Introduction
Breast cancer is one of the most frequent causes of
mortality among women, with a high incidence in both developed and
developing countries (1,2). Fifty percent of breast cancer cases
are diagnosed at an advanced stage and include lymph node
metastatic infiltration, necessitating more aggressive and costly
treatment (3) and increasing the
risk of post-treatment complications (4–8).
Aggressive surgery in advanced stage breast cancer
cases interrupts the main lymphatic drainage route in the upper
limbs and is the most important factor in the formation of
lymphedema (5). The pathogenesis
of post-mastectomy lymphedema associated with axillary dissection
is in particular attributed to the small number of lymphatic
vessels in the dissected area (9,10).
According to Glass et al, the more extensive the axillary
dissection, the greater the risk of complications (11).
Once lymphedema has become established, it is
incurable. Neither surgical nor drug treatments for lymphedema have
shown success (12,13). However, lymphedema can be avoided,
treated and controlled with daily preventive measures (14). The prevention of lymphedema has
been attempted using intraoperative techniques that take a more
conservative approach towards the axillary chain, such as the
investigation of the sentinel lymph nodes. Through this, selective
resection that is safe and less mutilating becomes possible, with
satisfactory results, although it is limited to patients without
evidence of lymph node macrometastasis (5,7,15–17).
Preventive measures, such as lymphatic self-massage,
hydration, kinesiotherapy, manual lymphatic drainage and the use of
elastic compression armbands, have been gaining increasing
attention (12,18). None is more important than the
others, but used together they may be effective in controlling and
preventing lymphedema. The only requirement is that the patient
must be taught how to carry out the procedures on a daily basis
(18–21). Self-massage, also known as simple
manual lymphatic drainage, is a version of manual lymphatic
drainage in which the patient learns the procedure and is able to
perform it alone, at home. It involves a series of gentle circular
movements, starting with stimulation of the axillary lymph node
chain contralateral to the surgical manipulation and of the
inguinal chain homolateral to the surgical manipulation. This is
followed by gentle movements at a site distant from the area of
congestion, moving gradually towards the swollen limb (12,18,22,23).
Lymphoscintigraphy is an effective technique for
evaluating the lymphatic systems of the upper and lower limbs
(5,24,25).
It provides functional information regarding drainage and
obstruction in this system, with details on the morphology of the
lymph vessels and lymph nodes (26). In relation to lymphedema following
radical mastectomy, lymphoscintigraphy has only been used to assess
lymphedema that has already become established. In such cases, it
is performed before and after physiotherapy (26–29)
in order to investigate the effectiveness of the therapy.
This study was inspired by the notion that the
prevention of lymphedema is the most effective way to diminish
morbidity, and that physiotherapeutic measures should begin as
early as possible for breast cancer patients undergoing axillary
dissection. It was in particular motivated by the scarcity of the
literature on the subject, and aimed to evaluate the effectiveness
of early physiotherapeutic stimulation on lymph flow progression in
these breast cancer patients.
Materials and methods
This randomized experimental study included 22 women
who underwent radical mastectomy or quadrantectomy associated with
ipsilateral axillary lymph node dissection between 2005 and 2008.
Patients were over 18 years of age and had unilateral breast
cancer. They were seen between 15 and 60 days after surgery, and
presented arm range of motion corresponding to more than 120° of
shoulder flexion. Patients were excluded if they had undergone
previous chemotherapy or radiotherapy, if they were known to have
had lymphatic disease prior to the axillary dissection, if they
presented inflammatory or infectious processes associated with the
upper limb, or if the lymphoscintigraphy results could not be
analyzed.
Patients gave their free and informed consent for
participation in the study. The study was approved by the Research
Ethics Committee of Barretos Cancer Hospital.
The 22 patients underwent two lymphoscintigraphy
examinations, for a total of 44 procedures. The first examination
on each patient was performed between 15 and 60 days after surgery.
