1. Introduction
Memory can be defined as the acquisition, storage
and retrieval of sensory information, and the ability to recall
past events at the conscious and unconscious levels. Recent
advancements in both molecular neurobiology and information
technology are prompting a re-evaluation of how memory is defined
(1). There are three stages of
memory processing: Encoding, storage and retrieval. Encoding
denotes the transformation of sensory information into neural code.
The second stage of memory processing is storage or consolidation,
which is the process of retaining encoded information over time.
and the third is retrieval, the process of accessing and recalling
stored information and bringing it back into conscious
awareness.
Memory can be influenced by different factors that
affect the three stages of memory (encoding, storage, and
retrieval). Pain can impair memory function; both animal and human
studies have indicated that physical discomfort can negatively
affect the memory process (2,3).
Pain selectively hinders the consolidation of object recognition
memory (a type of declarative memory), without affecting its
initial acquisition or later retrieval (4). Memory encoding is highly dependent on
attention. In the event that an individual distracted or does not
allow sufficient focus to new stimuli, the amount of information
successfully converted for storage will be diminished (5). The capacity to store memories can be
compromised by physical damage to a specific area of the brain. For
instance, trauma to the hippocampus, a structure critical for
memory consolidation, can corrupt the storage process (6). Even after information is stored, the
ability to retrieve it from long-term memory can be disrupted. This
can occur due to normal decay over time, where the neural traces
weaken, making the memory difficult or impossible to access
(7).
The crucial difference between short- and long-term
memory is that long-term memory requires the synthesis of new
proteins for its stabilization. A critical newly synthesized
protein in the process of long-term potentiation (LTP) is protein
kinase C (PKC) ζ type (PKMζ), an autonomously active variant of the
enzyme PKC (8). PKMζ is essential
for sustaining activity-dependent strengthening of synaptic
connections. Inhibiting PKMζ effectively erases established
long-term memory without harming short-term memory.
Furthermore, once the inhibitor is removed, the
ability to form new long-term memory is completely restored.
Another crucial molecule is brain-derived neurotrophic factor
(BDNF), which is crucial for the persistence of long-term memories.
The enduring stabilization of synaptic modifications requires a
simultaneous increase in both pre-and postsynaptic structures,
specifically the axonal bouton, dendritic spine and postsynaptic
density. This structural growth is supported at the molecular level
by an increase in postsynaptic scaffolding proteins. For example,
higher levels of PSD-95 and HOMER 1c have been shown to be
associated with the stabilization of synaptic enlargement (9).
2. Role of epigenetics in memory
Studies on rats show that learning triggers DNA
hypermethylation in the hippocampus. Although the hippocampus
serves as the initial storage site for these memories, they are
eventually relocated to the anterior cingulate cortex (10,11).
During memory formation, in the hippocampus, the activity of the
DNA methyltransferase enzyme increases. This leads to the
hypermethylation of genes that inhibit memory, effectively turning
them off. Simultaneously, genes that promote memory often undergo
hypomethylation (removal of methyl groups), keeping them active
(12).
DNA methylation at specific base pairs affects
synaptic plasticity by altering gene expression and splicing
patterns. DNA methylation can influence memory by altering synaptic
circuits (13,14). The epigenetic regulation silences
memory repressors and promotes memory enhancers. Eventually, this
balance contributes to the formation, consolidation and long-term
storage of memories by stabilizing synaptic plasticity.
3. Balance between memory formation and
forgetting: How dopamine neurons orchestrate memory
For effective adaptation in the environment, animals
need a balance between neurogenesis and the vanishing of old
memories. Dopaminergic neurons may be responsible for both the
formation of new memory and the forgetting of unconsolidated memory
by different signalling pathways. Research on Drosophila has
revealed that cyclic AMP (cAMP) signalling plays a crucial role in
creating memories, while cyclic guanine monophosphate (cGMP)
signalling actively triggers the forgetting of unsolidified
memories through these dopaminergic neurons (15).
Through genetic screening and proteomic analysis,
researchers have discovered that neuronal activation leads to the
formation of a complex that includes Kdm4B, a histone H3K9
demethylase, and Bur, a GMP synthetase (15). This Kdm4B/Bur complex is crucial
and sufficient for the forgetting of unconsolidated memories.
Nitric oxide (NO)-dependent cGMP signalling activates the Kdm4B/Bur
complex through phosphorylation via dopamine neurons. This
activation is essential for gene expression of kek2, which
codes for a presynaptic protein. Consequently, activating Kdm4B/Bur
brings about changes at the presynapse (15).
The present review discusses the association between
cGMP signalling and synaptic changes, mediated by gene expression,
in the process of forgetting. It suggests that the opposing
functions of memory formation and forgetting are achieved by
different signalling pathways via dopamine neurons, ultimately
influencing synaptic integrity and balancing adaptive animal
behaviour.
Bidirectional memory regulation and
dopamine neuron signalling
Animal behaviour is determined by memories stored in
the brain; however, survival depends on the ability to forget
negative memories. Since environments are always changing, and in a
changing environment, an efficient system is essential to weaken
older memories, as it provides the way for more adaptive, updated
behaviours (16,17). At the same time, the brain must
also solidify key memories through a process known as
consolidation, often involving gene expression (18).
The active process of memory decay, or ‘forgetting’,
has been extensively studied at the molecular level in
Drosophila. A classic example is observed in the olfactory
aversive training paradigm, where a specific odour is paired with
an electric shock (19). This
creates an associative memory within the olfactory memory centre of
the fruit fly, known as the mushroom body (MB) neurons (20,21).
This memory formation is facilitated by the simultaneous activation
of dopamine neurons (22,23).
Notably, this olfactory aversive memory in fruit
flies rapidly fades within ~3 h. This rapid forgetting is actively
driven by specific molecular players: Rac1, a small G protein
belonging to the Rho GTPase family, and Raf, an upstream kinase of
mitogen-activated protein kinase (24,25).
While the blocking of this Rac1-mediated forgetting pathway can
extend memory duration, the memory still eventually fades. This
raises the question of whether memory simply fades away over time,
or whether are there additional active processes contributing to
its decay.
The processes by which memories are formed must
inherently interact with the mechanisms that cause them to fade. A
fundamental principle in learning and memory involves dopamine and
its G-protein-coupled receptors activating adenylate cyclase (AC),
which in turn elevates the levels of cAMP. This pathway is a common
thread in memory creation (20,26).
At the synaptic level in Drosophila, the
connections between MB neurons and their downstream partners, the
MB output neurons, experience synaptic depression (27,28).
However, a key question remains: Namely, whether this synaptic
plasticity within the MBs is linked to memory decay. Specifically,
it remains still unclear how synaptic depression may be reversed or
restored by either active forgetting mechanisms or any other
processes that lead to memory decay.
Traditionally, researchers linked gene expression
mainly to the strengthening of memories, a process known as
consolidation. However, research reveals a direct connection
between gene expression and the weakening of memories through
synaptic plasticity (29).
Furthermore, this memory decay occurs independently of the Rac1 or
Raf pathways (30). Since this
particular decay occurs after Rac1-dependent forgetting and uses
different mechanisms, it is termed ‘gene expression-based
forgetting’. This type of forgetting is driven by cyclic guanine
monophosphate (cGMP) signalling within the MB neurons, which
receives a boost from dopamine neurons. The results in altered gene
expression in the MB neurons and changes at the presynapse
(15).
The specific role of cGMP signalling via
dopaminergic neurons helps bridge a crucial gap in the
understanding of how memories are both formed and forgotten.
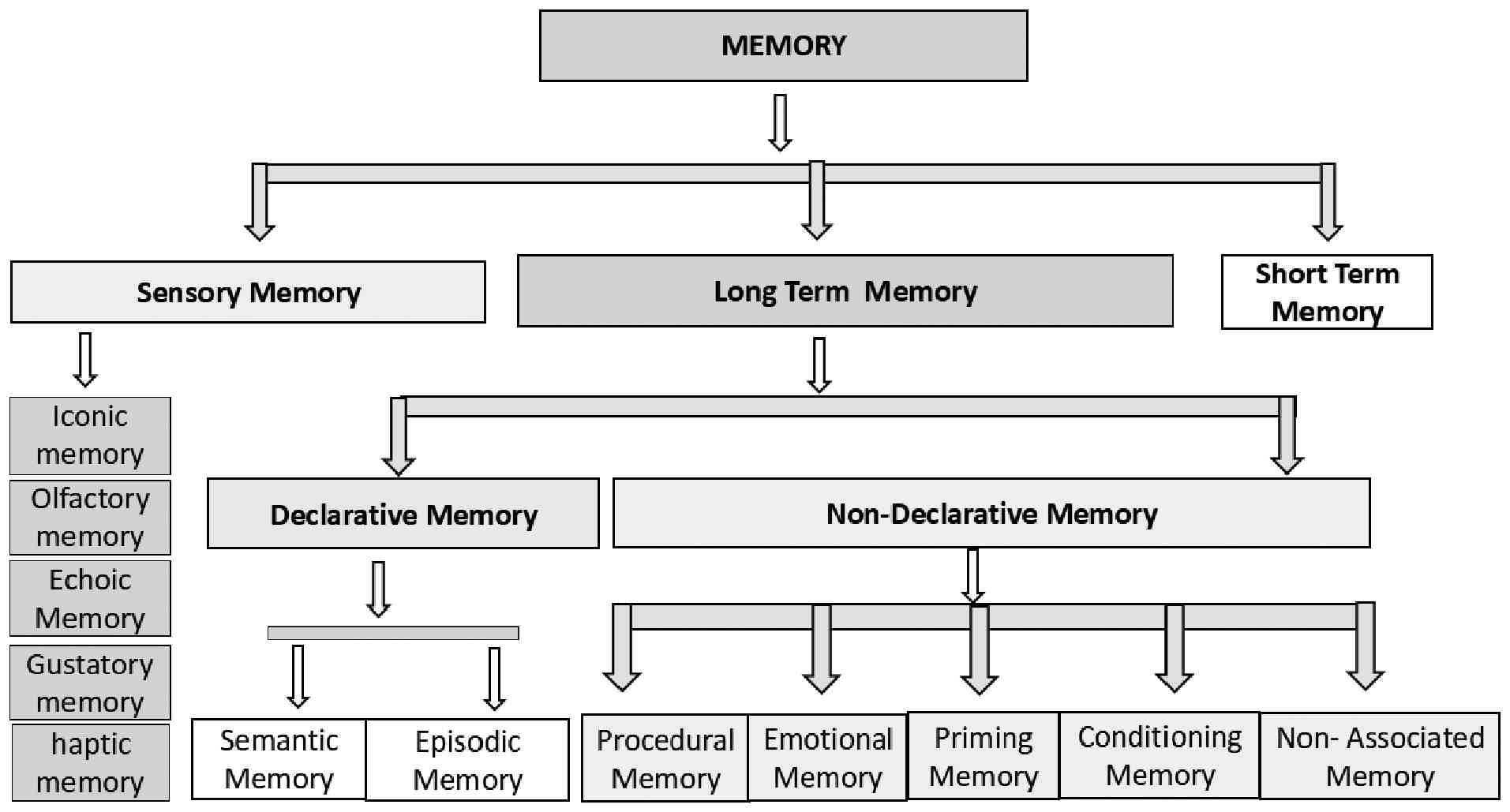
4. Classification of memory
Memory can be classified as sensory memory,
short-term memory or working memory, and long-term memory according
to the multistore (or modal) model proposed by Atkinson and
Shiffrin in 1968(31). As regards
sensory memory, in cognitive psychology, sensory memory is defined
as the primary stage of the memory process. It functions as a
modality-specific buffer that preserves a high-fidelity, isomorphic
(unprocessed) representation of environmental stimuli. It
essentially acts as a highly detailed, fleeting snapshot of the
environment that allows the brain a fraction of a second to process
incoming sensations before they are lost or moved to short-term
memory (32). Its range may from
milliseconds to 4 sec. Sensory memory can be classified as iconic
memory (vision). echoic memory (sound), haptic memory (touch),
olfactory memory (smell) and gustatory memory (taste). Short-term
memory and long-term memory are discussed in more detail in the
sections below.
5. Short-term memory
Short-term memory, also known as primary or active
memory, refers to the cognitive system responsible for holding a
limited amount of information for a brief period, generally up to
30 sec (33). This contrasts with
long-term memory, which can store an unlimited amount of
information indefinitely. The distinction between these two memory
systems lies not only in their duration, but also in their distinct
functions. Despite their differences, short-term memory and
long-term memory are closely interconnected.
Short-term memory refers to the active retention of
information for a brief duration, without any manipulation of that
information. When information is actively maintained and
manipulated, it is typically categorized as working memory, as
defined by Baddeley (34).
Research into short-term memory commonly employs
tasks where participants are presented with a series of items and
subsequently asked to either recall or recognize the information
they just encountered. This raises fundamental questions regarding
its operation: These questions pertain to how short-term memory
functions, and whether there are distinct processes and storage
mechanisms for different types of information, or for information
received through various sensory modalities, such as visual or
auditory input.
Investigating short-term memory across
modalities and materials
Much of the research on short-term memory has
focused on how verbal or visuospatial stimuli are processed, with
less attention paid to other types of information. Similarly, the
impact of sensory modality on short-term memory has been primarily
explored within the verbal domain.
The study by Talamini et al (35) aimed to bridge existing gaps by
comparing visual and auditory short-term memory across diverse
material types. Specifically, it investigated whether sensory
modality and the nature of the stimuli interact to influence memory
performance. Talamini et al (35) examined whether musical expertise
can modulate memory performance. As previous research has
demonstrated, musicians often have superior auditory memory and
visual memory to a certain extent, compared to non-musicians
(35). To test this hypothesis,
the researchers employed a consistent recognition paradigm across
various stimulus types, and in every trial, the researcher
presented two sequences of events separated by a silent delay
before the participants (musicians and non-musicians), and they
were asked to identify whether both sequences were identical or
different. They compared performance for auditory and visual
materials, which were categorized into three distinct groups: i)
First, a verbal group, which included syllables; ii) a second
non-verbal with contour group (these were stimuli that could not be
easily named but possessed a distinct contour (patterns of rising
and falling variations in loudness for auditory stimuli or
luminance for visual stimuli); and iii) a third non-verbal without
contour group; this category included pink noise sequences for
auditory stimuli and Kanji letter sequences for visual stimuli,
characterised by the absence of an obvious contour or pattern of
up-and-down changes based on non-pitch features (35). The results revealed a significant
advantage for musicians, mainly with auditory non-contour stimuli
and contour stimuli (visual or auditory). The findings of that
study suggest that musical training can enhance short-term memory
associated with musical skills. It was also concluded that the
encoding strategies play a crucial role in short-term memory
performance (35).
Investigating short-term memory
mechanisms
Short-term memory is a fascinating, complex
cognitive function that involves intricate processes of perceiving
information, forming memories, and then using that stored
information. Despite its critical role in daily life, the precise
mechanism by which short-term memories are formed, retained and
retrieved remain unclear.
To shed light on these mechanisms, a previous study
examined 41 healthy college students who participated in a
challenging memory task (36). The
researchers used functional near-infrared spectroscopy (fNIRS) to
monitor real-time changes in haemoglobin concentrations within
specific areas of the cortex in the brain. Alongside this, the
facial expressions and vital signs of the participants throughout
the memory tests were continuously recorded (36). The results revealed increased
activity in several key brain regions during the memory tasks: The
inferior prefrontal gyrus, the visual association cortex, the
pre-motor cortex and the supplementary motor cortex. They observed
a significant increase in functional connectivity between these
regions during task engagement, with differences in brain activity
among participants becoming less pronounced over time (36). Of note, participants who
demonstrated superior short-term memory performance exhibited fewer
negative emotional expressions and had higher heart rates compared
to those with weaker memory (36).
These findings strongly suggest that the robust interconnectivity
within the cortex of the brain and sufficient cerebral blood
oxygenation are crucial factors in boosting short-term memory
capacity.
The influence of proprioceptive
short-term memory on passive motor learning
When a physical trainer guides the limbs of an
individual to teach a specific movement, they are providing
proprioceptive information, the sense of body position and
movement. For the learner to perform the same type of movement,
they have to accurately perceive this information and store it for
later performance. This indicates two key abilities: Proprioceptive
acuity (the ability to accurately sense movement) and short-term
memory (the ability to hold that perceived information for a short
time). These two functions are required for active motor learning
(where the learner performs the movement themselves). The role of
these two functions is minimal in passive motor learning (where the
movement is physically guided).
To investigate this, a previous study experimented
on 21 healthy adults (aged an average of 25 years). That study was
performed to determine whether an the efficiency of an individual
in passively guided learning is linked to their proprioceptive
acuity and short-term memory capacity (37). That study revealed a significant
positive significance about learning efficiency. Learning
efficiency was strongly associated with short-term memory capacity.
