,
Magalhães TFF, Watanabe GA, Modolo LV, Sabino AA, de Fátima A and
de Resende MA: Curcumin as a promising antifungal of clinical
interest. J Antimicrob Chemother. 63:337–339. 2009.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Hettiarachchi SS, Perera Y, Dunuweera SP,
Dunuweera AN, Rajapakse S and Rajapakse RMG: Comparison of
antibacterial activity of nanocurcumin with bulk curcumin. ACS
Omega. 7:46494–46500. 2022.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Tyagi P, Singh M, Kumari H, Kumari A and
Mukhopadhyay K: Bactericidal activity of curcumin I is associated
with damaging of bacterial membrane. PLoS One.
10(e0121313)2015.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Song J, Choi B, Jin EJ, Yoon Y and Choi
KH: Curcumin suppresses Streptococcus mutans adherence to human
tooth surfaces and extracellular matrix proteins. Eur J Clin
Microbiol Infect Dis. 31:1347–1352. 2012.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Betts JW and Wareham DW: In vitro activity
of curcumin in combination with epigallocatechin gallate (EGCG)
versus multidrug-resistant Acinetobacter baumannii. BMC Microbiol.
14(172)2014.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Mun SH, Joung DK, Kim YS, Kang OH, Kim SB,
Seo YS, Kim YC, Lee DS, Shin DW, Kweon KT and Kwon DY: Synergistic
antibacterial effect of curcumin against methicillin-resistant
Staphylococcus aureus. Phytomedicine. 20:714–718.
2013.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Izui S, Sekine S, Maeda K, Kuboniwa M,
Takada A and Amano A: Antibacterial activity of curcumin against
periodontopathic bacteria. J Periodontol. 87:83–90. 2016.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Wang J, Zhou X, Li W, Deng X, Deng Y and
Niu X: Curcumin protects mice from Staphylococcus aureus
pneumonia by interfering with the self-assembly process of
α-hemolysin. Sci Rep. 6(28254)2016.PubMed/NCBI View Article : Google Scholar
|
|
87
|
De R, Kundu P, Swarnakar S, Ramamurthy T,
Chowdhury A, Nair GB and Mukhopadhyay AK: Antimicrobial activity of
curcumin against Helicobacter pylori isolates from India and during
infections in mice. Antimicrob Agents Chemother. 53:1592–1597.
2009.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Yun DG and Lee DG: Antibacterial activity
of curcumin via apoptosis-like response in Escherichia coli.
Appl Microbiol Biotechnol. 100:5505–5514. 2016.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Amol Marathe S, Balakrishnan A, Devi Negi
V, Sakorey D, Chandra N and Chakravortty D: Curcumin reduces the
motility of Salmonella enterica serovar Typhimurium by binding to
the flagella, thereby leading to flagellar fragility and shedding.
J Bacteriol. 198:1798–1811. 2016.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Bellio P, Brisdelli F, Perilli M, Sabatini
A, Bottoni C, Segatore B, Setacci D, Amicosante G and Celenza G:
Curcumin inhibits the SOS response induced by levofloxacin in
Escherichia coli. Phytomedicine. 21:430–434. 2014.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Zhang J, Tang L, Li GS and Wang J: The
anti-inflammatory effects of curcumin on renal ischemia-reperfusion
injury in rats. Renal Failure. 40:680–686. 2018.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Huang W, Li X, Wang D, Sun Y, Wang Q, Bu Y
and Niu F: Curcumin reduces LPS-induced septic acute kidney injury
through suppression of lncRNA PVT1 in mice. Life Sci.
254(117340)2020.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Fan Y, Chen H, Peng H, Huang F, Zhong J
and Zhou J: Molecular mechanisms of curcumin renoprotection in
experimental acute renal injury. Front Pharmacol.
8(912)2017.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Li L, Liu X, Li S, Wang Q, Wang H, Xu M
and An Y: Tetrahydrocurcumin protects against sepsis-induced acute
kidney injury via the SIRT1 pathway. Renal Failure. 43:1028–1040.
2021.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Maa F, Liu F, Ding L, You M, Yue H and
Zhou Y: Anti-inflammatory effects of curcumin are associated with
down regulating microRNA-155 in LPS-treated macrophages and mice.
Pharm Biol. 55:1263–1278. 2017.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Vachharajani V, Wang SW, Mishra N, el
Gazzar M, Yoza B and McCall C: Curcumin modulates leukocyte and
platelet adhesion in murine sepsis. Microcirculation. 17:407–416.
2010.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Kumari A, Dash D and Singh R: Curcumin
inhibits lipopolysaccharide (LPS)-induced endotoxemia and airway
inflammation through modulation of sequential release of
inflammatory mediators (TNF-α and TGF-β1) in murine model.
