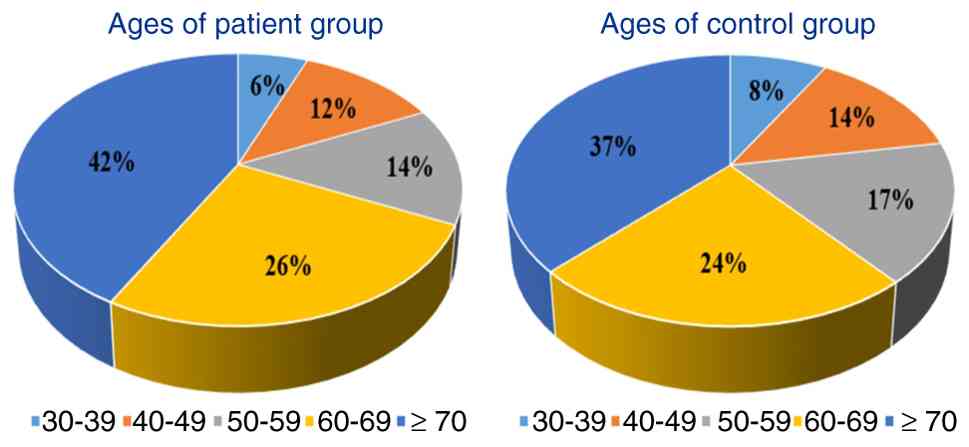Introduction
Bladder cancer is the ninth most common cancer
worldwide, with 614,298 new bladder cancer cases reported globally
in 2022; it is the sixth most common cancer in men (523,674 cases;
5.4% of all newly diagnosed cancer cases) and the 17th in women
(143,005 cases; age-standardized rate of 2.4 per 100,000), showing
a marked male predominance (1).
Bladder cancer accounts for ~2.7% of all cancer fatalities and 4.2%
of all newly discovered cancer cases, with a 5-year relative
overall survival rate of 77.9% (2). The prevalence of bladder cancer
increases with age progression and the majority of cases (80%)
occur in individuals aged >65 years (3). In total, >95% of bladder cancer
originates from the urothelium, the inner lining of the urinary
tract, and is referred to as urothelial carcinoma (4). As the urothelium spans both the upper
and lower urinary tracts, tumors can develop in either region.
Bladder cancer is classified by tumor invasiveness, as non-muscle
invasive bladder cancer or muscle-invasive blader cancer, and by
cellular grade (2). Hematuria is
the most common initial symptom leading to diagnosis; thus, an
assessment for bladder cancer is conducted only after presenting
with symptoms such as hematuria, and notably, 20% of patients will
have locally advanced or metastatic bladder cancer (5).
Cells in multicellular organisms communicate through
signaling mechanisms that regulate growth, development and
homeostasis (6). This occurs via
direct contact or chemical messengers that bind to specific
receptors on or inside the target cells. Cellular receptors can be
either intracellular or cell surface proteins; intracellular
receptors bind lipid-soluble messengers, while cell surface
receptors interact with water-soluble molecules (7). Disruptions in these pathways can lead
to cancer-related changes such as uncontrolled growth and
resistance to cell death (8).
Several tissues, including the testes, ovaries, endometrium,
prostate, kidneys, liver, circulatory system, brain, neurological
system, skeletal muscle, skin and bladder, have been shown to
express and act upon androgens and estrogen binding to their
receptors (9). Androgens, mainly
testosterone and its effective derivative dihydrotestosterone
(DHT), act through the androgen receptor (AR), a ligand-activated
transcription factor of the nuclear receptor superfamily (10). The AR, a 110 kDa protein with 919
amino acids, serves a key role in regulating the expression of
genes involved in numerous physiological and pathological processes
(11). Conversely, estrogens,
particularly estradiol (E2), act mainly via two estrogen receptors
(ERs), Erα and Erβ [which are encoded by estrogen receptor 1 (ESR1)
and estrogen receptor 2 (ESR2) genes, respectively]. Important
physiological processes, including reproduction, metabolism, bone
density maintenance and the development of estrogen-responsive
malignancies, such as endometrial and breast cancer, are known to
be mediated by these receptors (12). The ERs belong to a nuclear receptor
superfamily that has the capacity to convert extracellular signals
into transcriptional signals (13).
Understanding the interplay between
androgen/estrogen hormones and their receptors may not only clarify
the observed sex disparity in bladder cancer incidence but also
provide new therapeutic targets. Hormone receptor modulators, such
as AR antagonists or selective ER modulators, are being
investigated for their potential to halt disease progression and
improve treatment responses in patients with bladder cancer
(11). The present study aimed to
examine the levels of sex hormones, specifically estrogen and
testosterone, in women diagnosed with bladder cancer, and to
evaluate alterations in the expression of their corresponding
receptors, including AR and ESR1, within bladder tissue.
Furthermore, the present study aimed to investigate whether
hormonal and receptor disorder may serve as risk factors in the
pathogenesis and progression of bladder cancer in women.
Patients and methods
Study design and participants
In the present study, participants were
prospectively recruited between July 2024 and January 2025, from
various locations, including the Medicine City (Ghazi Al-Hariri
Surgical Specialties Hospital and Al-Amal National Hospital in
Baghdad, Iraq) as well as Al-Shifa Center for Oncology Treatment in
Maysan, Iraq. Sample collection and data acquisition were conducted
in accordance with standardized clinical procedures and predefined
study protocols.