This was considered to be a control examination, and was performed
without physiotherapeutic stimulation. The second examination was
performed 7±3 days after the first. For this second examination,
the patients were randomized into two groups: the ‘with
physiotherapeutic stimulation’ (WPS) group, in which patients
received physiotherapeutic stimulation while the lymphoscintigraphy
examination was being conducted, and the ‘without physiotherapeutic
stimulation’ (WOPS) group, in which patients received an
examination under conditions similar to the first.
Physiotherapeutic stimulation
Physiotherapeutic stimulation lasted for 5 min, from
the 5th to the 10th min of the dynamic phase of the second
examination in the randomized WPS group. It was performed by a
single physiotherapist in accordance with the technique proposed by
Földi (23), with stimulation of
the contralateral axillary lymph node chain and homolateral
inguinal chain relative to the manipulated breast and axilla using
circular movements with the palms of the hand to lightly and gently
move the skin.
Lymphoscintigraphy
Lymphoscintigraphy was performed using a two-head
gamma camera (GE Millennium VG Hawkeye) with a low-energy
high-resolution collimator, photopeak centered on 140 KeV, windows
of 20% and matrices of 128×128 for dynamic images, 256×256 for
static images and 256×1024 for the whole body scan, without
magnification. Patients were positioned in dorsal decubitus with
their arms raised above their head, then 37 MBq of
99mTc-phytate in a volume of 0.5 ml was administered
subcutaneously using an insulin needle and syringe in a fan-shaped
injection into the second interdigital space (25–27)
of the manipulated limb. Capture of dynamic images with a 1 min
exposure was initiated immediately after the injection and
continued for 20 min. Two static images with a 500 sec exposure
were acquired immediately after the end of the dynamic examination,
one in the same field of view as the dynamic examination, including
the area from the hands to the axillae, and the other including the
anterior thoracic region and axillae. A whole body scan at a
velocity of 7 cm/sec over the bed was initiated 90 min after the
injection of radioactive fluid and included anterior and posterior
projections, with the patient in the same position.
Patients attended the lymphoscintigraphy procedure
wearing clothes that would not restrict the superficial lymphatic
circulation, and were instructed not to perform any activity with
the limb until the end of the examination.
Qualitative analysis