Specifically, individuals who had good recall capacity of older
sensory stimuli demonstrated superior learning efficiency. However,
the researchers found no significant association between learning
efficiency and proprioceptive acuity. Using a causal graph model,
they observed that memory directly influenced learning, while
proprioceptive acuity had an indirect effect on learning, operating
through memory. These findings underscore the critical importance
of a learner's short-term memory for effective passive motor
learning (37).
6. Long-term memory
Long-term memory involves the capacity of the brain
to store information for extended periods, ranging from days to a
lifetime. It is the vast mental archive that allows the recalling
of past events, facts, skills and experiences. John Robert
Anderson, Professor and Psychologist at Carnegie Mellon University,
Pittsburgh, PA, USA, classified long term memory into two parts:
One is declarative (explicit), and the other is non-declarative
memory (implicit) (38).
Declarative memory can be divided into two parts: The first is
semantic memory, and the second is episodic memory, while
non-declarative memory can be classified into several types, such
as procedural memory, emotional memory, classical conditioning,
priming and non-associated memory, as illustrated in Fig. 1.
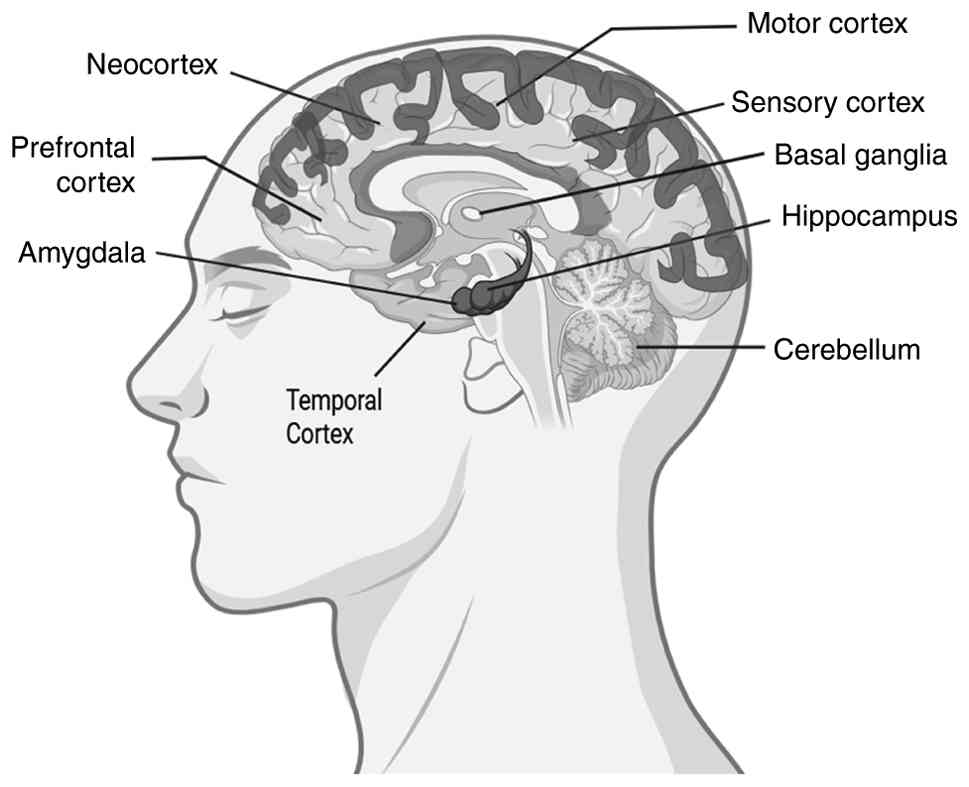
Certain regions of the brain encode different types
of memories. The hippocampus is the most critical region of the
brain responsible for declarative memories, semantic facts and
events remembrance is coded by the hippocampus (39). The basal ganglia are responsible
for spatial navigation memory (direction). Procedural learning,
including motor skills, is associated with the basal ganglia, such
as cycling, driving and learning a new language (40). The limbic system, including the
amygdala encodes emotional memories, such as weddings, birthdays,
winning an award ceremony and old school days. Laughing, crying and
weeping are associated with the limbic system and are encoded by
the amygdala (41). Short-term
memory is encoded by the cortex region, the prefrontal cortex, the
neocortex, sensory and motor cortex. Short-term memories are often
termed working memories (33,34).
The various parts of the brain responsible for different types of
memories are depicted in Fig.
2.
Long-term memory formation is a multi-stage journey,
transforming fleeting sensory experiences into lasting
recollections. It begins with encoding, the initial conversion of
what is seen, heard or felt into a neural language the brain can
understand and store. Then this newly encoded, fragile memories
stabilize, a process known as consolidation (42). This involves the following: i)
Cellular consolidation, which occurs rapidly, within minutes to
hours after learning, involves structural changes at individual
synapses (43). ii) System-level
consolidation, which is a much slower process, taking over days,
weeks, months, or even years (44,45).
System-level consolidation occurs across different brain regions.
In this consolidation, memories gradually move from the hippocampus
to the neocortex (46). Sleep
plays a critical role in the consolidation process, as the
hippocampus reiterates recent events, helping to strengthen and
update these cortical connections (47). Once memories are consolidated, they
become stored across various brain regions. Finally, retrieval is
the act of accessing and recalling this stored information whenever
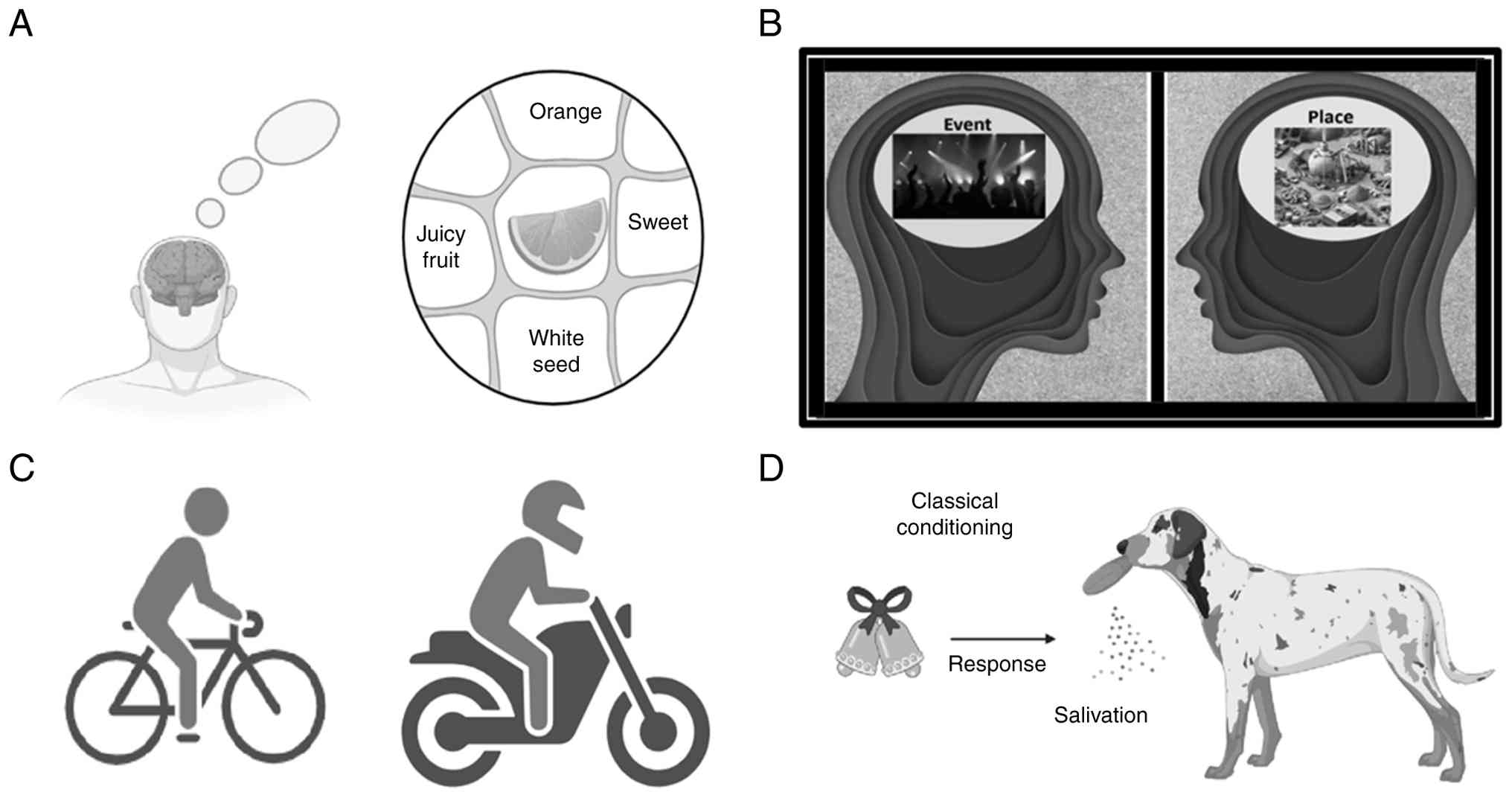
it's needed. Declarative memory (semantic and episodic memory) and
non-declarative memory (including procedural memory and classical
conditioning) are depicted in Fig.
3.
Mechanisms of long-term memory
The more in-depth understanding of long-term memory
formation, achieved through decades of research, provides exciting
new avenues for improving cognitive function. Researchers believe
that memory can be enhanced by targeting the mechanisms involved in
memory formation, including activating cAMP response
element-binding protein (CREB), managing
(alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid
(AMPA)/N-Methyl-D-aspartic acid (NMDA) receptor trafficking,
adjusting neuromodulators such as dopamine, adrenaline, cortisol,
or acetylcholine, and influencing metabolic processes through
substances, such as glucose and insulin (48).
The fundamental basis of long-term memory at the
cellular level is synaptic plasticity, the ability of connections
between neurons (synapses) to change in strength and structure. The
most well-studied mechanism is LTP. LTP is a persistent
strengthening of synapses that occurs when two neurons are
repeatedly and simultaneously activated. This phenomenon is often
summarised by ‘neurons that fire together, wire together’. The
first step of LTP is the induction of LTP. This typically involves
the activation of NMDA receptors (a type of glutamate receptor) on
the postsynaptic neuron. When these receptors are activated by
coincident presynaptic activity and strong postsynaptic
depolarisation, they allow an influx of calcium ions into the
neuron. Manipulating NMDA receptors has been shown to improve both
working memory and the consolidation of long-term memory (49). The second step is downstream
signalling. The increase in intracellular calcium triggers a
cascade of molecular events, activating various protein kinases
(such as PKA, PKC and CaMKII) (50). The third step involves gene
expression and protein synthesis (51,52).
For true long-term memory (lasting hours to days or longer), these
signalling pathways often lead to changes in gene expression and
the synthesis of new proteins, such as CREB and immediate early
genes (IEGs) (52).
CREB is activated in various signalling pathways,
contributing to memory consolidation and even enhancement. For
instance, when growth factors stimulate tyrosine kinase receptors,
CREB initiates a chain reaction involving Ras and extracellular
signal-regulated kinase (ERK) (53). CREB plays a crucial role in
long-term potentiation, which is a form of synaptic plasticity and
a fundamental mechanism for storing memories. CREB also contributes
in reconsolidation process (54).
GPCR activation induces adenylyl cyclase enzyme,
which converts ATP to cAMP, which activates PKA. Even stress
pathways and glutamate release can trigger CREB by causing the
release of intracellular calcium and subsequent events (55). Ultimately, all these pathways can
lead to the activation of CREB1, an active form of CREB. This
activation controls the expression of numerous target genes,
including IEGs. Some of these IEGs are themselves transcription
factors, which indicates that they regulate other genes responsible
for the synaptic changes that underpin synaptic plasticity. A good
example is the IEG and transcription factor C/EBPβ, a gene
regulated by CREB that is essential for memory consolidation
(56). Altering the function of
CREB1 has a direct impact on memory: When CREB1 is disrupted,
memory impairments occur, whereas boosting its activation leads to
improved memory (57).
These transcription factors then regulate the
expression of effector genes that lead to lasting structural
changes at the synapse (57).
These include the growth of new synaptic connections, changes in
the number and type of neurotransmitter receptors (e.g., more AMPA
receptors), and modifications to the shape of dendritic spines
(small protrusions on dendrites that receive synaptic input)
(58). These structural changes
are essential for the long-term stability of memories.
Role of NO in memory
consolidation
The role of NO in memory consolidation has been
recently reviewed (59). NO plays
various roles in the central nervous system, including blood vessel
dilation and neurotransmission to immune responses. The production
of NO in the brain is mainly catalysed by the NO synthase (NOS)
enzyme, which is found in three forms: Endothelial NOS (eNOS),
neuronal NOS (nNOS) and inducible NOS (iNOS). While eNOS and iNOS
are found in the central nervous system, nNOS is responsible for
generating NO within neurons (60).
Specificity protein 1 (Sp1) and CREB are crucial
transcription factors that regulate NO production by modulating the
expression of NOS enzymes related gene. nNOS can interact with Sp1,
and the interaction can inhibit Sp1 binding to DNA, providing a
mechanism for NO to modulate its own production. Once synthesised,
NO can diffuse to the postsynaptic cleft, where it exerts diverse
effects on neighbouring neurons (61).
Memory consolidation is the complex process by which
short-lived memories transform into a more stable memory (51). NO is crucial to this process,
largely through its involvement in synaptic plasticity (51,62,63).
Synaptic plasticity is an activity-dependent modification of the
efficacy of synaptic transmission at synapses. It plays a crucial
role in the capacity of the brain to include transient experiences
into persistent memory traces.
NO strengthens synaptic plasticity by enhancing LTP
in the hippocampus and cerebral cortex (64). It facilitates the conversion of
labile memories into a stable memory through several pathways. NO
functions as a retrograde messenger; it diffuses backwards from
postsynaptic neuron to presynaptic neuron through the synaptic
cleft and targets soluble guanylyl cyclase in the presynaptic
neuron, enhancing neurotransmitter release. These versatile roles
of NO ensure that neural circuits are optimally configured for the
long-term retention of information (65). This comprehensive involvement of NO
indicates the significance of cognitive functions and suggests its
potential as a target for future therapeutic strategies in treating
memory-related disorders.
In the hippocampus, NO functions as a retrograde
messenger following the activation of NMDA receptors (NMDARs)
during LTP (64). The process
begins when NMDARs on the postsynaptic neuron are activated,
leading to an influx of calcium ions (Ca2+) (66). This calcium flow activates nNOS,
which produces NO. NO rapidly diffuses back from the postsynaptic
terminal to the presynaptic terminal, where it activates soluble
guanylate cyclase. This enzyme then catalyses the conversion of GTP
to cGMP. The resulting increase in cGMP activates protein kinase G
(PKG) (67). PKG further
phosphorylates various proteins involved in neurotransmitter
release, such as glutamate, and finally strengthens the synapse,
promoting LTP. The complex signalling cascade plays a critical role
of NO in regulating synaptic plasticity and memory consolidation
(67).
7. Declarative memory
Declarative memory is also known as explicit memory,
a type of long-term memory that involves the conscious recollection
of facts, events and concepts (68). It is the type of memory that can
intentionally be called to mind and verbalised. It has been
indicated that the medial temporal lobe, particularly the
hippocampus, plays a crucial role in declarative memory (39). This form of memory is further
categorised into two main types: Semantic and episodic memory.
Semantic memory
Semantic memory deals with general knowledge and
facts about the world, independent of personal experience or
context. It is based on the concepts, principles and abstract
information, such as ‘the Moon revolves around the Earth’ or ‘a
Square has four sides’, relies on semantic memory. It is the
repository for all the knowledge which has been accumulated.
Research indicates that the semantic memory networks of older
adults has greater separation and segregation between concepts,
with fewer connections among them. Networks offer a powerful
strategy to model the organisation of conceptual representations
(69,70). These computationally modelled
semantic memory networks go beyond analysing simple word-to-word
relationships. They enable researchers to examine complex
interactions among multiple words and assess the overall structural
characteristics of a network, such as its efficiency. Researchers
have applied semantic memory network analyses in diverse fields,
including creativity, language acquisition, studies of clinical
populations and research on healthy ageing (70,71).
Evidence suggests that with advancing age, semantic memory exhibits
reduced efficiency, poorer organisation and fewer connections
(72,73). Ageing influences certain cognitive
functions, including language production, which can result in older
adults experiencing more difficulty with word retrieval.
How visual and semantic
representations influence memory
A previous study explored how different types of
visual and semantic information contribute to memory formation. The
researchers used a deep neural network to model three levels of
visual processing: Early, middle and late (74). For semantic representations, they
drew upon normative features (e.g., ‘is round’), taxonomic
categories (e.g., ‘is a fruit’) and encyclopaedic knowledge (e.g.,
‘is sweet’). The aim of that study was to identify brain regions
where each type of representation predicted either perceptual
memory (remembering what something looked like), conceptual memory
(remembering what something meant), or general memory (remembering
both) (74).
