Introduction
Liver cancer is the sixth most prevalent form of
cancer worldwide and the fourth most common cause of cancer-related
mortalities (1). Portal vein
thrombosis (PVT) is a prevalent complication in patients with
hepatocellular carcinoma (HCC), affecting ~40% of cases (2). PVT is a poor prognostic factor that
increases mortality rates by reducing blood flow in the liver,
resulting in portal hypertension, ascites, hepatic encephalopathy
and liver failure (3). PVT also
facilitates the dissemination of tumors throughout the liver
parenchyma (1).
The pathogenesis of PVT in HCC is highly intricate
and involves a combination of multiple factors. Venous stasis,
endothelial injury and hypercoagulability comprise Virchow's triad.
The velocity of portal blood flow is diminished in HCC as a result
of portal hypertension, tumor compression or the loss of normal
liver tissue structure. This stasis facilitates the formation of
thrombi. Furthermore, the tumor directly damages the endothelial
tissue and initiates a coagulation response by invading the portal
vein wall. The synthesis of natural anticoagulants (protein S,
protein C and antithrombin) is reduced in patients with HCC due to
liver injury, while cancer cells overproduce procoagulant factors,
such as tissue factors, resulting in increased activation of the
coagulation system (4). Currently,
chronic inflammation frequently plays a role in the pathogenesis of
thrombus formation. Numerous inflammatory mediators, including
TNF-α, interleukin (IL)-6, interferon-γ, transforming growth factor
β, IL-17, IL-9, IL-1β and chemokines, such as CXC motif chemokine
ligand (CXCL) 8/CXCL1 and C-C motif chemokine ligand 2, are
released by cancer cells, all of which contribute to endothelial
injury (5). Neutrophils are
activated by inflammatory cytokines, which facilitate their
adhesion to endothelial cells, promote inflammation and platelet
aggregation and result in the formation of thrombus (6-8).
Neutrophils are crucial in the inflammatory response and, as a
result, serve a substantial role in the formation of thrombus.
Obtained from a peripheral blood cell count, the
neutrophil-to-lymphocyte ratio (NLR) is a simple index that
indicates the immune and inflammatory functions of the body.
Researchers have investigated it as a possible predictive biomarker
for different types of cancer, such as HCC (1,9).
Nevertheless, there is a lack of studies examining the predictive
relevance of NLR in predicting thrombosis in patients with HCC,
although some researchers have mentioned the role of NLR in
predicting the prognosis of HCC (10). The objective of the present study
was to assess the predictive value of NLR in the recognition of PVT
in patients with HCC.
Patients and methods
Subjects and research design
The present cross-sectional investigation was
performed at Bach Mai Hospital (Hanoi, Vietnam) from February, 2024
to May, 2025. Patients who were ≥18 years of age, had a first-time
confirmed diagnosis of HCC according to the American Association
for the Study of Liver Diseases (AASLD) guidelines (11) and had not received treatment were
included in the study. The study excluded patients who were on
medication that affected their white blood cell count, had a
history of hematological disease or had an infection. The patients
were divided into two different groups as follows: Patients
diagnosed with PVT through computed tomography or magnetic
resonance imaging scans comprised group 1. The second group
consisted of patients who did not have PVT. The study protocol was
approved by the Institutional Review Board of Hanoi Medical
University (no. 1063/GCN-HMUIRB) in January, 2024.
Data collection
Prior to commencing treatment, peripheral blood cell
indices were obtained, which included the number of white blood
cells, neutrophils, lymphocytes, monocytes and platelets, as well
as the concentration of hemoglobin. Coagulation parameters,
including fibrinogen, D-dimer, prothrombin time (PT) and activated
partial thromboplastin time (APTT) were also collected. The study
also collected data on the following: Disease [age, sex, etiology
of HCC, body mass index (BMI)], liver function [Child-Pugh,
Barcelona Clinic Liver Cancer classification (BCLC)] (12,13),
α-fetoprotein (AFP) and tumor characteristics [size,
Tumor-Node-Metastasis staging system (TNM)] (14).