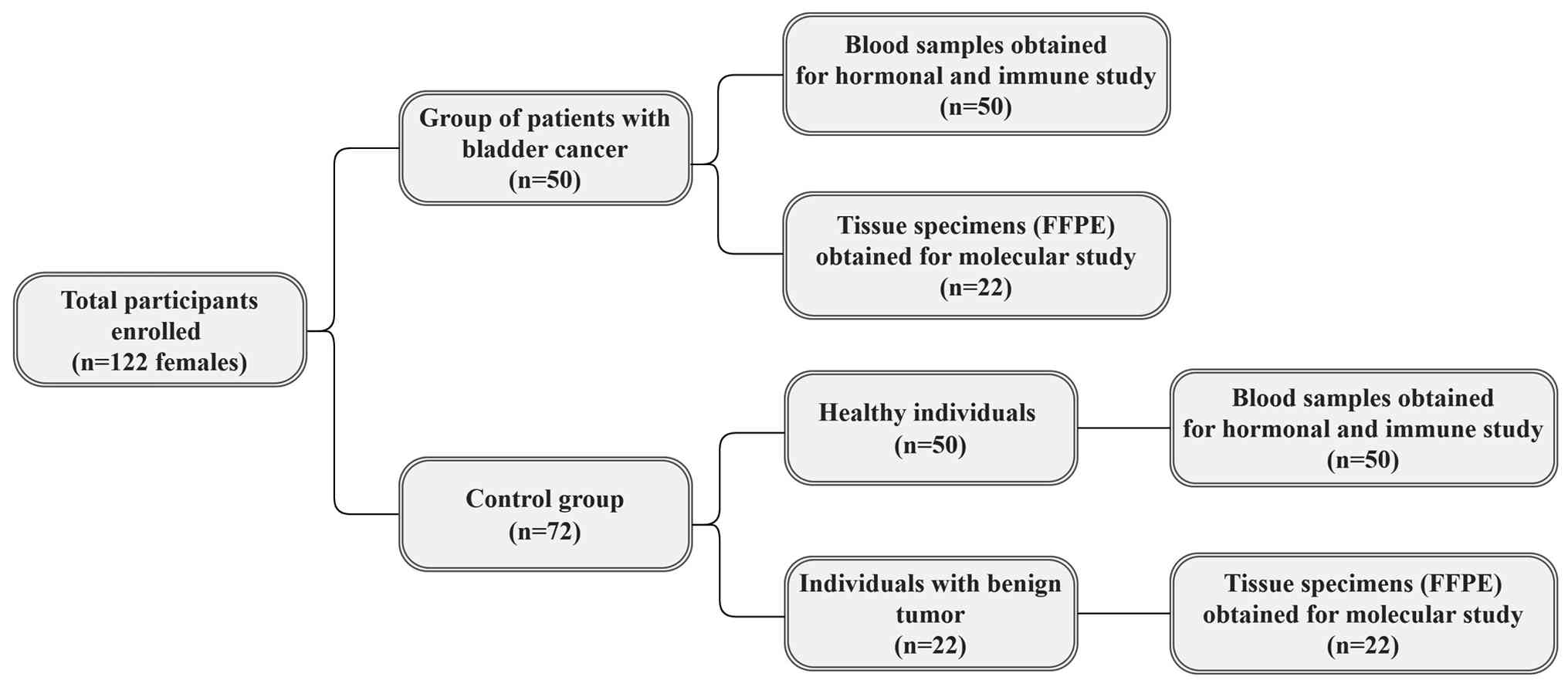
The present case-control study involved 122
participants, all of whom were women of different ages ranging from
35-80 years, with a median age of 64 years. Among the participants,
50 patients who were diagnosed with bladder cancer (Table I) provided blood samples for
hormonal analysis, of whom 22 also contributed tissue specimens [as
formalin-fixed, paraffin-embedded (FFPE) blocks] for molecular
studies. The control group included 72 age-matched individuals of
whom 50 provided blood samples for hormonal comparison with the
patient group, whereas the remaining 22 had benign tumors that were
histologically diagnosed as benign urothelial papilloma and
endometriosis of the urinary bladder. These diagnoses were
confirmed by histopathological examination, with no evidence of
malignancy or dysplasia. FFPE tissue specimens were collected from
these cases and used as a comparison group for the bladder cancer
tissue data (Fig. 1). For both the
patient and control groups, venous blood samples were obtained and
immediately processed by centrifugation at 3,000 x g for 10 min at
room temperature (20-25˚C) to separate the serum, which was then
aliquoted and stored in a deep freeze until further analysis. The
present study was performed in compliance with the Declaration of
Helsinki and approved by the Institutional Review Committee of
College of Science, in cooperation with the Local Ethics Committee
of the Biology Department, Mustansiriyah University (Baghdad, Iraq;
January 2024; approval. no. BCSMU/1024/0062Z).
 | Table IClinicopathological characteristics of
patients (n=50) with bladder cancer. |
Table I
Clinicopathological characteristics of
patients (n=50) with bladder cancer.
| Characteristic | No. of patients |
|---|
| Median age, years
(range) | 67 (37-80) |
| Sex | |
|
Female | 50(100) |
| T stage | |
|
Ta | 2(4) |
|
T1 | 19(38) |
|
T2 | 16(32) |
|
T3 | 13(26) |
| N stage | |
|
N0 | 50(100) |
| M stage | |
|
M0 | 50(100) |
| Tumor grade | |
|
G1 | 11(22) |
|
G2 | 15(30) |
|
G3 | 24(48) |
Inclusion criteria
The inclusion criteria were as follows: Patient
group: i) Female patients of any age with a confirmed diagnosis of
malignant bladder tumor based on histopathological examination; and
ii) participants who provided informed consent for study
participation. Disease stage or grade was not used as an
eligibility criterion as all stages and grades of bladder cancer
were included. The control group was: i) Healthy female volunteers
age-matched to the patient group; and ii) female patients of any
age with a confirmed diagnosis of benign bladder tumor based on
histopathological examination.