During the study, participants viewed objects, while
undergoing functional magnetic resonance imaging (fMRI).
Afterwards, they completed two memory tests: One conceptual
(word-based) and one perceptual (picture-based), and found that
visual representations were strong predictors of subsequent
perceptual memory within the visual cortices. Notably, they also
supported conceptual and general memory in more frontal brain
regions. Semantic representations, on the other hand, predicted
perceptual memory in the visual cortex, conceptual memory in the
perirhinal and inferior prefrontal cortex, and general memory in
the angular gyrus. These findings suggest that the way visual and
semantic information contributes to later memory is intricate,
depending on the specific type of information, the kind of memory
being tested, and where that information is stored in the brain
(74).
How brains organise knowledge about
individuals and places
Humans build up incredibly detailed semantic
knowledge about the world around them. This includes information
about individuals, such as their jobs and personalities, and
places, such as their cultural or historical importance. While
certain brain regions show a preference for images of individuals
and places, less is understood about whether these same areas also
specifically store the semantic knowledge associated with them. To
investigate this, a previous study developed a machine learning
model to gauge semantic similarity using information from
Wikipedia, then validated this model by comparing its similarity
ratings with those provided by human participants (75). Using the computational model,
researchers discovered that the brain represents semantic knowledge
about individuals and places in distinct networks: An anterior
temporal network for people and a posterior medial network for
places. Of note, the hippocampus, a crucial memory hub, was found
to represent semantic knowledge for both people and places
(75).
Episodic memory
Episodic memory represents all the previous events
of the life of an individual that occurred at a particular place
and specific time. These types of memories are often associated
with an individual emotion. For instance, remembering the first day
of school, a memorable vacation, or a significant life event, such
as winning a championship in any game or in event are to be
included in episodic memories, and these memories and moments from
life can be recalled (76,77). A previous study revealed that a
specific circuit, comprising the medial prefrontal cortex, lateral
entorhinal cortex and hippocampus, encodes episodic-like memories
by integrating event, place and time information (78). This process appears to involve
top-down regulation from the medial prefrontal cortex to the
hippocampus. This circuit is distinct from those processing
individual components of memory (object, time and place).
Episodic memory is vulnerable to decline in both
ageing and neurodegenerative conditions (79,80).
Research indicates that episodic memory performance typically
undergoes a consistent, linear decrease beginning at ~50 to 60
years of age (80). Ageing is
associated with declines in processing speed, working memory, and
episodic memory (81). With age,
the human brain shrinks slightly, and white matter integrity
diminishes, and these age-related changes also cause the loss of
episodic memory, especially related to the hippocampus. The
hippocampus, which is vital for encoding, consolidating, and
retrieving episodic memories, undergoes substantial volume loss,
particularly after age 50, alongside reduced synaptic plasticity
and other neurobiological alterations (82). Furthermore, the hippocampus appears
to become functionally disconnected from other brain regions, such
as the posterior and anterior cingulate cortices, with age
(83,84). These neural changes are linked to
age-related declines in overall cognition and episodic memory. In
fact, the hippocampus is often the most severely affected brain
structure to show pathological changes, such as volume loss, in
Alzheimer's disease (AD) and its prodromal stage (mild cognitive
impairment) (85,86). Its condition is also the strongest
predictor of episodic memory impairment in AD.
The prefrontal cortex is another key brain area
involved in how episodic memories are formed and recalled,
particularly those that require mental control, practice and the
ability to block out distractions (87,88).
Notably, with increasing age, the prefrontal cortex exhibits
greater activity during episodic memory tasks, and this activity
tends to spread out and involve both sides of the brain (89). This indicates that older adults do
not exhibit the same one-sided brain activity in the frontal and
prefrontal areas that younger adults typically do. This shift may
be a compensation mechanism for the brain for age-related changes.
In fact, older adults who perform well on memory tasks tend to use
both sides of their prefrontal cortex more strongly than those who
do not perform as well (87).
Furthermore, with age, the prefrontal cortex region experiences a
noticeable decrease in both white matter (the connections between
brain cells) and grey matter (the brain cells themselves) (90,91).
Episodic memory often declines as a natural part of
ageing, and this decline is also a key feature of age-related
memory disorders. A number of brain areas are known to be involved
in both episodic memory and age-related cognitive changes. The
recent study by Almeida et al (92) specifically demonstrated that the
cerebellum plays a causal role in episodic memory during ageing.
They found that healthy older adults who received a 12-day
neurostimulation program targeting the right cerebellum experienced
significant improvements in their episodic memory (92). These improvements were not only
immediate; they also lasted for at least four months after the
stimulation period ended. These findings underscore the direct
involvement of the cerebellum in long-term episodic memory
processes and suggest that it may be critical for regulating and
maintaining overall cognitive function.
Effect of episodic memory on delay
discounting
Episodic memory may slightly decrease delay
discounting in certain situations; this impact is generally minor
and easily disrupted (93). Delay
discounting describes the process through which the perceived value
of a reward diminishes with the increment in the delay to receiving
it (94). This often leads
individuals to opt for smaller, immediate rewards instead of
pursuing larger, future. Delay discounting is typically measured by
presenting participants with hypothetical choices: a smaller sum of
money available now vs. a larger sum available at a later date
(95). A greater tendency to
select the larger, later sum indicates lower levels of delay
discounting, often seen as a measure of self-control. Conversely,
consistently choosing the smaller, sooner option reflects higher
levels of delay discounting, which can be an indicator of
impulsivity.
Episodic future thinking (EFT) stands out as a
strategy that encourages individuals to choose larger, more distant
rewards (96). When prompted to
vividly imagine positive future events, people tend to favour a
larger sum of money available later (96,97).
EFT has been shown to reduce delay discounting in both adults and
children (98), and it can even
help decrease cigarette consumption (99) and food intake (100).
It raises a critical question of whether simply
recalling past experiences or episodic memory help reduce delay
discounting. A small number of studies on EFT have used episodic
memory as a comparison point. These studies have consistently
demonstrated that EFT is significantly more effective at decreasing
delay discounting than recalling past events (99,101). However, this does not completely
rule out the possibility that episodic memory could still boost
self-control compared to not engaging in any episodic thinking at
all. The impact of EFT on delay discounting is likely influenced by
several factors, including whether the thought is focused on the
future, its emotional tone (valence), and how episodic or vivid the
thinking is.
Developmental changes in episodic
memory
Episodic memory is the unique ability to recall
personal experiences, including not only the event itself (the
‘what’), but also the specific location (the ‘where’) and time (the
‘when’) it took place (102). For
instance, if one thinks their last anniversary, they may remember
who they celebrated with, where they had lunch, and whether you
they had dessert before or after opening presents.
To understand how this ability develops in early to
mid-childhood, a previous study explored the effectiveness of four
different lab-based tasks in characterising changes in episodic
memory in 200 typically developing children, aged 4 to 8 years
(103). Utilising longitudinal
data and a structural equation modelling framework, the findings of
that study indicated that multiple episodic memory tests can
effectively measure a consistent underlying latent construct of
episodic memory throughout this developmental period. Furthermore,
this ability consistently improves between the ages of 4 and 8
years (103). These results
underscore that episodic memory, as a measurable construct,
increases at a similar rate during early to mid-childhood. That
study also highlighted the advantages of employing diverse
laboratory tasks to track these developmental changes in episodic
memory (103).
Correlation of declarative memory and
children's math skills
Significant advancements have been made in
understanding the brain processes behind math learning. A
prevailing hypothesis suggests that declarative and procedural
memory, as fundamental learning and memory systems, play critical
roles in the acquisition of mathematical skills.
To investigate this, a previous longitudinal study
examined whether declarative and procedural memory predict the math
proficiency of children during their elementary school years
(104). The researchers of that
study followed 109 children from second to fourth grade, testing
them each year. The results revealed the following: i) Within each
grade, stronger declarative memory skills were linked to improved
math performance, but procedural memory skills were not. ii) The
declarative memory of a child in an earlier grade predicted their
math skills in later grades. For example, good declarative memory
in the second grade was connected to improved math skills in the
fourth grade (104). Sensitivity
analyses confirmed the robustness of these results, apart from when
accounting for stable individual differences through random
intercepts in the longitudinal prediction of later math skills.
These findings emphasise the foundational contribution of early,
general learning and memory mechanisms to children's mathematical
development (104).
How sex and education affect
declarative memory in older adults
Declarative memory, which aids in the learning and
recalling of everyday facts and events, typically weakens with age.
However, some research suggests that the sex of an individual and
the amount of education they received early in life may help lessen
these memory declines.
Previously, researchers conducted a study with 704
older adults in Taiwan, ranging from 58 to 98 years of age, with
varying levels of education (0 to 17 years) (105). They investigated their non-verbal
declarative memory by testing their ability to recognise drawings
of both familiar (such as a hairbrush) and unfamiliar made-up
objects after seeing them briefly. The findings of that study
revealed that age negatively affected declarative memory, although
this impact varied based on the person's sex and the type of
object. Memory decline was steeper for men than for women,
revealing that women had an advantage in remembering real objects
beginning at ~70 years of age. However, the remembering of made-up
objects exhibited a slower decline with no difference between the
sexes (105). Education was
linked to improved memory, although its benefits also depended on
sex and object type. Education helped women more than men, and it
was more beneficial for remembering real objects than made-up ones.
For men, each year of education brought memory gains twice as large
as the losses experienced each year due to ageing. For women, these
gains were 5-fold greater. These results suggest that in older
adults, non-verbal memory declines with age, but improves with more
education. However, both of these associations are influenced by
the sex of an individual and whether the information being learned
relates to existing knowledge or is entirely new. That study
highlights the lasting benefits of education, particularly for
women (105).
Movement behaviours and their
associations with declarative memory and hippocampal development in
early childhood
A previous study investigated whether 24-h movement
behaviours, specifically sedentary time, physical activity and
sleep, were linked to declarative memory and the size of the
hippocampus (a brain area crucial for memory) in young children
(105). They examined these
behaviours both individually and as a whole. That study collected
observational data from pre-schoolers at two points: First, with 35
children averaging 3.9 years of age and then 6 months later, with
28 of those children, who were then ~4.5 years of age. They
measured their movement using activity trackers (106). The main parameters investigated
were their declarative memory scores and the volumes of different
parts of their hippocampus. Using various statistical models, they
analysed the movement behaviours. When they examine each behaviour
on its own, they found that longer sleep durations were linked to a
larger total hippocampal volume, and moderate to vigorous physical
activity was associated with a larger right hippocampus (106). When they considered all movement
behaviours together, children who met recommended sleep guidelines
tended to have larger hippocampal volumes overall, including in the
right and left hemispheres, and specifically in the body and tail
regions of the hippocampus. In the group of pre-school children,
the researcher observed a clear positive connection between sleep
duration and hippocampal volume, regardless of the child's age
(106). To better understand how
these 24- behaviours relate to brain health in very young children,
future studies are required to include more participants and also
consider the specific types and settings of movement.
8. Non-declarative memory
Non-declarative memory, also known as implicit
memory, is a type of long-term memory that is not conscious. This
memory allows the performance of skills and routines automatically
without having to think about them (107). For example, riding a car, bike,
or cycle, DNA isolation from an onion, or typing are examples of
non-declarative memory. Non-declarative memory is acquired and
retrieved without conscious effort. It is built up through
repetition and practice, rather than through conscious recall,
while declarative memory (or explicit memory) involves the
conscious recollection of facts, events, and concepts. It can be
divided into various types, including procedural memory, emotional
memory, priming, and classical conditioning. Each memory is managed
by various brain areas and associated pathways (107,108). Non-declarative memory, also known
as implicit memory, supports the involuntary abilities and learned
reactions, operating without conscious recall (62).
Procedural memory
This is based on motor skills such as riding a motor
car, bicycle, typing, RNA isolation from blood, and playing a
musical instrument. After learning, the person can perform the task
without conscious effort. Research indicates that the basal ganglia
plays a role in procedural memory (109). Navigational memory is also
associated with the basal ganglia, although the basal ganglia are
not only responsible for procedural memory; other parts of the
brain also play a crucial role in navigation. The basal ganglia
consist of numerous subcortical nuclei present in the cerebrum, the
substantia nigra, and the subthalamic nucleus of the brain. The
primary components of the basal ganglia, found in the cerebrum,
include the caudate nucleus, putamen, globus pallidus and the
accumbens area (110,111). Additionally, the substantia nigra
(located in the midbrain) and the subthalamic nucleus (in the
diencephalon) are also included in the basal ganglia (112).
The basal ganglia play a crucial role in processing
and refining information from the cerebral cortex, contributing to
motor, cognitive and limbic functions (113,114). The basal ganglia serve as a
critical neural centre for movement; they are responsible for
activating the motor unit of muscle for the movement (115,116). The basal ganglia are also
responsible for habit formation, and the caudate nucleus (a
specific nuclei of the basal ganglia) especially facilitates
successful outcomes by promoting effective behavioural patterns and
selecting logical subgoals based on a continuous evaluation of
results (117).
Apart from the motor control function, the basal
ganglia also play a critical role in various cognitive processes,
such as decision-making, shifting attention, updating information
and adapting behaviour (118).
The reward processing function, which influences behaviour, choices
and motivation is also associated with the basal ganglia.
Furthermore, the basal ganglia are involved in processing emotions,
regulating affective states, and integrating emotional information
into decision-making.
Research indicates that the brain actively engages
conscious, explicit (or declarative) processes during the
development of procedural memories (119). It suggests that conscious
attention and cognitive effort play an important role in
skill-associated functions. Research suggests that the brain
depends on declarative memory during the early-stage encoding of a
new motor or cognitive procedure. The procedural learning is not
entirely unconscious, but can be achieved by practice or
repetition. It also involves intellectual ability (107), which helps in understanding and
strategizing during the learning phase. Episodic memory (120), allowing learners to recall
specific events or steps, especially when first acquiring the
skill. Executive functions (121), which include abilities such as
planning, problem-solving and working memory, all critical for
navigating the complexities of new tasks.
Role of the hippocampus and basal
ganglia in navigation
The nature of human navigational memory remains one
of the most persistent and fiercely contested puzzles in cognitive
science. It raises the fundamental question of whether the brain
builds a literal, unified map of the world (cognitive map), or
whether it merely integrates (patch) together a series of
directions and landmarks (graph or route). The debate is primarily
between two research groups of thought regarding how spatial
information is stored in the hippocampus and entorhinal cortex. The
cognitive map theory, proposed by Tolman (40) in 1948, suggests that navigation is
the result of the creation of an internal map inside the brain
(cognitive map) that allows organisms (rats or humans) to
understand and manipulate spatial relationships (40). Moving beyond simple turn-by-turn
sequences (egocentric navigation), the study by Tolman (40) with rodents suggested that animals
could locate objects by referencing multiple landmarks relative to
one another (allocentric navigation). The second theory proposed by
Wang and Hayden (122) states
that organisms do not have a unified map. Instead, they store
connected nodes (landmark A → right turn → landmark B) and navigate
by integrating these local memories (122). Wang and Hayden (122) also hypothesised a dual-process
model for curiosity-driven learning involving the orbitofrontal
cortex and the dorsal anterior cingulate cortex. Under this
framework, the orbitofrontal cortex is responsible for evaluating
the inherent worth of new data and integrating it into an internal
cognitive map. Moreover, the dorsal anterior cingulate cortex
monitors external constraints and the accessibility of information,
drawing upon the orbitofrontal cortex's cognitive map to direct
goal-oriented actions (122).
The 2014 Nobel Prize in Physiology or Medicine was
awarded to Dr John M. O'Keefe, Dr May-Britt Moser and Dr Edvard I.
Moser (123) They were recognized
for their ground-breaking discovery of the nerve cells in the brain
that make sense of place and navigation possible. They demonstrated
how complex cognitive functions and behaviours are represented by
specific cells found in a specific region of the brain. Dr John
O'Keefe discovered place cells in the hippocampal region of the
brain. These cells function as a type of GPS and provide the
location of an animal by signalling mechanisms, enabling the brain
to form spatial memories (124,125). Later, Dr May-Britt Moser and Dr
Edvard I. Moser discovered grid cells in the medial entorhinal
cortex, near the hippocampus. These grid cells generate a specific
internal coordinate system that is important for the navigation
process. These two types (hippocampal place cells and entorhinal
grid cells) form an interconnected network that is required for
making spatial maps and navigation (125). The work of these three scientists
has fundamentally changed the understanding of how brains perform
complex cognitive tasks and create spatial memory.