Statistical analysis
The following two categories were compared:
Non-thrombotic and PVT. The qualitative variables (sex, etiology of
HCC, Child-Pugh, BCLC and TNM) were analyzed using χ2 or
Fisher's exact test. The quantitative variables (hemoglobin
concentration; white blood cell, neutrophil, lymphocyte, monocyte
and platelet counts; NLR, fibrinogen and D-dimer concentrations;
AFP, tumor size and BMI) were analyzed using independent samples
t-tests or Mann-Whitney U tests, depending on their normal or
non-normal distribution. To determine whether the data follow a
normal distribution or not, the Kolmogorov-Smirnov test was used;
in the event that the P-value of the test was >0.05, the
assumption of normal distribution data was accepted. The Youden
index and receiver operating characteristic (ROC) analysis was used
to determine the most appropriate NLR threshold for distinguishing
between groups with and without PVT. Logistic regression analysis
was implemented to assess how well NLR predicted PVT in patients
with HCC. Multivariate regression analysis of NLR was performed
with adjustment for AFP and tumor size to determine the independent
significance of NLR in predicting the risk of PVT in patients with
HCC. The AFP threshold of ≥400 ng/ml was selected based on clinical
evidence of poor predictive relevance with increased risk of
recurrence or progression (15,16).
The tumor size threshold of ≥3 cm was used to separate risk groups,
based on the standard for small and large HCC, to determine the
effect of tumor size on thrombotic risk and survival (17). SPSS 22.0 software (IBM Corp.) was
employed to performed all analyses, with a value of P<0.05
considered to indicate a statistically significant difference.
Results
Patient characteristics
The present study included 197 patients diagnosed
with HCC. The prevalence of PVT was 30%. The patients were
predominantly male, the mean age of the non-thrombotic group was
higher compared with that of the PVT group (P=0.011), and the mean
total tumor volume (TTV) of the PVT group was higher than that of
the non-thrombotic group (P<0.001) (Table I). The causes of HCC, which include
alcohol consumption, hepatitis B and C virus infection, cirrhosis
and BMI, did not exhibit any significant differences between the
groups (all P>0.05). Significant differences were observed
between the group with PVT and the group without PVT in terms of
the HCC stage, according to Child-Pugh, TNM and BCLC (Table I).
 | Table IBaseline characteristics of the
patients with HCC. |
Table I
Baseline characteristics of the
patients with HCC.
| Characteristic | Thrombosis (PVT),
n=60 | No thrombosis,
n=137 | P-value |
|---|
| Age in years, mean ±
SD | 56.72±9.18 | 61.16±10.21 | 0.011 |
| Sex, n (%) | | | |
|
Male | 53 (88.3) | 119 (86.9) | 0.775 |
|
Female | 7 (11.7) | 18 (13.1) | |
| Alcohol abuse, n
(%) | | | |
|
Yes | 14 (23.3) | 19(14) | 0.106 |
|
No | 46 (76.7) | 117(86) | |
| HBV, n (%) | | | |
|
Yes | 47 (78.3) | 91 (66.4) | 0.093 |
|
No | 13 (21.7) | 46 (33.6) | |
| HCV, n (%) | | | |
|
Yes | 2 (3.3) | 8 (5.9) | 0.726 |
|
No | 58 (96.7) | 128 (94.1) | |
| Cirrhosis, n (%) | | | |
|
Yes | 27(45) | 53 (38.7) | 0.406 |
|
No | 33(55) | 84 (61.3) | |
| Child Pugh, n
(%) | | | |
|
A | 35 (59.3) | 112 (82.4) | 0.002 |
|
B | 19 (32.2) | 21 (15.4) | |
|
C | 5 (8.5) | 3 (2.2) | |
| TNM, n (%) | | | |
|
IA | 0 (0) | 11(8) |
<0.001 |
|
IB | 2 (3.3) | 41 (29.9) | |
|
II | 0 (0) | 22 (16.1) | |
|
IIIA | 0 (0) | 21 (15.3) | |
|
IIIB | 26 (43.3) | 8 (5.8) | |
|
IVA | 25 (41.7) | 26(19) | |
|
IVB | 7 (11.7) | 8 (5.8) | |
| BCLC, n (%) | | | |
|
0 | 0 (0) | 8 (5.8) |
<0.001 |
|
A | 0 (0) | 35 (25.5) | |
|
B | 0 (0) | 58 (42.3) | |
|
C | 57(95) | 33 (24.1) | |
|
D | 3(5) | 3 (2.2) | |
| TTV in
cm3, median (range) | 211.3 (5-1863) | 41.35
(0.5-1436.7) |
<0.001 |
| BMI, mean ± SD | 21.2±2.37 | 22.17±2.71 | 0.061 |
Peripheral blood cells and
biomarkers
As demonstrated in Table II, the PVT group had elevated
neutrophil counts, NLR, total bilirubin, AFP, aspartate
aminotransferase, alanine aminotransferase, PT-international
normalized ratio (INR) and D-dimer levels; however, lymphocyte
counts and albumin levels were diminished in comparison with the
non-thrombotic group (all P<0.05). No significant differences
were observed between the two groups as regards platelet count,
white blood cell count, fibrinogen and APTT (all P>0.05)
(Table II).