Exclusion criteria. The exclusion criteria
were as follows: i) Male participants; ii) individuals with any
other diseases, including malignancies other than bladder cancer,
autoimmune or chronic inflammatory conditions or infectious
diseases; iii) FFPE tissue samples with inadequate quantity, poor
preservation or RNA of insufficient quality for gene expression
analysis; and iv) participants who had recently undergone
chemotherapy, radiotherapy or immunotherapy.
Primer design and gene targets
Primers were designed using Primer 3 Plus (version
4; https://www.primer3plus.com) and their
reference sequences were validated against the National Center for
Biotechnology Information database using UCSC Genome Browser Basic
Local Alignment Search Tool (https://blast.ncbi.nlm.nih.gov/blast.cgi). The primers
were synthesized and lyophilized by Alpha DNA. Table II contains all of the primer
sequences utilized in the present study.
 | Table IISequences and specifications of the
primers used in the present study. |
Table II
Sequences and specifications of the
primers used in the present study.
| Primer | Sequence
(5'→3') | Primer size,
bp | Ta, ˚C |
|---|
| AR | | | 58 |
|
Forward |
GAGATGATACCCTCCCAGCA | 20 | |
|
Reverse |
CATTTGCAGGGTTTCTGGTT | 20 | |
| ESR1 | | | 58 |
|
Forward |
AGCACCCTGAAGTCTCTGGA | 20 | |
|
Reverse |
GATGTGGGAGAGGATGAGGA | 20 | |
| GAPDH | | | 60 |
|
Forward |
GGCCTCCAAGGAGTAAGACC | 20 | |
|
Reverse |
AGGGGTCTACATGGCAACTG | 20 | |
RNA extraction and reverse
transcription-quantitative PCR (RT-qPCR)
The TransZol Up Plus RNA Kit Reagent (Tissue)
(TransGen Biotech Co., Ltd.; cat. no. ER501-01) was used to extract
total RNA from each bladder tissue sample according to the
manufacturer's instructions. RNA concentration was determined using
a NanoDrop spectrophotometer (Thermo Fisher Scientific, Inc.) and
RNA purity was assessed by the A260/A280
ratio, which was ~2.0. Total RNA was reverse-transcribed to
complementary DNA (cDNA) using the EasyScript® One-Step
gDNA Removal and cDNA Synthesis SuperMix Kit (TransGen Biotech Co.,
Ltd.; cat. no. AE311-02) according to the manufacturer's
instructions. The reaction was performed at 25˚C for 10 min in a
final volume of 20 µl, with 4 µl of total RNA and a random
primer.
Target gene expression was confirmed by qPCR, a
sensitive technique for assessing steady-state mRNA levels. The
Qiagen Rotor Gene Q Real-time PCR System (Applied Biosystems;
Thermo Fisher Scientific, Inc.) was used to perform qPCR. The
expression levels and fold changes of the target genes and the
housekeeping gene, GAPDH, were quantified using the
TransStart® Top Green qPCR SuperMix Kit (TransGen
Biotech Co., Ltd.; cat. no. AQ131-01) in a 20 µl reaction mixture
consisting of 10 µl 2X qPCR SuperMix, 4 µl nuclease-free water, 1
µl of each forward and reverse primer (10 µM) and 4 µl of cDNA. The
thermocycler protocol included an initial enzyme activation step at
94˚C for 30 sec, followed by 40 cycles of denaturation at 94˚C for
5 sec, annealing at 58˚C for 15 sec (60˚C for GAPDH) and extension
at 72˚C for 20 sec. A dissociation curve analysis was performed at
55-95˚C to verify product specificity.
The RT-qPCR efficiency of each primer set was
determined by serial dilutions of cDNA template from the bladder
tissue of women that diagnosed with malignant and benign tumor.
standard curve was generated by preparing serial dilutions of a
cDNA template, and each primer pair was tested across the dilution
series. The slope of the linear equation was applied to calculate
the efficiency according to the equation
E=(10(-1/slope)-1) x100 where E=PCR efficiency (%) and
slope=slope of standard curve. The PCR efficiency of studied genes
ranged from 89.4 to 103.8%, slope from -3.61 to -3.23 and
R2 from 0.995 to 0.998.
GAPDH was selected as the internal reference gene
for normalization of the qPCR data; it encodes a key glycolytic
enzyme that is constitutively and ubiquitously expressed across
mammalian tissues, demonstrating high amplification efficiency and
stable expression in both normal and pathological conditions,
including malignant and benign tissues, ensuring reliable and
consistent detection across all samples (14). Fold changes in the quantified
expression levels of mRNA were determined using the relative cycle
threshold (2-∆∆Cq) method as originally described by
Livak and Schmittgen (15). Each
sample was run in duplicate and the mean Cq values for both GAPDH
and the target genes were recorded for the patients and
controls.
Statistical analysis
Statistical analyses were performed using GraphPad
Prism (version 9.2; Dotmatics). Additionally, a priori power
analysis was performed using G*Power (version 3.1.9.7;
Heinrich-Heine-Universität Düsseldorf, Germany). The present study
included 50 patients and 72 control participants. Based on a
priori power analysis assuming a medium effect size (Cohen's
d=0.5), a significance level of α=0.05 and a two-sided test, the
statistical power of the present sample was 80%. Normality of the
continuous variables was evaluated using the Kolmogorov-Smirnov
(KS) and Shapiro-Wilk (SW) tests. Based on the normality
evaluation, for variables that satisfied the assumption of
normality, comparisons between two independent groups were
performed using an unpaired two-tailed t-test. For variables that
did not satisfy the assumption of normality, non-parametric
statistical methods were applied: Comparisons between two
independent groups were performed using the Mann-Whitney U test.
Monotonic associations between variables were examined using
Spearman's rank correlation coefficient (ρ). P<0.05 was
considered to indicate a statistically significant difference.