An animal needs to build a cognitive map (a mental
representation of its environment) to navigate successfully
(126). Several brain regions and
different types of cells work together to form this internal
navigation system. The hippocampus is a crucial region of the brain
to be considered for navigation in rat models (127). Two key cells are place cells in
the hippocampus and grid cells in the medial entorhinal cortex
(125,128). Hippocampal place cells fire
specifically when an animal is in a particular spot, creating a
place field that signals its location (129). This function depends on both
sensory information, such as what the animal sees, and spatial cues
from the medial entorhinal cortex that are independent of its
senses (124).
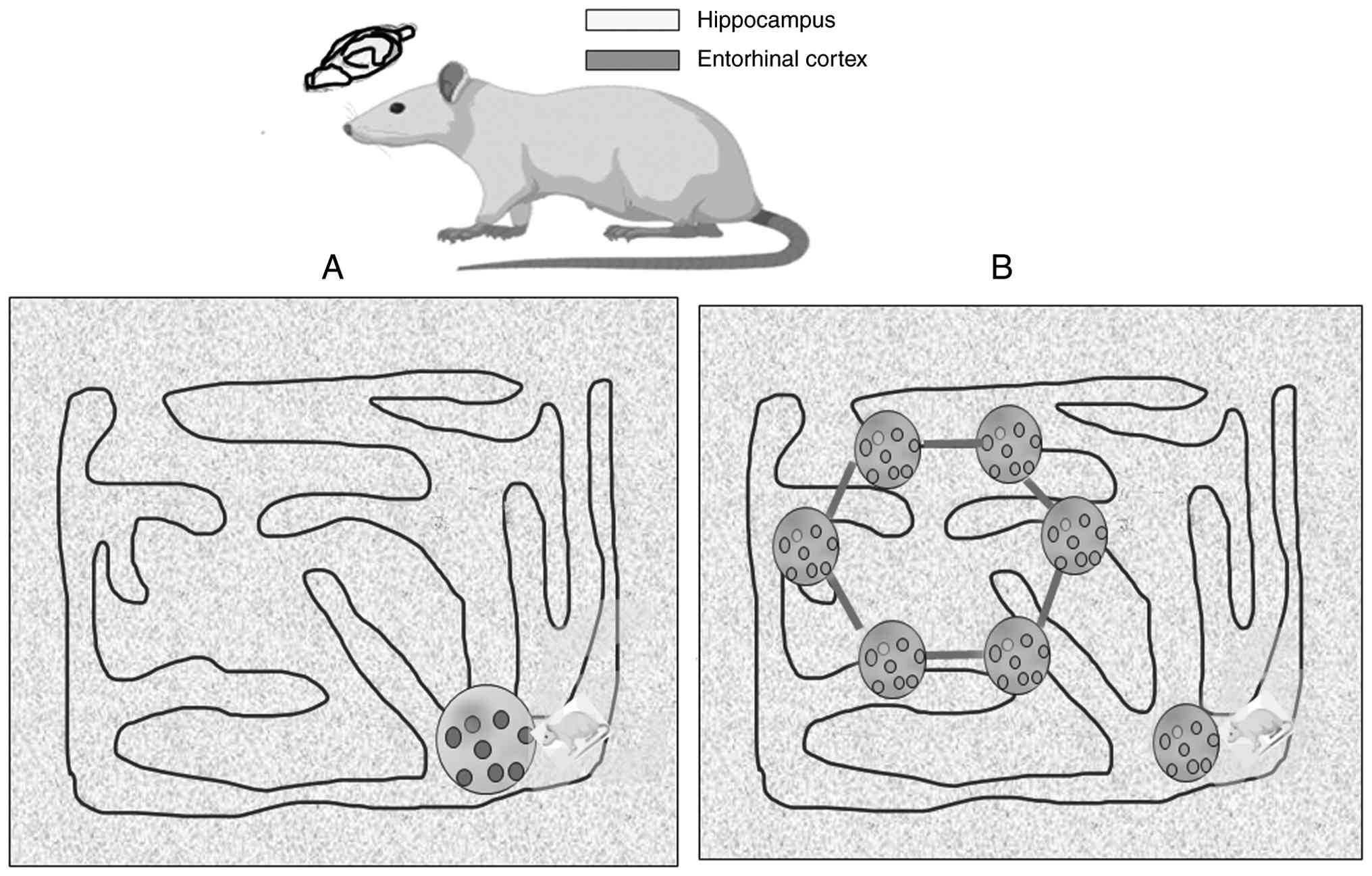
The medial entorhinal cortex uses grid cells to
create a sensory-independent spatial map (130,131). Head direction (HD) cells signal
the direction the head is facing, while grid cells fire in multiple
locations, creating a repeating, hexagonal pattern (132). These grid cells generate a unique
hexagonal firing pattern as an animal moves through an open space,
essentially forming a coordinate system in the brain (129) (Fig.
4).
The activity of grid cells is influenced by the
speed and direction of the animal. This suggests that they
integrate information about self-motion (known as idiothetic cues)
to calculate distance and direction, which is crucial for a process
called path integration (132,133). In a previous study, to determine
whether place cells help correct errors in a process called path
integration, researchers decoded the location of an animal using
grid cell activity (134). They
achieved this with and without input from place cells. They
discovered that when place fields emerged during exploration, the
accumulated error in the animal's position estimates decreased
(134). Place fields that were
closer to the current location of the animal were more effective at
reducing this error than those that were further away. That study
suggested that place cells function as spatial anchors and provide
precise information about the location of an animal that helps grid
cells maintain accurate path integration (134).
Previous research has suggested the coordination of
three regions of the brain for the navigation procedure. First, the
hippocampus and entorhinal cortex generate the core, map-like
spatial coordinates. Second, the parahippocampal and retrosplenial
cortices act as anchors, tethering these internal maps to stable
physical landmarks. Finally, these spatial codes are integrated
with frontal lobe processes to transform a mental map into an
actionable navigation plan (135).
The role of the basal ganglia in motor behaviour
has long been observed in mammals; however, the behavioural
functions are not exclusively motoric in nature. The basal ganglia
facilitate a form of learning in which stimulus-response (S-R)
associations or habits are acquired (136). There is evidence to indicate the
role of the basal ganglia, particularly the dorsal striatum, in
learning and memory (109). The
basal ganglia also function with other brain areas, such as the
hippocampus, to support various navigation methods (137). While the hippocampus creates a
mental map of one's surroundings, the basal ganglia enable more
rapid, more efficient navigation based on learned patterns. The
brain can then select which strategy to use, be it spatial or
habit-based, depending on the situation and environment.
Navigational technology such as GPS devices and
applications has become a normal part of daily life over the last
10 years. However, this technology affects the built-in navigation
system of the brain, also known as spatial memory. This internal
system relies on the hippocampus to function properly. A previous
study examined 50 regular drivers and recorded the extent of the
use of GPS throughout their lives, and tested various parts of
their spatial memory (138).
These tests included tasks that measured their navigation
strategies, ability to create mental maps and their use of
landmarks while navigating in a virtual environment. The initial
results indicated that those who had used GPS more often in their
lives tended to have a worse spatial memory (138). That study also followed-up 13 of
these participants 3 years later and found that those who had used
GPS more frequently since the first test exhibited a more
significant reduction in their hippocampus-dependent spatial memory
over that time. Of note, they found that those who more often used
GPS, did not do so because they already felt they had a poor sense
of direction. That study suggests that the heavy use of GPS itself
may be causing a decline in spatial memory, rather than being a
symptom of it (138). These
results are particularly important as society continues to
increasingly depend on GPS technology.
Priming
This phenomenon demonstrates how prior exposure to
a stimulus can unconsciously influence the response to a later
related stimulus. Priming may be perceptual or conceptual.
Perceptual priming occurs when our prior encounter with an item
makes it easier to process or identify that same item again. When
an individual sees a specific image, the next time this is seen,
even briefly, it will be recognized faster as the brain has been
primed. In conceptual (or semantic) priming, exposure to an item
improves the ability to recall or process a related item. For
instance, if one hears the word doctor, one can more quickly
recognize or think of the word nurse, as the two concepts are
linked in one's mind. The neocortex is involved in both the
encoding of memories and their subsequent consolidation, through
the interaction with the hippocampus (139).
Classical conditioning
This is a type of associative learning involves the
process through which a neutral stimulus becomes linked to a
specific response after being repeatedly paired with a meaningful
one. A classic example is Pavlov's dogs, which learned to salivate
at the sound of a bell as they associated it with food (140).
Experiences often create powerful associations that
can unconsciously influence one's reactions. In the same manner as
exams can induce anxiety for someone, being chased or threatened by
a dog in childhood could create a lifelong fear of canines. These
are instances where a specific stimulus becomes linked to an
emotional response known as learned associations and emotional
responses. One might find oneself reaching for his/her own
smartphone when another phone nearby rings with a familiar tone.
This is known as conditioned reactions to stimuli. These automatic
reactions demonstrate how one can be conditioned to respond to
certain cues. A well-known illustration of this is John Watson's
‘Little Albert’ experiment (141). In that study, a baby who
initially had no fear of a white rat was conditioned to fear it.
Researchers repeatedly paired the presence of the white rat with a
loud, startling noise. Over time, the baby began to exhibit fear
solely at the sight of the rat, demonstrating how a neutral
stimulus can become associated with a powerful emotional reaction.
It was an example of classical conditioning.
Emotional memory
The amygdala is responsible for emotional
responses, such as pleasure, weeping and anger. It directly
influences emotional learning and assists memory processes in other
areas, such as the hippocampus and prefrontal cortex. The
interaction of emotion and memory takes place across different
stages of information processing, from the initial formation and
strengthening of memories to their later recall (41).
A notable feature of human memory is its ability to
enhance explicit, consciously recallable memories for emotional
experiences. Research, drawing from neuroimaging,
neuropsychological, pharmacological and neural stimulation studies,
suggests that emotional stimuli activate particular cognitive and
neural processes that improve explicit memory (142). Emotional arousal impacts memory
through factors active during the initial encoding stage (such as
attention and elaboration) and through elements that adjust memory
consolidation (143,144). The role of the amygdala has been
identified in strengthening explicit memory for both positive and
negative emotional stimuli by modulating both encoding and
consolidation (145,146).
While heightened emotional states typically lead to
improved memory in healthy individuals, it remains unclear whether
this effect holds true for patients with AD. A previous study aimed
to compare emotional memory and emotional responses in individuals
with early AD against a group of older adults without the condition
(147). Participants in both
groups viewed a mix of emotionally evocative (both pleasant and
unpleasant) and neutral images while their cognitive and
electrophysiological reactions were monitored. Memory was later
assessed using both free recall and recognition tasks. The findings
of that study indicated that while patients with AD displayed
normal emotional reactions, their emotional memory effect was
impaired; specifically, they failed to remember emotional stimuli
better than neutral ones (147).
Electrical stimulation and emotional
processing
The dorsolateral prefrontal cortex is generally
considered to play a role in emotional processing, yet the precise
contribution of each hemisphere remains a topic of ongoing debate.
To investigate the unique role of the left dorsolateral prefrontal
cortex in the encoding and retrieval of emotional stimuli, a
previous study utilized unilateral transcranial direct current
stimulation (tDCS) in healthy individuals (148). A total of 42 right-handed
undergraduate students participated, receiving either anodal,
cathodal, or sham stimulation to their left dorsolateral prefrontal
cortex while viewing a series of neutral, pleasant and unpleasant
images. Following a filler task, participants were asked to recall
as many pictures as they could (148). The findings of that study
indicated that participants remembered more emotional pictures
(both pleasant and unpleasant) than neutral ones, irrespective of
the tDCS condition. Notably, those who received anodal stimulation
recalled significantly more pleasant images compared to
participants in the sham and cathodal conditions. No such
differences were observed for the recall of neutral or unpleasant
images. These results suggest that the left prefrontal cortex
contributes specifically to the encoding and retrieval of pleasant
stimuli (148).
Auditory cortex involvement in
emotional memory
Emotional memories are fundamental to human and
animal existence, shaping future decisions and actions.
Historically, brain lesion studies in animals have suggested the
involvement of the auditory cortex in emotional learning primarily
through processing auditory stimuli linked to emotional outcomes
and relaying this data to the amygdala (149). However, electrophysiological and
imaging research has revealed that emotional experiences induce
specific, associative, and long-lasting changes within the auditory
cortex itself. These findings indicate that the role of the
auditory cortex extends beyond simple stimulus processing and
transmission. Three important implications of these data have been
explored: i) The potential roles of the auditory cortex in
emotional learning; ii) its involvement in both early and late
stages of memory trace encoding; and iii) the functional
interaction between the auditory cortex and subcortical regions,
particularly the amygdala, which handles affective information. It
was concluded that the auditory cortex plays a more significant and
prominent role in emotional learning, particularly from the early
stages of memory encoding and through its connections with
subcortical nuclei, than previously understood (150).
The British philosopher, Gilbert Ryle, introduced
the important distinction between procedural and declarative memory
in his 1949 book, The Concept of Mind; Role of histamine in
emotional memory (151,152). As a key neuromodulator, histamine
responds to various emotional states, including stress, anxiety,
and reward-associated activity. We can therefore infer that
histamine is capable of encoding the emotional valence (the
intrinsic attractiveness/aversiveness) and salience (the
distinctiveness/importance) of stimuli. Consequently, histamine
appears to be significant in the consolidation and retrieval of
emotional memories, potentially through associative processes that
connect sensory experiences with the value of unconditioned stimuli
(153). Despite these insights,
the specific mechanisms by which histamine contributes to emotional
learning and memory are not yet fully understood. Future research
utilizing cutting-edge technologies, dissecting histaminergic
circuits, and employing spatiotemporally selective manipulation of
histamine receptors will be crucial for addressing these gaps and
for developing clinical strategies for emotional and memory
disorders.
Non-associative learning
This category includes changes in the response to a
single stimulus over time: Habituation is when a response to a
repeated stimulus decreases. For example, if one resides close to a
train track, eventually, that individual will stop noticing the
sound of passing trains. Sensitization is the opposite; it is an
increased response to a repeated stimulus. For example, when one is
anxious, a small unexpected noise may startle someone to a greater
extent than it usually would. Non-declarative memory is essential
for countless daily activities, allowing for the expert navigation
of one's surroundings and the mastering of intricate abilities
without the need for deliberate thought.
9. Neurogenesis and memory association
The hippocampus is associated with learning and
memory formation (154,155). However, researchers continue to
argue over its exact functional contributions. This uncertainty
arises from the involvement of hippocampal formation in various
processes, including spatial navigation (developing internal
cognitive maps), relational encoding (understanding the links
between different pieces of information), complex associations
(managing configural representations) and episodic memory (155).
Conceptual complexity arises in ongoing debate
regarding how specific subfields within the hippocampus contribute
to these functions. It is important to know that the dentate gyrus
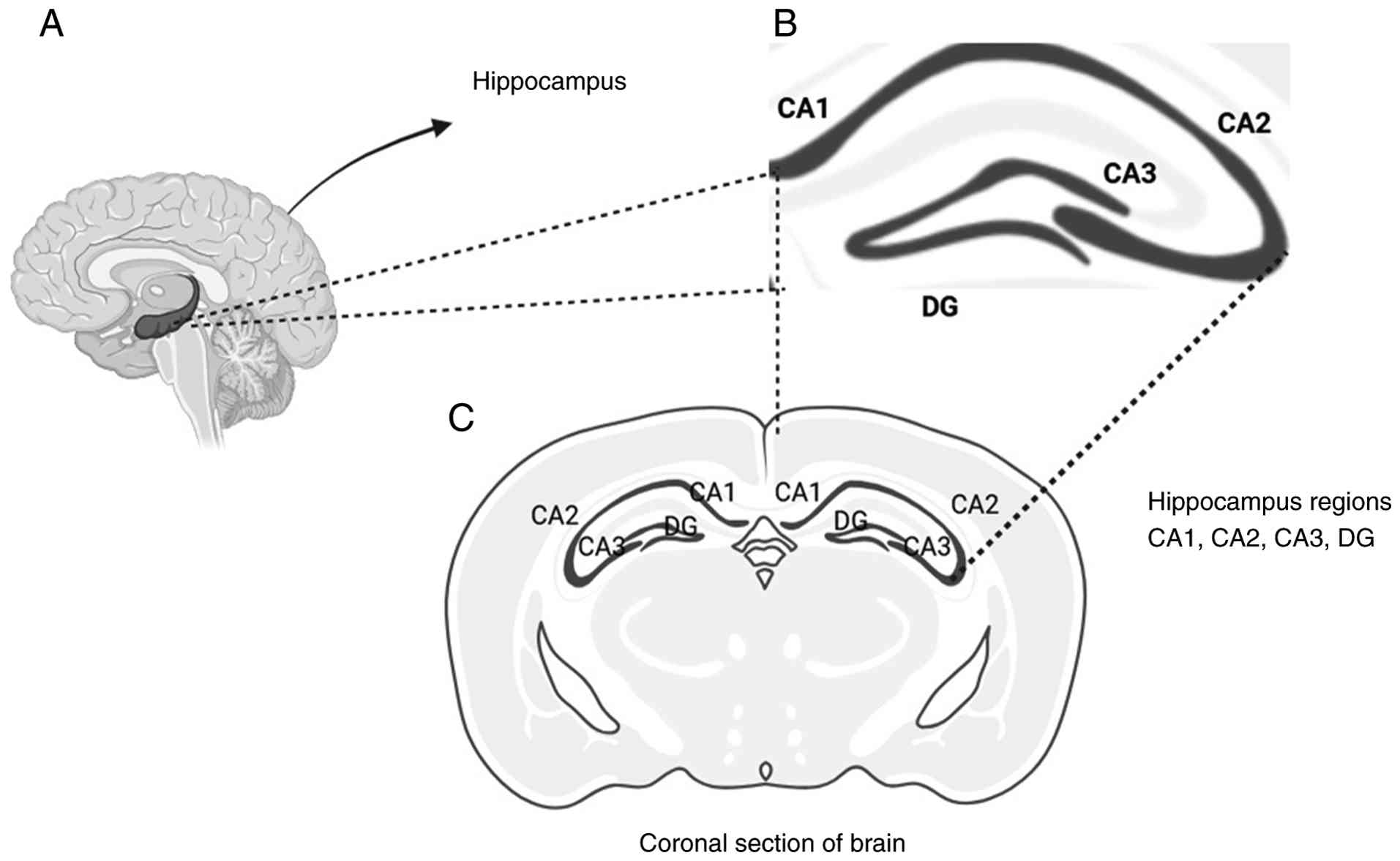
(DG), CA3, and CA1 are three primary regions of the hippocampus
that exhibit distinct properties. Specifically, the DG is unique
among them for its ability to generate new neurons throughout
adulthood, a process known as adult neurogenesis (156). The DG, CA3 and CA1 region in the
hippocampus is illustrated in Fig.