 | Table IILevels of peripheral blood cell and
biomarker in the patients with HCC. |
Table II
Levels of peripheral blood cell and
biomarker in the patients with HCC.
| Factor | Thrombosis
(PVT) | No thrombosis | P-value |
|---|
| Hemoglobin, g/l,
mean ± SD | 131.8±24.42 | 139 (53-165) | 0.071 |
| Platelet count,
109/l, median (range) | 188 (54-582) | 193.37±94.57 | 0.183 |
| WBC,
109/l, median (range) | 7.01
(3.13-15.99) | 6.4
(2.02-15.3) | 0.101 |
| Neu,
109/l, median (range) | 4.5
(1.38-13.6) | 3.54
(0.98-12.8) | 0.01 |
| Lymphocyte,
109/l, mean ± SD | 1.37±0.54 | 1.8(0.51-4.07) | 0.004 |
| NLR, median
(range) | 3.81
(0.78-15.6) | 2.34
(0.83-16.8) |
<0.001 |
| Total bilirubin,
µmol/l, median (range) | 18.2
(10.5-191.7) | 10.85
(3.5-278) |
<0.001 |
| Creatinine, µmol/l,
median (range) | 75 (42-171) | 77 (36-262) | 0.342 |
| Albumin, g/l,
median (range) | 36.8 (23-47) | 37.46±5.5 | 0.04 |
| AST, U/l, median
(range) | 106 (26-523) | 48 (14-834) |
<0.001 |
| ALT, U/l, median
(range) | 54 (9-439) | 41 (9-562) | 0.014 |
| AFP, ng/ml, median
(range) | 2135
(2.2-121000) | 72.3
(1.3-121000) |
<0.001 |
| Fibrinogen, g/l,
mean ± SD | 3.23±1 | 3.17±0.99 | 0.492 |
| PT-INR, median
(range) | 1.09
(0.92-2.37) | 1.03
(0.86-1.91) | 0.012 |
| APTT sec, mean ±
SD | 34.28±5.22 |
33.2(26.8-51.1) | 0.436 |
| D-Dimer, ng/ml FEU,
median (range) | 2123
(179-9552) | 854
(128-11015) |
<0.001 |
ROC curve analysis for PVT predictors
based on NLR
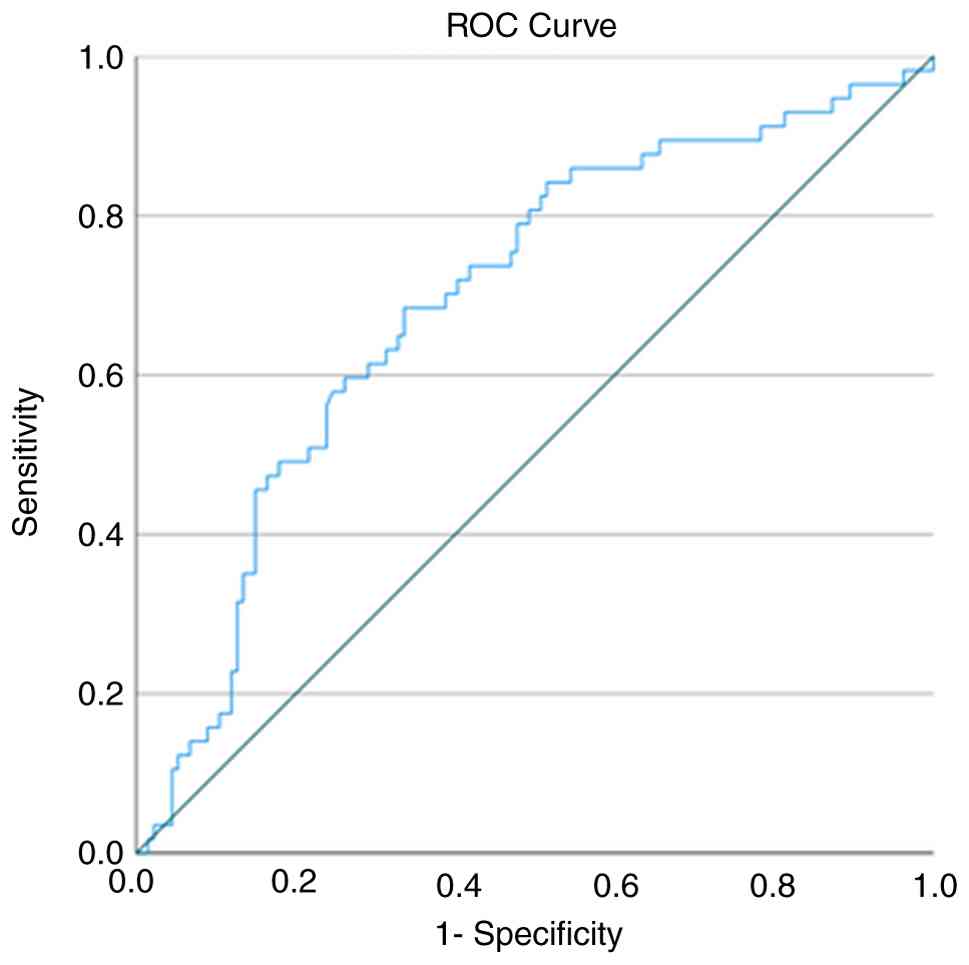
The ROC curve analysis of NLR for predicting the
occurrence of PVT in patients with HCC revealed an area under the
curve of 69.5% with a P-value of <0.001 (Fig. 1 and Table III). The ideal threshold value of
NLR, determined by the Youden index, was 2.817, exhibiting a
sensitivity of 68.4% and a specificity of 66.4%.
 | Table IIIROC parameters and optimal cut-off of
NLR for distinguishing the risk of PVT. |
Table III
ROC parameters and optimal cut-off of
NLR for distinguishing the risk of PVT.
| AUC | P-value | Cut-off | Sensitivity | Specificity |
|---|
| 69.5 | <0.001 | 2.817 | 68.4 | 66.4 |