Inflammopharmacology. 25:329–341. 2017.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Liu YF, Yang CW, Liu H, Sui SG and Li XD:
Efficacy and therapeutic potential of curcumin against
sepsis-induced chronic lung injury in male albino rats. J Nutr
Health Aging. 21:307–313. 2017.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Memis D, Hekimoglu S, Sezer A, Altaner S,
Sut N and Usta U: Curcumin attenuates the organ dysfunction caused
by endotoxemia in the rat. Nutrition. 24:1133–1138. 2008.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Lukita-Atmadja W, Ito Y, Baker GL and
McCuskey RS: Effect of curcuminoids as anti-inflammatory agents on
the hepatic microvascular response to endotoxin. Shock. 17:399–403.
2002.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Luo DD, Chen JF, Liu JJ, Xie JH, Zhang ZB,
Gu JY, Zhuo JY, Huang S, Su ZR and Sun ZH: Tetrahydrocurcumin and
octahydrocurcumin, the primary and final hydrogenated metabolites
of curcumin, possess superior hepatic-protective effect against
acetaminophen-induced liver injury: Role of CYP2E1 and Keap1-Nrf2
pathway. Food Chem Toxicol. 123:349–362. 2019.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Qiu F, Zeng C, Liu Y, Pan H and Ke C: J147
ameliorates sepsis-induced depressive-like behaviors in mice by
attenuating neuroinflammation through regulating the TLR4/NF-κB
signaling pathway. J Mol Histol. 54:725–738. 2023.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Gaddipati JP, Sundar SV, Calemine J, Seth
P, Sidhu GS and Maheshwari RK: Differential regulation of cytokines
and transcription factors in liver by curcumin following
hemorrhage/resuscitation. Shock. 19:150–156. 2003.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Shan B, Schaaf C, Schmidt A, Lucia K,
Buchfelder M, Losa M, Kuhlen D, Kreutzer J, Perone MJ, Arzt E, et
al: Curcumin suppresses HIF1A synthesis and VEGFA release in
pituitary adenomas. J Endocrinol. 214:389–398. 2012.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Zhao M, Wang Y, Yue Y, Zhang R, Wang S and
Zhang W: Curcumin alleviates neuronal apoptosis and cerebral
mitochondrial dysfunction in septic mice. Int. J Clin Exp Med.
9:6107–6113. 2016.
|
|
106
|
Wang Y, Shan X, Dai Y, Jiang L, Chen G,
Zhang Y, Wang Z, Dong L, Wu J, Guo G and Liang G: Curcumin analog
L48H37 prevents lipopolysaccharide-induced TLR4 signaling pathway
activation and sepsis via targeting MD2. J Pharmacol Exp Ther.
353:539–550. 2015.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Yang C, Wu K, Li SH and You Q: Protective
effect of curcumin against cardiac dysfunction in sepsis rats.
Pharm Biol. 51:482–487. 2013.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Zhu H, Zhang L, Jia H, Xu L, Cao Y, Zhai
M, Li K, Xia L, Jiang L, Li X, et al: Tetrahydrocurcumin improves
lipopolysaccharide-induced myocardial dysfunction by inhibiting
oxidative stress and inflammation via JNK/ERK signaling pathway
regulation. Phytomedicine. 104(154283)2022.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Rattis BAC, Piva HL, Duarte A, Gomes
FGFLR, Lellis JR, Soave DF, Ramos SG, Tedesco AC and Celes MRN:
Modulation of the mTOR pathway by curcumin in the heart of septic
mice. Pharmaceutics. 14(2277)2022.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Olszanecki R, Gebska A and Korbut R: The
role of haem oxygenase-1 in the decrease of endothelial
intercellular adhesion molecule-1 expression by curcumin. Basic
Clin Pharmacol Toxicol. 101:411–415. 2007.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Kumari A, Tyagi N, Dash D and Singh R:
Intranasal curcumin ameliorates lipopolysaccharide-induced acute
lung injury in mice. Inflammation. 38:1103–1112. 2015.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Carter Y, Liu G, Yang J, Fier A and Mendez
C: Sublethal hemorrhage induces tolerance in animals exposed to
cecal ligation and puncture by altering p38, p44/42, and SAPK/JNK
MAP kinase activation. Surg Infect (Larchmt). 4:17–27.
2003.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Yuan Z, Syed MA, Panchal D, Rogers D, Joo
M and Sadikot RT: Curcumin mediated epigenetic modulation inhibits
TREM-1 expression in response to lipopolysaccharide. Int J Biochem
Cell Biol. 44:2032–2043. 2012.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Sompamit K, Kukongviriyapan U, Nakmareong
S, Pannangpetch P and Kukongviriyapan V: Curcumin improves vascular
function and alleviates oxidative stress in non-lethal
lipopolysaccharide-induced endotoxaemia in mice. Eur J Pharmacol.
616:192–199. 2009.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Lu W, Jiang JP, Hu J, Wang J and Zheng MZ:
Curcumin protects against lipopolysaccharide-induced
vasoconstriction dysfunction via inhibition of thrombospondin-1 and
transforming growth factor-β1. Exp Ther Med. 9:377–383.