Images from the dynamic, static, and whole body scan
phases of the first and second examinations performed on each
patient were paired. The area reached by the radioactive fluid in
the lymphatic vessels of the upper limb, and the sites and numbers
of lymph nodes viewed in the three phases of lymphoscintigraphy
acquisition, were respectively compared. A sequential ordinal
classification for the area reached by the radiopharmaceutical was
applied from the injection point to the most distant point reached
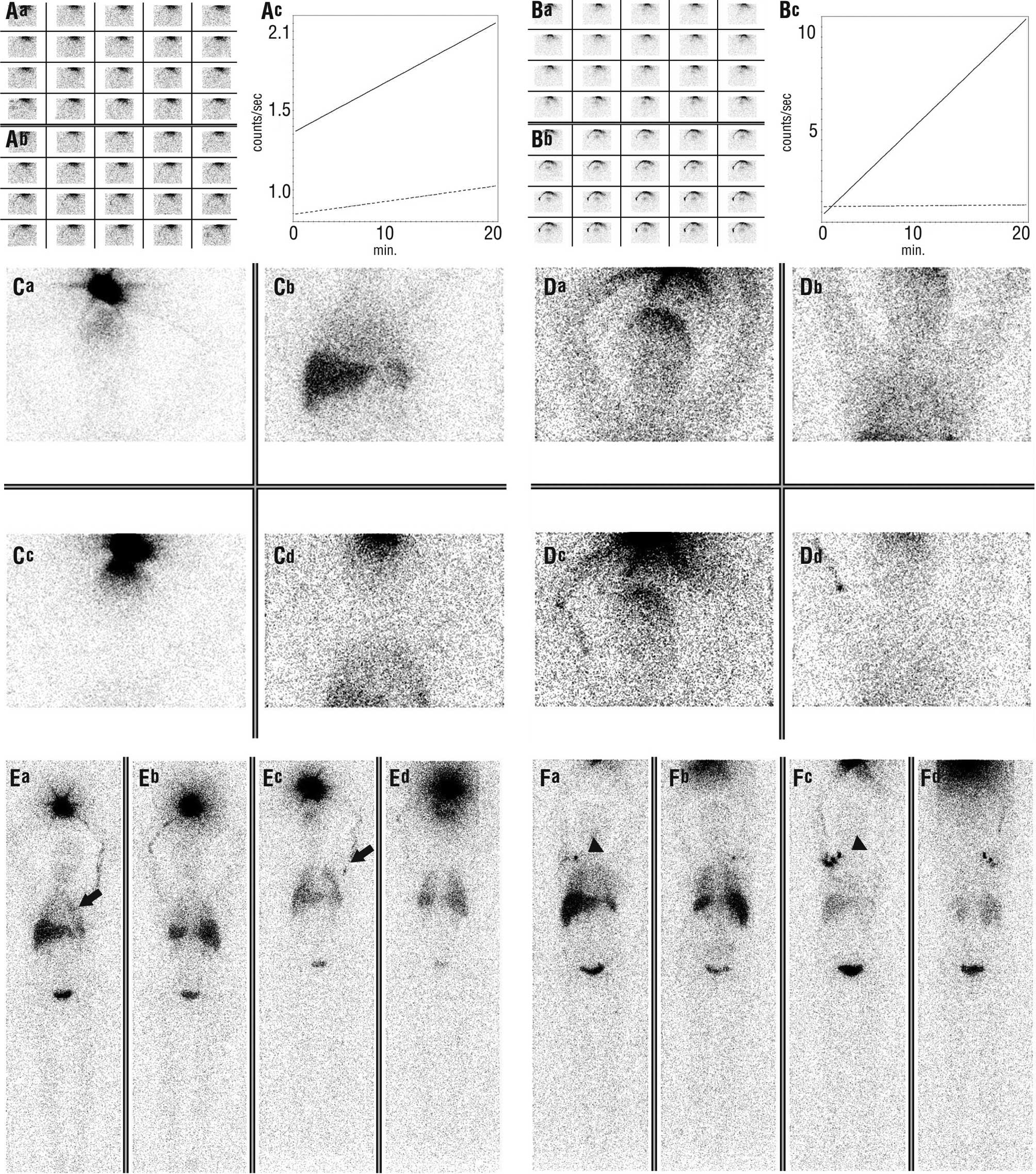
using a scale of 0–14 (Fig. 1)
(30).
 | Figure 1.Qualitative analysis: sequential
ordinal classification of upper limb. 0, injection site; 1, distal
third of forearm; 2, middle third of forearm; 3, proximal third of
forearm; 4, distal third of upper arm; 5, middle third of upper
arm; 6, proximal third of upper arm; 7, axilla without lymph nodes;
8, axillary lymph nodes; 9, lymph nodes in dorsolateral part of
upper arm; 10, apical lymph nodes; 11, central lymph nodes; 12,
internal thoracic lymph nodes; 13, lymph nodes in contralateral
axilla; 14, spleen. |
Patients were considered to present positive
progression of lymphatic flow if the area reached by the
radioactive fluid in the second examination was further from the
injection point than that reached in the first examination, or when
the number of lymph nodes apparent in the second examination was
greater than in the first examination.
Quantitative analysis in the dynamic
phase
Identical rectangular areas of interest were created
and laid out in the proximal region of the arm that received the
injection and the contralateral arm of each patient in both
examinations. Activity veresus time curves were generated for these
areas. The contralateral arm was taken to be the background.
Angular coefficients were obtained directly from processing
performed by the equipment. A value of 0.00005 was taken for every
angular coefficient calculated to have a value of 0.0000 by the
equipment. To determine the net coefficient for physiotherapeutic
stimulation, the background coefficient was subtracted from the
coefficient of the arm that received the injection. The velocity of
the progression of lymphatic fluid was represented by the net
angular coefficient. Analysis of the velocity of the progression of
lymphatic flow was performed by comparing the net angular
coefficients of the straight lines from both examinations on the
same patient. Positive progression was determined when there was a
higher velocity in the second examination than in the first
(Fig. 2).