As demonstrated in Table IV, in the univariate analysis,
characteristics associated with thrombosis occurrence included NLR
ratio >2.817, AFP concentration ≥400 ng/ml and tumor size ≥3 cm
(all P<0.05). The multivariate logistic regression results
indicated that the model was statistically significant
(χ2=46.066; P<0.001). The model included three
statistically significant independent variables: NLR ratio ≥2.817
[odds ratio (OR), 4.31; 95% confidence interval (CI), 1.98-9.42;
P<0.001], AFP levels ≥400 ng/ml (OR, 3.78; 95% CI, 1.74-8.23;
P<0.001) and tumor size ≥3 cm (OR, 11.19; 95% CI, 1.41-88.73;
P=0.022). A tumor size ≥3 cm was the most significant predictor,
elevating the risk of thrombosis by ~11-fold relative to the cohort
with smaller tumors. This suggests that indicators of inflammation
(NLR) and progression of tumors (AFP and size) significantly
contribute to the mechanism of thrombus formation in patients.
 | Table IVLogistic regression analysis for
predicting PVT. |
Table IV
Logistic regression analysis for
predicting PVT.
| | Univariate
analysis | Multivariate
analysis |
|---|
| Factor | P-value | OR | 95% CI | P-value | OR | 95% CI |
|---|
| NLR ≥2.817 | <0.001 | 4.285 | 2.207-8.321 | <0.001 | 4.31 | 1.98-9.42 |
| AFP ≥400 | <0.001 | 5.375 | 2.715-10.643 | <0.001 | 3.78 | 1.74-8.23 |
| Tumor size ≥3
cm | 0.003 | 20.427 | 2.725-153.125 | 0.022 | 11.19 | 1.41-88.73 |
Discussion
Numerous studies have illustrated the notable role
of the systemic inflammatory process in the pathogenesis of
thrombosis. According to mouse model experiments, neutrophils were
observed to modulate angiogenesis and tumor growth (18). The formation of a thrombus is
facilitated by the activation of endothelial cells, platelets and
leukocytes during the inflammatory process (19). In addition to secreting cytokines
that attract immune cells such as neutrophils and monocytes,
activated endothelial cells express P-selectin, which stimulates
the adhesion of leukocytes and platelets. Clusters of these cells
are formed and adhere to endothelial cells, which are essential
components of blood clots (20).