5.
The debate over adult hippocampal neurogenesis is
one of the most confrontational topics in modern neuroscience.
Rodents produce new neurons in the hippocampus throughout their
lives; the process, is known as adult hippocampal neurogenesis
(AHN), is a potent example of how mammalian brains remain adaptable
and plastic over time. Its existence in humans remain subjects of
intense disagreement among researchers (157,158). The current debate began in 2018
by two research groups that reached opposite conclusions using
similar post-mortem human tissue. i) Low neurogenesis view:
Sorrells et al (159)
argued that AHN sharply decreases during childhood and becomes
undetectable or extremely rare in adults. They suggested that the
human hippocampus may differ fundamentally from other mammals by
losing its neurogenic capacity early in life (160). ii) The persistent neurogenesis
view: By contrast, Boldrini et al (161) reported finding thousands of
immature neurons in the DG of humans across the lifespan, from the
age of 14 to 79 years. This view was later supported by
Moreno-Jiménez et al (162) who identified tens of thousands of
neuroblasts in healthy adult brains, although they noted a
significant decline in patients with AD. The controversy is
primarily driven by technical limitations in studying human brain
tissue: i) Post-mortem delay: Markers of immature neurons, such as
doublecortin (DCX), degrade rapidly after death. Critics of the no
neurogenesis view argue that long delays between death and tissue
fixation can make these markers vanish, leading to false negatives
(157,158). ii) Marker specificity: There is
debate over whether common markers such as DCX or PSA-NCAM
exclusively label new neurons or if they might also label
remodelling mature neurons or glial cells (163).
10. Memory modification technologies and
ethical considerations
Memory modification technologies (MMTs) are tools
that are being used to boost memory beyond its natural capacity.
While medical treatments focus on the restoration of memory loss.
Individuals have long used specific devices or technology to
enhance their memory. The current understanding of neuroscience
reveals that memory modulation can be achieved by either
pharmacological drugs, such as glutamate receptor agonists,
N-Methyl D-aspartic acid (NMDA), which can modulate memory by
enhancing neuroplasticity and facilitating memory consolidation, or
using neuromodulation techniques, such as deep brain stimulation
(DBS) (164,165). D-cycloserine (DCS) functions as a
partial agonist of the N-methyl-D-aspartate (NMDA) receptor,
potentially improving the outcomes of fear extinction and exposure
therapy. It achieves this either by boosting NMDA receptor activity
during the extinction process or by suppressing its function during
the initial consolidation of fear memories (166).
Previous research using animals demonstrated the
positive result of DBS on memory enhancement (167). The hippocampus is the first
region to undergo atrophy in dementia such as Alzheimer's disease
(AD). The hippocampus is a primary target for MMTs. DBS is
currently being refined as a clinical intervention for the early,
preclinical stages of the disease (168-170).
This technology is moving rapidly from laboratory research to human
application, with several clinical trials already assessing its
safety and efficacy. The hippocampus of the brain acts as a relay
station for turning short-term perceptions into long-term memories.
In AD this area is uniquely vulnerable due to deposition of
neurofibrillary tangles and amyloid plaques (171). Volume loss involves the visible
shrinking (atrophy) that is associated with the severity of memory
loss (172).
Despite its promise, DBS faces significant hurdles.
Phase II clinical trials for AD have yielded inconsistent results,
while patients >65 years of age exhibited a temporary
attenuation in cognitive decline, these improvements typically
vanished after 12 months, with overall memory continuing to
deteriorate (173). Alarmingly,
research suggests that applying DBS to patients <65 years of age
may inadvertently accelerate memory impairment (174). Furthermore, research has
documented paradoxical memory deficits following stimulation in
specific contexts. While non-invasive neuromodulation is being
explored as an alternative, DBS currently retains an advantage in
spatial and temporal specificity, offering more precise control
over neural circuits (175,176). However, this lead is being
challenged by emerging technologies like focused ultrasound, which
can non-invasively stimulate or suppress deep-brain activity with
high accuracy (177). Despite
these technological leaps, several foundational challenges remain.
i) Unknown mechanisms: The precise cellular mechanism that drive
memory changes during stimulation are not yet fully understand. ii)
Limited efficacy: Current outcomes are often modest and vary
significantly between individuals. iii) Developmental barriers:
Both invasive and non-invasive methods face rigorous regulatory and
safety hurdles that slow their translation to clinical use.
Nevertheless, these tools represent a frontier in cognitive
science, offering the potential to enhance human memory far beyond
the capabilities of traditional mnemonic strategies.
Modern strategies for memory improvement often
target synaptic plasticity through the stimulation of neurogenesis
or synaptogenesis. Other approaches focus on the modulation of
neurotransmitter systems, specifically their transmission and
receptor sensitivity, or the disinhibition of neural circuits
essential for memory formation. The most widely accepted mechanism
for these interventions is LTP. This process involves a sustained
increase in synaptic strength triggered by the simultaneous
activation of pre- and post-synaptic neurons. Theoretically, MMTs
could be engineered to improve the efficiency of LTP, thereby
sharpening overall cognitive performance.
A significant hurdle in developing these
technologies is the lack of anatomical and functional specificity.
As LTP occurs across virtually all excitatory pathways in the
hippocampus, a generalized boost to this process could lead to
unintended consequences: i) Memory overgeneralization: Instead of
enhancing only useful information, MMTs may inadvertently
strengthen every sensory input, rendering it difficult for the
brain to distinguish important memories from trivial noise. ii)
Pathological reinforcement: Global increases in plasticity can
exacerbate conditions such as addiction, where drug-related cues
become hyper-salient, or anxiety disorders, where traumatic
associations are strengthened rather than extinguished. iii)
Cognitive clutter: Enhancing synaptic excitability indiscriminately
may interfere with the natural ability of the brain to prune
unnecessary connections, potentially leading to cognitive
interference.
The current research progress in memory-related
neuroscience has transitioned from theoretical observation toward
the active modulation of mnemonic traces. Recent experimental
successes in animal models, specifically regarding the induction of
false memories (178,179) the suppression or silencing of
specific engrams (180,181), and the recalibration of emotional
valences (182), signal a
forthcoming experiment toward clinical human applications. However,
as the scientific community pursues these cognitive optimizations
through rigorous experimentation, the technical feasibility of
memory manipulation must be balanced against its profound ethical
ramifications (183). As MMTs,
ranging from pharmacological drugs to neural implants, move from
theory to reality, they raise several critical ethical dilemmas.
The core of the debate is a conflict between two major
philosophical perspectives: i) Transhumanism: This argues that
individuals have a moral obligation to use technology to overcome
biological limitations and improve human well-being (184). ii) Bioconservatism: This argues
that humans should preserve human nature and that essential
cognitive changes risk destroying the important things that make
life meaningful (e.g., vulnerability, effort and authentic
history).
11. Exercise, health and memory
Regular physical activity can be considered a key
lifestyle factor that enhances neurocognitive functions and brain
plasticity. It may also help reduce the risk of cognitive decline
linked to AD (185).
Research in animals consistently demonstrates that
regular exercise promotes the growth of new brain cells, a process
called neurogenesis, in the adult hippocampus. This, in turn,
improves learning and memory (186). While neurogenesis in the adult
human hippocampus has been frequently suggested (161,187), it has also faced recent
questioning (159).
A growing body of evidence indicates that
hippocampal synaptic plasticity (the ability of brain connections
to strengthen or weaken) is at least partly influenced by BDNF
(188). Physical exercise
increases gene expression of BNDF (mRNA and protein levels of BDNF)
in the hippocampus and other brain areas (188). Conversely, in the event that the
action of BDNF in the hippocampus is blocked, the positive effects
of exercise on memory are hindered (189).
The changes in BDNF levels related to exercise are
typically rapid and temporary (190). Specifically, BDNF levels rapidly
increase in hippocampal subfields following exercise, along with
improvements in LTP and synaptic plasticity (191,192). These rapid changes may contribute
to memory enhancement that lasts for a few hours.
Regular physical activity is a powerful tool for
improving brain function. It boosts memory and strengthens synaptic
plasticity within the hippocampus, a brain region critical for
learning and memory. Exercise also increases levels of BDNF, a
protein vital for neuron growth and survival.
Even short bursts of exercise, known as acute
exercise, have been shown to benefit hippocampal plasticity in
rodents. This is partly due to the release of endocannabinoids,
particularly anandamide (AEA), and an increase in BDNF. However, it
has not been clear if a single exercise session has similar effects
on BDNF and AEA levels in humans, and if these changes, in turn,
affect memory. To investigate this, a previous study was conducted
using blood biomarkers, behavioural tests, and fMRI measurements in
healthy male volunteers (193).
The researchers assessed how a single exercise session impacted
associative memory and the underlying brain mechanisms. Each
participant underwent memory tests after three different
conditions: rest, moderate-intensity exercise, or high-intensity
exercise. A long-term memory retest was conducted three months
later. The findings were significant: Both immediately after the
exercise and at the three-month retest, memory performance was
better after moderate-intensity exercise compared to rest. This
improvement in memory after moderate exercise was linked to an
increase in both AEA and BDNF levels. Specifically, AEA was
associated with hippocampal activity during memory recall, while
BDNF was found to enhance hippocampal memory representations and
long-term memory performance (193). These results demonstrate that a
single session of moderate-intensity exercise can indeed improve
the consolidation of hippocampal memory. That study suggests that
the signalling pathways involving endocannabinoids and BDNF likely
play a synergistic role in modulating the underlying neural
plasticity mechanisms of the brain (193).
12. Association between diet and memory
The connection between diet and memory is
bidirectional. Consuming a high-calorie diet and becoming obese can
harm both interoceptive processing and memory function in the
hippocampus of humans and rodents. These negative changes in brain
function can then contribute to even more excessive eating,
creating a self-perpetuating cycle that makes it difficult to
maintain a healthy weight (194).
There is a clear, bidirectional association between
eating habits and memory. Research in both humans and rodents shows
that a high-energy diet and obesity impair cognitive abilities,
specifically memory and the function of the hippocampus (195). This cognitive decline may, in
turn, lead to continued overconsumption, thereby initiating a
harmful cycle that encourages unhealthy weight gain.
Research in mice has identified specific neural
circuits that suppress food intake, originating from both the
ventral hippocampus (196) and
dorsal hippocampus (197) and
projecting to different septal nuclei. Since the vHC is primarily
linked to emotional and food-related memories and the dHC to
episodic and spatial memories, both regions likely contribute to
the memory of an eating episode. This shared memory representation
may then inhibit subsequent food consumption through both common
and unique mechanisms. Furthermore, memory is not contained in a
single area but is a composite of information from multiple brain
regions, each contributing different aspects of the same experience
(198,199). While most research in rodents
focuses on the hippocampus, other areas within the ‘core memory
network’ may also connect memory and eating. Future studies should
particularly investigate areas, such as the angular gyrus, that are
altered in human obesity (200).
The dried root of Rhodiola rosea (R.
rosea) was previously found to improve the ability of an insect
to link odours and tastes with rewards, and it also prevented the
typical memory decline associated with aging in adult flies. When
administered to larval Drosophila, dried R. rosea
root material improved their odour-taste reward associative memory
in a dose-dependent manner (201). It was also shown to prevent a
similar, age-related decline in this type of appetitive memory in
adult flies. The substance not only improved memory scores in
larvae but also mitigated the decline of this memory as the adult
flies aged (201).
A recent study investigated whether the human
tendency to remember the location of high-calorie foods more
accurately than low-calorie foods is consistent across different
cultures. Researchers conducted an online spatial memory test with
participants from the USA and Japan, and compared the results to a
previous study with a Dutch population (202). The findings revealed that
individuals in all three countries displayed this high-calorie
memory bias. That study concluded that the strength of this
cognitive trait is similar across diverse populations and is not
significantly influenced by sociodemographic factors (202).
Dietary magnesium (Mg2+) can improve
memory in both young and aging rats, primarily by increasing
synaptic density in the hippocampus and raising the expression of
the NR2B subunit of the NMDA receptor (203). A recent study in fruit flies
(Drosophila) found that magnesium also enhanced their
long-term memory. Notably, this effect in flies appears to be
independent of NMDA receptors in the mushroom body (204). Instead, it relies on a conserved
Mg2+ efflux transporter encoded by the unextended
(uex) gene. This gene is crucial for normal memory function and
contains a cyclic nucleotide-binding domain. The location of UEX is
altered in flies with mutations in cAMP-related genes, which are
known to cause memory deficits. Imaging suggests that UEX-dependent
efflux is necessary for the rhythmic maintenance of Mg2+
in Kenyon cells (204). This
research suggests that regulated neuronal magnesium efflux is vital
for both normal memory and memory enhanced by magnesium
supplementation.
13. Conclusion and future directions
The conclusion of modern memory research highlights
that memory is both a biological event (involving synaptic
plasticity and the hippocampus) and a cognitive feat. Memory is
supported by specialized circuits; the hippocampal-cortical network
directs declarative memories (facts and events), whereas the
auditory cortex, basal ganglia, and cerebellum facilitate
non-declarative skills and habits. Math skills are more associated
with declarative memory compared to nondeclarative memory. The
auditory cortex plays a more significant and prominent role in
emotional learning, particularly from the early stages of memory
encoding and through its connections with subcortical nuclei
amygdala. The role of the histamine neurotransmitter in emotional
memories has been evaluated. The intricate workings of the
hippocampus and entorhinal cortex navigate the spatial dimensions
of one's world. The hippocampus and entorhinal cortex generate the
spatial coordinates of the map. The parahippocampal and
retrosplenial cortices integrate maps to stable physical landmarks,
and these spatial codes are integrated with frontal lobe processes
to transform a mental map into a navigation plan.
Despite significant advances, a number of gaps and
future possibilities remain. While there is a strong understanding
of how the hippocampus lays down new memories, the precise
mechanisms by which they are consolidated and transferred to the
neocortex for long-term storage are still being elucidated.
Researchers are also finding the complexity between emotion and
memory, moving beyond general associations to understand how
specific emotional states, such a fear, can compress or distort our
perception of time and space in memory. Furthermore, the role of
sleep in memory consolidation is an emerging field, with new
research confirming its critical function in solidifying learned
information and skills.
In the case of short-term memory, brief bouts of
cardiovascular exercise (referred to as acute exercise) exert a
moderate positive effect. Acute exercise and long-term exercise,
both types of exercise, can benefit short-term memory. Acute
exercise may be particularly effective for boosting long-term
memory (205). Researchers have
also explored whether any exercise-induced changes in BDNF and VEGF
levels are linked to memory improvements (206). However, how acute exercise
affects emotional memory is less clear. Acute exercise appears to
be linked to increased memory confidence and accuracy for the most
important details of emotional stimuli. Additionally, certain
exercise types, such as treadmill exercise, are associated with a
higher occurrence of memory intrusions. Aerobic exercise training
can indeed reverse hippocampal volume loss in late adulthood, and
this positive change is accompanied by improved memory function. It
is a powerful reminder of how movement can directly impact brain
health. Acute exercise appears to be linked to increased memory
confidence and accuracy for the most important details of emotional
stimuli. Additionally, certain types of exercise, such as treadmill
exercise, are associated with a higher occurrence of memory
intrusions (207). The
hippocampus, a crucial brain region for memory, naturally shrinks
(84). This decline can lead to
impaired memory and a heightened risk of conditions like dementia
(208).