2015.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Chen L, Lu Y, Zhao L, Hu L, Qiu Q, Zhang
Z, Li M, Hong G, Wu B, Zhao G and Lu Z: Curcumin attenuates
sepsis-induced acute organ dysfunction by preventing inflammation
and enhancing the suppressive function of Tregs. Int
Immunopharmacol. 61:1–7. 2018.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Silva LS, Catalão CH, Felippotti TT,
Oliveira-Pelegrin GR, Petenusci S, de Freitas LA and Rocha MJ:
Curcumin suppresses inflammatory cytokines and heat shock protein
70 release and improves metabolic parameters during experimental
sepsis. Pharm Biol. 55:269–276. 2017.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Hu G, Wang D, Jiang L, Xu L, Zhao L and
Zhou M: Curcumin protects hepatocytes from sepsis by regulating
inflammatory response and hepatocyte apoptosis. Trop J Pharm Res.
21:67–71. 2022.
|
|
119
|
Yun SS, Kim SP, Kang MY and Nam SH:
Inhibitory effect of curcumin on liver injury in a murine model of
endotoxemic shock. Biotechnol Lett. 32:209–214. 2010.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Gradišar H, Keber MM, Pristovšek P and
Jerala R: MD-2 as the target of curcumin in the inhibition of
response to LPS. J Leukoc Biol. 82:968–974. 2007.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Chen HW, Kuo HT, Chai CY, Ou JL and Yang
RC: Pretreatment of curcumin attenuates coagulopathy and renal
injury in LPS-induced endotoxemia. J Endotoxin Res. 13:15–23.
2007.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Zhu H, Wang X, Wang X, Liu B, Yuan Y and
Zuo X: Curcumin attenuates inflammation and cell apoptosis through
regulating NF-κB and JAK2/STAT3 signaling pathway against acute
kidney injury. Cell Cycle. 19:1941–1951. 2020.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Wang S, Zhao P, Zhang Y, Zhu L, Zhu J, Luo
Y and Li Q: The therapeutic effects of curcumin in early septic
acute kidney injury: An experimental study. Drug Des Devel Ther.
15:4243–4255. 2021.PubMed/NCBI View Article : Google Scholar
|
|
124
|
Xiao X, Yang M, Sun D and Sun S: Curcumin
protects against sepsis-induced acute lung injury in rats. J Surg
Res. 176:e31–e39. 2012.PubMed/NCBI View Article : Google Scholar
|
|
125
|
Yılmaz Savcun G, Ozkan E, Dulundu E,
Topaloğlu U, Sehirli AO, Tok OE, Ercan F and Sener G: Antioxidant
and anti-inflammatory effects of curcumin against hepatorenal
oxidative injury in the experimental sepsis model created in rats.
Ulus Travma Acil Cerrahi Derg. 19:507–515. 2013.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Alexa ID, Ilie AC, Prada G, Herghelegiu
AM, Luca A and Rotaru TŞ: A comprehensive behavioural assessment of
curcumin's effect on inflammatory and non-inflammatory pain in
mice. Farmacia. 68:829–834. 2020.
|
|
127
|
Singla V, Mouli VP, Garg SK, Rai T,
Choudhury BN, Verma P, Deb R, Tiwari V, Rohatgi S, Dhingra R, et
al: Induction with NCB-02 (curcumin) enema for mild-to-moderate
distal ulcerative colitis-A randomized, placebo-controlled, pilot
study. J Crohns Colitis. 8:208–214. 2013.PubMed/NCBI View Article : Google Scholar
|
|
128
|
Laurindo LF, De Carvalho GM, De Oliveira
Zanuso B, Figueira ME, Direito R, de Alvares Goulart R, Buglio DS
and Barbalho SM: Curcumin-based nanomedicines in the treatment of
inflammatory and immunomodulated diseases: An evidence-based
comprehensive review. Pharmaceutics. 15(229)2023.PubMed/NCBI View Article : Google Scholar
|
|
129
|
Stohs SJ, Chen O, Ray SD, Ji J, Bucci LR
and Preuss HG: Highly bioavailable forms of curcumin and promising
avenues for curcumin-based research and application: A review.
Molecules. 25(1397)2020.PubMed/NCBI View Article : Google Scholar
|
|
130
|
Roney M and Mohd Aluwi MFF: Unraveling the
systems biology of curcumin: A Mini-review of its Anti-diabetic
potential through network pharmacology. Cell Biochem Biophys: Sep
10, 2025 doi: 10.1007/s12013-025-01888-y (Epub ahead of print).
|
|
131
|
Wahnou H, El Kebbaj R, Liagre B, Sol V,
Limami Y and Duval RE: Curcumin-based nanoparticles: Advancements
and challenges in tumor therapy. Pharmaceutics.
17(114)2025.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Xiang DB, Zhang KQ, Zeng YL, Yan QZ, Shi
Z, Tuo QH, Lin LM, Xia BH, Wu P and Liao DF: Curcumin: From a
controversial ‘panacea’ to effective antineoplastic products.
Medicine (Baltimore). 99(e18467)2020.PubMed/NCBI View Article : Google Scholar
|