Results
Age of the patient and control
groups
The age distribution of participants was divided
into five categories. Among the 50 patients with bladder cancer,
the age groups were 30-39 years (n=3; 6%), 40-49 years (n=6; 12%),
50-59 years (n=7; 14%), 60-69 years (n=13; 26%) and ≥70 years
(n=21; 42%) (Fig. 2). In the
control group (n=72), the age distributions were similar, with
30-39 years (n=6; 8%), 40-49 years (n=10; 14%), 50-59 years (n=12;
17%), 60-69 years (n=17; 24%) and ≥70 years (n=27; 37%) (Fig. 2). Unpaired t-test analysis revealed
no statistically significant differences in the age distribution
between the patients and controls (P>0.05), indicating
appropriate age-matching across the groups.
Normality testing
Prior to conducting the statistical analysis, the
distribution of the data was assessed using the KS and SW tests for
normality (Table III). Regarding
the serum hormone levels, testosterone demonstrated normal
distribution in both the control (KS test: P=0.199; SW test:
P=0.140) and patient (KS test: P=0.200; SW test: P=0.181) groups;
however, the estrogen levels in the control group showed a normal
distribution (KS test: P=0.200; SW test: P=0.955), while the
patient group exhibited significant deviation from normality (KS
test: P=0.006; SW test: P=0.001).
 | Table IIINormal distribution assessment of
serum estrogen and testosterone concentrations in patients (n=50)
with bladder cancer and healthy controls (n=50). |
Table III
Normal distribution assessment of
serum estrogen and testosterone concentrations in patients (n=50)
with bladder cancer and healthy controls (n=50).
| |
Kolmogorov-Smirnova | Shapiro-Wilk |
|---|
| Group | Statistic | df | P-value | Statistic | df | P-value |
|---|
| Testosterone | | | | | | |
|
Control | 0.217 | 50 | 0.199 | 0.883 | 50 | 0.140 |
|
Patient | 0.202 | 50 | 0.200 | 0.899 | 50 | 0.181 |
| Estrogen | | | | | | |
|
Control | 0.134 | 50 | 0.200 | 0.978 | 50 | 0.955 |
|
Patient | 0.302 | 50 | 0.006 | 0.723 | 50 | 0.001 |
For receptor gene expression in bladder tissues,
ESR1 expression showed mixed results, with the control group
demonstrating normal distribution (KS test: P=0.186; SW test:
P=0.045), while the patient group showed significant deviation from
normality (KS test: P=0.000; SW test: P=0.003). AR expression in
the control group showed normal distribution (KS test: P=0.200; SW
test: P=0.557), whereas the patient group demonstrated significant
deviation from normality (KS test: P=0.085; SW test: P=0.029).
These results are summarized in Table
IV.
 | Table IVNormal distribution assessment of
ESR1 and AR gene expression in bladder tissues from the patient
(n=22) and control groups (n=22). |
Table IV
Normal distribution assessment of
ESR1 and AR gene expression in bladder tissues from the patient
(n=22) and control groups (n=22).
| |
Kolmogorov-Smirnova | Shapiro-Wilk |
|---|
| Group | Statistic | df | P-value | Statistic | df | P-value |
|---|
| ESR1 | | | | | | |
|
Control | 0.220 | 22 | 0.186 | 0.841 | 22 | 0.045 |
|
Patient | 0.290 | 22 | 0.000 | 0.709 | 22 | 0.003 |
| AR | | | | | | |
|
Control | 0.192 | 22 | 0.200 | 0.940 | 22 | 0.557 |
|
Patient | 0.237 | 22 | 0.085 | 0.837 | 22 | 0.029 |
Serum reproductive hormone levels
The serum levels of estrogen and testosterone in the
patient (n=50) and healthy control (n=50) groups were evaluated and
compared using the Mann-Whitney U test (Table V). The serum testosterone levels
were significantly higher in the patient group (mean rank=54.40)
compared with the control group (mean rank=26.60), with a
Mann-Whitney U value of 244.000 (Z=-5.350; P<0.001). Similarly,
serum estrogen levels showed a significant increase in the patient
group (mean rank=55.52) relative to the control group (mean
rank=35.48), with a Mann-Whitney U value of 561.500 (Z=-3.640;
P<0.001). These findings indicate marked dysregulation of the
circulating reproductive hormones in female patients with bladder
cancer, characterized by elevated levels of both androgens and
estrogens.
 | Table VStatistical analysis of the serum
testosterone and estrogen levels in the patient (n=50) and control
(n=50) groups using the Mann-Whitney U test. |
Table V
Statistical analysis of the serum
testosterone and estrogen levels in the patient (n=50) and control
(n=50) groups using the Mann-Whitney U test.
| Statisitical
term | Testosterone | Estrogen |
|---|
| Mean rank | | |
|
Control | 26.60 | 35.48 |
|
Patient | 54.40 | 55.52 |
| Mann-Whitney U | 244.000 | 561.500 |
| Wilcoxon W | 1,064.000 | 1,596.500 |
| Z-value | -5.350 | -3.640 |
| P-value | <0.001 | <0.001 |
Analysis of gene expression
The expression levels of the target genes, AR and
ESR1, in the bladder cancer tissue samples were assessed using
RT-qPCR amplification curves, which were compared with the
housekeeping gene internal control, GAPDH (Fig. S1, S2 and S3). The stable and consistent expression
of GAPDH was confirmed by the amplification plots, which showed
early Cq values with little change between samples, demonstrating
its appropriateness for the normalization of the target genes.