Advanced neuroimaging techniques will continue to
map the dynamic brain networks responsible for different memory
types in real-time. As of early 2026, researchers are trying to
analyse the intersection of neuro-engineering, AI and cognitive
psychology. Researchers are finding ways to write and edit memories
using high technology tools such as optometry. Using light to
activate specific neurons (engrams), researchers can now activate
or suppress specific memories in animal models. Future research is
required to focus on translating these findings into non-invasive
therapies for humans with post-traumatic stress disorder or severe
phobias. A previous study revealed that slow gamma oscillations (40
hertz) are associated with the retrieval of spatial memory
(209). The researchers used
optogenetics in an Alzheimer model of mice to activate medial
septal parvalbumin neurons and found that optogenetic stimulation
restores hippocampal slow gamma oscillations amplitude, and
phase-amplitude coupling in mice (210).
When a memory is recalled, it becomes labile
(changeable) for a few hours. Research is investigating how to
safely intervene during this window to reduce the emotional pain
associated with traumatic memories. Extensive research is being
conducted on how our reliance on smartphones (external memory) is
physically reshaping our brains. A critical question is whether
humans are losing storage capacity, but gaining retrieval
efficiency. A new concept termed the ‘Third Way of Memory’ is
emerging. Generative AI can create photos, videos, or chatbots of
deceased loved ones. Researchers are studying how these ‘simulated
memories’ affect the grieving process and our collective sense of
history (211,212).
Brain-computer interfaces (BCIs) are being
developed to monitor brain signals in real-time. For patients with
AD, these devices could detect a ‘memory lapse’ and provide a tiny
electrical pulse to help the brain bridge the gap. Clinical trials
are exploring whether DBS techniques can enhance memory retrieval
in patients with traumatic brain injuries or early-stage dementia.
Emerging studies suggest our gut health directly impacts
neurogenesis in the hippocampus. Future research is looking into
‘psychobiotics’ dietary interventions designed specifically to
boost memory and slow cognitive decline.
Acknowledgements
The authors would like to thank Sharda University
(Greater Noida, India) for providing valuable resources, such as
the central library and internet resources for the present
study.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
RY and SK conceptualized the study and prepared the
original draft of the manuscript. AAC and RM reviewed and edited
the manuscript. RY and SK supervised the study. RY was involved in
figure illustration. All authors have read and agreed to the
published version of the manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zlotnik G and Vansintjan A: Memory: An
extended definition. Front Psychol. 10(2523)2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Moriarty O, McGuire BE and Finn DP: The
effect of pain on cognitive function: A review of clinical and
preclinical research. Prog Neurobiol. 93:385–404. 2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Low LA: The impact of pain upon cognition:
What have rodent studies told us? Pain. 154:2603–2605.
2013.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Liu X, Li L, Tang F, Wu S and Hu Y: Memory
impairment in chronic pain patients and the related
neuropsychological mechanisms: A review. Acta Neuropsychiatr.
26:195–201. 2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lazzarim MK, Targa A, Sardi NF, Hack GR,
Tobaldini G, Martynhak BJ and Fischer L: Pain impairs
consolidation, but not acquisition or retrieval of a declarative
memory. Behav Pharmacol. 31:707–715. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Squire LR and Wixted JT: The Cognitive
Neuroscience of Human Memory Since H.M. Annu Rev Neurosci.
34:259–288. 2011.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Eysenck MW: Attention and Arousal:
Cognition and Performance. 1st Edition, 1982. Springer Nature
Link.
|
|
8
|
Costa-Mattioli M and Sonenberg N:
Translational control of gene expression: A molecular switch for
memory storage. Prog Brain Res. 169:81–95. 2008.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Meyer D, Bonhoeffer T and Scheuss V:
Balance and stability of synaptic structures during synaptic
plasticity. Neuron. 82:430–443. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Duke CG, Kennedy AJ, Gavin CF, Day JJ and
Sweatt JD: Experience-dependent epigenomic reorganization in the
hippocampus. Learn Mem. 24:278–288. 2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Frankland PW, Bontempi B, Talton LE,
Kaczmarek L and Silva AJ: The involvement of the anterior cingulate
cortex in remote contextual fear memory. Science. 304:881–883.
2004.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Day JJ and Sweatt JD: DNA methylation and
memory formation. Nat Neurosci. 13:1319–1323. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Halder R, Hennion M, Vidal RO, Shomroni O,
Rahman RU, Rajput A, Centeno TP, van Bebber F, Capece V, Garcia
Vizcaino JC, et al: DNA methylation changes in plasticity genes
accompany the formation and maintenance of memory. Nat Neurosci.
19:102–110. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kim M and Costello J: DNA methylation: An
epigenetic mark of cellular memory. Exp Mol Med.
49(e322)2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Takakura M, Lam YH, Nakagawa R, Ng MY, Hu
X, Bhargava P, Alia AG, Gu Y, Wang Z, Ota T, et al: Differential
second messenger signaling via dopamine neurons bidirectionally
regulates memory retention. Proc Natl Acad Sci USA.
120(e2304851120)2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wixted JT: The psychology and neuroscience
of forgetting. Annu Rev Psychol. 55:235–269. 2004.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Davis RL and Zhong Y: The biology of
forgetting-a perspective. Neuron. 95:490–503. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
McGaugh JL: Memory-a century of
consolidation. Science. 287:248–251. 2000.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Yin JC, Wallach JS, Del Vecchio M, Wilder
EL, Zhou H, Quinn WG and Tully T: Induction of a dominant negative
CREB transgene specifically blocks long-term memory in Drosophila.
Cell. 79:49–58. 1994.PubMed/NCBI View Article : Google Scholar
|
|
20
|
McGuire SE, Deshazer M and Davis RL:
Thirty years of olfactory learning and memory research in
Drosophila melanogaster. Prog Neurobiol. 76:328–347.
2005.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Dubnau J, Grady L, Kitamoto T and Tully T:
Disruption of neurotransmission in Drosophila mushroom body blocks
retrieval but not acquisition of memory. Nature. 411:476–480.
2001.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Schwaerzel M, Monastirioti M, Scholz H,
Friggi-Grelin F, Birman S and Heisenberg M: Dopamine and octopamine
differentiate between aversive and appetitive olfactory memories in
Drosophila. J Neurosci. 23:10495–10502. 2003.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Kim YC, Lee HG and Han KA: D1 dopamine
receptor dDA1 is required in the mushroom body neurons for aversive
and appetitive learning in Drosophila. J Neurosci. 27:7640–7647.
2007.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Shuai Y, Lu B, Hu Y, Wang L, Sun K and
Zhong Y: Forgetting is regulated through Rac activity in
Drosophila. Cell. 140:579–589. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhang X, Li Q, Wang L, Liu ZJ and Zhong Y:
Active protection: Learning-activated Raf/MAPK activity protects
labile memory from rac1-independent forgetting. Neuron.
98:142–155.e4. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Yagishita S, Hayashi-Takagi A,
Ellis-Davies GC, Urakubo H, Ishii S and Kasai H: A critical time
window for dopamine actions on the structural plasticity of
dendritic spines. Science. 345:1616–1620. 2014.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Hige T, Aso Y, Modi MN, Rubin GM and
Turner GC: Heterosynaptic plasticity underlies aversive olfactory
learning in Drosophila. Neuron. 88:985–998. 2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Cohn R, Morantte I and Ruta V: Coordinated
and compartmentalized neuromodulation shapes sensory processing in
Drosophila. Cell. 163:1742–1755. 2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Crocker A, Guan XJ, Murphy CT and Murthy
M: Cell-type-specific transcriptome analysis in the Drosophila
mushroom body reveals memory-related changes in gene expression.
Cell Rep. 15:1580–1596. 2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wang W, Wang Z, Cao J, Dong Y and Chen Y:
Roles of Rac1-dependent intrinsic forgetting in memory-related
brain disorders: demon or angel. Int J Mol Sci.
24(10736)2023.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Wixted JT: Atkinson and Shiffrin's (1968)
influential model overshadowed their contemporary theory of human
memory. J Mem Lang. 136(104471)2024.
|
|
32
|
Tripathy SP and Öǧmen H: Sensory memory is
allocated exclusively to the current event-segment. Front Psychol.
9(1435)2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Cascella M and Khalili YA: Short-Term
Memory Impairment. In: StatPearls. StatPearls Publishing, Treasure
Island, FL, 2024.
|
|
34
|
Baddeley A: Working Memory, Thought, and
Action. In: Oxford Psychology. Oxford University Press, 2007.
|
|
35
|
Talamini F, Blain S, Ginzburg J, Houix O,
Bouchet P, Grassi M, Tillmann B and Caclin A: Auditory and visual
short-term memory: influence of material type, contour, and musical
expertise. Psychol Res. 86(421)2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Duan F, Yan X, Wang J, Wu Z, Zhang Y, Shu
Q, Liu F, Xu F and Han Q: Short-term memory retrieval enhances
brain functional connectivity. Front Behav Neurosci.
19(1578415)2025.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Chiyohara S, Furukawa JI, Noda T, Morimoto
J and Imamizu H: Proprioceptive short-term memory in passive motor
learning. Sci Rep. 13(20826)2023.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Anderson JR: Language, Memory, and
Thought. In: Erlbaum Associates. Halsted Press Division of Wiley,
New York, NY, 1976.
|
|
39
|
Tulving E and Markowitsch HJ: Episodic and
declarative memory: Role of the hippocampus. Hippocampus.
8:198–204. 1998.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Tolman EC: Cognitive maps in rats and men.
Psychol Rev. 55:189–208. 1948.PubMed/NCBI View Article : Google Scholar
|
|
41
|
LaBar KS and Cabeza R: Cognitive
neuroscience of emotional memory. Nat Rev Neurosci. 7:54–64.
2006.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Drachman DA and Arbit J: Memory and the
hippocampal complex. II. Is memory a multiple process? Arch Neurol.
15:52–61. 1966.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Wang SH and Morris RG:
Hippocampal-neocortical interactions in memory formation,
consolidation, and reconsolidation. Annu Rev Psychol. 61:49–79,
C1-4. 2010.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Mednick SC, Cai DJ, Shuman T, Anagnostaras
S and Wixted JT: An opportunistic theory of cellular and systems
consolidation. Trends Neurosci. 34:504–514. 2011.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Frankland PW and Bontempi B: The
organization of recent and remote memories. Nat Rev Neurosci.
6:119–130. 2005.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Takashima A, Nieuwenhuis IL, Jensen O,
Talamini LM, Rijpkema M and Fernández G: Shift from hippocampal to
neocortical centered retrieval network with consolidation. J
Neurosci. 29:10087–10093. 2009.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Abel M, Nickl AT, Reßle A, Unger C and
Bäuml KT: The role of sleep for memory consolidation: does sleep
protect memories from retroactive interference? Psychon Bull Rev.
30:2296–2304. 2023.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Stern SA and Alberini CM: Mechanisms of
memory enhancement. Wiley Interdiscip Rev Syst Biol Med. 5:37–53.
2013.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Riedel G, Platt B and Micheau J: Glutamate
receptor function in learning and memory. Behav Brain Res.
140:1–47. 2003.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Giese KP, Fedorov NB, Filipkowski RK and
Silva AJ: Autophosphorylation at Thr286 of the alpha
calcium-calmodulin kinase II in LTP and learning. Science.
279:870–873. 1998.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Squire LR, Genzel L, Wixted JT and Morris
RG: Memory consolidation. Cold Spring Harb Perspect Biol.
7(a021766)2015.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Adams JP and Dudek SM: Late-phase
long-term potentiation: Getting to the nucleus. Nat Rev Neurosci.
6:737–743. 2005.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Lonze BE and Ginty DD: Function and
regulation of CREB family transcription factors in the nervous
system. Neuron. 35:605–623. 2002.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Kandel ER: The molecular biology of
memory: cAMP, PKA, CRE, CREB-1, CREB-2, and CPEB. Mol Brain.
5(14)2012.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Popoli M, Yan Z, McEwen BS and Sanacora G:
The stressed synapse: the impact of stress and glucocorticoids on
glutamate transmission. Nat Rev Neurosci. 13:22–37. 2011.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Alberini CM and Kandel ER: The regulation
of transcription in memory consolidation. Cold Spring Harb Perspect
Biol. 7(a021741)2014.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Dinevska M, Widodo SS, Cook L, Stylli SS,
Ramsay RG and Mantamadiotis T: CREB: A multifaceted transcriptional
regulator of neural and immune function in CNS tumors. Brain Behav
Immun. 116:140–149. 2024.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Liao D, Hessler NA and Malinow R:
Activation of postsynaptically silent synapses during
pairing-induced LTP in CA1 region of hippocampal slice. Nature.
375:400–404. 1995.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Bahdar ZI, Abu-El-Rub E, Almazari R,
Alzu'bi A and Al-Zoubi RM: The molecular mechanism of nitric oxide
in memory consolidation and its role in the pathogenesis of
memory-related disorders. Neurogenetics. 26(22)2025.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Ghasemi M: Nitric oxide: Antidepressant
mechanisms and inflammation. Adv Pharmacol. 86:121–152.
2019.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Jomova K, Raptova R, Alomar SY, Alwasel
SH, Nepovimova E, Kuca K and Valko M: Reactive oxygen species,
toxicity, oxidative stress, and antioxidants: Chronic diseases and
aging. Arch Toxicol. 97:2499–2574. 2023.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Squire LR and Zola SM: Structure and
function of declarative and nondeclarative memory systems. Proc
Natl Acad Sci USA. 93:13515–13522. 1996.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Goto A: Synaptic plasticity during systems
memory consolidation. Neurosci Res. 183:1–6. 2022.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Bon CL and Garthwaite J: On the role of
nitric oxide in hippocampal long-term potentiation. J Neurosci.
23:1941–1948. 2003.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Hardingham N, Dachtler J and Fox K: The
role of nitric oxide in pre-synaptic plasticity and homeostasis.
Front Cell Neurosci. 7(190)2013.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Malenka RC, Lancaster B and Zucker RS:
Temporal limits on the rise in postsynaptic calcium required for
the induction of long-term potentiation. Neuron. 9:121–128.
1992.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Wittenborn EC and Marletta MA: Structural
perspectives on the mechanism of soluble guanylate cyclase
activation. Int J Mol Sci. 22(5439)2021.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Graf P and Schacter DL: Implicit and
explicit memory for new associations in normal and amnesic
subjects. J Exp Psychol Learn Mem Cogn. 11:501–518. 1985.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Siew CSQ, Wulff DU, Beckage NM and Kenett
YN: Cognitive Network Science: A review of research on cognition
through the lens of Network representations, processes, and
dynamics. In: Complexity. 2019. Available from: https://onlinelibrary.wiley.com/doi/10.1155/2019/2108423.
|
|
70
|
Castro N, Stella M and Siew CSQ:
Quantifying the interplay of semantics and phonology during
failures of word retrieval by people with aphasia using a multiplex
lexical network. Cogn Sci. 44(e12881)2020.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Wulff DU, De Deyne S, Jones MN and Mata R:
Aging Lexicon Consortium. New perspectives on the aging lexicon.
Trends Cogn Sci. 23:686–698. 2019.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Wulff DU, De Deyne S, Aeschbach S and Mata
R: Using network science to understand the aging lexicon: Linking
individuals' experience, semantic networks, and cognitive
performance. Top Cogn Sci. 14:93–110. 2022.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Kenett YN, Ungar L and Chatterjee A:
Beauty and wellness in the semantic memory of the beholder. Front
Psychol. 12(696507)2021.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Davis SW, Geib BR, Wing EA, Wang WC,
Hovhannisyan M, Monge ZA and Cabeza R: Visual and semantic
representations predict subsequent memory in perceptual and
conceptual memory tests. Cereb Cortex. 31:974–992. 2021.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Morton NW, Zippi EL, Noh SM and Preston
AR: Semantic knowledge of famous people and places is represented
in hippocampus and distinct cortical networks. J Neurosci.
41:2762–2779. 2021.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Szpunar KK: Episodic future thought: An
emerging concept. Perspect Psychol Sci. 5:142–162. 2010.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Schacter DL and Addis DR: The cognitive
neuroscience of constructive memory: Remembering the past and
imagining the future. Philos Trans R Soc B Biol Sci. 362:773–786.
2007.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Chao OY, de Souza Silva MA, Yang YM and
Huston JP: The medial prefrontal cortex - hippocampus circuit that
integrates information of object, place and time to construct
episodic memory in rodents: behavioral, anatomical and
neurochemical properties. Neurosci Biobehav Rev. 113:373–407.