Statistical analysis
Frequencies, percentages, central trend measurements
and dispersion measurements were used to characterize the sample.
The Kolmogorov-Smirnov test was used to investigate whether the
sample had normal distribution. Since it was found that it did not,
non-parametric tests were applied. The Mann-Whitney test was used
to evaluate differences between the means of the quantitative
variables of the WPS and WOPS groups. Fisher's exact test was used
to evaluate the association between the progression of lymphatic
drainage and the presence of physiotherapeutic stimulation.
Wilcoxon's non-parametric test was used to investigate the number
of lymph nodes observed before and after physiotherapeutic
stimulation in each group. For all the analyses, the level of
significance was set at 5%.
Results
Among the 22 patients studied, 12 were in the group
which received physiotherapeutic stimulation (WPS) and 10 were in
the group which did not (WOPS). The two groups were homogenous in
terms of the medians of the variables: age in years (47.5 vs. 52.0;
p=0.198), weight in kilograms (65.6 vs. 64.2; p=0.373), height in
meters (1.60 vs. 1.52; p=0.080), body mass index in
kg/m2 (26.7 vs. 26.3; p=0.843) and number of lymph nodes
dissected (17.5 vs. 18.0; p=0.691).
An association between physiotherapeutic stimulation
and the positive progression of lymphatic flow was present in all
three phases of the lymphoscintigraphic examinations. The images of
the dynamic and static phases were more representative of the
stimulation than the later whole body scan phase (Table I).
 | Table I.Lymphatic flow progression in the WPS
and WOPS groups categorized according to phase. |
Table I.
Lymphatic flow progression in the WPS
and WOPS groups categorized according to phase.
| Progression | WPS
| WOPS
| P-valueb |
|---|
| No. (%) | No. (%) |
|---|
| Dynamic
imagesa | | | <0.001 |
| Yes | 10 (100) | 0 (0.00) | |
| No | 0 (0.00) | 9 (100) | |
| Static imagesa | | | <0.001 |
| Yes | 11 (100) | 0 (0.00) | |
| No | 0 (0.00) | 8 (100) | |
| Whole body
scana | | | <0.001 |
| Yes | 11 (91.7) | 0 (0.00) | |
| No | 1 (8.30) | 7 (100) | |
| Total | 12 (100) | 10 (100) | |
In the WPS group, a significant increase in the
number of lymph nodes between the first and second whole body scan
examination was observed (p=0.007). In the WOPS group, there was no
significant difference between the medians (p=0.102) (Table II).
 | Table II.Number of lymph nodes apparent in the
first and second examination in the WPS and WOPS groups.a |
Table II.
Number of lymph nodes apparent in the
first and second examination in the WPS and WOPS groups.a
| Variable | First examination
| Second examination
| P-valueb |
|---|
| No. | Mean (SD) | Median | Min-Max | No. | Mean (SD) | Median | Min-Max |
|---|
| WPS | 11 | 2.1 (1.7) | 2.0 | 0–6 | 11 | 3.7 (2.1) | 4.0 | 0–7 | 0.007 |
| WOPS | 7 | 2.6 (2.0) | 3.0 | 0–5 | 7 | 1.9 (1.6) | 2.0 | 0–4 | 0.102 |
A significant increase in the velocity of the
lymphatic flow between the first and second examination was
observed in the WPS group compared to the WOPS group, as
represented by the difference in the median angular coefficient
ratio of the second examination compared to the first examination
(p=0.014) (Table III).
 | Table III.Descriptive statistics on the
variable of the ratio between the angular coefficients after/before
stimulation for the WOPS and WPS groups.a |
Table III.
Descriptive statistics on the
variable of the ratio between the angular coefficients after/before
stimulation for the WOPS and WPS groups.a
| Group | No. | Mean | Median | Variance | Standard
deviation | Min-Max |
|---|
| WPS | 10 | 44.0 | 2.75 | 6232.3 | 78.9 | 0.17–240 |
| WOPS | 8 | 0.8 | 0.75 | 0.5 | 0.7 | 0.08–2 |
Additional analysis of the drainage bundles in the
upper limb showed a predominance of lymphatic flow progression
through the cephalic bundle in both groups, which was intensified
after physiotherapeutic stimulation. Progression through the
basilic bundle as the only route was less frequent than progression
through both bundles simultaneously (Table IV).
 | Table IV.Descriptive statistics on the
drainage bundles of the upper limbs.a |
Table IV.