Through neutrophil extracellular traps (NETs), neutrophils
facilitate the formation of thrombus (20). Furthermore, NETs stimulate the
coagulation process by attracting coagulation factors, including
factor XIIa, which adhere to fibrin and von Willebrand factor. This
results in the recruitment of red blood cells and platelets to the
site of thrombus formation (21).
Numerous studies have also established an association between
systemic inflammation and cancer. The tumor induces the formation
of new blood vessels, DNA damage and the inhibitory effect of
apoptosis by increasing the production of cytokines and
inflammatory mediators (22-24).
Consequently, the process of systemic inflammation inextricably
links the mechanisms of thrombus formation and cancer.
Consequently, inflammatory markers can predict the risk of
thrombosis in patients with cancer. NLR is a non-specific
inflammatory marker that is indicative of the interaction between
neutrophils and lymphocytes during inflammation. A high neutrophil
count and a lower lymphocyte count are indicative of a higher
neutrophil count. A systemic inflammatory process is indicated by
an increase in the number of neutrophils, whereas a reduction in
the number of lymphocytes suggests a state of reduced immune
response (25). Therefore, an
elevated NLR may suggest a higher risk of thrombosis in patients
with cancer.
Currently, systemic treatment methods for patients
with HCC have undergone notable changes with the emergence of
immunotherapy regimens combining anti-angiogenic drugs. Regimens
such as atezolizumab combined with bevacizumab have become
first-line options for patients with advanced HCC, gradually
replacing sorafenib monotherapy, with marked improvements in
response rates and survival times (26). In addition, recent guidelines on
HCC treatment emphasize combined local-regional and systemic
therapy, individualized based on tumor burden, liver function and
vascular invasion (27). In this
context, PVT has become an increasingly crucial factor, not only
reflecting the advanced stage of the disease, but also directly
affecting the efficacy and safety of both local interventions and
systemic regimens containing anti-VEGF components. An elevated NLR
in patients with PVT (28) may be
indicative of a tumor microenvironment that promotes vascular
invasion and thrombus formation by stimulating thrombogenesis and
inflammation (29). The present
study demonstrated that patients with HCC and PVT had a much higher
NLR compared with those without thrombosis. Multivariate analysis
confirmed that NLR ≥2.817 increased the risk of PVT after adjusting
for other risk factors, such as AFP and tumor size.
Numerous studies have demonstrated that NLR is a
straightforward, cost-effective and user-friendly inflammatory
marker that has been assessed as a predictive instrument for the
risk of venous thromboembolism in patients with cancer (30,31,32).
The NLR has been shown to be higher in a venous thromboembolism
group with cancer compared with in a group without cancer (33). In a study involving 271
hospitalized patients with venous thromboembolism, Qian et
al (34) demonstrated that the
presence of an elevated NLR is a predictor of venous
thromboembolism in patients with breast cancer following treatment.
NLR is a predictor of response to anticoagulant treatment in
patients with lung cancer with thrombosis, as demonstrated in the
study conducted by Go et al (35). The predictive value of NLR in
patients with HCC has been demonstrated in a number of studies,
which have demonstrated its ability to predict treatment response,
disease-free survival and overall survival (36-39).
Several predictive ratings for survival time and treatment response
in patients with HCC also include NLR (40,41).
The predictive value of NLR in patients with HCC is the primary
focus of these studies, with only a small number of studies
examining the predictive ability of PVT.
AFP is a traditional tumor marker in HCC, which not
only reflects tumor burden but is also related to the degree of
invasion, growth and thrombosis risk (39). A high AFP level is an independent
predictive factor the risk of metastasis and recurrence and is also
associated with thrombosis, especially when HCC is accompanied by
increased vascular invasion (42,43).
A large tumor size is an indicator of a high tumor burden, often
accompanied by an increased risk of vascular invasion, particularly
of the portal vein, and is therefore directly related to the
incidence of thrombosis in HCC (44). Although PVT is a prognostic factor
in patients with HCC, the ability to predict the likelihood of PVT
can assist physicians in the implementation of early
thromboprophylaxis treatment strategies for patients, particularly
those with HCC who have an evident tendency towards
hypercoagulability (45). Applying
the NLR to predict the risk PVT in patients with HCC will help
physicians plan for closer imaging surveillance in high-risk
groups. This approach will also assist in selecting appropriate
systemic treatment regimens, considering the risk of thrombosis
associated with anti-VEGF drug therapies. Furthermore, combining
NLR, AFP and tumor size could potentially serve as a predictive
model for PVT in patients with HCC; however, more large-scale
prospective studies are required to validate this model. In
addition, impaired liver function (increased bilirubin, decreased
albumin) is a factor associated with a higher risk of thrombosis
and a worse prognosis (46).