2020.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Buckner RL: Memory and executive function
in aging and AD: Multiple factors that cause decline and reserve
factors that compensate. Neuron. 44:195–208. 2004.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Salthouse TA: When does age-related
cognitive decline begin? Neurobiol Aging. 30:507–514.
2009.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Park DC and Reuter-Lorenz P: The adaptive
brain: Aging and neurocognitive scaffolding. Annu Rev Psychol.
60:173–196. 2009.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Fjell AM, Sneve MH, Storsve AB, Grydeland
H, Yendiki A and Walhovd KB: Brain events underlying episodic
memory changes in aging: A longitudinal investigation of structural
and functional connectivity. Cereb Cortex. 26:1272–1286.
2016.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Damoiseaux JS, Viviano RP, Yuan P and Raz
N: Differential effect of age on posterior and anterior hippocampal
functional connectivity. NeuroImage. 133:468–476. 2016.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Salami A, Pudas S and Nyberg L: Elevated
hippocampal resting-state connectivity underlies deficient
neurocognitive function in aging. Proc Natl Acad Sci USA.
111:17654–17659. 2014.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Allen G, Barnard H, McColl R, Hester AL,
Fields JA, Weiner MF, Ringe WK, Lipton AM, Brooker M, McDonald E,
et al: Reduced hippocampal functional connectivity in Alzheimer
disease. Arch Neurol. 64:1482–1487. 2007.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Rosen AC, Prull MW, Gabrieli JD, Stoub T,
O'Hara R, Friedman L, Yesavage JA and deToledo-Morrell L:
Differential associations between entorhinal and hippocampal
volumes and memory performance in older adults. Behav Neurosci.
117:1150–1160. 2003.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Manenti R, Cotelli M, Robertson IH and
Miniussi C: Transcranial brain stimulation studies of episodic
memory in young adults, elderly adults and individuals with memory
dysfunction: A review. Brain Stimul. 5:103–109. 2012.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Simons JS and Spiers HJ: Prefrontal and
medial temporal lobe interactions in long-term memory. Nat Rev
Neurosci. 4:637–648. 2003.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Bai F, Zhang Z, Watson DR, Yu H, Shi Y,
Yuan Y, Zang Y, Zhu C and Qian Y: Abnormal functional connectivity
of hippocampus during episodic memory retrieval processing network
in amnestic mild cognitive impairment. Biol Psychiatry. 65:951–958.
2009.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Vidal-Piñeiro D, Valls-Pedret C,
Fernández-Cabello S, Arenaza-Urquijo EM, Sala-Llonch R, Solana E,
Bargalló N, Junqué C, Ros E and Bartrés-Faz D: Decreased default
mode network connectivity correlates with age-associated structural
and cognitive changes. Front Aging Neurosci. 6(256)2014.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Martins AR, Fregni F, Simis M and Almeida
J: Neuromodulation as a cognitive enhancement strategy in healthy
older adults: Promises and pitfalls. Neuropsychol Dev Cogn B Aging
Neuropsychol Cogn. 24:158–185. 2017.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Almeida J, Martins AR, Amaral L, Valério
D, Bukhari Q, Schu G, Nogueira J, Spínola M, Soleimani G, Fernandes
F, et al: The cerebellum is causally involved in episodic memory
under aging. Geroscience. 45:2267–2287. 2023.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Duff N, Olsen R, Walsh Z, Salmon K, Hunt M
and Macaskill A: A fragile effect: The influence of episodic memory
on delay discounting. Q J Exp Psychol (Hove). 78:514–533.
2025.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Garzón B, Kurth-Nelson Z, Bäckman L,
Nyberg L and Guitart-Masip M: Investigating associations of delay
discounting with brain structure, working memory, and episodic
memory. Cereb Cortex. 33:1669–1678. 2023.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Critchfield TS and Kollins SH: Temporal
discounting: Basic research and the analysis of socially important
behavior. J Appl Behav Anal. 34:101–122. 2001.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Rung JM and Madden GJ: Experimental
reductions of delay discounting and impulsive choice: A systematic
review and meta-analysis. J Exp Psychol Gen. 147:1349–1381.
2018.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Liu L, Feng T, Chen J and Li H: The value
of emotion: How does episodic prospection modulate delay
discounting? PLoS One. 8(e81717)2013.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Daniel TO, Said M, Stanton CM and Epstein
LH: Episodic future thinking reduces delay discounting and energy
intake in children. Eat Behav. 18:20–24. 2015.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Stein JS, Wilson AG, Koffarnus MN, Daniel
TO, Epstein LH and Bickel WK: Unstuck in time: Episodic future
thinking reduces delay discounting and cigarette smoking.
Psychopharmacology (Berl). 233:3771–3778. 2016.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Daniel TO, Stanton CM and Epstein LH: The
future is now: Comparing the effect of episodic future thinking on
impulsivity in lean and obese individuals. Appetite. 71:120–125.
2013.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Dassen FCM, Jansen A, Nederkoorn C and
Houben K: Focus on the future: Episodic future thinking reduces
discount rate and snacking. Appetite. 96:327–332. 2016.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Tulving E: Episodic memory: From mind to
brain. Annu Rev Psychol. 53:1–25. 2002.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Canada KL, Hancock GR and Riggins T:
Developmental changes in episodic memory across early- to
mid-childhood: Insights from a latent longitudinal approach.
Memory. 30:248–261. 2022.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Evans TM, Lipscomb DW, Earle FS, Del Tufo
SN, Lum JAG, Cutting LE and Ullman MT: Declarative memory supports
children's math skills: A longitudinal study. PLoS One.
19(e0304211)2024.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Reifegerste J, Veríssimo J, Rugg MD,
Pullman MY, Babcock L, Glei DA, Weinstein M, Goldman N and Ullman
MT: Early-life education may help bolster declarative memory in old
age, especially for women. Neuropsychol Dev Cogn B Aging
Neuropsychol Cogn. 28:218–252. 2021.PubMed/NCBI View Article : Google Scholar
|
|
106
|
St Laurent CW, Lokhandwala S, Allard T, Ji
A, Paluch A, Riggins T and Spencer RMC: Relations between 24-h
movement behaviors, declarative memory, and hippocampal volume in
early childhood. Sci Rep. 15(9205)2025.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Daum I and Ackermann H: Nondeclarative
memory-neuropsychological findings and neuroanatomic principles.
Fortschr Neurol Psychiatr. 65:122–132. 1997.PubMed/NCBI View Article : Google Scholar : (In German).
|
|
108
|
Harold PA: Handbook of Cerebrovascular
Diseases, Revised and Expanded. 2nd edition. CRC Press, Boca Raton,
FL, 2004.
|
|
109
|
Mizumori SJ, Puryear CB and Martig AK:
Basal ganglia contributions to adaptive navigation. Behav Brain
Res. 199:32–42. 2009.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Parent A: Extrinsic connections of the
basal ganglia. Trends Neurosci. 13:254–258. 1990.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Haber SN and Knutson B: The reward
circuit: linking primate anatomy and human imaging.
Neuropsychopharmacology. 35:4–26. 2010.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Bahrami S, Nordengen K, Rokicki J, Shadrin
AA, Rahman Z, Smeland OB, Jaholkowski PP, Parker N, Parekh P,
O'Connell KS, et al: The genetic landscape of basal ganglia and
implications for common brain disorders. Nat Commun.
15(8476)2024.PubMed/NCBI View Article : Google Scholar
|
|
113
|
DeLong MR and Wichmann T: Circuits and
circuit disorders of the basal ganglia. Arch Neurol. 64:20–24.
2007.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Redgrave P, Rodriguez M, Smith Y,
Rodriguez-Oroz MC, Lehericy S, Bergman H, Agid Y, DeLong MR and
Obeso JA: Goal-directed and habitual control in the basal ganglia:
Implications for Parkinson's disease. Nat Rev Neurosci. 11:760–772.
2010.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Yin HH and Knowlton BJ: The role of the
basal ganglia in habit formation. Nat Rev Neurosci. 7:464–476.
2006.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Grahn JA, Parkinson JA and Owen AM: The
cognitive functions of the caudate nucleus. Prog Neurobiol.
86:141–155. 2008.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Graybiel AM: Habits, rituals, and the
evaluative brain. Annu Rev Neurosci. 31:359–387. 2008.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Frank MJ: Dynamic dopamine modulation in
the basal ganglia: A neurocomputational account of cognitive
deficits in medicated and nonmedicated Parkinsonism. J Cogn
Neurosci. 17:51–72. 2005.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Beaunieux H, Eustache F, Busson P, de la
Sayette V, Viader F and Desgranges B: Cognitive procedural learning
in early Alzheimer's disease: Impaired processes and compensatory
mechanisms. J Neuropsychol. 6:31–42. 2012.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Pitel AL, Witkowski T, Vabret F,
Guillery-Girard B, Desgranges B, Eustache F and Beaunieux H: Effect
of episodic and working memory impairments on semantic and
cognitive procedural learning at alcohol treatment entry. Alcohol
Clin Exp Res. 31:238–248. 2007.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Beaunieux H, Desgranges B, Lalevée C, De
La Sayette V, Lechevalier B and Eustache F: Preservation of
cognitive procedural memory in a case of Korsakoff's Syndrome:
Methodological and theoretical insights. Percept Mot Skills. 86 (3
Pt 2):1267–1287. 1998.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Wang MZ and Hayden BY: Latent learning,
cognitive maps, and curiosity. Curr Opin Behav Sci. 38:1–7.
2021.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Burgess N: The 2014 nobel prize in
physiology or medicine: A spatial model for cognitive neuroscience.
Neuron. 84:1120–1125. 2014.PubMed/NCBI View Article : Google Scholar
|
|
124
|
O'Keefe J and Conway DH: Hippocampal place
units in the freely moving rat: Why they fire where they fire. Exp
Brain Res. 31:573–590. 1978.PubMed/NCBI View Article : Google Scholar
|
|
125
|
Edvardsen V, Bicanski A and Burgess N:
Navigating with grid and place cells in cluttered environments.
Hippocampus. 30:220–232. 2020.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Kumaran D and Maguire EA: The human
hippocampus: Cognitive maps or relational memory? J Neurosci.
25:7254–7259. 2005.PubMed/NCBI View Article : Google Scholar
|
|
127
|
Foster DJ and Knierim JJ: Sequence
learning and the role of the hippocampus in rodent navigation. Curr
Opin Neurobiol. 22:294–300. 2012.PubMed/NCBI View Article : Google Scholar
|
|
128
|
Moser EI, Kropff E and Moser MB: Place
cells, grid cells, and the brain's spatial representation system.
Annu Rev Neurosci. 31:69–89. 2008.PubMed/NCBI View Article : Google Scholar
|
|
129
|
Moser EI and Moser MB: Hippocampus and
neural representations. In: Encyclopedia of Neuroscience. Squire LR
(ed). Academic Press, Oxford, pp1129-1136, 2009.
|
|
130
|
Høydal ØA, Skytøen ER, Andersson SO, Moser
MB and Moser EI: Object-vector coding in the medial entorhinal
cortex. Nature. 568:400–404. 2019.PubMed/NCBI View Article : Google Scholar
|
|
131
|
Winter SS, Clark BJ and Taube JS: Spatial
navigation. Disruption of the head direction cell network impairs
the parahippocampal grid cell signal. Science. 347:870–874.
2015.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Gil M, Ancau M, Schlesiger MI, Neitz A,
Allen K, De Marco RJ and Monyer H: Impaired path integration in
mice with disrupted grid cell firing. Nat Neurosci. 21:81–91.
2018.PubMed/NCBI View Article : Google Scholar
|
|
133
|
Sargolini F, Fyhn M, Hafting T, McNaughton
BL, Witter MP, Moser MB and Moser EI: Conjunctive representation of
position, direction, and velocity in entorhinal cortex. Science.
312:758–762. 2006.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Fernandez-Leon JA, Uysal AK and Ji D:
Place cells dynamically refine grid cell activities to reduce error
accumulation during path integration in a continuous attractor
model. Sci Rep. 12(21443)2022.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Epstein RA, Patai EZ, Julian JB and Spiers
HJ: The cognitive map in humans: Spatial navigation and beyond. Nat
Neurosci. 20:1504–1513. 2017.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Packard MG and Knowlton BJ: Learning and
memory functions of the basal ganglia. Annu Rev Neurosci.
25:563–593. 2002.PubMed/NCBI View Article : Google Scholar
|
|
137
|
Sukumar D, Rengaswamy M and Chakravarthy
VS: Modeling the contributions of basal ganglia and hippocampus to
spatial navigation using reinforcement learning. PLoS One.
7(e47467)2012.PubMed/NCBI View Article : Google Scholar
|
|
138
|
Dahmani L and Bohbot VD: Habitual use of
GPS negatively impacts spatial memory during self-guided
navigation. Sci Rep. 10(6310)2020.PubMed/NCBI View Article : Google Scholar
|
|
139
|
Gagnepain P, Henson R, Chételat G,
Desgranges B, Lebreton K and Eustache F: Is neocortical-hippocampal
connectivity a better predictor of subsequent recollection than
local increases in hippocampal activity? New insights on the role
of priming. J Cogn Neurosci. 23:391–403. 2011.PubMed/NCBI View Article : Google Scholar
|
|
140
|
Jarius S and Wildemann B: And Pavlov still
rings a bell: Summarising the evidence for the use of a bell in
Pavlov's iconic experiments on classical conditioning. J Neurol.
262:2177–2178. 2015.PubMed/NCBI View Article : Google Scholar
|
|
141
|
Watson JB and Rayner R: Conditioned
emotional reactions. 1920. Am Psychol. 55:313–317. 2000.PubMed/NCBI View Article : Google Scholar
|
|
142
|
Hamann S: Cognitive and neural mechanisms
of emotional memory. Trends Cogn Sci. 5:394–400. 2001.PubMed/NCBI View Article : Google Scholar
|
|
143
|
Canli T, Zhao Z, Brewer J, Gabrieli JD and
Cahill L: Event-related activation in the human amygdala associates
with later memory for individual emotional experience. J Neurosci.
20(RC99)2000.PubMed/NCBI View Article : Google Scholar
|
|
144
|
Dolcos F, LaBar KS and Cabeza R:
Dissociable effects of arousal and valence on prefrontal activity
indexing emotional evaluation and subsequent memory: an
event-related fMRI study. NeuroImage. 23:64–74. 2004.PubMed/NCBI View Article : Google Scholar
|
|
145
|
Hamann SB, Ely TD, Grafton ST and Kilts
CD: Amygdala activity related to enhanced memory for pleasant and
aversive stimuli. Nat Neurosci. 2:289–293. 1999.PubMed/NCBI View Article : Google Scholar
|
|
146
|
Ritchey M, Dolcos F and Cabeza R: Role of
amygdala connectivity in the persistence of emotional memories over
time: An event-related FMRI investigation. Cereb Cortex.
18:2494–2504. 2008.PubMed/NCBI View Article : Google Scholar
|
|
147
|
Hamann SB, Monarch ES and Goldstein FC:
Memory enhancement for emotional stimuli is impaired in early
Alzheimer's disease. Neuropsychology. 14:82–92. 2000.PubMed/NCBI
|
|
148
|
Balzarotti S and Colombo B: Effects of
unilateral transcranial direct current stimulation of left
prefrontal cortex on processing and memory of emotional visual
stimuli. PloS One. 11(e0159555)2016.PubMed/NCBI View Article : Google Scholar
|
|
149
|
Grosso A, Cambiaghi M, Concina G, Sacco T
and Sacchetti B: Auditory cortex involvement in emotional learning
and memory. Neuroscience. 299:45–55. 2015.PubMed/NCBI View Article : Google Scholar
|
|
150
|
Concina G, Renna A, Grosso A and Sacchetti
B: The auditory cortex and the emotional valence of sounds.
Neurosci Biobehav Rev. 98:256–264. 2019.PubMed/NCBI View Article : Google Scholar
|
|
151
|
Douglas G and Saunders S: Ryle's Concept
of Mind. The Philosopher's Magazine. https://archive.philosophersmag.com/ryles-concept-of-mind/.
|
|
152
|
O'sullivan M: An Analysis of Gilbert
Ryle's The Concept of Mind. Macat Library, London, p96, 2017.
|
|
153
|
Fang Z, Chen J, Zheng Y and Chen Z:
Targeting histamine and histamine receptors for memory regulation:
An emotional perspective. Curr Neuropharmacol. 22:1846–1869.
2024.PubMed/NCBI View Article : Google Scholar
|
|
154
|
Eichenbaum H: A cortical-hippocampal
system for declarative memory. Nat Rev Neurosci. 1:41–50.
2000.PubMed/NCBI View Article : Google Scholar
|
|
155
|
Morris RGM: Elements of a neurobiological
theory of hippocampal function: The role of synaptic plasticity,
synaptic tagging and schemas. Eur J Neurosci. 23:2829–2846.