However, the AR and ESR1 amplification curves showed inconsistency
in the Cq values, suggesting that their transcript abundances were
different to GAPDH. The specificity and effectiveness of the
reactions were validated by the sigmoidal amplification patterns
shown for every gene, which were distinguished by discrete
exponential phases and notable separation from the negative
controls. The assumption of identical amplification efficiencies
was further supported by the comparable slopes of the target and
housekeeping gene curves, which guaranteed the accuracy of the
relative quantification.
Statistical analysis of gene expression in the
bladder tissues revealed contrasting patterns for AR and ESR1
expression (Table VI). AR gene
expression was significantly upregulated in the bladder cancer
tissues (mean rank=28.05) compared with the benign control tissues
(mean rank=14.30), with a Mann-Whitney U value of 76.000 (Z=-3.629;
P=0.002). This marked increase in AR expression suggests enhanced
androgenic signaling capacity within the bladder cancer tumor
microenvironment. By contrast, ESR1 gene expression was
significantly downregulated in the bladder cancer tissues (mean
rank=17.59) compared with the control tissues (mean rank=25.80),
with a Mann-Whitney U value of 134.000 (Z=-2.169; P=0.030). This
reduction in ESR1 expression, occurring despite elevated
circulating estrogen levels, indicates a disruption in the
estrogen-mediated signaling pathways.
 | Table VIStatistical comparison of the
androgen and estrogen receptor gene expression levels in the
bladder tissues from the patient (n=22) and control (n=22) groups
using the Mann-Whitney U test. |
Table VI
Statistical comparison of the
androgen and estrogen receptor gene expression levels in the
bladder tissues from the patient (n=22) and control (n=22) groups
using the Mann-Whitney U test.
| Statistical
term | Androgen
receptor | Estrogen
receptor-1 |
|---|
| Mean rank | | |
|
Control | 14.30 | 25.80 |
|
Patient | 28.05 | 17.59 |
| Mann-Whitney U | 76.000 | 134.000 |
| Wilcoxon W | 286.000 | 387.000 |
| Z-value | -3.629 | -2.169 |
| P-value | 0.002 | 0.030 |
Collectively, these findings demonstrate a distinct
pattern of hormone receptor dysregulation in female patients with
bladder cancer, characterized by enhanced AR expression coupled
with diminished ESR1 expression, which may contribute to an
imbalance favoring pro-oncogenic androgen signaling while reducing
the estrogen-mediated protective effects.
Correlation between hormone levels and
bladder cancer stage
To further explore the clinical significance of
hormonal dysregulation, Spearman's correlation analysis was
performed to examine the correlation between serum hormone levels
and bladder cancer stage in female patients (n=50; Table VII). The analysis revealed a weak
positive correlation between serum testosterone levels and cancer
stage (ρ=0.263; P=0.05), suggesting a pattern where higher
testosterone levels tended to be correlated with more advanced
disease stages. More notably, the serum estrogen levels
demonstrated a moderate positive correlation with cancer stage
(ρ=0.512; P=0.0015), indicating a significant correlation between
elevated estrogen levels and disease progression. This finding
suggests that estrogen levels may serve as a potential indicator of
tumor advancement in female patients with bladder cancer.
 | Table VIICorrelation of the serum testosterone
and estrogen levels with bladder cancer stage using Spearman's rank
correlation coefficient. |
Table VII
Correlation of the serum testosterone
and estrogen levels with bladder cancer stage using Spearman's rank
correlation coefficient.
| Statistical
term | Correlation with
stage of bladder cancer |
|---|
| Testosterone | |
|
Spearman's
correlation | 0.263 |
|
P-value
(two-tailed) | 0.05 |
|
No. of
samples | 50 |
| Estrogen | |
|
Spearman's
correlation | 0.512 |
|
P-value
(two-tailed) | 0.0015 |
|
No. of
samples | 50 |
Discussion
Age represents a key demographic parameter
consistently included in cancer epidemiological studies; it is
recognized as one of the influential and thoroughly investigated
determinants of cancer risk. Since the occurrence of most
malignancies notably rises with advancing years, cancer is widely
considered an age-related condition (16,17).
Beyond its role as a demographic factor, advancing age is
biologically linked to changes that may contribute to bladder
cancer development. For example, aging is associated with
cumulative genetic and epigenetic alterations, impaired DNA repair
mechanisms and increased oxidative stress, all of which can promote
carcinogenesis (18).
Additionally, age-related hormonal alterations, particularly
fluctuations in androgen and estrogen levels, can influence
urothelial cell function and proliferation, thereby increasing
vulnerability to bladder cancer (19). This age-hormone relationship
provides important context for understanding the hormonal
dysregulation observed in the present study.
Hormonal signals serve a critical role in regulating
the growth, differentiation and survival of cells throughout the
body. These same biological signals can also contribute to cancer
when they become dysregulated. Hormones act through highly specific
communication systems, including endocrine, paracrine and autocrine
signaling, that influence gene expression and cellular behavior
(20). The findings of the present
study demonstrated a clear hormonal imbalance in women with bladder
cancer, characterized by significantly elevated levels of both
estrogen and testosterone in serum compared with the control group.
Increased testosterone levels in women with bladder cancer suggests
dysregulation of androgen metabolism or adrenal androgen
overproduction. Testosterone and its more potent derivative, DHT,
signal through the AR, which is also expressed in bladder tissues
(21). Additionally, the increased
circulating estrogen levels in female patients with bladder cancer
may result from altered systemic hormone metabolism or local
estrogen synthesis within the tumor microenvironment, facilitated
by elevated aromatase enzyme activity that converts androgens to
estrogens (22).
Correspondingly, the results of the present study
demonstrated a significant increase in AR gene expression in
bladder cancer tissues compared with benign control tissues. This
elevation was consistently observed across the analyzed samples,
indicating a clear difference in expression levels between
malignant and non-malignant bladder tissues. The alignment between
elevated testosterone levels and increased AR expression suggests
that enhanced androgenic signaling may contribute to tumor
development or progression (21).