Prolonged PT (INR) and elevated D-dimer levels in patients with PVT
are also indicative of the activation of the coagulation system,
which suggests that fibrin formation and disintegration are
ongoing. This hypercoagulable state is consistent with reports of
hypercoagulable syndrome in HCC with vascular invasion.
The present study had certain limitations, which
should be mentioned. The sample size of the PVT group was small,
less than half that of the non-thrombotic group. The present study
was a cross-sectional study; therefore, the non-thrombotic group
was not followed-up. The authors could not assess whether
thrombotic progression occurred in this group.
In conclusion, the present study demonstrates an
association between the incidence of PVT in patients with HCC and
NLR. The occurrence of PVT is independently associated with an
elevated NLR. The value of NLR in preventing thrombosis in patients
with HCC remains an unresolved matter. Further research is required
in the future to ascertain the optimal NLR threshold for the
prevention, diagnosis and treatment of thrombosis, as well as the
effective use of this index.
Acknowledgements
No applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
All authors (TTMN, CPP and MPV) conceived and
designed the study. TTMN participated in data collection and
processing. TTMN, MPV and CPP participated in data analysis and
interpretation. All authors participated in the literature search
and wrote the manuscript. All authors have read and approved the
final manuscript. TTMN and MPV confirm the authenticity of all the
raw data.
Ethics approval and consent to
participate
The study protocol was approved by The Institutional
Review Board of Hanoi Medical University approved study number
1063/GCN-HMUIRB. Written informed consent was obtained from all
participants prior to enrollment in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that have no competing
interests.
References
|
1
|
Zhang M, Ding Q, Bian C, Su J, Xin Y and
Jiang X: Progress on the molecular mechanism of portal vein tumor
thrombosis formation in hepatocellular carcinoma. Exp Cell Res.
426(113563)2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Pan J, Wang L, Gao F, An Y, Yin Y, Guo X,
Nery FG, Yoshida EM and Qi X: Epidemiology of portal vein
thrombosis in liver cirrhosis: A systematic review and
meta-analysis. Eur J Intern Med. 104:21–32. 2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Quirk M, Kim YH, Saab S and Lee EW:
Management of hepatocellular carcinoma with portal vein thrombosis.
World J Gastroenterol. 21:3462–3471. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Galasso L, Cerrito L, Termite F, Mignini
I, Esposto G, Borriello R, Ainora ME, Gasbarrini A and Zocco MA:
The molecular mechanisms of portal vein thrombosis in
hepatocellular carcinoma. Cancers (Basel). 16(3247)2024.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Cavus UY, Yildirim S, Sönmez E, Ertan C
and Ozeke O: Prognostic value of neutrophil/lymphocyte ratio in
patients with pulmonary embolism. Turk J Med Sci. 44:50–55.
2014.PubMed/NCBI
|
|
6
|
Köse N, Yıldırım T, Akın F, Yıldırım SE
and Altun İ: Prognostic role of NLR, PLR, and LMR in patients with
pulmonary embolism. Bosn J Basic Med Sci. 20:248–253.
2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zhao J, Liu K, Li S, Gao Y, Zhao L, Liu H,
Fang H, Song B and Xu Y: Neutrophil-to-lymphocyte ratio predicts
the outcome of cerebral venous thrombosis. Curr Neurovasc Res.
18:204–210. 2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hu J, Cai Z and Zhou Y: The association of
neutrophil-lymphocyte ratio with venous thromboembolism: A
systematic review and meta-analysis. Clin Appl Thromb Hemost.
28(10760296221130061)2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Cupp MA, Cariolou M, Tzoulaki I, Aune D,
Evangelou E and Berlanga-Taylor AJ: Neutrophil-to-lymphocyte ratio
and cancer prognosis. BMC Med. 18(360)2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lin S, Hu S, Ran Y and Wu F:
Neutrophil-to-lymphocyte ratio predicts prognosis of patients with
hepatocellular carcinoma: A systematic review and meta-analysis.