2006.PubMed/NCBI View Article : Google Scholar
|
|
156
|
Abrous DN and Wojtowicz JM: Interaction
between Neurogenesis and hippocampal memory system: New vistas.
Cold Spring Harb Perspect Biol. 7(a018952)2015.PubMed/NCBI View Article : Google Scholar
|
|
157
|
Baptista P and Andrade JP: Adult
Hippocampal neurogenesis: Regulation and possible functional and
clinical correlates. Front Neuroanat. 12(44)2018.PubMed/NCBI View Article : Google Scholar
|
|
158
|
Petrik D and Encinas JM: Perspective: Of
mice and men - how widespread is adult neurogenesis? Front
Neurosci. 13(923)2019.PubMed/NCBI View Article : Google Scholar
|
|
159
|
Sorrells SF, Paredes MF, Cebrian-Silla A,
Sandoval K, Qi D, Kelley KW, James D, Mayer S, Chang J, Auguste KI,
et al: Human hippocampal neurogenesis drops sharply in children to
undetectable levels in adults. Nature. 555:377–381. 2018.PubMed/NCBI View Article : Google Scholar
|
|
160
|
Danzer SC: Adult neurogenesis in the human
brain: Paradise lost? Epilepsy Curr. 18:329–331. 2018.PubMed/NCBI View Article : Google Scholar
|
|
161
|
Boldrini M, Fulmore CA, Tartt AN, Simeon
LR, Pavlova I, Poposka V, Rosoklija GB, Stankov A, Arango V, Dwork
AJ, et al: Human hippocampal neurogenesis persists throughout
aging. Cell Stem Cell. 22:589–599.e5. 2018.PubMed/NCBI View Article : Google Scholar
|
|
162
|
Moreno-Jiménez EP, Terreros-Roncal J,
Flor-García M, Rábano A and Llorens-Martín M: Evidences for adult
hippocampal neurogenesis in humans. J Neurosci. 41:2541–2553.
2021.PubMed/NCBI View Article : Google Scholar
|
|
163
|
Ho NF, Hooker JM, Sahay A, Holt DJ and
Roffman JL: In vivo imaging of adult human hippocampal
neurogenesis: progress, pitfalls and promise. Mol Psychiatry.
18:404–416. 2013.PubMed/NCBI View Article : Google Scholar
|
|
164
|
Fell J, Staresina BP, Do Lam AT, Widman G,
Helmstaedter C, Elger CE and Axmacher N: Memory modulation by weak
synchronous deep brain stimulation: A pilot study. Brain Stimul.
6:270–273. 2013.PubMed/NCBI View Article : Google Scholar
|
|
165
|
Handford CE, Tan S, Lawrence AJ and Kim
JH: The effect of the mGlu5 negative allosteric modulator MTEP and
NMDA receptor partial agonist D-cycloserine on Pavlovian
conditioned fear. Int J Neuropsychopharmacol. 17:1521–1532.
2014.PubMed/NCBI View Article : Google Scholar
|
|
166
|
Norberg MM, Krystal JH and Tolin DF: A
meta-analysis of D-Cycloserine and the facilitation of fear
extinction and exposure therapy. Biol Psychiatry. 63:1118–1126.
2008.PubMed/NCBI View Article : Google Scholar
|
|
167
|
Stone SS, Teixeira CM, DeVito LM,
Zaslavsky K, Josselyn SA, Lozano AM and Frankland PW: Stimulation
of entorhinal cortex promotes adult neurogenesis and facilitates
spatial memory. J Neurosci. 31:13469–13484. 2011.PubMed/NCBI View Article : Google Scholar
|
|
168
|
Liu A, Jain N, Vyas A and Lim LW:
Ventromedial prefrontal cortex stimulation enhances memory and
hippocampal neurogenesis in the middle-aged rats. Elife.
4(e04803)2015.PubMed/NCBI View Article : Google Scholar
|
|
169
|
Titiz AS, Hill MRH, Mankin EA, M Aghajan
Z, Eliashiv D, Tchemodanov N, Maoz U, Stern J, Tran ME, Schuette P,
et al: Theta-burst microstimulation in the human entorhinal area
improves memory specificity. Elife. 6(e29515)2017.PubMed/NCBI View Article : Google Scholar
|
|
170
|
Suthana N, Haneef Z, Stern J, Mukamel R,
Behnke E, Knowlton B and Fried I: Memory enhancement and deep-brain
stimulation of the entorhinal area. N Engl J Med. 366:502–510.
2012.PubMed/NCBI View Article : Google Scholar
|
|
171
|
Murphy MP and LeVine H III: Alzheimer's
disease and the amyloid-beta peptide. J Alzheimers Dis. 19:311–323.
2010.PubMed/NCBI View Article : Google Scholar
|
|
172
|
Belder CRS, Boche D, Nicoll JAR,
Jaunmuktane Z, Zetterberg H, Schott JM, Barkhof F and Fox NC: Brain
volume change following anti-amyloid β immunotherapy for
Alzheimer's disease: amyloid-removal-related pseudo-atrophy. Lancet
Neurol. 23:1025–1034. 2024.PubMed/NCBI View Article : Google Scholar
|
|
173
|
Lozano AM, Fosdick L, Chakravarty MM,
Leoutsakos JM, Munro C, Oh E, Drake KE, Lyman CH, Rosenberg PB,
Anderson WS, et al: A phase II study of fornix deep brain
stimulation in mild Alzheimer's disease. J Alzheimers Dis.
54:777–787. 2016.PubMed/NCBI View Article : Google Scholar
|
|
174
|
Jacobs J, Miller J, Lee SA, Coffey T,
Watrous AJ, Sperling MR, Sharan A, Worrell G, Berry B, Lega B, et
al: Direct electrical stimulation of the human entorhinal region
and hippocampus impairs memory. Neuron. 92:983–990. 2016.PubMed/NCBI View Article : Google Scholar
|
|
175
|
Merkow MB, Burke JF, Ramayya AG, Sharan
AD, Sperling MR and Kahana MJ: Stimulation of the human medial
temporal lobe between learning and recall selectively enhances
forgetting. Brain Stimul. 10:645–650. 2017.PubMed/NCBI View Article : Google Scholar
|
|
176
|
Krauss JK, Lipsman N, Aziz T, Boutet A,
Brown P, Chang JW, Davidson B, Grill WM, Hariz MI, Horn A, et al:
Technology of deep brain stimulation: Current status and future
directions. Nat Rev Neurol. 17:75–87. 2021.PubMed/NCBI View Article : Google Scholar
|
|
177
|
Lacruz ME, Valentín A, Seoane JJ, Morris
RG, Selway RP and Alarcón G: Single pulse electrical stimulation of
the hippocampus is sufficient to impair human episodic memory.
Neuroscience. 170:623–632. 2010.PubMed/NCBI View Article : Google Scholar
|
|
178
|
Ramirez S, Liu X, Lin PA, Suh J,
Pignatelli M, Redondo RL, Ryan TJ and Tonegawa S: Creating a false
memory in the hippocampus. Science. 341:387–391. 2013.PubMed/NCBI View Article : Google Scholar
|
|
179
|
Vetere G, Tran LM, Moberg S, Steadman PE,
Restivo L, Morrison FG, Ressler KJ, Josselyn SA and Frankland PW:
Memory formation in the absence of experience. Nat Neurosci.
22:933–940. 2019.PubMed/NCBI View Article : Google Scholar
|
|
180
|
Nader K, Schafe GE and Le Doux JE: Fear
memories require protein synthesis in the amygdala for
reconsolidation after retrieval. Nature. 406:722–726.
2000.PubMed/NCBI View Article : Google Scholar
|
|
181
|
Nabavi S, Fox R, Proulx CD, Lin JY, Tsien
RY and Malinow R: Engineering a memory with LTD and LTP. Nature.
511:348–352. 2014.PubMed/NCBI View Article : Google Scholar
|
|
182
|
Redondo RL, Kim J, Arons AL, Ramirez S,
Liu X and Tonegawa S: Bidirectional switch of the valence
associated with a hippocampal contextual memory engram. Nature.
513:426–430. 2014.PubMed/NCBI View Article : Google Scholar
|
|
183
|
Tan SZK, Sheng V, Chan YS and Lim LW:
Eternal sunshine of the neuromodulated mind: Altering fear memories
through neuromodulation. Exp Neurol. 314:9–19. 2019.PubMed/NCBI View Article : Google Scholar
|
|
184
|
Sharma P and Upadhyay TN: Transhumanism,
human enhancement and ethical debate. Am J Arts Hum Sci. 4:1–6.
2025.
|
|
185
|
Lautenschlager NT, Cox KL, Flicker L,
Foster JK, van Bockxmeer FM, Xiao J, Greenop KR and Almeida OP:
Effect of physical activity on cognitive function in older adults
at risk for Alzheimer disease: A randomized trial. JAMA.
300:1027–1037. 2008.PubMed/NCBI View Article : Google Scholar
|
|
186
|
Wu CW, Chen YC, Yu L, Chen HI, Jen CJ,
Huang AM, Tsai HJ, Chang YT and Kuo YM: Treadmill exercise
counteracts the suppressive effects of peripheral
lipopolysaccharide on hippocampal neurogenesis and learning and
memory. J Neurochem. 103:2471–2481. 2007.PubMed/NCBI View Article : Google Scholar
|
|
187
|
Erickson KI, Voss MW, Prakash RS, Basak C,
Szabo A, Chaddock L, Kim JS, Heo S, Alves H, White SM, et al:
Exercise training increases size of hippocampus and improves
memory. Proc Natl Acad Sci USA. 108:3017–3022. 2011.PubMed/NCBI View Article : Google Scholar
|
|
188
|
Bekinschtein P, Oomen CA, Saksida LM and
Bussey TJ: Effects of environmental enrichment and voluntary
exercise on neurogenesis, learning and memory, and pattern
separation: BDNF as a critical variable? Semin Cell Dev Biol.
22:536–542. 2011.PubMed/NCBI View Article : Google Scholar
|
|
189
|
Vaynman S, Ying Z and Gomez-Pinilla F:
Hippocampal BDNF mediates the efficacy of exercise on synaptic
plasticity and cognition. Eur J Neurosci. 20:2580–2590.
2004.PubMed/NCBI View Article : Google Scholar
|
|
190
|
Rojas Vega S, Strüder HK, Vera Wahrmann B,
Schmidt A, Bloch W and Hollmann W: Acute BDNF and cortisol response
to low intensity exercise and following ramp incremental exercise
to exhaustion in humans. Brain Res. 1121:59–65. 2006.PubMed/NCBI View Article : Google Scholar
|
|
191
|
Neeper SA, Gómez-Pinilla F, Choi J and
Cotman C: Exercise and brain neurotrophins. Nature.
373(109)1995.PubMed/NCBI View Article : Google Scholar
|
|
192
|
Farmer J, Zhao X, van Praag H, Wodtke K,
Gage FH and Christie BR: Effects of voluntary exercise on synaptic
plasticity and gene expression in the dentate gyrus of adult male
Sprague-Dawley rats in vivo. Neuroscience. 124:71–79.
2004.PubMed/NCBI View Article : Google Scholar
|
|
193
|
Marin Bosch B, Bringard A, Logrieco MG,
Lauer E, Imobersteg N, Thomas A, Ferretti G, Schwartz S and Igloi
K: A single session of moderate intensity exercise influences
memory, endocannabinoids and brain derived neurotrophic factor
levels in men. Sci Rep. 11(14371)2021.PubMed/NCBI View Article : Google Scholar
|
|
194
|
Parent MB, Higgs S, Cheke LG and Kanoski
SE: Memory and eating: A bidirectional relationship implicated in
obesity. Neurosci Biobehav Rev. 132:110–129. 2022.PubMed/NCBI View Article : Google Scholar
|
|
195
|
Farruggia MC and Small DM: Effects of
adiposity and metabolic dysfunction on cognition: A review. Physiol
Behav. 208(112578)2019.PubMed/NCBI View Article : Google Scholar
|
|
196
|
Sweeney P and Yang Y: An excitatory
ventral hippocampus to lateral septum circuit that suppresses
feeding. Nat Commun. 6(10188)2015.PubMed/NCBI View Article : Google Scholar
|
|
197
|
Azevedo EP, Pomeranz L, Cheng J,
Schneeberger M, Vaughan R, Stern SA, Tan B, Doerig K, Greengard P
and Friedman JM: A role of Drd2 hippocampal neurons in
context-dependent food intake. Neuron. 102:873–886.e5.
2019.PubMed/NCBI View Article : Google Scholar
|
|
198
|
Gasbarri A, Pompili A, Packard MG and
Tomaz C: Habit learning and memory in mammals: Behavioral and
neural characteristics. Neurobiol Learn Mem. 114:198–208.
2014.PubMed/NCBI View Article : Google Scholar
|
|
199
|
White NM, Packard MG and McDonald RJ:
Dissociation of memory systems: The story unfolds. Behav Neurosci.
127:813–834. 2013.PubMed/NCBI View Article : Google Scholar
|
|
200
|
Cheke LG, Bonnici HM, Clayton NS and
Simons JS: Obesity and insulin resistance are associated with
reduced activity in core memory regions of the brain.
Neuropsychologia. 96:137–149. 2017.PubMed/NCBI View Article : Google Scholar
|
|
201
|
Michels B, Zwaka H, Bartels R, Lushchak O,
Franke K, Endres T, Fendt M, Song I, Bakr M, Budragchaa T, et al:
Memory enhancement by ferulic acid ester across species. Sci Adv.
4(eaat6994)2018.PubMed/NCBI View Article : Google Scholar
|
|
202
|
de Vries R, Boesveldt S and de Vet E:
Human spatial memory is biased towards high-calorie foods: A
cross-cultural online experiment. Int J Behav Nutr Phys Act.
19(14)2022.PubMed/NCBI View Article : Google Scholar
|
|
203
|
Slutsky I, Abumaria N, Wu LJ, Huang C,
Zhang L, Li B, Zhao X, Govindarajan A, Zhao MG, Zhuo M, et al:
Enhancement of learning and memory by elevating brain magnesium.
Neuron. 65:165–177. 2010.PubMed/NCBI View Article : Google Scholar
|
|
204
|
Wu Y, Funato Y, Meschi E, Jovanoski KD,
Miki H and Waddell S: Magnesium efflux from Drosophila Kenyon cells
is critical for normal and diet-enhanced long-term memory. Elife.
9(e61339)2020.PubMed/NCBI View Article : Google Scholar
|
|
205
|
Roig M, Nordbrandt S, Geertsen SS and
Nielsen JB: The effects of cardiovascular exercise on human memory:
A review with meta-analysis. Neurosci Biobehav Rev. 37:1645–1666.
2013.PubMed/NCBI View Article : Google Scholar
|
|
206
|
Kuhne LA, Ksiezarczyk AM, Braumann KM,
Reer R, Jacobs T, Röder B and Hötting K: The effects of acute
cardiovascular exercise on memory and its associations with
exercise-induced increases in neurotrophic factors. Front Aging
Neurosci. 13(750401)2021.PubMed/NCBI View Article : Google Scholar
|
|
207
|
Loprinzi P, Olafson D, Scavuzzo C, Lovorn
A, Mather M, Frith E and Fujiwara E: Effects of acute exercise on
emotional memory. Cogn Emot. 36:660–689. 2022.PubMed/NCBI View Article : Google Scholar
|
|
208
|
Jack CR Jr, Wiste HJ, Vemuri P, Weigand
SD, Senjem ML, Zeng G, Bernstein MA, Gunter JL, Pankratz VS, Aisen
PS, et al: Brain beta-amyloid measures and magnetic resonance
imaging atrophy both predict time-to-progression from mild
cognitive impairment to Alzheimer's disease. Brain. 133:3336–3348.
2010.PubMed/NCBI View Article : Google Scholar
|
|
209
|
Chen X, Shi X, Wu Y, Zhou Z, Chen S, Han Y
and Shan C: Gamma oscillations and application of 40-Hz audiovisual
stimulation to improve brain function. Brain Behav.
12(e2811)2022.PubMed/NCBI View Article : Google Scholar
|
|
210
|
Etter G, van der Veldt S, Manseau F,
Zarrinkoub I, Trillaud-Doppia E and Williams S: Optogenetic gamma
stimulation rescues memory impairments in an Alzheimer's disease
mouse model. Nat Commun. 10(5322)2019.PubMed/NCBI View Article : Google Scholar
|
|
211
|
Sri Takshara K and Bhuvaneswari G: The
role of death technologies in grief: An interdisciplinary
examination of AI, cognition, and human expression. Front Hum Dyn.
7(1582914)2025.
|
|
212
|
Kang S, Lee Y, Kim N, Ok SH, Kim P, Park
OH, Kim DM, Jang Y and Kim HJ: Beneficial effect of artificial
intelligence care call on memory and depression in community
dwelling individuals with dementia. Sci Rep.
15(27116)2025.PubMed/NCBI View Article : Google Scholar
|