Notably, even in women, androgen/AR signaling can influence bladder
cancer growth, indicating that androgens are not exclusively male
hormones in the context of cancer pathogenesis (11). These findings align with numerous
investigations examining AR expression variations in human bladder
tumors and the association with different disease characteristics
(23,24). Early research has shown that
bladder cancer tissue has higher levels of AR than control tissue,
indicating that AR is upregulated in malignancy (25). Furthermore, AR expression has been
strongly associated with tumor stage and grade, with patients
harboring AR-positive tumors demonstrating a worse prognosis than
those with AR-negative tumors (26). Analysis of both non-metastatic and
metastatic malignancies has revealed that a higher percentage of
metastatic lesions express AR compared with primary tumors,
supporting the concept that AR-positivity is associated with
metastatic potential (27).
However, other research has shown no statistically significant
difference in AR expression between male and female patients, with
AR expressed at comparable levels in both sexes (28).
The interplay between elevated androgen levels and
AR expression contributes to bladder cancer proliferation and
differentiation through multiple interconnected mechanisms that
promote cellular proliferation and malignant transformation
(29). Upon androgen binding, the
AR complex translocates to the nucleus and functions as a
transcription factor, upregulating cell cycle regulators, including
cyclins and cyclin-dependent kinases, that drive G1/S
phase transition while simultaneously suppressing apoptotic
pathways through increased Bcl-2 expression and decreased
pro-apoptotic factor activity (30). AR signaling also exhibits
cross-talks with critical growth factor pathways, enhancing EGFR
activity and activating the PI3K/AKT/mTOR cascade, both of which
are essential for cell survival and proliferation, while
stimulating VEGF expression to promote tumor angiogenesis (31). Given these findings, previous
hypotheses have emphasized androgenic activity, rather than AR
expression alone, as a key driver of bladder cancer biology, since
systemic testosterone levels strongly influence AR activation
(32).
In contrast to the upregulated androgen pathway, the
findings of the present study demonstrated elevated estrogen levels
accompanied by downregulation of ESR1 expression in bladder cancer
tissue, indicating a disruption in estrogen/ER signaling. This
disruption in the estrogen/ER axis has notable implications for
bladder cancer pathogenesis, as estrogen signaling has been
demonstrated to regulate critical cellular mechanisms including
cell cycle control, apoptosis and differentiation, thereby exerting
protective effects against tumorigenesis in various tissues
(33). The protective role of
estrogen in bladder carcinogenesis is supported by epidemiological
evidence showing that postmenopausal women have a higher risk of
bladder cancer than premenopausal women (34). Furthermore, women who reach
menopause at a younger age have a markedly increased risk of
bladder cancer, supporting the concept that estrogens may inhibit
bladder cancer incidence (35).
Additional clinical studies have demonstrated that higher
frequencies of estrogen exposure lead to lower bladder cancer
incidence. For example, parous women who have experienced elevated
E2 during pregnancy and those who used estrogen and progestin for
hormonal therapy have a lower risk of bladder cancer formation,
again suggesting that high estrogen exposure decreases bladder
cancer risk (36). Molecularly,
ESR1 expression has been found to be significantly downregulated in
high-grade or muscle-invasive bladder tumors (37). Consistently, Kontos et al
(38) reported a clear and
significant downregulation of ESR1 expression in bladder cancer
tissue, further highlighting the homogeneity in ESR1 expression
profiles across different bladder cancer subtypes and stages.
Several mechanisms have been proposed to explain how
altered ER expression and impaired ER signaling contribute to
bladder cancer progression. Gucalp et al (27) proposed that the observed reduction
in ER expression may result from epigenetic silencing through
promoter hypermethylation of the ESR1 gene, a well-characterized
phenomenon documented across various malignancies. A mechanistic
study has suggested that ESR1 controls the expression of inositol
polyphosphate-4-phosphatase type IIB to reduce AKT activity and
consequently suppress bladder cancer cell proliferation (39). Beyond transcriptional regulation,
ER signaling has been implicated in modulating non-coding RNA
networks, including microRNAs (miRs), circular RNAs (circRNAs) and
enhancer RNAs (eRNAs), all of which serve crucial roles in bladder
cancer progression. ESR1 has been demonstrated to induce miR-4324
expression through direct promoter binding in bladder cancer cells,
thereby suppressing cellular proliferation and metastatic potential
(40). Furthermore, ESR1
downregulates circ_0023642 expression by modulating its host gene,
UVRAG, which subsequently upregulates miR-490-5p and leads to
decreased EGFR expression, ultimately inhibiting bladder cancer
cell invasion (41). Conversely,
knockdown experiments targeting estrogen-responsive eRNAs,
specifically eGREB1 and P2RY2e, in bladder cancer cell lines
resulted in notable inhibition of proliferation, migration and
invasion, coupled with enhanced apoptosis, suggesting that these
eRNAs may exert oncogenic functions (42). The simultaneous elevation of AR
signaling and suppression of ER signaling observed in the present
study suggests that a critical hormonal imbalance may drive bladder
cancer progression in female patients by tipping cellular signaling
towards growth and survival rather than differentiation or
suppression.
The present study has certain limitations that
should be considered. Specifically, only AR and ESR1 were
evaluated, while other sex hormone receptors, including ESR2 and
progesterone receptor, which may also have role in the pathogenesis
of bladder cancer. Furthermore, receptor expression was assessed
only at the mRNA level through gene expression analysis, therefore,
the functional relevance of these findings needs to be further
confirmed at the protein level in future studies.