Descriptive statistics on the
drainage bundles of the upper limbs.a
| WPS
| WOPS
|
|---|
| First examination
no. (%) | Second examination
no. (%) | First examination
no. (%) | Second examination
no. (%) |
|---|
| Bundles not
shown | 6 (50.0) | 0 | 4 (50.0) | 6 (75.0) |
| Basilic lymphatic
bundle | 0 | 0 | 1 (12.5) | 0 |
| Cephalic lymphatic
bundle | 4 (33.3) | 9 (75.0) | 3 (37.5) | 1 (12.5) |
| Both bundles
shown | 2 (16.7) | 3 (25.0) | 0 | 1 (12.5) |
| Total | 12 (100) | 12 (100) | 8 (100) | 8 (100) |
Discussion
With advances in breast cancer treatment and
consequent increases in survival rates, quality of life has become
a major concern for breast cancer patients (31). The occurrence of post-mastectomy
lymphedema predisposes individuals to incapacitating diseases, with
great socioeconomic impact (10,16).
Early diagnosis and intervention, such as skin care (19,32),
kinesiotherapy and self-massage (12,18,19),
may significantly reduce the incidence of complications (32). Research on improving quality of
life has indicated that the prevention of lymphedema is the best
strategy for breast cancer patients. Prior knowledge of normal
lymphatic circulation and its changes in the presence of
obstruction has directed techniques of physiotherapeutic
stimulation. Various lymphatic drainage techniques have been used
for stimulating lymphatic flow and treating lymphedema, such as
those proposed by Vodder, Leduc and Földi. The technique proposed
by Földi (23) was chosen for the
present study, since it is routinely used at our institution.
Although several studies have used
lymphoscintigraphy to document the behavior of lymphedema and to
make before and after treatment comparisons (26–29),
there is a lack of documentation regarding the immediate
postoperative period. The effects of physiotherapeutic stimulation
during this period motivated the present investigation. The aim of
the study was to evaluate whether manual physiotherapeutic
stimulation, which can easily be performed by patients themselves,
might improve the progression of lymphatic flow.
We observed that the technique of early
physiotherapeutic stimulation was effective regarding lymphatic
flow progression in the WPS group compared with the WOPS group. In
this respect, several authors have reported increased lymphatic
transportation using the same procedure (33–36)
among patients with established lymphedema.
As shown in Table
I, all patients except one showed the progression of lymphatic
flow to the lymph node chain, even if this occurred at a later
stage. The one exception did not present progression even with
stimulation; however, this patient had the greatest number of lymph
nodes dissected (n=34). This finding is concordant with Clodius
et al (9), who reported
that the pathogenesis of the lymphedema is attributed to the
presence of fewer lymphatics in the dissected area.
There are two common drainage routes in the upper
limbs: the basilic route, which feeds into the axillary lymph
nodes, and the cephalic route, in the apical lymph nodes. There are
also two infrequent routes, the cervical and internal mammary
routes (30,37,38).
The lymphatic vessels of the upper limbs flow out into the lymph
nodes of the axillary region (37,38),
and therefore the basilic route is the principal drainage route. In
the event of obstruction in the axillary region, the drainage route
along the cephalic vein becomes important. This was observed in our
study: patients in the WPS group showed this collateral route,
while patients in the WOPS group did not.
As a strategy for stimulating lymphatic circulation
in cases of the obstruction of normal lymphatic flow, alternative
routes need to be considered, such as the cephalic route.
Furthermore, anastomoses of the lymphatic capillaries need to be
considered. These deviate lymphatic flow in a direction opposite to
the normal course (38), thereby
stimulating an alternative drainage route. In a systematic review,
Moseley et al concluded that, although self-massage only
promoted a small reduction in the volume of the limb, this was more
beneficial than doing nothing for the swollen limb (35). Williams et al also reported
that the technique was important when no treatment for lymphedema
was available (18).