Transl Cancer Res. 10:1667–1678. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Singal AG, Llovet JM, Yarchoan M, Mehta N,
Heimbach JK, Dawson LA, Jou JH, Kulik LM, Agopian VG, Marrero JA,
et al: AASLD practice guidance on prevention, diagnosis, and
treatment of hepatocellular carcinoma. Hepatology. 78:1922–1965.
2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Tsoris A and Marlar CA: Use of the child
pugh score in liver disease. In: StatPearls. StatPearls Publishing,
Treasure Island, FL, 2025.
|
|
13
|
Han K and Kim JH: Transarterial
chemoembolization in hepatocellular carcinoma treatment: Barcelona
clinic liver cancer staging system. World J Gastroenterol.
21:10327–10335. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Rosen RD and Sapra A: TNM Classification.
In: StatPearls. StatPearls Publishing, Treasure Island, FL,
2026.
|
|
15
|
Hsu CY, Liu PH, Lee YH, Hsia CY, Huang YH,
Lin HC, Chiou YY, Lee FY and Huo TI: Using serum AFP for prognostic
prediction in HCC. PLoS One. 10(e0118825)2015.
|
|
16
|
Chan MY, She WH, Dai WC, Tsang SHY, Chok
KSH, Chan ACY, Fung J, Lo CM and Cheung TT: Prognostic value of
preoperative AFP. Transl Gastroenterol Hepatol. 4(52)2019.
|
|
17
|
Liu H, Yang Y, Chen C, Wang L, Huang Q,
Zeng J, Lin K, Zeng Y, Guo P, Zhou W and Liu J: Reclassification of
tumor size for HBV-related HCC. World J Surg Oncol.
18(185)2020.
|
|
18
|
Jablonska J, Leschner S, Westphal K,
Lienenklaus S and Weiss S: Neutrophils responsive to IFN-β regulate
tumor angiogenesis. J Clin Invest. 120:1151–1164. 2010.PubMed/NCBI View
Article : Google Scholar
|
|
19
|
Branchford BR and Carpenter SL: The role
of inflammation in venous thromboembolism. Front Pediatr.
6(142)2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Borazan E, Balık AA, Bozdağ Z, Arık MK,
Aytekin A, Yılmaz L, Elçi M and Başkonuş İ: NLR and prognosis in
colorectal cancer. Turk J Surg. 33:185–189. 2017.
|
|
21
|
Cushman M: Epidemiology and risk factors
for venous thrombosis. Semin Hematol. 44:62–69. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Coussens LM and Werb Z: Inflammation and
cancer. Nature. 420:860–867. 2002.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Balkwill F and Mantovani A: Inflammation
and cancer: Back to Virchow? Lancet. 357:539–545. 2001.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Jaiswal M, LaRusso NF, Burgart LJ and
Gores GJ: Inflammatory cytokines induce DNA damage in
cholangiocarcinoma. Cancer Res. 60:184–190. 2000.PubMed/NCBI
|
|
25
|
Pfeiler S, Stark K, Massberg S and
Engelmann B: Propagation of thrombosis by neutrophils.
Haematologica. 102:206–213. 2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Jain A, Chitturi S, Peters G and Yip D:
Atezolizumab and bevacizumab as first line therapy in advanced
hepatocellular carcinoma: Practical considerations in routine
clinical practice. World J Hepatol. 13:1132–1142. 2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Li CB, Ning YT, Shen NY, Wang B, Xiao H
and Luo G: Systemic treatment of liver cancer: Current status and
future perspectives. World J Hepatol. 17(107520)2025.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Huang GQ, Zhu GQ, Liu YL, Wang LR,
Braddock M, Zheng MH and Zhou MT: NLR predicts mortality after
resection. Oncotarget. 7:5429–5439. 2016.
|
|
29
|
Li SH, Wang QX, Yang ZY, Jiang W, Li C,
Sun P, Wei W, Shi M and Guo RP: NLR in HCC with PVTT/HVTT. World J
Gastroenterol. 23:3122–3132. 2017.
|
|
30
|
Qian A, Zandi A, Bucciol R and Othman M:
The role of neutrophil-to-lymphocyte ratio and
platelet-to-lymphocyte ratio as venous thromboembolism predictors
in breast cancer patients pre- and post-therapy. Blood Coagul
Fibrinolysis. 36:62–67. 2025.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Du P: Neutrophil-lymphocyte ratio is a
predictor of venous thromboembolism in gastric cancer patients.