In conclusion, the findings of the present study
demonstrate a distinct pattern of hormonal dysregulation in female
patients with bladder cancer, characterized by elevated serum
levels of both testosterone and estrogen and significant changes in
receptor expression. Despite an observed increase in circulating
estrogen, ESR1 gene expression was downregulated in bladder cancer
tissues, indicating disrupted estrogen-mediated protective
signaling. Conversely, AR expression was significantly upregulated,
which together with elevated testosterone levels, suggests enhanced
pro-oncogenic androgenic activity within the tumor
microenvironment. Collectively, these findings revealed a critical
hormonal imbalance favoring pro-oncogenic androgen signaling while
diminishing estrogen-related protection, which may represent a
notable factor in bladder cancer pathogenesis and progression in
women. These results highlighted the potential importance of the
AR/ER axis as a therapeutic target and suggest that modulating AR
activity or restoring ER function may offer novel treatment
strategies for female patients with bladder cancer.
Supplementary Material
Reverse transcription-quantitative PCR
amplification plot showing the fluorescence intensity of androgen
receptor gene expression across different cycles in bladder tissue
samples from controls and patients with bladder cancer.
Reverse transcription-quantitative PCR
amplification plot showing the fluorescence intensity of estrogen
receptor 1 gene expression across different cycles in bladder
tissue samples from controls and patients with bladder cancer.
Reverse transcription-quantitative PCR
amplification plot showing the fluorescence intensity of
housekeeping gene GAPDH expression across different cycles in
bladder tissue samples from controls and patients with bladder
cancer.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
NML and IEA contributed to design and planning of
study. FGL collected and analyzed data and wrote the manuscript.
NML reviewed and edited the manuscript. IEA participated in data
processing. NML and IEA supervised the study. FGL and NML confirm
the authenticity of all the raw data. All authors read and approved
the final version of the manuscript.
Ethics approval and consent to
participate
Ethical approval was obtained from the relevant
institutional review board of Mustansiriyah University (approval
no. BCSMU/1024/0062Z). All data were prospectively obtained from
medical records in compliance with the principles to 1964 Helsinki
Declaration and its later amendments. Written informed consent was
obtained from all participants included in the present study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
GLOBOCAN 2022: Bladder cancer 9th most
common worldwide-World Bladder Cancer Patient Coalition.
|
|
2
|
Leslie SW, Soon-Sutton TL and Aeddula NR:
Bladder cancer. In: StatPearls. StatPearls Publishing, Treasure
Island, FL, 2025.
|
|
3
|
Mushtaq J, Thurairaja R and Nair R:
Bladder cancer. Surgery (Oxford). 37:529–537. 2019.
|
|
4
|
van Kessel K: Personalized bladder cancer
management. Erasmus Universiteit Rotterdam (EUR), 2018.
|
|
5
|
Kamat AM and Black PC: Bladder cancer: A
practical guide. Springer International Publishing, Cham, 2021.
|
|
6
|
Heldin CH, Lu B, Evans R and Gutkind JS:
Signals and receptors. Cold Spring Harb Perspect Biol.
8(a005900)2016.
|
|
7
|
Khalil B, Miller EJ and Lappin SL:
Physiology, cellular receptors. In: StatPearls. StatPearls
Publishing, Treasure Island, FL, 2025.
|
|
8
|
Iqbal MJ, Kabeer A, Abbas Z, Siddiqui HA,
Calina D, Sharifi-Rad J and Cho WC: Interplay of oxidative stress,
cellular communication and signaling pathways in cancer. Cell
Commun Signal. 22(7)2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hu H, Zhou H and Xu D: A review of the
effects and molecular mechanisms of dimethylcurcumin (ASC-J9) on
androgen receptor-related diseases. Chem Biol Drug Des. 97:821–835.
2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Estébanez-Perpiñá E, Bevan CL and McEwan
IJ: Eighty years of targeting androgen receptor activity in
prostate cancer: The fight goes on. Cancers (Basel).
13(509)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Li P, Chen J and Miyamoto H: Androgen
receptor signaling in bladder cancer. Cancers (Basel).
9(20)2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chen P, Li B and Ou-Yang L: Role of
estrogen receptors in health and disease. Front Endocrinol
(Lausanne). 13(839005)2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Eyster KM: The estrogen receptors: An
overview from different perspectives. Methods Mol Biol. 1366:1–10.
2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Joshi CJ, Ke W, Drangowska-Way A, O'Rourke
EJ and Lewis NE: What are housekeeping genes? PLoS Comput Biol.
18(e1010295)2022.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
16
|
White MC, Holman DM, Boehm JE, Peipins LA,
Grossman M and Henley SJ: Age and cancer risk: A potentially
modifiable relationship. Am J Prev Med. 46:S7–S15. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Al-Adilee MN and Al-Abassi HM: The Role of
Caspase-8, MLKL and RIPK1 in Iraqi Patients' women with breast
cancer. Al-Mustansiriyah J Sci. 36:22–33. 2025.
|
|
18
|
Rossi DJ, Jamieson CHM and Weissman IL:
Stems cells and the pathways to aging and cancer. Cell.
132:681–696. 2008.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Biagetti B and Puig-Domingo M: Age-related
hormones changes and its impact on health status and lifespan.
Aging Dis. 14:605–620. 2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ganguly S, Naik D, Muskara A and Mian OY:
The nexus of endocrine signaling and cancer: How steroid hormones
influence genomic stability. Endocrinology.
162(bqaa177)2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Miyamoto H, Yang Z, Chen YT, Ishiguro H,
Uemura H, Kubota Y, Nagashima Y, Chang YJ, Hu YC, Tsai MY, et al:
Promotion of bladder cancer development and progression by androgen
receptor signals. J Natl Cancer Inst. 99:558–568. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Cao J, Yang X, Li J, Wu H, Li P, Yao Z,
Dong Z and Tian J: Screening and identifying immune-related cells
and genes in the tumor microenvironment of bladder urothelial
carcinoma: Based on TCGA database and bioinformatics. Front Oncol.