Considering that inflammation due to surgical
aggression hinders the passage of lymphatic drainage (39) and causes increased limb volume,
protein stagnation and the risk of complications, the findings from
our study reinforce the need to stimulate early lymphatic drainage.
Self-massage is a technique that is easy for patients to perform,
and they should be given guidance in this respect by the
professionals involved in rehabilitation.
References
|
1.
|
National Cancer Institute (NCI), USA:
Progress shown in death rates from four leading cancers: decline in
overall mortality has slowed (monograph on the Internet). Available
from: http://www.nci.nih.gov/newscenter/pressreleases/2003ReportReleaseuri.
Cited March 2004.
|
|
2.
|
Instituto Nacional do Câncer (INCA):
Estimativa 2008: incidência de câncer no Brasil. INCA, Rio de
Janeiro,. 2008
|
|
3.
|
Johansson S, Svensson H and Denekamp J:
Dose response and latency for radiation-induced fibrosis, edema and
neuropathy in breast cancer patients. Int J Radiot Oncol Biol Phys.
52:1207–1219. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Bergmann A, Mattos IE and Koifman RJ:
Diagnóstico do linfedema: análise dos métodos empregados na
avaliação do membro superior após linfadenectomia axilar para
tratamento do câncer de mama. Rev Bras Canc. 50:311–320. 2004.
|
|
5.
|
Bourgeois P, Leduc O and Leduc A: Imaging
techniques in the management and prevention of posttherapeutic
upper limb edemas. Cancer. 83:2805–2813. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Paci E, Cardiddi A, Bacilli A, et al:
Long-term sequelae of breast. Tumori. 82:321–324. 1996.PubMed/NCBI
|
|
7.
|
Goffman TE, Laronga C, Wilson L and Elkins
D: Lymphedema of the arm and breast in irradiated breast in cancer
patients: risks in an era of dramatically changing axillary
surgery. Breast J. 10:405–411. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Carpentier PH: Physiopathologie des
lymphoedèmes. Rev Med Interne. 23:S371–S374. 2002.
|
|
9.
|
Clodius L, Piller NB and Casley-Smith JR:
The problems of lymphatic microsurgery for lymphedema. Lymphology.
14:69–76. 1981.PubMed/NCBI
|
|
10.
|
Filippetti M, Santoro E, Graziano F,
Petric M and Rinaldi G: Modern therapeutic approaches to
postmastectomy brachial lymphedema. Microsurgery. 15:604–610. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Glass EC, Essner R and Giuliano AE:
Sentinel node localization in breast cancer. Semin Nucl Med.
29:57–68. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Didem K, Ufuk YS, Serdar S and Zümre A:
The comparison of two different physiotherapy methods in treatment
of lymphedema after breast surgery. Breast Cancer Res Treat.
93:49–54. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Roucout S and Oliveira VM: Etiologia,
prevenção e tratamento do linfedema pós-mastectomia. Med Reab.
49:11–15. 1999.
|
|
14.
|
Linnitt N: Lymphoedema: recognition,
assessment and management. Br J Community Nurs. 10:S20–S26. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Clodius L: Minimizing secondary arm
lymphedema from axillary dissection. Lymphology. 34:106–110.
2001.PubMed/NCBI
|
|
16.
|
Bumpers HL, Best IM, Norman D and Weaver
WL: Debilitating lymphedema of the upper extremity after treatment
of breast cancer. Am J Clin Oncol. 25:365–367. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Rietman JS, Dijkstra PT, Geertzen JHB, et
al: Treatment-related upper limb morbidity 1 year after sentinel
lymph node biopsy or axillary lymph node dissection for stage I or
II breast cancer. Ann Surg Oncol. 11:1018–1024. 2004.
|
|
18.
|
Williams AF, Vadgama A, Franks PJ and
Mortimer PS: A randomized controlled crossover study of manual
lymphatic drainage therapy in women with breast cancer-related
lymphoedema. Eur J Cancer Care. 11:254–261. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Rich A: How to care for uncomplicated skin
and keep it free of complications. Br J Community Nurs. 12:S6–S9.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Hampton S: Elvarex compression garments in
the management of lymphoedema. Br J Nurs. 12:925–926. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
King B: Diagnosis and management of
lymphoedema. Nurs Times. 102:4749512006.
|
|
22.
|
Camargo MC and Marx AG: Linfoterapia.