Clin Lab 65: doi: 10.7754/Clin.Lab.2018.181005, 2019.
|
|
32
|
Selvaggio S, Brugaletta G, Abate A, Musso
C, Romano M, Di Raimondo D, Pirera E, Dattilo G and Signorelli SS:
Platelet-to-lymphocyte ratio, neutrophil-to-lymphocyte ratio and
monocyte-to-HDL cholesterol ratio as helpful biomarkers for
patients hospitalized for deep vein thrombosis. Int J Mol Med.
51(52)2023.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Signorelli SS: NLR, PLR, LMR in VTE with
cancer. Angiol Vasc Surg. 9:1–5. 2024.
|
|
34
|
Qian A, Zandi A, Bucciol R and Othman M:
NLR/PLR predict VTE in breast cancer. Blood Coagul Fibrinolysis.
36:62–67. 2025.
|
|
35
|
Go SI, Lee A, Lee US, Choi HJ, Kang MH,
Kang JH, Jeon KN, Park MJ, Kim SH and Lee GW: Clinical significance
of the neutrophil-lymphocyte ratio in venous thromboembolism
patients with lung cancer. Lung Cancer. 84:79–85. 2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Bruixola G, Niño OM, Diaz-Beveridge R,
Reche E, Salvador C, Escoin C, Akhoundova D, Segura A, Gimenez G
and Aparicio J: Baseline neutrophil-to-lymphocyte ratio (NLR) and
early toxicity as prognostic factors in advanced hepatocellular
carcinoma patients treated with sorafenib. J Clin Oncol. 33
(15_suppl)(e15159)2015.doi:10.1200/jco.2015.33.15_suppl.e15159.
|
|
37
|
Liao W, Zhang J, Zhu Q, Qin L, Yao W, Lei
B, Shi W, Yuan S, Tahir SA, Jin J and He S: Preoperative NLR in HCC
resection. Transl Oncol. 7:248–255. 2014.
|
|
38
|
Dan J, Zhang Y, Peng Z, Huang J, Gao H, Xu
L and Chen M: Postoperative NLR change after RFA. PLoS One.
8(e58184)2013.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Fan W, Zhang Y, Wang Y, Yao X, Yang J and
Li J: NLR/PLR predict survival after TACE. PLoS One.
10(e0119312)2015.
|
|
40
|
Agopian VG, Harlander-Locke M, Zarrinpar
A, Kaldas FM, Farmer DG, Yersiz H, Finn RS, Tong M, Hiatt JR and
Busuttil RW: Nomogram for recurrence after LT. J Am Coll Surg.
220:416–427. 2015.
|
|
41
|
Fu YP, Ni XC, Yi Y, Cai XY, He HW, Wang
JX, Lu ZF, Han X, Cao Y, Zhou J, et al: Inflammation-based score
predicts survival. Medicine (Baltimore). 95(e2784)2016.
|
|
42
|
Lee JS, Chon YE, Kim BK, Park JY, Kim DY,
Ahn SH, Han KH, Kang W, Choi MS, Gwak GY, et al: AFP after complete
response to TACE. Yonsei Med J. 62:12–20. 2020.
|
|
43
|
Pan YX, Sun XQ, Hu ZL, Xie W, Nie KX, Fang
AP, Zhang YY, Fu YZ, Chen JB, Wang JC, et al: Prognostic values of
alpha-fetoprotein and des-gamma-carboxyprothrombin in
hepatocellular carcinoma in China: An analysis of 4792 patients. J
Hepatocell Carcinoma. 8:657–670. 2021.PubMed/NCBI View Article : Google Scholar :
doi:10.2147/JHC.S316223.
|
|
44
|
Abdelhamed W, Shousha H and El-Kassas M:
Portal vein tumor thrombosis in HCC. Liver Res. 8:141–151.
2024.
|
|
45
|
Liu L, Liu D, He W and Zhang W: Incidence
of thrombosis in patients with hepatocellular carcinoma: systematic
review and meta-analysis. J Thromb Thrombolysis. 58:925–936.
2025.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Königsbrügge O, Posch F, Riedl J, Reitter
EM, Zielinski C, Pabinger I and Ay C: Association between decreased
serum albumin with risk of venous thromboembolism and mortality in
cancer patients. Oncologist. 21:252–257. 2016.PubMed/NCBI View Article : Google Scholar
|