9(1533)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chen J, Huang CP, Quan C, Zu X, Ou Z, Tsai
YC, Messing E, Yeh S and Chang C: The androgen receptor in bladder
cancer. Nat Rev Urol. 20:560–574. 2023.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Sottnik JL, Vanderlinden L, Joshi M,
Chauca-Diaz A, Owens C, Hansel DE, Sempeck C, Ghosh D and
Theodorescu D: Androgen receptor regulates CD44 expression in
bladder cancer. Cancer Res. 81:2833–2846. 2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Lombard AP and Mudryj M: The emerging role
of the androgen receptor in bladder cancer. Endocrine-related
cancer. 22:R265–R277. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Rangel N, Rondon-Lagos M, Annaratone L,
Osella-Abate S, Metovic J, Mano MP, Bertero L, Cassoni P, Sapino A
and Castellano I: The role of the AR/ER ratio in ER-positive breast
cancer patients. Endocr Relat Cancer. 25:163–172. 2018.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Gucalp A, Tolaney S, Isakoff SJ, Ingle JN,
Liu MC, Carey LA, Blackwell K, Rugo H, Nabell L, Forero A, et al:
Phase II trial of bicalutamide in patients with androgen
receptor-positive, estrogen receptor-negative metastatic breast
cancer. Clin Cancer Res. 19:5505–5512. 2013.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Mashhadi R, Pourmand G, Kosari F, Mehrsai
A, Salem S, Pourmand MR, Alatab S, Khonsari M, Heydari F, Beladi L
and Alizadeh F: Role of steroid hormone receptors in formation and
progression of bladder carcinoma: A case-control study. Urol J.
11:1968–1973. 2014.PubMed/NCBI
|
|
29
|
Godoy G, Gakis G, Smith CL and Fahmy O:
Effects of androgen and estrogen receptor signaling pathways on
bladder cancer initiation and progression. Bladder Cancer.
2:127–137. 2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
de Brot S and Mongan NP: The cell cycle
and androgen signaling interactions in prostate cancer. In:
Precision Molecular Pathology of Prostate Cancer. Robinson BD,
Mosquera JM, Ro JY and Divatia M (eds). Springer International
Publishing, Cham, pp381-404, 2018.
|
|
31
|
Izumi K, Zheng Y, Li Y, Zaengle J and
Miyamoto H: Epidermal growth factor induces bladder cancer cell
proliferation through activation of the androgen receptor. Int J
Oncol. 41:1587–1592. 2012.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Izumi K, Mizokami A, Lin WJ, Lai KP and
Chang C: Androgen receptor roles in the development of benign
prostate hyperplasia. Am J Pathol. 182:1942–1949. 2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Goto T and Miyamoto H: The role of
estrogen receptors in urothelial cancer. Front Endocrinol
(Lausanne). 12(643870)2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
McGrath M, Michaud DS and De Vivo I:
Hormonal and reproductive factors and the risk of bladder cancer in
women. Am J Epidemiol. 163:236–244. 2006.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Wolpert BJ, Amr S, Ezzat S, Saleh D, Gouda
I, Loay I, Hifnawy T, Mikhail NN, Abdel-Hamid M, Zhan M, et al:
Estrogen exposure and bladder cancer risk in Egyptian women.
Maturitas. 67:353–357. 2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Davis-Dao CA, Henderson KD,
Sullivan-Halley J, Ma H, West D, Xiang YB, Gago-Dominguez M, Stern
MC, Castelao JE, Conti DV, et al: Lower risk in parous women
suggests that hormonal factors are important in bladder cancer
etiology. Cancer Epidemiol Biomarkers Prev. 20:1156–1170.
2011.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Miyamoto H, Yao JL, Chaux A, Zheng Y, Hsu
I, Izumi K, Chang C, Messing EM, Netto GJ and Yeh S: Expression of
androgen and oestrogen receptors and its prognostic significance in
urothelial neoplasm of the urinary bladder. BJU Int. 109:1716–1726.
2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Kontos S, Kominea A, Melachrinou M,
Balampani E and Sotiropoulou-Bonikou G: Inverse expression of
estrogen receptor-β and nuclear factor-κB in urinary bladder
carcinogenesis. Int J Urol. 17:801–809. 2010.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Flores-Estrada JJ, Jiménez A,
Victoria-Acosta G, Cortés-Malagón EM, Ortiz-López MG,
Alvarez-Sánchez ME, Nuñez-Olvera SI, Pérez-Navarro YF,
Morales-Reyna M and Puente-Rivera J: Nuclear receptors in bladder
cancer: Insights into miRNA-mediated regulation and potential
therapeutic implications. Int J Mol Sci. 26(7340)2025.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Ge Q, Lu M, Ju L, Qian K, Wang G, Wu CL,
Liu X, Xiao Y and Wang X: miR-4324-RACGAP1-STAT3-ESR1 feedback loop
inhibits proliferation and metastasis of bladder cancer. Int J
Cancer. 144:3043–3055. 2019.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Wu L, Zhang M, Qi L, Zu X, Li Y, Liu L,
Chen M, Li Y, He W, Hu X, et al: ERα-mediated alterations in
circ_0023642 and miR-490-5p signaling suppress bladder cancer
invasion. Cell Death Dis. 10(635)2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Ding M, Liu Y, Li J, Yao L, Liao X, Xie H,
Yang K, Zhou Q, Liu Y, Huang W and Cai Z: Oestrogen promotes
tumorigenesis of bladder cancer by inducing the enhancer
RNA-eGREB1. J Cell Mol Med. 22:5919–5927. 2018.PubMed/NCBI View Article : Google Scholar
|