Reabilitação Física no Câncer de Mama. Editora Roca. (São Paulo).
98–99. 2000.
|
|
23.
|
Földi E, Földi M and Weissleder H:
Conservative treatment of lymphoedema of the limbs. Angiology.
36:171–180. 1985.
|
|
24.
|
Szuba A, Shin WS, Strauss HW and Rockson
S: The third circulation: radionuclide lymphoscintigraphy in the
evaluation of lymphedema. J Nucl Med. 44:43–57. 2003.PubMed/NCBI
|
|
25.
|
Baulieu F, Baulieu JL, Secchi V, Dabiens
J, Barsotti J and Itti R: Factorial analysis of dynamic
lymphoscintigraphy in lower limb limphoedema. Nucl Med Comm.
10:109–119. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Ferrandez JC, Laroche JP, Serin D,
Felix-Faure C and Vinot JM: Aspects lymphoscintigraphiques des
eddets du drainage lymphatique manuel. J Mal Vasc. 21:283–289.
1996.
|
|
27.
|
Perrymore WD and Harolds JA:
Technetium-99m-Albumin Colloid Lymphoscintigraphy in postoperative
lymphocele. J Nucl Med. 37:1517–1518. 1996.PubMed/NCBI
|
|
28.
|
Burnand KG, McGuinness CL, Lagattolla NR,
Browse NL, El-Aradi A and Nunan T: Value of isotope lymphography in
the diagnosis of lymphoedema of the leg. Br J Surg. 89:74–78. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Bull RH, Fenton DA and Mortimer PS:
Lymphatic function in the yellow nail syndrome. Br J Dermatol.
134:307–312. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Macea JR and Fregnani JHTG: Anatomy of the
thoracic wall, axilla and breast. Int J Morphol. 24:691–704. 2006.
View Article : Google Scholar
|
|
31.
|
Kwan W, Jackson J, Weir LM, Dingee C,
McGregor G and Olivotto IA: Chronic arm morbidity after curative
breast cancer treatment: prevalence and impact no quality of life.
J Clin Oncol. 20:4242–4248. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Linnitt N: Complex skin changes in chronic
oedemas. Br J Community Nurs. 12:S10–S15. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Badger C, Preston N, Seers K and Mortimer
P: Physical therapies for reducing and controlling lymphoedema of
the limbs. Cochrane Database Syst Rev. CD0031412004.PubMed/NCBI
|
|
34.
|
Huit M: A guide to treating lymphoedema.
Nurs Times. 96:42–43. 2000.
|
|
35.
|
Moseley AL, Carati CJ and Piller NB: A
systematic review of common conservative therapies for arm
lymphoedema secondary to breast cancer treatment. Ann Oncol.
18:639–646. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Andersen L, Hojris I, Erlandsen M and
Andersen J: Treatment of breast-cancer related lymphedema with or
without manual lymphatic drainage – a randomized study. Acta Oncol.
39:399–405. 2000.
|
|
37.
|
Suami H, Pan WR and Taylor GI: Changes in
the lymph structure of the upper limb after axillary dissection:
radiographic and anatomical study in a human cadaver. Plast
Reconstr Surg. 120:982–991. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Suami H, Pan WR and Taylor GI: The
lymphatic territories of the upper limb: anatomical study and
clinical implications. Plast Reconstr Surg. 119:1813–1822. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
39.
|
Tabibiazar R, Cheung L, Han J, et al:
Inflammatory manifestations of experimental lymphatic
insufficiency. PloS Med. 3:1114–1139. 2006. View Article : Google Scholar
|